- 1School of Nursing, University of Ottawa, Ottawa, ON, Canada
- 2Ottawa Hospital Research Institute, The Ottawa Hospital, Ottawa, ON, Canada
- 3Division of General and Community Pediatrics, Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, United States
- 4Department of Pediatrics, University of Cincinnati College of Medicine, Cincinnati, OH, United States
- 5Susan B. Meister Child Health Evaluation & Research (CHEAR) Center, University of Michigan, Ann Arbor, MI, United States
- 6Department of Learning Health Sciences, University of Michigan, Ann Arbor, MI, United States
- 7Patient/Parent/Caregiver Partners, Ann Arbor, MI, United States
- 8Department of Pediatrics, Donald and Barbara Zucker School of Medicine at Hofstra/Northwell, Hempstead, NY, United States
- 9Institute of Molecular Medicine, Feinstein Institute for Medical Research, Manhasset, NY, United States
- 10Division of Pediatric Psychiatry and Behavioral Health, Department of Pediatrics, University of Utah Spencer Fox Eccles School of Medicine, Salt Lake City, UT, United States
- 11Division of Urology, Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, United States
- 12Department of Pediatrics, Division of Pediatric Psychology, University of Michigan Medical School, Ann Arbor, MI, United States
Introduction: Parents and guardians of infants and young children with differences of sex development (DSD) often face numerous health and social decisions about their child’s condition. While proxy health decisions can be stressful in any circumstance, they are further exacerbated in this clinical context by significant variations in clinical presentation, parental lack of knowledge about DSD, irreversibility of some options (e.g., gonadectomy), a paucity of research available about long-term outcomes, and anticipated decisional regret. This study aimed to engage clinicians, parents, and an adult living with DSD to collaboratively co-design a suite of patient decision aids (PDAs) to respond to the decisional needs of parents and guardians of infants and young children diagnosed with DSD.
Methods: We used a systematic co-design process guided by the Ottawa Decision Support Framework and the International Patient Decision Aids Standards (IPDAS). The five steps were: literature selection, establish the team, decisional needs assessment, create the PDAs, and alpha testing.
Results: Our team of health professionals, parents, adult living with DSD and PDA experts, co-designed four PDAs to support parents/guardians of infants or young children diagnosed with DSD. These PDAs addressed four priority decisions identified through our decisional needs assessment: genetic testing, gender of rearing, genital surgery and gonadal surgery. All four PDAs include information for parents about DSD, the options, reasons to choose or avoid each option, and opportunities for parents/guardians to rate the importance of features of each option to clarify their values for these features. Qualitative feedback was positive from our team and when alpha tested with an interprofessional DSD speciality team in a single center.
Conclusions: These PDAs are clinical tools designed to support parents/guardians to be involved in making informed DSD decisions; next steps are to determine parents’ decisional outcomes. While these tools are specific to DSD, the process through which they were co-designed is transferable to co-design of PDAs in other pediatric populations.
1 Introduction
Differences of sex development (DSD), also called ‘disorders of sex development’, ‘intersex traits’, or “developmental variations of sex characteristics refers to a multitude of congenital conditions in which development of chromosomal, gonadal, and/or anatomical sex is atypical (1, 2). Within this definition, DSDs can have a wide range of gonadal phenotypes, such as partial or complete gonadal dysgenesis and ovotestis, and external genital phenotypes, such as hypospadias, clitoromegaly and ambiguous genitalia or fully masculinized or feminized genitalia that are discordant with karyotype or gonadal phenotypes. Using this inclusive definition, DSDs are estimated to occur in approximately one in 100 live births (2, 3). Parents and guardians of infants and young children newly diagnosed with DSD are often presented with numerous proxy decisions to make related to their child’s condition. Young children refers to those who are too young to be expected to meaningfully participate in the process of decision making. Although a previous legal case in the USA used the “rule of sevens” that children less than 7 years of age do not have decision making capacity (4), a study with parents and children, having experience living with diabetes, involved children as young as six years of age in the decision about changing insulin delivery (5).
Since the 1950s, it has been standard practice to perform ‘normalizing’ genital surgery in infancy based on the perception that this would lead to improved psychosocial and surgical outcomes (6). Recently, however, there has been growing controversy over early elective genital and gonadal surgeries, resulting in some medical centers ceasing to perform them and several Human Rights Organizations denouncing them as human rights violations (1, 7–9). While research into the long-term outcomes of these surgical decisions is lacking, there have been reports of decision regret in both parents and their grown children related to surgical decisions made in infancy. In a systematic review of five studies (N=783), 20% of parents who consented to hypospadias surgery for their young child reported moderate to severe decision regret and 45% reported mild decision regret (10). One modifiable decisional need associated with higher decision regret was pre-operative parental decisional conflict (10). Perspectives of parents who did not consent to this surgery were not formally assessed (10). Debate has also emerged amongst and between clinicians, advocates, patients, and families surrounding Western standards of care related to gender of rearing and the extent of investigations and treatment required (1).
Parents and guardians of an infant or young child with DSD require support to address their decisional needs. In a systematic review of 140 studies about decisional needs of parents facing a range of healthcare decisions, three key decisional needs were identified (11). First parents identified ‘needing information’ from different sources delivered in a way that facilitated its use. Parents also wanted to ‘talk with others’, including other parents; although they reported that these discussions sometimes resulted in their feeling pressured to make decisions based on the other parents’ opinions. Third, parents wanted ‘control over the process’ for these emotionally charged decisions and control in the consultation, without structural barriers interfering with their participation in making decisions. Patient decision aids (PDAs) are clinical tools that can address decisional needs and facilitate shared decision making (SDM) (12, 13). Particularly in clinical situations where multiple management options exist, studies have demonstrated that PDAs support high-quality decisions by presenting the evidence on options (including benefits and harms) in a balanced format and helping patients or proxies to clarify their values for features of options so that they can actively participate in SDM with their clinician(s). PDAs are different from parent information materials because they focus on a specific decision and provide information in a structured format that can prepare parents for making shared decisions with the clinician. However, few studies have evaluated PDAs in pediatrics. In one study, a PDA was designed to capture both the perspectives of the child with diabetes and the parent independently so that children as young as seven years old could participate in making the decision about selecting an insulin delivery method (5, 14). Parents and children who received decision coaching using this PDA were satisfied with the intervention and had lower decisional conflict (5).
