
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Public Health , 11 October 2024
Sec. Infectious Diseases: Epidemiology and Prevention
Volume 12 - 2024 | https://doi.org/10.3389/fpubh.2024.1419425
Background: Severe fever with thrombocytopenia syndrome (SFTS) is an acute infectious disease, which was first reported in 2009 in China. Previous studies have rarely quantitatively assessed the transmission and fatal risk of SFTS clusters.
Methods: Epidemiological information regarding SFTS clusters in Yantai city of Shandong province during 2013-2022 was obtained from the National Public Health Emergency Event Surveillance System (PHEESS) for Disease Control and Prevention information system. The secondary attack rate (SAR) and relative risk (RR) were used to assess the risk of human-to-human transmission of SFTS.
Results: A total of 20 SFTS clusters involving 51 laboratory-confirmed patients were reported between 2013 and 2022 in Yantai city, Shandong province. Most of the clusters occurred from May to October, and the patients were mainly distributed in four counties. Contact with blood or other fluids [RR = 14.06, 95% confidence interval (CI) = 3.29–70.65, p < 0.001] and using no personal protection equipment (PPE) [11.63% (10/86) vs. 2.22% (2/90), RR = 5.74, 95% CI = 1.17–55.44, p = 0.013] were significantly related with an increased risk of SFTS virus (SFTSV) transmission.
Conclusion: Our study may provide direct guidance on health education and behavioral interventions for the accompanying relatives and personnel of SFTS patients, both during their hospital stay and upon returning home after discharge.
Severe fever with thrombocytopenia syndrome (SFTS) is an acute infectious disease that seriously imposes substantial burden on public health. It was first reported in Hubei and Henan provinces in China from March to July 2009, and afterward, it rapidly spread to other provinces in the central, eastern, and northeastern regions (1). Later cases of SFTS were subsequently reported in countries of East Asia and South Asia (2, 3). In 2012, a novel phlebovirus was discovered in the Americas, the symptoms of which closely resembled those of SFTS (4). The World Health Organization has since recognized the critical nature of SFTS and listed it among the priority diseases for emergency research and development in 2018 (5).
The pathogen, known as SFTS virus (SFTSV), was initially isolated by the Chinese Center for Disease Control and Prevention (6). The International Committee on Taxonomy of Viruses named the newly discovered virus Dabie bandavirus in 2019, but the designation SFTSV continues to be used worldwide (7). Some of the early-diagnosed SFTS patients had a history of tick bites or found ticks in their living areas before showing symptoms of SFTS, and the sequences amplified from ticks and humans showed a high level of identity, indicating that SFTSV may be transmitted through ticks (8–10). SFTSV can be detected in ticks at different developmental stages and be transmitted by transstadial and transovarial routes (11, 12). SFTSV ribonucleic acid (RNA) has been detected in several tick species, including Haemaphysalis longicornis (H. longicornis), Haemaphysalis flava (H. flava), Ixodes nipponensis (I. nipponensis), and Amblyomma testudinarium (A. testudinarium), but it can only be isolated from H. longicornis ticks (6). Luo et al. (12) found that H. longicornis ticks are mainly a reservoir and vector of SFTSV in China. Domestic animals, encompassing goats, cattle, sheep, pigs, dogs, and chickens, are considered potential reservoir hosts for SFTSV. Niu et al. (13) conducted a study focusing on virus isolation and gene sequencing from H. longicornis ticks, SFTS patients, and domestic animals at Yantai city in Shandong Province, and it was found that all sequences shared more than 95% identity.
A substantial corpus of studies has filled the gap in the understanding of the transmission of SFTSV between humans and animals and between humans and humans. SFTSV can be detected in the serum, eye swab, saliva, rectal swab, and urine of animals, which underscores the potential for direct zoonotic transmission to humans from animals afflicted with SFTS (14, 15). Nam et al. reported a companion dog that exhibited fever, vomiting, and other SFTS symptoms, which were subsequently confirmed through real-time reverse transcription polymerase chain reaction (PCR) sequencing and an indirect immunofluorescence assay (16). A study from Japan reported that two veterinary personnel were infected with SFTSV from a sick cat, and the whole-genome sequences of the virus isolated from the infected individuals were found to exhibit 100% identity (17). In addition, SFTSV transmission can occur through direct contact of the unprotected skin or mucosa with the bodily fluids of infected humans (18). Exposure to blood or bloody secretions while accompanying patients or while handling a corpse after death is the most common mode of transmission. An observed dose–response relationship between the frequency of contact with blood and the incidence of SFTS infection highlights the elevated risk associated with increased exposure (19). Transmission through aerosols in enclosed environments can also cause SFTSV infections in individuals. Moon et al. (20) investigated the potential nosocomial aerosol transmission of SFTSV and suggested that airborne precautions be adopted to avoid direct contact and droplet transmission. A number of studies reported human-to-human transmission clusters, but only a few quantitatively assessed the human-to-human transmission risk among SFTS clusters.
In this study, we summarized all SFTS cluster outbreaks between 2013 and 2022 in Yantai city, explored the correlations of potential risk factors and SFTSV infection, and quantified the risk factors.
