
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Sustain. Food Syst. , 06 March 2025
Sec. Crop Biology and Sustainability
Volume 9 - 2025 | https://doi.org/10.3389/fsufs.2025.1511848
This article is part of the Research Topic Advancements in Post-Harvest Physiology and Technology of Fruits and Vegetables for Sustainable Food Production View all articles
 Sara Rached1
Sara Rached1 Amar Habsaoui1
Amar Habsaoui1 Khaoula Mzioud1
Khaoula Mzioud1 Mohammed Er-rajy2
Mohammed Er-rajy2 Feras Abujaber3
Feras Abujaber3 Hamada Imtara3,4*
Hamada Imtara3,4* Asmaa Oubihi5
Asmaa Oubihi5 Sara Haida1
Sara Haida1 Otman El-guourrami6
Otman El-guourrami6 Omar M. Noman7
Omar M. Noman7 Samah Ourras8
Samah Ourras8 Mahmoud Tarayrah9
Mahmoud Tarayrah9 Mohamed Ebn Touhami1
Mohamed Ebn Touhami1Mentha pulegium L., (MP) of the Lamiaceae family, endemic to Morocco, is a medicinal plant used in classical medicine. The aim of the present study is to enhance the value of this species by investigating the chemical composition of its essential oil (EO), antioxidant activity, toxicity, molecular docking and antimicrobial properties. In vitro measurement of antioxidant activity using the 2,2-diphenyl-1-picrylhydrazyl radical (DPPH) reduction assay and the 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid; ABTS) assay demonstrated the plant’s best performance. The toxicity test reveals that EO is non-toxic at an LD50 of 2000 mg/kg rat body weight. Evaluation of the in vitro antimicrobial effect by diffusion, direct contact, dilution and sporulation methods showed that all strains tested were sensitive to MP EO. Minimal inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) values for clinical strains ranged from 2 mg/mL to 10 mg/mL. MP could therefore be a good alternative in the treatment of infectious diseases.
The environment is generally a shining example of the obvious phenomenon of cohabitation. The treatment of human infections relies on the use of natural substances derived from plants, animals and minerals (Bunse et al., 2022). There is no doubt that plants make a significant contribution to ecosystems by providing necessary services. Man, and other living beings cannot exist, as they should without plants. In any case, the use of plants, particularly medicinal plants, has long served as a general indicator of ecosystem health (Jamshidi-Kia et al., 2018). Recourse to herbal medicine to prevent illness has a long history, which began when archeologists discovered the oldest trace of the use of medicinal plants for curative purposes, engraved on a Sumerian clay plate over 5.000 years ago. They then discovered other documents on the use of natural products throughout the ages, which were reported using different methods depending on the period of history in which they were used, such as the “Eber’s papyrus” of ancient Egypt, the Chinese Materia Medica, the Indian Ayurveda, and so on (Elkordy et al., 2021; Antoniou et al., 2023; Kloter et al., 2023). Various trials (sometimes by mistake) led to the discovery of plants and their use in the manufacture of food and medicines, and over time, man has been able to meet his needs by adapting to his environment. For a very long time, knowledge of medicinal plants was handed down from generation to generation, then with the development of civilizations and the evolution of techniques, the body of knowledge was gradually enriched (Ghareeb et al., 2020; Rached et al., 2022).
Morocco’s flora is richly varied, including a substantial potential for medicinal plants, many of which are endemic, due to its unique geographical location and favorable Mediterranean climate (Fennane and Tattou, 2008; Rhattas et al., 2016). According to reports, medicinal plant ethnobotanical use generated average annual revenues of 550 million Moroccan dirhams in 2015 (equivalent to $59.51 million). M. pulegium is the second most economically valuable of all aromatic and medicinal plants in the Taounat region (5,616 km2), after Capparis spinosa, at 6.12 million Moroccan dirhams per year (Lamiri et al., 2001; Mostafa, 2018). In the words of the Ministry of Agriculture and Maritime Fishing, M. spicata is planted on a surface area of 3,000 ha spread over several provinces, with a production of 85,000 tones, 5,000 tons of which are destined for export (Hassani, 2020).
In Morocco, for example, Mentha is found in almost every region, either spontaneously or cultivated, mainly in humid areas, so that its abundance is greatest in the mountains (Jafari-Sales et al., 2019). Varieties of Mentha, most commonly Mentha pulegium L., are widely used as insect repellents, herbal teas, aromatic plants and medicinal agents. Commonly consumed as an infusion, inhalation or chest poultice, it is used to treat colds, sore throats, coughs, bronchitis, lung infections and flu of all kinds, as well as a food condiment. It is the plant of choice for all winter ailments. It is also used to make scented tea, while its essential oils are blended with other EOs to produce fragrances, detergents and hygiene products (Di Stasi et al., 2002; Carlier-Loy, 2015). In infants suffering from aerophagia, compresses soaked in mint essence applied to the navel or massaged into the forehead relieve pain and migraine, respectively. However, the herbal tea of flowering branches and leaves has antispasmodic, antiseptic, cholagogue and insecticidal properties, while the flowering branch acts as an expectorant and disinfectant. Essential oils and menthol extracted from mint are widely used to flavor drinks, chewing gum and desserts (Aghfir et al., 2008; Mohammadi et al., 2024).
Essential oils and plant extracts from Mentha have been found in earlier studies to have strong antioxidant and insecticidal properties. Nevertheless, inappropriate use of essential oils can have harmful effects on humans, such as skin irritation, headaches and nausea. Generally speaking, caution is advised if essential oils are to be taken internally or used on food products, due to the possible carcinogenic effects of some of them. When applied in doses not recommended, essential oils can cause functional damage to organs such as the stomach and liver in animals, and probably in humans (Liaqat et al., 2018; Souza et al., 2022; Wu et al., 2023).
In traditional Moroccan medicine, Mentha pulegium L is widely used, particularly for respiratory diseases. It is considered the best remedy for winter illnesses such as colds of all kinds (Hseini and Kahouadji, 2007). For this reason, and given the widespread use of this plant, it is worth studying its toxicological properties with a view to optimizing its use. Despite the country’s abundant botanical diversity, there is a lack of pharmacological and phytochemical data. Therefore, it is essential for industry and science to contribute to the improvement of this biodiversity (Rached et al., 2022, 2023; Mzioud et al., 2023). Providing a thorough examination of the Mentha genus while highlighting the abundant biodiversity in western Morocco is the goal of this essay. Specifically, it aims to study the Zaër region’s Mentha pulegium L. (MP). The objective of this research is to identify and describe the bioactive substances found in this species in order to determine how they might be used in pharmacology, antimicrobial treatments, and natural antioxidants, among other areas.
The samples were harvested in the spring of 2021 in the Zaër area. After harvesting, the plant material was cleaned and rinsed to remove all debris. Drying was conducted under shade in an airy location and subsequently stored in dark, well-closed boxes until use. The botanical identification of the samples was performed by a team from Ibn Tofail University, Laboratory of Plant, Animal, and Agro-Industrial Production, Faculty of Science, under the supervision of Prof. ZIDANE Lahcen.
0.1 Kg of plant material (Mentha pulegium L) and distilled water (600 cm3) were placed in a flask, and the steam distillation process was carried out using the Clevenger device for 3 h. Our collected essential oil (volatile part) was stored in the dark at 277.15 K for later use. Oil storage required well-capped bottles protected from air and sunlight in order to prevent any risk of oxidation and polymerization. We calculated the essential oil content in relation to the total dry mass of the plant (Rached et al., 2022, 2023, 2024). The yield of EO was obtained by applying the following formula:
Where M1: mass of EO (Kg). M0: dry plant mass (Kg).
Physico-chemical properties enabled us to control and manage the quality of essential oils. For this reason, we calculated the following physico-chemical indices: refractive index, ethanol miscibility, peroxide value, acid value, ester value, and saponification value according to a methodology described in international standards (NM ISO) and the protocols of Rached et al. (2022).
The ratio between the sine angle of incidence and the sine angle of refraction of a light ray of a specified wavelength passing from the air into the essential oil is maintained at a constant temperature, as the refractometer would allow a direct reading of the refractive index.
An essential oil is considered miscible with V volumes or more of ethanol of determined alcoholic strength at a temperature of 293.15 K. To reach a limpid solution for 1 volume of essential oil, we need to progressively add at least 20 volumes of ethanol at 293.15 K.
This indicates the number of milligrams of potassium hydroxide required to neutralize the free acids contained in 1.10−3 Kg of essential oil. We weighted and introduced 2.10−3 Kg of oil into a 100 cm3 flask to which we added 5 cm3 of 95% ethanol and 5 drops of 0.2% phenolphthalein (PP). Then we titrated the solution with the potassium hydroxide solution (0.1 M) contained in the burette that we continuously added until the solution turned. The calculation of the AI is given following the formula:
Where, V is the volume in milliliters of potassium hydroxide solution used for the titration; c is the exact concentration in moles by liter of the potassium hydroxide solution; and m is the mass in grams of the test sample.
The ester indices are the number of milligrams of potassium hydroxide necessary to neutralize the acids released by the hydrolysis of the esters contained in 1.10−3 Kg of essential oil. We weighted and introduced 2.10−3 Kg of essential oil into a flask, then using a burette; we added 25 cm3 of the potassium hydroxide solution as well as fragments of pumice stone or porcelain. We fitted the condenser, placed the flask on the heating mantle and left it to heat for 1 h. After cooling the flask, we removed the tube and added 20 cm3 of water, followed by 5 drops of phenolphthalein solution. The excess potassium hydroxide that remained was titrated with the hydrochloric acid solution. At the same time, a blank was performed under the same conditions and with the same reagents. The calculation of the EI follows the formula:
Where, M: mass in grams of the test. V0: volume in mL of the HCl solution (0.5 M) used for the blank. V1: volume in cm3 of the HCl solution (0.5 M) used to determine the EI of the essential oil.
