
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Public Health , 16 September 2022
Sec. Infectious Diseases: Epidemiology and Prevention
Volume 10 - 2022 | https://doi.org/10.3389/fpubh.2022.978052
Purpose: Investigation of the community-level symptomatic onset risk regarding severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variants of concern, is crucial to the pandemic control in the new normal.
Methods: Investigated in this study is the spatiotemporal symptom onset risk with Omicron BA.1, BA.2, and hamster-related Delta AY.127 by a joint analysis of community-based human mobility, virus genomes, and vaccinations in Hong Kong.
Results: The spatial spread of Omicron BA.2 was found to be 2.91 times and 2.56 times faster than that of Omicron BA.1 and Delta AY.127. Identified has been an early spatial invasion process in which spatiotemporal symptom onset risk was associated with intercommunity and cross-community human mobility of a dominant source location, especially regarding enhancement of the effects of the increased intrinsic transmissibility of Omicron BA.2. Further explored is the spread of Omicron BA.1, BA.2, and Delta AY.127 under different full and booster vaccination rate levels. An increase in full vaccination rates has primarily contributed to the reduction in areas within lower onset risk. An increase in the booster vaccination rate can promote a reduction in those areas within higher onset risk.
Conclusions: This study has provided a comprehensive investigation concerning the spatiotemporal symptom onset risk of Omicron BA.1, BA.2, and hamster-related Delta AY.127, and as such can contribute some help to countries and regions regarding the prevention of the emergence of such as these variants, on a strategic basis. Moreover, this study provides scientifically derived findings on the impact of full and booster vaccination campaigns working in the area of the reduction of symptomatic infections.
Designated as a variant of concern by the World Health Organization (WHO) on November 26th 2021 (1), the Omicron variant has become the dominant variant circulating globally (2). Currently, the sub-variants of Omicron are mainly divided into five groups: BA.1, BA.2, BA.3, BA.4, and BA.5 (3, 4). Of these, the BA.1 subvariant was initially the predominant of the global Omicron lineage, but the global proportion of COVID-19 cases, associated with the BA.2 variant had been increasing rapidly (5–8). Hong Kong had generally well controlled the attack of all former prominent COVID-19 variants, but resultantly the people had acquired little immunity from infections. During the global Omicron wave, Hong Kong became one of the global cities that suffered the most from BA.1 and BA.2 variants one after another (9). The BA.2 sub-group in Hong Kong, in particular, is unique when compared to variants, seen in the rest of the world due to unique mutations found in ORF1a: A2909V and ORF3a: L140F (10). It is also of note that, at the same time, an unusual cluster of probable hamster-to-human SARS-CoV-2 transmission due to the SARS-CoV-2 AY.127 variant has appeared in Hong Kong (11). Hong Kong's COVID-19 case fatality rate was one of the highest in the world (12), at the peak of the previous wave of outbreaks. Faced with a surge in cases during the previous epidemic wave, Hong Kong and other regions, fearing a more severe development of the disease, focused on prevention, and the early investigation of symptomatic cases (13). This is, now, currently the aim and practice of other countries and regions of the world, which now have likewise, rapidly aimed to prevent the worsening cases and deaths regarding the latest epidemic wave of COVID-19 (14, 15). To achieve early detection and treatment of symptomatic cases, it is necessary to effectively monitor and predict the risk level of symptomatic cases at the community level, the latter now becoming the focus of current epidemic prevention work in various countries (16–18).
Thus, in order to support more effective control of the spread of SARS-CoV-2 sub-lineages in such as Hong Kong and other countries, it is worthwhile to effectively predict and understand the spatiotemporal symptom onset risk for Omicron BA.1, BA.2, and hamster-related Delta AY.127 during the early spread stages. More importantly, the impact of COVID-19 vaccination, at the time of spatiotemporal symptom onset risk with Omicron BA.1, BA.2, and Delta AY.127 would provide benefits, if further explored (19).
Omicron BA.1 was detected for the first time, in virus gene sequencing samples from local airline pilots in Hong Kong as early as December 28th, 2021 (20). After that, BA.1 spread rapidly within the territory and, in fact, became a dominant variant until mid-January 2022. On January 16th, 2022, BA.2 was first detected in a virus sample from a local case entering the community after being infected during quarantine. It replaced BA.1 as the current dominant virus strain in Hong Kong, for a very short period, as the current dominant virus strain in Hong Kong. During this period, beginning on January 17th, 2022, the Delta SARS-CoV-2 AY.127 variants were continuously detected in virus samples from a cohort of employees and customers of a hamster pet store (21). During the early stages of the spread of these variants in Hong Kong, multipronged measures were taken to increase the uptake and pace of the vaccination, in terms of the inactivated (Sinovac) or mRNA (BioNTech) COVID-19 vaccine (22). From mid-December 2021 to early February 2022, the full vaccination rate in Hong Kong was increased from 61.3 to 64.3%, and the booster vaccination rate was increased from 3.7 to 12.8% (23).
Thus, in order to understand the spatiotemporal spread of Omicron BA.1, BA.2, and Delta AY.127 in Hong Kong, firstly determined will be the SARS-CoV-2 sequences in these cases by whole-genome sequencing of respiratory specimens or deep throat saliva from the above cases, to determine the possible transmission linkage based on their phylogenetic relatedness (24–27). By the identified sequences and epidemiological link of these cases, the enhanced urban-community-level weighted kernel density estimation (WKDE) model (28–31) will then, be proposed to predict the spatiotemporal COVID-19 symptom onset risk of Omicron BA.1, BA.2, and Delta AY.127 in 291 Tertiary Planning Units (TPUs) of Hong Kong (Figure 1). (i) Locations with symptomatic cases resided/visited, (ii) locations with positive sewage testing results, (iii) time-varying vaccination rate and vaccination efficiency (6, 32–36) were incorporated to enhance the WKDE model. Based on the onset risk prediction results during the first 20 days, simulated were the early spatiotemporal spread of Omicron BA.1, BA.2, and Delta AY.127 under different scenarios and with different full and booster vaccination rate levels. The spatiotemporal data of the daily symptom onset cases in 291 TPUs of Hong Kong from December 26th, 2021, to February 4th, 2022, are utilized regarding the development of this study.
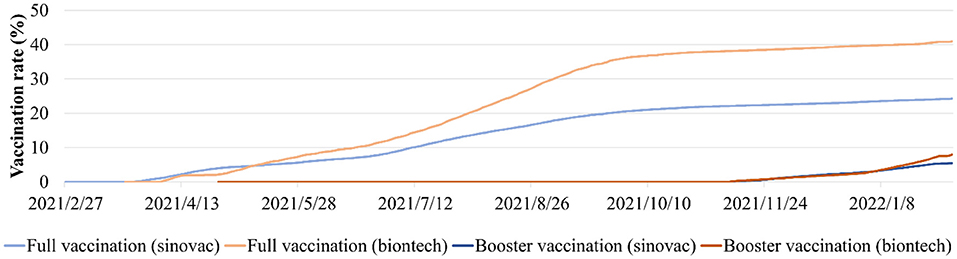
Figure 1. The daily full and booster variation rates of Sinovac and BioNTech vaccine in Hong Kong from February 22nd, 2021 to February 4th, 2022.
This current study, involved the full genome sequencing and thereby concerned respiratory specimens or deep throat saliva from laboratory-confirmed COVID-19 patients admitted to both the main hospital, and the temporary hospital adjacent to the community treatment facility in Hong Kong. To enable the whole viral genome sequencing of patients' specimens to be conducted, the PCR tiling of the SARS-CoV-2 virus with rapid barcoding protocol was used (Version: mrt_9127_v110_revH_14Jul2021) on Nanopore GridION MK1 (Oxford Nanopore Technologies) (37). Between Dec 2021 and Feb 2022, a total of 652 cases were reported in Hong Kong. Because of a high number of reported cases, only the symptomatic cases were admitted to hospitals for treatment, whereas asymptomatic cases were mostly quarantined at home. We recruited all the positive cases from five public hospitals during that period for whole viral genome analysis. A total of 573 cases was collected and sequenced, which account for 87.9% of the total cases during the study period. Throughout the whole-genome sequencing process, data on i) 34 local cases with Omicron BA.1, ii) 231 local cases with Omicron BA.2, and iii) 16 local cases with Delta AY.127 during the period from December 2021 to February 2022, along with spatiotemporal information, were used in this study. Transmission clusters were defined by clear epidemiological linkage and onset-time relationship (27). The detail of local cases and transmission clusters used in this study have been made public in the Next Strain dataset (38). In addition, location data collected from sewage sample(s) tested positive during the period from December 2021 to February 2022 has also been used in this study (39).
To quantify the daily human mobility effects on the COVID-19 epidemic in all 219 TPUs, daily traffic flow data covering all Hong Kong's strategic routes between and including the entirety of December 2021 to February 2022, were used in this study (40). The COVID-19 vaccines used in Hong Kong were the Sinovac Vaccines and BioNTech Vaccines. In order to measure the impact of full and booster vaccinations on the COVID-19 epidemic, Hong Kong's daily vaccination rates were used from February 22nd, 2021, to February 4th, 2022 (23) (Figure 1). The vaccination effectiveness of BioNTech and Sinovac against symptomatic diseases for Omicron and Delta was determined, based on previous studies (33, 41–43) (Supplementary Table 1). In addition, the daily COVID-19 effective reproductive number R was obtained from December 2021 to February 2022 from reports by The University of Hong Kong (44).
