
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Public Health , 03 March 2025
Sec. Infectious Diseases: Epidemiology and Prevention
Volume 13 - 2025 | https://doi.org/10.3389/fpubh.2025.1511374
This article is part of the Research Topic Epidemiology and Control of Blood-Borne Viruses in Low and Middle Income Countries: Genomic Insights and Public Health Strategies View all 3 articles
 Carlos Brites1†
Carlos Brites1† Prince Baffour Tonto2,3†
Prince Baffour Tonto2,3† Antonio C. Vallinoto4
Antonio C. Vallinoto4 Onayane dos Santos Oliveira4
Onayane dos Santos Oliveira4 Simone Simionatto5
Simone Simionatto5 Monica Bay6
Monica Bay6 Tania Reuter7
Tania Reuter7 Monica M. Gomes-da-Silva8
Monica M. Gomes-da-Silva8 Melissa Medeiros9,10
Melissa Medeiros9,10 Rafaela Mayoral1
Rafaela Mayoral1 Estela Luz1
Estela Luz1 Michael Rocha11
Michael Rocha11 Hareton Vechi12,13
Hareton Vechi12,13 Bobby Brooke Herrera1,2,3*
Bobby Brooke Herrera1,2,3*Background: Human T cell lymphotropic virus type 1 (HTLV-1) is highly endemic in Brazil, necessitating surveillance studies to understand its epidemiology. While previous research has focused on either specific cities or populations, there is a need for multicenter studies encompassing epidemiologically relevant populations to ascertain more accurate prevalence rates and predictors of HTLV-1 infection in the country.
Methods: We conducted a multicenter, cross-sectional study involving 3,184 participants across seven cities and five study populations in Brazil. Blood samples were collected, and the prevalence of HTLV-1 infection was determined by enzyme-linked immunosorbent assay (ELISA) and Western blot. Binary logistic regression analysis was used to determine risk factors of HTLV-1 infection.
Results: Among the total study population, 1,135 (35.7%) were aged >40 years and 1,704 (53.5%) were female. The overall prevalence of HTLV-1 infection was 0.5% (95% CI: 0.3–0.8), with variation observed among the cities or study populations. Factors associated with HTLV-1 infection included age > 40 years (OR, 8.867; 95% CI: 1.824–43.099), female gender (OR, 4.604; 95% CI: 1.184–17.903), and Hepatitis C virus (HCV) infection (OR, 13.995; 95% CI: 2.374–82.506). The identification of older age and female gender, coupled with the high prevalence of HTLV-1 in HIV-positive patients, suggests sexual transmission as the primary route of HTLV-1 infection.
Conclusion: Our study reveals varied prevalence rates of HTLV-1 infection across diverse populations and cities in Brazil. The association of older age, female gender, and HCV, emphasizes the need for tailored interventions to prevent HTLV-1 transmission.
Surveillance of infectious pathogens is fundamental to public health, enabling the development and implementation of strategies to curb their spread and alleviate disease burden. Effective surveillance requires diverse and robust testing methodologies capable of identifying pathogens, particularly those with the ability to establish latent infections within host cells and transmit infections inapparently (1). Among these pathogens, Human T cell lymphotropic virus type 1 (HTLV-1) emerges as a significant concern.
HTLV-1, the first human oncogenic retrovirus discovered (2), presents a multifaceted challenge to public health due to its modes of transmission and associated debilitating conditions. Transmission predominantly occurs through sexual intercourse (3), vertical transmission via pregnancy or prolonged breastfeeding (4), and exposure to contaminated blood products or shared needles or syringes (5). The repercussions of HTLV-1 are severe, encompassing conditions such as Adult T-cell leukemia/lymphoma (ATLL) (6), characterized by a dismal prognosis and a median survival of less than a year, and HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) (7, 8), associated with a reduced quality of life and lifespan. HTLV-1 infection can also cause inflammatory syndromes including arthritis (9), uveitis (10), and infective dermatitis (11).
The primary cellular target of HTLV-1 is CD4+ T-cells, although it can also infect CD8+ T-cells, dendritic cells and monocytes to a lesser extent. Unlike HIV, which efficiently infects CD4+ T-cells through cell-free virions, HTLV-1 relies on host cell machinery for cell-to-cell transmission of its targets. Following viral replication in CD4+ T cells and formation of virions, HTLV-1 transfer viral particles to uninfected cells through multiple mechanisms, including the formation of a viral synapse, formation of biofilm-like structures containing virions, the establishment of cell–cell conjugates, the trafficking of extracellular vesicles bearing viral components (12).
Despite its global prevalence with an estimated 5–20 million people infected, HTLV-1 infection is endemic in diverse regions, including but not limited to Japan, the Caribbean, Sub-Saharan Africa, Central and South America, and the Middle East (13–16). There are currently no approved vaccines or anti-retroviral therapeutics to prevent or help manage HTLV-1 infection, due in large part to the lack of funding dedicated to HTLV-1 research. Treatment options are limited to supportive care, symptomatic management, and addressing complications associated with HTLV-1-related diseases, particularly ATLL and HAM/TSP (17, 18).
Within South America, Brazil is a focal point for HTLV-1 endemicity with an estimated 800,000 people infected (19). However, prevalence rates vary considerably among different populations and geographic areas within the country. Notably, higher prevalence rates are typically observed in the northern and northeastern regions of the country compared to other areas (20). Historically, individuals with underlying comorbidities such as HIV infection and tuberculosis have been shown to be highly susceptible to HTLV-1 infection in Brazil, as evidenced by higher but variable prevalence rates across different geographical locations. Notably, reported HTLV-1 prevalence rates range from 1.4 to 21.1% among HIV patients (21), and 4.3 to 10.8% among tuberculosis (TB) patients (22). Furthermore, pregnant women (PW) represent a key population in HTLV-1 epidemiology due to the potential for vertical transmission of the virus. Studies have reported varying HTLV-1 prevalence rates, ranging from 0.1 to 1.1% among PW (23).
Despite numerous studies conducted in Brazil to understand HTLV-1 prevalence rates, many have been localized to specific areas with varying methodological approaches, hindering the accurate estimation of nationwide prevalence. Therefore, studies encompassing diverse regions and populations are imperative to provide a comprehensive understanding of the magnitude and potential predictors of HTLV-1 infection in the country. The present study is a multicenter, cross-sectional evaluation of HTLV-1 prevalence and associated risk factors across seven cities in five different regions (Salvador, Belem, Curitiba, Dourados, Fortaleza, Natal, Vitoria) and five epidemiologically relevant populations (general population, individuals with HIV infection, individuals with tuberculosis, pregnant women, and individuals on pre-exposure prophylaxis [PREP]) in Brazil.
Our multicenter, cross-sectional study included 3,184 participants aged from 11 to 85 years, recruited from seven distinct cities in Brazil from 2021 to 2023. These cities comprised six state capital cities: Fortaleza, Natal, and Salvador in the northeastern part of the country (capital cities of Ceará, Rio Grande do Norte, and Bahia states, respectively); Belem (capital city of Pará state); Vitória (capital of Espirito Santo state); Curitiba (capital of Paraná state); and Dourados (the second largest city of Mato Grosso do Sul state), representing the northern, southeastern, southern and central-western regions, respectively (Figure 1).
