
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Sustain. Food Syst. , 03 April 2025
Sec. Agro-Food Safety
Volume 9 - 2025 | https://doi.org/10.3389/fsufs.2025.1502841
 Petrina McKenzie-Reynolds1,2
Petrina McKenzie-Reynolds1,2 Patricia Millner3
Patricia Millner3 Fawzy Hashem2*
Fawzy Hashem2* Lurline Marsh2
Lurline Marsh2 Brett Smith2
Brett Smith2 Annette Kenney2
Annette Kenney2 Arthur Allen2
Arthur Allen2 Salina Parveen2
Salina Parveen2 Amy S. Collick4
Amy S. Collick4Introduction: Poultry litter (PL) compost and heat-treated PL-pellets (PP) are common organic fertilizer/soil amendments in specialty crop production and soil health management. However, such products may enhance survival and persistence of foodborne pathogens and their transfer to fresh produce.
Methods: A replicated (n = 4) 2-year, randomized complete block field-plot inoculated-challenge study, with PL treatments (composted APL and PP) and cover crop/soil placement, was conducted to assess soil persistence/transfer of generic gE.colirif-R to cantaloupe, cucumber, spinach, and table radish, in cover-cropped USDA-National Organic Program (NOP) 8-years managed as certified plots. Amended soils were analyzed monthly for E. coli up through 90-120-day NOP required wait periods between PL application and produce harvest. Cantaloupe, cucumber, spinach, and table radish, grown compliant with NOP standards, were assayed at harvest for pathogens (Salmonella, Listeria monocytogenes, Staphylococcus aureus) and gE. coli.
Results and discussion: All amended, inoculated soils were positive for the inoculated E. coli (6-log MPN/g) immediately after inoculation. At 90 dpi, all plot soils were positive for the inoculated gEcrif-R; by 120 dpi, gEcrif-R concentrations in soils declined significantly (p < 0.05) both in 2018 and 2019. No other pathogens (Salmonella, Listeria monocytogenes, Staphylococcus aureus) were detected at 0 or 90 dpi in plot soils. Generic Ecrif-R was detected on four of 16 cantaloupes, four of 16 cucumbers from APL plots; four of 20 radish bulbs from till incorporated and four from subsurface-applied APL tested positive for gEcrif-R at 90 dpi. By 120 dpi, only two spinach plants (n = 20) were positive from subsurface APL plots; no other pathogens or gEcrif-R were recovered for any crops in 2018–2019. Significant, consistent declines and decreasing survival throughout the growing seasons in populations of inoculated E. coli in soil, resulted in very low detection on any unwashed produce by 90 and 100 dpi in accord with NOP manure amendment-to-harvest wait times. Exposure to a combination of environmental conditions, including soil moisture, splash transfer of soil to produce, and temperatures (soil and air), impact E. coli populations in tilled and subsurface PL product-amended soils and on produce managed in compliance with organic production requirements.
Foodborne illnesses associated with fresh produce consumption have increased over recent years (Turner et al., 2019). Fresh produce (spinach, lettuce, cucumbers, radish, and other leafy greens) is frequently grown organically and consumed raw. Consumption of fresh produce is increasingly sought by consumers because it is recognized as a significant source of micronutrients, including vitamins, polyphenolics, carotenoids, minerals, and glucosinolates, along with macronutrients such as carbohydrates and fibers (Barrett et al., 2010).
Fresh produce labeled as ‘Organic’ indicates to consumers that the conditions of its production and processing comply with all USDA requirements stipulated by the National Organic Program (NOP). These include use of only organically approved inputs, e.g., seeds (non-GMO), crop, pest and weed control management practices with use of only approved synthetics. This includes an emphasis on ecological production fostering biodiversity, biological cycles and soil biological activity responsive to site-specific conditions by integrating cultural, biological and mechanical practices that foster cycling of resources, ecological balance, and conservation of biological diversity. Organic farming is a combination of natural techniques and substances to enhance productivity, biodiversity and sustainability in a healthy manner. This includes the use of cover crops (USDA-AMS NOP, 2011). Organic farming often includes the use of animal manure and biological soil amendments of animal origin (BSAAO) to provide plant nutrients, soil fertility, and organic matter, thereby contributing to grower sustainability (Ramos et al., 2019). Depending on the type of treatment animal manure is subjected to prior to land application, it may harbor pathogens that can transfer to fresh produce grown in manure-amended soils, this can lead to foodborne illnesses from surviving pathogens (Sharma et al., 2016). Consumption of contaminated fresh produce and consequent foodborne illness outbreaks from pathogens continues to pose a significant problem for the fresh produce industry and to human health, from Salmonella spp., Listeria monocytogenes, pathogenic Escherichia coli (E. coli), and E. coli 0157:H7 (Iwu and Okoh, 2019). This acknowledgment has led to concerns about risks associated with the production and consumption of fresh produce harvested from manured soils and possible steps to ensure safer management practices to minimize adverse environmental and human health impacts. The U.S. Food and Drug Administration (FDA) addressed concerns about the use of BSAAOs in fields of fresh produce through the produce safety rule of the Food Safety Modernization Act (FSMA) (FDA, 2015). The FDA recommends that growers who apply raw manure follow the guidance provided by the United States Department of Agriculture National Organic Program (USDA-NOP) that stipulates, organic biological soil amendments, such as poultry litter, need to be incorporated into soils at least 90 to 120 days before produce harvest (USDA-AMS NOP, 2011). The wait time between soil amendment and produce harvest depends on whether the specialty crop is directly or indirectly in contact with soil. These regulations also restrict surface application of PL and other manures and require tillage or subsurface incorporation to reduce nutrient runoff into waterways and to enhance soil health (Maryland Department of Agriculture, 2012). Nonetheless, survival, persistence, and transfer of manure pathogens to fresh produce are major concerns. While several published studies have reported on microbial food safety aspects associated with organically managed fresh produce systems (Harvey et al., 2016; Maffei et al., 2016; Chandler-Khayd et al., 2023; Neher et al., 2019; Limoges et al., 2022), in this study we evaluated the effect of placement of two different PL products (6-month aged PL and heated pelletized PL) on four specialty crops (cantaloupe, cucumber, table radish, and spinach) grown in the USA Delmarva region. This region is recognized as a leading supplier of poultry products and generator of poultry litter, which is used as a biological soil amendment and fertilizer for a wide variety of crops and farms, including those in the NOP-certified program. Cantaloupe, cucumber, table radish, and spinach are commonly produced in the Delmarva region, and the fruits, edible leaves, or bulbs can directly contact soils amended with various manure products.
The presence and persistence of pathogenic E. coli have been a major concern in manure-amended soils (Sharma et al., 2016). Non-pathogenic strains of E. coli have served as surrogates for gram-negative enteric pathogens that employ sophisticated strategies to colonize and infect mammalian hosts (Eblen et al., 2005). Heat-treated poultry litter pellets, a commonly incorporated fertilizer for specialty crops, have been shown to support the increased survival of Salmonella enterica Newport, not only in soil but also on spinach (Shah et al., 2019). The potential for microbial contamination of agricultural produce can occur pre- and post-harvest; a range of measures have been investigated to mitigate these situations, including effects of cover crops (Reed-Jones et al., 2016) and biological soil amendments of animal origin (Neher et al., 2019; Litt et al., 2021; Limoges et al., 2022). A replicated pot-scale simulation of the effects of soil (chase silt loam) incorporated cover crops (buckwheat, Sunn hemp, and mustard greens) on inoculated E. coli, showed significant declines from initial 5 log CFU/g to 3.96 log CFU/g between 10 and 30 (Zhao et al., 2023). Through collaborative efforts among various organizations, the One Health approach has emerged to address environmental and human health factors involved (Ohno and Hettiarachchi, 2018).
In this study, six open-field plot experiments were conducted during two (summer and fall) growing seasons (2018 and 2019) at the University of Maryland Eastern Shore (UMES) Agricultural Experiment Station, within the 2.02 ha NOP-certified organic field in Princess Anne, MD. A randomized complete block design was used on the two sites within the NOP-certified areas for both growing seasons. Each treatment used in this study consisted of four replications with three poultry products: two crops in summer, two crops in fall, and two cover crops. Cucumber and cantaloupe plots were amended with either aged poultry litter (APL) or heat-treated poultry pellets (PP) for crops planted in May as summer crops and previously cropped with hairy vetch (11 kg/ha) or forage radish (5 kg/ha) in October 2017 and 2018. Poultry litter products were applied to plots at 2300 kg/ha, consistent with nutrient management practices and regulations for cantaloupe and cucumber. Aged poultry litter (APL), poultry litter compost (PLC), or heat-treated poultry pellets (PP) were applied to the spinach and radish plots. Spinach and radishes were planted in August (2018 and 2019) as cool-season crops.
The PL was obtained from the UMES poultry open-sided litter storage shed, and PP (NPK, 4–3-4) was obtained from the distributor (Mighty Grow, Inc. P.O Box 526, 870 Edward Loper Rd. Fruitdale, Alabama, USA) and collected for weight measurement and field distribution. The aged poultry litter, APL, was aged in a stack for 6 months, and the composted poultry litter, PLC, was aged approximately 10 years after thermophilic composting was conducted by a validated process. These poultry litter products were well decomposed and did not contain elevated levels of ammonia that would ‘burn’ crops after planting. Treatments were analyzed for dry matter, N, and P content (Moebius-Clune et al., 2016). The Mighty Grow pellets (PP) were applied sub-surface (ss), in 5 cm deep trench bands (2/plot), flanking seed rows, and covered with plot soil. The PL was surface applied likewise in bands flanking seeded rows and then manually tilled into the soil before inoculation with a sterile PL extract with a 3-strain gEcrif-R cocktail. Cantaloupe and cucumber plots were left bare for 10 days, thereafter black plastic mulch and drip lines were installed prior to planting.
Seedlings of hybrid cantaloupe (Cucumis melo cv. Divergent F1 OG) and slicing cucumbers (Cucumis sativus cv. Marketmore 76 OG) seeds (Johnny’s Selected Seeds, Winslow, Maine) were produced in the UMES greenhouse. Seedlings were transplanted to plots on the NOP-certified organic research/teaching farm in June 2018 and 2019. Plants were irrigated/checked daily and replaced as needed.
Of 16 cantaloupe plots (each 26.79 m2), eight received 8.1 kg of PL, and eight others received 7.5 kg each PP. Amendments were applied either as flanking bands or subsurface trenches adjacent to each seeded row. Prior to planting, plots had been cover-cropped either with Hairy Vetch (HV) or Forage Radish (FR) as part of the soil quality/health maintenance program. Cover crops also preceded cucumber crops. Of 16 cucumber plots (each 7.44 m2), half were amended with PL product by flanking bands and the other half were amended by subsurface trenching with either 2.8 kg/plot of APL, or 2.7 kg/ plot of PP.
Spinach and radish were grown in subsections of 20 plots, 2 m × 1 m/plot, with 1 m x 1 m subplots, in 2018. Four full plots received 0.94 kg of TPL (tilled in by hoe). Four other plots also received 1 kg of PLss (subsurfaced)/plot, four plots received 1.0 kg PP (subsurfaced, ss)/plot, four plots received 1.2 kg of PLC (subsurfaced, ss)/plot and the remaining four plots received 1.24 kg PLC/plot. Poultry product amounts for different treatments were calculated based on the recommended nitrogen requirement for each crop type, the amount supplied by the type of treatment to be applied, and plot sizes.
