- 1Department of Human Nutrition, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, Multan, Pakistan
- 2University Institute of Food Science and Technology, The University of Lahore, Lahore, Pakistan
- 3Department of Food Science, Government College University Faisalabad, Faisalabad, Pakistan
- 4Department of Human Nutrition and Dietetics, Riphah International University, Faisalabad, Pakistan
- 5Faculty of Food Engineering, Ştefan Cel Mare University of Suceava, Suceava, Romania
- 6Faculty of Mechanical Engineering, Automotive, and Robotics, Ștefan Cel Mare University of Suceava, Suceava, Romania
Terminalia chebula is a valuable medicinal plant that can be used in human nutrition. The current study was conducted on different varieties of Terminalia chebula (Harad) and aimed to investigate their proximate composition, antioxidant activity (DPPH and FRAP assay), and total phenolic content (TPC) by using different solvents (ethanol, acetone, hexane, and distilled water) for further use in the development of functional muffins. Wheat flour was supplemented with the Harad varieties (at 5%, 7.5%, 10%, or 15%) for the preparation of muffins. The color tonality (L*, a*, b*, C*, and h*) of the final product was measured with a digital colorimeter, and the sensory analysis was performed by using a 15-point scale. The results showed that the DPPH values of the Harad samples varied from 22.16 ± 1.27 to 84.33 ± 1.29, while the FRAP values ranged from 18.65 ± 1.73 to 113.4 ± 2.1 mmol FeSO4/g, depending on the solvent type. TPC in different Harad varieties ranged from 10.77 ± 2.35 to 107.20 ± 1.80 mg GAE/100 g, whereas the total flavonoid content ranged from 3.78 ± 0.03 to 47.91 mg QE/100g. Among Harad varieties, the Kabuli variety contains the highest amount of Ca, Na, and K, namely 95.58, 2.16, and 113.66 mg/kg, followed by the black variety with 89.58, 1.83, and 105.08 mg/kg respectively. The muffins prepared with black Harad give the highest values of the color parameters compared to other samples. According to the consumer’s choice, the sensory profile assessment revealed that the muffins supplemented with 7.5% of different Harad varieties (green, black, and Kabuli) were the most acceptable. Therefore, Harad varieties could partially replace the white flour in muffins to improve their nutritional value. These results can be useful for the bakery industry interested in developing new functional foods.
1. Introduction
Herbal medicine gained popularization over the years, and it was the base of the modern medicine’s development. According to a survey, 70–80% of the world population directly or indirectly depends on medicinal herbs for primary health care and treatments (Panda et al., 2019; Ranjha et al., 2020; Shehzadi et al., 2020; Sabtain et al., 2021). The plants are used in several medicinal systems in many countries and are natural and available cheap sources of bioactive compounds with benefits in chronic disease prevention and treatment (Panda et al., 2019; Ranjha et al., 2020; Shehzadi et al., 2020; Sabtain et al., 2021). To make the best use of phytoconstituents, their extraction from plant materials (fruits, seeds, peels, leaves, etc.) is necessary (Ranjha et al., 2021; Sabtain et al., 2021; Nadeem et al., 2022).
Scientists screening many medicinal plants discovered that one of the most well-regarded medicinal plants (Mahmood, 2019; Shehzadi et al., 2020) is Terminalia chebula, called also Harad, or Chebulic Myrobalan. T. chebula contains many different kinds of phytoconstituents that unveiled a great number of medicinal activities. The myrobalan fruit has various health merits, being used as a household remedy against numerous human illnesses since ancient times (Majed et al., 2015; Van Wyk and Wink, 2018).
Globally, T. chebula has been identified in tropical regions (Saleem et al., 2002). It is a wild deciduous plant native to south Asia from India, Iran, Pakistan, Burma, southwest China, Vietnam, and Malaysia (Gaire and Kim, 2014; Sadeghnia et al., 2017). In Pakistan, there are three different varieties, namely green, black, and Kabuli, possessing different shapes and sizes. Mostly, the outer skin (pericarp) is used in different pharmacological preparations. T. chebula is cultivated from seed during the spring season in different soil types, from clay to sandy (Sharma et al., 2011).
In many food products, T. chebula is a functional ingredient because it plays a pharmacological role (Sharma et al., 2011). The T. chebula comprises therapeutic constituents such as chebulic acids (Lee et al., 2010), ellagic acid, chebulanin (Chattopadhyay and Bhattacharyya, 2007), gallic acid (Patel Madhavi et al., 2010), luteolin (Kumar, 2006), rutin, ethaedioic ethanoic acid, ethyl gallate, tannic acid (Juang et al., 2004), ascorbic acid, quercetin, neochebulinic acid (Han et al., 2006), ethaedioic acid (Reddy et al., 2009), corilagin, terpinenolsterpineol, terpinenes (Srivastav et al., 2010) chebulinic acids, casuarinin, 2,4-chebulyi–β-D-glucopyranose (Juang et al., 2004) 1, 6-di-O-galloyl-D-glucose, 3,4,6- tri-O-galloyl-D-glucose, 1,2,3,4,6-penta-O-galloyl-D-glucose, punicalaginterflavin-A, terchebin, anthraquinone (Srivastav et al., 2010). Moreover, it is recognized as having anti-inflammatory, antioxidant (Suchalatha and Devi, 2005), antiviral (Kim et al., 2001), antidiabetic (Rao and Nammi, 2006), hepatoprotective (Vidya et al., 2011), antibacterial (Kannan et al., 2009), antiviral (Kim et al., 2001), antifungal (Sahab and Sabbour, 2011), antiulcerogenic (Sharma et al., 2011), antinociceptive (Kaur and Jaggi, 2010), immunomodulatory (Aher and Wahi, 2011), radioprotective (Jagetia et al., 2002), cardioprotective (Suchalatha and Devi, 2005), anti-hyperglycemic (Shreedevi and Sampathkumar, 2011), antiplasmodial (Prakash et al., 2013), anti- arthritis (Seo et al., 2012), hypocholesterolemic (Maruthappan and Shree, 2010) and anticancer (Saleem et al., 2002) activities.
The interest of consumers for functional foods is increasing, and the use of medicinal plants in their development is an opportunity. The influence of such an ingredient on the quality of the final product should be considered in order to obtain the maximum health benefits, without affecting product’s quality. To our knowledge, there are no studies regarding the effects of different Harad varieties on the physical and sensory characteristics of muffins. Furthermore, the characterization of the Harad varieties justifies the possibility to include them in a baked product like muffins, to create a novel functional food. In this research, different varieties of Terminalia chebula were investigated, aiming to develop novel functional muffins by replacing wheat flour in various amounts. For this purpose, the proximate analysis was performed to compare the three varieties (green, black, and Kabuli) of Terminalia chebula, along with the TPC, DPPH, and FRAP employed to measure the phenolic and antioxidant contents, respectively. The color tonality (L*, b*, C*, a*, and h*), sensory texture, and other sensory parameters were determined in order to check the quality and consumer acceptance of muffins supplemented with Terminalia chebula.
2. Materials and methods
2.1. Materials
The present research was accomplished in the postgraduate laboratories of the Food Science and Technology Department at Bahauddin Zakariya University, Multan, Punjab, Pakistan. Different varieties of Harad (T. chebula) were procured from the Al-Hamad Pansare store, Multan. The raw materials for muffin preparation were bought from the local market. At the same time, the analytical grade chemicals were purchased from Merck and Sigma-Aldrich. The different varieties of Harad were manually removed from inessential materials. The gall or fruit particle size was reduced into fine powder through the local grinder and packed and stored at 37°C for further analysis.
2.2. Harad varieties characterization
2.2.1. Proximate analysis of different varieties of Harad
Different varieties of Harad were evaluated for proximate analysis in terms of crude protein, moisture, ash, crude fat, crude fiber, and nitrogen-free extract (NFE), according to their standard methods described by the AOAC (2016). NFE was calculated using the following equation (Eq. 1):
2.2.2. Quantification of minerals
Macro and microelements from T. chebula varieties (green, black, and Kabuli) were measured by using a Flame photometer and atomic absorption spectrophotometer according to the method presented in the AOAC (2016). The sample was digested with nitric acid (HNO3) and perchloric acid (HClO4). After that, it was diluted with de-ionized water and analyzed for specific elements by using a standard calibration.
2.2.3. Antioxidant potential of Terminalia chebula varieties
2.2.3.1. Preparation of the extract
The extracts were prepared with different solvents such as ethanol, acetone, hexane, and distilled water at a 1:10 ratio of powder: solvent. The samples stayed overnight at room temperature (about 25°C), then they were shacked at 280 rpm for 5–8 h, filtered with Whatman filter paper no.1, and evaporated with a vacuum rotary evaporator (Heidolph, Hei-Vap, Germany) at 20°C. Then, the extract was centrifuged at 3,000 rpm for 5 min. Finally, the concentrated extract was stored in 50 mL Falcon tubes at 2–8°C.
2.2.3.2. Total phenolic content determination
The total phenolic content (TPC) of T. chebula was determined according to Singleton et al. (1999) method, with slight modification. The extract reacted with 2.5 mL of 10 times concentrated Folin Ciocalteu Reagent (FCR) and 2 mL of 7.5% Na2CO3 for 30 min in darkness. The different extract samples were measured at 760 nm wavelength using a spectrophotometer and a standard gallic acid solution with 10–100 mg/L concentration.
2.2.3.3. DPPH assay
The free radical scavenging activity was evaluated trough DPPH assay, according to Brand-Williams et al. (1995) method with little modifications. The different extracts of T. chebula ranging between 50–100 μL were diluted up to 100 μL with methanol. A quantity of 2 mL of Harad extract with 3 mL DPPH was incubated for 30 min in a shaded place at room temperature. The calibration standard ascorbic acid was used for measuring free radical scavenging activity at an absorbance of 517 nm (Eq. 2).
