- 1Integrated Microbiome Core, Department of Dermatology, Case Western Reserve University, Cleveland, OH, United States
- 2University Hospitals Cleveland Medical Center, Cleveland, OH, United States
- 3BIOHM Health LLC, Cleveland, OH, United States
- 4Louis Stokes Cleveland Department of Veterans Affairs Medical Center, Cleveland, OH, United States
The significant stressors brought about and exacerbated by COVID-19 are associated with startling surges in mental health illnesses, specifically those related to depressive disorders. Given the huge impact of depression on society, and an incomplete understanding of impactful therapeutics, we have examined the current literature surrounding the microbiome and gut-brain axis to advance a potential complementary approach to address depression and depressive disorders that have increased during the COVID-19 pandemic. While we understand that the impact of the human gut microbiome on emotional health is a newly emerging field and more research needs to be conducted, the current evidence is extremely promising and suggests at least part of the answer to understanding depression in more depth may lie within the microbiome. As a result of these findings, we propose that a microbiome-based holistic approach, which involves carefully annotating the microbiome and potential modification through diet, probiotics, and lifestyle changes, may address depression. This paper's primary purpose is to shed light on the link between the gut microbiome and depression, including the gut-brain axis and propose a holistic approach to microbiome modification, with the ultimate goal of assisting individuals to manage their battle with depression through diet, probiotics, and lifestyle changes, in addition to offering a semblance of hope during these challenging times.
Introduction
Within a 1-month period at the beginning of the COVID-19 pandemic, there was a reported 34.1% increase in prescriptions for anti-anxiety medications, an 18.6% increase in antidepressant prescriptions, and a 14.8% increase in common anti-insomnia drugs including prescribed anti-insomnia medications including eszopiclone, zolpidem and zaleplonin the United States (1). During such a short period of time, this steep rise hints at the magnitude of COVID-19's immediate and widespread effect on mental health. In a recent Single Care survey taken from 1,000 U.S. respondents, 59% reported that their mental health had been affected by COVID-19 (2). Additionally, a spokesperson for the Substance Abuse and Mental Health Services Administration (SAMHSA)-Disaster Distress Helpline reported a 338% increase in the call volume for individuals experiencing emotional distress from February 2020 to March 2020, an 891% increase compared to the call volume from March of 2019 (3). This alarming rate of mental health crises is an extreme concern for countries worldwide. Experts from the United Nations World Health Organization (WHO) have urged governments to put tribulations regarding the upsurge and severity of mental health illnesses at the “front and center” of their COVID-19 response (4). The suggestion by the United Nations is not trivial, as it is based on the startling statistics that show the devastating toll the pandemic is having on individuals all around the world.
Increasing levels of depressive disorders are especially concerning for mental health. Prior to the pandemic, Major Depressive Disorder (MDD) affected > 350,000,000 people and was a leading cause of disability, morbidity, and mortality worldwide (5–7). To further highlight the severity of depressive disorders, an even larger proportion of the population suffers from undiagnosed subclinical symptoms of depression (8). This subclinical disorder is especially problematic, as individuals who are diagnosed with depression or experience depressive symptoms are not the only ones who suffer from the resulting detrimental effects. Instead, this disorder also adversely affects society as a whole. Depression and depressive symptoms are associated with decreased physical health, increased unemployment, impaired social functioning, reduced productivity, and suicide, all of which can result in high economic costs to society and increased demand on the healthcare system (9, 10).
Despite the toll that depression has on both individuals and society, understanding and effectively treating depressive disorders is difficult. Current research addressing the diagnosis and treatment of depression and mood disorders is ongoing, but needs more time to develop a full understanding of the complexities involved in these disorders and to develop novel strategies to treat these disorders. Currently, clinical trials show that ~30–40% of depressed patients do not respond to their first-line antidepressant treatment (11, 12), up to 70% do not achieve complete remission of symptoms (13), and 20% never show a reduction in depressive symptoms even when accounting for multiple antidepressants and various methods of action (14–16). Interestingly, some promising results of electroconvulsive therapy (ECT) have been noted (17), however, this therapy is also not without controversy (18, 19).
Notably, more than half of individuals prescribed antidepressants experience side effects (20), which can be so severe that some must stop the treatment altogether. Thus, as with numerous other drug treatments, side effects have a considerable toll on efficacy and proper usage. The damage caused by depression, combined with an incomplete understanding of the depth of this disease highlights the need to examine new potential methods to address depression and better understand how it influences our bodies.
While discovering effective approaches to depressive disorders has always been a priority, we believe it is especially important today. This urgency was highlighted by the recent COVID19 pandemic, where increased stress and mental health conditions including increased anxiety and sleep disruption/disorders were elevated, especially in healthcare workers worldwide (21, 22). A survey among an Indian population showed that the prevalence of anxiety among respondents (n = 392) was 25.3% which was very high compared to the estimated levels in a Indian National Mental Health Survey (2015–2016) for the general population (23, 24). Similar other online surveys have been conducted targeting various international groups (25–28). Although, the validity of answers is a general problem of online surveys, similar observations reflect a potential major issue. Furthermore, such problems cannot be avoided in these challenging times. Such stressors include, but are not limited to, the following: (i) the uncertainty caused by the worldwide COVID-19 pandemic; (ii) feelings of fear and isolation; (iii) disrupted workplaces and schedules; (iv) unemployment; (v) changes to daily routines; (vi) anxiety about future events and decisions, and (vii) other yet unknown world events. These significant stressors, associated with the pandemic, were abruptly placed on top of regular stressors caused by everyday living with little or no warning.
To make matters worse, the increased level of acute and chronic stress caused by COVID-19 does not appear to be abating (21, 22, 26–31). People are still afraid that they or a loved one will contract the virus, worry about the efficacy and side effects of vaccination, there is still not the same level of physical human interaction, a lack of a sense of normalcy lingers with social distancing rules and masks. People are struggling with unemployment and parents were required to adjust to a new normal regarding educating (at home) school age children. These worries were highlighted under the concept of COVID Stress Syndrome, a newly proposed syndrome that is still under investigation (32–34).
Here, we propose a potential approach to address depression, which involves analysis of the microbiome and potential modification through diet, probiotics, and lifestyle changes. This article discusses gut and brain communication and the potential connection to stress and depression, the potential association between COVID-19 and the microbiome, the reported association between the microbiome and depression, and how the microbiome and depression may relate to diet, probiotics, and lifestyle habits. Lastly, we conclude by offering a potential approach to addressing depression through lifestyle modification including diet, exercise and potential microbiome modification. The overall goal is to examine evidence suggesting a connection of the microbiome to the brain, which implicates potential effects on depression and depressive disorders, in order to address how individuals may contain and end their battle with depression and navigate these difficult times.
COVID-19 and Microbiome
Since the beginning of the COVID-19 pandemic extensive efforts have been made to combat this new virus. One of the main steps in studying any infectious agent is to know how it can infect human cells and consequently cause symptoms. In this regard, studies have found that COVID-19 targets exclusively the angiotensin-converting enzyme 2 (ACE2) receptor as an entry receptor into ACE2-expressing cells (35) which have been identified as the surface receptor for the spike proteins of acute respiratory syndrome coronavirus (SARS-CoV) (36), a first and essential step of viral infections of host cells (37). Moreover, the virus also uses serine protease TMPRSS2 for S protein priming that facilitates viral spread and pathogenesis (38, 39). Thus, in addition to the mode of transmission playing a major role (40), the expression of ACE2 on respiratory epithelia cells make it more plausible (39, 41).
In addition to the well-documented respiratory route of infection and subsequent consequences (respiratory sequalea) (42), ACE2 is expressed on other tissues especially the gastrointestinal tract (41). This may explain extra-pulmonary manifestations associated with COVID19 infection (43–52). Although respiratory syndrome is the main presentation of COVID19 infection, gastrointestinal (GI) symptoms have been reported in a number of COVID-19 cases (53–56). This concept has been further validated by data reported by Xiao et al., showing that 73 patients infected with COVID-19 tested positive for viral RNA in their stools (52). However, the epithelium lining the esophagus, stomach, duodenum, and rectum showed no significant damage on staining. Furthermore, continued viral shedding in stools even after negative nasal swabs has been reported by Xu et al., in a study that included 10 pediatric COVID-19 infection cases confirmed by real time reverse transcription PCR assay of viral RNA. Although, a limitation for this study is small sample size and there was no evidence of replication-competent virus in fecal swabs. In another study by Gu et al., examining the gut microbial diversity of 30 patients with COVID-19, a marked decrease in microbiome diversity was observed when compared to healthy controls. Although this is a modest number of observed events (N = 30) the overall observation that the gut microbiome of the COVID-19 group was dominated by Streptococcus, Rothia, Veillonella, Erysipelatoclostridium, and Actinomyces was notable. On the other hand, the abundance of the Fusicatenibacter, Anaerostipes, Agathobacter, unclassified Lachnospiraceae, and E. hallii group was reduced (57). This data agreed with results from Zuo et al., that showed an increase in opportunistic pathogens known to cause bacteremia including Clostridium hathewayi, Actinomyces viscosus, and Bacteroides nordii compared with controls, as well as depletion of Faecalibacterium prausnitzii, Lachnospiraceae bacterium, Eubacterium rectale, Ruminococcus obeum, and Dorea formicigenerans in COVID-19 patients receiving antibiotic therapy (58). However, again, the study had a limited sample size (n = 25). Importantly, this study showed that Clostridium ramosum and C. hathewayi, were the top bacteria positively associated with COVID-19 disease severity, which is supported by the observation that these microorganisms have been shown to strongly upregulate colonic expression of ACE2 in the murine gut (59). ACE2 not only plays a role in COVID-19 infection, but also plays an important role in regulating the gut microbiome (60) which in turn regulates the host immune response and gene expression (61). Additionally, a decrease in ACE2 expression was found to be associated with decreased tryptophan amino acid absorption in murine studies (60) which has also been linked to disturbances in the gut microbiome and consequently disruption of the gut-brain-axis using in silico analyses (62).
In a separate clinical trial designed to further understand the effect of COVID-19 infection on the gut microbiome, viral RNA extraction and shotgun metagenomics sequencing were performed on fecal samples of 15 patients hospitalized with COVID-19 (63). Analysis of the samples showed active viral infection signatures in the fecal samples even after clearance of the virus from respiratory samples for up to 6 days with a higher infectivity signature in samples that had greater abundances of the bacterial species Collinsella aerofaciens, Collinsella tanakaei, Streptococcus infantis, and Morganella morganii. Although this study involved a limited number of patients, these were emerging reports during the pandemic, and acquiring large sample numbers was unlikely to occur. Similarly, analysis of fecal samples from 30 patients with COVID-19 in Hong Kong showed increased proportions of opportunistic fungal pathogens such as Candida albicans, Candida auris, and Aspergillus flavus compared with controls, where detection of Aspergillus was found in 2 samples even after clearance of the virus from nasopharyngeal samples and resolution of respiratory symptoms (64). Yu et al., also reported that reduction in the Bifidobacterium, Lactobacillus, and Eubacterium, as well as significantly increased Corynebacterium of Actinobacteria and Ruthenibacterium of Firmicutes were observed in four COVID-19 patients who had decreased immune cells and refractory hypoxemia during hospitalization (65).
Collectively, the previous reports shed light on the potential correlation between COVID infection and microbiome disturbance, however, these reports have limitations that necessitate further investigations with larger sample sizes that can be used to approximate the general population.
Although there were reports of COVID-19 patients exhibiting GI symptoms, high rates of fever (>38.5°C), fatigue, shortness of breath and headache (56), a study by Lean et al., published during preparation of our manuscript, showed that patients with digestive symptoms experienced less severe disease (66) as defined by the need for mechanical ventilation, ICU admission or death, which is further supported by similar observation in other studies (55, 67, 68). Additionally, patients with digestive symptoms had a significantly longer time from onset to admission than patients without digestive symptoms (55, 68), necessitating a high level of caution in reaching an early diagnosis and setup of necessary infection control measures to control the transmission rate. This inconsistent observation reflects our poor understating of the nature of this novel virus and warrant further investigation.
Another clinical reported manifestation is COVID-19 induced neurological symptoms (51, 69, 70) including non-specific cognitive complaints, headache, numbness/tingling, dysgeusia, and anosmia (loss of smell) (71–74). These observations were thought to be due to ACE2 expression on neurons and glial cells of the brain (41) allowing the virus to directly reach the brain via the olfactory bulb following inhalation without the use of ACE2, however, this was strongly refuted by growing evidence that support these neurological findings are mainly attributed to the effects of hypoxia, coagulopathy, and multi-organ damage in severe infection, accompanied by virus-mediated inflammatory processes (74).
Gut-Brain Axis
In the past, scientists believed that the brain was the central control for all processes. However, recent findings suggest otherwise. Numerous studies have shown that the microbiota, or the communities of bacteria and fungi that live in our gut, impact our brain and emotions. The communication between the gut microbiome and the brain is bidirectional and occurs through neural, inflammatory, and hormonal signaling pathways. To further expand upon this relationship, murine studies have shown that the gut microbiome is actively involved in processes linked to brain development, physiology, psychology, and behavior (75, 76). Specifically, murine models have demonstrated that the gut microbiome plays a critical role in the regulation of mood, anxiety, and pain (77, 78). In addition, the brain-gut axis has been shown to modulate the following: stress responsiveness (79, 80), prefrontal myelination (81), brain biochemistry (78), immune function, neurotransmission, and neurogenesis (80) using murine models and germ free animal studies.
