
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Sports Act. Living , 01 April 2025
Sec. Exercise Physiology
Volume 7 - 2025 | https://doi.org/10.3389/fspor.2025.1533030
Introduction: This study compares energy expenditure, body composition, and metabolic rate between serial (SCG) and integrated (ICG) combined training in untrained middle-aged obese males. Method: Twenty untrained obese males (age: 50 ± 3 years) were randomly assigned to a SCG (n = 10) or an ICG (n = 10). Both groups completed a 3-month training program (3 sessions/week) consisting of walking and bodyweight exercises, differing only in the sequence of aerobic and strength training. In SCG, strength training was performed before aerobic training, while in ICG, aerobic and strength exercises alternated in a predetermined order.
Results: After a 3-month training period, both groups significantly improved all health indices compared to pre-training values (p < 0.005, g = 0.40–2.71), indicating small to large effects. Specifically, there were moderate reductions in body fat percentage, small decreases in body circumferences, and moderate-to-large reductions in arterial blood pressure and resting heart rate. Additionally, respiratory function showed a large improvement. No significant differences were observed between SCG and ICG in energy expenditure, resting metabolic rate, total daily energy expenditure, macronutrient composition, or health indices after the intervention (p > 0.05).
Conclusion: In conclusion, both SCG and ICG are effective strategies for improving metabolic health, respiratory function, and body composition in middle-aged obese males. These findings highlight the flexibility of combined training approaches in promoting overall health and fitness in this population and suggest that both combined training programs can be implemented in structured exercise programs to promote cardiometabolic health in middle-aged adults.
Physical inactivity increases the risk of cardiovascular diseases and the other chronic diseases as obesity, diabetes, hypertension and cancer (1, 2). Obesity, affecting millions worldwide, particularly children and adolescents, is a significant global health issue (3). To prevent obesity and other chronic diseases, the World Health Organization (4) recommends that untrained healthy adults engage in regular physical activity 3–5 times per week (5). Walking, in particular, is a popular, convenient, and cost-free form of aerobic exercise that can be easily integrated into daily routines and maintained throughout life (6). Numerous studies demonstrate the beneficial effects of walking on aerobic capacity, lipid profile, body composition, and blood pressure (7–9). In addition to aerobic activities like walking, incorporating strength training into the program can enhance health-related benefits (10). For this reason, over the past decades, numerous researchers have recommended designing and implementing various combined training programs that include both cardiorespiratory and neuromuscular exercises. These programs aim to improve overall health, functional capacity, and physical fitness parameters (10).
There are two forms of combined training programs: the serial method, where strength training is completed before or after aerobic exercise in each session, and the integrated method, where aerobic and strength routines are alternated repeatedly during the training session (1). Although numerous studies have examined the effects of serial combined aerobic (walking) and strength training programs on physical fitness and health indices (11–15), fewer studies have investigated integrated combined training programs. Additionally, while some research has directly compared serial combined training program (SCG) and integrated combined training program (ICG), these studies have primarily focused on different populations, such as trained individuals, female participants, or athletes. To our knowledge, only one study has directly compared the efficiency of SCG and ICG aerobic (walking) and strength training programs in middle-aged overweight untrained men (1). In this study, both combined training programs improved health indices and physical fitness, with no significant differences between them (1).
Various studies have compared the energy expenditure of healthy adults using newer aerobic devices with traditional forms of aerobic exercises, such as walking, running, or cycling (16, 17). Many studies have also compared the energy expenditure of modified forms of walking with regular walking (18–20). However, differences in energy expenditure between SCG and ICG may be influenced by their distinct physiological demands. The structured nature of SCG may allow for greater total work output in each exercise mode, while the intermittent alternation of aerobic and strength exercises in ICG may elicit greater cardiovascular and metabolic responses, potentially leading to differences in fat oxidation, oxygen consumption, and recovery dynamics. For example, Church et al. (20) compared regular walking with Nordic walking and found that Nordic walking induced significantly higher energy expenditure than regular walking. However, few studies compare the energy expenditure of training regimes that involve resistance training (21). Therefore, only a few studies have compared the energy expenditure using SCG or ICG aerobic and strength training programs. Hunter et al. (22) compared the energy expenditure of a combined aerobic and strength training program with that of aerobic training or strength training alone in untrained older women. They found that only the combined training program significantly decreased energy expenditure (−150 kcal/day). Similarly, Pichon et al. (23) compared the energy expenditure of traditional resistance training and circuit weight training in young adults. The results of this study suggest that the circuit training program induced higher energy expenditure than traditional resistance training. In another study, Monteiro et al. (24) compared the energy expenditure of circuit weight training with that of compounded circuit training (an integrated training program). The ICG induced higher energy expenditure than the typical circuit training. Additionally, no previous study has compared the energy expenditure of two outdoor combined exercise programs (serial and integrated) in obese middle aged untrained men.
In recent years, the measurement of energy expenditure using the latest sport watches has become increasingly prevalent. Therefore, a study by Roos et al. (25) estimated energy expenditure during aerobic running using three different sport watches. They found that the Polar V800 (the same watch used in this study) is recommended for accurate assessment of energy expenditure.
