- 1Department of Infectious Diseases, The First Affiliated Hospital of Henan University of Science and Technology, Henan Medical Key Laboratory of Gastrointestinal Microecology and Hepatology, Luoyang, China
- 2Institute of Psychiatry and Neuroscience, Xinxiang Medical University, Xinxiang, China
- 3Department of Genetics and Prenatal Diagnosis, Hainan Women and Children’s Medical Center, Haikou, Hainan, China
- 4Department of Surgical Oncology, Xinxiang Central Hospital, The Fourth Clinical of Xinxiang Medical University, Xinxiang, China
- 5Priority Area Chronic Lung Diseases, Research Center Borstel - Leibniz Lung Center, Members of the German Center for Lung Research (DZL), Borstel, Germany
Introduction: The neutrophil cytosolic factor 1 (NCF1) rs201802880 polymorphism is a missense mutation resulting in an amino acid substitution from arginine to histidine at position 90, which impairs the function of NADPH oxidase. This casual variant confers an increased risk for multiple autoimmune disorders, including primary Sjögren’s syndrome and systemic lupus erythematosus. Given the high prevalence of this autoimmune disease risk variant in East Asia, we hypothesized that it may confer an evolutionary advantage by providing protection against infectious diseases.
Methods: To test this hypothesis, we investigated whether the NCF1 rs201802880 variant offers a protective effect against tuberculosis (TB), a historically significant and deadly infectious disease. Our study included 490 healthy controls and 492 TB patients who were genotyped for the NCF1 rs201802880 polymorphism.
Results: Our results showed that the NCF1 rs201802880 AA genotype was associated with a reduced risk of TB in women (OR= 0.25, 95% CI: 0.09-0.68, p=0.0023). Additionally, healthy individuals with the NCF1 rs201802880 AA genotype had significantly lower circulating white blood cell (5.56 ± 1.78 vs 6.43 ± 1.59, p=0.003) and neutrophil (3.23 ± 1.20 vs 3.74 ± 1.23, p = 0.02) counts compared to those with the GG or GA genotypes, with this difference being more pronounced in women than in men.
Conclusion: This study demonstrates that the autoimmune disease-causal NCF1 variant is associated with a protective effect against TB infection.
Introduction
Genetic association studies have identified the GTF2I-NCF1 intergenic region on chromosome 7 as a significant susceptibility locus for various autoimmune disorders, including primary Sjögren’s syndrome (pSS) (1, 2), systemic lupus erythematosus (SLE) (3, 4), rheumatoid arthritis (5), systemic sclerosis (SSc) (6), and neuromyelitis optica spectrum disorder (NMOSD) (7, 8). In 2017, the causal polymorphism within this susceptibility locus was pinpointed as the neutrophil cytosolic factor 1 (NCF1) rs201802880 G>A variant. This missense mutation results in an amino acid substitution from arginine (Arg) to histidine (His) at position 90 (9, 10). Beyond its association with disease susceptibility, the NCF1 Arg90His variant has been linked to various clinical and immunological features in SLE. These include an earlier age at diagnosis, presence of anti-beta2 glycoprotein I and anticardiolipin antibodies, increased formation of neutrophil extracellular traps (NETs), elevated serum interferon activity, and impaired macrophage efferocytosis (9–12). This association is further supported by experimental data from NCF1-His90 knock-in (KI) mice, which exhibit reduced oxidative burst, diminished macrophage efferocytosis, splenomegaly, increased type I interferon (IFN-I) scores, and higher levels of plasma cells, as well as enhanced Pristane-induced kidney disease compared to wild-type littermates (11).
The NCF1 Arg90 residue is evolutionarily conserved within the p47phox subunit of the phagocyte NADPH oxidase complex. The frequency of the NCF1 rs201802880 A allele varies significantly globally, being less than 0.5% in Caucasian populations, while exceeding 15% in East Asian groups such as Chinese, Japanese, and Korean populations (13). Despite its association with autoimmune disorders, including those linked to infertility and pregnancy loss due to autoantibodies such as anti-beta2 glycoprotein I and anticardiolipin antibodies (14, 15), the variant persists in human populations. This suggests that the NCF1 Arg90His variant may confer an evolutionary advantage, possibly by offering protection against infectious diseases. Indeed, neutrophils with the homozygous AA genotype demonstrate markedly reduced production of reactive oxygen species (ROS) compared to those with GG or GA genotypes (9, 12). Consistent with this, splenocytes from NCF1-His90 KI mice show a reduced capacity for ROS generation (11). Considering the crucial role of ROS in the pathogenesis of tuberculosis (TB) (16), a contagious disease caused by infection with Mycobacterium tuberculosis (Mtb) bacteria and coexisted with human for more than 40,000 years history (17), we hypothesize that the homozygous AA genotype may confer resistance to TB. This study aims to investigate the relationship between the NCF1 Arg90His variation and susceptibility to tuberculosis.
Materials and methods
Patients and control subjects
All patients with tuberculosis (TB) and healthy control subjects were recruited from the First Affiliated Hospital of Henan University of Science and Technology, Luoyang, China. The diagnosis of TB was made in accordance with the Chinese Guidelines for the Diagnosis and Treatment of Tuberculosis (2020 edition) (18). Specifically, individuals meeting any of the following four criteria were classified as having TB: (1) positive sputum smear for acid-fast bacilli; (2) culture of Mycobacterium tuberculosis from sputum, bronchoalveolar lavage fluid, or pleural effusion; (3) positive nucleic acid test for M. tuberculosis and/or positive culture in sputum, bronchoalveolar lavage fluid, or pleural effusion; (4) positive acid-fast bacilli staining or nucleic acid test for M. tuberculosis in lung tissue specimens from the lesion site. Both pulmonary and extrapulmonary TB patients were included in this study. All procedures were conducted in accordance with the principles of the Declaration of Helsinki, and ethical approval for the study protocol was obtained from the Ethics Committee of the First Affiliated Hospital of Henan University of Science and Technology. The ethical approval batch number is 2024-03-K187.
