
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Nutr. , 12 March 2025
Sec. Clinical Nutrition
Volume 12 - 2025 | https://doi.org/10.3389/fnut.2025.1547655
Background: Stroke-associated pneumonia (SAP) is a common and serious complication in patients with spontaneous intracerebral hemorrhage (SICH), contributing to prolonged hospital stays and poor outcomes. Nutritional status has been linked to the development of SAP in patients with ischemic stroke, but its role in SICH patients remains understudied. This study aims to evaluate the predictive value of the Nutritional Risk Screening-2002 (NRS-2002) score for SAP in SICH patients and to compare it with other nutritional assessment tools.
Methods: This retrospective observational study included 404 consecutive SICH patients admitted to Dongyang People’s Hospital from January 2023 to May 2024. Nutritional risk was assessed using the NRS-2002 score upon admission, and SAP was diagnosed within the first 7 days of hospitalization. Univariate and multivariate logistic regression analyses identified risk factors for SAP, and receiver operating characteristic (ROC) curves were used to compare the predictive accuracy of the NRS-2002, Controlling Nutritional Status (CONUT) score, and Prognostic Nutritional Index (PNI) for SAP.
Results: Among the 404 patients, 97 developed SAP. A higher NRS-2002 score was significantly associated with an increased risk of SAP (OR: 1.575, 95% CI: 1.134–2.186, p = 0.007). ROC analysis showed that the NRS-2002 score (AUC: 0.768, 95% CI: 0.716–0.820) outperformed the CONUT (AUC: 0.597, 95% CI: 0.530–0.663) and PNI (AUC: 0.588, 95% CI: 0.519–0.657) in predicting SAP (p < 0.05). Subgroup analysis revealed that the NRS-2002 score ≥ 3 was particularly predictive of SAP in patients with weight loss, severe stroke, and those without hypertension or with diabetes.
Conclusion: The NRS-2002 score is a valuable predictor of pneumonia in SICH patients, with higher scores correlating with a significantly increased risk of SAP. This highlights the importance of early nutritional assessment in identifying high-risk patients and potentially guiding clinical interventions to reduce SAP incidence.
Stroke-associated pneumonia (SAP), defined as lower respiratory tract infection occurring within 7 days in non-mechanically ventilated stroke patients (1), is a common and serious complication that significantly increases adverse outcomes and mortality (2, 3). Muscle weakness after a stroke impairs swallowing and coughing, raising the risk of aspiration of food, saliva, or liquids, which introduces pathogens into the lungs (4). Moreover, stroke-induced immunosuppression weakens the body’s immune defenses, further facilitating lung infections (5). SAP not only prolongs hospital stays and delays functional recovery but also increases the risk of complications such as deep vein thrombosis, gastrointestinal bleeding, and atrial fibrillation (6). As a major contributor to poor stroke outcomes, understanding its underlying mechanisms and identifying high-risk groups is essential.
Malnutrition affects approximately 6 to 62% of stroke patients admitted to hospitals (7, 8). Recent studies have demonstrated a close association between nutritional status and the development of SAP. Nutritional indicators, including the albumin-to-globulin ratio, blood lipid levels, Body Mass Index (BMI), and fibrinogen-to-prealbumin ratio, have been found to be associated with the development of stroke-associated pneumonia in ischemic stroke (IS) patients (9–11). Additionally, nutritional assessment tools, which are methods for evaluating a patient’s nutritional status based on a combination of clinical data, such as the Nutritional Risk Screening-2002 (NRS-2002), Prognostic Nutritional Index (PNI), Controlling Nutritional Status (CONUT), and Geriatric Nutritional Risk Index (GNRI) have been identified as useful predictors of SAP risk in IS patients (12–14). However, research on the relationship between nutritional status and pneumonia associated with spontaneous intracerebral hemorrhage (SICH) remains limited. Although both IS and SICH share common risk factors for developing pneumonia, there are also distinct risk factors specific to pneumonia following SICH. First, SICH patients are typically required to remain bedridden in the acute phase to minimize the risk of worsening bleeding (15), while IS patients are encouraged to mobilize early to reduce complications like venous thromboembolism and promote rehabilitation (16). Prolonged bed rest in SICH patients is a significant risk factor for the development of aspiration pneumonia (17). Second, consciousness impairment caused by brain edema after a stroke increases the risk of pneumonia (18). Brain edema is generally more severe in SICH than in IS (19), making imaging factors closely related to edema, such as lesion volume and location, especially important in SICH-associated pneumonia. Furthermore, hemorrhagic infiltration into the ventricles is a distinctive imaging feature in SICH patients. Finally, while cardiac factors, such as atrial fibrillation, play a significant role in pneumonia following IS (20), they have relatively less influence on pneumonia after SICH (21). Evidence suggests that patients with hemorrhagic stroke are more likely to develop pneumonia compared to those with IS (22). Therefore, it is crucial to explore the relationship between nutritional status and pneumonia in patients with SICH.
The NRS2002 score is a nutritional screening tool based on over 100 randomized controlled trials (RCTs) as its evidence base (23). Since its introduction in 2002, numerous studies have further validated its effectiveness in clinical practice (24, 25). Many clinical nutrition guidelines, such as those from the European Society for Parenteral and Enteral Nutrition (ESPEN) and the Chinese Society for Parenteral and Enteral Nutrition (CSPEN), recommend the NRS2002 as the preferred tool for nutritional risk screening. The NRS2002 score evaluates patients’ nutritional status based on three parameters: impaired nutritional status, disease severity, and age (23). The CONUT and PNI scores evaluate nutritional status from the perspective of immune and inflammatory responses. Since stroke induces immune and inflammatory reactions (26), these scores are particularly relevant for assessing the nutritional status of affected patients. Additionally, the parameters involved are readily available in routine clinical practice, ensuring they do not impose an additional burden on healthcare providers. This study aims to explore the relationship between the NRS-2002 and SICH-associated pneumonia. Additionally, it seeks to compare the predictive performance of the NRS-2002 score with other nutritional scoring systems, including the CONUT and the PNI.
In this retrospective observational study, we focused solely on consecutive patients with spontaneous intracerebral hemorrhage admitted to Dongyang People’s Hospital from January 1, 2023, to May 31, 2024. The study received ethical approval from the Ethics Committee of Dongyang People’s Hospital (2024-YX-332) and was conducted in accordance with principles outlined in the Declaration of Helsinki. To safeguard patient privacy, all personal information was anonymized during data collection and analysis. SAP was diagnosed within the first 7 days of admission according to the criteria based on clinical and laboratory indices of respiratory tract infection (e.g., fever, cough, auscultatory respiratory crackles, new purulent sputum, or positive sputum culture), and supported by typical chest X-ray or chest computed tomography imaging (1). Patients were included based on the following criteria: (1) Aged 18 years or older. (2) Admission with a primary diagnosis of SICH, confirmed by cerebral computed tomography (CT) or magnetic resonance imaging (MRI). (3) Signed informed consent was obtained from the patients or their legally authorized representative. The exclusion criteria were: (1) Intracerebral hemorrhage resulting from brain tumors, aneurysms, arteriovenous malformations, cavernous hemangiomas, or traumatic brain injuries. (2) Patients who underwent mechanical ventilation. (3) Pre-existing pulmonary infection prior to admission. (4) Severe hepatic and renal insufficiency. (5) Hospitalization duration of less than 24 h. (6) Incomplete or missing data.
We estimated the minimum sample size required for the study using PASS software. Due to the lack of data on the NRS-2002 and stroke-related pneumonia in patients with SICH, we referred to data from studies on NRS-2002 and stroke-associated infections in patients with IS (12). The expected odds ratio (OR) for exposure was 2.3, and the proportion of patients with nutritional risk in the non-pneumonia group was 29%. The significance level (α) was set at 0.05, and the power (β) was set at 0.80. The minimum sample size was calculated using the “two independent proportions” module of PASS software with a 1:1 group allocation.
