
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Remote Sens. , 21 March 2025
Sec. Acoustic Remote Sensing
Volume 6 - 2025 | https://doi.org/10.3389/frsen.2025.1482244
This article is part of the Research Topic Detection and Characterization of Unidentified Underwater Biological Sounds, Their Spatiotemporal Patterns and Possible Sources. View all 10 articles
 Michelle-Nicole Havlik*
Michelle-Nicole Havlik* Anieka J. Parry
Anieka J. Parry Alexandra Steckbauer
Alexandra Steckbauer Marta Ezeta Watts
Marta Ezeta Watts Fabio Marchese
Fabio Marchese Francesca Benzoni
Francesca Benzoni Carlos M. Duarte
Carlos M. DuarteThrough sharing characteristics of chorus activity, especially in regions that are particularly data deficient, we can aim at a broader, global understanding of fish chorusing and consequently important spatiotemporal changes in habitat use by schooling fish. Here, we identify seasonal changes in fish chorusing activity using passive acoustic monitoring, in the central Red Sea mesophotic and adjacent shallow coral reef zones. For this study, recorders were placed in the mesophotic coral reef zone (70–80 m), and adjacent shallow reef sites (10 m), over 2 weeks during summer and winter seasons. A total of eleven choruses were identified and catalogued according to timing, location and acoustic characteristics of frequency and sound pressure levels. The presence of choruses in both deep and shallow reef sites is indicative of critical habitat for fish foraging, courtship, spawning, and/or migratory activity. All but two choruses were found to originate at or near the mesophotic sites. Four choruses unique to summer and winter (n=3 and 1 respectively) were most prevalent in soundscapes. Temperature and oxygen levels, measured to document conditions under which the choruses were present, showed little change across the mesophotic zone even between seasons, while daily fluctuation occurred in the adjacent shallow sites in both seasons.
Mesophotic Coral Ecosystems (MCEs) (Pérez-Rosales et al., 2022) have been largely understudied in comparison to their shallow counterparts, despite representing a significant percentage of coral reef ecosystem area globally (Lesser et al., 2009; Kahng et al., 2010; Loya et al., 2019; Pyle and Copus, 2019). This is largely due to the difficulty of access to these depths. MCEs mainly exist beyond the regular limits for SCUBA diving, invoking the need for technical diving or costly manned and unmanned vehicles to adequately explore these ecosystems. MCEs are both dependent on availability of light and temperature, ranging from 30 m to depths of up to 150 m, depending on the location (Kahng et al., 2010).
With rising sea temperatures, MCEs have also been suggested as a potential refuge for coral species against thermal bleaching (Pérez-Rosales et al., 2022; Kahng et al., 2010; Bongaerts et al., 2010). Like shallow reefs, they support a high diversity of corals, sponges, algae and other reef associated organisms (Lesser et al., 2009; Kahng et al., 2010; Loya et al., 2019; Rowley, 2018). However, the bioacoustic dynamics of these habitats, particularly in terms of fish communities, remain poorly understood. The upper mesophotic zone supports similar fish communities to shallow photic reefs, however a faunal shift occurs in the lower mesophotic zone, where unique fish assemblages exist (generally between 60 and 150 m) (Kahng et al., 2010; Raick et al., 2023). MCEs likely possess a unique soundscape fundamental to ecological processes, such as the attraction of larval organisms through phonotaxis (Montgomery et al., 2006; Vermeij et al., 2010; Jones et al., 2017; Lecchini et al., 2018; Anderson, 2021). As acoustic events which often cover a large spatial extent (McCauley and Cato, 2000), fish chorusing can form a part of this soundscape, giving an indication of habitat use by chorusing fish species. Soundscapes of mesophotic and deep coral reef ecosystems remain understudied in comparison to other habitats, such as shallow-water coral reefs, coastal ocean and pelagic sites (Havlik et al., 2022).
Capturing fish chorus activity using passive acoustic monitoring (PAM) can help define spatiotemporal trends in vocal species presence and habitat usage (Cato, 1978; Hawkins et al., 2023; Parsons et al., 2016a), information difficult to ascertain through traditional methods such as visual surveys or trawling (Lindseth and Lobel, 2018; Lobel, 2002). In this way, PAM can enable more effective management and zoning, especially in fisheries management (Luczkovich et al., 2008a). Sound is an intrinsic component for marine life, with many species perceiving sound and using it for essential behaviors such as communication (Amorim, 2006; Heimrich et al., 2021; Kaatz et al., 2017; Ladich, 2019), navigation (Montgomery et al., 2006; Vermeij et al., 2010; Simpson et al., 2005) and feeding (Gannon et al., 2005; Herzing and Elliser, 2014). Consequently, the largescale acoustic event formed by fish chorusing, can affect more than just the species participating. Chorusing can function as a beacon for marine fauna towards areas of high productivity and biomass (Pine et al., 2018; McCauley and Cato, 2016). While globally the characteristics of numerous choruses have been described (Lobel, 2002; Amorim, 2006), few studies have settled on the species responsible with a high degree of certainty. Previously it has been possible to estimate the number of individuals forming a chorusing shoal (Erisman and Rowell, 2017; Rowell et al., 2017; Rowell et al., 2012), however this remains difficult due to lack of species specific bioacoustic information such as calling rates, as well as differences in sound propagation. Nevertheless, observing chorusing behavior can provide presence/absence information of fish assemblages. Despite this value of PAM, its application to MCEs remains particularly sparse (Raick et al., 2023; Raick et al., 2024), further emphasizing the understudied nature of these unique habitats.
As fish chorusing constitutes an essential part of marine soundscapes (Cato, 1978; Pine et al., 2018; McCauley and Cato, 1998), understanding these acoustic phenomena at mesophotic depths can begin to understand seasonality and habitat use by soniferous groups of fish in this understudied zone of the Red Sea. A soundscape is defined by the International Standards Organization (ISO) as the “characterization of the ambient sound in terms of its spatial, temporal and frequency attributes, and the types of sources contributing to the sound field” (Schafer, 1969; Pijanowski et al., 2011; ISO, 2017). All fish are able to perceive sounds underwater (Amorim, 2006; Lobel, 1992; Parmentier and Fine, 2016) but active sound production and acoustic communication has been found in approximately 1000 fish species, from 133 fish families and 33 orders (Looby et al., 2022). The two main mechanisms for sound production in fish are stridulation (rubbing of bones), and the use of sonic muscles attached to the swim bladder (Amorim, 2006; Parmentier and Fine, 2016). While fish species may produce more than one sound (Amorim, 2006; Kaatz et al., 2017; Ladich, 2019; Aalbers and Drawbridge, 2008; Bertucci et al., 2015; Carriço et al., 2019), chorusing behavior by a single species usually consists of one repeated, coordinated sound (Lobel, 2002; Lobel, 1992), or mixed sounds by multiple species as is the case in dawn and dusk chorusing behavior similar to that seen in birds (Cato, 1978; McCauley and Cato, 1998). A chorus is defined as a sustained background noise, resulting when many individuals emit sound simultaneously in a designated area, and is clearly audible above ambient noise (Cato, 1978; Kaatz et al., 2017; McCauley and Cato, 2016). Chorusing in fish can occur passively during feeding (Pine et al., 2018), or to coordinate activities such as courtship, spawning and feeding between individuals of a species (Cato, 1978; McCauley and Cato, 1998; Lobel, 1992; D’spain and Batchelor, 2006; McCauley, 2012; Parsons et al., 2013) and many species have evolved nocturnality as a potential spawning strategy to lessen predatory risk and increase egg survival (Šmejkal et al., 2018).
Due to this nocturnality, the use of PAM is particularly well-suited to detect such coordinated sound production over space and time, offering valuable insights into fish activity where visual surveys are logistically challenging (Luczkovich et al., 2008a; Luczkovich et al., 2008b; Van Oosterom et al., 2016). Visual surveys of spawning aggregations depend on weather conditions, and are generally restricted to a certain area (Chérubin et al., 2020), while the aggregations may have an uncertain spatial range (Ikegami et al., 2014; McCauley and Cato, 2016). While weather also influences PAM recordings, in certain conditions filters may be applied to data to extract meaningful patterns (Erbe et al., 2022). PAM can non-invasively detect biological activity and changes that traditional methods, such as plankton net tows, may not accurately capture, as egg collection can be influenced by predation and currents (Chérubin et al., 2020). However, while fish choruses are often recorded, attributing them to specific species remains challenging due to the difficulty of direct observation (McCauley and Cato, 2016; Zhang and Katsnelson, 2021; Hawkins et al., 2023). Species identification can include using a camera/hydrophone set up where the shoal is estimated to be, or in an experimental tank. While a tank ensures the sound originates from the study species, fish may not conduct themselves naturally. With development, PAM could provide better insights into the activity and durationspawning events as has been seen in exsitu experiments (Luczkovich et al., 2008b; Montie et al., 2017).
Nevertheless, PAM offers the ability to non-invasively monitor changes in fish chorusing and detect these important biological events that may otherwise remain undocumented. Fish chorusing can function as an attractant, or a beacon towards an area of high productivity utilized by the chorusing species (McCauley and Cato, 2016), associated predators (Gannon et al., 2005; Remage-Healey et al., 2006; Luczkovich and Keusenkothen, 2008) and other marine life. Furthermore, the absence or reduction of fish chorusing may serve as an indicator of environmental change or habitat degradation, signaling broader ecological disturbances (Gordon et al., 2019; Rossi et al., 2017; Duarte et al., 2021). While fish chorusing has been documented globally, soundscape analyses of MCEs remain limited to a handful of studies, particularly in regions of French Polynesia and Okinawa, Japan (Raick et al., 2023; Raick et al., 2024; Lin et al., 2021; Bertucci et al., 2017; Akamatsu et al., 2018; Williams et al., 2024). The eastern coast of the Central Red Sea, where few courtship and spawning events have been described (El-Regal, 2013; Kattan et al., 2022), and no fish choruses have been documented, remains an understudied area for bioacoustics altogether. This study presents the first comprehensive investigation into the spatial and temporal components of fish chorusing within the mesophotic and shallow reef zones of the Red Sea.
The Red Sea is a unique ocean basin with highly diverse shallow and deep coastal reef ecosystems, flanking a central canyon with depths of up to 3,000 m (Berumen et al., 2013; Carvalho et al., 2019). The Red Sea exhibits high levels of biodiversity and endemism; of approximately 1166 fish species present, 165 species are exclusively endemic to the Red Sea (Bogorodsky and Randall, 2019). Additionally, of 46 species that live below 200 m, 22 species are endemic (Bogorodsky and Randall, 2019). Given increasing threats such as overfishing (Chérubin et al., 2020; Ikegami et al., 2014) and rising anthropogenic noise pollution from one of the world’s most active shipping lanes, the need for innovative monitoring strategies is paramount. It is documented that choruses lasting longer with increased fish calls, involved more productive spawning sessions (Montie et al., 2017; Sadovy De Mitcheson et al., 2008), and anthropogenic noise such as motor boats can directly overlap frequencies, disturbing activities surrounding communication and reproduction success (Holles et al., 2013; Nedelec et al., 2017; Popper and Hawkins, 2019; Radford et al., 2023; De Jong et al., 2018).
Here, we aimed to characterize temporal patterns of fish chorusing in soundscapes within the mesophotic and adjacent shallow reef ecosystems of the Red Sea. Using multiple hydrophones (three per location) within two study locations, deployed on a gradient from shallow to the adjacent mesophotic area, we aimed to understand the spatial extent and potential source location of fish choruses in these zones, as well as the seasonality and temporal patterns.
Suitable sites in the mesophotic zone were identified using high resolution multibeam bathymetry (unpublished raw data) and georeferenced ROV transects collected in 2021 by the Habitat and Benthic Biodiversity (HaBB) Laboratory team at KAUST (Marchese et al., 2021). Two locations were subsequently chosen: Al Fahal (AF) reef (22°18′03.0″N 38°57′35.6″E) and King Abdullah Economic City (KAEC/KA) reef (22°22′13.3″N 39°03′39.1″E). At each location one shallow site and two mesophotic sites were established, totaling six sites (Figures 1, 2; Table 1). In terms of the entire reef shelf, KAEC sites are considered to be inshore, while Al Fahal is a mid-shore fringing reef (13 km offshore) while more fringing reefs enclose the reef complex ∼20 km offshore).
