
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Med. , 12 March 2025
Sec. Geriatric Medicine
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1546988
This article is part of the Research Topic Sarcopenia and Nutrition in Chronic Kidney Disease View all 4 articles
 Renjie Lu1
Renjie Lu1 Shiyun Wang2
Shiyun Wang2 Pinghua Chen2
Pinghua Chen2 Fangfang Li1,2
Fangfang Li1,2 Pan Li2
Pan Li2 Qian Chen1
Qian Chen1 Xuefei Li1
Xuefei Li1 Fangyu Li1
Fangyu Li1 Suxia Guo1
Suxia Guo1 Jinlin Zhang1,3
Jinlin Zhang1,3 Dan Liu2*
Dan Liu2* Zhijun Hu2*
Zhijun Hu2*Background: Sarcopenia frequently occurs as a complication among individuals with chronic kidney disease (CKD), contributing to poorer clinical outcomes. This research aimed to create and assess a predictive model for the risk of sarcopenia in CKD patients, utilizing data obtained from the China Health and Retirement Longitudinal Study (CHARLS).
Methods: Sarcopenia was diagnosed based on the Asian Working Group for Sarcopenia (AWGS 2019) criteria, including low muscle strength, reduced physical performance, and low muscle mass. The 2015 CHARLS data were split randomly into a training set (70%) and a testing set (30%). Forty-nine variables encompassing socio-demographic, behavioral, health status, and biochemical factors were analyzed. LASSO regression identified the most relevant predictors, and a logistic regression model was used to explore factors associated with sarcopenia. A nomogram was developed for risk prediction. Model accuracy was evaluated using calibration curves, while predictive performance was assessed through receiver operating characteristic (ROC) and decision curve analysis (DCA). Four machine learning algorithms were utilized, with the optimal model undergoing hyperparameter optimization to evaluate the significance of predictive factors.
Results: A total of 1,092 CKD patients were included, with 231 (21.2%) diagnosed with sarcopenia. Multivariate logistic regression revealed that age, waist circumference, LDL-C, HDL-C, triglycerides, and diastolic blood pressure are significant predictors. These factors were used to construct the nomogram. The predictive model achieved an AUC of 0.886 (95% CI: 0.858–0.912) in the training set and 0.859 (95% CI: 0.811–0.908) in the validation set. Calibration curves showed good agreement between predicted and actual outcomes. ROC and DCA analyses confirmed the model’s strong predictive performance. The Gradient Boosting Machine (GBM) outperformed other machine learning models. Applying Bayesian optimization to the GBM achieved an AUC of 0.933 (95% CI: 0.913–0.953) on the training set and 0.932 (95% CI: 0.905–0.960) on the validation set. SHAP values identified age and waist circumference as the most influential factors.
Conclusion: The nomogram provides a reliable tool for predicting sarcopenia in CKD patients. The GBM model exhibits strong predictive accuracy, positioning it as a valuable tool for clinical risk assessment and management of sarcopenia in this population.
Sarcopenia, characterized by the progressive loss of skeletal muscle mass, strength, and functionality, poses an increasing health challenge, particularly for older adults and individuals with chronic illnesses. As kidney function declines, there is a notable deterioration in muscle health, which significantly affects physical capabilities and overall quality of life (1). Chronic kidney disease (CKD) affects millions worldwide and leads to various complications, including metabolic imbalances, nutritional deficiencies, and reduced physical capacity, all of which exacerbate muscle function decline (2–4). Sarcopenia frequently occurs as a complication among individuals with CKD, with a global prevalence of 24.5% across all disease stages (5). This progressive muscle deterioration contributes to poorer clinical outcomes, including increased mortality risk (6). While recent studies have developed nomograms for sarcopenia prediction in hemodialysis populations, reliable tools for non-dialysis CKD patients remain scarce. This gap underscores the need for models adaptable to broader CKD stages (7, 8). Developing an accurate risk prediction model would aid healthcare providers in identifying high-risk individuals early, facilitating the timely implementation of interventions focused on nutrition and physical rehabilitation. These interventions are crucial for enhancing the quality of life, minimizing the risk of additional health complications, and reducing overall healthcare costs.
The China Health and Retirement Longitudinal Study (CHARLS) provides a valuable opportunity to develop such a predictive model (9). It is a nationally representative survey of Chinese adults aged 45 and older, designed to collect comprehensive data on health, social, and economic factors (10). The dataset includes comprehensive information, such as biomarkers, physical measurements, and health histories, making it an ideal resource for developing models that predict adverse health outcomes associated with aging (11). The primary objective of this study is to develop and validate a predictive model for muscle health decline in patients with CKD using CHARLS data. The model is designed to enhance the early detection of high-risk individuals, facilitating prompt interventions to avert severe health decline.
This study draws on data from CHARLS. The CHARLS dataset includes extensive details about participants’ demographic profiles, health conditions, socioeconomic status, and lifestyle habits. This study analyzes data obtained from the publicly accessible 2015 wave of CHARLS, which provides comprehensive health-related measurements.
Participants eligible for inclusion in the study were individuals diagnosed with chronic kidney disease (CKD), identified through either self-reported confirmation by a physician or clinical diagnostic criteria, such as an estimated glomerular filtration rate (eGFR) of less than 60 mL/min/1.73 m2 (12). To uphold the robustness of the analysis, those with incomplete data on key variables—including sarcopenia-related metrics, renal function indicators, and other covariates integral to the predictive model—were excluded. Consequently, the final analytic sample comprised 1,092 CKD patients aged 45 years or older, each with comprehensive data suitable for evaluation (Figure 1).
Sarcopenia status was evaluated in accordance with the 2019 criteria established by the Asian Working Group for Sarcopenia (AWGS), encompassing three core components: muscle strength, appendicular skeletal muscle mass (ASM), and physical performance. The diagnosis of sarcopenia was assigned to individuals exhibiting reduced muscle mass in conjunction with either compromised muscle strength or diminished physical performance (13). Muscle Strength was measured using a Yuejian™ WL-1000 dynamometer. Participants stood upright, holding the dynamometer at a right angle to their body, and were instructed to squeeze the handle as firmly as possible for 3 s. Two trials were performed for each hand, with a minimum rest interval of 15 s between consecutive trials to minimize fatigue. The highest value across all four trials (left and right hands combined) was recorded as the final grip strength. Consistent with the AWGS 2019 criteria, low grip strength was defined as <28 kg for men and < 18 kg for women.
ASM was estimated using a validated anthropometric equation specifically designed for Chinese residents. Previous studies have demonstrated that this equation yields results closely aligned with those obtained from dual X-ray absorptiometry (DXA) (14, 15). Low muscle mass was defined as the sex-specific lowest 20% of height-adjusted ASM (ASM/Ht2) within the cohort, consistent with methodologies applied in large-scale Chinese sarcopenia studies (16). Specifically, for women, low muscle mass was defined as less than 5.37 kg/m2, and for men, it was less than 7.06 kg/m2. The ASM equation utilized is:
Additionally, physical performance was assessed through gait speed and a 5-time chair stand test. For gait speed, participants completed two trials of a 2.5-meter walk at their usual pace, with timing initiated when the first foot crossed the start line and stopped at the finish line; the maximum speed from either trial was used, with <1 m/s defined as low performance. 5-Time Chair Stand Test required participants to rise five times as quickly as possible with arms crossed over the chest. Timing began on the command “ready? stand” and ended when the participant’s buttocks touched the chair after the fifth stand. A practice trial was allowed prior to formal measurement. Low performance was defined as a completion time ≥ 12 s (13).
The demographic factors considered in this study were age, gender, marital status, and education level. Marital status was categorized as either married or not married. Educational attainment was categorized into primary, secondary, or tertiary education levels.
Lifestyle and behavioral factors encompassed smoking status, alcohol use, physical and social activities, sleep quality, and life satisfaction. Smoking status was categorized into two groups: current smoker and non-smoker. Alcohol consumption was categorized into two groups based on drinking history: individuals with a history of alcohol use and those without. Physical activity was categorized by frequency and intensity as high, moderate, or low. Social activity was classified into two categories based on engagement: those who participate in social activities and those who do not. Sleep quality was classified into four levels based on self-reported frequency of poor sleep: “Rarely or none of the time,” indicating poor sleep is infrequent or almost never occurs; “Some or a little of the time,” suggesting poor sleep occurs occasionally but not regularly; “Occasionally or a moderate amount of the time,” denoting that poor sleep is experienced somewhat regularly; and “Most or all of the time,” reflecting frequent or consistent poor sleep. Life satisfaction was measured and divided into three levels: “Fair,” indicating average life satisfaction; “Good,” indicating above-average satisfaction where individuals feel mostly content with their lives; and “Poor,” signifying below-average satisfaction, where individuals often feel dissatisfied.
Health-related factors included body index, self-perceived health status, ADL (Activities of Daily Living) disability, IADL (Instrumental Activities of Daily Living) disability, and depressive symptoms. Body index encompassed body mass index (BMI), waist circumference, height, and weight. BMI was determined by dividing weight in kilograms by height in meters squared. Self-reported health status was categorized as good, fair, or poor based on participants’ own assessment of their overall health. The ADL scale assesses participants’ ability to perform basic tasks such as dressing, bathing, feeding, transferring, toileting, and maintaining continence. If a participant is completely unable to perform any one of these tasks, they are considered to have an ADL disability. Similarly, the IADL scale evaluates more complex tasks, including doing chores, cooking, shopping, managing finances, and taking medication. If a participant is completely unable to perform any one of these tasks, they are considered to have an IADL disability (17–19). Depressive symptoms were assessed using a 10-item screening questionnaire derived from the Center for Epidemiologic Studies Depression Scale (CES-D), which evaluates depressive mood and behavior. A CES-D score greater than 10 was used to define the presence of depression (20, 21).