However, to our knowledge, only three studies (1, 26–29) directly compared the effectiveness of SCG and ICG reported equivocal findings. Grivas et al. (1) showed that SCG and ICG aerobic (walking) and strength training (body weight exercises) led to similar benefits in middle-aged males. Similarly, Karatrantou et al. (29) observed that SCG and ICG aerobic (aerobic dance) and strength training (body weight exercises) led to similar benefits in middle-aged females. On the other hand, Davis et al. (26–28) reported that ICG aerobic (treadmill running) and strength training (resistance exercises) was slightly superior in improving some fitness parameters in young collegiate track and field athletes. However, there is evidence that subjects' characteristics (e.g., age, sex) and physical fitness level are associated with different cardiovascular responses, peripheral fatigue development, and substrate utilization during exercise (30, 31), potentially leading to varying neuromuscular and aerobic training adaptations (32, 33). Additionally, the mode of aerobic exercise in a combined training program may influence the extent of neuromuscular adaptations (34).
Thus, the main objectives of this study were: (a) to examine and compare the energy expenditure, resting metabolic rate, total daily energy expenditure and macronutrient composition of SCG and ICG, and (b) to assess and compare the efficacy of SCG and ICG on health physical fitness indices.
The study participants were 20 healthy, untrained obese men aged between 44 and 54 years old. Obesity in this study was determined based on body mass index (BMI), body fat percentage, and waist-to-hip ratio, following the World Health Organization (WHO) and American College of Sports Medicine (ACSM) guidelines. Participants were classified as obese if they had a BMI ≥30 kg/m2, a BF% ≥25%, or a waist-to-hip ratio >0.90 for males. These criteria ensured that participants exhibited both general and central obesity, making them an appropriate population for investigating the effects of combined training modalities on metabolic and physiological adaptations. The sample size was determined based on the convenience. Prior to the study, participants' health (health history questionnaire, resting electrocardiogram, and echocardiogram examined by a cardiologist) and activity status were assessed. Their activity status was assessed using a structured questionnaire, which included questions regarding the frequency, duration, and intensity of physical activity over the past year. The questionnaire specifically evaluated participation in structured strength or endurance training programs, as well as occupational and recreational physical activity levels, to ensure that participants met the criteria for inactivity. Additionally, verbal confirmation of their inactivity status was obtained during an initial interview conducted by the research team. All participants met the following criteria: (1) inactive status (i.e., without systematic strength or endurance training for at least one year before the study), (2) free from chronic illnesses, and (3) no injuries or diseases. Participants were thoroughly informed about the potential risks, discomforts, and benefits of the study. Subsequently, they received detailed information about the study design, measurements, and procedures and provided written informed consent form. Participants were fully informed about the study's hypotheses and objectives as part of the informed consent process, as the study relied on objective physiological and metabolic measurements rather than self-reported outcomes. The procedures were following the Helsinki declaration of 1975, as revised in 2000, and approval was received from the Ethics Committee of the local university.
Two weeks before the beginning of the training protocol, subjects completed a laboratory familiarization session and were informed about the proper form of exercises. Thereafter, indices of health were assessed on 2 separate days, with a 24 h interval between sessions to minimize participant fatigue and ensure accurate data collection. Two days following the pre-training testing, the subjects were randomly divided into two equal groups (n = 10 per group): SCG and ICG (Table 1). During the study, SCG and ICG participants were involved in a 3-month outdoor combined training program. All measurements were repeated after 3-months of training. The measurements were made at the same time of the day to minimize the effects of circadian fluctuation and started two days after the end of the training intervention. Before testing, all participants were asked to refrain from: (a) alcohol or caffeine consumption within 3 h of testing, (b) smoking within 3 h of testing, and (c) strenuous physical strength and endurance activities within 48 h of testing.
Both groups participated in a 3-month combined training program (3 times/week). The duration of each training session was 58–88 min and consisted of 15 min of warm-up (10 min walking and 5 min stretching exercises), followed by 33–63 min of SCG or ICG. Each session was completed with a 10 min cool down period (5 min walking and 5 min stretching exercises). Both groups performed the same aerobic and strength exercises during the workouts, using equivalent intensity, duration, volume, and training frequency. Training intensity was controlled using an HR monitor, maintaining a target heart rate of 65%–80% HRmax throughout the training session. This intensity range was chosen based on established guidelines for improving cardiovascular fitness and metabolic health in untrained middle-aged individuals (1, 29). Thus, the only difference between the two groups was the sequence of aerobic and strength training. In SCG, the strength training was performed before aerobic training, while in ICG the aerobic and the strength training were alternated repeatedly in a predetermined order (3 min of walking/2 min of strength training) as described previously (1) (Figure 1).
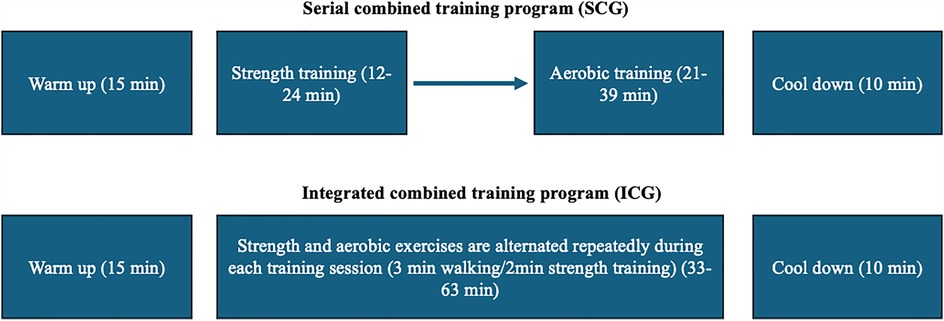
Figure 1. Schematic representation of the training protocols. Both groups performed a 15 min warm-up and a 10 min cool-down. In SCG, strength training was performed before aerobic training, whereas in ICG, strength and aerobic exercises were alternated (3 min walking/2 min strength training) throughout the session. Both groups performed the same aerobic and strength exercises during the workouts, using equivalent intensity, duration, volume, and training frequency.