As a result of the bidirectional interaction between the gut microbiome and the brain, each can send messages that impact the other. Cortisol, which is the primary stress hormone that dictates which functions are essential during a fight-or-flight situation (82), is an example of how the brain can send messages that impact the gut. Observations in animal studies showed that administration of dexamethasone mimicked in a dose dependent manner the effect of maternal separation in preterm male and female pups which showed enhanced plasma corticosterone, increased in vivo intestinal permeability, and induced bacterial translocation to liver and spleen (83). A similar observation was reported in other studies using rat models (84). Furthermore, germ free mice demonstrated plasma ACTH and corticosterone elevation in response to stress that was higher in comparison to specific pathogen free controls that was reversed by reconstitution with Bifidobacterium infantis, suggesting gut microbiota can affect the development of the hypothalamic–pituitary–adrenal stress response in mice (83, 85). In humans, patients with irritable bowel syndrome (IBS) were found to have increased adrenocorticotropic hormone and cortisol levels in response to corticotropin-releasing hormone infusion and elevated levels of the proinflammatory cytokines IL-6 compared to controls, an observation that suggest over activation of the hypothalamic-pituitary-adrenal axis (86). Although this study had a limited sample size (n = 151), this evidence suggests a communication between the brain and the gut.
On the other hand, an imbalance in the gut's microbes, as reviewed by Petersen and Round (87), is an example of how the gut can send messages that impact the brain. Microbial shifts in abundance and diversity (either increased or decreased) are often termed “dysbiosis” (87–90). In the present article, our reference to dysbiosis is to indicate a change from “baseline” microbiota for any given individual, not necessarily to indicate a pathological outcome. However, in general, decreased levels of diversity have been associated with pathological outcomes often identified as dysbiosis. When dysbiosis is observed, numerous targets are affected, such as the levels of dopamine and serotonin in the brain, both of which directly influence mood using germ free murine models (91–95). It is worth mentioning that dysbiosis is not the only factor that affects the level of these transmitters, however, this observation strongly suggests that microbiota may have a role in regulating these neurotransmitters. Such changes are associated with increased depression and anxiety and altered eating habits (96, 97). In summation, the bidirectional interaction between the gut microbiome and the brain highlights the relationship that exists between the two.
Is There an Association Between the Microbiome and Depression?
Given the reported increase in stress, anxiety and depression with the COVID19 pandemic, examining potential factors influencing depression is in order. One such factor is the microbiome connection to these disorders. The connection between the gut microbiome and depression is demonstrated by the subsequent factors that affect, and are affected by, the gut microbiome's composition and the onset/development of depression. The interaction between the gut microbiome and the brain lays the foundation for the specific link between the gut microbiome and depression. Changes to the gut microbiota in major depressive disorder adversely affect many dimensions of the gut-brain axis, including the following: hyperactivation of the Hypothalamic-Pituitary-Adrenal (HPA) axis, disruption of neural circuits and neurotransmitter levels, excess production of proinflammatory cytokines in the immune system, and disruption of the intestinal barrier, as reviewed by Bastiaanssen et al. (98).
Similarly, the gut microbiome, as reviewed by Rea et al., plays a critical role in the development and function of the HPA axis, which controls one's stress response and is involved in depression (99). Clinical evidence suggests that depressive episodes are associated with dysregulation of the HPA axis (100), and the stabilization of the HPA axis is connected to a reduction in depressive symptoms (101, 102). Additionally, the gut microbiome can affect both neurotransmission and the production of various neurotransmitters, some of which are related to depression (96, 97, 103). The microbiota has also been shown to directly impact the function of the central nervous system (CNS) via neuronal activation of stress circuits (98). For example, murine studies indicate gut microbes can regulate stress response through the activation of the vagal pathways when Citrobacter rodentium and Campylobacter jejuni are administered orally (104, 105). Mechanisms underlying the effects of the microbiome in psychopathological disorders (e.g., anxiety and depression) suggest that pro-inflammatory cytokines could act as mediators to induce anxious behavior as shown in clinical studies (106). In this regard, various cytokine receptors have been found on the vagus nerve and other peripheral and spinal nerves that may signal inflammation to the brain, which could lead to depression and anxiety (107). Alternatively, in individuals with vagotomy, cytokines could enter the brain via the circumventricular organs, which facilitate communication between the central nervous system and peripheral blood, or via cytokine transporters at the blood-brain barrier where pro-inflammatory cytokines could gain access to the brain through these saturable transport systems. Another pathway that may participate in inducing depression involves activation of interleukin-1 receptors (positioned on perivascular macrophages and endothelial cells of small blood vessels in the brain) by circulating cytokines leading to local production of prostaglandin E2 as determined in rat studies (108, 109).
The direct link between the gut microbiome and depression has also been supported through various studies with differing perspectives and experimental approaches including reviews and primary behavioral studies in rats (110–113). It has been discovered that around 40–60% of anxious and depressed individuals report having an intestinal function disturbance, like Irritable Bowel Syndrome (114). Furthermore, transferring fecal gut microbiota from humans with depression into rodents has been shown to induce depression-like states in the animals, highlighting the causal role that the gut microbiome plays in the onset of Major Depressive Disorders (MDD) (113, 115, 116).
While the findings above highlight the link between the microbiome and depression, more evidence to support this relationship has been established. For example, scientists can distinguish between individuals with and without depression by analyzing their microbiome makeup (49). In fact, studies using the 16S ribosomal RNA sequencing method have observed abnormalities in the gut bacterial microbiomes of individuals with MDD (117, 118). Specifically, depressed patients are shown to have reduced microbiome abundance and alpha diversity. For example, Aizawa et al. found that the amount of Bifidobacterium and Lactobacillus in the feces of patients with major depression is typically less than that of healthy individuals (119). In comparison, a study conducted by Valles-Colomer et al. examined the microorganisms present in 1,000 individuals and discovered that depressed individuals were often more likely to have a reduced abundance of Coprococcus and Dialister when compared to their non-depressed counterparts (120).
Another study conducted by Yang et al. identified 3 bacteriophages, 47 bacterial species, and 50 fecal metabolites showing notable differences in abundance between patients with MDD and healthy control subjects. Patients with MDD were mainly characterized by increased abundance of the genus Bacteroides spp. and decreased levels of the genera Blautia and Eubacterium. Furthermore, using a discovery and validation set, the authors identified a biomarker panel that robustly discriminated between MDD and healthy individuals (49). Additionally, most studies that have examined patients with MDD have found an association between depression and “increased levels of the phylum Actinobacteria, order Bacteroidales, family Enterobacteriaceae, genus Alistipes and deceased family Lachnospiraceae, and genus Faecalibacterium” (121).
The plethora of evidence supporting the gut-brain axis and the established relationship between depression and the gut microbiome is indicative of potential new and more effective depression management. Based on the findings above, it is clear that the gut microbiome's makeup in individuals with depressive disorders are disrupted and lack the appropriate levels of beneficial microorganisms. Thus, we believe that encouraging the growth of such beneficial microorganisms and rebalancing the gut microbiome in individuals with MDD may be a promising step toward assisting individuals ease their depression via the gut-brain axis. Therefore, we propose a multifaceted approach to manage depression that involves rebalancing and maintaining the gut microbiome through diet, probiotics, and specific lifestyle changes.
Diet and Depression
While acknowledging the complex and multidirectional nature of the relationship between diet and mental health, we propose that diet is an essential part of a balanced approach to combating depression. A large number of promising studies have examined the relationship between diet and depression. In particular, one study examined 16 eligible randomized controlled trials with outcome data for more than 45,000 participants. The findings from this study indicate that dietary intervention significantly reduced depressive symptoms in high-quality trials (g = 0.321, 95% CI = 0.12–0.53, p = 0.002) (122). Other meta analyses and clinical studies have revealed an association between diet quality and the probability of and risk for depression (123, 124). Additionally, pro-inflammatory dietary patterns have specifically been linked to a greater likelihood of developing depression and depressive symptoms in clinical studies (125–127).
Eating patterns during the COVID pandemic showed an increased adoption of unhealthy diets. A study by Robinson et al., examining the impact of COVID-19 on a large sample of UK adults (n = 2,002) during social lockdown showed that 82% reported an increase in the intake of unhealthy food (128). Furthermore, 36% of the participants reported an increase in alcohol intake compared to before lockdown which is known to cause disruption of the gut microbiome (129) by causing bacterial overgrowth (130, 131) and hyper-permeability of the intestinal membrane (i.e., leaky gut) (132). In this regards, Pollard et al., also reported an increase in alcohol intake of 14% over the baseline in 2019 during the COVID-19 pandemic in a sample that included 1,540 adults in the U.S. (133).
Studies have also established a relationship between the prevalence of depressive symptoms and certain eating habits. For example, depressive symptoms have been associated with a high glycemic index attributed to a high intake of sugars and refined carbohydrates (126, 127, 134). Conversely, healthy eating habits that limit sugar intake and refined carbohydrates are associated with a decreased risk of depression. Such findings align with several smaller and more recent trials where considerable improvements in depression levels were observed in people who followed the so-called “Mediterranean Diet” (135–137). For context, the Mediterranean diet, which adheres to healthy eating practices, is associated with a small intake of red meat; a high intake of vegetables, fruits, beans, and nuts; and a medium intake of eggs, dairy, and poultry (123, 126, 127).
We propose that the consistently significant and positive effects of dietary intervention on depressive symptoms reflect the impact that diet has on the microbiome. While recognizing the role that genetics and antibiotics play, we believe that diet is a potentially adjustable determining factor of the relative abundance, diversity, and functionality of organisms comprising the gut microbiome. In the same vein, no single treatment is likely to fully ameliorate depression and/or depressive symptoms, in fact, combined therapeutic approaches (a holistic approach) may include several areas including pharmacological interventions, psychiatric therapy and lifestyle and nutritional changes. For example, studies have shown that the artificial sweetener Splenda™ promotes gut dysbiosis and the increased growth of Proteobacteria, which is an indicator for inflammation (138). Additionally, decreased function of the gut barrier, also known as “leaky gut,” is linked to an “unhealthy” gut microbiome that is attributed to a diet high in saturated fats, refined sugars, artificial sweeteners, and low in fiber, aka the “Western” diet (125, 139, 140). On the other hand, a diet high in fibers, polyphenols (flavonoids and phenolic acid micronutrients found in berries, nuts, flaxseeds, vegetables, olives, coffee, and tea) and unsaturated fatty acids (fatty acids that have one or more carbon-carbon double bonds, comprised of monounsaturated fatty acids present in avocado, nuts, olive, peanut oils and vegetable oils, and polyunsaturated fatty acids present mainly in vegetable oils such as sunflower, sesame, soybean, and corn oils, the prototypical “Mediterranean” diet) is associated with improving and promoting the growth of beneficial gut microbial taxa (139, 141).
Although the exact underlying mechanisms through which diet impacts mental health have yet to be defined, there are several pathways by which diet can play a role. These pathways are related to oxidative stress, inflammation, and mitochondrial dysfunction, which tend to be disrupted in people with mental health disorders (142). A healthy diet typically includes various bioactive compounds that beneficially engage these pathways. For example, fruits and vegetables contain fiber, vitamins, minerals, and are packed with a large number of polyphenols. These nutritional factors appear to be associated with decreased depression rates, possibly due to their anti-inflammatory, neuroprotective, and prebiotic properties (143, 144). On the other hand, unhealthy diets are rich in nutritional factors that may negatively engage these pathways. Specifically, elements commonly found in processed foods like artificial sweeteners, emulsifiers, and saturated fatty acids may change the gut microbiome's composition and activate inflammatory pathways (136).
Probiotics and Depression
In addition to diet, we propose that probiotics are an essential part of an encompassing approach to treating depression. We assert that probiotics will help to serve the critical function of rebalancing the microbiome. While we recognize that the study of probiotics in humans is a newly emerging field that has recently gained an immense amount of attention, published evidence suggests that probiotics will play a promising role in combating depression (145). Numerous studies conducted on both humans and animals have indicated that probiotics are associated with a reduction in anxiety and depression (78, 80, 146–149). Other clinical studies have shown that probiotics have also effectively mitigated anxiety and depressive symptoms in a manner that is comparable to that of conventional prescription medications (150–152). Additionally, related studies have indicated that probiotic use reduced depressive symptoms and improved the functionality of both the HPA axis and was equivalent to treatment with the antidepressant Citalopram in murine studies (106, 146, 152). However, as with any emerging therapeutic option, some failed to achieve the desired effect, a result that has been reported in a number of studies (153–155).
One particular study gave patients suffering from chronic stress a 3-week probiotic regimen that contained Lactobacillus casei. Subjects in the bottom third of the elated/depressed scale demonstrated the most improvement as they consistently rated an overall happier mood (150). Additionally, a double-blind, placebo-controlled randomized study examined the effects of probiotics on behavior, brain function, and gut microbial composition in 45 healthy volunteers. At the end of the 4-week period, probiotic administration was associated with shifts in gut microbiome profiles and brain activation patterns. The probiotic arm in this study contained the following nine bacterial strains: Lactobacillus casei W56, Lactobacillus acidophilus W22, Lactobacillus paracasei W20, Bifidobacterium lactis W51, Lactobacillus salivarius W24, Lactococcus lactis W19, Bifidobacterium lactis W52, Lactobacillus plantarum W62 and Bifidobacterium bifidum W23 (156). Using probiotics that incorporate multiple strains may provide better benefits than those using single strains and may result in having synergistic effects that can substantially enhance the establishment and competitiveness of a putative probiotic strain in the GI tract. In this regard, a review by Sun and Buys showed that consumption of probiotics that have multiple strains in combination as well as of probiotics containing Lactobacillus acidophilus, a mixture of Lactobacillus acidophilus and Bifidobacterium lactis, or Lactobacillus plantarum led to a statistically significant reductions in total and low-density lipoprotein (LDL) cholesterol levels, while clinical trials that used a single strain did not (157). We speculate that each strain may contribute differently (e.g., some produce more beneficial short chain fatty acids (SCFA), while others antagonize pathogenic microorganisms present in the gut).
In addition to the strains previously mentioned, Bifidobacterium infantis (146), Lactobacillus helveticus (106, 147), Lactobacillus rhamnosus (78), and Bifidobacterium longum (137) have also been associated with a reduction in depression. Studies have suggested that probiotics with such bacterial strains modify the microbiome and potentially treat depression due to their ability to normalize cortisol levels, regulate the HPA axis, and reduce circulating pro-inflammatory cytokines (96, 106).