Chronic diseases were identified through self-reported diagnoses, including mental disease, stroke, heart disease, cardiovascular disease, arthritis, dyslipidemia, liver disease, digestive disease, hypertension, chronic lung disease, and asthma.
Laboratory test indicators included blood pressure, lipid profile, renal function markers, hematological indicators, and inflammatory markers. Blood pressure measurements included systolic and diastolic values. The lipid profile included low-density lipoprotein (LDL-C), triglycerides (TG), high-density lipoprotein (HDL-C), and total cholesterol (TC). Renal function markers included cystatin C, serum creatinine, blood urea nitrogen (BUN), and uric acid levels. The estimated glomerular filtration rate (eGFR) serves as an important indicator of kidney function. In this study, we used a formula based on serum cystatin C (Cys) levels, known for its accuracy. This method, derived from the CKD-EPI equation, provides a reliable assessment of kidney function, especially in populations where creatinine-based estimates are less accurate (22). Hematological indicators included glycated hemoglobin and C-reactive protein.
The CHARLS 2015 dataset was randomly divided into two subsets: a training set (70% of the data) and an internal validation set (30% of the data). LASSO (Least Absolute Shrinkage and Selection Operator) regression was performed on the training set to identify possible predictors of sarcopenia in individuals with chronic kidney disease (CKD). Variables that showed significant associations were included in a subsequent multivariate logistic regression analysis, where independent predictors were determined using a significance level of p < 0.05. These identified predictors were then utilized to create a nomogram for evaluating sarcopenia risk (23).
The model’s ability to discriminate was assessed using the area under the receiver operating characteristic curve (AUC). Calibration was examined through the use of calibration plots and the Hosmer-Lemeshow test, which assessed how well the predicted probabilities aligned with the actual outcomes. To determine the clinical utility of the nomogram, Decision Curve Analysis (DCA) was performed.
To evaluate the predictive performance of the identified predictors, four machine learning models were developed: Gradient Boosting Machine (GBM), Regularized Discriminant Analysis (RDA), Random Forest (RF), and Support Vector Machine (SVM). For further optimization, Bayesian optimization was employed for hyperparameter tuning. Additionally, SHapley Additive exPlanations (SHAP) values and variable importance metrics were used to assess feature contributions.
Principal Component Analysis (PCA) was conducted to reduce dimensionality and mitigate multicollinearity (24). The analysis included all predictor variables, excluding ID and the outcome variable (sarcopenia). The number of principal components (PCs) retained was determined based on cumulative explained variance, with the goal of capturing at least 75% of the total variance. K-means clustering was performed to identify subgroups with similar characteristics. The optimal number of clusters was determined using the Elbow method and Silhouette coefficient. The clusters were analyzed in relation to sarcopenia prevalence to explore potential group-specific differences (25).
All statistical analyses were performed using R version 4.3.1. Continuous variables are expressed as mean ± standard deviation (SD) or median (interquartile range, IQR), depending on suitability, whereas categorical variables are shown as frequencies and percentages. Appropriate statistical tests, including Student’s t-test, chi-square test, and Mann–Whitney U test, were employed for group comparisons. A two-tailed p-value below 0.05 was deemed statistically significant.
A total of 1,092 CKD patients were included in the final analysis, with 231 diagnosed with sarcopenia and 861 without, yielding a sarcopenia incidence of 21.2%. The demographic and clinical features of the sample, which were used for model development, are presented in Table 1. Notable differences were found between the groups in terms of age, marital status, BMI, and waist circumference (p < 0.05). Individuals with sarcopenia were generally older and exhibited a higher rate of comorbidities, including hypertension and dyslipidemia (p < 0.05).
The cohort exhibited significant gradients in clinical profiles across CKD stages (Supplementary Table S2). Participants with advanced CKD (G3b-G5) were older (mean age 71.6 vs. 60.9 years in G1-G2, p < 0.001) and had higher rates of hypertension (58.5% vs. 38.9%, p < 0.001) and IADL disability (24.5% vs. 13.0%, p = 0.001). Cognitive function declined progressively (8.13 vs. 10.38, p < 0.001), paralleled by elevated inflammatory markers (CRP: 4.49 vs. 2.70 mg/L, p < 0.001). Sarcopenia prevalence nearly doubled in G3b-G5 (29.9%) compared to G1-G2 (16.2%, p < 0.001), underscoring the multifactorial burden in advanced CKD.
The dataset, comprising 1,092 participants, was randomly divided into two subsets: a training set and an internal validation set. To ensure that the two subsets were comparable, we conducted thorough statistical comparisons, the results of which are provided in Supplementary Table S1. These analyses were aimed at evaluating the equivalence of the training and validation cohorts across key demographic and clinical characteristics. The findings revealed no significant differences (p > 0.05).
LASSO regression was applied to the training dataset (n = 764) to identify the most relevant predictors of sarcopenia in CKD patients (Figure 2). The following predictors were retained in the model: age, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglycerides (TG), waist circumference, and diastolic blood pressure (DBP). These variables were subsequently included in the multivariate logistic regression analysis.
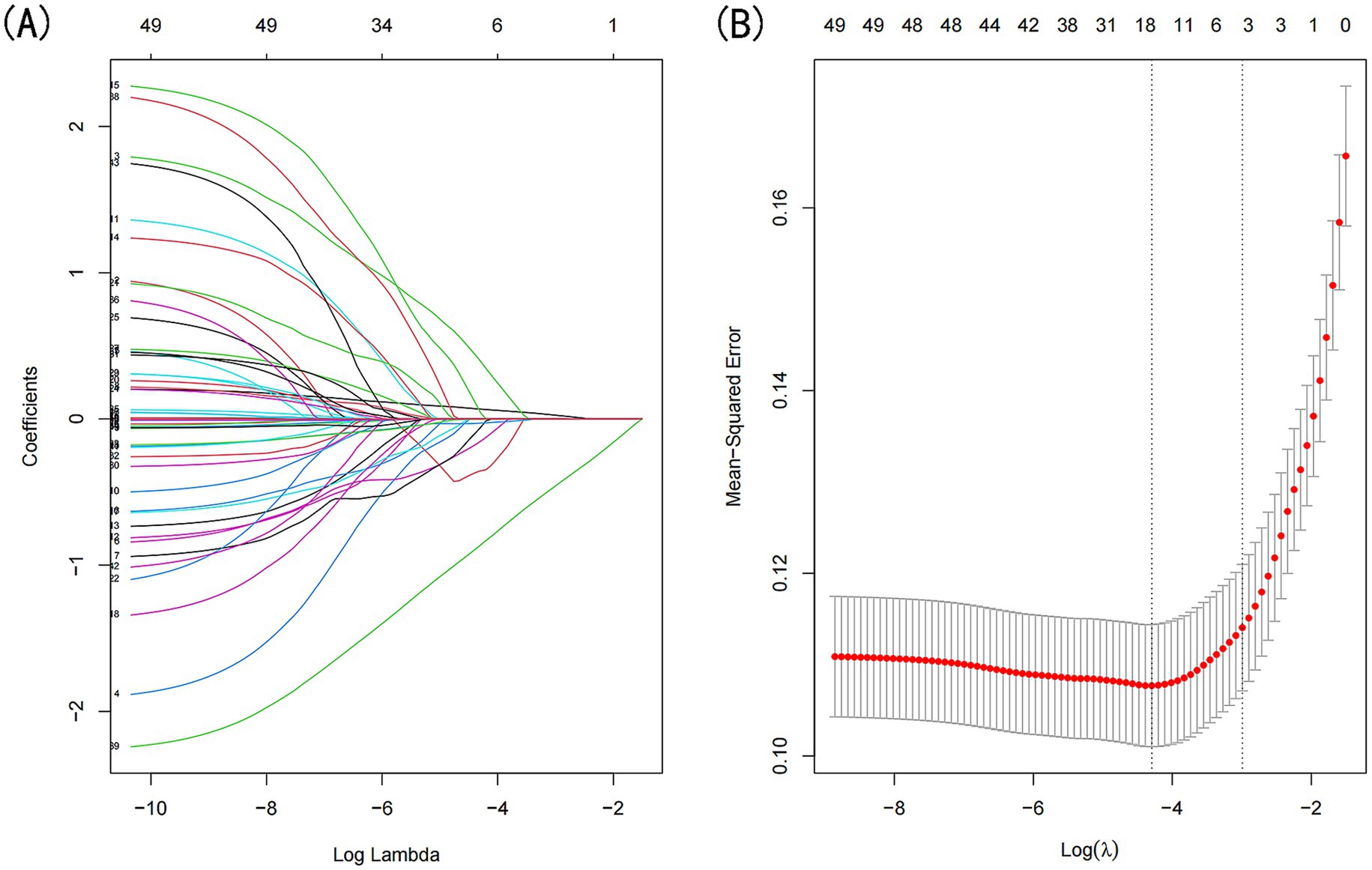
Figure 2. Demographic and clinical feature selection was performed using the LASSO regression model. (A) Coefficient profile was generated based on the logarithmic (lambda) sequence, with non-zero coefficients identified at the optimal lambda. (B) The optimal lambda parameter was selected through tenfold cross-validation using the minimum criteria. The partial likelihood deviation (binomial deviation) curve relative to log(lambda) was plotted, and a vertical line was drawn at the optimal value using the 1-SE criterion.
