
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Endocrinol. , 01 December 2022
Sec. Clinical Diabetes
Volume 13 - 2022 | https://doi.org/10.3389/fendo.2022.960343
 Mingyu Zhang1
Mingyu Zhang1 Taoyuan Yin2
Taoyuan Yin2 Feng Xia3
Feng Xia3 Suhong Xia1
Suhong Xia1 Wangdong Zhou1
Wangdong Zhou1 Yu Zhang1
Yu Zhang1 Xu Han1
Xu Han1 Kai Zhao1
Kai Zhao1 Lina Feng1
Lina Feng1 Ruonan Dong1
Ruonan Dong1 Dean Tian1
Dean Tian1 Yan Yu1*
Yan Yu1* Jiazhi Liao1*
Jiazhi Liao1*Hypertriglyceridemia (HTG) is one of the most common clinical dyslipidemia. Nevertheless, stroke and acute pancreatitis co-occurrence due to hypertriglyceridemia are extremely rare. We present a case of hypertriglyceridemia-associated stroke and pancreatitis in a 39-year-old woman. The patient’s laboratory tests reported high triglyceride concentrations beyond the instrument’s detection range, and radiological examination showed typical signs of cerebral infarction and acute pancreatitis. The patient received combined blood purification therapy, intravenous thrombolysis with urokinase, and conservative treatment of pancreatitis. We discuss the clinical features, pathogenesis, diagnosis, and treatment of hypertriglyceridemic stroke and pancreatitis combined with the relevant literature. We reviewed the mechanisms by which triglycerides contribute to atherosclerosis and acute pancreatitis. We point out the superiority of combined blood purification therapy and caution physicians about the effects of prescribed drugs on blood lipids.
Severe hypertriglyceridemia is a common risk factor for acute pancreatitis and atherosclerotic vascular disease (1, 2). A population-based study in Copenhagen revealed that triglyceride levels greater than 5 mmol/L were associated with a 3-fold increased risk of ischemic stroke and a 10-fold increased risk of acute pancreatitis compared to levels less than 1 mmol/L (3). The higher the triglyceride level, the higher the risk of suffering from acute pancreatitis. However, very severe and extremely high triglyceride levels may not further increase the risk of stroke (4). It has been mentioned that the co-occurrence of stroke and acute pancreatitis due to hypertriglyceridemia is rare (5). This article detailed such a rare case and searched for similar case reports. We present the first review of two complications of hypertriglyceridemia from the perspectives of clinical features, mechanism, diagnosis, and treatment.
A 39-year-old female patient was initially admitted to a local clinic for sudden-onset dizziness, unsteady standing, right-sided limb weakness, and choking cough when swallowing water. The local clinic found that her blood pressure was 180/140mmHg, but a CT head scan showed no significant abnormalities. That evening she experienced the same symptoms above, accompanied by vomiting, and the vomitus was dark green liquid. She was immediately admitted to the emergency department of Qichun County People’s Hospital for head CT. It did not show any significant abnormalities. Given the patient’s symptoms, she underwent further examination. The CDFI revealed left vertebral artery stenosis. Then cranial DWI revealed brainstem infarction (Figure 1A), and the patient was given urokinase thrombolysis, oxygen inhalation, antihypertensive, lipid-lowering, and intracranial pressure-lowering (diuretics and mannitol) treatments at once. The patient suddenly developed severe pain in the right abdomen accompanied by vomiting, and then she was taken to our emergency department.
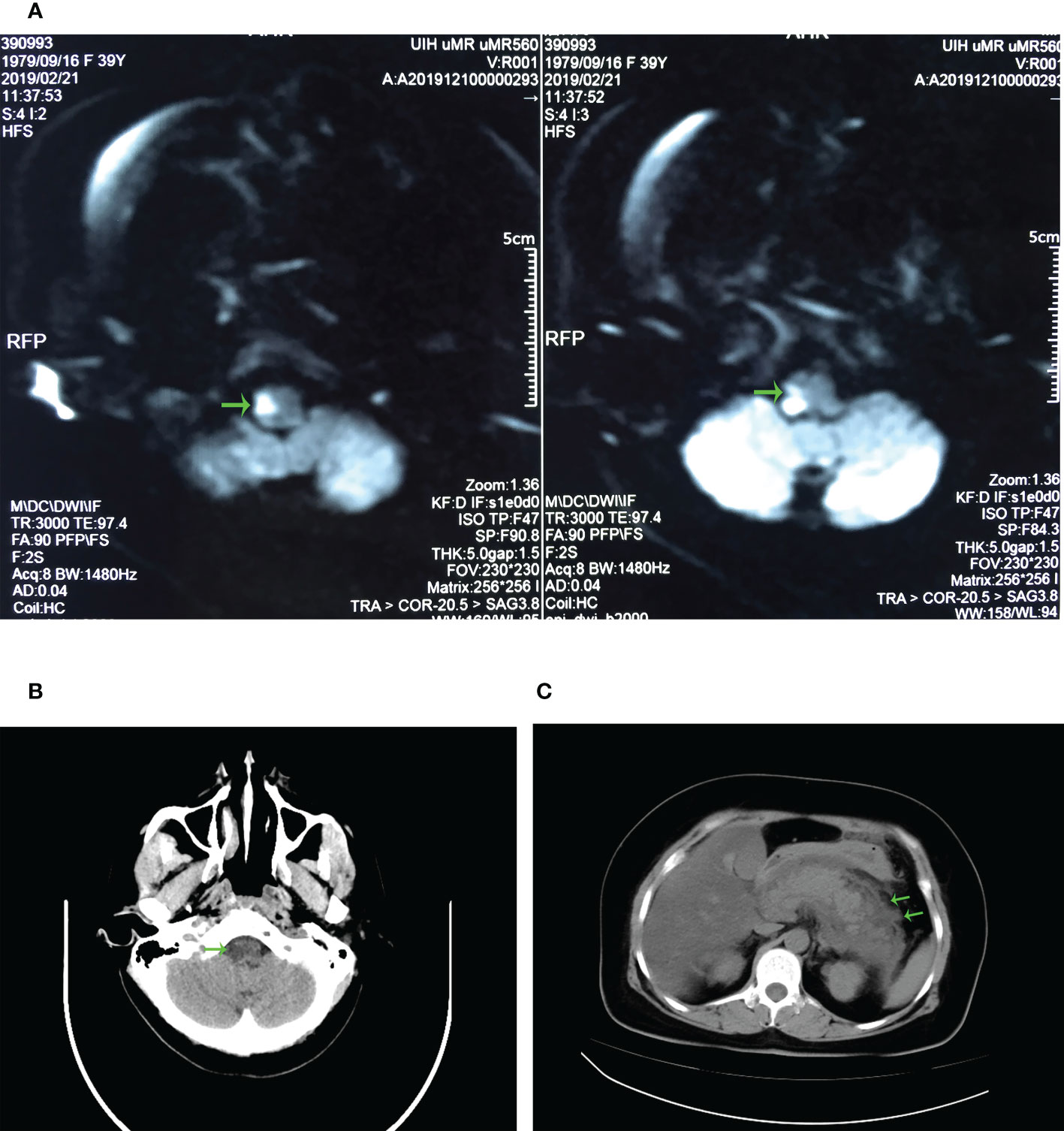
Figure 1 Radiology results. (A) Patient’s cranial DWI imaging. The green arrow marks the abnormal signal on the right side of the patient’s medulla oblongata, which is considered as acute cerebral infarction. (B) CT imaging of the patient’s head. The green arrow marks a small hypodense lesion on the right side of the medulla oblongata, suggesting the possibility of infarction on the right side of the medulla oblongata. (C) CT imaging of the patient’s abdomen. The green arrow marks the swelling and exudation of the pancreas, indicating acute pancreatitis.
