
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Psychiatry , 01 April 2025
Sec. Public Mental Health
Volume 16 - 2025 | https://doi.org/10.3389/fpsyt.2025.1555513
This article is part of the Research Topic The Intersection of Psychology, Healthy Behaviors, and its Outcomes View all 79 articles
 Liming Tang*
Liming Tang* Jinrong Zhong
Jinrong Zhong Mei’e Zeng
Mei’e Zeng Weiwei Deng
Weiwei Deng Chunmei Huang
Chunmei Huang Shuifen Ye
Shuifen Ye Fengjin Li
Fengjin Li Dongqin Lai
Dongqin Lai Wanling Huang
Wanling Huang Bin Chen
Bin Chen Xiaoyuan Deng
Xiaoyuan Deng Xiaoying Lai
Xiaoying Lai Lirong Wu
Lirong Wu Bilan Zou
Bilan Zou Hanzhong Qiu*
Hanzhong Qiu* Ying Liao*
Ying Liao*Background: Patients with somatization symptoms are at elevated risk of depression, yet underdiagnosis persists due to cultural tendencies (e.g., in China) to express psychological distress via physical complaints. Existing predictive models lack integration of sociocultural and physiological factors, particularly in non-Western populations.
Objective: To develop a culturally tailored risk-prediction model for depression in patients with somatization symptoms, emphasizing early identification and personalized intervention.
Methods: A prospective cohort study included 200 somatization patients (SSS≥38, PHQ-2<3) from a Chinese hospital (May 2020–August 2022). LASSO regression identified predictors from 18 variables, followed by multivariate logistic regression to construct a nomogram. Model performance was assessed via ROC-AUC, calibration curves, Hosmer-Lemeshow test, and decision curve analysis (DCA). Internal validation used 200 bootstrap resamples.
Results: Five independent predictors were identified: advanced age (OR=1.11, 95% CI: 1.02–1.20), poor self-rated health (OR=2.07, 95% CI: 1.04–4.30), lack of co-residence with children (OR=1.63, 95% CI: 1.10–2.42), low income (OR=1.45, 95% CI: 1.05–2.01), and self-medication (OR=1.32, 95% CI: 1.01–1.73). The nomogram demonstrated strong discrimination (AUC=0.810, 95% CI: 0.728–0.893) and calibration (Hosmer-Lemeshow p=0.32). DCA confirmed clinical utility: at threshold probabilities >5%, the model provided higher net benefit than “treat-all” or “treat-none” strategies.
Conclusion: This model integrates sociocultural (e.g., family structure) and behavioral factors to predict depression risk in somatizing patients, particularly in East Asian contexts. It offers a practical tool for clinicians to prioritize high-risk individuals, reducing diagnostic delays and healthcare burdens. Future multicenter studies should validate its generalizability and incorporate biomarkers (e.g., inflammatory markers) to enhance mechanistic insights.
Somatization, a prevalent phenomenon in healthcare, refers to the presentation of physical symptoms that cannot be explained by known medical conditions or physiological abnormalities. These symptoms are often manifestations of underlying psychological distress, such as anxiety and depression. Common somatic complaints include palpitations, non-cardiac chest pain, dyspnea, gastrointestinal disturbances (e.g., irritable bowel syndrome, altered bowel habits), chronic fatigue, and diffuse musculoskeletal pain (1, 2). Globally, approximately 16–25% of individuals in primary care settings report somatic symptoms severe enough to meet criteria for somatization disorders, with higher prevalence in populations where cultural norms discourage direct expression of emotional distress (3). Patients exhibiting somatic symptoms frequently seek recurrent medical care, contributing to a significant healthcare burden. However, the inability to identify organic causes often complicates diagnosis and treatment, delaying appropriate psychological interventions (1, 4).
The relationship between Somatization and depression is well-documented, with studies confirming that individuals with depressive disorders are likely to experience somatic symptoms (5, 6). Beyond the epidemiological associations, there are complex pathophysiological mechanisms linking somatization symptoms and depression. At the neurotransmitter level, alterations in the serotonin and dopamine systems play crucial roles. Serotonin is involved in regulating mood, sleep, and pain perception. In patients with both somatization symptoms and depression, serotonin levels are often decreased, which can lead to mood disturbances and enhanced pain sensitivity, contributing to the manifestation of somatic symptoms. Dopamine, on the other hand, is related to motivation and reward. Imbalances in dopamine can cause anhedonia, a common symptom in depression, and may also be associated with the development of somatic complaints.
The hypothalamic - pituitary - adrenal (HPA) axis, a key component of the neuroendocrine system, is also dysregulated in these patients. Chronic stress, which is often associated with depression, can over - activate the HPA axis, leading to increased cortisol secretion. Elevated cortisol levels can have widespread effects on the body, including inflammation and immune system dysregulation. Inflammation has been linked to the development of somatic symptoms such as fatigue, muscle pain, and cognitive impairment, further blurring the line between physical and psychological symptoms. Furthermore, emerging evidence from neuroimaging studies shows structural and functional changes in the brains of patients with somatization symptoms and depression. Regions such as the prefrontal cortex, amygdala, and hippocampus, which are involved in emotion regulation, stress response, and memory, exhibit abnormal activity and connectivity. These changes may contribute to the complex interplay between psychological distress and the manifestation of somatic symptoms.
In China, cultural norms and expectations profoundly influence the manifestation and expression of psychological distress. Depression is frequently communicated through somatic complaints rather than direct emotional expressions, largely due to the persistent stigma surrounding mental illness. Patients often present to primary care physicians with persistent physical symptoms despite the absence of identifiable organic causes, a phenomenon known as cultural somatization (7, 8). This pattern has been extensively documented, revealing that Chinese patients are more likely to seek medical attention for physical ailments while minimizing or concealing emotional symptoms (1, 5). Empirical studies indicate that approximately 18.2% of outpatients in general hospitals report somatic complaints that meet diagnostic criteria for somatization disorders, highlighting the critical need for improved diagnostic frameworks in clinical practice (9).
Despite the well-documented association between somatic symptoms and depression, there remains a significant gap in research examining the predictive factors for depression among patients with somatization, both within China and globally (5, 6). Early identification and intervention are paramount, as untreated depression associated with somatic symptoms not only exacerbates the duration and severity of the illness but also increases the risk of adverse outcomes, including suicidal behavior (10).
In this context, the present study aims to develop a predictive risk model for depression in patients presenting with somatic symptoms. Guided by the methodological framework proposed by Collins et al. (11) for the development, validation, and updating of clinical prediction models, this research will identify independent predictors of depression among somatizing patients. By establishing a robust and clinically applicable risk assessment tool, this study seeks to facilitate early diagnosis, timely intervention, and improved clinical outcomes. The ultimate goal is to provide a personalized, evidence-based approach to risk stratification, enabling targeted treatment strategies, reducing unnecessary healthcare utilization, and enhancing long-term patient prognosis (10, 11).
All patients enrolled were tracked throughout the 3, 6, and 12-month follow-up periods. No patients were lost to follow-up, ensuring the completeness of the dataset.
