
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Nutr. , 13 March 2025
Sec. Nutritional Epidemiology
Volume 12 - 2025 | https://doi.org/10.3389/fnut.2025.1519612
This article is part of the Research Topic Preventative Medicine: Nutritional and Lifestyle Interventions for Healthy Ageing and Chronic Diseases View all 37 articles
 Hongman Li1,2
Hongman Li1,2 Long Li1,2*
Long Li1,2*Background: Cardiorenal syndrome (CRS) represents a burgeoning global health concern characterized by its increasing prevalence. Life’s Crucial 9 (LC9), an innovative tool for cardiovascular health assessment, and the Dietary Inflammatory Index (DII), which quantifies diet’s impact on body inflammation, have not been previously studied in conjunction regarding their association with CRS.
Objective: This study aims to explore the relationship between LC9 and CRS, using data from the National Health and Nutrition Examination Survey (NHANES), and to examine whether DII serves as a mediator in this association.
Methods: This research included data from 25,792 NHANES participants spanning from 2005 to 2018. The study leverages the dataset’s comprehensive representativeness and robust statistical power to ensure generalizable and reliable findings. We employed weighted logistic regression to evaluate the association between LC9 scores and CRS presence, conducted subgroup analyses, and performed mediation analysis to investigate the role of DII.
Results: Our analysis demonstrated a significant inverse relationship between LC9 and CRS. Upon controlling for confounders, each 10-point rise in LC9 correlates with a 26% reduction in CRS prevalence (p < 0.001). Additionally, stratifying LC9 into tertiles with T1 as the reference group revealed that T2 (OR = 0.59, 95% CI = 0.48–0.72, p < 0.001) and T3 (OR = 0.57, 95% CI = 0.38–0.88, p < 0.001) exhibited a strong negative correlation trend. The dose–response curve illustrates a linear relationship between LC9 and CRS; as LC9 increases, the occurrence of CRS decreases. DII shows a significant positive connection with CRS (p < 0.001), but DII indicates a decreasing trend when LC9 rises (β = −0.65, p < 0.001). Mediation analysis reveals that DII mediates the association between LC9 and CRS, with a mediation proportion of 12.5% (p < 0.001).
Conclusion: The findings indicate a robust inverse correlation between LC9 scores and CRS incidence, with DII is associated with this relationship. This suggests potential preventive strategies against CRS through lifestyle modifications guided by LC9.
Cardiorenal syndrome (CRS) refers to a clinical syndrome caused by impaired interactions between the heart and kidneys (1). As the population ages and the burden of chronic diseases grows, the incidence and clinical impact of CRS are also increasing. CRS adversely impacts patients’ quality of life and substantially escalates the utilization of medical resources and associated healthcare costs. According to the 2009 Acute Dialysis Quality Initiative, it is divided into five subtypes (2), involving the interactions between acute and chronic heart and kidney conditions. Type 1 and 2 CRS arise from cardiogenic factors affecting renal function, whereas Type 3 and 4 are due to nephrogenic factors impacting cardiac function. Type 5 CRS includes cardiac and renal dysfunctions caused by systemic diseases. The global prevalence of CRS has increased (3), especially among heart failure patients, where the prevalence exceeds 50% (4). Cardiovascular-related deaths account for nearly half of all deaths among individuals with chronic kidney disease (CKD) (5). Furthermore, the simultaneous occurrence of heart failure and kidney disease heightens this risk (4, 6, 7). Between 2011 and 2020, the United States saw 97,135 deaths related to cardiorenal diseases in adults aged 15 and older (8). This presents considerable management difficulties for public health and healthcare systems. Current research has revealed the complexity of the pathophysiological mechanisms of CRS, involving various factors such as neuroendocrine activation, inflammatory pathways, oxidative stress, and biomechanical changes (9). These mechanisms are not only interconnected with each other but also interact with multi-organ dysfunction, leading to a vicious cycle of disease. To reduce the global disease burden and improve public health levels, identifying the potential risk factors of CRS through epidemiological research is crucial.
In 2010, the American Heart Association (AHA) introduced the public health initiative Life’s Simple 7 (LS7), designed to enhance cardiovascular health (CVH) by addressing seven determinants: smoking, weight, total cholesterol, blood glucose, physical activity, and food (10). Life’s Essential 8 (LE8) is a revised and more extensive health evaluation framework introduced by the AHA in 2022. This model incorporates the essential element of “sleep” into the prior LS7, highlighting the significance of enough and high-quality sleep for CVH (11). Current depression is a prevalent and serious mental health disorder, impacting approximately 300 million people globally. The World Health Organization predicts that by 2030, major depressive disorder will become the leading factor in the global burden of chronic diseases (12). Therefore, many studies emphasize the importance of depression in the prevention of cardiovascular disease (CVD) (13, 14) and introduce another new indicator, Life’s Crucial 9 (LC9), which integrates mental health into LE8 (15). The score range for these nine components is 0 to 100, and the total score for LC9 is determined as the arithmetic mean of the 9 indications.
Currently, there is no research on LC9 and CRS, but previous evidence suggests that higher LE8 levels are associated with a lower likelihood of CVD, CKD, and their related mortality rates (16–19). Additionally, research has established a correlation between depression and the incidence of CVD and CKD (13, 20). Alexander A. Huang and colleagues used data from the NHANES database for the years 2017–2020 to identify risk factors for coronary artery disease using the powerful machine learning model XGBoost. Out of a total of 684 features, 58 were found to be significant in univariate analysis (p < 0.0001), including components of LC9 such as blood pressure, blood lipids, blood sugar, smoking, obesity, depression, and diet (21). Therefore, we speculate that the LC9 guidelines are closely related to the prevention and management of CRS. By adhering to the LC9 guidelines, the risk of CRS can be effectively reduced, thereby improving the quality of life for patients.
Systemic inflammation is widely recognized as a critical factor in the onset and progression of CRS (9). Additionally, more and more studies are currently confirming that diet significantly influences the regulation of systemic inflammation (22). Dietary anti-inflammatory components such as omega-3 fatty acids (primarily found in deep-sea fish), high-quality dietary fibre, antioxidants (such as vitamin E, vitamin C, and beta-carotene), and polyphenols (abundant in fruits, vegetables, nuts, tea, and coffee) can reduce the levels of inflammation markers in the body. These nutrients work by reducing oxidative stress and regulating immune function to suppress inflammation (23). Additionally, consuming a diet rich in monounsaturated and polyunsaturated fatty acids, high in potassium, and low in sodium is associated with improvements in blood lipids, blood pressure, and blood sugar levels, thereby being related to a reduced risk of cardiovascular diseases and chronic kidney disease (24). The Dietary Inflammatory Index (DII) is a scientific metric introduced in 2014, derived from an extensive study of scientific literature, to evaluate the influence of diet on inflammatory status. It evaluates various food components according to their enhancing or suppressing effects on inflammatory markers (IL-1β, IL-4, IL-6, IL-10, TNF-α, and C-reactive protein) (25). A higher DII score signifies a diet with pronounced pro-inflammatory characteristics, whereas a lower or negative score denotes anti-inflammatory features in the diet. Since its inception, the DII has been extensively utilized in epidemiological and clinical research to investigate the correlation between dietary inflammatory potential and diverse health outcomes (26, 27). A healthy diet occupies an important position in LC9, emphasizing the intake of foods rich in fruits, vegetables, whole grains, low-fat proteins, and healthy fats. This dietary approach is usually associated with a low DII score. Nutrition significantly influences CVD and CKD. Unhealthy dietary patterns (most notably the high-fat, high-calorie Western diet) can exacerbate chronic low-grade inflammation, thereby promoting abnormal immune activation and contributing to the onset of CVD (28). A meta-analysis indicates that a 1-point increase in the DII score is related to an 8% rise in the risk and mortality linked to CVD, highlighting the substantial influence of an anti-inflammatory diet on CVD prevention (29). The inflammation associated with CKD has also been confirmed to be related to DII (30). At present, there is an absence of extensive epidemiological research capable of thoroughly investigating the pathophysiology of CRS to inform preventative and therapy efforts (31). Consequently, we undertook this study, employing a large cross-sectional methodology for the first time to examine the relationship between LC9 and the occurrence of CRS, while also investigating whether this relationship is mediated by DII, with the objective of determining whether enhancing LC9 is associated with a reduced occurrence of CRS.
