
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Psychol., 10 October 2022
Sec. Neuropsychology
Volume 13 - 2022 | https://doi.org/10.3389/fpsyg.2022.1014202
Post-traumatic stress disorder (PTSD) is a debilitating sequela of extraordinary traumatic sufferings that threaten personal health and dramatically attenuate the patient's quality of life. Accumulating lines of evidence suggest that functional disorders in the ventral tegmental area (VTA) dopaminergic system contribute substantially to PTSD symptomatology. Notably, music therapy has been shown to greatly ameliorate PTSD symptoms. In this literature review, we focused on whether music improved PTSD symptoms, based on VTA dopaminergic action, including the effects of music on dopamine (DA)-related gene expression, the promotion of DA release and metabolism, and the activation of VTA functional activities. In addition, the strengths and limitations of the studies concerning the results of music therapy on PTSD are discussed. Collectively, music therapy is an effective approach for PTSD intervention, in which the VTA dopaminergic system may hold an important position.
Post-traumatic stress disorder (PTSD) is a debilitating psychiatric disorder that occurs following exposure to extraordinary actual or threatened trauma (such as death, serious injury, or sexual violation), with four cardinal symptomatic clusters according to criteria of the Diagnostic and Statistical Manual of Mental Disorders (DSM)-5: re-experiencing (e.g., intrusive trauma-related imagery and reliving in nightmares), avoidance (e.g., avoiding reminders of the traumatic event), negative cognitions and moods (e.g., depression and anxiety), and arousal (e.g., prolonged hypervigilance that causes irritability or frequent outbursts of anger, difficulty in concentrating, trouble falling asleep and exaggerated startle response); and the duration of the disturbance lasts more than 1 month (American Psychiatric Association, 2013; Bisson et al., 2015; Shalev et al., 2017). PTSD remarkably impairs personal and social functions, causing, for example, obstacles in interpersonal relationships (Bisson et al., 2015; Yehuda et al., 2015; Shalev et al., 2017), family instability (Jordan et al., 1992), substance use disorders (Bisson et al., 2015; Hakvoort et al., 2020), high risks of suicidal ideation (Bisson et al., 2015; Brown et al., 2020), and considerable cost-effectiveness of clinical treatments (Bisson et al., 2015; Von Der Warth et al., 2020). In addition, the biological vulnerability to PTSD may be transmitted across generations through epigenetic processes (Ramo-Fernández et al., 2015; Yehuda et al., 2015).
It is estimated that the overall lifetime prevalence of PTSD ranges from 1.3 to 12.2% in civilians (Zhou et al., 2021), with higher prevalence in some populations such as in soldiers and rape survivors (Bisson et al., 2015; Shalev et al., 2017; Baranyi et al., 2018; Eiset et al., 2021). The reason why an individual is susceptible to PTSD while others exhibit resilience or recovery remains largely unknown.
There are several evidence-based therapies recommended for PTSD, such as psychological, pharmacological, and physiotherapeutic interventions (Bisson et al., 2015; Yehuda et al., 2015). Although such therapies are empirically validated, their actual feasibility remains highly debated (Harvey et al., 2003; Bradley et al., 2005; Wilson et al., 2018). None of the available methods are uniformly successful, and all of the methods have been reported with side effects (Masand and Gupta, 2002; Guina et al., 2015; Akiki and Abdallah, 2018). Moreover, some therapies are contraindicated in children, pregnant women, and individuals with special requirements, such as due to liver/kidney dysfunctions (Blanaru et al., 2012).
Notably, music therapy has been shown to exhibit significant effects on trauma symptoms (e.g., it improves wellbeing and sleep quality) in traumatized refugees (Jespersen and Vuust, 2012; Beck et al., 2018a, 2021). There is abundant theoretical and empirical evidence concerning the beneficial effects of music therapy on PTSD patients (Table 3). Considering that PTSD is strongly associated with mesolimbic dopaminergic dysfunction (Zhou et al., 2021), the aim of the present review was to detail whether and how music therapy affects PTSD associated with VTA dopaminergic action.
The previous studies published between January 1, 1960 and July 20, 2022 were searched using electronic databases, such as Google Scholar, PubMed, EMBASE, Cochrane, Web of Science, CINAHL (EBSCO), and PsycINFO (EBSCO). For studies involving music therapy on PTSD, the search strings were (music*) AND (PTSD OR post-traumatic stress disorder OR posttraumatic stress disorder). The inclusion criteria were as follows: (1) human studies, (2) a clear diagnosis of PTSD, and (3) music therapy used to treat the PTSD symptoms. To identify studies of music modulation on dopaminergic action, the search strings were (music*) AND (dopamine* level OR dopamine* concentration OR dopamine* release OR dopamine* regulat* OR dopamine* modulat* OR dopamine* gene*). The inclusion criteria were as follows: (1) either human studies or those in rats or mice, (2) studies that evaluated DA levels, DA metabolite levels, or DA-related gene expression following music therapy, and (3) with a clear method for measuring DA level or DA-related gene expression. The exclusion criteria were as follows: (1) review/meta-analyses, (2) non-full-text publications, or (3) articles not written in English. The flow diagram for the literature search is shown in Figure 1.
The title and abstract of each study were first screened to identify potential literatures on this topic. Full texts were then assessed for eligibility based on the inclusion and exclusion criteria. The articles selected to evaluate the effects of music therapy on PTSD and music modulation on dopaminergic action (DA level and DA-related gene expression) are presented in Tables 1–3.
Music listening and performance have been reported to alter the expression of multiple DA-related genes (Table 1). A recent study found that hsa-miR-23a was upregulated in music listeners (Nair et al., 2021), and as hsa-miR-23a was associated with dopaminergic neuronal activation and striatal DA stabilization (Nair et al., 2003, 2021), music may strengthen dopaminergic actions and induce feelings of pleasure (Menon and Levitin, 2005; Stegemöller, 2014; Zatorre, 2015).