The aerobic training program, for both exercise groups, consisted of walking at an intensity corresponding to 65%–80% of age-predicted HRmax (Polar Electro, Kempele, Finland) for 21–39 min. The intensity and duration of training progressively increased during the training program according to the procedures described previously (1). The strength training program included five resistance exercises targeting all major muscle groups: sit-ups, dorsals, push-ups, tricep dips, and lunges. Exercise intensity was progressively increased by adjusting the number of repetitions and sets throughout the intervention (see Table 2). Specifically, participants started with 2 sets of 8 repetitions per exercise and gradually progressed to 4 sets of 12 repetitions over the training period. This progression was implemented following established protocols (1) (see Table 2) to ensure safe and effective adaptation in untrained middle aged individuals. All training sessions were supervised by the project staff to ensure completion and adherence to the training program.

Table 2. Gradual increase of loading parameters during the serial and integrated combined training programs.
Before and after the completion of the 3-month training program, different health indices were measured.
All health-related measurements were conducted before and after the 3-month intervention following a standardized protocol to ensure comparability. To minimize learning effects and measurement variability, participants first attended a familiarization session, where they were introduced to all assessment procedures and instructed on the correct execution on the tests. The testing process was explained in detail, and participants had the opportunity to practice certain measurements to ensure proper technique and consistency before data collection. Testing was conducted over two consecutive days. On the first day, anthropometric and body composition measurements were performed. During anthropometric measurements, participants stood fully upright, looking straight ahead, with heels together, barefoot, and with minimal clothing. Body height was measured using a physical beam scale (Seca model 220, Seca, Hamburg, Germany), with participants standing fully erect, looking straight ahead, and their heels together. Body mass was recorded with the same scale, ensuring participants remained still while standing. Body composition was assessed using bioelectrical impedance analysis (Maltron 900). Participants were instructed to avoid food, beverages, and exercise for at least three hours before the test and to urinate before the measurement to minimize fluid-related variability. They stood barefoot on the analyzer with their hands placed on the designated electrodes while maintaining a neutral posture. Waist-to-hip ratio was measured following WHO guidelines. Waist circumference was taken at the midpoint between the iliac crest and the inferior border of the last rib at the end of a normal exhalation. Hip circumference was measured at the widest part of the buttocks while ensuring the measuring tape was horizontal and parallel to the ground. All measurements were performed with a precision of 0.1 cm and repeated twice to ensure accuracy (35).
On the second day, cardiovascular, pulmonary, and metabolic assessments were conducted. Blood pressure was measured using an electronic blood pressure monitor (A&D-UA-851) after participants remained seated and at rest for 5 min in a quiet environment. The measurement was taken from the participant's dominant arm, positioned at heart level, and was repeated after 1 min to confirm consistency. Pulmonary function was assessed using spirometry (Micro Medical Micro). Participants were instructed to take a deep breath and then exhale forcefully into the spirometer while wearing a nose clip to prevent nasal airflow. Three trials were performed for each participant, and the highest values of forced vital capacity (FVC) and forced expiratory volume in one second (FEV1) were recorded. Total energy expenditure and macronutrient composition was calculated using sport watch Polar V800 (25, 36). Resting metabolic rate (RMR) was calculated using the Katch-McArdle formula, which is based on the fat free mass (FFM): RMR = 370 + (21.6 × FFM (kg)) (37). The total daily energy expenditure (TDEE) was calculated using the Katch-McArdle equation to determine RMR and adjusted for physical activity using an activity factor. The activity factor was determined based on participants' reported physical activity levels, with multipliers ranging from 1.2 (sedentary) to 1.9 (very active). TEE = RMR × activity multiplier (38).
To maintain consistency, all measurements were conducted by the same trained technician, who provided real-time feedback, ensured proper execution, and corrected any errors in participant performance. Before testing, participants attended a familiarization session where they received step-by-step verbal instructions and live demonstrations for each assessment. This session allowed them to practice specific tests under supervision to ensure proper technique and compliance with standardized procedures. Post-intervention assessments followed the same standardized protocols as pre-testing to allow for direct comparisons and minimize variability.
Results are presented as means ± standard deviations. Data were analyzed with IBM SPSS Statistics v.23 software. The normality of data was examined using the Shapiro–Wilk test, while homogeneity of variance was examined with Levene's test. 2-way ANOVAs (group × time; 2 × 2) with repeated measures on the “time” factor were used to analyze the data. Sidak pairwise comparisons were applied to locate the significantly different means within and between groups. One-way ANOVAs were used between groups to compare the relative changes from pre- to post-training. The magnitude of the difference was assessed by Hedges' g (g). The magnitude of the difference was considered small (0.2 < g ≤ 0.5), moderate (0.5 < g ≤ 0.8), or large (g > 0.8). Statistical significance was set at p < 0.05 for all analyses.