The cross-sectional data comes from the National Health and Nutrition Examination Survey (NHANES), a series of research projects managed by the National Center for Health Statistics (NCHS). This survey intends to evaluate the health and nutritional status of the U.S. population by collecting health-related data to help formulate public health policies, guide disease prevention programs, and improve health services. This research employed data from seven cycles of the NHANES, covering the years 2005 to 2018, with a total of 70,190 participants. Individuals under 20 are still developing, with differences in metabolism, dietary habits, and physical activity compared to adults. Using 20 years as a cutoff helps focus on chronic disease risk factors (32). Pregnant women have distinct nutritional needs and metabolic changes, which affect their diet and health outcomes, and medications or supplements taken during pregnancy can confound results (33). This classification is commonly used in public health studies, allowing for easier comparison with other research (34). As a result, we excluded participants under 20, pregnant women (31,141), those missing LC9 and DII data (13,226), and those with missing CRS data, resulting in 25,792 participants for the final analysis (Figure 1). NHANES received approval from NCHS, and all subjects gave their informed consent. The information utilized for this investigation can be found at (https://www.cdc.gov/nchs/nhanes/index.htm).
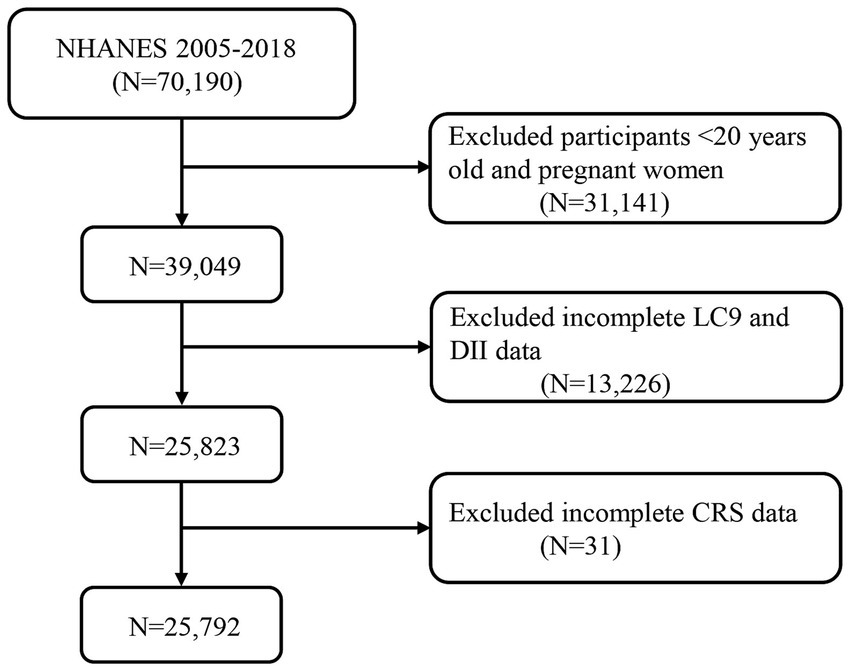
Figure 1. A flow diagram of eligible participant selection in the national health and nutrition examination survey. LC9, life’s crucial 9; DII, dietary inflammatory index; CRS, cardiorenal syndrome.
LC9 adds depression as an evaluation metric based on the LE8 score. LE8 consists of 4 health behaviors (diet, physical activity, nicotine exposure, and sleep duration) and 4 health factors (body mass index, non-high-density lipoprotein cholesterol, blood glucose, and blood pressure). The calculation method for the LE8 score of each indicator has been detailed in previous publications (35). In short, each of the 8 CVH indicators is allocated a score from 0 to 100, which is determined by expert panel members using a modified Delphi method based on health outcomes and risk associations. The overall LE8 score is the average of the 8 indicators. The increased depression score of LC9 is calculated based on the Patient Health Questionnaire-9 (PHQ-9) score, which is a validated structured questionnaire used for depression screening. A higher PHQ-9 score indicates the presence of a higher level of depressive symptoms. The depression scores are categorized as 100, 75, 50, 25, and 0, corresponding to PHQ-9 scores of 0–4, 5–9, 10–14, 15–19, and 20–27, respectively. The total LC9 score is calculated as the arithmetic mean of the 9 indicators (36). Detailed classification and scoring methods can be found in Supplementary Tables 1, 2.
The Mobile Examination Center recorded the types and quantities of food and beverages consumed by participants in the 24 h preceding the interview, which were used to calculate the DII (27). In this study, 28 out of 45 food parameters were included: alcohol, β-carotene, caffeine, carbohydrate, cholesterol, energy, total fat, fiber, folic acid, iron, magnesium, monounsaturated fatty acids, polyunsaturated fatty acids, n-3 fatty acids, n-6 fatty acids, protein, saturated fat, selenium, zinc, vitamin A, B1, B2, B3, B6, B12, C, D, and E (37). Shivappa and his colleagues indicated that utilizing no more than 30 dietary characteristics remains adequate for maintaining the prediction validity of the DII for diet-related inflammation (38). The DII calculation relies on dietary intake statistics, which are correlated with a geographically representative global database that offers a reliable estimate of the mean and standard deviation for each parameter (Supplementary material).
Based on prior research, we define CRS as people concurrently diagnosed with both CVD and CKD (39). CVD is identified through self-reported diagnoses of coronary heart disease, angina, congestive heart failure, myocardial infarction, or cerebrovascular accident. Chronic Kidney Disease (CKD) is defined by an estimated glomerular filtration rate (eGFR) of less than 60 mL/min/1.73 m2. We employed the CKD-Epidemiology Collaboration (EPI) equation to compute eGFR (40).
We included age, gender, race, married/live with partner, education level, poverty income ratio, smoking, drinking, hypertension, diabetes, and hyperlipidemia as covariates. Specific classifications can be found in Supplementary Table 3.
All data in this study were subjected to statistical analysis utilizing R software (version 4.3.1). We utilized weights for guaranteeing that our survey information is nationally representative. In descriptive statistical analysis, participants were classified into non-CRS and CRS groups, using mean ± standard deviation (SD) to describe continuous variables and applying the student’s t-test to evaluate differences between the two groups. For categorical variables, we utilize frequency and percentage descriptions, employing the chi-square test for comparison analysis.