The alpha-synuclein gene (SNCA), which is located in the region with the strongest linkage to musical aptitude on chromosome 4q22.1 (Kanduri et al., 2015b), is significantly up-regulated in individuals listening to and/or performing music (Järvelä, 2018; Nair et al., 2021). As SNCA is closely linked to DA metabolism, music therapy may regulate the dopaminergic pathway (Järvelä, 2018). GATA2 (encoding GATA binding protein 2) is abundantly expressed in dopaminergic neurons and is also up-regulated following stimuli with music (Järvelä, 2018). Interestingly, this gene regulates SNCA expression (Kanduri et al., 2015b), indicating that there may be a synergistic effect in both genes due to musical stimuli. It should be noted that over-expression of SNCA and GATA2 has been implicated in Parkinson's disease (Somayaji et al., 2021) and hematopoietic diseases (Zhou et al., 2019), respectively. We hypothesize that music-mediated up-regulation of these genes is limited to certain brain regions (e.g., music-mediated up-regulation principally in VTA) or the overall expression level is far below pathopoiesia.
Several other genes involved in DA secretion, transport, and signaling are also up-regulated following listening to music and/or musical performance. For example, RTN4 is involved in DA secretion (Kanduri et al., 2015b); SLC6A8 is associated with DA regulation, secretion, and transport (Kanduri et al., 2015b); PPP2R3A is involved in DA metabolism (Järvelä, 2018); and RGS9 is involved in the regulation of intracellular signaling in dopaminergic neurons (Rahman et al., 2003; Järvelä, 2018). However, some researchers reported inconsistent changes in the expressions of DA-related genes. For example, DRD2 and DRD4 have been found to have associations (Ukkola et al., 2009; Quarto et al., 2017) and inconclusive relationships with musical aptitudes (Ukkola et al., 2009; Mariath et al., 2017).
Collectively, several DA-related genes are up-regulated upon listening to music, and may facilitate dopaminergic release, activate the reward pathways, and ultimately correlate with pleasant feelings (Stegemöller, 2014; Zatorre, 2015; Moraes et al., 2018).
Endogenous DA release was markedly increased when individuals listened to pleasurable music; however, the DA release pattern was significantly different in different anatomical pathways, e.g., music-induced DA release in the caudate was more involved during the anticipation of listening to music, whereas DA release in the nucleus accumbens (NAc) was more involved during listening to music (Salimpoor et al., 2011).
Studies from animals and human beings have indicated that music listening enhanced the DA levels in the brain, such as increases in DA in the NAc, VTA, prefrontal cortex (PFC), and neostriatum (Sutoo and Akiyama, 2004; Menon and Levitin, 2005; Feduccia and Duvauchelle, 2008; Tasset et al., 2012). However, other studies reported no differences in DA levels in the NAc, piriform cortex, motor cortex, insular cortex, and somatosensory cortex after musical stimuli (Sutoo and Akiyama, 2004). Interestingly, dual pharmacological manipulation of DA through the use of a DA precursor (levodopa) and a DA antagonist (risperidone) positively and negatively affects human responses to music, thus, indicating a causal role of DA function in musical pleasure (Ferreri et al., 2019). Examples of DA and dopaminergic metabolite changes in response to musical stimuli are listed in Table 2.
Music-induced VTA activation has been detected with fMRI (Menon and Levitin, 2005). Music also activated the connectivity between the VTA and other regions, such as the NAc, hypothalamus, insula, orbitofrontal cortex, and bilateral inferior frontal cortex (Menon and Levitin, 2005). However, no data are available to illustrate whether and how music affects VTA neuronal activities, such as dopaminergic neuron firing or burst characteristics [burst represents a periodic high-discharge firing that produces robust dopamine release (Zhou et al., 2021)], or electrophysiological properties between dopaminergic and other neurons, such as long-term potentiation [persistent synaptic strengthening after stimulation (Ungless et al., 2001)].
Music therapy has been reported to have ameliorative effects on almost all PTSD symptomatic clusters. Examples of music therapy for PTSD patients are listed in Table 3.
Several studies reported that PTSD patients listening to music experienced remarkable reductions in PTSD symptoms (Blanaru et al., 2012; Pourmovahed et al., 2021), even when experienced through remotely delivered modalities (Lightstone et al., 2015). In general, group music therapy (GMT) may be more effective than single music treatment for the reduction of the severity of PTSD symptoms and improvement in quality of life (Bensimon et al., 2012; Carr et al., 2012; Macfarlane et al., 2019; de Witte et al., 2022).
Different modalities of music therapies exert different effects on PTSD phenotypes aside from the cardinal symptoms. Guided imagery and music (GIM) significantly decreases the symptoms of dissociation (Maack, 2012; Story and Beck, 2017); GIM in the trauma-focused group of patients enhanced PTSD patients' quality of life and wellbeing (Rudstam et al., 2017; Beck et al., 2021); songwriting treatment reduced the numbing and depressive symptoms of PTSD patients (Coulter, 2000; Precin, 2011; Hirschberg et al., 2020), and playing musical instruments, such as drums or guitars, increased the sense of openness and togetherness, and facilitated non-intimidating access to traumatic memories in PTSD patients (Bensimon et al., 2008; Pezzin et al., 2018).
Collectively, music therapy has idiosyncratic merit and benefits for PTSD intervention. Moreover, the therapeutic modality may also affect the effect of treatment.
Intrusion represents unwanted distressing memories or cues that evoke “flashbacks” of the traumatic events, causing the patient to not be able to distinguish the past from the present (Bisson et al., 2015; Shalev et al., 2017; Fenster et al., 2018). Carr et al. (2012) found that music therapy eliminated dissociative flashbacks in PTSD patients. Music may establish “a safe space” in the brain that strengthens the tolerance for traumatic experiences (Bensimon et al., 2008), helps PTSD patients remain grounded, and helps them distinguish the present moment from the past traumatic events (Volkman, 1993; Orth, 2001; Bensimon et al., 2008).