Population selection: Initially, a list of all patients admitted to the Department of General Medicine at xx Hospital from May 2020 to August 2022 was obtained. After excluding patients with incomplete medical records, those who had fully completed relevant examinations but still could not have their physical discomfort explained by known physiological or medical knowledge were identified. These patients were then evaluated using the Somatic Self Rating Scale (SSS) and PHQ-2. Only patients with SSS scores ≥ 38 (12) and PHQ - 2 scores<3 were included in the study. This step - by - step screening process ensured that the selected patients had significant somatization symptoms without current depressive symptoms, which was crucial for the development of the predictive model for depression risk.
Exclusion criteria: History of mental illness, paralysis, epilepsy, dementia, cognitive impairment, organ dysfunction, and other medical conditions.
This project uses SSS and PHQ-2 and subsequent PHQ-9 scales to evaluate Somatization symptoms and depressive status and conducts a self-made general questionnaire survey to screen patients with Somatization symptoms (SSS score ≥ 38) and non-depressive status (PHQ-2<3). Relevant factors are evaluated, and PHQ-2 questionnaires are used for 3-month, 6-month, and 12-month follow-ups to evaluate PHQ-2 and subsequent PHQ-9 scores and assess the patient’s depressive status.
Life satisfaction: Measured using a 5-point Likert scale assessing general contentment with life. For ease of analysis, a score of 1-3 is considered dissatisfied, and 4-5 is considered satisfied. Self-health assessment: Patients rated their health on a scale from 1 (excellent) to 5 (worst). For ease of analysis, a score of 1-3 is considered poor, and 4-5 is considered good.
The Patient Health Questionnaire-2 (PHQ-2) was selected as a preliminary screening instrument due to its brevity and ease of administration, which minimizes patient burden in hospital settings. Although not a diagnostic tool, it is widely recognized for its validity as an initial screening measure for depressive symptoms.
The Somatic Symptom Scale (SSS) was employed to assess the somatic symptoms of patients (13). This scale, developed by Mao Jialiang from the Department of Cardiology at Renji Hospital in Shanghai (4th edition), demonstrates strong psychometric properties with a Cronbach’s α coefficient of 0.89 and a reliability coefficient of 0.96. The scale comprises 20 items, each rated on a 4-point Likert scale without reverse scoring. The scoring system is as follows: “1” indicates “no symptoms,” “2” indicates “mild degree,” “3” indicates “moderate degree,” and “4” indicates “severe degree.” Interpretation of SSS scores is as follows: scores <30 indicate no psychological or emotional issues requiring treatment; scores ≥30 and <38 suggest potential psychological and emotional problems, warranting psychological counseling; scores ≥38 and <42 indicate moderate psychological and emotional problems, recommending pharmacological treatment; and scores ≥42 signify severe psychological and emotional problems, necessitating combined pharmacological treatment (12).
The PHQ-2 and PHQ-9 instruments were utilized to screen patients for depressive disorders (9). The PHQ-2 scale consists of two items assessing the following symptoms over the past two weeks: (1) anhedonia or diminished interest in activities, and (2) depressed mood or feelings of hopelessness. Each item is scored on a 4-point scale: “not at all” (0 points), “several days” (1 point), “more than half the days” (2 points), and “nearly every day” (3 points). The total score ranges from 0 to 6, with a cutoff score of 3 indicating potential depression. Patients who screened positive on the PHQ-2 were subsequently administered the PHQ-9, which served as the diagnostic criterion for depression.
A self-administered general questionnaire was developed to collect comprehensive demographic and psychosocial information, including social demographics, marital status, family background, economic status, chronic medical conditions, social support systems, and life satisfaction indicators (9).
While a formal prospective power analysis was not conducted due to the exploratory nature of this prediction model study, we ensured methodological rigor through alternative approaches: Sample Size Justification: Based on recommendations for prediction models requiring at least 10 events per predictor variable (EPV), our model included 5 predictors with 34 depression events (6.8 EPV), meeting the minimum threshold for stable estimation. Post-Hoc Power Analysis: Using the pROC package in R, a post-hoc power calculation based on the observed AUC (0.81) and sample size (n=200) indicated >80% power to detect an AUC significantly greater than 0.70 (null hypothesis) at α=0.05. Internal Validation: Bootstrap resampling (200 iterations) yielded a narrow optimism-corrected AUC confidence interval (0.728–0.893), confirming robust discrimination despite the moderate sample size.
Statistical analyses were performed using SPSS 26.0 and R 4.2 software. Continuous data that did not follow a normal distribution were expressed as medians with interquartile ranges (IQR). Group comparisons were conducted using independent two-sample t-tests.
Variable selection was performed using LASSO (Least Absolute Shrinkage and Selection Operator) regression, followed by the development of a predictive model through multivariate logistic regression analysis, which was subsequently visualized using a nomogram. Prior to LASSO regression, variables exhibiting a variance inflation factor (VIF) exceeding 5 were comprehensively evaluated to address multicollinearity. To further mitigate overfitting, a 10-fold cross-validation approach was employed during LASSO regression to optimize the penalty parameter (λ). Internal validation of the final logistic regression model was conducted using 200 bootstrap resamples to calculate optimism-adjusted performance metrics.
The predictive model was evaluated based on three key aspects: discrimination, calibration, and clinical utility. Discrimination was assessed using the area under the receiver operating characteristic curve (ROC-AUC). Calibration was evaluated using calibration curves and the Hosmer-Lemeshow goodness-of-fit test. Clinical utility was examined using Decision Curve Analysis (DCA) to determine the net benefit across different threshold probabilities. A p-value of less than 0.05 was considered statistically significant.
This study was conducted in accordance with the tenets of the Declaration of Helsinki. The procedures adopted were in accordance with the ethical standards formulated by the Ethics Committee of Longyan First Hospital, ethics approval number LYREC2022-006-01. Written informed consent will be obtained from all participants or their legal guardians before the study.
The study included 200 patients aged between 30 and 97 years, with a mean age of 67.77 ± 12.11 years. The cohort consisted of 110 males (55%) and 90 females (45%). Among the participants, 34 patients (17%) were diagnosed with depression. The mean Somatic Symptom Scale (SSS) score was 34.85 ± 8.31. Detailed demographic and clinical characteristics of the enrolled patients are presented in Table 1.
LASSO regression analysis was employed to screen predictive variables from 18 candidate variables, including general social information, family status, economic status, chronic disease history, and life satisfaction. At the optimal penalty parameter (λ = 0.038), five non-zero coefficient predictive variables were identified (Figures 1, 2). The selected variables included age, income, self-rated health status, cohabitation with children, and self-medication practices. The LASSO regression model demonstrated optimal performance at this λ value.
