
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
MINI REVIEW article
Front. Public Health , 27 October 2023
Sec. Injury Prevention and Control
Volume 11 - 2023 | https://doi.org/10.3389/fpubh.2023.1279118
This article is part of the Research Topic Novel Targets for Chronic Inflammatory Diseases: Focus On Therapeutic Drugs and Natural Compounds, volume II View all 13 articles
This review delves into the intersection of two prevalent conditions, hyperlipidemia and rotator cuff injuries, both of which bear substantial healthcare burdens. Our investigation begins with an exploration of rotator cuff injuries, common musculoskeletal disorders that severely impair shoulder functionality and quality of life. These injuries are notably pervasive among sports enthusiasts and the older adult, with an incidence rate estimated at 5–10% in the general population. Despite their widespread occurrence and the diverse, multifactorial etiological factors, effective treatment strategies remain elusive. We then examine hyperlipidemia, a metabolic disorder affecting approximately 40% of the global adult population. Characterized by elevated levels of cholesterol and triglycerides, hyperlipidemia can precipitate severe cardiovascular complications and presents a significant socioeconomic burden. Although current management strategies encompass lifestyle modifications and pharmacological interventions, the condition remains a formidable health challenge. Central to this review is the exploration of a potential association between hyperlipidemia and rotator cuff injuries. We aim to synthesize the current understanding of hyperlipidemia’s role in the pathophysiology of rotator cuff injuries, thereby offering fresh insights into their common etiological underpinnings, potential therapeutic targets, and drugs, such as Statins. The influence of other lipid-lowering therapeutics on tendon health is also considered, and further research into the molecular pathways and potential therapeutic benefits of these drugs is required. This pursuit aligns with broader efforts to enhance patient outcomes, minimize healthcare burdens, and contribute to the global understanding of these prevalent conditions.
The rotator cuff, a sophisticated orchestration of muscles and tendons that envelope the shoulder joint, serves an indispensable role in bestowing stability and enabling a diverse array of shoulder movements (1–3). Afflictions to this vital assembly, which span the spectrum from benign inflammation or tendonitis to partial or even full-throttle tendon ruptures, predominantly impact the shoulder’s soft tissues (4, 5). These injuries frequently culminate in acute pain, muscular weakness, and restricted mobility, thereby epitomizing a pervasive musculoskeletal disorder (6). Rotator cuff injuries manifest with particular frequency among sports aficionados and the geriatric population, often inducing substantial debilitation in shoulder functionality and a significant deterioration in the quality of life (3). The annual incidence rate of rotator cuff injuries hovers around 5–10% within the general populace, with a conspicuous surge in prevalence commensurate with advancing age (asymptomatic tears were found in more than 20% of individuals over the age of 60) (7). A rough estimate of the economic burden for a single patient undergoing surgery for a rotator cuff injury can be in the range of $10,000–$30,000 or even more, including both direct and indirect costs. Thus, this condition represents a substantial quota of visits to orthopedic clinics, exerting a hefty strain on healthcare resources, and thereby emerging as a noteworthy public health quandary.
The precise etiology of rotator cuff injuries remains nebulous, likely being multifactorial in nature. The onslaught of years is a well-entrenched risk factor, with degenerative alterations in the rotator cuff tendons becoming increasingly commonplace as individuals age (8). Furthermore, lifestyle determinants such as smoking have been implicated, with empirical evidence indicating that smokers are at an escalated risk of succumbing to rotator cuff injuries in contrast to their non-smoking counterparts (9). Medical conditions such as diabetes and hypertension have also been correlated with an augmented risk of these injuries, thereby suggesting a role for systemic health in the integrity of the rotator cuff (10, 11). The stewardship of rotator cuff injuries is multifarious, with therapeutic options customized to the individual patient’s needs, factoring in the severity of the injury, the patient’s overarching health status, and their functional exigencies. Conservative management approaches, encompassing physical therapy, pain mitigation with non-steroidal anti-inflammatory drugs (NSAIDs), and corticosteroid injections, typically constitute the initial line of treatment (12). However, in cases of severe injuries or when conservative management proves ineffective, surgical intervention may be necessitated (8). Despite these treatment modalities, a significant number of patients persist in experiencing pain and functional limitations, thereby highlighting the need for further research into innovative therapeutic strategies (11, 13).
Hyperlipidemia, a pervasive metabolic aberration, is characterized by escalated levels of total cholesterol (TC), triglycerides (TG), and low-density lipoprotein cholesterol (LDL-C), coupled with a reduction in high-density lipoprotein cholesterol (HDL-C) (1, 14, 15). This condition often manifests covertly, with no symptoms in its incipient stages. However, unaddressed hyperlipidemia can precipitate atherosclerosis, engendering grave cardiovascular ramifications such as heart disease, cerebrovascular accident, and peripheral artery disease (10, 16, 17). As a substantial global health quandary, the World Health Organization posits that approximately 40% of the global adult populace grapples with elevated total cholesterol levels, a key indicator of hyperlipidemia. This widespread disorder imposes a significant socioeconomic encumbrance, accounting for a considerable proportion of healthcare expenditure. Therapeutic stratagems encompass lifestyle modifications, including dietary adjustments and consistent physical exertion, along with pharmacological interventions like statins, fibrates, and niacin (16, 18, 19). Despite these measures, hyperlipidemia persists as a formidable health challenge, underscoring the exigency for continued exploration into innovative therapeutic approaches and prophylactic strategies. The potential correlation between hyperlipidemia and rotator cuff injuries further accentuates the necessity for a comprehensive understanding and efficacious management of this disorder (16).