We utilized a weighted multivariable logistic regression model to investigate the relationship between LC9 and CRS, with LC9 being the tertile. Further calculate the p-values for the trend test and linear trend to determine their relationship. This research established: Model 1, the unadjusted original model. Model 2 adjusted for age, gender, education level, marital status, and race. Model 3 further adjusted for income, smoking, drinking, hypertension, diabetes, and hyperlipidemia. We stratified LC9 and DII into tertiles, employing the initial tertile as the reference group for the trend analysis. A smooth curve fitting method has been used for analyzing the possible linear correlation of LC9 and CRS. Furthermore, odds ratios (ORs) were computed with each 10-point increment in LC9, and analyses of subgroups were performed. To verify the robustness of the results, this study performed sensitivity analysis using multiple imputation with chained equations (MICE). The main analysis was repeated using five imputed datasets to address missing data for LC9, DII, and CRS.
The R software’s “mediation” package is employed to assess indirect, direct, and total effects. Mediation analysis with 1,000 bootstrap resamples was performed, adjusting variables to explore if DII acts as a mediator in the relationship between LC9 and CRS. The proportion mediated is calculated using the equation: (indirect effect / (indirect effect + direct effect)) × 100% (41). The overall effect of LC9 on CRS (Path C), LC9’s direct effect on CRS when DII is included as a mediator (Path C′), the impact of LC9 on DII (Path A), the influence of DII on CRS (Path B), and the indirect influence of DII on the LC9-CRS relationship (Path A*B) are all expressed through regression coefficients.
A total of 25,792 participants satisfied the criteria for this cross-sectional study, with data appropriately weighted. Among these, 96% did not have CRS, while 4% were diagnosed with CRS. The prevalence of persons with CRS markedly escalated with age. A greater prevalence of CRS was noted in males, non-Hispanic White people, persons who were unmarried or not cohabiting, those living in poverty, smokers, and individuals with hypertension, diabetes, or hyperlipidemia (p < 0.05). Persons with CRS exhibited reduced LC9 scores (p < 0.05) and elevated DII scores (p < 0.05) (Table 1).
Table 2 of the multivariable logistic regression model illustrates the correlation between LC9 and CRS. In the modified covariate model 3, weighted logistic regression reveals that a rise in LC9 is negatively correlated with the prevalence of CRS (OR = 0.74, 95% CI = 0.67–0.83, p < 0.001). Dividing LC9 into tertiles, with T1 as the reference group, T2 (OR = 0.59, 95% CI = 0.48–0.72, p < 0.001) and T3 (OR = 0.57, 95% CI = 0.38–0.88, p < 0.001) exhibited a strong negative correlation trend, suggesting that higher LC9 scores are associated with a substantial reduction in the incidence of CRS (p < 0.001). In contrast to LC9, Model 3 indicates that when DII is treated as a continuous variable [OR = 1.13, 95% CI = (1.06, 1.20), p < 0.001], an increase in DII scores is associated with a higher incidence of CRS. Similarly, dividing DII into tertiles with T1 as the reference group, T2 (OR = 1.25, 95% CI = 1.00–1.55, p = 0.046), and T3 (OR = 1.48, 95% CI = 1.15–1.90, p = 0.002) showed a significant positive correlation trend. These associations have received consistent support across multiple adjusted models. Figure 2 employs a smooth curve fitting for demonstrating that the relationship of LC9 and CRS is a linear negative correlation (non-linearity = 0.163). The difference between LC9 and DII remained of statistical significance when adjusting variables (β = −0.54, 95% CI: −0.57, −0.51, p < 0.001) (Table 3), indicating that elevated LC9 scores correlate with diminished DII scores. This study performed multiple imputations to address missing LC9, DII, and CRS data, confirming the stability of the results, which were consistent with the original findings (Supplementary Tables 4, 5).
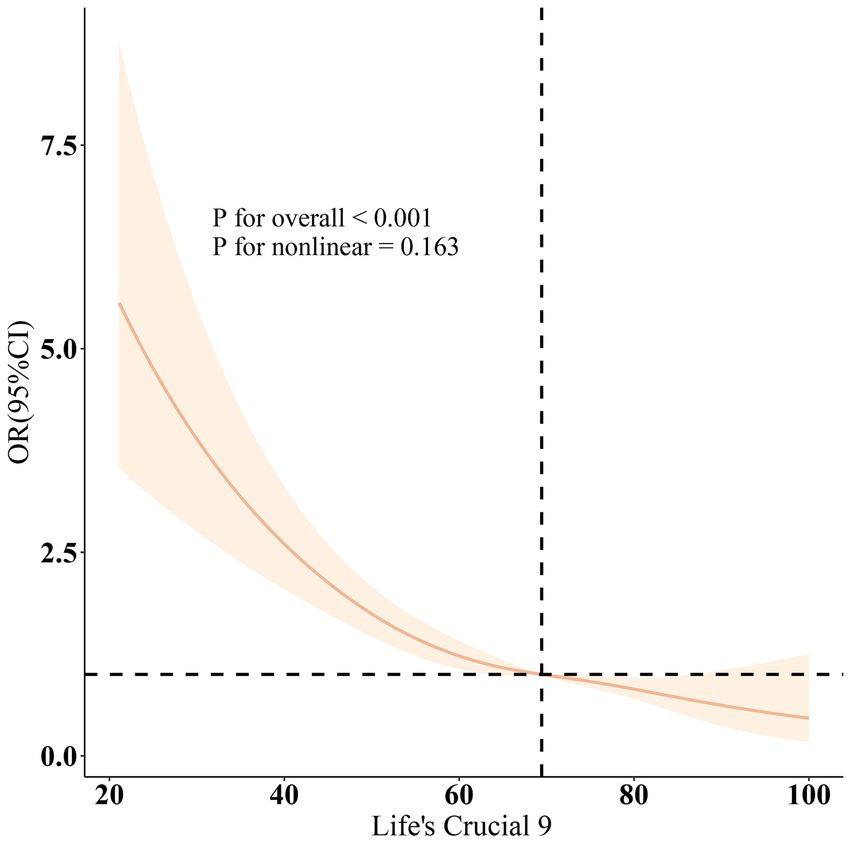
Figure 2. Dose–response relationships between LC9 and CRS. OR (solid lines) and 95% confidence levels (shaded areas) were adjusted for age, gender, education level, marital, PIR, race, smoking, drinking, hypertension, diabetes, and hyperlipidemia.
Figure 3 displays the subgroup association of LC9 with CRS. The subgroup analysis did not reveal any significant interactions between LC9 and the stratified factors, except for diabetes (p > 0.05).
