- 1Department of Medicine, Division of Hematology/Oncology, Icahn School of Medicine at Mount Sinai, Tisch Cancer Institute, New York, NY, Unites States
- 2Feil Family Brain and Mind Research Institute, Weill Cornell Medicine, New York, NY, Unites States
Upon accumulation of misfolded proteins in the mitochondria, the mitochondrial unfolded protein response (UPRmt) is activated. This review focuses on the role of this response in cancer. We discuss evidence that during transformation, the UPRmt may play an essential role in the maintenance of the integrity of the mitochondria in the face of increased oxidative stress. However, the role of the UPRmt in other diseases is also emerging and is therefore also briefly discussed.
Introduction
The proper folding of proteins is fundamental to cellular life. Perturbations to this process promote the formation of protein aggregates and underlie a number of human pathologies.
Aggregation of proteins in the lumen of organelles represents an additional challenge, as they cannot be directly presented to the ubiquitin/proteasome system. The discovery of the endoplasmic reticulum mediated degradation was originally made in yeast and revealed the first mechanism of how misfolded proteins in the lumen of the endoplasmic reticulum can be retro-translocated to the cytoplasm and ubiquitinated for their degradation by the proteasome [for a recent review (1)]. Further, the accumulation of misfolded proteins in the lumen of the endoplasmic reticulum was found to lead to the transcription of the endoplasmic reticulum chaperone, BiP (KAR2) (2). Using a genetic screen, the same authors identified inositol-requiring transmembrane kinase/endonuclease (IRE1) as the first sensor of the UPRER, which communicates the proteotoxic stress to the nucleus (3, 4). While IRE1 is the sole sensor of the UPRER in yeast, higher eukaryotes have three distinct axes of the UPRER—IRE, PERK, and ATF6, all of which represent independent signaling cascades that activate separate pathways to cumulatively reduce proteotoxic stress and maintain organelle homeostasis (5, 6). The study of the UPRER identified CHOP as a transcription factor essential for this response. More recently, a similar mechanism has been proposed to take place for the elimination of misfolded proteins inside the mitochondria (7, 8). The initial axis of the mitochondrial unfolded protein response (UPRmt) to be discovered also implicates CHOP (8). However, as described in details below, the effect of CHOP is context dependent and there is no overlap between the UPRER and UPRmt in that stress in the lumen of the endoplasmic reticulum do not activate the UPRmt and vice versa.
Further, in contrast to the endoplasmic reticulum, the mitochondrial matrix is rich in chaperones and proteases and is therefore well equipped to manage the accumulation of misfolded proteins. In fact, an even more recent study described a critical role of the mitochondria in the management of cytoplasmic proteostasis (9). In this study, the authors found that, upon heat shock stress in yeast, cytosolic proteins that are prone to aggregation are imported into the mitochondria for degradation (9), a phenomenon they named MAGIC (mitochondria as guardian in cytosol). While this concept is intriguing, how misfolded proteins are transported from the cytoplasm into the mitochondria remains unclear. One possibility is that cytosolic chaperones may partially unfold misfolded proteins allowing them to enter the mitochondria. Importantly for this review, the same study reported that import of cytosolic misfolded proteins was found in both the inter-membrane space (IMS) and the matrix of the mitochondria. How the localization of misfolded proteins within the mitochondria after import from the cytosol is determined also remains to be determined. Nevertheless, if cytosolic proteins can indeed be imported in both the matrix and the IMS, this would have major implications for the UPRmt since, unlike to matrix, the IMS has limited protein quality control and no heat shock proteins. Therefore, raising the question as to how accumulation of misfolded proteins in the IMS would help alleviate proteotoxic stress.
Misfolded Proteins in the Mitochondrial IMS
If MAGIC is a conserved mechanism in mammals, it is predicted to lead to the accumulation of misfolded proteins in the IMS, as a result of their import from the cytosol under stress conditions. In addition to import of cytosolic proteins in the mitochondria, accumulation of misfolded proteins in the IMS can also arise within the mitochondria itself. This effect may be especially true in cancer cells that are characterized by increased levels of reactive oxygen species (ROS), which cause oxidation of proteins and their misfolding.
