
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Anim. Sci. , 20 March 2025
Sec. Animal Nutrition
Volume 6 - 2025 | https://doi.org/10.3389/fanim.2025.1562308
 Hossein Rajaei-Sharifabadi1,2
Hossein Rajaei-Sharifabadi1,2 Ahmad Reza Seradj1
Ahmad Reza Seradj1 Saman Lashkari3
Saman Lashkari3 Deepak E. Velayudhan4
Deepak E. Velayudhan4 Ester Vinyeta4
Ester Vinyeta4 Tofuko Awori Woyengo3,5*
Tofuko Awori Woyengo3,5*Energy and nutrient availability in corn DDGS for pigs is partly limited by complex interactions between fiber and protein of the corn DDGS. Supplemental direct fed microbials (DFM) and protease can potentially improve energy and nutrient availability in corn DDGS-based diets for pigs. This study determined the effects of supplementing a corn DDGS-soybean meal (SBM)-based diet with a combination of protease and Bacillus-based DFMs on the growth performance and apparent total tract digestibility (ATTD) of nutrients in pigs. Eighty pigs (initial BW = 29.2 kg) housed in 20 pens were fed two diets (10 pens per diet), which were a corn DDGS-SBM-based diet without or with a combination of Bacillus subtilis protease at 5,000 U/kg and three-strain Bacillus-based DFMs at 1.5 × 105 CFU/g. The basal diet contained phytase at 750 FTU/kg and was formulated to meet the nutrient recommendations for grower–finisher pigs except for NE, Ca, and P contents, which were lower than the recommendations by 0.209 MJ/kg, 0.9 g/kg, and 0.9 g/kg, respectively. The diets were fed in three phases based on BW: phase 1 from 30 to 55 kg, phase 2 from 55 to 75 kg, and phase 3 from 75 to 100 kg. Growth performance was determined by phase, whereas the ATTD of nutrients was determined at the end of phase 1. Protease and DFM supplementation increased (P < 0.05) the ATTD of gross energy, nitrogen, and P by 8.0%, 10.3%, and 15.5%, respectively, but did not affect BW gain and feed intake. In conclusion, adding protease and DFMs to the corn-DDGS-SBM-based diet increased nutrient digestibility but did not affect the growth performance of pigs, implying that the basal diet was not deficient in energy. Thus, basal diets with appropriate low energy values should be developed to optimize the utilization of protease and DFMs in diets for pigs.
Bacillus strains are beneficial bacteria that can be used as direct-fed microbials (DFMs) in pig diets. By producing a variety of enzymes (such as phytase and fiber-degrading enzymes), promoting a balanced gut microbiome, and enhancing the population of microorganisms capable of degrading fiber, Bacillus strains can improve digestion and nutrient absorption, leading to better overall health and performance (Kabir, 2009). Additionally, they can help mitigate the emission of harmful environmental gases (Prenafeta-Boldú et al., 2017). Considering that different Bacillus strains vary in their enzyme production profiles, a combination of multiple strains may have greater potential to increase digestibility compared to individual strains (Cai et al., 2015; Augspurger et al., 2016; Jaworski et al., 2017; Lewton et al., 2021). This synergistic effect could arise from the complementary enzymatic activities of different strains, potentially addressing a broader range of dietary components. On the other hand, dietary supplementation with exogenous protease has been shown to improve growth rate and nutrient digestibility (Pan et al., 2016; Yu et al., 2016, 2020), as well as to increase intestinal fermentation capacity and transit time (Min et al., 2019; Aranda-Aguirre et al., 2021). Recent efforts have explored the potential complementary advantages of combining Bacillus-based DFMs with exogenous protease in pig diets, particularly those based on corn and soybean meal. Payling et al. (2019) reported that protease and DFMs worked synergistically to degrade fiber–protein complexes in soybean meal-based digesta. They proposed that protease hydrolyzes proteins, thereby enhancing access to fiber substrates for enzymes secreted by DFMs, indicating a complementary mode of action. Prior study demonstrated that incorporating Bacillus-based DFMs and protease into the diet of growing pigs resulted in synergistic improvements in the apparent total tract digestibility (ATTD) of nitrogen and energy (Payling et al., 2017).
Distillers’ dried grains with soluble (DDGS), a co-product from the ethanol industry, is rich in amino acids, phosphorus, and other essential nutrients (NRC, 2012), making it a sustainable and cost-effective option for incorporation into the diets of livestock animals (Reddy et al., 2021). However, DDGS (compared with soybean meal) has a lower availability of nutrients for pigs (NRC, 2012). The lower nutrient availability in DDGS has been suggested to be due to the formation of complexes between fiber, protein, and other components of DDGS that are poorly digested by pigs (Jha et al., 2015). Thus, the supplementation of DDGS-based diets with a combination of protease and Bacillus-based DFMs can potentially be effective in improving nutrient availability in DDGS for pigs. However, information is lacking on the effect of supplementing DDGS-based diets for pigs with a combination of protease and Bacillus-based DFMs. We hypothesized that this supplementation of DDGS-based diets with a combination of protease and Bacillus-based DFMs would improve growth performance and nutrient digestibility in growing pigs. Therefore, the present study aimed to evaluate the effects of supplementing corn DDGS-soybean meal-based diet for growing–finishing pigs with a combination of protease and Bacillus-based DFMs on growth rate, feed intake, feed efficiency, and ATTD of energy and nutrients.
All experimental procedures involving animals were reviewed and approved by the Institutional Animal Care and Use Committee at South Dakota State University (Protocol #15-083E).