We gathered a comprehensive set of baseline characteristics, including demographic data, vascular risk factors, comorbidities, laboratory results, and neuroimaging findings upon admission. Hypertension was identified by the prior use of antihypertensive medications (27). Diabetes mellitus was defined as the use of glucose-lowering medications or hemoglobin A1c ≥6.5% (28). Conditions such as ischemic heart disease (IHD), dementia, chronic obstructive pulmonary disease (COPD), and cancer were considered present if there was clear medical documentation or a confirmed diagnosis at discharge. A history of stroke was defined by a past transient ischemic attack or stroke (29). Current cigarette smoking and alcohol consumption of 15 g or more per day in the past year were defined accordingly (30). Neuroimaging was independently reviewed by two seasoned neurologists, with disagreements resolved through group discussions. Due to the heterogeneity in NIHSS and GCS score assessments in medical records, a third-party assessment team, blinded to the study group and treatment assignments, conducted a centralized review based on the available records. Any discrepancies in results led to intervention by a third assessor. A trained nurse or neurologist assessed the patients’ swallowing function using the Kubota Water Swallowing Test (31). Dysphagia was defined as a score of 3 or higher, or if the patient was unable to cooperate due to impaired consciousness. Based on patient information, the risk of stroke-associated pneumonia was calculated using the Intracerebral Hemorrhage-Associated Pneumonia Score (ICH-APS) (21).
The NRS-2002, CONUT, and PNI were used to assess patients’ nutritional status. The NRS-2002 was applied by trained nurses to evaluate the nutritional status of patients upon admission. The NRS-2002 score ranges from 0 to 7 and includes three components: (1) disease severity (indicating increased nutritional needs), ranging from 0 to 3 based on comorbidities and medical history; (2) nutritional impairment, based on BMI, body weight, and food intake, with a score range of 0 to 3; and (3) age, with one point assigned for patients aged 70 years or older. Patients with NRS-2002 score of 3 or greater are classified as being at risk of malnutrition (23). The CONUT is calculated using three indicators: serum albumin level, peripheral blood lymphocyte count, and total cholesterol level. Points are assigned based on laboratory results: (1) Serum albumin: ≥ 35 g/L = 0 points, 30–34 g/L = 2 points, 25–29 g/L = 4 points, < 25 g/L = 6 points; (2) Lymphocyte count: ≥ 1.6 × 109/L = 0 points, 1.2–1.6 × 109/L = 1 point, 0.8–1.1 × 109/L = 2 points, < 0.8 × 109/L = 3 points; (3) Total cholesterol: ≥ 180 mg/dL = 0 points, 140–179 mg/dL = 1 point, 100–139 mg/dL = 2 points, < 100 mg/dL = 3 points. The total score (0–12) reflects nutritional status, with higher scores indicating worse nutrition (32). The PNI is calculated by adding the serum albumin level to 5 times the peripheral blood lymphocyte count (33).
Continuous variables were presented as medians with interquartile ranges (IQRs). Categorical variables were expressed as numbers and percentages. Comparisons between two groups for continuous variables were made using the Mann–Whitney U-test. For categorical variables, differences between the groups were analyzed using Fisher’s exact test or the Chi-square (χ2) test, as appropriate. Univariate logistic regression was used to identify risk factors for SICH-associated pneumonia. Stepwise multivariate logistic regression, adjusting for variables with p < 0.05 in the univariate analysis, was then applied to further examine the relationship between the NRS-2002 score and pneumonia in patients with SICH. SHAP (SHapley Additive exPlanations) is used to assess the contribution of each variable to the risk of pneumonia in patients with SICH. Receiver operating characteristic (ROC) analyses were conducted to evaluate the predictive performance of various nutritional scores and to externally validate the well-established ICH-APS model. Restricted cubic splines were utilized to explore the linear relationship between the NRS-2002 score and SICH-associated pneumonia. Subgroup analysis was performed to investigate potential moderating variables in the association between the NRS-2002 score and SICH-associated pneumonia. All statistical tests were two-tailed, with a significance level set at p < 0.05. Data analysis was conducted using PASS version 15, R version 4.0.4, SPSS version 26, and GraphPad Prism 9.0.
Based on the sample size calculation result, a minimum of 95 pneumonia patients and 95 non-pneumonia patients were required to achieve reliable results. A total of 697 patients with spontaneous intracerebral hemorrhage were initially considered for inclusion. Among them, 65 patients were excluded due to intracerebral hemorrhage resulting from brain tumors, aneurysms, arteriovenous malformations, cavernous hemangiomas, or traumatic brain injuries; 189 patients were excluded because of mechanical ventilation; 6 patients were excluded for having pre-existing pulmonary infection prior to admission; 5 patients were excluded due to a hospital stay of less than 24 h; and 28 patients were excluded due to incomplete data. Ultimately, 404 patients met the inclusion criteria, comprising 97 with pneumonia and 307 without pneumonia, meeting the minimum sample size requirement. Baseline characteristics of these patients were presented in Table 1. The average age of the included patients was 65.2 years, and 280 (69.3%) were male. Of these, 155 had an NRS-2002 score ≥ 3. These patients were older, had a lower BMI, were more likely to have comorbidities, and exhibited lower levels of blood lipids and blood cell counts compared to those with an NRS-2002 score < 3.
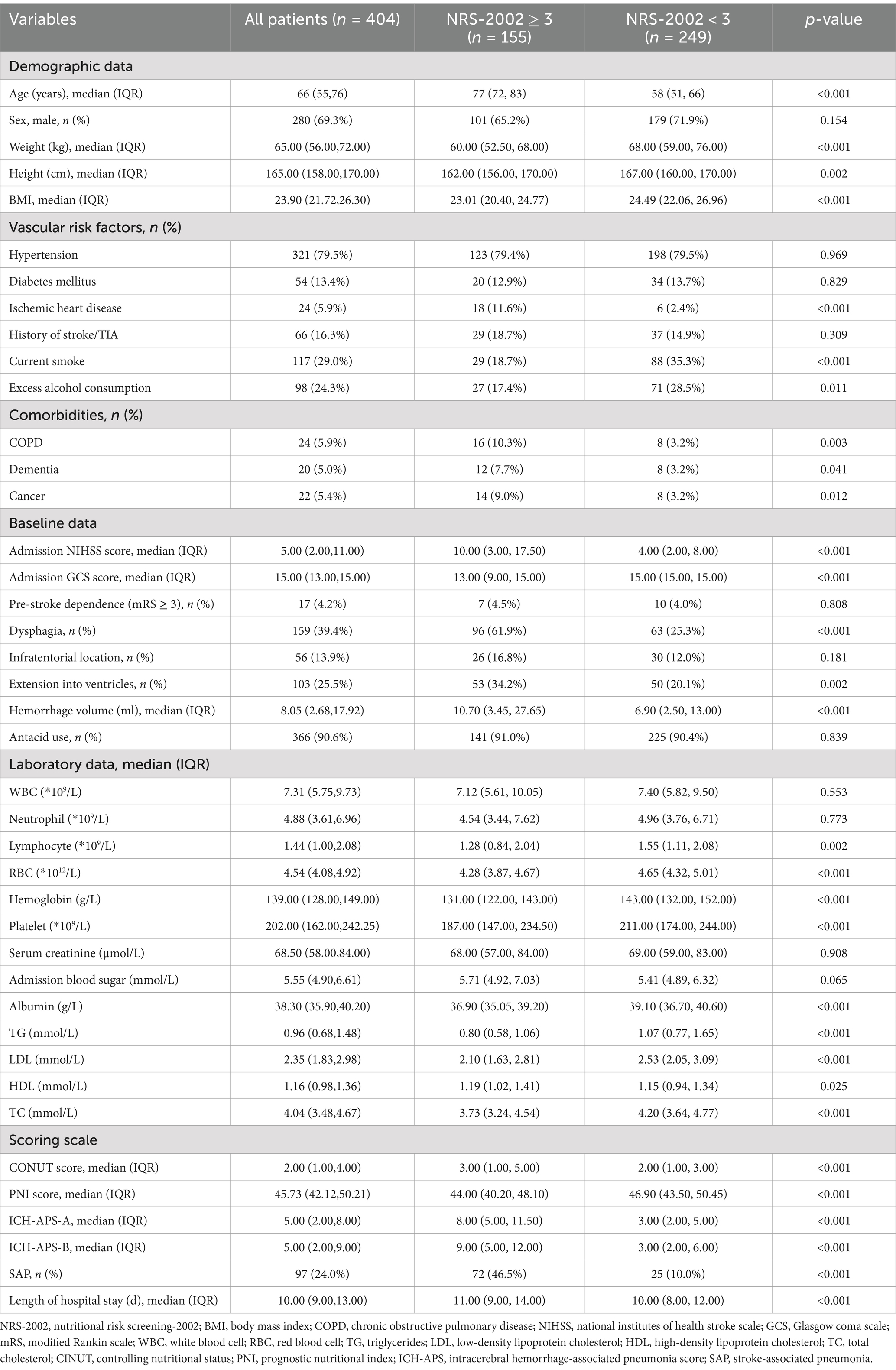
Table 1. Baseline characteristics of patients with spontaneous intracerebral hemorrhage: NRS-2002 ≥ 3 vs. NRS-2002 < 3.