This study was reviewed and approved by the Ethics Committee of the Yantai Center for Disease Control and Prevention. It is important to note that our research did not involve any direct interaction with human participants or experimental procedures on humans. The only human materials used were the blood samples collected from SFTS patients and their contacts for public health purposes, and written informed consent for the use of their clinical samples was obtained from these patients.
The diagnosis of confirmed SFTS patients requires three points. First, a person who has a history of working, living, or traveling in hilly, forested, mountainous, or other areas during the epidemic season or has a history of tick bites within 2 weeks of disease onset. Second, a person who develops clinical symptoms such as fever, fatigue, significant anorexia, nausea, and vomiting, accompanied by leukopenia and thrombocytopenia. Third, at least one of the following laboratory findings is required: (1) the identification of SFTSV ribonucleic acid in the patient’s blood samples via molecular diagnostic assays; (2) the successful isolation of the SFTSV; (3) seroconversion or a substantial (> fourfold) increase in the specific IgG antibody titer against SFTSV between acute and recovery serum samples.
SFTS clusters are identified by two or more reported cases of patients with SFTS who work or visit the same locations, such as villages, hills, forests, tea gardens, or scenic areas. In addition, clusters include related cases reported among the close contacts of the initial patient within the longest incubation period of SFTS.
The criteria for identifying human-to-human transmission clusters in SFTS are as follows: (1) Secondary cases of patients who have no history of tick bites, visible tick bite marks on their bodies, or contact with animals in the 2 weeks preceding symptom onset; (2) Secondary cases of patients who have had direct, unprotected contact with the blood, bloody secretions, or other bodily fluids of the primary SFTS case within 2 weeks before their symptom onset; and (3) Secondary cases of patients with the onset of symptoms occurring within the established incubation period following exposure to the primary case.
In our study, the term “index patient” refers to the individual who was first infected with SFTSV through exposure to ticks or other routes.
The secondary attack rate (SAR) refers to the percentage of the affected individuals among all close susceptible contacts between the shortest and longest incubation periods of certain infectious diseases after exposure to a primary case. The calculation formula is as follows:
SAR (%) = number of affected individuals between the shortest and longest incubation periods/total number of susceptible contacts × 100%.
The prevention and control of SFTS in China follows national guidelines for notifiable infectious diseases. All suspected and confirmed cases are required to be reported to the National Notifiable Diseases Surveillance System (NNDSS) within 24 h. The clusters are also required to be notified to the National Public Health Emergency Event Surveillance System (PHEESS). The reported data include age, sex, permanent address, occupation, and key dates related to the onset, diagnosis, and outcome. Laboratory-confirmed SFTS cases from 1 January 2013 to 31 December 2022 were retrieved from the NNDSS for analysis.
An epidemiological investigation was carried out by well-trained interviewers using a face-to-face approach. A structured questionnaire was used to collect detailed information about each SFTS cluster. The reacquired information contained demographical information (age, sex, permanent address, occupation, etc.), exposure history within the 1st month before the onset (time, location, cluster settings, and infection route), information about infected individuals and deaths, laboratory test results (IgG titer and whether the SFTSV RNA was positive), and control measures (hospital infection control measures and environmental disinfection).
Descriptive epidemiological methods were carried out to analyze the temporal and spatial distribution of the clusters. SARs and an χ2 test were used to evaluate the transmissibility and relative risk (RR). A normality test was carried out for between-group comparisons of age and the time interval from the onset to confirmation. In addition, t-tests were conducted for the normally distributed data, and χ2 tests were conducted for the categorical variables. All calculations were performed using R studio (version 4.3.2; R Foundation for Statistical Computing, Vienna, Austria), and a p-value <0.05 was considered to have statistical significance.
As shown in Table 1 and Figure 1, 20 SFTS clusters were reported between 2013 and 2022 in Yantai city, Shandong province, with 51 laboratory-confirmed cases, including nine fatalities (17.65%. 9/51). The SFTS clusters showed a seasonal distribution, and the proportions from May to October were 15.69% (8/51), 35.29% (18/51), 11.76% (6/51), 11.76% (6/51), 21.57% (11/51), and 3.92% (2/51), respectively.
