
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
BRIEF RESEARCH REPORT article
Front. Neurol. , 28 March 2025
Sec. Neurotrauma
Volume 16 - 2025 | https://doi.org/10.3389/fneur.2025.1541520
 Layan A. Elfaki1,2*
Layan A. Elfaki1,2* Bhanu Sharma3
Bhanu Sharma3 Liesel-Ann C. Meusel2
Liesel-Ann C. Meusel2 Isis So4
Isis So4 Brenda Colella2
Brenda Colella2 Anne L. Wheeler5,6
Anne L. Wheeler5,6 Jocelyn E. Harris7
Jocelyn E. Harris7 Robin E. A. Green2,8
Robin E. A. Green2,8In chronic moderate-to-severe TBI (msTBI), depression is one of the most common psychiatric consequences. Yet to date, there is limited understanding of its neural underpinnings. This study aimed to better understand this gap by examining seed-to-voxel connectivity in depression, with all voxel-wise associations seeded to the bilateral anterior prefrontal cortices (aPFC). In a secondary analysis of 32 patients with chronic msTBI and 17 age-matched controls acquired from the Toronto Rehab TBI Recovery Study database, the Personality Assessment Inventory Depression scale scores were used to group patients into an msTBI-Dep group (T ≥ 60; n = 13) and an msTBI-Non-Dep group (T < 60; n = 19). Resting-state fMRI scans were analyzed using seed-based connectivity analyses. F-tests, controlling for age and education, were used to assess differences in bilateral aPFC rsFC across the 3 groups. After nonparametric permutation testing, the left aPFC demonstrated significantly increased rsFC with the left (p = 0.041) and right (p = 0.013) fusiform gyri, the right superior temporal lobe (p = 0.032), and the right precentral gyrus (p = 0.042) in the msTBI-Dep group compared to controls. The msTBI-Non-Dep group had no significant rsFC differences with either group. To our knowledge, this study is the first to examine aPFC rsFC in a sample of patients with msTBI exclusively. Our preliminary findings suggest a role for the aPFC in the pathophysiology of depressive symptoms in patients with chronic msTBI. Increased aPFC-sensory/motor rsFC could be associated with vulnerability to depression post-TBI, a hypothesis that warrants further investigation.
Depression is one of the most frequently reported psychiatric sequelae of moderate-to-severe traumatic brain injury (msTBI), affecting 26–40% of survivors (1–3). A substantial portion of patients develop depressive symptoms in the chronic stages (operationalized here as >6 months) post-injury (4–7), which can impede ongoing cognitive and functional recovery and is associated with increased suicidal ideation (2, 8–10). A better understanding of the neural basis of depression in chronic msTBI informs treatment research through offering an objective and quantitative biomarker as a treatment target/outcome measure that can supplement behavioral outcome measures. As well, such a biomarker can be used for neuromodulation research e.g., transcranial direct current stimulation (11–13).
Resting-state functional connectivity (rsFC) is a correlation of the blood oxygen level-dependent timeseries of two voxels or regions of interest and may constitute a data-driven avenue to explore depression in msTBI (14, 15). rsFC patterns have been extensively studied in patients with major depressive disorder through non-invasive methods (16–18) and are reproducible across various task states (19, 20). With regard to the neuroimaging correlates of depression following TBI of all severities, a recent systematic review identified 10 rsFC studies on comorbid depression and TBI (21), although only three studies examined patients with a moderate-to-severe injury not exclusively (22–24). Irrespective of TBI severity, notably inconsistent results were reported across the 10 studies, potentially attributable to their variable data acquisition and neuroimaging processing methods, choice of regions of interest, and differing clinical and demographic sample characteristics. Further, different studies operationalize depression using different cutoffs, depressive phenotypes, and time post-injury. These discrepancies may explain the limited progress in identifying a biomarker of depression in TBI (14, 21, 25). This is similarly the case for non-TBI related depression despite it being extensively studied (26–29).
The anterior prefrontal cortex (aPFC; Brodmann area 10 (BA10)) is a relevant target given the documented occurrence of cognitive control deficits in both depression (30–32) and msTBI (33–35) independently. It is implicated in cognitive control as a hub within the frontoparietal control network (36, 37) as well as salience detection as part of the salience network (38–40). To our knowledge, only two studies have investigated aPFC rsFC in patients with comorbid depression and chronic TBI (22, 23). The first utilized seed-based connectivity analyses and reported reduced rsFC connectivity between the aPFC and regions including the superior temporal gyrus, frontal pole, and the inferior parietal lobule in patients with TBI, irrespective of depressive symptom severity, as compared to healthy controls (22). In a subsequent study by this group using an expanded participant cohort, the authors examined the effects of cognitive training on depressive symptoms in patients with chronic TBI and depressive symptoms (23). Results revealed that training-induced reductions in cognitive depressive symptom severity were positively associated with rsFC between the aPFC and the inferior temporal gyrus, precentral gyrus, and postcentral gyrus, underscoring the implication of the aPFC in the occurrence of depression post-TBI.
In the context of non-TBI related depression, very few studies have examined rsFC of the aPFC specifically within the PFC. Examining the PFC broadly, increased rsFC between the PFC - specifically the inferior frontal gyrus—and amygdala was reported in patients with major depressive disorder (MDD) as compared to healthy controls, which decreased after treatment with antidepressant medication (41). The hyperconnectivity was interpreted as the PFC increasing its inhibition of the amygdala in depressed states, simultaneously leading to an increased frequency of negative emotions. Conversely, another study examined rsFC of the corticolimbic system to report reduced dlPFC-amygdala rsFC in unmedicated patients with MDD as compared to healthy controls (42). Moreover, recent seminal work has parceled brain activity/connectivity of patients with depression and anxiety disorders into 6 distinct biotypes in a study of task-based MRI (43). The profiles were based on symptom severity, behavioral performance, and response to treatment. Although the medial superior, anterior medial, and ventromedial prefrontal cortices emerged as key in differentiating the biotypes, this circuit-level approach precludes us from understanding the unique contributions of specific brain regions including the aPFC (44, 45). Therefore, targeted investigation into the aPFC is necessary to better understand its role in comorbid depression and TBI.
Given the lack of studies in this area, this present study aimed to contribute groundwork research towards the identification of a neural signature of depressive symptoms in chronic msTBI by examining rsFC of the aPFC to other brain regions voxel-wise. Thus, aPFC seed-based connectivity analysis comparisons were undertaken between the groups of patients with and without depression as well as healthy controls to examine group differences in bilateral aPFC rsFC. Based on the findings of rsFC alterations in the frontoparietal control network (46–48) and salience network (30, 49, 50), in depression and msTBI (22), and the occurrence of cognitive control deficits in both comorbidities (30, 32–34), we hypothesized reduced rsFC between the aPFC and other brain regions in the chronic msTBI groups as compared to healthy controls. Additionally, we also hypothesized reduced right aPFC rsFC with other regions in the group of patients with chronic msTBI compared to those with comorbid msTBI and depression (23).
This is a secondary data analysis of prospectively collected data from the Toronto Rehab TBI Recovery Study, a longitudinal study examining neural, cognitive, and mental health recovery following msTBI (51–54). All patients provided written informed consent for their participation in the study.
Inclusion criteria for the parent study comprised: (1) diagnosis of msTBI; (2) between 18 and 80 years in age; (3) able to follow commands in English; (4) able to use at least 1 upper extremity; and (5) able to provide informed consent for participation or availability of a legal decision-maker. Exclusion criteria included: (1) prior history of TBI; (2) other pre-existing central nervous system disorder; (3) current diagnosis or history of a psychotic disorder; (4) persisting PTA at 6 weeks post-injury; and (5) metal implants that precluded MRI. Further details on the inclusion/exclusion criteria of the parent study are detailed in reports directly related to said study (51, 55). An additional inclusion criterion for the current study was availability of valid fMRI scans and a completed Personality Assessment Inventory (PAI) (56) questionnaire in the chronic stage of the msTBI (> 6 months post-injury). Pairwise deletions were made for patients with invalid responses on the PAI based on internal validity indices (56). Specifically, individuals with T scores ≥75, ≥ 73, ≥ 68, and ≥ 92 on the Infrequency, Inconsistency, Positive Impression Management, and/or Negative Impression Management (NIM-Tot) scale, respectively, were excluded (32). See Table 1.