Avoidance includes the intentional forgetting of past events, or rejecting to face the cues that might associate with traumatic events (Bisson et al., 2015; Shalev et al., 2017; Fenster et al., 2018). Music therapy is an effective approach to addressing the avoidant behavior of PTSD sufferers. Music improvization requires active participation, not just passive acceptance (Volkman, 1993; Orth, 2005), which requires the participants to engage and foster group commitments, and avoid distressing memories or cues associated with their trauma (Orth, 2005; Bensimon et al., 2008; Carr et al., 2012). Remarkably, during group music actions, PTSD patients listen to his/her playing, focus on another's creation, and enjoy the entire atmosphere simultaneously, which may distract from one's own traumatic memories (Carr et al., 2012; de Witte et al., 2022).
Negative cognition and mood include the inability to experience positive emotion and persistent negative beliefs (Bisson et al., 2015; Shalev et al., 2017; Fenster et al., 2018). Music is a helpful way of emotional expression, particularly on the distress caused by trauma. For example, playing musical instruments or singing songs elicit strong emotional responses to control negative moods, relieve bad feelings, and release physical energy (Orth, 2005; Bensimon et al., 2008; Carr et al., 2012). Music has been widely used to express feelings of anger and irritability by encouraging the tolerance of silence and loud sounds (Volkman, 1993; Orth, 2005; Bensimon et al., 2008), resulting in feelings of relief, satisfaction, and empowerment (Bensimon et al., 2008). Music-mediated excitation in the mesolimbic dopaminergic system may associate with reductions in negative cognition and mood improvements (Menon and Levitin, 2005; Salimpoor et al., 2011).
Hyperarousal includes high precaution, excessive startle responses, insomnia, irritability, aggression, lack of concentration, and lack of confidence (Shalev et al., 2017; Fenster et al., 2018). Listening to or playing music enables PTSD patients to create calmness, reduce tension, dismiss distressing reminders, and engages traumatized individuals to promote relaxation, perceive safety, and enjoy happiness (Orth, 2005; Macfarlane et al., 2019). Hyperarousal is reported to be associated with increased amygdala activity, while musical intervention is found to calm amygdalar actions that reduce hyperarousal (Hayes et al., 2012; Pitman et al., 2012).
The auditory cortex is the primary center for musical information processing, which carries out musical perceptual analyses and extracts abstract information from acoustic features (such as pitch, timbre, intensity, and roughness) (Koelsch and Siebel, 2005; Boso et al., 2006). Interestingly, acoustic projections to several areas of the limbic system, including the VTA (Koelsch and Siebel, 2005; Menon and Levitin, 2005; Kraus and Canlon, 2012), are associated with emotional modulation. Accumulating evidence illustrates that the auditory cortex causes functional modifications to the VTA through complicated projections (Salimpoor and Zatorre, 2013; Belfi and Loui, 2020).
The VTA has been shown to be activated due to music listening, and consequently, DA release is triggered (Menon and Levitin, 2005; Salimpoor et al., 2011). Several studies have revealed that PTSD was associated with reduced VTA dopaminergic activities and DA levels in the brain (Corralfrias et al., 2013). Interestingly, some medications that stabilize or enhance dopaminergic signaling in the brain can ameliorate PTSD symptoms in patients (Houlihan, 2011; McLaughlin et al., 2016; Zhou et al., 2021). A previous review summarizes that VTA dopaminergic activation rescues PTSD symptoms through several principal pathways, such as the VTA dopaminergic projections to the NAc, PFC, hippocampus, habenula, and amygdala; all of which are strongly involved in PTSD-related symptomatic processes (Zhou et al., 2021). Therefore, musical activation of VTA dopaminergic singling may also replicate actions of the pathways that theoretically compensate for PTSD-induced hypodopaminergia and conjecturally mitigate PTSD symptoms (Figure 2). This hypothesis is in line with previous theories: when an individual is confronted with traumatic events, the DA level is transiently increased, but followed by a persistent decrease (Corralfrias et al., 2013), and if the dopaminergic neurons are re-excited after the trauma, they may send safety signals to the fear circuits, which may prevent PTSD development (Lee et al., 2016). Nevertheless, the precise mechanism underlying music-mediated amelioration of PTSD symptoms through VTA dopaminergic action deserves further investigation.
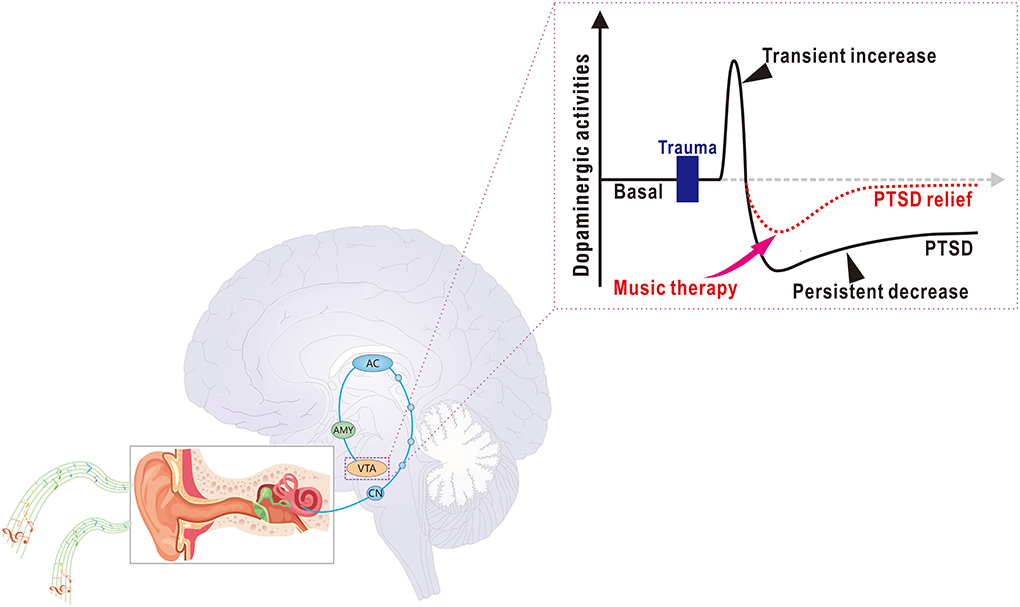
Figure 2. Schematic illustration of music therapy on post-traumatic stress disorder (PTSD) through dopaminergic action. A simplified neural pathway for acoustic processes from the cochlea to the auditory cortex. The auditory cortex sends projections to the amygdala and then to the ventral tegmental area (VTA) (Koelsch, 2014). Inset, the DA level is transiently elevated after trauma, followed by persistent hypodopaminergia (Corralfrias et al., 2013). Music activates the VTA function and stimulates the DA release, which may reduce PTSD-induced hypodopaminergia in the brain, which ameliorates PTSD symptoms. CN, cochlear nucleus; AC, auditory cortex; AMY, amygdala; VTA, ventral tegmental area.