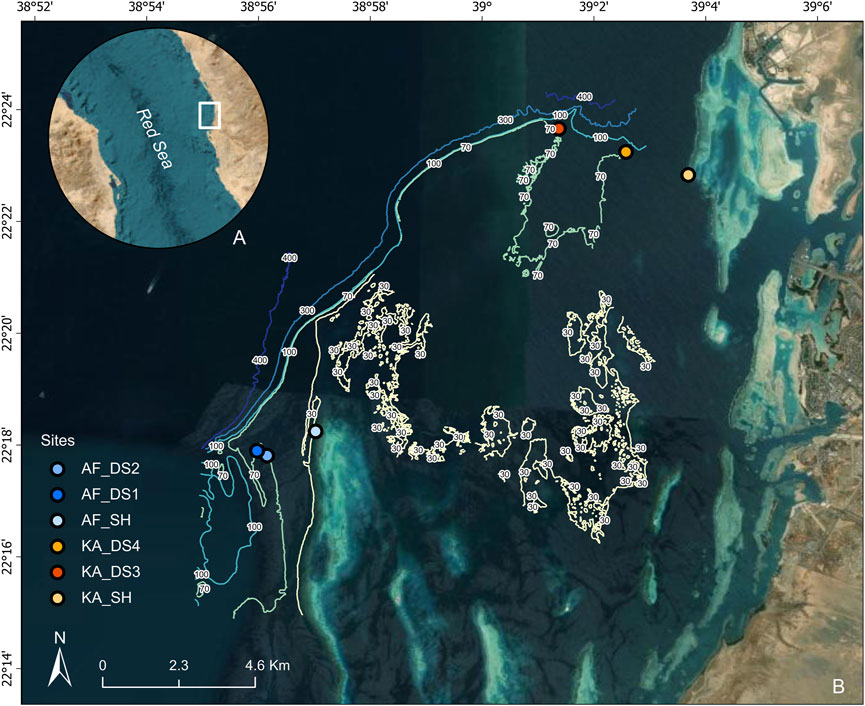
Figure 1. (A) Inset map showing study location in the Red Sea (B) Map of study site locations, off the coast of Thuwal and KAEC (Saudi Arabia) (depths in Table 1) with benthic elevation, of Al Fahal (AF) reef complex and KAEC (KA) reef complex (survey data in publication) showing a steep drop off from the mesophotic plateau (∼70–80 m) to deeper water (∼600 m depth). The cargo ship on the satellite image (below A) shows common passage area for large vessels.

Figure 2. (A) Coral at Al Fahal shallow site (12 m depth) (B) Frame deployment at KAEC shallow site (11.8 m) surrounded by patches of mostly dead coral. ROV photo of mesophotic site KAEC, DS3 (77.8 m), representative of all mesophotic sites, showing (C) surrounding rocky bommie on the sand flat, dominated by coralline crustose algae, corals such as Leptoseris sp. and sponges (KAEC, DS3, 77.8 m depth) (D) Gobiidae on sand flat usually in symbiosis with Alpheidae (snapping shrimp) (KAEC, DS3, 77.8 m depth).

Table 1. Frame deployment information of the two locations, Al Fahal reef (AF) and KAEC (KA), shallow (SH) and mesophotic (deep) sites (DS).
All mesophotic sites were 70–85 m deep, primarily sandy and flat bottomed with rocky patches. These rocky patches were dominated by algal and sponge cover, as well as low coral cover of mostly gorgonians and plating species. KAEC shallow site (11.8 m), was comprised of mainly dead coral, macroalgae, and a sandy bottom, while Al Fahal shallow site (12 m) was characterized by high live coral cover. It was observed visually that the Al Fahal shallow site had a greater abundance and biodiversity of fish than in KAEC (Figure 2). The distance between the KAEC sites was 1.8 km, and 2 km respectively. While the shallow Al Fahal site was similarly spaced at 1.5 km from the first mesophotic site, the second was set only 300 m away due to constraints of bathymetry. The flat, sandy bottom between 70 and 90 m, required for deployments only extended thus far, with a steep incline immediately adjacent. For both mesophotic and shallow sites, aluminum frames were each equipped with one SoundTrap ST300 hydrophone (Ocean Instruments, NZ), a CTD (Ocean Seven 310 plus) with temperature, oxygen, salinity and pH probes, and a miniDOT® dissolved oxygen and temperature logger (PME) set to record every 15 and 10 min respectively (Frame seen in Figure 2B; Supplementary Figure S2). Due to technical difficulties, data from both CTD and miniDOT®s were not available from all sites (Table 1; Supplementary Table S2, S3). Each frame was floated by a subsurface buoy and weighted with a biodegradable hessian sack filled with sand and gravel, and retrieved via an acoustic release (RT6-1000, Sonardyne Ltd.). Hydrophones were set to record continuously at a sampling rate of 96 kHz (High-gain setting), and the individual in-house factory calibration used for analysis (Supplementary Table S5). Each SoundTrap is calibrated by Ocean Instruments using a piston phone calibration at 250 Hz and is specified to have flat response across its full bandwith +/- 3 dB. Each of the mesophotic deployments were consequently surveyed using an ROV (BlueROV2, Blue Robotics Inc) with a GoPro Hero 9 attached to record in 4K resolution, while the shallow (10 m) were deployed and checked via SCUBA diving. The SeaTrac standard system (USBL) was used to track the position of the ROV in real time. The hydrophones were set to record for up to 15 days per season (recording dates for individual hydrophones in Table 1).
Firstly, audio files (.wav) were processed using CHORUS (Characterisation of Recorded Underwater Sound) (Gavrilov and Parsons, 2014) and long-term spectral averages (LTSA) of the whole recording period were observed to identify chorus patterns and anomalies (1,024 point Hanning window type, frequency resolution 1 Hz). To create the LTSAs, the data is preprocessed using the CHORUS script. As this requires entry of SoundTrap calibration data, the resulting Pressure Spectral Density plot of all recordings provides a calibrated view (averaging time of 0.08333, minimum level 40 dB, and a maximum level 110 dB). For each site, chorus patterns were visually identified, and the day/timing of highest chorus energy noted.
Secondly, to reduce power needed for processing, the first one minute of each five minute audio file was extracted using a custom MATLAB script. These one minute files were used to visualize each chorus in Raven Pro 1.6 (Cornell University, Lab of Ornithology). Using methods from Borie-Mojica, Rezende (Borie-Mojica et al., 2022), parameters of low and high frequency limits (Hz), peak frequency (Hz), and the stop and start times of each chorus and duration were subsequently manually extracted in Raven 1.6 (Table 1).
The start and stop times were classified as when the individual fish calls were overlapping, indicative of either shoaling activity (defined as individual fish grouping together for social reasons) for the choruses consisting of a single sound, or heightened activity/communication in the choruses with multiple calls. Broadband analysis (Hann window, 1 s window length, 50% window overlap, end-end calibration) was performed using PAMGuide in MATLAB (Merchant et al., 2015). The frequency band 40 Hz–3 kHz was used to identify the peak Sound Pressure Level (SPL) values, representing received levels (RL) (Erbe et al., 2022). As SPL is a time-average, it is a useful unit for sounds that last for a long time, or that can be considered continuous, like chorusing (Erbe et al., 2022). A custom MATLAB script averaged the results for each minute recording. The corresponding recording to peak level and time was checked in Raven Pro 1.6 to make sure there was no vessel interference or noise artefact during this time, and input to Supplementary Table S1. Choruses were assigned a unique label according to temporal, spectral and acoustic characteristics.
Finally, to produce overall chorus parameters (Table 2) for each chorus individually, data from all sites (Supplementary Table S1) was averaged to find a midpoint value, and the range represented (±), for start/end times, duration, low/high/peak frequency, and peak SPL values. The representative days, chosen for strong chorus activity and the least amount of vessel noise interference with the choruses, were January 13th – 14th for Winter, with the exception of KA_DS4, which only recorded until the 13th , and the 12th was used, and the 27th – 28th July for Summer. Pressure Spectral Density Plots (PSD, Figures 3, 5) for each main chorus type, were created using PAMGuide (Merchant et al., 2015) (Hann window, 1 s window length, 90% overlap).
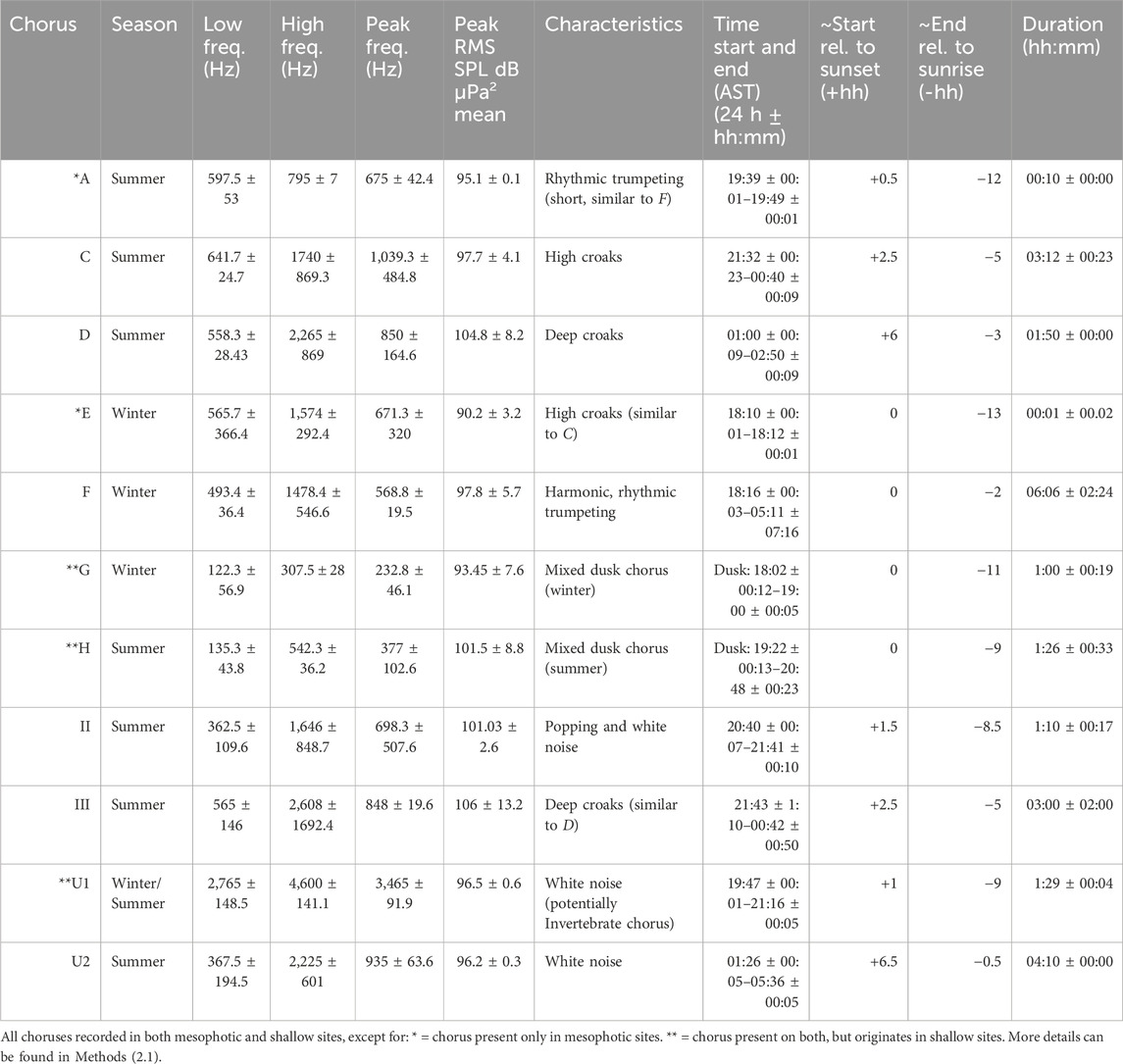
Table 2. Summary of fish chorus parameters from all sites and seasons. Results are the mean of from each site where the chorus is present (Supplementary Table S1) calculated using 1 min clips from peak days (without vessel presence in the clip) per site-specific frequency band (Supplementary Table S1) (NB: U1 is calculated from 40 Hz to 5 kHz).
