
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol. , 02 June 2022
Sec. Autoimmune and Autoinflammatory Disorders
Volume 13 - 2022 | https://doi.org/10.3389/fimmu.2022.909760
This article is part of the Research Topic Clinical Management, Pathogenesis and Biomarkers of Cardiovascular Disease Associated with Systemic Autoimmune Disorders View all 10 articles
 Daniella M. Schwartz1,2*
Daniella M. Schwartz1,2* Philip Parel3
Philip Parel3 Haiou Li3
Haiou Li3 Alexander V. Sorokin3
Alexander V. Sorokin3 Alexander R. Berg3
Alexander R. Berg3 Marcus Chen3
Marcus Chen3 Amit Dey4
Amit Dey4 Christin G. Hong3
Christin G. Hong3 Martin Playford3
Martin Playford3 McKella Sylvester1
McKella Sylvester1 Heather Teague3
Heather Teague3 Evan Siegel5
Evan Siegel5 Nehal N. Mehta3
Nehal N. Mehta3Background and Objectives: Psoriasis is a heterogeneous inflammatory disease that involves the skin, joints, liver, heart, and other organs. Psoriatic arthritis (PsA) is associated with cardiovascular disease (CVD), but the relative contributions of inflammatory and metabolic dysregulation to CVD are incompletely understood. We set out to discover novel potential contributors to CVD in PsA patients by comprehensively phenotyping a cohort of PsA patients using these advanced technologies.
Methods: In this cross-sectional analysis of a cohort study, we investigated associations of systemic inflammation and metabolic dysregulation with Coronary CT angiography (CCTA)-proven coronary artery disease (CAD) in 39 subjects with PsA. We measured traditional CVD risk factors [blood pressure, Body Mass Index (BMI), diabetes, age, sex, smoking], serum markers of systemic inflammation (hsCRP, GlycA) and metabolic dysfunction (cholesterol efflux capacity), and inflammatory cytokines (IL-1β, IL-6, IL-12/IL-23, IL-17A, TNF-α, IFN-γ). We also incorporated radiographic measures of metabolic dysfunction (visceral and subcutaneous adipose volume) and tissue-specific inflammation (positron emission tomography-computed tomography, PET-CT). To quantify relative contributions of FDG (fluorodeoxyglucose) uptake and adiposity to coronary plaque, we performed multiple linear regression, controlling for Framingham risk score (FRS) and FRS + visceral adiposity.
Results: Compared with non-psoriatic volunteers, subjects with PsA had elevated markers of metabolic and inflammatory disease, which was more pronounced in subjects with moderate-to-severe skin disease. This included visceral (p = 0.005) and subcutaneous (p = 0.004) adiposity, BMI (p = 0.001), hemoglobin A1C (p = 0.037), high sensitivity C-reactive protein (p = 0.005), IL-6 (p = 0.003), IFN-γ (p = 0.006), and liver FDG uptake (p = 0.03). In subjects with PsA, visceral adiposity correlated significantly with subclinical CAD (standardized β = 0.681, p = 0.002), as did FDG uptake in bone marrow (standardized β = 0.488, p = 0.008), liver (standardized β = 0.619, p < 0.001), spleen (standardized β = 0.523, p = 0.004), and subcutaneous adipose (standardized β = 0.524, p = 0.003).
Interpretation: Together, these findings reveal inflammatory and metabolic potential contributors to subclinical CAD in PsA, including adipose inflammation, and suggesting novel targets for CVD prevention and treatment in PsA.
Psoriasis is a complex disease with substantial clinical heterogeneity. In addition to skin inflammation, psoriasis can manifest with axial or peripheral arthritis (PsA), liver disease, lung disease, gastrointestinal inflammation, aortic root involvement, or nail disease (1). Cardiovascular disease (CVD) is the leading cause of death in patients with psoriasis and PsA and is often regarded as a component of the clinical spectrum encompassed by psoriatic disease (1, 2). However, the factors driving psoriatic atherogenesis are incompletely understood. Psoriatic diseases are associated with high rates of traditional CV risk factors (2). Systemic inflammation, which is often more severe in PsA, also promotes atherogenesis (2, 3). Additionally, psoriatic diseases –particularly PsA – are associated with metabolic dysregulation including visceral adipose tissue expansion and HDL dysfunction, leading to impaired cholesterol metabolism (4, 5). Metabolic dysregulation interacts with inflammation to promote atherosclerosis through as-yet unknown mechanisms (5, 6). Defining these mechanisms and interactions can address a major unmet need by identifying factors that can be targeted to prevent the high rates of CVD in patients with psoriatic diseases.
Traditionally, systemic inflammation is evaluated by clinical laboratory measurement of acute phase reactants, mainly high sensitivity C-reactive protein (hs-CRP) (7). More recently, 18-fluorodeoxyglucose positron emission tomography computed tomography (FDG-PET-CT) has been used to sensitively and noninvasively quantify systemic and tissue inflammation (3). Together with serum cytokine and glycoprotein acetylation (GlycA) levels, FDG-PET-CT can provide a more complete picture of inflammatory disease pathogenesis than hs-CRP alone (3, 6). Similarly, metabolic dysregulation is usually estimated using traditional CV risk factors but can be more sensitively quantified by incorporating radiographic measures of adiposity and advanced lipid phenotyping assays like cholesterol efflux capacity (8). Combined with coronary CT angiography (CCTA), a noninvasive angiographic technique that provides a quantifiable measure of coronary artery disease (CAD), these techniques allow for a multimodal real-time evaluation of potential contributors to psoriatic atherogenesis (9).
We hypothesized that combining CCTA with other advanced radiographic and serum-based assays might identify novel contributions to subclinical coronary disease in PsA (2). We performed a cross-sectional analysis of a prospective cohort study, utilizing data from a one-time initial visit. We measured metabolic dysregulation, inflammatory burden, and subclinical CAD in subjects with PsA and age-sex matched non-psoriatic volunteers, utilizing traditional clinical disease measures, serum-based assays, CCTA, and FDG-PET-CT.
358 subjects with psoriasis were recruited between Jan 2013-May 2021 as part of an IRB-approved prospective cohort study at the NIH Clinical Center (Figure 1, 13H-0065). Full inclusion and exclusion criteria were previously reported (10). 204 subjects who had received biologic disease modifying antirheumatic drugs (bDMARDs) were excluded. Age and sex matched volunteers were recruited through a separate IRB approved protocol at the NIH Clinical Center (NCT01934660). All subjects provided written informed consent.
