
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Psychiatry , 03 April 2025
Sec. Addictive Disorders
Volume 16 - 2025 | https://doi.org/10.3389/fpsyt.2025.1537822
This article is part of the Research Topic Case Reports in Psychopharmacology, volume III View all 5 articles
 Bojie Zhou1,2†
Bojie Zhou1,2† Shanghao Yang1,2†
Shanghao Yang1,2† Xiafeng Zhou1,2
Xiafeng Zhou1,2 Qian Chen1,3
Qian Chen1,3 Ewen Tu1,4
Ewen Tu1,4 Bo Zhang1,5
Bo Zhang1,5 Li Shi1,2
Li Shi1,2 Xuhui Zhou1,2*
Xuhui Zhou1,2*Background and objectives: Polydrug use has caused serious harm to public health, especially involving novel psychoactive substances. Tiletamine, an N-methyl-D-aspartate receptor (NMDAR) antagonist commonly used as a veterinary anesthetic, has recently emerged in China as an additive in e-cigarettes. However, the long-term impacts of tiletamine and its combined use with other substances remain poorly understood. This case report aims to provide further insight into the clinical manifestations and treatment of tiletamine abuse, particularly focusing on the tremors induced by polydrug use.
Case presentation: The patient had five years of intermittent alcohol use and five months of etomidate abuse. After combining tiletamine for two months, he was repeatedly hospitalized due to coarse tremors, poor sleep and appetite. Based on his substance use pattern and related outcomes, he was diagnosed with phencyclidine use disorder. Initially, intravenous diazepam (20 mg/day) effectively alleviated the tremors. During the second hospitalization, the same dose took longer to take effect, and by the third hospitalization, the dose was increased to 30 mg/day without reducing the tremors. Therefore, primidone was added and gradually titrated to 50 mg/day. The patient’s tremors began to improve by the eighth day and significantly diminished by the tenth day. As we gradually replaced diazepam with lorazepam, the patient insisted on discharge.
Conclusions: Polydrug users, particularly those using NMDAR antagonists and gamma-aminobutyric acid type A receptor (GABA-AR) agonists, may be at increased risk of developing tiletamine dependence, with more severe consequences due to cross-addiction. The combination of alcohol and tiletamine could exacerbate neuroexcitotoxicity during withdrawal, potentially contributing to severe tremors. The successful management of tremors with a combination of neuroinhibitory therapies suggested an effective strategy for complex cases. Further studies are needed to better understand the long-term impacts and risks of tiletamine dependence.
In recent years, there has been a significant rise in the incidence of polydrug use, varying from 3.7%–49.7% in different western countries (1–3), elevating it to a critical public health concern. The primary consequences encompass a heightened risk of accidents or injuries, drug overdose, and fatalities (4). Polydrug use is defined as the concurrent or sequential use of multiple legal or illegal addictive substances (4). Among these, alcohol holds a prominent position due to its widespread availability; an Italian study analyzing outpatient cases revealed that 60% of abuse instances involve alcohol (5). In China, with the rapid economic development, alcohol consumption has been increasing, which has raised concerns about alcohol-related issues (6). Long-term alcoholics often experience postural tremors when they reduce or stop drinking (7). In some cases, even without complete withdrawal, long-term drinking can lead to persistent tremors, which may be related to the chronic brain damage along with other neurological issues (8). The combination of alcohol with other drugs can exacerbate adverse effects such as withdrawal symptoms, cardiovascular disease, liver damage, and behavioral abnormalities (9).
Recently, the stringent regulation of traditional drugs has prompted illegal manufacturers to explore novel psychoactive substances (NPS) as substitutes for illicit drugs. NPS that are most closely linked to psychiatric consequences include synthetic cannabinoids, novel synthetic opioids, and ketamine analogs (10). The proliferation of these substances has been facilitated by the shift from street-level drug markets to online platforms, making them more accessible to a broader audience (11). Despite efforts to control their spread, NPS continue to pose a significant challenge due to their ever-evolving nature and the difficulty in regulating them effectively (12). In China, the drug abuse pattern has also transitioned from traditional opiates to a combination of new synthetic drugs, medical anesthetics, and psychoactive substances (13). Since 2022, reports have emerged concerning the abuse of etomidate e-cigarettes in China. Due to its severe addictive potential and harmful effects, etomidate was officially regulated in China as of October 1st, 2023. Subsequently, the veterinary anesthetic tiletamine has gradually emerged as a novel drug substitute, also sold and consumed in e-cigarette form, posing a significant threat to public health and safety.
