
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Public Health , 13 February 2025
Sec. Aging and Public Health
Volume 13 - 2025 | https://doi.org/10.3389/fpubh.2025.1443920
This article is part of the Research Topic Beyond Rest: Exploring the Bidirectional Relationship and Intersecting Pathways of Sleep and Physical Health View all 8 articles
 Tala Kaddoura1,2†
Tala Kaddoura1,2† Mohamad Hodroj1†
Mohamad Hodroj1† Bilal Chmeis1
Bilal Chmeis1 Fatima Rammal1
Fatima Rammal1 Sandrella Bou Malhab3
Sandrella Bou Malhab3 Sara Mansour1
Sara Mansour1 Amal Akour4,5
Amal Akour4,5 Sami El Khatib6,7
Sami El Khatib6,7 Basile Hosseini8
Basile Hosseini8 Souheil Hallit9,10,11‡
Souheil Hallit9,10,11‡ Diana Malaeb12*‡
Diana Malaeb12*‡ Hassan Hosseini13,14‡
Hassan Hosseini13,14‡Objectives: To estimate the rate of obstructive sleep apnea (OSA) and associated factors among Lebanese adults.
Methods: A cross-sectional study was conducted in February–April 2022; 1,202 Lebanese adults were enrolled. The sample was collected among several Lebanese governorates through an anonymous online self-reported questionnaire. The STOP-BANG questionnaire was used as a screening tool to estimate the OSA risk. “Low OSA risk” is established with 0–2 positive answers, “Intermediate OSA risk” is established by 3–4 positive answers while “High OSA risk” is established by 5–8 positive answers.
Results: This study showed that 743 (62.4%) of the sample had low risk for OSA, 357 (30.0%) had moderate, and 90 (7.6%) had high risk for OSA. Older age (aOR = 1.05) and having hypertension (aOR = 7.80) were associated with higher odds of moderate OSA. Female sex (aOR = 0.04) was significantly associated with lower odds of moderate OSA compared to males. Older age (OR = 1.17), higher BMI (OR = 1.14), hypertension (OR = 18.55), and having severe COVID-19 infection compared to mild (OR = 4.30) were significantly associated with higher odds of high OSA, whereas female sex (OR = 0.002) and being married compared to single (aOR = 0.23) were associated with lower odds of high OSA.
Conclusion: This study showed that most Lebanese adults have low risk for OSA. It also confirmed that sex, age, obesity, hypertension, and severe COVID-19 were associated with high OSA risk. These associated factors call for future research exploring the causes including the potential effect of social, economic, and political instability, local customs, and environmental factors. Moreover, future health campaigns should be assigned to increase awareness among Lebanese population regarding the prevention of OSA through lifestyle modifications.
Obstructive sleep apnea (OSA) is a sleep disorder characterized by recurrent episodes of complete (apnea) or partial (hypopnea) collapse of the upper airway which impairs regular ventilation during sleep. OSA increases the risk of metabolic, cardiovascular and neurocognitive disorders due to hypoxemia and oxidative stress (1). OSA accounts for more than 85% of sleeping disorders. It can be asymptomatic in majority of those having moderate to severe OSA (2). Estimates of the prevalence of OSA vary based on the diagnostic criteria used, a recent study showed that the estimated prevalence ranges from 9 to 38% in community-screened populations (3). According to the American Academy of Sleep Medicine (AASM), undiagnosed sleep apnea costs $27 billion annually linked to the comorbid health risks (4). Several risk factors contribute to OSA occurrence, which are divided into unmodifiable and modifiable factors. Unmodifiable risk factors include sex, age, and genetic predisposition, while modifiable ones include obesity, specific medication utilization, hypothyroidism disorders, smoking, and nasal congestion (5). According to previous studies, men demonstrate higher risk for developing sleep apnea and higher severity of the disease than women (6). In addition, patients older than 60 years and obese with BMI > 30 kg/m2 had higher prevalence of OSA (7, 8). Furthermore, OSA is highly interrelated with different morbidities such as diabetes mellitus (DM) (9), progressive renal function decline (10), depression (11, 12), as well as cardiovascular disorders such as: hypertension, heart failure, atrial fibrillation, stroke, and coronary artery disease (13). Indeed, cardiovascular diseases existence is highly interrelated and bidirectionally associated with OSA providing a strong causal and resultant association. It can be also correlated with poor pregnancy outcomes including postpartum hemorrhage, premature delivery, congenital abnormalities (14), in addition to other disorders like gastro-esophageal reflex disorder (GERD) (15). Not only physiological changes occur, but also patients will become vulnerable to excessive daytime drowsiness, inattention, and irritability which will increase their risk of falling asleep while doing their basic daily activities, putting them at a high risk of all-cause mortality and work-related accidents (16). According to a survey done in Lebanon, early AM hours fatal crashes were associated with the drivers’ fatigue and sleepiness (17). Afterwards, systematic and meta-analytic reviews provided robust evidence that OSA plays a critical role in the development of broad-spectrum cognitive dysfunctions such as attention and vigilance deficits, verbal and visual delayed long-term memory, and executive function decline (18). Lebanese patients today need to be aware of the common pathologies and symptoms that should lead them to consult a physician for early diagnosis of OSA. A research study done in Lebanon has shown that patients could be underdiagnosed to due asymptomatic presentation in most of the cases (19). Another study conducted in Beirut showed that although 31% participants were at high risk for sleep apnea, only 5% were diagnosed by a physician (20). Chronic stress from political, social and economic instability lead to high rates of smoking (21), and unhealthy dietary habits (22) which contribute to key OSA risk factors like obesity and airway inflammation. Therefore, the aim of this study is to thoroughly evaluate the risk of OSA in a large, sample of Lebanese adults, with a particular focus on how various health factors including comorbidities, lifestyle choices, and demographic characteristics contribute to the likelihood of developing OSA. By examining these factors in a comprehensive manner, this study seeks to identify key determinants that may influence OSA risk within the Lebanese population and provide a more clear understanding of its prevalence and contributing factors.