The patient’s acute cerebral infarction was properly treated in Qichun County People’s Hospital, her neurological symptoms were relatively stable, and she was transferred to our hospital mainly because of an abdominal emergency. After admission, relevant laboratory investigations were performed. The patient’s blood pressure was 137/97mmHg, INR was 1.14, APTT value was 30.5s. The patient’s blood pressure and these coagulation parameters were normalized thanks to treatment at other hospital. While the patient’s blood glucose was raised at 16.03 mmol/L, arterial pH was 7.334 with bicarbonate of 17.8 mmol/L, base excess -7.10 mmol/L, and lactate 3.80 mmol/L. Her initial plasma level of the coagulation biomarker D-dimer was 0.87 ug/mL FEU, thrombin-time (TT) was 20.8 seconds, neutrophil count was 18.67 *10^9/L (93.3%), lymphocyte count was 0.62*10^9/L (3.1%), red blood cell count was 5.19*10^12/L, platelet count was 216*10^9/L. C-reactive protein was 50.9 mg/L, pancreatic amylase was 1028 IU/L, lipase was 2509.8 IU/L, total cholesterol was >20.70 mmol/L, triglycerides levels was >50.00 mmol/L, HDL was 0.79mmol/L, LDL was 1.10mmol/L and calcium was 1.52 mmol/L. The hCG value was 2.9 mIU/ml.
CT scans of the head, abdomen and abdominal CT angiography were perfumed in our hospital within 24 hours. At this time, head CT revealed a small hypodense lesion on the right side of the medulla oblongata (Figure 1B), which was consistent with the presentation after intravenous thrombolysis of stroke. Abdominal CT revealed pancreatic swelling and exudation (Figure 1C), fatty liver, and double renal calculi. Since the patient had symptoms of sudden abdominal pain, we performed an abdominal CTA to investigate whether she had an aortic dissection. No apparent double-lumen structure was observed in the thoracoabdominal aorta.
The patient had a history of diabetes, which had not been treated regularly for five years. There was no history of smoking or drinking. Based on the clinical presentation, laboratory tests, and radiological examination, we can determine that the patient successively developed cerebral infarction and acute pancreatitis quickly. We centrally reviewed all laboratory test results, and the patient’s hyperlipidemia, especially the triglyceride level much higher than the reference value, gained our attention. We performed a literature search, we searched Google Scholar for keywords “(cerebral infarction OR embolism OR stroke) AND pancreatitis” and found several cases (Table 1). We conclude that it is a rare case, and the patient’s hypertriglyceridemia might cause two complications, cerebral infarction and acute pancreatitis, to co-occur.
We want to lower triglyceride levels in patients as early as possible. As the dual filtration plasma exchange has fewer overall adverse effects than the conventional ones, it saves much exogenous plasma and reduces the incidence of blood-borne infections. After obtaining the patient’s consent, we performed dual filtration plasma exchange treatment via the right internal jugular vein, using 30 mg of heparin for anticoagulation throughout the procedure, disposing of 3200 ml of plasma, discarding 400 ml of plasma, supplementing with 100 ml of albumin and 300 ml of saline. At the end of the treatment, heparin was neutralized with 50mg of protamine. Due to the patient’s recent history of cerebral infarction and the use of heparin in the dual filtration plasma exchange technique, we closely observed the patient for new bleeding spots throughout the body and were alert for cerebral hemorrhage after cerebral infarction.
As needed, the patient was treated the same day with hemoperfusion combined with continuous veno-venous hemofiltration (CVVH), which has been proven effective in many studies. The whole process took 6 hours, and the ultrafiltration volume was 0.5 liters. The coupled blood purification method reduces triglyceride levels while removing unwanted inflammatory factors, creatinine, blood urea nitrogen, and excess water from the body. Triglyceride level decreased to 4.03 mmol/L after treatment.
We performed two more hemoperfusions over the next few days combined with CVVH. In addition, we continued to maintain proper fluid balance, use insulin intravenous drip to lower blood sugar, apply of lipid-lowering medication (fibrates), administer antibiotics (biapenem, ornidazole and levofloxacin) to combat infection, inhibit pancreatic enzyme secretion, relieve pain (dizocine and diclofenac sodium), apply of antiplatelet therapy (aspirin and clopidogrel) and correct electrolyte disorders and acid-base balance disorders. We ended up with good laboratory results, we graphed the patient’s major lipid changes (Figure 2). The patient was discharged 23 days after admission. At the time of discharge, the patient’s upper and lower limbs were still unable to resist gravity, her NIHSS score was 6.