The study population comprised five groups: 926 HIV-positive patients attending HIV referral centers, 325 recently diagnosed TB patients receiving treatment at tuberculosis referral centers, 719 PW attending public antenatal care centers, and 931 patients seeking care for various other reasons at public health centers, categorized as the general population (GP) (Figure 1). An additional group of 238 participants attending an HIV prophylaxis center in Natal was included due to the lack of HTLV-1 prevalence data in this population. Furthermore, one site could only screen people with HIV due to local issues related to the COVID-19 pandemic. None of the study participants presented with symptoms of ATL or HAM/TSP, or other clinical signs associated with HTLV-1.
Participants were eligible for the study if they met any one of the inclusion criteria: (1) residence of the specified cities; (2) Diagnosis of HIV infection; (3) Diagnosis of tuberculosis; (4) Pregnancy; and (5) Attendance at public health centers for other medical reasons within these cities. The exclusion criterion was failure or refusal to provide informed consent.
Socio-demographic and non-infectious comorbidities were solicited from the participants using a well-structured and standardized written questionnaire. The socio-demographic factors were age, gender, family income, length of education, marital status, stable relationship, ethnicity, blood transfusion, intravenous drug use, and sexual orientation. For non-infectious comorbidities, factors including diabetes mellitus, arterial hypertension, obesity and cardiovascular diseases were collected. Information on infectious comorbidities that included hepatitis B (HBV) and C (HCV) was obtained through the medical history of the participants.
The required sample size was calculated based on an estimated HTLV-1 prevalence of 0.5% among the GP and PW, 4% among HIV-positive individuals, and 10% among tuberculosis patients, with a margin of error of 0.5% for GP and PW, 1.5% for HIV and 4% for Tuberculosis. Using a 97% confidence interval, the minimum sample size was calculated to be 2,693. We used the formula: N*p (1−p)/(d2/Z21−α/2)*(N−1) + p*(1−p) to calculate the sample size. We added an extra 10% to account for missing data and dropouts, increasing the sample size to 2,963. Additionally, 283 participants from an HIV PrEP program were included due to limited data on HTLV-1 infection within this population. 61 participants who initially satisfied the inclusion criteria, withdrew their consent, and were subsequently excluded from the study, resulting in a final sample size of 3,184 (98% response rate) (Figure 1).
Ethical clearance for demographic data and whole blood sample collection was obtained from the institutional review boards (IRB) from each clinical site, including Maternidade Climério de Oliveira, Universidade Federal da Bahia (approval number: 31650520.0.1001.5543), Universidade Federal da Grande Dourados (approval number: 31650520.0.2007.5160), Universitário Cassiano Antônio de Moraes da Universidade Federal do Espirito Santo (approval number: 31650520.0.2009.5071), Instituto de Ciências da Saúde da Universidade Federal do Para (approval number: 31650520.0.2008.0018), Hospital Universitário Onofre Lopes Da Universidade Federal do Rio Grande Do Norte (approval number: 31650520.0.2005.5292), Hospital de Clinicas da Universidade Federal do Parana (approval number: 31650520.0.2006.0096), and Hospital São Jose de Doenças Infecciosas – HSJ/Secretaria de Saúde de Fortaleza (approval number: 31650520.0.2001.5044). The study was conducted in accordance with the principles of Declaration of Helsinki. Formal written consent was obtained from all participants. This study received an exemption determination from the Rutgers Robert Wood Johnson Medical School IRB.
10 mL of whole blood was collected from each participant via venipuncture into heparin-containing tubes. Blood samples were subjected to centrifugation immediately after collection for 15 min at 2000 g. Serum samples were transferred into cryotubes and stored at −20°C until further analysis. The time interval between blood collection and testing varied depending on the study site and ranged from 24 to 48 h.
Detection of antibodies against HTLV was conducted by using an ELISA test kit (HTLV-1/II ELISA 4.0, MP Diagnostics, Eschwege, Germany), according to the manufacturer’s instructions. All reactive samples by ELISA were further analysed by Western blot (HTLV Blot 2.4, MP Diagnostics, Eschwege, Germany) to differentiate between exposure to HTLV-1 and HTLV-2, according to the manufacturer’s instructions.
Categorical variables were represented as case counts and proportions, with differences between categorical variables assessed using Chi-square or Fisher’s exact tests, as appropriate. Univariate and multivariate binary logistic regression analyses were performed to assess the association between HTLV-1 prevalence and background factors. Variables used in the univariate analysis were age group, gender, stable relationship, marital status, ethnicity, sexual orientation, blood transfusion, intravenous drug use, length of education, family income, comorbidities, city, and study population. Finally, variables with p ≤ 0.25 in the univariate analysis were included in the multivariate logistic regression analysis. Multiple imputation (MI) method was done to replace the missing values, and the variables included in the MI model were stable relationship, blood transfusion, intravenous drug use, HCV, HBV, and sexual orientation. Statistical analyses were conducted, and plots were generated using IBM SPSS statistics software version 28 and/or GraphPad Prism 10. Finally, the map of Brazil, illustrating HTLV-1 prevalence rates, was generated using R version 4.4.1.
A total of 3,184 individuals participated in the study. Of these, 1,135 (35.7%) were aged >40 years and 1,704 (53.5%) were female (Table 1). Regarding racial characteristics, 1,699 (53.4%) subjects identified themselves as Black or mixed race. With respect to relationship, approximately 48% reported being in a stable relationship, with a majority reporting being single and never married, accounting for approximately 57% of the study cohort (Table 1). Majority of participants also self-identified as heterosexual (1,949; 61.2%), and 85 (2.7%) participants reported a prior history of blood transfusion (Table 1). Concerning educational attainment, a substantial proportion of the participants reported receiving less than 12 years of formal education (2,461; 77.3%) (Table 1). Furthermore, 2,461 (47.1%) participants reported a family income of less than 2 minimum wages.
In terms of health status, a notable proportion of participants reported various infections and comorbidities. In addition to 955 (30%) participants previously diagnosed with HIV infection, 32 (1%) and 65 (2%) participants reported hepatitis C virus (HCV) and hepatitis B virus (HBV) infections, respectively (Table 1). Additionally, 554 (17.4%) participants reported a prior history of sexually transmitted infections. Furthermore, 268 (8.4%) participants reported a diagnosis of diabetes mellitus, while 110 (3.5%) and 265 (8.3%) reported cardiovascular diseases and obesity, respectively. Arterial hypertension was also reported by 495 (15.6%) participants (Table 1). Finally, 2.7% of participants reported a history of blood transfusion, while 0.4% reported intravenous drug use.
Among 3,184 participants, 28 tested positive for HTLV infection based on serological analyses. These cases were confirmed by Western blotting, with 15 being identified as HTLV-1, 2 indeterminate, and 11 negative. There were two subjects who were positive for HTLV-2 in Belem. The overall prevalence of HTLV-1 was estimated to be 0.5% (95% CI: 0.3–0.8) (Table 2). The baseline characteristics of participants who were HTLV-1 positive are comprehensively described in the Supplementary Table.