Depression status was operationalized using a T-score cutoff of 60 on the PAI’s Depression clinical scale’s total score (DEP-Tot), as employed by other studies assessing depressive symptoms in TBI populations (57, 58). Participants with DEP-Tot T-scores ≥60 were assigned to the msTBI-Dep group (n = 13; mean = 74.54 (SD = 13.61)) and those with DEP-Tot scores <60 were placed in the msTBI-Non-Dep group (n = 19; mean = 47.79 (SD = 5.96)).
The healthy control group comprised n = 25 healthy controls matched on age and education to the msTBI groups. Inclusion criteria were: (1) between 18 and 80 years in age; (2) able to follow commands in English; and (3) commitment to completing the imaging scans. Exclusion criteria included: (1) previous history of TBI requiring hospitalization including concussion; (2) history of any disease affecting the central nervous system; (3) current diagnosis of depression; and (4) presence of magnetic materials affecting MRI scan acquisition. Applying these criteria, we obtained a sample of n = 17 healthy controls.
The PAI is a 344-item self-report scale, with high internal consistency (56) and construct validity (59). The PAI has been validated for msTBI (54). To classify patients into study groups, we employed the total score of the Depression clinical scale (DEP-Tot), which represents a composite of its three subscales: Depression-Cognitive (DEP-Cog), Depression-Affective (DEP-Aff), and Depression-Physiological (DEP-Phys) symptoms.
We examined the performances of the msTBI-Dep and msTBI-Non-Dep groups on a range of neuropsychological tests to identify potential differences between the groups that might contribute to aPFC rsFC differences. A comprehensive clinical battery measuring attention, speed of processing, verbal and visuospatial memory, executive functioning, verbal and performance IQ, and estimated premorbid IQ was administered by a trained psychometrist under the supervision of a neuropsychologist. Additional details of the battery are listed in a previous publication by our group (52).
MRI data were obtained using a General Electric (GE) Signa-Echospeed 1.5 Tesla HD scanner (SIGNA EXCITE, GE Healthcare, Milwaukee, Wisconsin) with an 8-channel head coil configuration. First, high-resolution T1-weighted three-dimensional gradient-echo echo-planar images were acquired with the following parameters: sagittal T1-weighted spin echo, repetition time = 300 msec, echo time = 13 msec, slice thickness = 5 mm no gap, slice spacing of 2.5 mm, matrix = 256 × 128, and field of view (FOV) = 22 cm. Resting-state scans (rs-fMRI) lasting 5 min were then acquired via whole-brain gradient-echo echoplanar images with the following parameters: repetition time = 2,000 msec, echo time = 40 msec, flip angle = 85°, slice thickness = 5.0 mm and 150 axial slices, interleaved in order. Participants were instructed to keep their eyes closed for the 5-min resting scans.
Resting-state fMRI data were preprocessed using FMRIB Software Library (FSL version 5.0.1; http://fsl.fmrib.ox.ac.uk/) FEAT. This was done using similar steps to those performed in a previous study by our lab group (55). Preprocessing included: (1) discarding the first five functional volumes, resulting in 145 volumes for analysis; (2) skull stripping using FSL BET; (3) motion correction using FSL MCFLIRT; (4) spatial smoothing with a Gaussian kernel of 5 mm full-width at half-maximum; (5) Gaussian-weighted high pass temporal filter (100 s); (6) artifact denoising via ICA-AROMA (60) which has been validated for seed-based analyses (61); (7) cerebrospinal fluid and white matter signal regression with thresholds of 1 and 0.98, respectively, via FSL FAST; and (8) linear registration with 12 degrees of freedom to MNI152 through FSL FLIRT. The final outputs were in Montreal Neurological Institute (MNI) space (62). Global signal regression was not applied given that it can introduce anticorrelations into the functional data (60, 63). For quality assurance, the data were visually inspected by a neuroradiologist following registration to ensure that there were no contusions and/or excessive motion (n = 3 patients were excluded accordingly).
Following preprocessing, whole-brain seed-based connectivity analyses were implemented through FSL FEAT (64). Two 5 mm radius spheres were created and centered on the following MNI coordinates: left aPFC: −36, 57, 9; right aPFC: 34, 52, 10. These ROI were delineated following the approach of a previous study identifying significant aPFC rsFC differences in patients with msTBI in comparison to healthy controls (22). The average BOLD signal time series within each aPFC seed were extracted and entered as the primary regressors in a first-level general linear model (GLM) for the left and right aPFC, independently. In all first-level analyses, cluster-based thresholding was employed with a z-threshold of 3.1 and a significance level of 0.05. FSL FILM pre-whitening was applied to correct for time series autocorrelations and improve estimation accuracy.
To identify bilateral aPFC rsFC differences across the msTBI-Dep, msTBI-Non-Dep, and healthy control groups, higher-level group comparisons were conducted via an F-test in FSL (65). The F-test simultaneously assessed all possible pairwise differences between the three groups, controlling for age and education. For any significant comparisons that emerged (FWE-corrected p < 0.05), two-tailed unpaired t-tests were subsequently performed to investigate the directionality of findings.
To preserve the study’s statistical power, we undertook a data-driven approach in addition to consulting the literature to identify the covariates that were included, and thus, controlled for in the GLM of the seed-based analyses. First, education was included due to the high variability in education attained across participants (range: 9 to 18 years) as well as the differences between the msTBI-Dep, msTBI-Non-Dep, and healthy control groups in education with a moderate effect size (H(2, N = 49) = 2.31, p = 0.315, ε2 = 0.0.050). Second, it was important to control for age given its documented effects on rsFC of various brain regions and networks in depression (66, 67). Apart from its effect on rsFC, older age at injury also portends poorer functional outcomes post-TBI (53, 68). Despite the wide range in inclusion criteria for age, only 2 patients were 65+, distributed across the healthy control and msTBI-Dep groups. Altogether, age and education (both measured in years) were centered over the global mean of each group and added as covariates of non-interest. Total intracranial volume (TIV) was not added as a covariate because of the limited sample size. Despite this, all fMRI scans were normalized and registered to the standard MNI-152 template. This would’ve at least partially reduced potential influences of TIV (69).
Between-group differences in aPFC rsFC were assessed using FSL’s non-parametric permutation-based Randomize. Threshold-free cluster enhancement (TFCE) was used with 5,000 permutations to avoid the use of an arbitrary cluster-forming threshold, and family-wise error (FWE) correction was undertaken to correct for multiple comparisons. TFCE is preferred over traditional cluster thresholding methods given that it substantially reduces the rate of false positive results (70, 71). Significant clusters were localized and labeled using FSL’s Harvard-Oxford Cortical and Subcortical Structural Atlas (72). The sizes of these clusters were identified and denoted k. Effect sizes for the connectivity maps, that is, Cohen’s d values, were computed from t-statistic maps using the standard Cohen’s d calculation formula for two-sample t-tests.
Statistical analyses for all demographic, clinical, and neuropsychological assessment data were carried out on IBM SPSS Statistics (version 28.0.1.0). The data were first subjected to normality testing using the Shapiro–Wilk normality test. Subsequently, the msTBI-Dep group and the msTBI-Non-Dep group were compared on all demographic, clinical and neuropsychological variables to identify any significant group differences that could influence rsFC findings. An α level of 0.05 was utilized to identify significant differences. As for effect sizes, Cohen’s d was computed for independent-samples t-tests, effect size r was computed for Mann–Whitney U tests, KW-epsilon squared for the Kruskal-Wallis test, and Cramer’s V for Fisher’s exact test (see Table 1).