Several other lines of evidence may also support the mitigation of PTSD symptoms via musical therapy by affecting mesolimbic dopaminergic actions. For example, PTSD is featured with anhedonia, which may associate with deficits in reward functioning (Enman et al., 2015), whereas music-stimulated VTA dopaminergic activation involves reward-value coding by organizing music into precise reinforcements (Menon and Levitin, 2005; Stegemöller, 2014; Zatorre, 2015). In addition, PTSD is also accompanied with social dissociation, while VTA dopaminergic activation promotes prosocial interactions (Hung et al., 2017; McHenry et al., 2017; Harvey, 2020; Wang et al., 2021), such as the enhancement of interpersonal trust, positive cooperation, and social connectedness among individuals (Chanda and Levitin, 2013; Harvey, 2018), which may improve symptoms in PTSD patients.
To our knowledge, the present review is the first to summarize the effects of music therapy on PTSD associated with VTA dopaminergic action; that is, we assessed whether music enhances VTA dopaminergic action, which compensates for PTSD-induced hypodopaminergia and consequently ameliorates PTSD symptoms. This review provides suggestions for clinicians and/or policymakers to develop standardized guidelines for the use of music therapy on PTSD. In addition, although this review substantiates a role for music therapy on PTSD symptoms associated with VTA dopaminergic action, the precise mechanism underlying the process remains largely enigmatic. It is our hope that the present review will inspire additional studies that evaluate the use of music therapy on PTSD.
Notably, the current review has several limitations. First, the paper was not prepared following a rigorous methodology of Preferred Reporting Items for Systematic Reviews and Meta-Analyses due to the sample size. We searched 19 original articles concerning music therapy on PTSD, six of which reported cases or without controls, and eight included small populations (only 5–16) of participants. Small sample sizes may result in substantial heterogeneity among participants and result in heterogeneity in the intervention effects (de Witte et al., 2022). Second, it is challenging to reach a standardized protocol for music therapy on PTSD due to the diverse methods employed across investigations, such as music intervention (receptive listening vs. active creation), music induction (prerecorded music vs. live music), music selection (pre-selected by the therapist vs. preferred by the patients), and music familiarity (familiar music vs. unfamiliar music) (Landis-Shack et al., 2017; Pant et al., 2022). Third, the unavoidable factor of publication bias could also affect the accuracy of the conclusions, as positive and significant outcomes are more frequently published relative to the non-significant or negative results (de Witte et al., 2019, 2022); i.e., the results reported in publications do not represent all findings.
This review supports that music serves as an effective approach for treating PTSD that is associated with VTA dopaminergic activation. It should be noted that music therapy affects various aspects of physiological functions, such as various other brain circuits [e.g., mPFC-amygdala neurocircuit (Koelsch, 2014; Reybrouck et al., 2021)], neurotransmitters [e.g., oxytocin (Beck et al., 2018a; Harvey, 2020)], and immunological responsiveness [e.g., the reaction of the HPA axis (Reybrouck et al., 2018; Pant et al., 2022)], in addition to dopaminergic activities. Whether and how these different factors coordinate with VTA dopaminergic tone to treat PTSD symptoms warrants further investigation. Perhaps, music therapy could be combined with other methods to provide better therapeutic outcomes of PTSD intervention.
MN and CZ conceived and designed the manuscript. MN, SW, and PZ searched and investigated the literatures. PZ and SW prepared the original draft. PZ and CZ wrote the paper. All authors read and approved the final manuscript.
This work was supported by grants from the National Natural Science Foundation of China (81871062) and the Natural Science Foundation of Guangdong Province (2019A1515010331), and the key Project of Humanities and Social Sciences of Anhui Provincial Education Department (2017SK27).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Akiki, T. J., and Abdallah, C. G. (2018). Are there effective psychopharmacologic treatments for PTSD? J. Clin. Psychiatry 80, 12473. doi: 10.4088/JCP.18ac12473
American Psychiatric Association (2013). Diagnostic and Statistical Manual of Mental Disorders. 5th Edn. Washington, DC: American Psychiatric Association.