The prevalence of HTLV-1 infection in Salvador was estimated to be 0.4%, with all cases occurring among participants receiving treatment for tuberculosis, where the prevalence rate was notably higher at 2.1%. In Fortaleza and Natal, the prevalence rates were 0.4 and 0.2%, respectively, observed exclusively within the HIV study population, with a prevalence of 0.4% in Fortaleza and 0.9% in Natal. HTLV-1 prevalence in Belem was estimated to be 0.6%, distributed among the GP (1.1%), HIV-positive individuals (0.6%), and those being treated for tuberculosis (1.1%). In Dourados, the prevalence was 0.4%, identified within the HIV-positive (0.9%) and PW (0.5%) study populations. In Vitoria, the estimated prevalence of 0.6% was among the GP (1.7%) and HIV-positive individuals (1.8%). There were no HTLV-1 cases detected in the city of Curitiba (Table 2; Figures 2, 3).
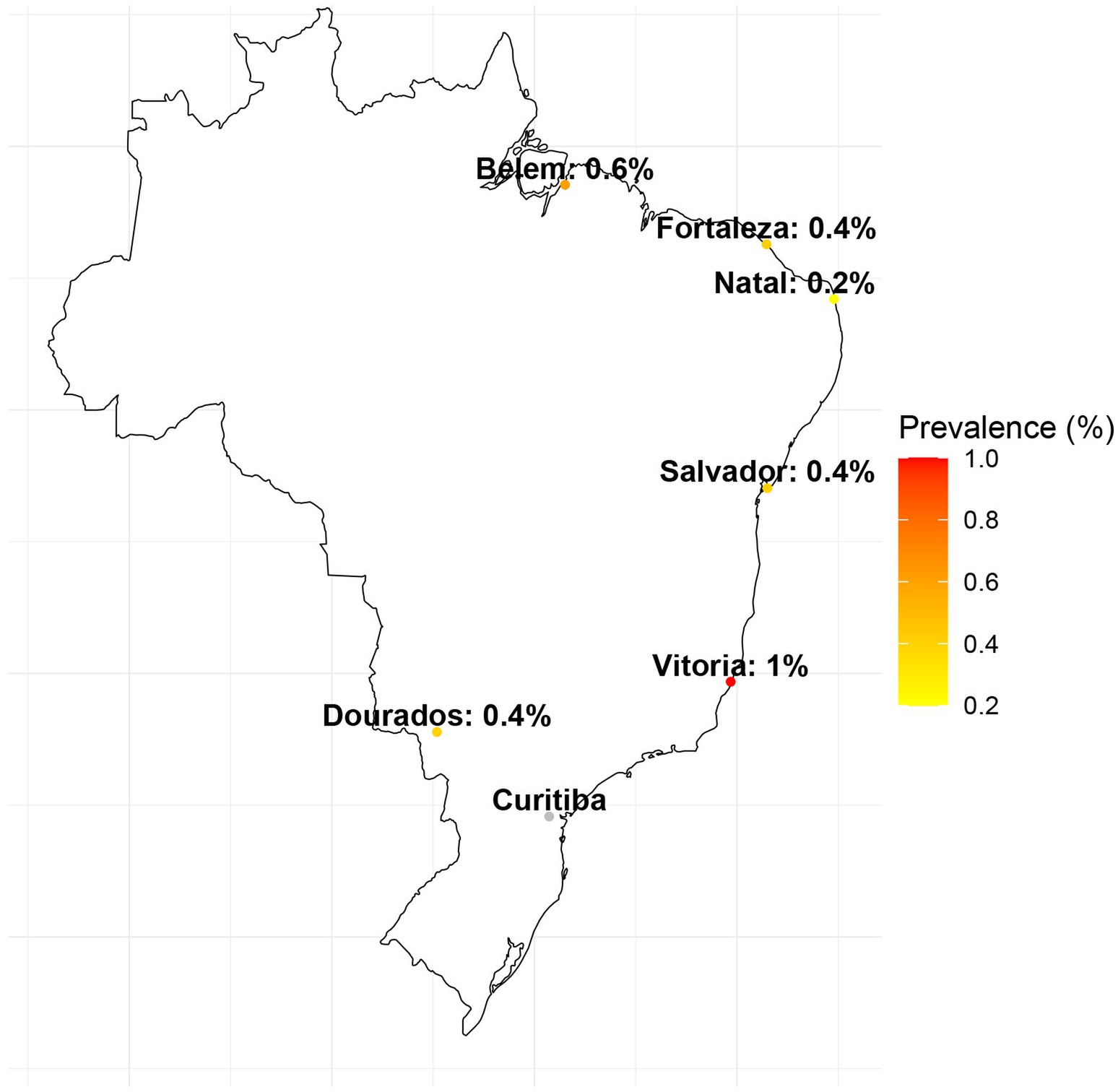
Figure 2. A map of Brazil showing the geographical location of the cities included in the study and their respective HTLV-1 prevalence. There was no case of HTLV-1 in the city of Curitiba.
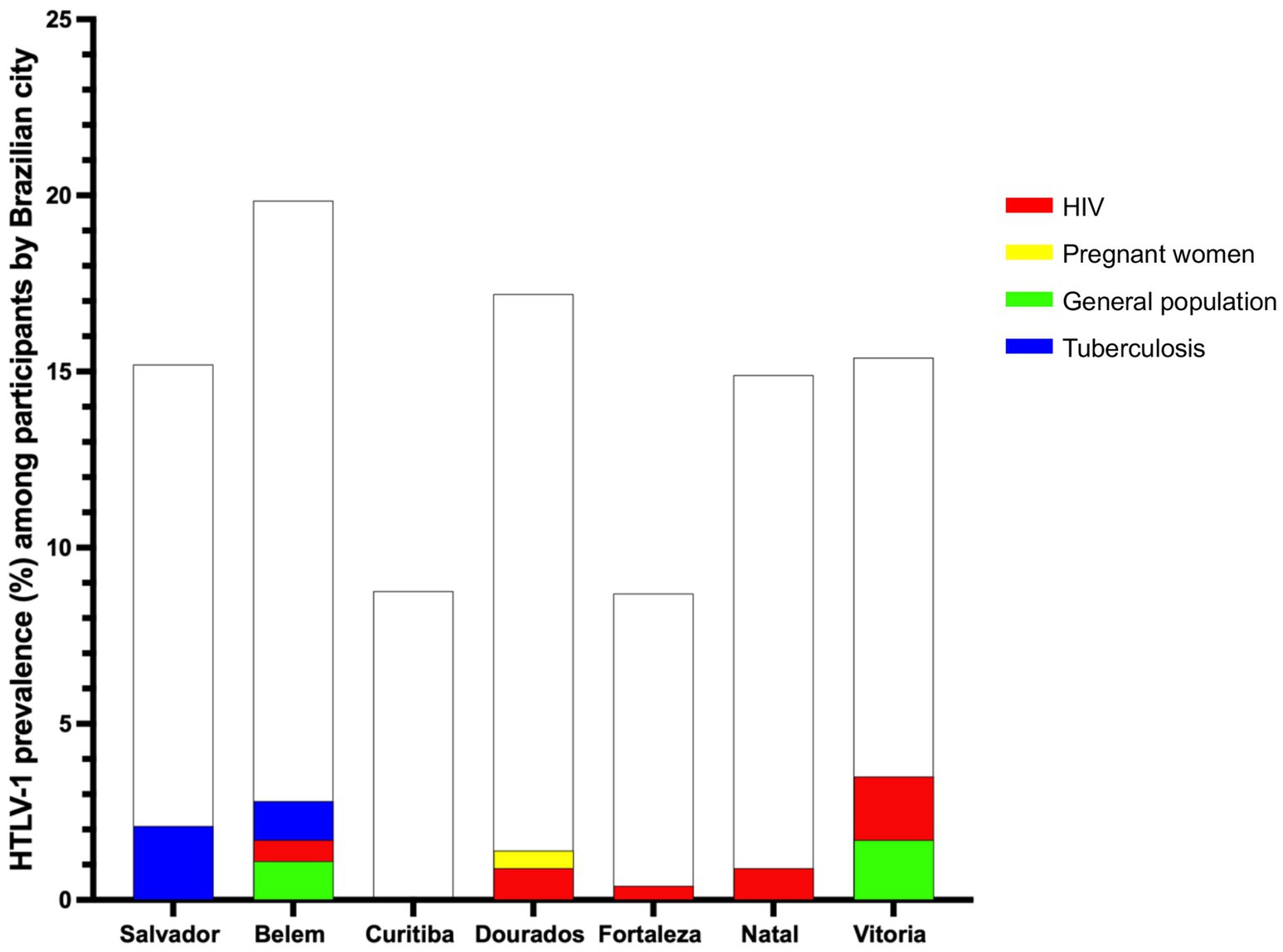
Figure 3. HTLV-1 prevalence among the study population, relative to the total sample size, by Brazilian city. The white bars represent the total sample size for each city relative to the overall study population. The colored bars indicate the HTLV-1 prevalence of subgroups within each city’s study population. Each color in the legend corresponds to a specific subgroup of the study population, with higher bars indicating a higher prevalence of HTLV-1 infection.