The SARS-CoV-2 has been found to have a high viral load and levels of transmissibility around the date of symptom onset (45, 46). Hence, it is necessary to adopt appropriate data-driven spatiotemporal models to dynamically and individually assess onset risk levels. Shi et al. developed an extended weighted kernel density estimation (WKDE) model (28–31). This model presents a retrospective analysis based on spatiotemporal information regarding onset cases, and presents an inference of the infection date of each onset case, and further infers the spatial distribution (i.e, the kernel density surface) of the infection risk to people by the onset cases at past dates, and finally to predict the distribution of onset risk at future dates (28–31). In addition, according to the transmission law of COVID-19 (i.e., mainly through direct, indirect, and close contact between people), data on the dynamic flow of people was introduced into the model to improve prediction accuracy (28–31). The improved model is an extended WKDE model (28–31).
As a further development of the original extended WKDE model, the urban-community-level WKDE model, proposed in this study, includes the following three steps (28–31):
a) Conducting a retrospective analysis of the historical existence likelihood of the infection in each community location in which an onset case had remained at the location and an onset case suffered by a visitor to the location;
b) Making inferences on the historical existence likelihood of the infection in the entire city;
c) Making predictions about the epidemic onset risk in the entire city on a given day in the near future.
The main difference between the urban-community-level WKDE model and the original extended WKDE model was that at step (b) of the model, the historical existence likelihood of an infection in a random location in the entire region was formulated as
where PInfection(S, ti) is the probability of any individual infected with COVID-19 infecting others in a random location, S, in the city on day ti; R(S, ti) denotes the COVID-19 effective reproductive number in the city on day ti; VE(S, ti) denotes the vaccine effectiveness against symptomatic diseases in the city on day ti (6, 32–36); VP(S, ti) is the proportion of the population who have been fully vaccinated in the city on day ti; Lj is the j-th location among the province places where the onset cases resided; PInfection(L, ti) denotes the probability that one onset case was infected on day ti in a location L; and Kh(S – Lj) denotes a Gaussian kernel between locations S and Lj(28–31). The values of PInfection (Lj, ti), Kh (S – Lj), and h were determined in earlier model procedures (28–31).
Mintra_TPU(S, ti) denotes a human mobility factor within a TPU containing location S on day ti, calculated as follows (28–31):
where Xk denotes the daily traffic flow within the TPU containing location S on day tk prior to ti.
MinterTPU(S, ti) denotes a human mobility factor from other TPUs to the TPU containing location S, calculated as follows (28–31):
where Yk denotes the daily traffic flow from other TPUs to the TPU containing location S on day tk prior to ti.
Finally, the predicted risk in each location was divided by the maximum predicted risk among all locations on a specific date and thereby standardized to a value between 0 and 1. Different levels of onset risk have been defined as follows: low onset risk (0–0.2), low-medium onset risk (0.2–0.4), medium onset risk (0.4–0.6), medium-high onset risk (0.6–0.8), and high onset risk (0.8–1). The reliability of the predicted COVID-19 onset risk was evaluated using its spatial significance, i.e., the percentage of symptom onset cases on a future date to be predicted that would occur in the areas with a predicted onset risk >0.8 (identified as onset hotspots) (28–31).
The whole genome sequencing results from, and including December 2021 to February 2022 have been used in this study. A total of 225 cases were identified as Omicron BA.1 from the period: December 2021 to January 2022. Of these cases, the majority (n = 191, 84.89%) were identified as imported cases and did not leak into the local community. Based on the phylogenetic analysis of the local cases, in late December 2021, two Omicron BA.1 strains were introduced to this community via aircrews exempt from quarantine. Each strain established discrete transmission chains: 8 local cases belonged to the Moon Palace cluster; 26 local cases belonged to the North Point cluster (Figure 2A). The locations of 34 local cases in these two clusters, are shown separately in Figure 2B.
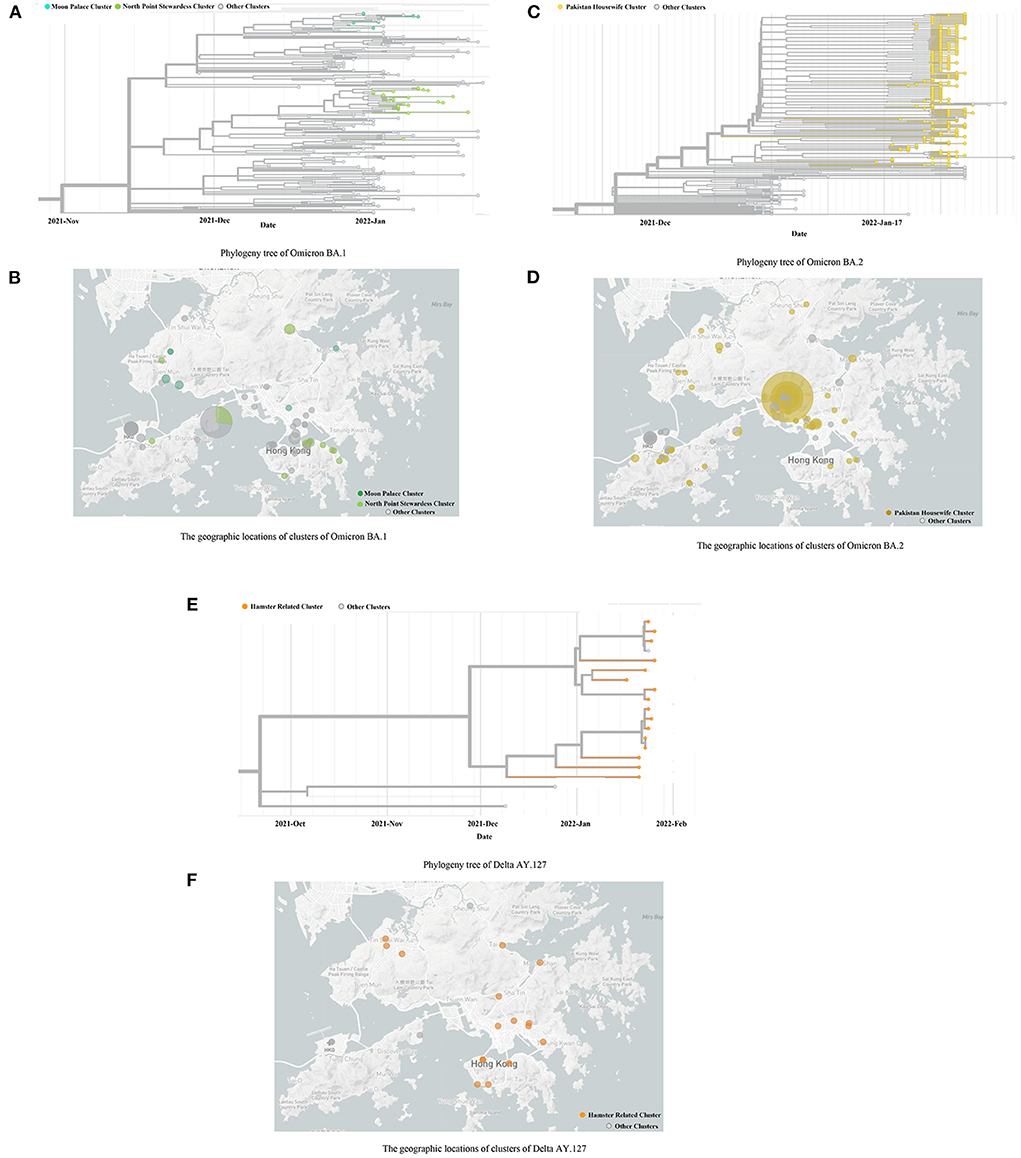
Figure 2. The Phylogeny trees and geographic locations of cases with Omicron BA.1, BA.2, and Delta AY.127. (A,B) indicates the Phylogeny trees and geographic locations of cases with Omicron BA.1. (C,D) indicates the Phylogeny trees and geographic locations of cases with Omicron BA.2. (E,F) indicates the Phylogeny trees and geographic locations of cases with Delta AY.127. (A) Phylogeny tree of Omicron BA.1. (B) The geographic locations of clusters of Omicron BA.1. (C) Phylogeny tree of Omicron BA.2. (D) The geographic locations of clusters of Omicron BA.2. (E) Phylogeny tree of Delta AY.127. (F) The geographic locations of clusters of Delta AY.127.
A total of 329 cases was identified as Omicron BA.2, in which 231 were local cases. The source was thought to be a Pakistan housewife infected by another returnee living in an adjacent room of the quarantine hotel. This particular woman further infected nine family members, following her completion of the compulsory quarantine period. The family cluster eventually sparked the largest scale of COVID-19 epidemic in Hong Kong (Figure 2C). The locations of the premises which suffered clusters of 329 cases are shown in Figure 2D.
In addition to the Omicron variant, a total of 16 local cases was genotyped as Delta AY.127 (Figure 2E). These cases were attributed as hamster-to-human transmission and occurred owing to evidence of the variant within pet shops (10). The locations of the premises visited by the 16 local cases are shown in Figure 2F.
The COVID-19 symptom onset risk in 291 TPUs in Hong Kong, during the early 20 days of the emergence and spread of Omicron BA.1, BA.2, Delta AY.127, was first predicted and further analyzed using the urban-community-level WKDE model. (i) 34 local Omicron BA.1 cases, (ii) 231 local Omicron BA.2 cases, and (iii) 16 local Delta AY.127 cases, with spatiotemporal information from Hong Kong during the period from December 26th, 2021, to February 4th, 2022, were used in the model. The prediction accuracy of the urban-community-level WKDE model was over 85% for symptom onset risk during the following seven days (28–31) (Figure 3). Such an ‘outlier-performance' should be attributed to the incorporation of (i) Locations with case resided/visited, (ii) locations with positive sewage testing results, (iii) time-varying vaccination rates, and vaccination efficiency.