Given the substantial prevalence and socio-economic burden of hyperlipidemia (20, 21), and its potential association with rotator cuff injuries, a comprehensive understanding of the interplay between these two conditions is essential. Therefore, this review aims to amalgamate the current understanding of the role of hyperlipidemia in the pathophysiology of rotator cuff injuries. Our objective is to elucidate this connection, thereby providing novel insights for etiological research and clinical management. By probing into the underlying mechanisms, we expect to shed light on potential therapeutic targets and preventive strategies, thereby contributing to the global efforts in addressing these prevalent conditions.
The etiology of rotator cuff injuries is a complex interplay of multiple factors, inclusive of age-related degenerative changes, disease progression, and undue physical strain. In the scientific discourse of recent years, a considerable emphasis has been placed on the potential correlation between hyperlipidemia and rotator cuff injuries. This connection was evidenced in a prospectively designed study by Skovgaard et al. (16), wherein hypercholesterolemia surfaced as a factor escalating the risk of upper limb tendon injuries by a factor of 1.5. A similar correlation was delineated in Kara’s research (10), where cardiovascular risk elements, including diabetes and hyperlipidemia, were identified as potential precipitators of rotator cuff injuries.
In a parallel vein, Djerbi et al. (22) uncovered a significant correlation between smoking, dyslipidemia, and the incidence rate of rotator cuff tears. An 11-year longitudinal follow-up study conducted by Lin et al. (15) further underscored hyperlipidemia as an independent risk factor for rotator cuff disorders, doubling the risk compared to non-hyperlipidemic cohorts. Recent studies echoed these findings, demonstrating a discernible link between hyperlipidemia and various manifestations of rotator cuff tendon diseases, with hyperlipidemic patients bearing a higher risk of complete tears.
However, it is imperative to note the existence of contrarian views, as several studies (23, 24) reported no significant correlation between dyslipidemia and rotator cuff tears. These incongruities might be attributed to variances in the demographic composition of the study populations, the parameters observed, and the criteria for grouping.
Hyperlipidemia, characterized by elevated lipid levels, has been increasingly identified as a critical factor that correlates not only with the onset of rotator cuff tears but also with the propensity for a re-tear following surgical intervention. This multifaceted influence of hyperlipidemia was substantiated in a large-scale, rigorous study conducted by Cancienne et al. (25). Their research, encompassing an extensive cohort of 30,638 patients subjected to rotator cuff repair, unveiled a significant correlation between perioperative total cholesterol and low-density lipoprotein cholesterol levels, and the rate of revision surgery following the initial repair. This finding underscores the potential for hyperlipidemia to complicate the post-operative trajectory and recovery in patients with rotator cuff tears.
Further amplifying this perspective, Garcia’s research (14) revealed a distinct disparity in the risk of re-tear between hyperlipidemic and non-hyperlipidemic patients. The study found that, compared to their non-hyperlipidemic counterparts, patients with hyperlipidemia faced a staggering fourfold increase in the risk of re-tear following rotator cuff repair. This finding is indicative of the profound influence of lipid metabolic disorders on the integrity and healing capacity of rotator cuff tissues.
Complementing these findings, Kim’s study (26) further reinforced the role of hyperlipidemia as a significant risk factor for re-tear subsequent to rotator cuff repair. The convergence of these findings across multiple studies underscores the necessity of considering hyperlipidemia in the strategic planning of both preventive and therapeutic interventions for rotator cuff injuries.
Taking this multifactorial approach a step further, Harada et al. (27) embarked on an investigation of the risk factors for re-tear following rotator cuff repair. The study’s findings highlighted that the combination of hyperlipidemia, a tear size of ≥40 mm, and a critical shoulder angle of ≥37° collectively forecasted the highest rate of re-tear. This discovery underscores the importance of a comprehensive, multi-parametric assessment in predicting the risk of re-tear and devising more effective post-operative management strategies.
The intricate role of hyperlipidemia in the pathogenesis of rotator cuff injury remains somewhat nebulous, but current research illuminates a series of molecular mechanisms that may underpin the deleterious effects of this metabolic disorder on tendon health (Figure 1).