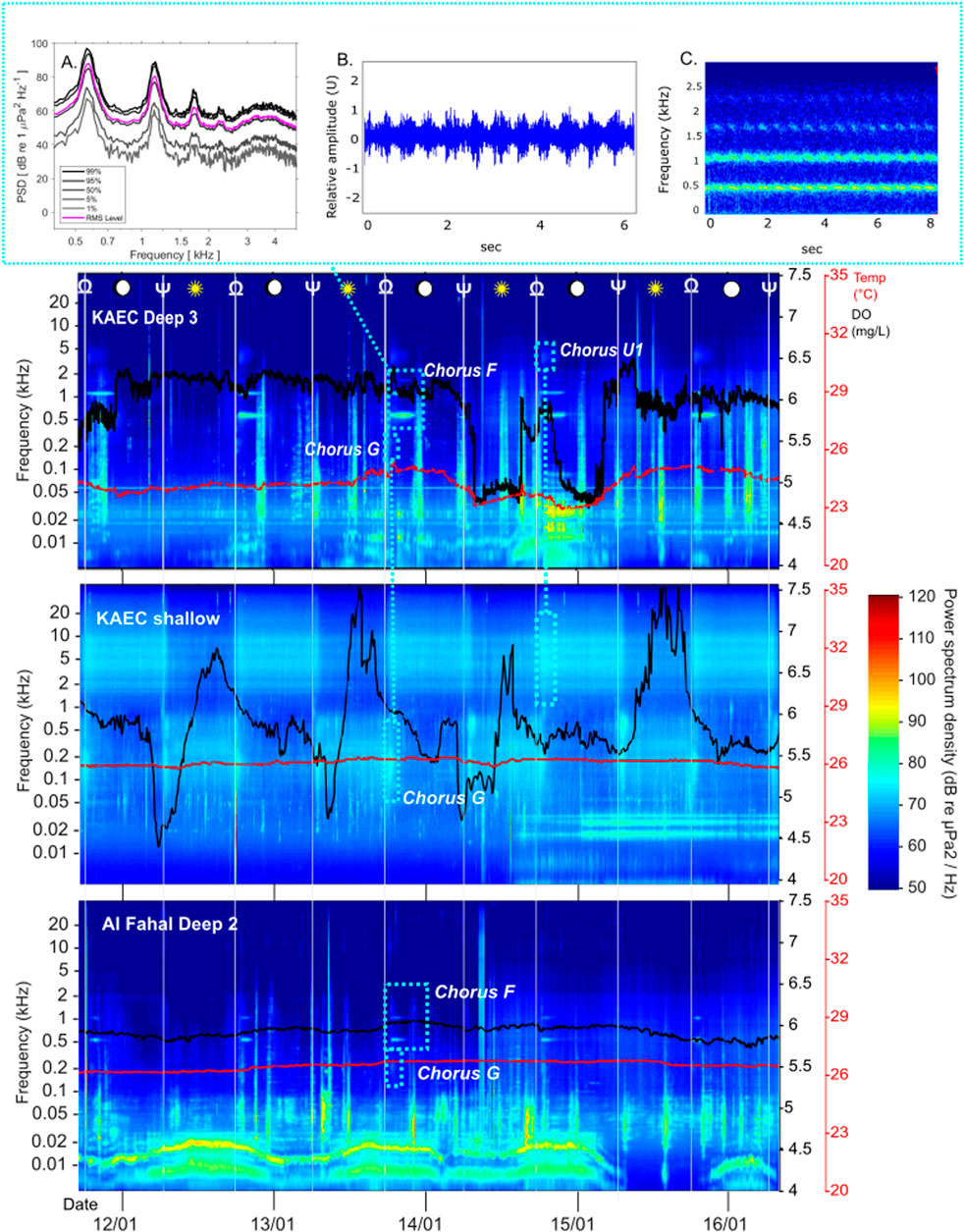
Figure 3. TOP: Main winter Chorus F (coordinated, rhythmic trumpeting pulses, with up to 4 harmonics at highest observed chorus strength (0.6 kHz, 1.15 kHz, 1.75 kHz, 2.3 kHz) at KAEC mesophotic site DS3 (A) Spectral density plot [90% overlap, Hann window, window length 0.08 ) of F (B) 6 s waveform of F (Raven Pro 1.6.5, Hann window size 5412)] (C) 6 s spectrogram of Chorus F showing rhythmic pulsed nature of coordinated calling (Raven Pro 1.6.5, Hann window, window size 5412) BOTTOM: Long term spectrogram of KAEC mesophotic site 3, KAEC shallow site, and Al Fahal mesophotic site 2 (CHORUS, parameters in methods). Temperature (red) and dissolved oxygen data (black) from the miniDOTs is overlaid. Chorus F, Chorus G and Chorus U1 are highlighted by blue dotted lines. Sunrise (Ψ) and sunset (Ω) are denoted by aforementioned symbols and white lines. Light blue dotted lines outline choruses present.
Data files from CTD and miniDOT®s were cleaned and trimmed to the deployment time windows (Table 1; additional data will be made available). Due to technical difficulties, data from CTD and miniDOT®s were not available from all sites. To correct for differences between instrument types, a correction value was applied based on locations were both datasets were available. Data were averaged per hour (due to different sampling intervals of 15 and 10 min for CTDs and miniDOT®s, respectively), and plotted using packages “ggplot2” and “lubridate” in R (version 4.1.1). Wind speeds (knots) for the time period were taken from Jeddah, and resulting Beaufort scale per day was calculated (https://mesonet.agron.iastate.edu/). Lunar phase was also noted by illumination percentage for the nearest coastal town with data, Thuwal (timeanddate.com), Saudi Arabia (Figures 3, 5; Supplementary Table S4).
Eleven fish choruses were identified, and were different between seasons. Fewer choruses were detected in winter (n = 3), than in summer (n = 8) (Table 1, 2).Less difference in chorus types was found within the locations, whereas variation in received levels per site was more prominent. Only Chorus U1 was ubiquitously present across seasons and sites, while Chorus F was the only one present across all sites during a season—winter—except for one shallow site. In both seasons respectively, interconnectivity of shallow and deep soundscapes was shown by the acoustic presence of certain choruses in both.
The main winter chorus, Chorus F, was characterized by melodic trumpeting sounds with rhythmic qualities and was present at all mesophotic sites (Figure 3; Table 2). While Chorus F was recorded across all mesophotic sites, variability in received levels, timing, and peak frequencies was observed between locations (Figures 3, 4; Supplementary Table S1). Chorus F was also briefly audible at the Al Fahal shallow site. The highest averaged received levels for F occurred at KA_DS3 (105.7 dB re 1 μPa2) and the inshore adjacent KA_DS4 (92.9 dB re 1 μPa2) (Supplementary Table S1). Received levels of F at AF_SH reached 102 dB re 1 μPa2, due to overlap with snapping shrimp energy. On average, Chorus F lasted 6:06 h (±02:24 h) during the evening, starting shortly after sunset (18:16PM ± 00:03), and reaching an average peak of 97.8 ± 5.7 dB re 1 μPa2 within 20 min. Chorus F consequently lasted until ∼01:30 AM, with a peak frequency of 568.8 ± 19.5 Hz (Table 2). On KA_DS4 where received levels were highest (Table 2; Figure 4), four harmonics were present in Chorus F at 600 Hz, 1.15 kHz, 1.75 kHz, and 2.3 kHz (Figure 3A), while on other sites, only the lowest three–two harmonics were present.
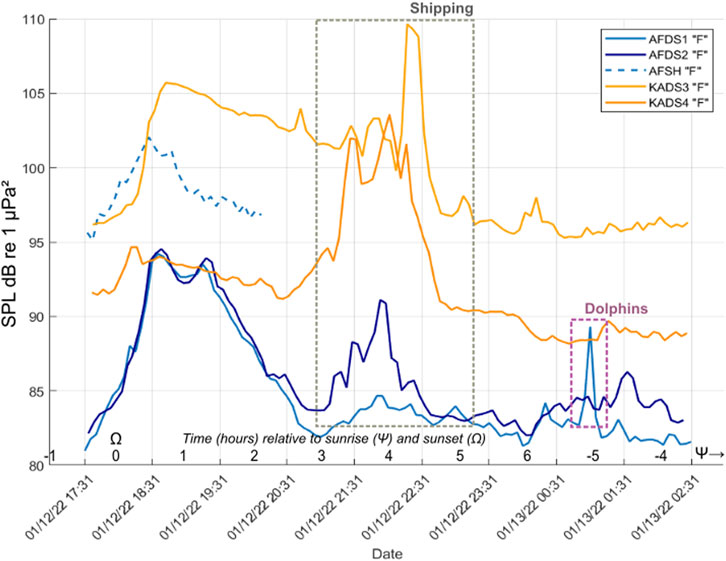
Figure 4. Received levels from start to end times of winter Chorus F at Al Fahal and KAEC mesophotic and shallow sites calculated within the respective frequency bands per site (Supplementary Table S1). Time is Arabian Standard Time (AST). The shallow site (AF_SH) is represented by a dotted blue line, grey squares show the interference of shipping noise during chorusing, falsely inflating the SPL, as well as a pink square for loud dolphin echolocation activity. Time is shown in hours relative to sunrise and sunset on the X-axis. Broadband analysis using specific frequency bands per chorus (Supplementary Table S1), 50% window overlap, Hann window, window length 1 s).
Chorus G, originating in both shallow sites was a dusk chorus consisting of many different sounds, starting at sunset, and lasting approximately 1 h (Table 2). Chorus G had a peak frequency of 232.8 ± 46.1 Hz, and average received level of 93.45 ± 7.6 dB re 1 μPa2 (Table 2).
Chorus E was shorter in length than other choruses (∼10 min) but had comparable characteristics to summer Choruses C and III. The peak frequency of Chorus E was 671.3 ± 320 Hz (C: 1,039.3 ± 484.8 Hz), and its peak SPL was 90.2 ± 3.2 dB re 1 μPa2 (C: 97.7 ± 4.1 dB re 1 μPa2) (Table 2). Chorus E was audible across all mesophotic sites (Supplementary Table S1) except KA_DS3.
Choruses C, D, and III exhibited differences in received levels, timing, and location (Figure 5), but shared similarities in the type of individual sound being a series of pulses (Figure 7). Chorus III was only found on KAEC sites. The highest received levels were observed for III at KA_DS3, peaking at 117.5 dB re 1 μPa2, followed by the adjacent inshore mesophotic site KA_DS4 (109 dB re 1 μPa2) (Supplementary Table S1). Choruses C and D, only observed on Al Fahal sites, exhibited average peak levels of 97.7 ± 4.1 and 104.8 ± 8.2 dB re 1 μPa2, respectively, with slightly higher levels at AF_DS2 than AF_DS1 (Figure 5). Choruses C and D were also audible at adjacent shallow site AF_SH, as was Chorus III at KA_SH (Figures 5, 6; Supplementary Table S1). Choruses III and C overlapped temporally.
