
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Nutr. , 04 March 2025
Sec. Nutritional Epidemiology
Volume 12 - 2025 | https://doi.org/10.3389/fnut.2025.1523171
Background: Existing studies on the association between the composite dietary antioxidant index (CDAI) and all-cause mortality are controversial. We aimed to analyze the association of CDAI with all-cause mortality, and determine the influence of gender on this association.
Methods: The data of adult participants (age ≥ 18) from the National Health and Nutrition Examination Survey (NHANES) cycles spanning 2001 to 2018 were analyzed. The NHANES-issued identifiers for participants enabled the linkage of data from the NHANES Public Use Linked Mortality File.
Results: The study encompassed a sample of 15,651 individuals. The mean CDAI was 0.52 ± 6.06. The restricted cubic spline revealed that the hazard ratio (HR) of all-cause mortality decreased significantly with increasing CDAI. However, this negative association existed only when the CDAI was less than 5. Multivariate Cox regression analysis showed that compared to the first CDAI quartile, the HR of all-cause mortality was significantly decreased in the third and fourth quartiles (both p < 0.001), and the p-value of the trend test was <0.001. In the subgroup analysis, a notably strong negative association between CDAI and the risk of all-cause mortality was only observed in men (p for interaction <0.001).
Conclusion: Higher CDAI is associated with a reduced risk of all-cause mortality exclusively in adult males, underscoring the substantial influence of gender on this relationship.
Excessive reactive oxygen species (ROS) production leads to oxidative stress, which surpasses the body’s antioxidant capabilities, resulting in cellular and DNA damage (1). Persistent damage from ROS can elevate the likelihood of chronic non-communicable diseases, notably cancer and cardiovascular diseases (CVDs), which are major contributors to global mortality rates. This highlights the importance of managing oxidative stress to reduce these health risks (2). Chronic inflammation plays a pivotal role in the initiation and advancement of several health disorders, such as obesity, CVDs, diabetes, and cancer. It is also linked to an increased risk of all-cause mortality (3, 4).
Dietary antioxidants, such as vitamins A, C and E, zinc, selenium, and carotenoids, can combat non-communicable diseases (NCDs) by neutralizing free radicals and reducing oxidative stress (5). In addition, an antioxidant-rich diet has been demonstrated to substantially decrease levels of inflammation, leading to improved conditions of NCDs and a lowered risk of mortality (6–8). The composite dietary antioxidant index (CDAI) is increasingly being recognized as a potential tool for mitigating oxidative stress and chronic inflammation (9). CDAI is calculated by summing the standardized intake values of six antioxidants: vitamin A, vitamin C, vitamin E, zinc, selenium, and total carotenoids (10, 11). Studies have demonstrated that CDAI is significantly associated with various health outcomes, including delaying aging, reducing the risk of chronic diseases, and improving metabolic health (12–15). As a comprehensive index, CDAI can evaluate the intake of multiple antioxidant substances in an individual’s diet more comprehensively and accurately reflect the overall dietary impact on oxidative stress, rather than focusing solely on single antioxidants. Additionally, CDAI provides a scientific basis for dietary interventions by identifying individuals with insufficient antioxidant intake, thereby promoting healthier dietary practices. In daily life, one can obtain vitamin A and carotenoids from dark green vegetables like spinach and kale, as well as orange vegetables and fruits such as carrots, sweet potatoes, mangoes, tomatoes, and sweet peppers (16). Nuts and seeds, including almonds, sunflower seeds, and Brazil nuts, are rich sources of zinc, vitamin E, and selenium (17). Animal-based foods such as eggs, oysters, and shrimp provide vitamins A and E (18). Citrus fruits like oranges and grapefruits are excellent sources of vitamin C and carotenoids (19). Whole grains and legumes, such as quinoa and lentils, contribute to the intake of zinc and selenium (20).
The association of CDAI with all-cause mortality remains an active area of research. Previous studies conducted in the USA have shown that CDAI is negatively correlated to the risk of all-cause mortality in adults, and with the risk of type 2 diabetes and osteoarthritis among the middle-aged and older adults (13, 21, 22). However, a large cohort study from the UK gave conflicting evidence, and subgroup analysis showed that CDAI was not significantly associated with either non-cancer or cancer-related deaths (23). Evidence from cross-sectional studies also supports this finding (10). Overall, the association between CDAI and all-cause mortality remains controversial, and studies on potential effect modification roles are lacking.