The peroxide is the number of micrograms of active peroxide contained in 1 gram of product and oxidizing potassium iodide to release iodine under the conditions of the method described. We put 1 gram of oil in a tube and placed it in an Erlenmeyer flask, then added 10 cm3 of chloroform and shaken the solution. We added 15 cm3 of acetic acid CH3COOH, followed by 1 cm3 of saturated aqueous KI, shaken the flask, and left it in the dark for 5 min. Add 75 cm3 of distilled water and shake for 1 min. Carefully titrate in the presence of starch and released iodine with a solution of Na2S2O3 (0.01 N) until complete discoloration of the solution. The calculation of the PI is given by the formula:
m is the mass of the test, V is the volume of the thiosulfate solution N/100.
The saponification index is the amount of soda expressed in grams needed to saponify 1.10−3 kg of oil. The saponification index is calculated according to the following formula:
Where, AI is the acid number and IE is the ester number.
The relative density of essential oils at 293.15 K was determined in accordance with the standard (NM ISO 279:2008). Using a 5 cm3 pycnometer, equal volumes of oil and water are weighed at a temperature of 20°C. The mass ratio of essential oil to water gives the density of our oily sample (Mzioud et al., 2023). This physical quantity is given by the following formula:
Where m0, m1 and m2 represent the mass of empty pycnometer, pycnometer filled with water and essential oil studied, respectively.
Our EO was analyzed at the Center Universitaire of Analysis, Expertise, Technology Transfer and Incubation CUE2TI, Université IBN Tofail Kenitra, and Technical Support Units for Scientific Research UATRS, CNRST Rabat, using a type 456-GC gas chromatograph coupled to an EVOQ TQ mass spectrometer. The energy applied was 70 eV, and the impact of the electrons caused fragmentation. The column is a capillary Rxi-5Sil MS column (30 m 0.25 mm ID 0.25 μm df). The column temperature was initially set at 313.15 K for 8 min, then increased by 275.15 K/min for the next 20 min. Helium was used as the carrier gas, with a flow rate of 1.50 mL/min. The injection volume was 1 μL and the injection temperature was maintained at 523.15 K. The instrument was managed by MS Data review software and connected to a computer system hosting a NIST MS Search 2.0 mass spectra collection.
The study protocol was approved by the Ethical Institutional Committee of the Faculty of Sciences at University Ibn Tofail, Kenitra, Morocco. All experimental procedures involving laboratory animals was carried out in compliance with the organization for economic cooperation and development (OECD) guidelines no. 42 (OCDE, 2002). An esophageal probed was used to administer the test substance in a single dosed. Three healthy, non-pregnant female swiss mice fasted for 3 to 4 h with watered were used at each stage of the experiment. The weight of the animals was measured after fasting, and the test substance was administered. In our case, a dosed of 300 mg/kg was administered, followed by a dosed of 2000 mg/kg, while distilled watered was administered to another group (the controlled group). After administration, the animals was again deprived of food for 1 to 2 h (OCDE, 2002; Oubihi et al., 2023).
Lethal dosed 50, often referred to as LD50, was a measured of a compound’s acute toxicity or short-term hazardous potential. The quantity of active ingredient was measured in milligrams per kilogram of animal body weight; the lowered the figure, the greater the toxicity of the product (OCDE, 2002). Mortality was recorded for 14 days after administration of the extracts tested, and the LD50 was determined.
We used the DPPH (1,1-diphenyl,2-picrylhydrazyl) technique to examine the antioxidant power of our EO (Laouini et al., 2012). By diluting the stock solution with ascorbic acid (0–0.2 mg/mL) used as a standard, solutions of increasing concentration were created. To prepare the DPPH solution, 3 mg DPPH was dissolved in 100 cm3 ethanol. We placed 0.1 cm3 of each solution in a tube containing 2 cm3 of freshly prepared DPPH solution, as per procedure. In addition to ethanol, a negative control (blank) was made. A spectrophotometer (UV-2005, Selecta, Barcelona, Spain) was used to measure the absorbance of the solution at 517 nm after it had been left in the dark for 30 min. Three readings was taken for each solution (Rached et al., 2022). The following formula describes how antioxidant activity is expressed as a percentage of DPPH radical inhibition (Said et al., 2018):
% IP means percentage inhibition and Abs means absorbance.
Using this formula, we plot the straight line (y = ax + b) representing the variation in percentage inhibition as a function of different concentrations in each sample. From this line, it was possible to deduce the concentration that reduces 50% of DPPH for the sample studied and for ascorbic acid. This concentration, called IC50, was calculated according to the following equation:
Where IC50 was the concentration required to reduce 50% of the DPPH, a was the slope of the line, and b was the intercept of the line.
The ABTS radical trapping test was carried out using the methodology of Obanor et al. (2013) and Eddine et al. (2016). Oxidation of ABTS by potassium persulfate (K2S2O8) produces the ABTS radical. To create ABTS cations, a solution of ABTS (7 mM) was combined with a solution of potassium persulfate (70 mM) in an equivalent proportion. The combination was maintained at room temperature and in the dark for 16 h. The ABTS radical cation was diluted with methanol to give an initial absorbance of around 0.700 at 734 nm before being used in the assay. Different amounts of each extract (100 μL) were added to 2 cm3of ABTS solution for the studied. Absorbance was measured at 734 nm and the % inhibition was determined as described above for DPPH.
The fungal strains studied, Fusarium pseudograminearum and Fusarium moniliforme, belong to the mycotheque of the plant, animal and agro-industrial production laboratory at the Faculty of Science, Kenitra. They were isolated from saffron and regularly transplanted onto PDA (potato dextrose agar) culture medium. Morphological characteristics was used for identification, which was then confirmed at CNRST in rabat.
The antifungal power of our essential oil was tested in our study using the direct contact method (Mzioud et al., 2023; Rached et al., 2023) on isolated mold species, namely Fusarium pseudograminearum and Fusarium moniliforme. The procedure involved pouring a volume V of essential oil or extract into 20 cm3 of PDA medium in test tubes. After shaking, glass Petri dishes with a diameter of nine cm was filled from the medium. For inoculation, a disk of mycelium measuring 0.6 cm in diameter was placed in the middle of the dish after preculture for 3 to 7 days at 301.15 K. As a control, a Petri dish containing 20 cm3 of PDA without EO was inoculated. For each concentration, three tests were performed. The antifungal index was determined after 7 days incubation at 28°C, taking into account the expansion of the control mycelium:
Where, Da and Db represent the dimension of the test growth zone and is the dimension of the control growth zone.
The activity of our essential oil on sporulation was evaluated with the cultures where the fungistatic effect was observed. The Malassez cell was used to count the spores recovered after dilution in distilled water, and the results was expressed as the number of spores/mL of suspension. The following formula was used to calculate the number of spores:
The Muller-Hinton agar diffusion method was used to measured antibacterial activity. The agar medium was placed on a petri dished with a microbiological solution having an optical density of 1 McFarland (Božović et al., 2015). The EO has been assessed was placed on 6 mm diameter Whatman absorbent paper disks. The disks were then placed on the surface of the seeded agar, and incubated for 24 h at 310.15 K. After incubation, the appearance of an area of inhibition surrounding the impregnated disks, in which no micro-organisms develop, demonstrates their sensitivity to EO. EO diffuses uniformly as soon as the impregnated disks were applied. The size of the inhibition area determines the sensitivity of a given pathogen. Penicillin (5 μg), oxacillin (5 μg) and amoxicillin (25 μg) were used individually as positive controls (Oubihi et al., 2020).
The macro-dilution approached in solid media was used to calculate MIC (Jaber et al., 2021). To ensured that extracts were evenly distributed in the medium, EO was emulsified in a 0. 2% agar solution. In this agar solution, dilutions of 100, 40, 20, 1, 5, 3.3, and 2 μL/mL were performed. 1.5 cm3 of each dilution was aseptically added to test tubes containing 13.5 cm3 of solid MH medium to give final concentrations of 10, 4, 2, 1, 0.5, 0.33, and 0.2 μL/mL. After 15 s’ agitation, the contents of each tube were instantly emptied into a sterile petri plate. In addition, controls comprising only culture medium was performed. Used a calibrated platinum loop to collected the same quantity of inoculum, inoculation was carried out in streaks. A 24-h incubation period was carried out at 310.15 K. The first plate in the range without bacterial growth was used to define the MIC. Each experiment was performed three times.
The smallest concentration of an EO that can destroy more than 99.9% of the bacterial population initially inoculated is called the Minimum Bactericidal Concentration (MBC). It describes the antibacterial properties of an essential oil. After reading the MIC the following day, or by plating on Mueller-Hinton agar, samples from each plate where growth was not observable to the naked eye were taken to determine the MBC. The latter represents the lowest dose that produces no subculture after the infected plates had been incubated for 24 h at 310.15 K (Jaber et al., 2021).
The docking technique was equipped with tools that facilitate understanding the interactions between a presumed rigid active site and a ligand molecule, making it a valuable approach in drug discovery (Er-rajy et al., 2023a, 2023b). Molecular docking investigations was conducted to evaluate the interactions of compounds, identified through GC–MS based on major peak areas, focusing on three activities: antifungal, antoxidant and antibacterial (Ahmed et al., 2018).
The two major ligands, Menthone and Pulegone, was downloaded from the PubChem database, and biomacromolecules (Code ID: 6PXX, 2BJI, 3K4Q and 2CDU) was acquired in protein data bank (PDB) format (Gill et al., 2005; Papp-Wallace et al., 2019). Ligands was prepared as PDB files to facilitate molecular research using various software tools, including Discovery Studio and ChemOffice (Baroroh et al., 2023). In addition, Discovery Studio was used to prepare the proteins obtained from the PDB base. The ligands’ structures was docked into the enzyme’s active site using AutoDockTools (Morris et al., 2008). The interactions between enzyme active-site residues and ligand molecules was analyzed using Discovery Studio Visualizer, which included examining 2D interactions (Er-rajy et al., 2023a).