ANOVAs showed significant “group × time” interaction effects on health indices (p < 0.005, Table 3). Compared to pre-training values, after a 3-month training period (post-training) both groups (SCG and ICG) significantly (p < 0.005, g = 0.40–2.71) improved all health indices (Figure 2). More specifically, found a moderate decrease in %BF, a small decrease in body circumferences, and a moderate—large decrease in arterial blood pressure and resting heart rate as well as a large increase in FVC and FEV1 were observed. Furthermore, RMR significantly decreased (p < 0.005), while TDEE significantly increased (p < 0.005) (Table 4). Macronutrient composition also showed significant increases, with higher consumption of protein, carbohydrate, and fat (p < 0.005) (Table 5). There were no significant differences between SCG and ICG in any of the measurements (p > 0.05). Additionally, no significant differences were observed in average energy expenditure between SCG and ICG after training sessions (p > 0.05) (Table 6).
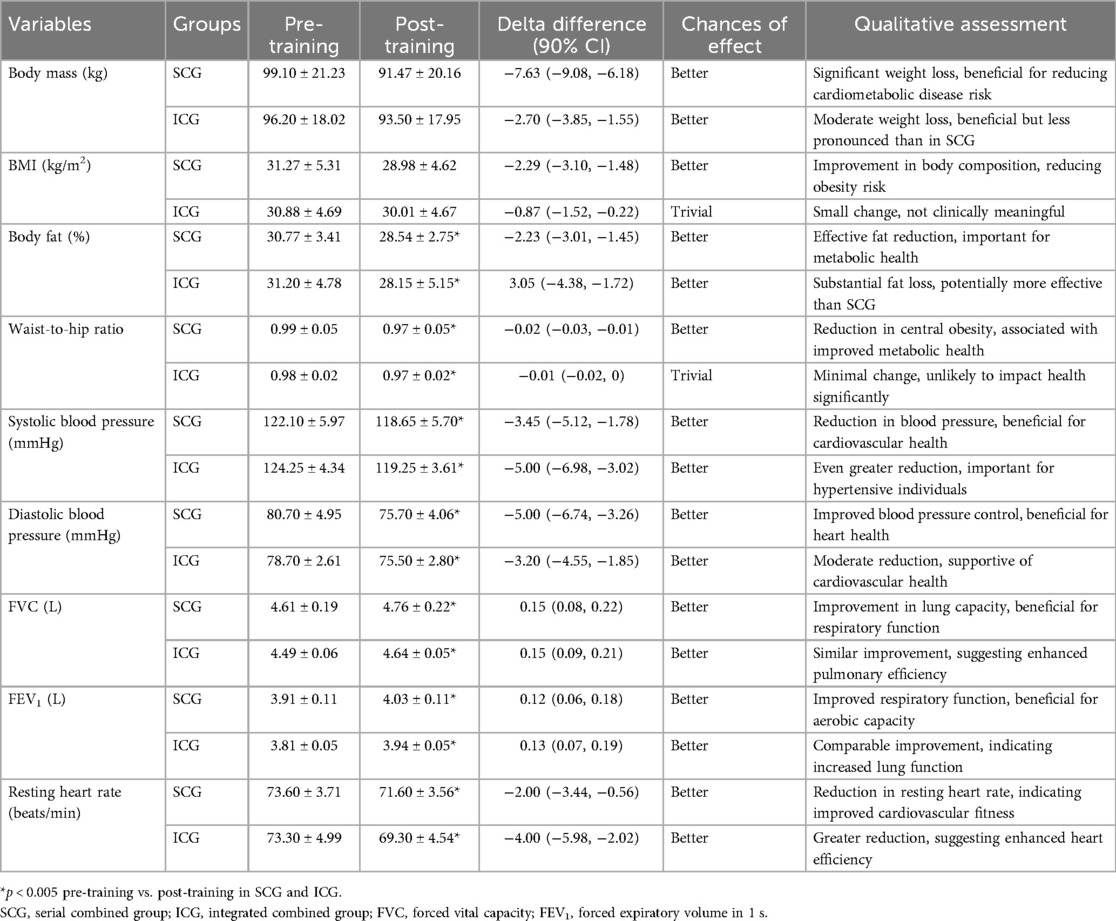
Table 3. Health indices in the two groups (SCG and ICG) before and after training sessions (values are means ± SD).

Figure 2. Standardized mean differences (SMDs) and 95% confidence intervals for the effects of SCG vs. ICG on physical health indices.
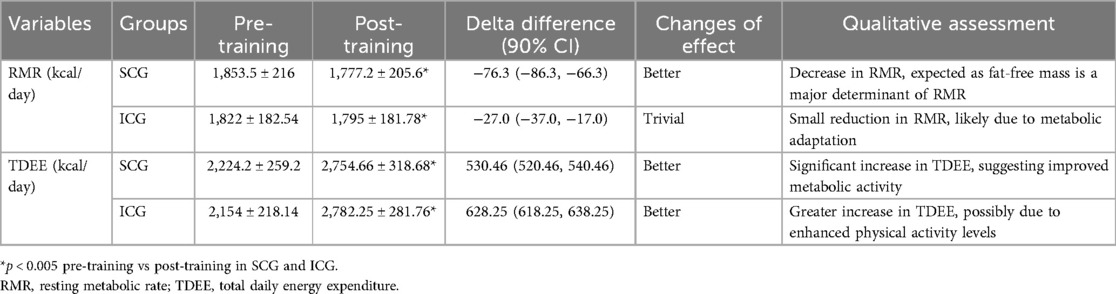
Table 4. Resting metabolic rate and total daily energy expenditure in the two groups (SCG and ICG) before and after training sessions (values are means ± SD).