Three rifampicin-resistant (Rif R) strains of non-pathogenic E. coli (TVS, 353 and 354 and 355) on tryptic soy agar with rifampicin cultured plates (TSAR) (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) were received from BARC (from Dr. Trevor Suslow, University of California, Davis). These E. coli strains were isolated from different sources: TVS 353 (E. coli W778) was isolated from irrigation water, TVS 354 (E. coli P149) was isolated from lettuce leaves, and TVS 355 (E. coli S19) was isolated from sandy loam soils (Tomás-Callejas et al., 2011). Two colonies of each E. coli strain were sub-cultured onto fresh MacConkey + rifampicin (MAC-R) agar plates (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) and incubated at 37°C. Cultured plates were retrieved after 24 h. Isolated colonies were then selected and transferred into 50 mL of half (½) strength tryptic soy broth (TSB + rifampicin)-(Becton, Dickinson, and Company, Sparks, Maryland/Gold Biotechnology, St. Louis, MO) and incubated at 37°C, 24 h. Retrieved turbid cultures (50 mL) were added to their own separate 750 mL sterile PLE (1PLE: 1dH2O). The solutions were replaced in the incubator for 48 h. at 37°C. Equivalent cell densities for low inoculum (6 log CFU/mL) by combining PLE cultures that were used in field experiments were selected. Bacteria cultures were enumerated by dilution plating of each strain in sterile half-strength TSB on MAC-R agar plates (2 plates /dilution) and incubated at 37°C for 24 h. Bacterial cell counts were performed to determine the dilution factor that was used in inoculation for plots.
Poultry litter extract (PLE) and inoculant preparation as well as application to plots, followed the procedure described for a three-strain cocktail of E. coli rifampicin-resistant (gEcrif-R) (Sharma et al., 2019). Calculated amounts of each E. coli at 6 log MPN/mL were added to sterile distilled water in a sterile carboy (20 L). This mixture was used to make up 18 L for cucumber and cantaloupe plot inoculant (sterile water and PLE was used as diluent). The contents were mixed thoroughly and poured into a backpack sprayer, which had been time calibrated to spray 1 L of the inoculum (Black and Decker Automatic Sprayer, battery operated 4 gals., 15.14 L).
A 2.54 cm diameter probe was used to collect five soil cores (composited) per plot per sampling event in an “X” pattern across each plot. Black plastic mulch was applied to cucumber and cantaloupe plots and slotted to provide access for soil sampling; access sites were closed after each soil core sampling event. Soil samples were collected from amended, inoculated areas on plots with gloved hands and a sterilized stainless-steel corer. Samples were stored in sealed, labeled sterile 680 g sterile sample bags and transported to the laboratory in an insulated chilled cooler. Soil samples collected on day zero before inoculation (D0B) served to evaluate soil health before amendment application and planting of crops. Samples were collected on D0B to determine the presence or absence of any E. coli or other bacterial pathogens in the soils/plots before inoculation or poultry litter product amendment. Soil samples were also collected immediately after manure and inoculum application (D0A). Subsequent soil sampling was conducted over 30, 60, 90, 120-, and 150-day post-amendment periods. The soil textural class of 2018 plots was sandy loam, and in 2019, plots were repeated on silty loam soil. This difference resulted from in-field rotations as required for organic cultivation by the National Organic Program (NOP).
Soil temperatures were measured periodically by placing a WatchDog weather station (Spectrum WatchDog 1,000 Series Micro Station, (Spectrum Technologies, Inc. East Plainfield, IL 60585) in the open fields for cantaloupe and cucumber. The weather station was pre-calibrated with unique external plug-in sensor ports. The instrument logs data from these sensors at a selected measurement interval (1–60 min). Soil moisture content reported in this study are based on wet and dry weight data from 25 g of each ‘as received’ sample were re-weighed after 24 h drying at 70°C. Dry weights were calculated by subtracting the recorded grams dry weight (gdw) from the original recorded grams wet weight. The moisture content of the soil was calculated as g/dw. This procedure was used to measure the soil’s gravimetric moisture percentage to normalize the bacterial content within the soil. Gravimetric soil moisture (percentage) was calculated as:
To detect and enumerate E. coli, 30 g of soil sample was weighed into a tared whirl-Pak sterile filter bag, and 120 mL buffered peptone water (BPW) (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) was added and the mixture was hand massaged to disperse the soil prior dilution. Enrichment and enumeration by MPN followed a previously described procedure (Sharma et al., 2019). Samples from immediately before and after inoculation were analyzed, along with those from 30, 60, 90, 120, and 150 dpi, for each crop type, plot, and treatment/amendment type. The mini-most probable number (mini-MPN) method was used for the quantitative determination of gEc populations. Homogenates for produce samples, 30 g of produce were placed into a sterile whirl-Pak filter bag and 120 mL of BPW was added and massaged for 1 min. One mL of sample solution was added to 96-well blocks containing double-strength tryptic soy broth with 80 μg/mL rifampicin (TSB/R). This solution was serial diluted into 5 columns of the well blocks containing single strength TSB/R (0.2 mL). Sample solutions were incubated for 24 h at 37°C, and then, 3 μL from each well/dilution of the 6 rows on the well block were dropped onto MAC/R plates; plates were incubated for 24 h at 37°C and then examined for growth. The MPN calculations were computed (US EPA, 2024), and the limit of detection was 0.1 MPN gEc/gdw soil.
Listeria: Pre-warmed Listeria enrichment broth (LEB 200 mL, was added to produce samples (1 whole cucumber, 100 g radish, 30 g spinach leaves, 30 g cantaloupe rind) and incubated for 24 h at 37°C; thereafter, 0.1 mL of the enriched sample solution was added to 5 mL of Fraser broth and incubated for 24 h at 37°C. One loop of Fraser broth that had turned black was streaked onto Brilliance Listeria agar (BLA) (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ). Isolated colonies with typical Listeria appearances (gray to black, surrounded by a black halo) were streaked onto trypticase soy agar (TSA) and incubated for 24 h at 37°C.
Salmonella: Cantaloupe rind samples were obtained from each of the cantaloupe plots by firmly swabbing the entire rind using a pre-moistened (25 mL BPW + 0.05% tween) sterile sponge. Excess BPW (Neongen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) was squeezed from the sponge. The sample sponge was returned to the sample bag with diluent. Other produce samples that were assayed included 1 whole cucumber, 100 g radish, and 30 g spinach leaves per plot. Each of these produce samples per plot was suspended in 200 mL of tryptic soy broth containing sodium pyruvate (TSB/Napyr) (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ). All these produce samples were hand-massaged for 1 min. Samples suspensions were incubated for 24 h at 42°C. One mL of TSB/Napyr sample solution was added to 9 mL of Tetrathionate Broth (TT broth) (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) and incubated at 42°C for 24 h. Samples (0.1 mL) of tryptic soy broth (TSB) were added to 10 mL of Rappaport Vassiliadis (RV) broth (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) and incubated at 42°C for 24 h. After incubation, a 10 μL loop of TT broth and Rappaport-Vassiliadis (RV) broth were streaked separately onto (XLT4) plates. Plates were incubated at 42°C for 24 h (plus an additional 24 h if no colonies appeared initially). Colonies appearing with Salmonella-like features (red with black color) were re-streaked onto XLT4 and incubated at 42°C for 24 h (2 colonies each). Colonies were re-streaked until they appeared red with black center or pure black, and one final colony/plate was streaked onto TSA. Plates were incubated for 24 h at 42°C and colonies were counted for the presence or absence of the targeted pathogen. Colonies were re-streaked onto agar plates until they appeared pure, one final colony per plate was streaked onto TSA and incubated 24 h at 42°C.
Staphylococcus aureus: Samples of cantaloupe rind, cucumber, radish, and spinach (20 g each) were added separately to two sterile whirl-Pak filter bags, and 180 mL of buffer peptone water (BPW) (for a 1:10 dilution) were added to produce samples. The sample solution was homogenized for 1 min at 260 rpm or massaged for 1 min. Sample solutions at 10−1 and 10−2 dilutions made up of BPW + Napyr were spiral plated onto Baird Parker agar (BPA) (Neogen, Lansing, MI; Fisher Scientific, Fair Lawn, NJ) and incubated for 24 h at 37°C. Negative cultures were re-incubated for a further 24 h at 37°C. Staphylococcus colonies appeared grey-black and shiny. Follow-up for confirmation tests were performed using a coagulase test.
Bacterial concentrations from assayed soil samples (1, 30, 60, 90, and 120 dpi) were expressed as Log10 from MPN values (US EPA, 2024). Data were exported, and Analysis of variance (ANOVA) was conducted using the Statistix 9 (Analytical Software, Tallahassee, FL) program to separate the observed data into different components and determine the relationship between the dependent and independent variables. Mean comparisons by controlling the error rate to the specified level were performed using the Tukey HSD All-Pairwise Comparisons Test. Means differences were considered significant at a level of (P < 0.05).
Evaluation of gEc rif-R populations on cantaloupe grown on cover-cropped soils amended with poultry manure under organic farming management practice at UMES (2018 and 2019).
Results of the gEc rif-R populations in cantaloupe field plots amended with PL and cover crops fluctuated between 1 and 150 days post inoculation (dpi) during 2018 and 2019 (Figures 1A,B). In 2018 cantaloupe season, the population of gEc rif-R from day 60 through to 150 dpi was very low even by 120 dpi (Figure 1A). The population of gEc rif-R remained very low, from 120 to 150 dpi in all treatments. All pair-wise comparisons detected a significantly higher level of decline for HVPL compared to FRPL, HVPP, and FRPP on 60 dpi sampling. There were no significant differences observed among all treatments for 1, 30, 90, 120, and 150 dpi (Figure 1A).
