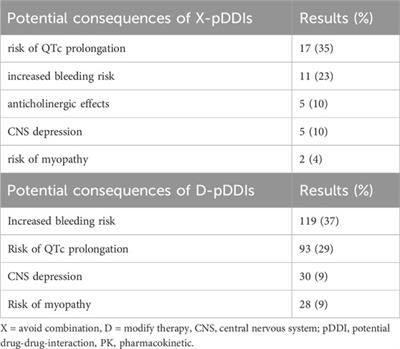
Review
Published on 06 Nov 2024
Chemical adherence testing in the clinical management of hypertension: a scoping review
in Drugs Outcomes Research and Policies

- 698 views
Review
Published on 06 Nov 2024
in Drugs Outcomes Research and Policies

Original Research
Published on 05 Nov 2024
in Drugs Outcomes Research and Policies
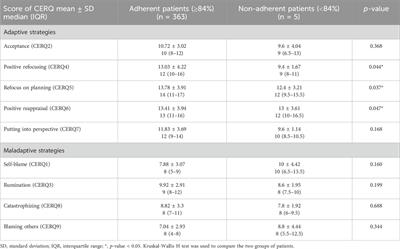
Editorial
Published on 05 Nov 2024
in Drugs Outcomes Research and Policies
Original Research
Published on 05 Nov 2024
in Drugs Outcomes Research and Policies

Correction
Published on 31 Oct 2024
in Drugs Outcomes Research and Policies
Review
Published on 31 Oct 2024
in Drugs Outcomes Research and Policies

Original Research
Published on 28 Oct 2024
in Drugs Outcomes Research and Policies

Original Research
Published on 25 Oct 2024
in Drugs Outcomes Research and Policies

Brief Research Report
Published on 25 Oct 2024
in Drugs Outcomes Research and Policies

Original Research
Published on 24 Oct 2024
in Drugs Outcomes Research and Policies

Original Research
Published on 23 Oct 2024
in Drugs Outcomes Research and Policies
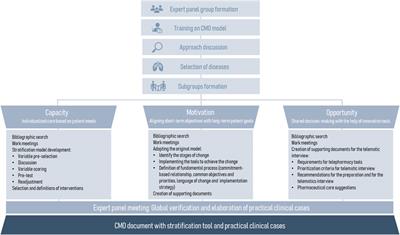
Original Research
Published on 21 Oct 2024
in Drugs Outcomes Research and Policies

Correction
Published on 17 Oct 2024
in Drugs Outcomes Research and Policies
Clinical Trial
Published on 17 Oct 2024
in Drugs Outcomes Research and Policies

Original Research
Published on 15 Oct 2024
in Drugs Outcomes Research and Policies
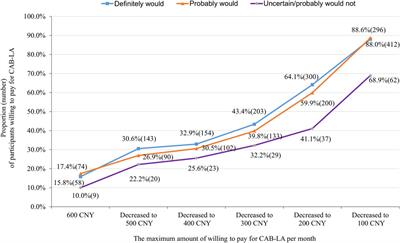
Original Research
Published on 15 Oct 2024
in Drugs Outcomes Research and Policies