Baranyi, G., Cassidy, M., Fazel, S., Priebe, S., and Mundt, A. P. (2018). Prevalence of posttraumatic stress disorder in prisoners. Epidemiol. Rev. 40, 134–145. doi: 10.1093/epirev/mxx015
Beck, B. D., Lund, S. T., Søgaard, U., Simonsen, E., Tellier, T. C., Cordtz, T. O., et al. (2018a). Music therapy versus treatment as usual for refugees diagnosed with posttraumatic stress disorder (PTSD): Study protocol for a randomized controlled trial. Trials 19, 301. doi: 10.1186/s13063-018-2662-z
Beck, B. D., Messel, C., Meyer, S. L., Cordtz, T. O., Søgaard, U., Simonsen, E., et al. (2018b). Feasibility of trauma-focused guided imagery and music with adult refugees diagnosed with PTSD: A pilot study. Nord. J. Music Ther. 27, 67–86. doi: 10.1080/08098131.2017.1286368
Beck, B. D., Meyer, S. L., Simonsen, E., Søgaard, U., Petersen, I., Arnfred, S. M. H., et al. (2021). Music therapy was noninferior to verbal standard treatment of traumatized refugees in mental health care: results from a randomized clinical trial. Eur. J. Psychotraumatol. 12, 1930960. doi: 10.1080/20008198.2021.1930960
Belfi, A. M., and Loui, P. (2020). Musical anhedonia and rewards of music listening: current advances and a proposed model. Ann. N. Y. Acad. Sci. 1464, 99–114. doi: 10.1111/nyas.14241
Bensimon, M., Amir, D., and Wolf, Y. (2008). Drumming through trauma: music therapy with post-traumatic soldiers. Art. Psychother. 35, 34–48. doi: 10.1016/j.aip.2007.09.002
Bensimon, M., Amir, D., and Wolf, Y. (2012). A pendulum between trauma and life: group music therapy with post-traumatized soldiers. Art. Psychother. 39, 223–233. doi: 10.1016/j.aip.2012.03.005
Bisson, J. I., Cosgrove, S., Lewis, C., and Robert, N. P. (2015). Post-traumatic stress disorder. Brit. Med. J. 351, h6161. doi: 10.1136/bmj.h6161
Blanaru, M., Bloch, B., Vadas, L., Arnon, Z., Ziv, N., Kremer, I., et al. (2012). The effects of music relaxation and muscle relaxation techniques on sleep quality and emotional measures among individuals with posttraumatic stress disorder. Ment. Illn. 4, e13. doi: 10.4081/mi.2012.e13
Boso, M., Politi, P., Barale, F., and Enzo, E. (2006). Neurophysiology and neurobiology of the musical experience. Funct. Neurol. 21, 187–191.
Bradley, R., Greene, J., Russ, E., Dutra, L., and Westen, D. (2005). A multidimensional meta-analysis of psychotherapy for PTSD. Am. J. Psychiatry 162, 214–227. doi: 10.1176/appi.ajp.162.2.214
Brown, L. A., Chen, S., Narine, K., Contractor, A. A., and Oslin, D. (2020). DSM-5 PTSD symptom clusters and suicidal ideation in veterans. Psychiatry Res. 288, 112942. doi: 10.1016/j.psychres.2020.112942
Carr, C., D'ardenne, P., Sloboda, A., Scott, C., Wang, D., and Priebe, S. (2012). Group music therapy for patients with persistent post-traumatic stress disorder–an exploratory randomized controlled trial with mixed methods evaluation. Psychol. Psychother. 85, 179–202. doi: 10.1111/j.2044-8341.2011.02026.x
Chanda, M. L., and Levitin, D. J. (2013). The neurochemistry of music. Trends Cogn. Sci. 17, 179–193. doi: 10.1016/j.tics.2013.02.007
Corralfrias, N. S., Lahood, R. P., Edelmanvogelsang, K. E., French, E. D., and Fellous, J. M. (2013). Involvement of the ventral tegmental area in a rodent model of post-traumatic stress disorder. Neuropsychopharmacology 38, 350–363. doi: 10.1038/npp.2012.189
Coulter, S. J. (2000). Effect of song writing versus recreational music on posttraumatic stress disorder (PTSD) symptoms and abuse attribution in abused children. J. Poetry Ther. 13, 189–208. doi: 10.1023/A:1021402618865
de Witte, M., Pinho, A. D. S., Stams, G. J., Moonen, X., Bos, A. E. R., and van Hooren, S. (2022). Music therapy for stress reduction: a systematic review and meta-analysis. Health Psychol. Rev. 16, 134–159. doi: 10.1080/17437199.2020.1846580
de Witte, M., Spruit, A., van Hooren, S., Moonen, X., and Stams, G. J. (2019). Effects of music interventions on stress-related outcomes: a systematic review and two meta-analyses. Health Psychol. Rev. 14, 294–324. doi: 10.1080/17437199.2019.1627897
Eiset, A. H., Aoun, M. P., Stougaard, M., Gottlieb, A. G., Haddad, R. S., Frydenberg, M., et al. (2021). The prevalence of post-traumatic stress disorder increased in refugees after long-distance migration. Eur. J. Public Health 22, 363. doi: 10.1186/s12888-022-03982-4
Emanuele, E., Boso, M., Cassola, F., Broglia, D., Bonoldi, I., Mancini, L., et al. (2010). Increased dopamine DRD4 receptor mRNA expression in lymphocytes of musicians and autistic individuals: bridging the music-autism connection. Neuroendocrinol. Lett. 31, 122–125. doi: 10.1159/000287255
Enman, N. M., Arthur, K., Ward, S. J., Perrine, S. A., and Unterwald, E. M. (2015). Anhedonia, reduced cocaine reward, and dopamine dysfunction in a rat model of posttraumatic stress disorder. Biol. Psychiatry 78, 871–879. doi: 10.1016/j.biopsych.2015.04.024
Feduccia, A. A., and Duvauchelle, C. L. (2008). Auditory stimuli enhance MDMA-conditioned reward and MDMA-induced nucleus accumbens dopamine, serotonin and locomotor responses. Brain Res. Bull. 77, 189–196. doi: 10.1016/j.brainresbull.2008.07.007
