
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Environ. Eng. , 02 April 2025
Sec. Environmental Catalysis
Volume 4 - 2025 | https://doi.org/10.3389/fenve.2025.1532795
 Yaxing Dai1
Yaxing Dai1 Zhihao Liu2*
Zhihao Liu2*Chemical oxidation remediation is a technique that involves the addition of chemical oxidants to contaminated soil to degrade total petroleum hydrocarbons (TPHs), with the goal of reducing soil organic matter content or eliminating organic contamination. This paper reviews the research progress of several major chemical oxidation remediation technologies, including hydrogen peroxide (H2O2)-based Fenton and Fenton-like systems, calcium peroxide (CaO2)-based Fenton and Fenton-like systems, and persulfate-activated oxidation systems (e.g., Na2S2O8). Among these, the persulfate-activated oxidation system has recently emerged as a research hotspot due to its potential in eliminating TPHs from soil. The efficiency of TPHs degradation depends significantly on the activation method employed and the oxidative capacity of the system. Consequently, future research should focus on two critical directions: (1) the development of highly efficient, cost-effective, and environmentally sustainable activation methods; and (2) the enhancement of oxidative performance in existing systems, such as Na2S2O8/CaO2 and Na2S2O8/H2O2. In discussing the advancements in these major chemical oxidation remediation technologies, this paper specifically examines various persulfate activation methods and their corresponding treatment efficiencies. The aim is to provide insights and references for the development of efficient, cost-effective, and environmentally friendly persulfate-activated oxidation systems, thereby promoting the application of chemical oxidation remediation technologies in the treatment of petroleum hydrocarbon-contaminated soils.
With the continuous development of industry, the demand for fossil fuels is constantly increasing (Dai et al., 2024; Zhou et al., 2024), consequently, the issue of environmental pollution is becoming increasingly severe (Liu et al., 2024; Liu et al., 2023b). The exploitation of crude oil, the use of petroleum products, industrial production activities, discharges of oily wastewater, and natural spills have all contributed to the increasingly severe phenomenon of environmental pollution caused by TPHs (Ashjar et al., 2021; Wu et al., 2020). The leakage of Total Petroleum Hydrocarbons (TPHs) into the environment has altered the structure of soil and impacted microbial diversity (Zhang et al., 2023). As a result, the soil becomes a carrier of hazardous substances, thereby posing risks to soil ecology, human health, and the wellbeing of plants and animals (Sayed et al., 2021; Pinedo et al., 2014; Kim et al., 2022). Consequently, the treatment and remediation of soils contaminated with TPHs, and the restoration of soil ecological stability, have emerged as urgent priorities that demand immediate, and targeted, interventions to address the growing environmental and ecological concerns (Yuan et al., 2023; Peng et al., 2017; Kim et al., 2021).
At present, physical restoration (Chang et al., 2022), bioremediation (Bidja Abena et al., 2019; Feng et al., 2021; Othman et al., 2022), and chemical remediation (Li et al., 2022; Effendi et al., 2022; Liu et al., 2023a; Chen et al., 2022) are commonly used to remediate TPHs-contaminated soils. Among these approaches, chemical remediation technology has emerged as a research hotspot, primarily due to its strong pertinence, high treatment efficiency, and relatively short implementation period. Chemical remediation technology mainly encompasses three key methods: chemical flushing technology (Naeem and Qazi, 2020), chemical stabilization technology (Yang et al., 2020), and chemical oxidation technology (Chang et al., 2022; Bu et al., 2023; Li et al., 2024a). The chemical oxidation systems commonly used for TPHs-contaminated soils, such as the H2O2 Fenton oxidation system, the CaO2-based Fenton oxidation system, the activated Na2S2O8 oxidation system and the Na2S2O8/CaO2 composite oxidation system (Ni et al., 2024).
This paper primarily reviews the advancements in chemical oxidation technology, which aims to eliminate or mitigate soil contamination by introducing chemical oxidizing agents and activators. This process effectively degrades pollutants and restores soil health. The review encompasses the practical applications and current research status of several key chemical oxidation technologies, while also exploring potential future directions for development.
In 1893, Fenton HJ initially proposed that a mixed solution of H2O2 and Fe2+ exhibits potent oxidising properties, and is capable of degrading the majority of organic substances into inorganic states. As a result of the extensive research conducted, the Fenton oxidation system has become a more sophisticated and well-developed process. With the depth of research, Fenton oxidation system has been developed more mature. H2O2 is used as the oxidant and mixed with Fe2+. Under acidic conditions, Fe2+ catalyzes the generation of highly oxidative hydroxyl radicals (·OH), which have a standard electrode potential of 2.76 V. The hydroxyl radical (·OH) is strongly electrophilic and highly oxidative, capable of undergoing electrophilic reactions with most organic pollutants. This ultimately leads to the mineralization of these pollutants into H2O and CO2. The mechanism of treatment can be described as follows (Equations 1.1–1.6):
The remediation of petroleum hydrocarbons in soils has been the subject of extensive study with regard to Fenton oxidation systems based on H2O2 (Apul et al., 2016). It has been demonstrated that the choice of catalysts and the influencing factors have a significant impact on the final degradation. The traditional hydrogen peroxide oxidation Fenton oxidation system employs hydrogen peroxide in conjunction with Fe2+, which reacts at low pH to produce -OH, before undergoing further reaction. The Fenton oxidation system has been the subject of extensive research with the objective of enhancing its efficacy and reducing any potential environmental impact (Priyadarshini et al., 2022).
Chen et al. (2022) employed hydrogen peroxide (Fe2+/H2O2) oxidation systems, utilising Fe2+ as an activator to treat TPHs pollutants, the schematic diagram of the experimental setup was given (Figure 1a). The effects of the oxidant concentration, Fe2+ dosage, and initial pH conditions of the removal rate of TPHs from oil-based drilling cuttings as shown in Figure 1b. The findings demonstrated that under room temperature conditions, liquid–solid ratio = 10:1, H2O2 = 10 mmol/g, Fe2+ = 10 mmol/g,initial pH = 3, the TPHs removal rates were found to be 45.04% for the (Fe2+/H2O2) system. And the (Fe2+/H2O2) system removed up to 80% of C10-C13 components. In previous studies, Fernando Pardo (Pardo et al., 2014) and colleagues employed H2O2 as an oxidising agent (400–4,000 mmol L−1), ferric ions as a catalyst (5–20 mmol L−1), and trisodium citrate (50 mmol L−1) as a chelating agent. The removal efficiencies of petroleum hydrocarbons (TPHs) were obtained at two different pollutant concentrations (1,000–10,000 mg diesel kg/soil), with efficacies reaching up to 75%.