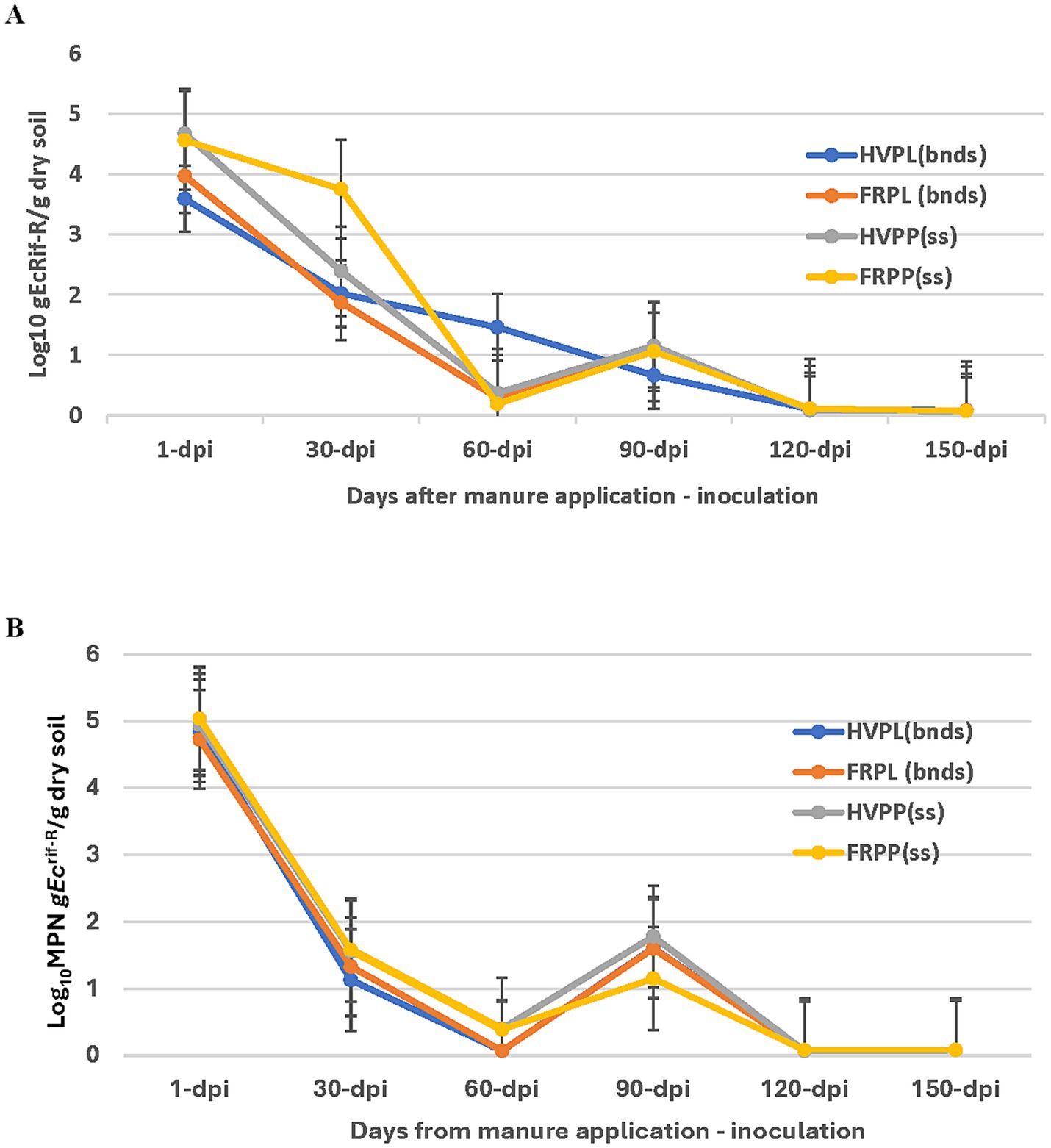
Figure 1. Effect of manure type and application method on survival and persistence of generic E. coli (gEcrif-R) in organic soil with cantaloupe in 2018 (A) and 2019 (B). HVPL, Hairy vetch + Poultry litter; FRPL, Forage radish + Poultry litter; HVPP, Hairy vetch + Poultry pellets; FRPP, Forage radish + Poultry pellets; bnds, Bands; ss, Subsurfacing. Error bars indicate standard error of the mean.
In the growing season of 2019, there were no significant differences in gEc rif-R population recoveries revealed by an all-pairwise comparison test (Figure 1B). Within treatments for sampling days (1–120) post-inoculation, there were differences observed in the gEc rif-R population declines in the cantaloupe plots. The population of gEc rif-R also showed some fluctuations over the sampling period and extremely low levels at 120–150 dpi (Figure 1B).
Data also showed a relationship between the decline in E. coli populations and the temperature decline over both research seasons (2018 and 2019). The months of June (Day 1) to August (Day 60) with temperature ranges between 25°C and 28°C, showed E. coli populations at their highest (Figure 2A). During this period, E. coli populations were declining, but E. coli recovery from hairy vetch and PL plots was significantly higher compared to all other treatments. The populations began to decline again in September although recovery increased in all treatments followed by a significant decline in succeeding months when soil and air temperatures declined seasonally. The E. coli populations followed the same trend (Figure 2A). In the 2019 experimental season, E. coli populations followed a different trend compared to 2018, declining from August (Day 120) to September (Day 150) even though the mean air temperature was low, and the soil temperature remained high (Figure 2B). The black plastic mulch on the plots covering the drip irrigation tubing may have helped retain soil moisture compared to un-mulched soil and likely increased soil temperature during periods of high solar radiation. Data show that the E. coli populations were very low in June (Day 60) when temperatures for both air and soil were high, but populations increased in July (Day 90) when the temperature was lower than in June. These data are consistent with that showing sampling dates for soil microbial analysis. During both years. Higher rainfall occurred from August to November of 2018 which may have influenced soil moisture. This had an adverse effect on E. coli populations. Soil moisture levels also may have contributed to the decline in E. coli populations. This is supported by the observed relationship between the soil moisture content and E. coli survival represented in Figure 3A. However, in 2019, E. coli recovery was greater at higher moisture content and decreased as moisture content decreased for all treatments, except for fluctuations in population in July (Figure 3B). These data show a contrasting pattern when compared to the 2018 experimental season. It can be concluded that no single factor determines the survival of E. coli populations in these soil amendment conditions.
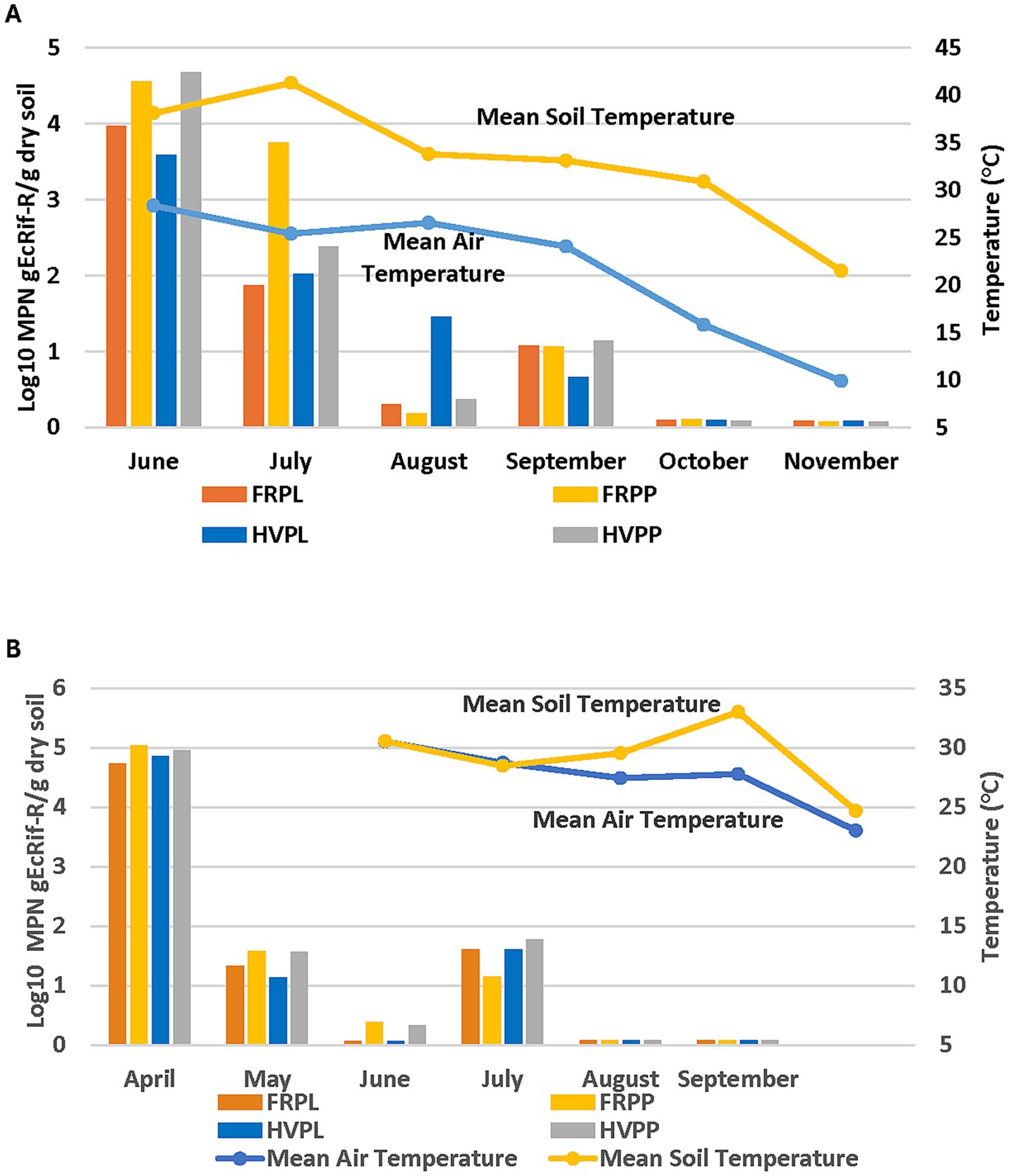
Figure 2. Temperature and temporal effects on gEcrif-Rsurvival for each sampling month in organic soil with cantaloupe and amended with PL with cover crops in 2018 (A) and 2019 (B) experimental seasons. HVPL, Hairy vetch + Poultry litter; FRPL, Forage radish + Poultry litter; HVPP, Hairy vetch + Poultry pellets; FRPP, Forage radish + Poultry pellets; bnds, Bands; ss, Subsurfacing. Error bars indicate standard error of the mean.
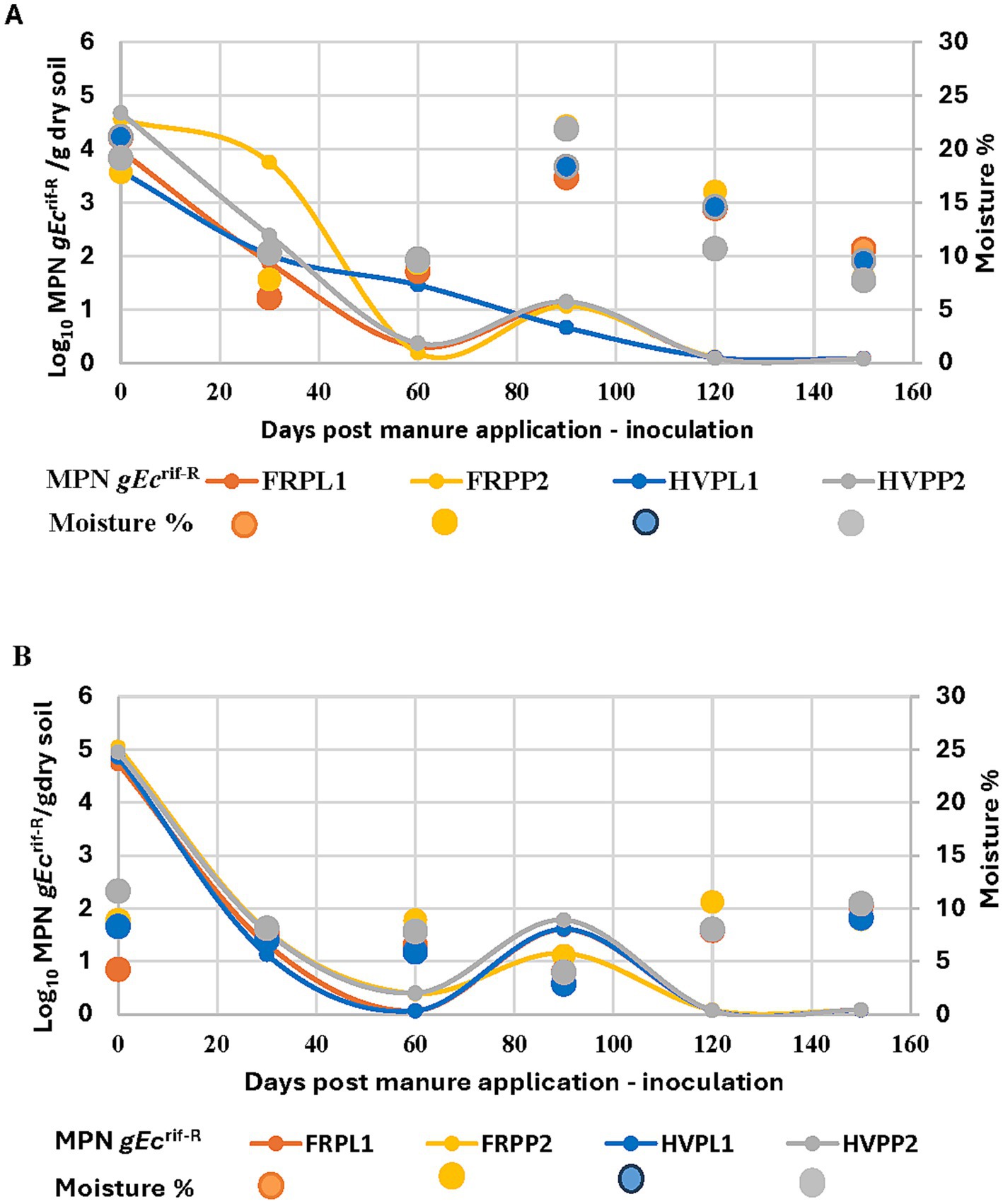
Figure 3. Volumetric water volume/soil moisture effect determined by wet and dry weight of soil assessment relative to gEcrif-R survival for each sampling month in organic soil with cantaloupe and amended with poultry manure with cover crops in the 2018 (A) and 2019 (B) experimental seasons. HVPL, Hairy vetch + Poultry litter; FRPL, Forage radish + Poultry litter; HVPP, Hairy vetch + Poultry pellets; FRPP, Forage radish + Poultry pellets; bnds, Bands; ss, Subsurfacing.