Tiletamine is a non-competitive NMDA receptor antagonist with structural and pharmacological similarities to phencyclidine (PCP) and ketamine, known for its potent analgesic and dissociative anesthetic properties (14). It is often combined with the GABA-A receptor agonist zolazepam, which provides mild sedation and effective muscle relaxation (14). Tiletamine and zolazepam are combined in a 1:1 ratio to form the compound anesthetic Telazol (Zoetis Inc., New Jersey, USA). In Asia, this anesthetic is commonly known as Zoletil (Virbac, Lyon, France). Telazol abuse has been reported since 1999 (15). Initially confined to veterinary workers (15, 16), Telazol use has expanded to include the general public (17). The primary routes of abuse include intravenous bolus (18), intramuscular injection (19), and nasal inhalation (17). Most reported cases involve recreational use, with two resulting in fatalities (15, 16), and one in a failed suicide attempt (18). Due to its societal harm, tiletamine has been classified as a controlled substance in countries including South Korea and the United States (20).
Although tiletamine is gaining popularity in other countries, its consequences remain insufficiently explored. In China, where tiletamine use is still emerging, there is no medical evidence documenting its abuse or addiction. Its long-term effects and potential dependence are not well understood, nor are the outcomes of co-use with e-cigarettes and alcohol, presenting significant challenges to treatment services and regulatory oversight. These developments highlight the urgent need for more case studies to identify the clinical symptoms and effective recovery strategies to mitigate the public health impact of tiletamine. A key concern is the movement disorders associated with tiletamine (Telazol or Zoletil) use (21, 22), which are primarily manifested as tremors in this case. Herein, we report a case of severe tremors resulting from polydrug use, primarily involving alcohol and tiletamine e-cigarettes, aiming to provide further insight into the clinical manifestations and treatment of tiletamine abuse.
A 40-year-old male presented with recurrent limb tremors, poor appetite, and sleep disturbances, leading to repeated hospitalizations over three months. He has a long history of substance use, beginning in 2005 with ketamine (4 years), followed by heroin (3 years), and methamphetamine (6 years). After each substance, he underwent compulsory detoxification for one year. In 2019, following his last detoxification, he intermittently consumed high-proof alcohol (400–500 ml per occasion), primarily during social gatherings. In June 2023, he began using etomidate-containing e-cigarettes, which led to a car accident due to hazardous driving. He discontinued etomidate after the accident but switched to tiletamine-containing e-cigarettes in October 2023, escalating his use from a few times a week to daily (from two to six cartridges). Despite experiencing euphoria initially, he later developed depressive moods and suicidal thoughts. His alcohol consumption continued, but he did not report withdrawal symptoms. Key milestones in the patient’s substance use history were illustrated in Table 1.
On November 28, 2023, the patient developed involuntary tremors in both upper limbs, which worsened with activity. He was prescribed diazepam(10mg/day), which alleviated the tremors, but his symptoms returned after resuming tiletamine use. He was hospitalized on December 4, 2023, and received intravenous diazepam(20mg/day), after which he improved and requested discharge. Upon discharge, he resumed alcohol consumption, which further reduced the tremors. He relapsed to tiletamine on January 21, 2024, and increased his dosage (eight cartridges per day). By January 27, 2024, he reduced his alcohol intake by half. On February 5, 2024, he experienced worsening tremors and was admitted to our hospital, where he received intravenous diazepam(20mg/day) with significant improvement. However, after discharge, he relapsed and resumed previous consumption levels without drinking alcohol. On February 20, 2024, he was readmitted after his tremors worsened due to abrupt discontinuation of tiletamine, along with chest tightness, nausea, and dizziness. A review of his medical history revealed two prior admissions for limb tremors, with details of diagnoses and interventions summarized in Table 2.