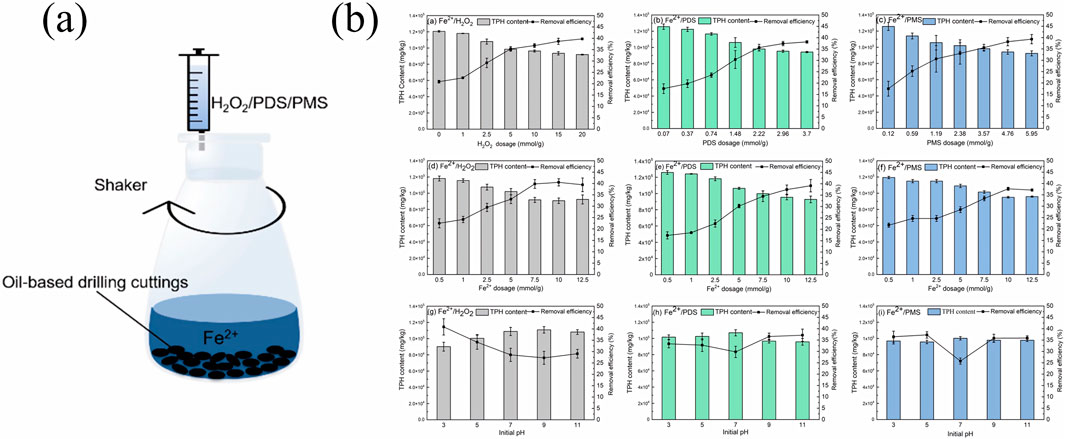
Figure 1. (a) The schematic diagram of the experimental setup and the effects of the oxidant concentration, Fe2+ dosage, (b) initial pH conditions of the removal rate of total petroleum hydrocarbons from oil-based drilling cuttings (Chen et al., 2022) Reproduced and modified with permission.
Akpoveta et al. (2018) achieved up to 87.6% degradation of TPHs (10% diesel) by Fenton oxidation with 350,000 mg/L H2O2 and 600 mg/L FeSO4, in the optimal room temperature range of 27°C–30°C, and at pH = 4.7, and the reaction products are environmentally friendly. Xu et al. (2011) employed three distinct oxidising reagents to address soil contamination resulting from cable insulating oil leaks. The efficacy of three different oxidising agents (H2O2, CaO2, and permanganate) in facilitating the oxidation of various carbon chain lengths, including C10-C12, C13-C16, C17-C20, C21-C24, C25-C40 (Table 1), and TPHs oxidation were evaluated. Among them H2O2 is the most effective of the three oxidants for chemical oxidation of cable oil contaminated soils under neutral pH conditions. Therefore, the experiments investigated the optimum concentration of oxidant, the optimum amount of iron catalyst and the addition of hydrogen peroxide for the treatment of TPHs with H2O2 as oxidant. The results showed that the optimum removal rate of 46% of cable oil contaminated soil was achieved when Fe(II) = 6.98 mmol/L and H2O2 at a concentration of 1,469 mmol/L was added at three times with a minimum time interval of 5 h. Yang et al. (2022) also pointed out that the optimal method of adding reagents is to add the iron catalyst in one step, then the stepwise addition of H2O2, which has no effect on the microbiological environment in the soil.
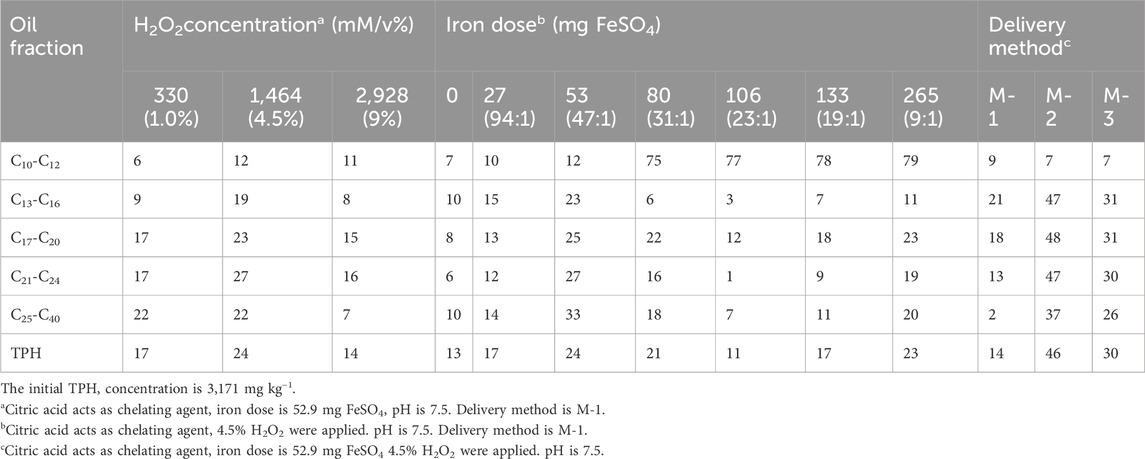
Table 1. Removal efficiency (%) of cable oil by liquid hydrogen peroxide pH 7.5 ((Xu et al., 2011) Reproduced and modified with permission).
In order to enhance the efficacy of the H2O2 Fenton oxidation system in the treatment of TPHs, various new Fenton composite remediation systems have been proposed. In a study conducted by Adhami et al. (2021), an electrokinetic-Fenton oxidation system (Figure 2) was employed to treat oil-based drilling waste. This resulted in a notable reduction in the TPHs content, from 31,600 mg/kg to 72,680 mg/kg, with an impressive degradation rate of 77%, and this electrokinetic-Fenton method has also been widely used in the treatment of organic matter in soils (Paixão et al., 2020).
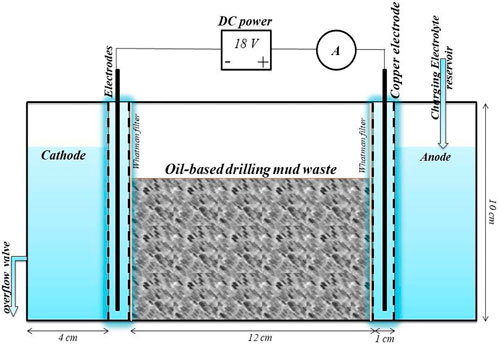
Figure 2. Schematic plot of the experimental EK cell (Adhami et al., 2021) Reproduced and modified with permission.
Sivagami et al. (2019) used the ultrasonic-Fenton method to treat TPHs in oil spill sludge, and under the conditions of pH = 3.0, sludge/water ratio = 1:100, ultrasonic power = 100 W and ultrasonic amplitude = 40–50%, and the weight ratio of H2O2/Fe2+ = 10:1, the removal of TPHs after ultrasonic treatment for 10 min was up to 84.25%, present excellent results.