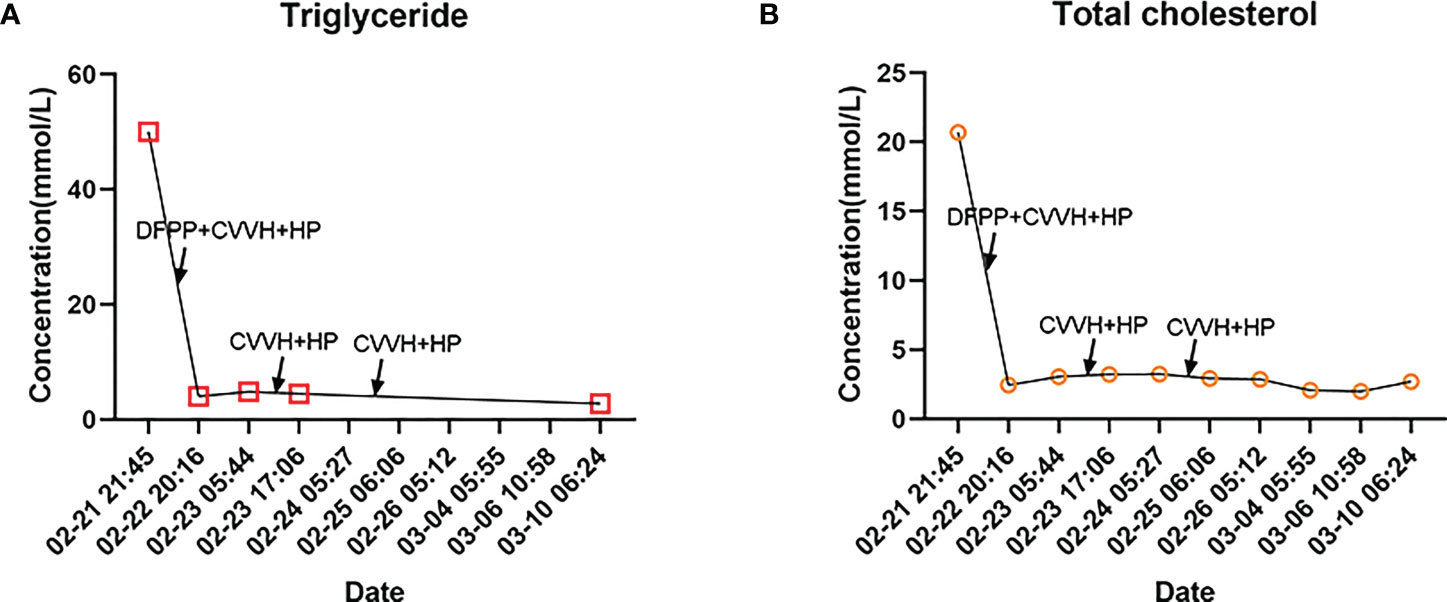
Figure 2 Major lipid trends in our patient. (A) Trends in triglyceride changes during hospitalization. Maximum lipid-lowering benefit was obtained in the first combined blood purification therapy, with further reduction in triglyceride levels in the subsequent treatment. (B) Trends in total cholesterol changes during hospitalization. The trend was generally consistent with triglyceride levels, and total cholesterol levels returned to normal after the first combined blood purification therapy. Abbreviations: Double Filtration Plasmapheresis (DFPP); Continuous veno-venous hemofiltration (CVVH); Hemoperfusion (HP).
In Table 1, patients in Case 1-3 all developed acute pancreatitis first, either alcohol-related or traumatic. These patients suffered sudden cerebral infarction about a week later, and all three patients had infarcts in the left middle cerebral artery. The cerebral infarction that occurred in these patients was attributed to fat embolism. In patients with severe acute pancreatitis, chylomicrons and very low-density lipoprotein (VLDL) are released from necrotic omental fat into the circulation, in response to the patient’s elevated C-reactive protein, calcium-dependent agglutination of chylomicrons and VLDL occurs, ultimately leading to vascular occlusion (11). When cerebral infarction occurs after acute pancreatitis, and the two conditions are separated by a period of time, and the patient does not have other thrombosis-prone risk factors, we reasonably presume that the cerebral infarction is a rare complication of acute pancreatitis in this case. Case 4 reported a 4-year-old child who developed recurrent acute pancreatitis and acute cerebral infarction. When the patient is very young, we should take genetic defects into account in the first instance. Case 5 and our case both had hyperlipidemia induced by diabetes, especially with triglyceride levels much higher than normal. Patients with hypertriglyceridemia must be alert to the occurrence of pancreatitis and cerebral infarction, and the application of blood purification techniques for rapid lipid lowering seems to be necessary.
Genetic and acquired factors influence triglyceride levels. VLDL and chylomicrons are both major transporters of triglycerides in the body. Some gene defects can lead to defective lipase activity so that triglyceride clearance is inhibited. Other genetic mutations cause reduced hepatic clearance of VLDL and chylomicron remnants. Diabetes, alcoholism, pregnancy and certain drugs can also lead to triglyceride overproduction (12).
Our patient was not tested for the relevant genetic defects. Genetic testing is not a commonly used test at the time of diagnosis. We can only indirectly understand the genetic conditions of patients by asking about family history. More attention should be paid to factors that can cause secondary metabolic disorders, such as poor control of diabetes, heavy drinking and medication use. Our patient did have a history of diabetes that had not been well treated for many years.
Hypertriglyceridemia is the most common cause of acute pancreatitis after biliary system disorders and alcohol (13). As many as 25.6% of acute pancreatitis is associated with dyslipidemia in patients (14).On the one hand, large amounts of triglycerides are hydrolyzed by lipase in the vascular bed of the pancreas to produce excess free fatty acids. Human plasma albumin is limited in amount, and free fatty acids that fail to bind to it aggregate into micellar structures with detergent properties. This micellar structure has toxic effects on platelets, vascular endothelial cells, and acinar cells, eventually inducing ischemia and acidosis. On the other hand, elevated levels of giant lipoproteins rich in triglycerides, namely chylomicrons, increase the viscosity of the blood. A marked rise in the viscosity of the plasma can result in capillary blockage, which exacerbates ischemia and acidosis, and triggers acute pancreatitis through activating trypsinogen (15–17).