A total of 80 pigs (40 barrows and 40 gilts; initial BW = 29.2 kg; Large White-Landrace female × Large White-Hampshire male; Pig Improvement Company, Hendersonville, Tennessee, USA) were used in this study. The pigs were obtained in two batches of 40 pigs each from a commercial farm. Pigs in each batch were individually weighed and placed in 10 pens of either four barrows or four gilts balanced for body weight. The pens were in two different rooms. The first room accommodated four pens (2.3 × 1.8 m) featuring a fully slatted concrete floor along with a single-space dry feeder and a nipple drinker. The second room housed six pens (4.88 × 1.2 m) equipped with a half-slated concrete floor, double-space dry feeders, and nipple drinkers. The temperature in each room was consistently maintained at 22°C throughout the study period, while continuous lighting was provided for 24 h without a dark period. The rooms were ventilated through negative pressure.
Two diets were fed. The diets were a corn DDGS-soybean meal-based diet (Table 1) without or with a combination of Bacillus subtilis protease at 5,000 U/kg and a three-strain Bacillus-based DFMs incorporating two strains of B. amyloliquefaciens and one strain of B. subtilis at 1.5 × 105 CFU/g (Danisco Animal Nutrition & Health, IFF, The Netherlands). The basal diets containing phytase (Axtra® PHY; Danisco Animal Nutrition & Health, IFF, The Netherlands) at 750 FTU/kg were formulated according to the NRC (2012) nutrient recommendations for grower–finisher pigs except for net energy (NE), Ca, and standardized total tract digestible P contents, which were slightly lower than the recommendations by 0.209 MJ/kg, 0.9 g/kg, and 0.9 g/kg, respectively (Table 2). The basal diet was formulated to be lower in NE value than the recommended value because supplementation of the combination of protease and DFMs was expected to increase the digestibility of energy-generating components of the diets and, hence, the NE value of the diet. The basal diet was also formulated to be lower in Ca and standardized total tract digestible P contents than the recommended values because the basal diet contained phytase. Diets were fed in three phases based on BW: phase 1 from 30 to 55 kg, phase 2 from 55 to 75 kg, and phase 3 from 75 to 100 kg. The diets did not contain antimicrobial agents and pharmacological levels of Zn and Cu. Pens of pigs were randomly allocated to the two dietary treatments within batch, sex, and room, for a total of five pens per dietary treatment per sex and, hence, 10 pens per diet for the study.
During the experimental period, diet were offered to the pigs ad libitum and pigs had unlimited access to water. Feed intake and individual body weight were monitored biweekly throughout the experimental period with the calculation of average daily gain (ADG), average daily feed intake (ADFI), and feed efficiency (G:F) by phase. To measure ATTD, titanium dioxide was added to the diets at a rate of 0.4% as an indigestible marker and fed during the last 7 days of phase 1 of feeding (days 21 to 28 of the experiment). A minimum of 100 g of fresh and clean fecal samples were collected from the floor of pens on days 26 and 27 of the study into plastic bags and stored at −20°C for subsequent analysis.
Fecal samples from each pen were thawed, pooled, and homogenized. A 100-g subsample of homogenized feces for each pen was then lyophilized at −40°C and 0.05 mbar for 24 h. Feed and lyophilized fecal samples were ground in a centrifugal mill (Retsch model ZMII; Brinkman Instruments, Rexdale, ON, Canada) to pass through a 0.75-mm sieve. Feed and fecal samples were subjected to analysis for dry matter (DM), gross energy (GE), nitrogen (N), neutral detergent fiber (NDF), acid detergent fiber (ADF), ether extract (EE), Ca, P, and titanium dioxide content. The determination of DM followed the protocol outlined by the AOAC (1990). Gross energy was quantified using an adiabatic bomb calorimeter (model AC600, Leco, St. Joseph, MI, USA). Nitrogen content was assessed using the method 984.13A-D, while EE was analyzed according to the method 920.39A. The NDF and ADF were determined based on the methodology by Holst (1973) and AOAC (2006), respectively. Calcium and P analyses involved ashing and digestion following the AOAC (1990) procedures, with measurement conducted using inductively coupled plasma mass spectrometry (Varian Inc., Palo Alto, CA, USA). Titanium dioxide content was determined through spectrophotometry (model Spectra MAX 190, Molecular Devices, Sunnyvale, CA, USA) at 408 nm after ashing at 525°C for 10 h (Myers et al., 2004).
Data were analyzed using ANOVA with the PROC MIXED procedure of SAS (SAS Institute Inc., Cary, NC, USA). Before ANOVA, the normal distribution of the data was evaluated using the UNIVARIATE procedure of SAS. Growth performance parameters for each phase were analyzed using a model that included batch, room, diet, sex, and diet × sex interaction as fixed effects, whereas the nutrient digestibility data were analyzed using a model with fixed effects of batch, room, and diet. For the entire study period, the fixed effects of batch, room, and diet were included in the model, while sex was considered a block effect. Initial BW was incorporated as a covariate factor for ADG, ADFI, and G:F, and each pen was considered as the experimental unit. Means were separated by the probability of difference. Hypotheses were tested at a significance level of P ≤0.05. Additionally, trends (0.05 < P ≤ 0.10) were reported where applicable.
The analyzed values of CP, NDF, Ca, and P for the phase 1 basal diet were close to the calculated values of the same diet (Table 2). The results of the growth performance are presented in Table 3. The main effects of the diet on ADG, ADFI, and G:F were not significant (P > 0.05). During phase 2, barrows showed a greater ADG (P = 0.051) compared to gilts. Moreover, higher ADFI was observed for barrows than for gilts during both phase 2 (P = 0.009) and phase 3 (P < 0.038) of feeding. No interaction was detected between diet and sex for any of the performance parameters. Additionally, the overall performance of the pigs was not affected by the dietary treatment (Figure 1).