Compared with non-pneumonia patients, those with pneumonia had a significantly higher proportion of NRS-2002 scores ≥3 (74.2% vs. 27.0%, p < 0.001). Moreover, there was a clear upward trend in the proportion of pneumonia patients with increasing NRS-2002 score (P-trend <0.001) (Figure 1). In multivariate logistic regression analysis, after adjusting for variables with p < 0.05 in the univariate analysis, the NRS-2002 score remained significantly associated with an increased risk of SAP (OR: 1.575, 95% CI: 1.134–2.186, p = 0.007). Additionally, other factors such as age (OR: 1.045, 95% CI: 1.017–1.073, p = 0.002), chronic obstructive pulmonary disease (COPD) (OR: 3.734, 95% CI: 1.198–11.643, p = 0.023), dysphagia (OR: 6.407, 95% CI: 3.023–13.582, p < 0.001), and NIHSS score (OR: 1.075, 95% CI: 1.031–1.120, p < 0.001) were also identified as significant risk factors for SAP (Table 2). The SHAP plot shows the importance of each feature (variables with p < 0.05 in multivariate logistic regression) in predicting stroke-associated pneumonia in patients with SICH (Figure 2). NRS-2002 is the fourth most significant positive contributor, while dysphagia is the strongest positive contributor. Restricted cubic spline (RCS) analysis showed that the incidence of SAP increased notably with higher NRS-2002 scores when the score was ≥3. This relationship became more pronounced as the NRS-2002 score increased (Figure 3).
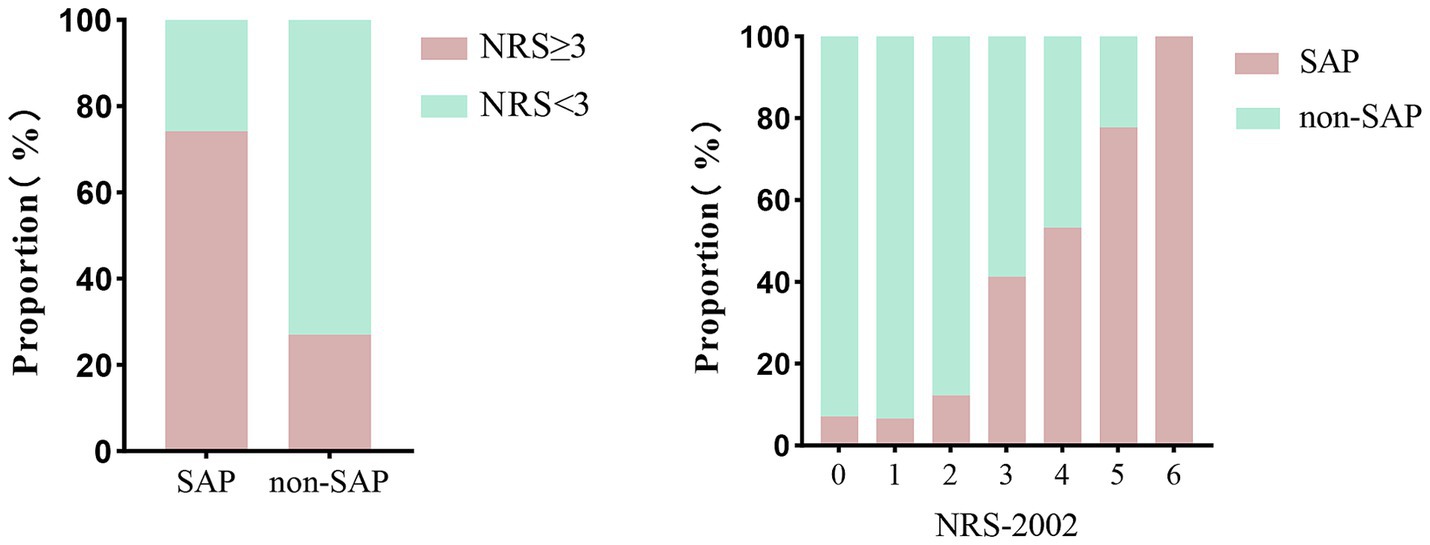
Figure 1. Probability distribution histogram illustrated the association of NRS-2002 and SAP. The proportion of patients with malnutrition (NRS-2002 ≥ 3) in SAP is higher than that in non-SAP (74.2% vs. 27.0%, p < 0.001). An increase in the NRS-2002 score is associated with a higher risk of pneumonia (P-trend <0.001). NRS-2002, nutritional risk screening-2002; SAP, stroke-associated pneumonia.

Table 2. Logistic regression analysis for predicting SAP in patients with spontaneous intracerebral hemorrhage.
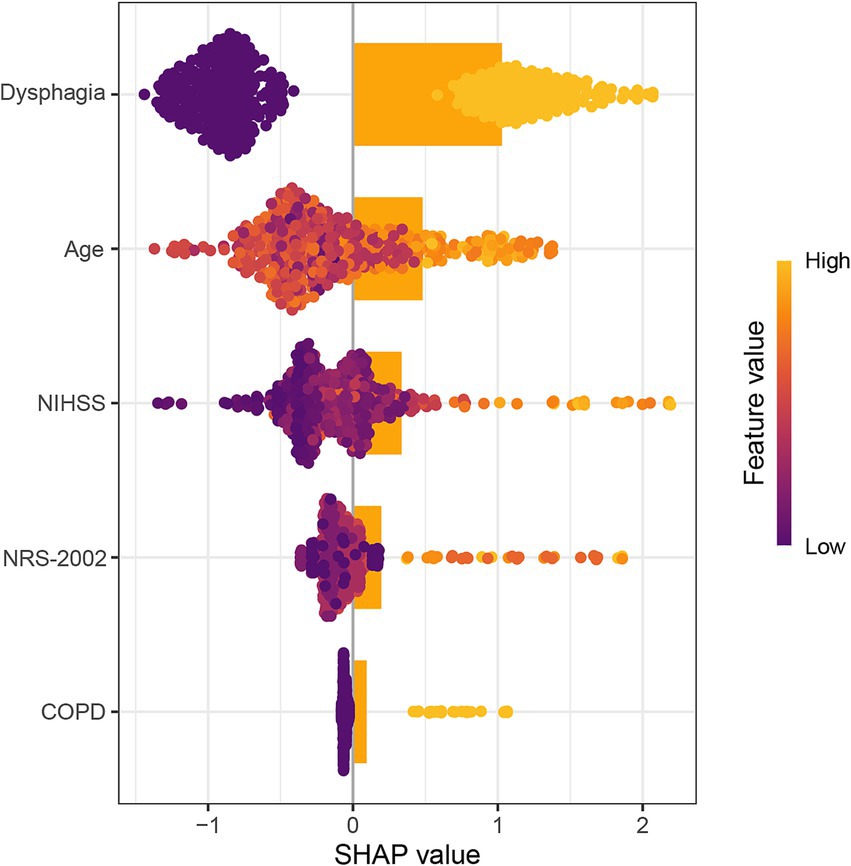
Figure 2. SHAP analysis of the contribution of each risk factor in predicting spontaneous intracerebral hemorrhage-related pneumonia. SHAP, SHapley Additive exPlanations; NRS-2002, nutritional risk screening-2002; NIHSS, national institutes of health stroke scale; COPD, chronic obstructive pulmonary disease.