Regarding the study population, the highest prevalence was observed among those receiving treatment for tuberculosis (0.9%), followed by HIV-positive individuals (0.7%), and the GP (0.5%). The prevalence among PW was 0.1% with no HTLV-1 case detected among those on PrEP (Table 2).
Risk factors for HTLV-1 infection were identified using univariate and multivariate logistic regression analyses. In the univariate analysis, participants over 40 years of age (OR, 11.789; 95% CI: 2.656–52.335) and those with a prior history of HCV infection (OR, 18.856; 95% CI: 3.808–93.881) were associated with significantly increased odds of HTLV-1 infection (Table 3). The prevalence rate of HTLV-1 infection among participants over 40 was significantly higher compared to those 40 years or younger (Figure 4A). Similarly, the prevalence rate of HTLV-1 infection among participants with a history of HCV infection was significantly higher compared to those without HCV infection (Figure 4C).
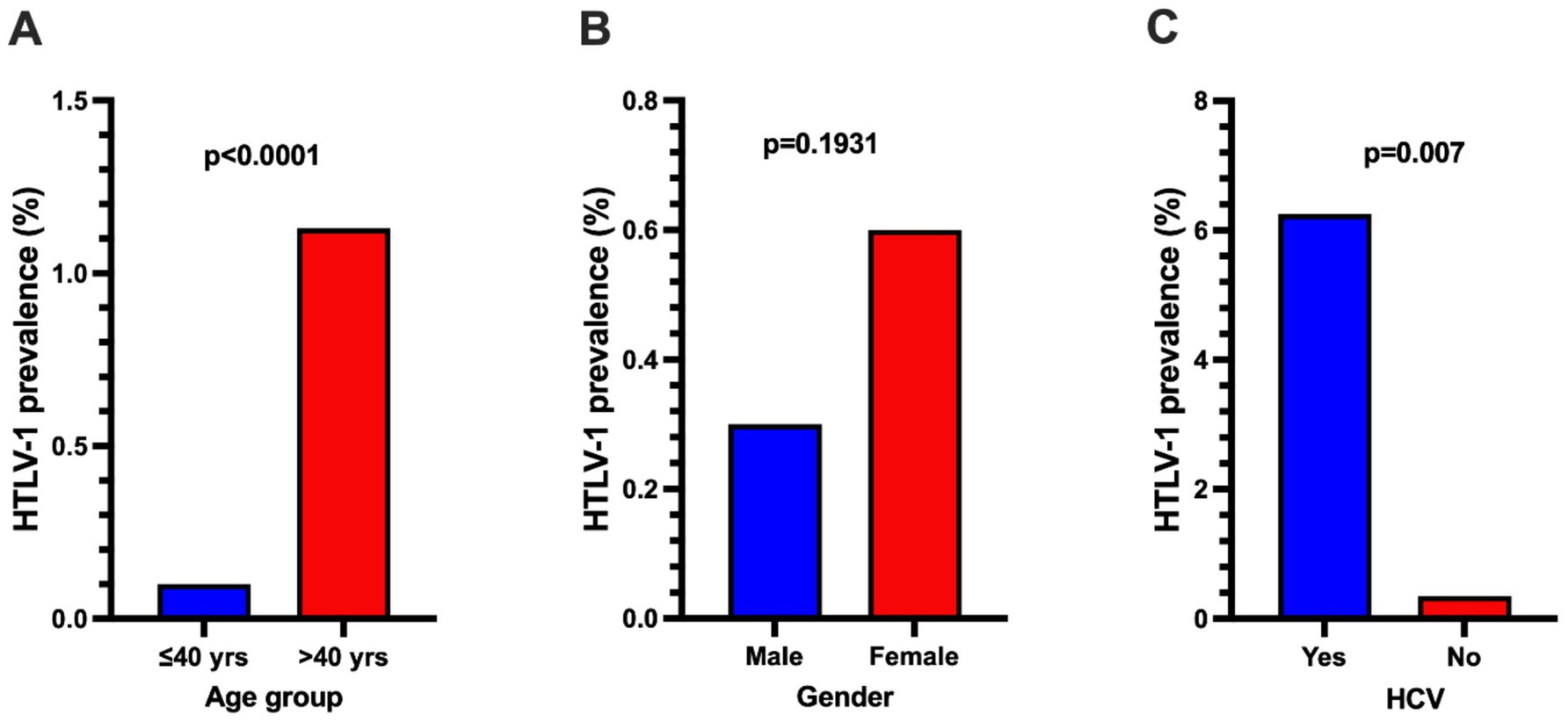
Figure 4. Comparison of HTLV-1 prevalence between participants with different age group (A), gender (B), and (C) those with or without prior history of HCV infection. Differences between the groups were assessed using Fisher’s Exact test, and p < 0.05 was considered statistically significant.
In the multivariate logistic regression model, participants over 40 years of age (OR, 8.867; 95% CI: 1.824–43.099), female gender (OR, 4.604; 95% CI: 1.184–17.903), and those with a prior history of HCV infection (OR, 13.995; 95% CI: 2.374–82.506) were identified as independent risk factors of HTLV-1 infection (Table 4). Additionally, the prevalence of HTLV-1 tended to be higher in females than in males (Figure 4B).
