
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Public Health , 18 February 2020
Sec. Infectious Diseases: Epidemiology and Prevention
Volume 8 - 2020 | https://doi.org/10.3389/fpubh.2020.00011
Leishmania tropica and Leishmania major are both the main cause of anthroponotic (ACL) and zoonotic cutaneous leishmaniasis (ZCL), respectively, in the Old World. Leishmania infantum and Leishmania donovani, which are important causes of visceral leishmaniasis, have also occasionally been reported in CL patients. The present study investigates the current distribution of causative species of CL in Iran and neighboring countries in the Middle East. There has been expansion of L. tropica into new urban and rural foci in Iran, with well-documented cases of visceralization, a substantial increase of CL in Syria, and the emergence of new foci and outbreaks in Turkey and Iraq, especially due to L. major. Civil war in Syria and Iraq, population movement, poverty, and climatic change play important roles in the changing CL distribution in this region. Control programs should adopt a multidisciplinary approach based on active surveillance and case finding, especially in vulnerable refugee populations, determination of hazard maps for CL hot points using GIS and other advanced technology, the free distribution of drugs, rodent control, and greater community engagement in poor and marginalized populations. Comprehensive molecular studies that could show the species and strains of Leishmania in different areas of each country can give a better view from the distribution of CL in this region.
Leishmaniasis is the complex disease with a spectrum of clinical manifestations, including cutaneous leishmaniasis (CL), mucocutaneous leishmaniasis (MCL), diffuse cutaneous leishmaniasis (DCL), and visceral leishmaniasis (VL, also known as kala-azar). It is transmitted by female nocturnally biting sand flies, Phlebotomus in the “Old World (OW)” and Lutzomia in the “New World (NW).” CL is more prevalent than VL and is distributed in more than 90 countries with the majority of cases occurring in South and Central America, the Mediterranean Basin and extending across the Middle East to Central Asia. The least burdensome regions include South Asia and Sub-Saharan Africa (SSA), but within SSA, East Africa (e.g., Ethiopia, Kenya) is endemic for CL and VL. VL is mostly distributed in South Asia, SSA, and South and Central America. High-burden countries, India, Bangladesh, Sudan, Ethiopia, and Brazil, account for 90% of VL patients while lower rates of VL occur in Southern Europe, Central Asia, China, and the Middle East including Iran (1).
Leishmaniasis is mainly zoonotic, but Leishmania donovani and Leishmania tropica, the causes of OWVL and OWCL, respectively, are transmitted from person to person and are also described as anthroponotic (A) VL and CL (2). The NWCL species such as Leishmania braziliensis, Leishmania guyanensis, Leishmania amazonensis, and Leishmania mexicana are zoonotic species and are endemic in Central and South America; their zoonotic reservoirs include forest rodents and sloths (3). The zoonotic reservoirs for zoonotic OWCL (ZCL) due to Leishmania major and Leishmania aethiopica are different rodent species (4) and hyraxes (2), respectively. Although humans are the main reservoir hosts of L. tropica, latterly it has been found in several animal species such as dogs (5), hyraxes (6), jackals, and foxes (7) and raise the question of a zoonotic cycle in some geographic regions of the Middle East.
The two principal OWCL species overlap in their geographical distribution. L. major extends from West Africa to Central Asia and L. tropica is common in the Eastern Mediterranean, Middle East, North India, Afghanistan, and northeast and South Africa (2). The clinical manifestations of OWCL vary by species. L. aethiopica is the main cause of DCL, which is characterized by chronicity and a poor response to conventional leishmaniasis treatments, like the antimonials but pentamdine appears more promising (8). Although in most cases the cutaneous lesions are self-healing, recovery time may last 2 to 5 years (2). DCL occurs mostly on the face (8) but oronasal mucocutaneous disease has also been documented (9).
In L. major infections, the ulcero-crusted form was the most frequent clinical form and occurs mainly on the lower extremities (10). The ulcers usually necrotize rapidly and multiple wet sores and severe inflammation are common and are slow to heal. Self-healing occurs in more than half of the patients within 2–8 months (2). L. major may also manifest as for mucocutaneous, lupoid, and sporotricoid forms.
L. tropica is common in the Eastern Mediterranean, Middle East, North India, Afghanistan, and northeast and South Africa (11). In patients infected by L. tropica, dry papular or dry ulcerative types are often observed (12) usually on the face and other exposed areas. A superficial spreading form, called leishmaniasis recidivans (LR), which presents as new lesions around an old L. tropica scar, and the plaque form lupoid leishmaniasis are predominantly caused by L. tropica (13, 14). L. tropica responds well to the antimonials and miltefosine (based on small case series) but LR is less responsive (13, 15). Without specific treatment, self-healing is generally longer compared to L. major and may last up to 1 year or more (2). Several cases of kala-azar by L. tropica have been reported from several countries, including India and Iran (16, 17), illustrating its propensity for invasive pathogenicity.
In recent years, there has been a substantial change in the distribution of L. tropica and L. major in Iran and its neighbors, which has challenged the prevention, control, and treatment of CL in this region. Climate change, notably drought in Iran and neighboring countries, the civil wars in Syria and Iraq, and the unstable political situation in Afghanistan have all contributed to the changes in the distribution of the disease in this endemic area of CL. Accordingly, we decided to review in detail the situation of OWCL in our region, which is one of the most important leishmaniasis foci in the world.
Iran, a country of more than 80 million people, lies in the heart of the Middle East, bordered by Azerbaijan, Armenia, Turkmenistan, and the Caspian Sea in the north; Afghanistan and Pakistan in the east; the Persian Gulf and the Oman Sea in the south; Iraq in the west; and Turkey in the northwest. Northern and western Iran are covered by the Zagros and Alborz mountain ranges, respectively, and the rest of the country is covered by forests, plains, and large deserts.
VL and CL are endemic in Iran. VL is endemic in certain regions of the country, notably the northwest and southwest and several small confined regions in central, western, and southeast Iran (18–20). A new endemic focus has been reported recently in the northeast (21). VL is caused mostly by L. infantum and a small number of cases have been due to L. tropica (22).
CL is widely distributed in Iran and has been reported in 25 of 31 Iran provinces in central, west, southwest, south, southeast, east, and northeast regions (23–29).
There are two forms of CL in Iran, ZCL caused by L. major and ACL caused by L. tropica. The main animal reservoirs for L. major infection are desert rodents, including Rhombomys opimus (great gerbil), Tatera indica (Indian gerbil or antelope rat), Meriones libycus (libyan jird), M. persicus (Persian jird), M. hurrianae (Indian desert jird), and Nesokia indica (short-tailed bandicoot) (4, 30, 31).
Therefore, L. major infection is mostly endemic in rural and arid areas and is transmitted mostly by Phlebotomus papatasi; however, urban foci in northeast, central, southeast, and southwest Iran are well documented as well as the reporting of sporadic cases.
By contrast, L. tropica is transmitted between humans and no animal reservoir host has been confirmed in Iran; however, it has been isolated rarely from dogs in this country (32). Phlebotomus sergenti sandfly is the main L. tropica vector in Iran and throughout the Middle East (33), and the ideal breeding conditions include semi-arid (annual precipitation: 226 mm) and more temperate (annual mean temperature 16.4°C) regions (34). ACL is endemic in the larger Iranian cities, including Kerman, Shiraz, Isfahan, Tehran, Mashhad, and some smaller cities such as Bam.
Data suggest overall that OWCL incidence rates have been decreasing in recent years from 37/100,000 in 2007 to 27/100,000 in 2010 and 22/100,000 in 2013 but, simultaneously, it seems that the distribution of L. tropica has expanded to new foci (29, 35).
The epidemiology and geographical distribution of CL in different parts of Iran are presented below.
The highest annual occurrence of CL in Kerman province in south–southeast Iran is reported from well-known ACL foci in Bam and Kerman counties, with 1787 and 369 cases in 2008–2009, respectively, followed by Jiroft (n = 136), Baft (n = 67), Sirjan (n = 41), Shahre-Babak (n = 38), Anbarabad (n = 27), and Roodbare-Jonoob (24). Sharifi et al. (36) conducted a cross-sectional survey in Kerman province, using PCR to identify the Leishmania species causing CL; all samples from Bam, Kerman, Jiroft, Shahre-Babak, Anbarabad, and Roodbare-Jonoob were identified as L. tropica, and only nine (4.4%) cases of L. major were detected in Baft and Sirjan districts. Phylogenetic studies showed a high degree of homogeneity among L. tropica strains in southeast Iran (37). Hormozgan, other southeast Iranian province, has been shown to be a major foci of L. major; e.g., a subsample of 18 of 40 ulcers were all found to be identified as L. major in a study in Jask county (38).
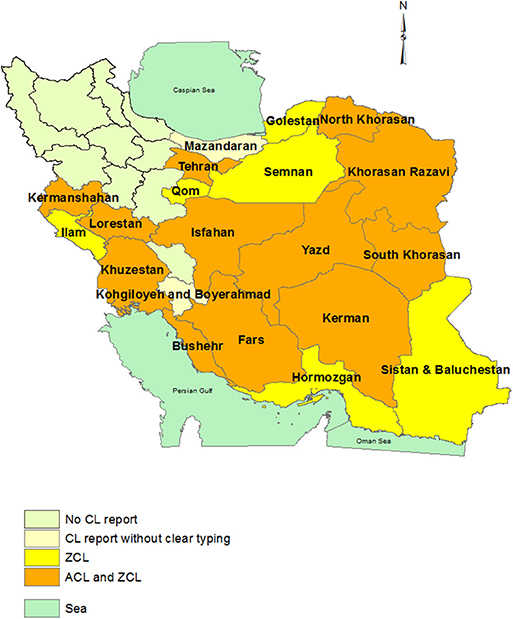
Figure 1. Province-based distribution of ACL and ZCL in Iran. ACL is confined to one or some counties in each province that was not shown here. The maps were generated based on the shape file layers of provinces of Iran and neighboring countries obtained from the DIVA-GIS website (http://www.diva-gis.org/gdata) by using ArcGIS version 10.1 (http://www.esri.com/arcgis).