This cross-sectional study was conducted between February and April 2022 after the lockdown period imposed by the government for the coronavirus disease 2019 (COVID-19) pandemic. We used an anonymous self-administered questionnaire developed on Google® Forms and posted on several social media platforms. The link was shared among participants using the social media and sent to people from all districts/governorates of Lebanon (Beirut, Mount Lebanon, North Lebanon/Akkar, South Lebanon/Nabatieh, and Bekaa/Hermel-Baalbeck) using the snowball technique. Participants were asked to fill the survey online which takes between 15 and 20 min to be complete and send the link to other participants too, which explains the snowball sampling technique used.
Eligibility criteria was mentioned in the first section of the survey that clearly specified the exclusion criteria. Excluded were, any participant younger than 18 years and patients with cognitive impairment or mental disorder. All participants knew the purpose of the study and gave their informed consent before starting the questionnaire. Participants received no compensation for their participation, which was completely voluntary.
We used G*Power software was used to determine the sample size. The minimum required sample size was 600 participants, considering an alpha error of 5%, a power of 95%, a minimal model r-square of 5% and allowing 20 predictors to be included in the model.
The questionnaire is divided into three sections. The first part included: socio-demographic characteristics including age, sex, weight, height, marital status, employment status, work type, monthly income, residency area (Beirut, Mount Lebanon, South, North, Akkar, Baalbek-Hermel, and Nabatieh), and household crowding index (calculated by taking the total number of co-residents per household excluding the newborn infant divided by the total number of rooms excluding the kitchen and bathrooms). Furthermore, it encompassed data about the social history for smoking and alcohol consumption. This section included also an assessment of health-related disorders including excessive drowsiness per day, waking up gasping and choking, dry mouth or sore throat after waking up, morning headaches, lack of concentration, mood swings, depression, forgetfulness, and swelling in the legs. The second part incorporated personal diseases and medication intake documented through assessing previous diagnosis of OSA, stroke, heart failure, diabetes mellitus, hypertension, and other chronic non-communicable diseases. Also, COVID 19 retraction history was documented, including symptoms, vaccination status, and the vaccine status.
The third part included the different scales used:
Patient Health Questionnaire (PHQ-9): an Arabic version validated scale in Lebanon (23) to retrieve information to screen for the presence and severity of depression. The questionnaire is composed of 9 multiple choice questions rated from 0 = not at all to 3 = nearly every day (24). Total score is calculated by summing all the choices together (24).
Lebanese Anxiety Scale (LAS-10) is a validated tool utilized to screen for anxiety in adults (25) and adolescents (26). It is composed of 10 self-reported items, the total score is calculated by summing all the answers, with higher scores reflecting that the participant is more anxious (25). Answers were scaled from 0 to 4 for each question with: 0 not present, 1 mild, 2 moderate, 3 severe, 4 very severe. The Cronbach’s alpha is 0.947.
Beirut Distress Scale (BDS): It is a valid and reliable tool to screen for psychological distress, influencing mood, and physical and cognitive functions among the Lebanese population (27). It is composed of 10 items each scored from 0 = never to 3 = always. The Cronbach’s alpha is 0.097.
STOP-BANG questionnaire: It is used to screen for OSA, which is a reliable, concise, an easy-to-use screening tool for obstructive sleep apnea. This questionnaire is valid in Arabic and reported snoring behavior, tiredness, gasping, hypertension, and neck circumference (28). It consists of eight dichotomous (yes: positive answer/no: negative answer) items related to the clinical features of sleep apnea. The total score ranges from 0 to 8. Patients can be classified for OSA risk based on their respective scores. “Low OSA risk” is established with 0–2 positive answers, “Intermediate OSA risk” is established by 3–4 positive answers while “High OSA risk” is established by 5–8 positive answers. Or a minimum of 2 on the STOP questions in addition to male sex or BMI > 35 kg/m2 or an elevated neck circumference (>43 cm in males or > 41 cm in females) (29). The Cronbach’s alpha is 0.7 (28).
Data was analyzed on the Statistical Package for Social Sciences version 25. Descriptive statistics was performed through the use of mean and standard deviation for continuous measures, counts, and percentages for categorical variables. Cronbach’s alpha values were recorded for the reliability of the analysis of all scales. Association between multiple categorical variables and OSA risk was evaluated using a Chi-square test in the bivariate analysis, whereas the ANOVA test was used to compare multiple means in the case of continuous variables. Bonferroni correction was applied for multiple testing by dividing 0.05 (5% risk of error) by the total number of variables included in the analysis (=22). The corrected p-value for significance was estimated at 0.05/22 = 0.002. Multinomial logistic regression was performed taking the obstructive sleep apnea risk (OSA risk) as the dependent variable. Factors that showed a significant p-value in the bivariate analysis were used as independent variables in the final model. Statistical significance was considered at p ≤ 0.05 in the final model.