The msTBI-Dep and msTBI-Non-Dep group differed in terms of the lowest GCS recorded, which was significantly higher in the msTBI-Non-Dep group with a medium effect size (U = 49.5, p = 0.024, d = 0.3972). Length of post-traumatic amnesia and acute care length of stay did not differ significantly between the two msTBI groups (p’s > 0.05). Otherwise, there were no significant differences between the three study groups in the remaining demographic, injury-related, and clinical variables.
With regards to the neuropsychological assessments examined, no significant difference was found between the msTBI-Dep group and the msTBI-Non-Dep group on any assessment, and the effect sizes were all small with the exception of the Stroop color-naming, word-reading, and Stroop interference tests which were medium (Cohen’s d = 0.4–0.6).
There were no significant differences between the msTBI-Non-Dep group and the msTBI-Dep group nor the msTBI-Non-Dep group and the healthy control group (FWE-corrected p < 0.5) in bilateral aPFC rsFC. However, rsFC of the left aPFC with 4 brain regions was significantly higher in the msTBI-Dep group as compared to the healthy control group, with large effect sizes. The 4 regions consisted of the left fusiform gyrus (BA37; t = 3.65, TFCE = 22,729, FWE-corrected p = 0.041, k = 119, Cohen’s d = 1.35), the right fusiform gyrus (BA37; t = 4.39, TFCE = 30,761, FWE-corrected p = 0.01, k = 11,867, Cohen’s d = 1.62), the right superior temporal lobe (STL; BA22; t = 4.43, TFCE = 24,941, FWE-corrected p = 0.032, k = 685; Cohen’s d = 1.63), and the right precentral gyrus (BA4; t = 4.34, TFCE = 22,443, FWE-corrected p = 0.042, k = 29, Cohen’s d = 1.60). These differences were non-significant between the two other study groups (p > 0.05). See Table 2 and Figure 1.
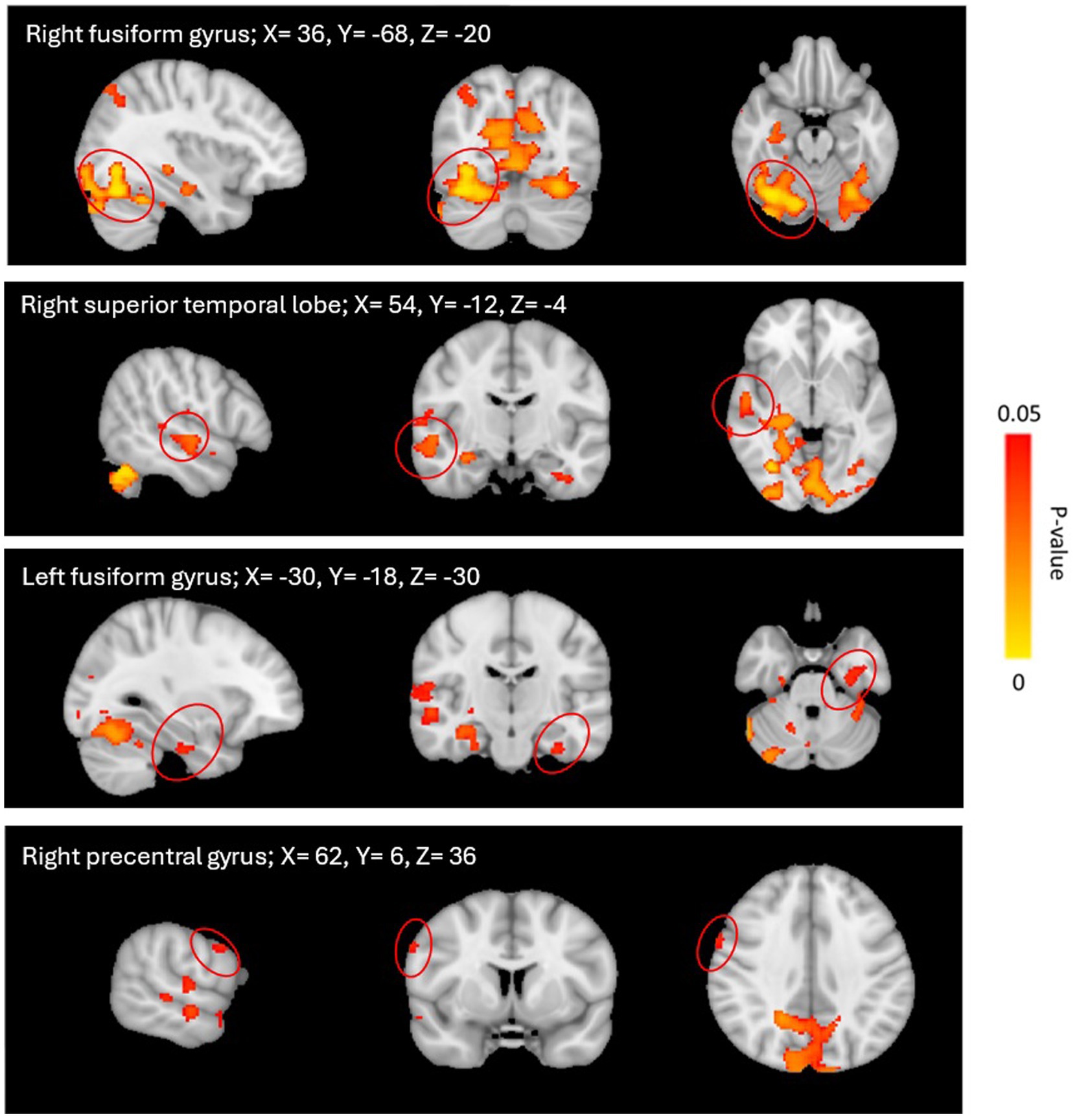
Figure 1. LaPFC rsFC Comparisons: msTBI-Dep Group > healthy control group. Significant clusters obtained from the F-test computed for the left anterior prefrontal cortex, specifically the msTBI-dep > healthy control contrast, after controlling for age and education. Images represent threshold-free cluster enhancement and family-wise error rate-corrected p-values thresholded at an α level of 0.05, overlaid on a standardized MNI-152 brain. The left hemisphere of the brain corresponds to the right side in this image and vice versa. See Table 2.
The overarching aim of this study was to address a gap in the research with regard to the clinical neuroimaging biomarkers of depression in msTBI (21). This was the first study to examine the question of biomarkers of depression in bilateral aPFC rsFC through seed-based connectivity analyses in (1) a homogenous group of patients with msTBI (i.e., not integrated with mild TBI) and (2) in the chronic stages of injury.
We found increased rsFC in the msTBI-Dep group as compared to the healthy control group between the left aPFC and four sensory and motor regions (bilateral fusiform gyri, right STL, and right precentral gyrus). In this preliminary study, the implication of the aPFC - a brain region involved in cognitive control and salience detection - and these 4 regions aligns with past literature reporting rsFC alterations to these aforementioned modalities in both msTBI (73) and depression (32, 74) populations.
Broadly, dysfunction of the PFC has been consistently implicated in affective disorders, including MDD (75). Focusing on the aPFC specifically, it is associated with the salience network, and thus, implicated in salience detection, attention control, and emotional regulation (39, 76). Additionally, as a key hub within the frontoparietal control network, it contributes to cognitive control (36) and action selection through reward tracking (77). Dysfunction of the salience and frontoparietal control networks are commonly reported in clinically depressed populations (30, 78, 79). Despite this, there exists a very limited range of studies examining aPFC rsFC in depression following chronic msTBI (22, 23). It is therefore important to interpret these findings in the context of the cognitive and affective roles that these implicated regions have.