Parents and guardians of infants with a DSD, however, are tasked with making decisions about their child’s future without knowing their child’s values or opinions and within a rapidly changing clinical context (8, 15). While proxy health decisions can be stressful in any circumstance, they are further exacerbated in DSD by significant variations in clinical presentation, lack of parents’/guardians’ knowledge about these conditions, the irreversibility of some of the options (e.g., genitoplasty, phalloplasty, gonadectomy), a paucity of high quality research available about long-term outcomes, anticipated decision regret, and pressure from external sources. For example, there is tension in DSD decision making between a human rights approach (i.e., the right to bodily autonomy) (16) and the responsibility and right of parents to make decisions that they believe to be in the best interest of their child and family. In 2019, the American Medical Association (17) maintained the status quo of parental rights. As such, an enormous responsibility is placed on parents to make complex decisions without standardized and evidence-based decisional support resources.
Currently, there is one PDA available for parents of a child born with hypospadias who are considering surgery; most of whom would not be considered to have DSD (18, 19). Parents described the PDA as easy to understand and thought it would help prepare them for decision making (19). Given the parents’ and guardians’ decisional needs and PDAs limited to one on hypospadias repair, there is a need to create a series of PDAs to respond to the decisional needs of parents and guardians of infants with DSD. The aim of this current manuscript is to outline and discuss the methods we employed to co-design a suite of PDAs, as guided by the Ottawa Decision Support Framework and International Patient Decision Aids Standards, so that it may serve as a methodological guide for others.
2 Methods
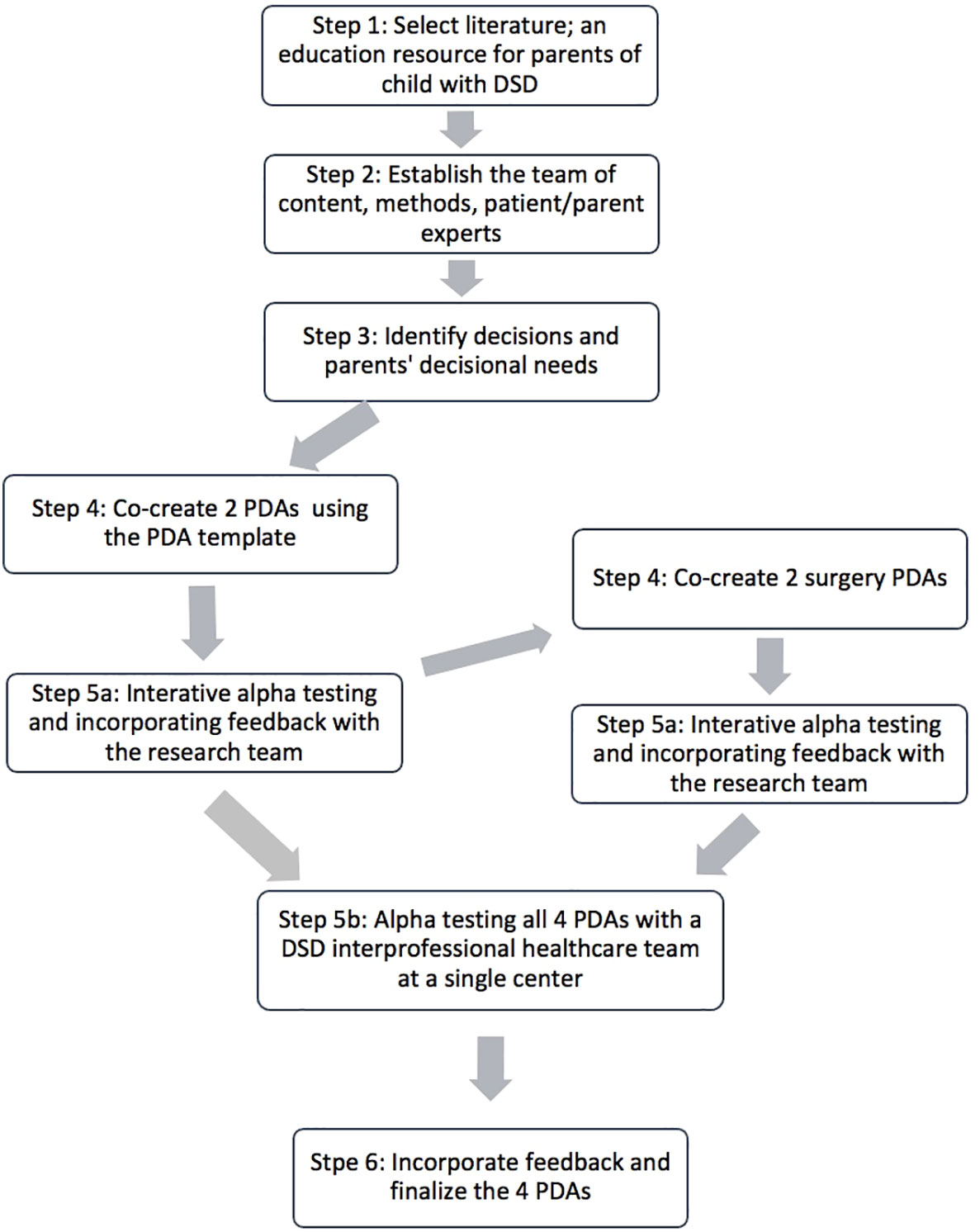
We used a systematic co-design process guided by the Ottawa Decision Support Framework and the International Patient Decision Aids Standards (IPDAS) (20–22). The Ottawa Decision Support Framework hypothesizes that decision support interventions such as PDAs that address patient/families’ decisional needs will lead to quality decisions that are implemented (22). According to IPDAS (21), creating a quality PDA requires the following steps: literature selection, establish the team, decisional needs assessment, and create the PDAs with iterative alpha testing (see Figure 1). The project was submitted to an Institutional Review Board (IRB) at Michigan Medical School and deemed exempt from IRB oversight due to the targeted sample (HCPs) and absence of collecting protected personal health information.
