- 1Department of Animal and Food Sciences, University of Kentucky, Lexington, KY, United States
- 2College of Veterinary Medicine, Lincoln Memorial University, Harrogate, TN, United States
- 3Division of Animal and Nutritional Science, West Virginia University, Morgantown, WV, United States
Despite the increased interest and widespread use of cannabidiol (CBD) in humans and companion animals, much remains to be learned about its effects on health and physiology. Metabolomics is a useful tool to evaluate changes in the health status of animals and to analyze metabolic alterations caused by diet, disease, or other factors. Thus, the purpose of this investigation was to evaluate the impact of CBD supplementation on the canine plasma metabolome. Sixteen dogs (18.2 ± 3.4 kg BW) were utilized in a completely randomized design with treatments consisting of control and 4.5 mg CBD/kg BW/d. After 21 d of treatment, blood was collected ~2 h after treat consumption. Plasma collected from samples was analyzed using CIL/LC-MS-based untargeted metabolomics to analyze amine/phenol- and carbonyl-containing metabolites. Metabolites that differed — fold change (FC) ≥ 1.2 or ≤ 0.83 and false discovery ratio (FDR) ≤ 0.05 — between the two treatments were identified using a volcano plot. Biomarker analysis based on receiver operating characteristic (ROC) curves was performed to identify biomarker candidates (area under ROC ≥ 0.90) of the effects of CBD supplementation. Volcano plot analysis revealed that 32 amine/phenol-containing metabolites and five carbonyl-containing metabolites were differentially altered (FC ≥ 1.2 or ≤ 0.83, FDR ≤ 0.05) by CBD; these metabolites are involved in the metabolism of amino acids, glucose, vitamins, nucleotides, and hydroxycinnamic acid derivatives. Biomarker analysis identified 24 amine/phenol-containing metabolites and 1 carbonyl-containing metabolite as candidate biomarkers of the effects of CBD (area under ROC ≥ 0.90; P < 0.01). Results of this study indicate that 3 weeks of 4.5 mg CBD/kg BW/d supplementation altered the canine metabolome. Additional work is warranted to investigate the physiological relevance of these changes.
Introduction
Cannabidiol (CBD) is one of over 100 phytocannabinoids produced by glandular trichomes of Cannabis sativa (1–3). Cannabidiol has been proposed to induce a plethora of beneficial health effects, including anxiolytic, antipsychotic, anti-inflammatory, analgesia, and immunomodulatory effects (4, 5). The wide range of potential therapeutic effects are thought to be a result of multiple mechanisms of action on receptors known to be a part of the endocannabinoid system [ECS; (6, 7)]. Due to the psychoactive effects caused by the action of Δ9-tetrahydrocannabinol (THC) on the CB1 receptor, hemp and all its products, including CBD, were classified as illegal, Schedule I drugs under the Controlled Substances Act (CSA) in 1970 (8, 9). This severely restricted access to CBD as well as the potential for research into the effects of CBD on mammalian physiological systems. As a result, there was little to no opportunity to investigate the potential effects of CBD until industrial hemp was removed from the CSA and CBD was removed from the Schedule I drug list in 2018 (10).
Despite the lack of research, public perception of the supposed health benefits of CBD has led to a rapid expansion of the market for industrial hemp-derived CBD products. In one survey of over 1,100 dog owners, 79.8% said they had purchased hemp or marijuana for their dogs for reasons such as pain relief, relieving anxiety or noise phobia, and reduction of inflammation (11). While this survey represents only a small portion of the population, it does demonstrate favorable perceptions of pet owners on the safety and efficacy of CBD use in pets. Yet despite public opinion, there remains a substantial lack of scientific literature to back these anecdotal claims, particularly regarding potential effects of CBD on long-term health and safety, which makes continued research into its potential benefits and risks all the more essential.
Mass spectrometry-based metabolomics has been increasingly used to assess the health and status of animals and to analyze metabolic alterations caused by diet, disease, or other factors (12–14). Targeted metabolomics can be used to quantify defined groups of metabolites, whereas untargeted metabolomics provides a comprehensive analysis of all measurable analytes in a sample, including any unknowns (15, 16). In instances where the specific metabolites of interest are unknown, untargeted metabolomics can also be used to discover specific biomarkers for later use in targeted metabolomics studies as well as pathway analysis (17, 18). In recent years, chemical isotope labeling (CIL) and liquid chromatography-mass spectrometry (LC-MS)-based untargeted metabolomics has provided an opportunity to analyze metabolites based on chemical groups, including metabolites containing the amine/phenol chemical group (amine/phenol metabolome) which are common intermediate products of amino acid metabolism, and metabolites containing a carbonyl group (carbonyl-metabolome) which common intermediate products of energy metabolism (19).
To date, there has been no evaluation of the effect of CBD on the canine metabolome. Therefore, the objective of this exploratory study was to evaluate the impact of CBD supplementation on the canine metabolome with the underlying hypothesis that after 3 weeks of supplementation, CBD would alter the canine metabolome compared with control. This was accomplished through the use of untargeted metabolomics and biomarker analysis of amine/phenol- and carbonyl-containing metabolites.
Materials and Methods
This study was approved by the Lincoln Memorial University (LMU) institutional animal care and use committee (protocol 1911-RES) before the start of the study. All housing and husbandry were provided in accordance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals (8th ed.), and all applicable LMU protocols.
Subjects and Diets
Sixteen dogs (eight male, eight female, 9 months to 4 years of age, 18.2 ± 3.4 kg BW) of various mixed breeds were received from a local shelter for inclusion in this study. Additionally, the shelter was informed of and gave consent for the use of the dogs for research purposes before their arrival. Prior to beginning the experiment, each dog had a complete blood count (CBC) and serum chemistry analysis (IDEXX Laboratories, Inc., Westbrook, Maine) performed, along with physical evaluation by the attending veterinarian and a fecal examination to rule out any underlying disease that might preclude enrollment. Dogs were excluded if they demonstrated serious behavioral issues, such as human aggression that would endanger research personnel, were severely emaciated, classified as a body condition score <3.5 on a 9-point scale (where one is emaciated and 9 is obese), or if their initial evaluations revealed an underlying disease that required more than routine treatments (such as heartworm positive dogs).
Dogs were individually housed in 1.2 × 1.8 m cages within one of two dog kennels at the LMU DeBusk Veterinary Teaching Center. They were stratified by treatment and sex and evenly distributed between the two kennels. Dogs were fed Purina Pro Plan EN Gastroenteric Fiber Balance Dry Dog Food (Nestle Purina, Inc., St. Louis, MO) to meet the daily metabolizable energy requirements of neutered adult dogs at maintenance, calculated as (70 * BW0.75) * 1.6 and split into two meals per day. Body weight and body condition score (5-point scale) were assessed weekly for the adjustment of diets. Dogs arrived from the shelter and were started on the study diet more than 37 days prior to starting treatments and 58 days before collecting samples for this study.
Experimental Design and Treatments
These dogs were participating in a concurrent study evaluating the impact of CBD on canine voluntary activity (20) with treatments consisting of 0 (placebo treats; CON) or 75.6 ± 5.86 mg CBD/d (CBD). Dogs were blocked by baseline activity before being stratified by age, weight, and sex and randomly assigned to treatments within each block. The CBD was a constituent of a proprietary industrial hemp extract (AgTech Scientific, Paris, KY) that was incorporated into treats and administered in the form of 2 treats daily, each containing half the daily dose. Both CON and CBD treats were composed of the following ingredients: chicken, chicken liver, Asian carp, catfish, and in the case of the CBD treats, industrial hemp extract. Cannabidiol was the primary constituent of the industrial hemp extract; however, trace THC was present in the CBD treatment (2.9 ± 0.22 mg THC/d). Based on the mean BW of dogs included in the study and analysis of the treats, mean dose of CBD ± SD was 4.5 ± 0.77 mg CBD/kg BW/d. Treats were offered solely as a reward upon kennel re-entry following twice-daily exercise within 30 min of meals.
Blood Sample Collection
After 21 d of treatment administration, ~6 mL of blood was collected via cephalic catheter or jugular venipuncture ~2 h after the final treat administration. The selection of this time point was based on previous work demonstrating the half-life of elimination of CBD to be between 1 and 4 h after oral administration (21–23). Blood samples were collected into tubes containing sodium heparin and were immediately centrifuged at 1,645 × g for 10 min. Plasma was collected after centrifugation then stored at −20°C (<12 h) before long-term storage at −80°C.
CIL/LC-MS-Based Untargeted Metabolomics Analysis
Untargeted metabolomic profiling was done using a CIL/LC-MS-based technique with an Agilent 1100 LC system (Palo Alto, CA) connected to a Bruker Impact HD quadrupole time-of-flight (QTOF) MS (Billerica, MA). This technique uses a differential isotope labeling (12C and 13C-labeling) to separate metabolites based on chemical groups followed by LC-MS analysis (19). Detailed information regarding sample preparation, labeling, normalization, LC-UV and LC-MS setup, and metabolite quantification have been reported elsewhere (24, 25). Typical coefficient of variation for this high-performance chemical isotope labeling LC-MS method for metabolome analysis is in the range of 5–10% for individual metabolites (26–28). In this study, amine/phenol- and carbonyl-containing metabolites were analyzed. A total of 19 LC-MS data files were generated (three quality control samples, eight CBD samples, and eight CON samples).