As it relates to brain-behavior relationships, the fusiform gyrus has been known to facilitate object and facial recognition (80) and plays pivotal roles in affect and emotional processing (81–83). Similarly, in addition to coordinating motor movements as part of the primary motor cortex (84, 85), the precentral gyrus plays a critical role in regulating emotional circuitry involving the amygdala (86, 87). Increased activity of the precentral gyrus was reported during the perception of threatening emotional stimuli as compared to neutral stimuli, supporting the engagement of this motor region in emotional processing (88). Not surprisingly, increased rsFC between motor regions including the supplementary motor area and self-related regions including the pregenual ACC was reported in patients with depression as compared to healthy controls (89). Lastly, as part of the affective network (90), the STL has been implicated with emotional processing in patients with MDD (91, 92) as well as healthy patients (93). Evidently, regardless of whether it is a primary function or not, these sensory and motor regions have documented roles in emotional processing and regulation.
Frontal regions, including the PFC, exert top-down control over emotion processing regions as a regulatory mechanism (94–96). In patients with mild TBI, Iraji et al. (97) reported increased rsFC between the aPFC and the thalamus at the acute stage post-injury. This hyperconnectivity was hypothesized to serve as a compensatory mechanism for depression through capitalizing on additional networks and connectivity. Therefore, given the roles of these four regions, it is possible that the increased rsFC between the aPFC and these sensory and motor regions reflects an attempt to increase top-down control over the excessive bottom-up emotional processing experienced in depression in the msTBI-Dep group. In other words, this hyperconnectivity could be a marker of compensatory mechanisms that down-regulate the heightened reactivity to negative emotional stimuli that emerges in depression following injury (97). However, such hyperconnectivity is ultimately without functional gain as reflected by the elevated PAI DEP-Tot scores, and hence, the presence of depressive symptoms in the msTBI-Dep group.
Conversely, the observed pattern of non-adaptive - or potentially maladaptive - hyperconnectivity between the aPFC and the four sensory and motor regions could itself be an underlying neuropathology of depressive symptomatology in the msTBI-Dep group. This hypothesis is supported by the literature findings wherein patients with MDD exhibited PFC-amygdala hyperconnectivity that decreased over the course of 8 weeks of treatment with antidepressant medication (41). Interestingly, hyperconnectivity within higher-order cognitive and sensory networks was also reported in chronic msTBI, which the authors hypothesized to be attributable to compensatory mechanisms and/or underlying TBI symptomatology (98). Because the msTBI groups had non-significant aPFC rsFC differences in the present study, it is thus plausible to consider that the aPFC hyperconnectivity in our msTBI-Dep group may be due to either compensatory mechanisms, underlying TBI symptomatology, or both. Although future research is needed to delineate the mechanisms behind these rsFC changes, the findings of this present study show that aPFC rsFC may have potential as a neuroimaging marker for characterizing the changes that occur in the presence of depressive symptoms in patients with chronic msTBI.
Despite being one of the most common sequelae of msTBI, current clinical treatment strategies for the management of depression post-TBI are lacking (10, 21). This renders chronic msTBI patients at an increased risk of developing depression and its associated functional impediments (99, 100). What is more, associations between rsFC and depression scores have been reported in chronic msTBI in the absence of associations between whole-brain grey matter volume and depression scores, contributing to the psychiatric sequelae that emerge after TBI while evading detection through structural imaging tools (14).
The observed aPFC hyperconnectivity in depression post-TBI informs treatment research through aiding the prediction and monitoring response to treatment. Consequently, non-invasive neuromodulation-based therapies, e.g., transcranial magnetic and deep brain stimulation can be utilized to normalize aberrant aPFC rsFC, alleviating depressive symptoms and augmenting quality of life (11–13). This is particularly important given previous research from our lab showing an escalation in the number of patients with clinically significant symptoms of depression in the chronic stages of msTBI (101).
In terms of strengths, employing seed-based connectivity analyses to identify a signal for depressive symptoms post-TBI signifies that our findings are restricted to a specific seed region as opposed to the network(s) with which the region is associated. Although network-based approaches such as independent component analysis enable a more holistic understanding of brain rsFC, the aPFC seed-based connectivity analyses undertaken in this study serve as a starting point for understanding brain rsFC alterations in a research area that is yet to be explored.
Small sample size was a notable limitation of this study, which may have limited our statistical power; it also precluded control for additional potential confounds within the models such as TIV, injury severity, history of depression or anxiety, and participant sex which may have affected the aPFC rsFC reported. However, although participant sex has been reported to impact rsFC in depression (102, 103), there were no significant differences in sex distribution between our three study groups via Fisher’s exact test (p > 0.05, Cramer’s V = 0.34). Given that the effect size was moderate, additional sensitivity analyses were run to complement this finding. This included correlational analyses between sex distribution and the PAI DEP-Tot depression scores which were also non-significant in our msTBI groups (p = 0.615). Similarly, although the inclusion criteria for age were 18–80, only 2 patients were 65+, with this variable controlled for within the GLM.
Because this study was a secondary analysis on prospectively collected data, we did not have control over the study parameters and outcomes, including the imaging scanner acquisition hardware and software. One notable limitation in this regard was that the neuroimaging data employed in this study were acquired with a 1.5 Tesla scanner, which may have limited our signal-to-noise ratio, and thus, spatial resolution.
Replication studies should address these limitations. To build additional mechanistic insight, future studies should consider exploring aPFC rsFC using a seed-to-region of interest (ROI) analysis inclusive of the bilateral aPFC seeds and the precentral gyrus, the fusiform gyrus, and the STL as ROI. Future analyses could also be expanded to include ROIs previously implicated in depression following chronic msTBI (14, 24) as well as ROIs implicated in non-TBI related depression (104–107). The latter is warranted in light of the reported similarities in neurophysiological responses to depression occurring within and outside the context of msTBI (14, 108, 109). Such analyses would enrich our understanding of rsFC alterations occurring between the left aPFC and sensory and motor regions following chronic msTBI and whether the observed hyperconnectivity constitutes vulnerability or a compensatory reaction to depression following chronic msTBI.
This present study, to our knowledge, is first to investigate aPFC rsFC in a group of patients with exclusively msTBI (i.e., without patients in the mild range of TBI) and in the chronic stages of msTBI. Our findings revealed that aPFC-sensory/motor rsFC was significantly increased in the msTBI-Dep group compared to the healthy control group, though the nature of this hyperconnectivity necessitates further investigation. Our results provide support to findings established in depression literature, in the context of comorbid depression and msTBI. Altogether, these preliminary findings contribute novel empirical data towards characterizing the functional basis of depression in chronic msTBI.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Toronto Rehabilitation Institute Research Ethics Board. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
LE: Conceptualization, Formal analysis, Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing, Data curation, Project administration. BS: Conceptualization, Methodology, Writing – review & editing, Validation. L-AM: Conceptualization, Methodology, Validation, Writing – review & editing. IS: Formal analysis, Writing – review & editing. BC: Data curation, Project administration, Writing – review & editing, Conceptualization, Investigation. AW: Conceptualization, Validation, Writing – review & editing, Supervision. JH: Writing – review & editing. RG: Conceptualization, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Validation, Writing – review & editing, Writing – original draft.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was funded by the Canadian Institutes of Health Research (#MOP-67072 and #MOP-86704), Physicians Services Incorporated Foundation (#12-43), and the Natural Sciences and Engineering Research Council of Canada (#UT458054). The funding agencies did not influence the study design or manuscript preparation.