To represent yield, a percentage is used, calculated on the basis of 100 g of dry plant material. By hydrodistillation, the essential oil yield for Mentha pulegium L., is 2.31%. This proportion is the same as that found by Bouhaddouda et al. (2.69%; Bouhaddouda et al., 2016). and higher than that reported by Derwich et al., (1.66%; Derwich et al., 2010) and Aljaiyash et al., between 1.10 and 1.78% (Ahmed et al., 2018). So if we compare the yields for the same species according to the condition of the fresh plant (1.43%) or dry, MP in the dry state is more reproducible (Rached et al., 2024). Several factors may explain this difference in performance, including variation in botanical origin, environmental factors and cultivation practices, maturity and the part of the plant used, external factors (temperature, humidity, drought, etc.), seasonal variation and photoperiod.
One of the criteria used to assess the quality of essential oils is their physical and chemical properties. The results of the physical and chemical properties are shown in Table 1. With reference to the characterization methods chosen, the physico-chemical analysis of our essential oil showed that MP’s oil is characterized by stability and freshness, with an index value of less than 2, and a low peroxide value, testifying to the high quality of our oil. In addition, MP recorded a high ester index of 28.050. The higher the quantity of ester in the oil, the higher its quality (Rached et al., 2022; Mzioud et al., 2023). The conclusion drawn from the results of the physicochemical characterization tests is that our essential oil is fresh and of good quality in relation to Moroccan normative values.
GC/MS analysis identified nine components from MP EO, corresponding to 92.50% of its composition. The results are shown in Table 2 and Figure 1. In terms of our chemical composition (Table 2), M. pulegium L is essentially made up of monoterpene compounds, the most prevalent of which, accounting for 80.038% of the total, is pulegone. Accompanied by α-pinene (0.137%), 3-Octanol (0.285%), D-Limonene (0.49%); L-Menthone (8.137%), Cis-Isopulegone (1.368%), Menthol (0.269%), Piperitenone (1.654%) and (+)-Mintlactone (0.124%). Therefore, MP EO from the Zaër region, Morocco has a pulegone chemotype.
The literature reveals that Moroccan MP EO is characterized by the preponderance of (+)-pulegone, which can be as high as 90%, associated with other monoterpenic ketones: isomenthone, menthone, piperenone, within which pulegone is a colorless liquid with an aromatic odor (Amalich et al., 2016). Our results are compatible with the majority of work already carried out in Morocco, which confirms that Moroccan spearmint EO is very rich in pulegone. Moreover, Reis-Vasco et al. (1999) and Agnihotri et al. (2005) also found similar primary chemicals with the same proportions for MP from Sintra [Portugal; Pulegone (78.3–80.9), Menthone (8.5–9.2)] and Jammu Region and Kashmir Valley [India; Pulegone (65.9–83.1), Menthone (8.3–8.7)] respectively. Menthol and pulegone, present in the EO of Mentha species, are the substances that give mints their characteristic aromas and flavors.
Uwineza et al. (2018) show that MP EO from Settat, Morocco, contains 84.75% pulegone, Menthone 0.21%, Geraniol 14.72%, Isopulegyl acetate 0.02%, Nopol 0.13%, and “trans-” Cinnamyl alcohol, Ocimenone E, Myristicin with percentages of 0.03; 0.05; 0.05, respectively. Meanwhile, research carried out in Tunisia by Agnihotri et al. (2005) and Hajlaoui et al. (2009), revealed that pulegone is the predominant component of MP, with concentrations of 44.27 and 61.11%, respectively. In fact, compared with the results of Boukhebti et al., ours is more than twice as good (Boukhebti et al., 2011). Chromatographic analysis of EO derived from pennyroyal mint from the Moroccan Middle Atlas revealed that pulegone (68.86%) and piperenone (24.79%) dominate Azrou EO. Other compounds identified, but in relatively small percentages, are chrysanthenol (1.03%), thymol (1.01%), limonene (0.9%) and menth-2-ene-1-ol (0.57%).
M’rirt EO is also distinguished by two components whose proportions exceed those of Azrou, namely pulegone (71.97%) and piperenone (26.04%), to which are added chrysanthenol (0.80%), menth-2-ene-1-ol (0.28%), α-pinene (0.14%) and β-pinene (0.13%). Nevertheless, compared with the other two regions, the EO from Khénifra is essentially loaded with pulegone at a higher rate (81.46%). The presence of other compounds was also identified, but at low levels, such as α-humelene (2.89%), E-caryophyllene (1.70%), isopulegol (0.55%), 10-epicubelol (0.51%), α-pinene (0.23%) and β-pinene (0.22%; Zekri et al., 2017). In comparison with these different stations, our EO from Zaër is richer in pulegone (80.038%) than those from M’rirt and Azrou, and equal to that from Khenifra.
In the field of pharmacology, the capacity of a plant or even a natural principle to act is not enough to justify its therapeutic use. Indeed, any bioactive molecule runs the risk of having negative effects on human health, at least over the long term and in high doses. Beyond its performance, the active dose must be purified of any toxicity and made to appear harmless. It is essential to determine the benefit/risk ratio of any substance within the framework of a therapeutic indication (El Omari, 2021; Maniben et al., 2023). Toxicity tests are essential for estimating the level of damage caused by compounds to biological and non-biological materials. This type of testing is often carried out on potential products in order to develop new drugs and determine the therapeutic potency of a drug molecule. Acute toxicity tests define the effect of a single dose on certain species of laboratory animals (Falya et al., 2020; Oubihi et al., 2023).
In accordance with OECD Guideline 423, the acute toxicity of MP HE was determined. The results are summarized in Figures 2A,B. General behavioral control is one of the essential indicators to be checked, as are changes in body weight, which are among the early signs of toxicity. During the 14 days of EO administration, the behavior of our mice was monitored and mortality recorded. We emphasize that our essential oil administered at levels of 300 mg/kg and 2000 mg/kg showed no symptoms of toxicity or mortality, and all animals tested survived the 14 days of observation.
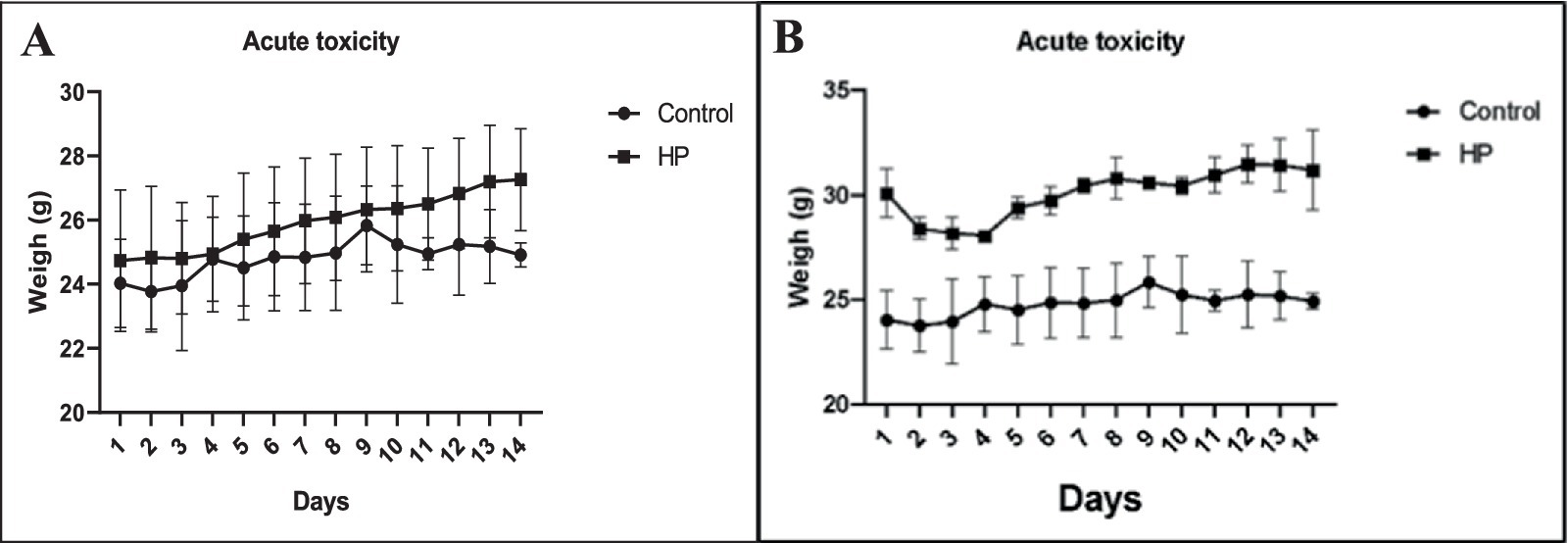
Figure 2. Development of the average weight of mice receiving MP essential oil orally versus time in days, as well as in the control group at 300 mg/kg (A) and 2000 mg/kg (B).
Monitoring the increase in body weight of mice given mint oil (Figure 2B), we noted a rapid and significant decrease in body weight, which stabilized from day 5 onwards. We know that the adverse effects of a drug or substance are accompanied by a change in body weight, which may explain the changes in body weight observed during the first 4 days of the experiment (Deyno et al., 2020). We assume that the majority pulegone in the essential oil (80.03%) is responsible for this effect. All these data imply that the lethal dose (LD50) of our sample is greater than 2000 mg/kg.