The elevation in ROS in cancer cells combined with the Warburg effect, which refers to the elevation in glycolysis for the production of ATP, have led to the misconception that cancer cells have defective mitochondria. In contrast, it is now recognized that most cancer cells continue to require oxidative phosphorylation. This observation has led to the idea of the reverse-Warburg effect, the recognition of oxidative tumors, and metabolic flexibility [for a recent review (10)]. ROS contribute to the reprogramming of the mitochondrial of cancer cells and have been shown to play a causative role in tumorigenesis and cancer progression (11–13). While elevation in ROS levels benefit cancer cells by promoting genomic instability and metabolic reprogramming, if left uncontrolled leading to excessive levels, ROS can cause severe DNA damage, oxidation of lipids and proteins, and cause cell death (13). Therefore, cancer cells must acquire mechanisms to keep their ROS levels within a window that is compatible with the maintenance of the integrity of the organelle. Given that the primary site of ROS production in the mitochondria is the electron transport chain (ETC) of the mitochondrial inner-membrane, the mitochondria of cancer cells are particularly vulnerable to oxidative stress. Mitochondria are double membrane bound organelles composed of the outer and inner-membranes, thereby creating two sub-compartments. The mitochondrial matrix contains the mitochondrial genome and mitochondrial-specific ribosomes for the translation of mitochondrial encoded proteins as well as a multitude of well-characterized enzymes involved in intermediary metabolism. The IMS, however, has been largely overlooked and is often perceived merely as a storage space for pro-apoptotic proteins, until they are released into the cytosol for the execution of apoptosis. In contrast, over 100 proteins reside in the IMS, representing roughly 10% of the mitochondrial proteome. IMS proteins are actively involved in metabolism, protein import, oxidative protein folding, ETC complex assembly, export of ferrous precursors, and transport of metabolites, metal ions, and lipids (14). Because ETC-generated ROS is produced on both sides of the inner-membrane (15), ROS-mediated misfolding of proteins also occurs in the IMS. In addition, oxidative protein folding, which occurs only in the IMS and the endoplasmic reticulum, is a process by which proteins are folded into proper conformations through the formation of disulfide bonds, and in so doing produce one molecule of ROS per cycle of folding (14). ROS produced by this process can also contribute to the misfolding and aggregation of IMS proteins (15). Therefore, given the fact that the IMS of the mitochondria has very little capacity to handle misfolded protein when compared with that of the mitochondrial matrix, the IMS appears poised for the accumulation of misfolded proteins.
Our group has previously sought to understand the mechanisms by which misfolded proteins in the IMS of the mitochondria are managed and resolved in the context of cancer cells. Using a mutant form of endonuclease G (EndoG), which misfolds and forms protein aggregates in the IMS, we first reported that the proteasome in the cytosol and the protease OMI in the IMS cooperate to limit the accumulation of misfolded proteins in the IMS (16). We proposed that the proteasome acts as a pre-import checkpoint, while OMI acts a post-import checkpoint (16).
The Brief Summary of the Discovery of the UPRmt
The UPRmt was originally identified in mammalian cells using the overexpression of mitochondrial matrix localized misfolded OTCΔ (17). This first axis of the UPRmt was found to be mediated through the transcription factor CHOP leading to the upregulation of a number of mitochondrial chaperones and proteases, such as ClpP, hsp10, and hsp60 (17). This effect was shown to be mediated through binding of CHOP to mitochondrial upstream elements (18). Further, it was shown that binding of CHOP to the promoters of target genes in response to mitochondrial proteotoxic stress was dependent on AP-1 (18–20). Mitochondrial matrix proteotoxic stress did not lead to activation of UPRER genes despite CHOP being implicated in the UPRER, suggesting that AP-1 provides the context specificity of CHOP.
Since its discovery, much of the work in the field of the UPRmt has been focused on this axis, with particular emphasis on the chaperone hsp60, which has been used extensively as a reporter of the UPRmt in genetic screens to identify players of the UPRmt in Caenorhabditis elegans. Using this model system, ATFS-1 and the DVE-1/UBL5 complex have been identified as important transcriptional activators of the UPRmt (21–33).
More recently, the transcription factor ATF5 was identified as the mammalian ortholog of ATFS-1 (34). As ATF5 has been shown to act downstream of CHOP (35) and both CHOP and ATF5/ATFS-1 activation leads to induction of mitochondrial chaperones and proteases, CHOP and ATF5 reside in the same axis of the UPRmt, which is therefore referred to as the CHOP axis (Figure 1). While a body of literature already exists around the role of ATF5 in cancer biology, notably in the regulation of apoptosis (36), it will be interesting to further investigate the role of ATF5 in the context of the UPRmt and cancer.