The authors thank Sakina Rizvi for their support and input on project design. This study is part of a larger Master of Science thesis, published within the University of Toronto Thesis Repository, Tspace (110).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Albrecht, JS, Lydecker, A, Peters, ME, and Rao, V. Treatment of depression after traumatic brain injury reduces risk of neuropsychiatric outcomes. J Neurotrauma. (2020) 37:2542–8. doi: 10.1089/neu.2019.6957
2. Choi, Y, Kim, EY, Sun, J, Kim, H-K, Lee, YS, Oh, B-M, et al. Incidence of depression after traumatic brain injury: a Nationwide longitudinal study of 2.2 million adults. J Neurotrauma. (2022) 39:390–7. doi: 10.1089/neu.2021.0111
3. Dams-O’Connor, K, Juengst, SB, Bogner, J, Chiaravalloti, ND, Corrigan, JD, Giacino, JT, et al. Traumatic brain injury as a chronic disease: insights from the United States traumatic brain injury model systems research program. Lancet Neurol. (2023) 22:517–28. doi: 10.1016/S1474-4422(23)00065-0
4. Bombardier, CH, Hoekstra, T, Dikmen, S, and Fann, JR. Depression trajectories during the first year after traumatic brain injury. J Neurotrauma. (2016) 33:2115–24. doi: 10.1089/neu.2015.4349
5. Hart, T, Hoffman, JM, Pretz, C, Kennedy, R, Clark, AN, and Brenner, LA. A longitudinal study of major and minor depression following traumatic brain injury. Arch Phys Med Rehabil. (2012) 93:1343–9. doi: 10.1016/j.apmr.2012.03.036
6. Ouellet, M-C, Beaulieu-Bonneau, S, Sirois, M-J, Savard, J, Turgeon, AF, Moore, L, et al. Depression in the first year after traumatic brain injury. J Neurotrauma. (2018) 35:1620–9. doi: 10.1089/neu.2017.5379
7. Scholten, AC, Haagsma, JA, Cnossen, MC, Olff, M, van Beeck, EF, and Polinder, S. Prevalence of and risk factors for anxiety and depressive disorders after traumatic brain injury: a systematic review. J Neurotrauma. (2016) 33:1969–94. doi: 10.1089/neu.2015.4252
8. Grauwmeijer, E, Heijenbrok-Kal, MH, Peppel, LD, Hartjes, CJ, Haitsma, IK, de Koning, I, et al. Cognition, health-related quality of life, and depression ten years after moderate to severe traumatic brain injury: a prospective cohort study. J Neurotrauma. (2018) 35:1543–51. doi: 10.1089/neu.2017.5404
9. Howlett, JR, Nelson, LD, and Stein, MB. Mental health consequences of traumatic brain injury. Biol Psychiatry. (2022) 91:413–20. doi: 10.1016/j.biopsych.2021.09.024
10. Izzy, S, Chen, PM, Tahir, Z, Grashow, R, Radmanesh, F, Cote, DJ, et al. Association of Traumatic Brain Injury with the risk of developing chronic cardiovascular, endocrine, neurological, and psychiatric disorders. JAMA Netw Open. (2022) 5:e229478. doi: 10.1001/jamanetworkopen.2022.9478
11. Klooster, D, Voetterl, H, Baeken, C, and Arns, M. Evaluating robustness of brain stimulation biomarkers for depression: a systematic review of magnetic resonance imaging and electroencephalography studies. Biol Psychiatry. (2024) 95:553–63. doi: 10.1016/j.biopsych.2023.09.009
12. Prompiengchai, S, and Dunlop, K. Breakthroughs and challenges for generating brain network-based biomarkers of treatment response in depression. Neuropsychopharmacology. (2024) 50:230–45. doi: 10.1038/s41386-024-01907-1
13. Voegeli, G, Cléry-Melin, ML, Ramoz, N, and Gorwood, P. Progress in elucidating biomarkers of antidepressant pharmacological treatment response: a systematic review and meta-analysis of the last 15 years. Drugs. (2017) 77:1967–86. doi: 10.1007/s40265-017-0819-9
14. Luo, L, Langley, C, Moreno-Lopez, L, Kendrick, K, Menon, DK, Stamatakis, EA, et al. Depressive symptoms following traumatic brain injury are associated with resting-state functional connectivity. Psychol Med. (2021) 53:2698–705. doi: 10.1017/s0033291721004724
15. Raimondo, L, Oliveira, ĹAF, Heij, J, Priovoulos, N, Kundu, P, Leoni, RF, et al. Advances in resting state fMRI acquisitions for functional connectomics. NeuroImage. (2021) 243:118503. doi: 10.1016/j.neuroimage.2021.118503
16. Korgaonkar, MS, Goldstein-Piekarski, AN, Fornito, A, and Williams, LM. Intrinsic connectomes are a predictive biomarker of remission in major depressive disorder. Mol Psychiatry. (2020) 25:1537–49. doi: 10.1038/s41380-019-0574-2
17. Lynch, CJ, Gunning, FM, and Liston, C. Causes and consequences of diagnostic heterogeneity in depression: paths to discovering novel biological depression subtypes. Biol Psychiatry. (2020) 88:83–94. doi: 10.1016/j.biopsych.2020.01.012
18. Tassone, VK, Demchenko, I, Salvo, J, Mahmood, R, Di Passa, A-M, Kuburi, S, et al. Contrasting the amygdala activity and functional connectivity profile between antidepressant-free participants with major depressive disorder and healthy controls: a systematic review of comparative fMRI studies. Psychiatry Res Neuroimaging. (2022) 325:111517. doi: 10.1016/j.pscychresns.2022.111517
19. Bondi, E, Maggioni, E, Brambilla, P, and Delvecchio, G. A systematic review on the potential use of machine learning to classify major depressive disorder from healthy controls using resting state fMRI measures. Neurosci Biobehav Rev. (2023) 144:104972. doi: 10.1016/j.neubiorev.2022.104972
20. Spronk, M, Keane, BP, Ito, T, Kulkarni, K, Ji, JL, Anticevic, A, et al. A whole-brain and cross-diagnostic perspective on functional brain network dysfunction. Cereb Cortex. (2021) 31:547–61. doi: 10.1093/cercor/bhaa242
21. Medeiros, GC, Twose, C, Weller, A, Dougherty, JW 3rd, Goes, FS, Sair, HI, et al. Neuroimaging correlates of depression after traumatic brain injury: a systematic review. J Neurotrauma. (2022) 39:755–72. doi: 10.1089/neu.2021.0374
22. Han, K, Chapman, SB, and Krawczyk, DC. Altered amygdala connectivity in individuals with chronic traumatic brain injury and comorbid depressive symptoms. Front Neurol. (2015) 6:231. doi: 10.3389/fneur.2015.00231
23. Han, K, Martinez, D, Chapman, SB, and Krawczyk, DC. Neural correlates of reduced depressive symptoms following cognitive training for chronic traumatic brain injury. Hum Brain Mapp. (2018) 39:2955–71. doi: 10.1002/hbm.24052
24. Moreno-López, L, Sahakian, BJ, Manktelow, A, Menon, DK, and Stamatakis, EA. Depression following traumatic brain injury: a functional connectivity perspective. Brain Inj. (2016) 30:1319–28. doi: 10.1080/02699052.2016.1186839
25. Dunlop, K, Talishinsky, A, and Liston, C. Intrinsic brain network biomarkers of antidepressant response: a review. Curr Psychiatry Rep. (2019) 21:87. doi: 10.1007/s11920-019-1072-6
26. Buch, AM, and Liston, C. Dissecting diagnostic heterogeneity in depression by integrating neuroimaging and genetics. Neuropsychopharmacology. (2021) 46:156–75. doi: 10.1038/s41386-020-00789-3