According to numerous scientific studies, antioxidant drugs are the only approach to preventing and treating oxidative stress-related diseases such as Alzheimer’s and cancer. However, given their side-effects and high cost, most people choose to treat themselves with herbal remedies, which have fewer side-effects (Eddine et al., 2015; Haida et al., 2022; Rached et al., 2022).
Plants are known to generate a number of antioxidant molecules, such as phenolic acids, flavonoids and tannins, whose ability to prevent ROS-induced oxidative stress is very important (Eddine et al., 2013; Ahmed et al., 2018). Compared with synthetic antioxidants, these are used in preventive medicine with few or no negative side effects (Laouini and Ouahrani, 2017; Omari et al., 2019). Table 3 shows the IC50 values representing the concentration of our EO to inhibit 50% of the DPPH / ABTS free radical. Analysis of the values obtained enabled us to conclude that ascorbic acid has a very powerful antiradical activity, with IC50 = 0.086 mg/mL and linear regression (R2 = 0.993). This value is similar to that obtained by Bentabet et al., (0.08 mg/mL; Bentabet et al., 2014).
In addition, our essential oil has a very interesting DPPH free radical scavenging capacity, with an IC50 equal to 1.550 mg/mL. This can be explained by the nature and type of chemical constituents present in our oil, e.g., Menthone, Eugenol, Menthol, Pulegone and Limonene, etc., which are known for their antioxidant effects. In our case, we can assume that the IC50 = 1.550 mg/mL of MP essential oil is due to pulegone (80.038%), the majority compound in this oil, which is known to be a good antioxidant, which may also justify the high antioxidant power of our EO, unlike that found by Bouhaddoda, where she noted the total absence of pulegone (Bouhaddouda, 2016). As a result, our findings on DPPH radical scavenging activity demonstrated that our tested plant could be used as a promising source of antioxidant molecules, while respecting the notion of dose dependency.
On the basis of the results illustrated in Table 3, we observe that the EO studied displays inhibition of ABTS●+ radicals. The IC50 value is 29.320 mg/mL. Nevertheless, it revealed a lower scavenging activity than that of the potent synthetic antioxidant ascorbic acid (IC50 = 0.002 mg/mL). We share similar results to that found by Ait chaouche, who revealed that Lamiaceae, principally pennyroyal mint, had the most potent antioxidant activity (Ait Chaouche, 2018).
Compared with other Moroccan aromatic and medicinal plants, our EO is more effective than Myrtus communis (Myrtaceae) essential oil with IC50 = 4748.33 μg/mL, Allium sativum (Liliaceae) essential oil with IC50 = 3.630 mg/mL (Liliaceae) and almost equal to Artemisia herba alba (Lamiaceae) IC50 = 1118.50 ± 3.97 μg/mL, Lippia citriodora (Verbenaceae) IC50 = 1335.63 μg/mL and Marrubium vulgare L., (Lamiaceae) with a 1.136 mg/mL (Bouyahya et al., 2017; Rached et al., 2022; Mzioud et al., 2023).
The antifungal power of plant drugs has been the subject of numerous in vitro experiments. Many of these have focused on the potential application of EOs and plant extracts in the biological control of microscopic fungi (Rached et al., 2022; Mzioud et al., 2023). The results of our research show the inhibitory effect of our M. pulegium L. plant on the fungal strains selected, and can be applied to inhibit the growth of these fungi.
Interpretation of the mycelial growth results of molds exposed to the action of our volatile fraction of different concentrations tested reveals that our results are positive and that the degree of inhibition is a function of both the concentration of EO used and the type of mold tested. In terms of figures, inhibition values range from 17.977 to 100.00% (Figure 3). MP EO was fungicidal against both fungi tested at concentrations of 2.5 and 5.00 μL/mL in Fusarium moniliforme and Fusarium pseudograminearum, respectively. This suggests that Fusarium moniliforme is more sensitive than Fusarium pseudograminearum to MP EO.
Plant extracts, and especially essential oils, have long been recognized for their antifungal properties, and the first biological control strategy was introduced with Antoferine®, a vine extract rich in polyphenols. Today, chemists, doctors, biologists and the food industry are increasingly interested in essential oils as a means of treating certain infectious diseases, for which synthetic antibiotics are becoming less and less effective, or as a means of preserving foodstuffs against oxidation, replacing synthesized chemical substances. These studies are part of the “Could plant extracts be the answer to mycotoxins, ask the plants: “Plants for plants” concept (Uwineza et al., 2018; Chelaghema, 2021).
In order to determine whether the effect of our essential oil and extracts also influenced the sporulation of our fungi, or only mycelial growth, we adopted a sporulation study. The results presented in Table 4 show sporulation rates decreasing in proportion to the concentration of the essential oil tested in the medium. However, at 0.08 and 2.50 μL/mL, the sporulation reduction rate is 100% for Fusarium pseudograminearum and Fusarium moniliforme, respectively. The quantity of spores is often greater in Fusarium moniliforme, which is more resistant to the antifungal activity of our essential oil. Fusarium pseudograminearum is therefore more sensitive to our essential oil than Fusarium moniliforme. Our essential oil therefore showed satisfactory results and excellent effects on sporulation of the two strains tested, and can be used as an antifungal agent, since the results reveal significant inhibition of spore production and mycelial growth of the two Fusarium genera tested. Nevertheless, their actions on sporulation are rather favorable.
We have determined that an essential oil is antibacterial if its zone of inhibition is greater than or equal to 8 mm, in accordance with the literature (Ponce et al., 2003). Our study revealed that MP EO was highly effective against all isolates, with inhibition zones of up to 14 mm against Staphylococcus epidermidis; 11 mm against Klebsiella pneumoniae; 10 mm against Staphylococcus aureus; and 9 mm against Escherichia coli; Acinetobacter baumannii; Enterobacter cloacae (Table 5). In addition, this EO revealed a significant antibacterial power to the strains examined in comparison with the antibiotics chosen as controls, namely Penicillin (5 μg), Oxacillin (5 μg) and Amoxicillin (25 μg). In general, Gram- bacteria were more resistant. It is known that EOs have a greater effect on Gram+ bacteria than on Gram- bacteria.
The limited antibacterial activity of our EO against Gram negative bacteria observed in this experiment is, however, consistent with previous research. This observation has been attributed to the property of these bacteria of possessing a hydrophilic polysaccharide chain that forms a barrier against hydrophobic essential oils (Boukhebti et al., 2011; Mahendran et al., 2021). EOs enriched with phenolic compounds are known to possess a high level of antimicrobial activity. In fact, these compounds are considered to have the highest antimicrobial activity, followed by aldehydes, ketones and alcohols.
The experimental data collected in Table 6 show that compared with the control group, we observed a drop in the aggressiveness caused by the bacteria tested in the presence of our essential oil.
By inhibiting bacterial growth according to a dose–response relationship, we demonstrated that MP EO has antibacterial activity. These results enabled us to identify the antibacterial parameters MIC and MBC. Staphylococcus aureus and Staphylococcus epidermidis showed a MIC = 2 μL/mL, Escherichia coli and Acinetobacter baumannii recorded MICs of 10 μL/mL and 4 μL/mL respectively, while Klebsiella pneumoniae showed a MIC>10 μL/mL. The bacteriostatic effect of our HE therefore varies according to the strain tested and the concentration. However, the bactericidal effect (MBC) against Staphylococcus aureus, Staphylococcus epidermidis and Acinetobacter baumannii is recorded at a concentration of 4 μL/mL. However, for Escherichia coli and Enterobacter cloacae, a concentration of 10 μL/mL is needed to completely kill these microbial germs, which are more resistant to our oil. MP EO is therefore active against the strains tested.
Zekri et al. (2017) found that minimum inhibitory doses differed according to the species tested and the germ targeted in his study of numerous Moroccan mints. They ranged from 0.30 to 1.2 μL/mL for Mentha spicata, from 1.20 to 9.60 μL/mL for MP, and from 9.60 to 19.20 μL/mL for Mentha suaveolens. Bacterial concentrations ranging from 19.20 to 76.80 μL/mL for MP, from 19.20 to 76.80 μL/mL for Mentha spicata, and from 38.40 to 76.80 μL/mL for Mentha suaveolens were used to destroy the bacteria. Meanwhile, Medjekal et al., (Samir et al., 2017) have noted that this oil has an antibacterial effect at a concentration of 20% against E. coli and Staphylococcus. However, MP of Portuguese origin demonstrated an inhibitory action with a MIC of 1.2 mg/mL.
Computer-aided drug design leverages molecular docking as a powerful technique for elucidating how protein receptors interact with ligands, providing crucial insights into binding affinities, interaction mechanisms, and the potential for optimizing drug efficacy and specificity. The antibacterial activity of the various extracts from our plant shows that the main compounds identified by GC–MS have an effect on the Klebsiella pneumoniae receptor (PDB ID: 6PXX). We performed molecular docking of these compounds, which revealed that all of them exhibited high negative binding energy values, ranging from −4.63 to −4.95 kcal/mol. The results of molecular docking of two molecules identified with the two target proteins are shown in Figure 4.
Figure 4 shows the results of the molecular docking study showed the Menthone compound exhibited one hydrogen bonding interactions with the protein residues Arg-214, with a distance greater than 2.06 Å, and also three Pi-alkyl and alkyl bonds with the protein residues Trp-105, Ala-69 and Val-120, with a distance greater than 3.96 Å, 3.88 Å and 4.52 Å, respectively. In addition, the results of the molecular anchoring study are shown in Figure 4, which demonstrated that the Pulegone compound exhibited a hydrogen bond interaction with protein residues Arg-250, with a distance greater than 6.13 Å, as well as one alkyl bonds with protein residues Ala-69, with a distance greater than 4.42 Å. The results suggest that menthone and pulegone are potential inhibitors of the Klebsiella pneumoniae receptor and could be used as antibacterial agents.