Potential disruption of gut microbiome caused by COVID-19 result in colonization of the gut with opportunistic pathogens, increasing the risk of biofilm formation that was shown to be associated with many GI diseases (158, 159) that are resistant to standard treatment. In a recent study a probiotic combination of Saccharomyces boulardii, Lactobacillus acidophilus, Bifidobacterium breve, and L. rhamnosus was shown to provide anti-biofilm activity in vitro against polymicrobial biofilms formed by C. albicans or C. tropicalis when combined with Escherichia coli and Serratia marcescens (160). Thus, use of probiotics may help in restoring gut microbiome balance, as well as, reducing the risk of biofilm formation.
The underlying mechanism/s for the contribution of probiotics to rebalance the microbiome, normalize neuroendocrine system, and reduce depression may be mediated by: (1) immunomodulatory effects of the probiotics, (2) ability to decrease intestinal translocation of microbial constituents across the gut intestinal barrier, and (3) inhibition of digestive biofilms. However, the studies that suggest these potential influences are mainly associative, with little mechanistic considerations. The potential involvement of immunomodulatory effects of probiotics (e.g., B. pseudocatenulatum) is indicated by a pronounced decrease of stress-induced interleukin (IL)-18 levels in the serum as well as decrease of basal and stress-induced interferon gamma (IFNγ) levels in the small intestine of probiotic-treated animals. Additionally, probiotic incorporation led to a reduction of corticosterone in stool and catecholamines in the hypothalamus, as well as anxiety-like behavior (161). Such findings indicate that probiotic therapies may produce certain benefits over current therapeutic drugs or play an adjunctive role. As a result of this, we propose that probiotics may play an important part of a comprehensive approach to rebalancing the microbiome and treating depression.
Lifestyle Habits and Depression
The last element proposed for combating depression focuses on 3 specific lifestyle habits; exercise, sleep, and stress management. The gut microbiota is an extremely dynamic system that constantly changes over time due to a combination of various factors and lifestyle habits. Since the beginning of COVID-19 pandemic, the fear of being infected, loss of loved ones, lockdown, and social isolation all together caused marked changes in individuals' behaviors in an attempt to combat the stress caused by these factors. This in turn caused a huge impact on daily activities in multiple aspects including physical activity (162), eating habits (128), and sleeping (163). We propose that these three lifestyle habits are critical as a result of the existing evidence that demonstrates their unique benefits in improving gut health and decreasing depression.
Exercise and Depression
The first lifestyle habit addressed is exercise. Studies showed that COVID-19 negatively affected daily physical activity, thereby shifting individuals to more sedentary lifestyle (164). Zheng et al., investigated the impact of the pandemic on physical activity in a sample of 631 young adults. In total, 70% of the participants reported decreased physical activity levels since the onset of the COVID-19 pandemic with a significant increase in sleep duration and sedentary behavior (e.g., computer/video games, sitting talking on telephone, doing computer/paper work), consistent with data reported by Moore et al., demonstrating significant decline in all types of physical activity among children and adolescents in Canada since the beginning of the pandemic (164). A recent review has shown that regular exercise plays an important role in increasing the diversity of the gut microbiota and maintaining the balance between beneficial and pathogenic bacterial communities (165). Consequently, this major decline in physical activity eventually causes significant disruption to the gut microbiota (i.e., dysbiosis) which in turn plays a role in multiple neuropsychiatric disorders (e.g., depression), as reviewed by Belizario and Faintuch (166).
We propose exercise as a significant lifestyle habit to consider due to its relationship with the microbiome and depression. Exercising is associated with various benefits that can be directly or indirectly related to microbiome health. For example, studies have indicated that many conditions involving inflammation, like depression, can be treated through exercise (167). Additionally, in clinical studies, exercise has been shown to reduce depressive symptoms (168–171) and the effects of stress-induced intestinal barrier dysfunction has been demonstrated using murine models (172).
The underlying mechanism/s responsible for the reduction in depression symptoms associated with exercise is unclear. However, a number of mechanisms have been proposed including the thermogenic, endorphin, and the monoamine hypothesis. Animal studies provide supportive evidence to the latter hypothesis where an increase in the availability of the brain neurotransmitters dopamine (a feel-good endorphin), norepinephrine, and serotonin have been observed in plasma, urine and various brain regions following exercise (168). Recent rodent studies continue to suggest that exercise may alleviate depression through common neuro-molecular mechanisms (173, 174), including increased expression of neurotrophic factors [i.e., Brain-derived neurotropic factor (BDNF)], regulation of HPA-axis activity (175), increased availability of serotonin and norepinephrine (176), and reduced systemic inflammatory signaling (177). These activities stimulate the development of new neurons, increase cerebral vasculature in human as well as animal studies (178, 179) and synaptic connections between neurons thereby boosting cognitive function with the ultimate result of reducing depression symptoms.
While the exact reasons are unknown, the evidence is abundantly clear that exercise helps to decrease depression and depressive symptoms. As a result of these findings, we propose that exercise is a critical lifestyle element to consider when dealing with depression. Specifically, performing a minimum of 150 min of moderate exercise every week, consistent with the U.S. government's Physical Activity Guidelines for Americans (180) and the American Heart Association's recommendations, may improve depressive symptoms (181).
Sleep and Depression
In addition to exercise, sleep is another essential lifestyle element to consider, as it is also associated with the microbiome and depression. When it comes to sleep and the microbiome, sleep has been shown to have a direct impact on the microbiome in many ways. For example, murine studies have demonstrated the link between sleep deprivation or disruption and microbial dysbiosis (182, 183).
Gualano et al., investigated the effects of Covid-19 lockdown on mental health and sleep disturbances in a clinical study in Italy and showed an increase in the prevalence of depression and anxiety symptoms and sleep disturbances during the Covid-19 lockdown compared to before the lockdown 24.7 and 23.2% of the participants reporting depression and anxiety symptoms, respectively; while 42.2% had sleep disturbances (163). This was in agreement with other studies that assessed the impact of COVID-19 lockdown on mental health in Italy and China (21, 30, 184–186).
Lack of sleep may lead to and exacerbate depression and depressive symptoms. In fact, numerous studies have shown that individuals with depression experience trouble falling or staying asleep (187, 188), and individuals who have sleeping problems are more likely to become depressed in the future (189–192). These findings highlight the critical role sleep plays in depression and its impact on the microbiome. As a result of this, we propose that obtaining adequate sleep is a vital lifestyle element to consider when treating depression. Specifically, adults should attempt to receive 7–9 h of sleep per night, as suggested by the National Sleep Foundation's recommendations (193).
The relationship between sleep and the microbiome is bi-directional where the microbiome can affect our sleep pattern and sleep can impact the microbiome composition. Smith et al. (194) investigated the associations between sleep and the microbiome in human clinical studies and showed that microbiome diversity (richness and Shannon diversity indices) was positively correlated with sleep efficiency, and total sleep time, while negatively correlated with sleep fragmentation (characterized by repetitive short interruption of sleep). These findings suggest that gut microbiome diversity promotes healthier sleep. Specifically, richness within the phyla Firmicutes and Bacteroidetes were positively correlated with sleep efficiency. Other authors showed that in humans these two phyla are associated with sleep quality, circadian rhythm (internal process that regulates the sleep-wake cycle) and food intake, factors known to influence sleep quality (195, 196). Unlike Firmicutes and Bacteroidetes phyla, richness within the Actinobacteria phylum was shown to negatively correlate with the number of awakenings. In other words, increased richness within this phylum contributes to high sleep quality. The positive effect of the identified phyla on sleep could be attributed to their ability to secret the metabolite γ-aminobutyric acid, a neurotransmitter known to promote sleep (197).
The effect of sleep on the gut microbiome has also been investigated. Studies by Li et al. showed that a shift in circadian rhythms can elicit a change in the intestinal microbiota (198). While a publication by Benedict et al. showed that after 2 days of partial sleep deprivation vs. after 2 days of normal sleep, subjects had a significant increase in Firmicutes:Bacteroidetes (F/B) ratio, a measure thought to influence the maintenance of normal intestinal homeostasis. An increase or decrease in F/B ratio is often regarded as dysbiosis, however newer studies also question the reproducibility of this ratio (199). Furthermore, 2 days partial sleep deprivation resulted in an increase in the abundance of Coriobacteriaceae and Erysipelotrichaceae families, and reduced levels of Tenericutes (all P < 0.05). However, partial sleep deprivation did not have any effect of ß-diversity. These findings suggest that sleep deprivation in human subjects can induce alterations in bacterial families residing in the gut (200).
Interestingly, Li et al. (198) suggested that a bidirectional connection between the gut microbiome, sleep and depression exists with inflammation and endocrine hormones contributing to this process. In this regard, sleep loss, prolonged disturbances of circadian rhythms, and depression affects the metabolism of commensal gut bacteria and leads to alterations in microbiome structure including a reduction in members of the Lactobacillaceae family and an increase in Enterococci, Bacteroides multiforme, Lachnospiraceae, and Ruminococcaceae. These changes lead to microbiome dysbiosis (201–203). Damage to the gut epithelial barrier, as happens in leaky gut, will allow bacterial cells and their harmful metabolites to stimulate inflammatory immune responses thereby exciting the vagus and spinal afferent nerves (204). It has been postulated that the underlying mechanism for this process may be that the inflammatory response brought about by gut microbiome dysbiosis exacerbates insomnia and depression (112, 205–208).
Stress and Depression
The last major lifestyle habit that comprises a complete approach to combating depression is effective stress management. We propose that stress management is an important lifestyle habit mainly because stress heavily impacts the microbiome and is related to depression. As a result of the gut-brain axis, there is always a link to the gut when a stress response occurs (97). For this reason, stress has been shown to have a significantly negative impact on the microbiome as it affects intestinal barrier function (209) and host-microbe interactions (210). Studies have demonstrated that various types of stressors, including the burden of COVID-19 pandemic (211–213), heat, social rejection, and separation, are responsible for compositional alterations to the gut microbiome (207, 214–218). In this regard, a murine study by Galley et al., showed that colonic lumen tissues were differentially affected by exposure to the psychological stressor imposition of restraint. Specifically, restraint stress significantly reduced the alpha diversity in the microbiome of stressed mice compared to controls. Moreover, a significant reduction in the relative abundance of the genera Lactobacillus and Adlercreutzia was observed (219).
In response to stress, the HPA axis produces cortisol to help the body deal with danger (220). This type of stress response relates to an array of neurological issues, including depression. In fact, studies have shown that the hyperactivity of the HPA axis is one of the most common neurobiological changes in patients suffering from depression (221, 222). Additional research has also demonstrated a “robust and causal” relationship between stressful life events and depression (223). Furthermore, a gastrointestinal connection exists as stress is a top risk factor for IBS, which is often linked to depression as outlined in recent review articles (224–226).
As such, we propose that effective stress management is an essential lifestyle habit to explore when combating depression. Specifically, the following stress management strategies, which include, but are not limited to, the following: taking time for hobbies, fostering healthy friendships, volunteering, seeking counseling when needed, and other relaxation techniques like yoga, meditation, deep breathing, and massage treatment.
While we understand that various lifestyle habits can positively impact depression, treatment should also include psychological, neuropsychiatric and pharmacological approaches. We propose that exercise, sleep, and stress management are among the most accessible methods to individuals that can be relatively easily implemented. Given the various research studies that have linked the microbiome and depression with these three particular lifestyle habits, we propose that this approach may hold promise.
Proposed Approach to Rebalance the Microbiome and Reduce Depression
Having highlighted a relationship between depression and the microbiome, and the given association of COVID19 with stress and depression, we propose that one approach to combat depression should include potential diet modification, nutritional supplements and lifestyle changes. Specifically, increasing the abundance of beneficial organisms that exhibit a reduction in their levels during stress and depression (e.g., F. prausnitzii, Bifidobacterium, and Lactobacillus) and decrease the levels of pathogens such as Candida, Corynebacterium and Ruthenibacterium should provide an improved gut microbiome.
A Prototypic Diet
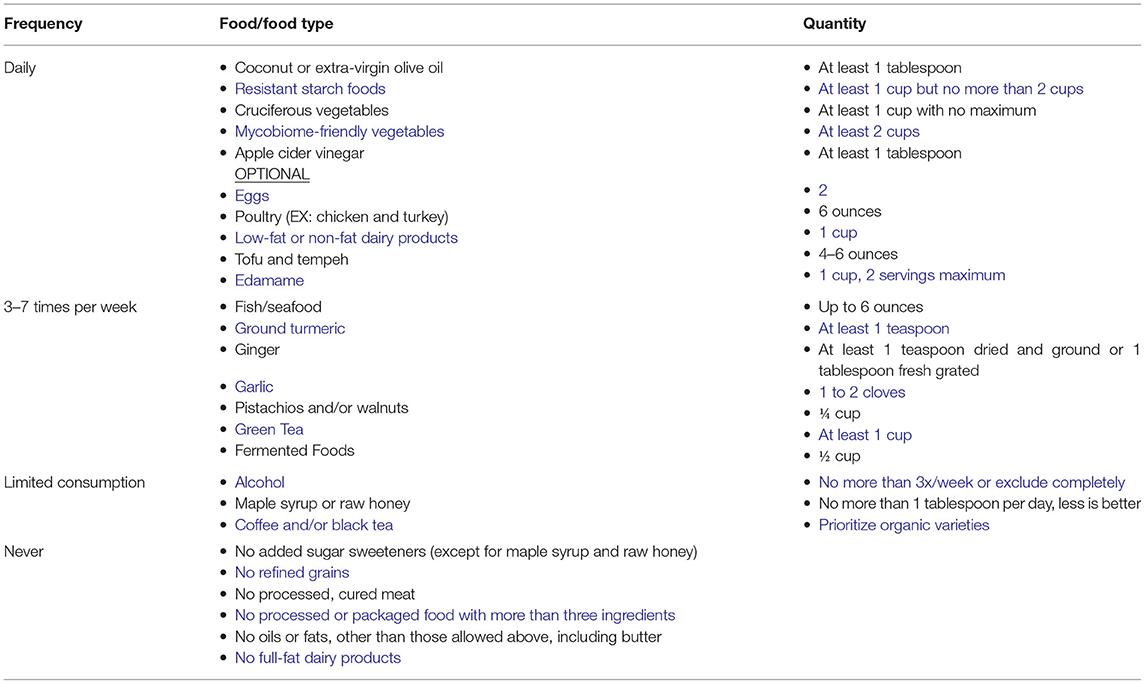
Healthy eating habits involving limiting consumption of sugar and refined carbohydrates and are associated with a decreased risk of depression. One such diet, the Total Gut Balance (TGB) (97) incorporates all aspects of the microbiome using foods and lifestyle practices to support a healthy gut and reduce overall inflammation with potent anti-inflammatory and antioxidant foods rich in targeted vitamins, minerals, and other beneficial constituents. This diet is nutritionally balanced, whole-food based, low- glycemic rich in fiber and resistant starches, low in sugar, full of mono- and polyunsaturated plant fats, low in saturated fat and animal fat, incorporating lean plant protein and animal protein primarily from seafood, with some poultry, as outlined in Table 1. As shown, this diet closely resembles the “Mediterranean” diet in the makeup of the constituents (227).