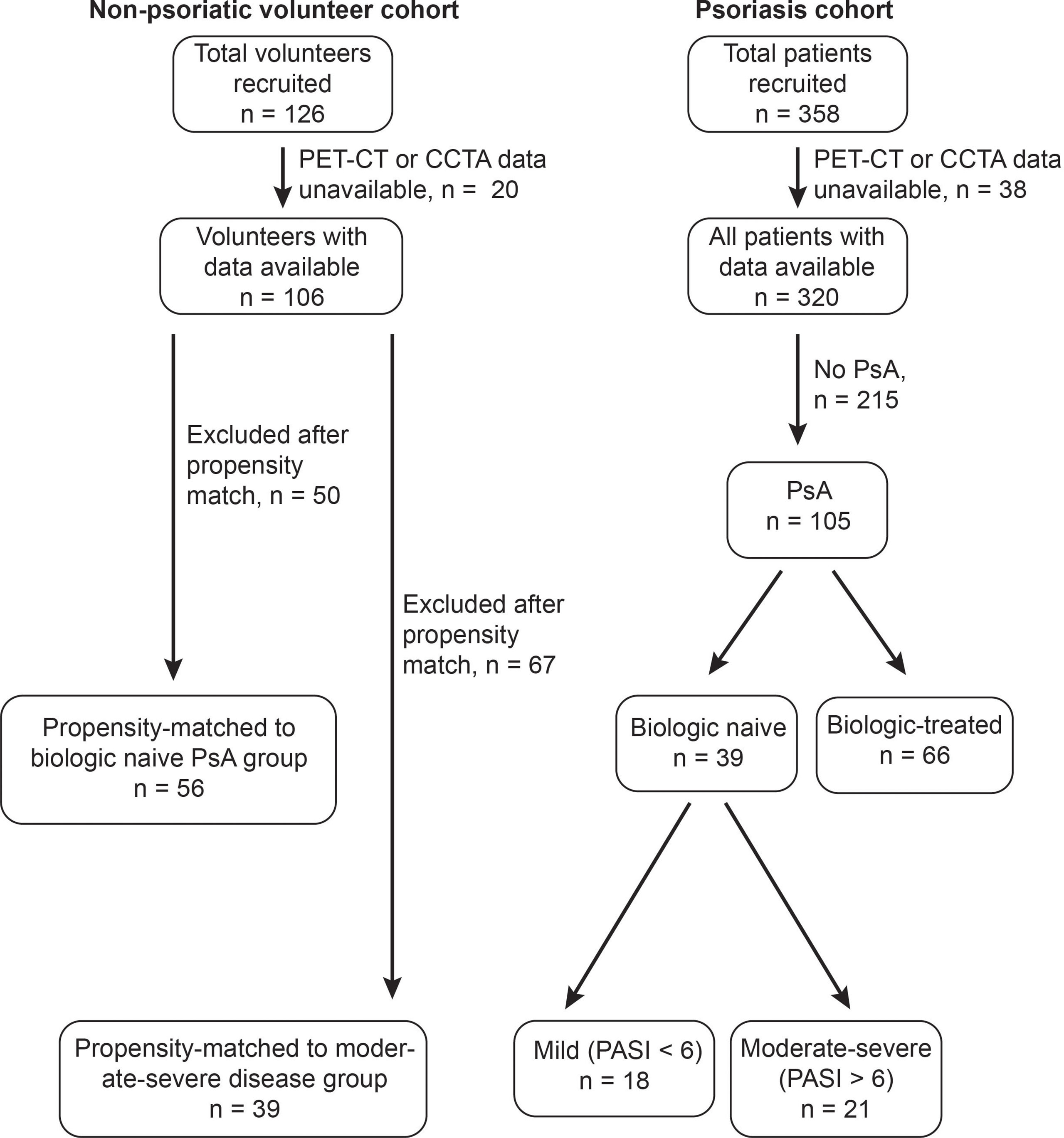
Figure 1 Identification of subjects with treatment-naive psoriatic arthritis (PsA) within a cohort of patients with psoriatic diseases, and of age-sex-matched non-psoriatic volunteers. 358 total patients with psoriatic diseases were recruited, of whom 320 were confirmed to have psoriatic disease and had PET-CT and CCTA data available. Of these 105 had psoriatic arthritis, 39 of whom were biologic-naive. 21 biologic naive patients with PsA had moderate-severe skin disease. 126 total non-psoriatic volunteers were recruited, of whom 106 had PET-CT and CCTA data available. 56 volunteers were age-sex-matched to the full treatment-naïve PsA cohort, whereas 39 volunteers were age-sex-matched to the 21 subjects with moderate-severe skin disease.
Full details of clinical assessment were previously described (10). Briefly, patients underwent evaluation by trained providers at the NIH Clinical Center to verify psoriasis onset, duration, and severity (Psoriasis Area and Severity Score, PASI). Psoriatic arthritis (PsA) was diagnosed by a rheumatologist based on diagnosis of psoriasis and seronegative inflammatory arthritis; all patients satisfied CASPAR criteria. Joint disease activity was assessed systematically by a rheumatologist in a subset of patients (n = 16). Clinical assessments included full medical histories, physical examinations, PASI quantification, medication evaluations, anthropometric measurements, and clinical laboratory assays.
All patients underwent CCTA using a 320-detector row Aquilion ONE ViSION (Toshiba); acquisition details were previously described (11). All scans were analyzed in a blinded fashion using QAngio CT (Medis). FDG-PET-CT imaging was performed using a Biograph mCT PET-CT 64-slice scanner (Siemens Medical Solutions). After an overnight fast (≥8 hours), images were acquired approximately 60 minutes (62 ± 1 minutes) after administration of FDG (10 mCi). All patients underwent identical PET-CT protocols with the same team of technologists. Standard bed (3 min each, scanning cranially to caudally) were obtained for each patient using 1.5-mm axial slices. Patients with fasting glucose >200 mg/dl were excluded. Analysis was done in a blinded fashion using dedicated PET-CT image analysis software (OsirixTM, Pixmeo SARL) as previously described (11). Visceral and subcutaneous adipose tissue volumes were analyzed using low-dose CT: 100 transverse slices from the caudal sternum to cranial pubic symphysis were interpreted using automated software with contour model algorithm. Visceral and subcutaneous adipose tissue volumes were demarcated as previously described (11).