Similarly, the antibacterial activity of the various extracts from our plant shows that the main compounds identified by GC–MS have an effect on the Staphylococcus epidermidis receptor. We performed molecular docking of these compounds, which revealed that all of them exhibited high negative binding energy values, ranging from −4.63 to −4.95 kcal/mol.
The molecular docking results of two frequently identified molecules with the Staphylococcus epidermidis receptor (PDB ID: 2BJ1) are presented in Figure 5, highlighting their binding affinities and interaction profiles.
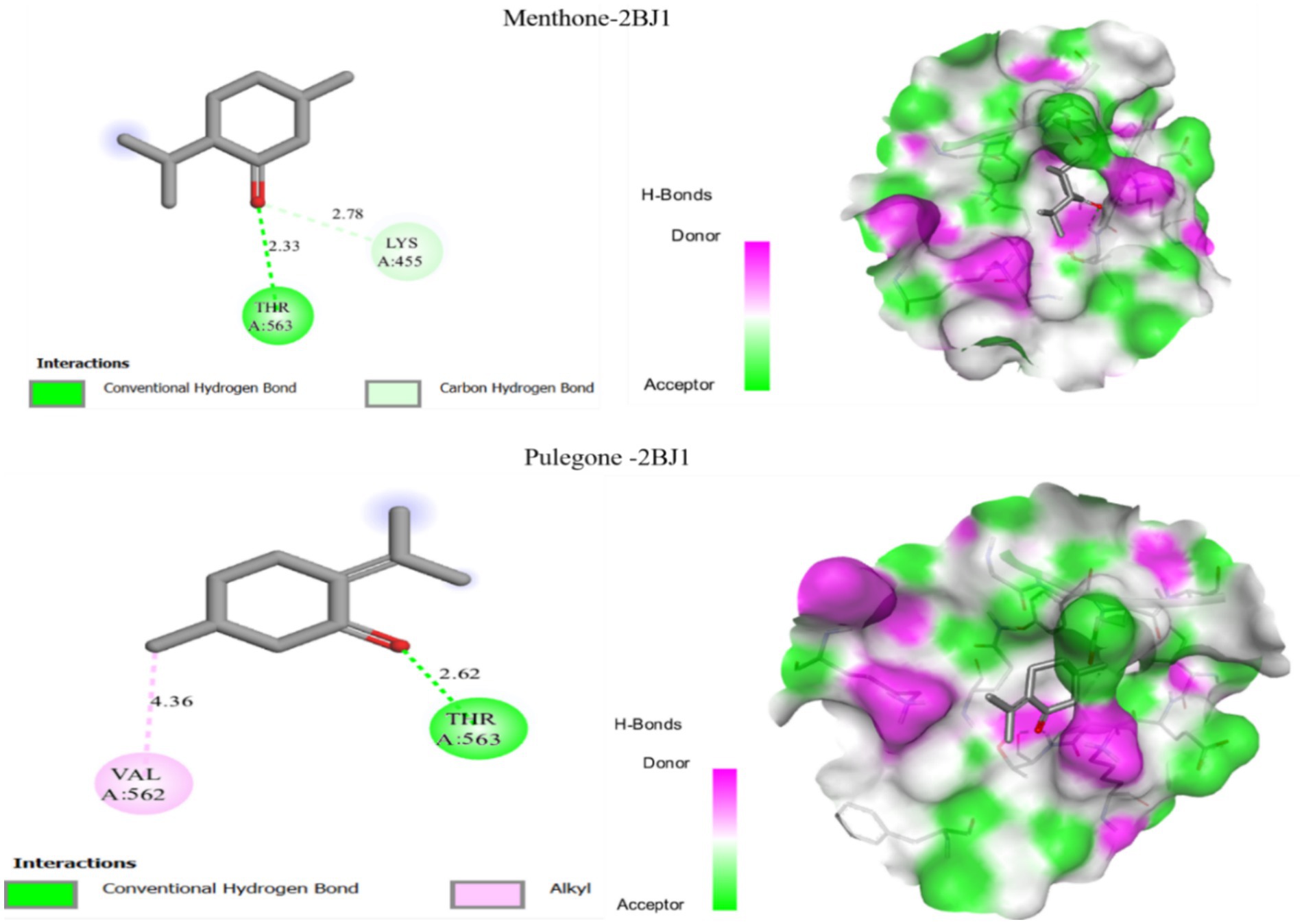
Figure 5. Outcomes of molecular docking of two ligands, menthone and pulegone, with the target protein 2BJ1.
Figure 5 presents the molecular docking results, revealing that the Menthone compound forms a hydrogen bond with the protein residue Thr-365 at a distance of more than 2.33 Å, as well as a carbon-hydrogen bond with Lys-455 at a distance greater than 2.78 Å. Additionally, the molecular docking results for the Pulegone compound indicate a hydrogen bond with Thr-563 at a distance of over 2.66 Å, and an alkyl bond with Val-562 at a distance greater than 4.36 Å. These findings suggest that both Menthone and Pulegone may serve as potential inhibitors of the Staphylococcus epidermidis receptor, highlighting their potential as antibacterial agents.
The fungal activity of the extract from our plant shows that the main compounds identified by GC–MS have an antifungal effect (PDB ID: 3K4Q). We performed molecular docking of these compounds, which revealed that all of them exhibited high negative binding energy values, ranging from −4.63 to −4.95 kcal/mol. The results of molecular docking of two molecules identified with the target proteins are shown in Figure 6.
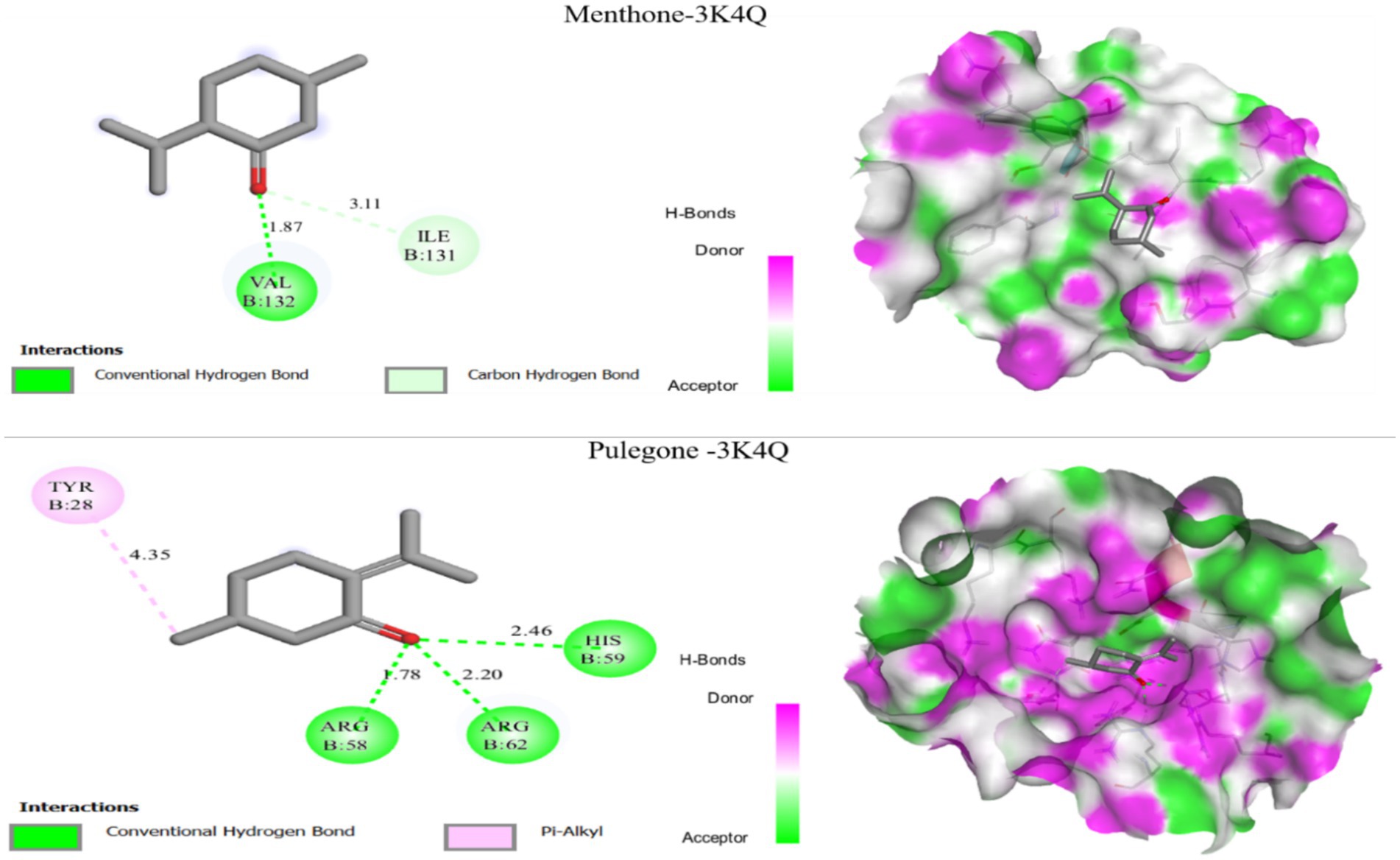
Figure 6. Molecular docking results of two ligands menthone and pulegone with the three target proteins 3K4Q.
Figure 6 presents the molecular docking results, showing that the Menthone compound forms a hydrogen bond with the protein residue Val-132 at a distance of over 1.87 Å, along with a carbon-hydrogen bond with Ile-131 at a distance greater than 3.11 Å. Additionally, the molecular docking analysis for the Pulegone compound revealed three hydrogen bonds with the protein residues His-59, Arg-69, and Arg-58, at distances of 2.46 Å, 2.20 Å, and 1.78 Å, respectively, as well as an alkyl bond with Tyr-28 at a distance greater than 4.35 Å. These findings suggest that menthone and pulegone could act as potential inhibitors of the fungal receptor, indicating their potential as antifungal agents.