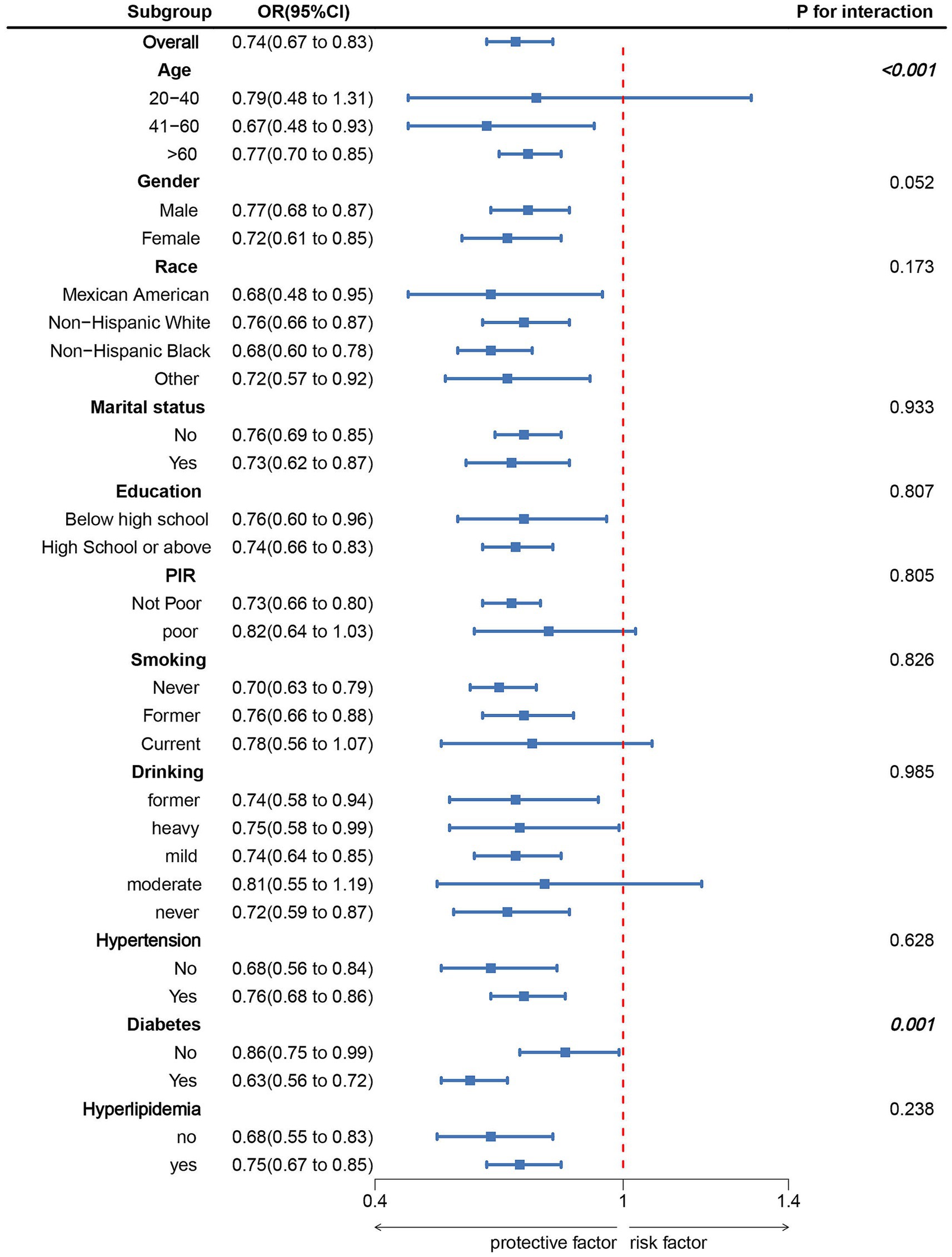
Figure 3. Subgroup analysis between LC9 and CRS. ORs were calculated as per 10-unit increase in LC9. Analyses were adjusted for age, gender, education level, marital, PIR, race, smoking, drinking, hypertension, diabetes, and hyperlipidemia.
The analysis supports the connection between the DII and CRS, and the relationship between LC9 and DII. Our findings meet the prerequisites for conducting a mediation analysis. After adjusting for all covariates, we discovered that DII acts as a mediator. DII (indirect effect = −0.00302, p = 0.030; direct effect = −0.0213, p < 0.001) accounted for 12.50% of the influence between LC9 and CRS, based on the mediation percentage calculated as (indirect effect / (indirect effect + direct effect) * 100%, p = 0.030). Thus, DII is considered a significant mediator in the relationship between LC9 and CRS (Figure 4).
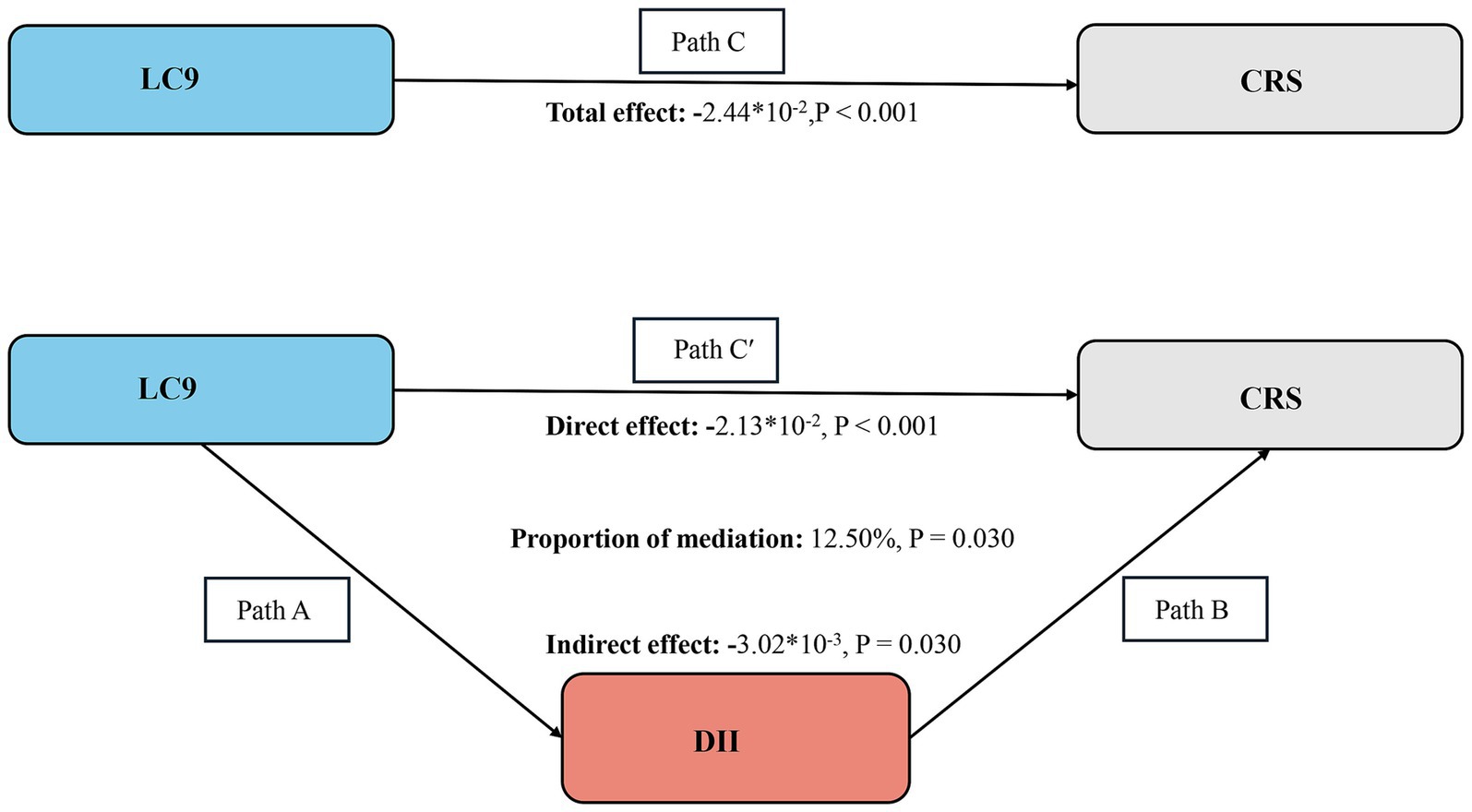
Figure 4. Schematic diagram of the mediation effect analysis. Path C indicates the total effect; path C′ indicates the direct effect. The indirect effect is estimated as the multiplication of paths A and B (path A*B). The mediated proportion is calculated as indirect effect/ (indirect effect + direct effect) × 100%. LC9, life’s crucial 9; DII, dietary inflammatory index; CRS, cardiorenal syndrome. Analyses were adjusted for age, gender, education level, marital, PIR, race, smoking, drinking, hypertension, diabetes, and hyperlipidemia.