Metabolite Data Processing
Raw data processing on the 19 LC-MS data files was performed using ISOMS Pro 1.0 according to procedures described by Mung and Li (25). Peak pairs whose mean (sample) / mean (blank) was ≤ 4.0 were filtered out. Peak pairs with no data present in at least 80% of the samples were filtered out. The final metabolite-intensity table was generated using IsoMS-Quant (29).
Metabolite Identification
A two-tier identification approach was used to perform metabolite identification. In tier 1, peak pairs were searched against a chemical isotope-labeled metabolite library (CIL Library) based on accurate mass and retention time (29). The CIL Library contains 1,213 experimental entries, including 711 amines/phenols and 90 carbonyls. In tier 2, a linked identity library (LI Library) was used for the identification of the remaining peak pairs. The LI Library includes over 2,000 human endogenous metabolites from 68 metabolic pathways, providing high-confidence putative identification results based on accurate mass and predicted retention time matches (30).
Statistical Analysis
The final metabolite intensity tables for the amine/phenol- and carbonyl-containing metabolome were imported separately into MetaboAnalyst 5.0 software package [www.metaboanalyst.ca; (31)] for statistical analysis. Before statistical analysis, the data were log-transformed, normalized by the median, and auto-scaled. Median scaling was performed to eliminate unwanted inter-sample variations to make the individual samples more comparable to each other. Auto-scaling was used to make the metabolites more comparable in magnitude to each other. Univariate (volcano plot) and multivariate analysis (Partial least squares discriminant analysis [PLS-DA] scores plot) were then generated to identify overall treatment differences across the multivariate dataset. The volcano plot was constructed by plotting the fold change (FC; CBD/CON) of each metabolite against its P-value. Metabolites with FC ≥ 1.2 or ≤ 0.83 having a false discovery ratio (FDR) ≤ 0.05 were considered to be differentially increased or decreased relative to CON, respectively. The utility of the metabolites with FC ≥ 1.2 or ≤ 0.83 and FDR ≤ 0.05 to serve as potential biomarkers of the effects of CBD was tested using a receiver operating characteristic (ROC) curves as calculated by the ROCCET web server using MetaboAnalyst 5.0 software package. Metabolites with an area under ROC (AUROC) ≥ 0.90 and a P ≤ 0.05 were considered excellent biomarkers as defined in Xia et al. (17).
Results
Amine/Phenol Metabolites
Within the amine/phenol analysis, a total of 2,681 unique peak pairs (representing different compounds) were detected. Of those peak pairs, 134 metabolites were positively identified in tier 1 (CIL Library; Supplementary Table 1) and 103 metabolites were putatively identified with high confidence in tier 2 (LI Library; Supplementary Table 2). The PLS-DA scores plot (Figure 1A) shows a clear separation between CON and CBD samples, and the permutation test (P < 0.01) confirms the validity of the PLS-DA model (Supplementary Figure 1).
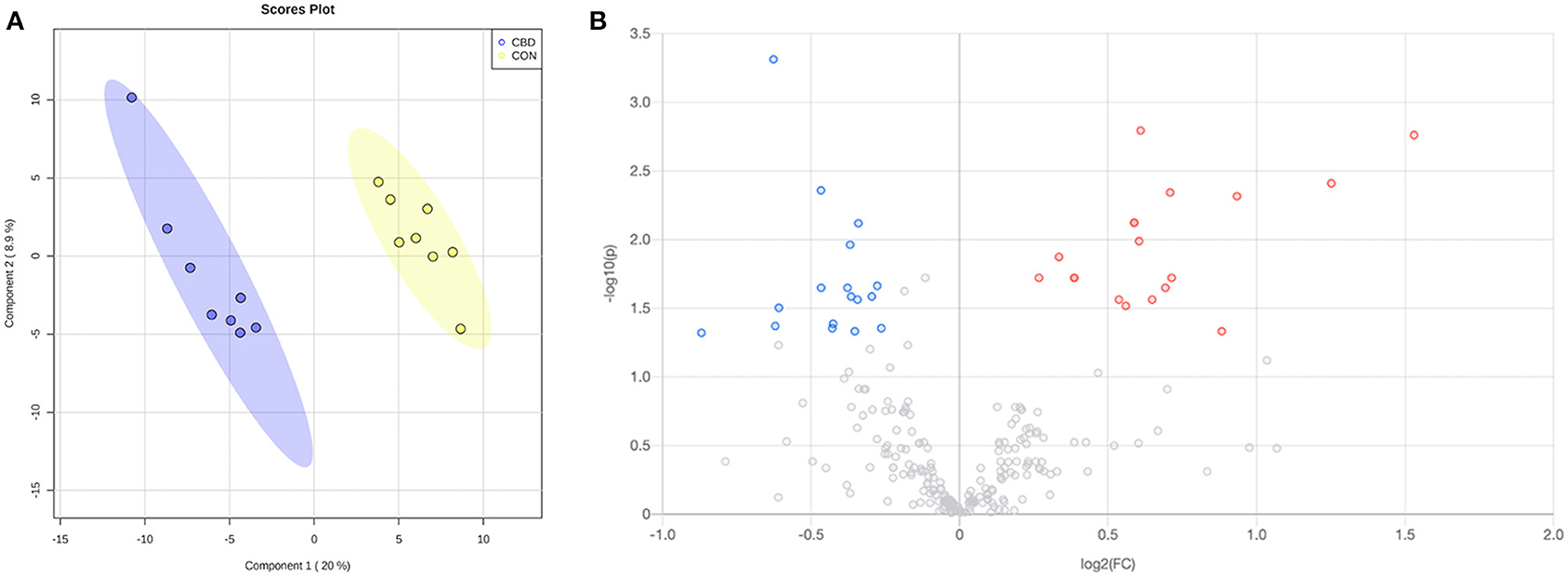
Figure 1. (A) Partial least squares discriminant analysis (PLS-DA) scores plot and (B) volcano plot showing the differential amine/phenol-containing metabolites. Fold change (FC) ≥ 1.2 (in red) or ≤ 0.83 (in blue) with false discovery ratio (FDR) ≤ 0.05 are differentially increased or reduced by cannabidiol (CBD) relative to control (CON).
Volcano plot analysis showed that 32 metabolites were differentially altered (FC ≥ 1.2 or ≤ 0.83, FDR ≤ 0.05) by CBD (Figure 1B; Table 1). Eighteen of those metabolites—pyrimidodiazepine, 4-amino-4-deoxychorismate, isoferulic acid, an isomer of D-glucosamine, 7-carboxy-7-carbaguanine, 2,4-dihydroxyhept-2-enedioate, ascorbate, 2′-deamino-2′-hydroxy-6′-dehydroparomamine, trans-2,3-dihydroxycinnamate, gamma-glutamyl-gamma-aminobutyraldehyde, 1,4-diaminobutane, tyramine, an isomer of 2-deoxy-scyllo-inosamine, isoleucyl-alanine, 3-(4-hydroxyphenyl)-pyruvate, aspartyl-threonine, vanillic acid, and D-lysopine—were differentially increased (FC ≥ 1.2, FDR ≤ 0.05) by CBD. The other 14 metabolites—N-acetyl-L-asparagine, alanyl-proline, asparaginyl-aspartic acid, seryl-aspartic acid, phenylalanyl-glycine, prolyl-glutamine, o-tyrosine, N-acetyl-L-adrenaline, L-threo-3-methylaspartate, Z-3-peroxyaminoacrylate, L-glutamate-5-semialdehyde, 2-methyl-3-hydroxy-5-formylpyridine-4-carboxylate, gamma-aminobutyric acid, and aspartyl-glutamine—were differentially reduced (FC ≤ 0.83, FDR ≤ 0.05) by CBD compared to CON.