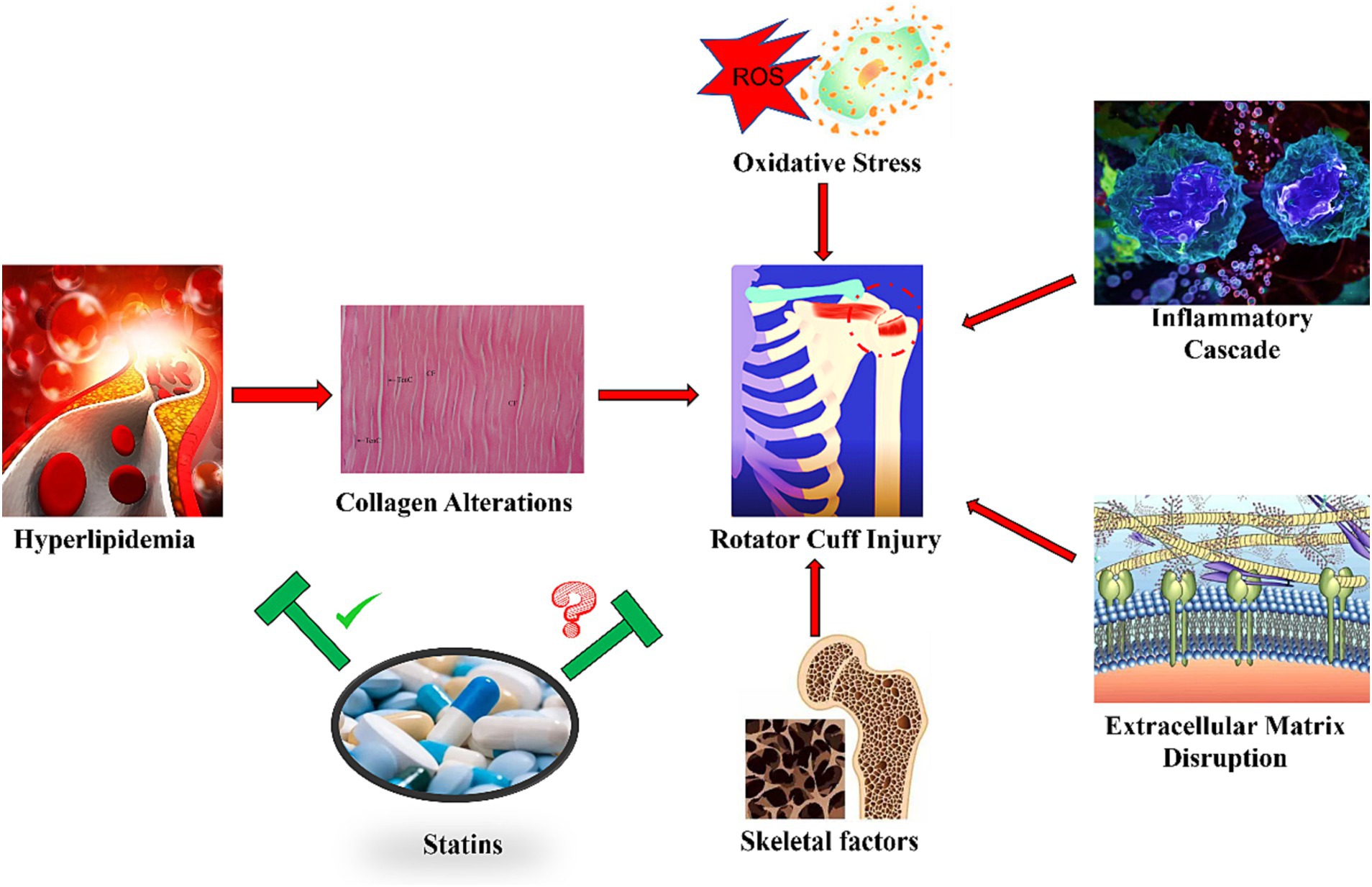
Figure 1. Mechanism of hyperlipidemia and statins on rotator cuff injury. The figure reflects several messages: 1. Hyperlipidemia may lead to a range of pathologic changes leading to a higher incidence of rotator cuff tears. 2. Collagen transformation, oxidative stress, inflammation, extracellular matrix disorders, and changes in the skeletal system underlie the co-morbidities of hyperlipidemia and rotator cuff injuries. 3. Statins have been clearly shown to intervene in hyperlipidemia, but the effect on rotator cuff injuries is controversial, and co-morbidities are not well understood. 4. The efficacy of statins in rotator cuff injury is controversial, and more research is needed to support the efficacy of statins in this co-morbid condition.
A multitude of studies affirm the primacy of oxidative stress in triggering tendon degeneration, fibrosis, and adhesion (28, 29). Hyperlipidemia, particularly characterized by high cholesterol, can suppress the expression of tendon-related genes in tendon stem/progenitor cells via the reactive oxygen species (ROS)-activated nuclear factor kappa-B (NF-κB) signaling pathway (30, 31). Concurrently, it can instigate cell apoptosis and autophagy through the ROS-activated AKT/FOXO1 signaling pathway in these cells, culminating in dysfunction and ultimately precipitating tendon degeneration and tendinopathy (32, 33).
Normal tendons are predominantly constituted of type I collagen, interspersed with a minor proportion of type III collagen. Aberrant cholesterol deposition in tendon tissues can provoke alterations in collagen synthesis and organization, thereby inducing structural and biomechanical alterations within the tendon matrix (34).