The patient’ s vital signs were normal upon first two admission. On the third hospitalization, he was clear with a blood pressure of 128/93 mmHg, pulse rate of 59 beats per minute, respiration rate of 20 breaths per minute, and body temperature of 36.9°C. The observable tremors in four limbs, about 5 centimeters in amplitude, intensified with voluntary movement; however, his muscle tone remained normal. He also exhibited an unsteady gait, head tremor, voice tremor, slurred articulation and variable voice intensity. Increased irritability was noted, but hallucinations and delusions were denied. Recent laboratory tests showed no abnormalities in routine blood tests, coagulation profiles, blood ammonia, electrolytes, liver and kidney function, cardiac enzymes, blood glucose, lipid profiles, or thyroid function. Urinalysis was negative for benzodiazepines, morphine, ketamine, ecstasy, methamphetamine, and buprenorphine. The electrocardiogram (ECG) showed a normal rhythm with a heart rate of 69 beats per minute. Brain magnetic resonance imaging (MRI) revealed a cavernous hemangioma in the right basal ganglia, and the electroencephalogram (EEG) showed a mildly abnormal fast-wave background. The images of the ECG and EEG are presented in Figures 1, 2 respectively.
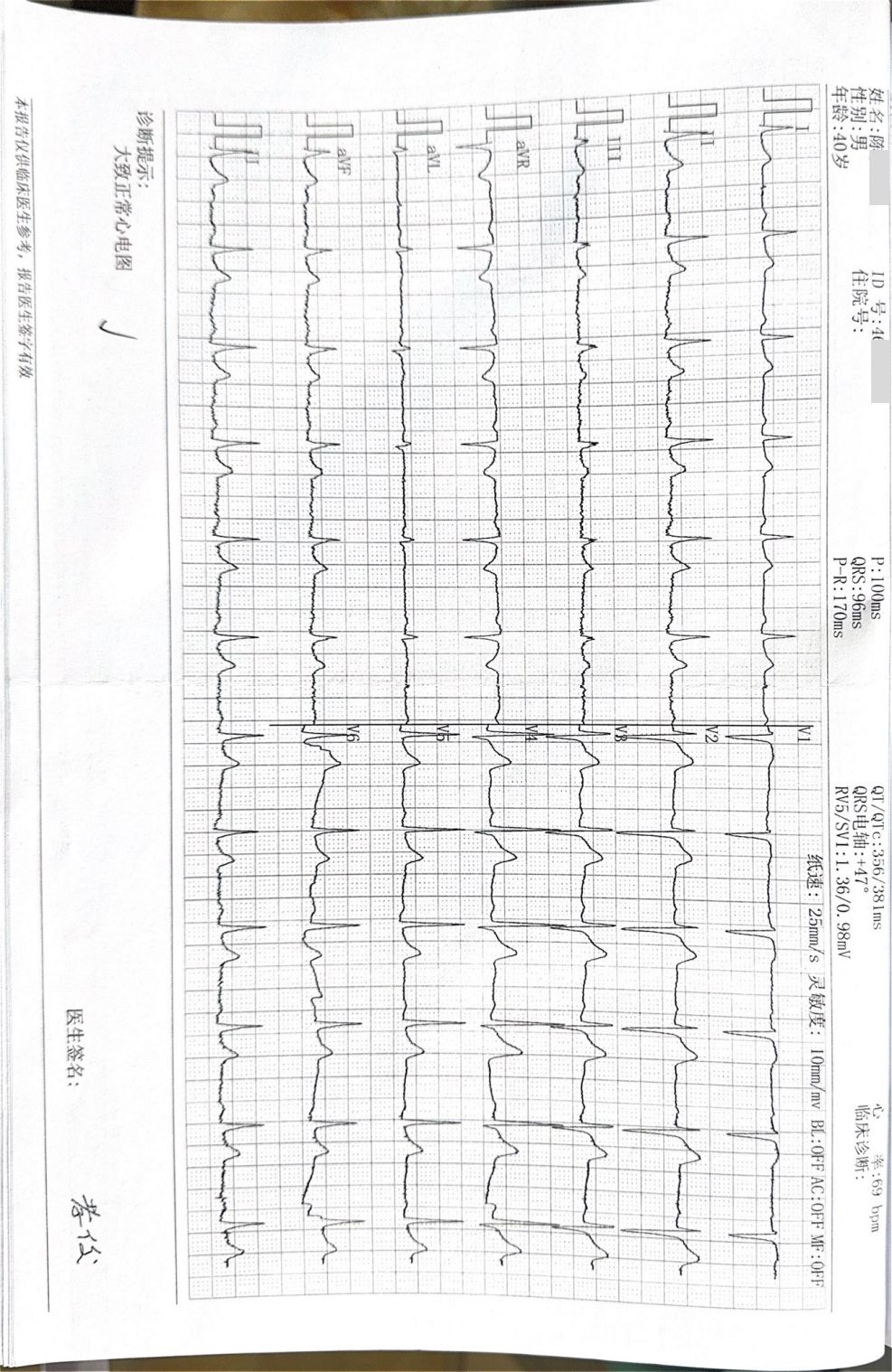
Figure 1. Electrocardiogram (ECG) images. The electrocardiogram showed a generally normal rhythm with a heart rate of 69 beats per minute.