A total number of 1,190 persons was enrolled in the study, with a mean age of 30.66 years (±12.56). Females represented 64.7% of the sample. We found that 743 (62.4%) of the study population had low risk for OSA, 357 (30.0%) had moderate risk, and 90 (7.6%) had high risk for OSA. Other characteristics of the sample can be found in Table 1.
The bivariate analysis results showed that a higher risk for obstructive sleep apnea was significantly found in males vs. females, in married persons compared to single ones, being employed, smokers compared to nonsmokers, those who drink alcohol vs. not, and in participants who have congestive heart failure, diabetes mellitus type 2, hypertension, lung diseases, hormonal disorders, and narrowed airways compared to not. Moreover, higher age, BMI, depression, anxiety and stress means were found in the group at high risk of OSA compared to the low and intermediate risk groups (Table 2).
A multinomial logistic regression, taking moderate vs. low OSA risk as the dependent variable, showed that older age (aOR = 1.05) and having hypertension vs. not (aOR = 7.80) were associated with higher odds of having moderate OSA. Female sex (aOR = 0.04) was significantly associated with lower odds of having moderate OSA compared to males (Table 3, Model 1).
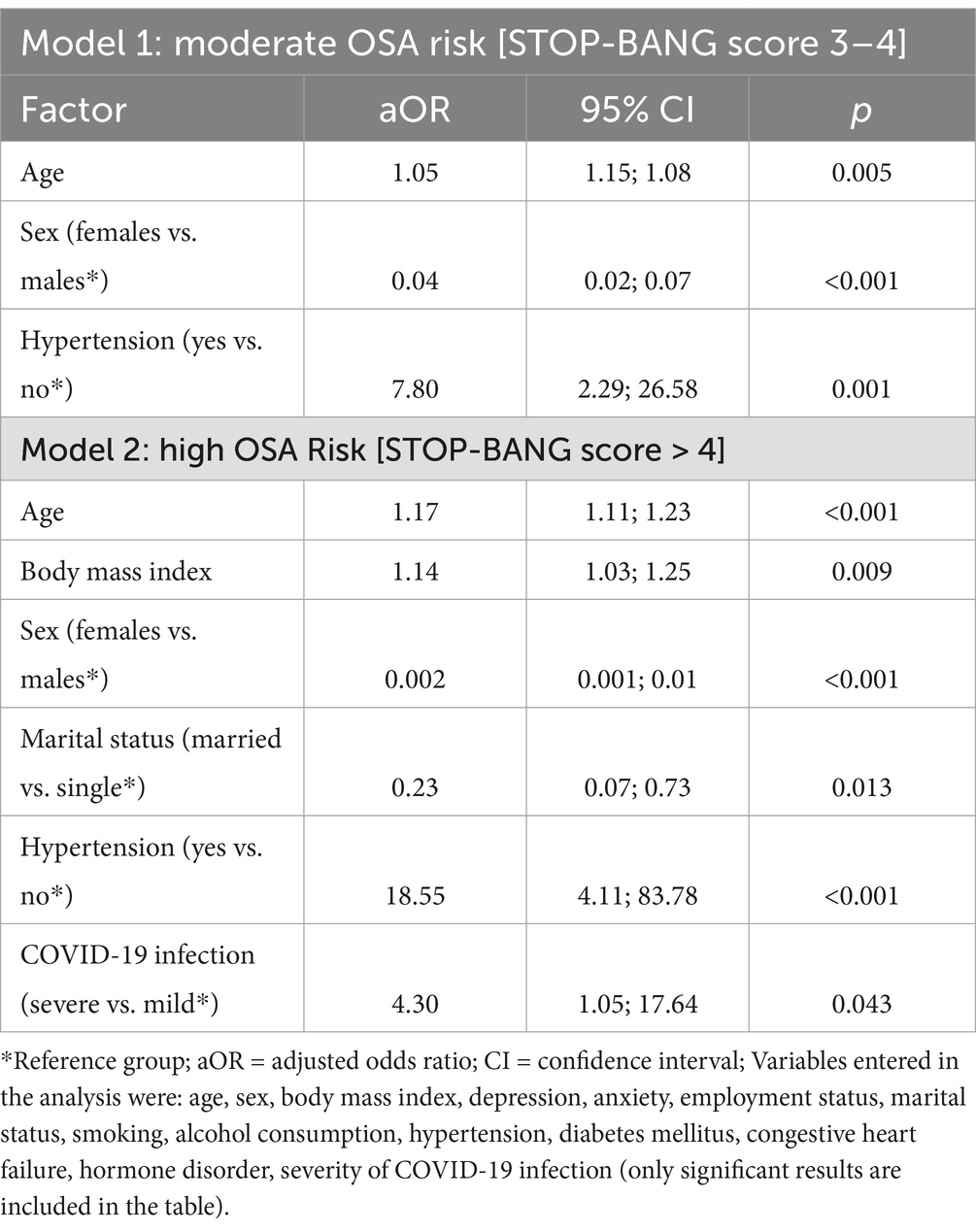
Table 3. Multinomial logistic regression taking OSA risk as the dependent variable (low OSA risk [STOP-BANG score 0–2] taken as the reference category).
A multinomial logistic regression, taking high vs. low OSA risk as the dependent variable, showed that older age (OR = 1.17), higher BMI (OR = 1.14), having hypertension vs. not (OR = 18.55), and having severe COVID-19 infection compared to mild (OR = 4.30) were significantly associated with higher odds of having high OSA, whereas female sex (OR = 0.002) and being married compared to single (aOR = 0.23) were significantly associated with lower odds of having high OSA (Table 3, Model 2).