Multivariate logistic regression was performed to validate the independent predictors of sarcopenia. The results are presented in Table 2. Age: OR = 4.39 (95% CI: 3.08–6.26), LDLC: OR = 0.66 (95% CI: 0.48–0.90), HDLC: OR = 1.40 (95% CI: 1.06–1.84), TG: OR = 0.53 (95% CI-0.37, 0.76), Waist: OR = 0.32 (95% CI: 0.23–0.43), DBP:OR = 0.62 (95% CI: 0.45–0.86) were identified as significant independent predictors of sarcopenia (p < 0.05).
The model’s performance was evaluated using several methods, including the area under the receiver operating characteristic curve (AUC) for both the training and validation datasets, as well as calibration curves and decision curve analysis (DCA). In the training set (Figure 3A), the model achieved an AUC of 0.886 (95% CI: 0.858–0.912), while in the validation set (Figure 3B), the AUC was 0.859 (95% CI: 0.811–0.908), demonstrating ability to distinguish between sarcopenia patients and non-patients. The Hosmer-Lemeshow goodness-of-fit test yielded p-values greater than 0.05 for both the training set (χ2 = 13.302, df = 8, p = 0.102) and validation set (χ2 = 6.748, df = 8, p = 0.564). The calibration curve was near the ideal line, further validating the model’s predictive accuracy (Figure 4). Decision curve analysis was performed to evaluate the clinical utility of the model. DCA was conducted to assess the clinical value of the model, with the resulting curve indicating substantial net benefit across a range of decision thresholds, underscoring its potential utility in clinical practice (Figure 5).
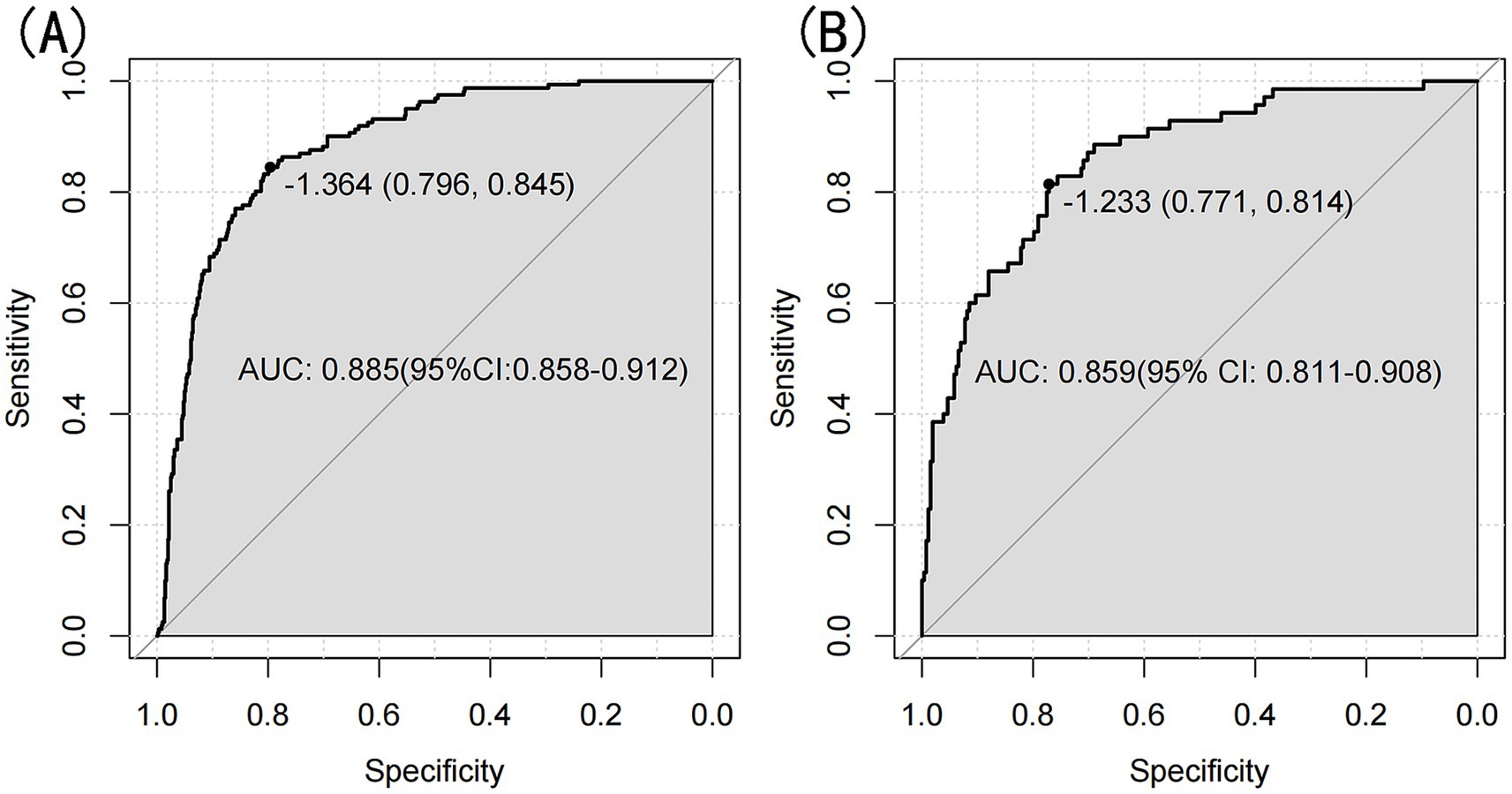
Figure 3. (A) Nomogram ROC curves generated from the training set. (B) Nomogram ROC curves generated using the validation set.
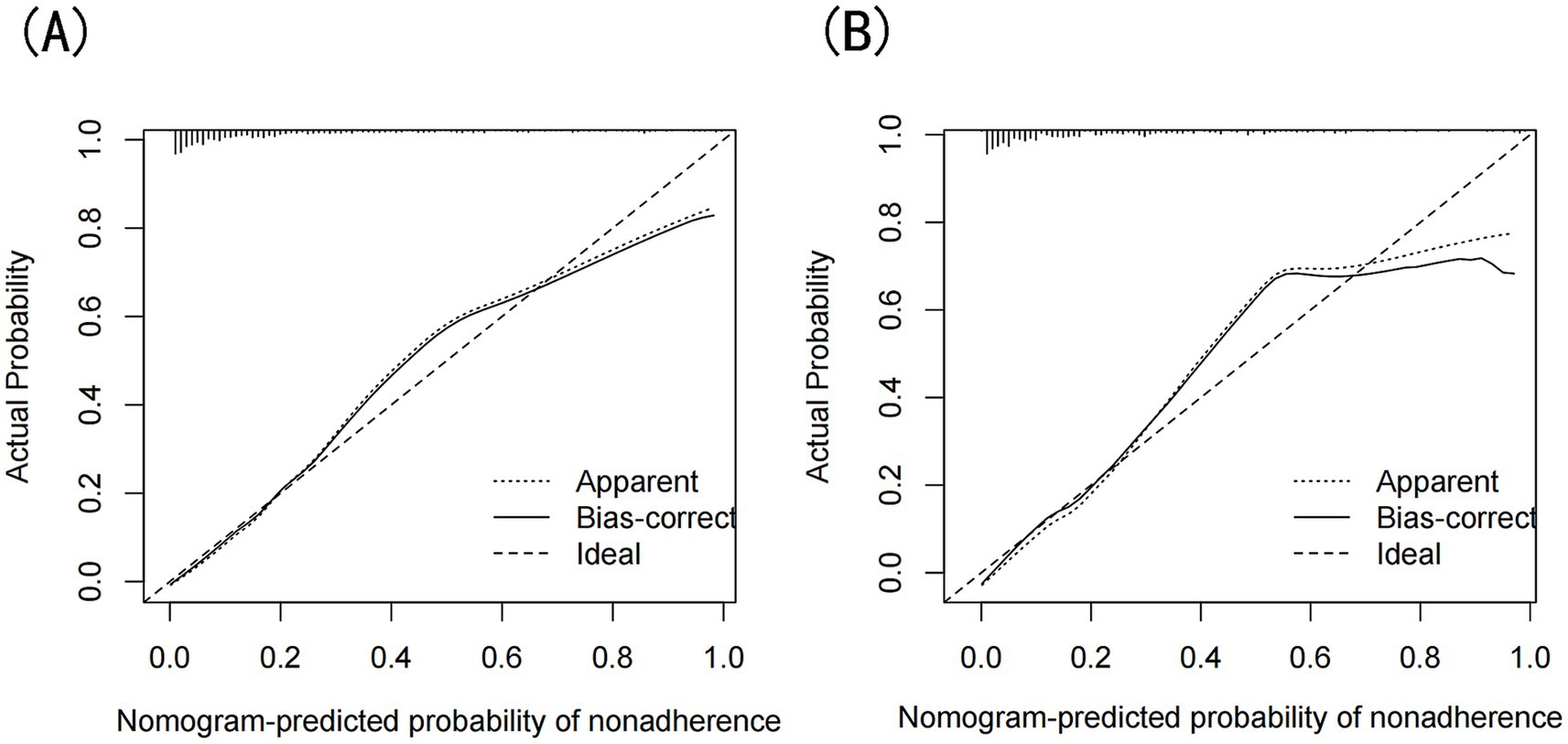
Figure 4. (A) Calibration curves of the nomogram prediction for the training set, (B) Calibration curves of the nomogram prediction for the validation set.
The nomogram is presented in Figure 6. Each predictor was assigned a score corresponding to its contribution to sarcopenia risk, and the total score was used to estimate the probability of sarcopenia.