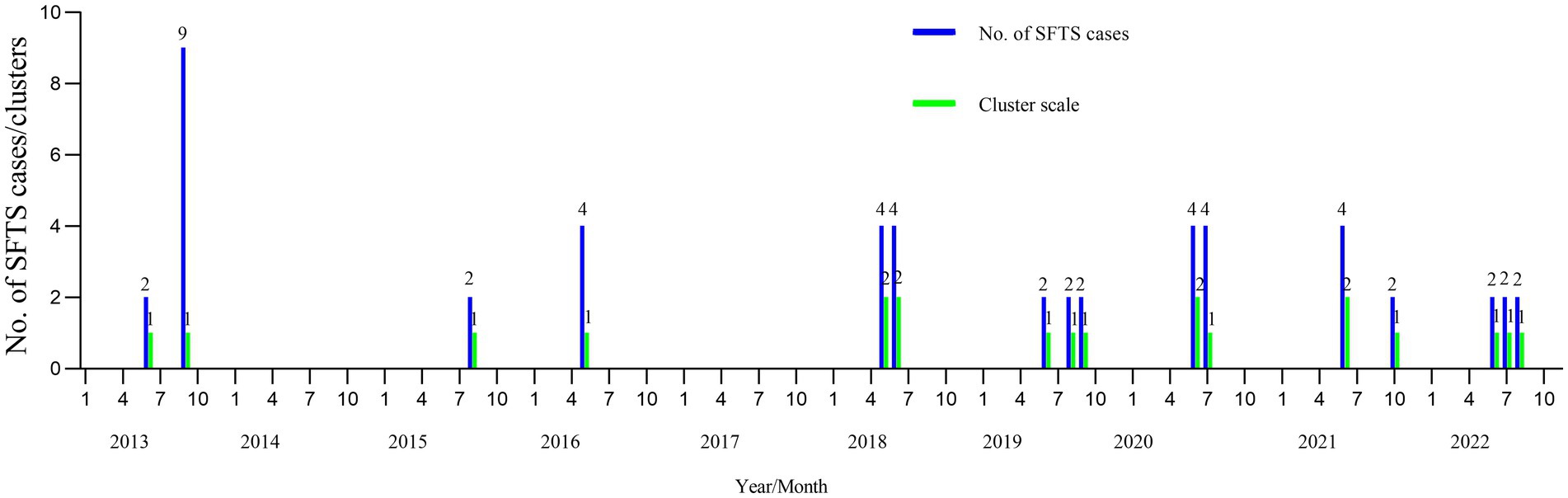
Figure 1. The seasonality of the SFTS clusters in Yantai city from 2013 to 2022. The horizontal axis represents January to December from 2013 to 2012. The vertical axis represents the number of cases and clusters. SFTS, severe fever with thrombocytopenia syndrome.
A total of eight counties reported the SFTS clusters. The cases shown in Figure 2 were mainly distributed in Laizhou (n = 14), Penglai (n = 13), Qixia (n = 6), Zhaoyuan (n = 6), Haiyang (n = 4), Laiyang (n = 4), Laishan (n = 2), and Fushan (n = 2). The distribution of sex was nearly equal, with 26 male patients (50.98%) and 25 female patients (49.02%). The majority of the patients were farmers (74.51%), and 76.47% of the patients were in the 50–79 age group.
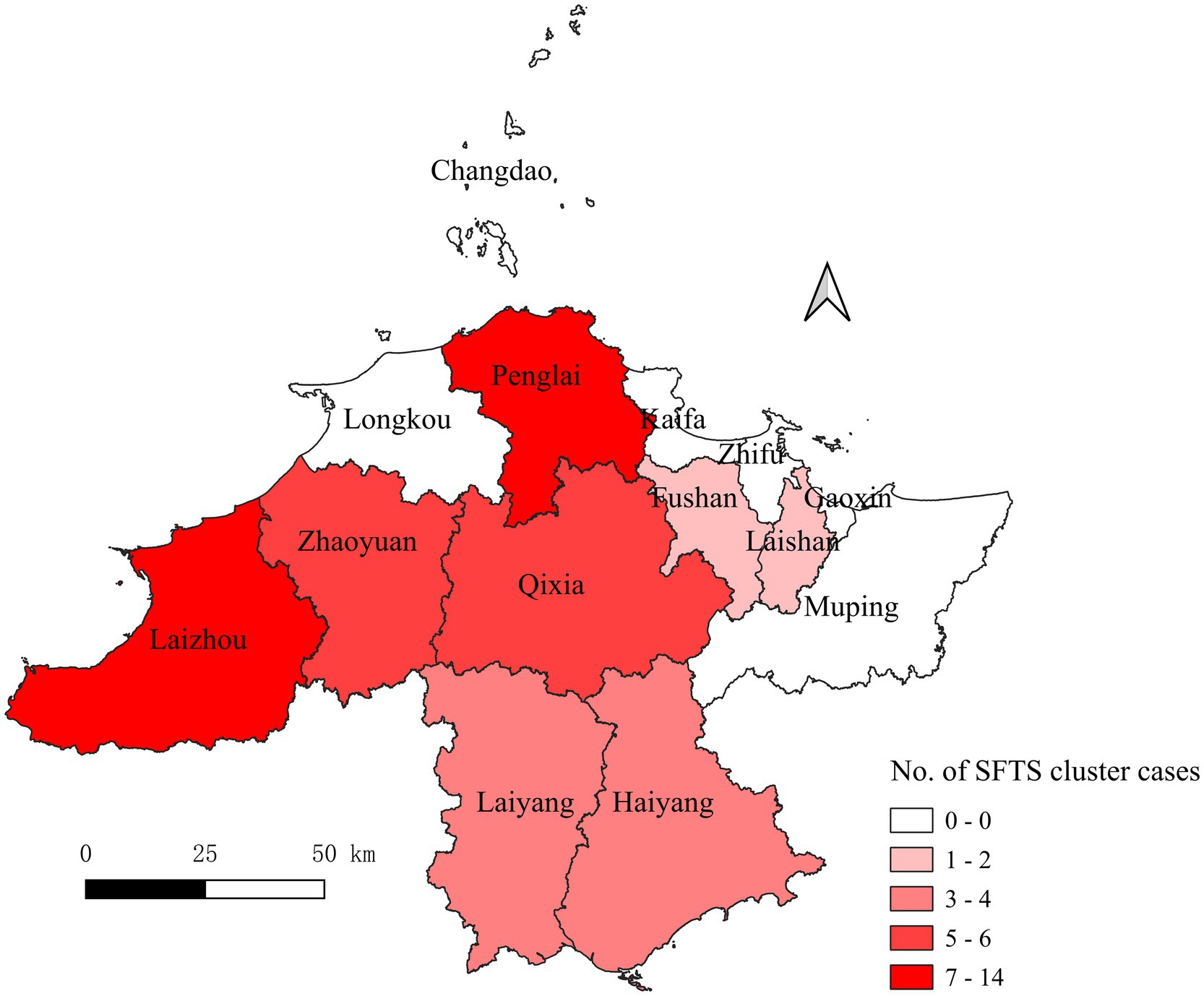
Figure 2. County distributions of the accumulative SFTS cases in Yantai city from 2013 to 2022. SFTS, severe fever with thrombocytopenia syndrome. The numbers indicate the patients from the clusters.