To the best of our knowledge, this is the first large-scale study that assessed the correlates of obstructive sleep apnea risk among the Lebanese population. The results showed that older age, higher BMI, male sex, hypertension, marital status, and severe COVID-19 infection were associated with higher odds of having high OSA risk.
Our study showed that OSA is higher among patients with advanced age consistent with the findings from a previous study (3). A similar finding was observed in a study conducted on Saudi individuals, where the prevalence of OSA increased with age, independent of other potential confounding factors (30). Our results can be explained by the fact that older individuals suffer from reduced tethering of the upper airway due to the loss of the elastic recoil effect produced by the lung (1). In addition, older individuals suffer from an easier collapse in the airways caused by a reduction in the collagen, negative pressure reflux, and arousal threshold (1). Thus, the deposition of the parapharyngeal fat will increase, the soft palate will widen, and the bony shape seen around the pharynx will change. Moreover, the efficiency of the upper dilator muscles will fall with age accompanied by an incline in several health morbidities that act as risk factors for OSA occurrence (31, 32). Furthermore, our findings can be interpreted by the fact that hypertension is more prevalent in advanced age which helps in the understanding why OSA is more encountered in the older adult population (33).
Our study findings showed that OSA risk is higher at a BMI level of ≥35 kg/m2 consistent with findings from a previous study (30, 34). The association between obesity and OSA risk is a linear relationship that can be explained that in obesity more pressure will be applied on the airway tract through the increased disposition of aft around the neck area impairing the normal airflow and obstructing the respiratory tract site (35).
It has been documented that a 10% increase in body weight results in a sixfold increase in moderate to severe OSA and increases the apnea-hypopnea index (AHI) by 32% (36). Furthermore, it can be explained that obesity is not only a direct factor triggering OSA, it also acts as a confounding factor promoting other health morbidities such as hypertension, diabetes increased insulin resistance, and coronary artery diseases, which in return acts as major risk factors for OSA occurrence (37).
Our findings confirmed that male sex is at a higher risk for developing OSA, which is consistence with the literature (3, 38–40). Our findings are consistent with those of Al Qattan et al., who evaluated the risk of OSA among Kuwaiti individuals (5). Our results can be interpreted by the fact that males have bigger lung volume and tends to gain weight more centrally than do females which probably results in men having more fat stored in the upper airway structures and abdomen sites than do women (41). Several studies have confirmed that men have larger airways than do women which confirms the propensity in the airway collapse in men more than women (1). It has been also highlighted that in the STOP-BANG questionnaire males receive an extra score placing them at a higher risk for OSA development (42). It is explained in the literature that men have more bulky tongues and soft palate and more fat deposition at the mandible site increasing the risk of developing OSA (41). In-addition to the unique anatomy and physiology pertaining to males, there are certain social and lifestyle factors which are more common in males that increase risk of OSA as smoking, alcohol consumption, and early work hour with diminished night sleep hours (43). There are different resources that justified the higher prevalence of OSA among males compared to females by elucidating the fact that females do not report symptoms of snoring and the reluctance of women to acknowledge symptoms of OSA and seek medical help (44).
Our study reported a strong association between hypertension and a higher risk of OSA consistent with the findings by Borsini et al. and Silva et al. (45, 46). A study conducted in Turkey by Ozdemir et al. confirmed that the prevalence of OSA was as high as 9 times in hypertension compared to hypertension-free patients (32). The underlying reason can be explained by the fact that fluids at night are redistributed from the lower limbs to the neck imposes more airway pressure, exacerbates obstruction, and increases levels of angiotensin II and aldosterone in the nasopharynx and upper airway tissues in hypertension patients triggers OSA (47). In this study, OSA and hypertension coexisted together so it is difficult to build a unidirectional relationship since both are known to contribute to each other.
Our results showed that OSA risk is lower among married compared to single status consistent with the findings from another study (48). In contrast to our results, an Emirati study found higher prevalence of OSA risk in married groups compared to single, divorced or widowed groups (49). Our findings can be explained by the fact that insomnia-related symptoms mainly difficulty falling asleep is higher among unmarried individuals which increases the risk of developing OSA (50). It has been documented that irregular sleep rhythm and stronger night and day symptoms related to sleep disorders are more common among unmarried individuals which increases the risk of developing OSA (51, 52).
According to our findings, severe COVID-19 infection was associated with a higher risk of OSA consistent with the findings in another study conducted among US adults (53). Our findings can be explained by the fact that COVID-19 triggers an inflammatory state, especially in the lungs which can cause concentric fibrosis and can narrow the airway (54). It has been documented that patients infected with COVID-19 manifest with hypoxia and immune dysfunction that increases the risk to develop OSA (55).
Our study identified a significant correlation between obstructive sleep apnea (OSA) and both depression and anxiety, while no such correlation was observed with stress. This was demonstrated through analyses using the PHQ-9 Depression Scale, the LAS Anxiety Scale, and the BDS Distress Scale. Starting with depression, our findings align with those of Lee et al., demonstrating that a high PHQ-9 score is significantly associated with a risk of OSA (56). Depression can be considered a risk factor for OSA, possibly due to the effects of OSA on insular neurons, which may experience damage or dysfunction (57). Sleep fragmentation and intermittent hypoxia associated with OSA further exacerbate this relationship by altering neuronal activity and reducing white and gray matter in the brain, thereby intensifying depressive symptoms (58). Anxiety is considered another risk factor for OSA occurrence, this is because anxiety may influence brain regions, including the ventromedial prefrontal cortex, cingulate, parietal, and insular cortices, as well as the hippocampus and amygdala. These regions regulate respiratory control (59). These results are consistent with those reported by Duan et al., who found a strong association between anxiety and the increased risk of OSA (60). The connection between stress and sleep apnea is mostly indirect and it varies from one person to the other. Our results show no correlation between stress and OSA risk, which may be due to individual differences in how stress is perceived and experienced. This contrasts with findings from other studies, which have reported a stronger association between stress and the risk of OSA (61).