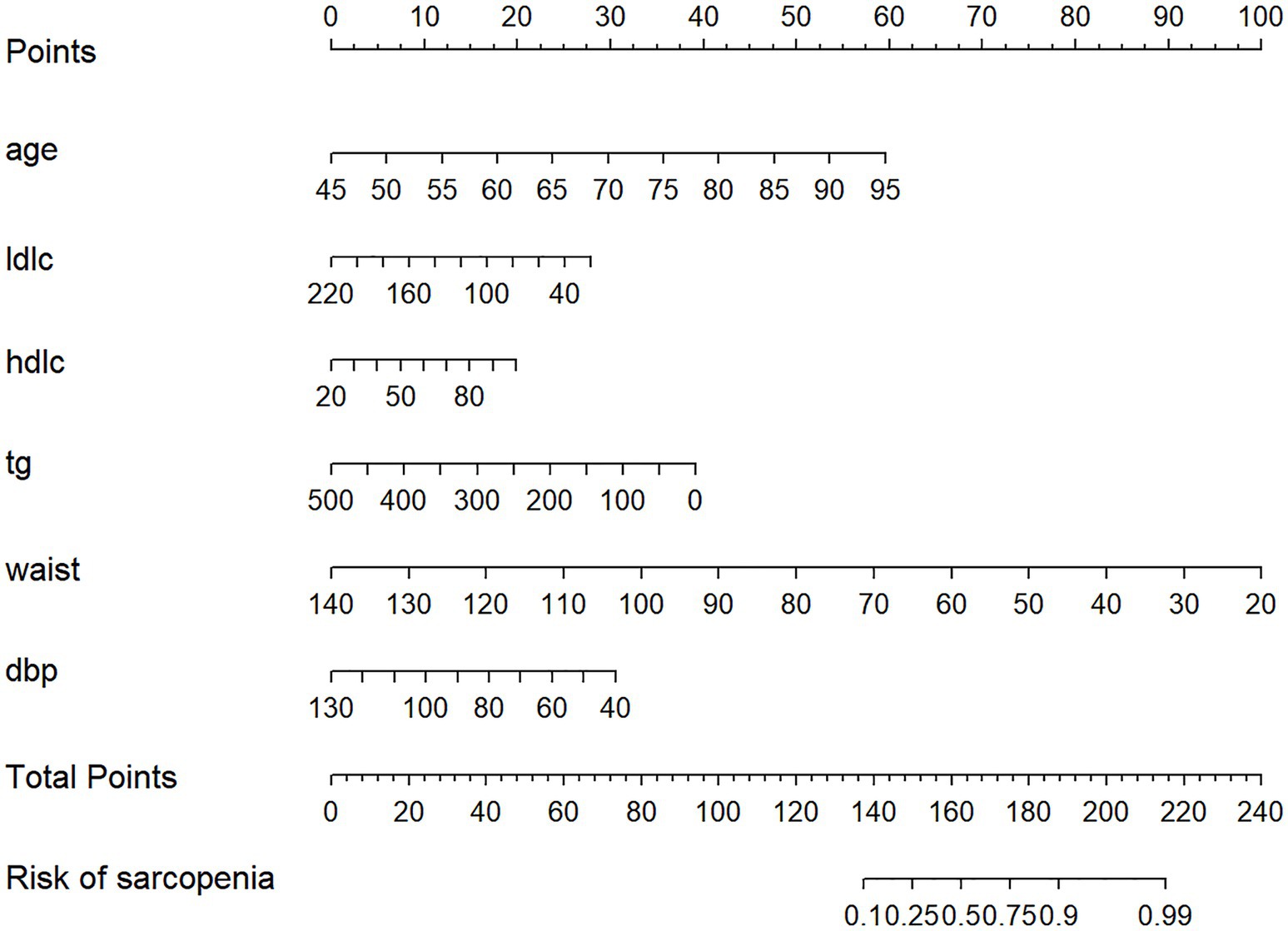
Figure 6. Nomogram to evaluate the risk of sarcopenia in patients with diabetes. LDLC mean low-density lipoprotein cholesterol. HDLC mean high-density lipoprotein cholesterol.TG mean triglycerides.
To further assess the predictive performance and significance of the predictors used in developing the nomogram, we constructed four machine learning models. As shown in Figures 7A,B, these models exhibited strong discriminative ability and predictive value in both the training and validation sets. The Random Forest (RF) model achieved the highest AUC of 1.000 in the training set, but performed moderately in the validation set (AUC = 0.917, 95% CI: 0.884–0.949). In contrast, the Gradient Boosting Machine (GBM) model demonstrated the best predictive performance in the validation set (AUC = 0.923, 95% CI: 0.893–0.953), while ranking second in the training set (AUC = 0.966, 95% CI: 0.952–0.981), just behind the RF model.
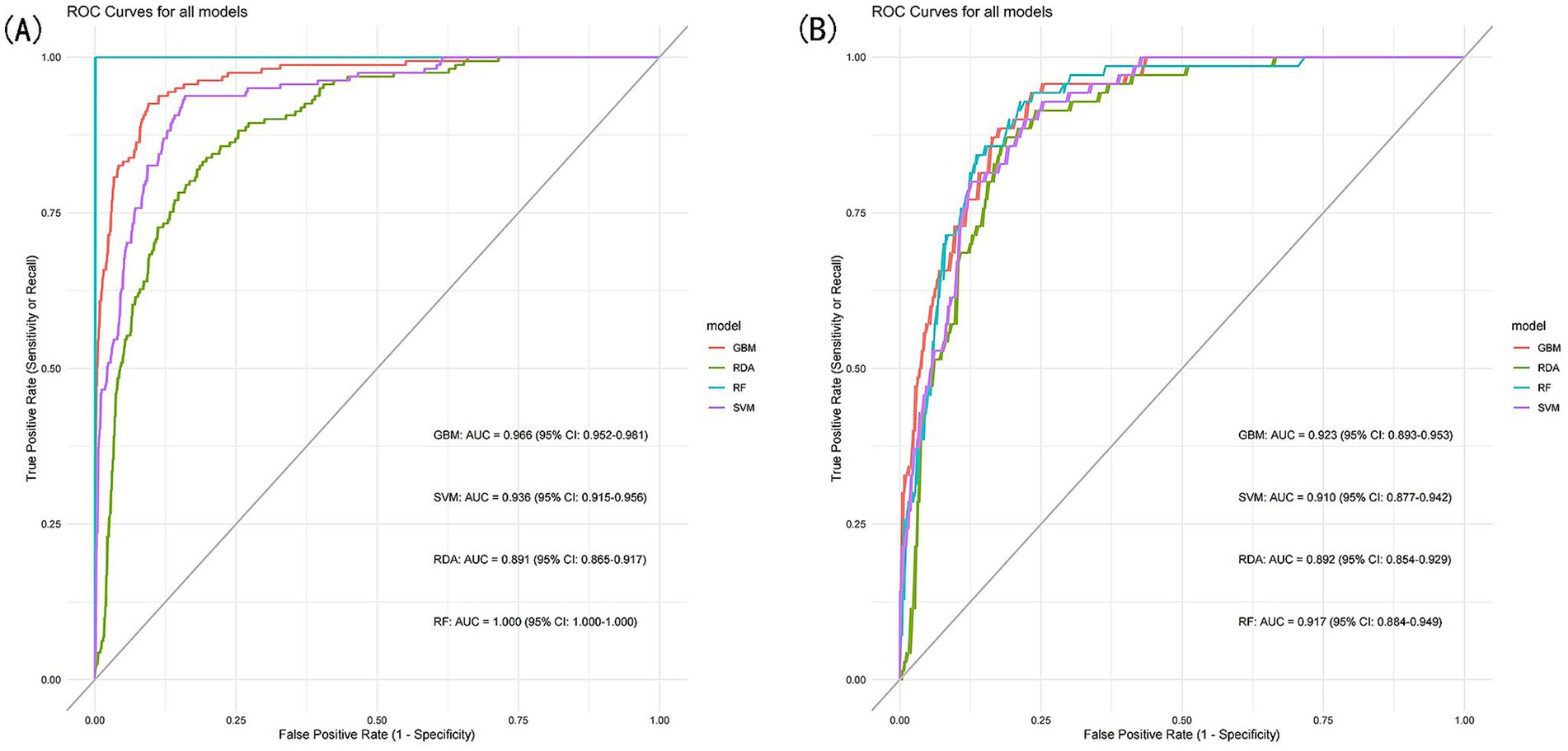
Figure 7. (A) ROC curves of the machine learning models for the training set, (B) ROC curves of the machine learning models for the validation set.
We ultimately selected the Gradient Boosting Machine (GBM) model for hyperparameter optimization using Bayesian optimization. This method is efficient for optimizing hyperparameters by constructing a probabilistic model of the objective function and using it to identify the most promising hyperparameters for evaluation. The optimization process identified the following optimal parameters: 56 trees, an interaction depth of 2, a shrinkage rate of 0.0777, and a minimum number of observations in a node of 30. After optimization, the model achieved an AUC of 0.933 (95% CI: 0.9132–0.9527) for the training set and 0.932 (95% CI: 0.9045–0.9595) for the validation set (Figure 8). Variable importance analysis revealed that waist circumference was the most significant predictor, followed by age, DBP, TG, LDL-C, and HDL-C. Specifically, waist circumference had a relative importance of 59.88%, age 26.92%, DBP 4.22%, TG 3.89%, LDL-C 3.77%, and HDL-C 1.32% (Figure 9A). Additionally, SHAP (Shapley Additive exPlanations) values were used to interpret the model’s predictions. Age and waist circumference were the primary drivers of the model’s output, with other variables contributing to a lesser extent, making smaller adjustments to the final prediction (Figure 9B).