Evaluation of gEc rif-R population in cucumber cultivated soils under organic farming management practice at UMES (2018, 2019).
Fluctuation in the gEc rif-R populations across all treatments was observed in the 2018 cucumber plots over a 1-150-day period (Figure 4A). The population continued to decrease to very low levels up to day 150, with no significant differences among FRPL, HVPP, and FRPP (Figure 4A). However, all-pairwise comparisons showed significantly higher populations for HVPL compared to FRPL, HVPP, and FRPP (Figure 4A) on day-60 sampling. The same patterns were observed in the cantaloupe field, and this can be attributed to the fact that both fields were exposed to the same environmental conditions. Fluctuations in the E. coli population were also observed for all treatments in the 2019 growing season (Figure 4B). Extremely low levels of E. coli were observed at day 90 and declines in populations of different treatments continued up to day 150. No significant differences (p < 0.05) were observed for E. coli populations among treatments.
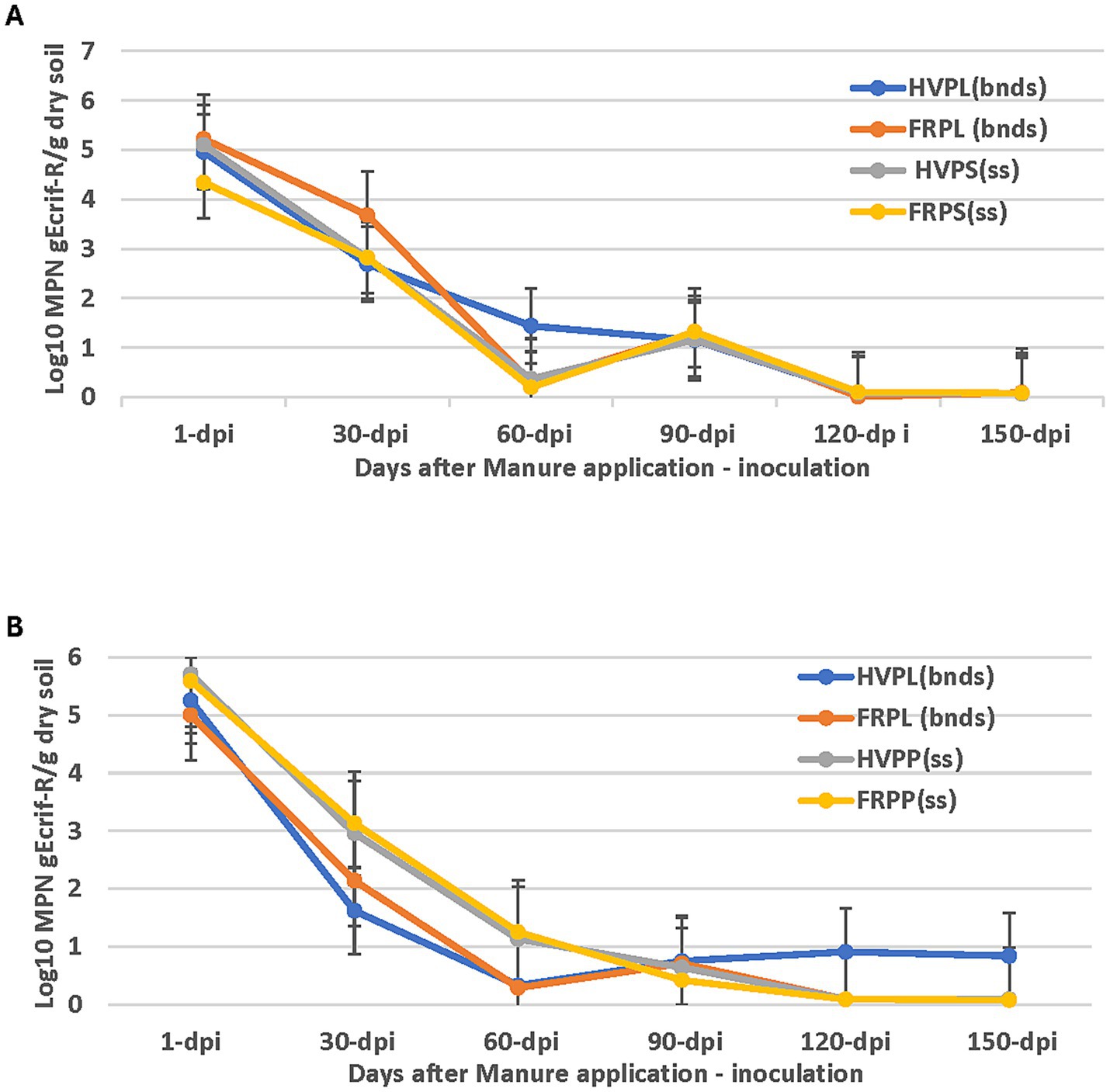
Figure 4. Effect of manure type and application method on survival and persistence of generic E. coli (gEcrif-R) in organic soil with cucumber in 2018 (A) and 2019 (B). HVPL, Hairy vetch + Poultry litter; FRPL, Forage radish + Poultry litter; HVPP, Hairy vetch + Poultry pellets; FRPP, Forage radish + Poultry pellets; bnds, Bandings; ss, Subsurfacing. Error bars indicate standard error of the mean.
Soil and air temperatures reflected the same patterns as those of the 2018 and 2019 data for the cantaloupe field. In 2018, E. coli populations declined as temperatures declined on sampling days 1–90 (Figures 5A,B). The 2019 temperature data for cucumber plots were like those for cantaloupe plots. Populations of E. coli declined throughout the sampling period (days 1–50) and were similar for all treatment types, except for August of 2018 which showed a higher population survival in comparison to other treatments. A definitive explanation for this result is not possible since temperature wasn’t a controlled factor in this cucumber study, nor in the cantaloupe field study. Both fields were exposed to the same environmental conditions and were affected by the same amount of rainfall contributing to the mean air and soil temperature in 2018 in contrast to 2019. More rain fell in the July–August period which coincided with lower air and soil temperature and with microbial survival. The soil moisture content also showed a similar relationship with E. coli recovery as that of temporal effects for the 2018 experimental season (Figure 6A). The opposite was observed during the 2019 experimental season, where E. coli survival decreased when soil moisture decreased (Figure 6B). These data show that environmental factors differentially impact population survival.
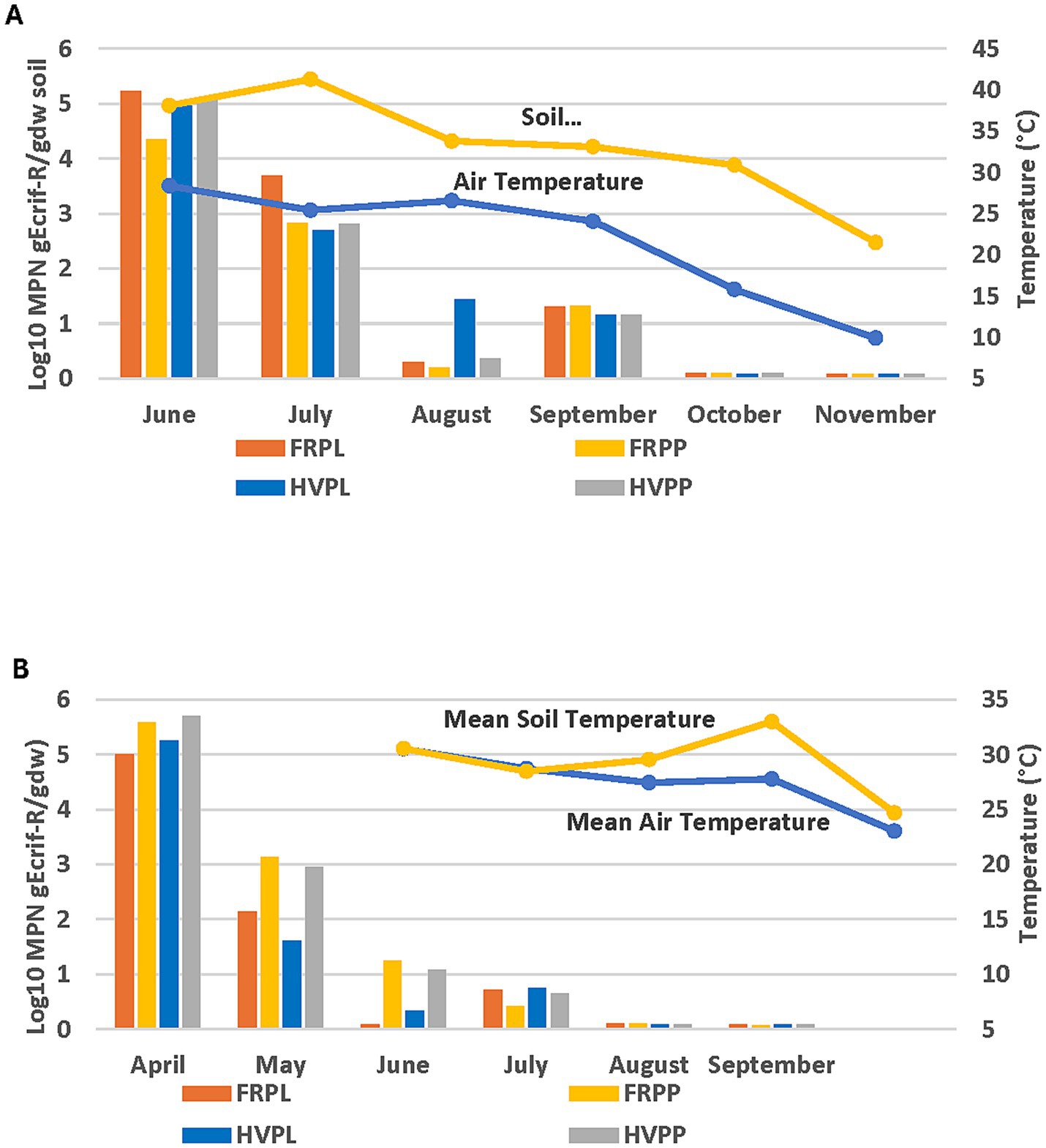
Figure 5. Temperature and temporal effects relative to gEcrif-R survival for each sampling month in organic soil with cucumber and amended with poultry manure + cover crops in 2018 (A) and 2019 (B) experimental seasons. HVPL, Hairy vetch + Poultry litter; FRPL, Forage radish + Poultry litter; HVPP, Hairy vetch + Poultry pellets; FRPP, Forage radish + Poultry pellets; bnds, Bandings; ss, Subsurfacing.