The epidemiological findings revealed that six clusters were due to tick bites, 10 were suspected to be tick-related, and four had unknown infection sources. Of them, three clusters resulted in SFTS secondary cases through human-to-human transmission (one occurred in the hospital and two occurred in both the hospital and the patients’ homes), and the remaining 17 SFTS clusters were caused by the same external environment. Among the three human-to-human clusters, the secondary patients were family members, relatives, and neighbors of the index patients. They were infected with SFTSV through blood contact (i.e., contact with blood or bloody fluids and secretions from the patients) and non-blood contact (i.e., contact with patients’ fluids or secretions other than blood or aerosol). As shown in Figure 3A, Patients I and E took care of the index patient. Along with Patient G, they participated in the removal of medical devices after Patient A died. Other secondary patients came in contact with Patient A’s corpse. Another human-to-human cluster was caused by blood contact of the index patient (Figure 3B). Figure 3C shows that Patients C and D were exposed to the blood of the index patient, while Patient B did not have direct contact with the blood of the index patient. They all remained in a closed space where the patient’s body and her blood-contaminated clothing were stored for a long time. Thus, we speculated that Patients C and D may have been infected with SFTSV through blood or aerosols and Patient B through aerosols.
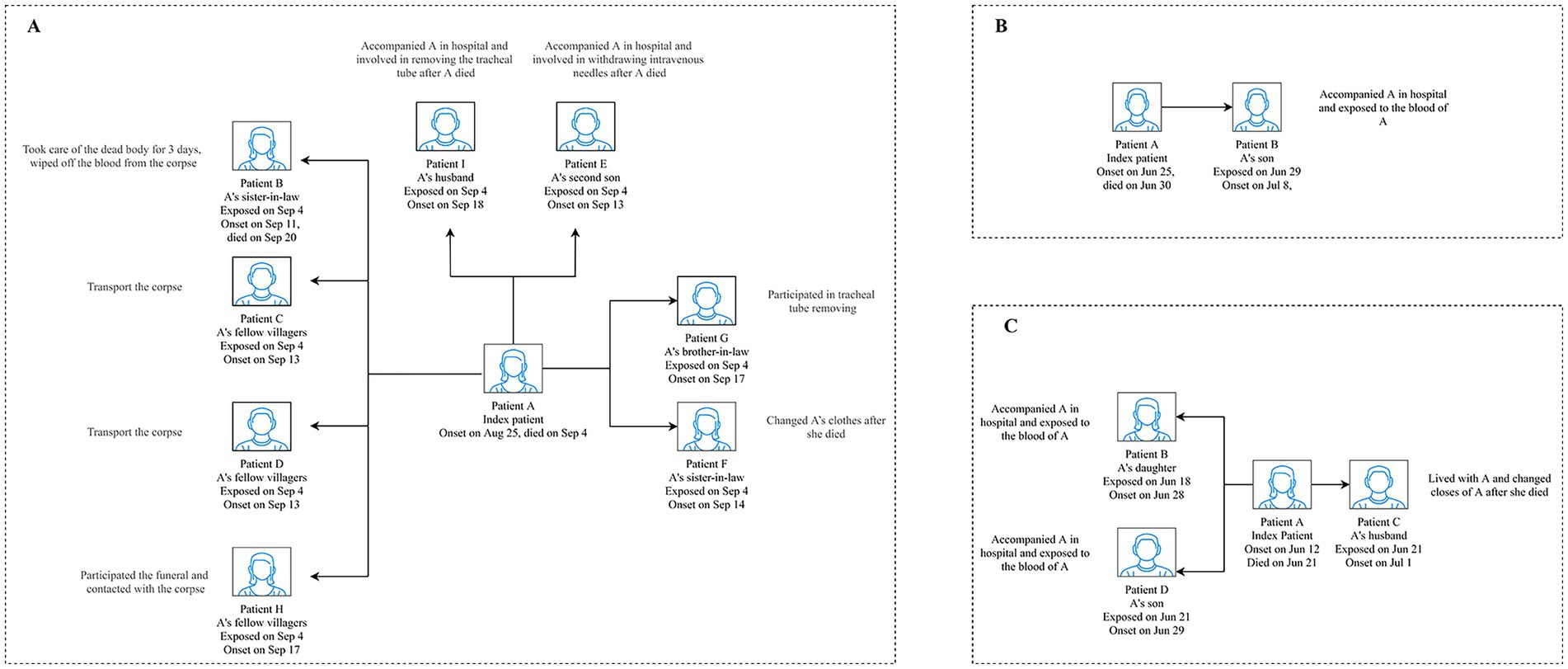
Figure 3. Transmission model of the three human-to-human transmission modes of the SFTS clusters in Yantai city. A displays the cluster that occurred in Penglai county in 2013. B displays the cluster that occurred in Laizhou county in 2019. C displays the cluster that occurred in Zhaoyuan county in 2020. SFTS, severe fever with thrombocytopenia syndrome.