LDL cholesterol is a recognized mediator of atherosclerosis, but atherosclerotic vascular events still occur in low LDL cholesterol levels, for which hypertriglyceridemia is a residual risk factor (18). Significant triglyceride increases may be associated with a hypercoagulable state, as factor VIIIc, factor VIIc, fibrinogen, and antithrombin III levels are significantly elevated (19). Hypertriglyceridemia independently promotes increased plasma viscosity and reduced blood flow (20). It has been reported that elevated triglyceride levels are associated with atherosclerotic vascular disease (21, 22). Surprisingly, triglycerides themselves do not directly promote atherosclerotic plaque formation, and in patients with mild to moderate hypertriglyceridemia, VLDL, their residues, and medium-density lipoproteins put individuals at increased risk of atherosclerosis. While in patients with severe hypertriglyceridemia, the cholesterol deposited in plaque may originate from triglyceride-rich chylomicron residues (23). Chylomicron and VLDL remnants are small in diameter and easily penetrate the arterial wall into the subendothelial space (24, 25). These residual particles carry more cholesterol than LDL and can be taken up directly by scavenger receptors on macrophages without oxidative modification, leading to the eventual formation of atherosclerotic plaques (22, 26–28). In addition, free fatty acids generated by triglyceride hydrolysis increase ROS production and induce mitochondrial dysfunction. Mitochondrial dysfunction impaired aerobic capacity, and increased ROS concentrations induce endothelial dysfunction/apoptosis and vascular smooth muscle cell proliferation/apoptosis, also leading to eventual atherosclerotic progression (29). Our patient had relatively low HDL levels, hypertriglyceridemia is usually accompanied by low HDL levels, making it challenging to analyze the effect of a single factor on atherosclerotic vascular events. Numerous Mendelian randomization analyses reveal that elevated levels of residual particulate cholesterol contribute to ischemic heart disease and suggest that HDL cholesterol may not be causally related to the risk of atherosclerotic vascular events (30–32). One study reported that the risk of coronary heart disease due to hypertriglyceridemia was attenuated or even eliminated after adjusting for other risk factors (33). Their data adjustment seems to be biased. They excluded HDL cholesterol and non-HDL cholesterol as confounding factors, whereas non-HDL cholesterol is primarily composed of TG-rich chylomicron remnants and VLDL remnants, with which triglycerides are inextricably linked. This study confirms from another perspective that TG indirectly, but not directly, promotes atheromatous plaque formation.
In the past, lipid disorder was only regarded as a secondary phenomenon of alcoholic or gallstone pancreatitis (34). However, high triglyceride levels are now a risk factor for developing pancreatitis (35). In patients with dyslipidemia, those with acute pancreatitis had higher maximal triglyceride levels than those without pancreatitis. However, there was no correlation between the level of triglycerides and the degree of pancreatitis (36). Controlling triglyceride levels could effectively reduce the risk of recurrence of pancreatitis (37). Moreover, because examination on admission to the hospital is later than the onset of the disease, the initial triglyceride level will be much higher than the laboratory values. Obtaining serum TG levels close to the onset of pain during the initial or recurrent episodes of acute pancreatitis is essential to identifying patients with HTG-induced acute pancreatitis (12). Triglyceride levels should be monitored as early as possible and daily at the onset of acute pancreatitis to avoid unnecessary missed diagnoses or misdiagnoses. Serum triglyceride levels in the first three days of onset are helpful for accurately diagnosing hypertriglyceridemia-induced pancreatitis (14). Triglyceride concentrations > 1000 mg/dl are generally considered to increase the risk of pancreatitis. The risk is even more significant in individuals with very severe hypertriglyceridemia (triglycerides >2000 mg/dl) (12).
Plasmapheresis can rapidly reduce the levels of chylomicrons and triglycerides in the serum. Several case reports and clinical guidelines demonstrated the feasibility of plasmapheresis for the treatment of patients with severe or deteriorating hypertriglyceridemia combined with complications (38–40). Long-term, regular dual filtration plasmapheresis can benefit patients with hypertriglyceridemia and related recurrent complications who are not well treated with conventional dietary therapy and medications (41) Dual filtration plasmapheresis is a technical innovation with high safety, broad applicability, low nutrient loss and low plasma usage. The initial lipid-lowering treatment in our case used this approach. By the way, measuring a patient’s total cholesterol level may predict the efficacy of triglyceride-lowering therapy (42). However, plasmapheresis is expensive and is not without risks. It requires the establishment of intravenous access and is an invasive treatment. It may develop complications related to bacteremia, deep vein thrombosis, and bleeding. It has been shown that there is no significant difference in morbidity and mortality whether patients are treated with plasmapheresis or non-invasive conservative treatment (43). Other studies revealed no difference in triglyceride reduction rates or clinical outcomes between patients who did or did not receive plasmapheresis (44, 45). These observational studies described above could not control for all possible confounding factors. Therefore, we need more rigorous randomized controlled trials to explore the value of plasmapheresis.
CVVH is a renal replacement therapy, a hemodialysis technique used to correct metabolic disorders and remove inflammatory mediators and toxic substances from patients in critical care (46). Successful treatment of hyperlipidemia-associated acute pancreatitis by CVVH has been reported (47). Another case reported hyperlipidemia and other serious side effects in children induced by propofol, a sedative drug, and CVVH resolved these abnormalities (48). A randomized controlled trial suggested that a subgroup classification of patients was needed to discuss the clinical benefits of CVVH for them. The efficacy of CVVH treatment is significant in patients with intra-abdominal pressure ≥20 mm Hg. However, the efficacy of CVVH in patients with intra-abdominal pressure <20 mm Hg is unclear (49). Serum IL-17 is somewhat predictive of the clinical benefit of CVVH in patients with severe pancreatitis (50).
Various combinations of blood purification techniques are commonly used in clinical practice. Hemoperfusion combined with CVVH significantly reduces triglyceride levels and removes excess inflammatory factors from the internal environment, significantly improving the prognosis of patients (51). In patients with hyperlipidemic severe acute pancreatitis, a prospective controlled study showed that hemoperfusion plus high-volume hemofiltration significantly improved serological parameters and clinical manifestations (52). The treatment, in our case, draws on their experience. In addition, an observational study reported that the use of plasma exchange combined with CVVH, a sequential hemodialysis modality, was effective in treating patients with hyperlipidemia-induced acute pancreatitis in a critical care setting (53).