27. Takahara, Y, Kashiwagi, Y, Tokuda, T, Yoshimoto, J, Sakai, Y, Yamashita, A, et al. Comprehensive evaluation of pipelines for diagnostic biomarkers of major depressive disorder using multi-site resting-state fMRI datasets. Biorxiv. (2024). doi: 10.1101/2024.03.17.584538
28. Taylor, JJ, Kurt, HG, and Anand, A. Resting state functional connectivity biomarkers of treatment response in mood disorders: a review. Front Psych. (2021) 12:565136. doi: 10.3389/fpsyt.2021.565136
29. Shin, D, Cho, WI, Park, CHK, Rhee, SJ, Kim, MJ, Lee, H, et al. Detection of minor and major depression through voice as a biomarker using machine learning. J Clin Med Res. (2021) 10:10. doi: 10.3390/jcm10143046
30. Jiao, K, Xu, H, Teng, C, Song, X, Xiao, C, Fox, PT, et al. Connectivity patterns of cognitive control network in first episode medication-naive depression and remitted depression. Behav Brain Res. (2020) 379:112381. doi: 10.1016/j.bbr.2019.112381
31. Villalobos, D, Pacios, J, and Vázquez, C. Cognitive control, cognitive biases and emotion regulation in depression: a new proposal for an integrative interplay model. Front Psychol. (2021) 12:628416. doi: 10.3389/fpsyg.2021.628416
32. Zhang, X, Zhang, R, Lv, L, Qi, X, Shi, J, and Xie, S. Correlation between cognitive deficits and dorsolateral prefrontal cortex functional connectivity in first-episode depression. J Affect Disord. (2022) 312:152–8. doi: 10.1016/j.jad.2022.06.024
33. Lennon, MJ, Brooker, H, Creese, B, Thayanandan, T, Rigney, G, Aarsland, D, et al. Lifetime traumatic brain injury and cognitive domain deficits in late life: the PROTECT-TBI cohort study. J Neurotrauma. (2023) 40:1423–35. doi: 10.1089/neu.2022.0360
34. Murphy, JM, Bennett, JM, de la Piedad, GX, and Willis, ML. Emotion recognition and traumatic brain injury: a systematic review and meta-analysis. Neuropsychol Rev. (2022) 32:520–36. doi: 10.1007/s11065-021-09510-7
35. Steward, KA, Kennedy, R, Novack, TA, Crowe, M, Marson, DC, and Triebel, KL. The role of cognitive Reserve in Recovery from Traumatic Brain Injury. J Head Trauma Rehabil. (2018) 33:E18–27. doi: 10.1097/HTR.0000000000000325
36. Dreher, JC, Koechlin, E, Tierney, M, and Grafman, J. Damage to the fronto-polar cortex is associated with impaired multitasking. PLoS One. (2008) 3:e3227. doi: 10.1371/journal.pone.0003227
37. Manassero, E, Concina, G, Caraig, MCC, Sarasso, P, Salatino, A, Ricci, R, et al. Medial anterior prefrontal cortex stimulation downregulates implicit reactions to threats and prevents the return of fear. eLife. (2024) 13:e85951. doi: 10.7554/eLife.85951
38. Seeley, WW, Menon, V, Schatzberg, AF, Keller, J, Glover, GH, Kenna, H, et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci. (2007) 27:2349–56. doi: 10.1523/JNEUROSCI.5587-06.2007
39. Uddin, LQ. Salience network of the human brain. University of Miami, FL, USA: Elsevier Inc. (2016).
40. Xia, J, Fan, J, Liu, W, Du, H, Zhu, J, Yi, J, et al. Functional connectivity within the salience network differentiates autogenous-from reactive-type obsessive-compulsive disorder. Prog Neuro Psychopharmacol Biol Psychiatry. (2020) 98:109813. doi: 10.1016/j.pnpbp.2019.109813
41. Zhang, A, Yang, C, Li, G, Wang, Y, Liu, P, Liu, Z, et al. Functional connectivity of the prefrontal cortex and amygdala is related to depression status in major depressive disorder. J Affect Disord. (2020) 274:897–902. doi: 10.1016/j.jad.2020.05.053
42. Hossein, S, Cooper, JA, DeVries, BAM, Nuutinen, MR, Hahn, EC, Kragel, PA, et al. Effects of acute stress and depression on functional connectivity between prefrontal cortex and the amygdala. Mol Psychiatry. (2023) 28:4602–12. doi: 10.1038/s41380-023-02056-5
43. Tozzi, L, Zhang, X, Pines, A, Olmsted, AM, Zhai, ES, Anene, ET, et al. Personalized brain circuit scores identify clinically distinct biotypes in depression and anxiety. Nat Med. (2024) 30:2076–87. doi: 10.1038/s41591-024-03057-9
44. Cappon, DB, and Pascual-Leone, A. Toward precision noninvasive brain stimulation. Am J Psychiatry. (2024) 181:795–805. doi: 10.1176/appi.ajp.20240643
45. Williams, LM, and Whitfield, GS. Neuroimaging for precision medicine in psychiatry. Neuropsychopharmacology. (2024) 50:246–57. doi: 10.1038/s41386-024-01917-z
46. Kaiser, RH, Andrews-Hanna, JR, Wager, TD, and Pizzagalli, DA. Large-scale network dysfunction in major depressive disorder: a meta-analysis of resting-state functional connectivity. JAMA Psychiatry. (2015) 72:603–11. doi: 10.1001/jamapsychiatry.2015.0071
47. Neufeld, N, Alexopoulos, G, Dickie, E, Jeyachandra, J, Mulsant, B, Whyte, E, et al. Resting state functional connectivity and relapse in psychotic depression. Biol Psychiatry. (2021) 89:S96–7. doi: 10.1016/j.biopsych.2021.02.252
48. Wang, Y, Wang, C, Zhou, J, Chen, X, Liu, R, Zhang, Z, et al. Contribution of resting-state functional connectivity of the subgenual anterior cingulate to prediction of antidepressant efficacy in patients with major depressive disorder. Transl Psychiatry. (2024) 14:399. doi: 10.1038/s41398-024-03117-1
49. Dunlop, BW, Cha, J, Choi, KS, Nemeroff, CB, Craighead, WE, and Mayberg, HS. Functional connectivity of salience and affective networks among remitted depressed patients predicts episode recurrence. Neuropsychopharmacology. (2023) 48:1901–9. doi: 10.1038/s41386-023-01653-w
50. Krug, S, Müller, T, Kayali, Ö, Leichter, E, Peschel, SKV, Jahn, N, et al. Altered functional connectivity in common resting-state networks in patients with major depressive disorder: a resting-state functional connectivity study. J Psychiatr Res. (2022) 155:33–41. doi: 10.1016/j.jpsychires.2022.07.040
51. Belchev, Z, Gilboa, A, Binns, M, Colella, B, Glazer, J, Mikulis, DJ, et al. Progressive neurodegeneration across chronic stages of severe traumatic brain injury. J Head Trauma Rehabil. (2022) 37:E144–56. doi: 10.1097/HTR.0000000000000696
52. Christensen, BK, Colella, B, Inness, E, Hebert, D, Monette, G, Bayley, M, et al. Recovery of cognitive function after traumatic brain injury: a multilevel modeling analysis of Canadian outcomes. Arch Phys Med Rehabil. (2008) 89:S3–S15. doi: 10.1016/j.apmr.2008.10.002
53. Green, RE, Colella, B, Christensen, B, Johns, K, Frasca, D, Bayley, M, et al. Examining moderators of cognitive recovery trajectories after moderate to severe traumatic brain injury. Arch Phys Med Rehabil. (2008) 89:S16–24. doi: 10.1016/j.apmr.2008.09.551
54. Till, C, Christensen, BK, and Green, RE. Use of the personality assessment inventory (PAI) in individuals with traumatic brain injury. Brain Inj. (2009) 23:655–65. doi: 10.1080/02699050902970794
55. So, I, Meusel, L-AC, Sharma, B, Monette, GA, Colella, B, Wheeler, AL, et al. Longitudinal patterns of functional connectivity in moderate-to-severe traumatic brain injury. J Neurotrauma. (2023) 40:665–82. doi: 10.1089/neu.2022.0242