Earlier studies have primarily examined the relationship between CDAI and all-cause mortality but have overlooked the impact of gender differences on this association. Given the significant role that sex differences play in various physiological and pathological processes, our study used the National Health and Nutrition Examination Survey (NHANES) database to analyze the association between CDAI and all-cause mortality, and determine the influence of gender on this association.
The data from nine 2-year cycles (2001–2018) of the NHANES were used. This survey used a stratified, multi-stage probabilistic design to obtain a nationally representative sample of approximately 5,000 individuals per year to assess the nutritional and health status of the population1. All participants had provided written informed consent.
The inclusion criterion for the participants was age ≥ 18 years. The exclusion criteria were as follows: (1) pregnant or lactating women; (2) unreliable dietary recall status, or being below the minimum standard, or breastfeeding; (3) missing CDAI data; (4) missing follow-up data; (5) missing data on education, alcohol use, smoking, hypertension, stroke, or coronary heart disease. A total of 15,651 study subjects were ultimately enrolled (Figure 1).
We used dietary information recorded in NHANES to calculate CDAI. The 24-h dietary recall interview documented information of six dietary antioxidants, including vitamin A, C and E, zinc, selenium, and total carotenoids (α-carotene, β-carotene, β-cryptoxanthin, lycopene, lutein, and zeaxanthin) (10, 11). Each participant was interviewed twice for the dietary intake, initially at the baseline health check and then over the next 3–10 days. The dietary recall data from both occasions were assessed for reliability and the mean daily intake was calculated. The consumption of each antioxidant was normalized by first subtracting the average value and then dividing by the standard deviation. The resulting normalized values were summed to calculate the CDAI (24).
To accurately assess all-cause mortality in the follow-up population, the NHANES serial numbers were used to link the data extracted from the NHANES Public Use Linked Mortality File on December 31, 2019 with the NHANES database. The relevant causes of death were defined using the 10th Revision of the National Statistical Classification of Diseases (ICD-10), and deaths from any cause were considered to be all-cause mortality (25).
Age, gender, race, and education level were self-reported by participants at the baseline health examination. Smoking status was derived from self-reported responses to the question “Do you currently smoke?.” The responses were categorized as “not at all,” “some days” and “every day.” The frequency of alcohol consumption in the previous year was also categorized as “not at all,” “some days” and “every day.” Diabetes mellitus was defined as positive diagnosis by a physician, current usage of glucose-lowering medication, fasting blood glucose ≥126 mg/dL, and glycated hemoglobin ≥6.5% (26). Hypertension was defined as diagnosis of high blood pressure by a physician, current usage of anti-hypertensive medications, systolic blood pressure ≥ 130 mmHg, and a diastolic blood pressure ≥ 80 mmHg (27). Stroke and coronary heart disease were defined according to clinical diagnosis (28).
The mean values along with their standard deviations (denoted as mean ± SD) were used to illustrate the numerical data, whereas the categorical data was depicted using percentage values. One-way ANOVA was used to compare population means and Pearson’s chi-square test was used to compare population rates. The shape of the association between CDAI and all-cause mortality was described by a 5-node (5.00th, 27.50th, 50.00th, 72.50th, 95.00th) restricted natural cubic splines model, and the hazard ratio (HR) and significance of the relationship between CDAI and all-cause mortality were further analyzed by multivariate Cox regression models. We also performed sensitivity analyses according to CDAI level tripartite grouping.
The survival rates of male and female participants grouped in the different CDAI quartiles were analyzed by plotting Kaplan–Meier curves. The potential effect of gender on the association between CDAI and all-cause mortality was determined by multiplicative interaction analyses. R software version 4.4.1 was used for all statistical analyses, and p-value <0.05 was considered statistically significant.
A total of 15,651 participants were included in the final data analysis. 9,427 (60.23%) were men, with a mean BMI of 29.00 ± 6.72. Some day drinker were 11,386 (72.75%), while some day smoker were 1,318 (8.42%). 2,846 (18.18%) were diabetes, 8,413 (53.75%) were hypertensive, 973 (6.22%) were coronary heart disease, 754 (4.82%) were Stroke, and 3,030 (19.36%) died of any cause. The mean values of vitamin A and selenium were 0.61 ± 0.81 (mg) and 0.11 ± 0.06 (mg), respectively, the mean values of vitamin C, vitamin E and zinc were 81.11 ± 94.44 (mg); 7.45 ± 6.04 (mg); 11.56 ± 9.20 (mg), respectively. The average CDAI of the study population was 0.52 ± 6.06. Participants in the highest CDAI quartile had fewer women, lower BMI, lower diabetes, hypertension, stroke, and all-cause mortality. The baseline data of the patients in the different CDAI quartiles are summarized in Table 1.