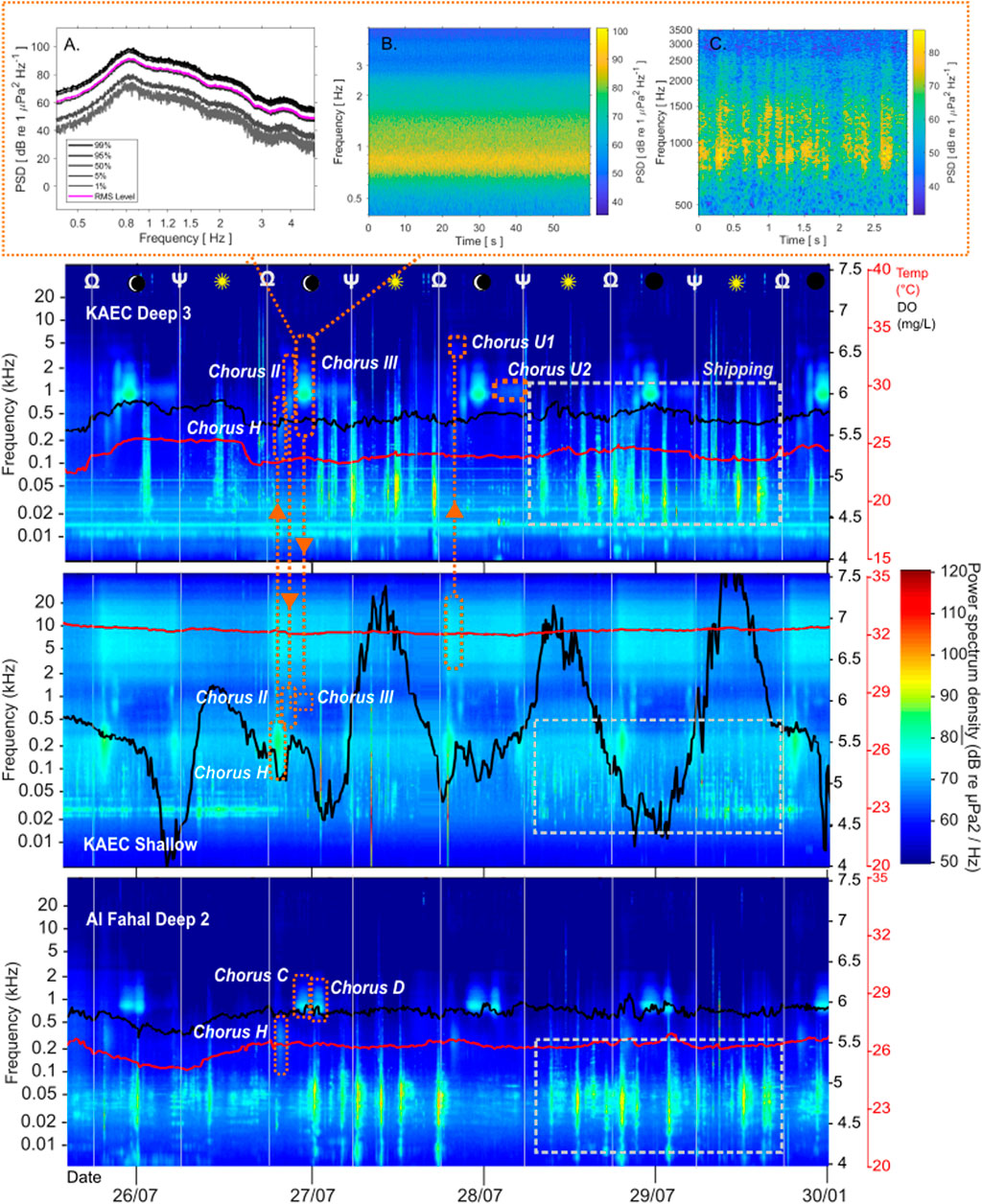
Figure 5. TOP: Main summer chorus at KA_DS3, Chorus III (dry croak pulses, peak frequency: 997 Hz) (D) Spectral density plot (90% overlap, Hann window, window length 0.08 secs) of Chorus III (E) 1 min spectrogram of III (90% overlap, Hann window, window length 0.08 secs) (F) 4 secs example of individual calls of III. BOTTOM: Long term spectrogram (made using CHORUS (Gavrilov and Parsons, 2014) (MATLAB) at KAEC mesophotic site 3, KAEC shallow site, and Al Fahal mesophotic site. Temperature (red) and dissolved oxygen data (black) from the miniDOTs is overlaid. Sunrise (Ψ) and sunset (Ω) are denoted by aforementioned symbols and white lines. Orange dotted lines outline choruses present.
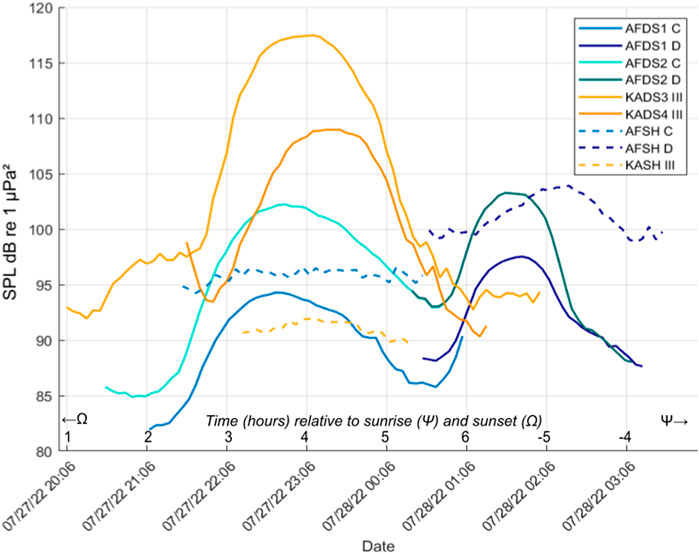
Figure 6. Received levels from half an hour before to half an hour after the start and end times of potentially related Choruses C, D, and III at Al Fahal and KAEC mesophotic and shallow sites. Time is Arabian Standard Time (AST). Time is shown in relation to sunrise (Ψ) and sunset (Ω) on the x-axis. Frequency bands used in analysis specific to each chorus (Supplementary Table S1).
Summer choruses Chorus C and Chorus D were most prevalent at the Al Fahal mesophotic sites but were also audible at lower levels in adjacent shallow sites (Figure 6). As Chorus C ended, Chorus D began, shifting from a higher peak frequency (1039.3 ± 484.8 Hz) to a lower one (850 ± 164.6 Hz) (Table 2; Figures 5, 6).
Individual sounds of III on KA_DS3, the site with highest received levels, showed 4-7 pulses (Figure 7C). On KA_DS4, further inshore, the sounds only had 2-4 pulses (Figure 7B). However, for C and D it was not possible to separate clean examples of individual sounds (Figures 7D, E).
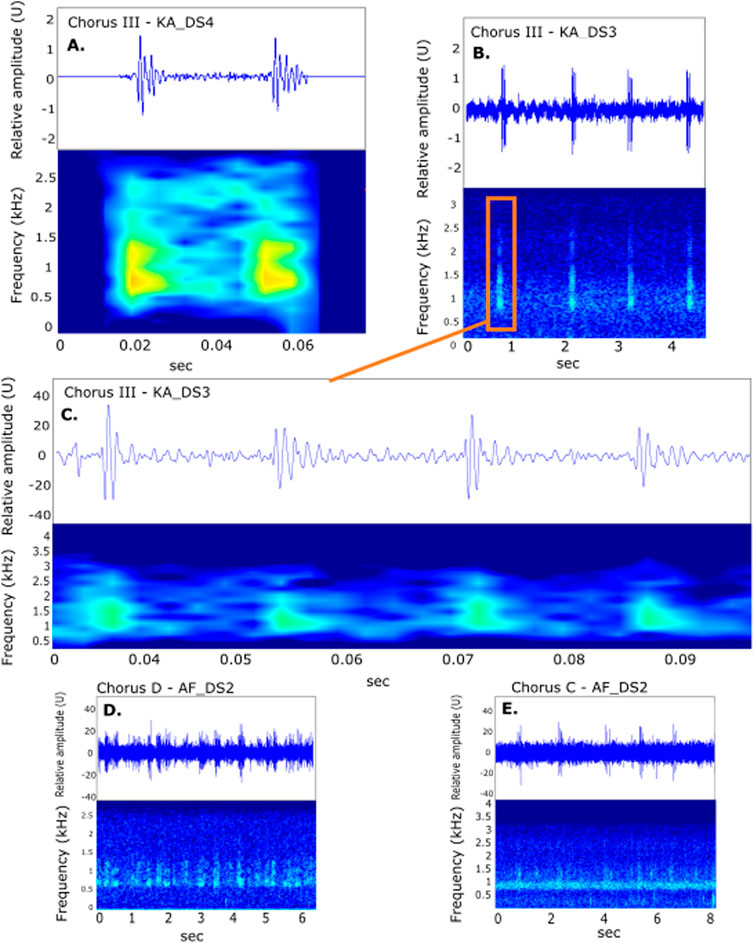
Figure 7. Spectrogram and waveform of individual calls from (A) Chorus III at KA_DS4 and (B) at KA_DS3, where the highest received levels occurred, with (C) zoomed in to show a structure of four pulses from KA_DS3. (D, E) Spectrogram and waveform of Chorus D and C respectively, where individual calls could not be separated for analysis. All spectrograms and waveforms created using Raven Pro 1.6 (Hann window, window size of 5412, band filter according to respective chorus parameters).
Chorus II was restricted to the KAEC shallow and mesophotic sites during the summer (Figure 5; Supplementary Table S1). Chorus II, perceived as popping/pulse sounds and white noise, had a variable peak frequency of 698.3 ± 507.6 Hz and peak SPL of 101.03 ± 2.6 dB re 1 μPa2 across sites.
Chorus A, occurring only in the summer, was very short in length (10 min) possessed a rhythmic quality similar to the winter Chorus F. It had a peak frequency of 675 ± 42.4 Hz (vs. F at 568.8 ± 19.5 Hz) and a peak SPL of 95.1 ± 0.1 dB re 1 μPa2 (vs. F at 97.8 ± 5.7 dB re 1 μPa2). A lastined only ∼10 min during the summer, compared to F, which lasts 06:06 ± 02:24 h during the winter. Chorus A was only present at both Al Fahal mesophotic sites.
Chorus H was the summer dusk chorus, starting at sunset (Supplementary Table S1) comprised of a mix of low-frequency fish sounds, such as pops, knocks, and grunts (Table 2, Figure 5). The peak frequency of Chorus H was 377 ± 102.6 Hz, and received levels were 101.5 ± 8.8 dB re 1 μPa2 (Table 2; Supplementary Table S1).
Choruses U1 and U2 were also present in summer at only the mesophotic sites. Both choruses sounded like white noise. Chorus U1 started at 19:47 (±0.01), one hour after sunset and ended at 21:16 (±0.05), with a peak frequency of 3,465 Hz (±92 Hz). Chorus U2, began at 01:26 (±0:05) 6 hours after sunset, and ended just before sunrise at 05:36 (±0:05). Chorus U2 had a peak frequency of 935 ± 64 Hz.
Water temperature in the mesophotic sites was relatively stable, ranging between 24–27°C across all sites, except for Al Fahal DS2 in summer (25.1–30.4°C) (Supplementary Figure S3; Supplementary Table S2). Shallow sites showed higher variability between seasons, with summer temperatures averaging 31°C and winter temperatures averaging 25.5°C, with diel range within ∼2°C (Figures 3, 5; Supplementary Figure S3). Dissolved oxygen saturation was more stable in mesophotic sites but highly variable in shallow sites regardless of season, particularly at KAEC in winter (68.2%–206% saturation relative to air), showing strong diel patterns (Supplementary Figure S3). Salinity and pH were stable across all depths and seasons at 39.54 ± 0.2 psu and 8.12 ± 0.06, respectively (Supplementary Table S3). Winter recordings coincided with a first quarter to full moon (59%–100% illumination) (Figure 3; Supplementary Table S4), while summer recordings occurred during a third quarter to new moon (0%–34.5% illumination) (Figure 5; Supplementary Table S4). During winter recordings, wind speed steadily increased from a daily average of 3.76 knots (Beaufort scale 2) on 9/1/22, to reach a peak of 10.8 knots (Beaufort scale 4) on 20/1/22 (Supplementary Figure S4). During the summer recording period, the average wind speed declined steadily from 21/7/22 at 8.8 knots (Beaufort scale 3), down to 4.3 knots (Beaufort scale of 2) on 31/7/22 (Supplementary Figure S4).
Eleven distinct chorus types and their characteristics were catalogued across winter and summer, revealing clear seasonal patterns in fish chorusing dynamics, as well as spatial characteristics. Water temperature and oxygen levels in the mesophotic zone, where the majority of choruses were observed, was relatively stable even between seasons (Supplementary Tables S2, S3).