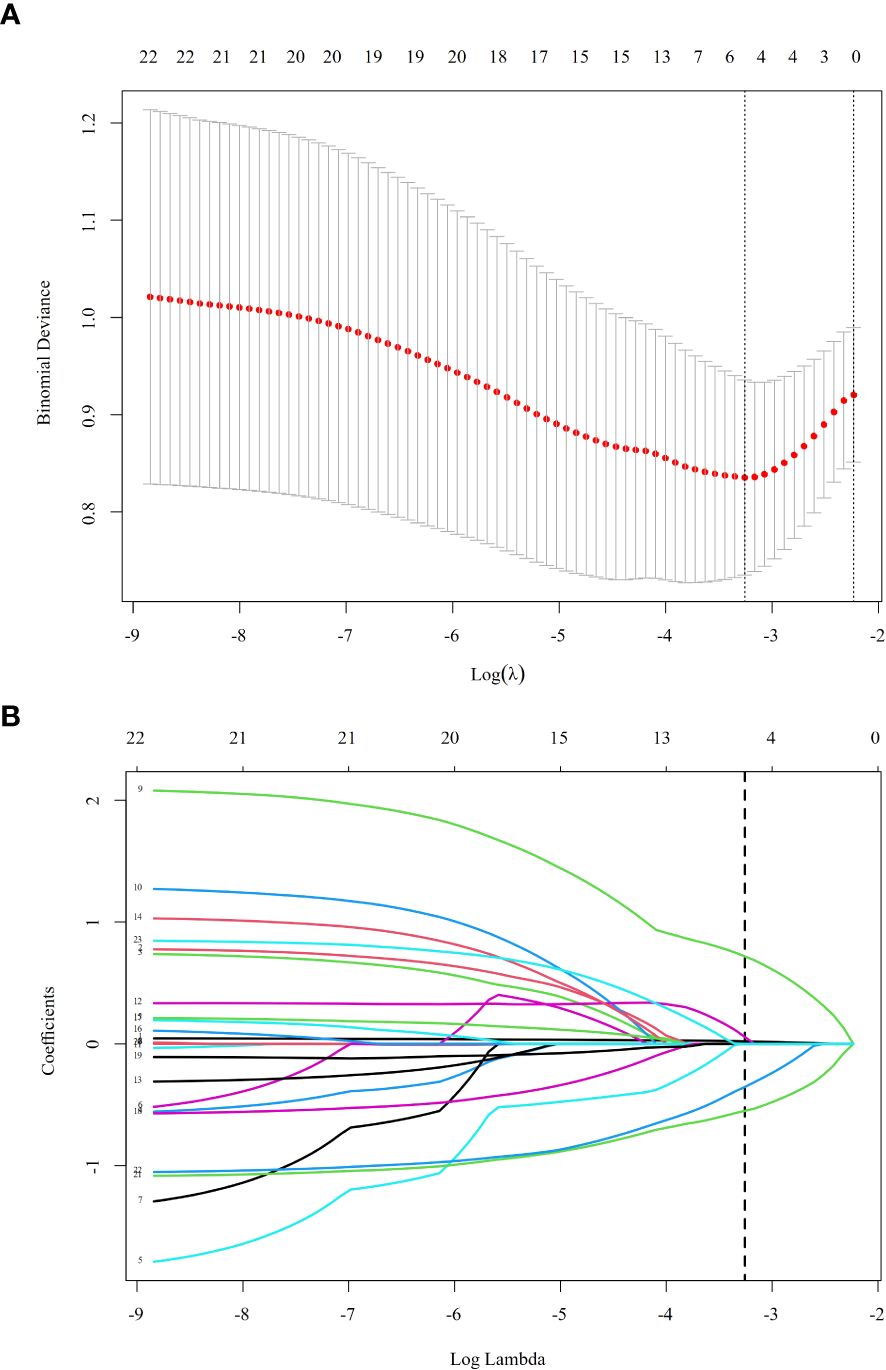
Figure 1. Using the LASSO regression model to screen predictive variables. (A) is the LASSO coefficient curve for 19 variables, and (B) is the process of screening λ through 10-fold cross-validation in the LASSO model. LASSO regression helps reduce the risk of overfitting by penalizing complex models and selecting only the most predictive variables for depression risk.
A multivariate logistic regression model was constructed to predict depression in patients with somatization symptoms. The dependent variable was the presence of depression, while the independent variables were the five predictors identified through LASSO regression analysis (Figure 3). The results indicated that self-rated health status, age, self-medication practices, and cohabitation with children were significant risk factors for depression in patients with somatization symptoms (p < 0.05), as detailed in Table 2.
A nomogram was developed based on the predictive variables to facilitate clinical application (Figure 4). The nomogram allows clinicians to assign scores to each variable based on patient-specific values, sum the scores, and determine the corresponding risk of depression by drawing a vertical line from the total score axis.
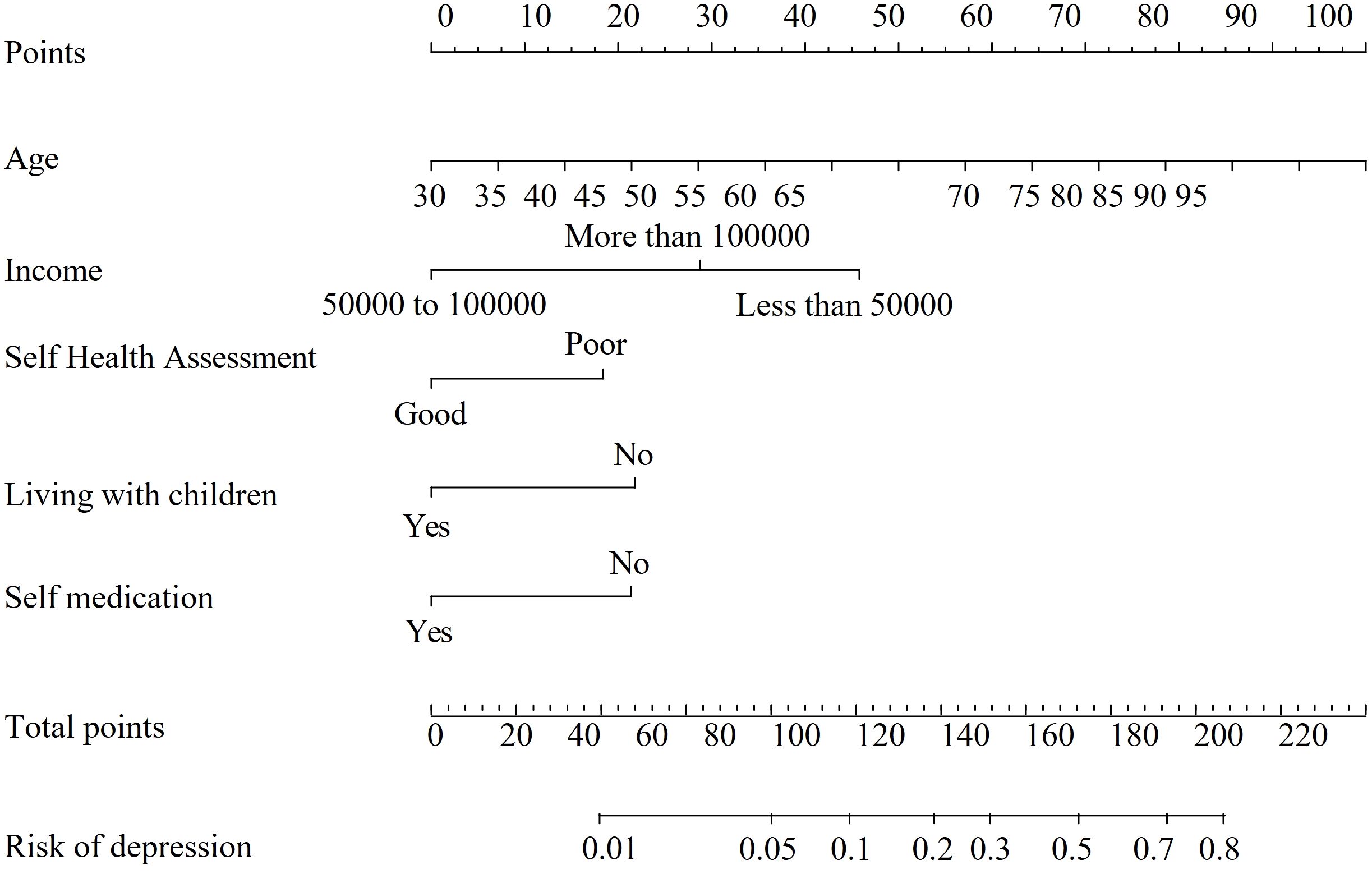
Figure 4. ROC curve of the predictive model for predicting depression in patients with somatization. Bootstrap resampling was performed 200 times, yielding an AUC of 0.795 (95% CI: 0.718-0.88). This curve demonstrates the trade-off between sensitivity and specificity, with higher AUC values indicating better model performance.