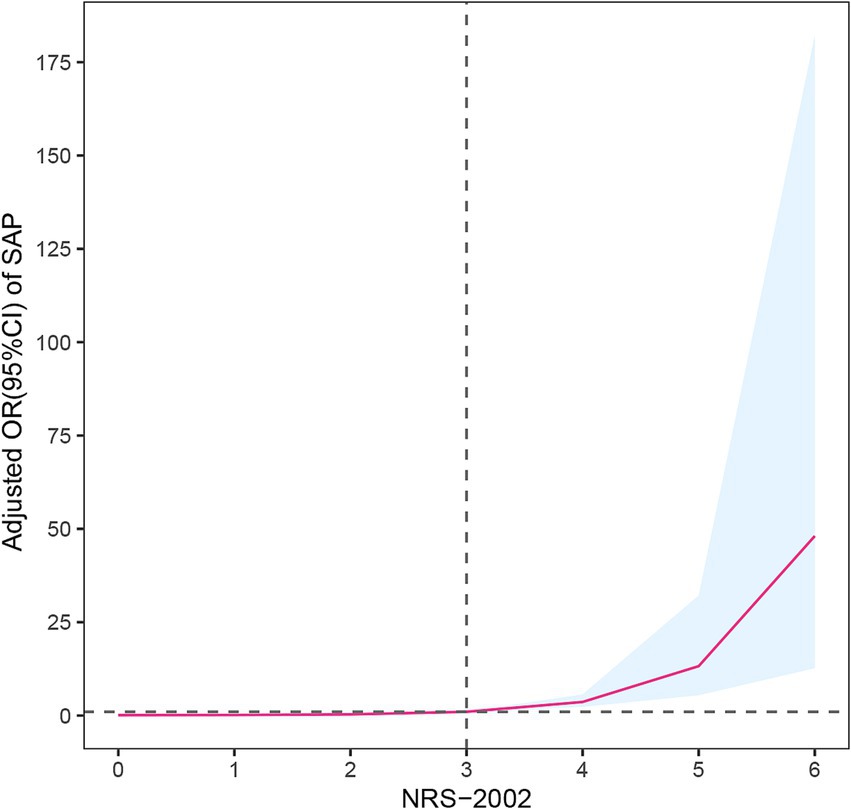
Figure 3. Restricted cubic spline model illustrated the association between NRS-2002 and SAP (P overall = 0.013, P non-linear = 0.151). The model accounts for confounding factors, including age, sex, NIHSS score, COPD, and dysphagia. The incidence of SAP increased significantly with higher NRS-2002 score, especially when the score was ≥3. NRS-2002, nutritional risk screening-2002; SAP, stroke-associated pneumonia; NIHSS, national institutes of health stroke scale; COPD, chronic obstructive pulmonary disease.
Figure 4 presented the predictive performance of the NRS-2002 score for SAP using AUC-ROC (Area Under the Receiver Operating Characteristic Curve) analysis (AUC: 0.768, 95% CI: 0.716–0.820). The NRS-2002 score demonstrated superior predictive capability for SAP compared to the PNI (AUC: 0.588, 95% CI: 0.519–0.657) and the CONUT score (AUC: 0.597, 95% CI: 0.530–0.663) (p < 0.01). External validation of the classic ICH-APS-A (AUC: 0.852, 95% CI: 0.807–0.896) and ICH-APS-B (AUC: 0.857, 95% CI: 0.813–0.901) models showed better predictive performance than the NRS-2002 score (p < 0.01). Incorporating the NRS-2002 score into the ICH-APS-A and ICH-APS-B models slightly increased the AUC, but this improvement was not statistically significant (p > 0.05). Table 3 summarized the optimal cut-off points, specificity, sensitivity, positive predictive value, and negative predictive value for each scoring system.
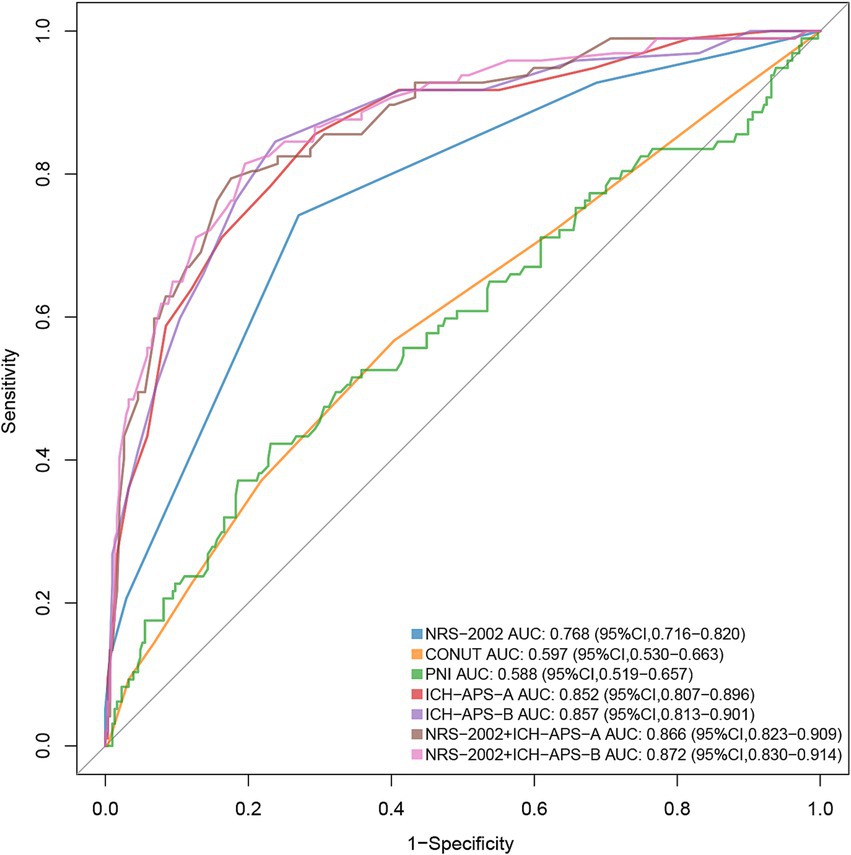
Figure 4. Comparison of AUCs between different predictive models. For predicting SAP, ICH-APS exhibited superior predictive performance compared to the nutritional assessment tools (p < 0.05). Among the nutritional scoring systems, NRS-2002 provided better predictive value than PNI and CONUT (p < 0.05). AUC, area under the curve; SAP, stroke-associated pneumonia; ICH-APS, intracerebral hemorrhage-associated pneumonia score; NRS-2002, nutritional risk screening-2002; PNI, prognostic nutritional index; CONUT, Controlling nutritional status.
A subgroup analysis stratified patients by age, sex, BMI, NIHSS score, hypertension, and diabetes (Figure 5). The results demonstrated that an NRS-2002 score ≥ 3 was associated with a higher likelihood of SAP in patients with a BMI <18.5 (OR: 22.00, 95% CI: 2.05–236.04, p = 0.011), NIHSS score > 5 (OR: 10.71, 95% CI: 5.27–21.78, p < 0.001), absence of hypertension (OR: 12.44, 95% CI: 3.20–48.46, p < 0.001), and diabetes (OR: 13.09, 95% CI: 2.44–70.12, p = 0.003). The severity of stroke might influence the relationship between the NRS-2002 score and SAP (P for interaction =0.011).