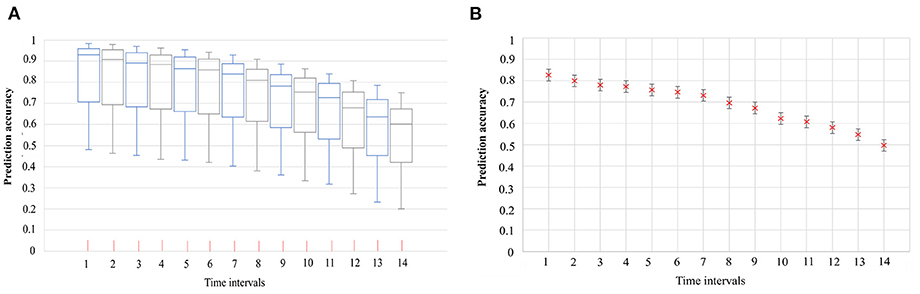
Figure 3. The accuracy of the predicted risk of COVID-19 symptom onset by urban-community-level WKDE models, and 95% confidence interval of prediction accuracy (28–31). (A) Accuracy of the predicted risk of COVID-19 symptom onset by urban-community-level WKDE models. The predicted onset risk is a normalized value of between 0 and 1, hence, indicating risk relative to the highest predicted risk among all locations, on the date for which the risk of symptom onset is predicted, hereafter termed “the prediction date”. The prediction accuracy is defined as the percentage of onset cases in those areas for which the predicted onset risk was higher than 0.8 on the prediction date. The time interval denotes the period between the base date (The base date refers to the date on which the onset risk for the next 14 days is predicted) and the date of prediction. The horizontal line in the box denotes the median, while the lower and upper edges of the box represent the respective first and third quartiles. The lines emanating from the box upwards and downwards represent the respective maximum and minimum values. (B) 95% confidence interval of the mean accuracy of the predicted risk of COVID-19 symptom onset by urban-community-level WKDE models.
The emergence and spread of Omicron BA.1, BA.2, and Delta AY.127 during the first 20 days after these sub-linages entered the community could be described by the spatiotemporal variation of the predicted risk of COVID-19 symptom onset and the intensity of intra-TPU mobility (i.e., the traffic flow within the community) and inter-TPU mobility (i.e., the traffic flow across communities) as follows (Figure 4). Starting from Omicron BA.1 entering the community in Hong Kong, it can be seen that the TPUs in Tuen Mun, in which the related cases first appeared, started to reach high and medium-high onset risk level. Since then, due to the high intra-TPU human mobility in Tuen Mun, the number of high-risk TPUs in Tuen Mun continued to expand (Figures 4, 5A). At the same time, Omicron BA.1 spread to other distant TPUs with a similar high human mobility to that in Tuen Mun, such as TPUs in North Point, Sham Shui Po, Tai Po, and Sha Tin (Figures 4, 5B,C). These TPUs also became or at high or medium-high onset risk (Figures 5B,C). By the 20th day after Omicron BA.1 entered the community, there were 53 TPUs at high onset risk (Figure 5D), and involving 2,191,586 people (Table 1). In addition, the areas around the above, with high population mobility were also at medium or medium-low onset risk (Figures 4, 5D).
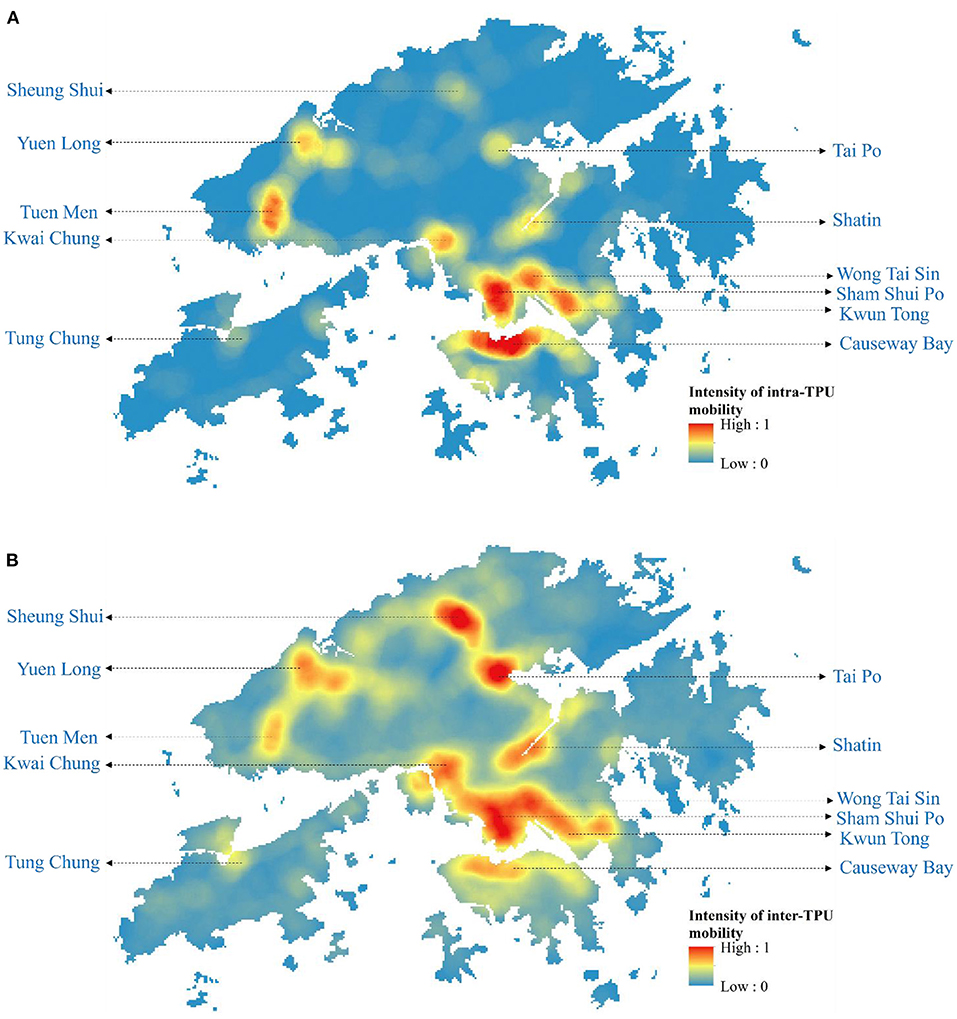
Figure 4. The intensity of intra-TPU and inter-TPU mobility of Hong Kong from December 2021 to February 2022. (A) The intensity of intra-TPU mobility of Hong Kong. (B) The intensity of inter-TPU mobility of Hong Kong.
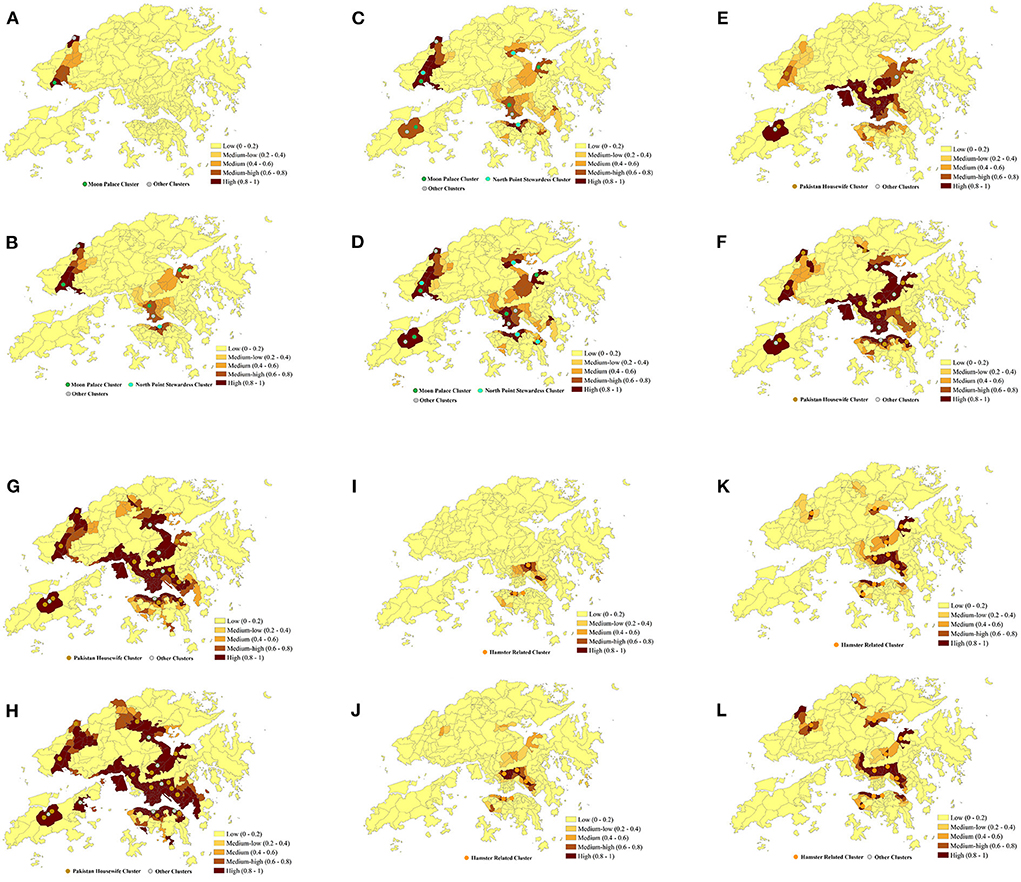
Figure 5. Predicted risk of COVID-19 symptom onset across 291 TPUs in Hong Kong within 20 days after Omicron BA.1, BA.2, and Delta AY.127 (A–L). The predicted COVID-19 symptoms onset risk was generated using the urban-community-level WKDE model. (A) 5th day after Omicron BA.1 entered the community. (B) 10th day after Omicron BA.1 entered the community. (C) 15th day after Omicron BA.1 entered the community. (D) 20th day after Omicron BA.1 entered the community. (E) 20th day after Omicron BA.2 entered the community. (F) 10th day after Omicron BA.2 entered the community. (G) 15th day after Omicron BA.2 entered the community. (H) 20th day after Omicron BA.2 entered the community. (I) 5th day after Delta AY.127 entered the community. (J) 10th day after Delta AY.127 entered the community. (K) 15th day after Delta AY.127 entered the community. (L) 20th day after Delta AY.127 entered the community.