56. Morey, LC. Personality assessment inventory. Odessa, FL: Psychological Assessment Resources (1991).
57. Keiski, MA, Shore, DL, and Hamilton, JM. The role of depression in verbal memory following traumatic brain injury. Clin Neuropsychol. (2007) 21:744–61. doi: 10.1080/13854040600775346
58. Lange, RT, Brickell, T, French, LM, Ivins, B, Bhagwat, A, Pancholi, S, et al. Risk factors for Postconcussion symptom reporting after traumatic brain injury in U.S. military service members. J Neurotrauma. (2013) 30:237–46. doi: 10.1089/neu.2012.2685
59. Slavin-Mulford, J, Sinclair, SJ, Stein, M, Malone, J, Bello, I, and Blais, MA. External validity of the personality assessment inventory (PAI) in a clinical sample. J Pers Assess. (2012) 94:593–600. doi: 10.1080/00223891.2012.681817
60. Pruim, RHR, Mennes, M, van Rooij, D, Llera, A, Buitelaar, JK, and Beckmann, CF. ICA-AROMA: a robust ICA-based strategy for removing motion artifacts from fMRI data. NeuroImage. (2015) 112:267–77. doi: 10.1016/j.neuroimage.2015.02.064
61. Seewoo, BJ, Joos, AC, and Feindel, KW. An analytical workflow for seed-based correlation and independent component analysis in interventional resting-state fMRI studies. Neurosci Res. (2021) 165:26–37. doi: 10.1016/j.neures.2020.05.006
62. Evans, AC, Collins, DL, Mills, SR, Brown, ED, Kelly, RL, and Peters, TM. 3D statistical neuroanatomical models from 305 MRI volumes. 1993 IEEE conference record nuclear science symposium and medical imaging conference; (1993).
63. Caballero-Gaudes, C, and Reynolds, RC. Methods for cleaning the BOLD fMRI signal. NeuroImage. (2017) 154:128–49. doi: 10.1016/j.neuroimage.2016.12.018
64. Woolrich, MW, Ripley, BD, Brady, M, and Smith, SM. Temporal autocorrelation in univariate linear modeling of FMRI data. NeuroImage. (2001) 14:1370–86. doi: 10.1006/nimg.2001.0931
65. Woolrich, MW, Behrens, TEJ, Beckmann, CF, Jenkinson, M, and Smith, SM. Multilevel linear modelling for FMRI group analysis using Bayesian inference. NeuroImage. (2004) 21:1732–47. doi: 10.1016/j.neuroimage.2003.12.023
66. Del Giacco, AC, Morales, AM, Jones, SA, Barnes, SJ, and Nagel, BJ. Ventral striatal-cingulate resting-state functional connectivity in healthy adolescents relates to later depression symptoms in adulthood. J Affect Disord. (2024) 365:205–12. doi: 10.1016/j.jad.2024.08.028
67. Dunlop, K, Victoria, LW, Downar, J, Gunning, FM, and Liston, C. Accelerated brain aging predicts impulsivity and symptom severity in depression. Neuropsychopharmacology. (2021) 46:911–9. doi: 10.1038/s41386-021-00967-x
68. Ponsford, JL, Downing, MG, Olver, J, Ponsford, M, Acher, R, Carty, M, et al. Longitudinal follow-up of patients with traumatic brain injury: outcome at two, five, and ten years post-injury. J Neurotrauma. (2014) 31:64–77. doi: 10.1089/neu.2013.2997
69. Matsumae, M, Kikinis, R, Mórocz, IA, Lorenzo, AV, Sándor, T, Albert, MS, et al. Age-related changes in intracranial compartment volumes in normal adults assessed by magnetic resonance imaging. J Neurosurg. (1996) 84:982–91. doi: 10.3171/jns.1996.84.6.0982
70. Eklund, A, Nichols, TE, and Knutsson, H. Cluster failure: why fMRI inferences for spatial extent have inflated false-positive rates. Proc Natl Acad Sci. (2016) 113:7900–5. doi: 10.1073/pnas.1602413113
71. Smith, SM, and Nichols, TE. Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage. (2009) 44:83–98. doi: 10.1016/j.neuroimage.2008.03.061
72. Desikan, RS, Ségonne, F, Fischl, B, Quinn, BT, Dickerson, BC, Blacker, D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage. (2006) 31:968–80. doi: 10.1016/j.neuroimage.2006.01.021
73. Raizman, R, Itzhaki, N, Sirkin, J, Meningher, I, Tsarfaty, G, Keren, O, et al. Decreased homotopic functional connectivity in traumatic brain injury. Cereb Cortex. (2022) 33:1207–16. doi: 10.1093/cercor/bhac130
74. Peters, AT, Burkhouse, K, Feldhaus, CC, Langenecker, SA, and Jacobs, RH. Aberrant resting-state functional connectivity in limbic and cognitive control networks relates to depressive rumination and mindfulness: a pilot study among adolescents with a history of depression. J Affect Disord. (2016) 200:178–81. doi: 10.1016/j.jad.2016.03.059
75. Pizzagalli, DA, and Roberts, AC. Correction: prefrontal cortex and depression. Neuropsychopharmacology. (2022) 47:609. doi: 10.1038/s41386-021-01160-w
76. Ramnani, N, and Owen, AM. Anterior prefrontal cortex: insights into function from anatomy and neuroimaging. Nat Rev Neurosci. (2004) 5:184–94. doi: 10.1038/nrn1343
77. Kovach, CK, Daw, ND, Rudrauf, D, Tranel, D, O’Doherty, JP, and Adolphs, R. Anterior prefrontal cortex contributes to action selection through tracking of recent reward trends. J Neurosci. (2012) 32:8434–42. doi: 10.1523/JNEUROSCI.5468-11.2012
78. Pannekoek, JN, van der Werff, SJA, Meens, PHF, van den Bulk, BG, Jolles, DD, Veer, IM, et al. Aberrant resting-state functional connectivity in limbic and salience networks in treatment-naïve clinically depressed adolescents. J Child Psychol Psychiatry. (2014) 55:1317–27. doi: 10.1111/jcpp.12266
79. Sylvester, CM, Barch, DM, Corbetta, M, Power, JD, Schlaggar, BL, and Luby, JL. Resting state functional connectivity of the ventral attention network in children with a history of depression or anxiety. J Am Acad Child Adolesc Psychiatry. (2013) 52:1326–36.e5. doi: 10.1016/j.jaac.2013.10.001
80. Nestmann, S, Karnath, H-O, and Rennig, J. The role of ventral stream areas for viewpoint-invariant object recognition. NeuroImage. (2022) 251:119021. doi: 10.1016/j.neuroimage.2022.119021
81. Geckeler, KC, Barch, DM, and Karcher, NR. Associations between social behaviors and experiences with neural correlates of implicit emotion regulation in middle childhood. Neuropsychopharmacology. (2022) 47:1169–79. doi: 10.1038/s41386-022-01286-5
82. Jamieson, AJ, Leonards, C, Davey, CG, and Harrison, BJ. Major depressive disorder associated alterations in the effective connectivity of the face processing network: A systematic review (2022). PsyArXiv. doi: 10.31234/osf.io/pc9s8
83. Pilmeyer, J, Huijbers, W, Lamerichs, R, Jansen, JFA, Breeuwer, M, and Zinger, S. Functional MRI in major depressive disorder: a review of findings, limitations, and future prospects. J Neuroimaging. (2022) 32:582–95. doi: 10.1111/jon.13011
84. Hikosaka, O. Neural systems for control of voluntary action—A hypothesis. Adv Biophys. (1998) 35:81–102. doi: 10.1016/S0065-227X(98)80004-X
85. Saito, T, Muragaki, Y, Tamura, M, Maruyama, T, Nitta, M, Tsuzuki, S, et al. Correlation between localization of supratentorial glioma to the precentral gyrus and difficulty in identification of the motor area during awake craniotomy. J Neurosurg. (2020) 134:1490–9. doi: 10.3171/2020.2.JNS193471
86. Kohn, N, Eickhoff, SB, Scheller, M, Laird, AR, Fox, PT, and Habel, U. Neural network of cognitive emotion regulation--an ALE meta-analysis and MACM analysis. NeuroImage. (2014) 87:345–55. doi: 10.1016/j.neuroimage.2013.11.001