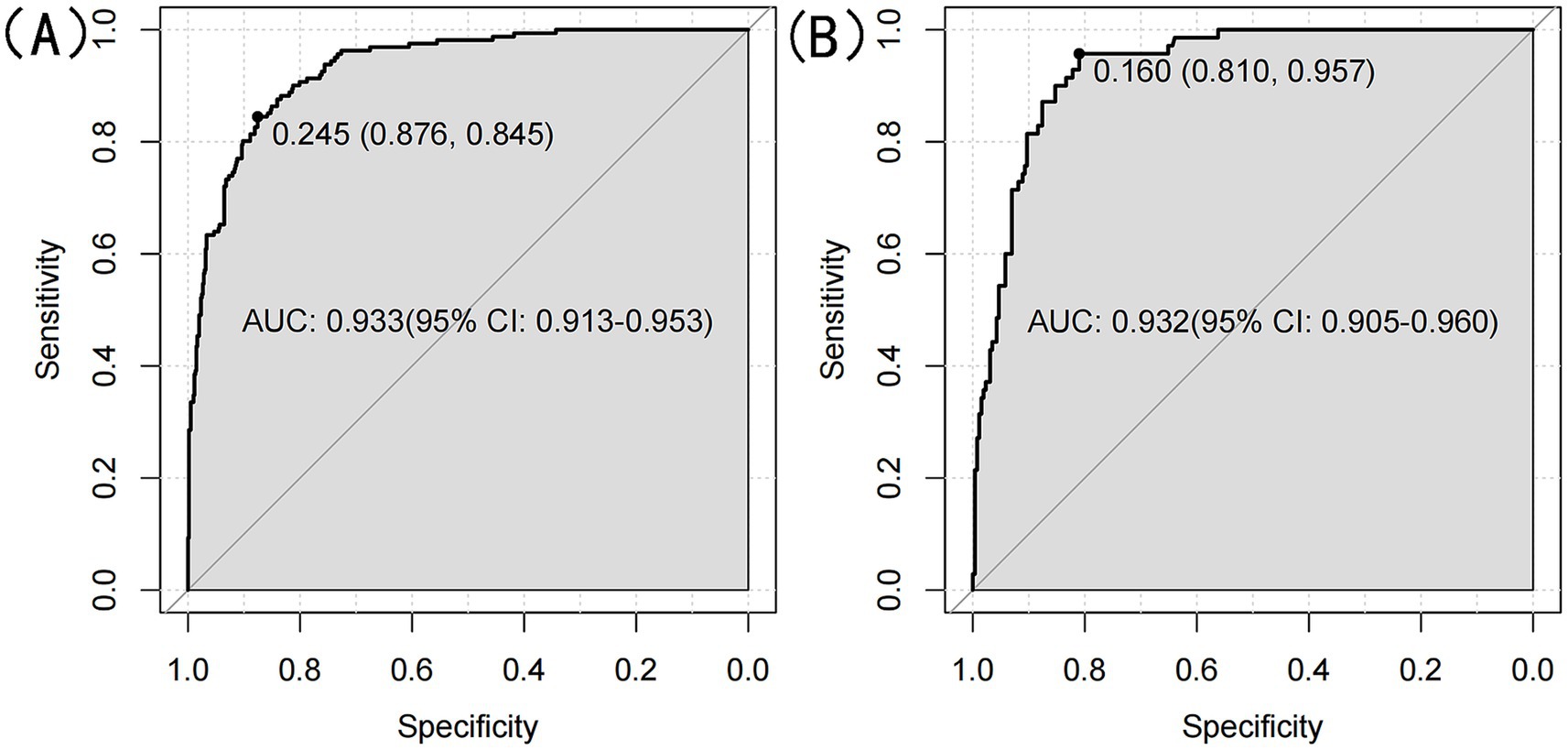
Figure 8. (A) ROC curve for optimized GBM model for the training set. (B) ROC curve for optimized GBM model for the validation set.
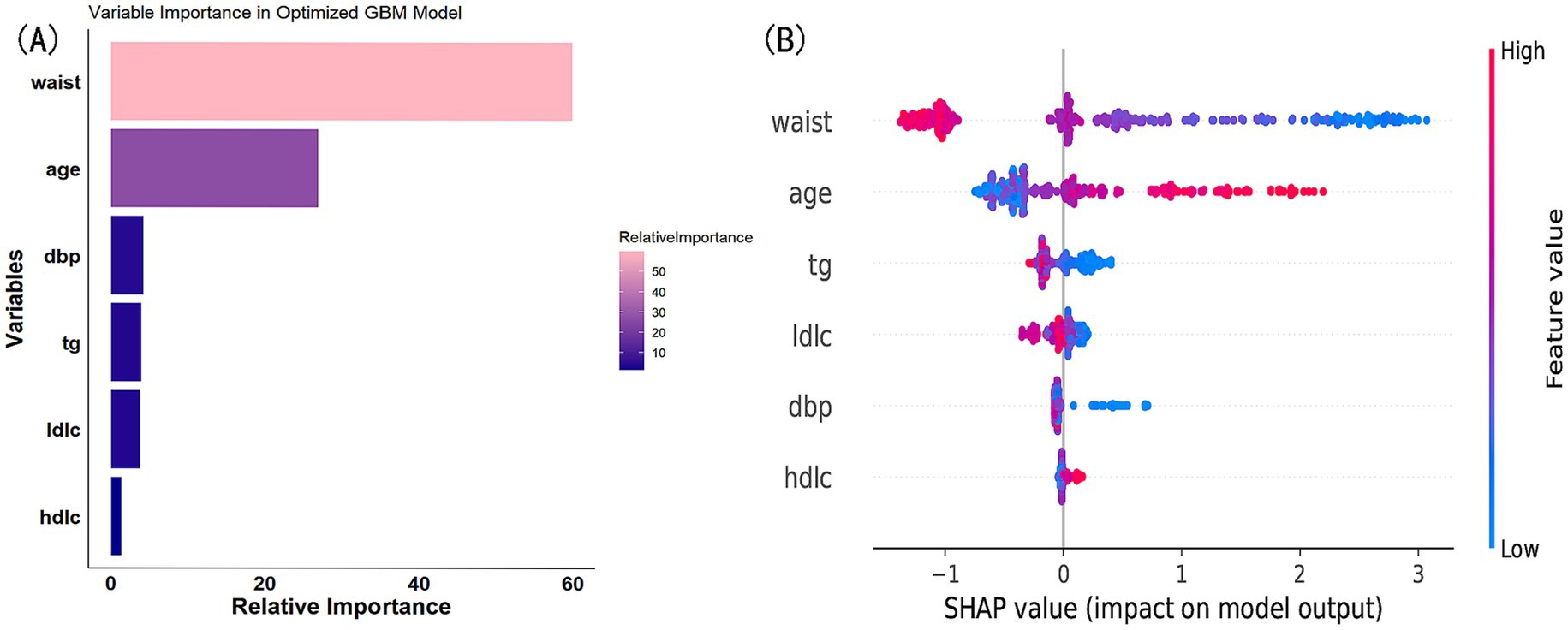
Figure 9. (A) Variable importance in optimized GBM model. (B) SHAP value contribution in optimized GBM model.
PCA reduced the original 52 features into 10 principal components, explaining a total of 75.8% of the variance. The first two principal components explained 26.5% of the variance. PC1 was most strongly influenced by eGFR, cystatin C, uric acid, age, and creatinine, whereas PC2 was primarily influenced by waist circumference, BMI, BRI, triglycerides, and immediate recall. The PCA biplot (Figure 10) illustrates the contribution of these features. K-means clustering identified four distinct patient subgroups. Cluster 1 had the highest prevalence of sarcopenia (53.4%), while Clusters 2 and 3 had lower prevalence (10.8 and 8.8%, respectively). Among the characteristics available at presentation, the clusters differed significantly across 82.4% (42/51) of all the admission variables recorded (Supplementary Table S3; Supplementary Figure S1).