1. Locate the patient’s value for each predictor variable on the corresponding axis.
2. Draw a vertical line to the points axis to determine the score for each variable.
3. Sum the scores for all predictors.
4. Draw a vertical line from the total score axis to the risk axis to estimate the probability of depression.
We then performed subgroup analyses to explore whether there was an interaction in the inclusion measures. In the subgroup analysis, there was no significant interaction between income level and age and depression risk (P for interaction = 0.272). Specifically, age was significantly associated with depression risk in people with incomes below $50,000 (OR = 1.11, 95% CI: 1.02-1 20, P = 0.015), but not in people with incomes between $50,000 and $100,000 and above $100,000 (P = 0.064 and 0.221, respectively). There was no significant interaction between age and depression risk in self-assessed health (P for interaction = 0.809), and age was significantly associated with depression risk in the self-assessed healthy population (OR = 1.07, 95% CI: 1.03-1, P = 0.002), but not in the self-assessed healthy population (P = 0.076). Self-medication did not have a significant interaction with age and depression risk (P for interaction = 0.144), and age was significantly associated with depression risk in the non-self-medication population (OR = 1.09, 95% CI: 1.03-1, P = 0.002), but not in the self-medication population (P = 0.312). Living with children did not have a significant interaction between age and depression risk (P for interaction = 0.596). Among those living without children, age was significantly associated with depression risk (OR = 1.08, 95% CI: 1.01-1, P = 0.028), while among those living with children, the association was nearly significant (P = 0.053). The results of the subgroup analysis are shown in Table 3.
Discrimination and calibration of the predictive model are the primary means of validation. Discrimination of the model is assessed by plotting ROC curves for the prediction of depressive states in patients with Somatization symptoms. The sample size of this study is 200 cases, not grouped, and all data are used as a modeling queue. The AUC of the predictive model is 0.810 (95% CI: 0.728 - 0.893), indicating good discrimination of the model (Figure 5). In this series, there was good consistency in the calibration curve of the depression risk nomogram for patients with Somatization symptoms (Figure 6).
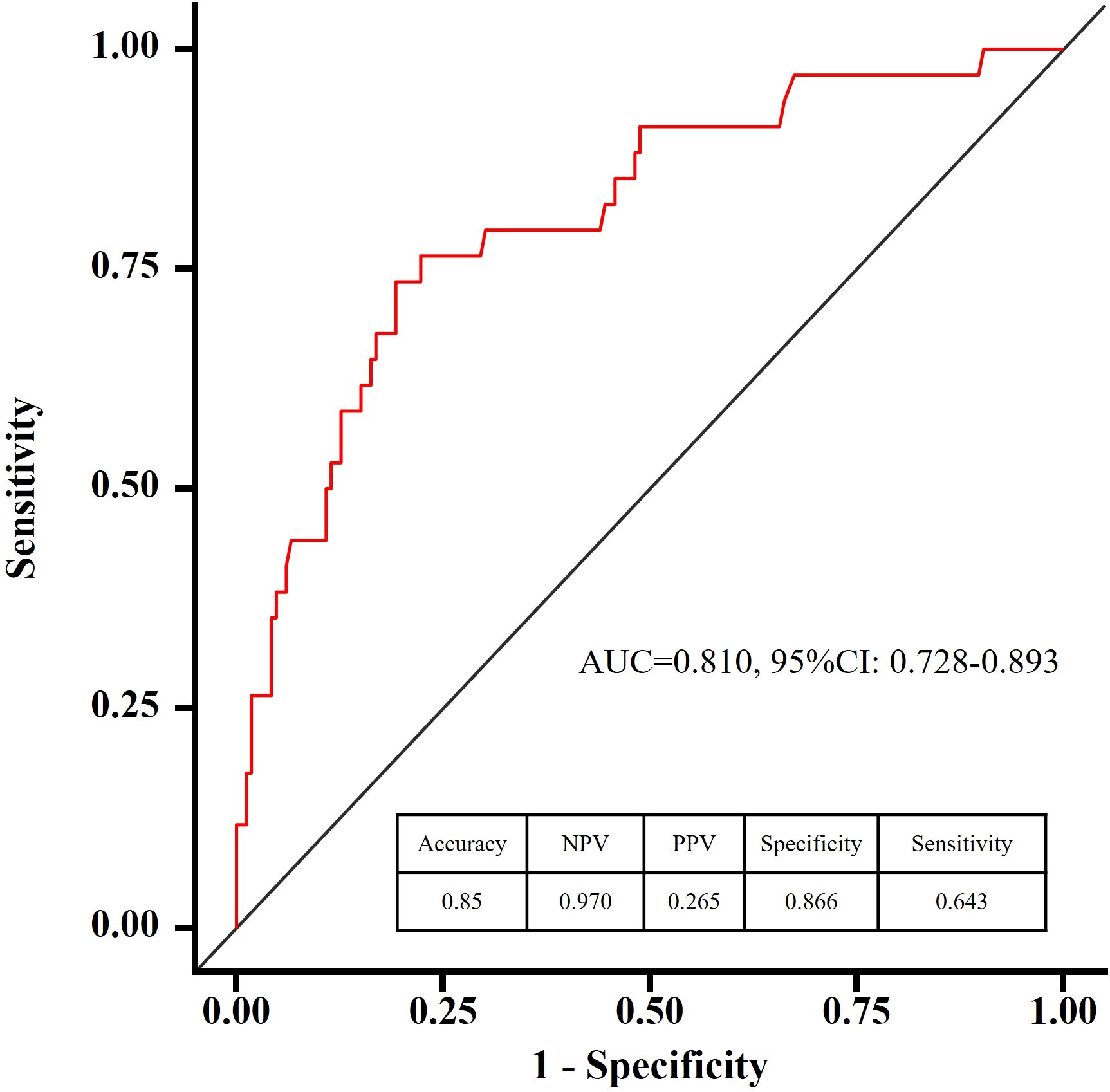
Figure 5. Calibration curve of the prediction model. Bootstrap resampling was performed 1000 times to validate calibration. This curve compares predicted versus observed probabilities. A model with perfect calibration will have predictions close to the diagonal line, indicating good agreement between predicted and actual risks.