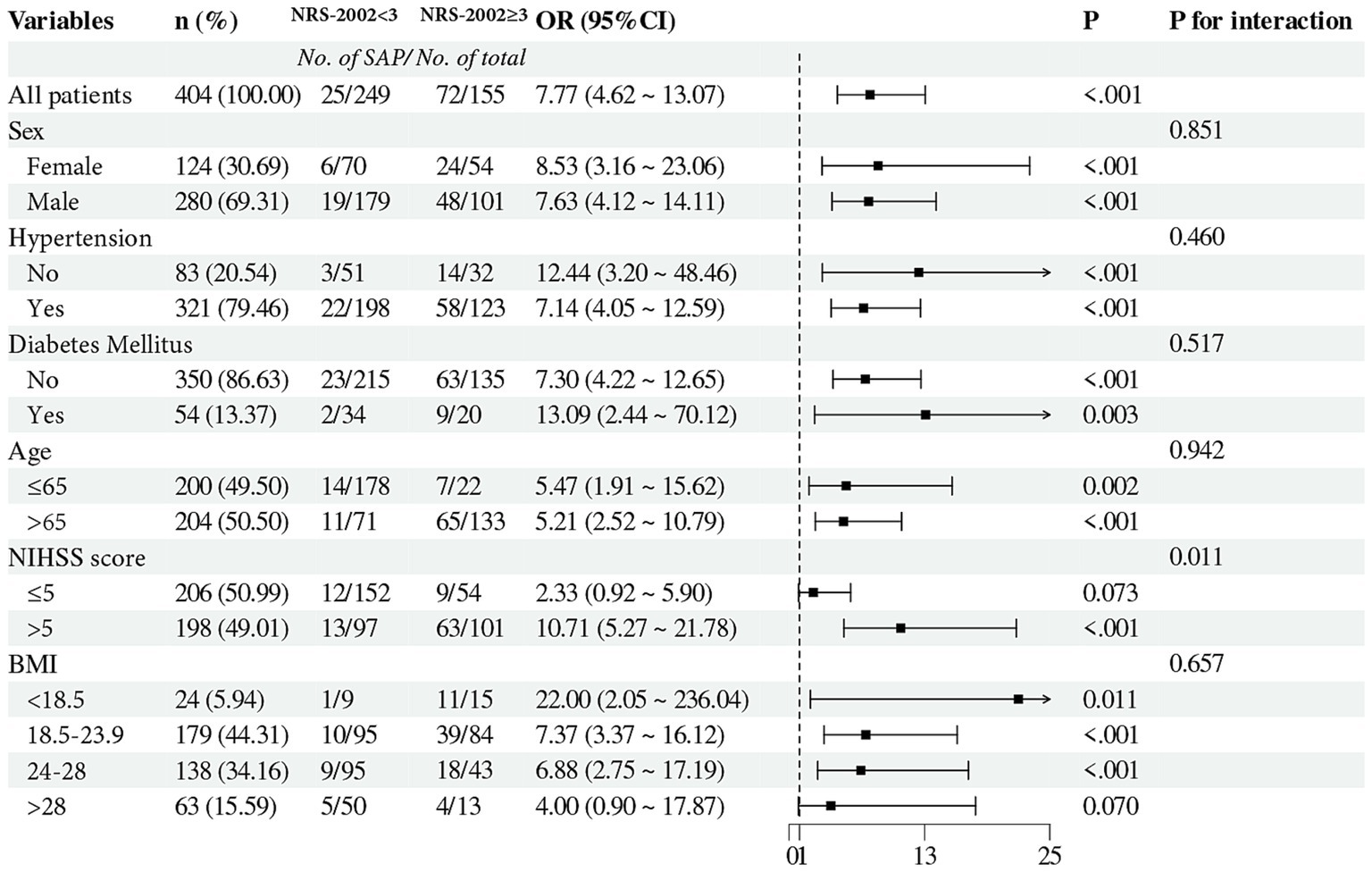
Figure 5. Subgroup analysis evaluating the interaction effect of NRS-2002 on the incidence of SAP across varied subgroups. The forest plot indicated that the NIHSS score might moderate the relationship between NRS-2002 and SAP occurrence (P for interaction = 0.011). NRS-2002, nutritional risk screening-2002; SAP, stroke-associated pneumonia; NIHSS, national institutes of health stroke scale; BMI, body mass index.
Studies have demonstrated that the NRS-2002 score was linked to the short-term and long-term prognosis of hemorrhagic stroke patients (34). However, few studies have focused on the connection between the NRS-2002 score and pneumonia related to SICH. To the best of our knowledge, this was the first study to explore the association between the NRS-2002 score and SICH-related pneumonia. Our results indicated that higher NRS-2002 score at the time of admission was significantly associated with an increased risk of developing SICH-related pneumonia. These findings underscored the pivotal role of nutritional status in the acute-phase management of spontaneous intracerebral hemorrhage.
In our study, 38.4% of patients with SICH were identified as malnourished, aligning with prior findings on nutritional status in cerebral infarction patients assessed using the NRS-2002 score (35). Malnourished patients in our study were typically older, had lower BMI, and exhibited more comorbidities. This trend might be attributed to the emphasis of the NRS-2002 scoring criteria. However, when alternative nutritional assessment tools were applied to stroke patients, malnourished individuals consistently displayed similar baseline characteristics (14), indicating potential concordance among different scoring systems. Blood lipid levels differ significantly between malnourished and healthy populations in our study. The Chinese Visceral Adiposity Index (CVAI), an indicator of visceral fat in the Chinese population, is strongly associated with stroke incidence (36). Unlike the PNI and CONUT scores, which assess nutritional status from immune and inflammatory perspectives, CVAI reflects nutritional status through blood lipid profiles. Its potential link to stroke-associated pneumonia warrants further investigation. Furthermore, our study observed lower levels of immune and blood cells in malnourished patients, providing evidence to support the use of immune and inflammatory indicators in evaluating nutritional status. The ICH-APS is a recognized predictor of pneumonia in patients with SICH and is recommended by the Chinese clinical management guidelines for intracerebral hemorrhage to evaluate the risk of pneumonia. In malnourished patients, ICH-APS scores were significantly higher, which further validated the use of the NRS-2002 score in assessing pneumonia risk in patients with SICH. Interestingly, the study found a lower proportion of smokers and alcohol consumers among malnourished patients. We believe this might be due to our definition of smoking and drinking as current smoking and excessive alcohol consumption. Malnourished patients had comorbid conditions like COPD and ischemic heart disease in our study, and in real-world situations, these individuals tend to quit smoking and reduce alcohol intake.