Data collection
Demographic and clinical data, including sex, age, clinical symptoms, purified protein derivative (PPD) skin test results, erythrocyte sedimentation rate (ESR), treatment regimens, and responses to treatment, were collected from electronic medical records. Hematological parameters, including counts of white blood cells (WBC), neutrophils (NEU), eosinophils (EOS), basophils (BAS), monocytes (MON), lymphocytes (LYM), red blood cells (RBC), and platelets (PLT), were measured for both TB patients and healthy controls using a Sysmex XN-1000 Analyzer (Sysmex, Japan).
DNA isolation and genotyping
Genomic DNA was extracted from peripheral blood leukocytes using the TaKaRa Blood Genome DNA Extraction Kit (Takara Biotechnology, Dalian Co., Ltd., China) following the manufacturer’s protocol. Genotyping of the NCF1 rs201802880 G>A polymorphism was conducted using nested PCR followed by a TaqMan assay, as previously described (10). Briefly, a specific NCF1 fragment was initially amplified through PCR by targeting the GTGT sequence in exon 2 of the gene. The resulting PCR product then served as the template for SNP genotyping using the TaqMan assay.
Statistical analysis
Statistical analyses were conducted using GraphPad Prism software (version 5.01, GraphPad Software Inc., La Jolla, CA, USA). Hardy–Weinberg equilibrium (HWE) was assessed using Fisher’s exact test, with p < 0.05 indicating a deviation from HWE. The Kolmogorov-Smirnov test was used to assess the normality of quantitative variables. For data following a normal distribution, comparisons between two groups were conducted using the Student’s t-test. For non-normally distributed data, the Mann-Whitney U test was applied. Genotype frequency differences were analyzed using Fisher’s exact test or chi-square test, as appropriate. Five genetic models—co-dominant, dominant, recessive, over-dominant, and additive—were applied for the genetic association analysis using the SNPSTATS program (https://www.snpstats.net/). The optimal inheritance model was determined based on the Akaike Information Criterion (AIC) and Bayesian Information Criterion (BIC), with the model yielding the lowest AIC and BIC values considered the best fit. Statistical significance was defined as p < 0.05.