Clinical laboratory assays including GlycA and serum lipid levels were measured at the NIH Clinical Center. Serum cytokines were measured using multiplex ELISA (Mesoscale). Cholesterol efflux capacity was measured using a cell-based assay in J774 macrophages as previously described (9).
PsA and non-psoriatic volunteers (controls) were age- and sex-matched as follows: non-psoriatic volunteers were matched in a 1:2 ratio according to propensity scores (age, sex) with a tolerance level of 0.2 to maximize sample size. Because the volunteer cohort is younger and contains more female subjects than the PsA cohort, we used a logistic regression model to perform matching. For matched data (Table 1 and Supplementary Table 1), baseline characteristics are presented mean ± SD for parametric variables, median (interquartile range [IQR]) for nonparametric variables, and percentages (%) for categorical variables. Data were assessed for normality via Shapiro-Wilks test. Statistical significance was assessed by Student t-test for parametric variables, Wilcoxon rank-sum test for nonparametric variables, and Pearson’s χ2 test or Fisher-exact test for categorical variables. The moderate-severe psoriasis subgroup analysis was performed by identifying patients with PASI score >6, which was the median value for this cohort (4), and repeating propensity score matching. To quantify relative contributions of FDG uptake and adiposity to coronary plaque, we performed multivariate linear regression for each PET-CT parameter, with Framingham risk score (Table 2) or Framingham risk score and visceral adiposity (Supplementary Table 2) as predictors and non-calcified coronary artery burden (NCB) as the outcome. Standardized ß coefficients, coefficient of partial determination (partial R2), and P-values were compared. All statistical analyses were performed using Stata 17 (StataCorp) and R 4.0.5. A two-tailed P-value ≤ 0.05 was considered significant.
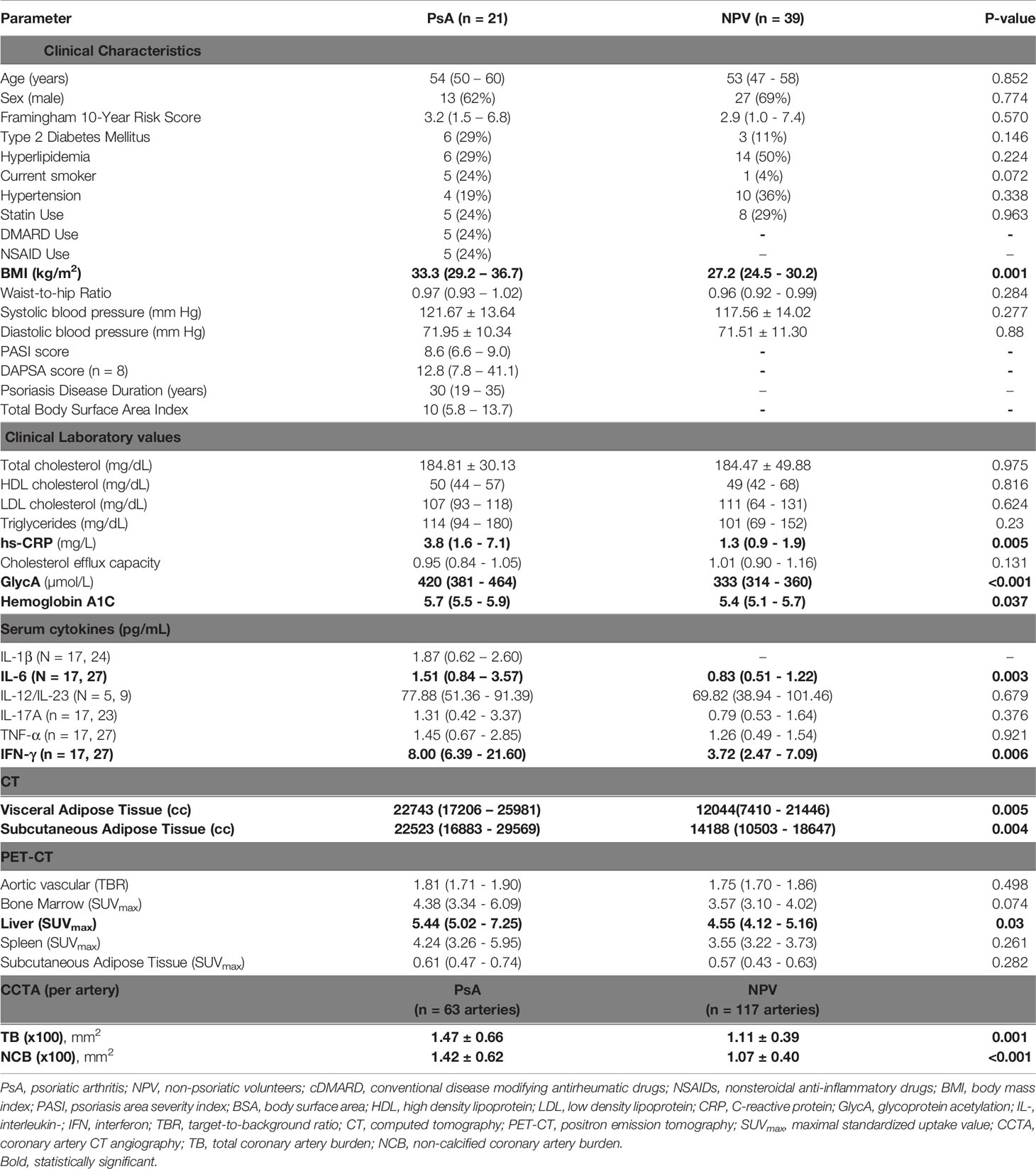
Table 1 Clinical, laboratory, immunological and imaging characteristics of biologic naïve subjects with PsA and moderate to severe skin disease (PASI > 6) and non-psoriatic volunteers.
