Artificial intelligence (AI) has great potential to improve the field of critical care and enhance patient outcomes. This paper provides an overview of current and future applications of AI in critical illness and its impact on patient care, including its use in perceiving disease, predicting changes in pathological processes, and assisting in clinical decision-making. To achieve this, it is important to ensure that the reasoning behind AI-generated recommendations is comprehensible and transparent and that AI systems are designed to be reliable and robust in the care of critically ill patients. These challenges must be addressed through research and the development of quality control measures to ensure that AI is used in a safe and effective manner. In conclusion, this paper highlights the numerous opportunities and potential applications of AI in critical care and provides guidance for future research and development in this field. By enabling the perception of disease, predicting changes in pathological processes, and assisting in the resolution of clinical decisions, AI has the potential to revolutionize patient care for critically ill patients and improve the efficiency of health systems.
Background: Sepsis-associated acute kidney injury (S-AKI) is considered to be associated with high morbidity and mortality, a commonly accepted model to predict mortality is urged consequently. This study used a machine learning model to identify vital variables associated with mortality in S-AKI patients in the hospital and predict the risk of death in the hospital. We hope that this model can help identify high-risk patients early and reasonably allocate medical resources in the intensive care unit (ICU).
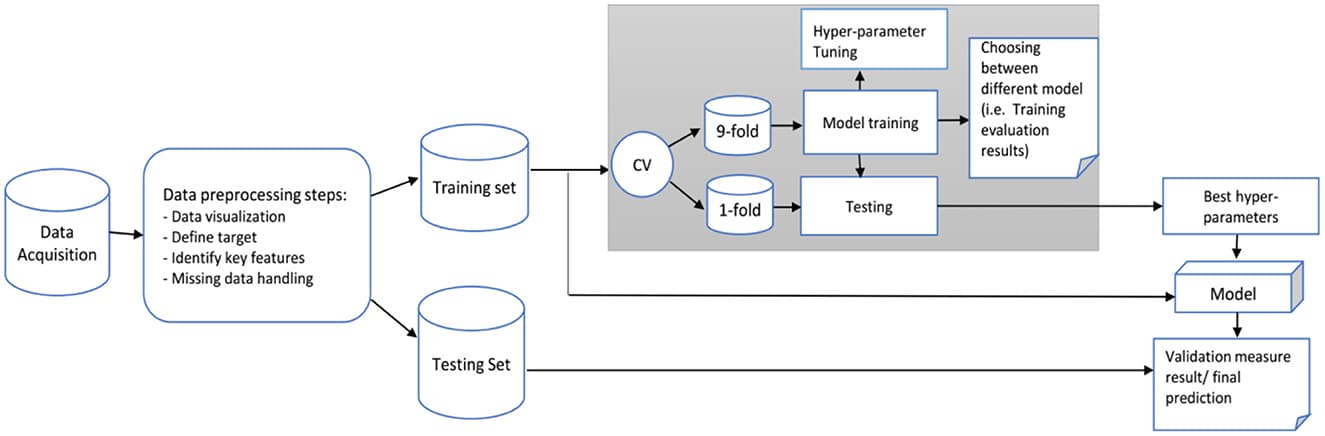