Multivariate Cox regression analysis was performed to quantify the association between CDAI and the risk of all-cause mortality. Using the first quartile of CDAI as the reference group, we observed a significant decrease in the HR of all-cause mortality in the third and fourth quartiles of the study participants (both p < 0.05). In addition, the p-value of the trend test was less than 0.05. The data are summarized in Table 2. Sensitivity analysis showed that compared with the low (−8.375 ~ −2.5033) group, the HR and p values of the middle (−2.5029 ~ 1.3304) and high (1.3305 ~ 195.5575) groups were 0.88 (0.81, 0.96), p = 0.006; 0.81 (0.74, 0.89), p < 0.001, and the trend test p-value was <0.001, as shown in Supplementary Table 1.
Restricted natural cubic splines showed a gradual decrease in the HR of all-cause mortality with the increase in CDAI. However, mortality due to all causes was not reduced by the CDAI at values higher than 5, indicating a saturation effect (see Figure 2).
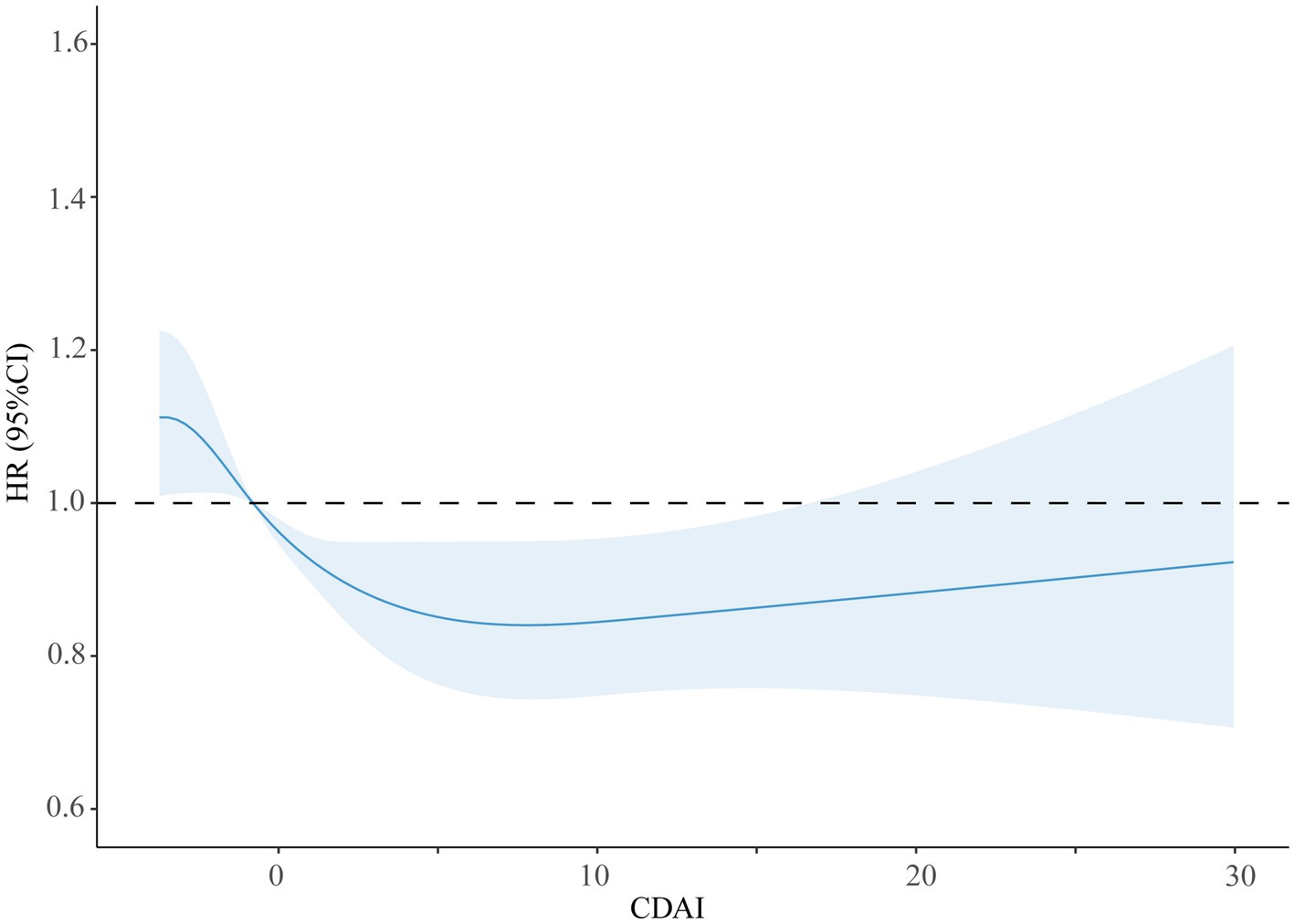
Figure 2. Restricted cubic splines of the association between CDAI and the risk of all-cause mortality.