Shiraz, the capital of Fars province, is an old established city and was the capital of the Zandieh Kingdom from 1751 to 1794. The city has long been recognized as endemic for ACL but no DNA-based molecular data have characterized the species until 2016. In one study, ACL was detected in the old and central parts of the city while L. major was reported in suburbs (27). Bushehr province is a known focus of L. major causing ZCL in southwest Iran (39), but a small study in Bushehr, the provincial capital, identified L. tropica in 6/8 CL patients (40). There are no data on the Leishmania species in the small province of Kohgiloyeh and Boyerahmad but a geospatial information systems (GIS)-based modeling study suggests a ZCL cycle in this part of southwest Iran (41).
Yazd city, the provincial capital of Yazd province, is in close proximity to vast deserts of “Kavir-e-Loot,” and is highly endemic for ZCL and has a low burden of ACL; one study reported an ACL rate of 0.2% in patients aged 6–16 years (42). In addition to Yazd city, Ardakan and Bafgh counties are also predominantly ZCL (24, 43). Yazd province was predicted as a potential hot spot for CL by spatial modeling (44).
Isfahan city in Isfahan province is another Old Iranian city and was the capital from 1598 to 1722 during the Safavieh kingdom. The “Zayandeh rood” river that originates from the western Zagros mountain range passes through the city and irrigates the gardens and fields in and around the city. Although Isfahan is known as a city for ACL (45), L. major has been found in the suburbs (28).
Several rural districts of Isfahan province are known to be the most important foci of ZCL in Iran. In 2007/2008, a new ZCL focus was found in the Aran-o-Bidgol region; L. major was detected in 10/14 cases and L. tropica was detected in the remaining 4. R. opimus was the reservoir and P. papatasi was identified as the vector (46).
Tehran, the current capital of Iran, has been known traditionally as an important ACL focus (47), but we could not find any molecular studies investigating the Leishmania species in CL patients. Recently, ZCL cases were detected in Pakdasht county in Tehran province (27) and L. major has been reported from Semnan and Qom provinces in central Iran (23, 48).
Karamian et al. (49) reported an emerging focus of ACL in Birjand county in South Khorasan province where L. tropica was identified in 89% (n = 51/57) of urban cases and 91% (n = 21/23) of ACL patients were rural dwellers. L. major was also detected in both urban and rural regions. In another study in 2016, ACL was also dominant (n = 55/60) in this county. In eastern Iran, L. tropica showed high genetic similarity with southeastern strains and may have originated from the southeast region (28).
Khuzestan province is endemic for ZCL and had an overall prevalence rate of 37.39/100,000. Recently, L. tropica has been detected in a minority of patients with prevalence rates ranging from 3 to 10%, including in the ancient city of Shush (10% rate) in northern Khuzestan (50, 51). Data in 2013 from the mountainous province of Lorestan showed a predominance of L. tropica; overall, 72% (45/64) of local CL patients were infected by L. tropica, notably from Poldokhtar (37/43, 86%) and Kouhdasht (4/5, 80%). In Noorabad (Delfan), only 1 and 9 of 10 patients were infected with L. tropica and L. major, respectively (52).
Kermanshah province, which borders Iraq to the east, had an overall CL prevalence rate of 7.4/100,000, with most cases (47%) coming from Ghasr-e-Shirin county (on the Iran Iraq border) where the prevalence was 264.5/100,000 (53). Most (84–89%) CL cases in Kermanshah province are caused by L. major (54, 55). Small studies have also found L. tropica in Kermanshah city and Sarpole-Zahab (2/6 isolates), while seven and five isolates from Ghasr-e-Shirin and Islamabad-e-Gharb, respectively, were L. major (54).
Ilam province, sandwiched between Kermanshah and Khuzestan provinces and Iraq, is also endemic for L. major. Two studies in Ilam and Mehran counties showed that all CL cases (n = 61/61 and n = 92/92, respectively) were infected by L. major (56, 57).
One study in 2010 reported 62 CL cases from different health centers in Mazandaran province but did not report the species (58). However, L. major was detected from most (98.4%, 23/125) CL patients in the Turkman sahra region in Golestan province (prevalence rate of 32.90/100,000 in 2013), which is a well-known focus for CL (29, 59).
Khorasn-Razavi province is an area of hyper-endemic CL that had a prevalence rate of 64/100,000 in 2013 (30). Mashhad city, the capital of the province, is a long-standing ACL endemic focus and a recent study reconfirmed the predominance (10:1) of ACL over ZCL in 94 patients (60). In another study of 164 CL cases from Torghabe-Shandiz, southwest of Mashhad city, all patients were infected by L. tropica (61). Also, 10/12, 20/23, 38/49, and 53/60 CL patients from Bardaskan, Gonabad, Kashmar, and Torbat-e-Heydarieh cities and counties, respectively, which are located in the central and southern regions of this province, were infected with L. tropica (62, 63).
In Sarakhs, a city in the center of Sarakhs county in the northeastern Khorasan Razavi, 12 of 62 CL patients were infected by L. tropica and 5/12 patients had not traveled to ACL endemic areas, suggesting local transmission of ACL (64).
North Khorasan has been endemic for ZCL for decades with a prevalence rate of 50/100,000 (65). L. tropica was only detected in 5% (2/43) of sampled patients in this province and 95% of cases were found as ZCL (66).
Phylogenetic studies showed that northeastern L. tropica strains were genetically distinct from southeast–east Iranian strains but had high genetic similarity with some strains of southwest Iran (28).
Although L. tropica is a well-recognized cause of CL, it has rarely been associated with VL. Since the first reported case from Kenya (67), additional reports have emerged in American soldiers deployed in Iraq (68), and patients from Iran and India (16, 69, 70). Moreover, canine VL due to L. tropica has also been described in Iran and Morocco (32, 71).
In Iran, viscerotropic disease was first reported in 2006 in a patient from southwestern Iran, and more cases have since been reported from several regions in Iran (23, 69). The frequencies of L. tropica-associated VL were 1.4% in 1993–1994 (69); 9% of bone marrow samples in patients were from southeast provinces (72) and 14% of patients were from southwest Iran (17).
Turkey, which borders northwestern Iran, is one of the main foci of L. tropica in the Middle East. Aside from L. tropica, L. infantum causes CL in some foci (73), while L. major accounts for few CL cases (74).
The vast majority of CL patients (99%) are reported from the southeastern Anatolia region (SE-AR) and the southern Mediterranean region (MR) (75). Sanliurfa province in SE-AR borders Syria and is a highly endemic area and responsible for more than 50% of the CL burden in Turkey (Figure 2). Other CL-affected provinces include Diarbakir (SE-AR), and several MR provinces, Adana, Osmaniye, Hatay, Diarbakir, and Mersin/Icel. Low CL endemic provinces include Kahramanmaras, Antalya, Aydin, Kayseri, Nigde, and Mus provinces (73).
Based on the molecular species identification, L. tropica was reported from Sanliurfa and Diarbakir (southeast); Nigde (MR, south); Izmir, Aydin, and Mugla (ER, southwest); Bingol (east); Konya (Central Anatolia region, south-central); and Adana and Hatay (south), while L. infantum was known as the main species in Hatay province and reported from one patient in Osmaniye (south) (76–81). L. major has also been reported in small numbers in Mus, Hatay, Adana, Sanlirufa, and Antalya (74, 82, 83). The presence of L. major in southern Turkey is due to the arrival of Syrian refugees since 2011 to these areas where L. major has not been identified previously (83).
There is high L. tropica heterogeneity in Turkey with distinct clades (84–86). There are genetic similarities between northwest Iranian L. infantum strains and hybrid strains of L. infantum/L. donovani in Cukurova in southeast Turkey (17). The recent arrival of refugees from Syria will add additional genetic heterogeneity to the L. tropica genetic pool in Turkey (86). Aside from humans, L. tropica, L. major, and L. infantum have been isolated frequently in cats in Turkey and may be influenced by different ecological cycles (87, 88).
Saudi Arabia is another important focus of CL (89), with most cases occurring in southwest, central, and eastern parts of the country. L. tropica is mainly endemic in Aseer province in the southwest (90) while L. major is reported from Hail, Al-Hasa, Eastern, and the Riyadh provinces (91, 92).
Areas of mixed ACL and ZCL occur in Al-Madinah (91) and Al-Baha (93) provinces, the Taif region of Makkah provinces (94) in the southwest and Al-Qasim province in central Saudi (93). CL is declining and reported at low frequencies (mean 1.6–16.6 cases annually from 2006 to 2012) or sporadically in Tabooq, Jazan, Najran Al-Jouf, and northern borders (Figure 3) (95).
Each year, tens of thousands of pilgrims from Iran and other CL endemic countries travel to Al Madinah and Al Makkah for the holy Haj pilgrimage and usually stay for 12 to 30 days. This population may become infected with Saudi strains of L. tropica and L. major and also contribute new strains of CL to the local population. One study reported that the internal transcriber spacer (ITS) gene of L. major isolates from Al Madinah were 100% concordant with some Iranian L. major strains (96).