2.1 Step 1: Select literature
Prior to this study, content expert members of our team conducted a thorough literature review to create educational resources for parents/guardians of children with DSD (23). Findings were used to create a comprehensive 185 page evidence-based education resource which was made freely available online. As the information was not presented in a structured way to guide decision making, clinicians were not using it to discuss decisions in clinical practice (24, 25). This education resource served as both the impetus and the evidence-based content for the PDAs.
2.2 Step 2: Establish the team
We established our research team with four pediatric clinicians (psychologist, general pediatrician, and pediatric endocrinologist and urologist), two parents of children with DSD, an adult living with DSD, and experts in PDA design, including a graduate student. The research team was invited to a virtual introductory meeting to describe the project and to discuss their experiences with DSD decisions. Previous research has shown that when knowledge users are involved on the research team, they are more likely to consider the findings relevant and to use them (20, 26). Our research team was responsible for the subsequent steps in co-creating the suite of PDAs.
2.3 Step 3: Identify decisions and parents’ decisional needs
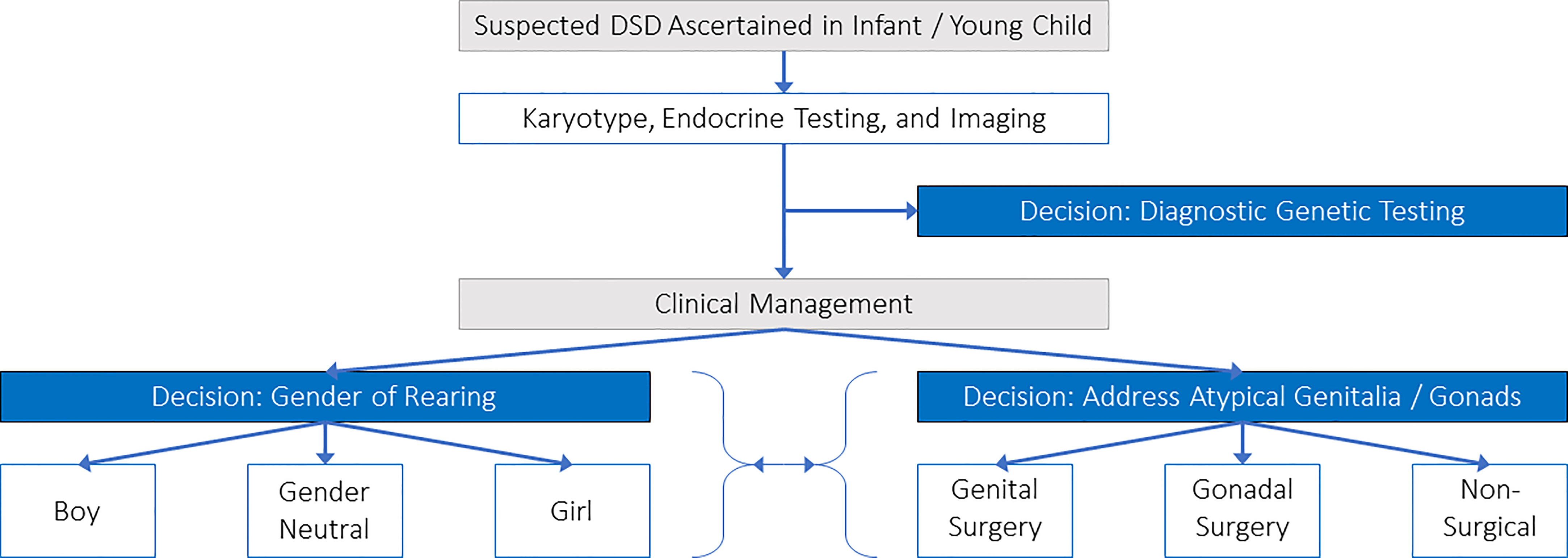
Our research team prepared and sent a survey to clinicians providing care to children with DSD to identify areas of greatest need for decision support (27). The survey was based on the Ottawa Decision Support Framework (22) and tested by members of the research team. We reviewed the results for the first round of respondents (28 clinicians) and identified three common difficult decisions that were relevant across a broad number of DSD conditions: 1) genetic testing, 2) gender of rearing, and 3) surgery (see Figure 2). Through team discussions about differences in the characteristics of surgeries, we identified two main types of surgical decisions: 3a) genital surgery and 3b) gonadal surgery. Other decisions included whether or not to have the child’s internal anatomy examined under anesthesia, share information about DSD with family/friends or their child, using medications, obtaining mental health services, and attending support groups. These decisions were not prioritized for the initial suite of PDAs because they were either too condition-specific, had limited harms, and/or were identified as ‘non-decisions’ because they were necessary for effective care (e.g., hormone replacement for a child with congenital adrenal hyperplasia).
Most respondents thought it was very important to have information on benefits/harms of all available options (89%) and access to an interprofessional healthcare team to discuss options with families/guardians (86%). Conversely, only half (54%) thought it was important to know what features of options were important to parents/guardians, and 39% wanted to have access to PDAs reflecting existing barriers to SDM. These identified decisional needs were consistent with a systematic review of 45 other decisional needs assessments based on the Ottawa Decision Support Framework (28). The research team discussed the results of the decisional needs assessment and agreed to create a suite of PDAs for the four identified difficult decisions.
2.4 Step 4: Co-create the PDAs using a PDA template
We drafted four PDAs using a two-phased process. For the first set of PDAs, on genetic testing and gender of rearing, the PDAs were initially drafted by the PDA experts on the research team using a previously well-tested standard template based on the Ottawa Decision Support Framework (22). This template provides a step-by-step way to make the decision and has been proven to improve knowledge, reduce decisional conflict and increase participation in decision making in 24 randomized controlled trials of PDAs for other decisions (29, 30). This template includes space to add the health condition, decision (stated explicitly), target audience, options, benefits, and harms; all of which are IPDAS criteria required to be defined as a PDA (31, 32). Then, it provides an interactive exercise for clarifying parents’/guardians’ values related to the outcomes of the potential options, a knowledge test to verify understanding, and the SURE test (Sure of myself; Understand information; Risk-benefit ratio; Encouragement) to screen for remaining decisional needs (33). The template also has prompts to meet the six criteria for minimizing risk of making a biased decision, which are: presenting information using equal detail, including citations for evidence, publication date, update policy, funding source, uncertainty around outcome probabilities (31). Evidence from the literature review was used to populate the PDA template. This process was repeated for the two surgical PDAs.