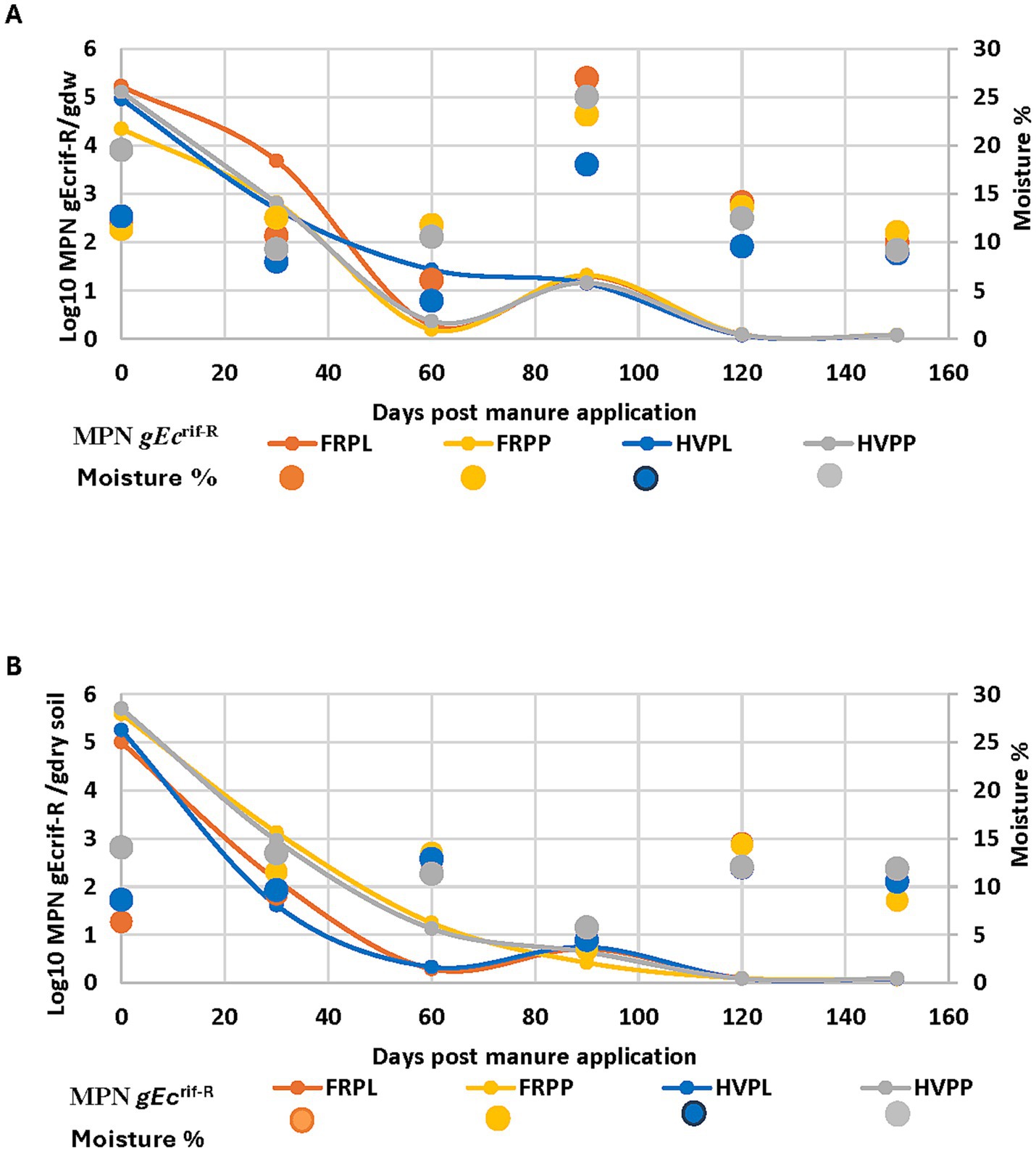
Figure 6. Volumetric water volume/soil moisture effect determined by wet and dry weight of soil assessment in relation to gEcrif-R survival for each sampling month in organic soil with cucumber and amended with poultry manure + cover crops in the 2018 (A) and 2019 (B) experimental seasons. HVPL, Hairy vetch + Poultry litter; FRPL, Forage radish + Poultry litter; HVPP, Hairy vetch + Poultry pellets; FRPP, Forage radish + Poultry pellets; bnds, Bands; ss, Subsurfacing.
Observations up to day 90 in the 2018 growing season showed a continuous decrease in gEc rif-R population in the spinach and radish soils (Figure 7A). There were no significant differences at (p < 0.05) in gEc rif-R populations at recovery among treatments for over 90 days (Figure 7A). Data from the 2019 experiment also showed continuous declines in gEc rif-R populations up to day 90 sampling (Figure 7B). No significant differences were observed in gEc rif-R populations across the treatments. Weather patterns and E. coli decline showed some association in the 2018 growing season. High temperatures (air and soil) were observed from Day 1 to Day 30 with a steady decline from October to November (Day 60 to Day 90). The same trend of decline was observed in the E. coli population over this period (Figure 8A). In 2019, the temperature pattern was different as both air and soil temperatures were steady from August to September (Figure 8B). There were some declines in September and a further decline in October. The soil moisture relationship with E. coli recovery was like that of temporal effects for the 2018 experimental season (Figure 9A). The same pattern was also observed during the 2019 experimental season, where E. coli survival decreased when soil moisture decreased (Figure 9B). These data show that environmental factors have a differential impact on E. coli survival.
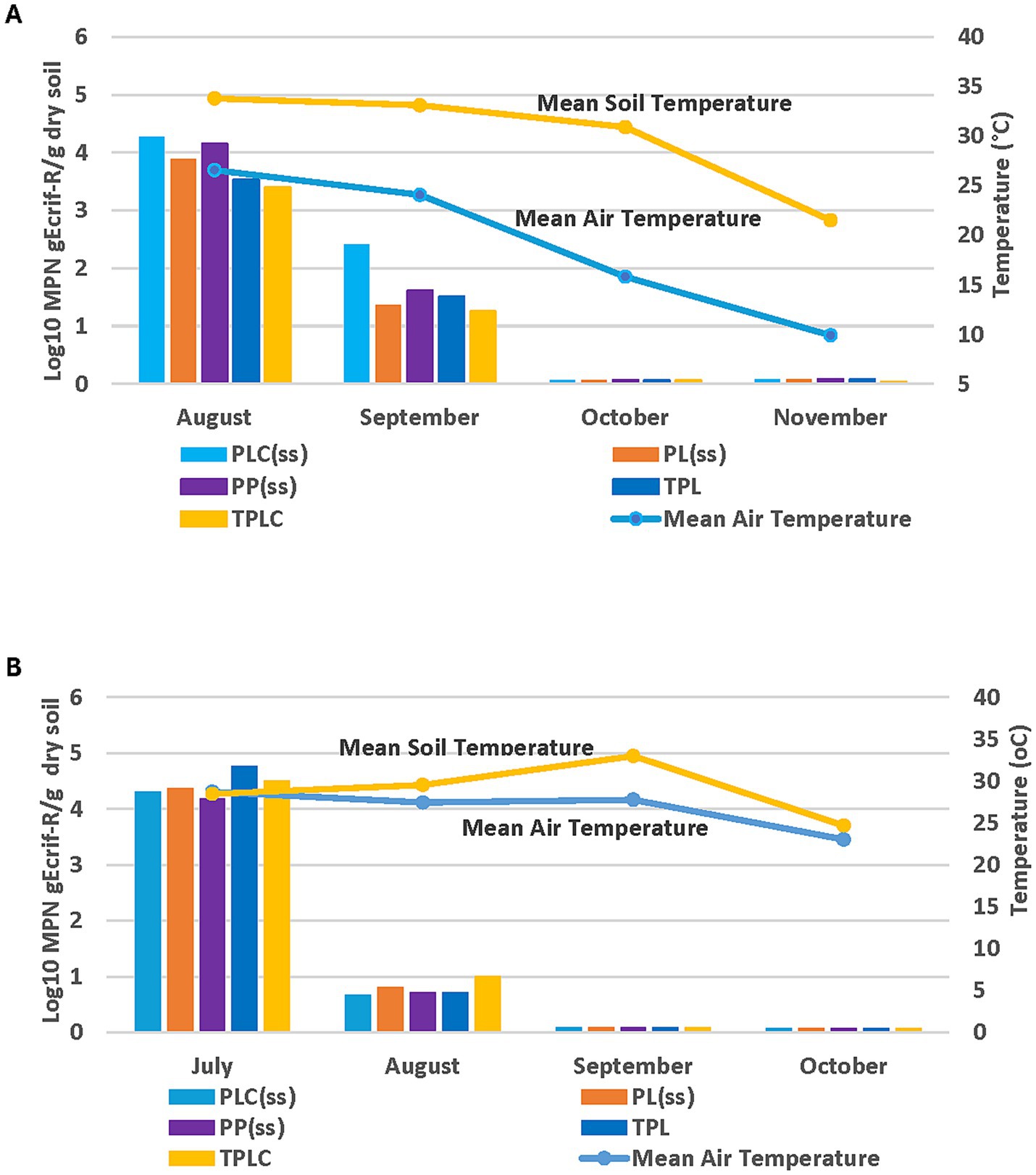
Figure 7. Effect of manure type and application method on survival and persistence of gEcrif-R in organic soil with spinach and radish in 2018 (A) and 2019 (B). PLC, Poultry litter compost; PL, Poultry litter; TPL, Tilled-in poultry litter; TPLC, Tilled-in poultry litter compost; PP, Poultry pellets. Error bars indicate standard error of the mean.