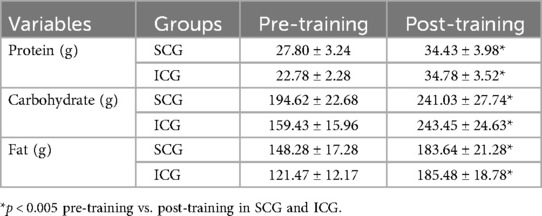
Table 5. Macronutrient composition in the two groups (SCG and ICG) before and after training sessions (values are means ± SD).

Table 6. Average energy expenditure in the two groups (SCG and ICG) after training sessions (values are means ± SD).
The purpose of this study was to examine and compare the energy expenditure, body composition, and metabolic rate between SCG and ICG in untrained middle-aged obese males. The main finding of this study was that there were no significant differences in average energy expenditure, resting metabolic rate, total energy expenditure and macronutrient composition between SCG and ICG after a 3-month combined training program.
This is the first study that compared the energy expenditure of SCG and ICG. However, several studies have examined the energy expenditure after the SCG. The study by Hunter et al. (22) found that 3 days/week of aerobic training and 3 day/week of resistance training significantly decreased energy expenditure by 150 kcal/day in untrained women aged 60–74 years. Dolezal and Potteiger (39) reported that resistance training and combined training groups (endurance and resistance training) showed significant increases in resting metabolic rate compared to the endurance training group.
Furthermore, only a few studies have examined the energy expenditure after the ICG. In a study by Monteiro et al. (24), compared the energy expenditure of circuit weight training with that of compounded circuit training (an integrated training program). The ICG induced higher energy expenditure than the typical circuit training.
Other important findings of the present study include that both training regimes significantly improved resting metabolic rate, with no significant differences observed between the two groups (SCG and ICG). This reduction in resting metabolic rate observed after a 3-month combined training program is a physiological adaptation, as fat-free mass is the main determinant of resting metabolic rate. Since body fat decreased, a concurrent reduction in resting metabolic rate was noted. The aligns with findings from Byrne and Wilmore (40), who reported a significant decrease in resting metabolic rate following 20 weeks of combined resistance and aerobic training (walking). This decline in RMR may be attributed to metabolic adaptation, a well-documented physiological response to weight loss and prolonged energy deficit. Specifically, reductions in body mass and fat stores often lead to compensatory decreases in energy expenditure, as the body attempts to preserve energy balance by downregulating thermogenesis and hormonal activity (e.g., leptin and thyroid hormones) (41, 42). These findings are consistent with previous research demonstrating that sustained weight loss and increased physical activity can induce a proportional decline in RMR, potentially as a survival mechanism to counteract energy deficits (43).
In this study, significantly improved total daily energy expenditure, with no significant differences between the two groups. The increase in total daily energy expenditure highlights the effectiveness of combined training programs in boosting daily energy expenditure, which is crucial for weight managements and improving metabolic health. These results align with prior studies indicating that exercise programs combining resistance and aerobic components significantly elevate total daily caloric expenditure (22). For example, the study by Hunter et al. (22) found that total daily energy expenditure increased after 16 weeks of combined aerobic and resistance training in older women.
Both groups demonstrated significant increases in macronutrient intake after the training period, with no significant differences between groups. These findings highlight the role of dietary adaptations in optimizing training outcomes, ensuring energy availability, and supporting metabolic health, while also aligning with previous literature emphasizing the importance of macronutrient balance in exercise performance and recovery. Macronutrient intake was estimated using a combination of smartwatch-based tracking and self-reported food records. While this approach provides insights into dietary patterns, it is important to acknowledge potential limitations, including variability in wearable-based estimations and potential recall inaccuracies in self-reported data. These factors should be considered when interpreting the impact of dietary adaptations on training outcomes (44, 45).
Other important findings of the present study were that both training regimes decreased body fat, body waist-to-hip ratio, and blood pressure, while increasing respiratory function. Our findings for the SCG aerobic (walking) and strength training (body weight strength exercises) program are in line with finding from previous studies reporting improvements in body fat, and body weight in middle-aged men following SCG (1, 11–15). Other studies, however, did not observed positive training adaptations in body weight, BMI, and waist circumference (13–15). The differences observed between the results from the earlier studies and the present study could be attribute to variations in subjects' characteristics and training loads. However, the primary factor is likely the order of exercises, which may amplify the interference effect and reduce the efficacy of SCG strength and aerobic training programs in improving neuromuscular performance (46, 47).
Furthermore, there are limited studies on ICG for middle-aged men. Previous studies showed similar improvements in body fat, body weight, and waist circumference in middle-aged men (1, 48–50). However, some of the previous studies have found no improvements in body fat, body weight, and waist circumference (48, 49). These findings highlight the need for careful consideration of exercise sequencing in combined training programs, as the interference effect may influence neuromuscular adaptations. Given the mixed results in body composition outcomes, exercise prescription for obese populations should emphasize individualized approaches, progressive overload, and long-term adherence strategies to maximize metabolic benefits. The discrepancies between the results of previous studies and the present study could be attributed to differences in aerobic training modalities, subject characteristics, loading parameters, and training frequency.