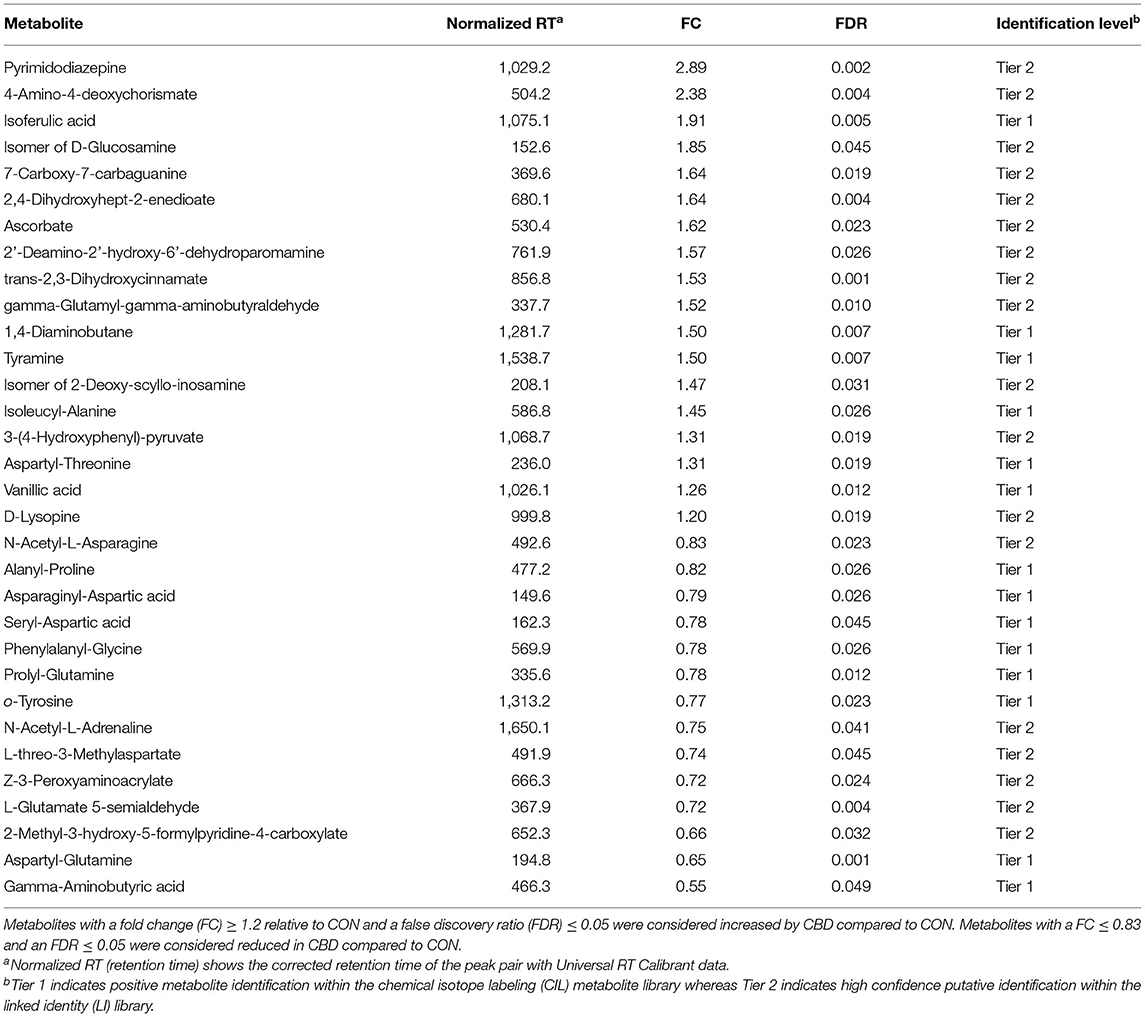
Table 1. Identified amine/phenol-containing metabolites affected by cannabidiol (CBD) compared to control (CON).
Univariate ROC analysis of the 32 identified amine/phenol-containing metabolites that were differentially increased or decreased by CBD revealed 24 metabolites—aspartyl-glutamine, gamma-aminobutyric acid, gamma-glutamyl-gamma-aminobutyraldehyde, L-glutamate-5-semialdehyde, prolyl-glutamine, pyrimidodiazepine, 4-amino-4-deoxychorismate, trans-2,3-dihydroxycinnamate, alanyl-proline, N-acetyl-L-asparagine, (Z)-3-peroxyaminoacrylate, 1,4-diaminobutane, 2′-deamino-2′-hydroxy-6′-dehydroparomamine, ascorbate, D-lysopine, o-tyrosine, phenylalanyl-glycine, 2,4-dihydroxyhept-2-enedioate, asparaginyl-aspartic acid, isoferulic acid, 7-carboxy-7-carbaguanine, 3-(4-hydroxyphenyl)-pyruvate, aspartyl-threonine, and isoleucyl-alanine—that appear to be highly predictive of the metabolomic changes between CBD and CON (AUROC ≥ 0.90; P < 0.001; Figure 2).

Figure 2. Box and whisker plots for candidate amine/phenol-containing biomarkers altered by cannabidiol (CBD; in blue) compared to control (CON; in yellow). Candidate amine/phenol biomarkers included (A) aspartyl-glutamine (AUROC = 1.00; P < 0.001); (B) gamma-aminobutyric acid (AUROC = 1.00; P = 0.005); (C) gamma-glutamyl-gamma-aminobutyraldehyde (AUROC = 1.00; P < 0.001); (D) L-glutamate-5-semialdehyde (AUROC = 1.00; P < 0.001); (E) prolyl-glutamine (AUROC = 1.00; P < 0.001); (F) pyrimidodiazepine (AUROC = 1.00; P < 0.001); (G) 4-amino-4-deoxychorismate (AUROC = 0.98; P < 0.001); (H) trans-2,3-dihydroxycinnamate (AUROC = 0.98; P < 0.001); (I) alanyl-proline (AUROC = 0.97; P = 0.002); (J) N-acetyl-L-asparagine (AUROC = 0.97; P < 0.001); (K) (Z)-3-peroxyaminoacrylate (AUROC = 0.95; P < 0.001); (L) 1,4-diaminobutane (AUROC = 0.95; P < 0.001); (M) 2'-deamino-2'-hydroxy-6'-dehydroparomamine (AUROC = 0.95; P = 0.004); (N) ascorbate (AUROC = 0.95; P = 0.003); (O) D-lysopine (AUROC = 0.95; P = 0.004); (P) o-tyrosine (AUROC = 0.95; P = 0.001); (Q) phenylalanyl-glycine (AUROC = 0.95; P = 0.002); (R) 2,4-dihydroxyhept-2-enedioate (AUROC = 0.94; P < 0.001); (S) asparaginyl-aspartic acid (AUROC = 0.94; P = 0.003); (T) isoferulic acid (AUROC = 0.94; P < 0.001); (U) 7-carboxy-7-carbaguanine (AUROC = 0.92; P = 0.001); (V) 3-(4-hydroxyphenyl)pyruvate (AUROC = 0.91; P = 0.004); (W) aspartyl-threonine (AUROC = 0.91; P = 0.005); and (X) isoleucyl-alanine (AUROC = 0.91; P = 0.007).
Carbonyl Metabolites
Within the carbonyl analysis, a total of 612 unique peak pairs were detected. Of those peak pairs, 6 peak pairs were positively identified in tier 1 (CIL Library; Supplementary Table 1) and 15 peak pairs were putatively identified with high confidence in tier 2 (LI Library; Supplementary Table 2). The PLS-DA scores plot (Figure 3A) shows clear separation between CON and CBD samples, and the permutation test (P < 0.01) confirms the validity of the PLS-DA model (Supplementary Figure 2).
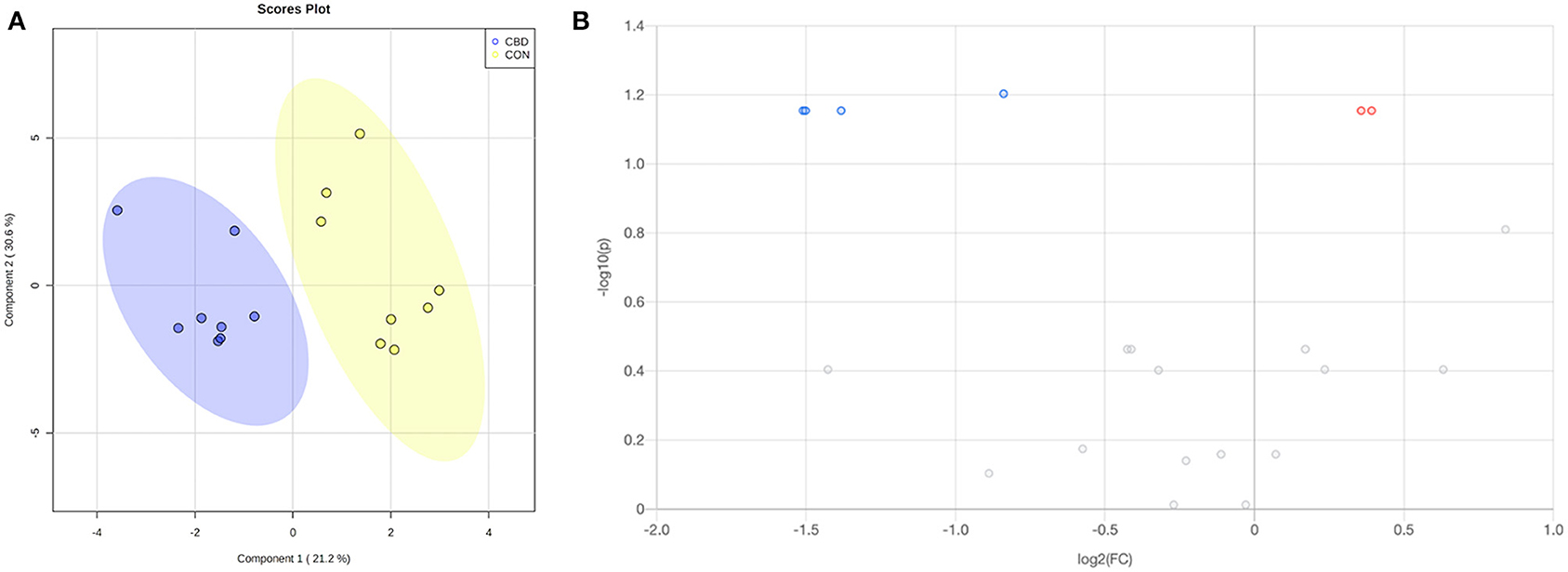
Figure 3. (A) Partial least squares discriminant analysis (PLS-DA) scores plot and (B) volcano plot showing the differential carbonyl-containing metabolites. Fold change (FC) ≥ 1.2 (in red) or ≤ 0.83 (in blue) with false discovery ratio (FDR) ≤ 0.05 are differentially increased or reduced by cannabidiol (CBD) relative to control (CON).
Volcano plot analysis showed that five metabolites were differentially altered (FC ≥ 1.2 or ≤ 0.83, FDR ≤ 0.05) by CBD (Figure 3B; Table 2). Glucose and 2-formylglutarate were differentially increased (FC ≥ 1.2, FDR ≤ 0.05) by CBD, while glyceraldehyde, isomer of glyceraldehyde, and 4-oxoglutaramate were differentially reduced (FC ≤ 0.83, FDR ≤ 0.05) by CBD compared to control.