The extracellular matrix (ECM) plays a pivotal role in maintaining tendon homeostasis (35). A hyperlipidemic milieu can induce changes in critical components of the ECM in tendon tissues and cells. The mechanism may involve low-density lipoprotein cholesterol (LDL-C) inducing dysfunction of tendon cells, contributing to alterations in the ECM components (36).
The inflammatory microenvironment is a key player in tendon degenerative damage (37–39), and hyperlipidemia has been closely linked to systemic inflammation (40). Cholesterol molecules can activate the NF-κB signaling pathway, augmenting the expression of pro-inflammatory cytokines such as tumor necrosis factor α (TNF-α), interleukin 6 (IL-6) (41–43).
Hyperlipidemia can exert a dual detrimental impact on bone health by inhibiting bone formation and accelerating bone resorption, thereby promoting the onset of osteoporosis (44). Notably, osteoporosis has been identified as an independent risk factor for both the genesis of rotator cuff tears and re-tear following surgical repair (45, 46). This underscores the intersection of metabolic, inflammatory, and skeletal factors in the pathogenesis and prognosis of rotator cuff injuries.
Statins, scientifically known as 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors, represent the principal lipid-lowering agents in clinical practice (47). By inhibiting HMG-CoA reductase, the rate-limiting enzyme in cholesterol synthesis, statins effectively curtail cholesterol production. This, in turn, upregulates the expression of LDL receptors on the cell surface, thereby accelerating the metabolic clearance of serum LDL. However, the potential influence of statins on rotator cuff injuries and their recovery trajectory engenders a degree of controversy in the current literature.
Several studies have elucidated a beneficial role for statins in the context of rotator cuff injuries and their post-injury repair. Dolkart et al. (48) employed a rat model of rotator cuff tear to probe the impact of atorvastatin on tendon-bone healing following rotator cuff repair surgery. The experimental findings suggested that atorvastatin could activate the COX2/PGE2/EP4 pathways, thereby fostering tendon cell proliferation, migration, and differentiation, and ultimately bolstering the biomechanical integrity of the tendon-bone interface. Parallel findings were obtained in an animal study conducted by Hao et al. (49), demonstrating that silk protein fortified with simvastatin could stimulate osteogenic differentiation of bone marrow mesenchymal stem cells and collagen synthesis via the β-catenin signaling pathway, thereby enhancing tendon-bone interface healing. Lin et al. (15) conducted an 11-year longitudinal study corroborating that hyperlipidemia constitutes a risk factor for rotator cuff injuries, and statin therapy in hyperlipidemic patients could mitigate this risk. A clinical study by Cancienne et al. (25) revealed a positive correlation between perioperative lipid levels and the rate of revision surgery following rotator cuff repair, suggesting that statin therapy could potentially decrease this revision rate. Similarly, a recent case–control study by Lee et al. (50) incorporated 104 hyperlipidemic patients who underwent arthroscopic rotator cuff repair, among whom 66 individuals were in the statin group and 38 in the non-statin group. The follow-up findings indicated a lower re-tear rate in the statin group.
Conversely, several studies have reported potentially detrimental effects of statins on tendon health. Animal experiments conducted by Oliveira et al. (51, 52) demonstrated that statins could disrupt collagen fiber organization and alter tendon matrix composition, thereby diminishing the biomechanical resilience of the tendon and increasing the risk of rupture. Kaleağasıoğlu et al. (53) discerned that statins could precipitate tendon calcification, which could in turn compromise its biomechanical properties. Animal experiments and clinical cohort studies by Eliasson et al. (54, 55) showed that statins could suppress tendon cell proliferation and elicit deleterious effects on tendon collagen and matrix. Furthermore, some studies found that statins did not improve the prognosis following rotator cuff repair surgery. In a study conducted by Amit et al. (1), 77 hyperlipidemic patients underwent arthroscopic rotator cuff repair, among whom 38 individuals were on statins and 39 were not. The follow-up found no significant differences in shoulder joint function, postoperative fatty infiltration, and re-tear rates between the two groups. Similarly, a study by Zeng et al. (56) found no significant difference in postoperative shoulder function scores between hyperlipidemic patients undergoing arthroscopic rotator cuff repair who were or were not on statin therapy perioperatively.
Potential reasons for the varied outcomes observed across different studies may be due to different baseline population data, different administered doses, and different treatment times, etc. Finally, it should be mentioned that there are still some limitations in the literature cited in this current review. (1) Most of the clinical studies have a low grade of evidence. (2) Animal studies are not sufficiently mechanistically insightful. (3) There is no direct evidence for the effect of statins on co-morbidities, and more clinical and basic experimental evidence is needed to support it.
The significance of hyperlipidemia in the context of rotator cuff injuries is progressively gaining momentum in medical research. The intricate interplay between hyperlipidemia and these injuries remains somewhat enigmatic, necessitating large-scale, multicentric prospective studies to conclusively ascertain whether hyperlipidemia augments the susceptibility to such injuries. The molecular pathways through which hyperlipidemia instigates rotator cuff injuries are yet to be fully elucidated, thereby requiring a more comprehensive exploration through animal models and cellular experiments.