A key limitation of this study is the reliance on predictive equations to estimate resting metabolic rate, total daily energy expenditure, and macronutrient intake. While these equations are widely used and validated for general populations, they may not fully account for individual metabolic variability, particularly in untrained, middle-aged obese males. Additionally, the study relied on self-reported dietary intake, which is subject to recall bias and potential underreporting, further impacting the accuracy of macronutrient estimations. Another limitation is the absence of a control group, which restricts the ability to directly attribute observed changes to the intervention. The relatively small sample size also limits the generalizability of the findings.
Future research should aim to improve measurement accuracy by incorporating gold-standard methods such as indirect calorimetry and doubly labeled water for metabolic assessments. The inclusion of direct calorimetry could provide an even more precise evaluation of energy expenditure, further refining predictive models used in metabolic research. Additionally, future studies should consider using objective dietary tracking tools, such as wearable devices or biomarkers, to minimize biases associated with self-reported intake and enhance measurement accuracy.
This study compared the effects of SCG and ICG on energy expenditure, body composition, and metabolic rate in untrained middle-aged obese males. The findings demonstrated no significant differences between the two training modalities in energy expenditure, resting metabolic rate, or macronutrient intake after three months. Both programs were effective in increasing total daily energy expenditure, optimizing macronutrient oxidation, and supporting metabolic health. In addition, both training regimens significantly decreased body fat, waist-to-hip ratio, and blood pressure while enhancing respiratory function. These results highlight the effectiveness of combined training in improving key health markers. Differences between this study and prior research may reflect variations in participants' characteristics, training protocols, and exercise modalities. Overall, both SCG and ICG appear to be viable strategies for promoting metabolic health and improving body composition in middle-aged obese males. Future research should focus on long-term outcomes, including training sustainability, exercise adherence, and metabolic adaptations, to further refine and optimize training programs for this population.
The raw data supporting the conclusions of this article will be made available by the author, without undue reservation.
The studies involving humans were approved by the Ethics Committee of the University of Thessaly. The procedures followed the Helsinki Declaration of 1975, as revised in 2000. The studies were conducted in accordance with local legislation and institutional requirements. All participants provided written informed consent to participate in this study.
GG: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The author would like to thank the study participants for their commitment.
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Grivas GV, Karatrantou K, Chasialis A, Batatolis C, Ioakimidis P, Gerodimos V. Serial vs. integrated outdoor combined training programs for health promotion in middle-aged males. Sports. (2022) 10:122. doi: 10.3390/sports10080122
2. Onagbiye S, Guddemi A, Baruwa OJ, Alberti F, Odone A, Ricci H, et al. Association of sedentary time with risk of cardiovascular diseases and cardiovascular mortality: a systematic review and meta-analysis of prospective cohort studies. Prev Med. (2024) 179:107812. doi: 10.1016/j.ypmed.2023.107812
3. Zhang X, Liu J, Ni Y, Yi C, Fang Y, Ning Q, et al. Global prevalence of overweight and obesity in children and adolescents. JAMA Pediatr. (2024) 178:800–13. doi: 10.1001/jamapediatrics.2024.1576
4. WHO Guidelines on Physical Activity and Sedentary Behaviour. Geneva: World Health Organization (2020). Available at: https://www.who.int/publications/i/item/9789240015128 (Accessed November 10, 2024).
5. Liguori G, Feito Y, Fountaine J, Roy B. ACSM’s Guidelines for Exercise Testing and Prescription, 11th ed. Philadelphia, PA: Wolters Kluwer (2021).
6. Ungvari Z, Fazekas-Pongor V, Csiszar A, Kunutsor SK. The multifaceted benefits of walking for healthy aging: from Blue Zones to molecular mechanisms. GeroScience. (2023) 45:3211–39. doi: 10.1007/s11357-023-00873-8
7. Nemoto K, Gen-no H, Masuki S, Okazaki K, Nose H. Effects of high-intensity interval walking training on physical fitness and blood pressure in middle-aged and older people. Mayo Clin Proc. (2007) 82:803–11. doi: 10.4065/82.7.803
8. Magistro D, Liubicich ME, Candela F, Ciairano S. Effect of ecological walking training in sedentary elderly people: act on aging study. Gerontologist. (2014) 54:611–23. doi: 10.1093/geront/gnt039
9. Bai X, Soh KG, Omar Dev RD, Talib O, Xiao W, Cai H. Effect of brisk walking on health-related physical fitness balance and life satisfaction among the elderly: a systematic review. Front Public Health. (2022) 9:829367. doi: 10.3389/fpubh.2021.829367
10. Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee I-M, et al. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. (2011) 43:1334–59. doi: 10.1249/MSS.0b013e318213fefb
11. Marzolini S, Oh PI, Thomas SG, Goodman JM. Aerobic and resistance training in coronary disease: single versus multiple sets. Med Sci Sports Exerc. (2008) 40:1557–64. doi: 10.1249/MSS.0b013e318177eb7f