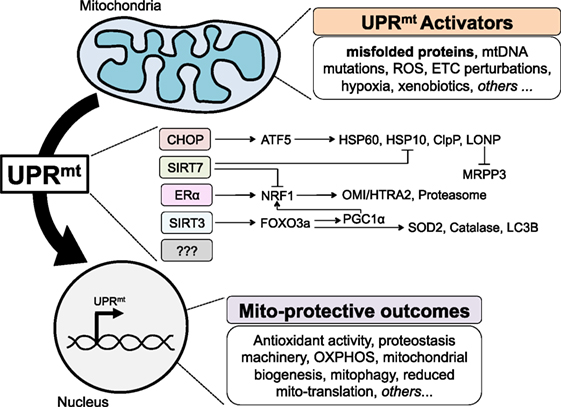
Figure 1. The mammalian mitochondrial unfolded protein response (UPRmt), to date. Various perturbations to mitochondria homeostasis, including misfolded proteins, activate currently known axes of the UPRmt to signal to the nucleus in a retrograde fashion, and induce gene expression that results in a number of mito-protective outcomes.
The CHOP axis of the UPRmt and its role in cancer biology has been recently addressed, mostly focusing on ClpP, the AAA+ peptidase subunit of the ClpXP, a complex that is induced by CHOP. ClpP was identified using a genetic screen for the viability of leukemic cells (37). The authors then showed that ClpP inhibition had potent antitumor effects both in vitro and in vivo (37). In a separate study, a proteomic screen for proteins associated with the oncoprotein survivin led to the identification of ClpP (38). Further, ClpP was found to be overexpressed in almost every solid tumor type and analysis of patient databases showed that elevated ClpP expression significantly correlated with worse clinical outcomes (38).
Because of the limited extent of the protein quality control mechanisms in the IMS, we wanted to determine whether the CHOP axis of the UPRmt is also activated upon proteotoxic stress in the IMS. A mutant form of the IMS resident protein EndoG was used as a tool to target misfolded proteins specifically to the IMS. To our surprise, we found that in estrogen receptor alpha (ERα) positive breast cancer cells, accumulation of misfolded proteins in the IMS did not activate the CHOP axis. While confirming in this model system that expression of OTCΔ in the matrix activates CHOP, we found that IMS stress activates AKT, which then phosphorylates the ERα to promote its transcriptional activity in a ligand-independent fashion (39). Further, we found that activation of the ERα following stress in the IMS leads to increased expression of OMI and the activation of the proteasome, therefore linking stress in the IMS to the elements of the protein quality control of the IMS we had previously identified. Moreover, as the ERα was previously shown to activate the transcription of NRF1, a major transcription factor involved in mitochondrial biogenesis (40), we also analyzed NRF1 in our EndoG model. We found that upon IMS stress, the transcription of NRF1 is elevated and dependent on the ERα, as inhibition of the ERα by shRNA prevents activation of NRF1 under these conditions (39).
Given that a significant percentage of breast cancers do not express the ERα, the finding of the ERα axis of the UPRmt raised the question as to how ERα negative breast cancer cells cope with misfolded protein in the IMS. To address this question, we used EndoG to induce stress in the IMS and OTCΔ to induce stress in the matrix in ERα negative breast cancer cells. These studies led to the identification of a third axis of the UPRmt regulated by the mitochondrial NAD-dependent sirtuin deacetylase (SIRT3) (41) (Figure 1). We found that in ERα negative cells, upon stress in either the IMS or the matrix, expression of SIRT3 is elevated and leads to the deacetylation of the transcription factor FOXO3a. While the deacetylation of FOXO3a in response to mitochondrial stress was found to be SIRT3-dependent, it likely occurs through an indirect mechanism, as FOXO3a and SIRT3 have different subcellular localization (41). We reported that deacetylated FOXO3a leads to the translocation of FOXO3a in the nucleus, where it promotes the transcription of target genes SOD2 and catalase (41). In these cells, either IMS-stress or matrix-stress activated CHOP and its downstream targets hsp60 and hsp10. Importantly, within the same year, a very similar axis of the UPRmt was also reported in C. elegans and found to influence lifespan (42).
In addition to the antioxidant machinery, we reported that the SIRT3 axis of the UPRmt induces the elimination of irreversibly damaged mitochondria through the process of mitophagy (41). As for SOD2 and catalase, the induction of mitophagy upon accumulation of misfolded proteins in the mitochondria was abolished when SIRT3 was inhibited by shRNA. However, inhibition of SIRT3 did not affect the induction of Hsp60 under these conditions and inhibition of CHOP did not inhibit the expression of SOD2, catalase, or markers of autophagy. We therefore concluded that CHOP and SIRT3 regulate different axes of the UPRmt.