During winter Chorus F was the most prominent, consistently present across mesophotic sites. Chorus F was present during all days of recording, coinciding with a first quarter lunar phase to full moon (59%–100% illumination) (Figure 3). Chorus F reached peak levels (117 dB re µPa2) at KA_DS3 (Table 2; Figures 3, 4), a site closest to the steep drop-off near the KAEC shelf (Figure 1). This characteristic suggests that the shoal responsible for Chorus F could be located in the deeper waters adjacent to the slope. It was not possible to identify individual calls at any part of the chorus, and the rhythmic quality of the sound (Figure 3) implied coordination of calling (Picciulin et al., 2024). While temperature in the mesophotic remained between 24°C and 27°C (Supplementary Figure S3), elevated wind and currents create an iso-speed profile and near surface duct. Water temperature in the mesophotic sites was relatively stable, ranging between 24–27°C across all sites (Supplementary Figure S3; Supplementary Table S2). Sound spreading in winter would have been affected by the presence of a near-surface sound duct (0–200 m)—where sound travels faster—formed by local conditions such as cooler water, higher currents and wind during this period in the Red Sea which also give rise to an iso-speed sound speed profile (SSP). (Erbe et al., 2022; Larsen and Radford, 2018; Larayedh et al., 2024). More efficient horizontal propagation and intensification of sound at the surface would occur along the duct, as well as reflection of sound from the bottom of the duct upwards (Erbe et al., 2022). The surface duct presence in the winter could imply that this chorus may originate shallower than 200m, as sound below would be more easily attenuated (Erbe et al., 2022).
Furthermore, the location of Chorus F could also be evident in the change in frequency components recorded between the sites. While the peak frequency of 568.8 ± 19.5 Hz was fairly consistent among sites, the harmonic components differed between sites. All four harmonics of Chorus F were received at KAEC mesophotic site 3 (Figure 3; Table 2), while these are attenuated to the two lower harmonics in the recordings of KAEC mesophotic site 4, Al Fahal mesophotic sites and Al Fahal shallow site (Figure 3; Supplementary Table S1). This degradation in frequency can occur when sound is picked up from the far-field (Erbe et al., 2022). Chorus F was only audible at Al Fahal shallow site for a brief period (∼1 h from 18:15), with elevated received levels (Figure 4) more likely due to the overlapping impulsive broadband sounds of snapping shrimp (Supplementary Table S1) (Butler et al., 2021; Lillis and Mooney, 2018). This shallow-to-mesophotic acoustic range highlights the broader spatial extent of Chorus F. The absence from the KAEC shallow site, despite being adjacent to the potential origin (KA_DS3), is most likely due to the sound attenuation in the soft absorptive sandy bottom, or by obstruction due to the complex morphology of the shallow, mostly dead reef around the recorder (Erbe et al., 2022).
The extended spatial scale of the fish choruses observed is further highlighted by the presence of shallow dusk Chorus G on mesophotic sites (Figure 3; Supplementary Table S1), as well as the dusk Chorus H during summer. Chorus G at shallow sites KA_SH and AF_SH was present during winter temperatures of 24.4°C–26.5°C (Figure 3; Supplementary Table S2), at a received level of 93.45 ± 7.6 dB re µPa2. This dusk chorus includes, among other sounds, identifiable pulse and knock sounds attributed to Pomacentridae (Akamatsu et al., 2018; Staaterman et al., 2013) and popping sounds characteristic of nocturnal species like Pempheridae (sweepers) (Kaplan et al., 2015; Lyon et al., 2019; Nedelec et al., 2015). Mixed-species assemblages with high site fidelity are commonly attributed to dawn and dusk chorusing (Anderson, 2021; Kaplan et al., 2015; Nedelec et al., 2015). Chorus U1 also appears to originate on the shallow reef site, and due to the high frequency nature and timing, is most likely attributed to the peak in crepuscular invertebrate (snapping shrimp) chorusing (Butler et al., 2021; Lillis and Mooney, 2018) (Figure 3; Table 2). The presence of both Chorus G and Chorus U1 in the deep soundscape recordings (Figure 3; Supplementary Table S1) affirms how shallow reef soundscapes can provide critical acoustic cues for larval settlement and reef navigation for marine fauna (Montgomery et al., 2006; Vermeij et al., 2010; Simpson et al., 2005). Previous experiments using light traps equipped with speakers playing nocturnal reef sounds, with a range of only 65 m, showed an increased catch of larval fish up to 1.6 times that of just light (Leis et al., 2003). Pelagic larvae or predators may rely on these choruses as navigation cues (Montgomery et al., 2006; Simpson et al., 2005; Remage-Healey et al., 2006), such as dolphins, with foraging echolocation visible in AF_DS1 sound levels (Figure 4) as well as communication sounds such as whistles and buzzes (Dibble et al., 2016; Janik, 2000) throughout all mesophotic sites.
Natural sources of sound underwater include wind, waves and currents, and higher wind speeds can affect ambient noise levels underwater, especially in low frequencies (>1 kHz) (Burnham et al., 2023). It is important to note that wind may also affect received levels of chorus activity (Burnham et al., 2023; Ainslie, 2005) and higher wind speeds have shown negative correlation to peak chorus levels (McWilliam et al., 2017). During winter recordings, wind speed steadily increased from a daily average of 3.76 knots (Beaufort scale 2) on 9/1/22, to reach a peak of 10.8 knots (Beaufort scale 4) on 20/1/22 (Supplementary Figure S4). A change in underwater conditions also occurs, during a period of higher wind, on the 15th January (Supplementary Figure S4). During higher wind days, the near-surface sound duct could be disrupted (Ainslie, 2005), allowing sound to travel less efficiently horizontally, influencing the received levels of winter choruses at the shallow hydrophone, or more distant mesophotic sites. If the conditions were more calm, recordings of choruses may present different received levels and spectral qualities. The varying effects of environmental factors makes it difficult to compare other fish sounds and choruses to identify the source species.
As is common with many fish choruses and sounds globally, there is no current evidence for a species responsible for the winter choruses. The tonal, harmonic sound structure of Chorus F (Figure 3A, C) shows similarity to the characteristics of sound produced actively in courtship and/or spawning coordination, as observed in Terapontidae species (Parsons et al., 2017; Laxminarsimha et al., 2020; Mahanty et al., 2015; Parsons et al., 2016b). The harmonic structure and frequency components (600 Hz–2.3 kHz) of Chorus F share similarities with choruses linked to swim bladder-driven sound production (Parsons et al., 2017). However, while comparison of waveforms (Figure 3B) show a similar pattern to Chorus VI (Parsons et al., 2017) in Western Australia, spectral comparison reveal differences in harmonic frequencies and peak values (568.8 ± 19.5 Hz for F vs. 440 Hz for VI), either suggesting a distinct source or difference in propagation conditions, or geography (Parmentier et al., 2005). Known Red Sea soniferous fish families which spawn during winter (e.g., Haemulidae, Gerridae, and Mugilidae (El-Regal, 2013; Bogorodsky and Randall, 2019)) have not been documented to produce harmonic choruses of this type (Amorim, 2006; Parmentier and Fine, 2016; Tricas and Boyle, 2014). Overall, Chorus F likely represents a key aggregation site off Thuwal, and underscores the spatial extent of fish chorusing in both mesophotic and shallow habitats.
Lastly, Chorus E present on all mesophotic sites except for KA_DS3, was short in length (10 min, Table 2) and sounded similar to the summer Choruses C and III. Although individual sounds could not be separated, Chorus E appeared to be comprised of pulsed calls, with a peak frequency within the range of III and C, of 671.3 ± 320 Hz. If attributed to the same source species, this may signal either migration or year-round residency with fluctuating numbers of individuals (Parsons et al., 2017). However, as fish choruses and sounds can show very similar characteristics (Hawkins et al., 2023), there is a chance this chorus can be attributed to a distinct species.
Summer recordings revealed the majority of choruses seen across the two seasons (n = 8). Al Fahal sites recorded adjacent Choruses C and D, at respective peak frequencies of 1039.3 ± 484.8 Hz, and850 ± 164.6 Hz), both characterized by dampened pulses or “croaks” (Table 2; Figures 5, 6). On KAEC sites, Chorus III exhibited a similar sound, at a peak frequency of 848 ± 19.6 Hz. The variation in received levels (Figure 6) and spectral differences (Supplementary Table S1) and timing were enough to label them different choruses.
Notably, Choruses C and D were present at both mesophotic and shallow sites at Al Fahal, whereas Chorus III was restricted to KAEC mesophotic sites (Figures 5, 6). These spatial differences could reflect site-specific shoaling behavior, differences in reef topography, orientation relative to the hydrophone, and sound propagation factors as discussed in the previous Section (4.1.1). In the summer, the Red Sea possesses a downward-refracting SSP where sound speed increases with depth, and there is a reduction in surface reflection of sound without the presence of a duct like in winter (Larayedh et al., 2024). This potentially means that soundwaves refracted downward are eventually absorbed or scattered by the seafloor, causing propagation loss (Erbe et al., 2022). Due to these conditions, it would be less likely for a chorus originating deeper than 200 m to be received at such high levels on KA_DS3 at 73.2 m (117.5 dB re µPa2, Supplementary Table S1). Chorus C, shares similar temporal characteristics as III (21:32 h ± 00:23–00:40 h ± 00:09, vs. 21:43 h ± 1:10–00:42 h ± 00:50, respectively), but exhibits a much lower received level of 97.7 ± 4.1 dB re µPa2 (III = 106 ± 13.2 dB re µPa2). Spectrally, Chorus C has a higher peak frequency (1,039.3 ± 484.8 Hz) than Chorus III (848 ± 19.6 Hz), the latter which is more similar to Chorus D (850 ± 164.6 Hz) (Table 2). As previously noted (Section 4.2), wind can also affect sound propagation (Ainslie, 2005) and contribute to ambient noise levels in the low frequencies. The average wind speed declined steadily from 21/7/22 at 8.8 knots (Beaufort scale 3), down to 4.3 knots (Beaufort scale of 2) on 31/7/22 (Supplementary Figure S4), and could have potentially scattered frequencies to reduce propagation of deep choruses to the shallow reef sites.
Alternatively, a short, consistent temporal gap between Chorus C and D suggests they may originate from two distinct species, although vertical movement within the water column by a single species remains a possibility (Hawkins et al., 2023; McCauley and Cato, 2016). A previous study observed frequency shifts in fish choruses as shoals moved higher in the water column, consistent with swim bladder-driven sounds (McCauley and Cato, 2016). If multiple species are responsible for these choruses, this pattern (C to D chorusing) may exemplify the acoustic niche hypothesis, wherein organisms partition vocal frequencies to avoid overlap while maximizing communication efficiency (Krause, 1993; Bertucci et al., 2020).
White noise Choruses U1 and U2 add further complexity to the mesophotic soundscape. Like in winter, Chorus U1 is most likely the attenuated sound of the shallow invertebrate chorus (Butler et al., 2021; Lillis and Mooney, 2018), as it coincides with the timing of greatest energy present in the shallow invertebrate chorus, just after dusk (Figures 3, 5). Chorus U1 is slightly more evident in winter than summer, likely due to the different sound propagation conditions, with the near-surface sound duct potentially increasing sound propagation from the shallow chorus (Larayedh et al., 2024). This difference is most likely not related to the difference in lunar phase: snapping shrimp sound pressure levels have been seen to increase during new moon (Gordon et al., 2019), as was observed during summer recordings (third quarter to new moon; 0%–34.5% illumination. Figure 5; Supplementary Table S4). Chorus U2, unlike U1 is lower in frequency (peak of 935 ± 63.6 Hz vs. 3,465 ± 91.9 Hz) and is only present on mesophotic sites. Starting at the end of Chorus III, one potential reason for Chorus U2 may arise from hydrodynamic shoal movements, possibly reflecting changes in position within the water column. Chorus U2 appearing at the end of Chorus III for 4:10 h, could suggest the source as hydrodynamic noise from diel vertical migration (DVM) by nocturnally active species like Myctophidae. DVM is whereby individuals stay at mesopelagic depths during the day to avoid predation, and travel to surface layers at night to feed (Dypvik and Kaartvedt, 2013; Klevjer et al., 2012). Alternatively, the farfield effect on distant, overlapping calls of a chorus, whether passive through feeding or active for courtship/spawning, may create a diffuse acoustic haze, or white noise (Cato, 1978; McCauley and Cato, 2016; McCauley and Cato, 1998). Echogram studies near the KAEC canyon (22.3°N, 39.03°E) detected deep scattering layers (DSL) dominated by Benthosoma pterotum (Myctophidae) ascending to feed near the surface at dusk (Dypvik and Kaartvedt, 2013; Klevjer et al., 2012; Sobradillo et al., 2022). The synchronized timing of Chorus III with this migration and the white noise Chorus U2 (Figure 5; Table 2) aligns with the white noise, post-chorus (Moulton, 1960). More likely, these sounds could potentially be by-products of foraging, or used for keeping loose structure in the school (McCauley and Cato, 2016). A long-term soundscape study explored potential Myctophid choruses, spanning 1–5 kHz, peaking in levels similarly between 0.75–5.25 h after sunset (Chorus III: 4 h after sunset), and composed of dampened pulses (McCauley and Cato, 2016). Furthermore, these choruses were not correlated with the moon phase, but rather periods of high primary productivity. Further studies would be required to assess their sound production characteristics and their link with primary productivity in the Red Sea.