This study, utilizing the NHANES database, identified a negative connection between LC9 and CRS prevalence after controlling for pertinent factors, with DII serving as a mediating factor, thereby corroborating our prediction. The findings from the RCS and subgroup analyses further suggest that elevated LC9 scores are associated with a decrease in the occurrence of CRS. This finding has significant clinical implications for public health interventions and individual health management since it indicates the potential to mitigate CRS risk through the enhancement of lifestyle factors. These findings offer important information for the roles of LC9 and DII in CRS and point to new research directions.
CRS denotes a series of intricate bidirectional pathophysiological mechanisms involving the malfunctioning of the heart and kidneys. The mechanism remains incompletely elucidated, involving numerous complex pathways that concentrate on critical elements such as acute cardiac decompensation, venous congestion, insufficient arterial perfusion, neurohormonal activation, inflammatory response, endothelial dysfunction, and diminished eGFR. The complexities of these pathways encompass high salt affinity, fluid retention, diminished renal clearance, and renal-associated endocrine processes. Furthermore, the difficulties may sustain the pathophysiology (42). The LC9 score is negatively correlated with the risk of CRS, which may be related to the health-promoting effects of the various components of LC9.
LC9 includes nine components: diet, physical activity, tobacco exposure, sleep quality, body mass index, blood pressure, blood glucose, blood cholesterol, and depression, all of which can be adjusted through personal lifestyle choices. While there are currently no studies reporting a direct relationship between LC9 and CRS, the individual components of LC9 have been widely studied and confirmed to be related to CRS or its components, CVD and CKD. First, results from a substantial perspective cohort study with a follow-up period of up to 32 years show that adherence to diverse healthy eating patterns correlates with a diminished risk of CVD (43). A meta-analysis of 18 published studies indicated that each additional point in HEI-2015 reduced the risk CVD mortality by 0.51% (44). A 24-year prospective study in the US showed that participants in the top quintile of the HEI-2015 score had a 17% reduced likelihood of developing of CKD relative to those in the lowest quintile, after adjusting for many covariates (45). Second, it is well-known that a cornerstone of CVD prevention is a healthy lifestyle, including habitual physical exercise (46), with ample scientific evidence supporting that physical exercise is associated with a reduced risk of CVD and CVD mortality (47, 48). Additionally, physical exercise has been repeatedly emphasized as being associated with a reduced prevalence of hypertension and diabetes, which are related to a lower incidence of CKD caused by hypertension and diabetes. Jacob W. Bruinius and colleagues indicated that increased self-reported physical activity correlates with a reduced risk of cardiovascular events and mortality in patients with CKD (49). The findings of a cohort study from the UK suggest that engaging in moderate to vigorous physical activity may significantly reduce the risk of developing CKD and AKI (50). Moreover, smoking is a globally recognized major health threat with significant negative impacts on both CVD and CKD. It damages heart and vascular health by promoting arteriosclerosis, raising blood pressure, accelerating thrombosis, and causing inflammation and oxidative stress, and it also accelerates renal function decline (51–54). Sleep also plays an important role in CRS, with studies confirming that poor quality (both quantity and quality) and sleep disorders correlate with a heightened risk of CVD and CKD (55, 56). Hypertension and diabetes have been confirmed by multiple studies to be associated with CRS (1), and metabolic factors such as body mass index, blood pressure, blood glucose, and blood lipids have been widely studied and confirmed to promote inflammation, oxidative stress, hemodynamic dysfunction, and ischemia, making them risk factors for CVD and CKD (57–61). In CKD patients, every 10-point increase in the LE8 score is associated with a 17% lower risk of all-cause mortality and an 18% lower risk of CVD mortality (17). Finally, from the substantial data of a cross-sectional analysis incorporating over five million people from the China Kadoorie Biobank and around two hundred thousand from the Dongfeng-Tongji cohort, it was found that depression is associated with higher rates of all-cause and CVD mortality compared to individuals without depression. The analysis further supports that depression significantly increases the risk for all-cause and CVD-related deaths even after adjusting for multiple variables (14). A separate investigation revealed a bidirectional association between depression and CVD (62). Research indicates that the inflammation and oxidative nitrosative stress (IO&NS) pathways underlie the shared pathophysiology of CVD and severe depressive disorder. The activation of these pathways may elevate the risk of both diseases and result in shared vulnerability (63). Moreover, depression is prevalent among CKD patients and is strongly linked to the likelihood of negative outcomes (64), with studies confirming a bidirectional relationship between them (65, 66).
This study found that enhancements in LC9 were positively correlated with a decrease in DII, suggesting that a healthy lifestyle correlates with a reduced inflammatory dietary load. Multiple prior investigations have demonstrated that dietary factors substantially influence the global burden of chronic diseases, rendering nutrition a vital modifiable target toward decreasing the incidence of these conditions (67). Dietary habits can affect the likelihood of chronic illnesses through different processes, such as managing gut microbiota, balancing oxidative stress, and maintaining energy balance. The basis of these mechanisms is the potential pro-inflammatory or anti-inflammatory properties of dietary patterns and individual dietary components (68). Studies indicate that elevated DII scores correlate with an increased risk of CVD, and mitigating the pro-inflammatory capacity of the diet may serve as an effective approach for CVD prevention (69). Another study showed that an elevation in DII correlates with a heightened likelihood of CKD stage advancement and a reduction in eGFR. After adjusting for confounding factors, for each unit increase in DII correlates with a 26% rise in the likelihood of CKD stage progression (70). Inflammation is considered a primary factor in the development of CRS. A study using a rat myocardial infarction model to examine the potential mechanisms of renal damage after cardiac dysfunction found that the mRNA expression of inflammatory cytokines IL-6 and TNF-α in the kidneys of myocardial infarction rats, as well as microvascular endothelial permeability and renal tubular cell apoptosis, were significantly increased, confirming the important role of the inflammatory response in the occurrence and development of CRS (71). Moreover, as chronic diseases such as hypertension, diabetes, and obesity play a significant role in the management of CRS, there is increasing evidence showing that patients with these chronic conditions have elevated levels of inflammatory biomarkers (including high-sensitivity C-reactive protein, various cytokines, and complement pathway products), which enhance the production of reactive oxygen species (ROS) in cells (72, 73). Therefore, mitigating inflammation through various methods is an effective measure for preventing CRS. Nonetheless, there have been no documented correlations between DII and CRS.
Our study revealed that the DII plays a mediating role between LC9 and CRS, contributing a mediation effect of 12.5%. While this percentage may appear modest, it is important to recognize the complexity of CRS’s pathogenesis, which involves multiple contributing factors. Given this complexity, the fact that dietary improvements are associated with a potential reduction in CRS represents a significant contribution. This underscores the crucial role of dietary habits in regulating systemic inflammation and managing CRS outcomes.
In summary, LC9 is a novel model for assessing cardiovascular health, DII is a dietary index, and CRS is a mechanism-complex disease closely related to a healthy lifestyle. Current research mainly focuses on the relationship between the LC9 model and DII and CRS, a topic that has not been previously explored. This study is the first investigation utilizing the NHANES database to examine the impact of DII on the relationship between LC9 and CRS, offering a novel perspective for forthcoming therapeutic therapies and public health policies. While these findings offer valuable insights into potential lifestyle interventions for CRS prevention, the cross-sectional nature of the study limits causal inferences. Future longitudinal studies are needed to confirm these relationships and explore their underlying mechanisms, providing a foundation for therapeutic and public health strategies.