Subgroup analysis was also performed to explore the potential impact of gender on the association. As shown in Table 3, women were not significantly associated with CDAI and all-cause mortality. Sensitivity analyses yielded similar results, the relationship was only observed in men (Supplementary Table 2). Furthermore, we also observed a multiplication interaction (p-value for interaction <0.001). Consistent with this, the survival probability was higher in the third and fourth quartile groups of CDAI among males, and the log-rank test showed a statistically significant difference (p < 0.001). However, no significant difference was observed in the survival probabilities of the different quartile groups among females (p = 0.940). The data is summarized in Figure 3.

Figure 3. Kaplan–Meier survival curves showing cumulative probability of all-cause mortality in the CDAI quartile groups among (A) female and (B) male participants.
We found a negative association between CDAI and the risk of all-cause mortality in the adult population of USA. However, the association was not linear, and CDAI >5 did not cause a reduction in the risk of all-cause mortality. Furthermore, we observed this negative relationship only in men, indicating a significant impact of gender.
Previous studies have also shown a significant negative association between CDAI and all-cause mortality (13, 21, 29). However, a study based on UK Biobank data showed that antioxidant supplementation did not have a significant impact on cancer or non-cancer deaths (23). In addition, CDAI less than −0.19 was significantly associated with all-cause mortality in 45-year-olds in the United States (22). Likewise, CDAI demonstrated a protective association with CVD-related mortality (13). CDAI has been shown to have no significant association with all-cause mortality in certain specific populations, for example, patients with metabolic syndrome (MetS) (10). However, the applicability of this association in other specific populations remains to be explored in further studies. The inconsistencies among these studies may be due to the different characteristics of the populations and the differences in covariates. Our findings are in line with most previous studies.
CDAI and all-cause mortality are linked by specific biological mechanisms, but the mechanisms are not fully understood, although some studies have put forth several hypotheses. Normal antioxidant defense mechanisms in the body result in a disrupted balance between the two due to excessive production of free radicals, which eventually manifest as oxidative stress. Given that antioxidants slow down the aging process by reducing free radical damage to cells (30), the results of several studies indicate that high CDAIs are associated with greater longevity and reduced death rates from all causes. Damage to cells by free radicals resulting from oxidative stress can be relieved by the ingestion of foods rich in antioxidants (31). Chronic inflammation also plays an important role in the development and progression of numerous diseases, such as CVDs, cancer, and diabetes, and is also strongly associated with an elevated risk of all-cause mortality (5, 32). The food can significantly reduce the levels of inflammatory markers and mitigate the symptoms of chronic diseases, resulting in lower mortality risk (33–35). The DNA repair pathway is another potential mechanistic link between CDAI and mortality risk. Antioxidants protect DNA from free radical damage and repair DNA lesions, which is able to prevent DNA related diseases to some extent (36). Diabetes- and CVD-related deaths are important components of all-cause mortality. CDAI may reduce the risk of type 2 diabetes by improving insulin resistance and glucose metabolism (37). CVDs, although not directly classified as metabolic diseases, can be caused or exacerbated by metabolic abnormalities. Increasing the intake of antioxidants can reduce the risk of CVD-related deaths by regulating plasma REDOX status and neutralizing the reactive oxygen and nitrogen species (38). Notably, the association between CDAI and risk of all-cause mortality in the present study was only demonstrated in adults with CDAI <5, however, the exact dose that does not produce any promotion of oxidation is unknown, and the exact mechanism remains to be elucidated.