Moreover, the influence of statins on rotator cuff injuries and the subsequent healing of the tendon-bone interface post-surgery continues to be a subject of debate (57). The impact of other widely used lipid-lowering therapeutics, such as ezetimibe and probucol, as well as novel lipid-lowering agents, on tendon health remains nebulous and warrants a more thorough investigation.
The unraveling of the mechanistic role of hyperlipidemia in rotator cuff diseases, coupled with a deeper understanding of the potential therapeutic benefits of lipid-lowering drugs, could potentially offer transformative insights into the etiological basis and clinical management of rotator cuff injuries. Such insights could pave the way for more personalized and effective therapeutic regimens, enhancing the prognosis for patients with these injuries.
YQ: Investigation, Methodology, Project administration, Resources, Writing – original draft, Writing – review & editing. HH: Conceptualization, Data curation, Investigation, Writing – original draft, Writing – review & editing. RW: Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing. YZ: Conceptualization, Data curation, Formal analysis, Writing – original draft. XF: Data curation, Formal analysis, Writing – original draft. FX: Funding acquisition, Supervision, Validation, Writing – review & editing. ZL: Conceptualization, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. QW: Conceptualization, Funding acquisition, Resources, Supervision, Validation, Visualization, Writing – original draft.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported Suzhou Clinical Key Disease Diagnosis and Treatment Technology Special Project (LCZX202127), Kunshan High-level Medical Talent Program Project (Kunshan Health [2019] No. 6), Kunshan Chinese Medicine Science and Technology Development Special Project (KZYY2202), and Kunshan Hospital of Traditional Chinese Medicine Golden Apricot Superior Talent Project (03rczc25).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Amit, P, Kuiper, JH, James, S, and Snow, M. Does statin-treated hyperlipidemia affect rotator cuff healing or muscle fatty infiltration after rotator cuff repair? J Shoulder Elb Surg. (2021) 30:2465–74. doi: 10.1016/j.jse.2021.05.014
2. Gavriilidis, I, Kircher, J, Magosch, P, Lichtenberg, S, and Habermeyer, P. Pectoralis major transfer for the treatment of irreparable anterosuperior rotator cuff tears. Int Orthop. (2010) 34:689–94. doi: 10.1007/s00264-009-0799-9
3. Luo, Z, Lin, J, Sun, Y, Zhu, K, Wang, C, and Chen, J. Outcome comparison of latissimus Dorsi transfer and pectoralis major transfer for irreparable subscapularis tendon tear: a systematic review. Am J Sports Med. (2022) 50:2032–41. doi: 10.1177/03635465211018216
4. Sanchez-Sotelo, J, and Athwal, GS. How to optimize reverse shoulder arthroplasty for irreparable cuff tears. Curr Rev Musculoskelet Med. (2020) 13:553–60. doi: 10.1007/s12178-020-09655-7
5. Wang, C, Song, W, Chen, B, Liu, X, and He, Y. Exosomes isolated from adipose-derived stem cells: a new cell-free approach to prevent the muscle degeneration associated with torn rotator cuffs. Am J Sports Med. (2019) 47:3247–55. doi: 10.1177/0363546519876323
6. Sun, Y, Lin, J, Luo, Z, and Chen, J. Preoperative lymphocyte to monocyte ratio can be a prognostic factor in arthroscopic repair of small to large rotator cuff tears. Am J Sports Med. (2020) 48:3042–50. doi: 10.1177/0363546520953427
7. Sun, Y, Lin, J, Luo, Z, Zhang, Y, and Chen, J. The serum from patients with secondary frozen shoulder following rotator cuff repair induces shoulder capsule fibrosis and promotes macrophage polarization and fibroblast activation. J Inflamm Res. (2021) 14:1055–68. doi: 10.2147/JIR.S304555
8. Sun, Y, Luo, Z, Chen, Y, Lin, J, Zhang, Y, Qi, B, et al. Si-Tgfbr1-loading liposomes inhibit shoulder capsule fibrosis via mimicking the protective function of exosomes from patients with adhesive capsulitis. Biomater Res. (2022) 26:39. doi: 10.1186/s40824-022-00286-2