Iraq borders Iran, Turkey, and Saudi Arabia. CL has been prevalent in all parts of country except in the three northeastern provinces of Sulaymaniyah, Dahuk, and Arbil (97). L. tropica is prevalent in the suburbs of major cities such as Baghdad and Mosul and L. major is reported from different regions throughout the country, although no clear species typing has been reported for most regions (Figure 4) (98). The Wasit governorate is one of the most important CL endemic areas in Iraq where the L. major–L. tropica ratio is 3:2 (99, 100). Wasit borders the western Iranian provinces of Ilam, Kermanshah, Khuzestan, and Lorestan, which harbor mostly L. major, and is on a main route to Karbala province, where millions of Iranian pilgrims travel annually. We could not find any phylogenetic studies of Iraqi L. tropica but it is likely that there are shared genotypes between eastern Iraq and western Iran. A genetic study of CL in Iraq identified L. major as the cause of a CL outbreak in the Garmian region in Sulaymaniyeh province from where there are no previous cases of CL (101). Garmian is close to Ghasr-e-Shirin county in Kermanshah province, Iran, a known focus of L. major (54). Moreover, the phylogenetic analysis of the Iraqi L. major showed a close genetic relationship of L. major from Ghasr-e-Shirin (101).
Pakistan, the southeast neighbor of Iran, is endemic for both ACL and ZCL. L. tropica is widely distributed in different parts of all provinces in this country, but notable areas of high endemicity include the Afghan refugee camps in northwest Pakistan (102), the west–northwest province of Khyber Pakhtunkhwa province (5.17%), which borders Afghanistan, and Baluchistan province, the south-SW province bordering Iran (103). L. tropica is also found in Punjab and Sind provinces. The main focus of L. major is Balochistan (104) and central parts of Sind province (Figure 5) (105) where it is mostly a disease in the plains whereas L. tropica is prevalent in mountainous regions and the cities (104, 106).
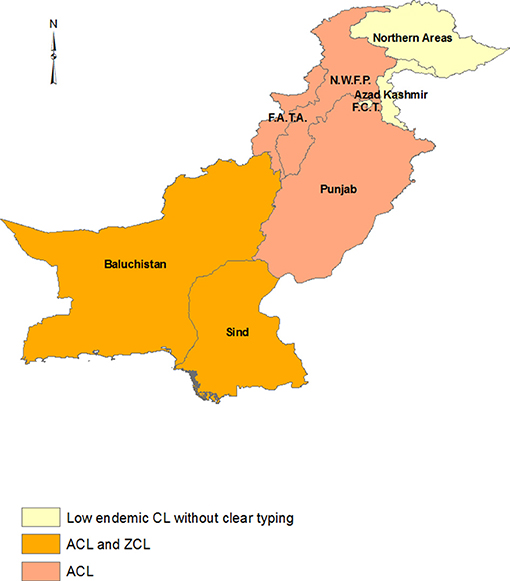
Figure 5. Distribution of CL types in Pakistan. The Khyber Pakhtunkhwa province was assumed the areas under N.W.F.P and F.A.T.A regions.
One genetic study revealed low genetic polymorphism for L. tropica from Balochistan and Sind (104). West Afghanistan (Herat province) and east (South Khorasan) and southeast (Kerman province) Iran are the main L. tropica foci in close proximity to Pakistan and showed higher homogeneity alongside central Iran isolates in comparison to other Iran's L. tropica foci (11, 28). There has not been a direct genetic comparison between Pakistan and Indian L. tropica, but interestingly, there is genetic similarity between southeast Iranian and Indian L. tropica strains (107), which raises the possibility that there may be a Pakistan–India genetic link.
Afghanistan, another country on Iran's eastern border, is well known as a high L. tropica endemic country. The main foci are Kabul city and province, Herat, Badakhshan, and Kandahar with lesser burdens in Kunduz and Balkh provinces. The ACL rate in Kabul province is the highest in the country with a reported prevalence rate of 21% (108, 109). In Herat province, L. tropica accounts for 96 to 98% of CL patients (11, 110) and shares genetic characteristics with east and southeast Iranian strains (11, 111). Badakhshan and Kandahar provinces border Khyber Pakhtunkhwa and Baluchistan provinces of Pakistan, respectively, but there are no genetic comparative studies of L. tropica.
L. major is reported as the minority species in Herat, Badakhshan, and Kandahar (112–114), but is more commonly found in Balkh and Kunduz provinces in northern Afghanistan along the border with Tajikistan (113). CL was reported from Parwan province in central Afghanistan but no data on the CL type has been published (Figure 6) (112). Afghanistan has suffered from political instability and periods of civil wars for decades, and a comprehensive view on the situation of CL in this country is not possible based on the existing data.
Turkmenistan, Iran's northeastern neighbor, has been known historically as an ACL endemic country (115). The town of Ashkabad was an important focus but following concerted vector control efforts against the sandfly, the incidence of ACL fell but appears to be making a comeback (116, 117). Between 2000 and 2009, 1562 cases of CL were reported in Turkmenistan, mostly in the southern and eastern parts of the country bordering Iran and Afghanistan (118). L. major is also reported from time to time in the local population and travelers (117, 119).
Recent years of conflict in Syria and the migration of large numbers of Syrian refugees to other countries have substantially affected the situation of leishmaniasis in the Middle East. By 1960, the prevalence of CL in Syria was limited to the two endemic regions of Aleppo (north) and Damascus (south) of Syria. In 2010, before the onset of the civil war, the incidence of CL was estimated at 23,000 cases a year (120), while after the onset of the war, the incidence of the disease almost doubled to 41,000 cases in 2013 (121). L. tropica-associated ACL is the main form of CL in Aleppo, whereas in Damascus, it is L. major ZCL (120). Overall, studies report that most cases of CL (about 90%) are caused by L. tropica (122). In 2009 (precivil war), of the 51,119 reported cases of CL in Syria, Jordan, Iraq, and Saudi Arabia, some 90.7% of cases were reported from Syria, making Syria the highest burden country for ACL in the region (123). Displacement of Syrian refugees to neighboring countries such as Lebanon, Iraq, Jordan, and Turkey has led to outbreaks of CL and altered the local epidemiology (124). In 2012, Saroufim et al. (125) sampled 948 Syrian patients residing in the refugee camps in Lebanon and reported that 85% of them were infected with L. tropica and 15% by L. major. According to the Lebanese Ministry of Health reports, only 6 cases of CL in Lebanon were reported between 2000 and 2012, while in 2011, 1033 new cases were reported; some 97% were Syrian refugees (124). Moreover, Syrian refugees have carried L. major and L. donovani to Turkey, two species that are not endemic to Turkey (83). Table 1 briefly shows the distribution of ACL and ZCL in the mentioned countries.
Our review reconfirms that CL remains an important disease in Iran and its neighboring countries (Figure 7). In this part of the Middle East, L. major ZCL and L. tropica ACL are present throughout the region in different ecological foci and with varying ratios of the two species. Data from 2012 suggest that Syria has the highest ACL burden followed by Iran, while Iran leads the ZCL burden followed closely by Saudi Arabia (126).
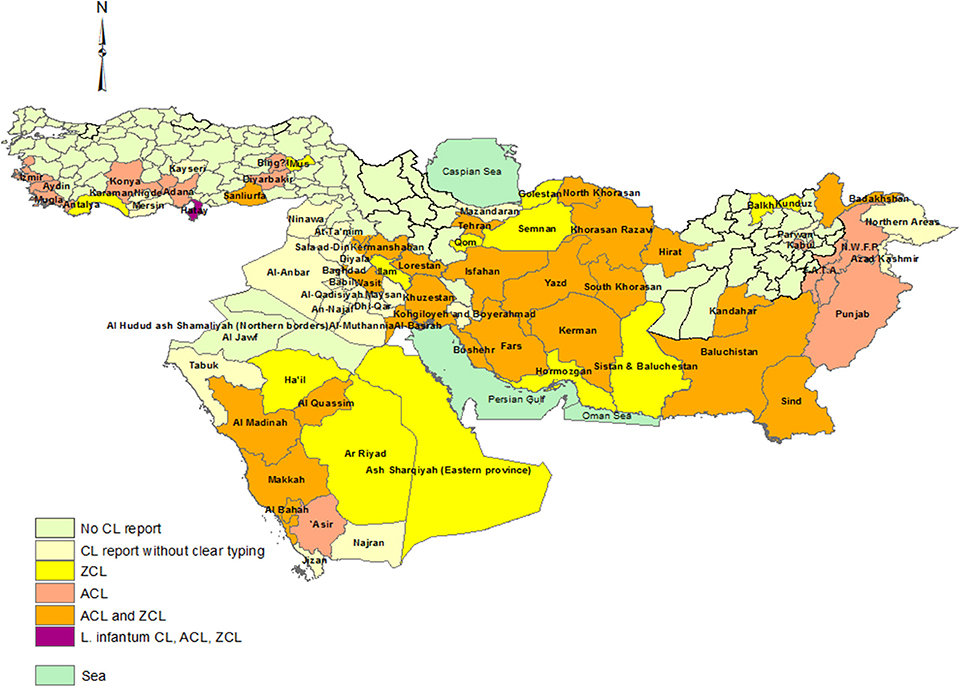
Figure 7. Distribution of CL types in the part of Middle East including Turkey, Iraq, Iran, Saudi Arabia, Afghanistan, and Pakistan in a view.
In this geographically heterogeneous region, CL has decreased in Iran and Saudi Arabia, increased dramatically in Syria, and re-emerged in Turkey and hitherto low endemic Lebanon. CL is geographically restricted to the southern coastline of Turkey and in selected provinces in Afghanistan while being extensively distributed in Iran, Saudi Arabia, and Iraq. Despite a reducing CL burden in Iran, new foci have merged including L. tropica, which is now reported from 10 provinces. Although L. tropica has long been known as an urban disease, it has expanded to rural areas and smaller towns in Iran (28, 127).
The relative lack of genotypic data makes it difficult to determine the patterns of spread and emergence in new foci. This is particularly important in border areas where traditionally there is much freedom of movement of populations. Phylogenetic data from Iran suggest that CL strains from SE Iran are related to strains from Herat in western Afghanistan, and strains from northwest Iran related to strains from SE Turkey (11, 28).