The significant negative association between CDAI and all-cause mortality was only observed in men, which was also reflected in the survival analysis. Furthermore, there was a significant multiplicative interaction. This is the first study to demonstrate that gender influences CDAI’s association with all-cause mortality. The most reasonable explanation is the differences in the levels of sex hormones between men and women, which may influence metabolism and antioxidant action. For example, estrogen is known to lower ROS production and oxidative stress by activating antioxidant pathways downstream of its specific receptors (estrogen receptors alpha and beta), which may reduce the benefit of dietary antioxidants on the risk of all-cause mortality (39, 40). Second, All-cause mortality is a significant long-term prognostic event for CVDs, and it is associated with a higher incidence of cardiovascular events in men compared to women (41). Given the substantial efficacy of CDAI in reducing deaths due to cardiovascular disease, this may be another reason why CDAI have a stronger protective effect in men (14).
This study presents several significant clinical implications. First, we observed a potential reduction in the risk of all-cause mortality among men associated with dietary antioxidant intake, highlighting the importance of gender differences in health interventions. The findings suggest that men should prioritize the consumption of foods rich in vitamins A, C, and E, as well as selenium and zinc (such as green vegetables, nuts, fish, etc.), to mitigate their risk of all-cause mortality. Instead, health interventions aimed at female populations should formulate comprehensive strategies that do not depend exclusively on diets rich in antioxidants. Second, the gender specificity inherent in CDAI may facilitate the establishment of mortality risk assessment models. Future studies could effectively identify individuals at high risk for death through CDAI and incorporate it into male health examination programs alongside other clinical indicators for a thorough evaluation. Finally, given that comorbidity is a recognized risk factor for all-cause mortality among older adults, it is advisable for clinicians to recommend an appropriate dietary regimen based on CDAI for male patients suffering from chronic conditions such as hypertension, hyperlipidemia, CVDs, and others. Future research should focus on conducting further clinical trials and mechanism-related investigations to develop and refine actionable public health strategies aimed at addressing the challenges posed by an aging population; thereby effectively extending healthy life expectancy.
We included a large number of covariables in the multifactor analysis to minimize any confounding bias. Furthermore, NHANES has a standardized method of collecting analytical data, along with strict quality control measures to ensure accuracy and reliability. Therefore, the information bias in this study was controlled within a reasonable range. Finally, the NHANES data is derived from a large sample that is representative of the population and can accurately reflect the health status.
There are some limitations in this study. First, the CDAI was derived from 24-h dietary recall interviews. Yet, the potential for recall bias and variability in daily dietary habits could introduce some imprecision into the findings. Second, it is unclear whether the dietary intake data collected at baseline changed during subsequent follow-up, which may have contributed to some information bias regarding CDAI. Third, although we included many confounding factors in the statistical analysis, we did not consider other compounds with antioxidant potential (e.g., phenolics and flavonoids), nor did we consider data on the consumption of supplements and related medications, due to a lack of data in the NHANES database. Fourth, this study was unable to further explore the independent effects of the six dietary antioxidants in reducing all-cause mortality due to a lack of relevant data as well as technical reasons. Finally, this study is only representative of U.S. adults, and it is necessary to verify the generalizability of the findings across culturally and geographically diverse populations.
In conclusion, CDAI showed a significant negative association with the risk of all-cause mortality. However, the protective effect of CDAI was not observed when it exceeded 5. In addition, CDAI levels were negatively related to all-cause mortality risk only in men and not in women when gender was a modifier.
Publicly available datasets were analyzed in this study. This data can be found at: https://www.cdc.gov/nchs/nhanes/.
The studies involving humans were approved by the NHANES and the Research Ethics Review Board of the NCHS. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
LD: Writing – original draft, Data curation, Supervision, Visualization. RZ: Writing – original draft, Data curation. JW: Writing – review & editing, Data curation. SH: Writing – review & editing, Supervision. WW: Writing – review & editing, Conceptualization, Supervision.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This work was financially supported by Jiangxi Provincial Natural Science Foundation (20232BAB206143) and Guiding Science and Technology Projects of Ji'an city (20233–023456).