An additional potential source for Choruses C, D and III includes the Carangidae family, which is known to produce grunt-like sounds (Parsons et al., 2016a; Carvalho et al., 2019). Species such as Caranx ignobilis (giant trevally) are important commercial fish in the region and have been observed aggregating at mesophotic depths (40 m) during summer spawning seasons in the Red Sea (Rice et al., 2020; Bogorodsky and Randall, 2019; Sadovy De Mitcheson et al., 2008). This species’ vertical shoaling behavior (Daly et al., 2018; Zhou et al., 2024) aligns well with the spatial and temporal features observed in these choruses.
Chorus II bears striking similarities to a chorus described in multiple studies (McCauley, 2012; Parsons et al., 2017) and attributed to crepuscular, planktivorous fish families Holocentridae, Priacanthidae and Apogonidae, and most likely related to feeding activity. As noted by the authors, this chorus shows spectral peaks between 600 and 900 Hz and contains a notch around 1,000 Hz, which suggests the shoal’s position near the surface (Parsons et al., 2017). The origin point of this chorus in our study is unknown. Chorus II shows variable spectral qualities in the mesophotic sites as compared to the shallow sites, contributing to the variable average low frequency of 362.5 ± 109.6 Hz to high frequency of 1,646 ± 848.7 Hz, and a peak frequency of 698.3 ± 507.6 Hz (Table 2).
Although not the primary focus of this study, vessel noise was found to be pervasive across all sites, originating mainly from large cargo ships and small fishing boats (highlighted in Figure 4). The proximity of both locations to the King Abdullah Economic City (KAEC) shipping lane (∼5 km away, Figure 1B) resulted in multiple daily detections, particularly at mesophotic sites, where low-frequency noise (40–200 Hz) reached levels up to 110 dB re µPa2/Hz (Figures 3–5). Vessel noise has been shown to significantly reduce fish communication space—for example, a large vessel <10 km away reduced the communication range of Pempheris adspersa by up to 99% (Putland et al., 2018). Most likely due to the presence of the near-surface sound duct (Section 4.1.1) focusing shallow noise (Larayedh et al., 2024), shipping noise exhibited increased effect on winter soundscapes than during summer, despite cooler temperatures. During the peak winter Chorus F, vessel activity coincided with fish sound production, inflating ambient noise levels within the chorus frequency band by up to 10 dB and masking the chorusing signal (Figures 3, 4). Similarly, in summer, nocturnal choruses were frequently overlapped by vessel noise. This pattern mirrors findings from coral reef studies in Japan, where deeper sites experienced disproportionately higher vessel noise levels compared to shallower habitats (Lin et al., 2021; Akamatsu et al., 2018).
Chronic vessel noise has detrimental effects on fish behavior, health, and reproductive success (Holles et al., 2013; Nedelec et al., 2017; Simpson et al., 2016). Increased ambient noise reduces the effective range of synchronized vocalizations needed for spawning coordination (Zhang and Katsnelson, 2021), which can normally span kilometers (Radford et al., 2023). In response, some species increase call volumes (up to 7–9 dB), potentially overexerting energy and reducing reproductive efficiency (Schafer, 1969; Carvalho et al., 2019; De Jong et al., 2018; Gavrilov and Parsons, 2014). Additionally, vessel noise can disrupt larval fish settlement by interfering with phonotaxis and masking reef acoustic cues critical for navigation (Montgomery et al., 2006; Vermeij et al., 2010; Rowell et al., 2017; Lobel, 1992). Further research into Red Sea fish chorusing should quantify the impacts of vessel noise on fish behavior and communication. As coastal development accelerates under Saudi Arabia’s “Vision 2030,” including rising construction and recreational watercraft noise, it is critical to establish baselines like these to detect and mitigate impacts on fish activity and habitat use.
As discussed, the results presented may be reflective of fish location and activity, but are also influenced heavily by ambient conditions, bathymetry, weather influences, and how these affect sound propagation and receiver geometry. These should be taken into account when planning future field studies (Biggs and Erisman, 2021). Within this study we have recorded the ambient conditions at the point of the receiver, but future studies in the central Red Sea could collect temperature and salinity profiles to create insitu SSPs, as well as use bathymetric information for sound propagation modelling (Erbe et al., 2022; Larsen and Radford, 2018), and investigate correlations of primary productivity through satellite imagery and insitu measurements. While depth increases complexity of calculations, a hydrophone array, or 4-element hydrophone, could help in localizing the shoal using Time Difference of Arrival (TDoA) (Erbe et al., 2022). Consequently, echo sounding using a boat sonar may be used to located the shoal. To then visually confirm the identity of the chorusing species, a PAM set-up equipped with an underwater camera (with lights or preferably infrared) could also be deployed.
This study demonstrates the utility of PAM in identifying critical habitats for fish, through cataloging seasonal and spatial variability in fish soundscapes. As the eleven documented choruses were most likely related to foraging, spawning and/or courtship, this establishes the study area of the central Red Sea as an important habitat for fish. Cataloging unknown choruses and linking them to specific taxa remain priorities for building global soundscape baselines. These efforts will be instrumental in designing effective spatial management strategies, protecting fragile reef ecosystems, and ensuring the sustainability of fish populations under increasing environmental pressures.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethical approval was not required for the study involving animals in accordance with the local legislation and institutional requirements. The research is non-invasive, passive acoustic monitoring that does not affect animals.
MN-H: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing–original draft, Writing–review and editing. AP: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Software, Validation, Visualization, Writing–original draft, Writing–review and editing. AS: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Project administration, Software, Writing–original draft, Writing–review and editing Validation, Visualization. MW: Investigation, Software, Visualization, Writing–original draft, Writing–review and editing. FM: Conceptualization, Investigation, Software, Visualization, Writing–original draft, Writing–review and editing. FB: Conceptualization, Funding acquisition, Investigation, Resources, Supervision, Writing–original draft, Writing–review and editing. CD: Conceptualization, Funding acquisition, Investigation, Methodology, Resources, Supervision, Writing–original draft, Writing - review and editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This research was funded by baseline funding by King Abdullah University of Science and Technology provided to CD and FB.
We thank the CMR staff, especially Andres Espinoza, Ramzi S. Al-Jahdali, and David Atienza for invaluable help in deployment and troubleshooting gear, as well as CMR skippers Mohammed Y. Alsheik, Issam Al-Jahdali, Eleazar G. Gonomit, Walid Al-Jahdali, Gazi Al-Jahdali, Abdullah Al-Jahdali; and fieldwork assistance from Nayra Pluma-Guerrero and Jennifer Thompson. We also thank Vanita Dighe and Eleonora Re for help with water sample analysis, as well as Miles Parsons for initial advice on fish choruses and using CHORUS, and Jennifer Miksis-Olds for initial discussion on identifying currents, anthropogenic noise and artefacts within soundscapes. Lastly (but certainly not least), thank you to both reviewers and Editor Lucia Di Iorio, whose valuable comments and advice helped improve the manuscript greatly. During preparation of the manuscript, ChatGPT (4o) was used to draft some of the code utilized in the work, as well as improve the readability of some paragraphs. All work was subsequently reviewed by the author/s, who take full responsibility for the content of the published article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/frsen.2025.1482244/full#supplementary-material
Supplementary audio 1 | Chorus_C_2230PM_. Summer Chorus C at AFDS2, 22:30PM.
Supplementary audio 2 | Chorus_III_DS3_2307PM_. Summer Chorus III at KADS3, 23:07PM.
Supplementary audio 3 | ChorusD_0225AM_. Summer Chorus D at AFDS2, 02:25AM.
Supplementary audio 4 | ChorusF_KADS3_2031PM_. Winter Chorus F at KADS3, 20:31PM.
Aalbers, S. A., and Drawbridge, M. A. (2008). White seabass spawning behavior and sound production. Trans. Am. Fish. Soc. 137 (2), 542–550. doi:10.1577/t04-058.1
Ainslie, M. A. (2005). Effect of wind-generated bubbles on fixed range acoustic attenuation in shallow water at 1–4kHz. J. Acoust. Soc. Am. 118 (6), 3513–3523. doi:10.1121/1.2114527
T. Akamatsu, T.-H. Lin, Y. Tsao, F. Sinniger, and S. Harii (2018). Coral reef soundscape to measure the species distribution and biodiversity (OCEANS-MTS/IEEE Kobe Techno-Oceans OTO; IEEE).
Anderson, E. R. (2021). The role of backreef soundscapes and their spatial structure for recruitment of tropical marine larvae.
Bertucci, F., Lejeune, P., Payrot, J., and Parmentier, E. (2015). Sound production by dusky grouper Epinephelus marginatus at spawning aggregation sites. J. Fish Biol. 87 (2), 400–421. doi:10.1111/jfb.12733
Bertucci, F., Maratrat, K., Berthe, C., Besson, M., Guerra, A. S., Raick, X., et al. (2020). Local sonic activity reveals potential partitioning in a coral reef fish community. Oecologia 193, 125–134. doi:10.1007/s00442-020-04647-3
Bertucci, F., Parmentier, E., Berthe, C., Besson, M., Hawkins, A. D., Aubin, T., et al. (2017). Snapshot recordings provide a first description of the acoustic signatures of deeper habitats adjacent to coral reefs of Moorea. PeerJ 5, e4019. doi:10.7717/peerj.4019
Berumen, M. L., Hoey, A., Bass, W. H., Bouwmeester, J., Catania, D., Cochran, J., et al. (2013). The status of coral reef ecology research in the Red Sea. Coral Reefs 32, 737–748. doi:10.1007/s00338-013-1055-8
Biggs, C. R., and Erisman, B. E. (2021). Transmission loss of fish spawning vocalizations and the detection range of passive acoustic sampling in very shallow estuarine environments. Estuaries Coasts 44 (7), 2026–2038. doi:10.1007/s12237-021-00914-5
Bogorodsky, S. V., and Randall, J. E. (2019). “Endemic fishes of the Red Sea,” in Oceanographic and biological aspects of the Red Sea, 239–265.
Bongaerts, P., Ridgway, T., Sampayo, E., and Hoegh-Guldberg, O. (2010). Assessing the ‘deep reef refugia’hypothesis: focus on Caribbean reefs. Coral reefs. 29, 309–327. doi:10.1007/s00338-009-0581-x
Borie-Mojica, A., Rezende, S. M., Padovani, F. B., Maida, M., and Travassos, P. (2022). Fish chorus and vessel noise in a marine protected coastal reef vary with lunar phase. Environ. Biol. Fishes 105 (5), 575–587. doi:10.1007/s10641-022-01267-z
Burnham, R. E., Vagle, S., Thupaki, P., and Thornton, S. J. (2023). Implications of wind and vessel noise on the sound fields experienced by southern resident killer whales Orcinus orca in the Salish Sea. Endanger. Species Res. 50, 31–46. doi:10.3354/esr01217
Butler, J., Pagniello, CMLS, Jaffe, J. S., Parnell, P. E., and Širović, A. (2021). Diel and seasonal variability in kelp forest soundscapes off the southern California coast. Front. Mar. Sci. 8 (357). doi:10.3389/fmars.2021.629643
Carriço, R., Silva, M. A., Menezes, G. M., Fonseca, P. J., and Amorim, M. C. P. (2019). Characterization of the acoustic community of vocal fishes in the Azores. PeerJ 7, e7772. doi:10.7717/peerj.7772
Carvalho, S., Kürten, B., Krokos, G., Hoteit, I., and Ellis, J. (2019). The red sea. World seas: An environmental evaluation: Elsevier, 49–74.