The H2O2 Fenton system is distinguished by a minimal environmental impact, a broad spectrum of applications, and straightforward operation. However, the efficacy of TPHs degradation is significantly influenced by the pH value. Therefore H2O2-based Fenton systems should require a combination of UV or visible light (Wang et al., 2016a), sono-, electro-, photo-electro-, sono-electro-, heterogeneous electro- and sono-photo- techniques to achieve higher TPHs removal rates (Priyadarshini et al., 2022; Khodaveisi et al. 2011).
The traditional H2O2 Fenton oxidation system has shortcomings such as dependence on the pH of the soil environment (applicable under acidic conditions) and poor H2O2 stability (Xue et al., 2018b). Therefore, to overcome the shortcomings of the traditional H2O2-based Fenton technology, researches have proposed the CaO2-based Fenton technology, which uses calcium peroxide (CaO2) to generate H2O2 (Wang et al., 2016b) and then participates in the degradation reaction to treat organic pollutants in soil (Xue et al., 2019). Upon dissolution in water, calcium peroxide (CaO2) generates hydrogen peroxide (H2O2), which is then activated by Fe2+ to produce reactive radicals (·OH), thereby forming the Fenton reaction. The mechanism of treatment can be described as follows (Equations 2.1–2.4):
The results of the study (Northup and Cassidy, 2008; Bogan et al., 2003) indicate that the degradation rate of organic matter was increased to some extent when calcium peroxide was used instead of H2O2. In a study conducted by Jiang et al. (2021), calcium peroxide powder with a purity of 89.1% was prepared and used as an oxidising agent for degradation experiments on diclofenac sodium. The results demonstrated that the degradation rate of diclofenac sodium was 97.5% within 180 min, which was more effective than oxidation using H2O2-based Fenton’s system.
Ndjou’ou prepared a hydrocarbon-contaminated soil which was treated in laboratory slurry reactors using two types of modified Fenton (MF) chemistry (Ndjou’ou and Cassidy, 2006). The degradation rates of two oxidation systems, namely, liquid hydrogen peroxide (HP) and calcium peroxide (CaO2) – based oxidant (Cool-Ox™), were compared for TPHs and the four individual fractions (C6-C10, C11-C16, C17-C34, >C34). The results shown that, in soil with TPHs concentration = 10,604± (850) mg/kg, PH = 8, 40% w/v soil slurry, after 1 week, the removals of liquid HP (100 mL of 50% HP) and CaO2-based (50 g) treatments were 73.7% and 95.6%, respectively. The results demonstrated that the CaO2-based treatment exhibited a superior effect compared to the H2O2 treatment. This team also investigated the treatment of PAHs in soil by a calcium peroxide system (Gryzenia et al., 2009), which resulted in a degradation rate of 92.3% in 10 days at 21,420 g mg/kg, 500 g Cool-Ox™ powder, 40% w/v soil slurry.
Xue et al. (2018a) used a CaO2-based Fenton system to treat a mixed system of several petroleum hydrocarbon substances in water, including: benzene, toluene, ethylbenzene, and xylene (marked as BTEX). The oxidation effect of BTEX was investigated by changing the ratio of CaO2/Fe(II)/BTEX. The results demonstrated that when the CaO2/Fe(II)/BTEX molar ratio was 5/5/1, BTEX removal was 35%. Furthermore, when the ratio was increased to 40/40/1, the removal rate of BTEX increased significantly, reaching 98% in the same condition. These findings offer insights into the degradation of TPHs in soil.
In water using a Fenton system with CaO2. The objective was to investigate the oxidation effect of BTEX by varying the ratio of CaO2/Fe(II)/BTEX. The removal of BTEX was found to be 35% when the molar ratio of CaO2/Fe(II)/BTEX was 5/5/1. However, when the molar ratio was increased to 40/40/1, the removal rate increased significantly, reaching 98% in the same condition. The findings of this type of investigation may offer insights into the degradation of TPHs in soil. Furthermore, the efficacy of calcium peroxide can be enhanced by preparing it into nanoscale, which increases its specific surface area and consequently accelerates the degradation of organic matter (Khodaveisi et al., 2011; Yavari-Bafghi et al., 2022).
The use of persulfate as an oxidising agent for the degradation of organic pollutants has been widely reported (Lominchar et al., 2018; Xu et al., 2023). The stimulated activation of persulfate results in the generation of sulfate-free radicals (SO4−) with strong oxidising properties, which exhibit a standard electrode potential of 2.60 V. The most common methods of activation are heat, ultrasound (Darsinou et al., 2015; Deng et al., 2015), electrical current (Chen et al., 2019), ultraviolet (UV) light, and alkali conditions. Sodium persulfate, when activated by ferrous ions, decomposes to generate sulfate radicals (·SO4−), which are highly oxidative with a standard electrode potential of 2.60 V. These sulfate radicals oxidize organic pollutants through mechanisms such as electron transfer, addition reactions, and hydrogen atom abstraction. The mechanism of Fe2+ activation can be described as (Equation 3.1). Persulfate dissociates in water to produce the S2O82-, with a standard redox potential of E0 = 2.01 V, and S2O82- generates sulfate radicals (·SO4−) with a redox potential of E0 = 2.60 V. In alkaline conditions, these sulfate radicals can further produce hydroxyl radicals (·OH) with a higher chemical oxidation potential (E0 = 2.80 V). These reactive radicals react with pollutants in the soil, thereby facilitating the removal of contaminants, the mechanism of alkaline activation can be described as (Equations 3.2–3.4). The S2O82- can be activated under sufficient thermal conditions, leading to the cleavage of the peroxide bond and the formation of SO4−, the mechanism of heat activation can be described as (Equation 3.5):
Persulfate was employed for the remediation of TPHs in soil by researchers (Wu et al., 2016). The degradation of TPHs was observed to reach up to 40.8% when the initial TPHs concentration was 14,432.5 mg/kg, and the reaction was conducted for 24 h, the degradation rate was 21% higher than H2O2 treatment. The oxidation process was observed to be essentially complete within 60 min, as determined by kinetic experiments. To further enhance the degradation rate, heat-stimulated persulfate activation represents a conventional method for the treatment of TPHs.