We thank all the NHANES study participants for their assistance. We also thank Miss Yiwa Liu of Guangdong Medical University as an independent third-party statistical analyst for validating the results of our data analysis.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fnut.2025.1523171/full#supplementary-material
1. Afzal, S, Abdul Manap, AS, Attiq, A, Albokhadaim, I, Kandeel, M, and Alhojaily, SM. From imbalance to impairment: the central role of reactive oxygen species in oxidative stress-induced disorders and therapeutic exploration. Frontiers in. Pharmacology. (2023) 14:1269581.4.4. doi: 10.3389/fphar.2023.1269581
2. Gwozdzinski, K, Pieniazek, A, and Gwozdzinski, L. Reactive oxygen species and their involvement in red blood cell damage in chronic kidney disease. Oxidative Med Cell Longev. (2021) 2021:6639199. doi: 10.1155/2021/6639199
3. Zhao, L, Hu, H, Zhang, L, Liu, Z, Huang, Y, Liu, Q, et al. Inflammation in diabetes complications: molecular mechanisms and therapeutic interventions. MedComm. (2024) 5:e516. doi: 10.1002/mco2.516
4. Yu, Y-C, Paragomi, P, Wang, R, Jin, A, Schoen, RE, Sheng, L-T, et al. Composite dietary antioxidant index and the risk of colorectal cancer: findings from the Singapore Chinese health study. Int J Cancer. (2022) 150:1599–1608. 5.7. doi: 10.1002/ijc.33925
5. Aboukhater, D, Morad, B, Nasrallah, N, Nasser, SA, Sahebkar, A, Kobeissy, F, et al. Inflammation and hypertension: underlying mechanisms and emerging understandings. J Cell Physiol. (2023) 238:1148–1159. 4.5. doi: 10.1002/jcp.31019
6. Possa, LO, Hinkelman, JV, Santos, CAD, Oliveira, CA, Faria, BS, Hermsdorff, HHM, et al. Association of dietary total antioxidant capacity with anthropometric indicators, C-reactive protein, and clinical outcomes in hospitalized oncologic patients. Nutrition. (2021) 90:3.2. doi: 10.1016/j.nut.2021.111359
7. Xu, X, Hall, J, Byles, J, and Shi, Z. Dietary pattern, serum magnesium, ferritin, C-reactive protein and anaemia among older people. Clin Nutr. (2017) 36:444–451. 6.6. doi: 10.1016/j.clnu.2015.12.015
8. Yang, M, Chung, S-J, Floegel, A, Song, WO, Koo, SI, and Chun, OK. Dietary antioxidant capacity is associated with improved serum antioxidant status and decreased serum C-reactive protein and plasma homocysteine concentrations. Eur J Nutr. (2013) 52:1901–11. doi: 10.1007/s00394-012-0491-5
9. Wu, Y, Cheng, S, Lei, S, Li, D, Li, Z, and Guo, Y. The association between the composite dietary antioxidant index and frailty symptoms: mediating effects of oxidative stress. Clinical interventions in. Aging. (2024) 19:3.5:163–73. doi: 10.2147/CIA.S448354
10. Zhou, Q, Zhou, L, Chen, X, Chen, Q, and Hao, L. Composite dietary antioxidant index is associated with reduced prevalence of metabolic syndrome but not mortality in metabolic syndrome: results from NHANES 2001-2018. Prev Med Rep. (2024) 41:2.4:102704. doi: 10.1016/j.pmedr.2024.102704
11. Wu, D, Wang, H, Wang, W, Qing, C, Zhang, W, Gao, X, et al. Association between composite dietary antioxidant index and handgrip strength in American adults: Data from National Health and nutrition examination survey (NHANES, 2011-2014). Front Nutr. (2023) 10:4.0:1147869. doi: 10.3389/fnut.2023.1147869
12. He, H, Chen, X, Ding, Y, Chen, X, and He, X. Composite dietary antioxidant index associated with delayed biological aging: a population-based study. Aging. (2024) 16:3.9:15–27. doi: 10.18632/aging.205232
13. Wang, L, and Yi, Z. Association of the Composite dietary antioxidant index with all-cause and cardiovascular mortality: a prospective cohort study. Front Cardiovasc Med. (2022) 9:2.8. doi: 10.3389/fcvm.2022.993930