Cato, D. H. (1978). Marine biological choruses observed in tropical waters near Australia. J. Acoust. Soc. Am. 64 (3), 736–743. doi:10.1121/1.382038
Chérubin, L. M., Dalgleish, F., Ibrahim, A. K., Schärer-Umpierre, M., Nemeth, R. S., Matthews, A., et al. (2020). Fish spawning aggregations dynamics as inferred from a novel, persistent presence robotic approach. Front. Mar. Sci. 6, 779. doi:10.3389/fmars.2019.00779
Daly, R., Daly, C., Bennett, R., Cowley, P., Pereira, M., and Filmalter, J. (2018). Quantifying the largest aggregation of giant trevally Caranx ignobilis (Carangidae) on record: implications for management. Afr. J. Mar. Sci. 40 (3), 315–321. doi:10.2989/1814232x.2018.1496950
De Jong, K., Amorim, M. C. P., Fonseca, P. J., Fox, C. J., and Heubel, K. U. (2018). Noise can affect acoustic communication and subsequent spawning success in fish. Environ. Pollut. 237, 814–823. doi:10.1016/j.envpol.2017.11.003
Dibble, D. S., Van Alstyne, K. R., and Ridgway, S. (2016). Dolphins signal success by producing a victory squeal. Int. J. Comp. Psychol. 29 (1). doi:10.46867/ijcp.2016.29.00.14
D’spain, G., and Batchelor, H. (2006). Observations of biological choruses in the Southern California Bight: a chorus at midfrequencies. J. Acoust. Soc. Am. 120 (4), 1942–1955. doi:10.1121/1.2338802
Duarte, C. M., Chapuis, L., Collin, S. P., Costa, D. P., Devassy, R. P., Eguiluz, V. M., et al. (2021). The soundscape of the Anthropocene ocean. Science 371 (6529), eaba4658. doi:10.1126/science.aba4658
Dypvik, E., and Kaartvedt, S. (2013). Vertical migration and diel feeding periodicity of the skinnycheek lanternfish (Benthosema pterotum) in the Red Sea. Deep Sea Res. Part I Oceanogr. Res. Pap. 72, 9–16. doi:10.1016/j.dsr.2012.10.012
El-Regal, M. A. (2013). Spawning seasons, spawning grounds and nursery grounds of some Red Sea fishes.
Erbe, C., Duncan, A., Hawkins, L., Terhune, J. M., and Thomas, J. A. (2022). “Introduction to acoustic terminology and signal processing,” in Exploring animal behavior through sound: volume 1: methods (Cham: Springer International Publishing), 111–152.
Erisman, B. E., and Rowell, T. J. (2017). A sound worth saving: acoustic characteristics of a massive fish spawning aggregation. Biol. Lett. 13 (12), 20170656. doi:10.1098/rsbl.2017.0656
Gannon, D. P., Barros, N. B., Nowacek, D. P., Read, A. J., Waples, D. M., and Wells, R. S. (2005). Prey detection by bottlenose dolphins, Tursiops truncatus: an experimental test of the passive listening hypothesis. Anim. Behav. 69 (3), 709–720. doi:10.1016/j.anbehav.2004.06.020
Gavrilov, A., and Parsons, M. (2014). A Matlab tool for the characterisation of recorded underwater sound (CHORUS). Acoust. Aust. 42 (3), 190–196.
Gordon, T. A., Radford, A. N., Davidson, I. K., Barnes, K., McCloskey, K., Nedelec, S. L., et al. (2019). Acoustic enrichment can enhance fish community development on degraded coral reef habitat. Nat. Commun. 10 (1), 5414. doi:10.1038/s41467-019-13186-2
Havlik, M. N., Predragovic, M., and Duarte, C. M. (2022). State of play in marine soundscape assessments. Front. Mar. Sci. 9. doi:10.3389/fmars.2022.919418
Hawkins, L. A., Saunders, B. J., Landero Figueroa, M. M., McCauley, R. D., Parnum, I. M., Parsons, M. J., et al. (2023). Habitat type drives the spatial distribution of Australian fish chorus diversity. J. Acoust. Soc. Am. 154 (4), 2305–2320. doi:10.1121/10.0021330
Heimrich, A. F., Halliday, W. D., Frouin-Mouy, H., Pine, M. K., Juanes, F., and Insley, S. J. (2021). Vocalizations of bearded seals (Erignathus barbatus) and their influence on the soundscape of the western Canadian Arctic. Mar. Mammal Sci. 37 (1), 173–192. doi:10.1111/mms.12732
Herzing, D. L., and Elliser, C. R. (2014). Nocturnal feeding of Atlantic spotted dolphins (Stenella frontalis) in the Bahamas. Mar. Mamm. Sci. 30 (1), 367–373. doi:10.1111/mms.12016
Hoare, D. J., Ruxton, G. D., Godin, J. G. J., and Krause, J. (2000). The social organization of free-ranging fish shoals. Oikos 89 (3), 546–554. doi:10.1034/j.1600-0706.2000.890314.x
Holles, S., Simpson, S. D., Radford, A. N., Berten, L., and Lecchini, D. (2013). Boat noise disrupts orientation behaviour in a coral reef fish. Mar. Ecol. Prog. Ser. 485, 295–300. doi:10.3354/meps10346
Ikegami, T., Takeuchi, Y., Hur, S.-P., and Takemura, A. (2014). Impacts of moonlight on fish reproduction. Mar. genomics 14, 59–66. doi:10.1016/j.margen.2013.11.007
ISO (2017). Underwater acoustics—terminology. Geneva, Switzerland: International Organization for Standardization.
Janik, V. M. (2000). Whistle matching in wild bottlenose dolphins (Tursiops truncatus). Science 289 (5483), 1355–1357. doi:10.1126/science.289.5483.1355
Jones, I. T., Suca, J., Llopiz, J., and Mooney, T. A. (2017). Characterizing soundscapes and larval fish settlement in tropical seagrass and mangrove habitats. J. Acoust. Soc. Am. 141 (5), 4003. doi:10.1121/1.4989181
Kaatz, I., Rice, A., and Lobel, P. (2017). “How fishes use sound: quiet to loud and simple to complex signaling,” in Reference model in life science: encyclopedia of physiology of fishes. Elsevier, 1–12.
Kahng, S., Garcia-Sais, J., Spalding, H., Brokovich, E., Wagner, D., Weil, E., et al. (2010). Community ecology of mesophotic coral reef ecosystems. Coral Reefs 29, 255–275. doi:10.1007/s00338-010-0593-6
Kaplan, M. B., Mooney, T. A., Partan, J., and Solow, A. R. (2015). Coral reef species assemblages are associated with ambient soundscapes. Mar. Ecol. Prog. Ser. 533, 93–107. doi:10.3354/meps11382
Kattan, A., Coker, D. J., Nowicki, J. P., Williams, C. T., and Berumen, M. L. (2022). Putative spawning aggregations of giant trevally in the Red Sea.
Klevjer, T. A., Torres, D. J., and Kaartvedt, S. (2012). Distribution and diel vertical movements of mesopelagic scattering layers in the Red Sea. Mar. Biol. 159, 1833–1841. doi:10.1007/s00227-012-1973-y
Krause, B. L. (1993). The niche hypothesis: a virtual symphony of animal sounds, the origins of musical expression and the health of habitats. Soundscape Newsl. 6 (5).
Ladich, F. (2019). Ecology of sound communication in fishes. Fish Fish. 20 (3), 552–563. doi:10.1111/faf.12368
Larayedh, R., Cornuelle, B. D., Krokos, G., and Hoteit, I. (2024). Numerical investigation of shipping noise in the Red Sea. Sci. Rep. 14 (1), 5851. doi:10.1038/s41598-024-56523-2
Larsen, O. N., and Radford, C. (2018). “Acoustic conditions affecting sound communication in air and underwater,” in Effects of anthropogenic noise on animals, 109–144.
Laxminarsimha, C. K., Sreekanth, G., Deshmukh, M., and Sharma, N. (2020). Marine soundscape and fish chorus in an archipelago ecosystem comprising bio-diverse tropical islands off Goa Coast, India. Aquat. Ecol. 54, 475–493. doi:10.1007/s10452-020-09754-0
Lecchini, D., Bertucci, F., Gache, C., Khalife, A., Besson, M., Roux, N., et al. (2018). Boat noise prevents soundscape-based habitat selection by coral planulae. Sci. Rep. 8 (1), 9283–9289. doi:10.1038/s41598-018-27674-w
Leis, J., Carson-Ewart, B., Hay, A., and Cato, D. (2003). Coral-reef sounds enable nocturnal navigation by some reef-fish larvae in some places and at some times. J. Fish Biol. 63 (3), 724–737. doi:10.1046/j.1095-8649.2003.00182.x
Lesser, M. P., Slattery, M., and Leichter, J. J. (2009). Ecology of mesophotic coral reefs. J. Exp. Mar. Biol. Ecol. 375 (1-2), 1–8. doi:10.1016/j.jembe.2009.05.009
Lillis, A., and Mooney, T. A. (2018). Snapping shrimp sound production patterns on Caribbean coral reefs: relationships with celestial cycles and environmental variables. Coral Reefs 37 (2), 597–607. doi:10.1007/s00338-018-1684-z
Lin, T.-H., Akamatsu, T., Sinniger, F., and Harii, S. (2021). Exploring coral reef biodiversity via underwater soundscapes. Biol. Conserv. 253, 108901. doi:10.1016/j.biocon.2020.108901
Lindseth, A. V., and Lobel, P. S. (2018). Underwater soundscape monitoring and fish bioacoustics: a review. Fishes 3 (3), 36. doi:10.3390/fishes3030036
Lobel, P. S. (1992). Sounds produced by spawning fishes. Environ. Biol. Fishes 33, 351–358. doi:10.1007/bf00010947
Lobel, P. S. (2002). Diversity of fish spawning sounds and the application of passive acoustic monitoring. Bioacoustics 12 (2-3), 286–289. doi:10.1080/09524622.2002.9753724
Looby, A., Cox, K., Bravo, S., Rountree, R., Juanes, F., Reynolds, L. K., et al. (2022). A quantitative inventory of global soniferous fish diversity. Rev. Fish Biol. Fish. 32 (2), 581–595. doi:10.1007/s11160-022-09702-1
Luczkovich, J. J., and Keusenkothen, M. A. (2008). Can longspine squirrelfish hear bottlenose dolphin? Bioacoustics. Bioacoustics 17 (1-3), 75–77. doi:10.1080/09524622.2008.9753771
Luczkovich, J. J., Mann, D. A., and Rountree, R. A. (2008a). Passive acoustics as a tool in fisheries science. Trans. Am. Fish. Soc. 137 (2), 533–541. doi:10.1577/t06-258.1
Luczkovich, J. J., Pullinger, R. C., Johnson, S. E., and Sprague, M. W. (2008b). Identifying sciaenid critical spawning habitats by the use of passive acoustics. Trans. Am. Fish. Soc. 137 (2), 576–605. doi:10.1577/t05-290.1
Lyon, R., Eggleston, D., Bohnenstiehl, D., Layman, C., Ricci, S., and Allgeier, J. (2019). Fish community structure, habitat complexity, and soundscape characteristics of patch reefs in a tropical, back-reef system. Mar. Ecol. Prog. Ser. 609, 33–48. doi:10.3354/meps12829
Mahanty, M., Kannan, R., Harikrishanan, C., and Latha, G. (2015). Terapon Theraps chorus observed in shallow water environment in the southeastern Arabian sea.
McCauley, R. (2012). “Fish choruses from the Kimberley, seasonal and lunar links as determined by long term sea noise monitoring,” in Proceedings of the acoustical society of Australia.