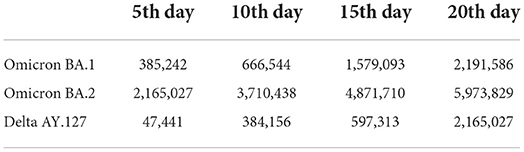
Table 1. Number of people living in high-onset-risk communities during the spread of Omicron BA.1, BA.2 and Delta AY.127 in Hong Kong.
Omicron BA.2 also spread from the TPUs in Kwai Chung to TPUs with high population mobility within the human mobility network (Figures 4, 5E–H). The speed of spatial spread of Omicron BA.2 was obviously faster than that of BA.1. When Omicron BA.2 entered the community on the 10th day, almost all TPUs of high human mobility were at high onset risk, such areas included Sham Shui Po, Tuen Mun, Shatin, Causeway Bay, Tai Po, Yuen Long, Sheung Shui (Figures 4, 5G). TPUs in high-risk areas were 7.83 times more likely than in the period of the BA.1 spread. In fact, 3,043,894 more people were at high-onset-risk TPUs (Table 1). By the 20th day after Omicron BA.2 entered the community, other TPUs around these high-mobility TPUs were founded to be also at high onset risk (Figure 5H), making a final total of 154 (Figure 5H). There were 3,782,243 more people in the high-risk area relative to the same period of the Omicron BA.1 spread (Table 1).
The spread speed of the hamster-related Delta AY.127, was slower than that of both Omicron BA.1 and BA.2, despite its spread throughout the human mobility network (Figures 5I–K). On the 20th day when Delta AY.127 entered the community, only 43 TPUs of high population mobility in Wong Tai Sin, Shatin, Kwun Tong, Causeway Bay were at high onset risk (Figure 5L). On the 20th day of the spread of Omicron BA.1 and BA.2, TPUs at high-onset-risk were respectively, 1.23 times and 3.55 times greater than that of Delta AY.127 during the same period. There were also 3,808,802 and 26,559 more people in high-onset-risk TPUs with the spread of Omicron BA.1 and BA.2 than with that with Delta AY.127 in the same period (Table 1). The impact of Delta AY.127 on those TPUs which surrounded these particular TPUs with high human mobility, appears to have been relatively limited.
In addition, during the 20 days of the spread of Omicron BA.1, BA.2, Delta AY.127, and according to the exploration of the correlation between the intensity of intra-TPU/inter-TPU mobility and the symptom onset risk (Table 2), it can be found that the intensity of population mobility within the community promotes spread of these variants to adjacent areas. For example, on the 20th day after the start of the Omicron BA.2 spread, the R2 between the intensity of intra-TPU mobility and the symptom onset risk reached 0.87, this being 0.0344 and 0.0967 higher than that of Omicron BA.1, Delta AY.127 (Table 2). Similarly, the intensity of population mobility across communities promotes the spread of variants to other communities with closely-connected traffic. For example, on the 20th day after the start of Omicron BA.2 spread, the R2 between the intensity of inter-TPU mobility and the symptom onset was 0.0394 and 0.1188 higher than that for Omicron BA.1, Delta AY.127 (Table 2). The effect of intra-TPU/inter-TPU human mobility on the spatial spread of Omicron BA.1, BA.2, and Delta AY.127 became more and more significant over time.
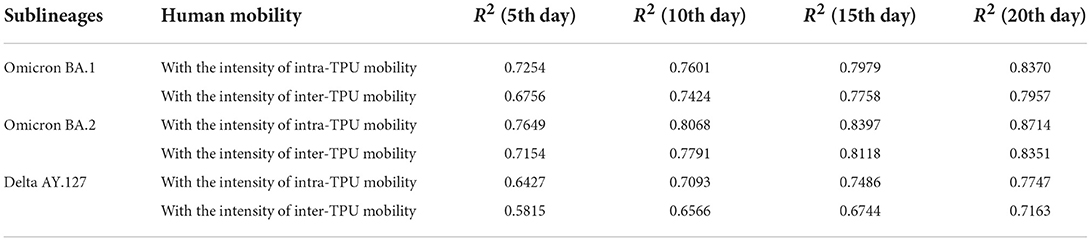
Table 2. The correlation between the symptom onset risk and the intensity of intra-TPU and inter-TPU mobility during the spread of Omicron BA.1, BA.2 and Delta AY.127 in Hong Kong.
We further compared the variations in the overall symptom onset risk of 291 TPUs within 20 days of Omicron BA.1, BA.2, and Delta AY.127 entering the community. The overall onset risk increased from 0.06 to 0.58 within 20 days of Omicron BA.1 spread (Figure 6). The overall risk of disease increased from 0.25 to 0.86 within 20 days of the spread of Omicron BA.2, which was a 1.16-fold increase over the risk of spread of Omicron BA.1 (Figure 6). By the 20th day of Omicron BA.2 entering the community, the overall symptom onset risk in Hong Kong was already at the high-onset-risk level. In contrast, the overall risk increases in the first 20 days of Delta AY.127 spread decreased by 58 and 78% relative to the Omicron BA.1 and BA.2 periods, respectively (Figure 6). By the 20th day of Delta AY.127 entering the community, the overall symptom onset risk in Hong Kong was still at low-medium onset risk level (Figure 6).
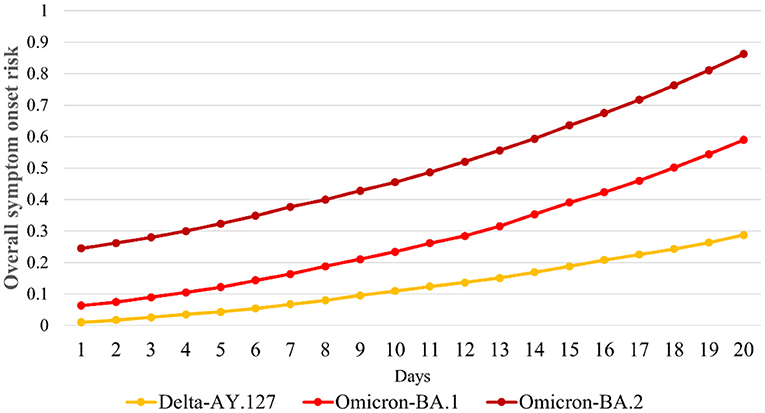
Figure 6. Daily overall risk of COVID-19 symptom onset in Hong Kong within 20 days after Omicron BA.1, BA.2, and Delta AY.127.
According to the variation of the overall onset risk in 291 TPUs in Hong Kong, the overall symptom onset risk (the mean value of the symptom onset risk in 291 TPUs) increased by 0.53, 0.62, and 0.28 within 20 days of Omicron BA.1, BA.2, and Delta AY.127 entering the community (Figure 6). The overall onset risk of Omicron BA.1 and BA.2 on the 20th day after entering the community even reached the medium onset risk level and the high-onset-risk level, respectively. Previous studies have found that vaccination, especially booster vaccination, has significantly improved protection against the symptomatic infection of Omicron and Delta. However, only limited full and booster vaccine occurred increased in Hong Kong because Sinovac and BioNTech vaccination appears to have limited effect in reducing overall onset risk. Thus, simulations were made on how to strengthen vaccination so as to further decrease the symptom onset risk of Omicron BA.1, BA.2, and Delta AY.127 in Hong Kong, in particular, the booster vaccination rates. The simulation was conducted in (i) the current full and booster vaccination rate, (ii) 5 times the full vaccination rate and the current booster vaccination rate, (iii) 5 times the full and booster vaccination rate, (iv) 10 times the full vaccination rate and the current booster vaccination rate, and (v) 10 times the full and booster vaccination rate. During the simulation process, the vaccine effectiveness of Sinovac and BioNTech Vaccine, which decreases over time, were also taken into account.
The temporal variation of the daily overall onset risk values within 20 days of Omicron BA.1, BA.2, and Delta AY.127 entering the community in Hong Kong under the above five scenarios, were explored to reflect the effects of improved full and booster vaccination rate (Figure 7). Within 20 days of Omicron BA.1 entering the community, the full vaccination rate was increased by 5 times (an increase of 175,000 people receiving the Sinovac vaccine, 170,000 people receiving the BioNTech vaccine) or 10 times (an increase of 394,000 people receiving the Sinovac vaccine, 383,000 people receiving the BioNTech vaccine), the overall symptom onset risk decreased by an average of 7.89 and 24.22% respectively (Figure 7A). On this basis, when the booster vaccination rate was increased, by 5 times (an increase of 323,000 people receiving the Sinovac vaccine, 444,000 people receiving the BioNTech vaccine) and 10 times (an increase of 726,000 people receiving the Sinovac vaccine, 1,000,000 people receiving the BioNTech vaccine), the overall symptom onset risk was further reduced by an average of 12.25 and 16.90% (Figure 7A). Compared with Omicron BA.1, the effect of increased vaccination rate on the reduction of overall symptom onset risk was relatively less within 20 days of Omicron BA.2 entering the community. When the full vaccination rate was increased by 5 times (an increase of 165,000 people receiving the Sinovac vaccine, 278,000 people receiving the BioNTech vaccine) and 10 times (an increase of 371,000 people receiving the Sinovac vaccine, 627,000 people receiving the BioNTech vaccine), the overall onset risk was reduced by 4.16 and 21.01% respectively (Figure 7B). When the booster vaccination rate was also increased by 5 times (an increase of 429,000 people receiving the Sinovac vaccine, 967,000 people receiving the BioNTech vaccine) and 10 times (an increase of 965,000 people receiving the Sinovac vaccine, 2176,000 people receiving the BioNTech vaccine), the overall onset risk was further reduced by 7.36 and 11.49% (Figure 7B). An increased vaccination rate was also effective in reducing the risk of symptom onset due to the hamster-related Delta AY.127. When the full vaccination rate was increased by 5 times (an increase of 148,000 people receiving the Sinovac vaccine, 266,000 people receiving the BioNTech vaccine) and 10 times (an increase of 334,000 people receiving the Sinovac vaccine, 600,000 people receiving the BioNTech vaccine), the overall onset risk was reduced by 25.30 and 58.54% (Figure 7C). When the booster vaccination rate was also increased by 5 times (an increase of 539,000 people receiving the Sinovac vaccine, 1,080,000 people receiving the BioNTech vaccine) and 10 times (an increase of 1,210,000 people receiving the Sinovac vaccine, 2,450,000 people receiving the BioNTech vaccine), the overall onset risk was further reduced by 32.37 and 23.63% (Figure 7C).