14. Jiang, Y, and Shen, Y. Composite dietary antioxidant index is inversely and non-linearly associated with cardiovascular disease, atherosclerotic cardiovascular disease, and cardiovascular mortality in people with dyslipidemia: evidence from NHANES 2001-2018. Front Nutr. (2024) 11:4.0. doi: 10.3389/fnut.2024.1478825
15. Miao, X, Li, B, Zhu, Z, and Yang, T. Sex differences in the association between composite dietary antioxidant index and hyperlipidemia: insights from NHANES. PLoS One. (2025) 20:e0316130. doi: 10.1371/journal.pone.0316130
16. Krinsky, NI, and Johnson, EJ. Carotenoid actions and their relation to health and disease. Mol Asp Med. (2005) 26:8.7:459–516. doi: 10.1016/j.mam.2005.10.001
17. Thomson, CD, Chisholm, A, McLachlan, SK, and Campbell, JM. Brazil nuts: an effective way to improve selenium status. Am J Clin Nutr. (2008) 87:6.5:379–84. doi: 10.1093/ajcn/87.2.379
18. Hambidge, M. Human zinc deficiency. J Nutr. (2000) 130:3.7:1344S–9S. doi: 10.1093/jn/130.5.1344S
19. Carr, AC, and Vissers, MCM. Synthetic or food-derived vitamin C--are they equally bioavailable? Nutrients. (2013) 5:4.8:4284–304. doi: 10.3390/nu5114284
20. Rayman, MP. Selenium and human health. Lancet. (2012) 379:98.4:1256–68. doi: 10.1016/S0140-6736(11)61452-9
21. Zhang, Y, Duan, Z, Lu, H, Lu, G, Fu, Y, Li, G, et al. Physical activity modifies the association of the composite dietary antioxidant index with all-cause mortality in the US osteoarthritis population. Frontiers in. Public Health. (2023) 11:3.0. doi: 10.3389/fpubh.2023.1297245
22. Luo, J, Xu, X, Sun, Y, Lu, X, and Zhao, L. Association of composite dietary antioxidant index with depression and all-cause mortality in middle-aged and elderly population. Sci Rep. (2024) 14:9809. 3.8. doi: 10.1038/s41598-024-60322-0
23. Behrendt, I, Eichner, G, and Fasshauer, M. Association of antioxidants use with all-cause and cause-specific mortality: a prospective study of the UK biobank. Antioxidants. (2020) 9:6.0. doi: 10.3390/antiox9121287
24. Wright, ME, Mayne, ST, Stolzenberg-Solomon, RZ, Li, Z, Pietinen, P, Taylor, PR, et al. Development of a comprehensive dietary antioxidant index and application to lung cancer risk in a cohort of male smokers. Am J Epidemiol. (2004) 160:5.0:68–76. doi: 10.1093/aje/kwh173
25. World Health Organization. (2015) International statistical classification of diseases and related health problems. 10th revision, Fifth edition, 2016. World Health Organization. Available at: https://iris.who.int/handle/10665/246208
26. Qiu, Z, Chen, X, Geng, T, Wan, Z, Lu, Q, Li, L, et al. Associations of serum carotenoids with risk of cardiovascular mortality among individuals with type 2 diabetes: results from NHANES. Diabetes Care. (2022) 45:14.8:1453–61. doi: 10.2337/dc21-2371
27. Cai, Y, Chen, M, Zhai, W, and Wang, C. Interaction between trouble sleeping and depression on hypertension in the NHANES 2005-2018. BMC Public Health. (2022) 22:481. 3.5. doi: 10.1186/s12889-022-12942-2
28. He, J, Zhu, Z, Bundy, JD, Dorans, KS, Chen, J, and Hamm, LL. Trends in cardiovascular risk factors in US adults by race and ethnicity and socioeconomic status, 1999-2018. JAMA. (2021) 326:63.1:1286–98. doi: 10.1001/jama.2021.15187
29. Yang, C, Yang, Q, Peng, X, Li, X, and Rao, G. Associations of composite dietary antioxidant index with cardiovascular disease mortality among patients with type 2 diabetes. Diabetol Metab Syndr. (2023) 15:131. 3.4. doi: 10.1186/s13098-023-01109-7
30. Mehdi, MM, Solanki, P, and Singh, P. Oxidative stress, antioxidants, hormesis and calorie restriction: the current perspective in the biology of aging. Arch Gerontol Geriatr. (2021) 95:3.5:104413. doi: 10.1016/j.archger.2021.104413