Results
Demographic, clinical and laboratory features of patients with active tuberculosis
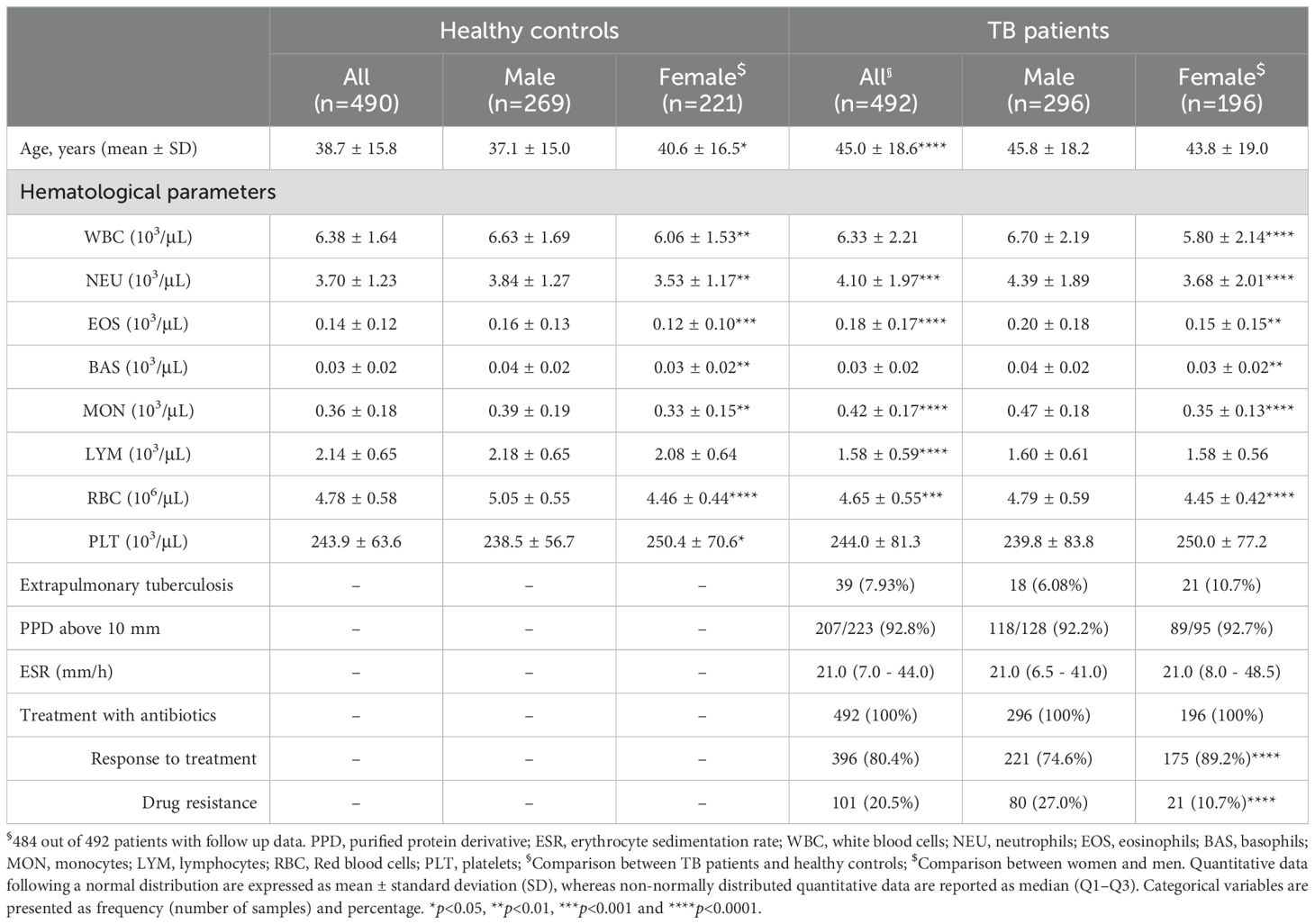
A total of 492 patients with active TB and 490 healthy control subjects were included in this study. The demographic, clinical, and laboratory characteristics of both groups are summarized in Table 1. The average age of TB patients was 45.0 ± 18.6 years, which was approximately 6 years older than that of the control subjects. Compared to the controls, TB patients exhibited higher levels of circulating neutrophils, eosinophils, and monocytes, while levels of lymphocytes and red blood cells were reduced. Among the 492 TB patients, 39 (7.93%) had extrapulmonary tuberculosis. The PPD skin test was administered to 223 patients, with 92.8% testing positive (defined as a reaction >10 mm). All patients received antibiotic treatment, with 54.9% showing a favorable response. Drug resistance was observed in 20.5% of patients, who were resistant to one or more antibiotics. The patient cohort included 196 females and 296 males, while the healthy control group comprised 221 females and 269 males. Stratified analysis revealed that females exhibited lower levels of WBC, neutrophils, eosinophils, basophils, monocytes, and RBC compared to males in both TB patients and healthy individuals. Furthermore, female TB patients demonstrated better treatment responses and a lower incidence of drug resistance compared to male patients (Table 1).
NCF1 rs201802880 AA genotype confers resistance to tuberculosis in women
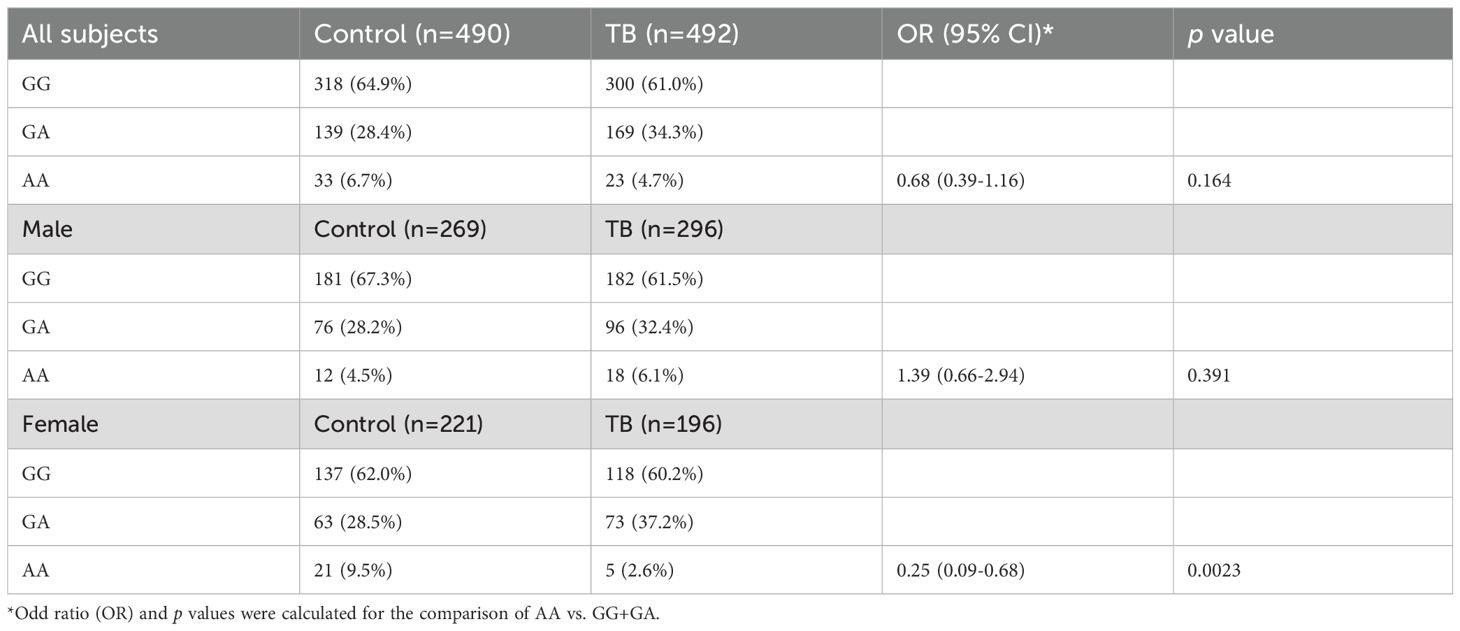
To assess the hypothesis that the NCF1 rs201802880 AA genotype offers protection against tuberculosis, we genotyped all 492 TB patients and 490 controls for this polymorphism. The genotype distribution for NCF1 rs201802880 was in Hardy-Weinberg equilibrium for both patient and control groups. Among the five genetic models, the recessive model demonstrated the lowest AIC and BIC values and was therefore selected for the association analysis (Supplementary Table 1).