As shown in Table 2, the SAR of the blood and other fluids contact transmission was higher than that of the non-blood contact transmission [23.33% (7/30) vs. 2.07% (4/193), RR = 14.06, 95% confidence interval (CI) = 3.29–70.65, p < 0.001]. In addition, not using personal protection equipment (PPE) showed a higher SAR for the contacts [11.63% (10/86) vs. 2.22% (2/90), RR = 5.74, 95% CI = 1.17–55.44, p = 0.013].
Supplementary Table 3 summarizes the risk factors for mortality in the SFTS clusters, which included sex, age, occupation, number of referrals, period from onset to diagnosis, and transmission route. Notably, no significant associations were found between these factors and SFTS-related deaths.
Our study analyzed 20 SFTS clusters in Yantai city (2013–2022), with 17 linked to environmental exposure and 3 to human-to-human transmission. Among the 51 lab-confirmed cases, nine were fatal (17.65%). The clusters were predominantly distributed in Laizhou, Penglai, Qixia, and Zhaoyuan, peaking in summer and autumn. Most patients were farmers aged 50–79 years. Coming in contact with the patients’ blood or other fluids and a lack of PPE significantly correlated with human-to-human SFTSV transmission.
Ticks thrive in mountainous, grassy, or forested areas, with climate factors such as temperature and rainfall influencing their populations, impacting tick-borne viral diseases (21, 22). A study from Brazil showed that female ticks optimally reproduce at 28°C and 80% RH, with lower efficiency and higher mortality at 4°C (23). The peak nymph activity coincides with increased human outdoor activity, elevating exposure risks (24). Deng et al. found that the incidence of SFTS showed an increasing trend as the tick density increased and that the latter was positively correlated with air temperature, wind speed, and sunshine duration (25). Our findings confirmed that SFTS infection peaks in summer and autumn, highlighting the need for heightened tick bite prevention. Yantai city is categorized by mountainous and hilly topography where farmers live and engage in agricultural labor, such as farming, mowing, and herding. These factors increase the risk of exposure to the same natural environment as that of ticks.
All three index patients died, while only one secondary patient died in the SFTS clusters in our study; this finding was similar to that of some studies (26, 27). The clinical symptoms and laboratory findings may explain the secondary patient’s death. Patient B (Figure 3A), along with exhibiting common SFTS symptoms, exhibited skin silt ecchymosis, limb tremor, and respiratory failure and died from disseminated intravascular coagulation. In addition, the patient’s viral load, activated partial thromboplastin time, alanine transaminase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), creatine kinase (CK), and creatinine levels were higher than those of the survivors, which may indicate impaired liver, heart, and kidney function. Numerous studies have provided epidemiological evidence of the relationship between these factors and mortality (28–31). The recovery of the other secondary patients could be attributed to more timely diagnosis and treatment, which potentially reduced the viral load compared to the index patients (27).
SFTS patients typically exhibit fever, thrombocytopenia, gastrointestinal symptoms, hemorrhagic tendency, and multiple organ dysfunctions, along with elevated SFTS RNA levels (7). Jeong et al. documented persistently positive SFTSV RNA levels in a patient’s serum samples, along with post-plasma exchange, with positive reactions also observed in the tracheal and gastric aspirates and the urine (S and M segment RNA Ct values ranging from 28.55 to 39.22) (32). In addition, a Japanese study reported SFTSV RNA levels in the cerebrospinal fluid and bronchoalveolar lavage fluid (4.10 and 2.51 log10 copies/mL, respectively), with sputum positivity lasting for roughly 4 months post-onset (33). Zhang et al. collected blood, throat swab, urine, or feces samples from patients with SFTS and found that SFTSV RNA levels can be detected in urine, fecal, and sputum specimens (34). The epidemiological data revealed that most SFTS patients exhibited hemorrhagic symptoms after death (35, 36). In some rural areas of China, when patients are on the brink of death, their families will take them home and after death, hold funerals and then cremate the bodies. The process includes changing clothes, applying makeup, and offering family condolences, and it is highly likely to cause human-to-human transmission of SFTSV. Fang et al. analyzed 27 SFTS clusters from China and Korea and found that direct blood contact, bloody secretion contact, and bloody droplet contact had a higher risk of transmitting SFTSV infection [OR = 6.35 (95% CI: 3.26–12.37), 38.01 (95% CI,19.73–73.23), and 2.27 (95% CI,1.01–5.19)] (26). Similarly, all of our deceased index patients in the SFTS clusters exhibited bleeding, with a significantly higher SAR among those exposed to blood or fluids [23.33% (7/30) vs. 2.07% (4/193)]. Furthermore, contact with blood and other fluids was found to be a significant risk factor for SFTSV transmission (RR = 14.06, 95% CI = 3.29–70.65, p < 0.001). This underscores the critical need for caution when handling the bodily fluids of SFTS patients.