87. Liu, P, Tu, H, Zhang, A, Yang, C, Liu, Z, Lei, L, et al. Brain functional alterations in MDD patients with somatic symptoms: a resting-state fMRI study. J Affect Disord. (2021) 295:788–96. doi: 10.1016/j.jad.2021.08.143
88. Lima Portugal, LC, Alves, RCS, Junior, OF, Sanchez, TA, Mocaiber, I, Volchan, E, et al. Interactions between emotion and action in the brain. NeuroImage. (2020) 214:116728. doi: 10.1016/j.neuroimage.2020.116728
89. Philippi, CL, Cornejo, MD, Frost, CP, Walsh, EC, Hoks, RM, Birn, R, et al. Neural and behavioral correlates of negative self-focused thought associated with depression. Hum Brain Mapp. (2018) 39:2246–57. doi: 10.1002/hbm.24003
90. Zeng, L-L, Shen, H, Liu, L, Wang, L, Li, B, Fang, P, et al. Identifying major depression using whole-brain functional connectivity: a multivariate pattern analysis. Brain. (2012) 135:1498–507. doi: 10.1093/brain/aws059
91. Kustubayeva, A, Eliassen, J, Matthews, G, and Nelson, E. FMRI study of implicit emotional face processing in patients with MDD with melancholic subtype. Front Hum Neurosci. (2023) 17:1029789. doi: 10.3389/fnhum.2023.1029789
92. Li, X, and Wang, J. Abnormal neural activities in adults and youths with major depressive disorder during emotional processing: a meta-analysis. Brain Imaging Behav. (2021) 15:1134–54. doi: 10.1007/s11682-020-00299-2
93. Mellem, MS, Jasmin, KM, Peng, C, and Martin, A. Sentence processing in anterior superior temporal cortex shows a social-emotional bias. Neuropsychologia. (2016) 89:217–24. doi: 10.1016/j.neuropsychologia.2016.06.019
94. Drysdale, AT, Grosenick, L, Downar, J, Dunlop, K, Mansouri, F, Meng, Y, et al. Erratum: resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. (2017) 23:264. doi: 10.1038/nm0217-264d
95. Dunlop, BW, Rajendra, JK, Craighead, WE, Kelley, ME, McGrath, CL, Choi, KS, et al. Functional connectivity of the subcallosal cingulate cortex and differential outcomes to treatment with cognitive-behavioral therapy or antidepressant medication for major depressive disorder. Am J Psychiatry. (2017) 174:533–45. doi: 10.1176/appi.ajp.2016.16050518
96. Yu, H, Li, M-L, Li, Y-F, Li, X-J, Meng, Y, Liang, S, et al. Anterior cingulate cortex, insula and amygdala seed-based whole brain resting-state functional connectivity differentiates bipolar from unipolar depression. J Affect Disord. (2020) 274:38–47. doi: 10.1016/j.jad.2020.05.005
97. Iraji, A, Benson, RR, Welch, RD, O’Neil, BJ, Woodard, JL, Ayaz, SI, et al. Resting state functional connectivity in mild traumatic brain injury at the acute stage: independent component and seed-based analyses. J Neurotrauma. (2015) 32:1031–45. doi: 10.1089/neu.2014.3610
98. Shumskaya, E, van Gerven, MAJ, Norris, DG, Vos, PE, and Kessels, RPC. Abnormal connectivity in the sensorimotor network predicts attention deficits in traumatic brain injury. Exp Brain Res. (2017) 235:799–807. doi: 10.1007/s00221-016-4841-z
99. Coxe, KA, Lee, G, Kagotho, N, and Eads, R. Mental health service utilization among adults with head injury with loss of consciousness: implications for social work. Health Soc Work. (2021) 46:125–35. doi: 10.1093/hsw/hlab005
100. Juengst, SB, Kumar, RG, and Wagner, AK. A narrative literature review of depression following traumatic brain injury: prevalence, impact, and management challenges. Psychol Res Behav Manag. (2017) 10:175–86. doi: 10.2147/PRBM.S113264
101. Heath, LM, Kidwai, MR, Colella, B, Monette, G, Tselichtchev, P, Tomaszczyk, JC, et al. Predictors and functional outcomes associated with longitudinal trajectories of anxiety and depression from 2 to ≥36 months after moderate to severe traumatic brain injury. J Neurotrauma. (2023) 40:2311–20. doi: 10.1089/neu.2023.0003
102. Chaudhary, S, Hu, S, Hu, K, Dominguez, JC, Chao, HH, and Li, C-SR. Sex differences in the effects of trait anxiety and age on resting-state functional connectivities of the amygdala. J Affect Disord Rep. (2023) 14:100646. doi: 10.1016/j.jadr.2023.100646
103. van der Wijk, G, Zamyadi, M, Bray, S, Hassel, S, Arnott, SR, Frey, BN, et al. Large individual differences in functional connectivity in the context of major depression and antidepressant pharmacotherapy. eNeuro. (2024) 11:ENEURO.0286–23.2024. doi: 10.1523/ENEURO.0286-23.2024
104. Hu, L, Xiao, M, Ai, M, Wang, W, Chen, J, Tan, Z, et al. Disruption of resting-state functional connectivity of right posterior insula in adolescents and young adults with major depressive disorder. J Affect Disord. (2019) 257:23–30. doi: 10.1016/j.jad.2019.06.057
105. Schmaal, L, Hibar, DP, Sämann, PG, Hall, GB, Baune, BT, Jahanshad, N, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA major depressive disorder working group. Mol Psychiatry. (2017) 22:900–9. doi: 10.1038/mp.2016.60
106. Cheng, B, Zhou, Y, Kwok, VPY, Li, Y, Wang, S, Zhao, Y, et al. Altered functional connectivity density and couplings in postpartum depression with and without anxiety. Soc Cogn Affect Neurosci. (2022) 17:756–66. doi: 10.1093/scan/nsab127
107. Wade, BSC, Loureiro, J, Sahib, A, Kubicki, A, Joshi, SH, Hellemann, G, et al. Anterior default mode network and posterior insular connectivity is predictive of depressive symptom reduction following serial ketamine infusion. Psychol Med. (2022) 52:2376–86. doi: 10.1017/S0033291722001313
108. Peng, X, Lau, WKW, Wang, C, Ning, L, and Zhang, R. Impaired left amygdala resting state functional connectivity in subthreshold depression individuals. Sci Rep. (2020) 10:17207. doi: 10.1038/s41598-020-74166-x
109. Scheuer, H, Alarcón, G, Demeter, DV, Earl, E, Fair, DA, and Nagel, BJ. Reduced fronto-amygdalar connectivity in adolescence is associated with increased depression symptoms over time. Psychiatry Res Neuroimaging. (2017) 266:35–41. doi: 10.1016/j.pscychresns.2017.05.012
Keywords: chronic moderate-to-severe traumatic brain injury, depression, anterior prefrontal cortex, seed-based connectivity analysis, personality assessment inventory, fusiform gyrus
Citation: Elfaki LA, Sharma B, Meusel L-AC, So I, Colella B, Wheeler AL, Harris JE and Green REA (2025) Examining anterior prefrontal cortex resting-state functional connectivity patterns associated with depressive symptoms in chronic moderate-to-severe traumatic brain injury. Front. Neurol. 16:1541520. doi: 10.3389/fneur.2025.1541520
Received: 07 December 2024; Accepted: 03 March 2025;
Published: 28 March 2025.
Edited by:
Tony L. Strickland, Center for Postconcussion Syndrome and PTSD Treatment, United StatesReviewed by:
Anthony P. Salvatore, University of Louisiana at Lafayette, United StatesCopyright © 2025 Elfaki, Sharma, Meusel, So, Colella, Wheeler, Harris and Green. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Layan A. Elfaki, bGF5YW4uZWxmYWtpQG1haWwudXRvcm9udG8uY2E=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.