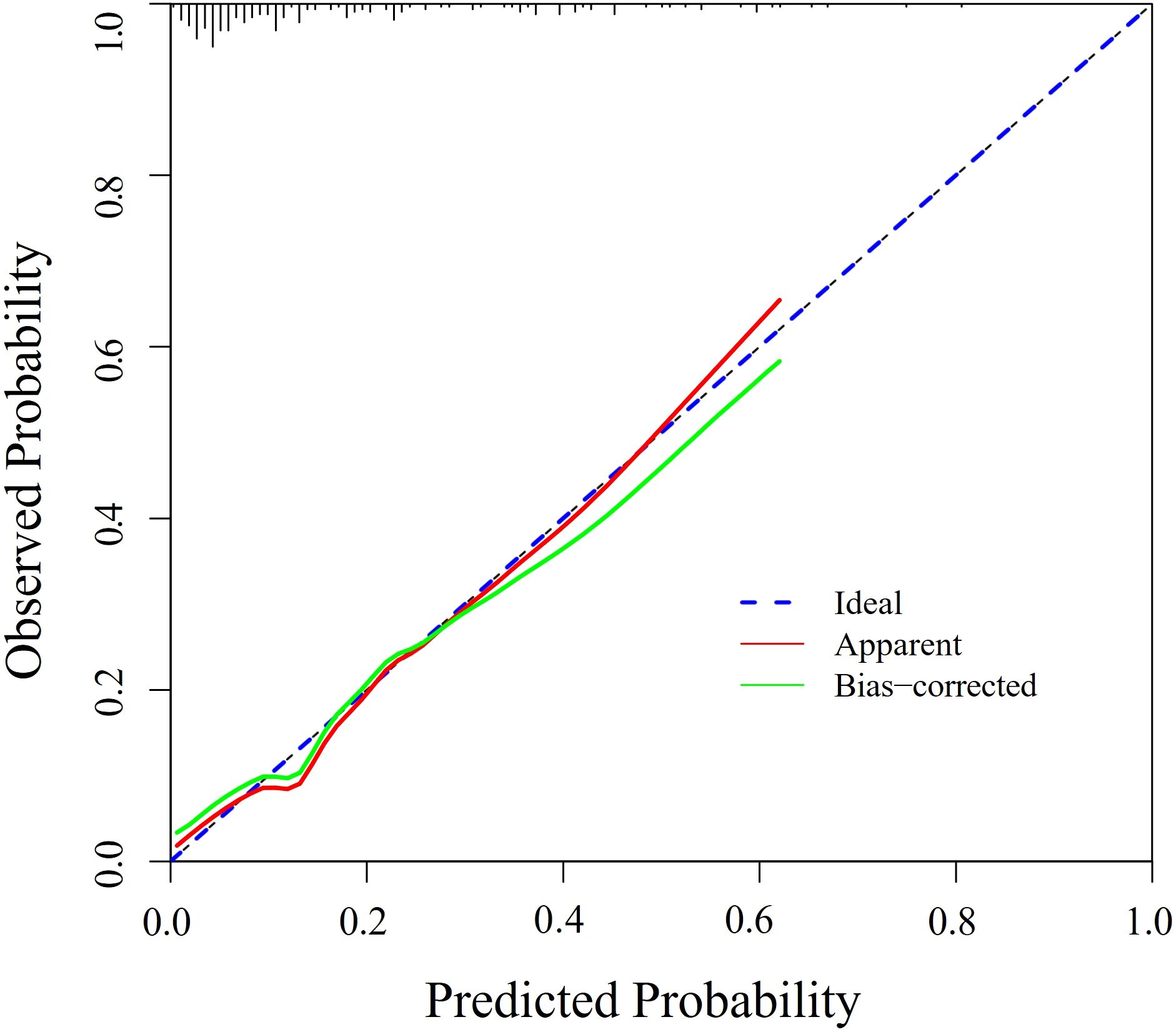
Figure 6. DCA analysis of the prediction model. Bootstrap resampling was performed 1000 times with 10-fold cross-validation. Decision Curve Analysis (DCA) illustrates the net clinical benefit of applying the model at different threshold probabilities. A higher net benefit across a range of thresholds indicates the model’s utility in clinical decision-making.
These findings indicate that the model demonstrates reasonable goodness of fit, with predicted probabilities closely aligning with observed probabilities, suggesting excellent calibration. In summary, the prediction model exhibits moderate predictive capability.
Figure 7 presents the decision curve analysis (DCA) for assessing the risk of depression in patients with somatization symptoms. DCA is a clinically relevant method for evaluating the net benefit and clinical utility of predictive models. The analysis revealed that when the threshold probabilities for both patients and clinicians exceed 5%, using this nomogram to predict depression risk provides greater clinical benefit compared to implementing intervention plans for all patients. Within this threshold range, the net benefit of the prediction model significantly surpasses that of the two extreme scenarios (intervening for all patients or no patients). This underscores the model’s potential to guide targeted clinical decision-making, optimizing resource allocation and improving patient outcomes.