This study has some limitations. First, it is a cross-sectional study that suggests a possible association where additional research is needed to establish a definitive causal relationship. Second, selection bias existed as individuals aware of using social media platforms and having internet access were able to participate in this study. In addition, the refusal rate cannot be known. Furthermore, the under-diagnosis of OSA in our study may be due to the young age of our participants along with the limited comorbidities at young compared to older ages. We also recognize the potential bias in our analysis group, specifically the higher proportion of women and the relatively young average age of participants. While these demographic characteristics may limit the generalizability of our findings to broader populations, we aim to perform future studies with more diverse and representative samples, including older populations and a more balanced sex distribution, which are essential to confirm and expand upon our findings. Similarly, reporting certain diseases as diabetes mellitus and hypertension could potentially influence the interpretation of our study, as individuals may be asymptomatic leading to undiagnosed hypertension or diabetes, which might not be fully accounted for in the analysis. Finally, the assessment was through a self-reported questionnaire, thus, the information reported may be biased given that the responders might have reported wrong answers based on misunderstanding of some questions. Future studies incorporating the performance of polysomnography and assessment about snoring or sleepiness during driving are still needed to confirm our results as it remains the gold standard for diagnosing OSA. Similarly, home sleep apnea testing can also be considered as a practical alternative for confirming estimates, offering a more accessible option providing data on the presence and severity of OSA.
Our study has certain strengths. This was the first study to evaluate the risk of OSA in a large sample of Lebanese adults residing in different provinces and to correlate this risk with various demographic and clinical characteristics. In addition, the association of OSA with the level of health, anxiety, and distress was assessed utilizing nation-specific validated questionnaires.
This study showed that most Lebanese adults have low risk for OSA where the risk is strongly associated with nonmodifiable and modifiable factors. Our findings highlight the need for an intervention targeted to reduce the risk of OSA in Lebanese adults. Further future studies are needed to confirm the associations seen in our findings taking into consideration more specific factors and a follow-up period to assess a strong unidirectional relationship. Moreover, future health campaigns should be conducted to increase awareness among the Lebanese population about preventing and slowing the progression of OSA through lifestyle modifications. This is particularly important given the evidence of clinical under recognition, low awareness and low referral rates of OSA within the medical community in Lebanon. Enhanced awareness and early diagnosis can significantly improve outcomes and reduce the societal and individual impacts of OSA.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving human/animal participants were reviewed and approved by the Research Committee at the Lebanese International University School of Pharmacy with an approval number of 2021RC-007-LIUSOP. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
TK: Data curation, Methodology, Writing – original draft. MH: Data curation, Methodology, Writing – original draft. BC: Data curation, Methodology, Writing – original draft. FR: Data curation, Methodology, Writing – original draft. SBM: Data curation, Software, Writing – review & editing. SM: Writing – review & editing, Formal analysis. AA: Writing – review & editing. SE: Writing – review & editing. BH: Writing – review & editing. SH: Writing – review & editing, Formal analysis, Software, Validation. DM: Formal analysis, Writing – review & editing, Conceptualization, Data curation, Supervision. HH: Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors would like to thank all those who participated in this study by filling up the online survey.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
OSA, Obstructive sleep apnea; AASM, American Academy of Sleep Medicine; COVID-19, coronavirus disease 2019; GERD, gastro-esophageal reflex disorder; AHI, apnea-hypopnea index.