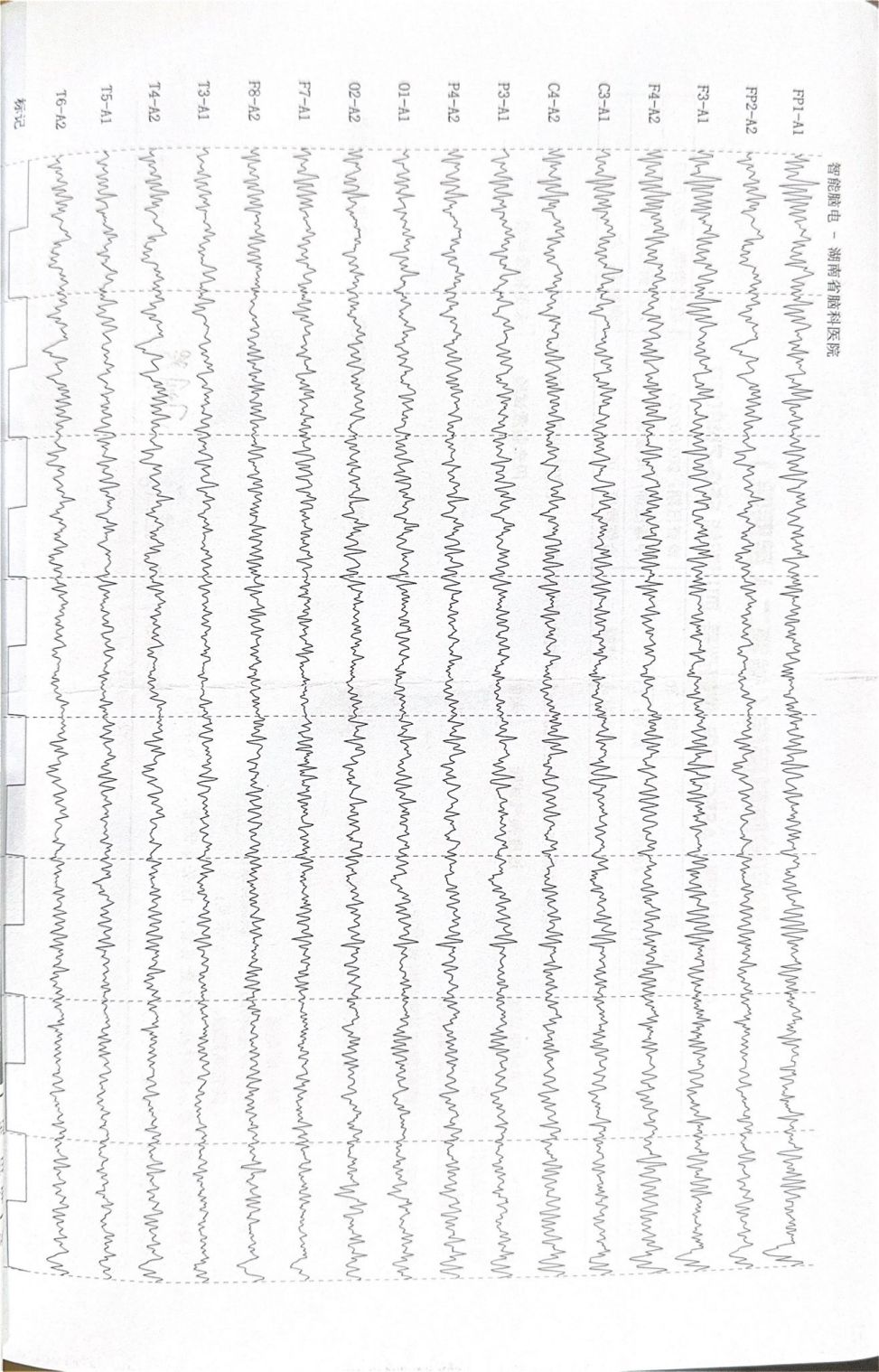
Figure 2. Electroencephalogram (EEG) images. The electroencephalogram showed abnormal findings with mild background activity. The primary rhythm observed during quiet wakefulness with eyes closed was a low-amplitude (5–10 μV), 15–20 Hz rhythmic activity, which appeared somewhat irregular. Bilateral symmetry was present.