Compared to healthy controls, TB patients exhibited a trend towards a lower frequency of the AA genotype, though this difference was not statistically significant (4.7% vs 6.7%, OR = 0.68, 95% CI: 0.39-1.17, p = 0.164) (Table 2). Given the association of NCF1 rs201802880 with autoimmune diseases, which predominantly affect women, we further explored the genotype-disease relationship by stratifying the analysis by gender. In women, the frequency of the AA genotype was significantly lower among TB patients compared to controls (2.6% vs 9.5%, OR = 0.25, 95% CI: 0.09-0.68, p=0.0023). Conversely, no significant difference was observed in men (Table 2).
Association between NCF1 rs201802880 and clinical features of TB
The observed association of the NCF1 rs201802880 variant with TB susceptibility in women prompted an investigation into its relationship with clinical features of TB. We compared patients with different genotypes (GG+GA vs AA) regarding clinical presentation, treatment responses, and follow-up outcomes. As detailed in Table 3, the two patient subgroups were comparable in terms of age, PPD test positivity, erythrocyte sedimentation rate (ESR), treatment response, drug resistance, and most hematological parameters. A significant difference was noted in platelet counts, with patients carrying the AA genotype exhibiting higher platelet levels compared to those with GG or GA genotypes (295.6 ± 107.9 vs 241.5 ± 79.5, p = 0.004). Additionally, although not statistically significant, there was a trend towards a lower rate of extrapulmonary tuberculosis (0.00% vs 8.32%) and a reduced female-to-male ratio (5/18 vs 191/278) in patients with the AA genotype (Table 3). Gender-stratified analysis did not reveal any additional difference between the two patient subgroups (Supplementary Table 2).

Table 3. Association of NCF1 Arg90His variation with clinical and immunological characteristics in patients with TB.
Association between NCF1 rs201802880 and neutrophil counts in healthy subjects
Given the substantial alterations in hematological parameters during Mtb infection, we next examined whether the NCF1 His90 variant is associated with hematological parameters in healthy subjects. Compared to those with GG or GA genotypes, individuals with the AA genotype had significantly lower white blood cell counts (5.56 ± 1.78 vs 6.43 ± 1.59, p = 0.003). This difference was primarily due to lower neutrophil counts in AA genotype carriers compared to GG+GA carriers (3.23 ± 1.20 vs 3.74 ± 1.23, p = 0.02), with no significant differences observed in other leukocyte types (Figure 1, Table 4). Additionally, healthy individuals with the AA genotype exhibit lower platelet counts compared to those with the GG or GA genotypes (Table 4). Stratified analysis by gender revealed that the reduction in circulating white blood cells and neutrophils associated with the AA genotype was more pronounced in women compared to men (Figure 1, Supplementary Table 3).
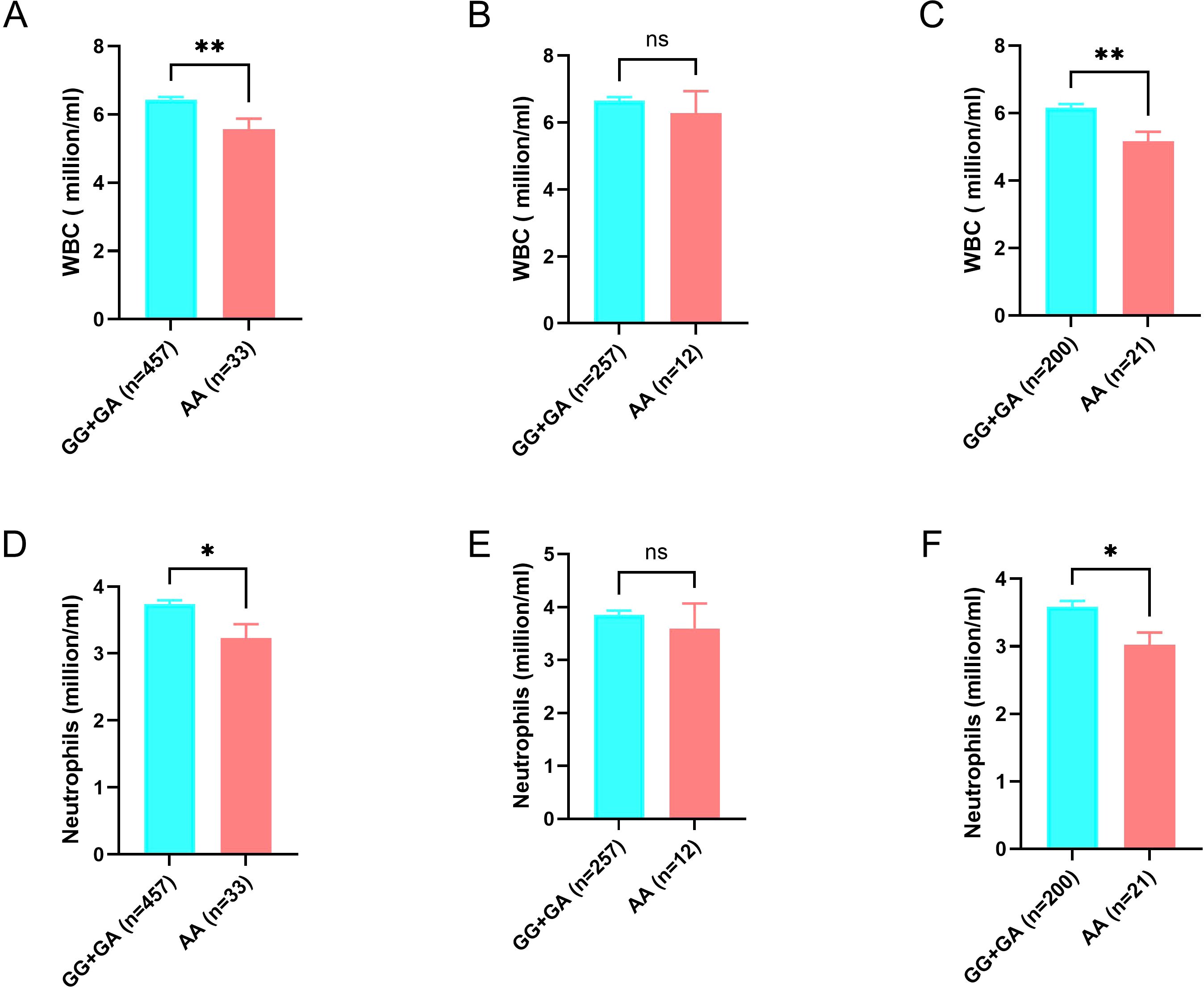
Figure 1. Association of NCF1 rs201802880 with circulating levels of white blood cells (WBC) and neutrophils in healthy control subjects. Comparison of levels of circulating WBC between AA and GG+GA genotypes in all subject (A), men (B) and women (C). Comparison of levels of circulating neutrophils between AA and GG+GA genotypes in all subject (D), men (E) and women (F). Data are presented as mean ± SEM. Statistical significance was determined using unpaired student’s t test. ns, not significant, *p<0.05 and **p<0.01.