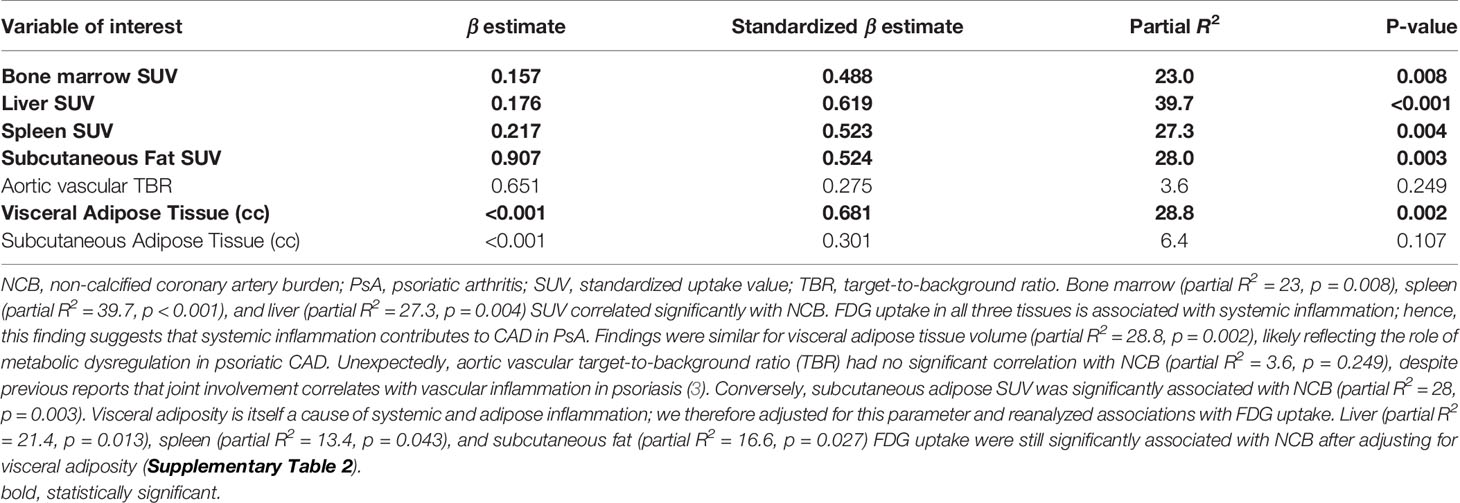
Table 2 Regression analysis of systemic inflammation and fat variables with NCB in biologic-naïve subjects with PsA (n = 34), adjusted for Framingham Risk Score.
39 subjects with biologic-naïve PsA were compared with 56 age-sex-matched non-psoriatic volunteers (Figure 1). Because patients with severe psoriasis are at an increased risk of CVD, we separately analyzed clinical, laboratory and radiographic characteristics of this subgroup (1). Clinical, immunological, and demographic characteristics of subjects with moderate-severe skin disease (PASI > 6, the median value for this cohort that has been previously used as a cutoff for moderate-severe disease (4)) are shown in Table 1, while characteristics of the full treatment-naïve PsA cohort are shown in Supplementary Table 1. Representative CCTA and FDG-PET-CT images are shown in Figures 2,3.
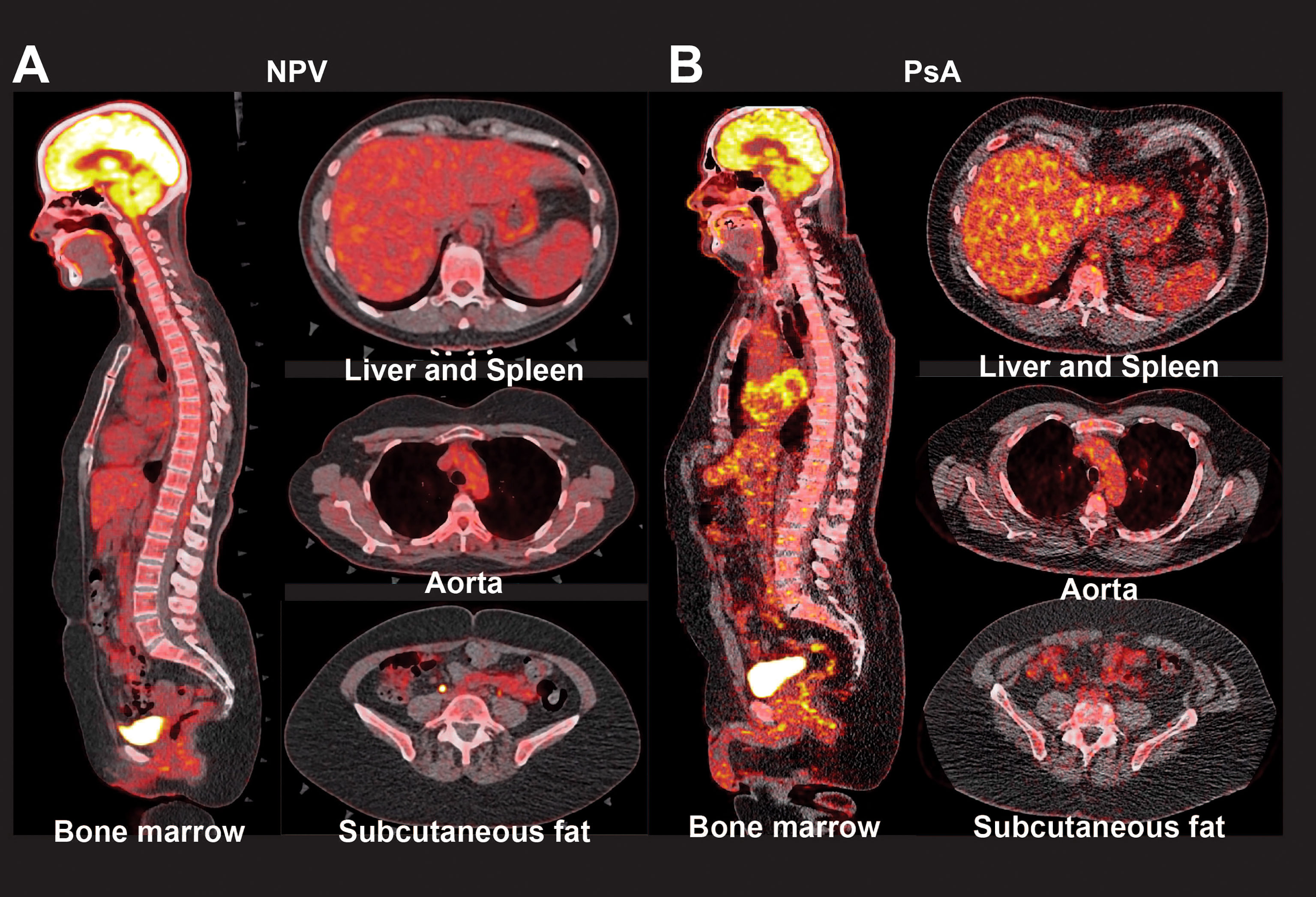
Figure 2 Radiographic markers of inflammation in patients with psoriatic arthritis. Representative PET-CT images show FDG uptake in the bone marrow, liver, spleen, aorta, and subcutaneous fat of non-psoriatic volunteers (NPV, A), and patients with psoriatic arthritis (PsA, B).