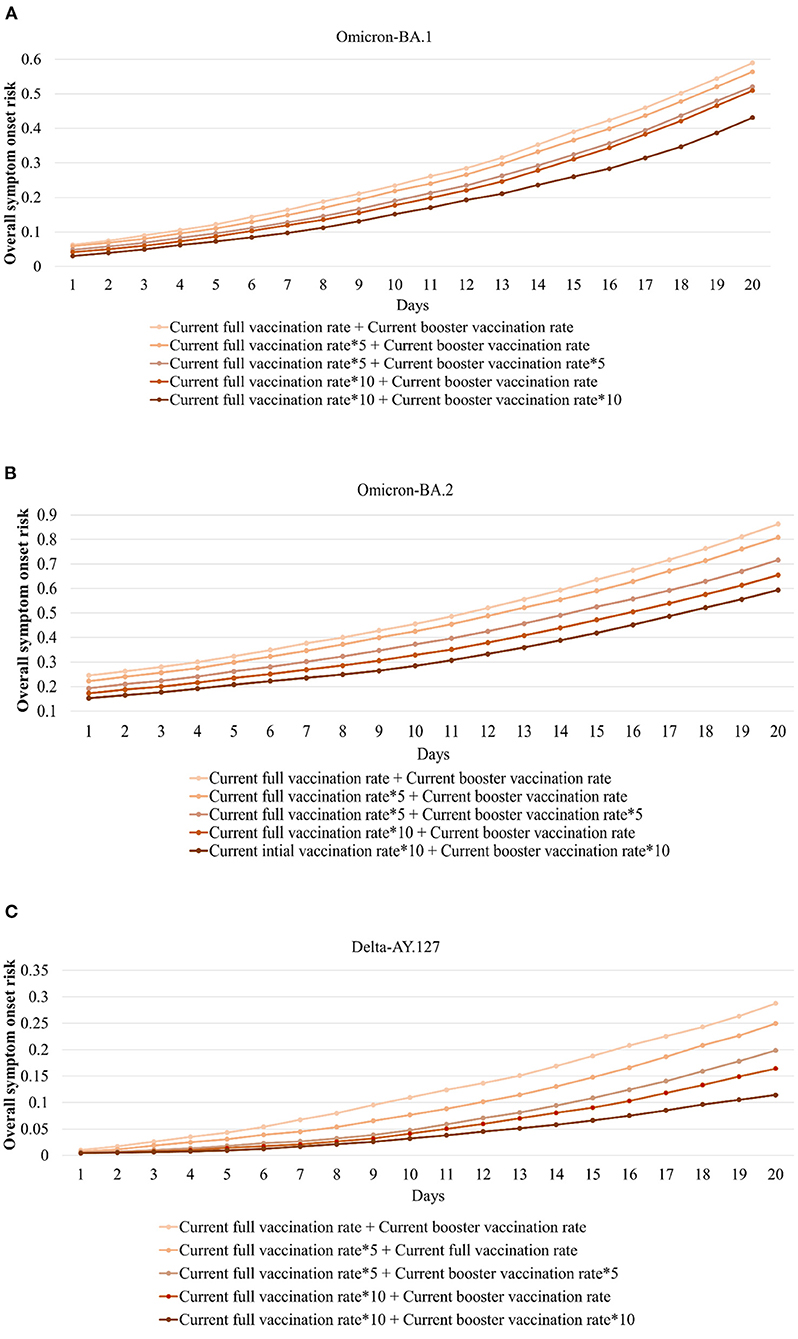
Figure 7. Daily overall symptom onset risk with the full and booster vaccination rate increased by 5 or 10 times during the 20 days when Omicron BA.1, BA.2, and Delta AY.127 entered the community. (A) indicates daily overall symptom onset risk of Omicron BA.1. (B) indicates daily overall symptom onset risk of Omicron BA.2. (C) indicates daily overall symptom onset risk of Delta AY.127.
Furthermore, the onset risk in all 291 TPUs which had 5 to 10 times the current daily full and booster vaccination rate, on the same date, was obviously lower than the risk with the current daily full and booster vaccination rate (Figure 8). When full vaccination rate was increased by 5 times and 10 times, the symptom onset risk in each TPU caused by Omicron BA.1 transmission decreased by 21.83 and 32.76% (Figures 8A–C), respectively, especially for medium and medium-low onset risk TPUs around areas of high human mobility. Over 91.79% of medium and medium-low onset risk TPUs had a maximum 90.15% reduction regarding symptom onset risk. When the booster vaccination rate increased by 5 or 10 times, the symptom onset risk would decrease by 32.45 and 43.12%, especially for these medium-high onset risk TPUs around areas of high human mobility (Figures 8B–D). Over 97.82% of the medium and medium-high onset risk TPUs had a maximum 56.48% reduction in symptom onset risk. Likewise, the increased vaccination rate had a clear effect on reducing the risk of Omicron BA.2 (Figures 8E–H). When the full and booster vaccination rate was increased 10 times, the symptom onset risk in each TPU was reduced by an average of 25.86% (Figure 8G). However, compared with Omicron BA.1, the effect of increasing the full and booster vaccination rate on the reduction of the symptom onset risk of Omicron BA.2 appears to be mainly for TPUs of medium-low and medium onset risk, respectively. The symptom onset risk in these TPUs was reduced by up to 82.34% (Figures 8B–D). In addition, increasing vaccination rates had the most significant effect on reducing the risk of hamster-associated Delta AY.127 (Figures 8I-L). When the full and booster vaccination rate was increased by 10, the symptom onset risk in each TPU was reduced by an average of 92.13% (Figure 8L). in fact, more than 80.97% of TPUs at high onset risk had an average of 83.21% lower symptom onset risk.
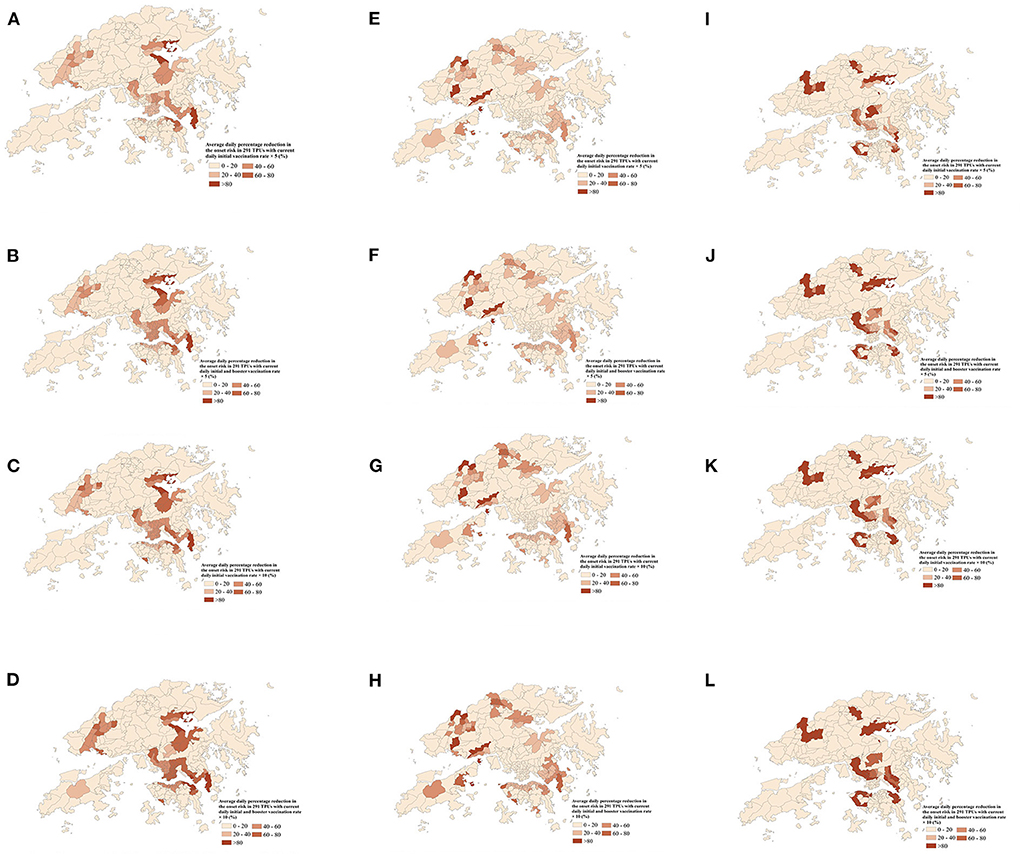
Figure 8. The average daily percentage reduction in the symptom onset risk in 291 TPUs of Hong Kong with 5 times or 10 times the current daily full and booster vaccination rate, compared with the current daily full and booster vaccination rate. (A-D) indicates the average daily percentage reduction in the symptom onset risk of Omicron BA.1 in 291 TPUs. (E-H) indicates the average daily percentage reduction in the symptom onset risk of Omicron BA.2 in 291 TPUs. (I-L) indicates the average daily percentage reduction in the symptom onset risk of Delta AY.127 in 291 TPUs. (A) Average daily percentage reduction in the onset risk in 291 TPUs with 5 times current daily full vaccination rate—Omicron BA.1. (B) Average daily percentage reduction in the onset risk in 291 TPUs with 5 times current daily full and booster vaccination rate—Omicron BA.1. (C) Average daily percentage reduction in the onset risk in 291 TPUs with 10 times current daily full vaccination rate—Omicron BA.1. (D) Average daily percentage reduction in the onset risk in 291 TPUs with 10 times current daily full and booster vaccination rate—Omicron BA.1. (E) Average daily percentage reduction in the onset risk in 291 TPUs with 5 times current daily full vaccination rate—Omicron BA.2. (F) Average daily percentage reduction in the onset risk in 291 TPUs with 5 times current daily full and booster vaccination rate—Omicron BA.2. (G) Average daily percentage reduction in the onset risk in 291 TPUs with 10 times current daily full vaccination rate—Omicron BA.2. (H) Average daily percentage reduction in the onset risk in 291 TPUs with 10 times current daily full and booster vaccination rate—Omicron BA.2. (I) Average daily percentage reduction in the onset risk in 291 TPUs with 5 times current daily full vaccination rate—Delta AY.127. (J) Average daily percentage reduction in the onset risk in 291 TPUs with 5 times current daily full and booster vaccination rate—Delta AY.127. (K) Average daily percentage reduction in the onset risk in 291 TPUs with 10 times current daily full vaccination rate—Delta AY.127. (L) Average daily percentage reduction in the onset risk in 291 TPUs with 10 times current daily full and booster vaccination rate—Delta AY.127.