2.5 Step 5: Alpha testing
With our research team, we conducted alpha-testing of the first two PDAs using virtual meetings and individual written feedback. Alpha-testing aims to determine the comprehensibility, usability, and acceptability of a PDA (21). The PDAs were iteratively revised after each of the three meetings. The two surgical PDAs were reviewed in a virtual meeting with our research team. We then met virtually with a DSD interprofessional healthcare team consisting of clinicians specializing in bioethics, genetic counselling, endocrinology, urology, and pediatrics at a large pediatric hospital to get feedback on all four PDAs. The PDAs were circulated prior to each meeting and summarized during the meeting with reference to key elements necessary to meet the IPDAS criteria (31). Based on these multiple, iterative rounds of feedback, four PDAs were finalized. Plans are in development for beta-testing the PDAs in the DSD clinic with parents/guardians and clinicians discussing the decisions. The University of Michigan Medical School Institutional Review Board reviewed the study and determined that it was exempt from ongoing review per federal exemption guidelines.
3 Results
3.1 Patient decision aids
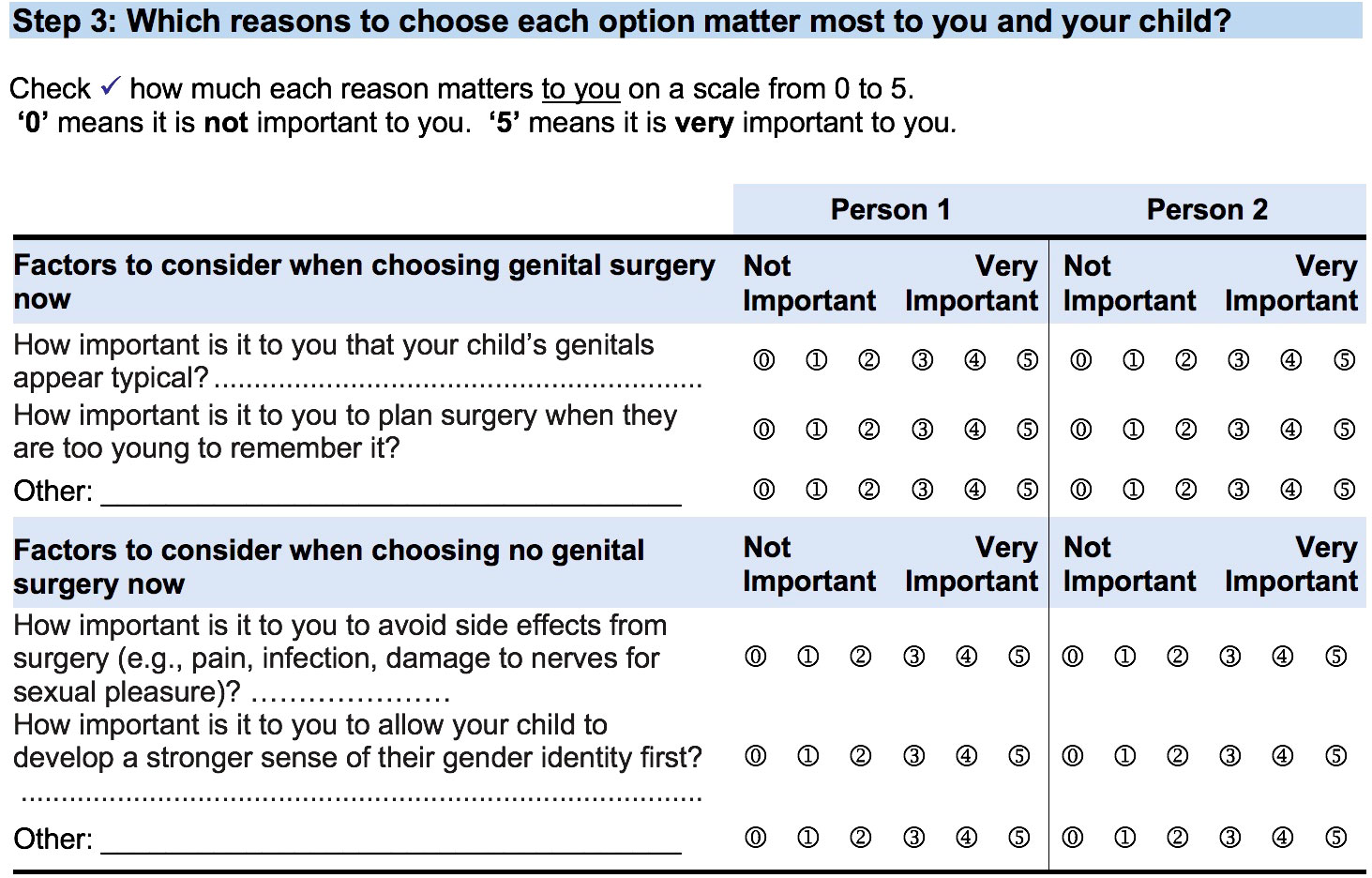
PDAs were created for four specific, difficult decisions faced by our target audience: parents/guardians of an infant or young child with DSD. Information on the condition, options, benefits and harms was presented in a format that made it possible to compare the positive and negative features of available options and efforts were made to transparently link information to the evidence sources. However, there is limited empirical evidence and the Western standard of DSD management has been based on expert opinion or consensus and with attention to ethical considerations. The PDAs also provide an interactive exercise to clarify parents’/guardians’ values for features of options by using a 0 to 5 scale where ‘0’ means it is not important and ‘5’ means it is very important (see Figure 3). The genetic testing PDA uses a similar exercise where users rate each of the pros and cons using stars to show how much each one matters them on a scale from ‘0’ stars which means not at all to ‘5’ stars which means a great deal. All research team members were identified as authors with their credentials. The following outlines specific details about each of the four PDAs.