Moreover, the opening of the border between Iran and Iraq appears to have played an important role in the spread and the transmission of Leishmania strains between the two countries (128). High genetic similarity between a newly emerged Iraqi L. major strain and an Iranian strain support this hypothesis (101). As a vector-borne disease with two predominant vectors, P. papatasi (ZCL) and P. sergenti (ACL), and animal and human reservoirs, respectively, multiple factors will affect disease prevalence and epidemic proneness that will be favor one species over another or be common to both. Climate and ecology are particularly important.
Our investigations show that the risk of CL declines with increasing northern latitude, consistent with the findings of Alvar et al. (118) who reported the number of CL cases of 99, 17, and 0/year in Turkmenistan, Azerbaijan, and Armenia; all of these countries are north of Iran. Given the nature of the CL life cycle, Ready (129) suggested that climate change affects the distribution of leishmaniasis either directly by the effect of temperature on the development of the parasite in the sandfly, or indirectly by affecting the distribution and density of the vector species; added to these factors are the social and economic changes in human interaction with the transmission cycle. Long-term climate change has been shown to spread sandfly species to areas not previously observed (130), e.g., climate warming has caused sandflies to migrate to the high altitudes of the Atlas Mountains in Morocco (131).
The density of P. papatasi, a vector of L. major, increases with the aridity, which, in turn, occurs in response to global warming. Cross and Hyams have predicted that a 1 to 5°C increase in ambient temperature due to global warming will lead to the geographical spread of P. papatasi in the Middle East and an extension of the L. major transmission season to become perennial in Saudi Arabia (132). Currently, in Saudi Arabia, P. papatasi thrives best in the summer months and peak indoor activity occurs in May and peak density is seen in the late evening (10–11 p.m.); by winter, its population decreases drastically but starts to pick up in early spring in March (133). Martin-Sanchez et al. (134) have suggested that global warming has a greater effect on disease transmission rather than an increase in sandfly density. The effect of climate and ecology is illustrated well from studies in ZCL provinces of Iran where favorable conditions included mildly sloping hills of limited elevation, flat lands, irrigated farmlands, and moderate degrees of rainfall (41, 135). Consistent findings are reported by Ramezankhani et al. (135) who also report that lower wind speeds of <14 m/s were associated with increased disease incidence, independent of other climatic and geographical factors. In Golestan, a humid province in northeast Iran, ZCL prevalence was higher in areas with the lower relative humidity (136), whereas in Fars province, continued drought was associated with a decline in CL (137). Gerbils are important rodent reservoirs for supporting continued ZCL transmission and include M. libycus (the Libyan jird), Psammomys obesus (the fat tailed sand rat) in Saudi Arabia, M. libycus, T. indica (the Indian gerbil or antelope rat), and R. opimus (great gerbil) in Iran (133, 138). In a remarkable feat of evolution, P. papatasi has adapted to living in burrows inhabited by the fat tailed sand rat wherein high humidity and cooler temperatures are essential for sandfly survival; thus, this vector reservoir combination is a powerful driver of ZCL (139). Caves, deep cracks in walls, and dark corners in houses are also good for sandfly survival, even when with extreme temperature and aridity.
Population movements are another important risk for CL emergence, spread, and epidemics. These are related to non-immune populations moving into endemic areas or when CL-infected individuals move to a low endemic area with competent vectors; in both scenarios, population density is important. ZCL epidemics have been linked to construction workers moving into arid areas for building projects and American soldiers becoming infected in Iraq, while ACL epidemics have been reported in aid workers responding to the earthquake in Bam (SE Iran), large cities like Kabul, Afghan refugees in camps in Pakistan, and Syrian refugees in Lebanon and Turkey. Our region is well known for its religious pilgrimages and festivals, and each year, millions of pilgrims attend the Haj and Umrah in Makkah, the Arba'een pilgrimage in Karbala, Iraq, and Christmas in Jerusalem/Al Quds and Bethlehem in Israel and Palestine. This provides the opportunity for mixing of CL genotypes and non-immunes to become infected. War is a particularly effective way of causing CL epidemics not just because of population displacement but because the health infrastructure breaks down, resulting in very limited access to health care, destruction of buildings, and a deterioration in sanitation (140). The civil war in Syria is a striking example with large numbers from highly CL endemic foci causing epidemics in neighboring countries, as well as the emergence of new foci of CL caused by non-indigenous Leishmania species. Similarly, since 2014 in Iraq, population displacement has led to CL outbreaks in non-endemic areas between north and central Iraq (101). CL is found more commonly in poor and marginalized populations due to a variety of factors that include small overcrowded poorly constructed dwellings with no screens on windows, poor nutritional status, lack of personal protection against insect bites, and the presence of garbage collection areas (141).
Limited access to health and diagnosis due to lack of money or war-related infrastructure destruction and displacement exacerbate the situation.
Under-reporting of CL is probably widespread and data based on passive detection, as used in Iran (137), are significantly lower compared to active surveillance (142) because patients do not always seek treatment centers. In a study in Jordan, covering 2001–2003, the estimated incidence of CL was 47 times higher than official reports (142). Active surveillance for CL may be implemented only in emergencies such as outbreaks of the disease, like the case finding project of the Lebanese Ministry of Health, after the onset of the civil war in Syria and the influx of Syrian refugees to this country. Another issue is diagnosis. Some countries lack access to molecular techniques so species identification is impossible; thus, CL burden data are based on microscopy or the clinical description of lesions, e.g., dry or wet cutaneous lesions (109).
Surveillances should be improved by determining the species of Leishmania causing CL in each region by using molecular methods. Moreover, in the map reports, the distribution of the disease should be specified based on the species of the parasite. Using GIS and performing geospatial research are advised to stratify areas based on the probability of the disease. By using these data and producing hazard maps, the disease control and prevention can be better managed. Moreover, undertaking active disease screening in suspected areas of CL especially in refugee camps and increasing access to health care to remote areas and marginalized populations and introducing free treatment of CL can be the keyways to reducing the burden of CL in this region of the world.
CL in the Middle East is a disease of complex interactions between parasite reservoirs, parasites, vectors, climate, ecology, political instability, poverty, and socioeconomic changes. Against this background is increasing and decreasing CL burden in different countries as well as newly emerging foci on low and high endemic countries. Effective control is challenging, must take all of these factors into account, and must adopt a multidisciplinary approach based on sound epidemiological evidence. This review sheds light on CL in our region with a focus on Iran, a country with a substantial burden.
MG conceptualized and wrote the initial draft. WT and MK critically appraised and revised the manuscript. All authors reviewed and approved the final manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors would like to thank Dr. Zahra Kanannejad for help in the data gathering.
1. Zijlstra EE. isceral leishmaniasis: a forgotten epidemic. Arch Dis Child. (2016) 101:561–7. doi: 10.1136/archdischild-2015-309302
2. Burza S, Croft SL, Boelaert M. Leishmaniasis. Lancet. (2018) 392:951–70. doi: 10.1016/S0140-6736(18)31204-2
3. Gramiccia M, Gradoni L. The current status of zoonotic leishmaniases and approaches to disease control. Int J Parasitol. (2005) 35:1169–80. doi: 10.1016/j.ijpara.2005.07.001
4. Akhavan AA, Yaghoobi-Ershadi MR, Khamesipour A, Mirhendi H, Alimohammadian MH, Rassi Y, et al. Dynamics of Leishmania infection rates in Rhombomys opimus (Rodentia: Gerbillinae) population of an endemic focus of zoonotic cutaneous leishmaniasis in Iran. Bull Soc Pathol Exot. (2010) 103:84–9. doi: 10.1007/s13149-010-0044-1
5. Dereure J, Rioux JA, Gallego M, Perieres J, Pratlong F, Mahjour J, et al. Leishmania tropica in Morocco: infection in dogs. Trans R Soc Trop Med Hyg. (1991) 85:595. doi: 10.1016/0035-9203(91)90356-4
6. Talmi-Frank D, Jaffe CL, Nasereddin A, Warburg A, King R, Svobodova M, et al. Leishmania tropica in rock hyraxes (Procavia capensis) in a focus of human cutaneous leishmaniasis. Am J Trop Med Hyg. (2010) 82:814–8. doi: 10.4269/ajtmh.2010.09-0513
7. Talmi-Frank D, Kedem-Vaanunu N, King R, Bar-Gal GK, Edery N, Jaffe CL, et al. Leishmania tropica infection in golden jackals and red foxes, Israel. Emerg Infect Dis. (2010) 16:1973. doi: 10.3201/eid1612.100953
8. Fikre H, Mohammed R, Atinafu S, van Griensven J, Diro E. Clinical features and treatment response of cutaneous leishmaniasis in North-West Ethiopia. Trop Med Int Health. (2017) 22:1293–301. doi: 10.1111/tmi.12928
9. van Griensven J, Gadisa E, Aseffa A, Hailu A, Beshah AM, Diro E. Treatment of cutaneous leishmaniasis caused by Leishmania aethiopica: a systematic review. PLoS Negl Trop Dis. (2016) 10:e0004495. doi: 10.1371/journal.pntd.0004495
10. Remadi L, Haouas N, Chaara D, Slama D, Chargui N, Dabghi R, et al. Clinical presentation of cutaneous leishmaniasis caused by Leishmania major. Dermatology. (2016) 232:752–9. doi: 10.1159/000456543
11. Fakhar M, Ghohe HP, Rasooli SA, Karamian M, Mohib AS, Hezarjaribi HZ, et al. Genetic diversity of Leishmania tropica strains isolated from clinical forms of cutaneous leishmaniasis in rural districts of Herat province, Western Afghanistan, based on ITS1- rDNA. Infect Genet Evol. (2016) 41:120–7. doi: 10.1016/j.meegid.2016.03.031
12. Bhutto AM, Soomro RA, Nonaka S, Hashiguchi Y. Detection of new endemic areas of cutaneous leishmaniasis in Pakistan: a 6-year study. Int J Dermatol. (2003) 42:543–8. doi: 10.1046/j.1365-4362.2003.01818.x
13. Sharifi I, Fekri AR, Aflatoonian MR, Khamesipour A, Mahboudi F, Dowlati Y, et al. Leishmaniasis recidivans among school children in Bam, South-east Iran, 1994–2006. Int J Dermatol. (2010) 49:557–61. doi: 10.1111/j.1365-4632.2010.04419.x
14. Gitari JW, Nzou SM, Wamunyokoli F, Kinyeru E, Fujii Y, Kaneko S, et al. Leishmaniasis recidivans by Leishmania tropica in central rift valley region in Kenya. Int J Infect Dis. (2018) 74:109–16. doi: 10.1016/j.ijid.2018.07.008
15. Dassoni F, Daba F, Naafs B, Morrone A. Leishmaniasis recidivans in Ethiopia: cutaneous and mucocutaneous features. J Infect Dev Ctries. (2017) 11:106–10. doi: 10.3855/jidc.8516
16. Khanra S, Datta S, Mondal D, Saha P, Bandopadhyay SK, Roy S, et al. RFLPs of ITS, ITS1 and hsp70 amplicons and sequencing of ITS1 of recent clinical isolates of Kala-azar from India and Bangladesh confirms the association of L. tropica with the disease. Acta Trop. (2012) 124:229–34. doi: 10.1016/j.actatropica.2012.08.017
17. Ghatee MA, Mirhendi H, Karamian M, Taylor WR, Sharifi I, Hosseinzadeh M, et al. Population structures of Leishmania infantum and Leishmania tropica the causative agents of kala-azar in Southwest Iran. Parasitol Res. (2018) 117:3447–58. doi: 10.1007/s00436-018-6041-1
18. Parvizi P, Alaeenovin E, Mohammadi S, Baghban N. Occurrence of low density of Leishmania infantum in sandflies from a new focus of visceral leishmaniasis in northwest of Iran. J Vector Borne Dis. (2013) 50:127–32.
