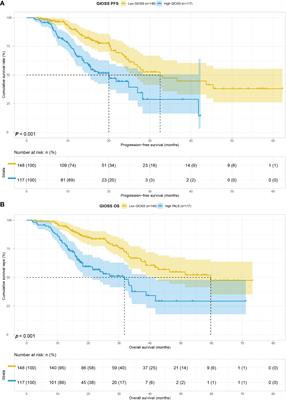
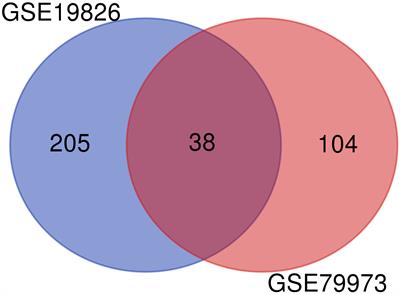
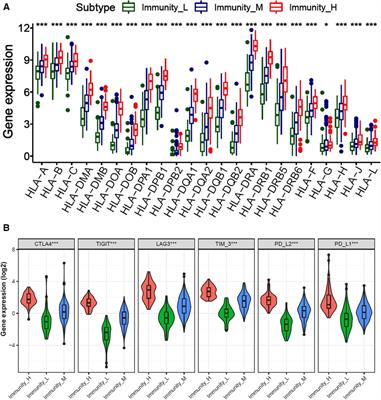
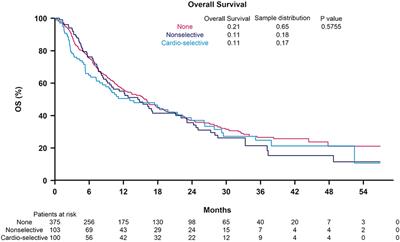
ORIGINAL RESEARCH
Published on 08 Sep 2023
Prediction of prognosis, immune infiltration, and personalized treatment of hepatocellular carcinoma by analysis of cuproptosis-related long noncoding RNAs and verification in vitro
doi 10.3389/fonc.2023.1159126
- 1,534 views
- 4 citations