While the identification of the CHOP axis by the Hoogenraad group and the ERα and SIRT3 axes by our group was obtained causing accumulation of misfolded proteins directly in the matrix or the IMS, the UPRmt has been found to be activated by other, more indirect, stressors. Notably, inhibition of the ETC, inhibition of mitochondrial translation, or inhibition of matrix chaperones can also activate the UPRmt. The use of these alternative stimuli has led to the identification of additional axes. First, in hematopoietic stem cells, SIRT7 has been shown to negatively regulate NRF1 activity and induce CHOP target genes—Hsp60, Hsp10, and ClpP (Figure 1). Second, global transcriptomics and proteomics performed on HeLa cells treated with a number of agents perturbing mitochondrial proteostasis identified a reduction in pre-RNA processing and an inhibition of mtDNA-encoded translation induced by the degradation of MRPP3 (43). The transcription factor responsible for this effect remains unknown and therefore whether MRPP3 is downstream of an already known axis or represents a novel axis remains to be determined (Figure 1).
Validation of the ERα and SIRT3 Axes of the UPRmt in Other Models
Following the identification of ERα and SIRT3 axes of the UPRmt using EndoG and OTCΔ overexpression as tools to induce stress in the IMS and matrix, respectively, we next aimed at validating these axes under more physiological conditions.
Validation of the ERα Axis in Familial ALS
The field of neurodegeneration has long appreciated the importance of misfolded proteins, as it has been identified as a common mechanism in a number of human neurodegenerative disorders, such as Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, and ALS (44, 45). While the majority of ALS cases are sporadic, 10% are familial and have been linked to pathogenic mutations in specific genes (46). Superoxide dismutase 1 (SOD1) was the first gene reported to be mutated in familial ALS and over 100 different mutations have been documented (46). Localized in both the cytosol and the IMS of the mitochondria, mutations in SOD1 cause misfolding and subsequent protein aggregation in both cellular compartments (46, 47). Mutant SOD1 aggregates are cytotoxic to the motor neurons of patients with familial ALS and drive disease progression. The SOD1G93A mutation is the best characterized mutation and it has been used to generate the first mouse model of familial ALS. These mice develop muscle atrophy and other symptoms of ALS and die within 130 days. In contrast, in mice where SOD1G93A is targeted exclusively within the IMS and is absent from the cytoplasm, symptoms are drastically reduced and survival prolonged to 360 days (48). This observation led us to postulate that the accumulation of SOD1G93A in the IMS may activate the UPRmt. Further, in this model, the absence of the SOD1G93A cytosolic aggregates, which we postulate may mitigate the ability of the UPRmt to protect the integrity of the mitochondria, may explain the longer survival in the IMS targeted model of SOD1G93A.
To test this hypothesis, we recently validated the activation of the ERα axis of the UPRmt in both the untargeted and IMS-targeted mouse models of SOD1G93A familial ALS (49). Interestingly, we found a significant gender difference in the activation of the proteasome as well as OMI (49). Further, in the absence of the ERα, mutant G93A-SOD1 failed to activate this response (39, 49). This finding therefore, does not only validate the ERα axis of the UPRmt in a disease relevant model in vivo but it also raises the distinct possibility that sex differences observed in several neurodegenerative diseases may be related to the ERα status of the affected tissue. We will actively pursue this possibility in the future.
Validation of the SIRT3 Axis in Cancer Cells under Endogenous Level of Mitochondrial Stress
While the use of EndoG and OTCΔ was instrumental in the discovery of several players of the UPRmt, in reality the accumulation of misfolded proteins is likely to be present in both the matrix and the IMS as ROS is produced on both sides of the inner-membrane simultaneously. Further, since our hypothesis is that activation of the UPRmt will increase mitochondrial fitness and adaptation to stress, we reasoned that the activation of UPRmt could be linked to a more aggressive cancer phenotype, such as increased invasion capacity. We therefore investigated whether the SIRT3 and CHOP axes of the UPRmt may be linked to the metastatic potential of breast cancer cells.
We found that markers of activation of the SIRT3 axis of the UPRmt could distinguish metastatic from non-metastatic cells in a panel of breast cancer cell lines (50). When overexpressed in non-invasive cells, SOD2, the antioxidant induced by the SIRT3 axis of the UPRmt, increased invasion. When SOD2 was inhibited by shRNA, the invasion of normally invasive cells was significantly reduced. Additionally, through the analysis of a collection of cybrids—cells lines with a common nuclear genome, but different mitochondrial genomes—we demonstrated that mitochondrial disease patient-derived mtDNA mutations influence the levels of mitochondrial stress and subsequently the levels of activation of the SIRT3 axis of the UPRmt. Activation of the SIRT3 axis of the UPRmt correlated with invasion capacity of the cybrids, further emphasizing the link between this pathway and metastasis. Importantly, activation of the SIRT3 axis of the UPRmt was seen in primary breast cancer patients and high expression, using SOD2 as a marker, was significantly associated with worse disease-free survival (50). Additionally, in a collection of 50 matched primary and metastatic lesions from breast cancer patients, using SOD2 as a marker, we observed a significant increase in activation of the SIRT3 axis of the UPRmt, in metastatic lesions when compared with primary lesions. These patient data strongly support our hypothesis that activation of the SIRT3 axis of the UPRmt increases the invasiveness and metastatic potential of cancer cells. In contrast the CHOP axis, monitored using hsp60 as a marker, did not distinguish the metastatic form the non-metastatic cells. Rather, hsp60 was found to be elevated very early after oncogene induction (50). This finding suggests the possibility that the number of axes of the UPRmt engaged in mounting a protective response to mitochondrial stress may increase over disease progression.