Consistent with earlier research (37), our study based on the NRS-2002 score showed that malnutrition increases the risk of pneumonia related to SICH. Malnutrition leads to immune suppression, resulting in an increased incidence of pneumonia. Malnutrition compromises immune function through various mechanisms. It reduces T-cell counts, particularly CD4+ T cells with interleukin-2 receptors and CD3+ and CD25+ T cells, leading to impaired cell-mediated immunity (38). Furthermore, malnutrition affects phagocyte function, diminishing the body’s ability to clear pathogens, and disrupts complement synthesis and activity, weakening antiviral defenses (39). Additionally, malnutrition impairs muscle tissue repair and energy supply, leading to respiratory muscle weakness or even atrophy. This results in difficulty coughing up sputum and breathing, further increasing the risk of pneumonia (40, 41). In our study, we observed that advanced age increased the risk of SICH-associated pneumonia. Since age was a component of the NRS-2002 score, we employed stepwise regression to mitigate the effects of collinearity. Despite this adjustment, age continued to show a significant association with an increased risk of pneumonia in patients with SICH. Many previous studies have identified a relationship between age and stroke-associated pneumonia (3, 21, 42). In the ICH-APS scoring model, age is positively associated with the risk of developing SAP, with the risk increasing by 4% for each additional year (21). Similarly, another study on pneumonia following SICH reported a comparable positive correlation between age and SAP risk (3), with the same odds ratio found in the ICH-APS scoring model. Our study also yielded a similar OR of 1.045 (95% CI: 1.017–1.073). We hypothesize that age-related nutritional risks may significantly contribute to this association. Additionally, aging leads to a decline in both cellular and humoral immunity, weakening the body’s ability to effectively combat pathogens (43, 44). This compromised nutrition and immune defense allows pathogens to proliferate in the respiratory tract, increasing the risk of lung infections. In the AIS-ASP model, COPD is significantly associated with the development of stroke-associated pneumonia (SAP) following IS (adjusted OR = 1.67, 95% CI: 1.09–2.55) (20). This correlation is even stronger in patients with SICH. COPD patients have more than a fivefold higher risk of developing SAP after ICH compared to non-COPD patients in the ICH-APS model (21). Consistent with previous studies (3, 21, 42), our findings also indicate that COPD patients are at a higher risk of developing SAP (adjusted OR = 3.734, 95% CI: 1.198–11.643). COPD is characterized by chronic bronchitis, emphysema, and other lung pathologies (45), leading to chronic inflammation and structural changes in the airways (46, 47). In the context of intracerebral hemorrhage, immune suppression exacerbates these existing respiratory complications, further elevating the risk of lung infections (5). Dysphagia has the greatest impact on the weight coefficient of pneumonia for SICH in ICH-LR2S2 score (3). In our study, dysphagia also showed the highest odds ratio, indicating that it was the most significant risk factor for SAP. This could be due to the increased risk of aspiration pneumonia caused by swallowing difficulties (48). Previous studies have nearly always identified an association between NIHSS score and SAP when NIHSS score was included in the analysis (3, 21, 42), and our study aligned with this finding. We suggest that future research investigate the association between specific NIHSS score components and stroke-associated pneumonia, with a particular focus on dysarthria and level of consciousness, as these may play a more significant role than other components.
There are numerous nutritional risk assessment tools, each with its own criteria. They can generally be classified into two categories: objective and subjective. The former focuses on hematological, biochemical, and anthropometric evaluations, while subjective assessments tend to rely on nutritional assessment questionnaires and medical history collection (49). Through the DeLong test comparison of AUC, we observed that the NRS-2002 score demonstrates superior predictive performance for pneumonia associated with SICH compared to the CONUT and PNI nutritional scores. This may be due to the fact that markers such as albumin and lymphocytes are affected not only by nutritional status but also by liver and kidney function, as well as hematopoietic activity (50, 51). Since we did not exclude patients with mild hepatic and renal dysfunction during enrollment, this may impact the true reflection of nutritional status by PNI and CONUT. In contrast, the NRS2002 evaluates nutritional status from multiple dimensions, overcoming the limitations of relying on a single marker, and provides a more comprehensive and accurate assessment of a patient’s nutritional risk. To further minimize subjective scoring variability, we provided systematic training for the assessors in advance to reduce potential bias. We externally validated the ICH-APS model through AUC-ROC analysis, indicating that it has excellent predictive value for pneumonia associated with SICH. Considering that the ICH-APS model does not fully account for the impact of nutritional status on pneumonia development, we integrated the NRS-2002 with the ICH-APS model for analysis and found that it did not statistically improve its predictive effectiveness. Based on the SHAP results in the study, the contribution of NRS-2002 is smaller than that of other indicators such as dysphagia, age, and the NIHSS score in predicting SAP. These indicators are all part of the ICH-SAP model, making the predictive efficacy of NRS-2002 incomparable to that of the ICH-SAP model. Compared to the ICH-APS model, which requires the inclusion of multiple parameters for prediction, the NRS-2002 score is simpler and more practical, thus offering high clinical applicability. We further used sensitivity, specificity, negative predictive value, and positive predictive value to demonstrate the characteristics of each prediction model. The NRS-2002 score had a threshold of 2.5 for predicting SICH-associated pneumonia in our study, with a score of 3 or higher indicating nutritional risk according to the guidelines (23). This further confirms the association between malnutrition and SICH-associated pneumonia. Notably, all the models demonstrated good negative predictive values, but their positive predictive values were relatively low. Therefore, the primary utility of these models is to help exclude patients with pneumonia in clinical practice.
The subgroup analysis identified a stronger correlation between malnutrition and SICH-associated pneumonia in patients with diabetes and those without hypertension. In diabetic patients, insulin resistance contributes to relative insulin deficiency and impaired glucose utilization, increasing the risk of malnutrition (12). Among non-hypertensive patients, a higher prevalence of non-hypertensive intracerebral hemorrhage may impact nutritional risk and the incidence of pneumonia. Future research should investigate and compare the prevalence of malnutrition and pneumonia across patients with intracerebral hemorrhage of varying etiologies. RCS analysis revealed that the relationship between malnutrition and pneumonia strengthens as NRS-2002 scores increase in malnourished patients. Since BMI is a key component of the NRS-2002 tool (23), individuals with low BMI are more likely to experience severe malnutrition. Additionally, due to the reduced expression of immune factors, patients with low BMI have weaker immune resistance (52), which increases their susceptibility to various infections during hospitalization. Besides pneumonia, they are also more prone to urinary tract infections, skin and soft tissue infections, and other complications (53, 54), which contribute to a higher disease severity and subsequently lead to an increased NRS-2002 score. As a result, underweight patients with malnutrition face a substantially higher risk of pneumonia compared to those with normal weight, overweight, or obesity. A significant interaction was observed between NRS-2002 and NIHSS scores in predicting the occurrence of SICH-associated pneumonia. Neurological deficits have been identified as risk factors for malnutrition (3, 21, 42, 55). Compared to patients with mild stroke, those with moderate to severe stroke are at a higher risk of malnutrition. As nutritional risk increases, the relationship between pneumonia and malnutrition becomes more pronounced, leading to a greater risk of pneumonia in malnourished patients with moderate to severe stroke.
In this study, we demonstrated that the NRS2002-based nutritional score can effectively predict the risk of stroke-associated pneumonia during hospitalization in patients with spontaneous intracerebral hemorrhage at the time of admission. For patients identified as being at risk of malnutrition, early consultation with a dietitian and timely, appropriate nutritional support, along with improved oral care and respiratory management, can be provided to reduce the risk of pneumonia. The NRS-2002 score does not require additional diagnostic procedures, making it feasible for implementation in both developing and developed countries. However, certain parameters in the score, such as disease severity, rely on the evaluator’s clinical judgment. Therefore, systematic and standardized training for evaluators is crucial. This study has several limitations. First, as a single-center retrospective study, it might be subject to selection bias. A multicenter study is essential for future research. Second, inflammatory markers such as C-reactive protein and procalcitonin were not included (56, 57), hindering the ability to explore the relationship between these markers, malnutrition, and stroke-associated pneumonia. Lastly, the study only focused on the relationship between baseline nutritional status and stroke-associated pneumonia, without tracking changes in nutritional status over time.