19. Sarkari B, Naraki T, Ghatee MA, Khabisi SA, Davami MH. Visceral leishmaniasis in southwestern Iran: a retrospective clinico-hematological analysis of 380 consecutive hospitalized cases (1999–2014). PLoS ONE. (2016) 11:e0150406. doi: 10.1371/journal.pone.0150406
20. Jahangir AB, Akhoundi B, Mohebali M, Ghaderipour A, Kakoee Z, Najafi F. Seroepidemiological survey of human visceral leishmaniasis in Ilam province, west of Iran in 2013. Iran J Parasitol. (2015) 10:56–61.
21. Arzamani K, Vatandoost H, Rassi Y, Abai MR, Akhavan AA, Alavinia M, et al. Susceptibility status of wild population of Phlebotomus sergenti (Diptera: Psychodidae) to different imagicides in a endemic focus of cutaneous leishmaniasis in northeast of Iran. J Vector Borne Dis. (2017) 54:282–6. doi: 10.4103/0972-9062.217621
22. Mohebali M. Visceral leishmaniasis in Iran: review of the epidemiological and clinical features. Iran J Parasitol. (2013) 8:348–58.
23. Tashakori M, Ajdary S, Kariminia A, Mahboudi F, Alimohammadian MH. Characterization of Leishmania species and L. major strains in different endemic areas of cutaneous leishmaniasis in Iran. Iran Biomed J. (2003) 7:43–50.
24. Mirzaie F, Eslami G, Yosefi MH, Pestehchian N. Molecular identification of Leishmania isolates obtained from patients suspected as having cutaneous leishmaniasis referred to reference laboratories from Yazd province in central Iran. Adv Biomed Res. (2013) 2:92. doi: 10.4103/2277-9175.122525
25. Akhlagh A, Salehzadeh A, Zahirnia AH, Davari B. 10-year trends in epidemiology, diagnosis, and treatment of cutaneous Leishmaniasis in Hamadan Province, West of Iran (2007–2016). Front Public Health. (2019) 7:27. doi: 10.3389/fpubh.2019.00027
26. Kolivand M, Fallah M, Salehzadeh A, Davari B, Poormohammadi A, Pazoki Ghohe H, et al. An epidemiological study of cutaneous leishmaniasis using active case finding among elementary school students in Pakdasht, Southeast of Tehran, Iran 2013-2014. J Res Health Sci. (2015) 15:104–8. doi: 10.13140/RG.2.1.2619.6643
27. Izadi S, Mirhendi H, Jalalizand N, Khodadadi H, Mohebali M, Nekoeian S, et al. Molecular epidemiological survey of cutaneous leishmaniasis in two highly endemic metropolises of Iran, application of FTA cards for DNA extraction from Giemsa-stained slides. Jundishapur J Microbiol. (2016) 9:e32885. doi: 10.5812/jjm.32885
28. Karamian M, Kuhls K, Hemmati M, Ghatee MA. Phylogenetic structure of Leishmania tropica in the new endemic focus Birjand in East Iran in comparison to other Iranian endemic regions. Acta Trop. (2016) 158:68–76. doi: 10.1016/j.actatropica.2016.02.010
29. Norouzinezhad F, Ghaffari F, Norouzinejad A, Kaveh F, Gouya MM. Cutaneous leishmaniasis in Iran: results from an epidemiological study in urban and rural provinces. Asian Pac J Trop Biomed. (2016) 6:614–9. doi: 10.1016/j.apjtb.2016.05.005
30. Akhoundi M, Mohebali M, Asadi M, Mahmodi MR, Amraei K, Mirzaei A. Molecular characterization of Leishmania spp. in reservoir hosts in endemic foci of zoonotic cutaneous leishmaniasis in Iran. Folia Parasitol. (2013) 60:218–24. doi: 10.14411/fp.2013.024
31. Saghafipour A, Vatandoost H, Zahraei-Ramazani AR, Yaghoobi-Ershadi MR, Jooshin MK, Rassi Y, et al. Epidemiological study on cutaneous leishmaniasis in an endemic area, of Qom province, central Iran. J Arthropod Borne Dis. (2017) 11:403–13.
32. Hajjaran H, Mohebali M, Zarei Z, Edrissian G. Leishmania tropica: another Etiological Agent of Canine Visceral Leishmaniasis in Iran. Iran J Public Health. (2007) 36:85–8.
33. Schnur LF, Nasereddin A, Eisenberger CL, Jaffe CL, El Fari M, Azmi K, et al. Multifarious characterization of Leishmania tropica from a Judean desert focus, exposing intraspecific diversity and incriminating Phlebotomus sergenti as its vector. Am J Trop Med Hyg. (2004) 70:364–72. doi: 10.4269/ajtmh.2004.70.364
34. Hanafi-Bojd AA, Yaghoobi-Ershadi MR, Haghdoost AA, Akhavan AA, Rassi Y, Karimi A, et al. Modeling the distribution of cutaneous leishmaniasis vectors (Psychodidae: Phlebotominae) in Iran: a potential transmission in disease prone areas. J Med Entomol. (2015) 52:557–65. doi: 10.1093/jme/tjv058
35. Shirzadi MR, Gouya MM. National Guidelines for cutaneous leishmaniasis surveillance in Iran. Ministry of Health and Medical Education (MOH). Tehran Iran: Zoonoses Control Department. (2012). p. 1–78.
36. Sharifi F, Sharifi I, Zarean M, Parizi MH, Aflatoonian MR, Harandi MF, et al. Spatial distribution and molecular identification of Leishmania species from endemic foci of south- eastern Iran. Iran J Parasitol. (2012) 7:45–52.
37. Ghatee MA, Mirhendi H, Marashifard M, Kanannejad Z, Taylor WR, Sharifi I. Population Structure of Leishmania tropica Causing Anthroponotic Cutaneous Leishmaniasis in Southern Iran by PCR-RFLP of Kinetoplastid DNA. Biomed Res Int. (2018) 2018:6049198. doi: 10.1155/2018/6049198
38. Azizi K, Soltani A, Alipour H. Molecular detection of Leishmania isolated from cutaneous leishmaniasis patients in Jask County, Hormozgan Province, Southern Iran, 2008. Asian Pac J Trop Med. (2012) 5:514–7. doi: 10.1016/S1995-7645(12)60090-X
39. Mohebali M, Darabi H, Hajjaran H, Shirzadi MR, Fouladvand M, Charehdar S, et al. Molecular and parasitological study of cutaneous leishmaniasis in Bushehr province, southwest of the Islamic Republic of Iran: a cross-sectional study during 2009–2012. J Parasit Dis. (2015) 39:371–6. doi: 10.1007/s12639-013-0370-x
40. Yaghoobi-Ershadi MR, Shahbazi F, Darvishi M, Akhavan AA, Jafari R, Khajeian M, et al. Molecular epidemiological study of cutaneous leishmaniasis in the focus of Bushehr city, southwestern Iran. J Arthropod Borne Dis. (2013) 7:113–21.
41. Ghatee MA, Haghdoost AA, Kooreshnia F, Kanannejad Z, Parisaie Z, Karamian M, et al. Role of environmental, climatic risk factors and livestock animals on the occurrence of cutaneous leishmaniasis in newly emerging focus in Iran. J Infect Public Health. (2018) 11:425–33. doi: 10.1016/j.jiph.2017.12.004
42. Yaghoobi-Ershadi MR, Hanafi-Bojd AA, Javadian E, Jafari R, Zahraei-Ramazani AR, Mohebali M. A new focus of cutaneous leishmaniasis caused by Leishmania tropica. Saudi Med J. (2002) 23:291–4.