Furthermore, this study possesses the subsequent limitations: (1) The nature of the cross-sectional data limits causal conclusions. Although LC9 might influence CRS through DII, more comprehensive studies, including prospective cohorts, controlled trials, and animal experiments, are needed to clarify the exact mechanisms involved. (2) Data Collection and Bias: The data for this study were collected primarily through self-administered questionnaires, which can introduce recollection bias. (3) Database Limitations: This study extensively relies on the NHANES database, which is representative of the U.S. population. However, the findings may not completely reflect the circumstances or environmental and genetic diversity found in other countries or regions. This limitation is significant because dietary habits, health behaviors, and disease prevalence can vary widely across different cultural and socio-economic backgrounds, potentially influencing the generalizability of the results to populations outside of the United States. (4) Despite including many covariates based on prior research to strengthen the robustness of our findings, the limitations of cross-sectional studies and the NHANES database prevent us from fully accounting for all potential confounders. Therefore, the results should be interpreted with caution and objectivity.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary material.
The studies involving humans were approved by the National Center for Health Statistics Ethics Review Board. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
HL: Conceptualization, Data curation, Formal analysis, Methodology, Software, Validation, Visualization, Writing – original draft, Writing – review & editing. LL: Methodology, Supervision, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
We express our gratitude to the participants of the National Health and Nutrition Examination Survey for their dedication to continuous research and the integration of electronic health records.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fnut.2025.1519612/full#supplementary-material
1. Rangaswami, J, Bhalla, V, Blair, JEA, Chang, TI, Costa, S, Lentine, KL, et al. Cardiorenal syndrome: classification, pathophysiology, diagnosis, and treatment strategies: a scientific statement from the American Heart Association. Circulation. (2019) 139:e840–78. doi: 10.1161/CIR.0000000000000664
2. Ronco, C, McCullough, P, Anker, SD, Anand, I, Aspromonte, N, Bagshaw, SM, et al. Cardio-renal syndromes: report from the consensus conference of the acute dialysis quality initiative. Eur Heart J. (2010) 31:703–11. doi: 10.1093/eurheartj/ehp507
3. Ronco, C, and Di Lullo, L. Cardiorenal syndrome in Western countries: epidemiology, diagnosis and management approaches. Kidney Dis. (2017) 2:151–63. doi: 10.1159/000448749
4. Damman, K, Valente, MAE, Voors, AA, O’Connor, CM, van Veldhuisen, DJ, and Hillege, HL. Renal impairment, worsening renal function, and outcome in patients with heart failure: an updated meta-analysis. Eur Heart J. (2014) 35:455–69. doi: 10.1093/eurheartj/eht386
5. Thompson, S, James, M, Wiebe, N, Hemmelgarn, B, Manns, B, Klarenbach, S, et al. Cause of death in patients with reduced kidney function. J Am Soc Nephrol. (2015) 26:2504–11. doi: 10.1681/ASN.2014070714
6. Yu, AS, Pak, KJ, Zhou, H, Shaw, SF, Shi, J, Broder, BI, et al. All-cause and cardiovascular-related mortality in CKD patients with and without heart failure: a population-based cohort study in Kaiser Permanente Southern California. Kidney Med. (2023) 5:100624. doi: 10.1016/j.xkme.2023.100624
7. Jankowski, J, Floege, J, Fliser, D, Böhm, M, and Marx, N. Cardiovascular disease in chronic kidney disease: pathophysiological insights and therapeutic options. Circulation. (2021) 143:1157–72. doi: 10.1161/CIRCULATIONAHA.120.050686
8. Shearer, JJ, Hashemian, M, Nelson, RG, Looker, HC, Chamberlain, AM, Powell-Wiley, TM, et al. Demographic trends of cardiorenal and heart failure deaths in the United States, 2011-2020. PLoS One. (2024) 19:e0302203. doi: 10.1371/journal.pone.0302203
9. Gallo, G, Lanza, O, and Savoia, C. New insight in Cardiorenal syndrome: from biomarkers to therapy. Int J Mol Sci. (2023) 24:5089. doi: 10.3390/ijms24065089
10. Hasbani, NR, Ligthart, S, Brown, MR, Heath, AS, Bebo, A, Ashley, KE, et al. American Heart Association’s Life’s simple 7: lifestyle recommendations, polygenic risk, and lifetime risk of coronary heart disease. Circulation. (2022) 145:808–18. doi: 10.1161/CIRCULATIONAHA.121.053730
11. Ma, H, Wang, X, Xue, Q, Li, X, Liang, Z, Heianza, Y, et al. Cardiovascular health and life expectancy among adults in the United States. Circulation. (2023) 147:1137–46. doi: 10.1161/CIRCULATIONAHA.122.062457
12. Malhi, GS, and Mann, JJ. Depression. Lancet. (2018) 392:2299–312. doi: 10.1016/S0140-6736(18)31948-2
13. Hare, DL, Toukhsati, SR, Johansson, P, and Jaarsma, T. Depression and cardiovascular disease: a clinical review. Eur Heart J. (2014) 35:1365–72. doi: 10.1093/eurheartj/eht462
14. Meng, R, Yu, C, Liu, N, He, M, Lv, J, Guo, Y, et al. Association of Depression with all-Cause and Cardiovascular Disease Mortality among Adults in China. JAMA Netw Open. (2020) 3:e1921043. doi: 10.1001/jamanetworkopen.2019.21043
15. Gaffey, AE, Rollman, BL, and Burg, MM. Strengthening the pillars of cardiovascular health: psychological health is a crucial component. Circulation. (2024) 149:641–3. doi: 10.1161/CIRCULATIONAHA.123.066132
16. Yi, J, Wang, L, Guo, X, and Ren, X. Association of Life’s essential 8 with all-cause and cardiovascular mortality among US adults: a prospective cohort study from the NHANES 2005-2014. Nutr Metab Cardiovasc Dis. (2023) 33:1134–43. doi: 10.1016/j.numecd.2023.01.021
17. Chen, H, Tang, H, Huang, J, Luo, N, Zhang, X, and Wang, X. Life’s essential 8 and mortality in US adults with chronic kidney disease. Am J Nephrol. (2023) 54:516–27. doi: 10.1159/000533257
18. Li, X, Ma, H, Wang, X, Feng, H, and Qi, L. Life’s essential 8, genetic susceptibility, and incident cardiovascular disease: a prospective study. Arterioscler Thromb Vasc Biol. (2023) 43:1324–33. doi: 10.1161/ATVBAHA.123.319290
19. Ren, Y, Cai, Z, Guo, C, Zhang, Y, Xu, H, Liu, L, et al. Associations between Life’s essential 8 and chronic kidney disease. J Am Heart Assoc. (2023) 12:e030564. doi: 10.1161/JAHA.123.030564