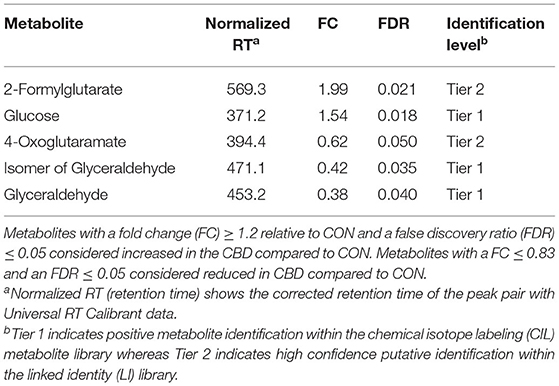
Table 2. Identified carbonyl-containing metabolites affected by cannabidiol (CBD) compared to control (CON).
Univariate ROC analysis of the five carbonyl metabolites positively and putatively identified that were differentially altered by CBD revealed that plasma glucose appears to be highly predictive of the metabolomic changes between CBD and CON (AUROC = 0.91; P = 0.020; Figure 4).
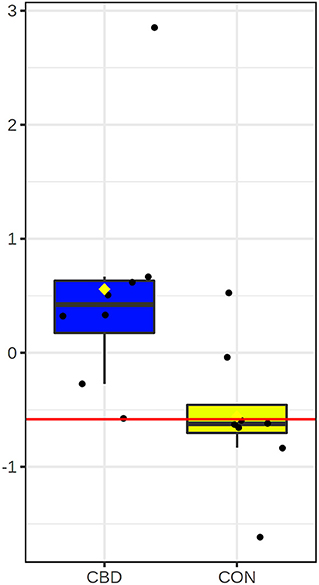
Figure 4. Box and whisker plot for candidate carbonyl-containing biomarker altered by cannabidiol (CBD; in blue) compared to control (CON; in yellow). Glucose (AUROC = 0.91; P = 0.020) was the only candidate biomarker for carbonyl-containing metabolites.
Discussion
Amino Acid Metabolism
Increased concentrations of tyramine, 3-(4-hydroxyphenyl)-pyruvate, 2,4-dihydroxyhept-2-enedioate, gamma-glutamyl-gamma-aminobutyraldehyde, 1,4-diaminobutane, and D-lysopine may indicate that CBD altered amino acid metabolism. Tyramine, 3-(4-hydroxyphenyl)-pyruvate, and 2,4-dihydroxyhept-2-enedioate are intermediates in tyrosine and phenylalanine metabolism (32–34). Tyramine, in particular, is also involved in the biosynthesis of many secondary metabolites in plants, such as isoquinoline alkaloids, flavonoids, and hydroxycinnamic acid amines (35, 36). Gamma-glutamyl-gamma-aminobutyraldehyde and 1,4-diaminobutane (i.e., putrescene) are intermediates in arginine, proline, and ornithine degradation pathways (37, 38). Putrescene is also known to play a role in the regulation of cell growth, protein synthesis, apoptosis, and other cellular processes (39, 40). D-lysopine is an amino opine derivative of L-lysine found in crown gall tumors produced by pathogenic bacteria that infect plants, including C. sativa. While not known to be produced in mammalian systems, other opines like saccharopine are known intermediates in the metabolism of lysine in mammals (41–43). The increase in these metabolites may suggest that CBD enhanced degradation of these amino acids.
Decreased concentrations of N-acetyl-L-asparagine, o-tyrosine, N-acetyl-L-adrenaline, L-threo-3-methylaspartate, L-glutamate-5-semialdehyde, and 4-oxoglutaramate may also suggest that CBD altered metabolism of other amino acids. N-acetyl-L-asparagine is a derivative of asparagine that is N-acetylated by N-acetyltransferase 1 (NAT1), one of several acetyltransferases known to play a role in drug metabolism (44). This enzyme has been suggested to play a role in the regulation of mTOR complex I activation, cancer cell proliferation, and mitochondrial function (44–46). As CBD is suspected to exert anti-cancer effects, it may be prudent in future work to investigate if CBD supplementation alters NAT1 activity. o-Tyrosine is a structural isomer of tyrosine and a phenylalanine derivative. It is considered a marker for oxidative stress as it is produced through free-radical hydroxylation of phenylalanine (47, 48). If the decrease in o-tyrosine was due to CBD supplementation, this may contribute to the suspected antioxidative effects of CBD. N-acetyl-L-adrenaline is a methylated form of epinephrine, an adrenal hormone involved in the regulation of visceral functions (49, 50).
L-threo-3-methylaspartate is an amino acid formed by glutamate mutase and can be metabolized by methylaspartate ammonia-lyase. It is found in the structures of the antibiotics friulimicin and vicenistatin and in carbon metabolism of haloarchaea (51, 52). L-glutamate-5-semialdehyde is a non-proteinogenic amino acid that is an intermediate in both proline and arginine biosynthesis from glutamate (53, 54). 4-oxoglutaramate is an intermediate in one of the histidine catabolism pathways that leads to the production of 2-oxoglutarate (i.e., α-ketoglutarate), which then feeds into the citric acid cycle (55, 56). Aspartyl-glutamine, aspartyl-threonine, alanyl-proline, asparaginyl-aspartic acid, isoleucyl-alanine, phenylalanyl-glycine, prolyl-glutamine, and seryl-glycine are products of the incomplete breakdown of protein digestion or catabolism. While some dipeptides are known to have physiological or cell-signaling effects, none of the affected dipeptides have been identified as one of these bioactive molecules (57, 58).
The altered concentrations of these metabolites suggest an effect of CBD on amino acid metabolism. However, since the relative concentrations of the individual amino acids (arginine, aspartate, glutamate, and proline) were unaffected by treatment, the biological significance of the changes in these metabolites is unclear. Additional research is needed to assess the potential for CBD to alter amino acid metabolism.
Glucose Metabolism
The increase in glucose, an isomer of glucosamine, 2′-deamino-2′-hydroxy-6′-dehydroparomamine, and an isomer of 2-deoxy-schyllo-inosamine may suggest that glucose metabolism was altered by CBD. The endocannabinoid system—by which CBD and other cannabinoids exert physiological effects—plays a well-established role in glucose and energy metabolism, marking it as a target for the treatment of metabolic diseases like type 2 diabetes (59). Cannabinoids like CBD and THC have been suggested to reduce hyperglycemia and increase insulin production in rodents (60, 61), but this has yet to be investigated in a canine model.
Glucosamine is an amino sugar that is readily synthesized in the body from glucose and glutamine. It is an essential component of mucopolysaccharides that are incorporated into connective tissue, mucous secretions, skin, tendons, ligaments, and cartilage. Additionally, it helps regulate the synthesis of collagen in cartilage (62–64). Because of its high concentration in joint tissues, glucosamine is commonly used as a dietary supplement in humans, horses, and dogs as a support for joint health and function and to relieve symptoms of osteoarthritis, though there is little scientific evidence supporting these effects (65). Both 2-deoxy-scyllo-inosamine and 2'-deamino-2'-hydroxy-6'-dehydroparomamine are intermediates in the biosynthesis of aminoglycoside antibiotics, like kanamycin, from glucose in Streptomyces bacterial species (66, 67). However, since these metabolites are not known to be generated in mammalian systems, the biological significance of this is unclear.
The decrease in gamma-aminobutyric acid (GABA), glyceraldehyde, and an isomer of glyceraldehyde may also indicate that CBD altered glucose metabolism. Best known as the primary inhibitory neurotransmitter in the central nervous system, GABA is also produced by insulin-producing β cells of the pancreas and immune cells (68, 69). In the pancreas, GABA inhibits glucagon secretion from neighboring α cells and modulates glucose homeostasis (68, 70, 71). This action of GABA in the pancreas has highlighted its potential as a target for diabetes treatment (72). It has also been shown to regulate cytokine secretion from human peripheral blood mononuclear cells (PBMCs) and CD4+ T cells and is thought to exert anti-inflammatory effects (73–75).
Glyceraldehyde, a triose monosaccharide, is an intermediate in glycolysis in its phosphorylated form (GAP). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH), the enzyme that catalyzes the conversion of GAP into 1,3-bisphosphoglycerate, is a major regulator of carbon flux in the body (76). It is also known to play a role in other cellular functions such as redox sensing, membrane fusion, iron homeostasis, and cell death (77). The decrease in glyceraldehyde may lend support to the suspected anti-obesity and anti-diabetic effects of CBD; however, the increase in glucose and decrease in GABA would appear to be incongruous with this potential effect. These results highlight a relatively unexplored avenue of CBD research that warrants further investigation.
Hydroxycinnamic Acid Derivatives
The increase in plasma isoferulic acid (IFA), trans-2,3-dihydroxycinnamate, and vanillic acid may suggest that CBD altered the metabolism of hydroxycinnamic acid derivatives. Isoferulic acid is a naturally occurring hydroxycinnamic acid derivative commonly found in Lobelia and Cimicifuga species. It is an isomer of ferulic acid, a phenolic compound that is a component of lignin, which is commonly found in cell walls of plants, including C. sativa (78). Ferulic acid has also been isolated from hemp seed meal, a byproduct of hemp oil processing (79). Both ferulic acid and IFA have been reported to have anti-inflammatory, anti-viral, anti-oxidative, and anti-diabetic properties and are commonly used as ingredients in herbal medicines in Japan and China (80, 81). Isoferulic acid has been shown to reduce plasma glucose in diabetic rats and to inhibit IL-8 production in mice (82, 83). Additionally, IFA has been suggested to act as an anti-glycation compound. Protein glycation is a non-enzymatic reaction associated with oxidative stress and reactive oxygen species (ROS) production; it is thought to be a contributor to age-related diseases (84). In several studies, IFA protected against fructose- and glucose-mediated glycation and inhibited ROS production in vitro (80, 85). In humans, IFA has been shown to be an intermediate in the metabolism of plant-derived phenolic compounds like caffeic acid (86), with humans obtaining the majority from dietary consumption.