Currently, with the widespread vaccination (47, 48) and the results of previous infections (49, 50), the COVID-19 immunity barrier in some regions was gradually established. Even for the Omicron variant, previous studies have shown that the COVID-19 vaccine is effective in preventing serious illness and death caused by Omicron (51–53). The pandemic, hence, entered another phase. As regards the advent of medically critical illnesses, one of the results is the necessity to minimize and prevent undue stress on the health care sector and thereby produce adequate care for patients, has become one of the keys to the development of epidemic prevention efforts (54). As a result, major countries in the world have begun to focus on symptomatic cases, especially severe cases, in epidemiological surveillance (17, 55). For example, the US CDC requires that case investigation should focus on symptomatic cases and assessing COVID-19 community levels (16). In Hong Kong, which when at the peak of the epidemic in March of 2022, due to the surge in cases, in order to avoid the overuse of medical resources, the focus of epidemic prevention and control has also been on the monitoring and treatment of symptomatic cases, especially severe cases. Hence, this study provides an analysis and comparison of the spatiotemporal symptom onset risks associated with Omicron BA.1, BA.2, and hamster-related Delta AY.127 during their early spread at 291 TPUs in Hong Kong, to support Hong Kong and the world to better deal with these new variants. Of importance, in this respect, is that, based on the SARS-CoV-2 sequences and the epidemiological link of symptomatic cases determined by whole-genome sequencing, this study used the enhanced urban-community-level WKDE model to enable the prediction of the symptom onset risk with SARS-CoV-2 variants. Furthermore, the spatiotemporal spread of Omicron BA.1, BA.2, and hamster-related Delta AY.127 under different full and booster vaccination scenarios have also been analyzed.
With the use of whole genome sequencing technology, the SARS-CoV-2 sequences and related cluster of symptomatic cases, within 20 days after Omicron BA.1, BA.2, and Delta AY.127 entered the community, were identified. Timely use of whole genome sequencing can greatly facilitate outbreak investigation and understanding of the cryptic chain of transmission, to further help to explore the spatiotemporal symptom onset risk. Based on i) locations with symptomatic cases, ii) locations with positive sewage testing results, and iii) time-varying vaccination rate and vaccination efficiency to strengthen the urban-community-level WKDE model, the model achieved a high accuracy with more than 85% in the onset risk prediction of future 7 days.
Thus, the spatiotemporal variation of the symptom onset risk with Omicron BA.1, BA.2, Delta AY.127 during the associated first 20 days of transmission in Hong Kong could be analyzed. The results are shown in this study:
i) The spatial transmissibility ability of Omicron BA.2 is obviously stronger than that of Omicron BA.1 and Delta AY.127. By the 20th day after Omicron BA.2 entered the community, a total of 154 TPUs were at high onset risk, which were 2.91 and 3.56 times faster than the spread of Omicron BA.1 and Delta AY.127. There were 3,782,243 and 3,808,802 more people in the high-risk area communities than during the same period of spread of Omicron BA.1 and Delta AY.127. Conversely, the spatiotemporal spread of hamster-relate Delta AY.127 was weaker than that of Omicron BA.1 and BA.2. On the 20th day when Delta AY.127 entered the community, only 43 TPUs of high population mobility were at high-onset-risk, which were 0.28 and 0.81 times of that in the same period as Omicron BA.2, BA.1 spread. There were also 3,808,802 and 26,559 fewer people in high-onset-risk TPUs than that with Omicron BA.2, BA.1 in the same period.
ii) Omicron BA.1, BA.2, and Delta AY.127 all started to emerge at the high human mobility community level linked with the traffic flow network. Afterwards, the intensity of human mobility within the community continued to promote the spread of the above SARS-CoV-2 variants to adjacent areas. The intensity of human mobility across communities further continued to promote the spread of SARS-CoV-2 variants to other communities of high human mobility levels, further away. Among these, the effect of inter-community and cross-community human mobility on the enhancement of the spatiotemporal spread of Omicron BA.2 was much more significant than that of Omicron BA.1 and Delta AY.127. This enhancement accumulated over time.
iii) The spread of Omicron BA.1, BA.2, and Delta AY.127 also had a certain impact on the temporal variation of the overall symptom onset risk in Hong Kong over time, but the impact intensity was different. The overall symptom onset risk increased from 0.25 to 0.86 within 20 days of the spread of Omicron BA.2, which was 1.16 times increase during the spread of Omicron BA.1. The overall symptom onset risk in Hong Kong was already at a high-onset-risk level. In contrast, by the 20th day of Delta AY.127 entering the community, the overall symptom onset risk in Hong Kong increased only to the low-medium risk level. The overall risk increases within 20 days of Delta AY.127 spread decreased by 58% and 78% relative to the respective Omicron BA.1 and BA.2 periods.
Through the simulation and comparison of the spatiotemporal symptom onset risk with Omicron BA.1, BA.2, and Delta AY.127 under different scenarios with different levels of full and booster vaccination rates, this study also provides a scientific reference for areas attacked by SARS-CoV-2 variants. The results show that:
1) When the full vaccination rate is increased, the overall symptom onset risk with Omicron BA. 1, BA.2, and Delta AY.127 in Hong Kong is effectively reduced. If the booster vaccination rate is further increased, the overall symptom onset risk can be likewise, further reduced. Within 20 days of Omicron BA.1 entering the community, when the full vaccination rate was increased by 394,000 people receiving the Sinovac vaccine and 383,000 people receiving the BioNTech vaccine, the overall symptom onset risk decreased by an average of 24.22%. On this basis, when the booster vaccination rate was increased by 726,000 people receiving the Sinovac vaccine, and 1,000,000 people receiving the BioNTech vaccine, the overall symptom onset risk was further reduced by an average of 16.90%. Compared with Omicron BA.1, the reduction effect of increased vaccination rate on the overall symptom onset risk of Omicron BA.2 was lower. When the full vaccination rate was increased by 371,000 people receiving the Sinovac vaccine, and 627,000 people receiving the BioNTech, the overall onset risk was only reduced by 4.16 and 21.01%. When the booster vaccination rate was further increased by 965,000 people receiving the Sinovac vaccine, and 2,176,000 people receiving the BioNTech vaccine, the overall onset risk was further reduced by 7.36 and 11.49%. Increased vaccination rates were most effective in reducing the risk of symptom onset due to hamster-related Delta AY.127. When the full vaccination rate was increased by 334,000 people receiving the Sinovac vaccine, and 600,000 people receiving the BioNTech vaccine, the overall onset risk was reduced by 25.30 and 58.54%. When the booster vaccination rate was increased by 1210,000 people receiving the Sinovac vaccine, and 2,450,000 people receiving the BioNTech vaccine, the overall onset risk was further reduced by 32.37 and 23.63%.
2) The increased full and booster vaccination rates can effectively reduce the symptom onset risk for each TPU. The above reduction effect is different for Omicron BA.1, BA.2, and Delta AY.127. For Omicron BA.1, when the full vaccination rate was increased by 10 times, the symptom onset risk in each TPU decreased by 32.76%, especially for medium and medium-low onset risk TPUs around areas of high human mobility. When the booster vaccination rate was also increased by 10 times, the symptom onset risk decreased by 43.12%, especially for the medium-high onset risk TPUs, around areas of high human mobility. But for Omicron BA.2, when the full and booster vaccination rates were increased by 10 times, although onset risk in each TPU was reduced by an average of 25.86%, the reduction effect seemed to be mainly for TPUs of medium-low and medium onset risk. The reduction effect of the symptom onset risk for TPUs originally at high and medium-high risk was more limited. Additionally, increasing vaccination rates could have the most significant effect on reducing the risk of hamster-associated Delta AY.127. When the full and booster vaccination rates were increased 10 times, the simulated symptom onset risk in each TPU was reduced by an average of 92.13%, even for TPUs of high onset risk.