Control AB is the absorbance of the control, and Sample AA is the absorbance of the tested samples.
2.2.3.4. Total flavonoids contents
The total flavonoid content (TFC) was measured by means of the method described by Dewanto et al. (2002). An amount of 0.3 mL of NaNO2 (5%) solution was mixed with 3 mL plant extract. Then, 0.6 mL of AlCl3 (10%) solution was added to 2 mL of (1 M) NaOH solution after 5 min. The solution was vortexed and measured at 510 nm wavelength using a spectrophotometer. The TFC was expressed in mg of quercetin equivalent (mg of QE).
2.2.3.5. FRAP assay
The ferric-reducing antioxidant potential of T. chebula extract was measured by using Zahin et al. (2010) with slight modifications. The FRAP reagent comprised 10 mM/L TPTZ solution, sodium acetate buffer solution, and 20 mM/L ferric chloride at a 1:1:10 ratio, at pH of 4.0. A quantity of 100 μL of T. chebula extract reacted with 2 mL of FRAP regent and then it was placed in a dark area for 20–30 min at 37°C. The ferrous sulfate solution with a concentration ranging from 100 to 1,000 μM was used for standard calibration and the absorbance was measured at 593 nm with a spectrophotometer. The results were expressed as μM of Fe (conversion of ions).
2.3. Muffins development
Muffins were prepared by replacing wheat flour (WF) with T. chebula of different varieties (green, black, and Kabuli) in various amounts (Table 1). Firstly, a foam was formed from eggs and sugar, then the powder ingredients were added to the foamy mixture to make the batter. The batter was put into muffins shape holding dish and baked at 149–200°C in the baking oven for 20–25 min. After that, the muffins were cooled at room temperature and kept in a polythene zipper bag for further analysis.
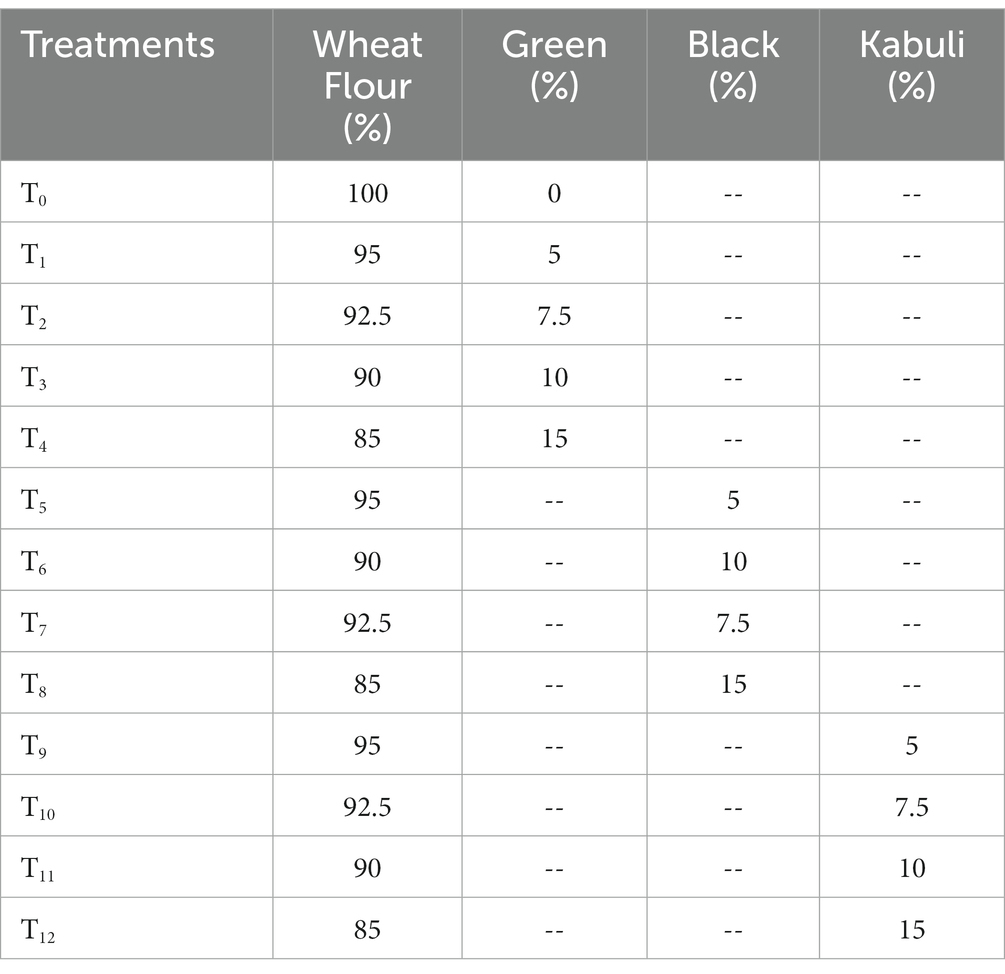
Table 1. Preparation of wheat and Harad (T. chebula) powder (green, black, and Kabuli) supplemented muffins.
2.4. Muffins characterization
2.4.1. Physical analysis
The muffins color was determined with a Chroma meter CR-400 (Konica Minolta INC. Japan). An amount of 55 g of muffins was taken, and the color values in terms of L* (Lightness), a* (+ redness, − greenish), and b (+ yellowness, − blueness) were measured. The C* (Chroma) and h* (Hue angle) were calculated according to Gouveia et al. (2007).
2.4.2. Sensory profile evaluation
The muffins were subjected to sensory evaluation by a trained panel from the Institute of Food Science and Nutrition (student and faculty members: aged from 20 to 55 years) BZU, Multan, Punjab, Pakistan. The total number of panelists was 30. A quantity of 55 g of muffins from each variety and with various addition levels of T. chebula was served at room temperature to the panelists. The T. chebula level used from each variety was unknown by the panelists who were asked to evaluate and record crust, crumb color, and appearance, volume, aroma, mouth feel, overall acceptability by using the 15-points scale method (Meilgaard et al., 1999). Three replications were made. The presentation order of the samples was randomized depending on the addition level and positional codes. Mouth palletizer water and crackers were provided to panelists to rinse their mouths between evaluations. Panelists were accommodated in different booths equipped with bright white lighting in a separate room for each assessment.
2.4.3. Sensory textural analysis
The textural characteristics (softness, hardness, springiness, and gumminess) of muffins were determined by sensory evaluation, as described in section 2.4.2.
2.5. Statistical analysis
Each parameter was attained by using complete randomized design (CRD) and analysis of variance (ANOVA) for all treatments, at significance levels of p ≤ 0.01 and p ≤ 0.05 with Statistix 8.1 software. Duncan’s preference test was used to differentiate between the treatments. Principal Component Analysis was carried out using XLSTAT software, 2022 version.
3. Results and discussion
3.1. Harad varieties characterization
3.1.1. Proximate analysis
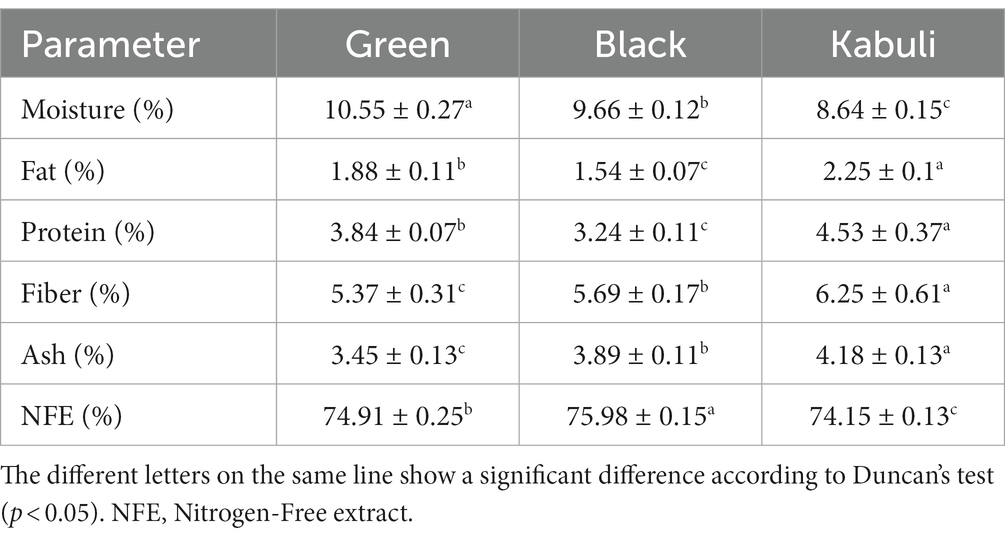
The proximate analysis of three varieties of T. chebula (green, black, and Kabuli) showed significant differences (p < 0.05; Table 2). The moisture content of Kabuli sample was lower (8.64%), while the green variety contained the highest water content (10.55%). The ash content of the Kabuli variety was the highest, 4.18%, with the lowest ash content being attained by the green sample (3.45%).
The highest fat content was recorded by Kabuli sample (2.25%), while the black one had the lowest value (1.54%). The amount of crude protein in T. chebula varieties ranged from 3 to 5%. The fiber content varied from 6.25% in Kabuli to 5.37% in the green variety. The NFE content was the highest in the black sample (75.98%), followed by 74.91% in green T. chebula, and 75.67% in Kabuli variety. Lwin et al. (2020) and Hussain et al. (2009) reported that the moisture, fat, protein, ash, fiber, and carbohydrate of T. chebula was 8.65, 1.45, 3.78, 2.68, 19.33, and 83.43%, respectively. The differences regarding the proximate composition of Harad varieties could be attributed to the phenotypic, genotypic and environmental variations (Navhale et al., 2011).