Trans-2,3-dihydroxycinnamate is a derivative of cinnamic acid, which is an intermediate in the biosynthesis of lignin, flavonoids, and other secondary metabolites produced by plants like C. sativa (87). Cinnamic acid and its derivatives, like trans-2,3-dihydroxycinnamate, possess antioxidizing activity (88). Vanillic acid is a dihydroxybenzoic acid derivative commonly used as a flavoring agent. It is also an intermediate in the synthesis of vanillin from ferulic acid and a phenolic compound that, like IFA, is a component of lignin present in the secondary cell wall of plants, including C. sativa (89, 90). Like other lignin-associated aromatic acids, vanillic acid has been reported to exert antimicrobial properties (91). If the increase in these metabolites is a result of CBD supplementation, it is possible that these compounds may contribute to the suspected anti-microbial, anti-inflammatory, and antioxidative effects of hemp. However, it is unlikely that the changes in these compounds are solely due to increased dietary consumption as the treats used in this study provided a small quantity of industrial hemp extract. Conversely, it may be possible that changes in the metabolism of these hydroxycinnamic acid derivatives occurred as a result of CBD; additional work is warranted to further investigate these potential effects.
Vitamin and Nucleotide Metabolism
Increased concentrations of pyrimidodiazepine, 4-amino-4-deoxychorismate, 7-carboxy-7-carbaguanine, 2-formylglutarate, and ascorbate may indicate an alteration of vitamin and nucleotide metabolism. Pyrimidodiazepine is a derivative of uracil and a substrate for pyrimidodiazepine synthase, an enzyme that can contribute to glutathione synthesis. Uracil derivatives like pyrimidodiazepine are also thought to possess antimicrobial and antioxidant properties (92). 4-amino-4-deoxychorismate is a precursor for para-aminobenzoic acid (pABA) biosynthesis, which is a precursor for folic acid (vitamin B9) synthesis in plants and microorganisms (93, 94). While vitamin B9 is an essential cofactor that facilitates methyl transfers, mammals do not possess the enzymes to produce folic acid and instead rely on dietary consumption of the vitamin (93). 7-carboxy-7-carbaguanine is pyrrolopyrimidine that, like 4-amino-4-deoxychorismate, is involved in the biosynthesis of vitamin B9 (95). 2-formylglutarate is an intermediate in nicotinamide metabolism in several bacterial species [(96, 97)]. Ascorbate, or vitamin C, can be synthesized in dogs from glucose or ingested in the diet (98, 99). Vitamin C serves as a cofactor in several essential reactions, including collagen synthesis and wound healing, as well as an antioxidant. If the increase in ascorbate was due to CBD supplementation, this may contribute to the suspected antioxidative effects of CBD.
Decreased concentrations of 2-methyl-3-hydroxy-5-formylpyridine-4-carboxylate and (Z)-3-peroxyaminoacrylate may also support an effect of CBD on vitamin and nucleotide metabolism. 2-methyl-3-hydroxy-5-formylpyridine-4-carboxylate is an intermediate in the pyridoxine (vitamin B6) degradation pathway (100). Vitamin B6 is an essential cofactor in several enzymatic reactions, including the synthesis of glutathione, an important antioxidant (101). (Z)-3-peroxyaminoacrylate is an intermediate in bacterial pyrimidine degradation pathway known as the Rut pathway. However, this pathway and intermediate are not known to play a role in mammalian pyrimidine metabolism (102).
Since the relative concentrations of pyridoxine, uracil, and folate were not affected by treatment, and since several of these metabolites are not known to be generated in mammalian systems, the biological significance of these changes is unclear. However, increased pyrimidodiazepine and ascorbate, along with decreased 2-methyl-3-hydroxy-5-formylpyridine-4-carboxylate, may indicate an influence of CBD on antioxidant status. Further studies are needed to determine the roles of these metabolites and the potential effects of CBD on these pathways.
Strengths and Limitations
Cannabidiol is already being supplemented to dogs for its potential therapeutic applications including osteoarthritis, separation anxiety, noise phobias, and epilepsy (4, 11). Several studies have evaluated its effectiveness in dogs with osteoarthritis (23, 103, 104) noise phobias (105), and epilepsy (106) with mixed results. Considerable work has also been done investigating pharmacokinetics (21–23, 107) and safety (108, 109) following oral CBD administration; however, this study is the first to evaluate the impact of CBD supplementation on the canine metabolome. This analysis provides a comprehensive scan of potential metabolic targets in dogs receiving CBD; however, because of the lack of metabolic profile with CBD it was not intended to be all-encompassing but rather a first look into the potential for CBD supplementation to alter the canine metabolome. Thus, this study may be limited by the relatively short duration of CBD supplementation, small sample size, lack of baseline measurement, and the use of only a single CBD dosage. Even so, identifying changes in the metabolome is essential for directing future targeted investigations into both the physiological relevance of these changes as well as elucidating potential mechanisms leading to these observed effects. It would be beneficial for future work to evaluate metabolomic changes in an unhealthy or diseased population of dogs supplemented with CBD and potential differences between acute and long-term CBD administration.
Conclusions
This study demonstrated the canine metabolome was altered with 4.5 mg CBD/kg BW/d supplementation for 3 weeks. Altered metabolites may suggest a potential for CBD to influence glucose, amino acid, vitamin, and nucleotide metabolism. Additionally, the increase in relative concentrations of metabolites like o-tyrosine, IFA, glucosamine, and pyrimidodiazepine may indicate potential pathways by which CBD may exert suspected anti-inflammatory, antioxidant, and antimicrobial effects. Several metabolites were identified as potential biomarkers for changes in the canine metabolome by CBD. Further studies with larger sample sizes, longer supplementation periods and baseline comparisons to refine metabolites are necessary to elucidate the physiological relevance of these changes.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
Ethics Statement
The animal study was reviewed and approved by LMU IACUC. Written informed consent was obtained from the owners for the participation of their animals in this study.
Author Contributions
DH, EM, KM, and SK-M contributed to the conception and design of the study. EM, SK-M, and DS facilitated data collection. EM and IO performed statistical analysis. EM wrote the first draft of the manuscript. All authors contributed to manuscript revision, read, and approved the submitted version.
Funding
The authors declare that this study received funding from AgTech Scientific, Paris, KY. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article, or the venue for publication.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Lincoln Memorial University students K. Athey, H. Barnhart, L. Calvin, K. Dubois, J. Gauldin, S. Swears, M. Kight, M. Mendoza, J. Steen, S. Swears, and K. Williams for their assistance in caring for dogs and facilitating data collection.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fvets.2021.685606/full#supplementary-material
Supplementary Table 1. Amine/phenol- and carbonyl-containing metabolites identified in the tier 1 chemical isotope labeling (CIL) library.
Supplementary Table 2. Amine/phenol- and carbonyl-containing metabolites identified in the tier 2 linked identity (LI) library.
Supplementary Figure 1. Partial least squares discriminant analysis (PLS-DA) model permutation for amine/phenol-containing metabolites.
Supplementary Figure 2. Partial least squares discriminant analysis (PLS-DA) model permutation for carbonyl-containing metabolites.
References
1. Gagne SJ, Stout JM, Liu E, Boubakir Z, Clark SM, Page JE. Identification of olivetolic acid cyclase from Cannabis sativa reveals a unique catalytic route to plant polyketides. Proc Natl Acad Sci USA. (2012) 109:12811–6. doi: 10.1073/pnas.1200330109
2. Stout JM, Boubakir Z, Ambrose SJ, Purves RW, Page JE. The hexanoyl-CoA precursor for cannabinoid biosynthesis is formed by an acyl-activating enzyme in Cannabis sativa trichomes. Plant J. (2012) 71:353–65. doi: 10.1111/j.1365-313X.2012.04949.x
3. Taura F, Sirikantaramas S, Shoyama Y, Yoshikai K, Shoyama Y, Morimoto S. Cannabidolic-acid synthase, the chemotype-determining enzyme in the fiber-type Cannabis sativa. FEBS Lett. (2007) 581:2929–34. doi: 10.1016/j.febslet.2007.05.043
4. Landa L, Sulcova A, Gbelec P. The use of cannabinoids in animals and therapeutic implications for veterinary medicine: a review. Veterinární Medicína. (2016) 61:111–22. doi: 10.17221/8762-VETMED
5. Mechoulam R, Parker LA, Gaillily R. Cannabidiol: an overview of some pharmacologial aspects. J Clin Pharmacol. (2002) 42:11S–9S. doi: 10.1002/j.1552-4604.2002.tb05998.x
6. Bergamaschi MM, Queiroz RHC, Crippa JAS, Zuardi AW. Safety and side effects of cannabidiol, a Cannabis sativa constituent. Curr Drug Saf. (2011) 6:237–49. doi: 10.2174/157488611798280924
7. Lu HC, Mackie K. An introduction to the endogenous cannabinoid system. Bio Psychiatry. (2015) 79:516–25. doi: 10.1016/j.biopsych.2015.07.028
8. Mead A. The legal status of cannabis (marijuana) and cannabidiol (CBD) under U.S. law. Epilepsy Behav. (2017) 70:288–91. doi: 10.1016/j.yebeh.2016.11.021
9. Corroon J, Kight R. Regulatory status of cannabidiol in the United States: a perspective. Cannabis Cannabinoid Res. (2018) 3:190–4. doi: 10.1089/can.2018.0030
10. Johnson R. Defining Hemp: A Fact Sheet Congressional Research Service Report R. Washington DC: Library of Congress (2019). p. 4472.