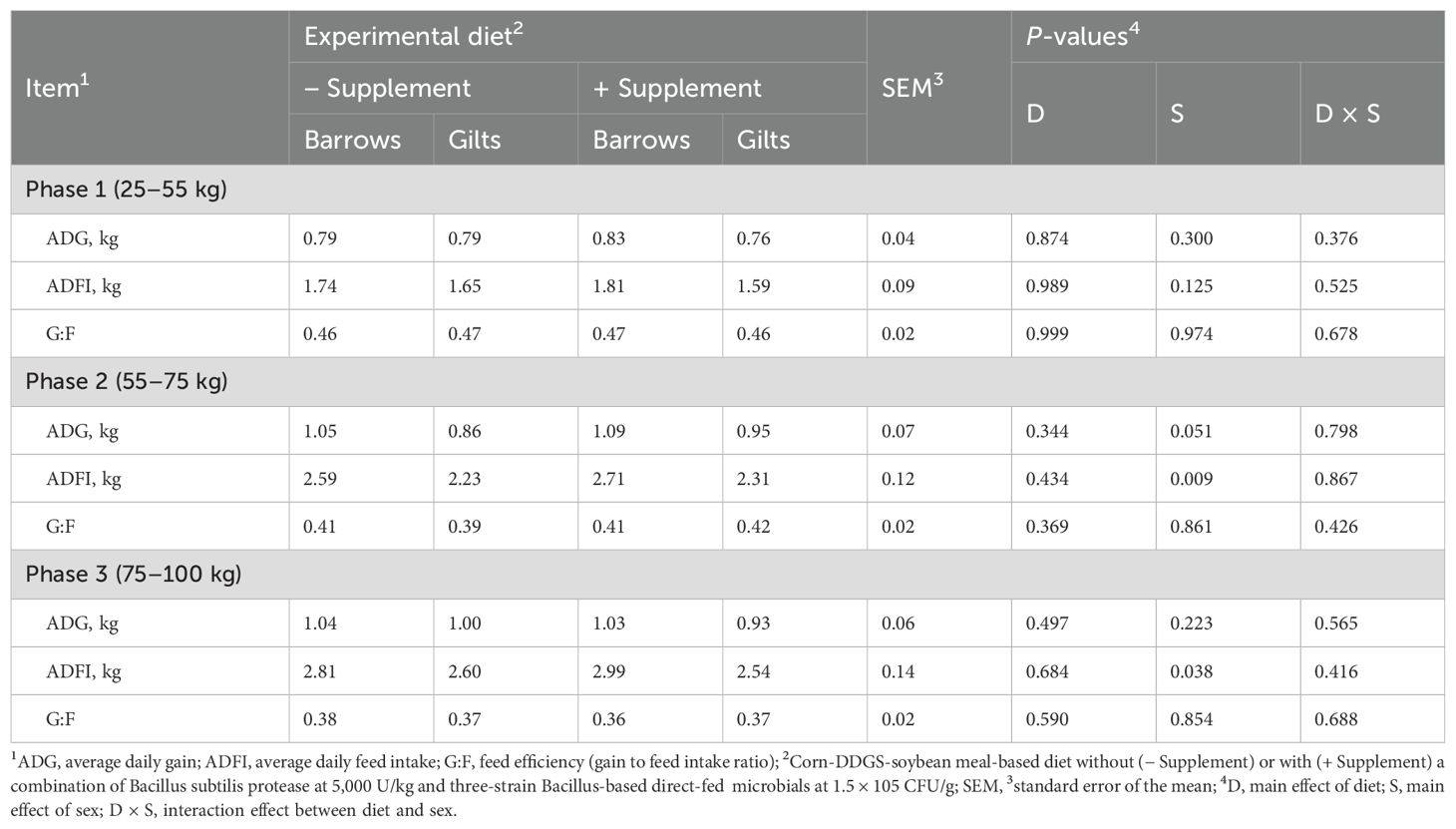
Table 3. Effects of experimental diets on growth performance of pigs across all three dietary phases.
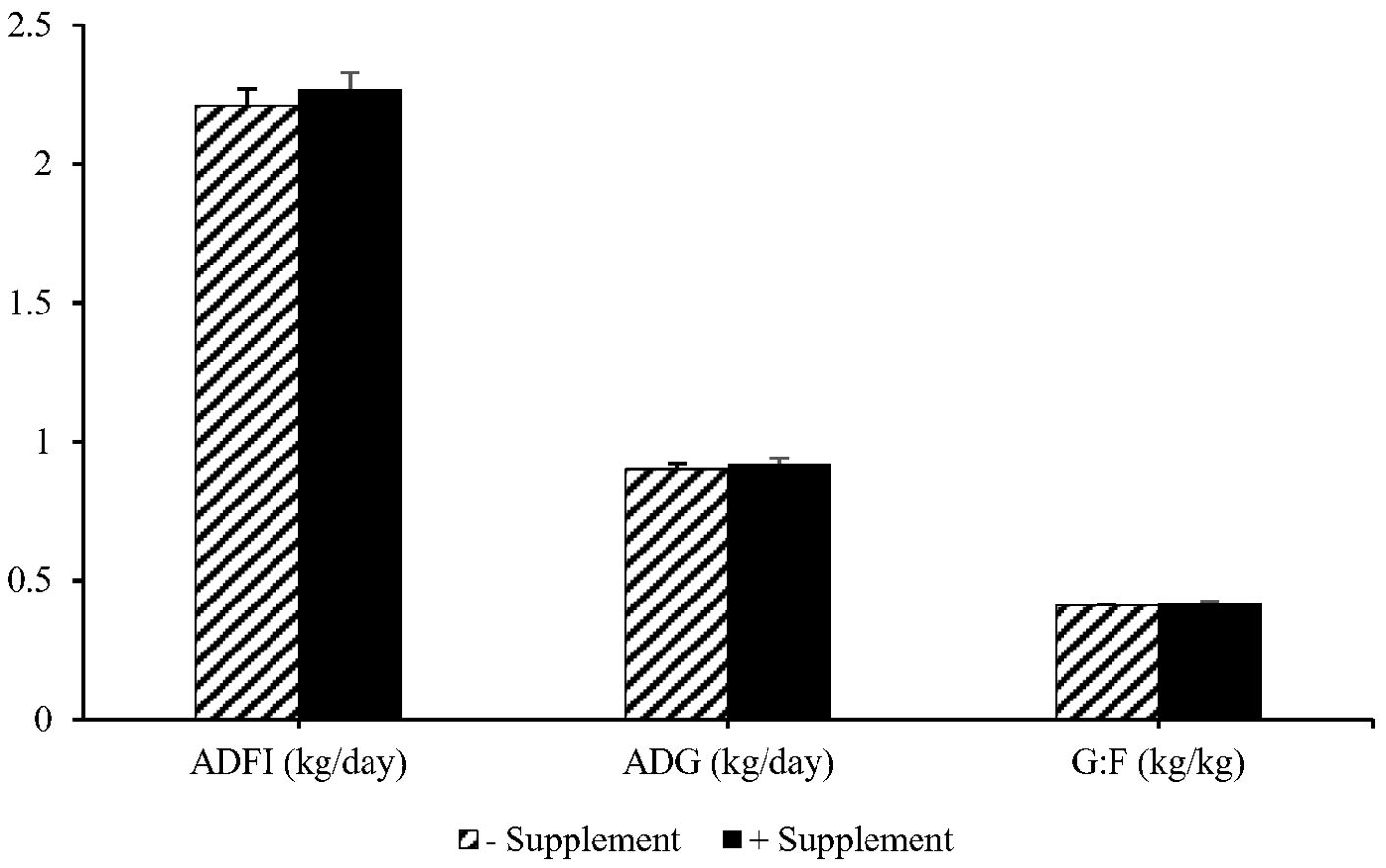
Figure 1. Effect of experimental diets on the overall average daily feed intake (ADFI), average daily gain (ADG), and feed efficiency (G: F) of pigs throughout the experiment. The experimental diets were a corn DDGS-soybean meal-based diet without (− Supplement) or with (+ Supplement) a combination of Bacillus subtilis protease at 5,000 U/kg and three-strain Bacillus-based direct-fed microbials at 1.5 × 105 CFU/g. No significant difference was observed between treatments.
Figure 2 shows the results of ATTD of energy and nutrients. Supplementing basal diet with a combination of protease and Bacillus-based DFMs resulted in an improvement (P < 0.05) in the ATTD of DM, GE, nitrogen, and EE. Moreover, pigs fed the diet supplemented with a combination of B. subtilis protease and multi-strain Bacillus-based DFMs had greater (P < 0.05) ATTD of NDF and ADF than those fed the non-supplemented diet. Although supplementation of protease and multi-strain Bacillus-based DFMs did not significantly increase the ATTD of Ca, it increased (P < 0.05) the ATTD of P.
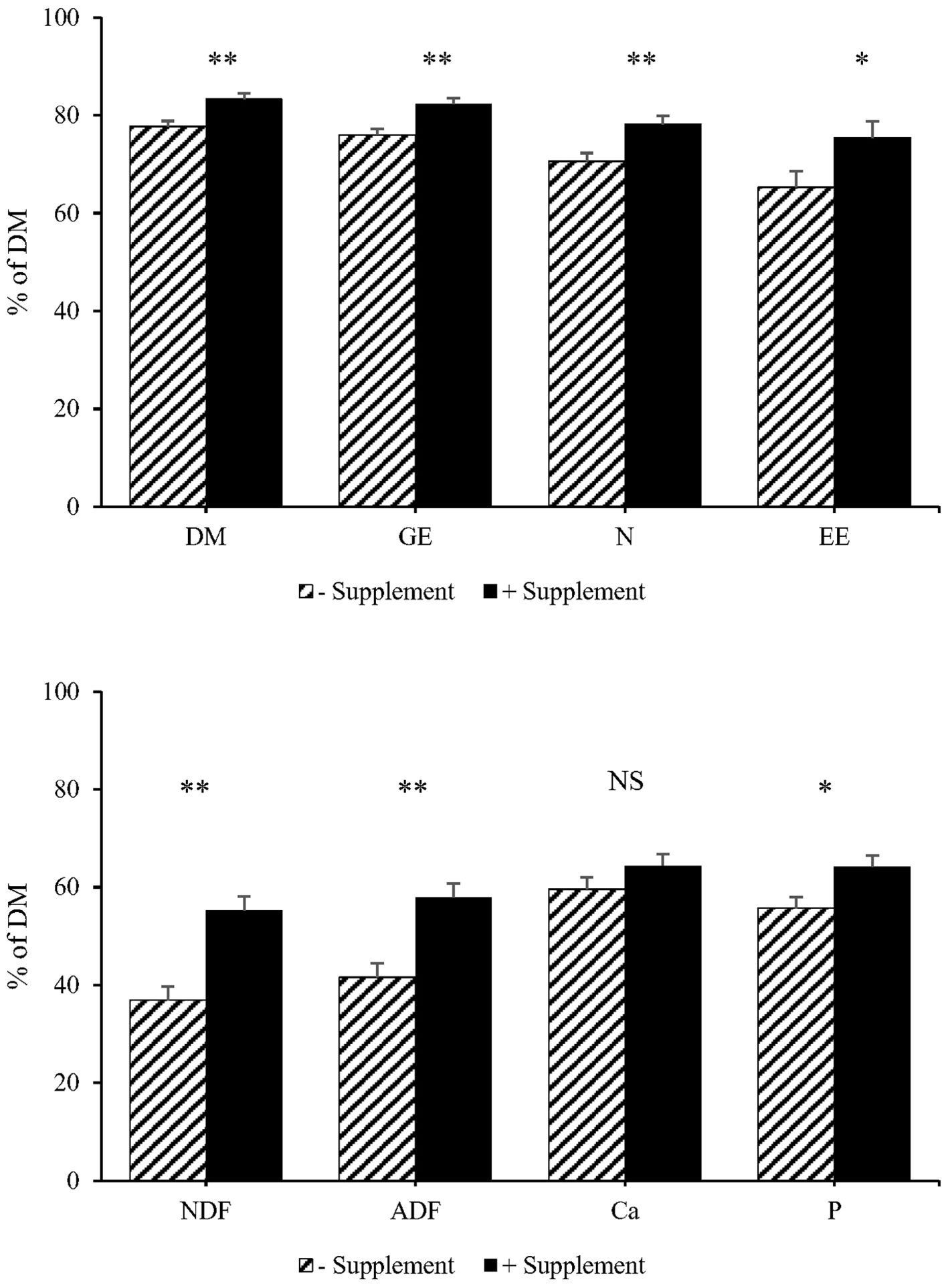
Figure 2. Effects of experimental diets on apparent total tract digestibility of dry matter (DM), gross energy (GE), nitrogen (N), ether extract (EE), neutral detergent fiber (NDF), acid detergent fiber (ADF), calcium (Ca), and phosphorous (P) in phase 1. The experimental diets were corn DDGS-soybean meal-based diet without (− Supplement) or with (+ Supplement) a combination of Bacillus subtilis protease at 5,000 U/kg and three-strain Bacillus-based direct-fed microbials at 1.5 × 105 CFU/g. *significant differences between treatments at P <0.05; **significant differences between treatments at P <0.01. NS, non-significant differences between treatments (P > 0.05).