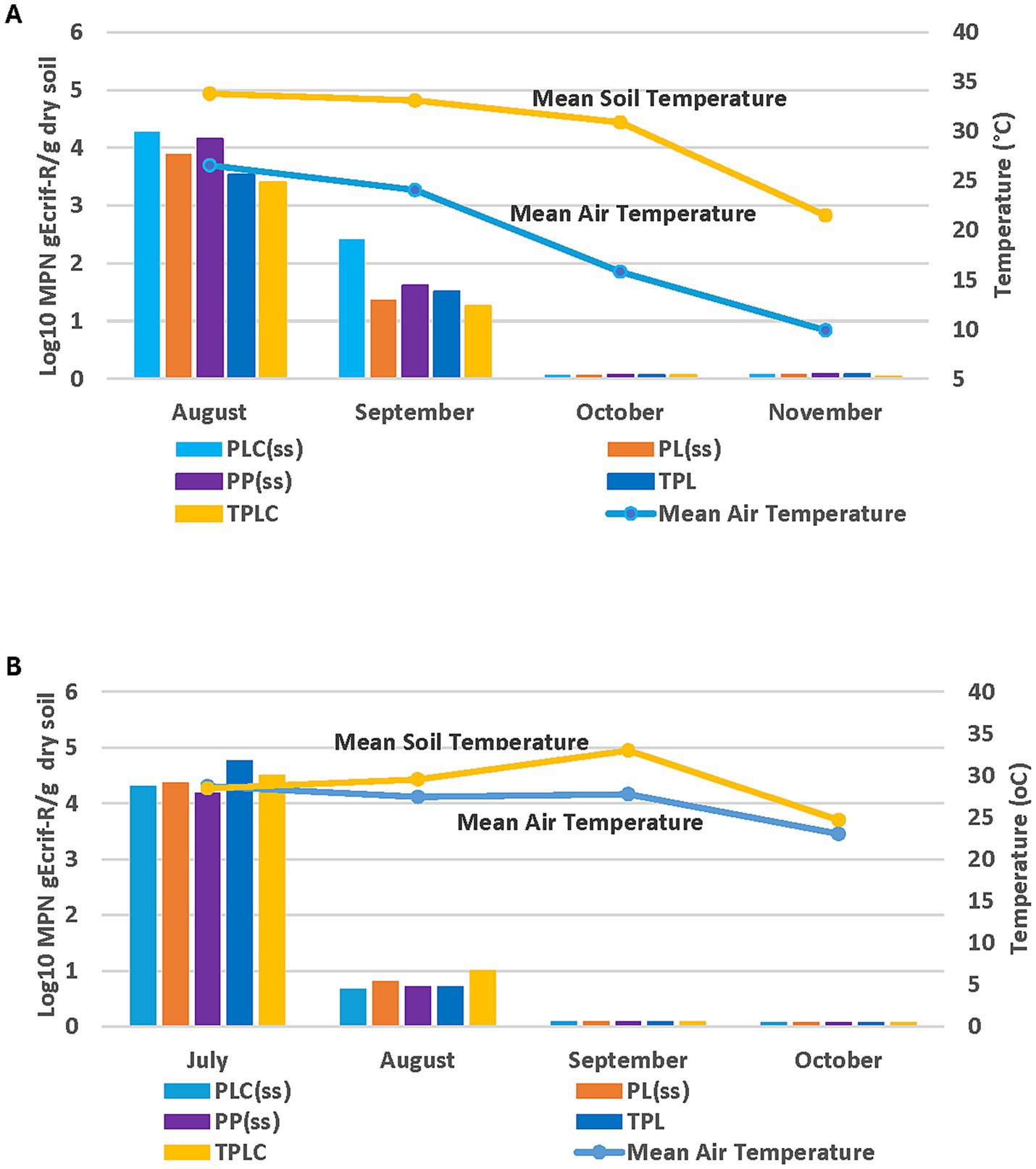
Figure 8. Temperature and temporal effects for gEcrif-R survival for each sampling month in organic soil with spinach/radish and amended with poultry manure in the 2018 (A) and 2019 (B) experimental seasons. PLC, Poultry litter compost; PL, Poultrylitter; TPL, Tilled-in poultry litter; TPLC, Tilled-in poultry litter compost; PP, Poultry pellets; ss, sub surfacing (no cover crop).
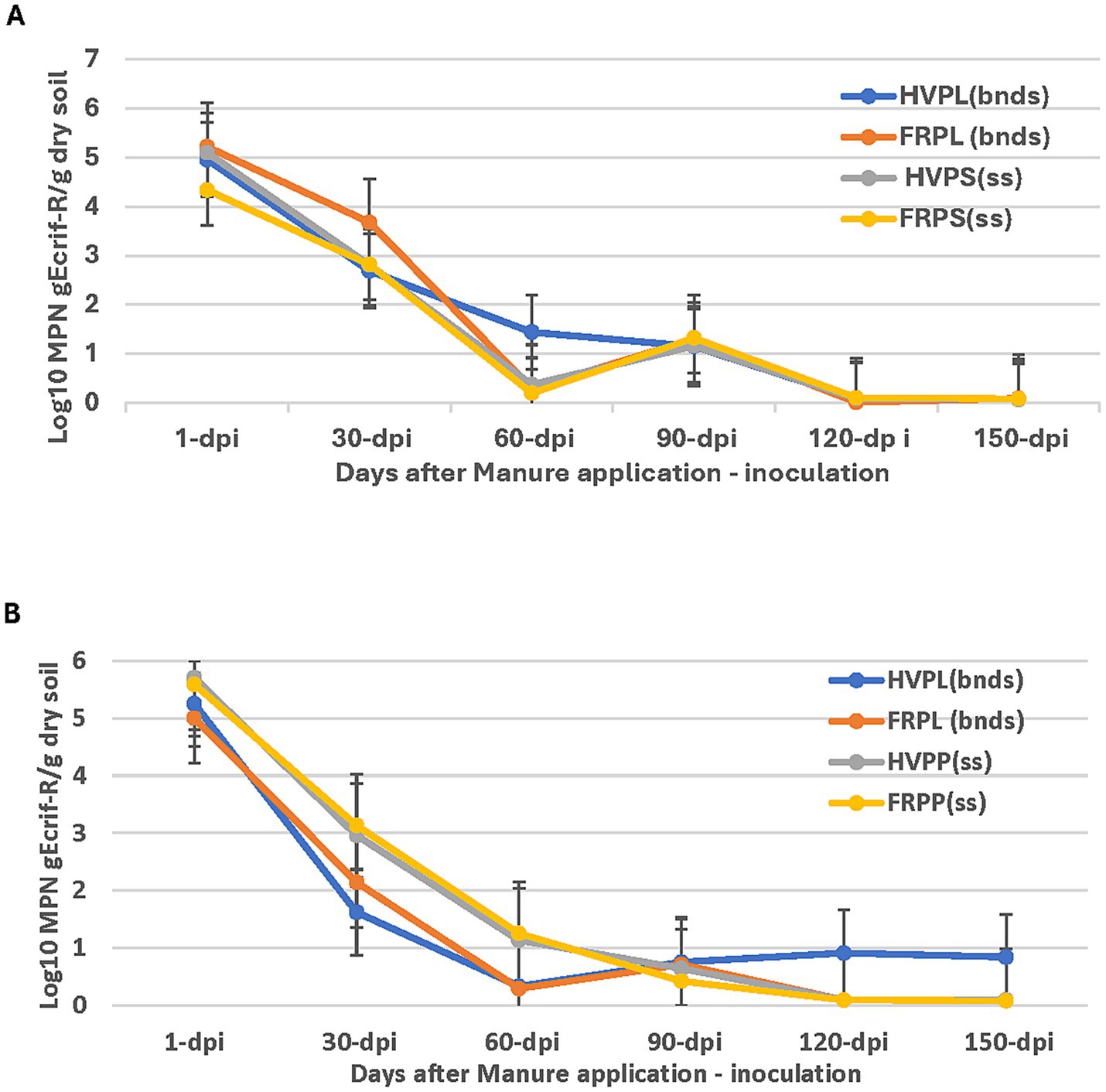
Figure 9. Volumetric water content/soil moisture effect determined by wet and dry weight of soil assessment in relation to gEcrif-R survival for each sampling month in organic soil cropped with spinach/radish and amended with poultry manure and cover crops in the 2018 (A) and 2019 (B) experimental seasons. PLC, Poultry litter compost; PL, Poultry litter; TPL, Tilled-in poultry litter; TPLC, Tilled-in poultry litter compost; PP, Poultry pellets; ss, sub surfacing (no cover crop).
Escherichia coli transfer and contamination of fresh produce (cantaloupes, cucumbers, spinach, and radish) grown organically in soils amended with poultry manure at UMES.
Data from these experiments show that none of the pathogens, Salmonella sp., Staphylococcus aureus, or Listeria monocytogenes, were detected on the produce in the growing seasons summer of 2018 and 2019. For cucumber and cantaloupe growing seasons 2018 and 2019, E. coli survival and transfer to produce support the appropriateness of the NOP 120-day harvest wait-time rule, but not the 90-day rule. This result is based on E. coli recovery from produce after poultry litter product applications, even with plastic mulch used on plots to keep cantaloupe and cucumber fruits from direct contact with the soil. These vines and fruits often grow off the plastic mulch and are subject to soil dispersal and rind contact from planting holes in the plastic mulch during rain events. Data showed the presence of E. coli on eight fruits (4 cucumbers and 4 cantaloupes from the FRPL treatment combination) from fruits sampled on day 90 in 2018 and 2019, but E. coli was undetectable at 120dpiFRPL.
Spinach and radish failure to germinate, even after reseeding attempts, in the 2018 research season precluded microbial analysis for both crops. In the 2019 growing season, eight sampled radish bulbs showed 4 bulbs from the PLss and TPL treatment combinations where E. coli was detected on day 90 and an absence of E. coli on all bulbs harvested and tested on day 120. For the 2019 growing season, spinach was processed and analyzed on day 120 only; this was after replanting a second batch of seeds. On day 120, E. coli was detected from two plants in the PLss plots. No other pathogens were detected on any samples (spinach or radish) on days 90 or 120.
Results for soils cropped with cantaloupe, cucumber, spinach, and radishes showed that survival and persistence of E. coli populations involve several environmental factors as well as on-farm management practices. Sharma et al. (2019) reported that seasonality also contributes to gEc rif-R survival duration in manure-amended soils. Populations of E. coli in these soils are affected by temperature which may account for population decline patterns (Semenov et al., 2007). Except for the 2018 season (Figures 1A, 4A), where there was a significant difference in E. coli population for HVPL compared to the other treatments on day 60, no other difference was observed for treatments to population survival for all amended plots (cantaloupes, cucumbers, spinach, and radishes) in the 2018 and 2019 seasons. Pre-season cover cropping with hairy vetch or forage radish and placement methods used as a combined treatment type did not show any major impact on the decline of E. coli populations in cantaloupe or cucumber-cropped soils. In another study, incorporation of three cover crops, mustard greens (Brassica juncea), Sunn hemp (Crotalaria juncea), and buckwheat (Fagopyrum esculentum) resulted in significantly reduced populations of the inoculated generic E. coli strain compared to the control within 10 to 30 days indicating a role for those cover crops in mitigating E. coli survival in soil (Zhao et al., 2023). When compared, aged poultry litter did not show any significant difference in population declines or survival patterns relative to thermophilic poultry litter compost and heat-treated poultry litter pellets (Figures 1A,B, 4A,B, 7B). There were no other forms of animal manure included in this study for comparison. However, studies have reported longer survival for E. coli in poultry litter-amended soils when compared to other types of animal manure such as dairy or horse (Sharma et al., 2016). Other studies by Sharma et al. (2016, 2019) have shown that even though the differences in E. coli survival were significantly noticeable in poultry litter compared to other untreated animal manure, the die-off timelines were within NOP-stipulated harvest wait times. Results from other studies also have confirmed that the NOP stipulations of 90 and 120-day harvest wait times are appropriate for crop production on soils amended with treated and untreated poultry manure.