The NRS-2002 score is associated with stroke-related pneumonia in patients with spontaneous intracerebral hemorrhage. Patients with a nutritional risk (NRS-2002 ≥ 3) are at a higher risk of developing pneumonia. Compared to other nutritional assessment tools, the NRS-2002 score shows greater predictive effectiveness.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Dongyang People’s Hospital Medical Ethics Committee. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
XZ: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. LK: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. PD: Conceptualization, Data curation, Investigation, Methodology, Supervision, Writing – original draft. DX: Methodology, Project administration, Supervision, Validation, Visualization, Writing – review & editing. HL: Conceptualization, Investigation, Methodology, Software, Writing – review & editing. ZJ: Data curation, Methodology, Software, Supervision, Writing – original draft.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
We would like to express our gratitude to all the participants and investigators for their valuable contributions in collecting the data for this project.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Smith, CJ, Kishore, AK, Vail, A, Chamorro, A, Garau, J, Hopkins, SJ, et al. Diagnosis of stroke-associated pneumonia: recommendations from the pneumonia in stroke consensus group. Stroke. (2015) 46:2335–40. doi: 10.1161/STROKEAHA.115.009617
2. Wu, B, Luo, H, Li, J, Chen, Y, Liu, J, Yu, P, et al. The relationship between the Barthel index and stroke-associated pneumonia in elderly patients and factors of sap. BMC Geriatr. (2024) 24:829. doi: 10.1186/s12877-024-05400-8
3. Yan, J, Zhai, W, Li, Z, Ding, L, You, J, Zeng, J, et al. Ich-Lr2s2: a new risk score for predicting stroke-associated pneumonia from spontaneous intracerebral hemorrhage. J Transl Med. (2022) 20:193. doi: 10.1186/s12967-022-03389-5
4. Qiao, X, Lu, C, Xu, M, Yang, G, Chen, W, and Liu, Z. Deepsap: a novel brain image-based deep learning model for predicting stroke-associated pneumonia from spontaneous intracerebral hemorrhage. Acad Radiol. (2024) 31:5193–203. doi: 10.1016/j.acra.2024.06.025
5. Li, D, Yuan, L, Wang, T, Rong, Y, Li, C, You, M, et al. Risk factors for stroke-related pneumonia in patients with Ischaemic stroke: a systematic evaluation and Meta-analysis. Clin Neurol Neurosurg. (2024) 246:108593. doi: 10.1016/j.clineuro.2024.108593
6. Strange, JE, Sindet-Pedersen, C, Staerk, L, Grove, EL, Gerds, TA, Torp-Pedersen, C, et al. All-cause mortality, stroke, and bleeding in patients with atrial fibrillation and Valvular heart disease. Eur Heart J Cardiovasc Pharmacother. (2021) 7:f93–f100. doi: 10.1093/ehjcvp/pvaa011
7. Martineau, J, Bauer, JD, Isenring, E, and Cohen, S. Malnutrition determined by the patient-generated subjective global assessment is associated with poor outcomes in acute stroke patients. Clin Nutr. (2005) 24:1073–7. doi: 10.1016/j.clnu.2005.08.010
8. Paquereau, J, Allart, E, Romon, M, and Rousseaux, M. The long-term nutritional status in stroke patients and its predictive factors. J Stroke Cerebrovasc Dis. (2014) 23:1628–33. doi: 10.1016/j.jstrokecerebrovasdis.2014.01.007
9. Chen, L, Xu, M, Huang, Q, Liu, Y, and Ren, W. Clinical significance of albumin to globulin ratio among patients with stroke-associated pneumonia. Front Nutr. (2022) 9:970573. doi: 10.3389/fnut.2022.970573
10. Liu, Y, Chen, Y, Zhi, Z, Wang, P, Wang, M, Li, Q, et al. Association between Tcbi (triglycerides, Total cholesterol, and body weight index) and stroke-associated pneumonia in acute ischemic stroke patients. Clin Interv Aging. (2024) 19:1091–101. doi: 10.2147/cia.S467577
11. Qiu, H, Luan, X, and Mei, E. High fibrinogen-Prealbumin ratio (Fpr) predicts stroke-associated pneumonia. J Stroke Cerebrovasc Dis. (2024) 33:107703. doi: 10.1016/j.jstrokecerebrovasdis.2024.107703
12. Chen, X, Li, D, Liu, Y, Zhu, L, Jia, Y, and Gao, Y. Nutritional risk screening 2002 scale and subsequent risk of stroke-associated infection in ischemic stroke: the remise study. Front Nutr. (2022) 9:895803. doi: 10.3389/fnut.2022.895803
13. Dai, C, Yan, D, Xu, M, Huang, Q, and Ren, W. Geriatric nutritional risk index is related to the risk of stroke-associated pneumonia. Brain Behav. (2022) 12:e2718. doi: 10.1002/brb3.2718
14. Li, D, Liu, Y, Jia, Y, Yu, J, Li, F, Li, H, et al. Association between malnutrition and stroke-associated pneumonia in patients with ischemic stroke. BMC Neurol. (2023) 23:290. doi: 10.1186/s12883-023-03340-1
15. Steiner, T, Al-Shahi Salman, R, Beer, R, Christensen, H, Cordonnier, C, Csiba, L, et al. European stroke organisation (Eso) guidelines for the management of spontaneous intracerebral hemorrhage. Int J Stroke. (2014) 9:840–55. doi: 10.1111/ijs.12309
16. Bernhardt, J, Dewey, H, Thrift, A, Collier, J, and Donnan, G. A very early rehabilitation trial for stroke (avert): phase ii safety and feasibility. Stroke. (2008) 39:390–6. doi: 10.1161/strokeaha.107.492363
17. Hua, J, Huang, P, Liao, H, Lai, X, and Zheng, X. Prevalence and clinical significance of occult pulmonary infection in elderly patients with type 2 diabetes mellitus. Biomed Res Int. (2021) 2021:3187388. doi: 10.1155/2021/3187388
18. Mégarbane, B, and Chevillard, L. The large Spectrum of pulmonary complications following illicit drug use: features and mechanisms. Chem Biol Interact. (2013) 206:444–51. doi: 10.1016/j.cbi.2013.10.011
19. Wan, Y, Holste, KG, Hua, Y, Keep, RF, and Xi, G. Brain edema formation and therapy after intracerebral hemorrhage. Neurobiol Dis. (2023) 176:105948. doi: 10.1016/j.nbd.2022.105948
20. Ji, R, Shen, H, Pan, Y, Wang, P, Liu, G, Wang, Y, et al. Novel risk score to predict pneumonia after acute ischemic stroke. Stroke. (2013) 44:1303–9. doi: 10.1161/strokeaha.111.000598
21. Ji, R, Shen, H, Pan, Y, Du, W, Wang, P, Liu, G, et al. Risk score to predict hospital-acquired pneumonia after spontaneous intracerebral hemorrhage. Stroke. (2014) 45:2620–8. doi: 10.1161/strokeaha.114.005023
22. Guo, R, Yan, S, Li, Y, Liu, K, Wu, F, Feng, T, et al. A novel machine learning model for predicting stroke-associated pneumonia after spontaneous intracerebral hemorrhage. World Neurosurg. (2024) 189:e141–52. doi: 10.1016/j.wneu.2024.06.001
23. Kondrup, J. Espen guidelines for nutrition screening 2002. Clin Nutr. (2003) 22:415–21. doi: 10.1016/s0261-5614(03)00098-0
24. Kondrup, J, Rasmussen, HH, Hamberg, O, and Stanga, Z. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. (2003) 22:321–36. doi: 10.1016/s0261-5614(02)00214-5
25. van Bokhorst-de van der Schueren, MA, Guaitoli, PR, Jansma, EP, and de Vet, HC. Nutrition screening tools: Does one size fit all? A systematic review of screening tools for the hospital setting. Clin Nutr. (2014) 33:39–58. doi: 10.1016/j.clnu.2013.04.008
26. Alsbrook, DL, Di Napoli, M, Bhatia, K, Biller, J, Andalib, S, Hinduja, A, et al. Neuroinflammation in acute ischemic and hemorrhagic stroke. Curr Neurol Neurosci Rep. (2023) 23:407–31. doi: 10.1007/s11910-023-01282-2
27. Unger, T, Borghi, C, Charchar, F, Khan, NA, Poulter, NR, Prabhakaran, D, et al. International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension. (2020) 75:1334–57. doi: 10.1161/hypertensionaha.120.15026