3.1.2. Mineral analysis
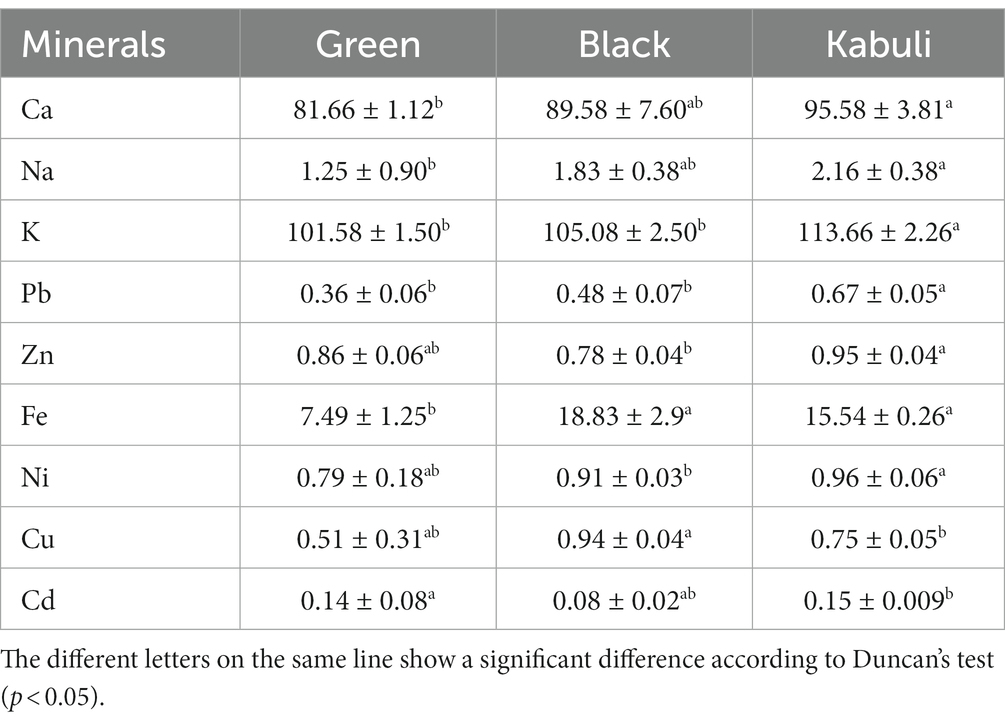
The results revealed that the Kabuli variety of T. chebula contains the highest amount of Ca, Na, and K, namely 95.58, 2.16, and 113.66 mg/kg respectively, followed by the black variety with 89.58, 1.83, and 105.08 mg/kg, respectively (Table 3). The lowest value of Ca, K, and Na was recorded in the green variety, ranging from 81.66, 101.58 to 1.25 mg/kg, respectively. The micro-elements, such as Pb and Zn, were in the highest amounts in Kabuli variety (0.67 and 0.95 mg/kg, respectively). At the same time, black variety contains a higher concentration of Fe, with 18.83 mg/kg, than the other two varieties.
Some trace metals or heavy metals, like Ni and Cd, were higher in Kabuli variety compared to the others, with values of 0.96 mg/kg and 0.15 mg/kg, respectively. In comparison to other varieties, the Cu content was higher (0.94 mg/kg) in black Harad of T. chebula (Table 3). The current concentration is similar to that reported by Hussain et al. (2009). The differences among varieties regarding the minerals content could be due to the plant genetics, location, climate and/or environmental factors (Waheed and Fatima, 2013). Furthermore, the high concentration of some elements could be also attributed to their uptake from the soil and water (Waheed and Fatima, 2013).
3.1.3. Antioxidant potential of Terminalia chebula varieties
3.1.3.1. Total phenolic content
The results of the total phenolic content revealed that the green variety contained a higher value of TPC (79.10 mg GAE/100 g) compared to the other varieties’ extracts in acetone and ethanol. In contrast, Kabuli variety exhibited the lowest value for TPC in ethanol, while the black T. chebula variety exhibited the highest TPC in water and the lowest in hexane and acetone (Table 4). The results showed that for the green variety, the best solvent was ethanol (71.31 mg GAE/100 g), while for the black and Kabuli variety water extraction is recommended since the greatest amount of TPC was obtained. Rekha et al. (2014) and Birur et al. (2015) reported that the range of total phenolic content was 21–107 mg GAE/100 g d.w. in different extract (ethanol, acetone, water, hexane, and methanol). Similarly, Khan et al. (2014) showed that the TPC in green and black varieties was 292.78 and 323.5 mg GAE/100 g, respectively. The efficiency of a certain solvent is driven by the polarity of the phenolic molecules, being known that polyphenols are more soluble in organic solvents because of the hydroxyl groups contained (Hassan Bulbul et al., 2022). The differences compared to the existing literature can be due to the presence of some carbohydrates, terpenoids, or ascorbic acid, or extraction protocol and/or the geographical origin of the plants (Aryal et al., 2019). It has been reported that T. chebula fruit contains 14 phenolic acids derivates such as caffeic acid, digallic acid, ellagic acid, ethyl gallate, eugenol, ferulic acid, gallic acid, melilotic acid, methyl gallate, p-coumaric acid, phloroglucinol, pyragallol, vanillic acid, and 4-O-methylgallic acid (Poudel et al., 2023). The polarity of solvents is a key factor in TPC extraction, being known that the relative polarity of the solvents is ranked as water > ethanol > acetone (Zhu et al., 2020). Thus, compared to the other solvents, acetone would have the worst extraction efficiency of TPC. However, our results do not confirm this hypothesis, a fact that was also supported by other studies and that proved the complex influence not only of the solvent type, but also of the chemical structure and physical properties of the sample (Zhu et al., 2020).
3.1.3.2. Total flavonoids content
The green Harad variety has the highest flavonoid content (34.5 mg QE/100 g) for the acetone extract, while the Kabuli and black varieties showed the highest values (31.35 and 47. 91 mg QE/100 g respectively) for the water extract (Table 4). Among the studied sample, the green one was proven to possess the greatest flavonoid content in ethanol and acetone, while in water the black sample had the most abundant TFC. Vardin and Yilmaz (2018) affirmed that the TF content of T. chebula can provide up to 88 mg QE/g pulp portion (dry weight). The antioxidant properties of the flavonoids which are secondary metabolites from plants depend on the number and position of the free -OH groups, while the variations in flavonoid content depend on genetic factors, and environmental conditions (Aryal et al., 2019). The literature reported that T. chebula contains 6 most important flavonoids like 3,4′-dimethoxy quercetin, 3-methoxy quercetin, isoquercetin, luteolin, quercetin and rutin (Poudel et al., 2023). Distinct extracts obtained with different solvents do not have similar phytochemical profiles, and thus will exhibit different biological activity (Moomin et al., 2023). Apart from the influence of solvent polarity, the extraction yields also depend on the temperature and extraction time (Moomin et al., 2023).
3.1.3.3. DPPH assay
The overall results showed that the free radical scavenging activity of the green variety was higher compared to the other varieties, except for the ethanol extract. The lowest value was recorded in Kabuli variety of T. chebula extracted in water and ethanol (Table 4). For all the studied varieties, the best solvent was proved to be water since the extracts exhibited the greatest antiradical activity. Sheng et al. (2018) and Parveen et al. (2018) also investigated the DPPH value in various extracts of T. chebula, in different solvents like water, hexane, acetone, methanol, ethyl acetate, and ethanol, and reported 77–85% inhibition. Similarly, Shaikh et al. (2016) also studied the DPPH inhibition in water, ethanol, and acetone and obtained 80 to 85% inhibition of chebulic myrobalan plant (T. chebula). The main components responsible for the inhibition of free radicals through hydrogen atoms donation are phenolic compounds and flavonoids (Aryal et al., 2019). The differences between the extraction methods using different solvents could be attributed to the ability of each solvent to better solubilize certain components. For example, the acetone extract of T. chebula had high amounts of hydroxybenzoic acid derivatives, hydroxycinnamic acid derivatives, flavonols, aglycon, and glycosides (Hassan Bulbul et al., 2022). On the other hand, the ethanol extract is rich in total terpenoid content, while the aqueous extract contains mainly phenolic compounds and tannins (Hassan Bulbul et al., 2022).
3.1.3.4. FRAP assay
The current results showed that the green variety contains a higher value for the extracts in ethanol, acetone and hexane compared to other varieties, while the black sample had the best antioxidant activity in water (Table 4). The best solvent for the green sample was acetone (73.41 mmol/100 g), while for the black and Kabuli water was the most effective (113.4 mmol/100 g and 76.33 mmol/100 g respectively). Arya et al. (2013) and Dixit et al. (2013) showed that the FRAP value ranged from 93.97 to 109.6 mmol/100 g, respectively. T. chebula is rich in tannins like gallic acid, ellagic acid, chebulic acid, chebulinic acid, punicalagin, terflavin A, corilagin, galloyl glucose, tannic acid, while among the most abundant flavonoids, quercetin, catechin, and kaempferol can be mentioned (Hassan Bulbul et al., 2022). These compounds contribute to the antioxidant properties of Harad. The differences among varieties regarding the best solvent could be attributed to the differences in the phenolic profile of the samples, being known that each solvent has an affinity for distinct phenolics category (Hassan Bulbul et al., 2022).