1. Jordan, AS, McSharry, DG, and Malhotra, A. Adult obstructive sleep apnoea. Lancet. (2014) 383:736–47. doi: 10.1016/s0140-6736(13)60734-5
2. Goodchild, TT, and Lefer, DJ. Obstructive sleep apnea: the not-so-silent killer. Circ Res. (2020) 126:229–31. doi: 10.1161/circresaha.119.316359
3. Senaratna, CV, Perret, JL, Lodge, CJ, Lowe, AJ, Campbell, BE, Matheson, MC, et al. Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev. (2017) 34:70–81. doi: 10.1016/j.smrv.2016.07.002
4. Gurubhagavatula, I, Sullivan, S, Meoli, A, Patil, S, Olson, R, Berneking, M, et al. Management of Obstructive Sleep Apnea in commercial motor vehicle operators: recommendations of the AASM sleep and transportation safety awareness task force. J Clin Sleep Med. (2017) 13:745–58. doi: 10.5664/jcsm.6598
5. Al-Qattan, H, Al-Omairah, H, Al-Hashash, K, Al-Mutairi, F, Al-Mutairat, M, Al-Ajmi, M, et al. Prevalence, risk factors, and comorbidities of obstructive sleep apnea risk among a working population in Kuwait: a cross-sectional study. Front Neurol. (2021) 12:620799. doi: 10.3389/fneur.2021.620799
6. Thompson, C, Legault, J, Moullec, G, Baltzan, M, Cross, N, Dang-Vu, TT, et al. A portrait of obstructive sleep apnea risk factors in 27, 210 middle-aged and older adults in the Canadian longitudinal study on aging. Sci Rep. (2022) 12:5127. doi: 10.1038/s41598-022-08164-6
7. Drager, LF, Togeiro, SM, Polotsky, VY, and Lorenzi-Filho, G. Obstructive sleep apnea: a cardiometabolic risk in obesity and the metabolic syndrome. J Am Coll Cardiol. (2013) 62:569–76. doi: 10.1016/j.jacc.2013.05.045
8. Young, T, Palta, M, Dempsey, J, Skatrud, J, Weber, S, and Badr, S. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. (1993) 328:1230–5. doi: 10.1056/nejm199304293281704
9. Reutrakul, S, and Mokhlesi, B. Obstructive sleep apnea and diabetes: a state of the art review. Chest. (2017) 152:1070–86. doi: 10.1016/j.chest.2017.05.009
10. Pochetti, P, Azzolina, D, Ragnoli, B, Tillio, PA, Cantaluppi, V, and Malerba, M. Interrelationship among obstructive sleep apnea, renal function and survival: a cohort study. Int J Environ Res Public Health. (2020) 17:4922. doi: 10.3390/ijerph17144922
11. Khawaja, IS, Kazaglis, L, Hashmi, A, Khurshid, KA, Eiken, C, and Hurwitz, TD. Obstructive sleep apnea and depression: a review. Psychiatr Ann. (2016) 46:187–91. doi: 10.3928/00485713-20160125-02
12. Peppard, PE, Szklo-Coxe, M, Hla, KM, and Young, T. Longitudinal association of sleep-related breathing disorder and depression. Arch Intern Med. (2006) 166:1709–15. doi: 10.1001/archinte.166.16.1709
13. Yeghiazarians, Y, Jneid, H, Tietjens, JR, Redline, S, Brown, DL, El-Sherif, N, et al. Obstructive sleep apnea and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. (2021) 144:e56–67. doi: 10.1161/cir.0000000000000988
14. Passarella, E, Czuzoj-Shulman, N, and Abenhaim, HA. Maternal and fetal outcomes in pregnancies with obstructive sleep apnea. J Perinat Med. (2021) 49:1064–70. doi: 10.1515/jpm-2020-0551
15. Ing, AJ, Ngu, MC, and Breslin, ABX. Obstructive sleep apnea and gastroesophageal reflux. Am J Med. (2000) 108:120–5. doi: 10.1016/S0002-9343(99)00350-2
16. Pan, L, Xie, X, Liu, D, Ren, D, and Guo, Y. Obstructive sleep apnoea and risks of all-cause mortality: preliminary evidence from prospective cohort studies. Sleep Breath. (2016) 20:345–53. doi: 10.1007/s11325-015-1295-7
17. Ghandour, AJ, Hammoud, H, and Al-Hajj, S. Analyzing factors associated with fatal road crashes: a machine learning approach. Int J Environ Res Public Health. (2020) 17:4111. doi: 10.3390/ijerph17114111
18. Olaithe, M, Bucks, RS, Hillman, DR, and Eastwood, PR. Cognitive deficits in obstructive sleep apnea: insights from a meta-review and comparison with deficits observed in COPD, insomnia, and sleep deprivation. Sleep Med Rev. (2018) 38:39–49. doi: 10.1016/j.smrv.2017.03.005
19. Coussa-Koniski, ML, Saliba, E, Welty, FK, and Deeb, M. Epidemiological characteristics of obstructive sleep apnea in a hospital-based historical cohort in Lebanon. PLoS One. (2020) 15:e0231528. doi: 10.1371/journal.pone.0231528
20. Chami, HA, Bechnak, A, Isma'eel, H, Talih, F, Nasreddine, L, Nasrallah, M, et al. Sleepless in Beirut: sleep difficulties in an urban environment with chronic psychosocial stress. J Clin Sleep Med. (2019) 15:603–14. doi: 10.5664/jcsm.7724
21. Haddad, NR, Aoun, CB, Mghames, A, Saleh, M, and Chahine, MN. Smoking habits and nicotine dependence among the general Lebanese population before and during both the economic crisis and COVID-19 pandemic. Tob Use Insights. (2024) 17:1179173x241253962. doi: 10.1177/1179173x241253962
22. Bouclaous, C, Azar, LJ, Barmo, N, Daher, R, Tabaja, J, El Hout, G, et al. Levels and correlates of numeracy skills in Lebanese adults with diabetes: a cross-sectional study. Int J Environ Res Public Health. (2022) 19:10557. doi: 10.3390/ijerph191710557