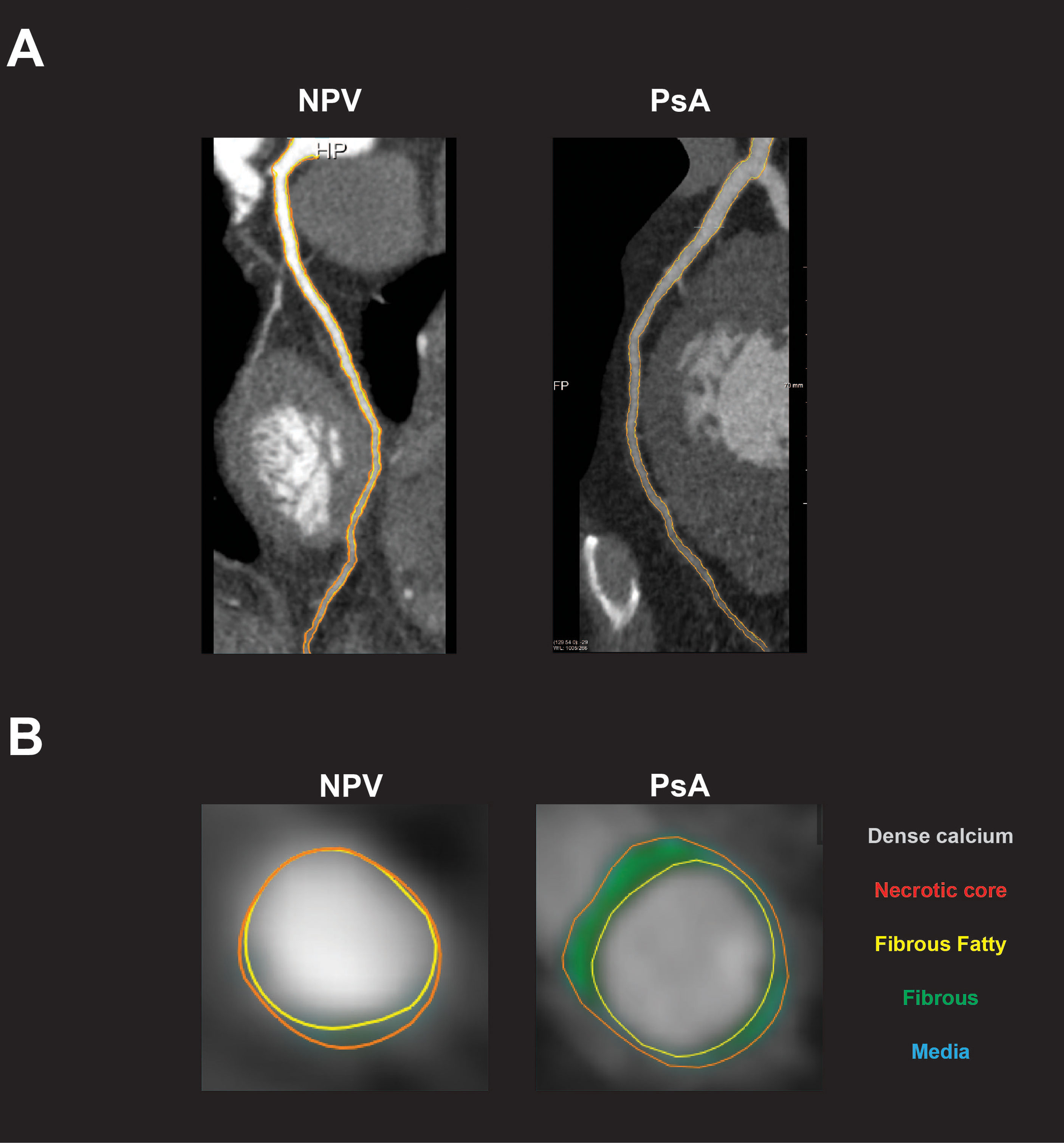
Figure 3 Subclinical coronary artery disease in patients with psoriatic arthritis. (A) Representative coronary artery CT angiogram (CCTA) images show left anterior descending (LAD) artery in non-psoriatic volunteers (NPV) and patients with psoriatic arthritis (PsA). (B) Representative CCTA images show coronary artery plaque in NPV and PsA. Total burden is determined by measuring the total plaque area (orange) around the lumen (yellow). Non-calcified coronary artery burden (NCB) is determined by calculating the difference between total and dense calcified burden. For these subjects, NCB x 100 = 0.602 (NPV) and 2.108 (PsA).
Disease-modifying antirheumatic drug (DMARD) and non-steroidal anti-inflammatory drug (NSAID) were both used in PsA, consistent with the use of these agents for joint pain and inflammation (12). PASI scores and total BSA indices suggested a moderate degree of skin disease, and DAPSA scores suggested a mild-to-moderate amount of joint disease. Both IL-6 and hs-CRP – an IL-6-dependent acute phase reactant – were elevated in PsA (7). IFN-γ levels were also elevated in PsA, as was GlycA, a more comprehensive inflammatory marker. PET maximal standardized uptake values (PET-SUVmax) quantify FDG uptake, which provides a radiographic measure of inflammation within tissues (3, 11). Bone marrow, liver, and spleen FDG update are associated with systemic inflammation; bone marrow and liver PET-SUVmax were significantly elevated in PsA (3, 11).