In this study, we observed an overall prevalence rate of HTLV-1 infection of 0.5% within the entire study population. Notably, analysis across different cities revealed varying HTLV-1 prevalence rates, with the highest prevalence rate observed in Vitória (1%). HTLV-1 prevalence was most frequent among individuals with tuberculosis and HIV. Despite the lower overall HTLV-1 prevalence observed in our study compared to other previously published literature, we noted higher rates in specific groups: 1.1% among the general population in Belem, 2.1% among tuberculosis patients in Salvador, and 1.7 and 1.8% among the general population and HIV patients in Vitoria, respectively. These findings are consistent with other studies conducted in Brazil (13, 24, 25).
Further investigation identified older age (>40 years), female gender, and history of HCV infection as significant risk factors for HTLV-1 infections, underscoring the importance of socio-demographic factors and co-infections in understanding HTLV-1 epidemiology. Most importantly, the identification of older age and female gender, coupled with the high prevalence of HTLV-1 among individuals with HIV, and none among injection drug users or individuals with prior history of blood transfusions, suggests sexual transmission may be the primary route of HTLV-1 infection. In fact, studies conducted elsewhere have demonstrated a clear link between sexual intercourse and HTLV-1 transmission, corroborating our findings (26, 27).
The first study of HTLV-1 seroprevalence in Brazil, conducted among Japanese migrants in Campo Grande, revealed high endemicity (28). Since then, numerous studies have investigated HTLV-1 prevalence and distribution, often focusing on specific populations or geographical areas. Among multicenter studies, prevalence rates have varied, reflecting differences in study populations, methodologies, and geographical locations. Our study uniquely surveyed diverse populations across multiple Brazilian cities, providing a more comprehensive understanding of HTLV-1 epidemiology in the country.
Of the few multicenter studies conducted, the HTLV-1 prevalence in Salvador was 2.48 and 1.76% among hemodialysis patients and the general population (29, 30), respectively, all higher than the rate observed in our study. Similarly, a study in four cities of the state of Para reported a HTLV-1 prevalence rate of 1.7% among female sex workers (13). Conversely, a study in Mato Grosso do Sul state reported a prevalence rate of 0.2% among prisoners (31). Moreover, studies conducted in other countries such as Gabon reported a HTLV-1 prevalence rate of 8.7% among rural inhabitants within 6 provinces (32). A nationwide survey in China reported a prevalence rate of 0.005% among blood donors (33). The discrepancy in the prevalence rates between these studies and ours may be attributed to differences in the target population, study design, methodological approach, and geographical location. Our study surveyed people from diverse populations in different Brazilian cities, while these studies surveyed individuals from different centers within the same city or different countries altogether.
Interestingly, the prevalence observed in individual cities differs from those of previous studies. Salvador is recognized as the city with the highest HTLV-1 endemicity, with a recent study among the general population reporting a prevalence rate of 1.48% (24), higher than observed in our study where HTLV-1 was only prevalent among individuals with tuberculosis. Similarly, studies in Belem (34), Dourados (35), Fortaleza (36), and Vitória (25) revealed varying prevalence rates, all differing from our observed rates. Notably, our study is the first to report HTLV-1 prevalence data in the city of Natal. While previous studies focused on specific populations or high-risk groups, our study surveyed individuals from five different populations, including high-risk groups, contributing to a broader understanding of HTLV-1 epidemiology in Brazil.
To capture the true prevalence rate of HTLV-1 infection in Brazil, our study was uniquely designed to include both the high-risk and the general populations. It is worth nothing that the prevalence rate in the general population differs from similar studies in Brazil. Notably, studies conducted in Salvador (24), and Belem (34) among the general population reported prevalence rates of 1.48 and 1.4%, respectively, all of which are higher than the observed rate in this study. The larger sample sizes in these studies may account for the higher HTLV-1 prevalence. However, findings were consistent with previous studies from Vitória, which reported a prevalence of 0.5%. Furthermore, our observed prevalence rate among HIV patients was higher compared to similar studies in Midwestern Brazil (37), but lower than the prevalence rate reported in São Paulo and Pernambuco (21, 38). Moreover, a recent meta-analysis of HTLV-1 prevalence among pregnant women in Brazil revealed a rate of 0.32% (39), which is higher than the observed rate in this study. Additionally, the prevalence of HTLV-1 among tuberculosis patients was comparable to similar studies conducted in Central-West Brazil (22), highlighting the dual role of co-infections in HTLV-1 transmission. Notably, the observed prevalence differed between Salvador (2.1%) and Belem (1.1%), potentially exacerbating the risk of TB in these regions due to immune dysregulation caused by HTLV-1.
Using logistic regression analysis, we identified older age as a significant risk factor for HTLV-1 infection, a finding consistent with previous studies conducted in Brazil (40–42) and elsewhere (32). In general, age serves as a crucial risk factor for contracting infectious pathogens due to longer cumulative exposure time. In the case of HTLV-1, transmission primarily occurs through sexual intercourse, blood transfusion/sharing needles, and breastfeeding. Hence, longer exposure to these routes over time can increase the likelihood of infection, especially in regions with high endemicity.
Our study showed that female gender is associated with an increased risk of contracting HTLV-1 infection, corroborating similar study in Brazil (43) and elsewhere (32). The virus replicates at a significantly higher rate in seminal fluid, resulting in higher levels of infected lymphocytes in semen than in vaginal fluids (44, 45). This disparity facilitates more efficient transmission of the virus from men to women than vice versa, thus making women more highly susceptible to contracting the virus though long-term exposure through the sexual route.
HCV infection was also identified as a significant risk factor for HTLV-1 infection, and this is consistent with a similar study in Brazil (46). Our data suggests a potential interaction between these two viruses. Although the underlying mechanism is not known, the association between HCV and HTLV-1 may be attributed to similar routes of transmission, including intravenous drug use, contaminated blood or blood products, and sexual contact. Additionally, HCV infection can lead to chronic liver disease, which may compromise the immune system and thus increase susceptibility to other infections, including HTLV-1. Moreover, HCV-infected individuals may be more likely to undergo regular medical monitoring and testing, leading to increased detection of co-existing HTLV-1 infection through routine screening. Conversely, we have shown that HTLV-1 infection increases the likelihood of HCV spontaneous clearance (47, 48). Future studies that focus on immune responses of HTLV-1 HCV co-infected subjects will help elucidate the interactions between these two viruses (49).
This study suggests that the primary mode of HTLV-1 transmission is likely through the sexual route, consistent with findings from studies in Brazil (24) and other countries (50, 51). This hypothesis is supported by the absence of HTLV-1 among participants with a history of blood transfusion or intravenous drug use, thus ruling out the parenteral route. Additionally, the identification of older age and female gender as significant predictors of HTLV-1 infection by logistic regression modeling rules out vertical transmission of the virus. In fact, about 87% of the HTLV-1 cases were among participants over 40 years, and 73% occurred in females. The prevalence was also higher among the HIV-positive individuals.