In general, the decline in gEcrif-R populations in both seasons (2018 and 2019) for manure placements may be due in part to the similarity in application types. All three methods involved poultry products being applied at a particular depth (approximately 6.35 cm) and eventually being covered by soil. All plots also had been cover cropped in this study. Incorporation of product into the soil at this depth could facilitate litter product decomposition and thereby cause stimulation of a competitive/inhibitory microbial population or a depletion of available, suitable nutrients to sustain E. coli populations (Mahler et al., 1994; Mahler and McDole, 2001). Likewise, the production of decomposition products inhibitory to E. coli may occur to a greater extent than the cumulative conditions affecting E. coli populations closer to the soil surface, such as with the broadcast of BSAAO products (Mahler et al., 1994; Mahler and McDole, 2001). According to studies carried out by Sharma et al. (2019), differences were observed in the survival of E. coli when applied on the surface in comparison to those involving subsurface or minimal tillage. These results indicate that incorporating raw manure into the soil is a technique that may help to limit the survival and persistence of E. coli bacteria. Hutchison et al. (2004) concluded that the amount of time raw manure remains on the soil surface influences the rate at which pathogen populations decline. Conversely, incorporating manure into the soil would prolong the time these pathogens remain viable. However, leaving manure on the soil surface may lead to increasing the risk of spreading pathogens in the environment and potentially to the fresh produce grown on the soil.
For both experimental seasons (2018 and 2019), data in this project showed a relationship between the gEcrif-R survival and temperature. Higher temperatures supported populations of E. coli, but as temperatures began to decline, E. coli populations declined in cantaloupe and cucumber plots (Figures 2A,B, 4A,B). Similar temporal and moisture patterns were observed in spinach and radish plots (Figures 8A,B) for both experimental seasons (2018 and 2019). Temperature and temporal data showed that E. coli populations survived in warmer months (Figures 2A,B, 5A,B, 8A,B). However, the contrasting soil moisture data from 2018 and 2019 relative to E. coli population changes, suggests the potential influence of confounding factors not measured in these experiments. One such factor is the different soil types present in the experimental plots for 2018 (sandy loam) and 2019 (silty loam). These soils have different structural components which may impact these organisms differently. Clearly, E. coli populations survived well in warm months when the temperature was at or above the maximum daily growing degree temperature (30°C); and populations declined as temperature declined. When temperatures fell below the minimum daily growing temperature (10°C), populations started to die off and eventually achieved undetectable levels. Fluctuations were most noticeable in cantaloupe and cucumber plots, but consistent patterns were present for the spinach and radish plots. The E. coli populations continued to decline after initial temperature declines. However, data for the 2019 experimental season in this project showed that E. coli population decreased along with a decrease in soil moisture. Environmental conditions at a given location and time impact pathogen survival and persistence. Temperature is a major factor, the pattern of which often coincides with the survival pattern of these microbes. Even though this study examined the interrelationship between temperature and soil moisture with the pattern of survivability, these findings also show that no single factor accounts for E. coli survival when levels hover in a moderate range. The data also show that one factor may have a different effect on the survival pattern in contrast to another (Williams et al., 2015). This means that these factors can be re-evaluated, and a more in-depth comparison made with other investigations carried out in other locations that have different environmental trends and patterns such as topography and the type of macro- and micro-flora and fauna present. These environmental factors, especially temperature patterns are complex and differ by region. Moreover, based on the findings in this study, the stipulations will satisfy the regulations of the NOP for the Delmarva region, but the experiment could be repeated in a different region to confirm the strength of these relationships and effectiveness in the agriculture industry.
Commonly, fresh produce production has occurred in an open field where they are continuously exposed to pre-harvested microbial contamination via irrigation water, agriculture soil, and raw or improperly composted manure. The agricultural soil used in production may naturally contain certain pathogens or have been populated by the application of animal manure, such as poultry litter (as used in this research). Animal manure can directly contaminate the food crops grown, especially if the fruit is damaged by bruises or cuts (Iwu and Okoh, 2019). Physical damage can wound the integrity of the produce surface and create a path for microbial contamination. The abrasions not only create areas that harbor contaminants but may also decrease the efficacy of washing this fresh produce for consumption. Therefore, it is important that farmers take into consideration the growth and harvesting of their produce in such a way that minimizes the damage and limits pathogen contamination. In a previous study conducted by Jensen et al. (2006), the transfer and survival of E. coli to lettuce in direct contact with soil amended with raw manure and grown under natural on-farm management practices was determined to be contaminated by E. coli from the surrounding environment and wildlife. In the same research, it was also determined that the fields with the shortest wait time intervals between application of manure and harvest supported the highest percentage of E. coli contamination. Several studies have reported on risk and protective factors associated with farm management procedures, environment, and weather that can collaboratively contribute to the contamination of fresh produce grown on manure amended soils (Park et al., 2014; Pang et al., 2020; Phan-Thien et al., 2020, Pires et al., 2023, Strawn et al., 2013; Xing et al., 2019). The cucumbers and cantaloupes used in the research were placed on plastic mulch to prevent direct contact with the soil, but contamination was detected at 90 dpi. This data explains the contamination of the produce from observed splash of soil particles during heavy rainfall onto the plants and fruits (Allende et al., 2017). In contrast, spinach and radish plots had no plastic mulch and thus were susceptible to contamination from direct contact with amended soil. Radish bulbs directly contacted the soil, so they were more susceptible to microbial contamination. However, as the E. coli population declined, so did transfer of E. coli. By 120 dpi, contamination potential, and its occurrence declined significantly from those immediately post-inoculation, resulting in no contaminated cucumbers, cantaloupes, and radish, with only two spinach plants (from 20 sampled) detected positive for E. coli. As the E. coli population declined, so did the potential of E. coli contamination. Clearly, contamination potential and occurrence declined by 120 dpi, resulting in undetectable contamination in cantaloupes and cucumbers. The probability of irrigation water as a source of microbial contamination was excluded since none of the E. coli inoculum strains (TVS-353, TVS-354, and TVS-355) were detected in the well water used for irrigation, which was tested at the initial stage of this project. Water quality was also tested every 30 days throughout the experiment in 2018 and 2019 to ensure contaminant-free irrigation water. Although contamination occurs at the pre-harvest stages in production, contamination can also happen at post-harvest stages through improper hygiene and handling, equipment, and processing. The pathways of contamination occur mainly through cuts and bruises on the edible portion of the produce (Iwu and Okoh, 2019).
Results from this research showed a gradual decline in the population of the generic rifampicin-resistant E. coli (gEcrif-R) over the sampling periods for both experimental seasons (2018 and 2019). These declines also occurred within the stipulated National Organic Program (NOP) manure application guidance periods for produce harvest dates (90 and 120-dpi). Results suggest that temperature was a major parameter associated with the decline of gEcrif-R populations. This data is in harmony with findings from other studies that examined the survival of E. coli and Salmonella in manure-amended soils (Bardsley et al., 2021; Cheong et al., 2024; Sharma et al., 2019; Tran et al., 2020). Ascribing a single factor for bacterial pathogen decline has been a challenge because assessing other environmental factors that may interact with temperature also can affect E. coli and can vary by location. Temperature also affects soil moisture content, which contributes to E. coli population dynamics. In addition to temperature, manure application methods can impact the survival of foodborne pathogens. Escherichia coli, when assessed under field conditions, has a greater opportunity to contaminate fresh produce when applied only to the soil surface as compared to incorporation into the soil. In this research, there was no comparison between surface and subsurface manure application. However, based on the data presented here, there were no significant differences in E. coli survival between soil with banded PL product trenching and banded cultivation. The comparison reveals that population decline patterns for E. coli were the same regardless of application/incorporation treatment type and were related to external environmental effects.
Initial levels of microorganisms in manure products can vary significantly. Previous studies have suggested that feedlot cattle manure can harbor up to 6.4 log CFU/g of pathogenic E. coli (Klein et al., 2010), and broiler litter can contain up to 5.0 log CFU/g of Salmonella spp. (Chinivasagam et al., 2010). Based on the average inactivation of gEcrif-R at field conditions by 90 and 120 dpi E. coli population declines show that a median response indicative of consistent and significant population declines within the period stipulated by the NOP guidelines for reduced risk of produce contamination. The population decline in soil and the low number of contaminated cantaloupe and cucumber samples detected occurred even though both these vine fruits exterior rind and skin had direct contact with the soil which had been initially inoculated with gEcrif-R. It is also notable that even though APL was not treated with a validated process to inactivate E. coli, it was only aged, and the litter and soil into which it was incorporated were free of Listeria monocytogenes, Salmonella, and Staphylococcus aureus.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
PM-R: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing. PM: Data curation, Formal analysis, Investigation, Methodology, Software, Validation, Visualization, Writing – original draft, Writing – review & editing, Conceptualization. FH: Conceptualization, Funding acquisition, Methodology, Resources, Supervision, Validation, Writing – original draft, Writing – review & editing. LM: Conceptualization, Funding acquisition, Project administration, Resources, Writing – original draft, Writing – review & editing. BS: Investigation, Methodology, Resources, Writing – review & editing. AK: Data curation, Investigation, Methodology, Resources, Visualization, Writing – review & editing. AA: Investigation, Supervision, Writing – review & editing. SP: Formal analysis, Methodology, Validation, Writing – original draft, Writing – review & editing. AC: Data curation, Formal analysis, Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was funded by the USDA-NIFA 1890 Capacity Building Program, (Award# 1012123).
The authors thank the persons responsible for organizing the project and also lab associates at the UMES Department of Agriculture, Food, and Resource Sciences and at USDA-ARS, Environmental Microbial and Food Safety Laboratory, Beltsville, MD.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Allende, A., Castro-Ibáñez, I., Lindqvist, R., Gil, M. I., Uyttendaele, M., and Jacxsens, L. (2017). Quantitative contamination assessment of Escherichia coli in baby spinach primary production in Spain: effects of weather conditions and agricultural practices. Int. J. Food Microbiol. 257, 238–246. doi: 10.1016/j.ijfoodmicro.2017.06.027
Bardsley, C. A., Weller, D. L., Ingram, D. T., Chen, Y., Oryang, D., Rideout, S. L., et al. (2021). Strain, soil-type, irrigation regimen, and poultry litter influence Salmonella survival and die-off in agricultural soils. Front. Microbiol. 12:590303. doi: 10.3389/fmicb.2021.590303
Barrett, D. M., Beaulieu, J. C., and Shewfelt, R. (2010). Color, flavor, texture, and nutritional quality of fresh-cut fruits and vegetables: desirable levels, instrumental and sensory measurement, and the effects of processing nutritional quality of fresh-cut fruits and vegetables. Crit. Rev. Food Sci. Nutr. 50, 369–389. doi: 10.1080/10408391003626322
Chandler-Khayd, C., Di Francesco, J., Baron, J. N., Ramos, T. D. M., Aminabadi, P., Jay-Russell, M. T., et al. (2023). Risk factors associated with the prevalence of Listeria monocytogenes in manured soils on certified organic farms in four regions of the United States. Front. Sustain. Food Syst. 7:1222192. doi: 10.3389/fsufs.2023.1222192
Cheong, S., Jay-Russell, M. T., Chandler-Khayd, C., Di Francesco, J., Haghani, V., Aminanadi, P., et al. (2024). Presence of foodborne pathogens and survival of generic Escherichia coli in an organic integrated crop-livestock system. Front. Sustain. Food Syst. 8:1343101. doi: 10.3389/fsufs.2024.1343101
Chinivasagam, H. N., Redding, M., Runge, G., and Blackall, P. J. (2010). Presence and incidence of food-borne pathogens in Australian chicken litter. Brit. Poultry Sci. 51, 311–318. doi: 10.1080/00071668.2010.499424
Eblen, D. R., Annous, B. A., and Sapers, G. M. (2005). Studies to select appropriate nonpathogenic surrogate Escherichia coli strains for potential use in place of Escherichia coli 0157:H7 and Salmonella in pilot plant studies. J. Food Protect. 68, 282–291. doi: 10.4315/0362-028X-68.2.282
FDA. (2015). Standards for the growing, harvesting, packing, and holding of produce for human consumption. Federal Register The Daily Journal of the United States Government. Available at: https://www.federalregister.gov/documents/2015/11/27/2015-28159/standards-for-the-growing-harvesting-packing-and-holding-of-produce-for-human-consumption (Accessed March 11, 2025).