Although diets were formulated for three phases and calculations were performed for all phases, chemical composition analysis was conducted only for phase 1, which is the phase that is typically the most critical and limiting for nutrient requirements and performance impacts of growing–finishing pigs. The analyzed nutrient composition of the basal diet for phase 1 closely matched the calculated (expected) composition, providing confidence in our diet formulation accuracy and ingredient quality.
In a prior study conducted by Kellner et al. (2021), it was noted that dietary supplementation of a combination of protease and DFMs (B. subtilis) significantly reduced mortality rates in wean-to-finish pigs, with no significant effects on growth performance under commercial conditions. In contrast, a meta-analysis of data from three studies on growing pigs (~25 kg initial BW, fed for 42 days) indicated that protease and multi-strain Bacillus-based DFMs, either individually or in combination, enhanced the growth performance of pigs (Payling et al., 2017). However, the variability among the experimental diets used in these studies, including differing levels of DDGS (0%, 5%, and 20%) and/or exogenous phytase, makes it challenging to draw definitive conclusions regarding the supplement’s overall effects. The growth performance responses of pigs to the inclusion of a blend of exogenous enzymes in DDGS-containing diets have also shown inconsistencies in previous studies. Li et al. (2012) fed grower–finisher pigs with diets containing 10% or 15% DDGS supplemented with a blend of exogenous enzymes, including protease. They found that the multi-enzyme blend tended to improve the growth performance in pigs fed diets with DDGS. In contrast, other researchers indicated no significant effects of multi-enzyme blends containing protease on the growth performance of grower–finisher pigs fed diets containing 15% to 60% DDGS (Thacker, 2009; Jacela et al., 2010; Agyekum et al., 2012; Kerr et al., 2013).