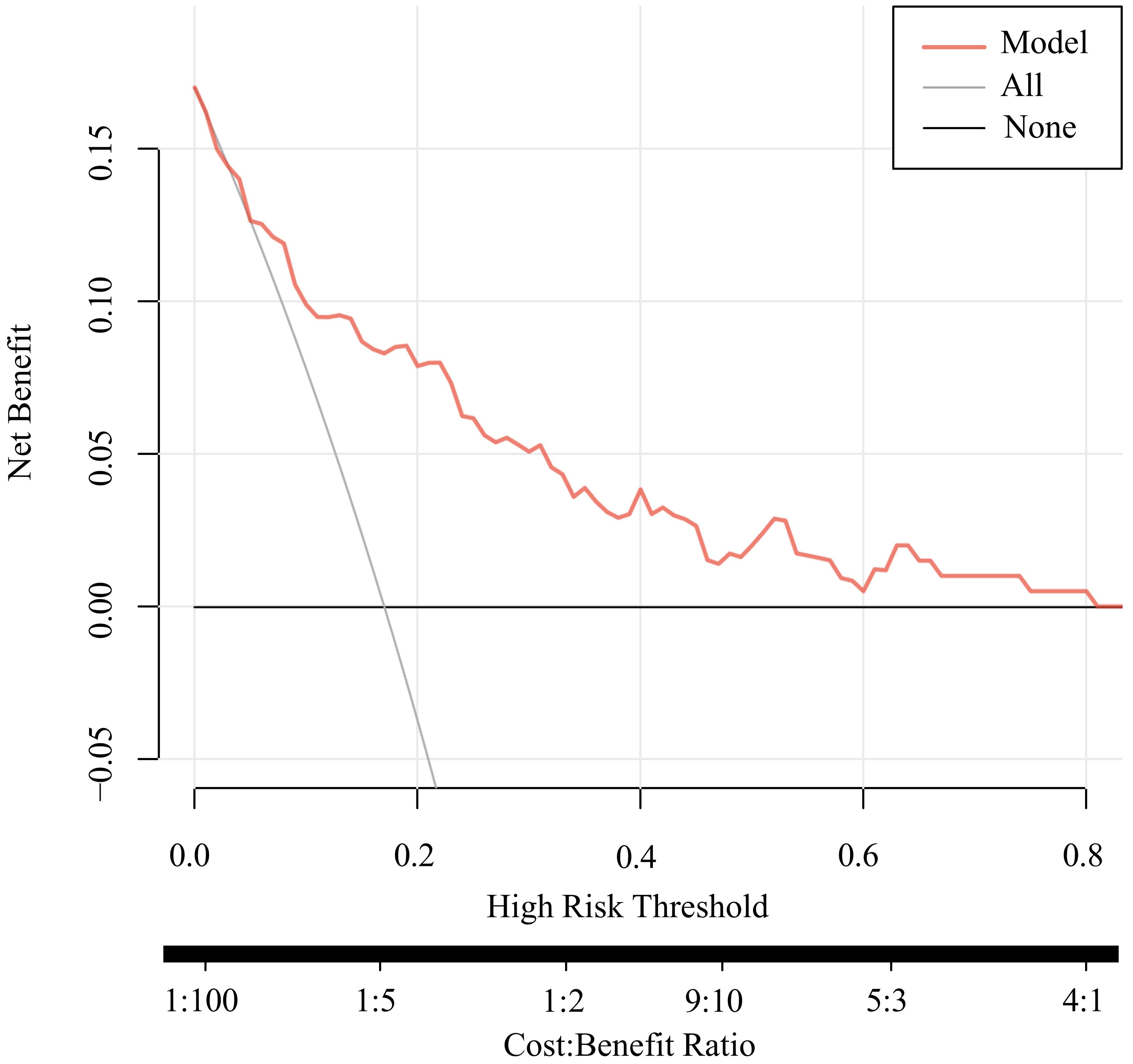
Figure 7. Line chart of the predictive model for the risk of depression in patients with Somatization symptoms. Self-health evaluation: 1 (good), 2 (average), 3 (poor); Living with children: 1 (Yes), 0 (No); Self-medication: 1 (Yes), 0 (No). To use the nomogram, identify the patient’s values on each predictor axis. Draw a vertical line to the points axis to assign a score. Sum the scores for all predictors and draw a vertical line from the total score axis to determine the corresponding risk of depression.
The study findings reveal that cohabitation with children and positive self-rated health evaluations serve as protective factors against depression, consistent with intergenerational studies highlighting the role of family support in mental health (14). Additionally, age-related physical decline significantly increases vulnerability to depression (15). These results underscore the importance of addressing both physical and social determinants to mitigate depression risk in patients with somatic symptoms (14–17).
Nomograms are widely recognized as a reliable and practical tool in clinical research for disease prediction, particularly in oncology and chronic disease management (18, 19). By analyzing risk factors associated with disease onset, progression, and prognosis, nomograms provide an intuitive and user-friendly interface for predicting disease probability, aiding clinicians in making informed decisions (20, 21).
In the developmental cohort of this study, 34 patients (17%) with somatic symptoms were diagnosed with depression. LASSO regression identified seven independent risk factors for depression, including self-rated health, memory decline, age, recent social status, annual family income, self-medication practices, and cohabitation with children. Multivariate logistic regression analysis confirmed that self-rated health, age, self-medication, and cohabitation with children were significant predictors of depression in patients with somatization symptoms (p < 0.05). The bootstrap-corrected area under the curve (AUC) for the nomogram in the training set was 0.810 (95% CI: 0.728–0.893), indicating strong discriminatory power. Furthermore, the Hosmer-Lemeshow goodness-of-fit test demonstrated excellent calibration, with predicted risks closely aligning with observed risks.
This predictive model highlights that improving self-rated health, fostering cohabitation with children, and addressing self-medication practices can reduce depression risk in patients with somatization symptoms, while advancing age exacerbates this risk. These findings align with prior studies in primary care and outpatient settings (22–24).
Existing literature suggests that aging increases depression risk due to factors such as multimorbidity, declining physical and cognitive function, and reduced socioeconomic status (16). Our study corroborates that older individuals with somatic symptoms face a higher risk of depression, likely due to physical frailty, increased dependency, social isolation, and financial insecurity (25–27). As China undergoes rapid population aging, the burden of depression among the elderly is expected to rise. Proactive prevention and intervention strategies are urgently needed to address this growing public health challenge.
Our study found that patients with somatic symptoms with low self-health evaluations had higher risk of developing depression compared to patients with high self-health evaluations. A study found that poor self-health assessment is a risk factor for developing depressive states in individuals aged 65 and above (28). As age increases, physiological functions gradually decline, self-care abilities decrease, and dependence increases, all of which may lead to poor self-health evaluations in the elderly. These factors have been reported as risk factors for the onset of depression in the elderly in other studies (16). When daily activities are restricted due to joint pain and prolonged sitting, physical function is impaired, and activity is restricted, they have a lower evaluation of their health, which is related to their occurrence of depression (29).
Research indicates that housing type does not directly influence the occurrence of depressive symptoms in older adults, whereas intergenerational support serves as a protective factor against depression (14). This suggests that while family living arrangements alone may not significantly impact depression in the elderly, enhancing the level of intergenerational support and improving older adults’ perceptions of such support are critical (14). The findings of this study further demonstrate that cohabitation with children is a protective factor against depression in patients with somatization symptoms. This may be attributed to the cultural context of the study population. In the Longyan region, a significant proportion of the population resides in rural and township areas, where multigenerational households are common. Studies have shown that such living arrangements facilitate greater emotional communication between older adults and their children or grandchildren, providing stronger family support (24). Higher levels of family support are associated with a lower incidence of depressive symptoms among older adults in China (30).
Patients often prioritize reporting physical symptoms over psychological concerns when consulting non-psychiatrists, as somatic symptoms can obscure the core manifestations of depression, thereby delaying its diagnosis (10, 31, 32). Our findings highlight the importance of alerting non-psychiatrists to the potential presence of mental health issues in patients presenting with somatization symptoms. Specifically, patients with somatization symptoms who report poor self-rated health, are older, lack self-treatment capabilities, and do not live with their children are at higher risk of depression. It is crucial to recognize that the severity of somatic symptoms does not necessarily reflect the severity of the underlying disease.
Existing research has documented the efficacy of pharmacological interventions (33) and psychological therapies (25, 26, 34, 35) for somatization symptoms. Accurate identification of these patients can significantly alleviate individual suffering and reduce the broader societal burden of disease. Clinicians should therefore adopt a holistic approach, integrating both physical and psychological assessments, to ensure timely and effective interventions for this vulnerable population.