Because metabolic dysregulation promotes atherogenesis, we also compared clinical and measures of metabolic disease in PsA vs. in volunteers (13). Subjects with PsA had elevated systolic blood pressures (SBP) and a trend towards higher body mass index (BMI) relative to non-psoriatic volunteers. Because overall and visceral adiposity are associated with metabolic syndrome, we measured these parameters radiographically (14). Subjects with PsA had significantly elevated subcutaneous adiposity compared to volunteers, and a trend towards increased body mass index (BMI) and visceral adiposity.
As in the general biologic-naïve cohort, subjects with PsA and moderate-severe skin disease had elevated GlycA, IFN-γ, hs-CRP and IL-6. PsA was more strongly associated with metabolic dysregulation in this subgroup than in the general cohort. BMI, visceral adiposity, and subcutaneous adiposity were significantly higher in PsA than in non-psoriatic volunteers. Hemoglobin A1c was also increased in PsA relative to volunteers, and there was a trend towards increased prevalence of type 2 diabetes mellitus.
CCTA reliably and noninvasively quantifies CAD, and psoriasis increases both total (TB) and non-calcified (NCB) coronary artery burden (9). In our cohort, TB and NCB were numerically increased in subjects with PsA relative to non-psoriatic volunteers, although differences were not statistically significant. In patients with moderate-severe skin disease, TB and NCB were significantly increased in PsA relative to volunteers.
Having characterized the radiographic measures of inflammatory and metabolic dysregulation in PsA, we next investigated associations of these factors with CAD in patients with PsA. One way to determine the contribution of a factor to CAD is to calculate the partial correlation (R2) and standardized β estimates of that variable with NCB (3, 4, 13, 15–17). We therefore measured the partial R2 and standardized β of tissue-specific FDG-PET SUV with NCB. Values are shown in Table 2.
PsA is a major CVD risk factor, but the exact pathophysiologic mechanisms are poorly understood, presenting a challenge to prevention and treatment (1, 3, 9, 13). Here, we used conventional and novel techniques to measure subclinical CAD, metabolic dysregulation, systemic inflammation, and tissue-specific FDG uptake in patients with PsA. PsA was characterized by metabolic dysregulation, systemic inflammation, and subclinical CAD compared to age-sex-matched volunteers. All three were worse in subjects with moderate-severe skin disease, suggesting that the combination of severe skin inflammation and joint disease may be especially atherogenic. Factor analysis revealed multiple potential contributors to CAD in PsA including visceral adiposity and FDG uptake in the liver, spleen, bone marrow, and fat, but not in the aorta.
A central finding of this study is that PsA is an atherogenic state. The association of psoriasis with CVD is well-described, but studies investigating the exact risks of and contributions to CVD in PsA have had disparate results (1, 2, 8). This may be because previous studies have used different outcome measures including traditional CV risk factors, major cardiovascular events (MACE), and carotid intimal medial thickness (cIMT). We employed CCTA, a direct noninvasive angiographic measurement of CAD that allowed us to quantify early disease more accurately, sensitively, and directly than a late binary outcome like MACE or an indirect measurement like cIMT (18–20). Another potential reason for conflicting results of prior studies is that PsA often treated with biologics (2). Because treatment with biologics can prevent psoriatic CVD, this could have affected results of previous studies (12). We avoided this potential confounder by investigating only biologic-naïve subjects.
Atherogenesis is a complex process with multiple drivers including metabolic dysregulation and systemic inflammation (10, 14). Psoriasis is an inflammatory disease that classically causes skin pathology but is clinically heterogeneous and can include systemic inflammation, as evidenced by elevated serum GlycA, hs-CRP, and cytokine levels. In our cohort, measures of IL-6-associated systemic inflammation were elevated in PsA. The IL-6 inhibitor clazakizumab has been shown to modestly improve musculoskeletal manifestations of PsA, but to have no effect on skin disease, so that its development was not pursued for PsA (21). Together with our data, this reported efficacy of clazakizumab for musculoskeletal psoriatic disease suggests that IL-6 may be a PsA-specific inflammatory mediator that is not as important for cutaneous inflammation. PsA is also associated with increased adiposity in population studies, including in this cohort, and adiposity can itself cause of elevated IL-6 and CRP (22). Because an extensive body of literature links IL-6-driven inflammation to CVD, targeting IL-6 may be a potential strategy to prevent PsA-associated CVD (7). Future studies will be needed to explore the underlying etiology of IL-6-driven inflammation and clinical implications in subjects with PsA.
Psoriasis also has strong associations with metabolic syndrome, although the reasons for this are poorly understood. In our cohort, PsA was significantly associated with visceral adiposity, subcutaneous adiposity, and glucose intolerance – particularly in subjects with moderate-severe skin disease. Increased adiposity may result from expansion of cutaneous inflammation to other compartments including the joints, intestine, fat, and liver. Future studies will be needed to determine whether visceral or subcutaneous adiposity promote extracutaneous psoriatic disease, including PsA. Notably, adipose tissue may itself serve as an inflammatory nidus in CVD pathogenesis. Previous studies have reported that psoriatic adipose tissue is infiltrated by immune cells, and that adipokine concentrations are elevated in PsA (5, 6). Accordingly, our regression analysis suggested that adipose inflammation likely contributed to CAD in PsA. We have previously reported that skin inflammation and sacroiliitis are both associated with aortic vascular inflammation, which is a known manifestation of psoriasis; however, our regression analysis suggests that aortic vascular inflammation was not associated with subclinical CAD in PsA (3). Future work will be needed to investigate the reasons for these associations, as well as the effects of bDMARD therapy on organ-specific inflammation. This will help determine whether targeting radiographic adipose inflammation might prevent atherogenesis in PsA.
One important limitation is the size of the treatment-naïve cohorts, which limited our power to detect differences between PsA and volunteers. However, the study was powered to detect significant differences in multiple parameters, particularly in the smaller group of subjects with moderate-severe skin disease. These variables included NCB, skin and systemic inflammation, visceral and subcutaneous adiposity, and other CVD risk factors.