The efficacy of feed additives in corn-soybean meal-DDGS-based diets has been a subject of ongoing research. Kerr et al. (2013) evaluated various commercial feed additives, including enzymes, probiotics, and yeast, in soybean meal-DDGS-based diets, similar to those used in our study. They found that these additives had minimal impact on nutrient digestibility and no effect on growth performance in both starter and finishing pigs when fed nutritionally adequate diets containing 30% corn DDGS. In the present study, although the NE, Ca, and P levels were slightly lower than the NRC (2012) nutrient recommendations (0.209 MJ/kg, 0.9 g/kg, and 0.9 g/kg, for NE, Ca, and P, respectively), the growth performance of the pigs was comparable to previous studies in which growing–finishing pigs were fed diets formulated to meet or exceed NRC recommendations (Yang et al., 2020). This indicates that pigs may have some capacity to adapt to marginally deficient diets without significant performance losses. However, the impact of feed additives may be more pronounced in diets with greater nutrient deficiencies or lower digestible ingredients. In a study conducted by Emiola et al. (2009) on pigs from 36.5 to 55 kg BW, soybean meal in a diet formulated to meet the NRC (1998) nutrient recommendations was completely replaced with 30% wheat DDGS. This substitution resulted in 4% and 5% reductions in digestible energy and lysine, respectively, and led to lower daily gain (76.2 vs. 90.5 g/day) and feed efficiency (0.33 vs. 0.39) compared to pigs fed a soybean meal-based control diet. These authors observed significant improvements in growth performance due to supplementation with a multi-enzyme preparation, which was associated with improvements in nitrogen, energy, and ether extract digestibility. Collectively, these findings indicate that the effectiveness of feed additives in improving growth performance through increased nutrient digestibility is contingent upon diet composition and nutrient adequacy. In marginally deficient diets, as in our study, benefits may be less apparent. Conversely, in diets with more pronounced deficiencies or less digestible ingredients, the positive effects of feed additives are likely to be more evident.