Chen et al. employed two different persulfate-based advanced oxidation processes (Figure 3), namely heat/PMS and heat/PDS, for the treatment of TPHs, respectively (Chen et al., 2024). The optimal conditions for the removal of TPHs by the heat/PMS process were found to be 1.48 mmol/g, 80°C, and 120 min, with a degradation rate of approximately 53%. Similarly, the optimal TPHs removal conditions for the heat/PDS process were identified as 3.57 mmol/g, 70°C, and 80 min, which resulted in the degradation of approximately 46%.
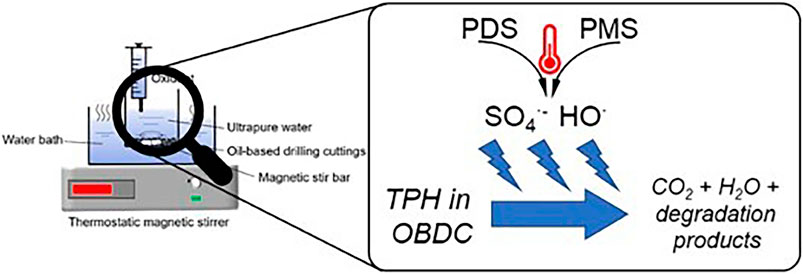
Figure 3. Schematic diagram of the experimental set-up for the heat/PDS and heat/PMS processes (Chen et al., 2024) Reproduced and modified with permission.
Li employed three different activated persulfate methods to treat TPHs in soil (Li et al., 2022), and investigated the effects of dosage, initial pH, temperature, ultrasonic power, and soil water ratio of persulfate on degradation, and the degradation mechanism of petroleum hydrocarbon compounds was also mentioned (Figure 4). After 72 h of treatment. Under the optimal conditions, the degradation efficiency of TPHs by ultrasound/heat activated persulfate (US/Heat/PS) was 78.2%. While the degradation efficiency of TPHs by heat activated persulfate (Heat/PS) system was 72.64%. The degradation efficiency of the single ultrasound activated persulfate (US/PS) system was 56.41%. The findings indicated that the concurrent utilisation of multiple activation methods to facilitate persulfate activation could potentially enhance the degradation rate of TPHs.
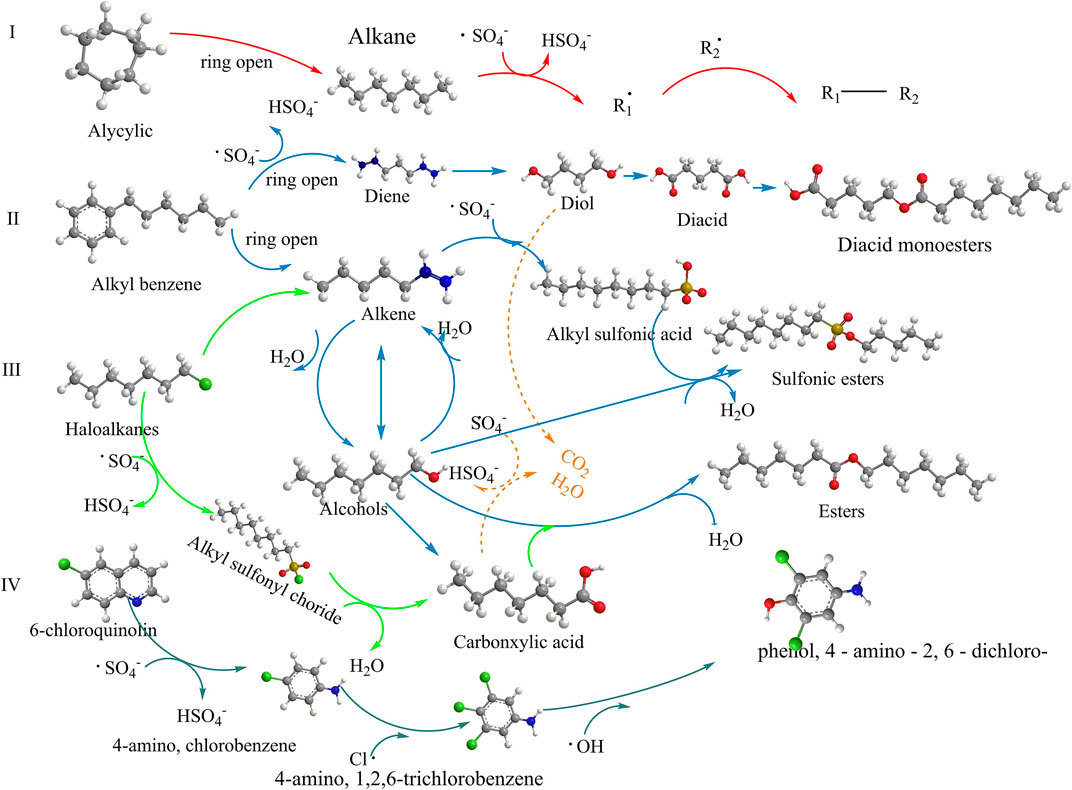
Figure 4. Proposed degradation mechanism of petroleum hydrocarbon compounds (Li et al., 2022) Reproduced and modified with permission.
Similarly, the use of different novel activation techniques can change the treatment effect of persulfate, also. Xue et al. (2022) mentioned a novel approach for the remediation of petroleum hydrocarbon-contaminated soil, which is utilising basic oxygen furnace slag (BOFs) and persulfate under electromagnetic induction heating (Figure 5b). Experimental findings demonstrated that persulfate can be effectively activated by BOFs or thermal induction under electromagnetic induction, thereby enhancing the oxidative degradation of BOFs/peroxysulfate and significantly accelerating the TPHs removal rate, the mechanism for removal of TPHs from soil under BOFs/persulfate system in presence of electromagnetic induction heating was given (Figure 5b).
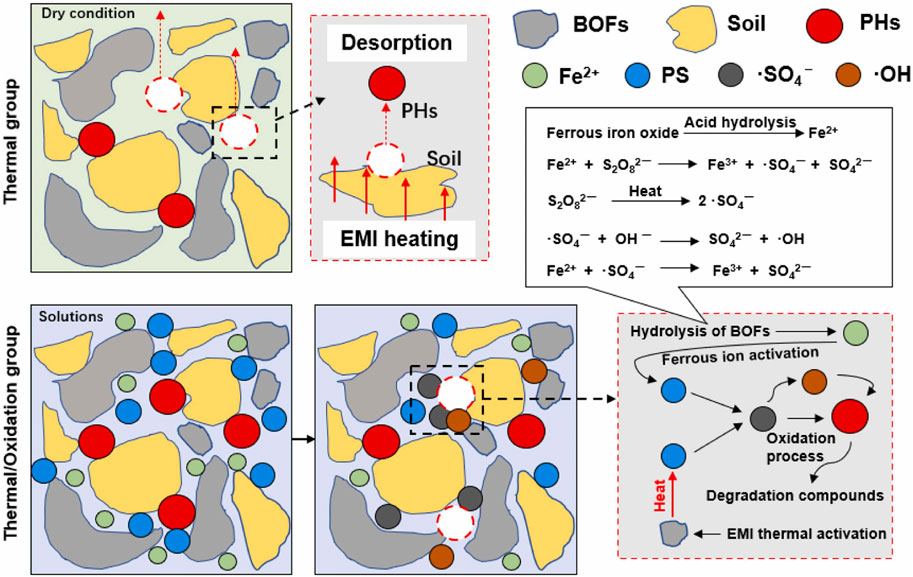
Figure 5. The schematic diagram of this work (a) and the mechanism for removal of TPHs from soil under BOFs/persulfate system in presence of electromagnetic induction heating (b) (Xue et al., 2022) Reproduced and modified with permission.