43. Yaghoobi-Ershadi MR, Jafari R, Hanafi-Bojd AA. A new epidemic focus of zoonotic cutaneous leishmaniasis in central Iran. Ann Saudi Med. (2004) 24:98–101. doi: 10.5144/0256-4947.2004.98
44. Holakouie-Naieni K, Mostafavi E, Boloorani AD, Mohebali M, Pakzad R. Spatial modeling of cutaneous leishmaniasis in Iran from 1983 to 2013. Acta Trop. (2017) 166:67–73. doi: 10.1016/j.actatropica.2016.11.004
45. Yaghoobi-Ershadi MR. Phlebotomine sand flies (Diptera: Psychodidae) in Iran and their role on Leishmania transmission. J Arthropod Borne Dis. (2012) 6:1–17.
46. Doroodgar A, Sadr F, Razavi MR, Doroodgar M, Asmar M, Doroodgar M. A new focus of zoonotic cutaneous leishmaniasis in Isfahan Province, Central Iran. Asian Pac J Trop Dis. (2015) 5:S54–8. doi: 10.1016/S2222-1808(15)60857-X
47. Motazedian H, Noamanpoor B, Ardehali S. Characterization of Leishmania parasites isolated from provinces of the Islamic Republic of Iran. East Mediterr Health J. (2002) 8:338–44.
48. Akhavan AA, Yaghoobi-Ershadi MR, Mehdipour D, Abdoli H, Farzinnia B, Mohebali M, et al. Epidemic outbreak of cutaneous leishmaniasis due to Leishmania major in Ghanavat rural district, Qom Province, Central Iran. Iran J Public Health. (2003) 32:35–41.
49. Karamian M, Bojd F, Salehabadi A, Hemmati M, Barati DA. Effectiveness of meglumine antimoniate against L. tropica in a recently emerged focus of cutaneous leishmaniasis in Birjand, eastern Islamic Republic of Iran. East Mediterr Health J. (2015) 21:280–6. doi: 10.26719/2015.21.4.280
50. Maraghi S, Mardanshah O, Rafiei A, Samarbafzadeh A, Vazirianzadeh B. Identification of cutaneous leishmaniasis agents in four geographical regions of Khuzestan province using Nested PCR. Jundishapur J Microbiol. (2013) 6:1–4. doi: 10.5812/jjm.4866
51. Maraghi S, Zadeh AS, Sarlak AA, Ghasemian M, Vazirianzadeh B. Identification of cutaneous leishmaniasis agents by nested Po-Lymerase chain reaction (Nested-PCR) in Shush City, Khuzestan Province, Iran. Iranian J Parasitol. (2007) 2:13–5.
52. Kheirandish F, Sharafi AC, Kazemi B, Mohebali M, Sarlak A, Tarahi MJ, et al. Identification of Leishmania species using PCR assay on giemsa-stained slides prepared from cutaneous leishmaniasis patients. Iran J Parasitol. (2013) 8:382–8.
53. Hamzavi Y, Khademi N. Trend of cutaneous leishmaniasis in Kermanshah Province, west of Iran from 1990 to 2012. Iran J Parasitol. (2015) 10:78–86. 54.
54. Alimoradi S, Hajjaran H, Mohebali M, Mansouri F. Molecular identification of Leishmania species isolated from human cutaneous leishmaniasis by RAPD-PCR. Iranian J Public Health. (2009) 38:44–50.
55. Akia A, Hamzavi Y. Diagnosis and molecular typing of leishmania in patients with cutaneous leishmaniasis. J Mazand Univ Med Sci. (2017) 26:22–30.
56. Haddad MH, Ghasemi E, Maraghi S, Tavala M. Identification of Leishmania species isolated from human cutaneous leishmaniasis in Mehran, Western Iran using nested PCR. Iran J Parasitol. (2016) 11:65–72.
57. Kermanjani A, Akhlaghi L, Oormazdi H, Hadighi R. Isolation and identification of cutaneous leishmaniasis species by PCR–RFLP in Ilam province, the west of Iran. J Parasit Dis. (2017) 41:175–9. doi: 10.1007/s12639-016-0772-7
58. Youssefi MR, Esfandiari B, Shojaei J, Jalahi H, Amoli SA, Ghasemi H, et al. Prevalence of cutaneous leishmaniasis during 2010 in Mazandaran Province, Iran. Afr J Microbiol Res. (2011) 5:5793–5. doi: 10.5897/AJMR11.528
59. Bordbar A, Parvizi P. High infection frequency, low diversity of Leishmania major and first detection of Leishmania turanica in human in northern Iran. Acta Trop. (2014) 133:69–72. doi: 10.1016/j.actatropica.2014.01.016
60. Mahmoodi MR, Mohajery M, Afshari JT, Shakeri MT, Panah MJ, Berenji F, et al. Molecular identification of Leishmania species causing cutaneous leishmaniasis in Mashhad, Iran. Jundishapur J Microbiol. (2010) 3:195–200.
61. Farash BR, Mohajery M, Fata A, Shamsian SA, Rezaee A, Yazdanpanah MJ. Anthroponotic cutaneous leishmaniasis in torghabeh-shandiz, a region with rural texture (a molecular study). Jundishapur J Microbiol. (2013) 6:1–3. doi: 10.5812/jjm.8274
62. Rezai A, Moghaddas E, Bagherpor MR, Naseri A, Shamsian SA. Identification of Leishmania Species for Cutaneous leishmaniasis in Gonabad, Bardaskan and Kashmar, Central Khorasan, 2015. Jundishapur J Microbiol. (2017) 10:e44469. doi: 10.5812/jjm.44469
63. Naseri A, Fata A, Rezai A, Hedayatimoghadam M, Berengi F, Akbarzadeh O, et al. Molecular identification of leishmania species in Torbat-e Heydarieh, Khorasan Razavi province, Iran. Health Sci. (2016) 5:87–92.
64. Shamsian S, Rezaei A, Akbarzadeh A, Hosseini Farash B. Molecular Identification of Leishmania tropica in an endemic border city for zoonotic cutaneous leishmaniasis (ZCL) in northeastern Iran. J Microb Exp. (2015) 2:53–8.
65. Javadian E, Nadim A, Tahvildare-Bidruni GH, Assefi V. Epidemiology of cutaneous leishmaniasis in Iran: B. Khorassan Part V: Report on a focus of zoonotic cutaneous leishmaniasis in Esferayen. Bull Soc Pathol Exot Filiales. (1976) 69:140–3.
66. Mahmoudzadeh-Niknam H, Ajdary S, Riazi-Rad F, Mirzadegan E, Rezaeian A, Khaze V, et al. Molecular epidemiology of cutaneous leishmaniasis and heterogeneity of Leishmania major strains in Iran. Trop Med Int Health. (2012) 17:1335–44. doi: 10.1111/j.1365-3156.2012.03078.x
67. Mebrahtu Y, Lawyer P, Githure J, Were JB, Muigai R, Hendricks L, et al. Visceral leishmaniasis unresponsive to pentostam caused by Leishmania tropica in Kenya. Am J Trop Med Hyg. (1989) 41:289–94. doi: 10.4269/ajtmh.1989.41.289
68. Magill AJ, Grogl M, Gasser RA Jr, Sun W, Oster CN. Visceral infection caused by Leishmania tropica in veterans of Operation Desert Storm. N Engl J Med. (1993) 328:1383–7. doi: 10.1056/NEJM199305133281904
69. Alborzi A, Rasouli M, Shamsizadeh A. Leishmania tropica–isolated patient with visceral leishmaniasis in southern Iran. Am J Trop Med Hyg. (2006) 74:306–7. doi: 10.4269/ajtmh.2006.74.306
70. Sacks DL, Kenney RT, Neva FA, Kreutzer RD, Jaffe CL, Gupta AK, et al. Indian kala-azar caused by Leishmania tropica. Lancet. (1995) 345:959–61. doi: 10.1016/S0140-6736(95)90703-3
71. Guessous-Idrissi N, Berrag B, Riyad M, Sahibi H, Bichichi M, Rhalem AN. Leishmania tropica: etiologic agent of a case of canine visceral leishmaniasis in northern Morocco. Am J Trop Med Hyg. (1997) 57:172–3. doi: 10.4269/ajtmh.1997.57.172
72. Hosseininasab A, Sharifi I, Mohammad Hossein DA, Zarean M, Dadkhah M. Causes of pediatric visceral leishmaniasis in southeastern Iran. Iran J Parasitol. (2014)9:584–7.
73. Gürel MS, Yesilova Y, Ölgen MK, Özbel Y. Cutaneous leishmaniasis in Turkey. Turkiye Parazitol Derg. (2012) 36:121–129. doi: 10.5152/tpd.2012.29
74. Gurses G, Ozaslan M, Zeyrek FY, Kiliç IH, Doni NY, Karagöz ID, et al. Molecular identification of Leishmania spp. isolates causes cutaneous leishmaniasis (CL) in Sanliurfa Province, Turkey, where CL is highly endemic. Folia Microbiol. (2018) 63:353–9. doi: 10.1007/s12223-017-0556-1
75. Ok ÜZ, Balcioglu IC, Özkan AT, Özensoy S, Özbel Y. Leishmaniasis in Turkey. Acta Trop. (2002) 84:43–8. doi: 10.1016/S0001-706X(02)00134-1
76. Culha G, Uzun S, Ozcan K, Memisoglu HR, Chang KP. Comparison of conventional and polymerase chain reaction diagnostic techniques for leishmaniasis in the endemic region of Adana, Turkey. Int J Dermatol. (2006) 45:569–72. doi: 10.1111/j.1365-4632.2006.02695.x
77. Akkafa F, Dilmec F, Alpua Z. Identification of Leishmania parasites in clinical samples obtained from cutaneous leishmaniasis patients using PCR-RFLP technique in endemic region, Sanliurfa province, in Turkey. Parasitol Res. (2008) 103:583–6. doi: 10.1007/s00436-008-1013-5
78. Toz SO, Nasereddin A, Ozbel Y, Ertabaklar H, Culha G, Sevil N, et al. Leishmaniasis in Turkey: molecular characterization of Leishmania from human and canine clinical samples. Trop Med Int Health. (2009) 14:1401–6. doi: 10.1111/j.1365-3156.2009.02384.x
79. Akpolat N, YILDIRIM IH, Çiçek M, Nergiz S, Özgür EZ, Özcan N, et al. Identification of causative species in patients with cuteneous leishmaniasis in Diyarbakir by polymerase chain reaction (pcr)-restriction fragment lenght polymorphism (RFLP). Int Arch Med Res. (2014) 6:17–20.