Fenster, R. J., Lebois, L. A. M., Ressler, K. J., and Suh, J. (2018). Brain circuit dysfunction in post-traumatic stress disorder: from mouse to man. Nat. Rev. Neurosci. 535–551. doi: 10.1038/s41583-018-0039-7
Ferreri, L., Mas-Herrero, E., Zatorre, R. J., Ripollés, P., Gomez-Andres, A., Alicart, H., et al. (2019). Dopamine modulates the reward experiences elicited by music. Proc. Natl. Acad. Sci. U. S. A. 116, 3793–3798. doi: 10.1073/pnas.1811878116
Guina, J., Rossetter, S. R., De, R. B., Nahhas, R. W., and Welton, R. S. (2015). Benzodiazepines for PTSD: a systematic review and meta-snalysis. J. Psychiatr. Pract. 21, 281–303. doi: 10.1097/PRA.0000000000000091
Hakvoort, L., De Jong, S., Van De Ree, M., Kok, T., Macfarlane, C., and De Haan, H. (2020). Music therapy to regulate arousal and attention in patients with substance use disorder and posttraumatic stress disorder: a feasibility study. J. Music Ther. 57, 353–378. doi: 10.1093/jmt/thaa007
Harvey, A. G., Bryant, R. A., and Tarrier, N. (2003). Cognitive behaviour therapy for posttraumatic stress disorder. Clin. Psychol. Rev. 23, 501–522. doi: 10.1016/S0272-7358(03)00035-7
Harvey, A. R. (2018). Music and the meeting of human minds. Front. Psychol. 9, 762. doi: 10.3389/fpsyg.2018.00762
Harvey, A. R. (2020). Links between the neurobiology of oxytocin and human musicality. Front. Hum. Neurosci. 14, 350. doi: 10.3389/fnhum.2020.00350
Hayes, J. P., Hayes, S. M., and Mikedis, A. M. (2012). Quantitative meta-analysis of neural activity in posttraumatic stress disorder. Biol. Mood Anxiety Disord. 2, 9. doi: 10.1186/2045-5380-2-9
Hirschberg, R., Sylvia, L. G., Wright, E. C., Gupta, C. T., Mccarthy, M. D., Harward, L. K., et al. (2020). Collaborative songwriting intervention for veterans with post-traumatic stress disorder. J. Altern. Complement. Med. 26, 198–203. doi: 10.1089/acm.2019.0368
Houlihan, D. J. (2011). Psychostimulant treatment of combat-related posttraumatic stress disorder. J. Psychopharmacol. 25, 1568–1572. doi: 10.1177/0269881110385600
Hung, L. W., Neuner, S., Polepalli, J. S., Beier, K. T., Wright, M., Walsh, J. J., et al. (2017). Gating of social reward by oxytocin in the ventral tegmental area. Science 357, 1406–1411. doi: 10.1126/science.aan4994
Järvelä, I. (2018). Genomics studies on musical aptitude, music perception, and practice. Ann. N. Y. Acad. Sci. 1423, 82–91. doi: 10.1111/nyas.13620
Jespersen, K. V., and Vuust, P. (2012). The effect of relaxation music listening on sleep quality in traumatized refugees: a pilot study. J. Music Ther. 49, 205–229. doi: 10.1093/jmt/49.2.205
Jordan, B. K., Marmar, C. R., Fairbank, J. A., Schlenger, W. E., and Al, E. (1992). Problems in families of male Vietnam veterans with posttraumatic stress disorder. J. Consult. Clin. Psychol. 60, 916–926. doi: 10.1037/0022-006X.60.6.916
Kanduri, C., Kuusi, T., Ahvenainen, M., Philips, A. K., Lähdesmäki, H., and Järvelä, I. (2015a). The effect of music performance on the transcriptome of professional musicians. Sci. Rep. 5, 9506. doi: 10.1038/srep09506
Kanduri, C., Raijas, P., Ahvenainen, M., Philips, A. K., Ukkola-Vuoti, L., Lähdesmäki, H., et al. (2015b). The effect of listening to music on human transcriptome. Peer J. 3, e830. doi: 10.7717/peerj.830
Koelsch, S. (2014). Brain correlates of music-evoked emotions. Nat. Rev. Neurosci. 15, 170–180. doi: 10.1038/nrn3666
Koelsch, S., and Siebel, W. A. (2005). Towards a neural basis of music perception. Trends Cogn. Sci. 9, 578–584. doi: 10.1016/j.tics.2005.10.001
Kraus, K. S., and Canlon, B. (2012). Neuronal connectivity and interactions between the auditory and limbic systems. Effects of noise and tinnitus. Hear. Res. 288, 34–46. doi: 10.1016/j.heares.2012.02.009
Landis-Shack, N., Heinz, A. J., and Bonn-Miller, M. O. (2017). Music therapy for posttraumatic stress in adults: a theoretical review. Psychomusicology 27, 334–342. doi: 10.1037/pmu0000192
Lee, J. C., Wang, L. P., and Tsien, J. Z. (2016). Dopamine rebound-excitation theory: putting brakes on PTSD. Front. Psychiatry 7, 163. doi: 10.3389/fpsyt.2016.00163
Lightstone, A. J., Bailey, S. K., and Voros, P. (2015). Collaborative music therapy via remote video technology to reduce a veteran's symptoms of severe, chronic PTSD. Arts Health 7, 123–136. doi: 10.1080/17533015.2015.1019895
Liu, X., Kanduri, C., Oikkonen, J., Karma, K., and Jrvel, I. (2016). Detecting signatures of positive selection associated with musical aptitude in the human genome. Sci. Rep. 6, 21198. doi: 10.1038/srep21198
Luo, C., Fan, H., Li, S., and Zou, Y. (2021). Therapeutic of candesartan and music therapy in diabetic retinopathy with depression in rats. Evid. Based Comp. Alternat. Med. 2021, 5570356. doi: 10.1155/2021/5570356
Maack, C. (2012). Outcomes and Processes of the Bonny Method of Guided Imagery and Music (GIM) and Its Adaptations and Psychodynamic Imaginative Trauma Therapy (PITT) for Women With Complex PTSD (Ph.D. thesis), Denmark: Aalborg University.