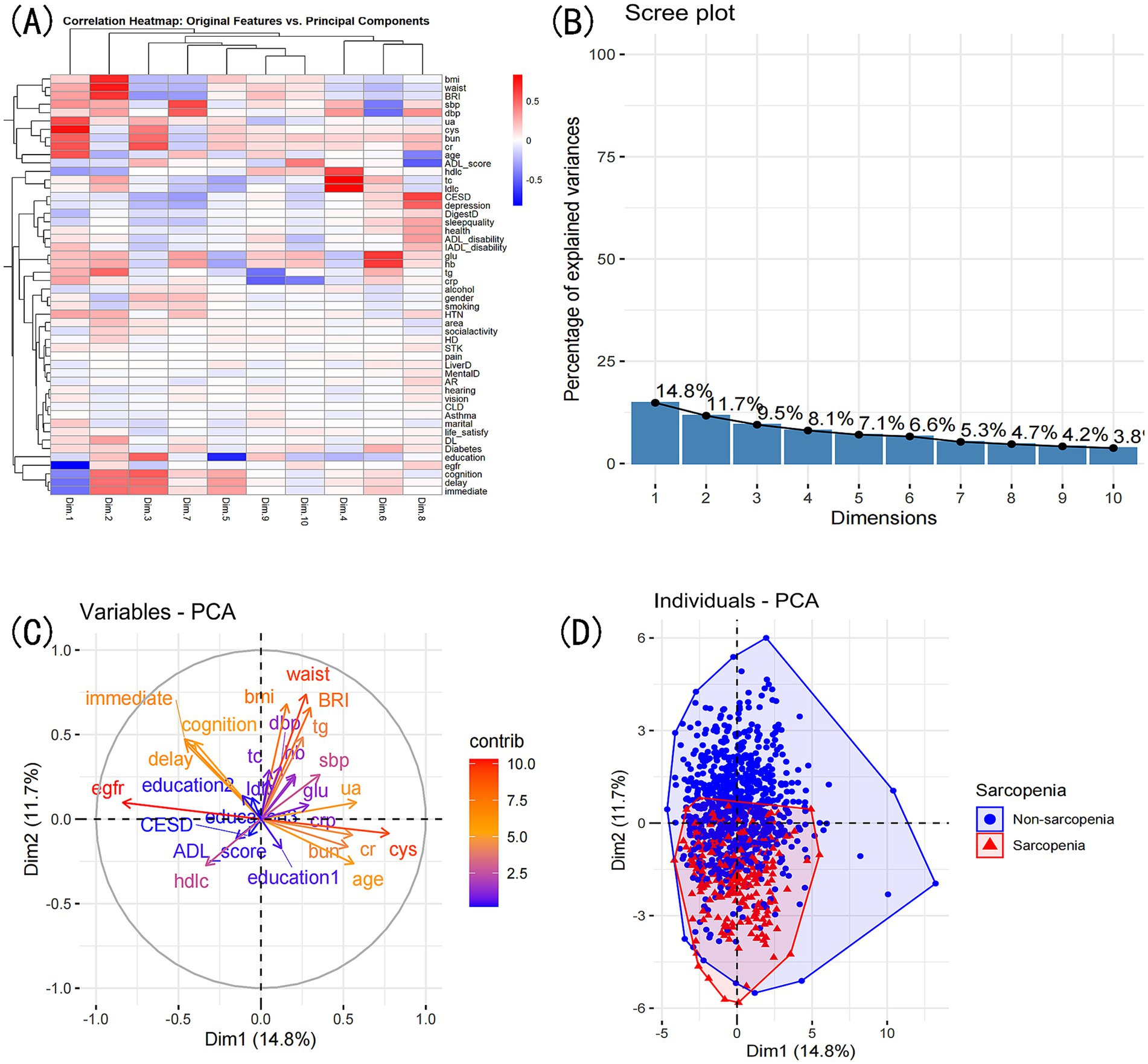
Figure 10. PCA comprehensive analysis. Exploring variable contributions and patient clustering: (A) Correlation heatmap of original variables and principal components. (B) Top contributing variables for each principal component. (C) Grouping of original variables based on positive or negative contributions to PCs. (D) Patient grouping based on PCA’s two principal dimensions: scatter plot.
CKD leads to muscle atrophy through a complex interaction of factors, primarily characterized by a disruption in the balance between protein synthesis and breakdown, along with impaired muscle regeneration (26, 27). A key driver of muscle wasting in CKD is the dysregulation of critical molecular pathways involved in muscle metabolism. The insulin-like growth factor-1 (IGF-1) pathway, which is essential for promoting muscle protein synthesis and cell proliferation, is often disrupted in CKD patients (28). This disruption creates an imbalance between the IGF-1 and myostatin pathways, with myostatin inhibiting muscle growth and promoting protein catabolism (29, 30). Consequently, accelerated catabolic processes contribute to increased muscle atrophy in these individuals (31). Inflammation plays a key role in muscle atrophy in CKD, with increased levels of pro-inflammatory cytokines like tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) driving muscle degradation (32, 33). These cytokines activate the ubiquitin-proteasome system (UPS) and the autophagy-lysosome pathway (ALP), both of which lead to increased proteolysis (34–36). Additionally, oxidative stress from excessive reactive oxygen species (ROS) exacerbates mitochondrial dysfunction, impairing energy production essential for muscle maintenance and repair (37). Other contributing factors include insulin resistance, vitamin D deficiency, and altered levels of angiotensin II, all of which further exacerbate the muscle-wasting process (38, 39). Insulin resistance, common in CKD, diminishes the anabolic effects of insulin on muscle tissue, while vitamin D deficiency is linked to muscle weakness and atrophy (40–44). Moreover, elevated levels of angiotensin II may promote muscle degradation through its effects on inflammation and fibrosis (45, 46).
This study focused on creating and validating a sarcopenia risk prediction model specifically for CKD patients, utilizing data from the 2015 CHARLS cohort. The final model determined that waist circumference, age, LDL-C, TG, HDL-C, and DBP are significant predictors of sarcopenia in CKD patients.
Age emerged as a significant predictor, aligning with the established link between aging and sarcopenia. As individuals age, they experience a progressive decline in muscle mass and strength, driven by several interconnected factors. Hormonal changes, particularly reductions in testosterone, estrogen, and growth hormone, play a crucial role in muscle metabolism and regeneration, leading to decreased muscle protein synthesis and increased muscle breakdown (47, 48). Additionally, the aging process often results in reduced physical activity levels, which further contributes to muscle atrophy and loss of strength. Furthermore, aging is associated with increased oxidative stress and inflammation, both of which can negatively impact muscle health. Elevated levels of reactive oxygen species (ROS) can damage muscle fibers and impair their function (49, 50).
Our findings indicate that in CKD patients, waist circumference, LDL-C, and TC are inversely related to sarcopenia risk, whereas HDL-C is positively associated with an increased risk, whereas high-density lipoprotein cholesterol is positively associated with an increased risk. This finding contrasts with much of the literature, which generally considers abdominal fat a risk factor for muscle loss (51, 52). Many studies have highlighted that excessive body fat, particularly visceral fat, is closely linked to declines in muscle mass, primarily due to the negative effects of inflammatory cytokines secreted by adipose tissue on muscle metabolism (53, 54). However, our results may reflect a protective role of moderate waist circumference and associated fat storage in maintaining muscle health among CKD patients. This hypothesis is further supported by findings from other studies, which showed that patients without sarcopenia had higher waist circumference, LDL-C, and TG levels compared to those with sarcopenia (55). Additionally, in a study constructing a predictive model for sarcopenia, low BMI was identified as a significant risk factor, consistent with findings from a Taiwanese population-based study (16, 56).
Our findings suggest that higher diastolic blood pressure may act as a protective factor against sarcopenia, potentially due to its impact on skeletal muscle perfusion. Chronic low blood pressure can impair blood flow to skeletal muscles, reducing muscle quality and contributing to the progression of sarcopenia. Maintaining adequate blood flow is crucial for delivering oxygen, hormones, and nutrients necessary for muscle maintenance and repair (57). Previous studies have indicated that insufficient perfusion can compromise muscle health, leading to increased muscle loss. Additionally, inadequate capillarization of skeletal muscle restricts the diffusion of substrates, oxygen, hormones, and nutrients, further exacerbating the risk of sarcopenia and the decline in physical function among older adults (58, 59). Consequently, higher diastolic blood pressure levels may reflect better vascular health and more efficient nutrient delivery, ultimately contributing to the preservation of muscle mass and function (60).
A notable strength of this study is the creation of the first nomogram specifically designed to assess sarcopenia risk across the CKD spectrum, leveraging data from the CHARLS. Our model incorporates six commonly available demographic, clinical, functional measures, along with key blood biomarkers such as LDL-C, HDL-C, and triglycerides, enabling simplified risk assessment in community and primary care settings. This broad applicability is particularly valuable given the scarcity of tools for non-dialysis CKD populations. Furthermore, the model’s robust predictive performance was achieved through advanced machine learning methodologies and comprehensive hyperparameter optimization, demonstrating parity with hemodialysis-specific tools despite its broader scope (7, 8).
The clinical applicability of our model is underscored by its capacity to identify CKD patients at elevated risk of sarcopenia through readily obtainable parameters, such as waist circumference, lipid profiles, and blood pressure. Considering the substantial prevalence of sarcopenia among CKD patients and its correlation with adverse clinical outcomes, early detection is imperative for timely and effective intervention. This predictive tool allows clinicians to categorize patients based on their risk and apply targeted strategies, such as personalized nutritional interventions, to improve body composition and support evidence-based dietary practices.
Nevertheless, this study has its limitations. Firstly, the cross-sectional design of the analysis limits our ability to infer causal relationships between the identified predictors and the onset of sarcopenia. Longitudinal research is required to clarify the temporal sequence and mechanistic pathways driving these associations. Second, the relatively modest sample size, while sufficient for model development, may limit the statistical power to detect weaker associations and could potentially influence the stability of the findings. Expanding datasets and conducting collaborative research across multiple cohorts would strengthen the robustness and reliability of the findings. Additionally, the study population was derived exclusively from the CHARLS, which may introduce selection bias and limit the applicability of the findings to other demographic and ethnic groups. External validation in independent CKD cohorts from diverse settings is critical to ensure the model’s generalizability and broader clinical utility. Furthermore, while the model utilizes easily accessible variables, the absence of more sophisticated biomarkers, such as serum myostatin or inflammatory markers, may restrict its ability to capture the complex biological processes driving sarcopenia.
Future studies should aim to increase the sample size and include longitudinal data to enhance causal inference. Integrating advanced omics data and imaging modalities could further refine the predictive accuracy of the model and deepen our understanding of sarcopenia’s pathophysiology. Additionally, while our percentile-based definition of low muscle mass aligns with precedents in aging research and mirrors AWGS DXA thresholds, future studies should harmonize sarcopenia criteria across methodologies to facilitate cross-population comparisons. Finally, clinical implementation research is needed to evaluate the nomogram’s effectiveness in improving patient outcomes, helping to bridge the gap between predictive modeling and real-world healthcare interventions.