28. American Diabetes Association R. 16. Diabetes advocacy: standards of medical Care in Diabetes-2019. Diabetes Care. (2020) 43:S203–4. doi: 10.2337/dc20-S016
29. Wang, Y, Cui, L, Ji, X, Dong, Q, Zeng, J, Wang, Y, et al. The China National Stroke Registry for patients with acute cerebrovascular events: design, rationale, and baseline patient characteristics. Int J Stroke. (2011) 6:355–61. doi: 10.1111/j.1747-4949.2011.00584.x
30. Lemarchand, E, Gauberti, M, Martinez de Lizarrondo, S, Villain, H, Repessé, Y, Montagne, A, et al. Impact of alcohol consumption on the outcome of ischemic stroke and thrombolysis: role of the hepatic clearance of tissue-type plasminogen activator. Stroke. (2015) 46:1641–50. doi: 10.1161/strokeaha.114.007143
31. Dong, Y, Zhang, CJ, Shi, J, Deng, J, and Lan, CN. Clinical application of Icf key codes to evaluate patients with dysphagia following stroke. Medicine. (2016) 95:e4479. doi: 10.1097/md.0000000000004479
32. Kuroda, D, Sawayama, H, Kurashige, J, Iwatsuki, M, Eto, T, Tokunaga, R, et al. Controlling nutritional status (Conut) score is a prognostic marker for gastric Cancer patients after curative resection. Gastric Cancer. (2018) 21:204–12. doi: 10.1007/s10120-017-0744-3
33. Zheng, Y, Wang, K, Ou, Y, Hu, X, Wang, Z, Wang, D, et al. Prognostic value of a baseline prognostic nutritional index for patients with prostate Cancer: a systematic review and Meta-analysis. Prostate Cancer Prostatic Dis. (2024) 27:604–13. doi: 10.1038/s41391-023-00689-9
34. Cong, W-J, Liu, Z-P, Liang, Y-X, Ye, S-L, Cai, Z-M, Chen, H-M, et al. Predictive value of nutritional risk for all-cause death and functional outcomes in Chinese elderly patients with acute stroke: a 3-year follow-up study. Clin Interv Aging. (2024) 19:109–18. doi: 10.2147/cia.S447038
35. Cai, Z-m, Wu, Y-z, Chen, H-m, Feng, R-q, Liao, C-w, Ye, S-l, et al. Being at risk of malnutrition predicts poor outcomes at 3 months in acute ischemic stroke patients. Eur J Clin Nutr. (2020) 74:796–805. doi: 10.1038/s41430-020-0605-8
36. Zhang, H, Zhan, Q, Dong, F, Gao, X, Zeng, F, Yao, J, et al. Associations of Chinese visceral adiposity index and new-onset stroke in middle-aged and older Chinese adults: an observational study. Lipids Health Dis. (2023) 22:74. doi: 10.1186/s12944-023-01843-x
37. Zhao, G, Chen, Y, Gu, Y, and Xia, X. The clinical value of nutritional and inflammatory indicators in predicting pneumonia among patients with intracerebral hemorrhage. Sci Rep. (2024) 14:16171. doi: 10.1038/s41598-024-67227-y
38. Committee on Military Nutrition Research. Military strategies for sustainment of nutrition and immune function in the field. Washington (DC): National Academies Press (US) (1999).
39. Marcos, A, Nova, E, and Montero, A. Changes in the immune system are conditioned by nutrition. Eur J Clin Nutr. (2003) 57:S66–9. doi: 10.1038/sj.ejcn.1601819
40. Fiaccadori, E, Zambrelli, P, and Tortorella, G. Physiopathology of respiratory muscles in malnutrition. Minerva Anestesiol. (1995) 61:93–9.
41. Rodríguez González-Moro, JM, de Lucas, RP, and Martínez, AY. Function of respiratory muscles in malnutrition and in the critically ill patient. Arch Bronconeumol. (2002) 38:131–6. doi: 10.1016/s0300-2896(02)75171-5
42. Hoffmann, S, Malzahn, U, Harms, H, Koennecke, HC, Berger, K, Kalic, M, et al. Development of a clinical score (A2ds2) to predict pneumonia in acute ischemic stroke. Stroke. (2012) 43:2617–23. doi: 10.1161/strokeaha.112.653055
43. Frasca, D, Diaz, A, Romero, M, Garcia, D, and Blomberg, BB. B Cell Immunosenescence. Annu Rev Cell Dev Biol. (2020) 36:551–74. doi: 10.1146/annurev-cellbio-011620-034148
44. Mittelbrunn, M, and Kroemer, G. Hallmarks of T cell aging. Nat Immunol. (2021) 22:687–98. doi: 10.1038/s41590-021-00927-z
45. Janssen, DJA, Bajwah, S, Boon, MH, Coleman, C, Currow, DC, Devillers, A, et al. European Respiratory Society clinical practice guideline: palliative Care for People with Copd or interstitial lung disease. Eur Respir J. (2023) 62:2202014. doi: 10.1183/13993003.02014-2022
46. Villaseñor-Altamirano, AB, Jain, D, Jeong, Y, Menon, JA, Kamiya, M, Haider, H, et al. Activation of Cd8(+) T cells in chronic obstructive pulmonary disease lung. Am J Respir Crit Care Med. (2023) 208:1177–95. doi: 10.1164/rccm.202305-0924OC
47. Tanabe, N, Vasilescu, DM, Hague, CJ, Ikezoe, K, Murphy, DT, Kirby, M, et al. Pathological comparisons of Paraseptal and Centrilobular emphysema in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. (2020) 202:803–11. doi: 10.1164/rccm.201912-2327OC
48. Chinese Expert Consensus Group of Dysphagia and Nutrition Management R. Chinese expert consensus on food and nutrition Management for Dysphagia (2019 version). Asia Pac J Clin Nutr. (2020) 29:434–44. doi: 10.6133/apjcn.202007_29(2).0026
49. Aliasghari, F, Izadi, A, Khalili, M, Farhoudi, M, Ahmadiyan, S, and Deljavan, R. Impact of premorbid malnutrition and dysphagia on ischemic stroke outcome in elderly patients: a community-based study. J Am Coll Nutr. (2018) 38:318–26. doi: 10.1080/07315724.2018.1510348
50. Schürch, CM, Caraccio, C, and Nolte, MA. Diversity, localization, and (Patho) physiology of mature lymphocyte populations in the bone marrow. Blood. (2021) 137:3015–26. doi: 10.1182/blood.2020007592
51. Sun, L, Yin, H, Liu, M, Xu, G, Zhou, X, Ge, P, et al. Impaired albumin function: a novel potential Indicator for liver function damage? Ann Med. (2019) 51:333–44. doi: 10.1080/07853890.2019.1693056
52. Ilavská, S, Horváthová, M, Szabová, M, Nemessányi, T, Jahnová, E, Tulinská, J, et al. Association between the human immune response and body mass index. Hum Immunol. (2012) 73:480–5. doi: 10.1016/j.humimm.2012.02.023
53. Li, Y, Ren, L, and Zou, J. Risk factors and prevention strategies of nosocomial infection in geriatric patients. Can J Infect Dis Med Microbiol. (2019) 2019:6417959–5. doi: 10.1155/2019/6417959
54. Kreitmann, L, Helms, J, Martin-Loeches, I, Salluh, J, Poulakou, G, Pène, F, et al. Icu-acquired infections in immunocompromised patients. Intensive Care Med. (2024) 50:332–49. doi: 10.1007/s00134-023-07295-2
55. Chen, Y, Yang, H, Lan, M, Wei, H, and Chen, Y. The controlling nutritional status score and risk factors associated with malnutrition in patients with acute ischemic stroke. Front Neurol. (2023) 14:14. doi: 10.3389/fneur.2023.1067706
56. Lu, Y, Liu, XY, Chen, YJ, Yu, J, and Yin, SJ. Serum Iron and a (2) ds (2) score in stroke-associated pneumonia. Int J Clin Exp Med. (2015) 8:6163–70.
Keywords: spontaneous intracerebral hemorrhage, stroke-associated pneumonia, nutritional risk screening-2002, controlling nutritional status, prognostic nutritional index, malnutrition
Citation: Zhang X, Kang L, Du P, Xu D, Li H and Jiang Z (2025) Association between nutritional status and pneumonia in patients with spontaneous intracerebral hemorrhage. Front. Nutr. 12:1547655. doi: 10.3389/fnut.2025.1547655
Received: 18 December 2024; Accepted: 26 February 2025;
Published: 12 March 2025.
Edited by:
Yi Chen, Xinjiang Medical University, ChinaReviewed by:
Wencai Liu, Shanghai Jiao Tong University, ChinaCopyright © 2025 Zhang, Kang, Du, Xu, Li and Jiang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lele Kang, MzYwMTE4NTI0QHFxLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.