One of the strengths of this study is the inclusion of participants from different cities and study populations, as it provides a broader perspective on the prevalence and risk factors associated with HTLV-1 infection. This approach allows for a more comprehensive understanding of the epidemiology of HTLV-1 in diverse settings and populations, enhancing the generalizability of our findings. However, it is important to acknowledge several limitations of the study. First, we focused exclusively on individuals attending specialized healthcare facilities, potentially introducing selection bias, and limiting the generalizability of our findings to the general population. Thus, including individuals outside these healthcare facilities would provide a more representative sample and a more accurate estimate of HTLV-1 prevalence. It is worth noting, however, that the included population should have a higher risk of HTLV infection, according to the available evidence, and a higher prevalence, which was not detected in our study. Second, the cross-sectional design of the study limits our ability to establish causality or temporal relationships between HCV and HTLV-1 infection. Longitudinal studies are needed to elucidate the dynamics of co-infection and the impact of one virus on the acquisition or progression of the other. Furthermore, the wider confidence interval observed for HCV as a predictor of HTLV-1 infection highlights uncertainty in the effect estimates. This wider interval may be due to relatively small sample size, limiting the precision of the findings. However, HCV has been shown to be a risk factor for HTLV-1 infection in Brazil (46). Finally, the reliance on self-reported data for risk factors may introduce recall bias and underreporting, potentially affecting the accuracy of our results. Future studies could incorporate more objective measures or validation techniques and larger sample size to improve the precision and reliability of risk factor data. Despite these limitations, our study provides valuable insights into the association between HCV and HTLV-1 infection and emphasizes the need for further research to elucidate the underlying mechanisms and implications of this relationship for clinical management and public health interventions. In addition, it shows that the prevalence of HTLV-1 infection in Brazil may be lower than that previously reported and reinforces the sexual exposure as the primary route of transmission.
In conclusion, our study provides compelling evidence for the variability of HTLV-1 infections across different geographical locations, and among diverse study populations in Brazil. We identified older age, female gender, and HCV as significant risk factors for HTLV-1 infection, highlighting the critical role of socio-demographic factors and co-infections in its epidemiology. Furthermore, we identified sexual transmission as the primary route of HTLV-1 infection. These findings emphasize the need for targeted interventions and public health strategies addressing age-related risks, socio-economic disparities, co-infection dynamics, and safe sex practices to effectively control HTLV-1 transmission. By understanding the interplay of these factors, we can better tailor prevention and screening programs to mitigate the burden of HTLV-1 infections and improve overall public health outcomes.
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author.
Ethical clearance for demographic data and whole blood sample collection was obtained from the institutional review boards (IRB) from each clinical site, including Maternidade Climério de Oliveira, Universidade Federal da Bahia (approval number: 31650520.0.1001.5543), Universidade Federal da Grande Dourados (approval number: 31650520.0.2007.5160), Universitário Cassiano Antônio de Moraes da Universidade Federal do Espirito Santo (approval number: 31650520.0.2009.5071), Instituto de Ciências da Saúde da Universidade Federal do Para (approval number: 31650520.0.2008.0018), Hospital Universitário Onofre Lopes Da Universidade Federal do Rio Grande Do Norte (approval number: 31650520.0.2005.5292), Hospital de Clinicas da Universidade Federal do Parana (approval number: 31650520.0.2006.0096), and Hospital São Jose de Doenças Infecciosas – HSJ/Secretaria de Saúde de Fortaleza (approval number: 31650520.0.2001.5044). The study was conducted in accordance with the principles of Declaration of Helsinki. Formal written consent was obtained from all participants. This study received an exemption determination from the Rutgers Robert Wood Johnson Medical School IRB. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
CB: Writing – review & editing, Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization. PT: Writing – original draft, Writing – review & editing, Data curation, Formal analysis, Investigation, Methodology, Software, Supervision, Validation, Visualization. AV: Writing – review & editing, Data curation, Investigation, Methodology, Validation. OS: Writing – review & editing, Data curation, Investigation, Methodology, Validation. SS: Writing – review & editing, Data curation, Investigation, Methodology, Validation. MB: Writing – review & editing, Data curation, Investigation, Methodology, Validation. TR: Writing – review & editing, Data curation, Investigation, Methodology, Validation. MG-d-S: Writing – review & editing, Data curation, Investigation, Methodology, Validation. MM: Writing – review & editing, Data curation, Investigation, Methodology, Validation. RM: Writing – review & editing, Data curation, Investigation, Methodology, Validation. EL: Writing – review & editing, Data curation, Investigation, Methodology, Validation. MR: Writing – review & editing, Data curation, Investigation, Methodology, Validation. HV: Writing – review & editing, Data curation, Investigation, Methodology, Validation. BH: Writing – review & editing, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study was funded by CNPq (National Research Council, Brazil) #442572/2019-0.
BH is a co-founder of Mir Biosciences, Inc., a biotechnology company focused on the development of diagnostics and vaccines for infectious diseases.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpubh.2025.1511374/full#supplementary-material
1. Herrera, BB, Mayoral, R, and Brites, C. Development and validation of a rapid screening test for HTLV-I IgG antibodies. Viruses. (2022) 15:129. doi: 10.3390/v15010129
2. Poiesz, BJ, Ruscetti, FW, Gazdar, AF, Bunn, PA, Minna, JD, and Gallo, RC. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci USA. (1980) 77:7415–9. doi: 10.1073/pnas.77.12.7415
3. Roucoux, DF, Wang, B, Smith, D, Nass, CC, Smith, J, Hutching, ST, et al. A prospective study of sexual transmission of human T lymphotropic virus (HTLV)-I and HTLV-II. J Infect Dis. (2005) 191:1490–7. doi: 10.1086/429410
4. Percher, F, Jeannin, P, Martin-Latil, S, Gessain, A, Afonso, PV, Vidy-Roche, A, et al. Mother-to-child transmission of HTLV-1 epidemiological aspects, mechanisms and determinants of mother-to-child transmission. Viruses. (2016) 8:40. doi: 10.3390/v8020040
5. Murphy, EL. Infection with human T-lymphotropic virus types-1 and -2 (HTLV-1 and -2): implications for blood transfusion safety. Transfus Clin Biol. (2016) 23:13–9. doi: 10.1016/j.tracli.2015.12.001
6. Takatsuki, K, Uchiyama, T, Sagawa, K, Yodoi, J, Seno, S, Takaku, F, et al., editors. Topics in hematology. The 16th International Congress of Hematology; (1977): Excerpta Medica: Amsterdam.