Macfarlane, C., Masthoff, E., and Hakvoort, L. (2019). Short-term music therapy attention and arousal regulation treatment (SMAART) for prisoners with posttraumatic stress disorder: a feasibility study. J. Forensic. Psychol. Res. Pract. 19, 376–392. doi: 10.1080/24732850.2019.1670023
Mariath, L. M., Silva, A. M., Kowalski, T. W., Gattino, G. S., Araujo, G. A., Figueiredo, F. G., et al. (2017). Music genetics research: association with musicality of a polymorphism in the AVPR1A gene. Genet. Mol. Biol. 40, 421–429. doi: 10.1590/1678-4685-gmb-2016-0021
Masand, P. S., and Gupta, S. (2002). Long-term side effects of newer-generation antidepressants: SSRIS, venlafaxine, nefazodone, bupropion, and mirtazapine. Ann. Clin. Psychiatry 14, 175–182. doi: 10.3109/10401230209147454
McHenry, J. A., Otis, J. M., Rossi, M. A., Robinson, J. E., Kosyk, O., Miller, N. W., et al. (2017). Hormonal gain control of a medial preoptic area social reward circuit. Nat. Neurosci. 20, 449–458. doi: 10.1038/nn.4487
McLaughlin, T., Febo, M., Badgaiyan, R. D., Barh, D., Dushaj, K., Braverman, E. R., et al. (2016). KB220Z™ a pro-dopamine regulator associated with the protracted, alleviation of terrifying lucid dreams. Can we infer neuroplasticity-induced changes in the reward circuit? J. Reward Defic. Syndr. Addict. Sci. 2, 3–13. doi: 10.17756/jrdsas.2016-022
Menon, V., and Levitin, D. J. (2005). The rewards of music listening: response and physiological connectivity of the mesolimbic system. Neuroimage 28, 175–184. doi: 10.1016/j.neuroimage.2005.05.053
Moraes, M. M., Rabelo, P. C. R., Pinto, V. A., Pires, W., Wanner, S. P., Szawka, R. E., et al. (2018). Auditory stimulation by exposure to melodic music increases dopamine and serotonin activities in rat forebrain areas linked to reward and motor control. Neurosci. Lett. 673, 73–78. doi: 10.1016/j.neulet.2018.02.058
Nair, P. S., Raijas, P., Ahvenainen, M., Philips, A. K., Ukkola-Vuoti, L., and Järvelä, I. (2021). Music-listening regulates human microRNA expression. Epigenetics 16, 554–566. doi: 10.1080/15592294.2020.1809853
Nair, V. D., Olanow, C. W., and Sealfon, S. C. (2003). Activation of phosphoinositide 3-kinase by D2 receptor prevents apoptosis in dopaminergic cell lines. Biochem. J. 373, 25–32. doi: 10.1042/bj20030017
Oikkonen, J., Huang, Y., Onkamo, P., Ukkola-Vuoti, L., Raijas, P., Karma, K., et al. (2015). A genome-wide linkage and association study of musical aptitude identifies loci containing genes related to inner ear development and neurocognitive functions. Mol. Psychiatry 20, 275–282. doi: 10.1038/mp.2014.8
Orth, J. (2001). Between abandoning and control: structure, security and expression in music therapy with traumatized refugees in a psychiatric clinic. Trauma Empowerment 1, 189–197.
Orth, J. (2005). Music therapy with traumatized refugees in a clinical setting. Voices 5, 1–13. doi: 10.15845/voices.v5i2.227
Pant, U., Frishkopf, M., Park, T., Norris, C. M., and Papathanassoglou, E. (2022). A neurobiological framework for the therapeutic potential of music and sound interventions for post-traumatic stress symptoms in critical illness survivors. Int. J. Environ. Res. Public Health 19, 3113. doi: 10.3390/ijerph19053113
Pezzin, L. E., Larson, E. R., Lorber, W., Mcginley, E. L., and Dillingham, T. R. (2018). Music-instruction intervention for treatment of post-traumatic stress disorder: a randomized pilot study. BMC Psychol. 6, 60. doi: 10.1186/s40359-018-0274-8
Pitman, R. K., Rasmusson, A. M., Koenen, K. C., Shin, L. M., Orr, S. P., Gilbertson, M. W., et al. (2012). Biological studies of post-traumatic stress disorder. Nat. Rev. Neurosci. 13, 769–787. doi: 10.1038/nrn3339
Polston, J. E., Rubbinaccio, H. Y., Morra, J. T., Sell, E. M., and Glick, S. D. (2011). Music and methamphetamine: conditioned cue-induced increases in locomotor activity and dopamine release in rats. Pharmacol. Biochem. Behav. 98, 54–61. doi: 10.1016/j.pbb.2010.11.024
Pourmovahed, Z., Ardekani, S., Roozbeh, B., and Ezabad, A. R. (2021). The effect of non-verbal music on posttraumatic stress disorder in mothers of premature neonates. Iran. J. Nurs. Midwifery Res. 26, 150. doi: 10.4103/ijnmr.IJNMR_37_20
Precin, P. (2011). Occupation as therapy for trauma recovery: a case study. Work 38, 77–81. doi: 10.3233/WOR-2011-1106
Quarto, T., Fasano, M. C., Taurisano, P., Fazio, L., Antonucci, L. A., Gelao, B., et al. (2017). Interaction between DRD2 variation and sound environment on mood and emotion-related brain activity. Neuroscience 341, 9–17. doi: 10.1016/j.neuroscience.2016.11.010
Rahman, Z., Schwarz, J., Gold, S. J., Zachariou, V., Wein, M. N., Choi, K. H., et al. (2003). RGS9 modulates dopamine signaling in the basal ganglia. Neuron 38, 941–952. doi: 10.1016/S0896-6273(03)00321-0
Ramo-Fernández, L., Schneider, A., Wilker, S., and Kolassa, I. T. (2015). Epigenetic alterations associated with war trauma and childhood maltreatment. Behav. Sci. Law 33, 701–721. doi: 10.1002/bsl.2200
Reybrouck, M., Vuust, P., and Brattico, E. (2018). Music and Brain Plasticity: How Sounds Trigger Neurogenerative Adaptations. Neuroplasticity - Insights of Neural Reorganization. London: IntechOpen.
Reybrouck, M., Vuust, P., and Brattico, E. (2021). Neural correlates of music listening: Does the music matter? Brain Sci. 11, 1553. doi: 10.3390/brainsci11121553
Rudstam, G., Elofsson, U., Sndergaard, H. P., Bonde, L. O., Amp, B., and Beck, D. (2017). Trauma-focused group music and imagery with women suffering from ptsd/complex ptsd: a feasibility study. Approaches 9, 202–216.