The oxidant activity of the various extracts from our plant shows that the main compounds identified by GC–MS have an antioxidant effect (PDB ID: 2CDU). We performed molecular docking of these compounds, which revealed that all of them exhibited high negative binding energy values, ranging from −5.95 to −5.65 kcal/mol. The results of molecular docking of two molecules identified with the target proteins are shown in Figure 7.
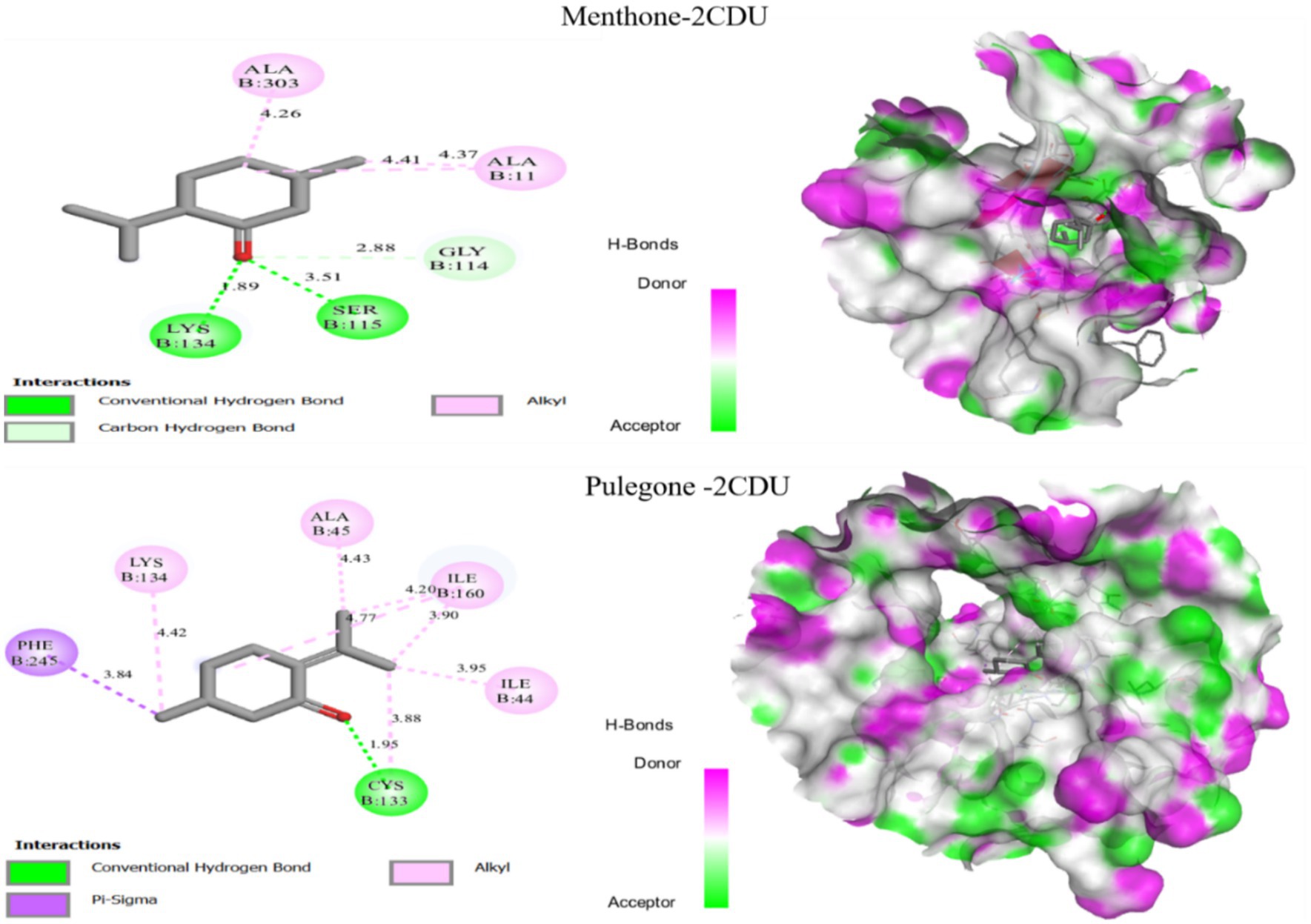
Figure 7. Results of the molecular docking of two ligands, menthone and pulegone, with the target protein 2CDU.
The results of molecular docking are presented in Figure 7, showing that the menthone compound forms two hydrogen bonds with the protein residues Ser-115 and Lys-134 at distances of 3.51 Å and 1.89 Å respectively, and two alkyl bonds with Ala-303 and Ala-11 at distances of 2.26 Å and 4.37 Å, respectively. Moreover, molecular anchoring analysis of the Pulegone compound revealed a hydrogen bond with the protein residue Cys-133 at a distance of 1.95 Å, as well as three alkyl bonds with Ile-44, Ile-60, Ala-45 and Lys-134, and a Pi-Sigma bond with the protein residue Phe-245. This suggests that menthone and pulegone may act as potential inhibitors of the oxidation receptor, indicating their potential as antioxidant agents. After an in-depth molecular docking study of the two most identified compounds in essential oils, we found that these compounds interacted and correlated with the binding sites of various selected proteins. Consequently, it can be concluded that these compounds exhibit significant antifungal, antioxidant, and antibacterial activities.
This study underscores the importance of preserving and valorizing traditional knowledge and biodiversity in the sustainable development of healthcare systems. The findings highlight that the essential oil (EO) of Mentha pulegium serves as a significant reservoir of bioactive compounds, such as pulegone, menthone, and limonene, and meets international standards of freshness and quality. Its notable antioxidant properties and potent antimicrobial activities against bacterial and fungal strains further emphasize its therapeutic potential. Specifically, the EO demonstrated bacteriostatic and bactericidal effects that varied according to the microbial strain and concentration, with strong activity against Staphylococcus aureus, Staphylococcus epidermidis, and Acinetobacter baumannii.
Additionally, its exceptional antifungal activity against Fusarium strains reinforces its broad- spectrum efficacy. Furthermore, molecular docking results corroborated the experimental outcomes, confirming the potential of Mentha pulegium EO as a promising candidate for therapeutic applications. This work not only contributes to advancing the scientific understanding of traditional herbal medicine but also provides a foundation for integrating this ancestral knowledge into modern healthcare practices.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/supplementary material.
The study protocol was approved by the Ethical Institutional Committee of the Faculty of Sciences at University Ibn Tofail, Kenitra, Morocco. All experimental procedures involving laboratory animals were carried out in compliance with the Organization for Economic Cooperation and Development (OECD) guidelines No. 42. The study was conducted in accordance with the local legislation and institutional requirements.
SR: Conceptualization, Formal analysis, Methodology, Writing – original draft. AH: Conceptualization, Formal analysis, Methodology, Writing – review & editing. KM: Methodology, Writing – original draft. ME-r: Formal analysis, Software, Validation, Writing – review & editing. FA: Formal analysis, Investigation, Resources, Writing – review & editing. HI: Data curation, Resources, Software, Validation, Writing – review & editing. AO: Formal analysis, Writing – review & editing. SH: Formal analysis, Writing – review & editing. OE-g: Formal analysis, Writing – review & editing. ON: Investigation, Resources, Writing – review & editing. SO: Data curation, Writing – review & editing. MT: Investigation, Resources, Writing – review & editing. MET: Conceptualization, Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. Researchers Supporting Project number (RSPD2025R1087) King Saud University, Riyadh, Saudi Arabia.
The authors extend their appreciation to the King Saud University, researchers supporting project number (RSPD2025R1087), King Saud University, Riyadh, Saudi Arabia.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Aghfir, A., Akkad, S., Rhazi, M., Kane, C. S. E., and Kouhila, M. (2008). Détermination du coefficient de diffusion et de l’énergie d’activation de la menthe lors d’un séchage conductif en régime continu. J. Renewable Energies 11, 385–394. doi: 10.54966/jreen.v11i3.90
Agnihotri, V. K., Agarwal, S. G., Dhar, P. L., Thappa, R. K., and Baleshwar, K. (2005). Essential oil composition of Mentha pulegium L. growing wild in the North-Western Himalayas India. Flavour Fragr. J. 20, 607–610. doi: 10.1002/ffj.1497
Ahmed, A., Ayoub, K., Chaima, A. J., Hanaa, L., and Abdelaziz, C. (2018). Effect of drying methods on yield, chemical composition and bioactivities of essential oil obtained from Moroccan Mentha pulegium L. Biocatal. Agric. Biotechnol. 16, 638–643. doi: 10.1016/j.bcab.2018.10.016
Ait Chaouche, F. S. (2018). Composition chimique et activité antioxydante, antimicrobienne et insecticide des huiles essentielles et des extraits de deux Lamiaceae. Available at: http://catalogue.ensa.dz/bib/30359
Amalich, S., Zerkani, H., Cherrat, A., Dédianhoua, N., Soro, K., Bourakhouadar, M., et al. (2016). Study on Mentha pulegium L. from M’rirt (Morocco): antibacterial and antifungal activities of a pulegone-rich essential oil. J. Chem. Pharm. Res. 8, 363–370.