The main challenge associated with incorporating DDGS into pig diets revolves around NSP, which can have a negative impact on nutrient digestibility and availability. Although there is some evidence indicating that feed additives such as exogenous enzymes, DFMs, and yeast may have no effect or even a negative impact on nutrient digestibility in pig diets containing DDGS (Kerr et al., 2013), the general consensus in the literature indicates that feed additives, particularly exogenous enzymes, enhance nutrient digestibility in DDGS-inclusive diets for growing pigs (Swiatkiewicz et al., 2016). In the present study, the blend of DFMs and protease had a significant positive effect on nutrient digestibility, with a 7% improvement in the ATTD of DM. This higher DM digestibility was associated with increased ATTD of CP, EE, NDF, ADF, and P by 11%, 15%, 33%, 28%, and 13%, respectively. The higher CP, EE, and fiber digestibility led to 8% greater GE digestibility compared to the non-supplemented group. Our findings show more pronounced effects than previous studies. For instance, Emiola et al. (2009) reported only a modest 1% to 2% improvement in DM digestibility when supplementing DDGS diets with multi-carbohydrase enzymes, though they observed improvements in nitrogen (8.8%), EE (25%), and NDF (5.4%) digestibility in growing pigs. Cai et al. (2015) found that multi-strain Bacillus species-based DFMs had minimal effects on the ATTD of DM and nitrogen in nursery pigs, without impacting GE digestibility. Consistent with our results, Payling et al. (2017) demonstrated that a combination of protease and Bacillus-based DFMs has the potential to enhance nutrient digestibility more effectively than either component alone, supporting the synergistic approach observed in our study.
The increased ATTD of nitrogen along with higher fiber (NDF and ADF) digestibility due to supplementation with the blend of DFMs and protease is likely attributed to a complementary mode of action where protease hydrolyzes proteins, providing increased access to fiber substrates for the enzymes secreted by DFMs as well as degrading the fiber–protein complexes and increase in protein solubilization. This synergistic action may explain the more pronounced improvements in nutrient digestibility observed in our study compared to those using single additives. The results from confocal laser scanning microscopy and scanning electron microscopy indicated that a combination of protease and Bacillus-based DFMs had a complementary effect in degrading the fiber–protein complexes in the SBM-based digesta, but not in wheat and corn-based digesta (Payling et al., 2019). This was supported by a significant increase in protein solubilization with the additive combination compared to the control, which is likely due to a complementary mode of action where protease hydrolyzes proteins, providing increased access to fiber substrates for the enzymes secreted by DFMs. Furthermore, the enhanced fiber digestibility observed in our study could have additional benefits beyond improved nutrient utilization. Increased fiber fermentation in the hindgut can lead to the production of short-chain fatty acids, which may contribute to improved gut health and potentially influence the microbial population in the gastrointestinal tract (Jha and Berrocoso, 2015). The positive results observed in our study with pigs fed DDGS-containing diets indicate that this synergistic approach could be particularly beneficial in diets with a high inclusion of fibrous by-products.
The synergistic effect of dietary enzyme additives as a blend of exogenous enzymes on nutrient digestibility in diets containing DDGS has been reported (Swiatkiewicz et al., 2016). Despite the comparable positive effects of enzyme blends and the combination of Bacillus-based DFMs and enzymes on nutrient digestibility, the combination of DFMs and enzymes is preferable due to its ability to enhance nutrient digestibility, reduce disease incidence, and maintain stability under various feed processing conditions (Latorre et al., 2017; Park et al., 2020). Moreover, porcine in-vitro fermentation studies have revealed complex interactions between exogenous enzyme feed additives. Although multi-carbohydrase enzymes were found to increase the fermentability of DDGS, their combination with protease hindered the efficacy of the multi-carbohydrase (Jha et al., 2015). This antagonistic interaction, which limits the additive benefits of protease when combined with certain enzymes, may be less pronounced in protease and DFM combinations. The potentially reduced interference between protease and DFMs could explain the positive results observed in our study.
Within plant cells, phytate is located in cell contents and not within cell walls (Tervilä-Wilo et al., 1996). Thus, the increase in P digestibility due to the supplementation of a combination of protease and the DFM product could be attributed to the fact that the protease and the DFM product increased the degradation of cell walls, leading to increased availability of phytate to phytase produced by the DFM product. Lee et al. (2019) reported increased P digestibility in growing–finishing pigs due to the addition of fiber-degrading enzymes to phytase-supplemented diets, implying that the efficacy of phytase can be enhanced by fiber-degrading enzymes.
Supplementing an energy-, Ca- and digestible P-deficient corn DDGS-soybean meal-based diet with a combination of protease and Bacillus-based DFMs enhanced the ATTD of nutrients, highlighting its potential as a feed additive to enhance nutrient utilization in growing–finishing pigs. However, the overall growth performance (e.g., ADG, ADFI, FE) was not affected by dietary supplements. Future research is needed to focus on exploring the effects of adding a combination of protease and DFM products fed in the current study on the growth performance of pigs fed diets with lower net energy and nutrient contents than those fed in the current study.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The animal study was approved by the Institutional Animal Care and Use Committee at South Dakota State University (Protocol #15-083E). The study was conducted in accordance with the local legislation and institutional requirements.
HR-S: Formal Analysis, Methodology, Writing – original draft. AS: Formal Analysis, Writing – review & editing. SL: Writing – review & editing. DV: Writing – review & editing. EV: Writing – review & editing. TW: Conceptualization, Data curation, Funding acquisition, Methodology, Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was funded by Danisco Animal Nutrition & Health (IFF).
The authors thank Danisco Animal Nutrition & Health (IFF) for funding the research.