The precise mechanisms of blood-to-body SFTSV transmission remain largely unexplored. Potential routes may involve open wounds, compromised skin integrity, and mucosal surfaces of the eyes, mouth, or nose (37). SFTSV disrupts the host’s immunity through a complex cascade, impacting the diverse immune cells, inflammatory mediators, inflammasomes, and signaling pathways (38). Moreover, the compromised adaptive immune response in SFTS patients indicates that the virus induces severe immunological dysregulation by affecting the differentiation of the targeted immune cells (7).
Few secondary patients present open wounds upon exposure to index patients with SFTS, suggesting transmission via oral, nasal, or ocular mucous membranes. The results of a survey on an SFTS cluster indicated that all secondary patients wore gloves and masks during blood contact with the index patient, but none used eye protection (39). Zhou et al. demonstrated that SFTSV could be transmitted through oral or ocular membranes (40). Therefore, it is necessary to wear a full set of PPE when in contact with SFTS patients. In our study, none of the healthcare workers were infected with SFTSV, which may have been due to their standard personal protection. In addition, using PPE during contact could have significantly protected them from SFTSV infection (RR = 5.74, 95% CI = 1.17–55.44, p = 0.03).
This study has some limitations, which are worth considering when interpreting its conclusions. First, the data of our study were from the PHEESS, which might not reflect the real-world situation due to the sensitivity of the monitoring system and report bias. However, the information in our study is the best available database regarding SFTS clusters. Second, owing to laboratory constraints, we were unable to obtain viral loads or conduct gene sequence alignments for human-to-human clusters. Therefore, memory biases might have obscured precise exposure histories, although homologous exposures are deemed unlikely. Finally, we did not analyze the risk indicators for the patients who died due to a lack of clinical information.
In conclusion, we reported 20 SFTS clusters involving 51 laboratory-confirmed patients, which occurred in Yantai city from 2013 to 2022. Contact with patients’ blood or other bodily fluids, along with the absence of PPE, escalates the human-to-human transmission of SFTSV. Thus, health education and behavioral interventions for the relatives and caregivers of SFTS patients, both in hospitals and post-discharge, are crucial.
The original contributions presented in the study are included in the article/Supplementary material. Further inquiries can be directed to the corresponding author.
The studies involving humans were approved by Yantai Center for Disease Control and Prevention. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
NZ: Formal analysis, Investigation, Methodology, Software, Visualization, Writing – original draft. XM: Investigation, Methodology, Supervision, Visualization, Writing – review & editing. JL: Investigation, Resources, Supervision, Validation, Visualization, Writing – review & editing. TL: Conceptualization, Funding acquisition, Resources, Supervision, Validation, Writing – review & editing.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study was funded by the Yantai Science and Technology Innovation Development Plan Policy Guidance Project (Grant number 2020YD061).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpubh.2024.1419425/full#supplementary-material
1. Yu, XJ, Liang, MF, Zhang, SY, Liu, Y, Li, JD, Sun, YL, et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N Engl J Med. (2011) 364:1523–32. doi: 10.1056/NEJMoa1010095
2. Takahashi, T, Maeda, K, Suzuki, T, Ishido, A, Shigeoka, T, Tominaga, T, et al. The first identification and retrospective study of severe fever with thrombocytopenia syndrome in Japan. J Infect Dis. (2014) 209:816–27. doi: 10.1093/infdis/jit603
3. Tran, XC, Yun, Y, Van An, L, Kim, SH, Thao, NTP, Man, PKC, et al. Endemic severe fever with thrombocytopenia syndrome. Vietnam Emerg Infect Dis. (2019) 25:1029–31. doi: 10.3201/eid2505.181463
4. McMullan, LK, Folk, SM, Kelly, AJ, MacNeil, A, Goldsmith, CS, Metcalfe, MG, et al. A new phlebovirus associated with severe febrile illness in Missouri. N Engl J Med. (2012) 367:834–41. doi: 10.1056/NEJMoa1203378
5. Organization, W.H . (2018). Annual review of diseases prioritized under the Research and Development Blueprint 2018. Available from: https://www.who.int/news-room/events/detail/2018/02/06/default-calendar/2018-annual-review-of-diseases-prioritized-under-the-research-anddevelopment-blueprint (Accessed January 12, 2024).