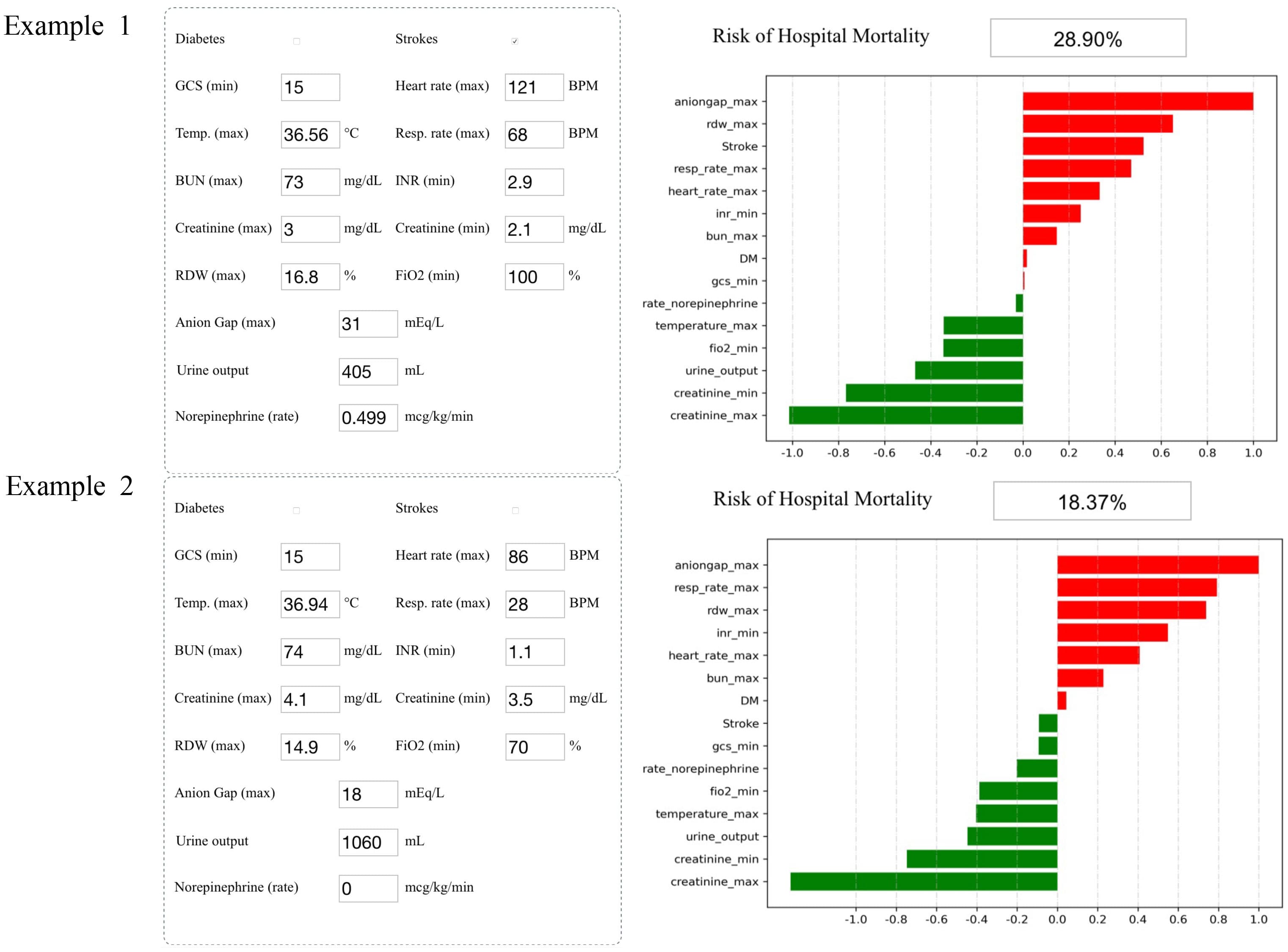
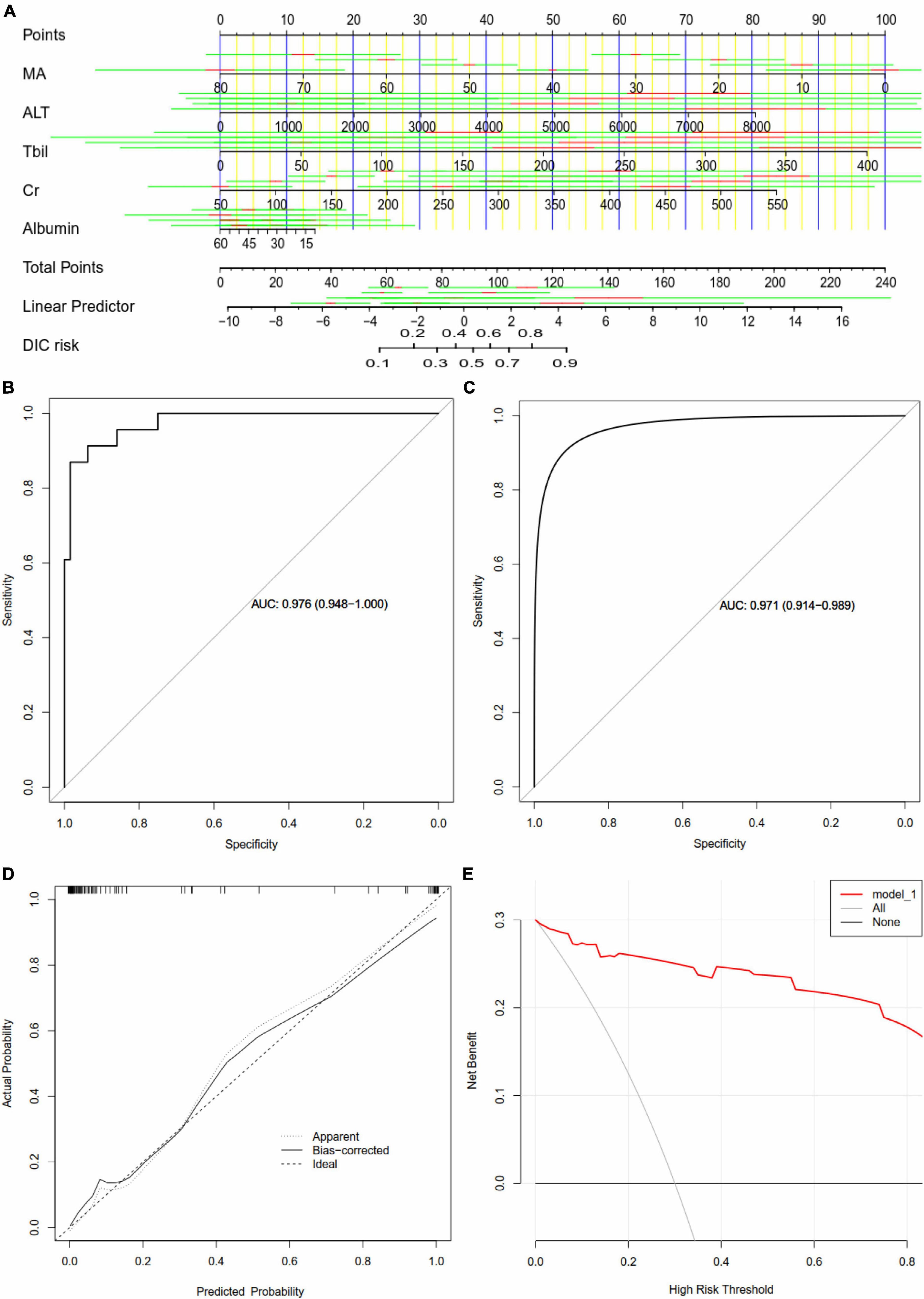
Methods: A total of 16,154 S-AKI patients from the Medical Information Mart for Intensive Care IV database were examined as the training set (80%) and the validation set (20%). Variables (129 in total) were collected, including basic patient information, diagnosis, clinical data, and medication records. We developed and validated machine learning models using 11 different algorithms and selected the one that performed the best. Afterward, recursive feature elimination was used to select key variables. Different indicators were used to compare the prediction performance of each model. The SHapley Additive exPlanations package was applied to interpret the best machine learning model in a web tool for clinicians to use. Finally, we collected clinical data of S-AKI patients from two hospitals for external validation.