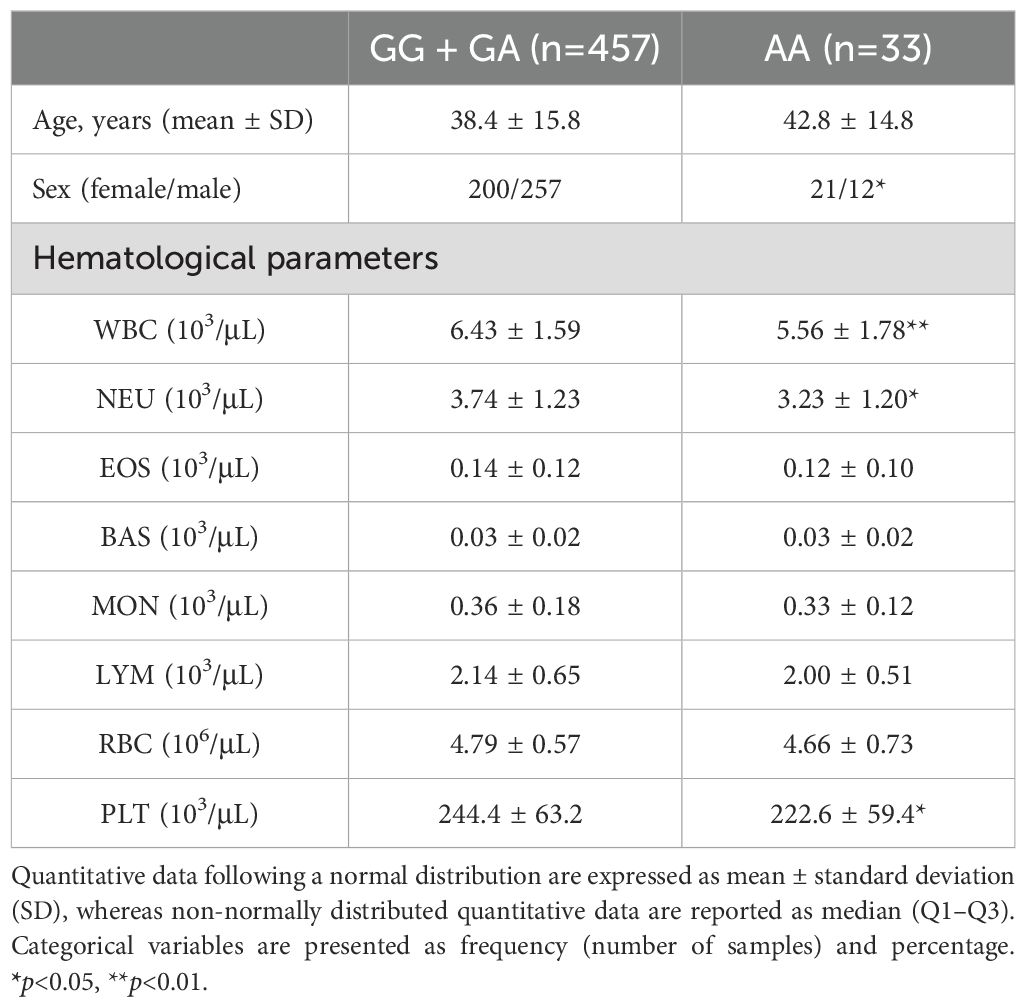
Table 4. Association of NCF1 Arg90His variation with laboratory characteristics in healthy subjects.
Discussion
In this study, we investigated the association between the autoimmune disease-causal variant NCF1 rs201802880 A and tuberculosis, a persistent global infectious disease. Our findings suggest that the AA genotype of the NCF1 rs201802880 polymorphism is associated with a protective effect against active TB in women. Furthermore, the AA genotype correlates with reduced levels of white blood cells and neutrophils in healthy individuals. To date, three genome-wide association studies (GWAS) have been conducted to investigate TB susceptibility in Chinese populations, identifying more than ten genetic loci associated with the disease (19–21). However, the genetic region encompassing the NCF1 gene on chromosome 7 has not been identified as a susceptibility locus for TB. This discrepancy may be attributed to the fact that all prior GWAS analyses were based solely on allele frequencies and did not incorporate stratified analyses by sex (19–21). Given that only the NCF1 AA genotype, rather than the NCF1 A allele, is associated with TB in women, it is unsurprising that this genetic association was not detected in previous studies.