6. Casel, MA, Park, SJ, and Choi, YK. Severe fever with thrombocytopenia syndrome virus: emerging novel phlebovirus and their control strategy. Exp Mol Med. (2021) 53:713–22. doi: 10.1038/s12276-021-00610-1
7. Luo, N, Li, M, Xu, M, Shi, C, Shi, X, Ni, R, et al. Research Progress of fever with thrombocytopenia syndrome. Intensive Care Res. (2023) 3:172–81. doi: 10.1007/s44231-023-00035-6
8. Liu, L, Guan, XH, Xing, XS, Shen, XF, Xu, JQ, Yue, JL, et al. Epidemiologic analysis on severe fever with thrombocytopenia syndrome in Hubei province, 2010. Zhonghua Liu Xing Bing Xue Za Zhi. (2012) 33:168–72. doi: 10.3760/cma.j.issn.0254-6450.2012.02.009
9. Zhang, YZ, Zhou, DJ, Qin, XC, Tian, JH, Xiong, Y, Wang, JB, et al. The ecology, genetic diversity, and phylogeny of Huaiyangshan virus in China. J Virol. (2012) 86:2864–8. doi: 10.1128/jvi.06192-11
10. Ding, F, Guan, XH, Kang, K, Ding, SJ, Huang, LY, Xing, XS, et al. Risk factors for bunyavirus-associated severe fever with thrombocytopenia syndrome, China. PLoS Negl Trop Dis. (2014) 8:e3267. doi: 10.1371/journal.pntd.0003267
11. Wang, S, Li, J, Niu, G, Wang, X, Ding, S, Jiang, X, et al. SFTS virus in ticks in an endemic area of China. Am J Trop Med Hyg. (2015) 92:684–9. doi: 10.4269/ajtmh.14-0008
12. Luo, LM, Zhao, L, Wen, HL, Zhang, ZT, Liu, JW, Fang, LZ, et al. Haemaphysalis longicornis ticks as reservoir and vector of severe fever with thrombocytopenia syndrome virus in China. Emerg Infect Dis. (2015) 21:1770–6. doi: 10.3201/eid2110.150126
13. Niu, G, Li, J, Liang, M, Jiang, X, Jiang, M, Yin, H, et al. Severe fever with thrombocytopenia syndrome virus among domesticated animals. China Emerg Infect Dis. (2013) 19:756–63. doi: 10.3201/eid1905.120245
14. Park, ES, Shimojima, M, Nagata, N, Ami, Y, Yoshikawa, T, Iwata-Yoshikawa, N, et al. Severe fever with thrombocytopenia syndrome Phlebovirus causes lethal viral hemorrhagic fever in cats. Sci Rep. (2019) 9:11990. doi: 10.1038/s41598-019-48317-8
15. Sun, Y, Zhang, D, Liu, H, Ruan, C, Dou, X, Ren, Z, et al. The first reported cases of severe fever with thrombocytopenia syndrome virus from domestic sick camel to humans in China. Emerg Microbes Infect. (2024) 13:2309990. doi: 10.1080/22221751.2024.2309990
16. Nam, SJ, Oh, YI, Kim, HJ, Cheon, DS, Noh, SJ, and Hong, YJ. Unusual case of severe fever with thrombocytopenia syndrome showing clinical manifestations in a companion dog. Vet Med Sci. (2020) 6:353–8. doi: 10.1002/vms3.261
17. Yamanaka, A, Kirino, Y, Fujimoto, S, Ueda, N, Himeji, D, Miura, M, et al. Direct transmission of severe fever with thrombocytopenia syndrome virus from domestic cat to veterinary personnel. Emerg Infect Dis. (2020) 26:2994–8. doi: 10.3201/eid2612.191513
18. Ye, C, and Qi, R. Risk factors for person-to-person transmission of severe fever with thrombocytopenia syndrome. Infect Control Hosp Epidemiol. (2021) 42:582–5. doi: 10.1017/ice.2020.1258
19. Tang, X, Wu, W, Wang, H, Du, Y, Liu, L, Kang, K, et al. Human-to-human transmission of severe fever with thrombocytopenia syndrome bunyavirus through contact with infectious blood. J Infect Dis. (2013) 207:736–9. doi: 10.1093/infdis/jis748
20. Moon, J, Lee, H, Jeon, JH, Kwon, Y, Kim, H, Wang, EB, et al. Aerosol transmission of severe fever with thrombocytopenia syndrome virus during resuscitation. Infect Control Hosp Epidemiol. (2019) 40:238–41. doi: 10.1017/ice.2018.330
21. Estrada-Peña, A, and de la Fuente, J. The ecology of ticks and epidemiology of tick-borne viral diseases. Antivir Res. (2014) 108:104–28. doi: 10.1016/j.antiviral.2014.05.016
22. Zintl, A, Moutailler, S, Stuart, P, Paredis, L, Dutraive, J, Gonzalez, E, et al. Ticks and tick-borne diseases in Ireland. Ir Vet J. (2017) 70:4. doi: 10.1186/s13620-017-0084-y
23. Oshiro, LM, da Silva Rodrigues, V, Garcia, MV, de Oliveira Souza, L, Higa, A, Suzin, JCB, et al. Effect of low temperature and relative humidity on reproduction and survival of the tick Rhipicephalus microplus. Exp Appl Acarol. (2021) 83:95–106. doi: 10.1007/s10493-020-00576-1
24. Barbour, AG, and Fish, D. The biological and social phenomenon of Lyme disease. Science. (1993) 260:1610–6. doi: 10.1126/science.8503006
25. Deng, B, Rui, J, Liang, SY, Li, ZF, Li, K, Lin, S, et al. Meteorological factors and tick density affect the dynamics of SFTS in Jiangsu province, China. PLoS Negl Trop Dis. (2022) 16:e0010432. doi: 10.1371/journal.pntd.0010432