Authors DV, EV were employed by the company Danisco Animal Nutrition & Health (IFF).
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Agyekum A., Slominski B., Nyachoti C. (2012). Organ weight, intestinal morphology, and fasting whole-body oxygen consumption in growing pigs fed diets containing distillers dried grains with solubles alone or in combination with a multienzyme supplement. J. Anim. Sci. 90, 3032–3040. doi: 10.2527/jas.2011-4380
AOAC (1990). Official Methods of Analysis of AOAC International. 15th ed (Arlington, VA: Association of Official Analytical Chemists).
AOAC (2006). Official Methods of Analysis of AOAC International. 18th ed (Gaithersburg, MD: Association of Official Analytical Chemists).
Aranda-Aguirre E., Robles-Jimenez L. E., Osorio-Avalos J., Vargas-Bello-Pérez E., Gonzalez-Ronquillo M. (2021). A systematic-review on the role of exogenous enzymes on the productive performance at weaning, growing and finishing in pigs. Vet. Anim. Sci. 14, 100195. doi: 10.1016/j.vas.2021.100195
Augspurger N. R., Spencer J. D., Son S., Ley J. A., King M. R. (2016). Improved growth performance of nursery pigs fed diets supplemented with a Bacillus subtilis-based direct-fed microbial feed additive. J. Anim. Sci. 94, 76–76. doi: 10.2527/msasas2016-162
Cai L., Indrakumar S., Kiarie E., Kim I. (2015). Effects of a multi-strain Bacillus species–based direct-fed microbial on growth performance, nutrient digestibility, blood profile, and gut health in nursery pigs fed corn–soybean meal–based diets. J. Anim. Sci. 93, 4336–4342. doi: 10.2527/jas.2015-9056
Emiola I. A., Opapeju F. O., Slominski B. A., Nyachoti C. M. (2009). Growth performance and nutrient digestibility in pigs fed wheat distillers dried grains with solubles-based diets supplemented with a multicarbohydrase enzyme1. J. Anim. Sci. 87, 2315–2322. doi: 10.2527/jas.2008-1195
Holst D. O. (1973). Holst filtration apparatus for Van Soest detergent fiber analyses. J. Assoc. Off Anal. Chem. 56, 1352–1356. doi: 10.1093/jaoac/56.6.1352
Jacela J., Dritz S., DeRouchey J., Tokach M., Goodband R., Nelssen J. (2010). Effects of supplemental enzymes in diets containing distillers dried grains with solubles on finishing pig growth performance. Prof. Anim. Sci. 26, 412–424. doi: 10.15232/S1080-7446(15)30623-9
Jaworski N., Owusu-Asiedu A., Walsh M., McCann J., Loor J., Stein H. (2017). Effects of a 3 strain Bacillus-based direct-fed microbial and dietary fiber concentration on growth performance and expression of genes related to absorption and metabolism of volatile fatty acids in weanling pigs. J. Anim. Sci. 95, 308–319. doi: 10.2527/jas.2016.0557
Jha R., Berrocoso J. (2015). Dietary fiber utilization and its effects on physiological functions and gut health of swine. Animal 9, 1441–1452. doi: 10.1017/S1751731115000919
Jha R., Woyengo T., Li J., Bedford M., Vasanthan T., Zijlstra R. (2015). Enzymes enhance degradation of the fiber–starch–protein matrix of distillers dried grains with solubles as revealed by a porcine in vitro fermentation model and microscopy. J. Anim. Sci. 93, 1039–1051. doi: 10.2527/jas.2014-7910
Kabir S. M. L. (2009). The role of probiotics in the poultry industry. Int. J. Mol. Sci. 10, 3531–3546. doi: 10.3390/ijms10083531
Kellner T. A., Ellingson J., de Souza A. L., Remus J. C. (2021). Does feeding a protease and probiotic (Bacillus Subtilis) combination (Syncra® SWI) tesult in improved growth performance and mortality in a commercial wean-to-finish swine feeding program? J. Anim. Sci. 99, 77–78. doi: 10.1093/jas/skab054.127
Kerr B., Weber T., Shurson G. (2013). Evaluation of commercially available enzymes, probiotics, or yeast on apparent total-tract nutrient digestion and growth in nursery and finishing pigs fed diets containing corn dried distillers grains with solubles. Prof. Anim. Sci. 29, 508–517. doi: 10.15232/S1080-7446(15)30272-2
Latorre J., Hernandez-Velasco X., Vicente J., Wolfenden R., Hargis B., Tellez G. (2017). Effects of the inclusion of a Bacillus direct-fed microbial on performance parameters, bone quality, recovered gut microflora, and intestinal morphology in broilers consuming a grower diet containing corn distillers dried grains with solubles. Poult. Sci. 96, 2728–2735. doi: 10.3382/ps/pex082
Lee J. W., Patterson R., Rogiewicz A., Woyengo T. A. (2019). Nutrient digestibility of multi-enzyme supplemented low-energy and AA diets for grower pigs1. J. Anim. Sci. 97, 2979–2988. doi: 10.1093/jas/skz178
Lewton J. R., Woodward A. D., Moser R. L., Thelen K. M., Moeser A. J., Trottier N. L., et al. (2021). Effects of a multi-strain Bacillus subtilis-based direct-fed microbial on weanling pig growth performance and nutrient digestibility. Transl. Anim. Sci. 5, txab058. doi: 10.1093/tas/txab058
Li G., Wang X., Lin M., Lu Z., Yao W. (2012). Effects of corn DDGS in combination with compound enzymes on growth performance, carcass fat quality, and plasma and tissue redox homeostasis of growing-finishing pigs. Livest Sci. 149, 46–52. doi: 10.1016/j.livsci.2012.06.019
Min Y., Choi Y., Kim Y., Jeong Y., Kim D., Kim J., et al. (2019). Effects of protease supplementation on growth performance, blood constituents, and carcass characteristics of growing-finishing pigs. J. Anim. Sci. Technol. 61, 234. doi: 10.5187/jast.2019.61.4.234
Myers W., Ludden P., Nayigihugu V., Hess B. (2004). A procedure for the preparation and quantitative analysis of samples for titanium dioxide. J. Anim. Sci. 82, 179–183. doi: 10.2527/2004.821179x
NRC. (1998). Nutrient Requirements of Swine. Tenth Revised Edition (Washington, DC: The National Academic Press).