Concluding Remarks and Future Perspectives
In light of the recent findings discussed in this review, it becomes increasingly clear that the UPRmt is a complex transcriptional pathway that expands well beyond the activation of proteases and chaperones of the matrix. We propose that the IMS plays a central role in initiating this pathway. One critical question that remains to be answered is what are the sensors/transducers of the UPRmt. Proteins able to translocate from the mitochondria to the nucleus, such as ATF5 (34), are prime candidates but others, such as GPS2 (51–55), which interestingly regulates the activity of the ERα (56), may be implicated. In addition, we cannot exclude the possibility that proteins at the surface of the mitochondria can also act as sensors and transducers of the UPRmt or that signaling cascades such as the one recently described, initiated by Lyn kinase in the IMS by ROS (57), may also be implicated in the UPRmt. Additional in vivo validation of the UPRmt in mammalian systems in the context of cancer and other pathologies is an important future direction for the new pathway. Further effort should be made to mechanistically integrate the currently known axes of the UPRmt as there is undoubtedly complex orchestration of multiple responses activated in response to mitochondrial stress. Notably, the name mitochondrial stress response has been recently proposed (58). However, misfolded proteins can arise directly or indirectly from a number of mitochondrial perturbagens, including mutation in mitochondrial genome. For instance, bacterial infection by Psedumonas aeruginosa, which causes mitochondrial dysfunction and leads to UPRmt activation in C. elegans and the transcriptional upregulation of innate immunity genes (31). Therefore, the field may benefit from expanding the term UPRmt to integrated mitochondrial stress response to more fully capture the numerous retrograde signaling cascades that are activated in response to changes in mitochondrial homeostasis, including proteostasis.
Author Contributions
TK has contributed to the writing of this review and made the figure. GM has contributed to the writing of this review. DG is the corresponding author and has contributed to the writing and finalized this review.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
This work is funded by RO1CA172046 to DG and R01NS084486 to DG and GM.
References
1. Morito D, Nagata K. Pathogenic hijacking of ER-associated degradation: is ERAD flexible? Mol Cell (2015) 59:335–44. doi:10.1016/j.molcel.2015.06.010
2. Mori K, Sant A, Kohno K, Normington K, Gething MJ, Sambrook JF. A 22 bp cis-acting element is necessary and sufficient for the induction of the yeast KAR2 (BiP) gene by unfolded proteins. EMBO J (1992) 11:2583–93.
3. Kohno K, Normington K, Sambrook J, Gething MJ, Mori K. The promoter region of the yeast KAR2 (BiP) gene contains a regulatory domain that responds to the presence of unfolded proteins in the endoplasmic reticulum. Mol Cell Biol (1993) 13:877–90. doi:10.1128/MCB.13.2.877
4. Cox JS, Shamu CE, Walter P. Transcriptional induction of genes encoding endoplasmic reticulum resident proteins requires a transmembrane protein kinase. Cell (1993) 73:1197–206. doi:10.1016/0092-8674(93)90648-A
5. Schröder M. Endoplasmic reticulum stress responses. Cell Mol Life Sci (2008) 65:862–94. doi:10.1007/s00018-007-7383-5
6. Frakes AE, Dillin A. The UPRER: sensor and coordinator of organismal homeostasis. Mol Cell (2017) 66:761–71. doi:10.1016/j.molcel.2017.05.031
7. Martinus RD, Garth GP, Webster TL, Cartwright P, Naylor DJ, Høj PB, et al. Selective induction of mitochondrial chaperones in response to loss of the mitochondrial genome. Eur J Biochem (1996) 240:98–103. doi:10.1111/j.1432-1033.1996.0098h.x