Another limitation is that patients were not uniformly assessed for joint disease activity using clinical measures (i.e., DAPSA). We could not quantify arthritis disease activity radiographically because our PET/CT protocol uses planar imaging, which is not sensitive enough to reliably detect joint inflammation. We were therefore unable to measure the potential contribution of joint inflammation to subclinical CAD in our cohort using clinical or radiographic measures. Multiple studies suggest that joint and nail inflammation may contribute to CVD in patients with PsA (23–25). However, CVD is also linked to multiple other risk factors in patients with psoriatic disease (26, 27). One such risk factor is systemic inflammation: hsCRP and GlycA are elevated in many patients with psoriatic disease and correlate with subclinical CAD but do not usually correlate with joint disease activity (26, 27). Moreover, psoriatic inflammation can involve multiple organs including the aortic root, liver, eye, gastrointestinal tract, and adipose tissue (1). For this reason, it is important to also think about inflammation outside of the skin, joint, and nails when considering potential drivers of CVD in patients with PsA. Although we could not assess the potential contribution of joint inflammation to CVD, our approach did identify several other tissue-specific loci of inflammation that might promote subclinical CVD in PsA. Further studies will be needed to investigate the roles of these different inflammatory foci to CVD pathogenesis in patients with PsA relative to the contributions of skin, joints, and nail inflammation.
A final limitation is that DMARDs and NSAIDs were both used in PsA, which could have affected the risk of CAD PsA relative to non-psoriatic volunteers. However, clinical practice guidelines for PsA recommend both DMARD and NSAID therapy (12). Thus, these treatments reflect real-world PsA management that affects the relative risk of CAD. Important strengths include the specific focus on treatment-naïve subjects, high accuracy of clinical phenotyping, inclusion of mechanistic inflammatory and metabolic biomarkers, and use of cutting-edge imaging studies. Together, these allowed us to identify novel potential inflammatory and metabolic contributors to atherogenesis in PsA, representing an important advance in the field.
In conclusion, psoriasis is atherogenic, dysmetabolic, and inflammatory state, which is most pronounced in patients with moderate to severe skin disease. IL-6-driven systemic inflammation is increased in patients with PsA, providing a potential mechanistic link to atherogenesis in this population. Systemic inflammation is associated with CVD in PsA, suggesting that targeting IL-6 might be effective to prevent CVD in this population. Visceral adiposity is also associated with PsA-related CVD, as is adipose inflammation, suggesting a potential link between metabolic dysregulation and systemic inflammation. Future studies will be needed to follow up on the significance of this finding and to determine the effects of targeting these potential contributors to CVD in patients with PsA.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
This study was approved by the Institutional Review Board of the National Heart Lung and Blood Institute, NIH. The patients/participants provided their written informed consent to participate in this study.
Data collection: AB, AS, MC, CH, MP, and HT; Data analysis: DS, PP, and HL; Writing: DS, ES, MS, and NM. All authors contributed to the article and approved the submitted version.
This work was funded by the intramural research programs of NIAID and NHLBI, and by the National Psoriasis Foundation.
NM has served as a consultant for Amgen, Eli Lilly, and Leo Pharma, receiving grants and other payments.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The authors thank Drs. Joel Gelfand, Mike Ward, and Robert Colbert for critical reading of the manuscript and the nurses on OP7 for their efforts for patient recruitment.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2022.909760/full#supplementary-material
1. Ogdie A, Yu Y, Haynes K, Love TJ, Maliha S, Jiang Y, et al. Risk of Major Cardiovascular Events in Patients With Psoriatic Arthritis, Psoriasis and Rheumatoid Arthritis: A Population-Based Cohort Study. Ann Rheum Dis (2015) 74:326–32. doi: 10.1136/annrheumdis-2014-205675
2. Husted JA, Thavaneswaran A, Chandran V, Eder L, Rosen CF, Cook RJ, et al. Cardiovascular and Other Comorbidities in Patients With Psoriatic Arthritis: A Comparison With Patients With Psoriasis. Arthritis Care Res (Hoboken) (2011) 63:1729–35. doi: 10.1002/acr.20627
3. Rose S, Dave J, Millo C, Naik HB, Siegel EL, Mehta NN. Psoriatic Arthritis and Sacroiliitis are Associated With Increased Vascular Inflammation by 18-Fluorodeoxyglucose Positron Emission Tomography Computed Tomography: Baseline Report From the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative. Arthritis Res Ther (2014) 16:R161. doi: 10.1186/ar4676
4. Salahuddin T, Natarajan B, Playford MP, Joshi AA, Teague H, Masmoudi Y, et al. Cholesterol Efflux Capacity in Humans With Psoriasis is Inversely Related to non-Calcified Burden of Coronary Atherosclerosis. Eur Heart J (2015) 36:2662–5. doi: 10.1093/eurheartj/ehv339
5. Eder L, Jayakar J, Pollock R, Pellett F, Thavaneswaran A, Chandran V, et al. Serum Adipokines in Patients With Psoriatic Arthritis and Psoriasis Alone and Their Correlation With Disease Activity. Ann Rheum Dis (2013) 72:1956–61. doi: 10.1136/annrheumdis-2012-202325
6. Rose S, Stansky E, Dagur PK, Samsel L, Weiner E, Jahanshad A, et al. Characterization of Immune Cells in Psoriatic Adipose Tissue. J Transl Med (2014) 12:258. doi: 10.1186/s12967-014-0258-2
7. Ridker PM, Rane M. Interleukin-6 Signaling and Anti-Interleukin-6 Therapeutics in Cardiovascular Disease. Circ Res (2021) 128:1728–46. doi: 10.1161/CIRCRESAHA.121.319077
8. Gladman DD, Ang M, Su L, Tom BD, Schentag CT, Farewell VT. Cardiovascular Morbidity in Psoriatic Arthritis. Ann Rheum Dis (2009) 68:1131–5. doi: 10.1136/ard.2008.094839
9. Lerman JB, Joshi AA, Chaturvedi A, Aberra TM, Dey AK, Rodante JA, et al. Coronary Plaque Characterization in Psoriasis Reveals High-Risk Features That Improve After Treatment in a Prospective Observational Study. Circulation (2017) 136:263–76. doi: 10.1161/CIRCULATIONAHA.116.026859
10. Harrington CL, Dey AK, Yunus R, Joshi AA, Mehta NN. Psoriasis as a Human Model of Disease to Study Inflammatory Atherogenesis. Am J Physiol Heart Circ Physiol (2017) 312:H867–73. doi: 10.1152/ajpheart.00774.2016
11. Manyak GA, Patel NH, Dey AK, Svirydava M, Parel PM, Teague HL, et al. Abdominal Visceral Adiposity is Associated With Coronary Artery Lipid-Rich Necrotic Core Partly Mediated by Bone Marrow Uptake of (18)F-FDG PET / CT in Psoriasis. J Invest Dermatol (2021). doi: 10.1016/j.jid.2021.10.031
12. Choi H, Uceda DE, Dey AK, Abdelrahman KM, Aksentijevich M, Rodante JA, et al. Treatment of Psoriasis With Biologic Therapy Is Associated With Improvement of Coronary Artery Plaque Lipid-Rich Necrotic Core: Results From a Prospective, Observational Study. Circ Cardiovasc Imaging (2020) 13:e011199. doi: 10.1161/CIRCIMAGING.120.011199
13. Teklu M, Zhou W, Kapoor P, Patel N, Dey AK, Sorokin AV, et al. Metabolic Syndrome and its Factors are Associated With Noncalcified Coronary Burden in Psoriasis: An Observational Cohort Study. J Am Acad Dermatol (2021) 84:1329–38. doi: 10.1016/j.jaad.2020.12.044
14. Powell-Wiley TM, Poirier P, Burke LE, Despres JP, Gordon-Larsen P, Lavie CJ, et al. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation (2021) 143:e984–e1010. doi: 10.1161/CIR.0000000000000973