7. Gessain, A, Barin, F, Vernant, JC, Gout, O, Maurs, L, Calender, A, et al. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet. (1985) 2:407–10. doi: 10.1016/S0140-6736(85)92734-5
8. Osame, M, Izumo, S, Igata, A, Matsumoto, M, Matsumoto, T, Sonoda, S, et al. Blood transfusion and HTLV-I associated myelopathy. Lancet. (1986) 328:104–5. doi: 10.1016/S0140-6736(86)91636-3
9. Umekita, K, and Okayama, A. HTLV-1 infection and rheumatic diseases. Front Microbiol. (2020) 11:152. doi: 10.3389/fmicb.2020.00152
10. Takeda, A, Ishibashi, T, and Sonoda, KH. Epidemiology of uveitis, caused by HTLV-1, toxoplasmosis, and tuberculosis; the three leading causes of endemic infectious uveitis in Japan. Ocul Immunol Inflamm. (2017) 25:S19–s23. doi: 10.1080/09273948.2016.1253851
11. Schierhout, G, McGregor, S, Gessain, A, Einsiedel, L, Martinello, M, and Kaldor, J. Association between HTLV-1 infection and adverse health outcomes: a systematic review and meta-analysis of epidemiological studies. Lancet Infect Dis. (2020) 20:133–43. doi: 10.1016/S1473-3099(19)30402-5
12. Kalinichenko, S, Komkov, D, and Mazurov, D. HIV-1 and HTLV-1 transmission modes: mechanisms and importance for virus spread. Viruses. (2022) 14:152. doi: 10.3390/v14010152
13. de Souza, RL, Pereira, MVS, da Silva, RM, Sales, JBL, Gardunho, DCL, Monteiro, JC, et al. Molecular epidemiology of HIV-1 and HTLV-1/2 among female sex Workers in Four Cities in the state of Para, northern Brazil. Front Microbiol. (2020) 11:602664. doi: 10.3389/fmicb.2020.602664
14. Gessain, A, Ramassamy, JL, Afonso, PV, and Cassar, O. Geographic distribution, clinical epidemiology and genetic diversity of the human oncogenic retrovirus HTLV-1 in Africa, the world's largest endemic area. Front Immunol. (2023) 14:1043600. doi: 10.3389/fimmu.2023.1043600
15. Iwanaga, M. Epidemiology of HTLV-1 infection and ATL in Japan: an update. Front Microbiol. (2020) 11:1124. doi: 10.3389/fmicb.2020.01124
16. Proietti, FA, Carneiro-Proietti, AB, Catalan-Soares, BC, and Murphy, EL. Global epidemiology of HTLV-I infection and associated diseases. Oncogene. (2005) 24:6058–68. doi: 10.1038/sj.onc.1208968
17. Araujo, A, Bangham, CRM, Casseb, J, Gotuzzo, E, Jacobson, S, Martin, F, et al. Management of HAM/TSP: systematic review and consensus-based recommendations 2019. Neurol Clin Pract. (2021) 11:49–56. doi: 10.1212/CPJ.0000000000000832
18. Mashkani, B, Jalili Nik, M, Rezaee, SA, and Boostani, R. Advances in the treatment of human T-cell lymphotropic virus type-I associated myelopathy. Expert Rev Neurother. (2023) 23:1233–48. doi: 10.1080/14737175.2023.2272639
19. Gessain, A, and Cassar, O. Epidemiological aspects and world distribution of HTLV-1 infection. Front Microbiol. (2012) 3:388. doi: 10.3389/fmicb.2012.00388
20. das Infecções, CG, Transmissíveis, S, Rosadas, C, Espinosa Miranda, A, Utsch Gonçalves, D, Caterino-de-Araujo, A, et al. Prevalência da infecção por HTLV-1/2 no Brasil-Boletim Epidemiológico. SVS/MS. (2020) 51:102274.
21. Ribeiro, ML, Gonçales, JP, Morais, VMS, Moura, L, and Coêlho, M. HTLV 1/2 prevalence and risk factors in individuals with HIV/AIDS in Pernambuco, Brazil. Rev Soc Bras Med Trop. (2019) 52:e20180244. doi: 10.1590/0037-8682-0244-2018
22. Kozlowski, AG, Carneiro, MA, Matos, MA, Teles, SA, Araújo, JA, Otsuki, K, et al. Prevalence and genetic characterisation of HTLV-1 and 2 dual infections in patients with pulmonary tuberculosis in central-West Brazil. Mem Inst Oswaldo Cruz. (2014) 109:118–21. doi: 10.1590/0074-0276130230
23. Rosadas, C, Malik, B, Taylor, GP, and Puccioni-Sohler, M. Estimation of HTLV-1 vertical transmission cases in Brazil per annum. PLoS Negl Trop Dis. (2018) 12:e0006913. doi: 10.1371/journal.pntd.0006913
24. Nunes, D, Boa-Sorte, N, Grassi, MF, Taylor, GP, Teixeira, MG, Barreto, ML, et al. HTLV-1 is predominantly sexually transmitted in Salvador, the city with the highest HTLV-1 prevalence in Brazil. PLoS One. (2017) 12:e0171303. doi: 10.1371/journal.pone.0171303
25. Orletti, MPS, Assone, T, Sarnaglia, GD, Martins, ML, Rosadas, C, Casseb, J, et al. Prevalence of infection by human T cell lymphotropic viruses (HTLV-1/2) in adult population in Vitória-ES. Braz J Infect Dis. (2021) 25:101631. doi: 10.1016/j.bjid.2021.101631
26. Ayerdi, O, Benito, R, Ortega, D, Aguilera, A, Montiel, N, Pintos, I, et al. HTLV infection in persons with sexually transmitted diseases in Spain. Front Immunol. (2023) 14:1277793. doi: 10.3389/fimmu.2023.1277793
27. Kamoi, K, Uchimaru, K, Nannya, Y, Tojo, A, Watanabe, T, and Ohno-Matsui, K. Sexual transmission of HTLV-1 resulting in uveitis with short-term latency and low proviral load. J Med Virol. (2024) 96:e70000. doi: 10.1002/jmv.70000
28. Kitagawa, T, Fujishita, M, Taguchi, H, Miyoshi, I, and Tadokoro, H. Antibodies to HTLV-I in Japanese immigrants in Brazil. JAMA. (1986) 256:2342. doi: 10.1001/jama.1986.03380170058009
29. Santos, RF, Conceição, GC, Martins, MS, Kraychete, A, Penalva, MA, Carvalho, EM, et al. Prevalence and risk factors for human T-Lymphotropic virus type 1 (HTLV-1) among maintenance hemodialysis patients. BMC Nephrol. (2017) 18:64. doi: 10.1186/s12882-017-0484-y
30. Dourado, I, Alcantara, LC, Barreto, ML, da Gloria, TM, and Galvão-Castro, B. HTLV-I in the general population of Salvador, Brazil: a city with African ethnic and sociodemographic characteristics. J Acquir Immune Defic Syndr. (2003) 34:527–31. doi: 10.1097/00126334-200312150-00013
31. Melo Bandeira, L, Moreira Puga, MA, Croda, J, Pompílio, MA, Amianti, C, Rocha de Rezende, G, et al. Human T-Lymphotropic Virus-1/2 infection in Central Brazil prisons: a multicenter study. Front Microbiol. (2021) 12:740245. doi: 10.3389/fmicb.2021.740245
32. Djuicy, DD, Mouinga-Ondémé, A, Cassar, O, Ramassamy, JL, Idam Mamimandjiami, A, Bikangui, R, et al. Risk factors for HTLV-1 infection in Central Africa: a rural population-based survey in Gabon. PLoS Negl Trop Dis. (2018) 12:e0006832. doi: 10.1371/journal.pntd.0006832