The NCF1 rs201802880 A variant, while conferring protection against TB, is associated with an increased susceptibility to various autoimmune disorders, exemplifying evolutionary trade-offs (22). Throughout human history, our immune system has evolved under the selective pressure of infectious diseases such as TB, which posed significant threats to survival (23). Consequently, genetic variants that enhanced resistance to infections were positively selected. In modern contexts, where infectious diseases are less prevalent, these same variants may lead to overactive immune responses and contribute to autoimmune disorders (24).
Notably, the AA genotype of NCF1 rs201802880, which confers protection against TB in women, is linked to decreased circulating neutrophil levels in healthy individuals. This decrease is more pronounced in women than in men. Neutrophils, the most abundant leukocytes in the blood, play a crucial role in the early immune response to Mtb infection (25). In both human TB and animal models, lung disease manifestations are characterized by neutrophilic inflammation (25, 26), highlighting the critical role of neutrophils in TB pathogenesis.
Neutrophils are thought to play a dual role in the development of TB (27). On one hand, they are highly efficient pathogen-killing cells, employing both direct and indirect mechanisms to contribute significantly to the clearance of Mtb infection. On the other hand, neutrophils have been implicated in promoting Mtb growth and facilitating TB progression. For instance, studies have demonstrated that the risk of TB infection is inversely and independently associated with peripheral blood neutrophil count (28). Additionally, CXCL5 deficiency in murine TB models results in resistance to Mtb infection, attributed to impaired neutrophil recruitment from the bloodstream (29). Thus, it is plausible that the NCF1 rs201802880 AA genotype may confers protection against TB, at least in part, by reducing circulating neutrophil levels.
Both human studies and animal experiments have demonstrated that the NCF1 rs201802880 AA genotype results in reduced ROS production in neutrophils (9, 11, 12). While ROS are essential for neutrophil-mediated elimination of Mtb (30), pathogenic Mtb strains can exploit ROS to enhance their survival (31). For instance, it has been observed that virulent Mtb can persist within human neutrophils despite their rapid activation. This survival is associated with the necrotic death of infected neutrophils, a process entirely dependent on ROS production (32). Therefore, while a complete deficiency in neutrophil-derived ROS impairs the clearance of Mtb, a moderate reduction in neutrophil ROS production may paradoxically confer resistance to TB development.
Notably, the AA genotype is associated with TB specifically in women, but not in men. This sex-specific association suggests that the effect of the NCF1 variant is more pronounced in females than in males. Experimental evidence from knock-in mice supports this hypothesis, as female mice carrying the NCF1 AA genotype exhibit splenomegaly, increased IFN scores, the development of autoantibodies, and lupus-like kidney disease following pristane injection. In contrast, male mice with the same genotype show no evidence of autoimmune disease manifestation (11). The present study further supports this notion, as the decrease in circulating neutrophil levels associated with the AA genotype is more pronounced in women than in men.
Based on these findings, we propose a hypothetical mechanism for the protective effect of the NCF1 rs201802880 AA genotype against TB infection in women. The AA genotype may lead to lower circulating neutrophil levels, thereby impairing neutrophil recruitment to the lung and reducing the transition from Mtb infection to active TB and subsequent neutrophil-mediated tissue damage. Additionally, decreased ROS production in neutrophils may prevent Mtb-triggered ROS-dependent necrotic cell death and facilitate bacterial elimination. It is important to note that this is a simplified model, and other mechanisms involving various immune cells, such as macrophages and dendritic cells, and their dysregulation may also contribute to the observed protective effect against TB (9, 11, 12). For instance, the NCF1 variant may contribute to TB development by affecting the function of antigen-presenting cells. Evidence indicates that NADPH oxidase regulates the activity of cysteine cathepsins by modulating the lumenal redox potential, thereby influencing the production of the MHC II repertoire. This, in turn, impacts antigen presentation and CD4+ T cell-mediated immunity (33, 34). Therefore, it is plausible that the NCF1 AA genotype, which impairs ROS production, may alter the presentation of Mtb antigens to CD4+ T cells. Given the pivotal role of CD4+ T cells in controlling Mtb infection (35), it is reasonable to propose that the NCF1 variant confers resistance to TB in women by modulating antigen presentation.
This study has two main limitations. Firstly, the association between the NCF1 rs201802880 polymorphism was examined in a single case-control study, and the lack of replication compromises the robustness of the findings. Secondly, although the sample size is substantial, it may still be insufficient for stratified analyses. For example, examining associations between the NCF1 rs201802880 polymorphism and clinical features of TB in subgroups with relatively small sample sizes may reduce statistical power. Therefore, further validation in independent case-control studies with larger sample sizes is warranted.