We can use some medications to lower triglyceride levels, such as fibrates, nicotinic acid, and omega-3 fatty acids/fish oil (12, 54, 55), and drug combinations may be more beneficial than single drugs. Previous studies have reported the occurrence of rhabdomyolysis, a malignant event caused by a combination of fibrates and statins (56). In the latest study, the safety profile of fenofibrate in combination with a statin was acceptable, and this combination significantly reduced the risk of vascular events in patients with typical diabetic dyslipidemia (57). In recent years, researchers have made significant progress in developing targeted therapies, such as volanesorsen (58, 59), evinacumab (60, 61), lomitapide (62) and icosapent ethyl (63).
Diabetes is one of the most common acquired factors causing hypertriglyceridemia. Diabetes can independently increase a patient’s risk of developing acute pancreatitis after controlling for other risk factors (64). Insulin effectively increases the peripheral activity of lipoprotein lipase while controlling blood glucose and reverses the effects of insulin resistance on the liver (12).
It has been shown that continuous intravenous administration of heparin to patients leads to a decrease in lipoprotein lipase activity, which may ultimately lead to the accumulation of chylomicrons in the circulation (65). Therefore, the use of heparin is controversial. More experiments are needed to investigate whether it can be used or find the recommended dose.
It is well documented that chronic heavy alcohol consumption leads to a significant increase in triglyceride levels (66). Abstinence from alcohol and sugar, a low-fat diet, and enhanced physical exercise have been written into the treatment algorithm of an Australian hospital (67). Monitoring for distant complications associated with this acute event, such as chronic pancreatitis or pancreatic pseudocysts, will continue to be required (68), so regular follow-up is advisable.
During our literature search, we unexpectedly found a variety of prescribed medications that can cause a spike in triglyceride levels in patients, which must be taken into account by clinicians. The relevant drugs are summarized in the table (Table 2).
This case has many limitations in the diagnostic work-up and treatment options for the patient’s neurological symptoms. We lacked some outcome measures that are important for the prognosis of stroke patients. Before the patient was transferred to our hospital, cerebral angiography, magnetic resonance angiography and transcranial Doppler could not be performed due to the local hospital’s lack of medical equipment and the patient’s financial constraints. After admission, the patient’s neurological symptoms were stable, while the abdominal symptoms were severe. On the one hand, we were unable to wean the patient off the ventilator, and on the other hand, the patient had limited economic conditions. So we did not perform further magnetic resonance spectrum, we performed general care for the patient after thrombolysis and focused our treatment on the management of severe acute pancreatitis.
In addition to LDL, the main driver of atherosclerosis, high levels of triglycerides also need to be taken seriously by the public for their potential risk of severe vascular events. The diagnosis of dyslipidemia at the beginning of the disease is crucial. It is also important to use blood purification techniques to reduce triglyceride levels as early as possible. Multiple coupled hemodialysis techniques may be superior to single techniques. Genetic analysis can be performed for those patients with poor response to conventional lipid-lowering therapy, patients with familial dyslipidemia, or patients with recurrent hyperlipidemic complications. Once genetic defects are found, we can apply biological agents to target the disease. Hospitalized patients often have multiple comorbidities, and physicians must be aware of the side effects of different medications when prescribing.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
MZ, TY and FX contributed to the conception and design of the manuscript. SX, WZ, YZ, XH and KZ searched databases for information on the hypertriglyceridemia. MZ, LF and RD wrote the first draft of the manuscript. DT, YY and JL revised the primary manuscript. YY and JL provided funds for open access. All authors contributed to the article and approved the submitted version.
This work is supported by the National Natural Science Foundation of China NO:81900476(YY), No. 81672392 (JL); the National Key R&D Program of China (2021YFC2600203).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Hegele RA, Ginsberg HN, Chapman MJ, Nordestgaard BG, Kuivenhoven JA, Averna M, et al. The polygenic nature of hypertriglyceridaemia: Implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol (2014) 2:655–66. doi: 10.1016/S2213-8587(13)70191-8
2. Sung LC, Chang CC, Lin CS, Yeh CC, Cherng YG, Chen TL, et al. Risk of acute atherosclerotic cardiovascular disease in patients with acute and chronic pancreatitis. Sci Rep (2021) 11:20907. doi: 10.1038/s41598-021-99915-4
3. Parhofer KG, Chapman MJ, Nordestgaard BG. Efficacy and safety of icosapent ethyl in hypertriglyceridaemia: A recap. Eur Heart J Suppl (2020) 22:J21–33. doi: 10.1093/eurheartj/suaa116
4. Zafrir B, Jubran A, Hijazi R, Shapira C. Clinical features and outcomes of severe, very severe, and extreme hypertriglyceridemia in a regional health service. J Clin Lipidol (2018) 12:928–36. doi: 10.1016/j.jacl.2018.03.086
5. Ota K, Oniki A, Kobayashi Z, Ishihara S, Tomimitsu H, Shintani S. Acute pancreatitis is a very rare comorbidity of acute ischemic stroke. J Rural Med (2018) 13:72–5. doi: 10.2185/jrm.2956
6. Vinod KV, Verma SP, Karthikeyan B, Kishore A, Dutta TK. Cerebral infarction leading to hemiplegia: A rare complication of acute pancreatitis. Indian J Crit Care Med (2013) 17:308–10. doi: 10.4103/0972-5229.120325
7. Bhalla A, Sachdev A, Lehl SS, Singh R, D'Cruz S. Cerebral fat embolism as a rare possible complication of traumatic pancreatitis. JOP (2003) 4:155–7.
8. Ludwig DR, Austin MJ, Wallace AN, Kamran M, Kansagra AP, Osbun JW, et al. Isolated internal carotid artery thrombus and cerebral infarction in a patient with necrotizing pancreatitis: Case report. J Stroke Cerebrovasc Dis (2018) 27:e1–4. doi: 10.1016/j.jstrokecerebrovasdis.2017.08.012
9. Muroi J, Yorifuji T, Uematsu A, Nakahata T. Cerebral infarction and pancreatitis: Possible complications of patients with 3-hydroxy-3-methylglutaryl-CoA lyase deficiency. J Inherit Metab Dis (2000) 23:636–7. doi: 10.1023/a:1005642316174
10. King P, Smith PJ, Betteridge J, Brown M. 'A lipaemic mystery': A patient with hypertriglyceridaemic pancreatitis and cerebral infarction. BMJ Case Rep (2011) 2011:bcr0920114819. doi: 10.1136/bcr.09.2011.4819
11. Wysocki A, Panek J. [Unusual complication in the course of acute pancreatitis]. Przegl Lek (1999) 56:537–8.
12. Scherer J, Singh VP, Pitchumoni CS, Yadav D. Issues in hypertriglyceridemic pancreatitis: An update. J Clin Gastroenterol (2014) 48:195–203. doi: 10.1097/01.mcg.0000436438.60145.5a
13. Fortson MR, Freedman SN, Webster PD 3rd. Clinical assessment of hyperlipidemic pancreatitis. Am J Gastroenterol (1995) 90:2134–9.