80. Culha G, Akyar I, Zeyrek FY, Kurt Ö, Gündüz C, Töz SÖ, et al. Leishmaniasis in Turkey: determination of Leishmania species by matrix-assisted laser desorption ionization time-of- flight mass spectrometry (MALDI-TOF MS). Iran J Parasitol. (2014) 9:239–48.
81. Beyhan YE, Karakus M, Karagoz A, Mungan M, Ozkan AT, Hokelek M. Detection and identification of cutaneous leishmaniasis isolates by culture, polymerase chain reaction and sequence analyses in Syrian and Central Anatolia patients. Saudi Med J. (2017) 38:968–71. doi: 10.15537/smj.2017.9.21025
82. Özbilgin A, Çulha G, Uzun S, Harman M, Topal SG, Okudan F, et al. Leishmaniasis in Turkey: first clinical isolation of Leishmania major from 18 autochthonous cases of cutaneous leishmaniasis in four geographical regions. Trop Med Int Health. (2016) 21:783–91. doi: 10.1111/tmi.12698
83. Koltas IS, Eroglu F, Alabaz D, Uzun S. The emergence of Leishmania major and Leishmania donovani in southern Turkey. Trans R Soc Trop Med Hyg. (2014) 108:154–8. doi: 10.1093/trstmh/trt119
84. Toz SO, Culha G, Zeyrek FY, Ertabaklar H, Alkan MZ, Vardarli AT, et al. A real-time ITS1- PCR based method in the diagnosis and species identification of Leishmania parasite from human and dog clinical samples in Turkey. PLoS Negl Trop Dis. (2013) 7:e2205. doi: 10.1371/journal.pntd.0002205
85. Eroglu F, Koltas IS, Alabaz D, Uzun S, Karakas M. Clinical manifestations and genetic variation of Leishmania infantum and Leishmania tropica in Southern Turkey. Exp Parasitol. (2015) 154:67–74. doi: 10.1016/j.exppara.2015.04.014
86. Ekşi F, Özgöztaşi O, Karsligil T, Saglam M. Genotyping Leishmania promastigotes isolated from patients with cutaneous leishmaniasis in south-eastern Turkey. J Int Med Res. (2017) 45:114–22. doi: 10.1177/0300060516677155
87. Paşa S, Vardarli AT, Erol N, Karakuş M, Töz S, Atasoy A, et al. Detection of Leishmania major and Leishmania tropica in domestic cats in the Ege Region of Turkey. Vet Parasitol. (2015) 212:389–92. doi: 10.1016/j.vetpar.2015.07.042
88. Can H, Döşkaya M, Özdemir HG, Sahar EA, Karakavuk M, Pektaş B, et al. Seroprevalence of Leishmania infection and molecular detection of Leishmania tropica and Leishmania infantum in stray cats of Izmir, Turkey. Exp Parasitol. (2016) 167:109–14. doi: 10.1016/j.exppara.2016.05.011
89. Morsy TA, Shoura MI. Some aspects of cutaneous leishmaniasis in Riyadh. Saudi Arabia. J Trop Med Hyg. (1976) 79:137–9.
90. Mondragon-Shem K, Al-Salem WS, Kelly-Hope L, Abdeladhim M, Al-Zahrani MH, Valenzuela JG, et al. Severity of old world cutaneous leishmaniasis is influenced by previous exposure to sandfly bites in Saudi Arabia. PLoS Negl Trop Dis. (2015) 9:e0003449. doi: 10.1371/journal.pntd.0003449
91. El-Beshbishy HA, Al-Ali KH, El-Badry AA. Molecular characterization of cutaneous leishmaniasis in Al-Madinah Al-Munawarah province, western Saudi Arabia. Int J Infect Dis. (2013) 17:e334-8. doi: 10.1016/j.ijid.2012.11.015
92. Haouas N, Amer O, Alshammri FF, Al-Shammari S, Remadi L, Ashankyty I. Cutaneous leishmaniasis in northwestern Saudi Arabia: identification of sand fly fauna and parasites. Parasit Vectors. (2017) 10:544. doi: 10.1186/s13071-017-2497-6
93. Shalaby I, Gherbawy Y, Jamjoom M, Banaja AE. Genotypic characterization of cutaneous leishmaniasis at al Baha and Al Qasim provinces (Saudi Arabia). Vector Borne Zoonotic Dis. (2011) 11:807–13. doi: 10.1089/vbz.2010.0213
94. Khan w, Zakai HA. Epidemiology, pathology and treatment of cutaneous leishmaniasis in taif region of Saudi Arabia. Iran J Parasitol. (2014) 9:365–73.
95. Zakai HA. Cutaneous leishmaniasis in Saudi Arabia: current status. J Adv Lab Res Biol. (2014) 5:29–34.
96. Abdalla NM, Abdelgani A, Osman AA, Sarhan MA. Cutaneous Leishmaniasis in El-Madinna Manowra region, Saudi Arabia in 2012. Bangladesh J Med Sci. (2013) 12:325–30. doi: 10.3329/bjms.v12i3.13189
97. Salam N, Al-Shaqha WM, Azzi A. Leishmaniasis in the Middle East: incidence and epidemiology. PLoS Negl Trop Dis. (2014) 8:e3208. doi: 10.1371/journal.pntd.0003208
98. Korzeniewski K, Olszanski R. Leishmaniasis among soldiers of stabilization forces in Iraq. Review article. Int Marit Health. (2004) 55:155–63.
99. Ali MA, Rahi AA, Khamesipour A. Species typing with PCR–RFLP from cutaneous leishmaniasis patients in Iraq. Donnish J Med Med Sci. (2015) 2:026–31.
100. Rahi AA. Genetic characterization of Leishmania species causing cutaneous leishmaniasis in Iraq. Int Res J Eng Technol. (2015) 2:1057–62.
101. Al-Bajalan MM, Al-Jaf SM, Niranji SS, Abdulkareem DR, Al-Kayali KK, Kato H. An outbreak of Leishmania major from an endemic to a non-endemic region posed a public health threat in Iraq from 2014-2017: Epidemiological, molecular and phylogenetic studies. PLoS Negl Trop Dis. (2018) 12:e0006255. doi: 10.1371/journal.pntd.0006255
102. Kolaczinski J, Brooker S, Reyburn H, Rowland M. Epidemiology of anthroponotic cutaneous leishmaniasis in Afghan refugee camps in northwest Pakistan. Trans R Soc Trop Med Hyg. (2004) 98:373–8. doi: 10.1016/j.trstmh.2003.11.003
103. Hussain M, Munir S, Ayaz S, Khattak BU, Khan TA, Muhammad N, et al. First report on molecular characterization of Leishmania species from cutaneous leishmaniasis patients in southern Khyber Pakhtunkhwa province of Pakistan. Asian Pac J Trop Med. (2017) 10:718–21. doi: 10.1016/j.apjtm.2017.07.015
104. Marco JD, Bhutto AM, Soomro FR, Baloch JH, Barroso PA, Kato H, et al. Multilocus enzyme electrophoresis and cytochrome b gene sequencing–based identification of Leishmania isolates from different foci of cutaneous leishmaniasis in Pakistan. Am J Trop Med Hyg. (2006) 75:261–6. doi: 10.4269/ajtmh.2006.75.261
105. Bhutto AM, Soomro FR, Baloch JH, Matsumoto J, Uezato H, Hashiguchi Y, et al. Cutaneous leishmaniasis caused by Leishmania (L.) major infection in Sindh province, Pakistan. Acta trop. (2009) 111:295–8. doi: 10.1016/j.actatropica.2009.05.009
106. Myint CK, Asato Y, Yamamoto YI, Kato H, Bhutto AM, Soomro FR, et al. Polymorphisms of cytochrome b gene in Leishmania parasites and their relation to types of cutaneous leishmaniasis lesions in Pakistan. J Dermatol. (2008) 35:76–85. doi: 10.1111/j.1346-8138.2008.00419.x
107. Ghatee MA, Sharifi I, Kuhls K, Kanannejad Z, Harandi MF, de Almeida ME, et al. Heterogeneity of the internal transcribed spacer region in Leishmania tropica isolates from southern Iran. Exp Parasitol. (2014) 144:44–51. doi: 10.1016/j.exppara.2014.06.003
108. Reithinger R, Mohsen M, Aadil K, Sidiqi M, Erasmus P, Coleman PG. Anthroponotic cutaneous leishmaniasis, Kabul, Afghanistan. Emerg Infect Dis. (2003) 9:727–9. doi: 10.3201/eid0906.030026
109. Faulde M, Schrader J, Heyl G, Amirih M. Differences in transmission seasons as an epidemiological tool for characterization of anthroponotic and zoonotic cutaneous leishmaniasis in northern Afghanistan. Acta Trop. (2008) 105:131–8. doi: 10.1016/j.actatropica.2007.10.011
110. Mosawi SH, Dalimi A. Molecular detection of Leishmania spp. isolated from cutaneous lesions of patients referred to Herat regional hospital, Afghanistan. East Mediterr Health J. (2015) 21:878–84. doi: 10.26719/2015.21.12.878
111. Fakhar M, Karamian M, Ghatee MA, Taylor WR, Ghohe HP, Rasooli SA. Distribution pattern of anthroponotic cutaneous leishmaniasis caused by Leishmania tropica in Western Afghanistan during 2013-2014. Acta Trop. (2017) 176:22–8. doi: 10.1016/j.actatropica.2017.07.028
112. Nadim A, Javadian E, Noushin MK, Nayil AK. Epidemiology of cutaneous leishmaniasis in Afghanistan. Part 2. Anthroponotic cutaneous leishmaniasis. Bull Soc Pathol Exot Filiales. (1979) 72:461–6.