Rudstam, G., Elofsson, U. O. E., Söndergaard, H. P., Bonde, L. O., and Beck, B. D. (2022). Trauma-focused group music and imagery with women suffering from PTSD/Complex PTSD: a randomized controlled study. Eur. J. Trauma Dissoc. 6, 100277. doi: 10.1016/j.ejtd.2022.100277
Salimpoor, V. N., Benovoy, M., Larcher, K., Dagher, A., and Zatorre, R. J. (2011). Anatomically distinct dopamine release during anticipation and experience of peak emotion to music. Nat. Neurosci. 14, 257–262. doi: 10.1038/nn.2726
Salimpoor, V. N., and Zatorre, R. J. (2013). Neural interactions that give rise to musical pleasure. Psychol. Aesthet. CreaT. 7, 62–75. doi: 10.1037/a0031819
Shalev, A., Liberzon, I., and Marmar, C. (2017). Post-traumatic stress disorder. N. Engl. J. Med. 376, 2459–2469. doi: 10.1056/NEJMra1612499
Somayaji, M., Lanseur, Z., Choi, S. J., Sulzer, D., and Mosharov, E. V. (2021). Roles for α-Synuclein in gene expression. Genes 12, 1166. doi: 10.3390/genes12081166
Stegemöller, E. L. (2014). Exploring a neuroplasticity model of music therapy. J. Music Ther. 51, 211–227. doi: 10.1093/jmt/thu023
Story, K. M., and Beck, B. D. (2017). Guided imagery and music with female military veterans: an intervention development study. Art. Psychother. 55, 93–102. doi: 10.1016/j.aip.2017.05.003
Sutoo, D. E., and Akiyama, K. (2004). Music improves dopaminergic neurotransmission: demonstration based on the effect of music on blood pressure regulation. Brain Res. 1016, 255–262. doi: 10.1016/j.brainres.2004.05.018
Tasset, I., Quero, I., García-Mayórgaz, D., Río, M., Túnez, I., and Montilla, P. (2012). Changes caused by haloperidol are blocked by music in Wistar rat. J. Physiol. Biochem. 68, 175–179. doi: 10.1007/s13105-011-0129-8
Ukkola, L. T., Onkamo, P., Raijas, P., Karma, K., and Järvelä, I. (2009). Musical aptitude is associated with AVPR1A-haplotypes. PLoS ONE 4, e5534. doi: 10.1371/journal.pone.0005534
Ungless, M. A., Whistler, J. L., Malenka, R. C., and Bonci, A. (2001). Single cocaine exposure in vivo induces long-term potentiation in dopamine neurons. Nature 411, 583–587. doi: 10.1038/35079077
Volkman, S. (1993). Music therapy and the treatment of trauma-induced dissociative disorders. Art. Psychother. 20, 243–251. doi: 10.1016/0197-4556(93)90019-X
Von Der Warth, R., Dams, J., Grochtdreis, T., and König, H. H. (2020). Economic evaluations and cost analyses in posttraumatic stress disorder: a systematic review. Eur. J. Psychotraumatol. 11, 1753940. doi: 10.1080/20008198.2020.1753940
Wang, J., Li, J., Yang, Q., Xie, Y. K., Wen, Y. L., Xu, Z. Z., et al. (2021). Basal forebrain mediates prosocial behavior via disinhibition of midbrain dopamine neurons. Proc. Natl. Acad. Sci. U. S. A. 118, e2019295118. doi: 10.1073/pnas.2019295118
Wilson, G., Farrell, D., Barron, I., Hutchins, J., Whybrow, D., and Kiernan, M. D. (2018). The use of eye-movement desensitization reprocessing (EMDR) therapy in treating post-traumatic stress disorder-A systematic narrative review. Front. Psychol. 9, 923. doi: 10.3389/fpsyg.2018.00923
Yamamoto, T., Ohkuwa, T., Itoh, H., Kitoh, M., Terasawa, J., Tsuda, T., et al. (2003). Effects of pre-exercise listening to slow and fast rhythm music on supramaximal cycle performance and selected metabolic variables. Arch. Physiol. Biochem. 111, 211–214. doi: 10.1076/apab.111.3.211.23464
Yehuda, R., Hoge, C. W., Mcfarlane, A. C., Vermetten, E., Lanius, R. A., Nievergelt, C. M., et al. (2015). Post-traumatic stress disorder. Nat. Rev. Dis. Primers 1, 15057. doi: 10.1038/nrdp.2015.57
Zatorre, R. J. (2015). Musical pleasure and reward: mechanisms and dysfunction. Ann. N. Y. Acad. Sci. 1337, 202–211. doi: 10.1111/nyas.12677
Zergani, E. J., and Naderi, F. (2016). The effectiveness of music on quality of life and anxiety symptoms in the post traumatic stress disorder in Bustan hospital of Ahvaz city. Rev. Eur. Stud. 8, 237–243. doi: 10.5539/res.v8n3p237
Zhou, P., Deng, M., Wu, J., Lan, Q., Yang, H., and Zhang, C. (2021). Ventral tegmental area dysfunction and disruption of dopaminergic homeostasis: implications for post-traumatic stress disorder. Mol. Neurobiol. 58, 2423–2434. doi: 10.1007/s12035-020-02278-6
Keywords: music therapy, post-traumatic stress disorder, symptom improvement, ventral tegmental area, dopaminergic action
Citation: Ning M, Wen S, Zhou P and Zhang C (2022) Ventral tegmental area dopaminergic action in music therapy for post-traumatic stress disorder: A literature review. Front. Psychol. 13:1014202. doi: 10.3389/fpsyg.2022.1014202
Received: 08 August 2022; Accepted: 21 September 2022;
Published: 10 October 2022.
Edited by:
Lawrence M. Parsons, The University of Sheffield, United KingdomReviewed by:
Carina Freitas, University of Madeira, PortugalCopyright © 2022 Ning, Wen, Zhou and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Changzheng Zhang, bmV1cm96aGFuZ0AxNjMuY29t; Peiling Zhou, cGVpbGluZ3pob3V2aXBAMTYzLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.