Brain MRI showed nonspecific abnormalities in the basal ganglia, which could not suggest any organic brain disorder or explain the onset of the tremors. The patient’s alcohol consumption pattern did not meet the criteria for alcohol dependence, and he did not exhibit symptoms such as sweating, elevated blood pressure, or tachycardia, thereby ruling out alcohol withdrawal as the cause of the tremors. Although we were unable to detect tiletamine using our current techniques, considering that the patient exhibited a pattern of self-reported tiletamine use (structure and potency similar to phencyclidine), characterized by high-dose consumption, intense cravings, increased tolerance, and recurrent physical and interpersonal issues meeting over two criteria from the DSM-5, we diagnosed the patient with phencyclidine use disorder.
To manage his tremors and associated symptoms, pharmacologic treatments outlined in Table 3 were administered. He received intravenous diazepam at 30 mg/day. Due to irritability, poor sleep and appetite, oral sodium valproate (0.25 g twice daily) and olanzapine (5 mg nightly) were started. Additionally, 500 ml of intravenous fluids were given twice daily to maintain physiological stability. As his appetite improved, intravenous fluid administration was gradually discontinued. By the sixth day, his sleep, anxiety and irritability had significantly improved, with sodium valproate at 1 g/day, olanzapine at 10 mg/night and the Hamilton Anxiety Rating Scale score dropping from 25 to 10. However, tremors showed no improvement (5 centimeters in amplitude). Consequently, primidone was initiated at 25 mg nightly and gradually increased to 50 mg/day. By the eighth day, tremors began to subside (3 centimeters in amplitude), with marked improvement by the tenth day (1 centimeters in amplitude). During this period, diazepam was gradually replaced with oral lorazepam. By the twelfth day, the patient was able to walk normally, though mild tremors persisted in both upper limbs. The following day, the patient requested discharge and declined further medication. No significant adverse drug reactions occurred throughout treatment.
In the six-month follow-up conducted by phone, the patient reported no use of tiletamine e-cigarettes or tremors, while maintaining his previous pattern of alcohol consumption.
To the best of our knowledge, this is the first documented case of polydrug use involving alcohol and e-cigarettes containing etomidate and tiletamine. The existing literature lacks detailed descriptions of tiletamine dependence and effective treatment recommendations. Compared to previously reported cases of tiletamine or Zoletil abuse, the primary symptom in this case was severe limb tremors. Consequently, our treatment focused primarily on addressing the tremors and successfully managed the acute withdrawal phase.
Animal studies suggest that tiletamine, when used as a sole anesthetic, can induce muscle rigidity and seizures (23), similar to the effects observed in humans after a single large dose (17). Acute tiletamine poisoning is often fatal and can result in changes in mental status, nystagmus, cardiac dysfunction, and metabolic disruption (15, 16, 24, 25), while chronic use has been associated with psychoses and behavioral abnormalities (19), with rare reports of movement disorders. For instance, Lee reported a 35-year-old man who intermittently used heroin and inhaled Telazol to reduce heroin dosage, gradually developing choreatic movements after two weeks, which were alleviated with clonazepam after two weeks (21). Similarly, a 30-year-old male who consumed 2,250 mg of Zoletil over 9 days developed involuntary tremors of the lower jaw, failing eyesight, and sialorrhea one day after discontinuing the injection. The symptoms self-resolved without any intervention (22). In our case, tremors in four limbs were consistently observed in relation to tiletamine and alcohol use.