Adherence to the TGB diet was evaluated in a small clinical study of 10 individuals (228) where it was noted that the TGB diet counteracts microbiome imbalance.
Overabundance of Proteobacteria in the GI tract reflects microbiome imbalance and an unstable gut microbiome structure that could lead to inflammatory symptoms. Moreover, previous animal studies suggest that the microbial dysbiosis in IBD is associated with an increase in the levels of pathogenic Proteobacterial taxa such as Enterobacteriaceae (e.g., E. coli) and a reduction in protective Firmicutes species such as F. prausnitzii (229).
Individuals participating in the TGB exhibited an increase in F. prausnitzii, an organism reduced in depressed subjects. In contrast, high levels of F. prausnitzii are found in healthy subjects (230). F. prausnitzii is a butyrate producer that has shown anti-inflammatory effects using in vitro and in vivo mouse colitis models. Finally, a decrease in the abundance of pathogenic Candida spp. was also observed in TGB participants.
The observed increase in the level of Roseburia, a Gram-positive anaerobic bacterium, may have a positive impact on the microbiome of depressed individuals. Roseburia is a commensal bacterium that produces short chain fatty acids (SCFA), especially butyrate, which are anti-inflammatory and shown to help maintain immunity (231).
In addition to the described shift in the bacterial and fungal gut communities of TGB participants, an increase in abundance of beneficial fungi and bacteria and a corresponding decrease in harmful microorganisms, a shift associated with reported improved GI symptoms, less fatigue, more energy, better sleep, and fewer cravings for empty-calorie foods was also reported (228).
Studies have shown that bacteria inhabiting our gut have the ability to produce and/or consume a wide range of mammalian neurotransmitters, including dopamine, norepinephrine, serotonin, or gamma-aminobutyric acid (GABA) (91–95, 232). Since it is well-known that disturbance of these neurotransmitters is linked to a variety of mental disorders including depression (233–237), a target of many antidepressant pharmacological treatments, a possible approach to rebalance these neurotransmitters, although not completely discovered, is by modulating the abundance of these bacteria. However, some gut-derived neuro transmitters function differently from brain-derived neurotransmitters (238). It has been reported that Bifidobacterium improved the expression of Tph1 and secretion of 5-hydroxytryptophan (5-HTP) in RIN14B cells, significantly reduced depressive behaviors of mice, and increased the level of 5-hydroxytryptamine and brain-derived neurotrophic factor concentration in brain (239). Furthermore, in some studies, autistic patients were found to have increased level of Clostridia that were associated with production of toxic compounds, such as 3-hydroxypropionic acid (240) and p-cresol (241), found to inhibit the dopamine beta-hydroxylase that converts dopamine to norepinephrine in neurons in the brain and in the sympathetic nervous system (242). This increase in dopamine in neurons leads to production of oxidative species, depletion of glutathione and subsequently brain damage (240, 243, 244). Thus, increasing these beneficial organisms while decreasing pathogenic ones through diet may help in rebalancing neurotransmitter and reducing symptoms of some mental disorders including depression. However, investigations in this regard is still ongoing and thus can't be used as a solo treatment option.
Probiotics
Based on association between COVID-19 infection and gut microbiome disruption, a logical approach to help balance the gut microbiome is administration of probiotics. Probiotic, as defined by World Health Organization, is “live microorganisms which when administered in adequate amounts confer a health benefit on the host” (245, 246). Not only should probiotic consumption restore the gut balance, it may also decrease the likelihood of colonization of the gut by opportunistic pathogens (247), as reported in many studies that analyzed the gut microbiome in COVID-19 infected patients (57, 58). Published reports have shown that use of a probiotic helped in supporting the immune system (248), as well as leading to a marked reduction in the incidence of respiratory tract infection (249) Furthermore, studies have also shown that lactic acid bacteria probiotics possess antiviral properties (250) which has been used in many fields including agriculture (251), poultry (252), and medicine (253).
Lifestyle Recommendations
Eating to enhance the microbiome may help to achieve better health, but there is compelling research to show that other aspects of life, from the environment you live in, as well as sleep, stress, and exercise, that can significantly impact all gut microbes. While genetics is certainly part of this equation, and diet is another component, research continues to demonstrate that lifestyle factors play an important role in chronic diseases (254, 255). Because depression has been shown to be linked to dysbiosis in the microbiome, knowing what you can do to make your lifestyle habits work for you instead of against you is also important. To aid this approach, we propose a number of lifestyle recommendations that may enhance the microbiome balance which should result in reduced depression. A summation of the recommended lifestyle targets is listed in Table 2.
Conclusion
As a result of the toll COVID-19 has had on mental health, and society's imperfect understanding and inability to adequately treat depression, we propose it is important to examine more thoroughly new potential methods to prevent and treat depression. While this topic still needs further examination, research is extremely promising and should offer a glimpse of hope. The overall goal of the present manuscript is to offer suggestions that incorporate an encompassing approach to combat depression. As such, we want to emphasize the importance of rebalancing the gut through probiotics, diet, and lifestyle changes. However, we also propose that more can and should be done to achieve this goal and improve the lives of individuals who are struggling.
Regardless of whether people have seen a decrease in their own mental well-being since the start of COVID-19, everyone needs to be responsive and work together to effectively bring an end to this mental health crisis. We propose that the first step toward progress is awareness. However, each of us cope with increased stressors in our own unique ways, such as remaining busy, spending time with family and friends, etc. It is critical to note that some community members are overwhelmed and are desperately in need of assistance. For these reasons, we recommend checking in on your loved ones and asking how you might be able to assist them during these unprecedented times.
Since the start of time, humans have continued to evolve and adapt to whatever situations they are presented with. We propose that the COVID-19 pandemic is no different from these situations. Therefore, if you find yourself constantly worrying about the future, we hope this paper has eased your concerns and allowed you to adopt our optimistic view that we will get through this and be better because of it.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
Ethical review and approval was not required for the study on human participants in accordance with the local legislation and institutional requirements. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
MG: conceptualization and funding acquisition. MF and MG: literature acquisition and writing—original draft. MG, TM, RB, and AG: writing—review and editing. All authors contributed to the article and approved the submitted version.
Funding
This work was supported in part by a National Institutes of Health Grant # R01AI145289- 01A1 to MG.
Conflict of Interest
MF was an employee at BIOHM Health LLC. MG was the Co-founder of BIOHM Health LLC and author of the Total Gut Balance Book.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. AMERICA'S STATE OF MIND. U.S. Trends in Medication Use for Depression, Anxiety and Insomnia. (2020). Available online at: https://www.express-scripts.com/corporate/americas-state-of-mind-report (accessed April 23, 2021).
2. Mental health survey 2020. (2020). Available online at: https://www.singlecare.com/blog/news/mental-health-survey/ (accessed May 10, 2020).
3. A crisis mental-health hotline has seen an 891% spike in calls. (2020). Available online at: https://www.cnn.com/2020/04/10/us/disaster-hotline-call-increase-wellness-trnd/index.html (accessed June 10, 2021).
4. Launch of the Policy Brief on COVID-19 and Mental Health. United Nations (2020). Available online at: https://www.un.org/en/coronavirus/mental-health-services-are-essential-part-all-government-responses-covid-19 (accessed October 1, 2021).
5. aan het Rot M, Mathew SJ, Charney DS. Neurobiological mechanisms in major depressive disorder. CMAJ. (2009) 180:305–13. doi: 10.1503/cmaj.080697
6. Gerhard DM, Wohleb ES, Duman RS. Emerging treatment mechanisms for depression: focus on glutamate and synaptic plasticity. Drug Discov Today. (2016) 21:454–64. doi: 10.1016/j.drudis.2016.01.016
7. Ledford H. Medical research: if depression were cancer. Nature. (2014) 515:182–4. doi: 10.1038/515182a
8. Johnson J, Weissman MM, Klerman GL. Service utilization and social morbidity associated with depressive symptoms in the community. JAMA. (1992) 267:1478–83. doi: 10.1001/jama.1992.03480110054033
9. Hawton K, Casanas ICC, Haw C, Saunders K. Risk factors for suicide in individuals with depression: a systematic review. J Affect Disord. (2013) 147:17–28. doi: 10.1016/j.jad.2013.01.004
10. McCrone P, Dhanasiri S, Patel A, Knapp M, Lawton-Smith S. Paying the Price: The Cost of Mental health Care in England to 2026. London: The King's Fund (2008).
11. Nierenberg AA, Amsterdam JD. Treatment-resistant depression: definition and treatment approaches. J Clin Psychiatry. (1990) 51Suppl:39–47.
12. Thase M, Rush A. Treatment-resistant depression In: Bloom FE, editor. The Fourth Generation of Progress. New York, NY: Raven Press (1995).
13. Amsterdam J. Treatment-resistant depression: progress and limitations. Psych Ann. (1998) 28:633–40. doi: 10.3928/0048-5713-19981101-08
14. Holtzheimer PE, Mayberg HS. Stuck in a rut: rethinking depression and its treatment. Trends Neurosci. (2011) 34:1–9. doi: 10.1016/j.tins.2010.10.004
15. Keller MB, Shapiro RW, Lavori PW, Wolfe N. Relapse in major depressive disorder: analysis with the life table. Arch Gen Psychiatry. (1982) 39:911–5. doi: 10.1001/archpsyc.1982.04290080031005
16. Paykel E. Epidemiology of refractory depression. In: Nolen WA ZJ, Roose SP, Amsterdam JD, editors. Refractory Depression: Current Strategies and Future Directions. New York, NY: Wiley (1994).
17. Group UER. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. (2003) 361:799–808. doi: 10.1016/S0140-6736(03)12705-5
18. Fink M. ECT has proved effective in treating depression. Nature. (2000) 403:826. doi: 10.1038/35002776
19. Sterling P. ECT damage is easy to find if you look for it. Nature. (2000) 403:242. doi: 10.1038/35002188
20. How effective are antidepressants? (2020). Available online at: https://www.ncbi.nlm.nih.gov/books/NBK361016? (accessed September 16, 2020).
21. Huang Y, Zhao N. Generalized anxiety disorder, depressive symptoms and sleep quality during COVID-19 outbreak in China: a web-based cross-sectional survey. Psychiatry Res. (2020) 288:112954. doi: 10.1016/j.psychres.2020.112954
22. Abdulah DM, Musa DH. Insomnia and stress of physicians during COVID-19 outbreak. Sleep Med X. (2020) 2:100017. doi: 10.1016/j.sleepx.2020.100017
23. Ahmad A, Rahman I, Agarwal M. Factors influencing mental health during Covid-19 outbreak: an exploratory survey among Indian population. medRxiv. (2020) 2020.05.03.20081380. doi: 10.1101/2020.05.03.20081380
24. Murthy RS. National Mental Health Survey of India 2015-2016. Indian J Psychiatry. (2017) 59:21–6. doi: 10.4103/psychiatry.IndianJPsychiatry_102_17
25. Bai Y, Wang X, Huang Q, Wang H, Gurarie D, Ndeffo-Mbah M, et al. SARS-CoV-2 infection in health care workers: a retrospective analysis and model study. medRxiv. (2020) 2020.03.29.20047159. doi: 10.1101/2020.03.29.20047159
26. Beck F, Leger D, Fressard L, Peretti-Watel P, Verger P, Coconel G. Covid-19 health crisis and lockdown associated with high level of sleep complaints and hypnotic uptake at the population level. J Sleep Res. (2021) 30:e13119. doi: 10.1111/jsr.13119
27. Casagrande M, Favieri F, Tambelli R, Forte G. The enemy who sealed the world: effects quarantine due to the COVID-19 on sleep quality, anxiety, and psychological distress in the Italian population. Sleep Med. (2020) 75:12–20. doi: 10.1016/j.sleep.2020.05.011
28. Bohlken J, Schomig F, Seehagen T, Kohler S, Gehring K, Roth-Sackenheim C, et al. [Experience of practice-based psychiatrists and neurologists during the COVID-19 pandemic]. Psychiatr Prax. (2020) 47:214–7. doi: 10.1055/a-1159-5575
29. Ammar A, Brach M, Trabelsi K, Chtourou H, Boukhris O, Masmoudi L, et al. Effects of COVID-19 home confinement on eating behaviour and physical activity: results of the ECLB-COVID19 international online survey. Nutrients. (2020) 12:61583. doi: 10.3390/nu12061583
30. Cellini N, Canale N, Mioni G, Costa S. Changes in sleep pattern, sense of time and digital media use during COVID-19 lockdown in Italy. J Sleep Res. (2020) 29:e13074. doi: 10.1111/jsr.13074
31. Priego-Parra BA, Triana-Romero A, Pinto-Gálvez SM, Ramos CD, Salas-Nolasco O, Reyes MM, et al. Anxiety, depression, attitudes, and internet addiction during the initial phase of the 2019 coronavirus disease (COVID-19) epidemic: A cross-sectional study in México. medRxiv. (2020) 2020:2020.05.10.20095844. doi: 10.1101/2020.05.10.20095844
32. Taylor S. COVID stress syndrome: clinical and nosological considerations. Curr Psychiatry Rep. (2021) 23:19. doi: 10.1007/s11920-021-01226-y
33. Taylor S, Landry CA, Paluszek MM, Fergus TA, McKay D, Asmundson GJG. COVID stress syndrome: concept, structure, and correlates. Depress Anxiety. (2020) 37:706–14. doi: 10.1002/da.23071
34. Taylor S, Landry CA, Paluszek MM, Fergus TA, McKay D, Asmundson GJG. Development and initial validation of the COVID Stress Scales. J Anxiety Disord. (2020) 72:102232. doi: 10.1016/j.janxdis.2020.102232
35. Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. (2020) 579:270–3. doi: 10.1038/s41586-020-2012-7
36. Li W, Moore MJ, Vasilieva N, Sui J, Wong SK, Berne MA, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. (2003) 426:450–4. doi: 10.1038/nature02145
37. Baranowski E, Ruiz-Jarabo CM, Domingo E. Evolution of cell recognition by viruses. Science. (2001) 292:1102–5. doi: 10.1126/science.1058613
38. Iwata-Yoshikawa N, Okamura T, Shimizu Y, Hasegawa H, Takeda M, Nagata N. TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection. J Virol. (2019) 93:18. doi: 10.1128/JVI.01815-18
39. Hoffmann M, Kleine-Weber H, Schroeder S, Kruger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. (2020) 181:271–80.e8. doi: 10.1016/j.cell.2020.02.052
40. Modes of transmission of virus causing COVID-19: implications for IPC precaution recommendations. (2020). Available online at: https://www.who.int/news-room/commentaries/detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations (accessed April 01, 2020).