23. Dagher, D, Samaha, S, Mhanna, M, Azzi, R, Mina, A, Soufia, M, et al. Depressive symptoms among a sample of Lebanese adolescents: scale validation and correlates with disordered eating. Arch Pediatr. (2023) 30:401–7. doi: 10.1016/j.arcped.2023.04.007
24. Sawaya, H, Atoui, M, Hamadeh, A, Zeinoun, P, and Nahas, Z. Adaptation and initial validation of the patient health questionnaire - 9 (PHQ-9) and the generalized anxiety disorder - 7 questionnaire (GAD-7) in an Arabic speaking Lebanese psychiatric outpatient sample. Psychiatry Res. (2016) 239:245–52. doi: 10.1016/j.psychres.2016.03.030
25. Hallit, S, Obeid, S, Haddad, C, Hallit, R, Akel, M, Haddad, G, et al. Construction of the Lebanese anxiety scale (LAS-10): a new scale to assess anxiety in adult patients. Int J Psychiatry Clin Pract. (2020) 24:270–7. doi: 10.1080/13651501.2020.1744662
26. Merhy, G, Azzi, V, Salameh, P, Obeid, S, and Hallit, S. Anxiety among Lebanese adolescents: scale validation and correlates. BMC Pediatr. (2021) 21:288. doi: 10.1186/s12887-021-02763-4
27. Malaeb, D, Farchakh, Y, Haddad, C, Sacre, H, Obeid, S, Hallit, S, et al. Validation of the Beirut distress scale (BDS-10), a short version of BDS-22, to assess psychological distress among the Lebanese population. Perspect Psychiatr Care. (2022) 58:304–13. doi: 10.1111/ppc.12787
28. BaHammam, AS, Al-Aqeel, AM, Alhedyani, AA, Al-Obaid, GI, Al-Owais, MM, and Olaish, AH. The validity and reliability of an Arabic version of the STOP-Bang questionnaire for identifying obstructive sleep apnea. Open Respir Med J. (2015) 9:22–9. doi: 10.2174/1874306401509010022
29. Chung, F, Yegneswaran, B, Liao, P, Chung, SA, Vairavanathan, S, Islam, S, et al. STOP questionnaire: a tool to screen patients for obstructive sleep apnea. Anesthesiology. (2008) 108:812–21. doi: 10.1097/ALN.0b013e31816d83e4
30. Wali, SO, Abalkhail, B, and Krayem, A. Prevalence and risk factors of obstructive sleep apnea syndrome in a Saudi Arabian population. Ann Thorac Med. (2017) 12:88–94. doi: 10.4103/1817-1737.203746
31. Malhotra, A, Huang, Y, Fogel, R, Lazic, S, Pillar, G, Jakab, M, et al. Aging influences on pharyngeal anatomy and physiology: the predisposition to pharyngeal collapse. Am J Med. (2006) 119:72.e9–14. doi: 10.1016/j.amjmed.2005.01.077
32. Marcus, CL. Obstructive sleep apnea syndrome: differences between children and adults. Sleep. (2000) 23:S140–1.
33. Chaudhary, SC, Gupta, P, Sawlani, KK, Gupta, KK, Singh, A, Usman, K, et al. Obstructive sleep apnea in hypertension. Cureus. (2023) 15:e38229. doi: 10.7759/cureus.38229
34. Sareli, AE, Cantor, CR, Williams, NN, Korus, G, Raper, SE, Pien, G, et al. Obstructive sleep apnea in patients undergoing bariatric surgery--a tertiary center experience. Obes Surg. (2011) 21:316–27. doi: 10.1007/s11695-009-9928-1
35. Patial, K, Mishra, HP, Pal, G, Suvvari, TK, Ghosh, T, Mishra, SS, et al. Understanding the association between obesity and obstructive sleep apnea syndrome: a case-control study. Cureus. (2023) 15:e45843. doi: 10.7759/cureus.45843
36. Peppard, PE, Young, T, Palta, M, Dempsey, J, and Skatrud, J. Longitudinal study of moderate weight change and sleep-disordered breathing. JAMA. (2000) 284:3015–21. doi: 10.1001/jama.284.23.3015
37. Mitra, AK, Bhuiyan, AR, and Jones, EA. Association and risk factors for obstructive sleep apnea and cardiovascular diseases: a systematic review. Diseases. (2021) 9:88. doi: 10.3390/diseases9040088
38. Hoflstein, V. Relationship between smoking and sleep apnea in clinic population. Sleep. (2002) 25:519–24.
39. Ong, JC, Gress, JL, San Pedro-Salcedo, MG, and Manber, R. Frequency and predictors of obstructive sleep apnea among individuals with major depressive disorder and insomnia. J Psychosom Res. (2009) 67:135–41. doi: 10.1016/j.jpsychores.2009.03.011
40. Young, T, Shahar, E, Nieto, FJ, Redline, S, Newman, AB, Gottlieb, DJ, et al. Predictors of sleep-disordered breathing in community-dwelling adults: the sleep heart health study. Arch Intern Med. (2002) 162:893–900. doi: 10.1001/archinte.162.8.893
41. Whittle, AT, Marshall, I, Mortimore, IL, Wraith, PK, Sellar, RJ, and Douglas, NJ. Neck soft tissue and fat distribution: comparison between normal men and women by magnetic resonance imaging. Thorax. (1999) 54:323–8. doi: 10.1136/thx.54.4.323
42. Chung, F, Abdullah, HR, and Liao, P. STOP-Bang questionnaire: a practical approach to screen for obstructive sleep apnea. Chest. (2016) 149:631–8. doi: 10.1378/chest.15-0903
43. Esen, AD, and Akpinar, M. Relevance of obstructive sleep apnea and smoking: obstructive sleep apnea and smoking. Fam Pract. (2021) 38:180–5. doi: 10.1093/fampra/cmaa112
44. Wimms, A, Woehrle, H, Ketheeswaran, S, Ramanan, D, and Armitstead, J. Obstructive sleep apnea in women: specific issues and interventions. Biomed Res Int. (2016) 2016:1764837–9. doi: 10.1155/2016/1764837