We developed a novel nomogram and machine learning framework for sarcopenia prediction in CKD. The Gradient Boosting Machine model demonstrated optimal performance, providing a validated tool for early risk stratification. This framework offers potential clinical utility in identifying high-risk patients and guiding personalized interventions. Future studies could explore its implementation in clinical practice and further validate its effectiveness in diverse patient populations.
Publicly available datasets were analyzed in this study. This data can be found at: http://charls.pku.edu.cn.
The studies involving humans were approved by Peking University Ethical Review Committee (IRB00001052-11015). The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
RL: Writing – original draft, Writing – review & editing. SW: Writing – review & editing, Data curation, Formal Analysis. PC: Data curation, Formal analysis, Writing – review & editing. FfL: Data curation, Formal analysis, Methodology, Writing – review & editing. PL: Data curation, Formal analysis, Writing – review & editing. QC: Data curation, Formal analysis, Methodology, Writing – review & editing. XL: Data curation, Investigation, Writing – review & editing. FyL: Data curation, Investigation, Writing – review & editing. SG: Data curation, Methodology, Writing – review & editing. JZ: Data curation, Methodology, Writing – review & editing. DL: Formal analysis, Funding acquisition, Methodology, Writing – review & editing. ZH: Formal analysis, Funding acquisition, Methodology, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by several funding sources, including the Shanghai Traditional Chinese Medicine (TCM) Inheritance and Innovation Development Three-Year Action Plan [ZY(2021–2023)-0201–01], the Pudong New Area Health System’s Pudong Distinguished TCM Doctor Training Program (PWRzm2020-15), and the TCM Rehabilitation Service Capacity Enhancement Project (Medical Affairs 032). Additional support was provided by the Longhua Hospital Innovation Project: Clinical Study on Acupotomy Combined with “Knee Joint Balance Exercise” for Knee Osteoarthritis (CX202045), the Pudong New Area Health Commission’s Special Disease Project (PWZzb2022-16), and the Advanced TCM Mentorship Talent Training Program (PDZY-2023-0806).
The authors would like to thank the CHARLS research team for providing the data used in this study. We also acknowledge the participants of the CHARLS study for their contributions, which made this research possible.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1546988/full#supplementary-material
1. Cruz-Jentoft, AJ, Bahat, G, Bauer, J, Boirie, Y, Bruyere, O, Cederholm, T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. (2019) 48:16–31. doi: 10.1093/ageing/afy169
2. Ammirati, AL. Chronic kidney disease. Rev Assoc Med Bras (1992). (2020) 66:s3–09. doi: 10.1590/1806-9282.66.S1.3
3. Fahal, IH. Uraemic sarcopenia: aetiology and implications. Nephrol Dial Transplant. (2014) 29:1655–65. doi: 10.1093/ndt/gft070
4. Wang, K, Liu, Q, Tang, M, Qi, G, Qiu, C, Huang, Y, et al. Chronic kidney disease-induced muscle atrophy: molecular mechanisms and promising therapies. Biochem Pharmacol. (2023) 208:115407. doi: 10.1016/j.bcp.2022.115407
5. Duarte, MP, Almeida, LS, Neri, S, Oliveira, JS, Wilkinson, TJ, Ribeiro, HS, et al. Prevalence of sarcopenia in patients with chronic kidney disease: a global systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. (2024) 15:501–12. doi: 10.1002/jcsm.13425
6. Ribeiro, HS, Neri, S, Oliveira, JS, Bennett, PN, Viana, JL, and Lima, RM. Association between sarcopenia and clinical outcomes in chronic kidney disease patients: a systematic review and meta-analysis. Clin Nutr. (2022) 41:1131–40. doi: 10.1016/j.clnu.2022.03.025
7. Cai, G, Ying, J, Pan, M, Lang, X, Yu, W, and Zhang, Q. Development of a risk prediction nomogram for sarcopenia in hemodialysis patients. BMC Nephrol. (2022) 23:319. doi: 10.1186/s12882-022-02942-0
8. Xie, D, Zhu, Q, Lu, J, Hu, C, Niu, J, Yu, C, et al. Development and validation of a diagnostic nomogram for sarcopenia in chinese hemodialysis patients. Nephrol Dial Transplant. (2023) 38:1017–26. doi: 10.1093/ndt/gfac260
9. Nakano, Y, Mandai, S, Naito, S, Fujiki, T, Mori, Y, Ando, F, et al. Effect of osteosarcopenia on longitudinal mortality risk and chronic kidney disease progression in older adults. Bone. (2024) 179:116975. doi: 10.1016/j.bone.2023.116975
10. Zhao, Y, Hu, Y, Smith, JP, Strauss, J, and Yang, G. Cohort profile: the China health and retirement longitudinal study (charls). Int J Epidemiol. (2014) 43:61–8. doi: 10.1093/ije/dys203
11. Zhu, X, Zhang, X, Ding, L, Tang, Y, Xu, A, Yang, F, et al. Associations of pain and sarcopenia with successful aging among older people in China: evidence from charls. J Nutr Health Aging. (2023) 27:196–201. doi: 10.1007/s12603-023-1892-2
12. Kdigo 2024 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. (2024) 105:S117–314. doi: 10.1016/j.kint.2023.10.018
13. Chen, LK, Woo, J, Assantachai, P, Auyeung, TW, Chou, MY, Iijima, K, et al. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. (2020) 21:300–307.e2. doi: 10.1016/j.jamda.2019.12.012
14. Wen, X, Wang, M, Jiang, CM, and Zhang, YM. Anthropometric equation for estimation of appendicular skeletal muscle mass in chinese adults. Asia Pac J Clin Nutr. (2011) 20:551–6.
15. Newman, AB, Kupelian, V, Visser, M, Simonsick, E, Goodpaster, B, Nevitt, M, et al. Sarcopenia: alternative definitions and associations with lower extremity function. J Am Geriatr Soc. (2003) 51:1602–9. doi: 10.1046/j.1532-5415.2003.51534.x
16. Li, Q, Cheng, H, Cen, W, Yang, T, and Tao, S. Development and validation of a predictive model for the risk of sarcopenia in the older adults in China. Eur J Med Res. (2024) 29:278. doi: 10.1186/s40001-024-01873-w
17. Katz, S, Ford, AB, Moskowitz, RW, Jackson, BA, and Jaffe, MW. Studies of illness in the aged. The index of adl: a standardized measure of biological and psychosocial function. JAMA. (1963) 185:914–9. doi: 10.1001/jama.1963.03060120024016
18. Lawton, MP, and Brody, EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. (1969) 9:179–86. doi: 10.1093/geront/9.3_Part_1.179
19. Zhang, Y, Xiong, Y, Yu, Q, Shen, S, Chen, L, and Lei, X. The activity of daily living (adl) subgroups and health impairment among chinese elderly: a latent profile analysis. BMC Geriatr. (2021) 21:30. doi: 10.1186/s12877-020-01986-x
20. Andresen, EM, Malmgren, JA, Carter, WB, and Patrick, DL. Screening for depression in well older adults: evaluation of a short form of the ces-d (center for epidemiologic studies depression scale). Am J Prev Med. (1994) 10:77–84. doi: 10.1016/S0749-3797(18)30622-6
21. Zhou, L, Ma, X, and Wang, W. Relationship between cognitive performance and depressive symptoms in chinese older adults: the China health and retirement longitudinal study (charls). J Affect Disord. (2021) 281:454–8. doi: 10.1016/j.jad.2020.12.059
22. Inker, LA, Schmid, CH, Tighiouart, H, Eckfeldt, JH, Feldman, HI, Greene, T, et al. Estimating glomerular filtration rate from serum creatinine and cystatin c. N Engl J Med. (2012) 367:20–9. doi: 10.1056/NEJMoa1114248
23. Wu, WT, Li, YJ, Feng, AZ, Li, L, Huang, T, Xu, AD, et al. Data mining in clinical big data: the frequently used databases, steps, and methodological models. Mil Med Res. (2021) 8:44. doi: 10.1186/s40779-021-00338-z
25. Li, L. Dimension reduction for high-dimensional data. Methods Mol Biol. (2010) 620:417–34. doi: 10.1007/978-1-60761-580-4_14
26. Sun, H, Sun, J, Li, M, Qian, L, Zhang, L, Huang, Z, et al. Transcriptome analysis of immune receptor activation and energy metabolism reduction as the underlying mechanisms in interleukin-6-induced skeletal muscle atrophy. Front Immunol. (2021) 12:730070. doi: 10.3389/fimmu.2021.730070
27. Wang, XH, Mitch, WE, and Price, SR. Pathophysiological mechanisms leading to muscle loss in chronic kidney disease. Nat Rev Nephrol. (2022) 18:138–52. doi: 10.1038/s41581-021-00498-0
28. Widajanti, N, Soelistijo, S, Hadi, U, Thaha, M, Aditiawardana, W, et al. Association between sarcopenia and insulin-like growth factor-1, myostatin, and insulin resistance in elderly patients undergoing hemodialysis. J Aging Res. (2022) 2022:1327332. doi: 10.1155/2022/1327332