Liu et al. (2023a) investigated the efficacy of a degradation system combining persulfate and microorganisms in the treatment of high concentrations of crude oil (12,835 ± 572.76 mg/kg) contaminated soil. The combined effect of different doses of persulfate (PS) and hydrocarbon-degrading mixed bacteria was investigated as well. The results showed that, use 1% PS oxidation combined with bioremediation, the degradation rate was 80.05% after 180 days of degradation under optimal conditions, and the degradation rate is 4.88% higher than that of single-use biodegradation, 20.94% higher than that of natural attenuation of a single 1% persulfate. The addition of microorganisms has been observed to promote the secretion of enzymes, thereby enhancing the degradation rate of TPHs.
The effectiveness of FeS@BC (iron sulfide@biochar) in activating persulfate (PS) for the remediation of TPHs in petroleum-contaminated soil was systematically investigated (Xia et al., 2022). The optimal TPHs removal efficiency of 61.83% was achieved under the following conditions: a mass ratio of FeS to BC of 1:5, a PS dosage of 0.08 mmol/g, an FeS@BC dosage of 14 mg/g, and an initial pH value of 5. The study demonstrated that an appropriate amount of BC can enhance the dispersion of FeS and increase the number of active sites. However, an excess of BC can lead to agglomeration, thereby reducing the reaction efficiency. An adequate amount of PS can generate sufficient radicals to degrade TPHs, but an excessive amount of PS can cause radical quenching, which decreases the degradation efficiency. An appropriate amount of FeS@BC can promote the activation of PS, but an excess of FeS@BC can also lead to radical quenching and reduced degradation efficiency. The weakly acidic condition (pH = 5) was found to be the most favorable for TPHs degradation, as it ensures the optimal generation and stability of sulfate radicals (SO4−). Notably, the TPHs removal efficiency of the FeS@BC/PS system was significantly higher than that of the FeS/PS system (47.91%).
Li et al. (2024b) investigated the effectiveness of zero-valent iron (ZVI)-activated persulfate (PS) combined with electrokinetics (EK) for the remediation of TPHs petroleum-contaminated soil. The soil used in the study had a pH of 7.64, an organic matter content of 61.24 g/kg, and a total petroleum hydrocarbon concentration of 12,045 mg/kg. The study explored the impact of ZVI dosage on the TPHs removal efficiency of the ZVI/PS system, revealing that the optimal removal rate of 11.17% was achieved at a ZVI dosage of 0.95 g (PS:ZVI molar ratio of 10:1). Additionally, the influence of PS injection location on the TPHs removal efficiency of the EK/PS system was examined, with results indicating that simultaneous addition of PS to both the anode and cathode yielded the best performance, achieving a TPHs removal rate of 17.02%. When the EK-ZVI/PS system was employed for TPHs remediation under optimized conditions (0.95 g ZVI, PS added to both electrodes), a TPHs removal rate of 23.72% was obtained. Furthermore, the incorporation of 5% humic acid (HA) into the EK-ZVI/PS system enhanced the TPHs removal rate to 27.74%. The results demonstrated that the EK-ZVI/PS system outperformed the individual ZVI/PS and EK/PS systems in terms of TPHs removal efficiency, highlighting the significant synergistic effect of the combined technology. The optimal TPHs removal rate of 27.74% was achieved under the conditions of 0.95 g ZVI, simultaneous PS addition to both electrodes, and 5% HA addition.
Similarly, for the removal of TPHs from soil (Cao et al., 2024), investigated the effectiveness of in situ formed Fe/Mn oxide cross-linked with soil organic matter (Fe/Mn-SOM) complexes in activating persulfate (PS) for the remediation of TPHs soil. The soil (pH = 7.9) with a TPHs content of 30,421 mg/kg. The Fe/Mn-SOM complexes were formed in situ by adding Fe(OH)2 and Mn(OH)2 colloidal solutions to the soil, followed by the addition of H2O2. After the formation of the Fe/Mn-SOM complexes, PS solution was added to achieve oxidative degradation of TPHs. The study explored the effects of various factors on TPHs degradation efficiency, including the Mn:Fe molar ratio, PS concentration, initial pH, soil-to-water ratio, coexisting anions (Cl−, NO3−, HCO3−, H2PO4−), and organic matter (humic acid, urea, lignin). The results indicated that the highest TPHs removal efficiency of 75.74% was achieved under the conditions of a Mn:F molar ratio of 1:1, PS concentration of 2.0 mmol/L, initial pH of 7, and a soil-to-water ratio of 4:1. This efficiency was significantly higher than that of systems using PS alone (12.52%), Fe2+/PS (22.64%), Mn2+/PS (18.63%), and Fe2++Mn2+/PS (43.46%). Notably, the system also demonstrated a high removal rate for polycyclic aromatic hydrocarbons (PAHs) at 80.60%. The study found that the in situ formed Fe/Mn-SOM complexes could directly contact petroleum hydrocarbons in the soil, avoiding the ineffective consumption of free radicals during diffusion. The Fe/Mn-SOM complexes exhibited a certain tolerance to coexisting anions and organic matter, but high concentrations of HCO3−, H2PO4−, urea, and lignin significantly reduced TPHs removal efficiency. As an in situ formed PS activator, the Fe/Mn-SOM complexes are characterized by high efficiency, cost-effectiveness, and environmental friendliness, providing a novel technical approach for the remediation of petroleum hydrocarbon-contaminated soil.
The chemical oxidative remediation techniques in this paper are summarized below (Table 2).