20. Iida, H, Fujimoto, S, Wakita, T, Yanagi, M, Suzuki, T, Koitabashi, K, et al. Psychological flexibility and depression in advanced CKD and Dialysis. Kidney Med. (2020) 2:684–691.e1. doi: 10.1016/j.xkme.2020.07.004
21. Huang, AA, and Huang, SY. Use of machine learning to identify risk factors for coronary artery disease. PLoS One. (2023) 18:e0284103. doi: 10.1371/journal.pone.0284103
22. Giugliano, D, Ceriello, A, and Esposito, K. The effects of diet on inflammation: emphasis on the metabolic syndrome. J Am Coll Cardiol. (2006) 48:677–85. doi: 10.1016/j.jacc.2006.03.052
23. Beam, A, Clinger, E, and Hao, L. Effect of diet and dietary components on the composition of the gut microbiota. Nutrients. (2021) 13:2795. doi: 10.3390/nu13082795
24. Badimon, L, Chagas, P, and Chiva-Blanch, G. Diet and cardiovascular disease: effects of foods and nutrients in classical and emerging cardiovascular risk factors. Curr Med Chem. (2019) 26:3639–51. doi: 10.2174/0929867324666170428103206
25. Shivappa, N, Steck, SE, Hurley, TG, Hussey, JR, and Hébert, JR. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. (2014) 17:1689–96. doi: 10.1017/S1368980013002115
26. Mao, Y, Weng, J, Xie, Q, Wu, L, Xuan, Y, Zhang, J, et al. Association between dietary inflammatory index and stroke in the US population: evidence from NHANES 1999-2018. BMC Public Health. (2024) 24:50. doi: 10.1186/s12889-023-17556-w
27. Wu, L, Shi, Y, Kong, C, Zhang, J, and Chen, S. Dietary inflammatory index and its association with the prevalence of coronary heart disease among 45,306 US adults. Nutrients. (2022) 14:4553. doi: 10.3390/nu14214553
28. Christ, A, Lauterbach, M, and Latz, E. Western diet and the immune system: an inflammatory connection. Immunity. (2019) 51:794–811. doi: 10.1016/j.immuni.2019.09.020
29. Shivappa, N, Godos, J, Hébert, JR, Wirth, MD, Piuri, G, Speciani, AF, et al. Dietary inflammatory index and cardiovascular risk and mortality-a Meta-analysis. Nutrients. (2018) 10:200. doi: 10.3390/nu10020200
30. Guo, M, Lei, Y, Liu, X, Li, X, Xu, Y, and Zheng, D. Association between dietary inflammatory index and chronic kidney disease in middle-aged and elderly populations. Front Nutr. (2024) 11:1335074. doi: 10.3389/fnut.2024.1335074
31. Chung, EYM, Trinh, K, Li, J, Hahn, SH, Endre, ZH, Rogers, NM, et al. Biomarkers in Cardiorenal syndrome and potential insights into novel therapeutics. Front Cardiovasc Med. (2022) 9:868658. doi: 10.3389/fcvm.2022.868658
32. Sun, M, Wang, L, Wang, X, Tong, L, Fang, J, Wang, Y, et al. Interaction between sleep quality and dietary inflammation on frailty: NHANES 2005-2008. Food Funct. (2023) 14:1003–10. doi: 10.1039/D2FO01832B
33. King, JC. Physiology of pregnancy and nutrient metabolism. Am J Clin Nutr. (2000) 71:1218S–25S. doi: 10.1093/ajcn/71.5.1218s
34. Pei, H, Li, S, Su, X, Lu, Y, Wang, Z, and Wu, S. Association between triglyceride glucose index and sleep disorders: results from the NHANES 2005-2008. BMC Psychiatry. (2023) 23:156. doi: 10.1186/s12888-022-04434-9
35. Lloyd-Jones, DM, Allen, NB, Anderson, CAM, Black, T, Brewer, LC, Foraker, RE, et al. Life’s essential 8: updating and enhancing the American Heart Association’s construct of cardiovascular health: a presidential advisory from the American Heart Association. Circulation. (2022) 146:e18–43. doi: 10.1161/CIR.0000000000001078
36. Ge, J, Peng, W, and Lu, J. Predictive value of Life’s crucial 9 for cardiovascular and all-cause mortality: a prospective cohort study from the NHANES 2007 to 2018. J Am Heart Assoc. (2024) 13:e036669. doi: 10.1161/JAHA.124.036669
37. Meng, X, Sha, W, Lou, X, and Chen, J. The relationship between dietary inflammatory index and osteoporosis among chronic kidney disease population. Sci Rep. (2023) 13:22867. doi: 10.1038/s41598-023-49824-5
38. Shivappa, N, Steck, SE, Hurley, TG, Hussey, JR, Ma, Y, Ockene, IS, et al. A population-based dietary inflammatory index predicts levels of C-reactive protein in the seasonal variation of blood cholesterol study (SEASONS). Public Health Nutr. (2014) 17:1825–33. doi: 10.1017/S1368980013002565
39. Lin, J, Li, Z, Xu, J, Pan, M, Yin, T, Wang, J, et al. Independent and joint associations of monocyte to high-density lipoprotein-cholesterol ratio and body mass index with cardiorenal syndrome: insights from NHANES 2003-2020. Lipids Health Dis. (2024) 23:153. doi: 10.1186/s12944-024-02149-2
40. Levey, AS, Stevens, LA, Schmid, CH, Zhang, YL, Castro, AF, Feldman, HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. (2009) 150:604–12. doi: 10.7326/0003-4819-150-9-200905050-00006
41. Huang, S, He, Q, Wang, X, Choi, S, and Gong, H. Associations of the planetary health diet index (PHDI) with asthma: the mediating role of body mass index. BMC Public Health. (2024) 24:2305. doi: 10.1186/s12889-024-19856-1
42. McCallum, W, and Sarnak, MJ. Cardiorenal syndrome in the hospital. Clin J Am Soc Nephrol. (2023) 18:933–45. doi: 10.2215/CJN.0000000000000064
43. Shan, Z, Li, Y, Baden, MY, Bhupathiraju, SN, Wang, DD, Sun, Q, et al. Association between healthy eating patterns and risk of cardiovascular disease. JAMA Intern Med. (2020) 180:1090–100. doi: 10.1001/jamainternmed.2020.2176
44. Hao, X, and Li, D. The healthy eating Index-2015 and all-cause/cause-specific mortality: a systematic review and dose-response Meta-analysis. Adv Nutr. (2024) 15:100166. doi: 10.1016/j.advnut.2023.100166
45. Hu, EA, Steffen, LM, Grams, ME, Crews, DC, Coresh, J, Appel, LJ, et al. Dietary patterns and risk of incident chronic kidney disease: the atherosclerosis risk in communities study. Am J Clin Nutr. (2019) 110:713–21. doi: 10.1093/ajcn/nqz146
46. Labarthe, DR, Kubzansky, LD, Boehm, JK, Lloyd-Jones, DM, Berry, JD, and Seligman, MEP. Positive cardiovascular health: a timely convergence. J Am Coll Cardiol. (2016) 68:860–7. doi: 10.1016/j.jacc.2016.03.608
47. Piercy, KL, Troiano, RP, Ballard, RM, Carlson, SA, Fulton, JE, Galuska, DA, et al. The physical activity guidelines for Americans. JAMA. (2018) 320:2020–8. doi: 10.1001/jama.2018.14854
48. Perry, AS, Dooley, EE, Master, H, Spartano, NL, Brittain, EL, and Pettee, GK. Physical activity over the Lifecourse and cardiovascular disease. Circ Res. (2023) 132:1725–40. doi: 10.1161/CIRCRESAHA.123.322121