The predictive model in this study primarily identified the contribution of sociobehavioral factors (e.g., self-health assessments, living with children) to depression risk, but acknowledged that the physiological basis of somatization symptoms (e.g., immune activation, neuroendocrine disorders) may indirectly affect outcomes through unmeasured pathways. For example, chronic inflammatory states may simultaneously cause physical discomfort (e.g., joint pain, fatigue) and low mood (36), while social support (e.g., living with children) may buffer such physiological risks by modulating stress responses (e.g., lowering cortisol levels) (37). Future studies should combine biomarkers (e.g. IL-6, CRP) and psychosocial variables to uncover the multilevel mechanisms underlying the somatization-depression relationship. Future research needs to obtain more comprehensive biopsychosocial data through multicenter collaboration to validate and extend the explanatory power of this model.
In clinical applications, it is important to note that, first, the protective effect of “living with children” may be closely related to traditional Chinese family values (e.g., filial piety, intergenerational support) (14); second, somatization as an expression of psychological symptoms is more prevalent in East Asian cultures (10), while Western patients may report emotional symptoms more directly. Therefore, caution is required when applying this model directly to Western populations, and it is recommended to adjust variable definitions (e.g., replacing “living with children” with “social support network density”) and perform cross-cultural calibration.
It should be noted that this study has certain limitations. The data used were from a single hospital in China, which may lead to bias in patient characteristics due to regional differences, different medical service levels, and unique cultural backgrounds. As a result, the generalizability of our findings may be limited. Future studies could consider a multi - center design to overcome these limitations and obtain more comprehensive and representative results. This would enhance the applicability of the predictive model in diverse healthcare settings. Another major limitation of this study is related to the assessment of subliminal depressive status evaluated by PHQ - 2 and subsequent PHQ-9. The MINI (Mini-International Neuropsychiatric Interview), was better, but it is not used because it is not convenient enough.
This study developed a personalized risk prediction model incorporating four key variables: self-rated health, age, self-treatment practices, and cohabitation with children. The model demonstrates robust predictive performance and clinical utility, as evidenced by its ROC curve, calibration curve, and decision curve analysis (DCA). By leveraging these factors, healthcare providers can make evidence-based decisions to identify and mitigate depression risk in patients with somatic symptoms. This approach not only enhances patient outcomes but also alleviates the individual and systemic burden on healthcare resources.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Ethics Committee of Longyan First Hospital, Longyan, China. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
LT: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. JZ: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. M’EZ: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. WD: Conceptualization, Data curation, Formal Analysis, Methodology, Resources, Software, Validation, Writing – original draft, Writing – review & editing. CH: Conceptualization, Data curation, Investigation, Methodology, Project administration, Resources, Validation, Writing – original draft, Writing – review & editing. SY: Conceptualization, Data curation, Methodology, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. FL: Data curation, Methodology, Resources, Software, Writing – original draft, Writing – review & editing, Conceptualization. DL: Conceptualization, Data curation, Formal Analysis, Methodology, Resources, Writing – original draft, Writing – review & editing. WH: Conceptualization, Formal Analysis, Methodology, Software, Validation, Writing – original draft, Writing – review & editing. BC: Conceptualization, Data curation, Investigation, Software, Writing – original draft, Writing – review & editing. XD: Data curation, Investigation, Project administration, Validation, Writing – original draft, Writing – review & editing. XL: Data curation, Formal Analysis, Methodology, Software, Writing – original draft, Writing – review & editing. LW: Data curation, Resources, Validation, Writing – original draft, Writing – review & editing. BZ: Data curation, Investigation, Software, Writing – original draft, Writing – review & editing. HQ: Data curation, Investigation, Validation, Writing – original draft, Writing – review & editing. YL: Data curation, Investigation, Validation, Writing – original draft, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study is funded by two research projects: 1. Sponsored by Longyan City Science and Technology Plan Project in 2021(Grant number 2021LYF17030). 2. Startup Fund for scientific research, Fujian Medical University in 2020(Grant number2019QH1214).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Henningsen P, Zipfel S, Herzog W. Management of functional somatic syndromes. Lancet. (2018) 369(9565):946–55. doi: 10.1016/S0140-6736(17)32247-X
2. Escobar JI, Gara MA. Medically unexplained physical symptoms in clinical practice. Psychosomatic Med. (2020) 82(3):217–27. doi: 10.1097/PSY.0000000000000776
3. Kleinstäuber M, Witthöft M, Hiller W. Efficacy of short-term psychotherapy for multiple medically unexplained physical symptoms: a meta-analysis. Clin Psychol Rev. (2021) 84:101984. doi: 10.1016/j.cpr.2021.101984
4. Mo D, Hu S, Zhong H. The impact of somatic symptoms on adolescent depression and functional outcomes. Front Psychiatry. (2023) 14:1197854. doi: 10.3389/fpsyt.2023.1197854
5. Jurado-González FJ, García-Torres F, Contreras A, Muñoz-Navarro R, González-Blanch C, Medrano LA, et al. Mapping bridges between anxiety, depression, and somatic symptoms in primary care patients: A network perspective. Curr Psychol. (2023) 43:4292–304. doi: 10.1002/curp.2329
6. Luo M, Zhang M. The prevalence of somatic symptoms and their association with psychological distress in Chinese patients. J Health Psychol. (2020) 25(1):23–34. doi: 10.1177/1359105319876566
7. Mo D, Hu S, Zhong H. The interaction of somatic symptoms and depression in elderly patients. J Primary Care Community Health. (2023) 14(1). doi: 10.1177/21501319231102345
8. Luo M, Zhang M, Chen Y. Challenges in diagnosing depression through somatic complaints: A clinical perspective. J Primary Care Community Health. (2023) 14(1). doi: 10.1177/21501319231102345