The patient’s tremors were initially mild and gradually worsened. Similar symptoms have been reported in ketamine users. In a 10-year cross-sectional study in Italy involving 7,897 cases of illegal substance use, 74 cases were ketamine-related. Tremors accounted for 6.8% of the reasons ketamine users sought emergency department care, and these tremors were typically mild and transient (26). This case, however, presented with intentional tremors, dysarthria, and ataxia, suggesting a cerebellar dysfunction. Interestingly, alcohol alleviated these tremors, similar to essential tremor (ET), where alcohol activates GABA-A receptors in cerebellar granule cells, reducing tremors (27–29). Additionally, repeated ketamine administration has been shown to upregulate NMDAR expression in the frontal cortex while downregulating GABA-AR expression, paralleling the modulation observed in chronic alcohol consumption models (30). Furthermore, the increase in its subunit GRN1 and the resultant neurotoxicity are dependent on both the duration and dosage of ketamine exposure (30). Thus, we hypothesize that repeated high-dose tiletamine use may cause neurotoxicity in cerebellar granule cells, increasing excitotoxicity during withdrawal. Alcohol, a GABA-A agonist, could counterbalance this excitotoxicity. However, with prolonged use, the balance between inhibitory and excitatory neurotransmission may be reestablished and then disrupted by abrupt cessation or dose reduction of either substance, leading to tremors.
However, the patient’s tremors were more severe than those induced by either ketamine or alcohol withdrawal, suggesting that polydrug use may have contributed to their intensity. The combination of alcohol and tiletamine likely resulted in synergistic neuroinhibition, heightening glutamatergic activity and excitotoxicity. Previous reports have shown that tiletamine abusers are typically polydrug users with histories of NMDAR antagonist or GABAR agonist use (31). Animal studies have shown that prior exposure to alcohol significantly enhances Zoletil place preference and self-administration, highlighting the cross-addiction and compensatory effects between ethanol and NMDA antagonists (32, 33). This cross-addiction may lead to increased consumption of both substances at high doses, potentially explaining the heightened severity of tremors. Although direct human studies are limited, preclinical data suggest that alcohol and tiletamine may together produce even stronger neuroexcitotoxic effects, similar to those observed with the combination of ketamine and ethanol (34, 35). Given tiletamine’s potency and effective duration between PCP and ketamine in animals (36), it is reasonable to expect tiletamine might produce even stronger neurotoxic effects in humans.
Accordingly, the tremors likely resulted from an imbalance between the excitatory glutamatergic and inhibitory GABAergic systems, exacerbated by acute withdrawal. During the first two hospitalizations, intravenous diazepam was initially effective in controlling the tremors. However, as tolerance developed, diazepam became less effective over time, necessitating higher doses. Given that the tremors were likely linked to excitotoxicity, we administered sodium valproate to increase GABA levels and stabilize mood, in addition to primidone to alleviate the tremors. This combination led to significant improvement, suggesting that neuroinhibitory therapies could effectively address the patient’s symptoms.
In addition, while deaths related to vaping products containing synthetic cannabinoids have been reported (37), and some studies have suggested that harmful constituents in vaping aerosols may cause cardiopulmonary dysfunction (38), it remains unclear whether e-cigarettes, as a medium, contribute to the dependence and neurotoxicity of tiletamine. Given the widespread use of e-cigarettes and the lack of regulatory policies on tiletamine in China, an increase in tiletamine abuse and dependence via e-cigarettes is expected, along with a potential rise in the risks associated with polydrug use. It remains uncertain whether e-cigarette use of tiletamine is more toxic than traditional methods. Vigilance is needed regarding the concurrent use of tiletamine and GABA-A agonists.