Antoniou, V., Gauhar, V., Modi, S., and Somani, B. K. (2023). Role of Phytotherapy in the management of BPH: A summary of the literature. J. Clin. Med. 12:1899. doi: 10.3390/jcm12051899
Baroroh, U., Biotek, M., Muscifa, Z. S., Destiarani, W., Rohmatullah, F. G., and Yusuf, M. (2023). Molecular interaction analysis and visualization of protein-ligand docking using biovia discovery studio visualizer. Indonesian J. Computational Biol. 2, 22–30. doi: 10.24198/ijcb.v2i1.46322
Bentabet, N., Boucherit-Otmani, Z., and Boucherit, K. (2014). Composition chimique et activité antioxydante d’extraits organiques des racines de Fredolia aretioides de la région de Béchar en Algérie. Phytothérapie 12, 364–371. doi: 10.1007/s10298-014-0834-x
Bouhaddouda, N. (2016). Activités antioxydante et antimicrobienne de deux plantes du sol local: Origanum vulgare et Mentha pulegium. Diplôme de Doctorat, Univ Badji Mokhtar, Annaba.
Bouhaddouda, N., Aouadi, S., and Labiod, R. (2016). Evaluation of chemical composition and biological activities of essential oil and methanolic extract of Origanum vulgare L. ssp. glandulosum (DESF.) Ietswaart from Algeria. Int. J. Pharmacogn. Phytochem. Res 8, 104–112.
Boukhebti, H., Chaker, A. N., Belhadj, H., Sahli, F., Ramdhani, M., Laouer, H., et al. (2011). Chemical composition and antibacterial activity of Mentha pulegium L. and Mentha spicata L. essential oils. Pharm. Lett. 3, 267–275.
Bouyahya, A., Bakri, Y., Khay, E. O., Edaoudi, F., Talbaoui, A., Et-Touys, A., et al. (2017). Antibacterial, antioxidant and antitumor properties of Moroccan medicinal plants: a review. Asian Pac J Trop Dis 7, 57–64. doi: 10.12980/apjtd.7.2017D6-294
Božović, M., Pirolli, A., and Ragno, R. (2015). Mentha suaveolens Ehrh. (Lamiaceae) essential oil and its Main constituent Piperitenone oxide: biological activities and chemistry. Molecules 20, 8605–8633. doi: 10.3390/molecules20058605
Bunse, M., Daniels, R., Gründemann, C., Heilmann, J., Kammerer, D. R., Keusgen, M., et al. (2022). Essential oils as multicomponent mixtures and their potential for human health and well-being. Front. Pharmacol. 13:956541. doi: 10.3389/fphar.2022.956541
Carlier-Loy, P. (2015). Mentha spicata: description et utilisations en thérapeutique et en agriculture comme antigerminatif sur la pomme de terre : Universite De Picardie Jules Verne. Available at: https://dumas.ccsd.cnrs.fr/dumas-01379512v1
Chelaghema, A. (2021). Utilisation d’extraits végétaux pour la maîtrise du risque mycotoxique dans les systèmes agro-alimentaires. Université Montpellier. Available at: https://theses.hal.science/tel-03719985 (Accessed April 23, 2023).
Derwich, E., Benziane, Z., and Taouil, R. (2010). GC/MS Analysis of Volatile Compounds of the Essential Oil of the Leaves of Mentha pulegium growing in Morocco, 55.
Deyno, S., Abebe, A., Tola, M. A., Hymete, A., Bazira, J., Makonnen, E., et al. (2020). Acute and sub-acute toxicity of Echinops kebericho decoction in rats. BMC Complementary Med. Therapies 20:2. doi: 10.1186/s12906-019-2794-z
Di Stasi, L. C., Oliveira, G. P., Carvalhaes, M. A., Queiroz-Junior, M., Tien, O. S., Kakinami, S. H., et al. (2002). Medicinal plants popularly used in the Brazilian tropical Atlantic Forest. Fitoterapia 73, 69–91. doi: 10.1016/S0367-326X(01)00362-8
Eddine, L. S., Djamila, B., and Redha, O. M. (2016). Solvent pH extraction effect on phytochemical composition and antioxidant properties of Algerian Matricaria Pubescens. J. Pharm. Res. 10, 106–112.
Eddine, L. S., Segni, L., Noureddine, G., Redha, O. M., and Sonia, M. (2013). Antioxidant, anti-inflammatory and diabetes related enzyme inhibition properties of leaves extract from selected varieties of Phoenyx dactylifera L. Innovare J Life Sci 1, 14–18.
Eddine, L. S., Segni, L., and Ridha, O. M. (2015). In vitro assays of the antibacterial and antioxidant properties of extracts from Asphodelus tenuifolius Cav and its main constituents: a comparative study. Int J Pharm Clin Res 7, 119–125.
El Omari, N. (2021). Étude phytochimique et toxicologique et evaluation de l’activite antioxydante, antidiabetique et antibacterienne des racines d’Aristolochia longa l. Available at: http://ao.um5.ac.ma/xmlui/handle/123456789/19489 (Accessed January 21, 2023).
Elkordy, A. A., Haj-Ahmad, R. R., Awaad, A. S., and Zaki, R. M. (2021). An overview on natural product drug formulations from conventional medicines to nanomedicines: past, present and future. J. Drug Delivery Sci. Technol. 63:102459. doi: 10.1016/j.jddst.2021.102459
Er-rajy, M., El Fadili, M., Mujwar, S., Zarougui, S., and Elhallaoui, M. (2023a). Design of novel anti-cancer drugs targeting TRKs inhibitors based 3D QSAR, molecular docking and molecular dynamics simulation. J. Biomol. Struct. Dyn. 41, 11657–11670. doi: 10.1080/07391102.2023.2170471
Er-rajy, M., Fadili, M. E., Mujwar, S., Lenda, F. Z., Zarougui, S., and Elhallaoui, M. (2023b). QSAR, molecular docking, and molecular dynamics simulation–based design of novel anti-cancer drugs targeting thioredoxin reductase enzyme. Struct. Chem. 34, 1527–1543. doi: 10.1007/s11224-022-02111-x
Falya, Y., Sumiwi, S. A., and Levita, J. (2020). Mini review: toxicity study of plant extracts. IOSR-JPBS 15, 25–32.
Fennane, M., and Tattou, M. I. (2008). Statistiques et commentaires sur l’inventaire actuel de la flore vasculaire du Maroc. Flora 1986. Available at: http://www.israbat.ac.ma/wp-content/uploads/2015/01/01-Fennane_BIS_SV34_1_1.pdf
Ghareeb, M. A., Tammam, M. A., El-Demerdash, A., and Atanasov, A. G. (2020). Insights about clinically approved and Preclinically investigated marine natural products. Current Res. Biotechnol. 2, 88–102. doi: 10.1016/j.crbiot.2020.09.001
Gill, R., Mohammed, F., Badyal, R., Coates, L., Erskine, P., Thompson, D., et al. (2005). High-resolution structure of myo-inositol monophosphatase, the putative target of lithium therapy. Acta Crystallogr. D Biol. Crystallogr. 61, 545–555. doi: 10.1107/S0907444905004038
Haida, S., Kribii, A., Ait Daoud, N., Belakhmima, R., and Abderahim, K. (2022). Antioxidant activity of Haloxylon scoparium alkaloid extracts from Figuig region (southeastern of Morocco). Brazilian J. Pharm. Sci. 58. doi: 10.1590/s2175-97902022e19494
Hajlaoui, H., Trabelsi, N., Noumi, E., Snoussi, M., Fallah, H., Ksouri, R., et al. (2009). Biological activities of the essential oils and methanol extract of tow cultivated mint species (Mentha longifolia and Mentha pulegium) used in the Tunisian folkloric medicine. World J. Microbiol. Biotechnol. 25, 2227–2238. doi: 10.1007/s11274-009-0130-3
Hassani, F. Z. E. (2020). Characterization, activities, and ethnobotanical uses of Mentha species in Morocco. Heliyon 6:e05480. doi: 10.1016/j.heliyon.2020.e05480
Hseini, S., and Kahouadji, A. (2007). Étude ethnobotanique de la flore médicinale dans la région de Rabat (Maroc occidental). Lazaroa 28, 79–93.
Jaber, H., Oubihi, A., Ouryemchi, I., Boulamtat, R., Oubayoucef, A., Bourkhiss, B., et al. (2021). Chemical composition and antibacterial activities of eight plant essential oils from Morocco against Escherichia coli strains isolated from different Turkey organs. Biochem. Res. Int. 2021:e6685800, 1–9. doi: 10.1155/2021/6685800
Jafari-Sales, A., Hossein-Nezhad, P., and Bolouri, P. (2019). Identification of chemical composition of essential oil and evaluation of antimicrobial effects of ethanolic extract of Mentha pulegium on Staphylococcus aureus and Escherichia coli. Health Biotechnol. Biopharma 3, 29–38.
Jamshidi-Kia, F., Lorigooini, Z., and Amini-Khoei, H. (2018). Medicinal plants: past history and future perspective. J herbmed pharmacol 7, 1–7. doi: 10.15171/jhp.2018.01
Kloter, E., Albanese, F., Schweighoffer, R., and Wolf, U. (2023). Phytotherapy in paediatric skin disorders-a systematic literature review. Complement. Ther. Med. 74:102942. doi: 10.1016/j.ctim.2023.102942
Lamiri, A., Lhaloui, S., Benjilali, B., and Berrada, M. (2001). Insecticidal effects of essential oils against hessian fly, Mayetiola destructor (say). Field Crop Res. 71, 9–15. doi: 10.1016/S0378-4290(01)00139-3
Laouini, S. E., and Ouahrani, M. R. (2017). Phytochemical screening, in vitro antioxidant and antibacterial activity of Rumex vesicarius L. extract. Scientific Study & Res. Chem. Chem. Eng. Biotechnol. Food Industry 18, 367–376.