NRC. (2012). Nutrient Requirements of Swine: Eleventh Revised Edition (Washington, DC: The National Academies Press).
Pan L., Zhao P. F., Yang Z. Y., Long S. F., Wang H. L., Tian Q. Y., et al. (2016). Effects of coated compound proteases on apparent total tract digestibility of nutrients and apparent ileal digestibility of amino acids for pigs. Asian-Australas J. Anim. Sci. 29, 1761–1767. doi: 10.5713/ajas.16.0041
Park S., Lee J. W., Jerez Bogota K., Francis D., González-Vega J. C., Htoo J. K., et al. (2020). Growth performance and gut health of Escherichia coli-challenged weaned pigs fed diets supplemented with a Bacillus subtilis direct-fed microbial. Transl. Anim. Sci. 4, txaa172. doi: 10.1093/tas/txaa172
Payling L., Kim I. H., Walsh M. C., Kiarie E. (2017). Effects of a multi-strain Bacillus spp. direct-fed microbial and a protease enzyme on growth performance, nutrient digestibility, blood characteristics, fecal microbiota, and noxious gas emissions of grower pigs fed corn-soybean-meal-based diets—A meta-analysis1. J. Anim. Sci. 95, 4018–4029. doi: 10.2527/jas.2017.1522
Payling L., Woyengo T. A., Nielsen M., Stein H. H., Walsh M. C., Romero L., et al. (2019). Microscopy and protein solubilization of digesta from pigs fed wheat, corn, or soybean meal-based diets, with or without protease and a Bacillus spp. direct-fed microbial. Anim. Feed Sci. Technol. 247, 183–193. doi: 10.1016/j.anifeedsci.2018.11.013
Prenafeta-Boldú F., Fernández B., Viñas M., Lizardo R., Brufau J., Owusu-Asiedu A., et al. (2017). Effect of Bacillus spp. direct-fed microbial on slurry characteristics and gaseous emissions in growing pigs fed with high fibre-based diets. Animal 11, 209–218. doi: 10.1017/S1751731116001415
Reddy P. P. R., Chakrawarthi M. K., Reddy D. M., Venkateswarlu S., Reddy J. B., Babu P. R., et al. (2021). Effect of dried distillers’ grain with solubles as a replacer of peanut cake for sheep fed on low quality forage. Trop. Anim. Health Prod. 53, 374. doi: 10.1007/s11250-021-02821-0
Swiatkiewicz S., Swiatkiewicz M., Arczewska-Wlosek A., Jozefiak D. (2016). Efficacy of feed enzymes in pig and poultry diets containing distillers dried grains with solubles: a review. J. Anim. Physiol. Anim. Nutr. 100, 15–26. doi: 10.1111/jpn.12351
Tervilä-Wilo A., Parkkonen T., Morgan A., Hopeakoski-Nurminen M., Poutanen K., Heikkinen P., et al. (1996). In vitro digestion of wheat microstructure with xylanase and cellulase from trichoderma reesei. J. Cereal Sci. 24, 215–225. doi: 10.1006/jcrs.1996.0054
Thacker P. (2009). Effects of supplementary threonine, canola oil or enzyme on nutrient digestibility, performance and carcass traits of growing-finishing pigs fed diets containing wheat distillers grains with solubles. Asian-Australas J. Anim. Sci. 22, 1676–1685. doi: 10.5713/ajas.2009.90295
Yang Z., Urriola P. E., Hilbrands A., Johnston L. J., Shurson G. C. (2020). Effects of feeding high-protein corn distillers dried grains and a mycotoxin mitigation additive on growth performance, carcass characteristics, and pork fat quality of growing–finishing pigs1. Transl. Anim. Sci. 4, 666–681. doi: 10.1093/tas/txaa051
Yu G., Chen D., Yu B., He J., Zheng P., Mao X., et al. (2016). Coated protease increases ileal digestibility of protein and amino acids in weaned piglets. Anim. Feed Sci. Technol. 214, 142–147. doi: 10.1016/j.anifeedsci.2016.02.006
Keywords: apparent total tract digestibility, Bacillus subtilis protease, direct-fed microbial, feed additive, fattening pigs
Citation: Rajaei-Sharifabadi H, Seradj AR, Lashkari S, Velayudhan DE, Vinyeta E and Woyengo TA (2025) Growth performance and nutrient digestibility of grower–finisher pigs fed corn DDGS-soybean meal-based diets supplemented with a combination of protease and multi-strain Bacillus-based direct-fed microbial. Front. Anim. Sci. 6:1562308. doi: 10.3389/fanim.2025.1562308
Received: 17 January 2025; Accepted: 26 February 2025;
Published: 20 March 2025.
Edited by:
Michael D. Flythe, United States Department of Agriculture, United StatesReviewed by:
Ravikanth Reddy Poonooru, University of Missouri, United StatesCopyright © 2025 Rajaei-Sharifabadi, Seradj, Lashkari, Velayudhan, Vinyeta and Woyengo. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Tofuko Awori Woyengo, woyengo@anivet.au.dk
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.