12. Mandic S, Tymchak W, Kim D, Daub B, Quinney HA, Taylor D, et al. Effects of aerobic or aerobic and resistance training on cardiorespiratory and skeletal muscle function in heart failure: a randomized controlled pilot trial. Clin Rehabil. (2009) 23:207–16. doi: 10.1177/0269215508095362
13. Libardi CA, Souza GV, GÁspari AF, Santos CFD, Leite ST, Dias R, et al. Effects of concurrent training on interleukin-6, tumour necrosis factor-alpha and C-reactive protein in middle-aged men. J Sports Sci. (2011) 29:1573–81. doi: 10.1080/02640414.2011.609896
14. Libardi CA, De Souza GV, Cavaglieri CR, Madruga VA, Chacon-Mikahil MPT. Effect of resistance, endurance, and concurrent training on TNF-α, IL-6, and CRP. Med Sci Sports Exerc (2012) 44:50–6. doi: 10.1249/MSS.0b013e318229d2e9
15. Mendonca GV, Pereira FD, Fernhall B. Effects of combined aerobic and resistance exercise training in adults with and without down syndrome. Arch Phys Med Rehabil. (2011) 92:37–45. doi: 10.1016/j.apmr.2010.09.015
16. Falcone PH, Tai C-Y, Carson LR, Joy JM, Mosman MM, McCann TR, et al. Caloric expenditure of aerobic, resistance, or combined high-intensity interval training using a hydraulic resistance system in healthy men. J Strength Cond Res. (2015) 29:779–85. doi: 10.1519/JSC.0000000000000661
17. Nakagata T, Yamada Y, Naito H. Energy expenditure, recovery oxygen consumption, and substrate oxidation during and after body weight resistance exercise with slow movement compared to treadmill walking. Physiol Int. (2018) 105:371–85. doi: 10.1556/2060.105.2018.4.27
18. Porcari JP, Hendrickson TL, Walter PR, Terry L, Walsko G. The physiological responses to walking with and without power Poles™ on treadmill exercise. Res Q Exerc Sport. (1997) 68:161–6. doi: 10.1080/02701367.1997.10607992
19. Jeukendrup A, Thielen J, Wagenmakers A, Brouns F, Saris W. Effect of medium-chain triacylglycerol and carbohydrate ingestion during exercise on substrate utilization and subsequent cycling performance. Am J Clin Nutr. (1998) 67:397–404. doi: 10.1093/ajcn/67.3.397
20. Church TS, Earnest CP, Morss GM. Field testing of physiological responses associated with nordic walking. Res Q Exerc Sport. (2002) 73:296–300. doi: 10.1080/02701367.2002.10609023
21. Bloomer RJ. Energy cost of moderate-duration resistance and aerobic exercise. J Strength Cond Res. (2005) 19:878–92. doi: 10.1519/R-16534.1
22. Hunter GR, Bickel CS, Fisher G, Neumeier WH, McCarthy JP. Combined aerobic and strength training and energy expenditure in older women. Med Sci Sports Exerc. (2013) 45:1386–93. doi: 10.1249/MSS.0b013e3182860099
23. Pichon CE, Hunter GR, Morris M, Bond RL, Metz J. Blood pressure and heart rate response and metabolic cost of circuit versus traditional weight training. J Strength Cond Res. (1996) 10:153–6.
24. Monteiro AG, Alveno DA, Prado M, Monteiro GDA, Ugrinowitsch C, Aoki MS, et al. Acute physiological responses to different circuit training protocols. J Sports Med Phys Fitness. (2008) 48:438–42.18997645
25. Roos L, Taube W, Beeler N, Wyss T. Validity of sports watches when estimating energy expenditure during running. BMC Sports Sci Med Rehabil. (2017) 9:22. doi: 10.1186/s13102-017-0089-6
26. Davis WJ, Wood DT, Andrews RG, Elkind LM, Davis WB. Concurrent training enhances Athletes’ strength, muscle endurance, and other measures. J Strength Cond Res. (2008) 22:1487–502. doi: 10.1519/JSC.0b013e3181739f08
27. Davis WJ, Wood DT, Andrews RG, Elkind LM, Davis WB. Concurrent training enhances Athletes’ cardiovascular and cardiorespiratory measures. J Strength Cond Res. (2008) 22:1503–14. doi: 10.1519/JSC.0b013e3181739f9f
28. Davis WJ, Wood DT, Andrews RG, Elkind LM, Davis WB. Elimination of delayed-onset muscle soreness by Pre-resistance cardioacceleration before each set. J Strength Cond Res. (2008) 22:212–25. doi: 10.1519/JSC.0b013e31815f93a1
29. Karatrantou K, Gerodimos V, Häkkinen K, Zafeiridis A. Health-promoting effects of serial vs. Integrated combined strength and aerobic training. Int J Sports Med. (2016) 38:55–64. doi: 10.1055/s-0042-116495
30. Johnson L, Kraemer R, Kraemer G, Haltom R, Cordill A, Welsch M, et al. Substrate utilization during exercise in postmenopausal women on hormone replacement therapy. Eur J Appl Physiol. (2002) 88:282–7. doi: 10.1007/s00421-002-0686-1
31. Farinatti PTV, Monteiro WD. Walk–run transition in young and older adults: with special reference to the cardio-respiratory responses. Eur J Appl Physiol. (2010) 109:379–88. doi: 10.1007/s00421-010-1366-1
32. McGuire DK, Levine BD, Williamson JW, Snell PG, Blomqvist CG, Saltin B, et al. A 30-year follow-up of the Dallas bed rest and training study: II. Effect of age on cardiovascular adaptation to exercise training. Circulation. (2001) 104:1358–66. doi: 10.1161/hc3701.096099
33. Deschenes MR, Kraemer WJ. Performance and physiologic adaptations to resistance training. Am J Phys Med Rehabil. (2002) 81:S3–S16. doi: 10.1097/00002060-200211001-00003
34. Schumann M, Küüsmaa M, Newton RU, Sirparanta A-I, Syväoja H, Häkkinen A, et al. Fitness and lean mass increases during combined training independent of loading order. Med Sci Sports Exerc. (2014) 46:1758–68. doi: 10.1249/MSS.0000000000000303
35. Moosaie F, Fatemi Abhari SM, Deravi N, Karimi Behnagh A, Esteghamati S, Dehghani Firouzabadi F, et al. Waist-to-height ratio is a more accurate tool for predicting hypertension than waist-to-hip circumference and BMI in patients with type 2 diabetes: a prospective study. Front Public Health. (2021) 9:726288. doi: 10.3389/fpubh.2021.726288
36. Kloss EB, Givens A, Palombo L, Bernards J, Niederberger B, Bennett DW, et al. Validation of polar grit X pro for estimating energy expenditure during military field training: a pilot study. J Sports Sci Med. (2023) 22:658. doi: 10.52082/jssm.2023.658