31. Lobo, V, Patil, A, Phatak, A, and Chandra, N. Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev. (2010) 4:118–26. doi: 10.4103/0973-7847.70902
32. Leuti, A, Fazio, D, Fava, M, Piccoli, A, Oddi, S, and Maccarrone, M. Bioactive lipids, inflammation and chronic diseases. Adv Drug Deliv Rev. (2020) 159:15.2:133–69. doi: 10.1016/j.addr.2020.06.028
33. Vahid, F, Rahmani, D, and Davoodi, SH. The correlation between serum inflammatory, antioxidant, glucose handling biomarkers, and dietary antioxidant index (DAI) and the role of DAI in obesity/overweight causation: population-based case-control study. Int J Obes. (2005) 45:4.2:2591–9. doi: 10.1038/s41366-021-00944-w
34. Wu, J-R, Song, EK, Moser, DK, and Lennie, TA. Dietary antioxidant insufficiency is associated with increased inflammatory markers and poorer health-related quality of life in patients with heart failure. The. J Cardiovasc Nurs. (2023) 38:6–12. doi: 10.1097/JCN.0000000000000912
35. Liu, Z, Li, J, Chen, T, Zhao, X, Chen, Q, Xiao, L, et al. Association between dietary antioxidant levels and chronic obstructive pulmonary disease: a mediation analysis of inflammatory factors. Front Immunol. (2023) 14:5.7. doi: 10.3389/fimmu.2023.1310399
36. Beetch, M, Harandi-Zadeh, S, Shen, K, Lubecka, K, Kitts, DD, O'Hagan, HM, et al. Dietary antioxidants remodel DNA methylation patterns in chronic disease. Br J Pharmacol. (2020) 177:1382–408. doi: 10.1111/bph.14888
37. Franzini, L, Ardigò, D, and Zavaroni, I. Dietary antioxidants and glucose metabolism. Curr Opin Clin Nutr Metab Care. (2008) 11:471–6. doi: 10.1097/MCO.0b013e328303be79.
38. Wada, Y, Sato, Y, Miyazaki, K, Takeda, Y, and Kuwahata, M. The reduced/oxidized state of plasma albumin is modulated by dietary protein intake partly via albumin synthesis rate in rats. Nutr Res. (2017) 37:46–57. doi: 10.1016/j.nutres.2016.12.003
39. Xiang, D, Liu, Y, Zhou, S, Zhou, E, and Wang, Y. Protective effects of estrogen on cardiovascular disease mediated by oxidative stress. Oxidative Med Cell Longev. (2021) 2021:5523516. doi: 10.1155/2021/5523516
40. Aryan, L, Younessi, D, Zargari, M, Banerjee, S, Agopian, J, Rahman, S, et al. The role of estrogen receptors in cardiovascular disease. Int J Mol Sci. (2020) 21:4.9. doi: 10.3390/ijms21124314
41. Walli-Attaei, M, Rosengren, A, Rangarajan, S, Breet, Y, Abdul-Razak, S, Sharief, WA, et al. Metabolic, behavioural, and psychosocial risk factors and cardiovascular disease in women compared with men in 21 high-income, middle-income, and low-income countries: an analysis of the PURE study. Lancet. (2022) 400:811–21. doi: 10.1016/S0140-6736(22)01441-6
Keywords: all-cause mortality, antioxidant index, composite dietary, gender difference, NHANES
Citation: Duan L, Zeng R, Wang J, Hu S and Wang W (2025) Gender difference in the association between composite dietary antioxidant index and all-cause mortality. Front. Nutr. 12:1523171. doi: 10.3389/fnut.2025.1523171
Received: 05 November 2024; Accepted: 19 February 2025;
Published: 04 March 2025.
Edited by:
Camelia Munteanu, University of Agricultural Sciences and Veterinary Medicine of Cluj-Napoca, RomaniaReviewed by:
Alessandro De Oliveira, Universidade Federal de São João del-Rei, BrazilCopyright © 2025 Duan, Zeng, Wang, Hu and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Weiye Wang, d3d5dGVjaEBmb3htYWlsLmNvbQ==
†These authors have contributed equally to this work and share first authorship
‡ORCID: Weiye Wang, orcid.org/0000-0001-6540-1364
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.