41. Hikmet F, Mear L, Edvinsson A, Micke P, Uhlen M, Lindskog C. The protein expression profile of ACE2 in human tissues. Mol Syst Biol. (2020) 16:e9610. doi: 10.15252/msb.20209610
42. Kumar S, Nyodu R, Maurya VK, Saxena SK. Morphology, genome organization, replication, and pathogenesis of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). In: Saxena SK, editor. Coronavirus Disease 2019 (COVID-19): Epidemiology, Pathogenesis, Diagnosis, and Therapeutics. Singapore: Springer Singapore (2020). doi: 10.1007/978-981-15-4814-7_3
43. Fox SE, Akmatbekov A, Harbert JL, Li G, Quincy Brown J, Vander Heide RS. Pulmonary and cardiac pathology in African American patients with COVID-19: an autopsy series from New Orleans. Lancet Respir Med. (2020) 8:681–6. doi: 10.1016/S2213-2600(20)30243-5
44. Bradley BT, Maioli H, Johnston R, Chaudhry I, Fink SL, Xu H, et al. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet. (2020) 396:320–32. doi: 10.1016/S0140-6736(20)31305-2
45. Diao B, Wang C, Wang R, Feng Z, Tan Y, Wang H, et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. medRxiv. (2020) 2020:2020.03.04.20031120. doi: 10.1101/2020.03.04.20031120
46. Su H, Yang M, Wan C, Yi LX, Tang F, Zhu HY, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. (2020) 98:219–27. doi: 10.1016/j.kint.2020.04.003
47. Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. (2020) 8:420–2. doi: 10.1016/S2213-2600(20)30076-X
48. Yao XH, Li TY, He ZC, Ping YF, Liu HW, Yu SC, et al. [A pathological report of three COVID-19 cases by minimal invasive autopsies]. Zhonghua Bing Li Xue Za Zhi. (2020) 49:411–7. doi: 10.3760/cma.j.cn112151-20200312-00193
49. Zhang W, Du RH, Li B, Zheng XS, Yang XL, Hu B, et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. (2020) 9:386–9. doi: 10.1080/22221751.2020.1729071
50. Paniz-Mondolfi A, Bryce C, Grimes Z, Gordon RE, Reidy J, Lednicky J, et al. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). J Med Virol. (2020) 92:699–702. doi: 10.1002/jmv.25915
51. Moriguchi T, Harii N, Goto J, Harada D, Sugawara H, Takamino J, et al. A first case of meningitis/encephalitis associated with SARS-coronavirus-2. Int J Infect Dis. (2020) 94:55–8. doi: 10.1016/j.ijid.2020.03.062
52. Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. (2020) 158:1831–3 e3. doi: 10.1053/j.gastro.2020.02.055
53. Dietrich CG, Hubner D, Marx G, Bickenbach J, Bootsveld A. Primary presentation of COVID-19 solely with gastrointestinal symptoms: a problem for the containment of the disease. Eur J Gastroenterol Hepatol. (2020) 32:1475–8. doi: 10.1097/MEG.0000000000001922
54. An P, Chen H, Ren H, Su J, Ji M, Kang J, et al. Gastrointestinal symptoms onset in COVID-19 patients in Wuhan, China. Dig Dis Sci. (2020) 12:1–10. doi: 10.1007/s10620-020-06693-6
55. Pan L, Mu M, Yang P, Sun Y, Wang R, Yan J, et al. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: a descriptive, cross-sectional, multicenter study. Am J Gastroenterol. (2020) 115:766–73. doi: 10.14309/ajg.0000000000000620
56. Jin X, Lian JS, Hu JH, Gao J, Zheng L, Zhang YM, et al. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019. (COVID-19) with gastrointestinal symptoms. Gut. (2020) 69:1002–9. doi: 10.1136/gutjnl-2020-320926
57. Gu S, Chen Y, Wu Z, Chen Y, Gao H, Lv L, et al. Alterations of the gut microbiota in patients with coronavirus disease 2019 or H1N1 influenza. Clin Infect Dis. (2020) 71:2669–78. doi: 10.1093/cid/ciaa709
58. Zuo T, Zhang F, Lui GCY, Yeoh YK, Li AYL, Zhan H, et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. (2020) 159:944–55.e8. doi: 10.1053/j.gastro.2020.05.048
59. Geva-Zatorsky N, Sefik E, Kua L, Pasman L, Tan TG, Ortiz-Lopez A, et al. Mining the human gut microbiota for immunomodulatory organisms. Cell. (2017) 168:928–43.e11. doi: 10.1016/j.cell.2017.01.022
60. Hashimoto T, Perlot T, Rehman A, Trichereau J, Ishiguro H, Paolino M, et al. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature. (2012) 487:477–81. doi: 10.1038/nature11228
61. Li N, Ma WT, Pang M, Fan QL, Hua JL. The commensal microbiota and viral infection: a comprehensive review. Front Immunol. (2019) 10:1551. doi: 10.3389/fimmu.2019.01551
62. Kaur H, Bose C, Mande SS. Tryptophan metabolism by gut microbiome and gut-brain-axis: an in silico analysis. Front Neurosci. (2019) 13:1365. doi: 10.3389/fnins.2019.01365
63. Zuo T, Liu Q, Zhang F, Lui GC-Y, Tso EYK, Yeoh YK, et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut. (2021) 70:276. doi: 10.1136/gutjnl-2020-322294
64. Zuo T, Zhan H, Zhang F, Liu Q, Tso EYK, Lui GCY, et al. Alterations in fecal fungal microbiome of patients with COVID-19 during time of hospitalization until discharge. Gastroenterology. (2020) 159:1302–10.e5. doi: 10.1053/j.gastro.2020.06.048
65. Yu L, Tong Y, Shen G, Fu A, Lai Y, Zhou X, et al. Immunodepletion with hypoxemia: a potential high risk subtype of coronavirus disease 2019. medRxiv. (2020) 2020:2020.03.03.20030650. doi: 10.1101/2020.03.03.20030650
66. Leal T, Costa E, Arroja B, Goncalves R, Alves J. Gastrointestinal manifestations of COVID-19: results from a European centre. Eur J Gastroenterol Hepatol. (2021) 33:691–4. doi: 10.1097/MEG.0000000000002152
67. Luo S, Zhang X, Xu H. Don't overlook digestive symptoms in patients with 2019 novel coronavirus disease (COVID-19). Clin Gastroenterol Hepatol. (2020) 18:1636–7. doi: 10.1016/j.cgh.2020.03.043
68. Han C, Duan C, Zhang S, Spiegel B, Shi H, Wang W, et al. Digestive symptoms in COVID-19 patients with mild disease severity: clinical presentation, stool viral RNA testing, and outcomes. Am J Gastroenterol. (2020) 115:916–23. doi: 10.14309/ajg.0000000000000664
69. Filatov A, Sharma P, Hindi F, Espinosa PS. Neurological complications of coronavirus disease (COVID-19): encephalopathy. Cureus. (2020) 12:e7352. doi: 10.7759/cureus.7352
70. Karimi N, Sharifi Razavi A, Rouhani NJIRCMJ. Frequent convulsive seizures in an adult patient with COVID-19: a case report. Iran Red Crescent Med J. (2020) 3:20200301. doi: 10.5812/ircmj.102828
71. Sheraton M, Deo N, Kashyap R, Surani S. A review of neurological complications of COVID-19. Cureus. (2020) 12:e8192. doi: 10.7759/cureus.8192
72. Bagheri SH, Asghari A, Farhadi M, Shamshiri AR, Kabir A, Kamrava SK, et al. Coincidence of COVID-19 epidemic and olfactory dysfunction outbreak in Iran. Med J Islam Repub Iran. (2020) 34:62. doi: 10.1101/2020.03.23.20041889
73. Graham EL, Clark JR, Orban ZS, Lim PH, Szymanski AL, Taylor C, et al. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized Covid-19 “long haulers”. Ann Clin Transl Neurol. (2021) 8:1073–85. doi: 10.1002/acn3.51350
74. Thakur KT, Miller EH, Glendinning MD, Al-Dalahmah O, Banu MA, Boehme AK, et al. COVID-19 neuropathology at Columbia University Irving Medical Center/New York Presbyterian Hospital. Brain. (2021) 15:awab148. doi: 10.1093/brain/awab148
75. Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. (2012) 13:701–12. doi: 10.1038/nrn3346
76. Osadchiy V, Martin CR, Mayer EA. Gut microbiome and modulation of CNS function. Compr Physiol. (2019) 10:57–72. doi: 10.1002/cphy.c180031
77. Amaral FA, Sachs D, Costa VV, Fagundes CT, Cisalpino D, Cunha TM, et al. Commensal microbiota is fundamental for the development of inflammatory pain. Proc Natl Acad Sci USA. (2008) 105:2193–7. doi: 10.1073/pnas.0711891105
78. Bravo JA, Forsythe P, Chew MV, Escaravage E, Savignac HM, Dinan TG, et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc Natl Acad Sci USA. (2011) 108:16050–5. doi: 10.1073/pnas.1102999108
79. Dinan TG, Cryan JF. Regulation of the stress response by the gut microbiota: implications for psychoneuroendocrinology. Psychoneuroendocrinology. (2012) 37:1369–78. doi: 10.1016/j.psyneuen.2012.03.007
80. Marx W, Moseley G, Berk M, Jacka F. Nutritional psychiatry: the present state of the evidence. Proc Nutr Soc. (2017) 76:427–36. doi: 10.1017/S0029665117002026
81. Hoban AE, Stilling RM, Ryan FJ, Shanahan F, Dinan TG, Claesson MJ, et al. Regulation of prefrontal cortex myelination by the microbiota. Transl Psychiatry. (2016) 6:e774. doi: 10.1038/tp.2016.42
82. Berger J, Heinrichs M, von Dawans B, Way BM, Chen FS. Cortisol modulates men's affiliative responses to acute social stress. Psychoneuroendocrinology. (2016) 63:1–9. doi: 10.1016/j.psyneuen.2015.09.004
83. Moussaoui N, Braniste V, Ait-Belgnaoui A, Gabanou M, Sekkal S, Olier M, et al. Changes in intestinal glucocorticoid sensitivity in early life shape the risk of epithelial barrier defect in maternal-deprived rats. PLoS ONE. (2014) 9:e88382. doi: 10.1371/journal.pone.0088382
84. O'Mahony SM, Marchesi JR, Scully P, Codling C, Ceolho AM, Quigley EM, et al. Early life stress alters behavior, immunity, and microbiota in rats: implications for irritable bowel syndrome and psychiatric illnesses. Biol Psychiatry. (2009) 65:263–7. doi: 10.1016/j.biopsych.2008.06.026
85. Sudo N, Chida Y, Aiba Y, Sonoda J, Oyama N, Yu XN, et al. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J Physiol. (2004) 558:263–75. doi: 10.1113/jphysiol.2004.063388
86. Dinan TG, Quigley EM, Ahmed SM, Scully P, O'Brien S, O'Mahony L, et al. Hypothalamic-pituitary-gut axis dysregulation in irritable bowel syndrome: plasma cytokines as a potential biomarker? Gastroenterology. (2006) 130:304–11. doi: 10.1053/j.gastro.2005.11.033
87. Petersen C, Round JL. Defining dysbiosis and its influence on host immunity and disease. Cell Microbiol. (2014) 16:1024–33. doi: 10.1111/cmi.12308
88. Hedayat KM, Lapraz J-C, Schuff B. Dysbiosis. In: Hedayat KM, Lapraz J-C, Schuff B, editors. The Theory of Endobiogeny. Cambridge, MA: Academic Press (2020). doi: 10.1016/B978-0-12-816965-0.00028-7
89. Wilkins LJ, Monga M, Miller AW. Defining dysbiosis for a cluster of chronic diseases. Sci Rep. (2019) 9:12918. doi: 10.1038/s41598-019-49452-y
90. Human Microbiome Project C. Structure, function and diversity of the healthy human microbiome. Nature. (2012) 486:207–14. doi: 10.1038/nature11234
91. Shishov Va, Fau - Kirovskaia TA, Kirovskaia Ta, Fau - Kudrin VS, Kudrin Vs, Fau - Oleskin AV, Oleskin AV. [Amine neuromediators, their precursors, and oxidation products in the culture of Escherichia coli K-12]. Prikl Biokhim Mikrobiol. (2009) 45:550–4. doi: 10.1134/S0003683809050068
92. Tsavkelova EA, Botvinko IV, Kudrin VS, Oleskin AV. Detection of neurotransmitter amines in microorganisms with the use of high-performance liquid chromatography. Dokl Biochem. (2000) 372:115–7.