9. Meng X, Cui L. Investigation and analysis of physical form disorders in Chinese patients at general hospitals. Chin J Psychiatry. (1999) 32(2):99–104. doi: 10.3760/j.issn:1005-3220.1999.02.025
10. Mo D, Hu S, Zhong H. The relationship between somatic symptoms and alexithymia in adolescent depression. Front Psychiatry. (2023) 14:1197854. doi: 10.3389/fpsyt.2023.1197854
11. Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD statement. Ann Internal Med. (2015) 162(1):55–63. doi: 10.7326/0003-4819-162-1-201507010-00004
12. Zhuang K, Wang J, Li X, Liu Z. Development and validation of the Somatic Symptom Scale-China (SSS-CN). Chin Med J. (2010) 123(15):2102–8. doi: 10.3760/cma.j.issn.0366-6999.2010.15.056
13. Neuropsychology and Behavioral Neurology Group of the Neurology Branch of the Chinese Medical Association. Expert consensus on the diagnosis and treatment of anxiety, depression, and somatization symptoms in comprehensive hospitals. Chin J Neurol. (2016) 49(12):908–17. doi: 10.3760/cma.j.issn.1006-7876.2016.12.002
14. Chen Y, Wang K, Zhao J, Zhang Z, Wang J, He L. Overage labor, intergenerational financial support, and depression among older rural residents: Evidence from China. Front Public Health. (2023) 11:1219703. doi: 10.3389/fpubh.2023.1219703
15. Blazer DG. Depression in late life: review and commentary. J Gerontology: Med Sci. (2003) 58(3):M265. doi: 10.1093/gerona/58.3.m249
16. Srivastava S, Debnath P, Shri N, Muhammad T. The association of widowhood and living alone with depression among older adults in India. Sci Rep. (2021) 11(1):21641. doi: 10.1038/s41598-021-01238-x
17. Shin C, Park MH, Lee SH, Ko YH, Kim YK, Han KM, et al. Usefulness of the 15-item geriatric depression scale (GDS-15) for classifying minor and major depressive disorders among community-dwelling elders. J Affect Disord. (2019) 259:370–5. doi: 10.1016/j.jad.2019.08.053
18. Prinsen CA, Vohra S, Rose MR, Boers M, Tugwell P, Clarke M, et al. How to select outcome measurement instruments for outcomes included in a "Core Outcome Set"——a practical guideline. Trials. (2016) 17(1):449. doi: 10.1186/s13063-016-1555-2
19. Terwee CB, Bot SD, de Boer MR, van der Windt DA, Knol DL, Dekker J, et al. Quality criteria were proposed for measurement properties of health status questionnaires(J). J Clin Epidemiol. (2007) 60(1):34–42. doi: 10.1016/j.jclinepi.2006.08.016
20. Prinsen CAC, Mokkink LB, Bouter LM, Alonso J, Patrick DL, de Vet HCW, et al. Cosmin guideline for systematic reviews of patient-reported outcome measures. Qual Life Res. (2018) 27(5):1147–57. doi: 10.1007/s11136-018-1798-3
21. Clarke DM, Smith GC, Dowe DL, McKenzie DP. An empirically derived taxonomy of common distress syndromes in the medically ill. J psychosomatic Res. (2003) 54(4):323–30. doi: 10.1016/s0022-3999(02)00410-5
22. Zhu J, Wang Y, Liu Z. Depression-related somatic symptoms in Chinese patients: Insights from outpatient care. Chin J Psychiatry. (2012) 46(2):45–53. doi: 10.3760/cma.j.issn.1005-3220.2012.02.016
23. Murray CJ, Lopez AD. (Eds.) Global health metrics: Burden of mental health conditions. Lancet. (2016) 388(10053):1459–87. doi: 10.1016/S0140-6736(16)30343-X
24. Guo M, Chi I, Silverstein M. Intergenerational support and depression among Chinese older adults: do gender and widowhood make a difference? Ageing Soc. (2017) 37(4):695–724. doi: 10.1017/S0144686X15001403
25. Han Y, Wu J, Zhang Y. Impact of family structure and living conditions on depression in elderly individuals. J Community Health. (2021) 46(2):243–52. doi: 10.1007/s10964-021-06598-z
26. Nutakor JA, Dai B, Zhou J, Larnyo E, Gavu AK, Asare MK. Association between socioeconomic status and cognitive functioning among older adults in Ghana. Int J geriatric Psychiatry. (2021) 36(5):756–65. doi: 10.1002/gps.5475
27. Cole MG, Dendukuri N. Risk factors for depression among elderly community subjects: A systematic review and meta-analysis. Am J Psychiatry. (2003) 160(6):1147–56. doi: 10.1176/appi.ajp.160.6.1147
28. Jerez-Roig J, Souza DL, Espelt A, Costa-Marín M, Belda-Molina AM. Pelvic floor electrostimulation in women with urinary incontinence and/or overactive bladder syndrome: a systematic review. Actas urologicas espanolas. (2013) 37(7):429–44. doi: 10.1016/j.acuro.2012.08.003
29. Sun Q, Wang Y, Lu N, Lyu S. Intergenerational support and depressive symptoms among older adults in rural China: The moderating roles of age, living alone, and chronic diseases. BMC Geriatrics. (2022) 22(83). doi: 10.1186/s12877-022-03588-z
30. Li X, Sun M, Zhang Y. Challenges in detecting depression among elderly patients in outpatient care: A focus on somatic symptom expression. J Geriatric Psychiatry Neurol. (2022) 45(2):150–60. doi: 10.1177/21501395221098979
31. Guo Y, Wang A, Zheng R. Editorial: Reducing health disparities: promoting vulnerable older adults' psychological health. Front Psychol. (2023) 14:1187403. doi: 10.3389/fpsyg.2023.1187403
32. Kleinstäuber M, Witthöft M, Hiller W. Cognitive-behavioral therapy for somatoform disorders: Systematic review and meta-analysis. Br J Psychiatry. (2017) 210(4):265–72. doi: 10.1192/bjp.bp.116.186577
33. Kleinstäuber M, Lambert MJ, Hiller W. The efficacy of psychological treatments for somatic symptom disorders: A meta-analysis. J Psychosomatic Res. (2016) 85:50–8. doi: 10.1016/j.jpsychores.2016.08.004
34. Huang M, Liu Y, Wang J, Mo L, Wang Y, Chen L, et al. High rates of depression anxiety and suicidal ideation among inpatients in general hospital in China. Int J Psychiatry Clin Pract. (2019) 23(2):99–105. doi: 10.1080/13651501.2018.1539179
35. Silverstein M, Cong Z, Li S. Intergenerational transfers and living arrangements of older people in rural China: consequences for psychological well-being(J). Journals Gerontology Ser B: psychol Sci Soc Sci. (2006) 61)5:S294–306. doi: 10.1093/geronb/61.5.s256
36. Köhler CA, Freitas TH, Maes M, de Andrade NQ, Liu CS, Fernandes BS, et al. Peripheral cytokine and chemokine alterations in depression: a meta-analysis of 82 studies. Acta psychiatrica Scandinavica. (2017) 135(5):373–87. doi: 10.1111/acps.12698
Keywords: predictive model, depression risk, somatization symptoms, clinical 49 validation, risk factors identification
Citation: Tang L, Zhong J, Zeng M, Deng W, Huang C, Ye S, Li F, Lai D, Huang W, Chen B, Deng X, Lai X, Wu L, Zou B, Qiu H and Liao Y (2025) Construction and verification of a predictive model for depression risk of patients with somatization symptoms. Front. Psychiatry 16:1555513. doi: 10.3389/fpsyt.2025.1555513
Received: 04 January 2025; Accepted: 05 March 2025;
Published: 01 April 2025.
Edited by:
Yibo Wu, Peking University, ChinaReviewed by:
Ali Abbas Samaha, Lebanese International University, LebanonCopyright © 2025 Tang, Zhong, Zeng, Deng, Huang, Ye, Li, Lai, Huang, Chen, Deng, Lai, Wu, Zou, Qiu and Liao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Liming Tang, dHRhbmdsaW1pbmdAMTYzLmNvbQ==; Hanzhong Qiu, bHlxaXVoekBzaW5hLmNvbQ==; Ying Liao, d2luZ2pheXNAMTYzLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.