29. Cohen, S, Nathan, JA, and Goldberg, AL. Muscle wasting in disease: molecular mechanisms and promising therapies. Nat Rev Drug Discov. (2015) 14:58–74. doi: 10.1038/nrd4467
30. Han, DS, Chen, YM, Lin, SY, Chang, HH, Huang, TM, Chi, YC, et al. Serum myostatin levels and grip strength in normal subjects and patients on maintenance haemodialysis. Clin Endocrinol. (2011) 75:857–63. doi: 10.1111/j.1365-2265.2011.04120.x
31. Bataille, S, Chauveau, P, Fouque, D, Aparicio, M, and Koppe, L. Myostatin and muscle atrophy during chronic kidney disease. Nephrol Dial Transplant. (2021) 36:1986–93. doi: 10.1093/ndt/gfaa129
32. Fang, WY, Tseng, YT, Lee, TY, Fu, YC, Chang, WH, Lo, WW, et al. Triptolide prevents lps-induced skeletal muscle atrophy via inhibiting nf-kappab/tnf-alpha and regulating protein synthesis/degradation pathway. Br J Pharmacol. (2021) 178:2998–3016. doi: 10.1111/bph.15472
33. Ma, W, Zhang, R, Huang, Z, Zhang, Q, Xie, X, Yang, X, et al. Pqq ameliorates skeletal muscle atrophy, mitophagy and fiber type transition induced by denervation via inhibition of the inflammatory signaling pathways. Ann Transl Med. (2019) 7:440. doi: 10.21037/atm.2019.08.101
34. Mitch, WE, and Goldberg, AL. Mechanisms of muscle wasting. The role of the ubiquitin-proteasome pathway. N Engl J Med. (1996) 335:1897–905. doi: 10.1056/NEJM199612193352507
35. Sandri, M. Protein breakdown in muscle wasting: role of autophagy-lysosome and ubiquitin-proteasome. Int J Biochem Cell Biol. (2013) 45:2121–9. doi: 10.1016/j.biocel.2013.04.023
36. Wang, Y, and Le, WD. Autophagy and ubiquitin-proteasome system. Adv Exp Med Biol. (2019) 1206:527–50. doi: 10.1007/978-981-15-0602-4_25
37. Xu, T, Yang, X, Wu, C, Qiu, J, Fang, Q, Wang, L, et al. Pyrroloquinoline quinone attenuates cachexia-induced muscle atrophy via suppression of reactive oxygen species. J Thorac Dis. (2018) 10:2752–9. doi: 10.21037/jtd.2018.04.112
38. Ho, HJ, and Shirakawa, H. Oxidative stress and mitochondrial dysfunction in chronic kidney disease. Cells. (2022) 12:88. doi: 10.3390/cells12010088
39. Molina, P, Carrero, JJ, Bover, J, Chauveau, P, Mazzaferro, S, and Torres, PU. Vitamin d, a modulator of musculoskeletal health in chronic kidney disease. J Cachexia Sarcopenia Muscle. (2017) 8:686–701. doi: 10.1002/jcsm.12218
40. Abdulla, H, Smith, K, Atherton, PJ, and Idris, I. Role of insulin in the regulation of human skeletal muscle protein synthesis and breakdown: a systematic review and meta-analysis. Diabetologia. (2016) 59:44–55. doi: 10.1007/s00125-015-3751-0
41. Garcia, LA, King, KK, Ferrini, MG, Norris, KC, and Artaza, JN. 1,25(oh)2vitamin d3 stimulates myogenic differentiation by inhibiting cell proliferation and modulating the expression of promyogenic growth factors and myostatin in c2c12 skeletal muscle cells. Endocrinology. (2011) 152:2976–86. doi: 10.1210/en.2011-0159
42. Girgis, CM, Clifton-Bligh, RJ, Mokbel, N, Cheng, K, and Gunton, JE. Vitamin d signaling regulates proliferation, differentiation, and myotube size in c2c12 skeletal muscle cells. Endocrinology. (2014) 155:347–57. doi: 10.1210/en.2013-1205
43. Spoto, B, Pisano, A, and Zoccali, C. Insulin resistance in chronic kidney disease: a systematic review. Am J Physiol Ren Physiol. (2016) 311:F1087–108. doi: 10.1152/ajprenal.00340.2016
44. Watson, EL, Wilkinson, TJ, O'Sullivan, TF, Baker, LA, Gould, DW, Xenophontos, S, et al. Association between vitamin d deficiency and exercise capacity in patients with ckd, a cross-sectional analysis. J Steroid Biochem Mol Biol. (2021) 210:105861. doi: 10.1016/j.jsbmb.2021.105861
45. Cohn, RD, van Erp, C, Habashi, JP, Soleimani, AA, Klein, EC, Lisi, MT, et al. Angiotensin ii type 1 receptor blockade attenuates tgf-beta-induced failure of muscle regeneration in multiple myopathic states. Nat Med. (2007) 13:204–10. doi: 10.1038/nm1536
46. Song, YH, Li, Y, Du, J, Mitch, WE, Rosenthal, N, and Delafontaine, P. Muscle-specific expression of igf-1 blocks angiotensin ii-induced skeletal muscle wasting. J Clin Invest. (2005) 115:451–8. doi: 10.1172/JCI22324
47. Buratto, J, Kirk, B, Phu, S, Vogrin, S, and Duque, G. Safety and efficacy of testosterone therapy on musculoskeletal health and clinical outcomes in men: a systematic review and meta-analysis of randomized placebo-controlled trials. Endocr Pract. (2023) 29:727–34. doi: 10.1016/j.eprac.2023.04.013
48. Critchlow, AJ, Hiam, D, Williams, R, Scott, D, and Lamon, S. The role of estrogen in female skeletal muscle aging: a systematic review. Maturitas. (2023) 178:107844. doi: 10.1016/j.maturitas.2023.107844
49. Mellen, RH, Girotto, OS, Marques, EB, Laurindo, LF, Grippa, PC, Mendes, CG, et al. Insights into pathogenesis, nutritional and drug approach in sarcopenia: a systematic review. Biomedicine. (2023) 11:136. doi: 10.3390/biomedicines11010136
50. Wilkinson, DJ, Piasecki, M, and Atherton, PJ. The age-related loss of skeletal muscle mass and function: measurement and physiology of muscle fibre atrophy and muscle fibre loss in humans. Ageing Res Rev. (2018) 47:123–32. doi: 10.1016/j.arr.2018.07.005
51. Park, SH, Park, JH, Park, HY, Jang, HJ, Kim, HK, Park, J, et al. Additional role of sarcopenia to waist circumference in predicting the odds of metabolic syndrome. Clin Nutr. (2014) 33:668–72. doi: 10.1016/j.clnu.2013.08.008
52. Zhou, H, Su, H, Gong, Y, Chen, L, Xu, L, Chen, G, et al. The association between weight-adjusted-waist index and sarcopenia in adults: a population-based study. Sci Rep. (2024) 14:10943. doi: 10.1038/s41598-024-61928-0
53. Kim, JY, Choi, J, Vella, CA, Criqui, MH, Allison, MA, and Kim, NH. Associations between weight-adjusted waist index and abdominal fat and muscle mass: multi-ethnic study of atherosclerosis. Diabetes Metab J. (2022) 46:747–55. doi: 10.4093/dmj.2021.0294
54. Kim, NH, Park, Y, Kim, NH, and Kim, SG. Weight-adjusted waist index reflects fat and muscle mass in the opposite direction in older adults. Age Ageing. (2021) 50:780–6. doi: 10.1093/ageing/afaa208
55. Zou, M, and Shao, Z. Construction and evaluation of sarcopenia risk prediction model for patients with diabetes: a study based on the China health and retirement longitudinal study (charls). Diabetol Metab Syndr. (2024) 16:230. doi: 10.1186/s13098-024-01467-w
56. Wu, LC, Kao, HH, Chen, HJ, and Huang, PF. Preliminary screening for sarcopenia and related risk factors among the elderly. Medicine (Baltimore). (2021) 100:e25946. doi: 10.1097/MD.0000000000025946
57. Phillips, BE, Atherton, PJ, Varadhan, K, Limb, MC, Wilkinson, DJ, Sjoberg, KA, et al. The effects of resistance exercise training on macro- and micro-circulatory responses to feeding and skeletal muscle protein anabolism in older men. J Physiol. (2015) 593:2721–34. doi: 10.1113/JP270343
58. Prior, SJ, Ryan, AS, Blumenthal, JB, Watson, JM, Katzel, LI, and Goldberg, AP. Sarcopenia is associated with lower skeletal muscle capillarization and exercise capacity in older adults. J Gerontol A Biol Sci Med Sci. (2016) 71:1096–101. doi: 10.1093/gerona/glw017
59. Timmerman, KL, Lee, JL, Fujita, S, Dhanani, S, Dreyer, HC, Fry, CS, et al. Pharmacological vasodilation improves insulin-stimulated muscle protein anabolism but not glucose utilization in older adults. Diabetes. (2010) 59:2764–71. doi: 10.2337/db10-0415
Keywords: sarcopenia, chronic kidney disease, predictive model, nomogram, machine learning, CHARLS
Citation: Lu R, Wang S, Chen P, Li F, Li P, Chen Q, Li X, Li F, Guo S, Zhang J, Liu D and Hu Z (2025) Predictive model for sarcopenia in chronic kidney disease: a nomogram and machine learning approach using CHARLS data. Front. Med. 12:1546988. doi: 10.3389/fmed.2025.1546988
Received: 23 December 2024; Accepted: 26 February 2025;
Published: 12 March 2025.
Edited by:
Maryanne Zilli Canedo Silva, São Paulo State University, BrazilReviewed by:
Thiago Santos Rosa, Catholic University of Brasilia (UCB), BrazilCopyright © 2025 Lu, Wang, Chen, Li, Li, Chen, Li, Li, Guo, Zhang, Liu and Hu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhijun Hu, aHpqejEwNjJAMTYzLmNvbQ==; Dan Liu, ZGFubGl1MDIxMkAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.