3.1.1 Genetic testing
The PDA presented two options: having genetic testing (beyond karyotype) or not having genetic testing. We provided information on the different types of genetic tests available (e.g., chromosomal microarrays, single gene or panel testing, targeted gene testing, whole exome to genome sequencing). Although there is a move to include costs in PDAs, this information was not publicly available and appeared to be quite variable depending on the testing facility. Benefits included potential to guide healthcare management and provide insights about long-term health outcomes. Potential harms included risk for discrimination (e.g., life insurance, romantic partnership, competitive sports) and discovering information that is not helpful, including future unrelated illnesses and previously unknown paternity. The final reading level was Flesch-Kincaid 7.6 (i.e., between 5th and 6th grade in a US school). The genetic testing PDA met all IPDAS criteria for being defined as a PDA and five of six criteria for minimizing risk of bias. The sixth item, describing uncertainty around the probabilities, was not applicable.
3.1.2 Gender of rearing
The PDA presented two options: raising the child as a binary gender (girl, boy) or gender-neutrally. Information regarding current understandings of sex and gender identity were included to orient the parents/guardians. While the impact of gender of rearing on physical and mental health is not well understood, general DSD-specific trends related to future gender identity were outlined. Furthermore, parents/guardians were invited to reflect upon their values surrounding existing gender norms as they related to their family, culture, religion, and community. The final reading level was Flesch-Kincaid 5.8. The gender of rearing PDA met all IPDAS criteria for being defined as a PDA and five of six criteria for minimizing risk of bias. The sixth item, describing uncertainty around the probabilities, was not applicable.
3.1.3 Genital surgery
The PDA presented two options: genital surgery as a baby/young child or no genital surgery now and may revisit the decision with my child later. We provided information on genitals, genital surgery, and emerging concerns over the risk to the cognitive development of children caused by general anesthesia in early life (34). A nuanced discussion about common parental concerns (e.g., being teased for atypical genitals, being upset as an older child or adult about the decisions made without their involvement, anesthetic risks) was organized into advantages (benefits) and disadvantages (harms) in an effort to provide the specific and balanced information parents/guardians wanted, while acknowledging the complexity of this values-laden decision. The final reading level was Flesch-Kincaid 9.2. The genital surgery PDA met all IPDAS criteria for being defined as a PDA and five of six criteria for minimizing risk of bias. The sixth item, describing uncertainty around the probabilities, was not applicable.
3.1.4 Gonadal surgery
The PDA described two options: gonadal surgery as a baby/young child or no gonadal surgery now and may revisit the decision with my child later. We provided information on gonads, gonadal surgery, and general anesthesia. Similar to the genital surgery PDA, we reported common parental/guardian questions and concerns (e.g., relative risk of cancer if not removed, infertility and future need of hormone replacement therapy if removed, anesthesia risks) which we discussed pragmatically and organized into benefits and harms. The final reading level was Flesch-Kincaid 8.5. The gonadal PDA met all IPDAS criteria for being defined as a PDA and for minimizing risk of bias.
3.2 Feedback findings
The research and clinical teams were generally supportive of the PDAs and although they were willing to use them, they described being unsure of how, when, and by whom to introduce them within the decision-making process. The parents and adult diagnosed with DSD on the research team were also supportive of the PDAs and reported that they would have found them relevant and useful when they were facing DSD-related decisions. The combination of insider (content expertise and lived experience) and outsider (methodological expertise) group members contributed to the rigor of this work. Because the standard of care had been largely established based on expert consensus, the PDA experts challenged clinicians to specify the evidence of some cited claims such as, infants recover better from early surgery than older children/adults, and that early surgery is preferable because the child will have no memory of it. We cited empirical evidence when it was available. Where expert consensus existed but there was significant uncertainty, such as the psychosocial impact of growing up with atypical genitalia on mental health and family dynamics, we were transparent about the limited evidence.
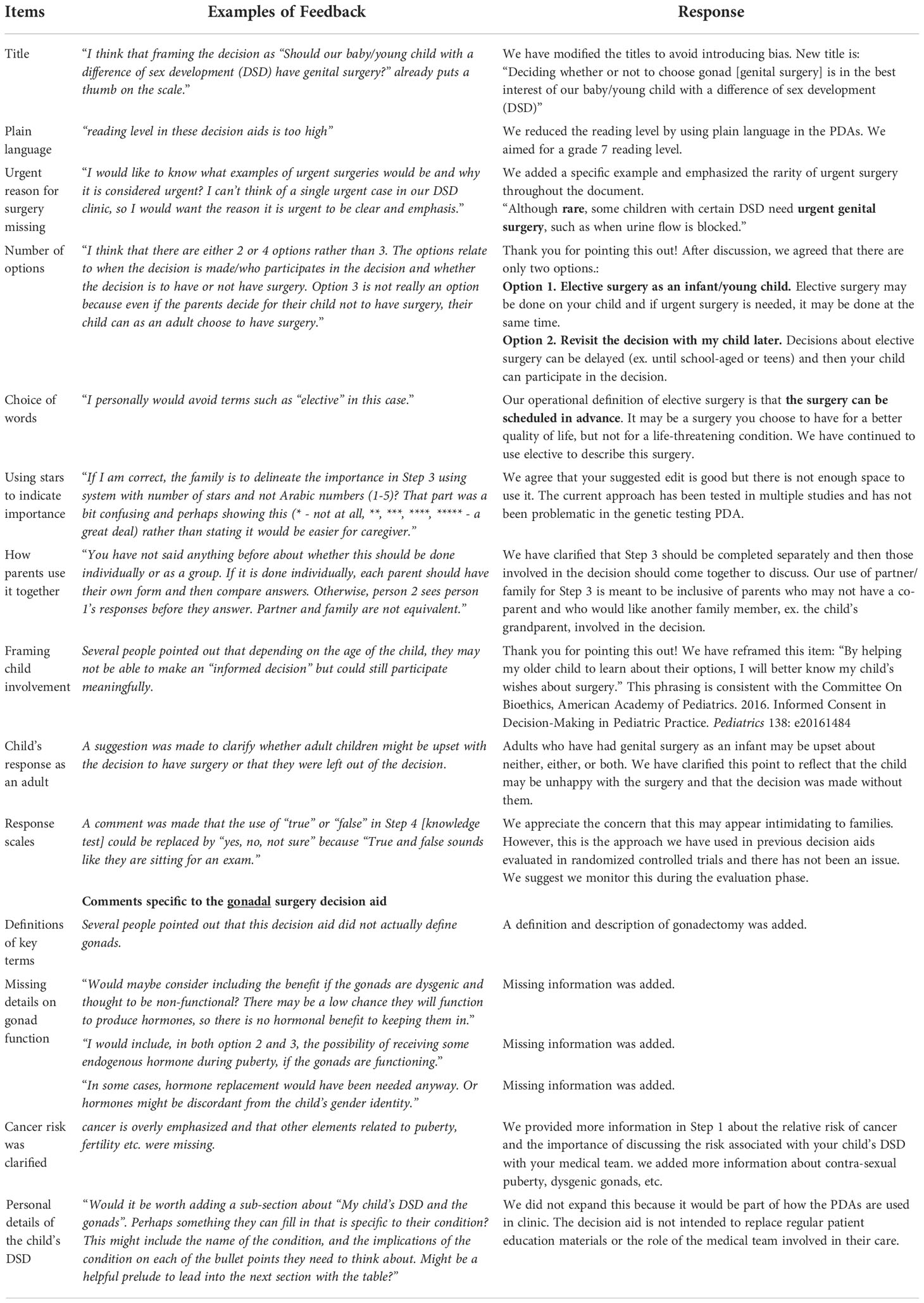
3.2.1 Changes made
We incorporated findings iteratively during the alpha-testing to enhance clarity of the information presented (see Table 1). Across PDAs, committee members identified that the language level was too high but there were challenges simplifying the language given the complex nature of the descriptions of these conditions and options. Through discussion, we reached consensus on how to simplify some of the language. Ultimately, we were unable to meet our goal of a seventh grade reading level for two out of the four PDAs due to the medical terms included. We agreed that it was important to use the same medical terms that would be used by DSD clinical teams, and we provided a brief description of them in the PDA.