93. Özogul F, Kuley E, ÖZOGUL Y, ÖZOGUL IJFS, Research T. The function of lactic acid bacteria on biogenic amines production by food-borne pathogens in arginine decarboxylase broth. Food Sci Technol Res. (2012) 18:795–804. doi: 10.3136/fstr.18.795
94. Özogul F. Production of biogenic amines by Morganella morganii, Klebsiella pneumoniae and Hafnia alvei using a rapid HPLC method. Eur Food Res Technol. (2004) 219:465–9. doi: 10.1007/s00217-004-0988-0
95. Clarke G, Grenham S, Scully P, Fitzgerald P, Moloney RD, Shanahan F, et al. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry. (2013) 18:666–73. doi: 10.1038/mp.2012.77
96. Clapp M, Aurora N, Herrera L, Bhatia M, Wilen E, Wakefield S. Gut microbiota's effect on mental health: The gut-brain axis. Clin Pract. (2017) 7:987. doi: 10.4081/cp.2017.987
98. Bastiaanssen TFS, Cussotto S, Claesson MJ, Clarke G, Dinan TG, Cryan JF. Gutted! unraveling the role of the microbiome in major depressive disorder. Harv Rev Psychiatry. (2020) 28:26–39. doi: 10.1097/HRP.0000000000000243
99. Rea K, Dinan TG, Cryan JF. The microbiome: A key regulator of stress and neuroinflammation. Neurobiol Stress. (2016) 4:23–33. doi: 10.1016/j.ynstr.2016.03.001
100. Barden N. Implication of the hypothalamic-pituitary-adrenal axis in the physiopathology of depression. J Psychiatry Neurosci. (2004) 29:185–93.
101. Heuser IJ, Schweiger U, Gotthardt U, Schmider J, Lammers CH, Dettling M, et al. Pituitary-adrenal-system regulation and psychopathology during amitriptyline treatment in elderly depressed patients and normal comparison subjects. Am J Psychiatry. (1996) 153:93–9. doi: 10.1176/ajp.153.1.93
102. Nickel T, Sonntag A, Schill J, Zobel AW, Ackl N, Brunnauer A, et al. Clinical and neurobiological effects of tianeptine and paroxetine in major depression. J Clin Psychopharmacol. (2003) 23:155–68. doi: 10.1097/00004714-200304000-00008
103. Roshchina G, Koroleva VI, Davydov VI. [Changes in the high-frequency electrical brain activity in the animal hypnosis state in rabbits]. Zh Vyssh Nerv Deiat Im I P Pavlova. (2010) 60:352–63. doi: 10.1007/s11055-011-9486-6
104. Goehler LE, Park SM, Opitz N, Lyte M, Gaykema RP. Campylobacter jejuni infection increases anxiety-like behavior in the holeboard: possible anatomical substrates for viscerosensory modulation of exploratory behavior. Brain Behav Immun. (2008) 22:354–66. doi: 10.1016/j.bbi.2007.08.009
105. Lyte M, Li W, Opitz N, Gaykema RP, Goehler LE. Induction of anxiety-like behavior in mice during the initial stages of infection with the agent of murine colonic hyperplasia Citrobacter rodentium. Physiol Behav. (2006) 89:350–7. doi: 10.1016/j.physbeh.2006.06.019
106. Messaoudi M, Violle N, Bisson JF, Desor D, Javelot H, Rougeot C. Beneficial psychological effects of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in healthy human volunteers. Gut Microbes. (2011) 2:256–61. doi: 10.4161/gmic.2.4.16108
107. Goehler LE. Cytokines in neural signaling to the brain. Front Neuroendocrinol. (2008) 6:337–52. doi: 10.1016/S1567-7443(07)10016-8
108. Konsman JP, Vigues S, Mackerlova L, Bristow A, Blomqvist A. Rat brain vascular distribution of interleukin-1 type-1 receptor immunoreactivity: relationship to patterns of inducible cyclooxygenase expression by peripheral inflammatory stimuli. J Comp Neurol. (2004) 472:113–29. doi: 10.1002/cne.20052
109. Schiltz JC, Sawchenko PE. Distinct brain vascular cell types manifest inducible cyclooxygenase expression as a function of the strength and nature of immune insults. J Neurosci. (2002) 22:5606–18. doi: 10.1523/JNEUROSCI.22-13-05606.2002
110. Bastiaanssen TFS, Cowan CSM, Claesson MJ, Dinan TG, Cryan JF. Making sense of the microbiome in psychiatry. Int J Neuropsychopharmacol. (2019) 22:37–52. doi: 10.1093/ijnp/pyy067
111. Dash S, Clarke G, Berk M, Jacka FN. The gut microbiome and diet in psychiatry: focus on depression. Curr Opin Psychiatry. (2015) 28:1–6. doi: 10.1097/YCO.0000000000000117
112. Foster JA, McVey Neufeld KA. Gut-brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. (2013) 36:305–12. doi: 10.1016/j.tins.2013.01.005
113. Kelly JR, Borre Y, C OB, Patterson E, El Aidy S, Deane J, et al. Transferring the blues: Depression-associated gut microbiota induces neurobehavioural changes in the rat. J Psychiatr Res. (2016) 82:109–18. doi: 10.1016/j.jpsychires.2016.07.019
114. Irritable bowel syndrome: Introduction. (2020). Available online at: https://www.hopkinsmedicine.org/health/conditions-and-diseases/irritable-bowel-syndrome-ibs (accessed September 09, 2020).
115. Kelly JR, Keane VO, Cryan JF, Clarke G, Dinan TG. Mood and microbes: gut to brain communication in depression. Gastroenterol Clin North Am. (2019) 48:389–405. doi: 10.1016/j.gtc.2019.04.006
116. Zheng P, Zeng B, Zhou C, Liu M, Fang Z, Xu X, et al. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host's metabolism. Mol Psychiatry. (2016) 21:786–96. doi: 10.1038/mp.2016.44
117. Jiang H, Ling Z, Zhang Y, Mao H, Ma Z, Yin Y, et al. Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav Immun. (2015) 48:186–94. doi: 10.1016/j.bbi.2015.03.016
118. Zheng P, Yang J, Li Y, Wu J, Liang W, Yin B, et al. Gut microbial signatures can discriminate unipolar from bipolar depression. Adv Sci (Weinh). (2020) 7:1902862. doi: 10.1002/advs.201902862
119. Aizawa E, Tsuji H, Asahara T, Takahashi T, Teraishi T, Yoshida S, et al. Possible association of Bifidobacterium and Lactobacillus in the gut microbiota of patients with major depressive disorder. J Affect Disord. (2016) 202:254–7. doi: 10.1016/j.jad.2016.05.038
120. Valles-Colomer M, Falony G, Darzi Y, Tigchelaar EF, Wang J, Tito RY, et al. The neuroactive potential of the human gut microbiota in quality of life and depression. Nat Microbiol. (2019) 4:623–32. doi: 10.1038/s41564-018-0337-x
121. Huang TT, Lai JB, Du YL, Xu Y, Ruan LM, Hu SH. Current understanding of gut microbiota in mood disorders: an update of human studies. Front Genet. (2019) 10:98. doi: 10.3389/fgene.2019.00098
122. Firth J, Marx W, Dash S, Carney R, Teasdale SB, Solmi M, et al. The effects of dietary improvement on symptoms of depression and anxiety: a meta-analysis of randomized controlled trials. Psychosom Med. (2019) 81:265–80. doi: 10.1097/PSY.0000000000000673
123. Lassale C, Batty GD, Baghdadli A, Jacka F, Sanchez-Villegas A, Kivimaki M, et al. Healthy dietary indices and risk of depressive outcomes: a systematic review and meta-analysis of observational studies. Mol Psychiatry. (2019) 24:965–86. doi: 10.1038/s41380-018-0237-8
124. Koliada A, Syzenko G, Moseiko V, Budovska L, Puchkov K, Perederiy V, et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. (2017) 17:120. doi: 10.1186/s12866-017-1027-1
125. Oddy WH, Allen KL, Trapp GSA, Ambrosini GL, Black LJ, Huang RC, et al. Dietary patterns, body mass index and inflammation: Pathways to depression and mental health problems in adolescents. Brain Behav Immun. (2018) 69:428–39. doi: 10.1016/j.bbi.2018.01.002
126. Phillips CM, Shivappa N, Hebert JR, Perry IJ. Dietary inflammatory index and mental health: A cross-sectional analysis of the relationship with depressive symptoms, anxiety and well-being in adults. Clin Nutr. (2018) 37:1485–91. doi: 10.1016/j.clnu.2017.08.029
127. Shivappa N, Hebert JR, Veronese N, Caruso MG, Notarnicola M, Maggi S, et al. The relationship between the dietary inflammatory index (DII®) and incident depressive symptoms: A longitudinal cohort study. J Affect Disord. (2018) 235:39–44. doi: 10.1016/j.jad.2018.04.014
128. Robinson E, Boyland E, Chisholm A, Harrold J, Maloney NG, Marty L, et al. Obesity, eating behavior and physical activity during COVID-19 lockdown: A study of UK adults. Appetite. (2021) 156:104853. doi: 10.1016/j.appet.2020.104853
129. Qamar N, Castano D, Patt C, Chu T, Cottrell J, Chang SL. Meta-analysis of alcohol induced gut dysbiosis and the resulting behavioral impact. Behav Brain Res. (2019) 376:112196. doi: 10.1016/j.bbr.2019.112196
130. Mutlu EA, Gillevet PM, Rangwala H, Sikaroodi M, Naqvi A, Engen PA, et al. Colonic microbiome is altered in alcoholism. Am J Physiol Gastrointest Liver Physiol. (2012) 302:G966–78. doi: 10.1152/ajpgi.00380.2011
131. Bode JC, Bode C, Heidelbach R, Durr HK, Martini GA. Jejunal microflora in patients with chronic alcohol abuse. Hepatogastroenterology. (1984) 31:30–4.