3.2. Muffins characterization
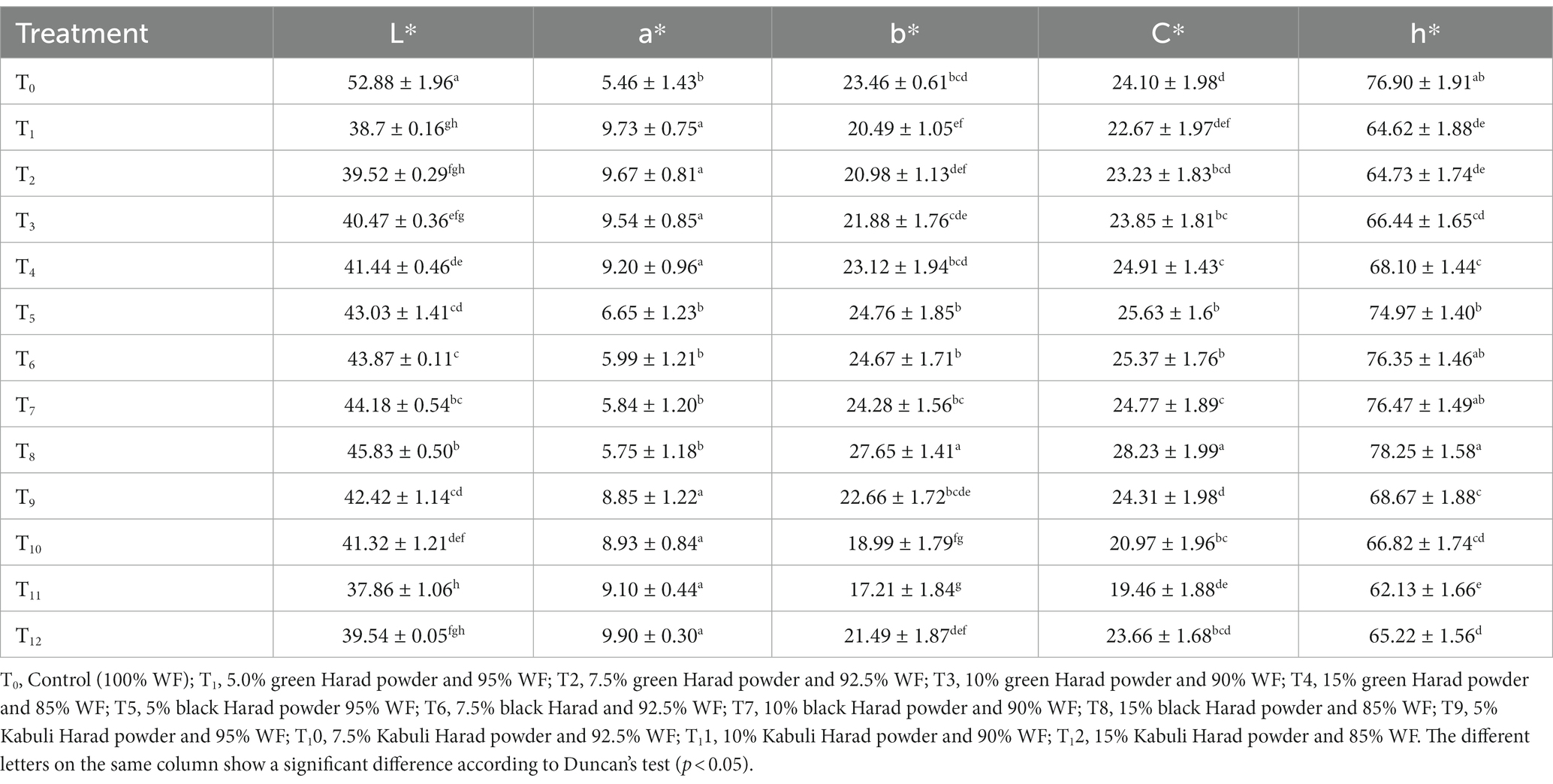
3.2.1. Color analysis of Terminalia chebula-supplemented muffins
The results for color showed that the lightness L* of T. chebula-supplemented muffins ranged from 37.86 to 45.83 (Table 5). The highest lightness value (52.88) was detected in the control and the lowest value in T11 (10% Kabuli T. chebula). The redness value of muffins varied from 5.46 to 9.90. The T0 (control) contains the lowest value of 5.46, while T12 (15% Kabuli T. chebula) showed the highest color tonality 9.90. The yellowness value fluctuated from 17.21 to 27.65 in the samples with different addition levels of the three varieties of T. chebula. A maximum value of 27.65 was recorded in T8 (15% black T. chebula), and the lowest value of 17.21 in T11 (10% Kabuli).
The calculated chroma value of different formulations of T. chebula varied from 19.46 to 28.23. The highest C* value was recorded in T8 (15% black-supplemented muffins) - 28.23, while T11 (10% Kabuli T. chebula) showed the lowest value - 19.46. The h* value of T. chebula-supplemented muffins ranged from 78.25 to 62.13. The highest value was observed in T8 (15% black) - 78.25, and for T11 (10% Kabuli), the lowest value was obtained (62.13). Arifin et al. (2019) and Topkaya and Isik (2019) investigated the color tonality value in supplemented muffins with pumpkin puree and pomegranate peel at different concentrations and reported similar values. The color differences could be due to the intensification of Maillard reactions and/or to the pigments present like carotenoids and gallotannins in T. chebula (Saxena and Saxena, 2004). Other tannins that could have contributed to the color changes are gallic acid, ellagic acid, chebulic acid, chebulinic acid, punicalagin, terflavin A, corilagin, galloyl glucose, tannic acid (Hassan Bulbul et al., 2022). Color changes could be also due to the transformation of anthocyanins to other compounds during baking (De Oliveira et al., 2014).
3.2.2. Sensory textural parameters
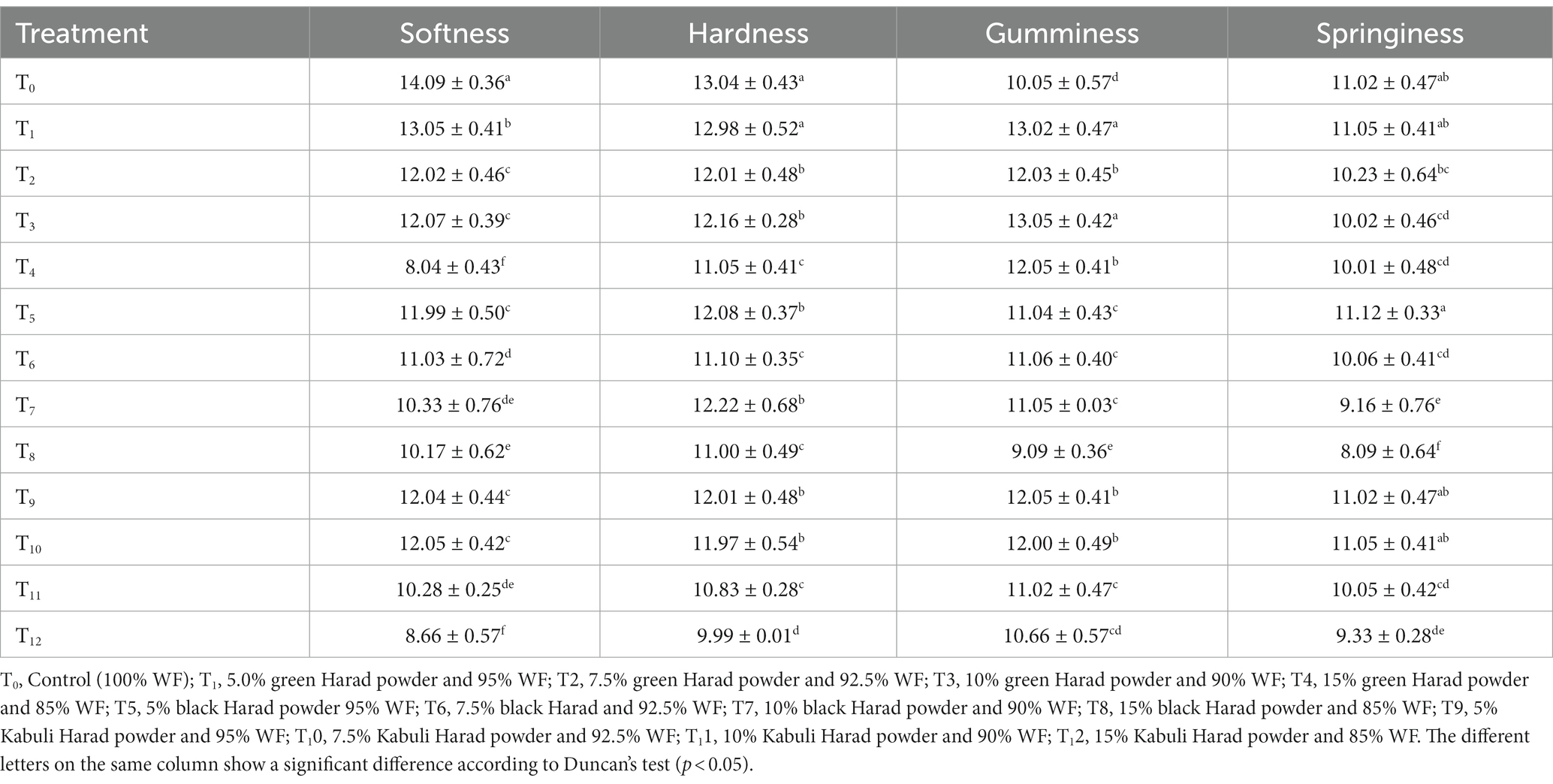
Some textural parameters (softness, hardness, gumminess, and springiness) of T. chebula-supplemented muffins were determined and presented in Table 6. The results of softness and hardness showed significant variations (p < 0.05), the green-containing sample T1 exhibiting the highest value of 13.05 and 12.98 respectively, and the T12 Kabuli sample the lowest (8.66 and 9.99 respectively). Regarding the gumminess, T3 with green variety sample showed the greatest value, while in terms of springiness T5 with 5% black variety presented the best score.