This current study has several limitations worthy of further examination. Firstly, the traffic flow data used in this study to measure daily human mobility is composed only of the official total population traffic flow on major arterial roads in Hong Kong. This restricts our exploration of COVID-19 symptom onset risk in different age groups and genders facing Omicron BA.1, BA.2, and Delta AY.127. Recently, we are applying to China Mobile Hong Kong for the main relevant mobile phone signaling data of different age groups to analyze their daily mobility, which has the potential to support the further exploration of COVID-19 symptom onset risk in people of different ages and genders. Secondly, lack of vaccine efficacy data related to sub-variants, the vaccination effectiveness against Omicron and Delta has been used in this study. However, the predictive performance of this proposed model can be even further improved if further sufficiently reliable vaccine effectiveness data against Omicron BA.1, BA.2, and Delta AY.127, especially for Sinovac vaccines, can be achieved. Importantly the acquisition of reliable effective vaccine data against Omicron BA.1, BA.2, and Delta AY.127 related to different age groups and genders, will further strengthen the additional study of COVID-19 symptom onset risk for different ages and different genders.
WHO requests countries and regions to continue to be vigilant, to monitor and report new sequences, as well as to conduct independent and comparative analyses of the different Omicron sub-lineages (2). Furthermore, WHO also encourages countries and regions to share available data on transmissibility and severity of variants, and their impact on diagnostics and vaccines (2). As the study has provided a comprehensive investigation about the spatiotemporal symptom onset risk of Omicron BA.1, BA.2, and Delta AY.127, we hope that this study can assist countries and regions to prevent the emergence of these variants on a strategic basis. Moreover, for countries and regions where the new SARS-CoV-2 variant Omicron has appeared, this study provides scientifically derived findings on the impact of the full and booster vaccination campaigns on reducing the symptomatic infection.
Publicly available datasets were analyzed in this study. The Whole Genome Sequencing data can be found at the Nextstrain repository: https://nextstrain.org/groups/ncovHK/ncov/Latest?c=transmission_alert&p%24=%24full; The real-time effective reproductive number data can be found at https://covid19.sph.hku.hk/; The vaccination data can be found at https://www.covidvaccine.gov.hk/en/dashboard. The traffic Data of Strategic/Major Roads can be found at https://data.gov.hk/en-data/dataset/hk-td-sm_4-traffic-data-strategic-major-roads.
WS conceived and designed the study, interpreted the results, and helped develop the computation models, analyze the data, and write the manuscript. CT collected the data, developed the computation models, wrote the manuscript, and helped interpreted the results. GS collected the data, conducted the whole gene sequencing, and helped write the manuscript. AZ helped develop the computation models, analyze the data, and write the manuscript. ZS wrote the manuscript, helped collect the data, and analyze the data and interpret the results. All authors contributed to the article and approved the submitted version.
This study was supported by National Key R&D Program of China (2019YFB2103102), Hong Kong Research Grants Council (C5079-21G), and Otto Poon Charitable Foundation Smart Cities Research Institute, The Hong Kong Polytechnic University (Work Program: CD03).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpubh.2022.978052/full#supplementary-material
1. World Health Organization. Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. (2021). Available online at: https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-SARS-CoV-2-variant-of-concern (accessed December 1, 2021).
2. World Health Organization. Statement on Omicron sublineage BA.2. (2022). Available online at: https://www.who.int/news/item/22-02-2022-statement-on-omicron-sublineage-ba.2 (accessed February 23, 2022).
3. Yu J, Collier AY, Rowe M, Mardas F, Ventura JD, Wan H, et al. Neutralization of the SARS-CoV-2 Omicron BA.1 and BA.2 Variants. N Engl J Med. (2022) 386:1579–180. doi: 10.1056/NEJMc2201849
4. Saxena SK, Kumar S, Ansari S, Paweska JT, Maurya VK, Tripathi AK, et al. Transmission dynamics and mutational prevalence of the novel Severe acute respiratory syndrome coronavirus-2 Omicron Variant of Concern. J Med Virol. (2022) 94:2160–6. doi: 10.1002/jmv.27611
5. Fonager J, Bennedbæk M, Bager P, Wohlfahrt J, Ellegaard KM, Ingham AC, et al. Molecular epidemiology of the SARS-CoV-2 variant Omicron BA.2 sub-lineage in Denmark, 29 November 2021 to 2 January 2022. Eurosurveillance. (2022) 27:1–7. doi: 10.2807/1560-7917.ES.2022.27.10.2200181
6. Bruel T, Hadjadj J, Maes P, Planas D, Seve A, Staropoli I, et al. Serum neutralization of SARS-CoV-2 Omicron sublineages BA.1 and BA.2 in patients receiving monoclonal antibodies. Nat Med. (2022) 28: 1297–302. doi: 10.1101/2022.03.09.22272066
7. Huang J, Zeng G. Letter to the editor: Epidemiology of the SARS-CoV-2 variant Omicron BA2 – vigilance needed. Eurosurveillance. (2022) 27:1–2. doi: 10.2807/1560-7917.ES.2022.27.13.2200254
8. Callaway E. Why does the Omicron sub-variant spread faster than the original? Nature. (2022) 602:556–7. doi: 10.1038/d41586-022-00471-2
9. Mallapaty S. COVID-19: how omicron overtook delta in three charts. Nature. (2022) 12:1–6. doi: 10.1038/d41586-022-00632-3
10. Kok K-H, Wong S-C, Chan W-M, Wen L, ChuA W-H, Ip JD, et al. Co-circulation of two SARS-CoV-2 variant strains within imported pet hamsters in Hong Kong. Emerg Microbes Infect. (2022) 11:689–98. doi: 10.1080/22221751.2022.2040922
11. Yen H-L, Sit THC, Brackman CJ, Chuk SSY, Gu H, Tam KWS, et al. Transmission of SARS-CoV-2 delta variant (AY127) from pet hamsters to humans, leading to onward human-to-human transmission: a case study. Lancet. (2022) 399:1070–8. doi: 10.1016/S0140-6736(22)00326-9
12. Taylor L. Covid-19: Hong Kong reports world's highest death rate as zero covid strategy fails. BMJ. (2022) 376:o707. doi: 10.1136/bmj.o707
13. Burki T. Hong Kong's fifth COVID-19 wave—the worst yet. Lancet Infect Dis. (2022) 22:455–6. doi: 10.1016/S1473-3099(22)00167-0
14. New Zealand Government. Releases (/ releases) COVID-19 Protection Framework Supported by New Testing Contact Tracing Strategy. (2021). Available online at: https://www.beehive.govt.nz/release/covid-19-protection-framework-supported-new-testing-and-contact-tracing-strategy (accessed December 1, 2021).
15. Pan American Health Organization. As COVID-19 Cases Nearly Double in the Americas, Health Workers Must be Protected, PAHO Says - PAHO/WHO. Pan American Health Organization. (2022). Available online at: https://www.paho.org/en/news/12-1-2022-covid-19-cases-nearly-double-americas-health-workers-must-be-protected-paho-says (accessed February 1, 2022).
16. The Centers for Disease Control Prevention. Prioritizing Case Investigation and Contact Tracing for COVID-19. (2022). Available online at: https://www.cdc.gov/coronavirus/2019-ncov/php/contact-tracing/contact-tracing-plan/prioritization.html (accessed February 20, 2022).
17. European Centre for Disease Prevention and Control. COVID-19 Surveillance Guidance Key Messages Surveillance Objectives. Solna: European Centre for Disease Prevention and Control (2021).
18. European Centre for Disease Prevention Control. Assessment of the further spread and potential impact of the SARS-CoV-2 Omicron variant of concern in the EU / EEA, 19th update Risk assessed. (2022). Available online at: https://www.ecdc.europa.eu/sites/default/files/documents/RRA-19-update-27-jan-2022.pdf (accessed February 1, 2022).
19. Cheng SMS, Mok CKP, Leung YWY, Ng SS, Chan KCK, Ko FW, et al. Neutralizing antibodies against the SARS-CoV-2 Omicron variant BA1 following homologous and heterologous CoronaVac or BNT162b2 vaccination. Nat Med. (2022) 28:486–9. doi: 10.1038/s41591-022-01704-7
20. Gu H, Krishnan P, Ng DYM, Chang LDJ, Liu GYZ, Cheng SSM, et al. Probable transmission of SARS-CoV-2 omicron variant in quarantine hotel, Hong Kong, China, November 2021. Emerg Infect Dis. (2022) 28:460–2. doi: 10.3201/eid2802.212422
21. Mallapaty S. How sneezing hamsters sparked a COVID outbreak in Hong Kong. Nature. (2022) 3–7. doi: 10.1038/d41586-022-00322-0
22. Government of Hong Kong. Multi-Pronged Measures to Increase COVID-19 Vaccination Uptake. (2022). Available online at: https://www.info.gov.hk/gia/general/202201/07/P2022010700565.htm (accessed February 1, 2022).
23. The Government of the Hong Kong Special Administrative Region. Hong Kong Vaccination Dashboard. (2022). Available online at: https://www.covidvaccine.gov.hk/en/dashboard (accessed February 1, 2022).