15. Kutner MH, Nachtsheim CJ, Neter J, Li W. Applied Linear Statistical Models. New York: McGraw-Hill (2005).
16. Sajja A, Abdelrahman KM, Reddy AS, Dey AK, Uceda DE, Lateef SS, et al. Chronic Inflammation in Psoriasis Promotes Visceral Adiposity Associated With Noncalcified Coronary Burden Over Time. JCI Insight (2020) 5:e142534. doi: 10.1172/jci.insight.142534
17. Dey AK, Joshi AA, Chaturvedi A, Lerman JB, Aberra TM, Rodante JA, et al. Association Between Skin and Aortic Vascular Inflammation in Patients With Psoriasis: A Case-Cohort Study Using Positron Emission Tomography/Computed Tomography. JAMA Cardiol (2017) 2:1013–8. doi: 10.1001/jamacardio.2017.1213
18. Yiu KH, Yeung CK, Zhao CT, Chan JC, Siu CW, Tam S, et al. Prevalence and Extent of Subclinical Atherosclerosis in Patients With Psoriasis. J Intern Med (2013) 273:273–82. doi: 10.1111/joim.12002
19. Fischer C, Hulten E, Belur P, Smith R, Voros S, Villines TC. Coronary CT Angiography Versus Intravascular Ultrasound for Estimation of Coronary Stenosis and Atherosclerotic Plaque Burden: A Meta-Analysis. J Cardiovasc Comput Tomogr (2013) 7:256–66. doi: 10.1016/j.jcct.2013.08.006
20. Cheng IT, Wong KT, Li EK, Wong PCH, Lai BT, Yim IC, et al. Comparison of Carotid Artery Ultrasound and Framingham Risk Score for Discriminating Coronary Artery Disease in Patients With Psoriatic Arthritis. RMD Open (2020) 6:e001364. doi: 10.1136/rmdopen-2020-001364
21. Mease PJ, Gottlieb AB, Berman A, Drescher E, Xing J, Wong R, et al. The Efficacy and Safety of Clazakizumab, an Anti-Interleukin-6 Monoclonal Antibody, in a Phase IIb Study of Adults With Active Psoriatic Arthritis. Arthritis Rheumatol (2016) 68:2163–73. doi: 10.1002/art.39700
22. Bastard J-P, Jardel C, Delattre J, Hainque B, Bruckert E, Oberlin F. Evidence for a Link Between Adipose Tissue Interleukin-6 Content and Serum C-Reactive Protein Concentrations in Obese Subjects. Circulation (1999) 99:2219c–22. doi: 10.1161/circ.99.16.2219/c
23. Eder L, Wu Y, Chandran V, Cook R, Gladman DD. Incidence and Predictors for Cardiovascular Events in Patients With Psoriatic Arthritis. Ann Rheum Dis (2016) 75:1680–6. doi: 10.1136/annrheumdis-2015-207980
24. Lam SHM, Cheng IT, Li EK, Wong P, Lee J, Yip RM, et al. DAPSA, Carotid Plaque and Cardiovascular Events in Psoriatic Arthritis: A Longitudinal Study. Ann Rheum Dis (2020) 79:1320–6. doi: 10.1136/annrheumdis-2020-217595
25. Colunga-Pedraza IJ, Galarza-Delgado DA, Azpiri-Lopez JR, Rodriguez-Romero AB, Guajardo-Jauregui N, Cardenas-de la Garza JA, et al. Nail Involvement in Psoriatic Arthritis Patients is an Independent Risk Factor for Carotid Plaque. Ann Rheum Dis (2021) 80:1629–31. doi: 10.1136/annrheumdis-2021-220782
26. Connelly MA, Otvos JD, Shalaurova I, Playford MP, Mehta NN. GlycA, a Novel Biomarker of Systemic Inflammation and Cardiovascular Disease Risk. J Transl Med (2017) 15:219. doi: 10.1186/s12967-017-1321-6
Keywords: psoriasis, psoriatic arthritis, cardiovascular disease, atherogenesis, PSA, imaging
Citation: Schwartz DM, Parel P, Li H, Sorokin AV, Berg AR, Chen M, Dey A, Hong CG, Playford M, Sylvester M, Teague H, Siegel E and Mehta NN (2022) PET/CT-Based Characterization of 18F-FDG Uptake in Various Tissues Reveals Novel Potential Contributions to Coronary Artery Disease in Psoriatic Arthritis. Front. Immunol. 13:909760. doi: 10.3389/fimmu.2022.909760
Received: 31 March 2022; Accepted: 02 May 2022;
Published: 02 June 2022.
Edited by:
Carlos Pérez-Sánchez, Maimonides Biomedical Research Institute of Cordoba (IMIBIC), SpainReviewed by:
Federico Diaz-Gonzalez, University of La Laguna, SpainCopyright © 2022 Schwartz, Parel, Li, Sorokin, Berg, Chen, Dey, Hong, Playford, Sylvester, Teague, Siegel and Mehta. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Daniella M. Schwartz, Daniella.Schwartz@nih.gov; Daniella.Schwartz@pitt.edu
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.