The chemical oxidation remediation of total petroleum hydrocarbons (TPHs) in soil primarily relies on the use of strong oxidants, which generate oxidative radicals under various activation conditions. These radicals facilitate the oxidative degradation of TPHs. This paper reviews the mechanisms and remediation efficiencies of hydrogen peroxide-based Fenton oxidation, calcium peroxide-based Fenton and Fenton-like systems, and activated persulfate oxidation in the context of TPH-contaminated soil. The hydrogen peroxide-based Fenton oxidation system is highly sensitive to the pH of the reaction environment and is primarily suitable for soil remediation under acidic conditions. However, its applicability and degradation efficiency can be enhanced by combining it with different activation methods. The calcium peroxide-based Fenton and Fenton-like systems serve as alternative technologies to the hydrogen peroxide Fenton process, offering a broader pH range and addressing the limitations of hydrogen peroxide in neutral to alkaline environments. Notably, activated persulfate oxidation exhibits advantages such as strong oxidative capacity, long-term stability, and diverse activation methods. To improve the removal efficiency of TPHs in soil and develop effective TPHs oxidation systems, future research should focus on leveraging the high oxidative potential of activated persulfate systems by selecting composite oxidation systems, such as PS/CaO2 or PS/H2O2, and optimizing activation methods, including efficient thermal activation, iron-based composite activation, and alkaline activation. The integration of multiple activation techniques and novel activation methods represents a critical research direction for enhancing TPHs removal efficiency.
YD: Formal Analysis, Funding acquisition, Writing–original draft, Writing–review and editing. ZL: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Writing–review and editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Fund 2024 Chongqing “Graduate Student Research Innovation Project”, CYB240041 and Science and Technology Research Program of Chongqing Municipal Education Commission (KJQN202313101).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Adhami, S., Jamshidi-Zanjani, A., and Darban, A. K. (2021). Remediation of oil-based drilling waste using the electrokinetic-Fenton method. Process Saf. Environ. Prot. 149, 432–441. doi:10.1016/j.psep.2020.11.018
Akpoveta, O. V., Medjor, W. O., and Medjor, E. A. (2018). Fenton treatment via oxidative mechanism and its kinetics on soil polluted with automatic gas oil. Petroleum 4, 452–456. doi:10.1016/j.petlm.2018.03.001
Apul, O. G., Dahlen, P., Delgado, A. G., Sharif, F., and Westerhoff, P. (2016). Treatment of heavy, long-chain petroleum-hydrocarbon impacted soils using chemical oxidation. J. Environ. Eng. 142, 04016065. doi:10.1061/(asce)ee.1943-7870.0001139
Ashjar, N., Keshavarzi, B., Moore, F., Soltani, N., Hooda, P. S., and Mahmoudi, M. R. (2021). TPH and PAHs in an oil-rich metropolis in SW Iran: implication for source apportionment and human health. Hum. Ecol. Risk Assess. An Int. J. 29, 1–21. doi:10.1080/10807039.2021.2015285
Bidja Abena, M. T., Li, T., Shah, M. N., and Zhong, W. (2019). Biodegradation of total petroleum hydrocarbons (TPH) in highly contaminated soils by natural attenuation and bioaugmentation. Chemosphere 234, 864–874. doi:10.1016/j.chemosphere.2019.06.111
Bogan, B. W., Trbovic, V., and Paterek, J. R. (2003). Inclusion of vegetable oils in Fenton’s chemistry for remediation of PAH-contaminated soils. Chemosphere 50, 15–21. doi:10.1016/s0045-6535(02)00490-3
Bu, Y., Wu, N., Qi, Y., Wei, J., Tang, X., Allam, A. A., et al. (2023). Degradation of PCB77 in spiked soils by ferrous sulfide-activated persulfate combined with potassium permanganate. Chem. Eng. J. 467, 143477. doi:10.1016/j.cej.2023.143477
Cao, Z. Z., Xu, J. L., Dai, J., Liu, C. Y., and Tang, H. (2024). Direct oxidation of adsorbed petroleum hydrocarbons by activating persulfate with Fe/Mn-SOM complex preformed in-situ for heavily contaminated soil remediation. Chem. Eng. J. 496, 154049. doi:10.1016/j.cej.2024.154049
Chang, Y. C., Peng, Y. P., Chen, K. F., Chen, T. Y., and Tang, C. T. (2022). The effect of different in situ chemical oxidation (ISCO) technologies on the survival of indigenous microbes and the remediation of petroleum hydrocarbon-contaminated soil. Process Saf. Environ. Prot. 163, 105–115. doi:10.1016/j.psep.2022.05.019
Chen, F., Li, X., Ma, J., Qu, J., Yang, Y., and Zhang, S. (2019). Remediation of soil co-contaminated with decabromodiphenyl ether (BDE-209) and copper by enhanced electrokinetics-persulfate process. J. Hazard. Mater. 369, 448–455. doi:10.1016/j.jhazmat.2019.02.043
Chen, X., Mu, S., and Luo, Y. (2022). Degradation of petroleum pollutants in oil-based drilling cuttings using an Fe2+-based Fenton-like advanced oxidation processes. Environ. Sci. Pollut. Res. 30, 37669–37678. doi:10.1007/s11356-022-24925-2
Chen, X., Mu, S., and Luo, Y. (2024). Removal of total petroleum hydrocarbons from oil-based drilling cuttings by a heat activation persulfate-based process. Environ. Technol. 45, 835–844. doi:10.1080/09593330.2022.2128894
Dai, Y. X., Zhang, X. L., Liu, S. Y., Zhang, F. R. Z., Zhang, Y. X., Sang, Y. T., et al. (2024). Syntheses and properties of associative acrylamide copolymers containing short hydrophobic chains used in a friction reducer for slick-water fracturing. Petroleum Sci. 21, 1889–1901. doi:10.1016/j.petsci.2024.03.013
Darsinou, B., Frontistis, Z., Antonopoulou, M., Konstantinou, I., and Mantzavinos, D. (2015). Sono-activated persulfate oxidation of bisphenol A: kinetics, pathways and the controversial role of temperature. Chem. Eng. J. 280, 623–633. doi:10.1016/j.cej.2015.06.061