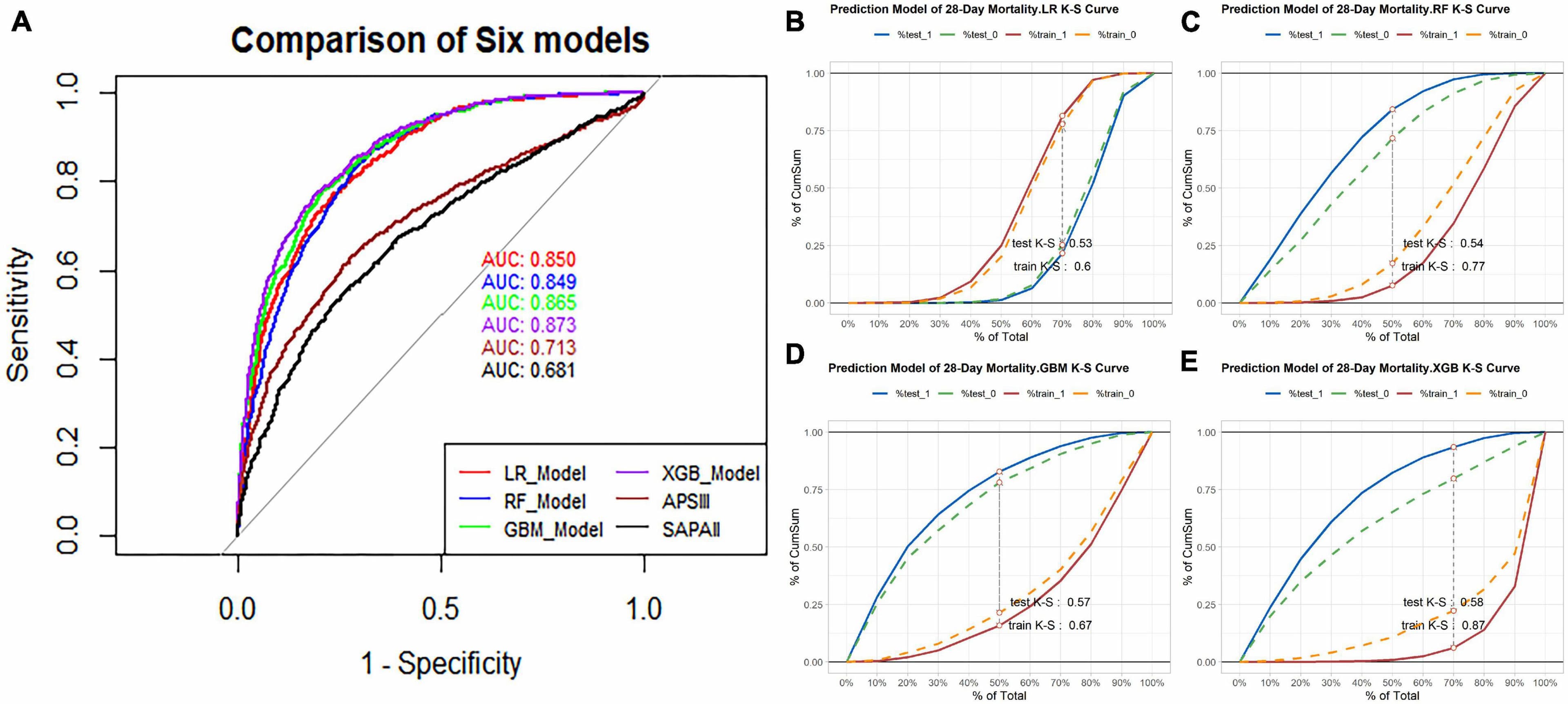
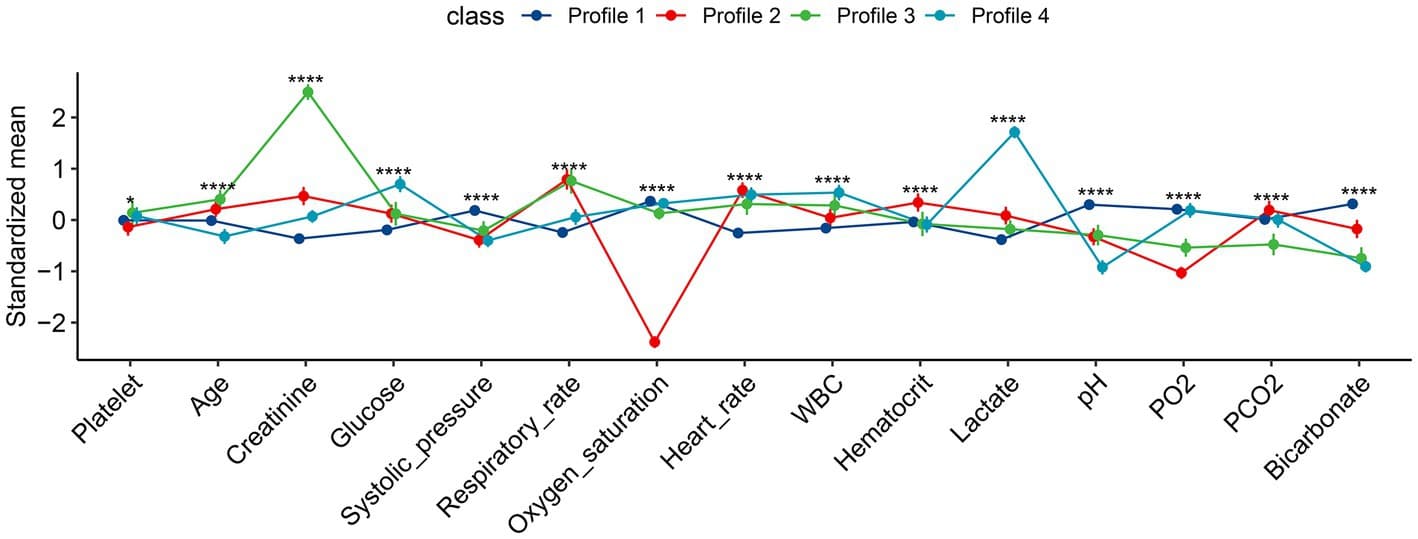
Results: In this study, 15 critical variables were finally selected, namely, urine output, maximum blood urea nitrogen, rate of injection of norepinephrine, maximum anion gap, maximum creatinine, maximum red blood cell volume distribution width, minimum international normalized ratio, maximum heart rate, maximum temperature, maximum respiratory rate, minimum fraction of inspired O2, minimum creatinine, minimum Glasgow Coma Scale, and diagnosis of diabetes and stroke. The categorical boosting algorithm model presented significantly better predictive performance [receiver operating characteristic (ROC): 0.83] than other models [accuracy (ACC): 75%, Youden index: 50%, sensitivity: 75%, specificity: 75%, F1 score: 0.56, positive predictive value (PPV): 44%, and negative predictive value (NPV): 92%]. External validation data from two hospitals in China were also well validated (ROC: 0.75).
Conclusions: After selecting 15 crucial variables, a machine learning-based model for predicting the mortality of S-AKI patients was successfully established and the CatBoost model demonstrated best predictive performance.
Background: Artificial intelligence (AI) and machine learning (ML) models continue to evolve the clinical decision support systems (CDSS). However, challenges arise when it comes to the integration of AI/ML into clinical scenarios. In this systematic review, we followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA), the population, intervention, comparator, outcome, and study design (PICOS), and the medical AI life cycle guidelines to investigate studies and tools which address AI/ML-based approaches towards