In conclusion, this study is the first to demonstrate that the autoimmune disease-causal NCF1 variant is associated with a protective effect against TB infection. This finding exemplifies the evolutionary trade-offs where genetic variations that were positively selected for protection against infectious diseases may also increase the risk of autoimmune disorders.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
Ethics statement
The studies involving humans were approved by the Ethics Committee of the First Affiliated Hospital of Henan University of Science and Technology. The ethical approval batch number is 2024-03-K187. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
Author contributions
XH: Data curation, Investigation, Resources, Validation, Writing – original draft. SL: Data curation, Formal analysis, Investigation, Methodology, Validation, Writing – original draft. RH: Formal analysis, Investigation, Validation, Writing – original draft. ZC: Data curation, Formal analysis, Investigation, Writing – original draft. CM: Investigation, Writing – original draft. ZF: Formal analysis, Investigation, Writing – original draft. HH: Data curation, Writing – original draft. QZ: Data curation, Writing – original draft. FP: Supervision, Writing – original draft. XY: Conceptualization, Data curation, Funding acquisition, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. JZ: Funding acquisition, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing, Formal analysis.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was supported by National Natural Science Foundation of China (81571593), Henan Science and Technique Foundation (SB201903014, LHGJ20200511), Science and Technology Research and Development Plan of Henan Province (242103810039), Hainan Province Clinical Medical Center (QWYH202175), the Excellent Talent Team of Hainan Province (QRCBT202121), Deutsche Forschungsgemeinschaft (DFG) project YU 142/1-3 (272606465), and Bundesministerium für Bildung und Forschung (BMBF) via German Center for Lung Research (DZL), Airway Research Center North (ARCN).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1514296/full#supplementary-material
References
1. Li Y, Zhang K, Chen H, Sun F, Xu J, Wu Z, et al. A genome-wide association study in Han Chinese identifies a susceptibility locus for primary Sjogren’s syndrome at 7q11.23. Nat Genet. (2013) 45:1361–5. doi: 10.1038/ng.2779
2. Zheng J, Huang R, Huang Q, Deng F, Chen Y, Yin J, et al. The GTF2I rs117026326 polymorphism is associated with anti-SSA-positive primary Sjogren’s syndrome. Rheumatol (Oxford). (2015) 54:562–4. doi: 10.1093/rheumatology/keu466
3. Li Y, Li P, Chen S, Wu Z, Li J, Zhang S, et al. Association of GTF2I and GTF2IRD1 polymorphisms with systemic lupus erythematosus in a Chinese Han population. Clin Exp Rheumatol. (2015) 33:632–8.
4. Sun C, Molineros JE, Looger LL, Zhou XJ, Kim K, Okada Y, et al. High-density genotyping of immune-related loci identifies new SLE risk variants in individuals with Asian ancestry. Nat Genet. (2016) 48:323–30. doi: 10.1038/ng.3496
5. Kim K, Bang SY, Ikari K, Yoo DH, Cho SK, Choi CB, et al. Association-heterogeneity mapping identifies an Asian-specific association of the GTF2I locus with rheumatoid arthritis. Sci Rep. (2016) 6:27563. doi: 10.1038/srep27563
6. Liu C, Yan S, Chen H, Wu Z, Li L, Cheng L, et al. Association of GTF2I, NFKB1, and TYK2 regional polymorphisms with systemic sclerosis in a Chinese han population. Front Immunol. (2021) 12:640083. doi: 10.3389/fimmu.2021.640083
7. Shu Y, Guo J, Ma X, Yan Y, Wang Y, Chen C, et al. Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis is associated with IRF7, BANK1 and TBX21 polymorphisms in two populations. Eur J Neurol. (2021) 28:595–601. doi: 10.1111/ene.14596
8. Xie JL, Liu J, Lian ZY, Chen HX, Shi ZY, Zhang Q, et al. Association of GTF2IRD1-GTF2I polymorphisms with neuromyelitis optica spectrum disorders in Han Chinese patients. Neural Regener Res. (2019) 14:346–53. doi: 10.4103/1673-5374.244800
9. Olsson LM, Johansson AC, Gullstrand B, Jonsen A, Saevarsdottir S, Ronnblom L, et al. A single nucleotide polymorphism in the NCF1 gene leading to reduced oxidative burst is associated with systemic lupus erythematosus. Ann Rheum Dis. (2017) 76:1607–13. doi: 10.1136/annrheumdis-2017-211287
10. Zhao J, Ma J, Deng Y, Kelly JA, Kim K, Bang SY, et al. A missense variant in NCF1 is associated with susceptibility to multiple autoimmune diseases. Nat Genet. (2017) 49:433–7. doi: 10.1038/ng.3782
11. Geng L, Zhao J, Deng Y, Molano I, Xu X, Xu L, et al. Human SLE variant NCF1-R90H promotes kidney damage and murine lupus through enhanced Tfh2 responses induced by defective efferocytosis of macrophages. Ann Rheum Dis. (2022) 81:255–67. doi: 10.1136/annrheumdis-2021-220793
12. Linge P, Arve S, Olsson LM, Leonard D, Sjowall C, Frodlund M, et al. NCF1-339 polymorphism is associated with altered formation of neutrophil extracellular traps, high serum interferon activity and antiphospholipid syndrome in systemic lupus erythematosus. Ann Rheum Dis. (2020) 79:254–61. doi: 10.1136/annrheumdis-2019-215820
13. Zhang L, Wax J, Huang R, Petersen F, Yu X. Meta-analysis and systematic review of the association between a hypoactive NCF1 variant and various autoimmune diseases. Antioxidants (Basel). (2022) 11(8):1589. doi: 10.3390/antiox11081589