Harvey, R. R., Zakhour, C. M., and Gould, L. H. (2016). Foodborne disease outbreaks associated with organic foods in the United States. J. Food Protect. 79, 1953–1958. doi: 10.4315/0362-028X.JFP-16-204
Hutchison, M. L., Walters, L. D., Moore, A., Crookes, K. M., and Avery, S. M. (2004). Effect of length of time before incorporation on survival of pathogenic bacteria present in livestock wastes applied to agricultural soil. Appl. Environ. Microbiol. 70, 5111–5118. doi: 10.1128/AEM.70.9.5111-5118.2004
Iwu, C. D., and Okoh, A. I. (2019). Preharvest transmission routes of fresh produce associated bacterial pathogens with outbreak potentials. Int. J. Environ. Res. Publ. Health 16, 4407–4444. doi: 10.3390/ijerph16224407
Jensen, A. N., Dalsgaard, A., Stockmarr, A., Nielsen, E. M., and Baggesen, D. L. (2006). Survival and transmission of Salmonella enterica serovar Typhimurium in an outdoor organic pig farming environment. Appl. Environ. Microbiol. 72, 1833–1842. doi: 10.1128/AEM.72.3.1833-1842.2006
Klein, M., Brown, L., Tucker, R. W., Ashbolt, N. J., Stuetz, R. M., and Roser, D. J. (2010). Diversity and abundance of zoonotic pathogens and indicators in manures of feedlot cattle in Australia. Appl. Environ. Microbiol. 76, 6947–6950. doi: 10.1128/AEM.01095-10
Limoges, M. A., Neher, D. A., Weicht, T. R., Millner, P. D., Sharma, M., and Donnelly, C. (2022). Differential survival of Escherichia coli and Listeria spp. in northeastern U.S. soils amended with dairy manure compost, poultry litter compost, and heat-treated poultry pellets and fate in raw edible radish crops. J. Food Protect. 85, 1708–1715. doi: 10.4315/JFP-21-261
Litt, P. K., Kelly, A., Omar, A., Johnson, G., Vinyard, B. T., Kniel, K. E., et al. (2021). Temporal and agricultural factors influence Escherichia coli survival in soil and transfer to cucumbers. Appl. Environ. Microbiol. 87, e02418–e02420. doi: 10.1128/AEM.02418-20
Maffei, D. F., Batalha, E. Y., Landgraf, M., Schaffner, D. W., and Franco, B. (2016). Microbiology of organic and conventionally grown fresh produce. Brazil. J. Microbiol. 47, 99–105. doi: 10.1016/j.bjm.2016.10.006
Mahler, R. L. (1994). Nitrogen source, timing of application and placement: effects on winter wheat production. Agron. J. 86, 637–642. doi: 10.2134/agronj1994.00021962008600040010x
Mahler, R. L., and McDole, R. E. (2001). Fertilizer placement. Department of Plant Soil and Entomological Sciences. Moscow: University of Idaho. Current information series. Available at: https://www.lib.uidaho.edu/digital/uiext/items/uiext29736.html (Accessed March 26, 2025).
Maryland Department of Agriculture (2012) Fact sheet Maryland’s revised nutrient management regulations. Available online at: http://mda.maryland.gov/resource_conservation/counties/NMPqanda.pdf. (Accessed March 11, 2025).
Moebius-Clune, B. N., Moebius-Clune, D. J., Gugino, B. K., Idowu, O. J., Schindelbeck, R. R., and Ristow, H. M., et al. (2016). Comprehensive assessment of soil health: the Cornell framework. Edition 3.2. Geneva, NY: Cornell University. Available at: https://cornell.app.box.com/s/ryy6xgtwok5l85zzy9fgms1276drud0y (Accessed March 11, 2025).
Neher, D. A., Cutler, A. J., Weicht, T. R., Sharma, M., and Millner, P. D. (2019). Composts of poultry litter or dairy manure differentially affect survival of enteric bacteria in fields with spinach. J. Appl. Microbiol. 126, 1910–1922. doi: 10.1111/jam.14268
Ohno, T., and Hettiarachchi, G. M. (2018). Soil chemistry and one health initiative: introduction to the special section. J. Environ. Qual. 47, 1305–1309. doi: 10.2134/jeq2018.08.0290
Pang, H., Mokhtari, A., Chen, Y., Oryang, D., Ingram, D. T., Sharma, M., et al. (2020). A predictive model for survival of Escherichia coli O157:H7 and generic E. coli in soil amended with untreated animal manure. Risk Anal. 40, 1367–1382. doi: 10.1111/risa.13491
Park, S., Navratil, S., Gregory, A., Bauer, A., Srinath, I., Szonyi, B., et al. (2014). Farm management, environment, and weather factors jointly affect the probability of spinach contamination by generic Escherichia coli at the preharvest stage. Appl. Environ. Microbiol. 80, 2504–2515. doi: 10.1128/AEM.03643-13
Phan-Thien, K., Metaferia, M. H., Bell, T. L., Bradbury, M. I., Sassi, H. P., van Ogtrop, F. F., et al. (2020). Effect of soil type and temperature on survival of Salmonella enterica in poultry manure-amended soils. Lett. Appl. Microbiol. 71, 210–217. doi: 10.1111/lam.13302
Pires, A. F. A., Ramos, T. D. M., Baron, J. N., Millner, P. D., Pagliari, P. H., Hutchinson, M., et al. (2023). Risk factors associated with the prevalence of Shiga-toxin-producing Escherichia coli in manured soils on certified organic farms in four regions of the USA. Front. Sustain. Food Syst. 7:1125996. doi: 10.3389/fsufs.2023.1125996
Ramos, T. B., Darouich, H., Šimůnek, J., Goncalves, M. C., and Martins, J. C. (2019). Soil salinization in very high-density olive orchards grown in southern Portugal: current risks and possible trends. Agri. Water Manage. 217, 265–281. doi: 10.1016/j.agwat.2019.02.047
Reed-Jones, M. L., Marine, S. C., Everts, K. L., and Mecallef, S. A. (2016). Effects of cover crop species and season on population dynamics of Escherichia coli and Listeria innocua in soil. Appl. Environ. Microbiol. 82, 1767–1777. doi: 10.1128/AEM.03712-15
Semenov, A. V., van Bruggen, A. H. C., van Overbeek, L., Termorshuizen, A. J., and Semenov, A. M. (2007). Influence of temperature fluctuations on Escherichia coli O157:H7 and Salmonella enterica serovar Typhimurium in cow manure. FEMS Microbiol. Ecol. 60, 419–428. doi: 10.1111/j.1574-6941.2007.00306.x
Shah, M. K., Bradshaw, R., Nyarko, E., Handy, E. T., East, C., Millner, P. D., et al. (2019). Salmonella enterica in soils amended with heat-treated poultry pellets survived longer than bacteria in unamended soils and more readily transferred to and persisted on spinach. Appl. Environ. Microbiol. 85, e00334–e00319. doi: 10.1128/AEM.00334-19
Sharma, M., Millner, P. D., Hashem, F., Camp, M., Whyte, C., Graham, L., et al. (2016). Survival and persistence of nonpathogenic Escherichia coli and attenuated Escherichia coli O157:H7 in soils amended with animal manure in a greenhouse environment. J. Food Protect. 79, 913–922. doi: 10.4315/0362-028X.JFP-15-421
Sharma, M., Millner, P. D., Hashem, F., Vinyard, B., East, C. L., Handy, E., et al. (2019). Survival of Escherichia coli in manure-amended soils is affected by spatiotemporal, agricultural, and weather factors in the mid-Atlantic United States. Applied. Environ. Microbiol. 85, 1–23. doi: 10.1128/AEM.02392-18
Strawn, L. K., Gröhn, Y. T., Warchocki, S., Worobo, R. W., Bihn, E. A., and Wiedmann, M. (2013). Risk factors associated with Salmonella and Listeria monocytogenes contamination of produce fields. Appl. Environ. Microbiol. 79, 7618–7627. doi: 10.1128/AEM.02831-13
Tomás-Callejas, A., López-Velasco, G., Camacho, A. B., Artés, F., Artés Hernández, F., and Suslow, T. V. (2011). Survival and distribution of Escherichia coli on diverse fresh-cut baby leafy greens under preharvest through postharvest conditions. Int. J. Food Microbiol. 151, 216–222. doi: 10.1016/j.ijfoodmicro.2011.08.027
Tran, D., Bradbury, M., Van Ogtrop, F., Bozkurt, H., Jones, B., and Mcconchie, R. (2020). Environmental drivers for persistence of Escherichia coli and Salmonella in manure-amended soils: a meta-analysis. J. Food Protect. 83, 1268–1277. doi: 10.4315/0362-028X.JFP-19-460
Turner, K., Moua, C. N., Hajmeer, M., Barnes, A., and Needham, M. (2019). Overview of leafy, greens-related food safety incidents with a California link: 1996 to 2016. J. Food Protect. 82, 405–414. doi: 10.4315/0362-028X.JFP-18-316
US EPA. (2024). Most probable number (MPN) calculator. Available online at: https://mostprobablenumbercalculator.epa.gov/mpnForm (Accessed September 12, 2024).
USDA-AMS NOP. (2011). NOP 5006. Guidance. Processed animal manures in organic crop production. Available at: https://www.ams.usda.gov/sites/default/files/media/5006.pdf (Accessed March 11, 2025).
Williams, M. L., LeJeune, J. T., and Gardener, B. M. (2015). Soil conditions that can alter natural suppression of Escherichia coli O157:H7 in Ohio specialty crop soils. Appl. Environ. Microbiol. 81, 4634–4641. doi: 10.1128/AEM.00125-15
Xing, J., Wang, H., Brookes, P. C., Salles, J. F., and Xu, J. (2019). Soil pH and microbial diversity constrain the survival of E. coli in soil. Soil Biol. Biochem. 128, 139–149. doi: 10.1016/j.soilbio.2018.10.013
Keywords: food safety, E. coli, Salmonella, Listeria monocytogenes, Staphylococcus aureus, poultry litter biological soil amendments, organic agriculture, cover crops food safety
Citation: McKenzie-Reynolds P, Millner P, Hashem F, Marsh L, Smith B, Kenney A, Allen A, Parveen S and Collick AS (2025) Survival and transfer of Escherichia coli to fresh produce from organically managed soils amended with poultry litter. Front. Sustain. Food Syst. 9:1502841. doi: 10.3389/fsufs.2025.1502841
Received: 27 September 2024; Accepted: 18 February 2025;
Published: 03 April 2025.
Edited by:
John J. Maurer, Virginia Tech, United StatesReviewed by:
Junia Jean-Gilles Beaubrun, United States Department of Agriculture (USDA), United StatesCopyright © 2025 McKenzie-Reynolds, Millner, Hashem, Marsh, Smith, Kenney, Allen, Parveen and Collick. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Fawzy Hashem, Zm1oYXNoZW1AdW1lcy5lZHU=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.