This study has several limitations. Due to constraints in testing methods, we were unable to conduct drug testing for tiletamine or quantify its amount in the e-cigarette. Given that both alcohol and tiletamine can cause neurological damage, we did not explicitly assess peripheral nerve damage in this patient, nor did we investigate potential deficiencies in vitamin B1 or B12. These factors should be considered in future clinical practice. Despite the effectiveness of pharmacological treatments in managing symptoms, improving treatment adherence in patients with complex polysubstance abuse remains challenging. In this case, the patient failed to complete the detoxification treatment during all three hospitalizations, suggesting a lack of recognition of the dangers of polysubstance abuse and insufficient motivation for withdrawal. A multi-disciplinary approach, including psychological therapy, behavioral interventions, and support groups, is crucial but difficult to implement consistently in clinical practice. Future efforts may need to focus more on prevention, with regular health education, substance use assessments, and psychological evaluations for individuals with a history of polysubstance abuse. This proactive approach would help identify problematic behaviors, the risk of drug dependence, and any potential side effects, allowing for timely intervention and improving the long-term management of polysubstance abusers to prevent relapse.
Polysubstance abusers, particularly those using NMDA antagonists and GABA-A agonists, could be at increased risk of developing addiction to tiletamine. The concurrent use of these substances could lead to more severe consequences due to cross-addiction and their ability to counteract each other’s negative effects. Long-term alcohol use induces adaptive changes in NMDA and GABA receptors, leading to faster tolerance development, requiring higher doses of tiletamine to achieve desired effects and complicating withdrawal treatment. Combined use of alcohol and tiletamine may exacerbate neuroexcitotoxicity during withdrawal by upregulating glutamatergic and NMDA receptor activity, potentially contributing to severe tremors as seen in this case. The effectiveness of adding primidone in treating tremors suggests that a combination of neuroinhibitory therapies may more effectively manage the complex tremors induced by polydrug use involving tiletamine and GABA-A receptor agonists. Further studies on tiletamine are needed to better understand its effects and potential risks on humans.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Written informed consent was obtained from the individual for the publication of any potentially identifiable images or data included in this article. Written informed consent was obtained from the participant/patient(s) for the publication of this case report.
BJZ: Conceptualization, Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing. SY: Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing. XFZ: Investigation, Methodology, Resources, Writing – review & editing. QC: Investigation, Writing – review & editing. ET: Supervision, Writing – review & editing. BZ: Methodology, Writing – review & editing. LS: Methodology, Writing – review & editing. XHZ: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was financially supported by the Clinical Research Center For Addiction Disorder in Hunan Province (No. 2023SK4055), the Scientific Research Project of the Hunan Health Commission (No. A202303096949), the Foundation of Hunan Provincial Administration of Traditional Chinese Medicine (No. B2024102), Hunan Provincial Health High-Level Talent Scientific Research Project (No. R2023178), National Cultivation Project of Key Clinical Specialty (Addiction medicine), Hunan Province clinical key specialty (Addiction medicine), Key Clinical Specialty Construction Project of the Hunan Health Commission (Improvement of Diagnosis and Treatment Ability of Severe Psychiatric Diseases in Hunan Province).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that Generative AI was used in the creation of this manuscript.
During the preparation of this work the authors used ChatGPT-4o in order to check grammar errors, refine language, and enhance readability to ensure that the manuscript is well-structured and adheres to academic standards. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Hassan AN, Le Foll B. Polydrug use disorders in individuals with opioid use disorder. Drug Alcohol Depend. (2019) 198:28–33. doi: 10.1016/j.drugalcdep.2019.01.031
2. Tassoni G, Cippitelli M, Mietti G, Cerioni A, Buratti E, Bury E, et al. Hair analysis to evaluate polydrug use. Healthcare (Basel). (2021) 9(8):972. doi: 10.3390/healthcare9080972
3. Karjalainen K, Kuussaari K, Kataja K, Tigerstedt C, Hakkarainen P. Measuring concurrent polydrug use in general populations: A critical assessment. Eur Addict Res. (2017) 23:163–9. doi: 10.1159/000477802
4. EMCDDA. Polydrug use: health and social responses 2021(2021). Available online at: https://www.emcdda.europa.eu/publications/mini-guides/polydrug-use-health-and-social-responses_en (Accessed March 23, 2024).