The original working PDA titles were criticized as introducing bias and we rephrased them to reflect more neutral or non-directive language. Clinicians highlighted the risk of cancer in the gonadal surgery PDA were over-represented, particularly given the relatively low risk of gonadal cancer for most children with DSD. Clinicians also suggested including more information about contra-sexual puberty and dysgenetic gonads. These changes significantly reduced the bias that could be introduced when overly emphasizing cancer – a term that was felt to hold significant swaying power in parents’/guardians’ decisions. In response to the genetic testing PDA, a genetic counsellor suggested including the limitations of testing given that, currently, only half will learn of the cause for a 46, XY DSD from genetic testing. Changes incorporated into the PDAs reflected the value of having a research team that was interprofessional and also included parents and an adult living with DSD.
3.2.2 Changes not made
All alpha testing results were considered, though not always incorporated. In these instances, we responded with the rationale for our decision while remaining open if the person(s) were unsatisfied with our response. In response to the gender of rearing PDA, several clinicians questioned whether it was relevant to include information about the infants’ capacity for full sexual function as an adolescent and adult. However, parent members of the research team revealed that, in fact, their child’s capacity for full sexual function as an adolescent and adult had been an important factor in their decision-making. Some clinicians questioned the rationale of including a gender-neutral option given that there was no evidence base to draw from. Parents advised that they felt that this option had the potential to prompt a valuable conversation, again reflecting the benefit of including a broad range of stakeholders in co-designing the PDAs. To meet IPDAS criteria, a PDA must, at minimum, present relevant options; therefore, focusing only on raising the child as a girl or boy and leaving out the gender-neutral option would have compromised the quality of the PDA.
Several suggestions were made to modify the actual template of the PDAs. These included: a) replacing the system of stars in Step 3 values clarification exercise in the genetic testing PDA with Arabic numbers to enhance clarity; and b) replacing the use of ‘true’ or ‘false’ in Step 4 knowledge test with ‘yes, no, not sure’ to render this step less intimidating. The concerns were reviewed by the PDA experts and the respondents were reassured that these approaches have been well-tested in multiple PDAs evaluated in randomized controlled trials without any issues. The research team plans to monitor these concerns when the PDAs undergo beta-testing with parents/guardians and clinicians discussing these decisions in the DSD clinic.
4 Discussion
This study was designed to engage end users in co-designing a suite of PDAs to support parents and guardians of infants and young children diagnosed with DSD. Overall, the PDAs were positively received. Parents reported that they would have found them beneficial, and clinicians stated that they could imagine using them in their practice. This degree of buy-in is likely due to the participatory approach used in their design. We went one step further to alpha test them with a naïve interprofessional DSD clinical team at another large teaching hospital. Our findings lead us to the following points of discussion.
Throughout the co-design process, it became evident that though clinicians might think favorably about PDAs, there are many misconceptions about involving parents/guardians in decision making. A PDA is a clinical tool that supports SDM (35). At several stages, clinicians requested that specific information be included about the individual child’s condition and expressed concern that the families would not be able to make these decisions by themselves based on the PDAs. The PDA experts clarified that PDAs are not intended to replace regular patient education materials or the role of the healthcare team involved in the care. Rather, PDAs are adjuncts to clinical practice and shown to be effective interventions for providing information on a specific decision using a structured format that guides patients/families in a process of decision making (12). It is possible that this confusion has contributed to the general hesitancy to incorporate PDAs into clinical practice in previous studies despite over 100 studies demonstrating their effectiveness (36).
Navigating the co-creation of PDAs in this clinical context included particular challenges due to the highly politicized and polarizing views held by patients, families, clinicians, and advocates alike. Debates about use of language were frequent and highlighted the potentially inflammatory nature of terms such as ‘elective’ and ‘unnecessary’ surgeries. Ultimately, following a rigorous methodological process, including parents who had chosen different options for their children, and engaging with a variety of experts with diverging perspectives contributed to reducing any conscious or subconscious bias held by team members. Future PDA co-design in similarly contested clinical contexts might benefit from diversifying the stakeholders and end users with whom they collaborate.
4.1 Strengths and limitations
This study has strengths and limitations to consider. Although our research team members had clinical and lived experiences and were from several DSD centers, the subsequent alpha testing was done with an interprofessional DSD clinical team within a single centre. Despite this limitation, we expect the findings to be transferable to other DSD specialty programs. The methods roadmap to co-design PDAs used well tested Ottawa Decision Support Framework (ODSF) template that meets the International Patient Decision Aid Standards, but the four PDAs have not been formally evaluated to determine their effect on parents’ decision making outcomes. Evaluation on parents’ outcomes is planned in the next study and the PDAs are expected to have similar outcomes to other PDAs designed using this well-tested template (23).