14. Carp HJ, Selmi C, Shoenfeld Y. The autoimmune bases of infertility and pregnancy loss. J Autoimmun. (2012) 38:J266–274. doi: 10.1016/j.jaut.2011.11.016
15. Hickman RA, Gordon C. Causes and management of infertility in systemic lupus erythematosus. Rheumatol (Oxford). (2011) 50:1551–8. doi: 10.1093/rheumatology/ker105
16. Shastri MD, Shukla SD, Chong WC, Dua K, Peterson GM, Patel RP, et al. Role of oxidative stress in the pathology and management of human tuberculosis. Oxid Med Cell Longev. (2018) 2018:7695364. doi: 10.1155/2018/7695364
17. Riccardi N, Canetti D, Martini M, Diaw MM, DIB A, Codecasa L, et al. The evolution of a neglected disease: tuberculosis discoveries in the centuries. J Prev Med Hyg. (2020) 61:E9–E12. doi: 10.15167/2421-4248/jpmh2020.61.1s1.1353
18. Jin F, Li Q, Bai C, Wang H, Li S, Song Y, et al. Chinese expert recommendation for diagnosis and treatment of massive hemoptysis. Respiration. (2020) 99:83–92. doi: 10.1159/000502156
19. Li M, Hu Y, Zhao B, Chen L, Huang H, Huai C, et al. A next generation sequencing combined genome-wide association study identifies novel tuberculosis susceptibility loci in Chinese population. Genomics. (2021) 113:2377–84. doi: 10.1016/j.ygeno.2021.05.035
20. Qi H, Zhang YB, Sun L, Chen C, Xu B, Xu F, et al. Discovery of susceptibility loci associated with tuberculosis in Han Chinese. Hum Mol Genet. (2017) 26:4752–63. doi: 10.1093/hmg/ddx365
21. Zheng R, Li Z, He F, Liu H, Chen J, Chen J, et al. Genome-wide association study identifies two risk loci for tuberculosis in Han Chinese. Nat Commun. (2018) 9:4072. doi: 10.1038/s41467-018-06539-w
22. Benton ML, Abraham A, LaBella AL, Abbot P, Rokas A, Capra JA. The influence of evolutionary history on human health and disease. Nat Rev Genet. (2021) 22:269–83. doi: 10.1038/s41576-020-00305-9
23. Liston A, Humblet-Baron S, Duffy D, Goris A. Human immune diversity: from evolution to modernity. Nat Immunol. (2021) 22:1479–89. doi: 10.1038/s41590-021-01058-1
24. Brinkworth JF, Barreiro LB. The contribution of natural selection to present-day susceptibility to chronic inflammatory and autoimmune disease. Curr Opin Immunol. (2014) 31:66–78. doi: 10.1016/j.coi.2014.09.008
25. Chandra P, Grigsby SJ, Philips JA. Immune evasion and provocation by Mycobacterium tuberculosis. Nat Rev Microbiol. (2022) 20:750–66. doi: 10.1038/s41579-022-00763-4
26. Capuano SV 3rd, Croix DA, Pawar S, Zinovik A, Myers A, Lin PL, et al. Experimental Mycobacterium tuberculosis infection of cynomolgus macaques closely resembles the various manifestations of human M. tuberculosis infection. Infect Immun. (2003) 71:5831–44. doi: 10.1128/IAI.71.10.5831-5844.2003
27. Lyadova IV. Neutrophils in tuberculosis: heterogeneity shapes the way? Mediators Inflammation. (2017) 2017:8619307. doi: 10.1155/2017/8619307
28. Martineau AR, Newton SM, Wilkinson KA, Kampmann B, Hall BM, Nawroly N, et al. Neutrophil-mediated innate immune resistance to mycobacteria. J Clin Invest. (2007) 117:1988–94. doi: 10.1172/JCI31097
29. Nouailles G, Dorhoi A, Koch M, Zerrahn J, Weiner J 3rd, Fae KC, et al. CXCL5-secreting pulmonary epithelial cells drive destructive neutrophilic inflammation in tuberculosis. J Clin Invest. (2014) 124:1268–82. doi: 10.1172/JCI72030
30. Segal AW. How neutrophils kill microbes. Annu Rev Immunol. (2005) 23:197–223. doi: 10.1146/annurev.immunol.23.021704.115653
31. Hilda JN, Das S, Tripathy SP, Hanna LE. Role of neutrophils in tuberculosis: A bird’s eye view. Innate Immun. (2020) 26:240–7. doi: 10.1177/1753425919881176
32. Corleis B, Korbel D, Wilson R, Bylund J, Chee R, Schaible UE. Escape of Mycobacterium tuberculosis from oxidative killing by neutrophils. Cell Microbiol. (2012) 14:1109–21. doi: 10.1111/j.1462-5822.2012.01783.x
33. Allan ER, Tailor P, Balce DR, Pirzadeh P, McKenna NT, Renaux B, et al. NADPH oxidase modifies patterns of MHC class II-restricted epitopic repertoires through redox control of antigen processing. J Immunol. (2014) 192:4989–5001. doi: 10.4049/jimmunol.1302896
34. Ewanchuk BW, Yates RM. The phagosome and redox control of antigen processing. Free Radic Biol Med. (2018) 125:53–61. doi: 10.1016/j.freeradbiomed.2018.03.040
Keywords: autoimmune diseases (AD), infectious diseases, neutrophil cytosolic factor 1 (NCF1), genetic association, evolutionary trade-offs, tuberculosis
Citation: Hu X, Li S, Huang R, Fu Z, Ma C, Cheng Z, Hu H, Zhou Q, Petersen F, Yu X and Zheng J (2025) The autoimmune disease risk variant NCF1-His90 is associated with a reduced risk of tuberculosis in women. Front. Immunol. 16:1514296. doi: 10.3389/fimmu.2025.1514296
Received: 20 October 2024; Accepted: 08 January 2025;
Published: 23 January 2025.
Edited by:
Selvakumar Subbian, Rutgers, The State University of New Jersey, United StatesReviewed by:
Stephen Carpenter, Case Western Reserve University, United StatesRamalingam Bethunaickan, National Institute of Research in Tuberculosis (ICMR), India
Copyright © 2025 Hu, Li, Huang, Fu, Ma, Cheng, Hu, Zhou, Petersen, Yu and Zheng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Junfeng Zheng, MTcxMDEzQHh4bXUuZWR1LmNu; Xinhua Yu, eGluaHVheXVAZnotYm9yc3RlbC5kZQ==
†These authors have contributed equally to this work
 Xinjun Hu
Xinjun Hu Shasha Li2†
Shasha Li2† Renliang Huang
Renliang Huang Frank Petersen
Frank Petersen Xinhua Yu
Xinhua Yu Junfeng Zheng
Junfeng Zheng