Deng, D., Lin, X., Ou, J., Wang, Z., Li, S., Deng, M., et al. (2015). Efficient chemical oxidation of high levels of soil-sorbed phenanthrene by ultrasound induced, thermally activated persulfate. Chem. Eng. J. 265, 176–183. doi:10.1016/j.cej.2014.12.055
Effendi, A. J., Ramadan, B. S., and Helmy, Q. (2022). Enhanced remediation of hydrocarbons contaminated soil using electrokinetic soil flushing – landfarming processes. Bioresour. Technol. Rep. 17, 100959. doi:10.1016/j.biteb.2022.100959
Feng, L., Jiang, X., Huang, Y., Wen, D., Fu, T., and Fu, R. (2021). Petroleum hydrocarbon-contaminated soil bioremediation assisted by isolated bacterial consortium and sophorolipid. Environ. Pollut. 273, 116476. doi:10.1016/j.envpol.2021.116476
Gryzenia, J., Cassidy, D., and Hampton, D. (2009). Production and accumulation of surfactants during the chemical oxidation of PAH in soil. Chemosphere 77, 540–545. doi:10.1016/j.chemosphere.2009.07.012
Jiang, Y. Y., Chen, Z. W., Li, M. M., Xiang, Q. H., Wang, X. X., Miao, H. F., et al. (2021). Degradation of diclofenac sodium using Fenton-like technology based on nano-calcium peroxide. Sci. Total Environ. 773, 144801. doi:10.1016/j.scitotenv.2020.144801
Khodaveisi, J., Banejad, H., Afkhami, A., Olyaie, E., Lashgari, S., and Dashti, R. (2011). Synthesis of calcium peroxide nanoparticles as an innovative reagent for in situ chemical oxidation. J. Hazard. Mater. 192, 1437–1440. doi:10.1016/j.scitotenv.2020.144801
Kim, J. W., Hong, Y. K., Kim, H. S., Oh, E. J., Park, Y. H., and Kim, S. C. (2021). Metagenomic Analysis for evaluating change in bacterial diversity in TPH-contaminated soil after soil remediation. Toxics 9, 319. doi:10.3390/toxics9120319
Kim, S. H., Woo, H., An, S., Chung, J., Lee, S., and Lee, S. (2022). What determines the efficacy of landfarming for petroleum-contaminated soils: significance of contaminant characteristics. Chemosphere 290, 133392. doi:10.1016/j.chemosphere.2021.133392
Li, X., Bu, Y., Xu, J., Alfassam, H. E., Rudayni, H. A., Allam, A. A., et al. (2024a). Degradation of 2-(2H-benzotriazol-2-yl)-4,6-di-tert-pentylphenol (UV328) in soil by FeS activated persulfate: kinetics, mechanism, and theoretical calculations. Environ. Pollut. 363, 125154. doi:10.1016/j.envpol.2024.125154
Li, Y. T., Sui, Q., Li, X., Wang, Y. Q., Liu, X. Y., Liu, H., et al. (2024b). Enhanced in-situ zero-valent iron activated persulfate oxidation with electrokinetics for the remediation of petroleum hydrocarbon contaminated soil. J. Environ. Chem. Eng. 12, 113781. doi:10.1016/j.jece.2024.113781
Li, Y. T., Zhang, J. J., Li, Y. H., Chen, J. L., and Du, W. Y. (2022). Treatment of soil contaminated with petroleum hydrocarbons using activated persulfate oxidation, ultrasound, and heat: a kinetic and thermodynamic study. Chem. Eng. J. 428, 131336. doi:10.1016/j.cej.2021.131336
Liu, N., Wang, L., Cao, D., Li, D., Zhu, Y., Huang, S., et al. (2023a). Remediation of petroleum contaminated soil by persulfate oxidation coupled with microbial degradation. J. Environ. Chem. Eng. 11, 109910. doi:10.1016/j.jece.2023.109910
Liu, Z. H., Chen, Z., Wang, Z., Zhang, D., and Ni, B. J. (2024). Recycling and repurposing of waste carbon nanofiber polymers: a critical review. Environ. Sci. Nano. 11, 1394–1411. doi:10.1039/d3en00784g
Liu, Z. H., Chen, Z., Zhang, D., and Ni, B. J. (2023b). Carboxyl and polyamine groups functionalized polyacrylonitrile fibers for efficient recovery of copper ions from solution. Environ. Sci. Pollut. Res. 31, 2243–2257. doi:10.1007/s11356-023-31227-8
Lominchar, M. A., Lorenzo, D., Romero, A., and Santos, A. (2018). Remediation of soil contaminated by PAHs and TPH using alkaline activated persulfate enhanced by surfactant addition at flow conditions. J Chem. Tech and Biotech 93, 1270–1278. doi:10.1002/jctb.5485
Naeem, U., and Qazi, M. A. (2020). Leading edges in bioremediation technologies for removal of petroleum hydrocarbons. Environ. Sci. Pollut. Res. 27, 27370–27382. doi:10.1007/s11356-019-06124-8
Ndjou’ou, A. C., and Cassidy, D. (2006). Surfactant production accompanying the modified Fenton oxidation of hydrocarbons in soil. Chemosphere 65, 1610–1615. doi:10.1016/j.chemosphere.2006.03.036
Ni, Y., Zhou, C., Xing, M., and Zhou, Y. (2024). Oxidation of emerging organic contaminants by in-situ H2O2 fenton system. Green Energy and Environ. 9, 417–434. doi:10.1016/j.gee.2023.01.003
Northup, A., and Cassidy, D. (2008). Calcium peroxide (CaO2) for use in modified Fenton chemistry. J. Hazard. Mater. 152, 1164–1170. doi:10.1016/j.jhazmat.2007.07.096
Othman, A. R., Ismail, N. S., Abdullah, S. R. S., Hasan, H. A., Kurniawan, S. B., Sharuddin, S. S. N., et al. (2022). Potential of indigenous biosurfactant-producing fungi from real crude oil sludge in total petroleum hydrocarbon degradation and its future research prospects. J. Environ. Chem. Eng. 10, 107621. doi:10.1016/j.jece.2022.107621
Paixão, I. c., López-Vizcaíno, R., Solano, A. M. S., Martínez-Huitle, C. a., Navarro, V., Rodrigo, M. A., et al. (2020). Electrokinetic-Fenton for the remediation low hydraulic conductivity soil contaminated with petroleum. Chemosphere 248, 126029. doi:10.1016/j.chemosphere.2020.126029
Pardo, F., Rosas, J. M., Santos, A., and Romero, A. (2014). Remediation of a biodiesel blend-contaminated soil by using a modified Fenton process. Environ. Sci. Pollut. Res. 21, 12198–12207. doi:10.1007/s11356-014-2997-2
Peng, H., Xu, L., Zhang, W., Liu, L., Liu, F., Lin, K., et al. (2017). Enhanced degradation of BDE209 in spiked soil by ferrous-activated persulfate process with chelating agents. Environ. Sci. Pollut. Res. 24, 2442–2448. doi:10.1007/s11356-016-7965-6