5 Conclusion
Our study methods pathway was successful in guiding the research team of clinicians, parents of a child with DSD, an adult living with DSD, and methodological experts to co-design a series of PDAs for parents and guardians of an infant or young child with DSD. The PDAs were co-designed for difficult decisions about genetic testing, gender of rearing, gonad surgery, and genital surgery that were identified as priorities in our decisional needs assessment. These PDAs are clinical tools designed to support parents/guardians to participate in making shared DSD-related decisions and the next steps are to evaluate them to determine the effect on parents’ decisional outcomes. While these tools are specific for DSD, the co-design process are transferable to co-design of PDAs in other pediatric populations.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Ethics statement
Ethical review and approval was not required for the study involving human participants in accordance with the local legislation and institutional requirements. Written informed consent to participate in this study was not required from the participants in accordance with the national legislation and the institutional requirements. Patient/Parent/Caregiver affiliations have been pseudonymized to protect the identities of vulnerable participants.
Author contributions
SL and DS co-wrote the first draft of the manuscript. MC, MG, KS-J, DES, and DS contributed to the conception and design of the study. All authors co-designed the patient decision aids, reviewed the manuscript, and approved the submitted version.
Funding
This work was supported by the Eunice Kennedy Shriver National Institute for Child Health and Human Development (R01 HD086583, R01 HD086583 S1, and R01 HD093450) and GME Innovation grant (U072007) from the University of Michigan Medical School.
Acknowledgments
We would like to acknowledge the contributions of the healthcare professionals who took the time to participate in the survey during the COVID-19 pandemic that had a huge negative impact on staff workload and moral.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Lee PA, Nordenström A, Houk CP, Ahmed SF, Auchus R, Baratz A, et al. Global disorders of sex development update since 2006: perceptions, approach and care. Horm Res Paediatr (2016) 85:158–80. doi: 10.1159/000442975
2. Lee PA, Houk CP, Ahmed SF, Hughes IA, International Consensus Conference on Intersex organized by the Lawson Wilkins Pediatric Endocrine S and the European Society for Paediatric E. Consensus statement on management of intersex disorders. international consensus conference on intersex. Pediatrics (2006) 118:e488-500. doi: 10.1542/peds.2006-0738
3. Arboleda VA, Sandberg DE, Villain E. DSDs: genetics, underlying pathologies and psychosexual differentiation. Nat Rev Endocrinol. (2014) 10(10):603–15. doi: 10.1038/nrendo.2014.130
4. Katz AL, Webb SA, Committee on Bioethics. Informed consent in decision-making in pediatric practice. Pediatrics (2016) 138:e20161485. doi: 10.1542/peds.2016-1485
5. Lawson ML, Shephard AL, Feenstra B, Boland L, Sourial N and Stacey D. Decision coaching using a patient decision aid for youth and parents considering insulin delivery methods for type 1 diabetes: a pre/post study. BMC Pediatr (2020) 20:1. doi: 10.1186/s12887-019-1898-4
6. Money J, Hampson JG and Hampson JL. Hermaphroditism: recommendations concerning assignment of sex, change of sex and psychologic management. Bull Johns Hopkins Hosp (1955) 97:284–300.
7. United Nations Human Rights Office of the High Commissioner. United nations human rights report 2019 (2019). Available at: https://www.ohchr.org/sites/default/files/Documents/Publications/OHCHRreport2019.pdf (Accessed September 19, 2022).
8. Human Rights Watch. I Want to be like nature made me.” medically unnecessary surgeries on intersex children in the US. Washington, DC (2017). Available at: https://www.hrw.org/sites/default/files/report_pdf/lgbtintersex0717_web_0.pdf.
9. Australian Human Rights Commission. Australian Human rights commission annual report 2020-2021 (2021). Available at: https://www.transparency.gov.au/annual-reports/australian-human-rights-commission/reporting-year/2020-21 (Accessed September 19, 2022).
10. Vavilov S, Smith G, Starkey M, Pockney P and Deshpande AV. Parental decision regret in childhood hypospadias surgery: A systematic review. J Paediatr Child Health (2020) 56:1514–20. doi: 10.1111/jpc.15075
11. Jackson C, Cheater FM and Reid I. A systematic review of decision support needs of parents making child health decisions. Health Expect (2008) 11:232–51. doi: 10.1111/j.1369-7625.2008.00496.x
12. Stacey D, Legare F and Lewis KB. Patient decision aids to engage adults in treatment or screening decisions. JAMA (2017) 318:657–8. doi: 10.1001/jama.2017.10289
13. Stacey D, Volk RJ. The international patient decision aid standards (IPDAS) collaboration: evidence update 2.0. Med Decis Making (2021) 41:729–33. doi: 10.1177/0272989x211035681
14. Feenstra B, Lawson ML, Harrison D, Boland L and Stacey D. Decision coaching using the Ottawa family decision guide with parents and their children: a field testing study. BMC Med Inform Decis Mak (2015) 15:1–10. doi: 10.1186/s12911-014-0126-2
15. Lurie Children's Hospital. Update on intersex care at lurie children’s and our sex development clinic (2021). Available at: https://www.luriechildrens.org/en/blog/update-on-intersex-care-at-lurie-childrens-and-our-sex-development-clinic/ (Accessed September 11,2022).
16. Gillam LH, Hewitt JK and Warne GL. Ethical principles for the management of infants with disorders of sex development. Horm Res Paediatr (2010) 74:412–8. doi: 10.1159/000316940
17. American Medical Association. CEJA report 3-I-18. report 3 on the council on ethical and judicial affairs (1-I-19): Amendment to e-2.2.1, “Pediatric decision making” (Resolution 3-A-16, “Supporting autonomy for patients with differences of sex development [DSD]”) (Resolution 13-A-18, “Opposing surgical sex assignment of infants with differences of sex development”). In: Association AM, editor. CEJA report 3-I-18 (Opinion 2.2.1). American Medical Association, Council on Ethical and Judicial Affairs 330 N Wabash Ave, Chicago, IL 60611 (2019). Available at: https://www.ama-assn.org/system/files/2019-12/i18-ceja-report-3.pdf.