14. Yin G, Cang X, Yu G, Hu G, Ni J, Xiong J, et al. Different clinical presentations of hyperlipidemic acute pancreatitis: A retrospective study. Pancreas (2015) 44:1105–10. doi: 10.1097/MPA.0000000000000403
15. Havel RJ. Pathogenesis, differentiation and management of hypertriglyceridemia. Adv Intern Med (1969) 15:117–54.
16. de Pretis N, Amodio A, Frulloni L. Hypertriglyceridemic pancreatitis: Epidemiology, pathophysiology and clinical management. United Eur Gastroenterol J (2018) 6:649–55. doi: 10.1177/2050640618755002
17. Valdivielso P, Ramirez-Bueno A, Ewald N. Current knowledge of hypertriglyceridemic pancreatitis. Eur J Intern Med (2014) 25:689–94. doi: 10.1016/j.ejim.2014.08.008
18. Nordestgaard BG. Triglyceride-rich lipoproteins and atherosclerotic cardiovascular disease: New insights from epidemiology, genetics, and biology. Circ Res (2016) 118:547–63. doi: 10.1161/CIRCRESAHA.115.306249
19. Chan P, Huang TY, Shieh SM, Lin TS, Tsai CW. Thrombophilia in patients with hypertriglyceridemia. J Thromb Thrombolysis (1997) 4:425–9. doi: 10.1023/a:1008857618659
20. Rosenson RS, Shott S, Lu L, Tangney CC. Hypertriglyceridemia and other factors associated with plasma viscosity. Am J Med (2001) 110:488–92. doi: 10.1016/s0002-9343(01)00643-x
21. Dron JS, Hegele RA. Genetics of triglycerides and the risk of atherosclerosis. Curr Atheroscler Rep (2017) 19:31. doi: 10.1007/s11883-017-0667-9
22. Sandesara PB, Virani SS, Fazio S, Shapiro MD. The forgotten lipids: Triglycerides, remnant cholesterol, and atherosclerotic cardiovascular disease risk. Endocr Rev (2019) 40:537–57. doi: 10.1210/er.2018-00184
23. Hegele RA. Multidimensional regulation of lipoprotein lipase: Impact on biochemical and cardiovascular phenotypes. J Lipid Res (2016) 57:1601–7. doi: 10.1194/jlr.C070946
24. Boren J, Matikainen N, Adiels M, Taskinen MR. Postprandial hypertriglyceridemia as a coronary risk factor. Clin Chim Acta (2014) 431:131–42. doi: 10.1016/j.cca.2014.01.015
25. Shaikh M, Wootton R, Nordestgaard BG, Baskerville P, Lumley JS, La Ville AE, et al. Quantitative studies of transfer in vivo of low density, sf 12-60, and sf 60-400 lipoproteins between plasma and arterial intima in humans. Arterioscler Thromb (1991) 11:569–77. doi: 10.1161/01.atv.11.3.569
26. Boren J, Taskinen MR, Olofsson SO, Levin M. Ectopic lipid storage and insulin resistance: A harmful relationship. J Intern Med (2013) 274:25–40. doi: 10.1111/joim.12071
27. Nordestgaard BG, Varbo A. Triglycerides and cardiovascular disease. Lancet (2014) 384:626–35. doi: 10.1016/S0140-6736(14)61177-6
28. Nakajima K, Nakano T, Tanaka A. The oxidative modification hypothesis of atherosclerosis: The comparison of atherogenic effects on oxidized LDL and remnant lipoproteins in plasma. Clin Chim Acta (2006) 367:36–47. doi: 10.1016/j.cca.2005.12.013
29. Madamanchi NR, Runge MS. Mitochondrial dysfunction in atherosclerosis. Circ Res (2007) 100:460–73. doi: 10.1161/01.RES.0000258450.44413.96
30. Varbo A, Benn M, Tybjaerg-Hansen A, Jorgensen AB, Frikke-Schmidt R, Nordestgaard BG. Remnant cholesterol as a causal risk factor for ischemic heart disease. J Am Coll Cardiol (2013) 61:427–36. doi: 10.1016/j.jacc.2012.08.1026
31. Musunuru K, Kathiresan S. Surprises from genetic analyses of lipid risk factors for atherosclerosis. Circ Res (2016) 118:579–85. doi: 10.1161/CIRCRESAHA.115.306398
32. Voight BF, Peloso GM, Orho-Melander M, Frikke-Schmidt R, Barbalic M, Jensen MK, et al. Plasma HDL cholesterol and risk of myocardial infarction: A mendelian randomisation study. Lancet (2012) 380:572–80. doi: 10.1016/S0140-6736(12)60312-2
33. Neil HA, Cooper J, Betteridge DJ, Capps N, McDowell IF, Durrington PN, et al. All-cause and cardiovascular mortality in treated patients with severe hypertriglyceridaemia: A long-term prospective registry study. Atherosclerosis (2010) 211:618–23. doi: 10.1016/j.atherosclerosis.2010.03.006
34. Haber PS, Wilson JS, Apte MV, Hall W, Goumas K, Pirola RC. Lipid intolerance does not account for susceptibility to alcoholic and gallstone pancreatitis. Gastroenterol (1994) 106:742–8. doi: 10.1016/0016-5085(94)90710-2
35. Carr RA, Rejowski BJ, Cote GA, Pitt HA, Zyromski NJ. Systematic review of hypertriglyceridemia-induced acute pancreatitis: A more virulent etiology? Pancreatology (2016) 16:469–76. doi: 10.1016/j.pan.2016.02.011
36. Lloret Linares C, Pelletier AL, Czernichow S, Vergnaud AC, Bonnefont-Rousselot D, Levy P, et al. Acute pancreatitis in a cohort of 129 patients referred for severe hypertriglyceridemia. Pancreas (2008) 37:13–2. doi: 10.1097/MPA.0b013e31816074a1
37. Athyros VG, Giouleme OI, Nikolaidis NL, Vasiliadis TV, Bouloukos VI, Kontopoulos AG, et al. Long-term follow-up of patients with acute hypertriglyceridemia-induced pancreatitis. J Clin Gastroenterol (2002) 34:472–5. doi: 10.1097/00004836-200204000-00020