Pinedo, J., Ibáñez, R., Lijzen, J. P. A., and Irabien, Á. (2014). Human Risk Assessment of contaminated soils by oil products: total TPH content versus fraction approach. Hum. Ecol. Risk Assess. An Int. J. 20, 1231–1248. doi:10.1080/10807039.2013.831264
Priyadarshini, M., Das, I., Ghangrekar, M. M., and Blaney, L. (2022). Advanced oxidation processes: performance, advantages, and scale-up of emerging technologies. J. Environ. Manag. 316, 115295. doi:10.1016/j.jenvman.2022.115295
Sayed, K., Baloo, L., and Sharma, N. K. (2021). Bioremediation of total petroleum hydrocarbons (TPH) by bioaugmentation and biostimulation in water with floating oil spill containment booms as bioreactor basin. Int. J. Environ. Res. Public Health 18, 2226. doi:10.3390/ijerph18052226
Sivagami, K., Anand, D., Divyapriya, G., and Nambi, I. (2019). Treatment of petroleum oil spill sludge using the combined ultrasound and Fenton oxidation process. Ultrason. Sonochemistry 51, 340–349. doi:10.1016/j.ultsonch.2018.09.007
Wang, H., Zhao, Y., Li, T., Chen, Z., Wang, Y., and Qin, C. (2016a). Properties of calcium peroxide for release of hydrogen peroxide and oxygen: a kinetics study. Chem. Eng. J. 303, 450–457. doi:10.1016/j.cej.2016.05.123
Wang, N., Zheng, T., Zhang, G., and Wang, P. (2016b). A review on Fenton-like processes for organic wastewater treatment. J. Environ. Chem. Eng. 4, 762–787. doi:10.1016/j.jece.2015.12.016
Wu, H., Sun, L., Wang, H., and Wang, X. (2016). Persulfate Oxidation for the remediation of petroleum hydrocarbon-contaminated soils. Pol. J. Environ. Stud. 25, 851–857. doi:10.15244/pjoes/60857
Wu, N., Qu, R., Li, C., Bin-Jumah, M., Allam, A. A., Cao, W., et al. (2020). Enhanced oxidative degradation of decabromodiphenyl ether in soil by coupling Fenton-persulfate processes: insights into degradation products and reaction mechanisms. Sci. Total Environ. 737, 139777. doi:10.1016/j.scitotenv.2020.139777
Xia, C., Liu, Q., Zhao, L., Wang, L., and Tang, J. (2022). Enhanced degradation of petroleum hydrocarbons in soil by FeS@ BC activated persulfate and its mechanism. Sep. Purif. Technol. 282, 120060. doi:10.1016/j.seppur.2021.120060
Xu, J., Pancras, T., and Grotenhuis, T. (2011). Chemical oxidation of cable insulating oil contaminated soil. Chemosphere 84, 272–277. doi:10.1016/j.chemosphere.2011.03.044
Xu, Y., Wang, L., Wang, M., Xie, X., Xue, J., and Yao, M. (2023). Sequential reuse of remediated soil from petroleum-contaminated site to recover transition heavy metals and activate peroxysulphate oxidation. J. Water Process Eng. 56, 104354. doi:10.1016/j.jwpe.2023.104354
Xue, Y., Liu, J., Zhou, Y., Wang, T., and Xiang, L. (2022). Treatment of petroleum hydrocarbon contaminated soil by basic oxygen furnace slag activated persulfate oxidation in presence of electromagnetic induction heating. J. Environ. Chem. Eng. 10, 107267. doi:10.1016/j.jece.2022.107267
Xue, Y., Lu, S., Fu, X., Sharma, V. K., Mendoza-Sanchez, I., Qiu, Z., et al. (2018a). Simultaneous removal of benzene, toluene, ethylbenzene and xylene (BTEX) by CaO2 based Fenton system: enhanced degradation by chelating agents. Chem. Eng. J. 331, 255–264. doi:10.1016/j.cej.2017.08.099
Xue, Y., Sui, Q., Brusseau, M. L., Zhang, X., Qiu, Z., and Lyu, S. (2018b). Insight on the generation of reactive oxygen species in the CaO2/Fe (II) Fenton system and the hydroxyl radical advancing strategy. Chem. Eng. J. 353, 657–665. doi:10.1016/j.cej.2018.07.124
Xue, Y., Sui, Q., Brusseau, M. L., Zhou, W., Qiu, Z., and Lyu, S. (2019). Insight into CaO2-based Fenton and Fenton-like systems: strategy for CaO2-based oxidation of organic contaminants. Chem. Eng. J. 361, 919–928. doi:10.1016/j.cej.2018.12.121
Yang, B., Zhou, M., Meng, Y., Chen, K., Xu, J., Huang, X., et al. (2022). Hydrocarbons removal and microbial community succession in petroleum-contaminated soil under hydrogen peroxide treatment. Environ. Sci. Pollut. Res. 30, 27081–27091. doi:10.1007/s11356-022-23875-z
Yang, Z. H., Verpoort, F., Dong, C. D., Chen, C. W., Chen, S., and Kao, C. M. (2020). Remediation of petroleum-hydrocarbon contaminated groundwater using optimized in situ chemical oxidation system: batch and column studies. Process Saf. Environ. Prot. 138, 18–26. doi:10.1016/j.psep.2020.02.032
Yavari-Bafghi, M., Shavandi, M., Dastgheib, S. M. M., and Amoozegar, M. A. (2022). Simultaneous application of CaO2 nanoparticles and microbial consortium in Small Bioreactor Chambers (SBCs) for phenol removal from groundwater. Process Saf. Environ. Prot. 160, 465–477. doi:10.1016/j.psep.2022.02.039
Yuan, L., Wu, Y., Fan, Q., Li, P., Liang, J., Liu, Y., et al. (2023). Remediating petroleum hydrocarbons in highly saline–alkali soils using three native plant species. J. Environ. Manag. 339, 117928. doi:10.1016/j.jenvman.2023.117928
Zhang, D., Hu, Q., Wang, B., Wang, J., Li, C., You, P., et al. (2023). Effects of single and combined contamination of total petroleum hydrocarbons and heavy metals on soil microecosystems: insights into bacterial diversity, assembly, and ecological function. Chemosphere 345, 140288. doi:10.1016/j.chemosphere.2023.140288
Keywords: total petroleum hydrocarbons, chemical oxidative remediation technologies, fentonbased reaction, activated persulfate, advanced oxidation
Citation: Dai Y and Liu Z (2025) Research progress on remediation of total petroleum hydrocarbons in soil by chemical oxidation - a review. Front. Environ. Eng. 4:1532795. doi: 10.3389/fenve.2025.1532795
Received: 22 November 2024; Accepted: 03 March 2025;
Published: 02 April 2025.
Edited by:
Eleonora Aneggi, University of Udine, ItalyCopyright © 2025 Dai and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhihao Liu, emhpaDk4bGl1QDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.