9. Pietrzak, M. Adhesive capsulitis: an age related symptom of metabolic syndrome and chronic low-grade inflammation? Med Hypotheses. (2016) 88:12–7. doi: 10.1016/j.mehy.2016.01.002
10. Applegate, KA, Thiese, MS, Merryweather, AS, Kapellusch, J, Drury, DL, Wood, E, et al. Association between cardiovascular disease risk factors and rotator cuff tendinopathy: a cross-sectional study. J Occup Environ Med. (2017) 59:154–60. doi: 10.1097/JOM.0000000000000929
11. Giri, A, O’Hanlon, D, and Jain, NB. Risk factors for rotator cuff disease: a systematic review and meta-analysis of diabetes, hypertension, and hyperlipidemia. Ann Phys Rehabil Med. (2023) 66:101631. doi: 10.1016/j.rehab.2022.101631
12. Luo, Z, Sun, Y, Qi, B, Lin, J, Chen, Y, Xu, Y, et al. Human bone marrow mesenchymal stem cell-derived extracellular vesicles inhibit shoulder stiffness via let-7a/Tgfbr1 axis. Bioact Mater. (2022) 17:344–59. doi: 10.1016/j.bioactmat.2022.01.016
13. Leong, HT, Fu, SC, He, X, Oh, JH, Yamamoto, N, and Hang, S. Risk factors for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. (2019) 51:627–37. doi: 10.2340/16501977-2598
14. Garcia, GH, Liu, JN, Wong, A, Cordasco, F, Dines, DM, Dines, JS, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elb Surg. (2017) 26:2086–90. doi: 10.1016/j.jse.2017.05.009
15. Lin, TT-L, Lin, C-H, Chang, C-L, Chi, C-H, Chang, S-T, and Sheu, WH-H. The effect of diabetes, hyperlipidemia, and statins on the development of rotator cuff disease: a nationwide, 11-year, longitudinal, population-based follow-up study. Am J Sports Med. (2015) 43:2126–32. doi: 10.1177/0363546515588173
16. Skovgaard, D, Siersma, VD, Klausen, SB, Visnes, H, Haukenes, I, Bang, CW, et al. Chronic hyperglycemia, hypercholesterolemia, and metabolic syndrome are associated with risk of tendon injury. Scand J Med Sci Sports. (2021) 31:1822–31. doi: 10.1111/sms.13984
17. Yao, Y. S., Li, T.Di, and Zeng, Z. H. (2020). Mechanisms underlying direct actions of hyperlipidemia on myocardium: an updated review. Lipids Health Dis 19,:23. doi: 10.1186/s12944-019-1171-8
19. Miao, H, Chen, H, Pei, S, Bai, X, Vaziri, ND, and Zhao, Y-Y. Plasma lipidomics reveal profound perturbation of glycerophospholipids, fatty acids, and sphingolipids in diet-induced hyperlipidemia. Chem Biol Interact. (2015) 228:79–87. doi: 10.1016/j.cbi.2015.01.023
20. Lan, Z, Tan, L, Wang, G, Tang, M, Wang, R, and Wang, S. Active compounds in RenShenJian decoction ameliorate insulin resistance in vitro. Tradit Med Res. (2022) 7:32. doi: 10.53388/tmr20221022002
21. Pan, B, Zhang, H, Li, H, Yuan, E, Luo, J, Zhang, T, et al. Advances in Chinese medicine treatment and research on endocrine diseases in 2021. Tradit Med Res. (2022) 7:49. doi: 10.53388/tmr20220208001
22. Djerbi, I, Chammas, M, Mirous, M-P, Lazerges, C, and Coulet, B. Impact of cardiovascular risk factor on the prevalence and severity of symptomatic full-thickness rotator cuff tears. Orthop Traumatol Surg Res. (2015) 101:S269–73. doi: 10.1016/j.otsr.2015.06.011
23. Longo, UG, Franceschi, F, Spiezia, F, Forriol, F, Maffulli, N, and Denaro, V. Triglycerides and total serum cholesterol in rotator cuff tears: do they matter? Br J Sports Med. (2010) 44:948–51. doi: 10.1136/bjsm.2008.056440
24. Yamamoto, N, Mineta, M, Kawakami, J, Sano, H, and Itoi, E. Risk factors for tear progression in symptomatic rotator cuff tears: a prospective study of 174 shoulders. Am J Sports Med. (2017) 45:2524–31. doi: 10.1177/0363546517709780
25. Cancienne, JM, Brockmeier, SF, Rodeo, SA, and Werner, BC. Perioperative serum lipid status and statin use affect the revision surgery rate after arthroscopic rotator cuff repair. Am J Sports Med. (2017) 45:2948–54. doi: 10.1177/0363546517717686
26. Kim, Y-K, Jung, K-H, Kim, J-W, Kim, U-S, and Hwang, D-H. Factors affecting rotator cuff integrity after arthroscopic repair for medium-sized or larger cuff tears: a retrospective cohort study. J Shoulder Elb Surg. (2018) 27:1012–20. doi: 10.1016/j.jse.2017.11.016