Laouini, S. E., Segni, L., Ouahrani, M. R., Gherraf, N., and Mokni, S. (2012). Phytochemical analysis, antioxidant and antimicrobial activities of leaves extract of date palm grown in Algeria. J. Fundamental Applied Sci. 4, 142–154. doi: 10.4314/jfas.v4i2.4
Liaqat, I., Riaz, N., Saleem, Q.-A., Tahir, H. M., Arshad, M., and Arshad, N. (2018). Toxicological evaluation of essential oils from some plants of Rutaceae Family. Evid. Based Complement. Alternat. Med. 2018:4394687. doi: 10.1155/2018/4394687
Mahendran, G., Verma, S. K., and Rahman, L.-U. (2021). The traditional uses, phytochemistry and pharmacology of spearmint (Mentha spicata L.): A review. J. Ethnopharmacol. 278:114266. doi: 10.1016/j.jep.2021.114266
Maniben, B. P. N., Nguele, M. O. O., Ndongo, M. N., Nko’O, J. H. M., Ombga, Y. L. T., Ngameni, B., et al. (2023). P14 Criblage phytochimique, toxicité aiguë et évaluation de l’activité antidiabétique de l’écorce du tronc de Cylicodiscus gabunensis (Fabaceae). J. Afr. Technol. Pharm. Biopharmacie JATPB 2. doi: 10.57220/jatpb.v2i3.144
Mohammadi, F., Rahimi, K., Ahmadi, A., Hooshmandi, Z., Amini, S., and Mohammadi, A. (2024). Anti-inflammatory effects of Mentha pulegium L. extract on human peripheral blood mononuclear cells are mediated by TLR-4 and NF-κB suppression. Heliyon 10:e24040. doi: 10.1016/j.heliyon.2024.e24040
Morris, G. M., Huey, R., and Olson, A. J. (2008). Using AutoDock for ligand-receptor docking. Curr. Protoc. Bioinforma. Chapter 8, Unit 8.14. doi: 10.1002/0471250953.bi0814s24
Mostafa, E. (2018). “Ethnobotany/Ethnopharmacology, and bioprospecting” in Natural products and drug discovery: An Integrated Approach, 105–118. doi: 10.1016/B978-0-08-102081-4.00005-8
Mzioud, K., Habsaoui, A., Imtara, H., Haida, S., Rached, S., Msairi, S., et al. (2023). Physicochemical characterization, antioxidant and antifungal activities of essential oils of Urginea maritima and Allium sativum. Open Chemistry 21:20230149. doi: 10.1515/chem-2023-0149
Obanor, F., Neate, S., Simpfendorfer, S., Sabburg, R., Wilson, P., and Chakraborty, S. (2013). Fusarium graminearum and fusarium pseudograminearum caused the 2010 head blight epidemics in Australia. Plant Pathol. 62, 79–91. doi: 10.1111/j.1365-3059.2012.02615.x
OCDE (2002). Guidelines for the testing of chemicals, revised draft guidelines 423; acute oral toxicity-acute toxic class method, revised document. Available at: https://www.google.com/search?q=OECD%2FOCDE+(2002).+Guidelines+for+the+testing+of+chemicals%2C+revised+draft+guidelines+423%3B+acute+oral+toxicity-acute+toxic+class+method%2C+revised+document.&oq=OECD%2FOCDE+(2002).+Guidelines+for+the+testing+of+chemicals%2C+revised+draft+guidelines+423%3B+acute+oral+toxicity-acute+toxic+class+method%2C+revised+document.&aqs=chrome..69i57j69i58.1120j0j15&sourceid=chrome&ie=UTF-8 (Accessed March 11, 2023).
Omari, E. N., Sayah, K., Fettach, S., el, O., Bouyahya, A., Faouzi, M. E. A., et al. (2019). Evaluation of in vitro antioxidant and antidiabetic activities of Aristolochia longa extracts. Evid. Based Complement. Alternat. Med. 2019:e7384735, 1–9. doi: 10.1155/2019/7384735
Oubihi, A., Ballaoui, F. Z., Imtara, H., Jaber, H., Ettouil, A., Haida, S., et al. (2023). Phytochemical compounds, acute toxicity, anti-inflammatory and antioxidant activities of Thymus leptobotrys Murb essential oil. Molecules 28:1355. doi: 10.3390/molecules28031355
Oubihi, A., Ouryemchi, I., Nounah, I., Tarfaoui, K., Harhar, H., Ouhssine, M., et al. (2020). Chemical composition, antibacterial and antifungal activities of Thymus leptobotrys Murb essential oil. Adv. Tradit. Med. 20, 673–679. doi: 10.1007/s13596-020-00488-w
Papp-Wallace, K. M., Kumar, V., Zeiser, E. T., Becka, S. A., and van den Akker, F. (2019). Structural analysis of the OXA-48 carbapenemase bound to a “poor” carbapenem substrate, doripenem. Antibiotics 8:145. doi: 10.3390/antibiotics8030145
Ponce, A. G., Fritz, R., Del Valle, C., and Roura, S. I. (2003). Antimicrobial activity of essential oils on the native microflora of organic Swiss chard. LWT-Food Sci. Technol. 36, 679–684. doi: 10.1016/S0023-6438(03)00088-4
Rached, S., Habsaoui, A., Mzioud, K., Lachhab, R., Haida, S., Errahmany, N., et al. (2023). Valorization of the green corrosion inhibitor Marrubium vulgare L.: electrochemical, thermodynamic, theoretical & surface studies. Chemical Data Collections 48:101099. doi: 10.1016/j.cdc.2023.101099
Rached, S., Imatara, H., Habsaoui, A., Mzioud, K., Haida, S., Saleh, A., et al. (2022). Characterization, chemical compounds and biological activities of Marrubium vulgare L. essential oil. PRO 10:2110. doi: 10.3390/pr10102110
Rached, S., Mzioud, K., Habsaoui, A., Galai, M., Dahmani, K., Ouakki, M., et al. (2024). Inhibition of copper corrosion in sulfuric acid by Mentha Pulegium L. Port. Electrochim. Acta 42, 137–153. doi: 10.4152/pea.2024420205
Reis-Vasco, E. M. C., Coelho, J. A. P., and Palavra, A. M. F. (1999). Comparison of pennyroyal oils obtained by supercritical CO2 extraction and hydrodistillation. Flavour Fragr. J. 14, 156–160. doi: 10.1002/(SICI)1099-1026(199905/06)14:3<156::AID-FFJ793>3.0.CO;2-J
Rhattas, M., Douira, A., and Zidane, L. (2016). Étude ethnobotanique des plantes médicinales dans le Parc National de Talassemtane (Rif occidental du Maroc). J. Applied Biosc. 97, 9187–9211. doi: 10.4314/jab.v97i1.5
Said, S., Noureddine, G., Eddine, L. S., Abdelmadjid, G., Djamel, B., and Tliba, A. (2018). Phenolic content, HPLC analysis and antioxidant activity extract from Tamarix gallica and Tamarix articulata growing in southeast of Algeria. Res. J. Pharmacy Technol 11, 3826–3832. doi: 10.5958/0974-360X.2018.00701.1
Samir, M., Imen, S., Mouloud, G., Laid, B., and Hacène, B. (2017). Etude phytochimique et activités biologiques d’une plante médicinale de la région de m’sila Mentha pulegium L. Actes du 5ème Meeting International sur l’Aridoculture et les Cultures Oasiennes: Biotechnologie végétale en zones arides et oasiennes. Available at: https://num.univ-msila.dz/DWE/public/attachements/2020/02/03/etude-phytochimique-2017pdf-7odtqald1580716509.pdf
Souza, L. P. D., Zuim, V., Stinguel, P., Pinheiro, P. F., and Zago, H. B. (2022). Toxicity of essential oil of Mentha piperita (Lamiaceae) and its Monoterpenoid menthol against Tetranychus urticae Kogan 1836 (Acari: Tetranychidae). An. Acad. Bras. Ciênc. 94:e20200427. doi: 10.1590/0001-3765202220200427
Uwineza, M. S., Yousfi, B. E., and Lamiri, A. (2018). Activités antifongiques in vitro des huiles essentielles de Mentha pulegium, Eugenia aromatica et Cedrus atlantica sur Fusarium culmorum et Bipolaris sorokiniana. Rev. Marocaine Prot. Plantes. Available at: https://revues.imist.ma/index.php/RMPP/article/view/14145.
Wu, Z., Jin, C., Chen, Y., Yang, S., Yang, X., Zhang, D., et al. (2023). Mentha spp. essential oils: A potential toxic fumigant with inhibition of acetylcholinesterase activity on Reticulitermes dabieshanensis. Plan. Theory 12:4034. doi: 10.3390/plants12234034
Zekri, N., El Hajjaji, S., Zair, T., El Hilali, F., Tabyaoui, M., El Azzouzi, M., et al. (2017). Étude Phytochimique et Activités Biologiques des Huiles Essentielles et des Extraits des M. pulegium (L.), M. suaveolens (Ehrh.), et M. spicata (L.) du Moyen-Atlas Marocain. Rabat: Université Mohammed V.
Keywords: Mentha pulegium L., toxicity, docking, essential oil, GC–MS, biological activities
Citation: Rached S, Habsaoui A, Mzioud K, Er-rajy M, Abujaber F, Imtara H, Oubihi A, Haida S, El-guourrami O, Noman OM, Ourras S, Tarayrah M and Touhami ME (2025) Chemical profiling, safety assessment, bioactive properties, and molecular interactions of the essential oil derived from Mentha pulegium L. Front. Sustain. Food Syst. 9:1511848. doi: 10.3389/fsufs.2025.1511848
Received: 15 October 2024; Accepted: 10 February 2025;
Published: 06 March 2025.
Edited by:
Anka Trajkovska Petkoska, University St. Clement of Ohrid, North MacedoniaReviewed by:
Laouini Salah Eddine, El-Oued University, AlgeriaCopyright © 2025 Rached, Habsaoui, Mzioud, Er-rajy, Abujaber, Imtara, Oubihi, Haida, El-guourrami, Noman, Ourras, Tarayrah and Touhami. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hamada Imtara, aGFtYWRhLmltdGFyYUBhYXVwLmVkdQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.