11. Kogan LR, Hellyer PW, Schoenfeld-Tacher R. Dog owners' use and perceptions of cannabis products. J Am Holist Vet Med Assoc. (2018) 51:26–33.
12. Sethi S, Brietzke E. Omics-based biomarkers: application of metabolomics in neuropsychiatric disorders. Int J Neuropsychopharmacol. (2015) 19:1–13. doi: 10.1093/ijnp/pyv096
13. Ogunade IM, Jiang Y, Adeyemi J, Oliveira A, Vyas D, Adesogan AT. Biomarker of aflatoxin ingestion: 1H NMR-based plasma metabolomics of dairy cows fed aflatoxin B1 with or without sequestering agents. Toxins. (2018) 10:545. doi: 10.3390/toxins10120545
14. Yang Y, Dong G, Wang Z, Wang J, Zhang Z, Liu J. Rumen and plasma metabolomics profiling by UHPLC-QTOF/MS revealed metabolic alterations associated with a high-corn diet in beef steers. PLoS ONE. (2018) 13:e0208031. doi: 10.1371/journal.pone.0208031
15. Adeyemi JA, Harmon DL, Paulus Compart DM, Ogunade IM. Effects of a blend of Saccharomyces cerevisiae-based direct-fed microbial and fermentation products in the diet of newly weaned beef steers: growth performance, whole-blood immune gene expression, serum biochemistry, and plasma metabolome. J Anim Sci. (2019) 97:4657–67. doi: 10.1093/jas/skz308
16. Roberts LD, Souza AL, Gerszten RE, Clish CB. Targeted metabolomics. Curr Protoc Mol Biol. (2012) 98:30.2.1–24. doi: 10.1002/0471142727.mb3002s98
17. Xia J, Broadhurst D, Wilson M, Wishart D. Translational biomarker discovery in clinical metabolomics: an introductory tutorial. Metabolomics. (2013) 9:280–99. doi: 10.1007/s11306-012-0482-9
18. Goldansaz SA, Markus S, Berjanskii M, Rout M, Guo AC, Wang Z, et al. Candidate serum metabolite biomarkers of residual feed intake and carcass merit in sheep. J Anim Sci. (2020) 98:1–15. doi: 10.1093/jas/skaa298
19. Zhao S, Li H, Han W, Chan W, Li L. Metabolomic coverage of chemical-group-submetabolome analysis: group classification and four-channel chemical isotope labeling LC-MS. Anal Chem. (2019) 91:12108–15. doi: 10.1021/acs.analchem.9b03431
20. Morris EM, Kitts-Morgan SE, Spangler DM, Gebert J, Vanzant ES, McLeod KR, et al. Feeding cannabidiol (CBD)-containing treats did not affect canine daily voluntary activity. Front Vet Sci. (2021) 8:645667. doi: 10.3389/fvets.2021.645667
21. Bartner LR, McGrath S, Rao S, Hyatt LK, Wittenburg LA. Pharmacokinetics of cannabidiol administered by 3 delivery methods at 2 different dosages to healthy dogs. Can J Vet Res. (2018) 82:178–83.
22. Deabold KA, Schwark WS, Wolf S, Wakshlag JJ. Single-dose pharmacokinetics and preliminary safety assessment with use of CBD-rich hemp neutraceutical in healthy dogs and cats. Animals. (2019) 9:832. doi: 10.3390/ani9100832
23. Gamble LJ, Boesch JM, Frye CW, Schwark WS, Mann S, Wolfe L, et al. Pharmacokinetics, safety, and clinical efficacy of cannabidiol treatment in osteoarthritic dogs. Front Vet Sci. (2018) 5:165. doi: 10.3389/fvets.2018.00165
24. Wu Y, Li L. Determination of total concentration of chemically labeled metabolites as a means of metabolome sample normalization and sample loading optimization in mass spectrometry-based metabolomics. Anal Chem. (2012) 84:10723–31. doi: 10.1021/ac3025625
25. Mung D, Li L. Development of chemical isotope labeling LC-MS for milk metabolomics: comprehensive and quantitative profiling of the Amine/Phenol submetabolome. Anal Chem. (2017) 89:4435–43. doi: 10.1021/acs.analchem.6b03737
26. Guo K, Li L. Differential 12C-/13C-isotope dansylation labeling and fast liquid chromatography/mass spectrometry for absolute and relative quantification of the metabolome. Anal Chem. (2009) 81:3919–32. doi: 10.1021/ac900166a
27. Zhao S, Luo X, Li L. Chemical isotope labeling LC-MS for high coverage and quantitative profiling of the hydroxyl submetabolome in metabolomics. Anal Chem. (2016) 88:10617–23. doi: 10.1021/acs.analchem.6b02967
28. Zhao S, Dawe M, Guo K, Li L. Development of high-performance chemical isotope labeling LC-MS for profiling the carbonyl metabolome. Anal Chem. (2017) 89:6758–65. doi: 10.1021/acs.analchem.7b01098
29. Huan T, Li L. Quantitative metabolome analysis based on chromatographic peak reconstruction in chemical isotope labeling liquid chromatography mass spectrometry. Anal Chem. (2015) 87:7011–6. doi: 10.1021/acs.analchem.5b01434
30. Li L, Li R, Zhou J, Zuniga A, Stanislaus AE, Wu Y, et al. MyCompoundID:using an evidence-based metabolome library for metabolite identification. Anal Chem. (2013) 85:3401–8. doi: 10.1021/ac400099b
31. Chong J, Wishart DS, Xia J. Using MetaboAnalyst 4.0 for comprehensive and integrative metabolomics data analysis. Curr Protocols Bioinformat. (2019) 68:e86. doi: 10.1002/cpbi.86
32. Lehmann T, Pollmann S. Gene expression and characterization of a stress-induced tyrosine decarboxylase from Arabidopsis thaliana. FEBS Lett. (2009) 583:1895–900. doi: 10.1016/j.febslet.2009.05.017
33. Miller JE, Litwack G. Purification, properties, and identity of liver mitochondrial tyrosine aminotransferase. J Biol Chem. (1971) 246:3234–40. doi: 10.1016/S0021-9258(18)62219-7
34. Wang W, Baker P, Seah SYK. Comparison of two metal-dependent pyruvate aldolases related by convergent evolution: substrate specificity, kinetic mechanism, and substrate channeling. Biochem. (2010) 49:3774–82. doi: 10.1021/bi100251u
35. Leonard W, Zhang P, Ying D, Fang Z. Tyramine-derived hydroxycinnamic acid amides in plant foods: sources, synthesis, health effects and potential applications in food industry. Crit Rev Food Sci Nutr. (2020) 1–18. doi: 10.1080/10408398.2020.1845603
36. Sato F, Inui T, Takemura T. Metabolic engineering in isoquinoline alkaloid biosynthesis. Curr Pharm Biotechnol. (2007) 8:211–8. doi: 10.2174/138920107781387438
37. Kurihara S, Oda S, Kato K, Kim HG, Koyanagi T, Kumagai H, et al. A novel putrescine utilization pathway involves γ-glutamylated intermediates of Escherichia coli K-12*. J Biol Chem. (2005) 280:4602–8. doi: 10.1074/jbc.M411114200
38. Jo J-E, Raj SM, Rathnasingh C, Selvakumar E, Jung W-C, Park S. Cloning, expression, and characterization of an aldehyde dehydrogenase from Escherichia coli K-12 that utilizes 3-Hydroxypropionaldehyde as a substrate. Appl Microbiol Biotechnol. (2008) 81:51–60. doi: 10.1007/s00253-008-1608-x
39. Igarashi K, Kashiwagi K. Cellular function by polyamines. Int J Biochem Cell Biol. (2010) 42:39–51. doi: 10.1016/j.biocel.2009.07.009
40. Larqué E, Sabater-Molina M, Zamora S. Biological significance of dietary polyamines. Nutrition. (2007) 23:87–95. doi: 10.1016/j.nut.2006.09.006
41. Darling S, Larsen PO. Saccharopine, a new amino acid in baker's and brewer's yeast: I. Isolation and properties Acta Chem Scand. (1961) 15:743–9. doi: 10.3891/acta.chem.scand.15-0743
42. Lippincott JA, Lippincott BB, Chang C-C. Promotion of crown-gall tumor growth by lysopine, octopine, nopaline, and carnosine. Plant Physiol. (1972) 49:131–7. doi: 10.1104/pp.49.2.131
43. Moore LW, Chilton WS, Canfield ML. Diversity of opines and opine-catabolizing bacteria isolated from naturally occurring crown gall tumors. Appl Environ Microbiol. (1997) 63:201–7. doi: 10.1128/aem.63.1.201-207.1997
44. Carlisle SM, Trainor PJ, Hein DW. Identification of L-asparagine as a novel endogenous substrate for human arylamine N-acetyltransferase 1. FASEB J. (2018) 31:821–8. doi: 10.1096/fasebj.31.1_supplement.821.8
45. Butcher NJ, Minchin RF. Arylamine N-acetyltransferase 1: a novel drug target in cancer development. Pharmacol Rev. (2012) 64:147–65. doi: 10.1124/pr.110.004275
46. Camporez JP, Wang Y, Faarkrog K, Chukijrungroat N, Petersen KF, Shulman GI. Mechanism by which arylamine N-acetyltransferase 1 ablation causes insulin resistance in mice. Proc Natl Acad Sci USA. (2017) 114:E11285–92. doi: 10.1073/pnas.1716990115
47. Molnár GA, Kun S, Selley E, Kertesz M, Szelig L, Csontos C, et al. Role of tyrosine isomers in acute and chronic diseases leading to oxidative stress—a review. Curr Med Chem. (2016) 23:667–85. doi: 10.2174/0929867323666160119094516
48. Molnár GA, Wagner Z, Marko L, Szegi TK, Mohas M, Kocsis B, et al. Urinary ortho-tyrosine excretion in diabetes mellitus and renal failure: evidence for hydroxyl radical production. Kidney Int. (2005) 68:2281–7. doi: 10.1111/j.1523-1755.2005.00687.x
49. Smith GK, Duch DS, Edelstein MP, Bigham EC. New inhibitors of sepiapterin reductase. Lack of an effect of intracellular tetrahydrobiopterin depletion upon in vitro proliferation of two human cell lines. J Biol Chem. (1992) 267:5599–607. doi: 10.1016/S0021-9258(18)42807-4
50. Malenka RC, Nestler EJ, Hyman SE. Chapter 6: widely projecting systems: monoamines, acetylcholine, and orexin. In: Sydor A, Brown RY, editors, Molecular Neuropharmacology: A Foundation for Clinical Neuroscience. 2nd ed. New York, NY: McGraw-Hill Medical (2009). p. 157.