The lowest values for gumminess and springiness were observed in T8 (15% black) (9.09 and 8.09). Topkaya and Isik (2019) and Jeong and Chung (2019) reported that the textural characteristics (springiness, hardness, softness, and gumminess) of pomegranate peel-supplemented muffins and legume-based muffins decreased with the increase of the powder concentration. The changes in the texture profile of muffins could be related to the fiber content of the ingredients added which could be responsible for disturbance of the gluten matrix. Generally, fruit fiber has a great affinity for water and the addition of fruit ingredients in muffins reduces the cake volume and causes a firmer, gummier, and less cohesive final product (Quiles et al., 2018). Another major component of T. chebula that might have influenced the texture of the muffins is sugars. Sugar is responsible for disturbance in gluten matrix development and increases starch gelatinization and protein denaturation temperatures which led to the tenderizing of muffins texture (Quiles et al., 2018).
3.2.3. Sensory evaluation
The sensory profile of muffins indicated significant variations (p < 0.05) among the different amounts of T. chebula varieties supplementation. The data are presented in Table 7. The crust color was dark due to the Maillard reaction, and the results of the Kabuli and green varieties were close (13.28 and 13.07, respectively).
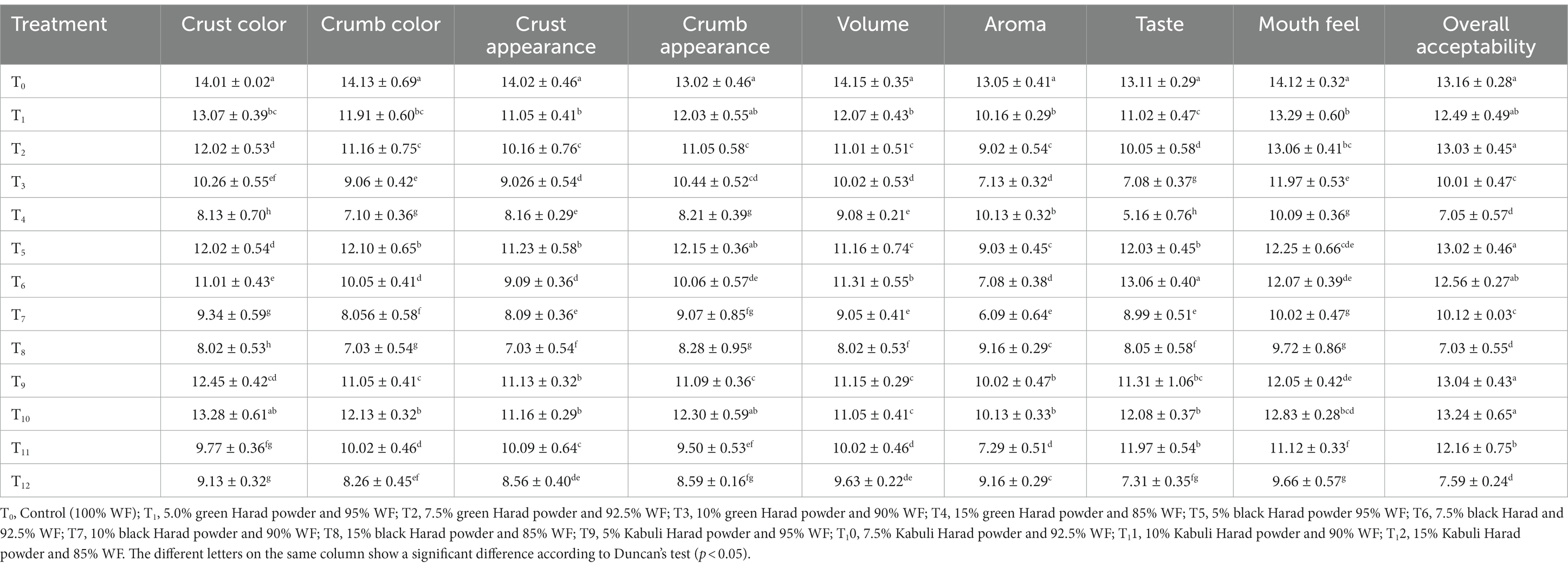
Table 7. Effect of different Harad varieties supplementation on the sensory characteristics of muffins.
The black T8 sample showed the lowest score of crust color (8.02), while the highest value was recorded in the control (14.01) and in T10 (7.5% Kabuli T. chebula) (13.28). The crumb was a small portion of the muffins and represented the inner part just below the crust. The black T8 and green T4 showed close values for crumb color of 7.03 and 7.10, while the highest value was recorded in the control T0 (14.13). The highest score for crust appearance was in T0 (14.02), while T8 (15% black) showed the lowest score (7.03). The greatest value for crumb appearance was observed for T0 (control) (13.02), and the lowest for T4 (15% green T. chebula) (8.21).
The volume and aroma were important parameters in the sensory evaluation. The results of volume showed that among the supplemented muffins, the highest value was observed in green T. chebula containing sample T1 (12.07). In contrast, the lowest value was recorded in black T8 (8.02). The T0 (control) exhibited the highest value for volume and aroma (14.15 and 13.05, respectively). The worst score for aroma was obtained by T7 sample (6.09). The taste characteristics results showed that among the supplemented muffins, the highest score was obtained by black T6 sample (13.06). At the same time, the lowest value was recorded in green T4 sample (5.16). T0 (Control) showed the highest value in both parameters, taste and mouth feel (13.11 and 14.12), respectively. With respect to mouthfeel, the best score was obtained by control T0 (14.12), followed by the T1 sample (13.29), while the smallest score was exhibited by T12 (9.66). The over-acceptability parameter revealed the significant impact of different T. chebula varieties, showing that the Kabuli-containing muffins obtained better scores compared to the black variety. The lowest value was observed for T8 (15% black) (7.03), while the highest value was obtained for T10 (7.5% Kabuli) (13.24). Shabeer et al. (2016) and Arya et al. (2013) conducted a study in which mango kernel, casein, and egg white were added to cookies to investigate the sensory profile. The authors reported significant variations in color, and a decrease in flavor and overall acceptability scores, changes that were related to the Maillard reactions and to the presence of high amounts of fiber, ash, and low protein level of the ingredient added. The taste, aroma, and consequently general acceptability of muffins supplemented with T. chebula could have been influenced by the presence of polyphenols. There are some phenolic compounds that give a bitter taste even in very small concentrations (De Oliveira et al., 2014). The sweet and bitter tastes are mutually suppressed in blends, while the astringency and bitterness can be considered negative features by the panelists (De Oliveira et al., 2014). Thus, the decrease in sensory scores of muffins could be attributed to the intake of phenolic compounds from T. chebula.
3.3. Relationships between characteristics and samples
3.3.1. Harad varieties characteristics
The relationships between the evaluated characteristics of Harad varieties (Figure 1) and supplemented muffins (Figure 2) were evaluated through Principal Component Analysis. For Harad varieties characteristics, the first principal component (PC1) explained 58.35% of data variation, while the second explained 41.63% of the total variance (Figure 1). Green and Kabuli varieties were associated with PC1, while the Black variety was associated with PC2. The contents of Ni, Na, Ca, Pb, K, Fiber, Ash, DPPH in all solvents, TPC and TFC in ethanol and acetone were associated with PC1, while Cd, Zn, protein, fat, TPC, FRAP, and TFC in hexane and water, NFE and DPPH in ethanol were associated to PC2. TPC, TFC, and FRAP in ethanol are opposed to K, fiber, and Pb content, while TPC, TFC, FRAP, and DPPH in acetone are placed in opposition to Fe, Cu, FRAP, opposed to Fe, Cu, FRAP, respectively TPC and TFC in water. The Zn, TPC in hexane, protein, and fat variables oppose DPPH ethanol and NFE (Figure 1). Moisture was positively correlated with TPC and TFC in ethanol (p < 0.05, 0.59 < r < 0.91), while ash content was strongly correlated with Ca and Na (p < 0.05, r = 0.99). Some strong correlations were observed between some TPC and TFC and DPPH and FRAP variables (p < 0.05). Fe content was negatively correlated with TPC acetone (p < 0.05, r = 0.99). At the same time, Zn, fat, and protein were strongly positively correlated with TPC hexane (p < 0.05, r = 0.99) and negatively with DPPH ethanol (p < 0.05, r = 0.99). The literature also reported significant correlations between the TPC and TFC with the antiradical activity of plants (Aryal et al., 2019).