clinical decision support (CDS) for monitoring cardiovascular patients in intensive care units (ICUs). We further discuss recent advances, pitfalls, and future perspectives towards effective integration of AI into routine practices as were identified and elaborated over an extensive selection process for state-of-the-art manuscripts.
Methods: Studies with available English full text from PubMed and Google Scholar in the period from January 2018 to August 2022 were considered. The manuscripts were fetched through a combination of the search keywords including AI, ML, reinforcement learning (RL), deep learning, clinical decision support, and cardiovascular critical care and patients monitoring. The manuscripts were analyzed and filtered based on qualitative and quantitative criteria such as target population, proper study design, cross-validation, and risk of bias.
Results: More than 100 queries over two medical search engines and subjective literature research were developed which identified 89 studies. After extensive assessments of the studies both technically and medically, 21 studies were selected for the final qualitative assessment.
Discussion: Clinical time series and electronic health records (EHR) data were the most common input modalities, while methods such as gradient boosting, recurrent neural networks (RNNs) and RL were mostly used for the analysis. Seventy-five percent of the selected papers lacked validation against external datasets highlighting the generalizability issue. Also, interpretability of the AI decisions was identified as a central issue towards effective integration of AI in healthcare.
Frontiers in Medicine
The Future of Artificial Intelligence in Acute Kidney Injury