51. Khomyakova M, Bukmez O, Thomas LK, Erb TJ, Berg IA. A methylaspartate cycle in haloarchaea. Science. (2011) 331:334–7. doi: 10.1126/science.1196544
52. Raj H, Poelarends GJ. The roles of active site residues in the catalytic mechanism of methylaspartate ammonia-lyase. FEBS Open Bio. (2013) 3:285–90. doi: 10.1016/j.fob.2013.07.002
53. Fons M, Cami B, Chippaux M. Possible involvement of a L-Δ1-pyrroline-5-carboxylate (P5C) reductase in the synthesis of proline in Desulfovibrio desulfuricans Norway. Biochem Biophys Res Commun. (1991) 179:1088–94. doi: 10.1016/0006-291X(91)91931-2
54. Ginguay A, Cynober L, Curis E, Nicolis I. Ornithine aminotransferase, an important glutamate-metabolizing enzyme at the crossroads of multiple metabolic pathways. Biology. (2017) 6:18. doi: 10.3390/biology6010018
55. Brown DD, Kies MW. The mammalian metabolism of L-histidine. I The enzymatic formation of L-hydantion-5-propionic acid. J Biol Chem. (1959) 234:3182–7. doi: 10.1016/S0021-9258(18)69645-0
56. Hassall H, Greenberg DM. Studies on the enzymic decomposition of urocanic acid. V. The formation of 4-oxoglutaramic acid, a non-enzymic oxidation product of 4-imidazolone-5-propionic acid. J Biol Chem. (1963) 238:1423–31. doi: 10.1016/S0021-9258(18)81200-5
57. Naka K, Jomen Y, Ishihara K, Kim J, Ishimoto T, Bae E-J, et al. Dipeptide species regulate p38MAPK–Smad3 signaling to maintain chronic myelogenous leukaemia stem cells. Nat Commun. (2015) 6:8039. doi: 10.1038/ncomms9039
58. Nakato J, Aoki H, Tokuyama Y, Yamamoto Y, Iwakura H, Matsumura S, et al. Comprehensive analysis of a dipeptide library to identify ghrelin release-modulating peptides. FEBS Lett. (2019) 593:2637–45. doi: 10.1002/1873-3468.13522
59. Bielawiec P, Harasim-Symbor E, Chabowski A. Phytocannabinoids: useful drugs for the treatment of obesity? special focus on cannabidiol. Front Endocrinol. (2020) 11:114. doi: 10.3389/fendo.2020.00114
60. Jadoon KA, Ratcliffe SH, Barrett DA, Thomas EL, Stott C, Bell JD, et al. Efficacy and safety of cannabidiol and tetrahydrocannabivarin on glycemic and lipid parameters in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled, parallel group pilot study. Diabetes Care. (2016) 39:1777–86. doi: 10.2337/dc16-0650
61. Zorzenon MRT, Santiago AN, Mori MA, Piovan S, Jansen CA, Padilha MEP, et al. Cannabidiol improves metabolic dysfunction in middle-aged diabetic rats submitted to a chronic cerebral hypoperfusion. Chem Biol Interact. (2019) 312:108819. doi: 10.1016/j.cbi.2019.108819
62. Anderson JW, Nicolosi RJ, Borzelleca JF. Glucosamine effects in humans: a review of effects on glucose metabolism, side effects, safety considerations and efficacy. Food Chem Toxicol. (2005) 43:187–201. doi: 10.1016/j.fct.2004.11.006
63. Beale BS. Use of nutraceuticals and chondroprotectants in osteoarthritic dogs and cats. Vet Clin North Am Small Anim Pract. (2004) 34:271–89. doi: 10.1016/j.cvsm.2003.09.008
64. Bhathal A, Spryszak M, Louizos C, Frankel G. Glucosamine and chondroitin use in canines for osteoarthritis: a review. Open Vet J. (2017) 7:36–49. doi: 10.4314/ovj.v7i1.6
65. Henrotin Y, Mobasheri A, Marty M. Is there any scientific evidence for the use of glucosamine in the management of human osteoarthritis? Arthritis Res. Ther. (2012) 14:201. doi: 10.1186/ar3657
66. Kudo F, Yamamoto Y, Yokoyama K, Eguchi T, Kakinuma K. Biosynthesis of 2-deoxystreptamine by three crucial enzymes in Streptomyces fradiae NBRC 12773. J Antibiot. (2005) 58:766–74. doi: 10.1038/ja.2005.104
67. Park JW, Park SR, Nepal KK, Han AR, Ban YH, Yoo YJ, et al. Discovery of parallel pathways of kanamycin biosynthesis allows antibiotic manipulation. Nat Chem Biol. (2011) 7:843–52. doi: 10.1038/nchembio.671
68. Bansal P, Wang S, Liu S, Xiang Y-Y, Lu W-Y, et al. GABA coordinates with insulin in regulating secretory function in pancreatic INS-1 β-cells. PLoS ONE. (2011) 6:e26225. doi: 10.1371/journal.pone.0026225
69. Franklin IK, Wollheim CB. GABA in the endocrine pancreas: its putative role as an islet cell paracrine-signalling molecule. J Gen Physiol. (2004) 123:185–90. doi: 10.1085/jgp.200409016
70. Purwana I, Zheng J, Li X, Deurloo M, Son DO, Zhang Z, et al. GABA promotes human b-cell proliferation and modulates glucose homeostasis. Diabetes. (2014) 63:4197–205. doi: 10.2337/db14-0153
71. Rorsman P, Berggren P-O, Bokvist K, Ericson H, Mohler H, Ostenson C-G, et al. Glucose-inhibition of glucagon secretion involves activation of GABA[[SB]]A[[/S]]-receptor chloride channels. Nature. (1989) 341:233–6. doi: 10.1038/341233a0
72. Wan Y, Wang Q, Prud'homme GJ. GABAergic system in the endocrine pancreas: a new target for diabetes treatment. Diabetes Metab Syndr Obes. (2015) 8:79–87. doi: 10.2147/DMSO.S50642
73. Bhandage AK, Jin Z, Korol SV, Shen Q, Pei Y, Deng S, et al. GABA regulates release of inflammatory cytokines from peripheral blood mononuclear cells and CD4+ T Cells and is immunosuppressive in type 1 diabetes. EBioMedicine. (2018) 30:283–94. doi: 10.1016/j.ebiom.2018.03.019
74. Jin Z, Mendu SK, Birnir B. GABA is an effective immunomodulatory molecule. Amino Acids. (2013) 45:87–94. doi: 10.1007/s00726-011-1193-7
75. Prud'homme G, Glinka Y, Wang Q. GABA exerts anti-inflammatory and immunosuppressive effects. J Immunol. (2013) 190(1 Suppl.):68.15.