24. Lu L, Chu AW-H, Zhang RR, Chan W-M, Ip JD, Tsoi H-W, et al. The impact of spike N501Y mutation on neutralizing activity and RBD binding of SARS-CoV-2 convalescent serum. EBioMed. (2021) 71:103544. doi: 10.1016/j.ebiom.2021.103544
25. Chan CT-M, Leung JS-L, Lee L-K, Lo HW-H, Wong EY-K, Wong DS-H, et al. A low-cost TaqMan minor groove binder probe-based one-step RT-qPCR assay for rapid identification of N501Y variants of SARS-CoV-2. J Virol Methods. (2022) 299:114333. doi: 10.1016/j.jviromet.2021.114333
26. Cheng VC-C, Siu GK-H, Wong S-C, Au AK-W, Ng CS-F, Chen H, et al. Complementation of contact tracing by mass testing for successful containment of beta COVID-19 variant (SARS-CoV-2 VOC B1351) epidemic in Hong Kong. Lancet Reg Heal West Pacific. (2021) 17:100281. doi: 10.1016/j.lanwpc.2021.100281
27. Leung KSS, Ng TTL, Wu AKL, Yau MCY, Lao HY, Choi MP, et al. Territorywide study of early coronavirus disease outbreak, Hong Kong, China. Emerg Infect Dis. (2021) 27:196–204. doi: 10.3201/eid2701.201543
28. Tong C, Shi W, Zhang A, Shi Z. Tracking and controlling the spatiotemporal spread of SARS-CoV-2 Omicron variant in South Africa. Travel Med Infect Dis. (2022) 46:102252. doi: 10.1016/j.tmaid.2021.102252
29. Shi W, Tong C, Zhang A, Wang B, Shi Z, Yao Y, et al. An extended Weight Kernel Density Estimation model forecasts COVID-19 onset risk and identifies spatiotemporal variations of lockdown effects in China. Commun Biol. (2021) 4:26. doi: 10.1038/s42003-021-01677-2
30. Shi W, Tong C, Zhang A, Shi Z. A Spatial and Dynamic Solution for how to Allocate the COVID-19 Vaccine in the Context of Limited Supply. Commun Med. (2021) 1:23. doi: 10.1038/s43856-021-00023-1
31. Tong C, Shi W, Zhang A, Shi Z. Tracking and controlling the spatiotemporal spread of SARS-CoV-2 Lineage B.1.1.7 in COVID-19 reopenings. GeoHealth. (2021) 5:e2021GH000517. doi: 10.1029/2021GH000517
32. Moreira ED Jr, Kitchin N, Xu X, Dychter SS, Lockhart S, Gurtman A, et al. Safety and efficacy of a third dose of BNT162B2 covid-19 vaccine. N Engl J Med. (2022) 386:1910-21. doi: 10.1056/NEJMoa2200674
33. Andrews N, Stowe J, Kirsebom F, Toffa S, Rickeard T, Gallagher E, et al. Covid-19 vaccine effectiveness against the omicron (B.1.1.529) variant. N Engl J Med. (2022) 386:1532–46. doi: 10.1056/NEJMoa2119451
34. Abu-Raddad LJ, Chemaitelly H, Ayoub HH, AlMukdad S, Yassine HM, Al-Khatib HA, et al. Effect of mRNA vaccine boosters against SARS-CoV-2 omicron infection in Qatar. N Engl J Med. (2022) 386:1804–16. doi: 10.1056/NEJMoa2200797
35. Prunas O, Warren JL, Crawford FW, Gazit S, Patalon T, Weinberger DM, et al. Vaccination with BNT162b2 reduces transmission of SARS-CoV-2 to household contacts in Israel. Science. (2022) 375:1151–4. doi: 10.1126/science.abl4292
36. Buchan SA, Chung H, Brown KA, Austin PC, Fell DB, Gubbay JB, et al. Effectiveness of COVID-19 vaccines against Omicron or Delta symptomatic infection and severe outcomes. medRxiv. [Preprint]. (2022). doi: 10.1101/2021.12.30.21268565
37. Siu GK, Lee L, Leung KS-S, Leung JS, Ng TT-L, Chan CT-M, et al. Will a new clade of SARS-CoV-2 imported into the community spark a fourth wave of the COVID-19 outbreak in Hong Kong? Emerg Microbes Infect. (2020) 9:2497–500. doi: 10.1080/22221751.2020.1851146
38. Siu GK, Wong TF. Genomic Epidemiology of Novel Coronavirus - Hk-Focused Subsampling. Nextstrain. (2022). Available online at: https://nextstrain.org/groups/ncovHK/ncov/Latest?c=transmission_alert&p%24=%24full (accessed February 1, 2022).
39. The Centre for Health Protection. Places with Sewage Sample(s) Tested Positive. (2022). Available online at: https://www.chp.gov.hk/en/features/105294.html?page=9 (accessed February 1, 2022).
40. Transport Department of Hong Kong. 737 Closed Circuit Televisions (CCTV) and Traffic detectors in Hong Kong. (2021). Available online at: https://data.gov.hk/en-data/dataset/hk-td-tis_2-traffic-snapshot-images (accessed February 1, 2022).
41. © McMenamin ME, Nealon J, Lin Y, Wong JY, Cheung JK, Lau EHY, et al. Vaccine effectiveness of one, two, and three doses of BNT162b2 and CoronaVac against COVID-19 in Hong Kong: a population-based observational study. Lancet Infect Dis. (2022) S1473–3099:00345-0. doi: 10.1016/S1473-3099(22)00345-0
42. Wu D, Zhang Y, Tang L, Wang F, Ye Y, Ma C, et al. Effectiveness of inactivated COVID-19 vaccines against symptomatic, pneumonia, and severe disease caused by the delta variant: real world study and evidence - China, 2021. China CDC Wkly. (2022) 4:57–65. doi: 10.46234/ccdcw2022.009
43. D24H@HKSTP and HKU WHO Collaborating Centre on Infectious Disease Epidemiology and Modelling. Update report dated February 21, 2022. (2022). Available online at: https://www.med.hku.hk/en/news/press/-/media/575E4CA7F5584164B108386D3EF7A6E3.ashx (accessed February 1, 2022).
44. School of Public Health of Hong Kong University. Real-Time Effective Reproductive Number for Local Cases in Hong Kong. (2022). Available online at: https://covid19.sph.hku.hk/ (accessed February 1, 2022).
45. Torjesen I. Covid-19: Peak of viral shedding is later with omicron variant, Japanese data suggest. BMJ. (2022) 376:o89. doi: 10.1136/bmj.o89
46. Singanayagam A, Hakki S, Dunning J, Madon KJ, Crone MA, Koycheva A, et al. Community transmission and viral load kinetics of the SARS-CoV-2 delta (B16172) variant in vaccinated and unvaccinated individuals in the UK: a prospective, longitudinal, cohort study. Lancet Infect Dis. (2022) 22:183–95. doi: 10.1016/S1473-3099(21)00648-4
47. Du Z, Wang L, Pandey A, Lim WW, Chinazzi M, Piontti AP y, et al. Modeling comparative cost-effectiveness of SARS-CoV-2 vaccine dose fractionation in India. Nat Med. (2022) 28:934–8. doi: 10.21203/rs.3.rs-855843/v1
48. Lobinska G, Pauzner A, Traulsen A, Pilpel Y, Nowak MA. Evolution of resistance to COVID-19 vaccination with dynamic social distancing. Nat Hum Behav. (2022) 6:193–206. doi: 10.1038/s41562-021-01281-8
49. Madhi SA, Kwatra G, Myers JE, Jassat W, Dhar N, Mukendi CK, et al. Population Immunity and Covid-19 Severity with Omicron Variant in South Africa. N Engl J Med. (2022) 386:1314–26. doi: 10.1056/NEJMoa2119658
50. Sigal A. Milder disease with Omicron: is it the virus or the pre-existing immunity? Nat Rev Immunol. (2022) 22:69–71. doi: 10.1038/s41577-022-00678-4
51. Nemet I, Kliker L, Lustig Y, Zuckerman N, Erster O, Cohen C, et al. Third BNT162b2 vaccination neutralization of SARS-CoV-2 Omicron infection. N Engl J Med. (2022) 386:492–4. doi: 10.1056/NEJMc2119358
52. Lauring AS, Tenforde MW, Chappell JD, Gaglani M, Ginde AA, McNeal T, et al. Clinical severity of, and effectiveness of mRNA vaccines against, covid-19 from omicron, delta, and alpha SARS-CoV-2 variants in the United States: prospective observational study. BMJ. (2022) 376:e069761. doi: 10.1136/bmj-2021-069761
53. Nyberg T, Ferguson NM, Nash SG, Webster HH, Flaxman S, Andrews N, et al. Comparative analysis of the risks of hospitalisation and death associated with SARS-CoV-2 omicron (B11529) and delta (B16172) variants in England: a cohort study. Lancet. (2022) 399:1303–12. doi: 10.1016/S0140-6736(22)00462-7
54. World Health Organization. Strategic Preparedness, Readiness and Response Plan to End the Global COVID-19 Emergency in 2022. (2022). Available online at: https://www.who.int/publications/m/item/strategic-preparedness-readiness-and-response-plan-to-end-the-global-covid-19-emergency-in-2022 (accessed February 1, 2022).
55. European Centre for Disease Prevention Control. Guidance for the Prevention and Control of COVID-19 in Temporary Reception Centres in the Context of the Large Numbers of People Fleeing Ukraine Key Messages. (2022). Available online at: https://www.ecdc.europa.eu/sites/default/files/documents/Guidance-for-the-prevention-and-control-of-COVID-19-in-temporary-reception-centres-in-context-large-numbers-people-fleeing-Ukraine.pdf (accessed February 1, 2022).
Keywords: Omicron BA.1 and BA.2, hamster-related Delta AY.127, spatiotemporal symptom onset risk, full vaccination, booster vaccination
Citation: Tong C, Shi W, Siu G-H, Zhang A and Shi Z (2022) Understanding spatiotemporal symptom onset risk of Omicron BA.1, BA.2 and hamster-related Delta AY.127. Front. Public Health 10:978052. doi: 10.3389/fpubh.2022.978052
Received: 25 June 2022; Accepted: 22 August 2022;
Published: 16 September 2022.
Edited by:
Hamad Ali, Kuwait University, KuwaitReviewed by:
Anwar Mohammad, Dasman Diabetes Institute, KuwaitCopyright © 2022 Tong, Shi, Siu, Zhang and Shi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wenzhong Shi, am9obi53ei5zaGlAcG9seXUuZWR1Lmhr; Zhicheng Shi, c2hpemhpY2hlbmdAc3p1LmVkdS5jbg==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.