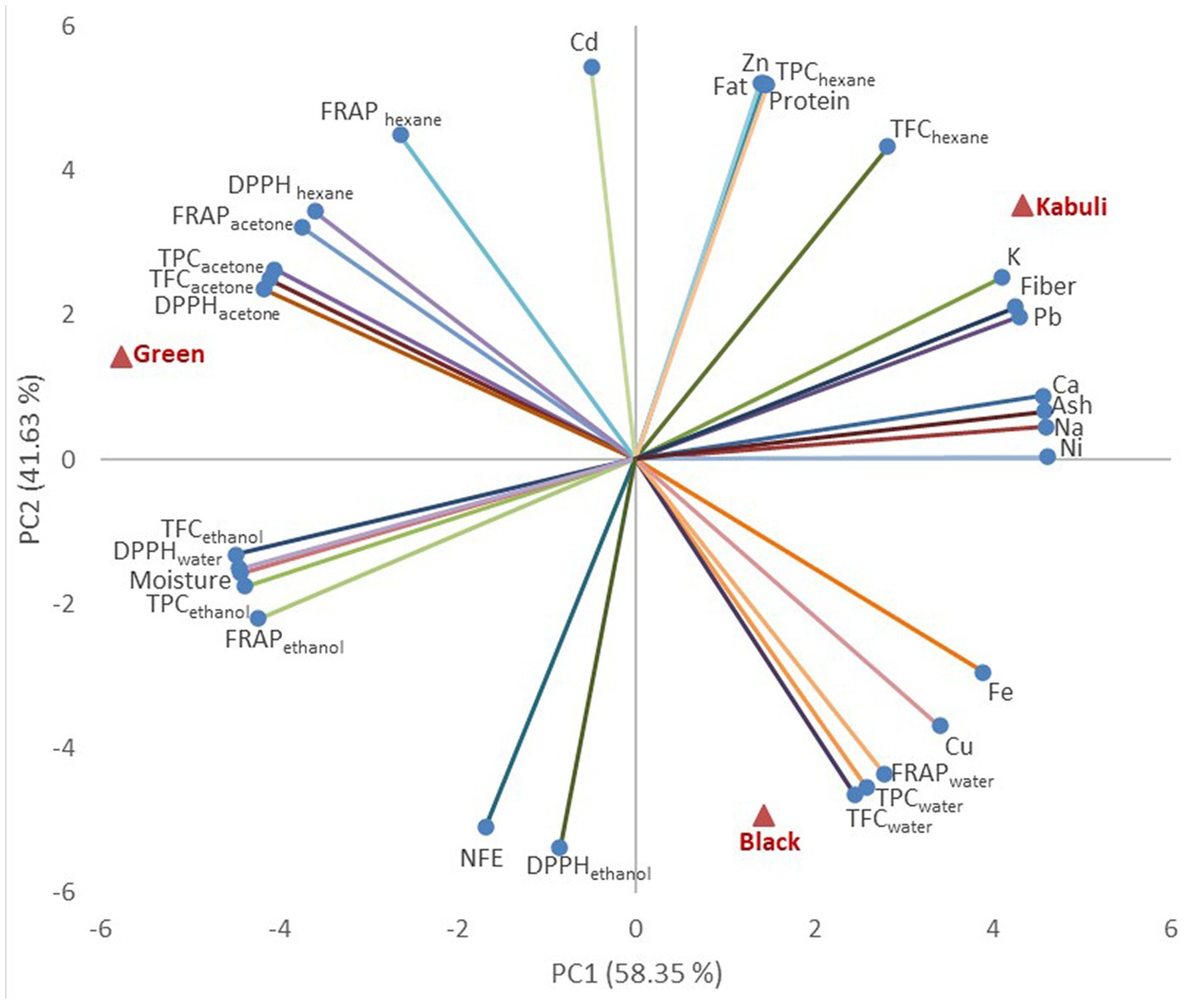
Figure 1. Principal Component Analysis bi-plot for the Harad varieties (red triangles) and the evaluated variables (blue bullets): NFE-nitrogen free extract, TPC-total phenolic content, TFC-total flavonoid content, FRAP- ferric reducing antioxidant power assay, DPPH- 2,2-diphenylpicrylhydrazyl assay.
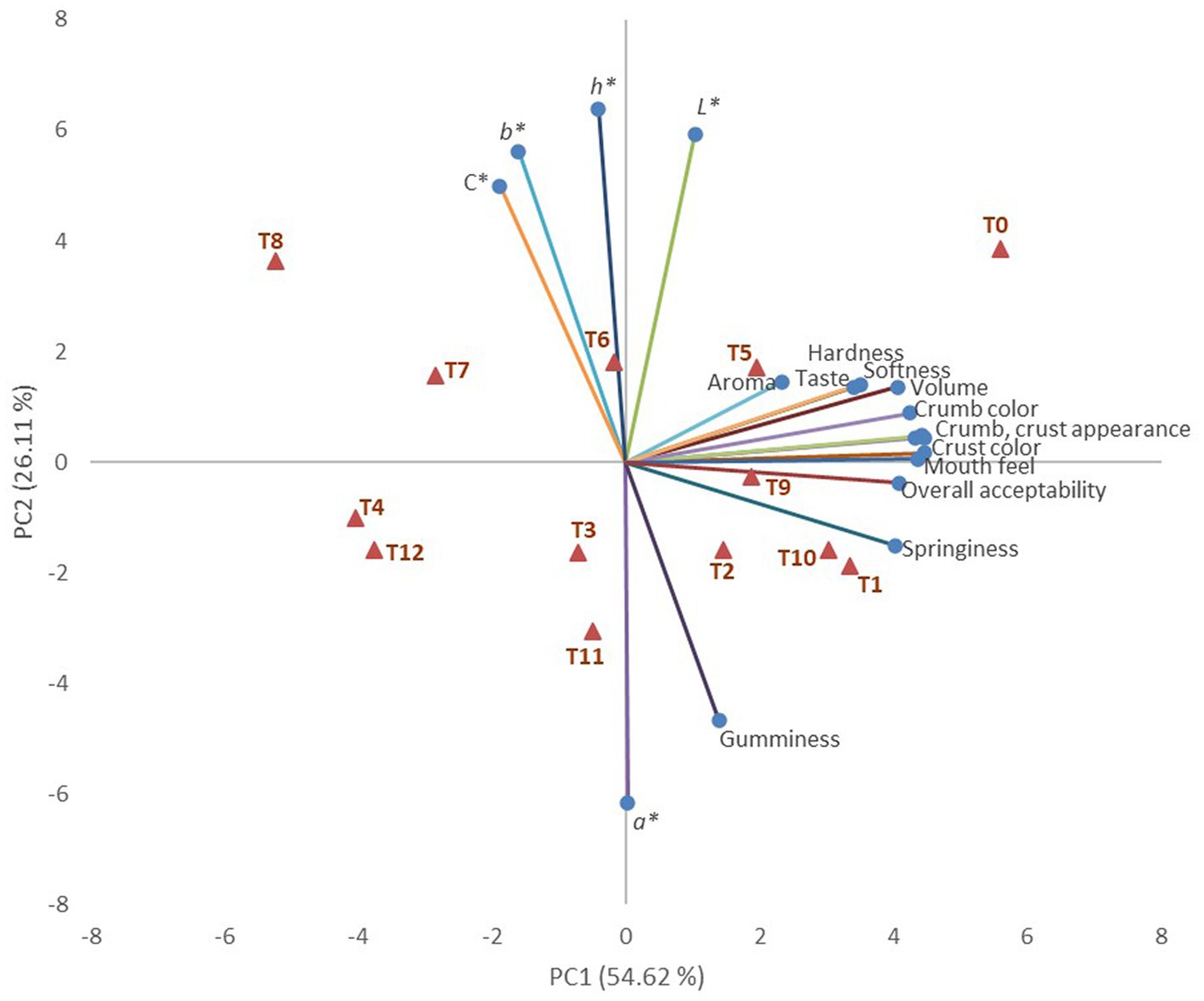
Figure 2. Principal Component Analysis bi-plot for the enriched muffins (red triangles) and the studied variables (blue bullets): T0-Control (100% WF), T1-5.0% green Harad powder and 95% WF, T2-7.5% green Harad powder and 92.5% WF, T3-10% green Harad powder and 90% WF, T4-15% green Harad powder and 85% WF, T5-5% black Harad powder 95% WF, T6-7.5% black Harad and 92.5% WF, T7:10% black Harad powder and 90% WF, T8:15% black Harad powder and 85% WF, T9:5% Kabuli Harad powder and 95% WF, T10-7.5% Kabuli Harad powder and 92.5% WF, T11-10% Kabuli Harad powder and 90% WF, T12-15% Kabuli Harad powder and 85% WF.
3.3.2. Muffins characteristics
Data variability regarding muffin samples and their characteristics was explained 54.62% by PC1 and 26.11% by PC2. The sensory and texture properties, except gumminess, were associated with PC1, while the color parameters and gumminess were associated with PC2. The muffins containing the black Harad variety were placed in the upper part of the graphic, while those containing green and Kabuli Harad were positioned in the lower part of the plot (Figure 2).
The control sample was differentiated from the others, being placed in the right positive quadrant. Softness, hardness, and springiness were positively correlated (p < 0.05, 0.61 < r < 0.91) with the sensory characteristics of crust and crumb (color and appearance), volume mouth feel, and overall acceptability. Softness and springiness were positively correlated with taste (p < 0.05, r = 0.59, and r = 0.69, respectively). Positive correlations were also found between crust or crumb color or appearance and overall acceptability, mouth feels, taste, and volume (p < 0.05, 0.72 < r < 0.94).
4. Conclusion
It is concluded that the Kabuli Harad and green varieties have higher proximate and mineral content than the black variety. The black Harad contained a higher antioxidant potential. The color tonality of the supplemented muffins showed that the black variety had the highest value compared to the other varieties at different levels. In the case of product development, the 7.5% level for each of the three varieties (Kabuli, black, and green) exhibited good acceptability of muffins. Considering these results, the present study has confirmed the feasibility of supplementing white flour with different Terminalia chebula varieties to develop functional muffins. Further investigation of the effects of Terminalia chebula on the rheological properties of the batter, final product microstructure, and consumption benefits is needed.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding authors.
Author contributions
MTS and EK: conceptualization and investigation. WK: methodology, software, and data curation. WK and MZK: validation. SRS and MAR: formal analysis. IC: resources. EK and MU-I: writing-original draft preparation. CM, MU-I, and IC: writing-review and editing. MI-L, AB, and CM: visualization. SM: supervision, project administration, and funding acquisition. All authors contributed to the article and approved the submitted version.
Funding
This work was funded by the Ministry of Research, Innovation, and Digitalization within Program 1–Development of national research and development system, Subprogram 1.2–Institutional Performance–RDI excellence funding projects, under contract No. 10PFE/2021.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer TT declared a shared affiliation with the author WK to the handling editor at the time of review.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Aher, V., and Wahi, A. (2011). Immunomodulatory activity of alcohol extract of Terminalia chebula retz combretaceae. Trop. J. Pharm. Res. 10, 567–575. doi: 10.4314/tjpr.v10i5.5
AOAC. (2016). Official methods of analysis of AOAC international. Rockville, MD: AOAC International.