McCauley, R. D., and Cato, D. H. (1998). Evening fish choruses near coral reef systems in the Great Barrier Reef, Australia. J. Acoust. Soc. Am. 103 (5_Suppl. ment), 2864. doi:10.1121/1.421614
McCauley, R. D., and Cato, D. H. (2000). Patterns of fish calling in a nearshore environment in the Great Barrier Reef. Philosophical Trans. R. Soc. Lond. Ser. B Biol. Sci. 355 (1401), 1289–1293. doi:10.1098/rstb.2000.0686
McCauley, R. D., and Cato, D. H. (2016). Evening choruses in the Perth Canyon and their potential link with Myctophidae fishes. J. Acoust. Soc. Am. 140 (4), 2384–2398. doi:10.1121/1.4964108
McWilliam, J. N., McCauley, R. D., Erbe, C., and Parsons, M. J. G. (2017). Patterns of biophonic periodicity on coral reefs in the Great Barrier Reef. Sci. Rep. 7 (1), 17459. doi:10.1038/s41598-017-15838-z
Merchant, N. D., Fristrup, K. M., Johnson, M. P., Tyack, P. L., Witt, M. J., Blondel, P., et al. (2015). Measuring acoustic habitats. Methods Ecol. Evol. 6 (3), 257–265. doi:10.1111/2041-210x.12330
Montgomery, J. C., Jeffs, A., Simpson, S. D., Meekan, M., and Tindle, C. (2006). Sound as an orientation cue for the pelagic larvae of reef fishes and decapod crustaceans. Adv. Mar. Biol. 51, 143–196. doi:10.1016/s0065-2881(06)51003-x
Montie, E. W., Hoover, M., Kehrer, C., Yost, J., Brenkert, K., O’Donnell, T., et al. (2017). Acoustic monitoring indicates a correlation between calling and spawning in captive spotted seatrout (Cynoscion nebulosus). PeerJ 5, e2944. doi:10.7717/peerj.2944
Mooney, T. A., Di Iorio, L., Lammers, M., Lin, T.-H., Nedelec, S. L., Parsons, M., et al. (2020). Listening forward: approaching marine biodiversity assessments using acoustic methods. R. Soc. open Sci. 7 (8), 201287. doi:10.1098/rsos.201287
Moulton, J. M. (1960). Swimming sounds and the schooling of fishes. Biol. Bull. 119 (2), 210–223. doi:10.2307/1538923
Nedelec, S. L., Mills, S. C., Radford, A. N., Beldade, R., Simpson, S. D., Nedelec, B., et al. (2017). Motorboat noise disrupts co-operative interspecific interactions. Sci. Rep. 7 (1), 6987. doi:10.1038/s41598-017-06515-2
Nedelec, S. L., Simpson, S. D., Holderied, M., Radford, A. N., Lecellier, G., Radford, C., et al. (2015). Soundscapes and living communities in coral reefs: temporal and spatial variation. Mar. Ecol. Prog. Ser. 524, 125–135. doi:10.3354/meps11175
Parmentier, E., and Fine, M. L. (2016). “Fish sound production: insights,” in Vertebrate sound production and acoustic communication, 19–49.
Parmentier, E., Lagardere, J.-P., Vandewalle, P., and Fine, M. (2005). Geographical variation in sound production in the anemonefish Amphiprion akallopisos. Proc. R. Soc. B Biol. Sci. 272 (1573), 1697–1703. doi:10.1098/rspb.2005.3146
M. Parsons, C. Erbe, R. McCauley, J. McWilliam, S. Marley, A. Gavrilovet al. (2016a). “Long-term monitoring of soundscapes and deciphering a usable index: examples of fish choruses from Australia,” Proceedings of meetings on acoustics (Acoustical Society of America, AIP Publishing LLC – FMS, NY).
Parsons, M., McCauley, R., and Thomas, F. (2013). The sounds of fish off cape naturaliste, western Australia. Acoust. Aust. 41 (1), 58–64.
Parsons, M. J., Salgado, K. C. P., Recalde-Salas, A., and McCauley, R. D. (2017). Fish choruses off port hedland, western Australia. Bioacoustics 26 (2), 135–152. doi:10.1080/09524622.2016.1227940
Parsons, M. J., Salgado-Kent, C. P., Marley, S. A., Gavrilov, A. N., and McCauley, R. D. (2016b). Characterizing diversity and variation in fish choruses in Darwin Harbour. ICES J. Mar. Sci. 73 (8), 2058–2074. doi:10.1093/icesjms/fsw037
Pérez-Rosales, G., Pichon, M., Rouzé, H., Villéger, S., Torda, G., Bongaerts, P., et al. (2022). Mesophotic coral ecosystems of French Polynesia are hotspots of alpha and beta generic diversity for scleractinian assemblages. Divers. Distributions 28 (7), 1391–1403. doi:10.1111/ddi.13549
Picciulin, M., Bolgan, M., and Burchardt, L. S. (2024). Rhythmic properties of Sciaena umbra calls across space and time in the Mediterranean Sea. Plos one 19 (2), e0295589. doi:10.1371/journal.pone.0295589
Pijanowski, B. C., Farina, A., Gage, S. H., Dumyahn, S. L., and Krause, B. L. (2011). What is soundscape ecology? An introduction and overview of an emerging new science. Landsc. Ecol. 26, 1213–1232. doi:10.1007/s10980-011-9600-8
Pine, M. K., Wang, D., Porter, L., and Wang, K. (2018). Investigating the spatiotemporal variation of fish choruses to help identify important foraging habitat for Indo-Pacific humpback dolphins, Sousa chinensis. ICES J. Mar. Sci. 75 (2), 510–518. doi:10.1093/icesjms/fsx197
Popper, A. N., and Hawkins, A. D. (2019). An overview of fish bioacoustics and the impacts of anthropogenic sounds on fishes. J. fish Biol. 94 (5), 692–713. doi:10.1111/jfb.13948
Putland, R. L., Merchant, N. D., Farcas, A., and Radford, C. A. (2018). Vessel noise cuts down communication space for vocalizing fish and marine mammals. Glob. change Biol. 24 (4), 1708–1721. doi:10.1111/gcb.13996
Pyle, R. L., and Copus, J. M. (2019). Mesophotic coral ecosystems: introduction and overview. Mesophotic coral Ecosyst., 3–27. doi:10.1007/978-3-319-92735-0_1
Radford, C. A., Constantine, R., Pine, M. K., and Farcas, A. (2023). Impact of small boat sound on the listening space of Pempheris adspersa, Forsterygion lapillum, Alpheus richardsoni and Ovalipes catharus. Sci. Rep. 13, 7007. doi:10.1038/s41598-023-33684-0
Raick, X., Di Iorio, L., Gervaise, C., Lossent, J., Lecchini, D., and Parmentier, É. (2021). From the reef to the ocean: revealing the acoustic range of the biophony of a coral reef (moorea island, French Polynesia). J. Mar. Sci. Eng. 9 (4), 420. doi:10.3390/jmse9040420
Raick, X., Di Iorio, L., Lecchini, D., Gervaise, C., Hédouin, L., Consortium, U. T. P., et al. (2023). Fish sounds of photic and mesophotic coral reefs: variation with depth and type of island. Coral Reefs 42 (2), 285–297. doi:10.1007/s00338-022-02343-7
Raick, X., Parmentier, É., Gervaise, C., Lecchini, D., Pérez-Rosales, G., Rouzé, H., et al. (2024). Invertebrate sounds from photic to mesophotic coral reefs reveal vertical stratification and diel diversity. Oecologia 205, 307–322. doi:10.1007/s00442-024-05572-5
Remage-Healey, L., Nowacek, D. P., and Bass, A. H. (2006). Dolphin foraging sounds suppress calling and elevate stress hormone levels in a prey species, the Gulf toadfish. J. Exp. Biol. 209 (22), 4444–4451. doi:10.1242/jeb.02525
Rice, A. N., Farina, S. C., Makowski, A. J., Kaatz, I. M., Lobel, P. S., Bemis, W. E., et al. (2020). Evolution and ecology in widespread acoustic signaling behavior across fishes. bioRxiv [Preprint] 2020 (09. 14), 296335. doi:10.1101/2020.09.14.296335
Rossi, T., Connell, S. D., and Nagelkerken, I. (2017). The sounds of silence: regime shifts impoverish marine soundscapes. Landsc. Ecol. 32 (2), 239–248. doi:10.1007/s10980-016-0439-x
Rowell, T. J., Demer, D. A., Aburto-Oropeza, O., Cota-Nieto, J. J., Hyde, J. R., and Erisman, B. E. (2017). Estimating fish abundance at spawning aggregations from courtship sound levels. Sci. Rep. 7 (1), 3340. doi:10.1038/s41598-017-03383-8
Rowell, T. J., Schärer, M. T., Appeldoorn, R. S., Nemeth, M. I., Mann, D. A., and Rivera, J. A. (2012). Sound production as an indicator of red hind density at a spawning aggregation. Mar. Ecol. Prog. Ser. 462, 241–250. doi:10.3354/meps09839
Rowley, S. J. (2018). Environmental gradients structure gorgonian assemblages on coral reefs in SE Sulawesi, Indonesia. Coral Reefs 37 (2), 609–630. doi:10.1007/s00338-018-1685-y
Sadovy De Mitcheson, Y., Cornish, A., Domeier, M., Colin, P. L., Russell, M., and Lindeman, K. C. (2008). A global baseline for spawning aggregations of reef fishes. Conserv. Biol. 22 (5), 1233–1244. doi:10.1111/j.1523-1739.2008.01020.x
Simpson, S. D., Meekan, M., Montgomery, J., McCauley, R., and Jeffs, A. (2005). Homeward sound. Science. 308 (5719), 221. doi:10.1126/science.1107406
Simpson, S. D., Radford, A. N., Nedelec, S. L., Ferrari, M. C., Chivers, D. P., McCormick, M. I., et al. (2016). Anthropogenic noise increases fish mortality by predation. Nat. Commun. 7 (1), 10544. doi:10.1038/ncomms10544
Šmejkal, M., Souza, A. T., Blabolil, P., Bartoň, D., Sajdlová, Z., Vejřík, L., et al. (2018). Nocturnal spawning as a way to avoid egg exposure to diurnal predators. Sci. Rep. 8 (1), 15377. doi:10.1038/s41598-018-33615-4
Sobradillo, B., Christiansen, S., Røstad, A., and Kaartvedt, S. (2022). Individual daytime swimming of mesopelagic fishes in the world's warmest twilight zone. Deep Sea Res. Part I Oceanogr. Res. Pap. 190, 103897. doi:10.1016/j.dsr.2022.103897
Staaterman, E., Rice, A., Mann, D., and Paris, C. (2013). Soundscapes from a tropical eastern pacific reef and a caribbean sea reef. Coral Reefs 32, 553–557. doi:10.1007/s00338-012-1007-8
Tricas, T. C., and Boyle, K. S. (2014). Acoustic behaviors in Hawaiian coral reef fish communities. Mar. Ecol. Prog. Ser. 511, 1–16. doi:10.3354/meps10930
Van Oosterom, L., Montgomery, J., Jeffs, A., and Radford, C. (2016). Evidence for contact calls in fish: conspecific vocalisations and ambient soundscape influence group cohesion in a nocturnal species. Sci. Rep. 6 (1), 19098–8. doi:10.1038/srep19098
Vermeij, M. J., Marhaver, K. L., Huijbers, C. M., Nagelkerken, I., and Simpson, S. D. (2010). Coral larvae move toward reef sounds. PloS one 5 (5), e10660. doi:10.1371/journal.pone.0010660
Williams, B., Belvanera, S. M., Sethi, S. S., Lamont, T. A., Jompa, J., Prasetya, M., et al. (2024). Unlocking the soundscape of coral reefs with artificial intelligence: pretrained networks and unsupervised learning win out. bioRxiv. [Preprint] 578582. doi:10.1101/2024.02.02.578582
Zhang, Q., and Katsnelson, B. (2021). A fish chorus on the margin of New Jersey Atlantic continental shelf. Front. Mar. Sci. 8, 671965. doi:10.3389/fmars.2021.671965
Keywords: fish chorus, acoustic monitoring, soundscape, mesophotic, coral, reef
Citation: Havlik M-N, Parry AJ, Steckbauer A, Watts ME, Marchese F, Benzoni F and Duarte CM (2025) Nocturnal fish chorusing activity in the central Red Sea mesophotic reef zone and adjacent shallow sites. Front. Remote Sens. 6:1482244. doi: 10.3389/frsen.2025.1482244
Received: 17 August 2024; Accepted: 03 March 2025;
Published: 21 March 2025.
Edited by:
Lucia Di Iorio, UMR5110 Centre de formation et de recherche sur les environnements méditerranéens (CEFREM), FranceReviewed by:
Gerald D’Spain, University of California, United StatesCopyright © 2025 Havlik, Parry, Steckbauer, Watts, Marchese, Benzoni and Duarte. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Michelle-Nicole Havlik, bWljaGVsbGVuaWNvbGUuaGF2bGlrQGthdXN0LmVkdS5zYQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.