38. Joury A, Alshehri M, Mahendra A, Anteet M, Yousef MA, Khan AM. Therapeutic approaches in hypertriglyceridemia-induced acute pancreatitis: A literature review of available therapies and case series. J Clin Apher (2020) 35:131–7. doi: 10.1002/jca.21763
39. Stefanutti C, Di Giacomo S, Labbadia G. Timing clinical events in the treatment of pancreatitis and hypertriglyceridemia with therapeutic plasmapheresis. Transfus Apher Sci (2011) 45:3–7. doi: 10.1016/j.transci.2011.06.013
40. Padmanabhan A, Connelly-Smith L, Aqui N, Balogun RA, Klingel R, Meyer E, et al. Guidelines on the use of therapeutic apheresis in clinical practice - evidence-based approach from the writing committee of the American society for apheresis: The eighth special issue. J Clin Apher (2019) 34:171–354. doi: 10.1002/jca.21705
41. Grupp C, Beckermann J, Koster E, Zewinger S, Knittel M, Walek T, et al. Relapsing and progressive complications of severe hypertriglyceridemia: Effective long-term treatment with double filtration plasmapheresis. Blood Purif (2020) 49:457–67. doi: 10.1159/000506506
42. Chen Z, Huang X, Han N, Guo Y, Chen J, Ning Y, et al. Total cholesterol concentration predicts the effect of plasmapheresis on hypertriglyceridemic acute pancreatitis: A retrospective case-control study. BMC Gastroenterol (2021) 21:3. doi: 10.1186/s12876-020-01572-w
43. Nakhoda S, Zimrin AB, Baer MR, Law JY. Use of the APACHE II score to assess impact of therapeutic plasma exchange for critically ill patients with hypertriglyceride-induced pancreatitis. Transfus Apher Sci (2017) 56:123–6. doi: 10.1016/j.transci.2016.10.005
44. Miyamoto K, Horibe M, Sanui M, Sasaki M, Sugiyama D, Kato S, et al. Plasmapheresis therapy has no triglyceride-lowering effect in patients with hypertriglyceridemic pancreatitis. Intensive Care Med (2017) 43:949–51. doi: 10.1007/s00134-017-4722-3
45. Berberich AJ, Ziada A, Zou GY, Hegele RA. Conservative management in hypertriglyceridemia-associated pancreatitis. J Intern Med (2019) 286:644–50. doi: 10.1111/joim.12925
46. Cole L, Bellomo R, Hart G, Journois D, Davenport P, Tipping P, et al. A phase II randomized, controlled trial of continuous hemofiltration in sepsis. Crit Care Med (2002) 30:100–6. doi: 10.1097/00003246-200201000-00016
47. Batashki I, Milchev N, Uchikova E, Pekhlivanov B, Chifligarov K, Uchikov A. [Hyperlipidemic pancreatitis during pregnancy–a case report]. Akush Ginekol (Sofiia) (2006) 45 Suppl 1:41–3.
48. Cray SH, Robinson BH, Cox PN. Lactic acidemia and bradyarrhythmia in a child sedated with propofol. Crit Care Med (1998) 26:2087–92. doi: 10.1097/00003246-199812000-00046
49. Xie Y, Yuan Y, Su W, Qing N, Xin H, Wang X, et al. Effect of continuous hemofiltration on severe acute pancreatitis with different intra-abdominal pressure: A cohort study. Med (Baltimore) (2021) 100:e27641. doi: 10.1097/MD.0000000000027641
50. Dai SR, Li Z, Zhang JB. Serum interleukin 17 as an early prognostic biomarker of severe acute pancreatitis receiving continuous blood purification. Int J Artif Organs (2015) 38:192–8. doi: 10.5301/ijao.5000406
51. Li MQ, Shi ZX, Xu JY, Lu B, Li JQ, Xu YJ, et al. Hemodiafiltration combined with resin-mediated absorption as a therapy for hyperlipidemic acute pancreatitis. Cell Biochem Biophys (2014) 69:699–702. doi: 10.1007/s12013-014-9855-1
52. Sun S, He L, Bai M, Liu H, Li Y, Li L, et al. High-volume hemofiltration plus hemoperfusion for hyperlipidemic severe acute pancreatitis: A controlled pilot study. Ann Saudi Med (2015) 35:352–8. doi: 10.5144/0256-4947.2015.352
53. Wang HL, Yu KJ. Sequential blood purification therapy for critical patients with hyperlipidemic severe acute pancreatitis. World J Gastroenterol (2015) 21:6304–9. doi: 10.3748/wjg.v21.i20.6304
54. Brahm AJ, Hegele RA. Chylomicronaemia–current diagnosis and future therapies. Nat Rev Endocrinol (2015) 11:352–62. doi: 10.1038/nrendo.2015.26
55. Hauenschild A, Bretzel RG, Schnell-Kretschmer H, Kloer HU, Hardt PD, Ewald N. Successful treatment of severe hypertriglyceridemia with a formula diet rich in omega-3 fatty acids and medium-chain triglycerides. Ann Nutr Metab (2010) 56:170–5. doi: 10.1159/000283561
56. Amend KL, Landon J, Thyagarajan V, Niemcryk S, McAfee A. Incidence of hospitalized rhabdomyolysis with statin and fibrate use in an insured US population. Ann Pharmacother (2011) 45:1230–9. doi: 10.1345/aph.1Q110
57. Elam MB, Ginsberg HN, Lovato LC, Corson M, Largay J, Leiter LA, et al. Association of fenofibrate therapy with long-term cardiovascular risk in statin-treated patients with type 2 diabetes. JAMA Cardiol (2017) 2:370–80. doi: 10.1001/jamacardio.2016.4828
58. Yang X, Lee SR, Choi YS, Alexander VJ, Digenio A, Yang Q, et al. Reduction in lipoprotein-associated apoC-III levels following volanesorsen therapy: Phase 2 randomized trial results. J Lipid Res (2016) 57:706–13. doi: 10.1194/jlr.M066399
59. Kolovou G, Kolovou V, Katsiki N. Volanesorsen: A new era in the treatment of severe hypertriglyceridemia. J Clin Med (2022) 11:982. doi: 10.3390/jcm11040982