33. Chang, L, Ou, S, Shan, Z, Zhu, F, Ji, H, Rong, X, et al. Seroprevalence of human T-lymphotropic virus infection among blood donors in China: a first nationwide survey. Retrovirology. (2021) 18:2. doi: 10.1186/s12977-020-00546-w
34. Silva, IC, Pinheiro, BT, Nobre, AFS, Coelho, JL, Pereira, CCC, Ferreira, LSC, et al. Moderate endemicity of the human T-lymphotropic virus infection in the metropolitan region of Belém, Pará, Brazil. Rev Bras Epidemiol. (2018) 21:e180018. doi: 10.1590/1980-549720180018
35. Amianti, C, Bandeira, LM, Cesar, GA, Weis-Torres, S, Tanaka, TSO, Machado, IR, et al. HTLV infection in Brazil's second-largest indigenous reserve. Sci Rep. (2022) 12:16701. doi: 10.1038/s41598-022-21086-7
36. Santos Tde, J, Costa, CM, Goubau, P, Vandamme, AM, Desmyter, J, Van Doren, S, et al. Western blot seroindeterminate individuals for human T-lymphotropic virus I/II (HTLV-I/II) in Fortaleza (Brazil): a serological and molecular diagnostic and epidemiological approach. Braz J Infect Dis. (2003) 7:202–9. doi: 10.1590/S1413-86702003000300005
37. Kozlowski, AG, Matos, MA, Carneiro, MA, Lopes, CL, Teles, SA, Vicente, CP, et al. SEROPREVALENCE of HTLV in a population of HIV1-infected patients in MIDWESTERN Brazil. Rev Inst Med Trop Sao Paulo. (2016) 58:80. doi: 10.1590/S1678-9946201658080
38. Caterino-de-Araujo, A, Sacchi, CT, Gonçalves, MG, Campos, KR, Magri, MC, and Alencar, WK. Short communication: current prevalence and risk factors associated with human T Lymphotropic virus type 1 and human T Lymphotropic virus type 2 infections among HIV/AIDS patients in São Paulo, Brazil. AIDS Res Hum Retrovir. (2015) 31:543–9. doi: 10.1089/aid.2014.0287
39. Vieira, BA, Bidinotto, AB, Dartora, WJ, Pedrotti, LG, de Oliveira, VM, and Wendland, EM. Prevalence of human T-lymphotropic virus type 1 and 2 (HTLV-1/−2) infection in pregnant women in Brazil: a systematic review and meta-analysis. Sci Rep. (2021) 11:15367. doi: 10.1038/s41598-021-94934-7
40. Abreu, IN, Lima, CNC, Sacuena, ERP, Lopes, FT, da Silva Torres, MK, Santos, BCD, et al. HTLV-1/2 in indigenous peoples of the Brazilian Amazon: Seroprevalence, molecular characterization and Sociobehavioral factors related to risk of infection. Viruses. (2022) 15:22. doi: 10.3390/v15010022
41. Bandeira, LM, Uehara, SN, Asato, MA, Aguena, GS, Maedo, CM, Benites, NH, et al. High prevalence of HTLV-1 infection among Japanese immigrants in non-endemic area of Brazil. PLoS Negl Trop Dis. (2015) 9:e0003691. doi: 10.1371/journal.pntd.0003691
42. Bandeira, LM, Uehara, SNO, Puga, MAM, Rezende, GR, Vicente, ACP, Domingos, JA, et al. HTLV-1 intrafamilial transmission among Japanese immigrants in Brazil. J Med Virol. (2018) 90:351–7. doi: 10.1002/jmv.24938
43. Carneiro-Proietti, AB, Sabino, EC, Leão, S, Salles, NA, Loureiro, P, Sarr, M, et al. Human T-lymphotropic virus type 1 and type 2 seroprevalence, incidence, and residual transfusion risk among blood donors in Brazil during 2007-2009. AIDS Res Hum Retrovir. (2012) 28:1265–72. doi: 10.1089/aid.2011.0143
44. Martel, M, and Gotuzzo, E. HTLV-1 is also a sexually transmitted infection. Front Public Health. (2022) 10:840295. doi: 10.3389/fpubh.2022.840295
45. Moriuchi, M, and Moriuchi, H. Seminal fluid enhances replication of human T-cell leukemia virus type 1: implications for sexual transmission. J Virol. (2004) 78:12709–11. doi: 10.1128/JVI.78.22.12709-12711.2004
46. Etzel, A, Shibata, GY, Rozman, M, Jorge, ML, Damas, CD, and Segurado, AA. HTLV-1 and HTLV-2 infections in HIV-infected individuals from Santos, Brazil: seroprevalence and risk factors. J Acquir Immune Defic Syndr. (2001) 26:185–90. doi: 10.1097/00126334-200102010-00015
47. Le Marchand, C, Bahia, F, Page, K, and Brites, C. Hepatitis C virus infection and spontaneous clearance in HTLV-1 and HIV co-infected patients in Salvador, Bahia, Brazil. Braz J Infect Dis. (2015) 19:486–91. doi: 10.1016/j.bjid.2015.06.007
48. Moreira, M, Ramos, A, Netto, EM, and Brites, C. Characteristics of co-infections by HCV and HBV among Brazilian patients infected by HIV-1 and/or HTLV-1. Braz J Infect Dis. (2013) 17:661–6. doi: 10.1016/j.bjid.2013.04.009
49. Bahia, F, Novais, V, Evans, J, Le Marchand, C, Netto, E, Page, K, et al. The impact of human T-cell lymphotropic virus I infection on clinical and immunologic outcomes in patients coinfected with HIV and hepatitis C virus. J Acquir Immune Defic Syndr. (2011) 57:S202–7. doi: 10.1097/QAI.0b013e31821e9a1e
50. Satake, M, Iwanaga, M, Sagara, Y, Watanabe, T, Okuma, K, and Hamaguchi, I. Incidence of human T-lymphotropic virus 1 infection in adolescent and adult blood donors in Japan: a nationwide retrospective cohort analysis. Lancet Infect Dis. (2016) 16:1246–54. doi: 10.1016/S1473-3099(16)30252-3
Keywords: multicenter, cross-sectional study, HTLV-1 prevalence, risk factors, epidemiologically relevant groups
Citation: Brites C, Tonto PB, Vallinoto AC, dos Santos Oliveira O, Simionatto S, Bay M, Reuter T, Gomes-da-Silva MM, Medeiros M, Mayoral R, Luz E, Rocha M, Vechi H and Herrera BB (2025) Multicenter cross-sectional study of HTLV-1 prevalence and associated risk factors in epidemiologically relevant groups across Brazil. Front. Public Health. 13:1511374. doi: 10.3389/fpubh.2025.1511374
Received: 14 October 2024; Accepted: 19 February 2025;
Published: 03 March 2025.
Edited by:
Marceline Djuidje Ngounoue, University of Yaounde I, CameroonReviewed by:
Maria Grazia Romanelli, University of Verona, ItalyCopyright © 2025 Brites, Tonto, Vallinoto, dos Santos Oliveira, Simionatto, Bay, Reuter, Gomes-da-Silva, Medeiros, Mayoral, Luz, Rocha, Vechi and Herrera. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bobby Brooke Herrera, YmhlcnJlcmFAZ2xvYmFsaGVhbHRoLnJ1dGdlcnMuZWR1
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.