113. Faulde M, Schrader J, Heyl G, Amirih M, Hoerauf A. Zoonotic cutaneous leishmaniasis outbreak in Mazar-e Sharif, northern Afghanistan: an epidemiological evaluation. Int J Med Microbiol. (2008) 298:543–50. doi: 10.1016/j.ijmm.2007.07.015
114. Plourde M, Coelho A, Keynan Y, Larios OE, Ndao M, Ruest A, et al. Genetic polymorphisms and drug susceptibility in four isolates of Leishmania tropica obtained from Canadian soldiers returning from Afghanistan. PLoS Negl Trop Dis. (2012) 6:e1463. doi: 10.1371/journal.pntd.0001463
115. Desjeux P. Leishmaniasis: current situation and new perspectives. Comp Immunol Microbiol Infect Dis. (2004) 27:305–18. doi: 10.1016/j.cimid.2004.03.004
116. Dobrzhanskaia RS, Khuseiinova K, Saf'ianova VM, Emel'ianova LP, Sukhanova TA. The activization of an Ashkhabad focus of anthroponotic cutaneous leishmaniasis. Med Parazitol. (1989) 5:71–4.
117. Larréché S, Launay G, Weibel Galluzzo C, Bousquet A, Eperon G, Pilo JE, et al. Cluster of zoonotic cutaneous leishmaniasis (Leishmania major) in European travelers returning from Turkmenistan. J Travel Med. (2013) 20:400–2. doi: 10.1111/jtm.12065
118. Alvar J, Vélez ID, Bern C, Herrero M, Desjeux P, Cano J, et al. Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE. (2012) 7:e35671. doi: 10.1371/journal.pone.0035671
119. Pratlong F, Dereure J, Ravel C, Lami P, Balard Y, Serres G, et al. Geographical distribution and epidemiological features of Old World cutaneous leishmaniasis foci, based on the isoenzyme analysis of 1048 strains. Trop Med Int Health. (2009) 14:1071–85. doi: 10.1111/j.1365-3156.2009.02336.x
120. Hayani K, Dandashli A, Weisshaar E. Cutaneous leishmaniasis in Syria: clinical features, current status and the effects of war. Acta Derm Venereol. (2015) 95:62–6. doi: 10.2340/00015555-1988
121. Al-Salem WS, Pigott DM, Subramaniam K, Haines LR, Kelly-Hope L, Molyneux DH, et al. Cutaneous leishmaniasis and conflict in Syria. Emerg Infect Dis. (2016) 22:931–3. doi: 10.3201/eid2205.160042
122. Jaber SM, Ibbini JH, Hijjawi NS, Amdar NM. An exploratory comparative study of recent spatial and temporal characteristics of cutaneous leishmaniasis in the Hashemite Kingdom of Jordan and Syrian Arab Republic pre-Arab Spring and their health policy implications. Appl Spat Anal Policy. (2014) 7:337–60. doi: 10.1007/s12061-014-9113-3
123. Jaber SM, Ibbini JH, Hijjawi NS, Amdar NM, Huwail MJ, Al-Aboud K. Exploring recent spatial patterns of cutaneous leishmaniasis and their associations with climate in some countries of the Middle East using geographical information systems. Geospat Health. (2013) 8:143–58. doi: 10.4081/gh.2013.62
124. Alawieh A, Musharrafieh U, Jaber A, Berry A, Ghosn N, Bizri AR. Revisiting leishmaniasis in the time of war: the Syrian conflict and the Lebanese outbreak. Int J Infect Dis. (2014) 29:115–9. doi: 10.1016/j.ijid.2014.04.023
125. Saroufim M, Charafeddine K, Issa G, Khalifeh H, Habib RH, Berry A, et al. Ongoing epidemic of cutaneous leishmaniasis among Syrian refugees, Lebanon. Emerg Infect Dis. (2014) 20:1712–5. doi: 10.3201/eid2010.140288
126. Hotez PJ, Savioli L, Fenwick A. Neglected tropical diseases of the Middle East and North Africa: review of their prevalence, distribution, and opportunities for control. PLoS Negl Trop Dis. (2012) 6:e1475. doi: 10.1371/journal.pntd.0001475
127. Mirahmadi H, Khorashad AS, Sohrabnahad A, Heydarian P, Bizhani N. Species identification and molecular typing of Leishmania spp. using targeting HSP70 gene in suspected patients of cutaneous leishmaniasis from Sistan and Baluchestan Province, Southeast Iran. Iran J Parasitol. (2016) 11:489–98.
128. Hamzavi Y, Nazari N, Khademi N, Hassani K, Bozorgomid A. Cutaneous leishmaniasis in Qasr-e Shirin, a border area in the west of Iran. Vet World. (2018) 11:1692–7. doi: 10.14202/vetworld.2018.1692-1697
129. Ready PD. Leishmaniasis emergence and climate change. Rev Sci Tech. (2008) 27:399–412. doi: 10.20506/rst.27.2.1803
131. Guernaoui S, Boumezzough A, Laamrani A. Altitudinal structuring of sand flies (Diptera: Psychodidae) in the High-Atlas mountains (Morocco) and its relation to the risk of leishmaniasis transmission. Acta Trop. (2006) 97:346–51. doi: 10.1016/j.actatropica.2006.01.001
132. Cross ER, Hyams KC. The potential effect of global warming on the geographic and seasonal distribution of Phlebotomus papatasi in southwest Asia. Environ Health Perspect. (1996) 104:724–7. doi: 10.1289/ehp.96104724
133. Abuzaid AA, Abdoon AM, Aldahan MA, Alzahrani AG, Alhakeem RF, Asiri AM, et al. Cutaneous leishmaniasis in Saudi Arabia: a comprehensive overview. Vector Borne Zoonotic Dis. (2017) 17:673–84. doi: 10.1089/vbz.2017.2119
134. Martín-Sánchez J, Morales-Yuste M, Acedo-Sánchez C, Barón S, Díaz V, Morillas- Márquez F. Canine leishmaniasis in southeastern Spain. Emerg Infect Dis. (2009) 15:795–8. doi: 10.3201/eid1505.080969
135. Ramezankhani R, Sajjadi N, Jozi SA, Shirzadi MR. Climate and environmental factors affecting the incidence of cutaneous leishmaniasis in Isfahan, Iran. Environ Sci Pollut Res Int. (2018) 25:11516–26. doi: 10.1007/s11356-018-1340-8
136. Shirzadi MR, Mollalo A, Yaghoobi-Ershadi MR. Dynamic relations between incidence of zoonotic cutaneous leishmaniasis and climatic factors in Golestan Province, Iran. J Arthropod Borne Dis. (2015) 9:148–60.
137. Sharafi M, Ghaem H, Tabatabaee HR, Faramarzi H. Forecasting the number of zoonotic cutaneous leishmaniasis cases in south of Fars province, Iran using seasonal ARIMA time series method. Asian Pac J Trop Med. (2017) 10:79–86. doi: 10.1016/j.apjtm.2016.12.007
138. Yaghoobi-Ershadi MR, Akhavan AA, Zahraei-Ramazani AR, Jalali-Zand AR, Piazak N. Bionomics of Phlebotomus papatasi (Diptera: Psychodidae) in an endemic focus of zoonotic cutaneous leishmaniasis in central Iran. J Vector Ecol. (2005) 30:115–8.
139. Ready PD. Biology of phlebotomine sand flies as vectors of disease agents. Annu Rev Entomol. (2013) 58:227–50. doi: 10.1146/annurev-ento-120811-153557
140. Du R, Hotez PJ, Al-Salem WS, Acosta-Serrano A. Old world cutaneous leishmaniasis and refugee crises in the Middle East and North Africa. PLoS Negl Trop Dis. (2016) 10:e0004545. doi: 10.1371/journal.pntd.0004545
141. Alvar J, Yactayo S, Bern C. Leishmaniasis and poverty. Trends Parasitol. (2006) 22:552–7. doi: 10.1016/j.pt.2006.09.004
Keywords: Leishmania tropica, Leishmania major, Iran, Turkey, Iraq, Afghanistan, Saudi Arabia, Syria
Citation: Ghatee MA, Taylor WR and Karamian M (2020) The Geographical Distribution of Cutaneous Leishmaniasis Causative Agents in Iran and Its Neighboring Countries, A Review. Front. Public Health 8:11. doi: 10.3389/fpubh.2020.00011
Received: 14 August 2019; Accepted: 14 January 2020;
Published: 18 February 2020.
Edited by:
Herbert Leonel de Matos Guedes, Federal University of Rio de Janeiro, BrazilReviewed by:
Valter Viana Andrade-Neto, Oswaldo Cruz Foundation (Fiocruz), BrazilCopyright © 2020 Ghatee, Taylor and Karamian. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mehdi Karamian, a2FyYW1pYW5tQHlhaG9vLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.