Arifin, N., Izyan, S. N., and Huda-Faujan, N. (2019). Physical properties and consumer acceptability of basic muffin made from pumpkin puree as butter replacer. Food Res. 3, 840–845. doi: 10.26656/fr.2017.3(6).090
Arya, A., Looi, C. Y., Wong, W. F., Noordin, M. I., Nyamathulla, S., Mustafa, M. R., et al. (2013). In vitro antioxidant, PTP-1B inhibitory effects and in vivo hypoglycemic potential of selected medicinal plants. Int. J. Pharmacol. 9, 50–57. doi: 10.3923/ijp.2013.50.57
Aryal, S., Baniya, M. K., Danekhu, K., Kunwar, P., Gurung, R., and Koirala, N. (2019). Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from western Nepal. Plan. Theory 8:96. doi: 10.3390/plants8040096
Birur, R. S., Ramachandra, Y. L., Subaramaihha, S. R., Subbaiah, S. G. P., Austin, R. S., and Dhananjaya, B. L. (2015). Antioxidant activities of leaf galls extracts of Terminalia chebula (Gaertn.) Retz. (Combretaceae). Acta Sci. Pol. Technol. Aliment. 14, 97–105. doi: 10.17306/J.AFS.2015.2.11
Brand-Williams, W., Cuvelier, M.-E., and Berset, C. (1995). Use of a free radical method to evaluate antioxidant activity. LWT 28, 25–30. doi: 10.1016/S0023-6438(95)80008-5
Chattopadhyay, R. R., and Bhattacharyya, S. K. (2007). PHCOG REV: plant review. Terminalia chebula: An update. Pharmacogn. Rev. 1, 151–157.
De Oliveira, L. D. L., De Carvalho, M. V., and Melo, L. (2014). Health promoting and sensory properties of phenolic compounds in food. Rev Ceres. 61, 764–779. doi: 10.1590/0034-737X201461000002
Dewanto, V., Wu, X., Adom, K. K., and Liu, R. H. (2002). Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 50, 3010–3014. doi: 10.1021/jf0115589
Dixit, D., Dixit, A. K., Lad, H., Gupta, D., and Bhatnagar, D. (2013). Radioprotective effect of Terminalia chebula Retzius extract against γ-irradiation-induced oxidative stress. Biomed. Aging Pathol. 3, 83–88. doi: 10.1016/j.biomag.2012.10.008
Gaire, B. P., and Kim, H. (2014). Neuroprotective effects of Fructus Chebulae extracts on experimental models of cerebral ischemia. J. Tradit. Chin. Med. 34, 69–75. doi: 10.1016/S0254-6272(14)60057-1
Gouveia, L., Batista, A. P., Miranda, A., Empis, J., and Raymundo, A. (2007). Chlorella vulgaris biomass used as colouring source in traditional butter cookies. Innov. Food Sci. Emerg. Technol. 8, 433–436. doi: 10.1016/j.ifset.2007.03.026
Han, Q., Song, J., Qiao, C., Wong, L., and Xu, H. (2006). Preparative isolation of hydrolysable tannins chebulagic acid and chebulinic acid from Terminalia chebula by high-speed counter-current chromatography. J. Sep. Sci. 29, 1653–1657. doi: 10.1002/jssc.200600089
Hassan Bulbul, M. R., Uddin Chowdhury, M. N., Naima, T. A., Sami, S. A., Imtiaj, M. S., Huda, N., et al. (2022). A comprehensive review on the diverse pharmacological perspectives of Terminalia chebula Retz. Heliyon. 8:e10220. doi: 10.1016/j.heliyon.2022.e10220
Hussain, J., Khan, A. L., Rehman, N., Hamayun, M., Shinwari, Z. K., Ullah, W., et al. (2009). Assessment of herbal products and their composite medicinal plants through proximate and micronutrients analyses. J. Med. Pl. Res 3, 1072–1077.
Jagetia, G. C., Baliga, M. S., Malagi, K. J., and Kamath, M. S. (2002). The evaluation of the radioprotective effect of Triphala (an ayurvedic rejuvenating drug) in the mice exposed to γ-radiation. Phytomedicine 9, 99–108. doi: 10.1078/0944-7113-00095
Jeong, D., and Chung, H.-J. (2019). Physical, textural and sensory characteristics of legume-based gluten-free muffin enriched with waxy rice flour. Food Sci. Biotechnol. 28, 87–97. doi: 10.1007/s10068-018-0444-8
Juang, L., Sheu, S., and Lin, T. (2004). Determination of hydrolyzable tannins in the fruit of Terminalia chebula Retz. By high-performance liquid chromatography and capillary electrophoresis. J. Sep. Sci. 27, 718–724. doi: 10.1002/jssc.200401741
Kannan, P., Ramadevi, S. R., and Hopper, W. (2009). Antibacterial activity of Terminalia chebula fruit extract. African J. Microbiol. Res. 3, 180–184.
Kaur, S., and Jaggi, R. K. (2010). Antinociceptive activity of chronic administration of different extracts of Terminalia bellerica Roxb. and Terminalia chebula Retz. fruits. Indian J. Exp. Biol. 48, 925–930.
Khan, A., Nazar, H., Sabir, S. M., Irshad, M., Awan, S. I., Abbas, R., et al. (2014). Antioxidant activity and inhibitory effect of some commonly used medicinal plants against lipid per-oxidation in mice brain. Afr. J. Tradit. Complement. Altern. Med. 11, 83–90. doi: 10.4314/ajtcam.v11i5.14
Kim, T. G., Kang, S. Y., Jung, K. K., Kang, J. H., Lee, E., Han, H. M., et al. (2001). Antiviral activities of extracts isolated from Terminalis chebula Retz., Sanguisorba officinalis L., Rubus coreanus Miq. and Rheum palmatum L. against hepatitis B virus. Phyther. Res. 15, 718–720. doi: 10.1002/ptr.832
Kumar, K. J. (2006). Effect of geographical variation on contents of tannic acid, gallic acid, chebulinic acid and ethyl gallate in Terminalia chebula. Nat. Prod. 2, 170–175.
Lee, H.-S., Koo, Y.-C., Suh, H. J., Kim, K.-Y., and Lee, K.-W. (2010). Preventive effects of chebulic acid isolated from Terminalia chebula on advanced glycation endproduct-induced endothelial cell dysfunction. J. Ethnopharmacol. 131, 567–574. doi: 10.1016/j.jep.2010.07.039
Lwin, W. W., Myint, C. Y., M., Maung, M., and Myint, K. T. Y. (2020). Formulation of capsule dosage form containing Ethanolic fruit extract of Terminalia chebula Retz. (Hpan-ga) having potent antioxidant activity. Myanmar Health Sci. Res. J. 32, 66–72.
Mahmood, S. (2019). A critical review on pharmaceutical and medicinal importance of ginger. Acta Sci. Nutr. Health 3, 78–82.
Majed, F., Nafees, S., Rashid, S., Ali, N., Hasan, S. K., Ali, R., et al. (2015). Terminalia chebula attenuates DMBA/croton oil-induced oxidative stress and inflammation in Swiss albino mouse skin. Toxicol. Int. 22, 21–29. doi: 10.4103/0971-6580.172252
Maruthappan, V., and Shree, K. S. (2010). Hypolipidemic activity of Haritaki (Terminalia chebula) in atherogenic diet induced hyperlipidemic rats. J. Adv. Pharm. Technol. Res. 1, 229–235.
Meilgaard, M. C., Carr, B. T., and Civille, G. V. (1999). Sensory evaluation techniques. Boca Raton, FL: CRC Press.
Moomin, A., Russell, W. R., Knott, R. M., Scobbie, L., Mensah, K. B., Adu-Gyamfi, P. K. T., et al. (2023). Season, storage and extraction method impact on the phytochemical profile of Terminalia ivorensis. BMC Plant Biol. 23, 162–118. doi: 10.1186/s12870-023-04144-8
Nadeem, M., Tehreem, S., Ranjha, M. M. A., Ahmad, A., Din, A., Ud Din, G. M., et al. (2022). Probing of ultrasonic assisted pasteurization (UAP) effects on physicochemical profile and storage stability of jambul (Syzygium cumini L.) squash. Int. J. Food Prop. 25, 661–672. doi: 10.1080/10942912.2022.2058532
Navhale, V. C., Sonone, N. G., Jangam, P. S., Jadhav, S. T., and Bhave, S. G. (2011). Genetic variability and selection of candidate plus trees in chebulic myrobalan (Terminalia chebula retz.). Electron J Plant Breed. 2, 157–163.
Panda, S. K., Mohanta, Y. K., Padhi, L., and Luyten, W. (2019). Antimicrobial activity of select edible plants from Odisha, India against food-borne pathogens. LWT 113:108246. doi: 10.1016/j.lwt.2019.06.013
Parveen, R., Shamsi, T. N., Singh, G., Athar, T., and Fatima, S. (2018). Phytochemical analysis and in-vitro biochemical characterization of aqueous and methanolic extract of Triphala, a conventional herbal remedy. Biotechnol. Rep. 17, 126–136. doi: 10.1016/j.btre.2018.02.003
Patel Madhavi, G., Patel Vishal, R., and Patel Rakesh, K. (2010). Development and validation of improved RP-HPLC method for identification and estimation of ellagic and gallic acid in Triphala churna. Int. J. ChemTech Res. 2, 1486–1493.
Poudel, P., Thapa, R., Lamichhane, S., Paudel, H. R., and Devkota, H. P. (2023). Terminalia chebula Retz.. Amsterdam, Netherlands: Elsevier Ltd.