26. Fang, X, Hu, J, Peng, Z, Dai, Q, Liu, W, Liang, S, et al. Epidemiological and clinical characteristics of severe fever with thrombocytopenia syndrome bunyavirus human-to-human transmission. PLoS Negl Trop Dis. (2021) 15:e0009037. doi: 10.1371/journal.pntd.0009037
27. Tao, M, Liu, Y, Ling, F, Zhang, R, Shi, X, Ren, J, et al. Characteristics of three person-to-person transmission clusters of severe fever with thrombocytopenia syndrome in southeastern China. Am J Trop Med Hyg. (2021) 105:794–800. doi: 10.4269/ajtmh.21-0366
28. Zhang, S, Wang, J, Zhang, Q, Pan, Y, Zhang, Z, Geng, Y, et al. Association of liver function and prognosis in patients with severe fever with thrombocytopenia syndrome. PLoS Negl Trop Dis. (2024) 18:e0012068. doi: 10.1371/journal.pntd.0012068
29. Wang, Y, Song, Z, Wei, X, Yuan, H, Xu, X, Liang, H, et al. Clinical laboratory parameters and fatality of severe fever with thrombocytopenia syndrome patients: a systematic review and meta-analysis. PLoS Negl Trop Dis. (2022) 16:e0010489. doi: 10.1371/journal.pntd.0010489
30. Ding, YP, Liang, MF, Ye, JB, Liu, QH, Xiong, CH, Long, B, et al. Prognostic value of clinical and immunological markers in acute phase of SFTS virus infection. Clin Microbiol Infect. (2014) 20:O870–8. doi: 10.1111/1469-0691.12636
31. Xu, X, Sun, Z, Liu, J, Zhang, J, Liu, T, Mu, X, et al. Analysis of clinical features and early warning indicators of death from severe fever with thrombocytopenia syndrome. Int J Infect Dis. (2018) 73:43–8. doi: 10.1016/j.ijid.2018.05.013
32. Jeong, EJ, Song, JY, Lim, CS, Lee, I, Park, MS, Choi, MJ, et al. Viral shedding from diverse body fluids in a patient with severe fever with thrombocytopenia syndrome. J Clin Virol. (2016) 80:33–5. doi: 10.1016/j.jcv.2016.04.018
33. Akagi, K, Miyazaki, T, Oshima, K, Umemura, A, Shimada, S, Morita, K, et al. Detection of viral RNA in diverse body fluids in an SFTS patient with encephalopathy, gastrointestinal bleeding and pneumonia: a case report and literature review. BMC Infect Dis. (2020) 20:281. doi: 10.1186/s12879-020-05012-8
34. Zhang, YZ, He, YW, Dai, YA, Xiong, Y, Zheng, H, Zhou, DJ, et al. Hemorrhagic fever caused by a novel Bunyavirus in China: pathogenesis and correlates of fatal outcome. Clin Infect Dis. (2012) 54:527–33. doi: 10.1093/cid/cir804
35. Chen, H, Hu, K, Zou, J, and Xiao, J. A cluster of cases of human-to-human transmission caused by severe fever with thrombocytopenia syndrome bunyavirus. Int J Infect Dis. (2013) 17:e206–8. doi: 10.1016/j.ijid.2012.11.006
36. Huang, D, Jiang, Y, Liu, X, Wang, B, Shi, J, Su, Z, et al. A cluster of symptomatic and asymptomatic infections of severe fever with thrombocytopenia syndrome caused by person-to-person transmission. Am J Trop Med Hyg. (2017) 97:396–402. doi: 10.4269/ajtmh.17-0059
37. Jiang, XL, Zhang, S, Jiang, M, Bi, ZQ, Liang, MF, Ding, SJ, et al. A cluster of person-to-person transmission cases caused by SFTS virus in Penglai. China Clin Microbiol Infect. (2015) 21:274–9. doi: 10.1016/j.cmi.2014.10.006
38. Yang, T, Huang, H, Jiang, L, and Li, J. Overview of the immunological mechanism underlying severe fever with thrombocytopenia syndrome (review). Int J Mol Med. (2022) 50:118. doi: 10.3892/ijmm.2022.5174
39. Wu, YX, Yang, X, Leng, Y, Li, JC, Yuan, L, Wang, Z, et al. Human-to-human transmission of severe fever with thrombocytopenia syndrome virus through potential ocular exposure to infectious blood. Int J Infect Dis. (2022) 123:80–3. doi: 10.1016/j.ijid.2022.08.008
Keywords: severe fever with thrombocytopenia syndrome, SFTSV, human-to-human transmission, risk, cluster
Citation: Zhang N, Mu X, Liu J and Liu T (2024) Risk assessment of human-to-human transmission of severe fever with thrombocytopenia syndrome virus based on 10-year clustered analysis. Front. Public Health. 12:1419425. doi: 10.3389/fpubh.2024.1419425
Received: 18 April 2024; Accepted: 10 September 2024;
Published: 11 October 2024.
Edited by:
Nam-Hyuk Cho, Seoul National University, Republic of KoreaReviewed by:
Keun Hwa Lee, Hanyang University, Republic of KoreaCopyright © 2024 Zhang, Mu, Liu and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Tao Liu, bHRfMzQwMkAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.