8. Zhao Q, Wang J, Levichkin IV, Stasinopoulos S, Ryan MT, Hoogenraad NJ. A mitochondrial specific stress response in mammalian cells. EMBO J (2002) 21:4411–9. doi:10.1093/emboj/cdf445
9. Ruan L, Zhou C, Jin E, Kucharavy A, Zhang Y, Wen Z, et al. Cytosolic proteostasis through importing of misfolded proteins into mitochondria. Nature (2017) 543:443–6. doi:10.1038/nature21695
10. Obre E, Rossignol R. Emerging concepts in bioenergetics and cancer research: metabolic flexibility, coupling, symbiosis, switch, oxidative tumors, metabolic remodeling, signaling and bioenergetic therapy. Int J Biochem Cell Biol (2015) 59:167–81. doi:10.1016/j.biocel.2014.12.008
11. Diebold L, Chandel NS. Mitochondrial ROS regulation of proliferating cells. Free Radic Biol Med (2016) 100:86–93. doi:10.1016/j.freeradbiomed.2016.04.198
12. Hamanaka RB, Chandel NS. Mitochondrial reactive oxygen species regulate cellular signaling and dictate biological outcomes. Trends Biochem Sci (2010) 35:505–13. doi:10.1016/j.tibs.2010.04.002
13. Schieber M, Chandel NS. ROS function in redox signaling and oxidative stress. Curr Biol (2014) 24:R453–62. doi:10.1016/j.cub.2014.03.034
14. Herrmann JM, Riemer J. The intermembrane space of mitochondria. Antioxid Redox Signal (2010) 13:1341–58. doi:10.1089/ars.2009.3063
15. Germain D. Sirtuins and the estrogen receptor as regulators of the mammalian mitochondrial UPR in cancer and aging. Adv Cancer Res (2016) 130:211–56. doi:10.1016/bs.acr.2016.01.004
16. Radke S, Chander H, Schäfer P, Meiss G, Krüger R, Schulz JB, et al. Mitochondrial protein quality control by the proteasome involves ubiquitination and the protease Omi. J Biol Chem (2008) 283:12681–5. doi:10.1074/jbc.C800036200
17. Zhao Q, Wang J, Levichkin IV, Stasinopoulos S, Ryan MT, Hoogenraad NJ. A mitochondrial specific stress response in mammalian cells. EMBO J (2002) 21:4411–9. doi:10.1093/emboj/cdf445
18. Aldridge JE, Horibe T, Hoogenraad NJ. Discovery of genes activated by the mitochondrial unfolded protein response (mtUPR) and cognate promoter elements. PLoS One (2007) 2(9):e874. doi:10.1371/journal.pone.0000874
19. Horibe T, Hoogenraad NJ. The chop gene contains an element for the positive regulation of the mitochondrial unfolded protein response. PLoS One (2007) 2(9):e835. doi:10.1371/journal.pone.0000835
20. Ryan MT, Hoogenraad NJ. Mitochondrial-nuclear communications. Annu Rev Biochem (2007) 76:701–22. doi:10.1146/annurev.biochem.76.052305.091720
21. Benedetti C, Haynes CM, Yang Y, Harding HP, Ron D. Ubiquitin-like protein 5 positively regulates chaperone gene expression in the mitochondrial unfolded protein response. Genetics (2006) 174:229–39. doi:10.1534/genetics.106.061580
22. Durieux J, Wolff S, Dillin A. The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell (2011) 144:79–91. doi:10.1016/j.cell.2010.12.016
23. Gitschlag BL, Kirby CS, Samuels DC, Gangula RD, Mallal SA, Patel MR. Homeostatic responses regulate selfish mitochondrial genome dynamics in C. elegans. Cell Metab (2016) 24:91–103. doi:10.1016/j.cmet.2016.06.008
24. Haynes CM, Petrova K, Benedetti C, Yang Y, Ron D. ClpP mediates activation of a mitochondrial unfolded protein response in C. elegans. Dev Cell (2007) 13:467–80. doi:10.1016/j.devcel.2007.07.016
25. Haynes CM, Yang Y, Blais SP, Neubert TA, Ron D. The matrix peptide exporter HAF-1 signals a mitochondrial UPR by activating the transcription factor ZC376.7 in C. elegans. Mol Cell (2010) 37:529–40. doi:10.1016/j.molcel.2010.01.015
26. Lin Y-F, Schulz AM, Pellegrino MW, Lu Y, Shaham S, Haynes CM. Maintenance and propagation of a deleterious mitochondrial genome by the mitochondrial unfolded protein response. Nature (2016) 533:416–9. doi:10.1038/nature17989
27. Lisanti S, Garlick DS, Bryant KG, Tavecchio M, Mills GB, Lu Y, et al. Transgenic expression of mitochondrial chaperone TRAP1 accelerates prostate cancer development. J Biol Chem (2016) 291:25247–54. doi:10.1074/jbc.M116.745950