27. Harada, N, Gotoh, M, Ishitani, E, Kakuma, T, Yano, Y, Tatara, D, et al. Combination of risk factors affecting retear after arthroscopic rotator cuff repair: a decision tree analysis. J Shoulder Elb Surg. (2021) 30:9–15. doi: 10.1016/j.jse.2020.05.006
28. Miao, Y, Chen, R, Zhang, Z, Liu, Y, Yang, F, and Zhang, J. Network pharmacology analysis of Xuanfei Baidu granule in the treatment of intestinal Flora disorder. Adv Gut Microbiome Res. (2022) 2022:1–13. doi: 10.1155/2022/7883756
29. Singh, T, Kaur, G, and Kaur, A. Dysbiosis—an etiological factor for cardiovascular diseases and the therapeutic benefits of gut microflora. Adv Gut Microbiome Res. (2023) 2023:1–8. doi: 10.1155/2023/7451554
30. Wang, Z-H, Lin, Z, Lai, Y, Ding, L, Wang, H, Chen, X, et al. The immunomodulatory effects of bone marrow-derived mesenchymal stem cells on lymphocyte in spleens of aging rats. Biomed Eng Commun. (2023) 2:13. doi: 10.53388/bmec2023013
31. Wu, Y, Hu, C, and Wang, Y-B. Recent advances in the application of biomimetic nanomedicines in disease treatment. Biomed Eng Commun. (2022) 1:4. doi: 10.53388/bmec2022004
32. Li, K, Deng, G, Deng, Y, Chen, S, Wu, H, Cheng, C, et al. High cholesterol inhibits tendon-related gene expressions in tendon-derived stem cells through reactive oxygen species-activated nuclear factor-κB signaling. J Cell Physiol. (2019) 234:18017–28. doi: 10.1002/jcp.28433
33. Li, K, Deng, Y, Deng, G, Chen, P, Wang, Y, Wu, H, et al. High cholesterol induces apoptosis and autophagy through the ROS-activated AKT/FOXO1 pathway in tendon-derived stem cells. Stem Cell Res Ther. (2020) 11:131. doi: 10.1186/s13287-020-01643-5
34. Steplewski, A, Fertala, J, Tomlinson, R, Hoxha, K, Han, L, Thakar, O, et al. The impact of cholesterol deposits on the fibrillar architecture of the Achilles tendon in a rabbit model of hypercholesterolemia. J Orthop Surg Res. (2019) 14:172. doi: 10.1186/s13018-019-1217-7
35. Figueiredo, EA, Loyola, LC, Belangero, PS, Campos Ribeiro-dos-Santos, ÂK, Emanuel Batista Santos, S, Cohen, C, et al. Rotator cuff tear susceptibility is associated with variants in genes involved in tendon extracellular matrix homeostasis. J Orthop Res. (2020) 38:192–201. doi: 10.1002/jor.24455
36. Fang, W, Sekhon, S, Teramoto, D, Fung, C, la, V, Duong, C, et al. Pathological alterations in the expression status of rotator cuff tendon matrix components in hyperlipidemia. Mol Cell Biochem. (2023) 478:1887–98. doi: 10.1007/s11010-022-04643-6
37. Chen, H, Li, H, Shi, W, Qin, H, and Zheng, L. The roles of m6A RNA methylation modification in cancer stem cells: new opportunities for cancer suppression. Cancer Insight. (2022) 1:1–18. doi: 10.58567/ci01020001
38. Liu, C, Qin, Q, and Cong, H. Research Progress on the relationship between mitochondrial Deoxyguanosine kinase and apoptosis and autophagy in lung adenocarcinoma cells. Cancer Insight. (2022) 1:53–62. doi: 10.58567/ci01010004
39. Shindle, MK, Chen, CCT, Robertson, C, DiTullio, AE, Paulus, MC, Clinton, CM, et al. Full-thickness supraspinatus tears are associated with more synovial inflammation and tissue degeneration than partial-thickness tears. J Shoulder Elb Surg. (2011) 20:917–27. doi: 10.1016/j.jse.2011.02.015
40. Luo, Z-W, Sun, Y-Y, Lin, J-R, Qi, B-J, and Chen, J-W. Exosomes derived from inflammatory myoblasts promote M1 polarization and break the balance of myoblast proliferation/differentiation. World J Stem Cells. (2021) 13:1762–82. doi: 10.4252/wjsc.v13.i11.1762
41. Bhatt, BA, Dube, JJ, Dedousis, N, Reider, JA, and O’Doherty, RM. Diet-induced obesity and acute hyperlipidemia reduce IkappaBalpha levels in rat skeletal muscle in a fiber-type dependent manner. Am J Physiol Regul Integr Comp Physiol. (2006) 290:R233–40. doi: 10.1152/ajpregu.00097.2005
42. Hu, Y, Lin, H, Dib, B, Atik, A, Bouzika, P, Lin, C, et al. Cholesterol crystals induce inflammatory cytokines expression in a human retinal pigment epithelium cell line by activating the NF-κB pathway. Discov Med. (2014) 18:7–14.