18. Chan KH, Misseri R, Carroll A, Frankel RM, Moore C, Cockrum B, et al. User testing of a hypospadias decision aid prototype at a pediatric medical conference. J Pediatr Urol (2020) 16:685.e681–685.e688. doi: 10.1016/j.jpurol.2020.08.006
19. Binion K, Miller A, Misseri R, Kaefer M, Longtin K, Carroll A, et al. Ask the parents: Testing the acceptability and usability of a hypospadias decision aid. J Pediatr Urol (2022) 18:170.e171–170.e179. doi: 10.1016/j.jpurol.2022.01.004
20. Witteman HO, Maki KG, Vaisson G, Finderup J, Lewis KB, Dahl Steffensen K, et al. Systematic development of patient decision aids: An update from the IPDAS collaboration. Med Decis Making (2021) 41:736–54. doi: 10.1177/0272989x211014163
21. Coulter A, Stilwell D, Kryworuchko J, Mullen PD, Ng CJ, van der Weijden T. A systematic development process for patient decision aids. BMC Med Inform Decis Mak (2013) 13:1–5. doi: 10.1186/1472-6947-13-S2-S2
22. Stacey D, Legare F, Boland L, Lewis KB, Loiselle MC, Hoefel L, et al. 20th anniversary Ottawa decision support framework part 3: overview of systematic reviews and updated framework. Med Decis Making (2020) 40:379–98. doi: 10.1177/0272989x20911870
23. Gardner M, Floody E and Sandberg DE. Support information for parents: Thinking about your child's sex development (2022). Available at: https://drive.google.com/file/d/1YZsUw7tAu_UKLeLQDt7EzfBuliI1nK1G/view (Accessed September 26, 2022).
24. Timmermans S, Yang A, Gardner M, Keegan CE, Yashar BM, Fechner PY, et al. Does patient-centered care change genital surgery decisions? the strategic use of clinical uncertainty in disorders of sex development clinics. J Health Soc Behav (2018) 59:520–35. doi: 10.1177/0022146518802460
25. Sandberg DE, Gardner M, Kopec K, Urbanski M, Callens N, Keegan CE, et al. Development of a decision support tool in pediatric differences/disorders of sex development. Semin Pediatr Surg (2019) 28:150838. doi: 10.1016/j.sempedsurg.2019.150838
26. Bowen S, Graham ID. Integrated knowledge translation. In: Straus SE, Tetroe J, Graham ID, editors. Knowledge translation in health care: moving from evidence to practice. Oxford: Wiley Blackwell (2013). p. 14–23.
27. Suorsa-Johnson KI, Gardner MD, Brinkman W, Carley M, Gruppen L, Liang N, et al. (2022). A survey of healthcare professionals’ perceptions of the decisional needs of parents with an infant born with a disorder/difference of sex development. J Pediatrics Urol. doi: 10.1016/j.jpurol.2022.09.015
28. Hoefel L, O'Connor AM, Lewis KB, Boland L, Sikora L, Hu J, et al. 20th anniversary update of the Ottawa decision support framework part I: A systematic review of the decisional needs of people making health or social decisions. Med Decis Making (2020) 40:555–81. doi: 10.1177/0272989x20936209
29. Hoefel L, Lewis KB, O'Connor AM and Stacey D. 20th anniversary update of the Ottawa decision support framework part 2: sub-analysis of a systematic review of patient decision aids. Med Decis Making (2020) 40:522–39. doi: 10.1177/0272989x20924645
30. O'Connor A, Stacey D, Saarimaki A, Pardo Pardo J, Rader T, Welch V, et al. Ottawa Patient decision aid development eTraining.
31. Joseph-Williams N, Newcombe R, Politi M, Durand MA, Sivell S, Stacey D, et al. Toward minimum standards for certifying patient decision aids: A modified Delphi consensus process. Med Decis Making (2013) 34:699–710. doi: 10.1177/0272989x13501721
32. Martin RW, Brogård Andersen S, O’Brien MA, Bravo P, Hoffmann T, Olling K, et al. Providing balanced information about options in patient decision aids: An update from the international patient decision aid standards. Med Decis Making (2021) 41:780–800. doi: 10.1177/0272989x211021397
33. Legare F, Kearing SA, Clay K, Gagnon S, D'Amours D, Rousseau M, et al. Are you SURE? assessing patient decisional conflict with a 4-item screening test. Can Fam Physician (2009) 56:e308-314.
34. SmartTots. SmartTots funds research into pediatric anesthesia with the goal of making usrgery safer for infants and children (2022). Available at: https://smarttots.org/ (Accessed August 11, 2022).
35. Stacey D, Légaré F, Lewis K, Barry MJ, Bennett CL, Eden KB, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev (2017) 4:CD001431. doi: 10.1002/14651858.cd001431.pub5
Keywords: patient decision aid, disorders of sex development (DSD), differences of sex development (DSD), intersex, decisional needs, shared decision making, decision making, decisional conflict
Citation: Lightfoot S, Carley M, Brinkman W, Gardner MD, Gruppen LD, Liang N, Pinkelman K, Speiser PW, Suorsa-Johnson KI, VanderBrink B, Wisniewski J, Sandberg DE and Stacey D (2023) Co-creating a suite of patient decision aids for parents of an infant or young child with differences of sex development: A methods roadmap. Front. Urol. 2:1020152. doi: 10.3389/fruro.2022.1020152
Received: 15 August 2022; Accepted: 26 October 2022;
Published: 04 January 2023.
Edited by:
Kiarash Taghavi, Monash Children’s Hospital, AustraliaReviewed by:
Nathalie Webb, Monash Health & Monash University, AustraliaSakineh Hajebrahimi, Tabriz University of Medical Sciences, Iran
Copyright © 2023 Lightfoot, Carley, Brinkman, Gardner, Gruppen, Liang, Pinkelman, Speiser, Suorsa-Johnson, VanderBrink, Wisniewski, Sandberg and Stacey. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dawn Stacey, ZHN0YWNleUB1b3R0YXdhLmNh
 Sophie Lightfoot
Sophie Lightfoot Meg Carley2
Meg Carley2 Melissa D. Gardner
Melissa D. Gardner Phyllis W. Speiser
Phyllis W. Speiser Kristina I. Suorsa-Johnson
Kristina I. Suorsa-Johnson David E. Sandberg
David E. Sandberg Dawn Stacey
Dawn Stacey