132. Elamin EE, Masclee AA, Dekker J, Jonkers DM. Ethanol metabolism and its effects on the intestinal epithelial barrier. Nutr Rev. (2013) 71:483–99. doi: 10.1111/nure.12027
133. Pollard MS, Tucker JS, Green HD. Changes in adult alcohol use consequences during the COVID-19 pandemic in the US. JAMA Netw Open. (2020) 3:e2022942. doi: 10.1001/jamanetworkopen.2020.22942
134. Gangwisch JE, Hale L, Garcia L, Malaspina D, Opler MG, Payne ME, et al. High glycemic index diet as a risk factor for depression: analyses from the Women's Health Initiative. Am J Clin Nutr. (2015) 102:454–63. doi: 10.3945/ajcn.114.103846
135. Francis HM, Stevenson RJ, Chambers JR, Gupta D, Newey B, Lim CK. A brief diet intervention can reduce symptoms of depression in young adults - A randomised controlled trial. PLoS ONE. (2019) 14:e0222768. doi: 10.1371/journal.pone.0222768
136. Jacka FN. Nutritional Psychiatry: Where to Next? EBioMedicine. (2017) 17:24–9. doi: 10.1016/j.ebiom.2017.02.020
137. Parletta N, Zarnowiecki D, Cho J, Wilson A, Bogomolova S, Villani A, et al. A Mediterranean-style dietary intervention supplemented with fish oil improves diet quality and mental health in people with depression: A randomized controlled trial (HELFIMED). Nutr Neurosci. (2019) 22:474–87. doi: 10.1080/1028415X.2017.1411320
138. Rodriguez-Palacios A, Harding A, Menghini P, Himmelman C, Retuerto M, Nickerson KP, et al. The artificial sweetener splenda promotes gut proteobacteria, dysbiosis, and myeloperoxidase reactivity in Crohn's disease-like ileitis. Inflamm Bowel Dis. (2018) 24:1005–20. doi: 10.1093/ibd/izy060
139. Noble EE, Hsu TM, Kanoski SE. Gut to brain dysbiosis: mechanisms linking western diet consumption, the microbiome, and cognitive impairment. Front Behav Neurosci. (2017) 11:9. doi: 10.3389/fnbeh.2017.00009
140. Sonnenburg ED, Sonnenburg JL. The ancestral and industrialized gut microbiota and implications for human health. Nat Rev Microbiol. (2019) 17:383–90. doi: 10.1038/s41579-019-0191-8
141. Ghosh TS, Rampelli S, Jeffery IB, Santoro A, Neto M, Capri M, et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut. (2020) 69:1218–28. doi: 10.1136/gutjnl-2019-319654
142. Morris G, Walder K, McGee SL, Dean OM, Tye SJ, Maes M, et al. A model of the mitochondrial basis of bipolar disorder. Neurosci Biobehav Rev. (2017) 74:1–20. doi: 10.1016/j.neubiorev.2017.01.014
143. Chang SC, Cassidy A, Willett WC, Rimm EB, O'Reilly EJ, Okereke OI. Dietary flavonoid intake and risk of incident depression in midlife and older women. Am J Clin Nutr. (2016) 104:704–14. doi: 10.3945/ajcn.115.124545
144. Gonzalez R, Ballester I, Lopez-Posadas R, Suarez MD, Zarzuelo A, Martinez-Augustin O, et al. Effects of flavonoids and other polyphenols on inflammation. Crit Rev Food Sci Nutr. (2011) 51:331–62. doi: 10.1080/10408390903584094
145. Noonan S, Zaveri M, Macaninch E, Martyn K. Food & mood: a review of supplementary prebiotic and probiotic interventions in the treatment of anxiety and depression in adults. BMJ Nutr Prev Health. (2020) 3:351–62. doi: 10.1136/bmjnph-2019-000053
146. Desbonnet L, Garrett L, Clarke G, Kiely B, Cryan JF, Dinan TG. Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression. Neuroscience. (2010) 170:1179–88. doi: 10.1016/j.neuroscience.2010.08.005
147. Liang S, Wang T, Hu X, Luo J, Li W, Wu X, et al. Administration of Lactobacillus helveticus NS8 improves behavioral, cognitive, and biochemical aberrations caused by chronic restraint stress. Neuroscience. (2015) 310:561–77. doi: 10.1016/j.neuroscience.2015.09.033
148. Messaoudi M, Lalonde R, Violle N, Javelot H, Desor D, Nejdi A, et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr. (2011) 105:755–64. doi: 10.1017/S0007114510004319
149. Rao AV, Bested AC, Beaulne TM, Katzman MA, Iorio C, Berardi JM, et al. A randomized, double-blind, placebo-controlled pilot study of a probiotic in emotional symptoms of chronic fatigue syndrome. Gut Pathog. (2009) 1:6. doi: 10.1186/1757-4749-1-6
150. Benton D, Williams C, Brown A. Impact of consuming a milk drink containing a probiotic on mood and cognition. Eur J Clin Nutr. (2007) 61:355–61. doi: 10.1038/sj.ejcn.1602546
151. Lyte M. Probiotics function mechanistically as delivery vehicles for neuroactive compounds: Microbial endocrinology in the design and use of probiotics. Bioessays. (2011) 33:574–81. doi: 10.1002/bies.201100024
152. Steenbergen L, Sellaro R, van Hemert S, Bosch JA, Colzato LS. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav Immun. (2015) 48:258–64. doi: 10.1016/j.bbi.2015.04.003
153. Liu Y, Steinhausen K, Bharwani A, Mian MF, McVey Neufeld K-A, Forsythe P. Increased persistence of avoidance behaviour and social deficits with L. rhamnosus JB-1 or selective serotonin reuptake inhibitor treatment following social defeat. Sci Rep. (2020) 10:13485. doi: 10.1038/s41598-020-69968-y
154. Kelly JR, Allen AP, Temko A, Hutch W, Kennedy PJ, Farid N, et al. Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects. Brain Behav Immun. (2017) 61:50–9. doi: 10.1016/j.bbi.2016.11.018
155. Barrera-Bugueno C, Realini O, Escobar-Luna J, Sotomayor-Zarate R, Gotteland M, Julio-Pieper M, et al. Anxiogenic effects of a Lactobacillus, inulin and the synbiotic on healthy juvenile rats. Neuroscience. (2017) 359:18–29. doi: 10.1016/j.neuroscience.2017.06.064
156. Bagga D, Reichert JL, Koschutnig K, Aigner CS, Holzer P, Koskinen K, et al. Probiotics drive gut microbiome triggering emotional brain signatures. Gut Microbes. (2018) 9:486–96. doi: 10.1080/19490976.2018.1460015
157. Sun J, Buys N. Effects of probiotics consumption on lowering lipids and CVD risk factors: a systematic review and meta-analysis of randomized controlled trials. Ann Med. (2015) 47:430–40. doi: 10.3109/07853890.2015.1071872
158. Chassaing B, Garenaux E, Carriere J, Rolhion N, Guerardel Y, Barnich N, et al. Analysis of the sigmaE regulon in Crohn's disease-associated Escherichia coli revealed involvement of the waaWVL operon in biofilm formation. J Bacteriol. (2015) 197:1451–65. doi: 10.1128/JB.02499-14
159. Macfarlane S, Dillon JF. Microbial biofilms in the human gastrointestinal tract. J Appl Microbiol. (2007) 102:1187–96. doi: 10.1111/j.1365-2672.2007.03287.x
160. Hager CL, Isham N, Schrom KP, Chandra J, McCormick T, Miyagi M, et al. Effects of a novel probiotic combination on pathogenic bacterial-fungal polymicrobial biofilms. mBio. (2019) 10:19. doi: 10.1128/mBio.00338-19
161. Moya-Perez A, Perez-Villalba A, Benitez-Paez A, Campillo I, Sanz Y. Bifidobacterium CECT 7765 modulates early stress-induced immune, neuroendocrine and behavioral alterations in mice. Brain Behav Immun. (2017) 65:43–56. doi: 10.1016/j.bbi.2017.05.011
162. van Bakel BMA, Bakker EA, de Vries F, Thijssen DHJ, Eijsvogels TMH. Impact of COVID-19 lockdown on physical activity and sedentary behaviour in Dutch cardiovascular disease patients. Neth Heart J. (2021) 29:273–9. doi: 10.1007/s12471-021-01550-1
163. Gualano MR, Lo Moro G, Voglino G, Bert F, Siliquini R. Effects of Covid-19 lockdown on mental health and sleep disturbances in Italy. Int J Environ Res Public Health. (2020) 17. doi: 10.3390/ijerph17134779
164. Zheng C, Huang WY, Sheridan S, Sit CH, Chen XK, Wong SH. COVID-19 pandemic brings a sedentary lifestyle in young adults: a cross-sectional and longitudinal study. Int J Environ Res Public Health. (2020) 17:108255. doi: 10.1101/2020.05.22.20110825
165. Monda V, Villano I, Messina A, Valenzano A, Esposito T, Moscatelli F, et al. Exercise modifies the gut microbiota with positive health effects. Oxid Med Cell Longev. (2017) 2017:3831972. doi: 10.1155/2017/3831972
166. Belizario JE, Faintuch J. Microbiome and gut dysbiosis. Exp Suppl. (2018) 109:459–76. doi: 10.1007/978-3-319-74932-7_13
167. Ho SS, Dhaliwal SS, Hills AP, Pal S. Effects of chronic exercise training on inflammatory markers in Australian overweight and obese individuals in a randomized controlled trial. Inflammation. (2013) 36:625–32. doi: 10.1007/s10753-012-9584-9
168. Craft LL, Perna FM. The benefits of exercise for the clinically depressed. Prim Care Companion J Clin Psychiatry. (2004) 6:104–11. doi: 10.4088/PCC.v06n0301
169. Dimeo F, Bauer M, Varahram I, Proest G, Halter U. Benefits from aerobic exercise in patients with major depression: a pilot study. Br J Sports Med. (2001) 35:114–7. doi: 10.1136/bjsm.35.2.114
170. Doyne EJ, Chambless DL, Beutler LE. Aerobic exercise as a treatment for depression in women. Behav Therapy. (1983) 14:434–40. doi: 10.1016/S0005-7894(83)80106-3
171. McNeil JK, LeBlanc EM, Joyner M. The effect of exercise on depressive symptoms in the moderately depressed elderly. Psychol Aging. (1991) 6:487–8. doi: 10.1037/0882-7974.6.3.487
172. Luo B, Xiang D, Nieman DC, Chen P. The effects of moderate exercise on chronic stress-induced intestinal barrier dysfunction and antimicrobial defense. Brain Behav Immun. (2014) 39:99–106. doi: 10.1016/j.bbi.2013.11.013
173. Garza AA, Ha TG, Garcia C, Chen MJ, Russo-Neustadt AA. Exercise, antidepressant treatment, and BDNF mRNA expression in the aging brain. Pharmacol Biochem Behav. (2004) 77:209–20. doi: 10.1016/j.pbb.2003.10.020
174. Russo-Neustadt AA, Beard RC, Huang YM, Cotman CW. Physical activity and antidepressant treatment potentiate the expression of specific brain-derived neurotrophic factor transcripts in the rat hippocampus. Neuroscience. (2000) 101:305–12. doi: 10.1016/S0306-4522(00)00349-3
175. Lopresti AL, Hood SD. A review of lifestyle factors that contribute to important pathways associated with major depression: diet, sleep and exercise. J Affect Disord. (2013) 15:12–27. doi: 10.1016/j.jad.2013.01.014
176. Lin TW, Kuo YM. Exercise benefits brain function: the monoamine connection. Brain Sci. (2013) 3:39–53. doi: 10.3390/brainsci3010039
177. Mathur N, Pedersen BK. Exercise as a mean to control low-grade systemic inflammation. Mediators Inflamm. (2008) 2008:109502. doi: 10.1155/2008/109502
178. Erickson KI, Leckie RL, Weinstein AM. Physical activity, fitness, and gray matter volume. Neurobiol Aging. (2014) 35 Suppl 2:S20–8. doi: 10.1016/j.neurobiolaging.2014.03.034
179. Voss MW, Vivar C, Kramer AF, van Praag H. Bridging animal and human models of exercise-induced brain plasticity. Trends Cogn Sci. (2013) 17:525–44. doi: 10.1016/j.tics.2013.08.001
180. Physical activity guidelines for Americans. (2020). Available online at: https://www.hhs.gov/fitness/be-active/physical-activity-guidelines-for-americans/index.html#:~:text=For%20substantial%20health%20benefits%2C%20adults,or%20an%20equivalent%20combination%20of
181. American Heart Association recommendations for physical activity in adults and kids. (2018). Available online at: https://www.heart.org/en/healthy-living/fitness/fitness-basics/aha-recs-for-physical-activity-in-adults
182. Voigt RM, Forsyth CB, Green SJ, Mutlu E, Engen P, Vitaterna MH, et al. Circadian disorganization alters intestinal microbiota. PLoS ONE. (2014) 9:e97500. doi: 10.1371/journal.pone.0097500
183. Voigt RM, Summa KC, Forsyth CB, Green SJ, Engen P, Naqib A, et al. The circadian clock mutation promotes intestinal dysbiosis. Alcohol Clin Exp Res. (2016) 40:335–47. doi: 10.1111/acer.12943
184. Ahmed MZ, Ahmed O, Aibao Z, Hanbin S, Siyu L, Ahmad A. Epidemic of COVID-19 in China and associated psychological problems. Asian J Psychiatr. (2020) 51:102092. doi: 10.1016/j.ajp.2020.102092
185. Lei L, Huang X, Zhang S, Yang J, Yang L, Xu M. Comparison of prevalence and associated factors of anxiety and depression among people affected by versus people unaffected by quarantine during the COVID-19 epidemic in Southwestern China. Med Sci Monit. (2020) 26:e924609. doi: 10.12659/MSM.924609
186. Mazza C, Ricci E, Biondi S, Colasanti M, Ferracuti S, Napoli C, et al. A Nationwide survey of psychological distress among Italian people during the COVID-19 pandemic: immediate psychological responses and associated factors. Int J Environ Res Public Health. (2020) 17:3165. doi: 10.3390/ijerph17093165
187. Hamilton M. Frequency of symptoms in melancholia depressive illness. Br J Psychiatry. (1989) 1989:201–6. doi: 10.1192/bjp.154.2.201
188. Yates WR, Mitchell J, John Rush A, Trivedi M, Wisniewski SR, Warden D, et al. Clinical features of depression in outpatients with and without co-occurring general medical conditions in STAR*D: confirmatory analysis. Prim Care Companion J Clin Psychiatry. (2007) 9:7–15. doi: 10.4088/PCC.v09n0102
189. Breslau N, Roth T, Rosenthal L, Andreski P. Sleep disturbance and psychiatric disorders: a longitudinal epidemiological study of young adults. Biol Psychiatry. (1996) 39:411–8. doi: 10.1016/0006-3223(95)00188-3
190. Chang PP, Ford DE, Mead LA, Cooper-Patrick L, Klag MJ. Insomnia in young men and subsequent depression. The Johns Hopkins Precursors Study. Am J Epidemiol. (1997) 146:105–14. doi: 10.1093/oxfordjournals.aje.a009241
191. Ford DE, Kamerow DB. Epidemiologic study of sleep disturbances and psychiatric disorders. An opportunity for prevention? JAMA. (1989) 262:1479–84. doi: 10.1001/jama.262.11.1479
192. Morphy H, Dunn KM, Lewis M, Boardman HF, Croft PR. Epidemiology of insomnia: a longitudinal study in a UK population. Sleep. (2007) 30:274–80. doi: 10.1093/sleep/30.3.274
193. National Sleep Foundation recommends new sleep times. (2015). Available online at: https://www.sleepfoundation.org/press-release/national-sleep-foundation-recommends-new-sleep-times (accessed March 12, 2021).