43. Luo, Z, Sun, Y-Y, Xia, W, Xu, J-Y, Xie, D-J, Jiao, C-M, et al. Physical exercise reverses immuno-cold tumor microenvironment via inhibiting SQLE in non-small cell lung cancer. Mil Med Res. (2023) 10:39. doi: 10.1186/s40779-023-00474-8
44. Mu, Y, Gao, W, Zhou, Y, Xiao, L, and Xiao, Y. Physiological and pathological/ectopic mineralization: from composition to microstructure. Microstructures. (2023) 3:2023030. doi: 10.20517/microstructures.2023.05
45. Chung, SW, Oh, JH, Gong, HS, Kim, JY, and Kim, SH. Factors affecting rotator cuff healing after arthroscopic repair: osteoporosis as one of the independent risk factors. Am J Sports Med. (2011) 39:2099–107. doi: 10.1177/0363546511415659
46. Hong, J-P, Huang, S-W, Lee, C-H, Chen, H-C, Charoenpong, P, and Lin, H-W. Osteoporosis increases the risk of rotator cuff tears: a population-based cohort study. J Bone Miner Metab. (2022) 40:348–56. doi: 10.1007/s00774-021-01293-4
47. Mao, W, Cai, Y, Chen, D, Jiang, G, Xu, Y, Chen, R, et al. Statin shapes inflamed tumor microenvironment and enhances immune checkpoint blockade in non–small cell lung cancer. JCI Insight. (2022) 7:e161940. doi: 10.1172/jci.insight.161940
48. Dolkart, O, Liron, T, Chechik, O, Somjen, D, Brosh, T, Maman, E, et al. Statins enhance rotator cuff healing by stimulating the COX2/PGE2/EP4 pathway: an in vivo and in vitro study. Am J Sports Med. (2014) 42:2869–76. doi: 10.1177/0363546514545856
49. Hao, L, Chen, J, Shang, X, and Chen, S. Surface modification of the simvastatin factor-loaded silk fibroin promotes the healing of rotator cuff injury through β-catenin signaling. J Biomater Appl. (2021) 36:210–8. doi: 10.1177/0885328221995926
50. Lee, S, Lee, N, and Shin, S-J. Relationship of missed statin therapy and 10-year atherosclerotic cardiovascular disease risk score to Retear rate after arthroscopic rotator cuff repair. Am J Sports Med. (2023) 51:1988–96. doi: 10.1177/03635465231175476
51. de Oliveira, LP, Vieira, CP, Guerra, FD, Almeida, MS, and Pimentel, ER. Structural and biomechanical changes in the Achilles tendon after chronic treatment with statins. Food Chem Toxicol. (2015) 77:50–7. doi: 10.1016/j.fct.2014.12.014
52. Oliveira, LP, Vieira, CP, Marques, PP, and Pimentel, ER. Do different tendons exhibit the same response following chronic exposure to statins? Can J Physiol Pharmacol. (2017) 95:333–9. doi: 10.1139/cjpp-2016-0133
53. Kaleağasıoğlu, F, Olcay, E, and Olgaç, V. Statin-induced calcific Achilles tendinopathy in rats: comparison of biomechanical and histopathological effects of simvastatin, atorvastatin and rosuvastatin. Knee Surg Sports Traumatol Arthrosc. (2017) 25:1884–91. doi: 10.1007/s00167-015-3728-z
54. Eliasson, P, Dietrich-Zagonel, F, Lundin, A-C, Aspenberg, P, Wolk, A, and Michaëlsson, K. Statin treatment increases the clinical risk of tendinopathy through matrix metalloproteinase release - a cohort study design combined with an experimental study. Sci Rep. (2019) 9:17958. doi: 10.1038/s41598-019-53238-7
55. Eliasson, P, Svensson, RB, Giannopoulos, A, Eismark, C, Kjær, M, Schjerling, P, et al. Simvastatin and atorvastatin reduce the mechanical properties of tendon constructs in vitro and introduce catabolic changes in the gene expression pattern. PLoS One. (2017) 12:e0172797. doi: 10.1371/journal.pone.0172797
56. Zeng, GJS, Lee, MJH, Chen, JY, Ang, BFH, Hao, Y, and Lie, DTT. Dyslipidemia with perioperative statin usage is not associated with poorer 24-month functional outcomes after arthroscopic rotator cuff surgery. Am J Sports Med. (2020) 48:2518–24. doi: 10.1177/0363546520937266
57. Cai, J, Xu, J, Ye, Z, Wang, L, Zheng, T, Zhang, T, et al. Exosomes derived from Kartogenin-preconditioned mesenchymal stem cells promote cartilage formation and collagen maturation for enthesis regeneration in a rat model of chronic rotator cuff tear. Am J Sports Med. (2023) 51:1267–76. doi: 10.1177/03635465231155927
Keywords: rotator cuff injury, hyperlipidemia, statin, mechanism, treatment, inflammation
Citation: Qian Y, Huang H, Wan R, Zhou Y, Feng X, Xu F, Luo Z and Wang Q (2023) Progress in studying the impact of hyperlipidemia and statins on rotator cuff injury and repair. Front. Public Health. 11:1279118. doi: 10.3389/fpubh.2023.1279118
Received: 21 August 2023; Accepted: 12 October 2023;
Published: 27 October 2023.
Edited by:
Yan Huang, Anhui Medical University, ChinaReviewed by:
Dan Liu, General Hospital of Ningxia Medical University, ChinaCopyright © 2023 Qian, Huang, Wan, Zhou, Feng, Xu, Luo and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Feng Xu, eGY3MDEyMjhAMTYzLmNvbQ==; Zhiwen Luo, emhpd2VuLmx1b19mdWRhbkBob3RtYWlsLmNvbQ==; Qing Wang, RG9jdG9yd3ExOTgzQDE2My5jb20=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.