45. Borsini, E, Blanco, M, Bosio, M, Schrappe, M, Ernst, G, Nosetto, D, et al. Prevalence of sleep apnea and cardiovascular risk factors in patients with hypertension in a day hospital model. Clin Exp Hypertens. (2018) 40:231–7. doi: 10.1080/10641963.2017.1356841
46. Silva, R, Brito, TP, Wanderley Neto, AC, Frota, RB, and Melo, JC. Major risk factors for obstructive sleep apnea monitored in the home. A cross-sectional study. Sao Paulo medical journal =. Rev Paul Med. (2021) 139:643–7. doi: 10.1590/1516-3180.2020.0689.R1.22042021
47. Bangash, A, Wajid, F, Poolacherla, R, Mim, FK, and Rutkofsky, IH. Obstructive sleep apnea and hypertension: a review of the relationship and pathogenic association. Cureus. (2020) 12:e8241. doi: 10.7759/cureus.8241
48. Goosmann, M, Williams, AM, Springer, K, and Yaremchuk, KL. The impact of marital status and race in obstructive sleep apnea. Ear Nose Throat J. (2022). doi: 10.1177/01455613221120068, [Online ahead of print]
49. Taimah, M, Ahmad, A, Al-Houqani, M, Al Junaibi, A, Idaghdour, Y, Abdulle, A, et al. Association between obstructive sleep apnea risk and type 2 diabetes among Emirati adults: results from the UAE healthy future study. Front Endocrinol. (2024) 15:1395886. doi: 10.3389/fendo.2024.1395886
50. Matsumoto, Y, Uchimura, N, and Ishitake, T. The relationship between marital status and multifactorial sleep in Japanese day workers. Sleep Biol Rhythms. (2022) 20:211–7. doi: 10.1007/s41105-021-00357-2
51. Kim, Y, Ramos, AR, Carver, CS, Ting, A, Hahn, K, Mossavar-Rahmani, Y, et al. Marital status and gender associated with sleep health among Hispanics/Latinos in the US: results from HCHS/SOL and Sueño ancillary studies. Behav Sleep Med. (2022) 20:531–42. doi: 10.1080/15402002.2021.1953499
52. Matsumoto, Y, Uchimura, N, Ishida, T, Toyomasu, K, Morimatsu, Y, Mori, M, et al. Day workers suffering from a wider range of sleep problems are more likely to experience suicidality. Sleep Biol Rhythms. (2016) 14:369–76. doi: 10.1007/s41105-016-0067-5
53. Quan, SF, Weaver, MD, Czeisler, M, Barger, LK, Booker, LA, Howard, ME, et al. Associations between obstructive sleep apnea and COVID-19 infection and hospitalization among US adults. J Clin Sleep Med. (2023) 19:1303–11. doi: 10.5664/jcsm.10588
54. Elicker, BM. What are the long-term pulmonary sequelae of COVID-19 infection? Radiology. (2022) 304:193–4. doi: 10.1148/radiol.220449
55. Dinh, Y, Alawady, A, Alhazmi, H, Altabtbaei, K, Freire, M, Alghounaim, M, et al. Association between risk of obstructive sleep apnea severity and risk of severe COVID-19 symptoms: insights from salivary and serum cytokines. Front Public Health. (2024) 12:1348441. doi: 10.3389/fpubh.2024.1348441
56. Lee, MR, and Jung, SM. Obstructive sleep apnea related to mental health, health-related quality of life and multimorbidity: a nationwide survey of a representative sample in Republic of Korea. PLoS One. (2023) 18:e0287182. doi: 10.1371/journal.pone.0287182
57. Kang, J, Tian, Z, and Li, M. Changes in insular cortex metabolites in patients with obstructive sleep apnea syndrome. Neuroreport. (2018) 29:981–6. doi: 10.1097/wnr.0000000000001065
58. Wheaton, AG, Perry, GS, Chapman, DP, and Croft, JB. Sleep disordered breathing and depression among U.S. adults: National Health and nutrition examination survey, 2005-2008. Sleep. (2012) 35:461–7. doi: 10.5665/sleep.1724
59. Macey, KE, Macey, PM, Woo, MA, Henderson, LA, Frysinger, RC, Harper, RK, et al. Inspiratory loading elicits aberrant fMRI signal changes in obstructive sleep apnea. Respir Physiol Neurobiol. (2006) 151:44–60. doi: 10.1016/j.resp.2005.05.024
60. Duan, X, Zheng, M, Zhao, W, Huang, J, Lao, L, Li, H, et al. Associations of depression, anxiety, and life events with the risk of obstructive sleep apnea evaluated by Berlin questionnaire. Front Med. (2022) 9:799792. doi: 10.3389/fmed.2022.799792
Keywords: obstructive sleep apnea, hypertension, sex, correlates, COVID-19
Citation: Kaddoura T, Hodroj M, Chmeis B, Rammal F, Malhab SB, Mansour S, Akour A, El Khatib S, Hosseini B, Hallit S, Malaeb D and Hosseini H (2025) Assessment of obstructive sleep apnea rate and associated factors among Lebanese adults: a cross-sectional study. Front. Public Health. 13:1443920. doi: 10.3389/fpubh.2025.1443920
Received: 04 June 2024; Accepted: 31 January 2025;
Published: 13 February 2025.
Edited by:
Jie Sun, Peking University, ChinaReviewed by:
Mohammed Al-Abri, Sultan Qaboos University, OmanCopyright © 2025 Kaddoura, Hodroj, Chmeis, Rammal, Malhab, Mansour, Akour, El Khatib, Hosseini, Hallit, Malaeb and Hosseini. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Diana Malaeb, RHIuZGlhbmFAZ211LmFjLmFl
†These authors share first authorship
‡These authors share last authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.