60. Ling P, Zheng X, Luo S, Ge J, Xu S, Weng J. Targeting angiopoietin-like 3 in atherosclerosis: From bench to bedside. Diabetes Obes Metab (2021) 23:2020–34. doi: 10.1111/dom.14450
61. Ahmad Z, Banerjee P, Hamon S, Chan KC, Bouzelmat A, Sasiela WJ, et al. Inhibition of angiopoietin-like protein 3 with a monoclonal antibody reduces triglycerides in hypertriglyceridemia. Circulation (2019) 140:470–86. doi: 10.1161/CIRCULATIONAHA.118.039107
62. Brahm AJ, Hegele RA. Lomitapide for the treatment of hypertriglyceridemia. Expert Opin Investig Drugs (2016) 25:1457–63. doi: 10.1080/13543784.2016.1254187
63. Bhatt DL, Steg PG, Miller M, Brinton EA, Jacobson TA, Ketchum SB, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med (2019) 380:11–22. doi: 10.1056/NEJMoa1812792
64. Lai SW, Muo CH, Liao KF, Sung FC, Chen PC. Risk of acute pancreatitis in type 2 diabetes and risk reduction on anti-diabetic drugs: A population-based cohort study in Taiwan. Am J Gastroenterol (2011) 106:1697–704. doi: 10.1038/ajg.2011.155
65. Weintraub M, Rassin T, Eisenberg S, Ringel Y, Grosskopf I, Iaina A, et al. Continuous intravenous heparin administration in humans causes a decrease in serum lipolytic activity and accumulation of chylomicrons in circulation. J Lipid Res (1994) 35:229–38. doi: 10.1016/S0022-2275(20)41211-8
66. Van de Wiel A. The effect of alcohol on postprandial and fasting triglycerides. Int J Vasc Med (2012) 2012:862504. doi: 10.1155/2012/862504
67. Tan HLE, McDonald G, Payne A, Yu W, Ismadi Z, Tran H, et al. Incidence and management of hypertriglyceridemia-associated acute pancreatitis: A prospective case series in a single Australian tertiary centre. J Clin Med (2020) 9:3954. doi: 10.3390/jcm9123954
68. Sisman G, Erzin Y, Hatemi I, Caglar E, Boga S, Singh V, et al. Familial chylomicronemia syndrome related chronic pancreatitis: A single-center study. Hepatobiliary Pancreat Dis Int (2014) 13:209–14. doi: 10.1016/s1499-3872(14)60033-3
69. Sakhri J, Ben Salem C, Harbi H, Fathallah N, Ltaief R. Severe acute pancreatitis due to tamoxifen-induced hypertriglyceridemia with positive rechallenge. JOP (2010) 11:382–4.
70. Yoneyama K, Nakagawa M. Severe acute pancreatitis due to tamoxifen-induced hypertriglyceridemia. Breast J (2019) 25:788–9. doi: 10.1111/tbj.13279
71. Hashmi A, Smith EI, Ciutac A, Smith JC. Lesson of the month: Acute pancreatitis due to hypertriglyceridaemia in a transgender woman: A complication of high-dose oral oestrogen therapy? Clin Med (Lond) (2021) 21:228–30. doi: 10.7861/clinmed.2021-0075
72. Shipley LC, Steele DT, Wilcox CM, Burski CM. A rare cause of acute pancreatitis in a transgender female. J Investig Med High Impact Case Rep (2020) 8:2324709620921333. doi: 10.1177/2324709620921333
73. Kim DS, O'Hayer PJ, Rubenfire M, Brook RD. Hypertriglyceridaemia-induced pancreatitis prompted by acute corticosteroid treatment: Caution for clinicians. Intern Med J (2019) 49:411–2. doi: 10.1111/imj.14228
74. Ashraf M. Acute pancreatitis caused by isotretinoin. Cureus (2020) 12:e8710. doi: 10.7759/cureus.8710
75. Atiq MU, Raza A, Ashfaq A. Idiosyncratic reaction causing a rare side effect: Isotretenoin-induced pancreatitis. Cureus (2019) 11:e6102. doi: 10.7759/cureus.6102
76. Sahu KK, Lal A, Mishra AK, Zhang P. Adalimumab-related hypertriglyceridemia and acute pancreatitis. QJM (2020) 113:298–9. doi: 10.1093/qjmed/hcz267
77. Leung J, Brady JL, Crook MA. The clinical importance of recognizing capecitabine-induced hypertriglyceridemia: A case report and review of the literature. J Clin Lipidol (2018) 12:1371–3. doi: 10.1016/j.jacl.2018.07.005
78. Maradana S, Akella P, Nalluru SS, Jindal V, Siddiqui AD. Hypertriglyceridemia induced pancreatitis due to brentuximab therapy: First case report. Cureus (2019) 11:e5138. doi: 10.7759/cureus.5138
79. Durrington PN, Cairns SA. Acute-pancreatitis: A complication of beta-blockade. Br Med J (Clin Res Ed) (1982) 284:1016. doi: 10.1136/bmj.284.6321.1016
Keywords: hypertriglyceridemia, stroke, pancreatitis, metabolic disorders, blood purification
Citation: Zhang M, Yin T, Xia F, Xia S, Zhou W, Zhang Y, Han X, Zhao K, Feng L, Dong R, Tian D, Yu Y and Liao J (2022) Hypertriglyceridemia may contribute to stroke and pancreatitis: A case report and review of the literature. Front. Endocrinol. 13:960343. doi: 10.3389/fendo.2022.960343
Received: 02 June 2022; Accepted: 17 November 2022;
Published: 01 December 2022.
Edited by:
Hwi Seung Kim, Chung-Ang University Gwangmyeong Hospital, South KoreaReviewed by:
Naishi Li, Peking Union Medical College Hospital (CAMS), ChinaCopyright © 2022 Zhang, Yin, Xia, Xia, Zhou, Zhang, Han, Zhao, Feng, Dong, Tian, Yu and Liao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yan Yu, d2h0ajIwMjAwNzIwQDE2My5jb20=; Jiazhi Liao, bGlhb2ppYXpoaUB0amgudGptdS5lZHUuY24=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.