49. Bruinius, JW, Hannan, M, Chen, J, Brown, J, Kansal, M, Meza, N, et al. Self-reported physical activity and cardiovascular events in adults with CKD: findings from the CRIC (chronic renal insufficiency cohort) study. Am J Kidney Dis. (2022) 80:751–761.e1. doi: 10.1053/j.ajkd.2022.05.007
50. Ye, Z, Liu, M, Yang, S, Zhang, Y, Zhang, Y, He, P, et al. Association of accelerometer-derived “weekend warrior” moderate to vigorous physical activity, chronic kidney disease and acute kidney injury. Prev Med. (2024) 187:108120. doi: 10.1016/j.ypmed.2024.108120
51. Mallah, MA, Soomro, T, Ali, M, Noreen, S, Khatoon, N, Kafle, A, et al. Cigarette smoking and air pollution exposure and their effects on cardiovascular diseases. Front Public Health. (2023) 11:967047. doi: 10.3389/fpubh.2023.967047
52. Kondo, T, Nakano, Y, Adachi, S, and Murohara, T. Effects of tobacco smoking on cardiovascular disease. Circ J. (2019) 83:1980–5. doi: 10.1253/circj.CJ-19-0323
53. Lee, S, Kang, S, Joo, YS, Lee, C, Nam, KH, Yun, H-R, et al. Smoking, smoking cessation, and progression of chronic kidney disease: results from KNOW-CKD study. Nicotine Tob Res. (2021) 23:92–8. doi: 10.1093/ntr/ntaa071
54. Kelly, JT, Su, G, Zhang, L, Qin, X, Marshall, S, González-Ortiz, A, et al. Modifiable lifestyle factors for primary prevention of CKD: a systematic review and Meta-analysis. J Am Soc Nephrol. (2021) 32:239–53. doi: 10.1681/ASN.2020030384
55. Miller, MA, and Howarth, NE. Sleep and cardiovascular disease. Emerg Top Life Sci. (2023) 7:457–66. doi: 10.1042/ETLS20230111
56. Wen, W, Cai, X, Zhu, Q, Hu, J, Hong, J, Zhang, X, et al. Linear relationship between hepatic steatosis index and major adverse cardiovascular events in hypertensive patients with obstructive sleep apnea: a real-world cohort study from China. Rev Cardiovasc Med. (2023) 24:280. doi: 10.31083/j.rcm2410280
57. Lo, R, Narasaki, Y, Lei, S, and Rhee, CM. Management of traditional risk factors for the development and progression of chronic kidney disease. Clin Kidney J. (2023) 16:1737–50. doi: 10.1093/ckj/sfad101
58. Silveira Rossi, JL, Barbalho, SM, Reverete de Araujo, R, Bechara, MD, Sloan, KP, and Sloan, LA. Metabolic syndrome and cardiovascular diseases: going beyond traditional risk factors. Diabetes Metab Res Rev. (2022) 38:e3502. doi: 10.1002/dmrr.3502
59. Mottillo, S, Filion, KB, Genest, J, Joseph, L, Pilote, L, Poirier, P, et al. The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol. (2010) 56:1113–32. doi: 10.1016/j.jacc.2010.05.034
60. Zhao, J, Cai, X, Hu, J, Song, S, Zhu, Q, Shen, D, et al. J-shaped relationship between weight-adjusted-waist index and cardiovascular disease risk in hypertensive patients with obstructive sleep apnea: a cohort study. Diabetes Metab Syndr Obes. (2024) 17:2671–81. doi: 10.2147/DMSO.S469376
61. Cai, X, Song, S, Hu, J, Zhu, Q, Shen, D, Yang, W, et al. Association of the trajectory of plasma aldosterone concentration with the risk of cardiovascular disease in patients with hypertension: a cohort study. Sci Rep. (2024) 14:4906. doi: 10.1038/s41598-024-54971-4
62. Bradley, SM, and Rumsfeld, JS. Depression and cardiovascular disease. Trends Cardiovasc Med. (2015) 25:614–22. doi: 10.1016/j.tcm.2015.02.002
63. Maes, M, Ruckoanich, P, Chang, YS, Mahanonda, N, and Berk, M. Multiple aberrations in shared inflammatory and oxidative & nitrosative stress (IO&NS) pathways explain the co-association of depression and cardiovascular disorder (CVD), and the increased risk for CVD and due mortality in depressed patients. Prog Neuro-Psychopharmacol Biol Psychiatry. (2011) 35:769–83. doi: 10.1016/j.pnpbp.2010.06.008
64. Bautovich, A, Katz, I, Smith, M, Loo, CK, and Harvey, SB. Depression and chronic kidney disease: a review for clinicians. Aust N Z J Psychiatry. (2014) 48:530–41. doi: 10.1177/0004867414528589
65. Liu, M, Zhang, Y, Yang, S, Wu, Q, Ye, Z, Zhou, C, et al. Bidirectional relations between depression symptoms and chronic kidney disease. J Affect Disord. (2022) 311:224–30. doi: 10.1016/j.jad.2022.05.104
66. Zheng, X, Wu, W, and Shen, S. Prospective bidirectional associations between depression and chronic kidney diseases. Sci Rep. (2022) 12:10903. doi: 10.1038/s41598-022-15212-8
67. Afshin, A, Sur, PJ, Fay, KA, Cornaby, L, Ferrara, G, Salama, JS, et al. Health effects of dietary risks in 195 countries, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet. (2019) 393:1958–72. doi: 10.1016/S0140-6736(19)30041-8
68. Tosti, V, Bertozzi, B, and Fontana, L. Health benefits of the Mediterranean diet: metabolic and molecular mechanisms. J Gerontol A Biol Sci Med Sci. (2018) 73:318–26. doi: 10.1093/gerona/glx227
69. Li, J, Lee, DH, Hu, J, Tabung, FK, Li, Y, Bhupathiraju, SN, et al. Dietary inflammatory potential and risk of cardiovascular disease among men and women in the U.S. J Am Coll Cardiol. (2020) 76:2181–93. doi: 10.1016/j.jacc.2020.09.535
70. Xu, Z, Li, L, Jiang, L, Zhai, Y, Tang, Y, Liu, D, et al. Association of Dietary Inflammatory Index with CKD progression and estimated glomerular filtration rate in the American CKD population: a cross-sectional study. PLoS One. (2024) 19:e0297916. doi: 10.1371/journal.pone.0297916
71. Cho, E, Kim, M, Ko, YS, Lee, HY, Song, M, Kim, MG, et al. Role of inflammation in the pathogenesis of cardiorenal syndrome in a rat myocardial infarction model. Nephrol Dial Transplant. (2013) 28:2766–78. doi: 10.1093/ndt/gft376
72. Xiao, L, and Harrison, DG. Inflammation in hypertension. Can J Cardiol. (2020) 36:635–47. doi: 10.1016/j.cjca.2020.01.013
Keywords: cardiorenal syndrome, Life’s Crucial 9, dietary inflammatory index, mediation analysis, NHANES
Citation: Li H and Li L (2025) Inverse associations of the lifestyle critical 9 with cardiorenal syndrome: the mediating role of the dietary inflammatory index. Front. Nutr. 12:1519612. doi: 10.3389/fnut.2025.1519612
Received: 30 October 2024; Accepted: 26 February 2025;
Published: 13 March 2025.
Edited by:
Macarena Lozano-Lorca, University of Granada, SpainReviewed by:
Ken-Ichi Kobayashi, Notre Dame Seishin University, JapanCopyright © 2025 Li and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Long Li, Z3p5eHlsbEBtZWRtYWlsLmNvbS5jbg==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.