28. Merkwirth C, Jovaisaite V, Durieux J, Shaw RJ, Auwerx J, Dillin A, et al. Two conserved histone demethylases regulate mitochondrial stress-induced longevity. Cell (2016) 165:1209–23. doi:10.1016/j.cell.2016.04.012
29. Nargund AM, Pellegrino MW, Fiorese CJ, Baker BM, Haynes CM, Chacinska A, et al. Mitochondrial import efficiency of ATFS-1 regulates mitochondrial UPR activation. Science (2012) 337:587–90. doi:10.1126/science.1223560
30. Nargund AM, Fiorese CJ, Deng P, Haynes CM, Pellegrino MW. Mitochondrial and nuclear accumulation of the transcription factor ATFS-1 promotes OXPHOS recovery during the UPRmt. Mol Cell (2015) 58:123–33. doi:10.1016/j.molcel.2015.02.008
31. Pellegrino MW, Nargund AM, Kirienko NV, Gillis R, Fiorese CJ, Haynes CM. Mitochondrial UPR-regulated innate immunity provides resistance to pathogen infection. Nature (2014) 516:414–7. doi:10.1038/nature13818
32. Rauthan M, Ranji P, Aguilera Pradenas N, Pitot C, Pilon M. The mitochondrial unfolded protein response activator ATFS-1 protects cells from inhibition of the mevalonate pathway. Proc Natl Acad Sci USA (2013) 110:5981–6. doi:10.1073/pnas.1218778110
33. Tian Y, Garcia G, Bian Q, Steffen KK, Joe L, Wolff S, et al. Mitochondrial stress induces chromatin reorganization to promote longevity and UPRmt. Cell (2016) 165:1197–208. doi:10.1016/j.cell.2016.04.011
34. Fiorese CJ, Schulz AM, Lin Y-F, Rosin N, Pellegrino MW, Haynes Correspondence CM. The transcription factor ATF5 mediates a mammalian mitochondrial UPR. Curr Biol (2016) 26:2037–43. doi:10.1016/j.cub.2016.06.002
35. Teske BF, Fusakio ME, Zhou D, Shan J, McClintick JN, Kilberg MS, et al. CHOP induces activating transcription factor 5 (ATF5) to trigger apoptosis in response to perturbations in protein homeostasis. Mol Biol Cell (2013) 24:2477–90. doi:10.1091/mbc.E13-01-0067
36. Sheng Z, Li L, Zhu LJ, Smith TW, Demers A, Ross AH, et al. A genome-wide RNA interference screen reveals an essential CREB3L2-ATF5-MCL1 survival pathway in malignant glioma with therapeutic implications. Nat Med (2010) 16:671–7. doi:10.1038/nm.2158
37. Cole A, Wang Z, Coyaud E, Voisin V, Gronda M, Jitkova Y, et al. Inhibition of the mitochondrial protease ClpP as a therapeutic strategy for human acute myeloid leukemia. Cancer Cell (2015) 27:864–76. doi:10.1016/j.ccell.2015.05.004
38. Seo JH, Rivadeneira DB, Caino MC, Chae YC, Speicher DW, Tang H-Y, et al. The mitochondrial unfoldase-peptidase complex ClpXP controls bioenergetics stress and metastasis. PLoS Biol (2016) 14:e1002507. doi:10.1371/journal.pbio.1002507
39. Papa L, Germain D. Estrogen receptor mediates a distinct mitochondrial unfolded protein response. J Cell Sci (2011) 124:1396–402. doi:10.1242/jcs.078220
40. Mattingly KA, Ivanova MM, Riggs KA, Wickramasinghe NS, Barch MJ, Klinge CM. Estradiol stimulates transcription of nuclear respiratory factor-1 and increases mitochondrial biogenesis. Mol Endocrinol (2008) 22:609–22. doi:10.1210/me.2007-0029
41. Papa L, Germain D. SIRT3 regulates the mitochondrial unfolded protein response. Mol Cell Biol (2014) 34:699–710. doi:10.1128/MCB.01337-13
42. Mouchiroud L, Houtkooper RH, Moullan N, Katsyuba E, Ryu D, Cantó C, et al. The NAD+/sirtuin pathway modulates longevity through activation of mitochondrial UPR and FOXO signaling. Cell (2013) 154:430–41. doi:10.1016/j.cell.2013.06.016
43. Münch C, Harper JW. Mitochondrial unfolded protein response controls matrix pre-RNA processing and translation. Nature (2016) 534:710–3. doi:10.1038/nature18302
44. Ciechanover A, Kwon YT. Degradation of misfolded proteins in neurodegenerative diseases: therapeutic targets and strategies. Exp Mol Med (2015) 47:e147. doi:10.1038/emm.2014.117