Prakash, A., Sharma, S. K., Mohapatra, P. K., Bhattacharjee, K., Gogoi, K., Gogoi, P., et al. (2013). In vitro and in vivo antiplasmodial activity of the root extracts of Brucea mollis wall. Ex Kurz. Parasitol. Res. 112, 637–642. doi: 10.1007/s00436-012-3178-1
Quiles, A., Campbell, G. M., Struck, S., Rohm, H., and Hernando, I. (2018). Fiber from fruit pomace: a review of applications in cereal-based products. Food Rev. Int. 34, 162–181. doi: 10.1080/87559129.2016.1261299
Ranjha, M. M. A., Amjad, S., Ashraf, S., Khawar, L., Safdar, M. N., Jabbar, S., et al. (2020). Extraction of polyphenols from apple and pomegranate peels employing different extraction techniques for the development of functional date bars. Int. J. Fruit Sci. 20, S1201–S1221. doi: 10.1080/15538362.2020.1782804
Ranjha, M. M. A. N., Irfan, S., Lorenzo, J. M., Shafique, B., Kanwal, R., Pateiro, M., et al. (2021). Sonication, a potential technique for extraction of phytoconstituents: a systematic review. PRO 9:1406. doi: 10.3390/pr9081406
Rao, N. K., and Nammi, S. (2006). Antidiabetic and renoprotective effects of the chloroform extract of Terminalia chebula Retz. Seeds in streptozotocin-induced diabetic rats. BMC Complement. Altern. Med. 6, 1–6. doi: 10.1186/1472-6882-6-17
Reddy, D. B., Reddy, T. C. M., Jyotsna, G., Sharan, S., Priya, N., Lakshmipathi, V., et al. (2009). Chebulagic acid, a COX–LOX dual inhibitor isolated from the fruits of Terminalia chebula Retz., induces apoptosis in COLO-205 cell line. J. Ethnopharmacol. 124, 506–512. doi: 10.1016/j.jep.2009.05.022
Rekha, V., Vijayalakshmi, D., Kumar, N., and Manipal, S. (2014). Anti cariogenic effect of Terminalia chebula. J. Clin. diagnostic Res. 8:ZC51. doi: 10.7860/JCDR/2014/9844.4765
Sabtain, B., Farooq, R., Shafique, B., Modassar, M., and Ranjha, A. N. (2021). A narrative review on the phytochemistry, nutritional profile and properties of prickly pear fruit. Open Access J. Biog. Sci. Res 7:7. doi: 10.46718/JBGSR.2021.07.000164
Sadeghnia, H. R., Jamshidi, R., Afshari, A. R., Mollazadeh, H., Forouzanfar, F., and Rakhshandeh, H. (2017). Terminalia chebula attenuates quinolinate-induced oxidative PC12 and OLN-93 cell death. Mult. Scler. Relat. Disord. 14, 60–67. doi: 10.1016/j.msard.2017.03.012
Sahab, A., and Sabbour, M. M. (2011). Virulence of four Entomopathogenic Fungi on some cotton pests with special reference to impact of some pesticides, nutritional and environmental factors on fungal growth. Egypt. J. Biol. pest Control 21, 61–67.
Saleem, A., Husheem, M., Härkönen, P., and Pihlaja, K. (2002). Inhibition of cancer cell growth by crude extract and the phenolics of Terminalia chebula retz. Fruit. J. Ethnopharmacol. 81, 327–336. doi: 10.1016/S0378-8741(02)00099-5
Saxena, S., and Saxena, R. K. (2004). Statistical optimization of tannase production from Penicillium variable using fruits (chebulic myrobalan) of Terminalia chebula. Biotechnol. Appl. Biochem. 39, 99–106. doi: 10.1042/ba20030097
Seo, J. B., Jeong, J.-Y., Park, J. Y., Jun, E. M., Lee, S.-I., Choe, S. S., et al. (2012). Anti-arthritic and analgesic effect of NDI10218, a standardized extract of Terminalia chebula, on arthritis and pain model. Biomol. Ther. (Seoul). 20, 104–112. doi: 10.4062/biomolther.2012.20.1.104
Shabeer, M., Sultan, M. T., Abrar, M., Suffyan Saddique, M., Imran, M., Saad Hashmi, M., et al. (2016). Utilization of defatted mango kernel in wheat-based cereals products: nutritional and functional properties. Int. J. Fruit Sci. 16, 444–460. doi: 10.1080/15538362.2016.1166094
Shaikh, R. U., Pund, M. M., and Gacche, R. N. (2016). Evaluation of anti-inflammatory activity of selected medicinal plants used in Indian traditional medication system in vitro as well as in vivo. J. Tradit. Complement. Med. 6, 355–361. doi: 10.1016/j.jtcme.2015.07.001
Sharma, P., Prakash, T., Kotresha, D., Ansari, M. A., Sahrm, U. R., Kumar, B., et al. (2011). Antiulcerogenic activity of Terminalia chebula fruit in experimentally induced ulcer in rats. Pharm. Biol. 49, 262–268. doi: 10.3109/13880209.2010.503709
Shehzadi, K., Rubab, Q., Asad, L., Ishfaq, M., Shafique, B., Ranjha, M. M. A., et al. (2020). A critical review on presence of polyphenols in commercial varieties of apple peel, their extraction and health benefits. Open Access J. Biog. Sci. Res 6:18. doi: 10.46718/JBGSR.2020.06.000141
Sheng, Z., Zhao, J., Muhammad, I., and Zhang, Y. (2018). Optimization of total phenolic content from Terminalia chebula Retz. Fruits using response surface methodology and evaluation of their antioxidant activities. PLoS One 13:e0202368. doi: 10.1371/journal.pone.0202368
Shreedevi, M. S., and Sampathkumar, B. (2011). Effect of siddha herbal paste on wound healing in induced Derman wounds in rat. Int. J. Ayurvedic Herb. Med. 1, 86–91.
Singleton, V. L., Orthofer, R., and Lamuela-Raventós, R. M. (1999). “analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent” in Methods in enzymology. eds. J. D. Everette and Q. M. Bryant, vol. 299 (Amsterdam, Netherlands: Elsevier), 152–178.
Srivastav, A., Chandra, A., Singh, M., Jamal, F., Rastogi, P., Rajendran, S. M., et al. (2010). Inhibition of hyaluronidase activity of human and rat spermatozoa in vitro and antispermatogenic activity in rats in vivo by Terminalia chebula, a flavonoid rich plant. Reprod. Toxicol. 29, 214–224. doi: 10.1016/j.reprotox.2009.11.001
Suchalatha, S., and Devi, C. S. S. (2005). Antioxidant activity of ethanolic extract of Terminalia chebula fruit against isoproterenol-induced oxidative stress in rats. Indian J. Biochem. Biophys. 42, 246–249.
Topkaya, C., and Isik, F. (2019). Effects of pomegranate peel supplementation on chemical, physical, and nutritional properties of muffin cakes. J. Food Process. Preserv. 43:e13868. doi: 10.1111/jfpp.13868
Vardin, H., and Yilmaz, F. M. (2018). The effect of blanching pre-treatment on the drying kinetics, thermal degradation of phenolic compounds and hydroxymethyl furfural formation in pomegranate arils. Ital. J. food Sci. 30:947. doi: 10.14674/IJFS-947
Vidya, S., Verma, E., Kumar, J. S., Anilkumar, V. T., and Ramesh, A. (2011). Hepato-protective activity of Terminalia chebula leaves in paracetamol induced hepatotoxicity in rats. Int. J. Adv. Pharm. Res. 2, 127–132. doi: 10.1016/S1995-7645(11)60127-2
Waheed, S., and Fatima, I. (2013). Instrumental neutron activation analysis of Emblica officinalis, Terminalia belerica and Terminalia chebula for trace element efficacy and safety. Appl. Radiat. Isot. 77, 139–144. doi: 10.1016/j.apradiso.2013.03.007
Zahin, M., Aqil, F., and Ahmad, I. (2010). Broad spectrum antimutagenic activity of antioxidant active fraction of Punica granatum L. peel extracts. Mutat. Res. Toxicol. Environ. Mutagen. 703, 99–107. doi: 10.1016/j.mrgentox.2010.08.001
Zhu, H., Zhang, J., Li, C., Liu, S., and Wang, L. (2020). Morinda citrifolia L. leaves extracts obtained by traditional and eco-friendly extraction solvents: relation between phenolic compositions and biological properties by multivariate analysis. Ind. Crops Prod. 153:112586. doi: 10.1016/j.indcrop.2020.112586
Keywords: Harad, antioxidant potential, medicinal plant, wheat muffins, Principal Component Analysis
Citation: Khalil E, Sultan MT, Khalid W, Khalid MZ, Rahim MA, Saleem SR, Luca M-I, Mironeasa C, Batariuc A, Ungureanu-Iuga M, Coţovanu I and Mironeasa S (2023) Evaluation of different Terminalia chebula varieties and development of functional muffins. Front. Sustain. Food Syst. 7:1227851. doi: 10.3389/fsufs.2023.1227851
Edited by:
Muhammad Faisal Manzoor, Foshan University, ChinaReviewed by:
Tabussam Tufail, University of Lahore, PakistanSeydi Yıkmış, Namik Kemal University, Türkiye
Sonia Morya, Lovely Professional University, India
Copyright © 2023 Khalil, Sultan, Khalid, Khalid, Rahim, Saleem, Luca, Mironeasa, Batariuc, Ungureanu-Iuga, Coţovanu and Mironeasa. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Muhammad Tauseef Sultan, dGF1c2VlZnN1bHRhbkBienUuZWR1LnBr; Waseem Khalid, d2FzZWVtLmtoYWxpZEB1aWZzdC51b2wuZWR1LnBr; Silvia Mironeasa, c2lsdmlhbUBmaWEudXN2LnJv; Mădălina Ungureanu-Iuga, bWFkYWxpbmEuaXVnYUB1c20ucm8=
 Ejaz Khalil1
Ejaz Khalil1 Waseem Khalid
Waseem Khalid Muhammad Zubair Khalid
Muhammad Zubair Khalid Muhammad Abdul Rahim
Muhammad Abdul Rahim Samavia Rashid Saleem
Samavia Rashid Saleem Ana Batariuc
Ana Batariuc Mădălina Ungureanu-Iuga
Mădălina Ungureanu-Iuga Ionica Coţovanu
Ionica Coţovanu Silvia Mironeasa
Silvia Mironeasa