76. Hildebrandt T, Knuesting J, Berndt C, Morgan B, Scheibe R. Cytosolic thiol switches regulating basic cellular functions: GAPDH as an information hub? Biol. Chem. (2015) 396:523–37. doi: 10.1515/hsz-2014-0295
77. White MR, Garcin ED. D-Glyceraldehyde-3-phosphate dehydrogenase structure and function. Subcell Biochem. (2017) 83:413–53. doi: 10.1007/978-3-319-46503-6_15
78. Zimniewska M, Rozańska W, Gryszczynska A, Romanowska B, Kicinska-Jakuowska A. Antioxidant potential of hemp and flax fibers depending on their chemical composition. Molecules. (2018) 23:1993. doi: 10.3390/molecules23081993
79. Pojić M, Mišan A, Sakač M, Hadnadev TD, Šarić B, Milovanović I, et al. Characterization of byproducts originating from hemp oil processing. J Agric Food Chem. (2014) 62:12436–42. doi: 10.1021/jf5044426
80. Meeprom A, Sompong W, Chan CB, Adisakwattana S. Isoferulic acid, a new anti-glycation agent, inhibits fructoseand glucose-mediated protein glycation in vitro. Molecules. (2013) 18:6439–54. doi: 10.3390/molecules18066439
81. Zhao Z, Moghadasian MH. Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: a review. Food Chem. (2008) 109:691–702. doi: 10.1016/j.foodchem.2008.02.039
82. Liu I-M, Hsu F-L, Chen C-F, Cheng J-T. Antihyperglycemic action of isoferulic acid in streptozotocin-induced diabetic rats. Br J Pharmacol. (2000) 129:631–6. doi: 10.1038/sj.bjp.0703082
83. Hirabayashi T, Ochiai H, Sakai S, Nakajima K, Terasawa K. Inhibitory effect of ferulic acid and isoferulic acid on murine interleukin-8 production in response to influenza virus infections in vitro and in vivo. Planta Med. (1994) 61:221–6. doi: 10.1055/s-2006-958060
84. Meeprom A, Sompong W, Suantawee T, Thilavech T, Chan CB, Adisakwattana S. Isoferulic acid prevents methylglyoxalinduced protein glycation and DNA damage by free radical scavenging activity. BMC Complement Altern Med. (2015) 15:346. doi: 10.1186/s12906-015-0874-2
85. Arfin S, Siddiqui GA, Naeem A, Moin S. Inhibition of advanced glycation end products by isoferulic acid and its free radical scavenging capacity: an in vitro andmolecular docking study. Int J Biol Macromol. (2018) 118:1479–87. doi: 10.1016/j.ijbiomac.2018.06.182
86. Clifford MN, Kerimi A, Williamson G. Bioavailability and metabolism of chlorogenic acids (acyl-quinic acids) in humans. Compr Rev Food Sci Food Saf. (2019) 19:1299–352. doi: 10.1111/1541-4337.12518
88. Santos PMP, Vieira AJSC. Antioxidising activity of cinnamic acid derivatives against oxidative stress induced by oxidising radicals. J Phys Org Chem. (2013) 26:432–9. doi: 10.1002/poc.3104
89. Civolani C, Barghini P, Roncetti AR, Ruzzi M, Schiesser A. Bioconversion of ferulic acid into vanillic acid by means of a vanillate-negative mutant of Pseudomonas fluorescens strain BF13. Appl Environ Microbiol. (2000) 66:2311–7. doi: 10.1128/AEM.66.6.2311-2317.2000
90. Khan BA, Warner P, Wang H. Antibacterial properties of hemp and other natural fibre plants: a review. Bioresources. (2014) 9:3642–59. doi: 10.15376/biores.9.2.3642-3659
91. Yemis GP, Pagotto F, Bach S, Delaquis P. Effect of vanillin, ethyl vanillin, and vanillic acid on the growth and heat resistance of Cronobacter species. J Food Prot. (2011) 74:2062–9. doi: 10.4315/0362-028X.JFP-11-230
92. El-Kalyoubi SA, Fayed EA, Abdel-Razek AS. One pot synthesis, antimicrobial and antioxidant activities of fused uracils: pyrimidodiazepines, lumazines, triazolouracil and xanthines. Chem Cent J. (2017) 11:66. doi: 10.1186/s13065-017-0302-4
93. Sahr T, Ravanel S, Basset G, Nichols BP, Hanson AD, Rebeille F. Folate synthesis in plants: purification, kinetic properties, and inhibition of aminodeoxychorismate synthase. Biochem J. (2006) 396:157–62. doi: 10.1042/BJ20051851
94. Wegkamp A, van Oorschot W, de Vos WM, Smid EJ. Characterization of the role of para-aminobenzoic acid biosynthesis in folate production by Lactococcus lactis. Appl Environ Microbiol. (2007) 73:2673–81. doi: 10.1128/AEM.02174-06
95. McCarty RM, Somogyi A, Lin G, Jacobsen NE, Bandarian V. The deazapurine biosynthetic pathway revealed: in vitro enzymatic synthesis of PreQ(0) from guanosine 5′-triphosphate in four steps. Biochem. (2009) 48:3847–52. doi: 10.1021/bi900400e
96. Kress D, Alhapel A, Pierik AJ, Essen L-O. The crystal structure of enamidase: a bifunctional enzyme of the nicotinate catabolism. J Mol Biol. (2008) 384:837–47. doi: 10.1016/j.jmb.2008.09.002
97. Alhapel A, Darley DJ, Wagener N, Eckel E, Elsner E, Pierik AJ. (2006). Molecular and functional analysis of nicotinate catabolism in Eubacterium barkeri. Proc. Natl. Acad. Sci. USA. 103:12341–12346. doi: 10.1073/pnas.0601635103
98. Linster CL, Schaftingen EV. Vitamin C. Biosynthesis, recycling and degradation in mammals. FEBS J. (2007) 274:1–22. doi: 10.1111/j.1742-4658.2006.05607
99. Meister A. Glutathione-ascorbic acid antioxidant system in animals. J Biol Chem. (1994) 269:9397–400. doi: 10.1016/S0021-9258(17)36891-6
100. Yokochi N, Yoshikane Y, Matsumoto S, Fujisawa M, Ohnishi K, Yagi T. Gene identification and characterization of 5-formyl-3-hydroxy-2-methylpyridine 4-carboxylic acid 5-dehydrogenase, an NAD+-dependent dismutase. J Biochem. (2009) 145:493–503. doi: 10.1093/jb/mvp007
101. Dalto DB, Matte J-J. Pyridoxine (Vitamin B6) and the glutathione peroxidase system; a link between one-carbon metabolism and antioxidation. Nutrients. (2017) 9:189. doi: 10.3390/nu9030189
102. Knapik AA, Petkowski JJ, Otwinowski Z, Cymborowski MT, Cooper DR, Chruszcz M, et al. Structure of Escherichia coli RutC, a member of the YjgF family and putative aminoacrylate peracid reductase of the rut operon. Acta Crystallogr Sect F Struct Biol Cryst Commun. (2012) 68:1294–9. doi: 10.1107/S1744309112041796
103. Brioschi FA, Di Cesare F, Gioeni D, Rabbogliatti V, Ferrari F, D'Urso ES, et al. Oral transmucosal cannabidiol oil formulation as part of a multimodal analgesic regimen: effects on pain relief and quality of life improvement in dogs affected by spontaneous osteoarthritis. Animals. (2020) 10:1505. doi: 10.3390/ani10091505
104. Verrico CD, Wesson S, Konduri V, Hofferek CJ, Vazquez-Perez J, Blair E, et al. A randomized, double-blinded, placebo-controlled study of daily cannabidiol for the treatment of canine osteoarthritis pain. Pain. (2020) 161:2191–202. doi: 10.1097/j.pain.0000000000001896
105. Morris EM, Kitts-Morgan SE, Spangler DM, McLeod KR, Costa JHC, Harmon DL. Feeding cannabidiol (CBD)-containing treats did not affect canine daily voluntary activity. Front Vet Sci. (2020) 7:569565. doi: 10.3389/fvets.2020.569565
106. McGrath S, Bartner LR, Rao S, Packer RA, Gustafson DL. Randomized blinded controlled clinical trial to assess the effect of oral cannabidiol administration in addition to conventional antiepileptic treatment on seizure frequency in dogs with intractable idiopathic epilepsy. J Am Vet Med Assoc. (2019) 254:1301–8. doi: 10.2460/javma.254.11.1301
107. Wakshlag JJ, Schwark WS, Deabold KA, Talsma BN, Cital S, Lyubimov A, et al. Pharmacokinetics of cannabidiol, cannabidiolic acid, Δ9-tetrahydrocannabinol, tetrahydrocannabinolic acid and related metabolites in canine serum after dosing with three oral forms of hemp extract. Front Vet Sci. (2020) 7:505. doi: 10.3389/fvets.2020.00505
108. Chicoine A, Illing K, Vuong S, Pinto KR, Alcorn J, Cosford K. Pharmacokinetic and safety evaluation of various oral doses of a novel 1:20 THC:CBD cannabis herbal extract in dogs. Front Vet Sci. (2020) 7:583404. doi: 10.3389/fvets.2020.583404
Keywords: cannabidiol, canine, metabolomics, biomarkers, metabolites
Citation: Morris EM, Kitts-Morgan SE, Spangler DM, Ogunade IM, McLeod KR and Harmon DL (2021) Alteration of the Canine Metabolome After a 3-Week Supplementation of Cannabidiol (CBD) Containing Treats: An Exploratory Study of Healthy Animals. Front. Vet. Sci. 8:685606. doi: 10.3389/fvets.2021.685606
Received: 25 March 2021; Accepted: 18 June 2021;
Published: 16 July 2021.
Edited by:
Anna Katharine Shoveller, University of Guelph, CanadaCopyright © 2021 Morris, Kitts-Morgan, Spangler, Ogunade, McLeod and Harmon. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: David L. Harmon, ZGF2aWQuaGFybW9uQHVreS5lZHU=
 Elizabeth M. Morris
Elizabeth M. Morris Susanna E. Kitts-Morgan
Susanna E. Kitts-Morgan Dawn M. Spangler2
Dawn M. Spangler2 Ibukun M. Ogunade
Ibukun M. Ogunade Kyle R. McLeod
Kyle R. McLeod David L. Harmon
David L. Harmon