45. Lindquist SL, Kelly JW. Chemical and biological approaches for adapting proteostasis to ameliorate protein misfolding and aggregation diseases-progress and prognosis. Cold Spring Harb Perspect Biol (2011) 3:a004507. doi:10.1101/cshperspect.a004507
46. Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci (2014) 17:17–23. doi:10.1038/nn.3584
47. Münch C, Bertolotti A. Exposure of hydrophobic surfaces initiates aggregation of diverse ALS-causing superoxide dismutase-1 mutants. J Mol Biol (2010) 399:512–25. doi:10.1016/j.jmb.2010.04.019
48. Magrané J, Hervias I, Henning MS, Damiano M, Kawamata H, Manfredi G. Mutant SOD1 in neuronal mitochondria causes toxicity and mitochondrial dynamics abnormalities. Hum Mol Genet (2009) 18:4552–64. doi:10.1093/hmg/ddp421
49. Riar AK, Burstein SR, Palomo G, Arreguin A, Manfredi G, Germain D. Sex specific activation of the ERα axis of the mitochondrial UPR (UPRmt) in the G93A-SOD1 mouse model of familial ALS. Hum Mol Genet (2017) 26:1318–27. doi:10.1093/hmg/ddx049
50. Kenny TC, Hart P, Ragazzi M, Sersinghe M, Chipuk J, Sagar MAK, et al. Selected mitochondrial DNA landscapes activate the SIRT3 axis of the UPRmt to promote metastasis. Oncogene (2017). doi:10.1038/onc.2017.52
51. Cardamone MD, Tanasa B, Chan M, Cederquist CT, Andricovich J, Rosenfeld MG, et al. GPS2/KDM4A pioneering activity regulates promoter-specific recruitment of PPAR? Cell Rep (2014) 8:163–76. doi:10.1016/j.celrep.2014.05.041
52. Cederquist CT, Lentucci C, Martinez-Calejman C, Hayashi V, Orofino J, Guertin D, et al. Systemic insulin sensitivity is regulated by GPS2 inhibition of AKT ubiquitination and activation in adipose tissue. Mol Metab (2016) 6:125–37. doi:10.1016/j.molmet.2016.10.007
53. Cardamone MD, Krones A, Tanasa B, Taylor H, Ricci L, Ohgi KA, et al. A protective strategy against hyperinflammatory responses requiring the nontranscriptional actions of GPS2. Mol Cell (2012) 46:91–104. doi:10.1016/j.molcel.2012.01.025
54. Huang J, Cardamone MD, Johnson HE, Neault M, Chan M, Floyd ZE, et al. Exchange factor TBL1 and arginine methyltransferase PRMT6 cooperate in protecting G protein pathway suppressor 2 (GPS2) from proteasomal degradation. J Biol Chem (2015) 290:19044–54. doi:10.1074/jbc.M115.637660
55. Lentucci C, Belkina AC, Cederquist CT, Chan M, Johnson HE, Prasad S, et al. Inhibition of Ubc13-mediated ubiquitination by GPS2 regulates multiple stages of B cell development. J Biol Chem (2017) 292:2754–72. doi:10.1074/jbc.M116.755132
56. Cheng X, Kao H-Y. G protein pathway suppressor 2 (GPS2) is a transcriptional corepressor important for estrogen receptor-mediated transcriptional regulation. J Biol Chem (2009) 284:36395–404. doi:10.1074/jbc.M109.062109
57. Patterson HC, Gerbeth C, Thiru P, Vögtle NF, Knoll M, Shahsafaei A, et al. A respiratory chain controlled signal transduction cascade in the mitochondrial intermembrane space mediates hydrogen peroxide signaling. Proc Natl Acad Sci USA (2015) 112:E5679–88. doi:10.1073/pnas.1517932112
Keywords: mitochondria, mitochondrial unfolded protein response, cancer, ALS, sirtuin deacetylase, estrogen receptor
Citation: Kenny TC, Manfredi G and Germain D (2017) The Mitochondrial Unfolded Protein Response as a Non-Oncogene Addiction to Support Adaptation to Stress during Transformation in Cancer and Beyond. Front. Oncol. 7:159. doi: 10.3389/fonc.2017.00159
Received: 10 April 2017; Accepted: 10 July 2017;
Published: 26 July 2017
Edited by:
Matiullah Khan, Asian Institute of Medicine, Science & Technology, MalaysiaReviewed by:
Abhishek D. Garg, KU Leuven, BelgiumChakrabhavi Dhananjaya Mohan, University of Mysore, India
Copyright: © 2017 Kenny, Manfredi and Germain. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Doris Germain, ZG9yaXMuZ2VybWFpbkBtc3NtLmVkdQ==
 Timothy C. Kenny1
Timothy C. Kenny1 Giovanni Manfredi
Giovanni Manfredi Doris Germain
Doris Germain