194. Smith RP, Easson C, Lyle SM, Kapoor R, Donnelly CP, Davidson EJ, et al. Gut microbiome diversity is associated with sleep physiology in humans. PLoS ONE. (2019) 14:e0222394. doi: 10.1371/journal.pone.0222394
195. Parkar SG, Kalsbeek A, Cheeseman JF. Potential role for the gut microbiota in modulating host circadian rhythms and metabolic health. Microorganisms. (2019) 7:41. doi: 10.3390/microorganisms7020041
196. Singh RK, Chang HW, Yan D, Lee KM, Ucmak D, Wong K, et al. Influence of diet on the gut microbiome and implications for human health. J Transl Med. (2017) 15:73. doi: 10.1186/s12967-017-1175-y
197. Gottesmann C. GABA mechanisms and sleep. Neuroscience. (2002) 111:231–9. doi: 10.1016/S0306-4522(02)00034-9
198. Li Y, Hao Y, Fan F, Zhang B. The role of microbiome in insomnia, circadian disturbance and depression. Front Psychiatry. (2018) 9:669. doi: 10.3389/fpsyt.2018.00669
199. Tseng CH, Wu CY. The gut microbiome in obesity. J Formos Med Assoc. (2019) 118 Suppl 1:S3–S9. doi: 10.1016/j.jfma.2018.07.009
200. Benedict C, Vogel H, Jonas W, Woting A, Blaut M, Schurmann A, et al. Gut microbiota and glucometabolic alterations in response to recurrent partial sleep deprivation in normal-weight young individuals. Mol Metab. (2016) 5:1175–86. doi: 10.1016/j.molmet.2016.10.003
201. Leone V, Gibbons SM, Martinez K, Hutchison AL, Huang EY, Cham CM, et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism. Cell Host Microbe. (2015) 17:681–9. doi: 10.1016/j.chom.2015.03.006
202. Poroyko VA, Carreras A, Khalyfa A, Khalyfa AA, Leone V, Peris E, et al. Chronic sleep disruption alters gut microbiota, induces systemic and adipose tissue inflammation and insulin resistance in mice. Sci Rep. (2016) 6:35405. doi: 10.1038/srep35405
203. Thaiss CA, Zeevi D, Levy M, Zilberman-Schapira G, Suez J, Tengeler AC, et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. (2014) 159:514–29. doi: 10.1016/j.cell.2014.09.048
204. Yarandi SS, Peterson DA, Treisman GJ, Moran TH, Pasricha PJ. Modulatory effects of gut microbiota on the central nervous system: how gut could play a role in neuropsychiatric health and diseases. J Neurogastroenterol Motil. (2016) 22:201–12. doi: 10.5056/jnm15146
206. Dibner C, Schibler U, Albrecht U. The mammalian circadian timing system: organization and coordination of central and peripheral clocks. Annu Rev Physiol. (2010) 72:517–49. doi: 10.1146/annurev-physiol-021909-135821
207. Hsiao EY, McBride SW, Hsien S, Sharon G, Hyde ER, McCue T, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. (2013) 155:1451–63. doi: 10.1016/j.cell.2013.11.024
208. Yi P, Li L. The germfree murine animal: an important animal model for research on the relationship between gut microbiota and the host. Vet Microbiol. (2012) 157:1–7. doi: 10.1016/j.vetmic.2011.10.024
209. Gareau MG, Silva MA, Perdue MH. Pathophysiological mechanisms of stress-induced intestinal damage. Curr Mol Med. (2008) 8:274–81. doi: 10.2174/156652408784533760
210. Soderholm JD, Yang PC, Ceponis P, Vohra A, Riddell R, Sherman PM, et al. Chronic stress induces mast cell-dependent bacterial adherence and initiates mucosal inflammation in rat intestine. Gastroenterology. (2002) 123:1099–108. doi: 10.1053/gast.2002.36019
211. Mosheva M, Hertz-Palmor N, Dorman Ilan S, Matalon N, Pessach IM, Afek A, et al. Anxiety, pandemic-related stress and resilience among physicians during the COVID-19 pandemic. Depress Anxiety. (2020) 37:965–71. doi: 10.1002/da.23085
212. Kujawa A, Green H, Compas BE, Dickey L, Pegg S. Exposure to COVID-19 pandemic stress: Associations with depression and anxiety in emerging adults in the United States. Depress Anxiety. (2020) 37:1280–8. doi: 10.1002/da.23109
213. Dorman-Ilan S, Hertz-Palmor N, Brand-Gothelf A, Hasson-Ohayon I, Matalon N, Gross R, et al. Anxiety and depression symptoms in COVID-19 isolated patients and in their relatives. Front Psychiatry. (2020) 11:581598. doi: 10.3389/fpsyt.2020.581598
214. Bailey MT. Influence of stressor-induced nervous system activation on the intestinal microbiota and the importance for immunomodulation. Adv Exp Med Biol. (2014) 817:255–76. doi: 10.1007/978-1-4939-0897-4_12
215. Bailey MT, Dowd SE, Galley JD, Hufnagle AR, Allen RG, Lyte M. Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor-induced immunomodulation. Brain Behav Immun. (2011) 25:397–407. doi: 10.1016/j.bbi.2010.10.023
216. Bharwani A, Mian MF, Foster JA, Surette MG, Bienenstock J, Forsythe P. Structural & functional consequences of chronic psychosocial stress on the microbiome & host. Psychoneuroendocrinology. (2016) 63:217–27. doi: 10.1016/j.psyneuen.2015.10.001
217. De Palma G, Collins SM, Bercik P, Verdu EF. The microbiota-gut-brain axis in gastrointestinal disorders: stressed bugs, stressed brain or both? J Physiol. (2014) 592:2989–97. doi: 10.1113/jphysiol.2014.273995
218. Moloney RD, Desbonnet L, Clarke G, Dinan TG, Cryan JF. The microbiome: stress, health and disease. Mamm Genome. (2014) 25:49–74. doi: 10.1007/s00335-013-9488-5
219. Galley JD, Yu Z, Kumar P, Dowd SE, Lyte M, Bailey MT. The structures of the colonic mucosa-associated and luminal microbial communities are distinct and differentially affected by a prolonged murine stressor. Gut Microbes. (2014) 5:748–60. doi: 10.4161/19490976.2014.972241
220. Stephens MA, Wand G. Stress and the HPA axis: role of glucocorticoids in alcohol dependence. Alcohol Res. (2012) 34:468–83.
221. Holsboer F. The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology. (2000) 23:477–501. doi: 10.1016/S0893-133X(00)00159-7
222. Pariante CM, Lightman SL. The HPA axis in major depression: classical theories and new developments. Trends Neurosci. (2008) 31:464–8. doi: 10.1016/j.tins.2008.06.006
223. Hammen C. Stress and depression. Annu Rev Clin Psychol. (2005) 1:293–319. doi: 10.1146/annurev.clinpsy.1.102803.143938
224. Dinan TG, Cryan J, Shanahan F, Keeling PW, Quigley EM. IBS: An epigenetic perspective. Nat Rev Gastroenterol Hepatol. (2010) 7:465–71. doi: 10.1038/nrgastro.2010.99
225. Elsenbruch S. Abdominal pain in Irritable Bowel Syndrome: a review of putative psychological, neural and neuro-immune mechanisms. Brain Behav Immun. (2011) 25:386–94. doi: 10.1016/j.bbi.2010.11.010
226. Mayer EA. The neurobiology of stress and gastrointestinal disease. Gut. (2000) 47:861–9. doi: 10.1136/gut.47.6.861
227. Davis C, Bryan J, Hodgson J, Murphy K. Definition of the mediterranean diet; a literature review. Nutrients. (2015) 7:9139–53. doi: 10.3390/nu7115459
228. Ghannoum M, Smith C, Adamson E, Isham N, Salem I, M R. Effect of mycobiome diet on gut fungal and bacterial communities of healthy adults. J Prob Health. (2019) 7:215. doi: 10.35248/2329-8901.20.8.215
229. Vester-Andersen MK, Mirsepasi-Lauridsen HC, Prosberg MV, Mortensen CO, Trager C, Skovsen K, et al. Increased abundance of proteobacteria in aggressive Crohn's disease seven years after diagnosis. Sci Rep. (2019) 9:13473. doi: 10.1038/s41598-019-49833-3
230. Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, et al. Diversity of the human intestinal microbial flora. Science. (2005) 308:1635–8. doi: 10.1126/science.1110591
231. Markowiak-Kopec P, Slizewska K. The effect of probiotics on the production of short-chain fatty acids by human intestinal microbiome. Nutrients. (2020) 12:1107. doi: 10.3390/nu12041107
232. Oleskin AV, El'-Registan GI, Shenderov BA. Role of neuromediators in the functioning of the human microbiota: “Business talks” among microorganisms and the microbiota-host dialogue. Microbiology. (2016) 85:1–22. doi: 10.1134/S0026261716010082
233. Kraus C, Castren E, Kasper S, Lanzenberger R. Serotonin and neuroplasticity - Links between molecular, functional and structural pathophysiology in depression. Neurosci Biobehav Rev. (2017) 77:317–26. doi: 10.1016/j.neubiorev.2017.03.007
234. Nemeroff CB. Recent advances in the neurobiology of depression. Psychopharmacol Bull. (2002) 36 (Suppl. 2):6–23.
235. Chiriţă AL, Gheorman V, Bondari D, Rogoveanu I. Current understanding of the neurobiology of major depressive disorder. Roman. J. Morphol. Embryol. (2015) 56(2 Suppl.):651–8
236. Nutt DJ. Relationship of neurotransmitters to the symptoms of major depressive disorder. J Clin Psychiatry. (2008) 69 Suppl E1:4–7.
237. Cryan JF, Slattery DA. GABAB receptors and depression. Current status. Adv Pharmacol. (2010) 58:427–51. doi: 10.1016/S1054-3589(10)58016-5
238. Mittal R, Debs LH, Patel AP, Nguyen D, Patel K, O'Connor G, et al. Neurotransmitters: the critical modulators regulating gut-brain axis. J Cell Physiol. (2017) 232:2359–72. doi: 10.1002/jcp.25518
239. Tian P, Wang G, Zhao J, Zhang H, Chen W. Bifidobacterium with the role of 5-hydroxytryptophan synthesis regulation alleviates the symptom of depression and related microbiota dysbiosis. J Nutr Biochem. (2019) 66:43–51. doi: 10.1016/j.jnutbio.2019.01.007
240. Shaw W. Clostridia bacteria in the GI tract affecting dopamine and norepinephrine metabolism. In: Greenblatt JM, Brogan K, editors. Integrative Psychiatry for Depression: Redefining Models for Assessment, Treatment, and Prevention of Mood Disorders. CRC Press (2016).
241. Persico AM, Napolioni V. Urinary p-cresol in autism spectrum disorder. Neurotoxicol Teratol. (2013) 36:82–90. doi: 10.1016/j.ntt.2012.09.002
242. Goodhart PJ, DeWolf WE, Kruse LI. Mechanism-based inactivation of dopamine beta-hydroxylase by p-cresol and related alkylphenols. Biochemistry. (1987) 26:2576–83. doi: 10.1021/bi00383a025
243. Munoz P, Huenchuguala S, Paris I, Segura-Aguilar J. Dopamine oxidation and autophagy. Parkinsons Dis. (2012) 2012:920953. doi: 10.1155/2012/920953
244. Baez S, Segura-Aguilar J, Widersten M, Johansson AS, Mannervik B. Glutathione transferases catalyse the detoxication of oxidized metabolites (o-quinones) of catecholamines and may serve as an antioxidant system preventing degenerative cellular processes. Biochem J. (1997) 324:25–8. doi: 10.1042/bj3240025
245. Guarner F, Khan AG, Garisch J, Eliakim R, Gangl A, Thomson A, et al. World Gastroenterology Organisation Global Guidelines: probiotics and prebiotics October 2011. J Clin Gastroenterol. (2012) 46:468–81. doi: 10.1097/MCG.0b013e3182549092
246. Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. (2014) 11:506–14. doi: 10.1038/nrgastro.2014.66
247. Kechagia M, Basoulis D, Konstantopoulou S, Dimitriadi D, Gyftopoulou K, Skarmoutsou N, et al. Health benefits of probiotics: a review. ISRN Nutr. (2013) 2013:481651. doi: 10.5402/2013/481651
248. Yan F, Polk DB. Probiotics and immune health. Curr Opin Gastroenterol. (2011) 27:496–501. doi: 10.1097/MOG.0b013e32834baa4d
249. Luoto R, Ruuskanen O, Waris M, Kalliomaki M, Salminen S, Isolauri E. Prebiotic and probiotic supplementation prevents rhinovirus infections in preterm infants: a randomized, placebo-controlled trial. J Allergy Clin Immunol. (2014) 133:405–13. doi: 10.1016/j.jaci.2013.08.020
250. Al Kassaa I, Hober D, Hamze M, Chihib NE, Drider D. Antiviral potential of lactic acid bacteria and their bacteriocins. Probiotics Antimicrob Proteins. (2014) 6:177–85. doi: 10.1007/s12602-014-9162-6
251. Lakshmi B, Viswanath B, Sai Gopal DV. Probiotics as antiviral agents in shrimp aquaculture. J Pathog. (2013) 2013:424123. doi: 10.1155/2013/424123
252. Seo BJ, Rather IA, Kumar VJ, Choi UH, Moon MR, Lim JH, et al. Evaluation of Leuconostoc mesenteroides YML003 as a probiotic against low-pathogenic avian influenza (H9N2) virus in chickens. J Appl Microbiol. (2012) 113:163–71. doi: 10.1111/j.1365-2672.2012.05326.x
253. Villena J, Oliveira ML, Ferreira PC, Salva S, Alvarez S. Lactic acid bacteria in the prevention of pneumococcal respiratory infection: future opportunities and challenges. Int Immunopharmacol. (2011) 11:1633–45. doi: 10.1016/j.intimp.2011.06.004
254. Myles M, Kral TVE. Social Environmental and Genetic Influences on Obesity and Obesity- Promoting Behaviors: Fostering Research Integration. Genes, Behavior, and the Social Environment: Moving Beyond the Nature/Nurture Debate Washington, DC: National Academies Press (2006).
Keywords: gut microbiome, depression, holistic approach, diet, probiotics, exercise, sleep, stress management
Citation: Ghannoum MA, Ford M, Bonomo RA, Gamal A and McCormick TS (2021) A Microbiome-Driven Approach to Combating Depression During the COVID-19 Pandemic. Front. Nutr. 8:672390. doi: 10.3389/fnut.2021.672390
Received: 25 February 2021; Accepted: 30 July 2021;
Published: 24 August 2021.
Edited by:
Gustavo Pacheco-Lopez, Autonomous Metropolitan University, Lerma, MexicoReviewed by:
Kiran Veer Sandhu, University College Cork, IrelandKhemlal Nirmalkar, Arizona State University, United States
Copyright © 2021 Ghannoum, Ford, Bonomo, Gamal and McCormick. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mahmoud A. Ghannoum, bWFnM0BjYXNlLmVkdQ==
 Mahmoud A. Ghannoum
Mahmoud A. Ghannoum MaryKate Ford
MaryKate Ford Robert A. Bonomo
Robert A. Bonomo Ahmed Gamal
Ahmed Gamal Thomas S. McCormick
Thomas S. McCormick