37. McArdle WD. Exercise Physiology: Energy, Nutrition, and Human Performance, 4th ed. Baltimore: Lea & Febiger (1996).
38. Garcia MB. Plan-cook-eat: a meal planner app with optimal macronutrient distribution of calories based on personal total daily energy expenditure. 2019 IEEE 11th Int Conf Humanoid Nanotechnol Inf Technol Commun Control Environ Manag HNICEM (2019). Available online at: http://manuelgarcia.info/publication/plan-cook-eat (accessed November 20, 2024).
39. Dolezal BA, Potteiger JA. Concurrent resistance and endurance training influence basal metabolic rate in nondieting individuals. J Appl Physiol. (1998) 85:695–700. doi: 10.1152/jappl.1998.85.2.695
40. Byrne HK, Wilmore JH. The effects of a 20-week exercise training program on resting metabolic rate in previously sedentary, moderately obese women. Int J Sport Nutr Exerc Metab. (2001) 11:15–31. doi: 10.1123/ijsnem.11.1.15
41. Müller MJ, Bosy-Westphal A, Heymsfield SB. Is there evidence for a set point that regulates human body weight? F1000 Med Rep (2010) 2:59. doi: 10.3410/M2-59
42. Dulloo AG, Schutz Y. Adaptive thermogenesis in resistance to obesity therapies: issues in quantifying thrifty energy expenditure phenotypes in humans. Curr Obes Rep. (2015) 4:230–40. doi: 10.1007/s13679-015-0156-9
43. Nunes CL, Casanova N, Francisco R, Bosy-Westphal A, Hopkins M, Sardinha LB, et al. Does adaptive thermogenesis occur after weight loss in adults? A systematic review. Br J Nutr. (2022) 127:451–69. doi: 10.1017/S0007114521001094
44. Banna JC, McCrory MA, Fialkowski MK, Boushey C. Examining plausibility of self-reported energy intake data: considerations for method selection. Front Nutr. (2017) 4:45. doi: 10.3389/fnut.2017.00045
45. Burrows TL, Ho YY, Rollo ME, Collins CE. Validity of dietary assessment methods when compared to the method of doubly labeled water: a systematic review in adults. Front Endocrinol. (2019) 10:850. doi: 10.3389/fendo.2019.00850
46. Cadore EL, Izquierdo M, Alberton CL, Pinto RS, Conceição M, Cunha G, et al. Strength prior to endurance intra-session exercise sequence optimizes neuromuscular and cardiovascular gains in elderly men. Exp Gerontol. (2012) 47:164–9. doi: 10.1016/j.exger.2011.11.013
47. Cadore EL, Izquierdo M, Pinto SS, Alberton CL, Pinto RS, Baroni BM, et al. Neuromuscular adaptations to concurrent training in the elderly: effects of intrasession exercise sequence. Age (Omaha). (2013) 35:891–903. doi: 10.1007/s11357-012-9405-y
48. Lambers S, Van Laethem C, Van Acker K, Calders P. Influence of combined exercise training on indices of obesity, diabetes and cardiovascular risk in type 2 diabetes patients. Clin Rehabil. (2008) 22:483–92. doi: 10.1177/0269215508084582
49. Schiffer T, Kleinert J, Sperlich B, Schulte S, Struder H. Effects of aerobic dance and fitness programme on physiological and psychological performance in men and women. Int J Fit. (2009) 5:37–46.
Keywords: energy expenditure, concurrent training, exercise, obesity, physical fitness
Citation: Grivas GV (2025) Comparative analysis of combined exercise programs in middle-aged obese males: impact on energy expenditure, body composition, and metabolic rate. Front. Sports Act. Living 7:1533030. doi: 10.3389/fspor.2025.1533030
Received: 22 November 2024; Accepted: 20 March 2025;
Published: 1 April 2025.
Edited by:
Xu Yan, Victoria University, AustraliaReviewed by:
Karuppasamy Govindasamy, Symbiosis International (Deemed University), IndiaCopyright: © 2025 Grivas. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gerasimos V. Grivas, Z3JpdmFzZ2VyQGhvdG1haWwuY29t
†Present Address: Gerasimos V. Grivas, Department of Physical Education, Hellenic Naval Academy, Piraeus, Greece
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.