5. Bonfiglio NS, Portoghese I, Renati R, Mascia ML, Penna MP. Polysubstance use patterns among outpatients undergoing substance use disorder treatment: A latent class analysis. Int J Environ Res Public Health. (2022) 19(24):16759. doi: 10.3390/ijerph192416759
6. Tao R, Jiang F, Min K, Liu T, Liu Y, Xia L, et al. Alcohol use among mental health professionals in China: A nationwide cross-sectional survey. Alcohol Alcohol. (2021) 56:351–9. doi: 10.1093/alcalc/agaa065
7. Tiglao SM, Meisenheimer ES, Oh RC. Alcohol withdrawal syndrome: outpatient management. Am Fam Physician. (2021) 104:253–62.
8. Planas-Ballvé A, Grau-López L, Morillas RM, Planas R. Neurological manifestations of excessive alcohol consumption. Gastroenterol Hepatol. (2017) 40:709–17. doi: 10.1016/j.gastrohep.2017.05.011
9. Singh AK. Alcohol interaction with cocaine, methamphetamine, opioids, nicotine, cannabis, and γ-hydroxybutyric acid. Biomedicines. (2019) 7(1):16. doi: 10.3390/biomedicines7010016
10. Schifano F, Napoletano F, Chiappini S, Guirguis A, Corkery JM, Bonaccorso S, et al. New/emerging psychoactive substances and associated psychopathological consequences. Psychol Med. (2021) 51:30–42. doi: 10.1017/S0033291719001727
11. Schifano F. Recent changes in drug abuse scenarios: the new/novel psychoactive substances (NPS) phenomenon. Brain Sci. (2018) 8(12):221. doi: 10.3390/brainsci8120221
12. Vicknasingam B, Narayanan S, Singh D, Corazza O. Global strategy for New Psychoactive Substances: an update. Curr Opin Psychiatry. (2020) 33:295–300. doi: 10.1097/YCO.0000000000000612
13. Sun HQ, Bao YP, Zhou SJ, Meng SQ, Lu L. The new pattern of drug abuse in China. Curr Opin Psychiatry. (2014) 27:251–5. doi: 10.1097/YCO.0000000000000073
14. de la Peña JB, Cheong JH. The abuse liability of the NMDA receptor antagonist-benzodiazepine (tiletamine-zolazepam) combination: evidence from clinical case reports and preclinical studies. Drug Testing Analysis. (2016) 8:760–7. doi: 10.1002/dta.1987
15. Cording CJ, DeLuca R, Camporese T, Spratt E. A fatality related to the veterinary anesthetic telazol. J Anal Toxicol. (1999) 23:552–5. doi: 10.1093/jat/23.6.552
16. Chung H, Choi H, Kim E, Jin W, Lee H, Yoo Y. A fatality due to injection of tiletamine and zolazepam. J Anal Toxicol. (2000) 24:305–8. doi: 10.1093/jat/24.4.305
17. Yip WL, Rex Pui Kin L, Magdalene YT, Nike Kwai Cheung L, Matthew Sik HT. Tiletamine detected in a ketamine abuser with altered mental status. Clin Toxicology. (2019) 58:430–1. doi: 10.1080/15563650.2019.1650938
18. Cha K, Jeong WJ, Kim HM, So BH. Intravenous zoletil administration for the purpose of suicide. Clin Exp Emerg Med. (2021) 8:149–51. doi: 10.15441/ceem.20.050
19. Huang YT, Cheng YC, Yang TW, Huang MC. Psychosis associated with high dose of Zoletil. Psychiatry Clin Neurosci. (2016) 70:364–5. doi: 10.1111/pcn.2016.70.issue-8
20. Kim J, Yum H, Jang M, Shin I, Yang W, Baeck S, et al. A comprehensive and sensitive method for hair analysis in drug-facilitated crimes and incorporation of zolazepam and tiletamine into hair after a single exposure. Anal Bioanal Chem. (2016) 408:251–63. doi: 10.1007/s00216-015-9099-y
21. Lee C-C, Lin Y-Y, Hsu C-W, Chu S-J, Tsai S-H. Movement disorder caused by abuse of veterinary anesthesia containing tiletamine. Am J Emergency Med. (2009) 27:1022.e5–.e6. doi: 10.1016/j.ajem.2008.12.030
22. Wang I, Yeom S, Kim Y. A case of zoletil, veterinary anesthetic poisoning. J Korean Soc Emergency Med. (2013) 24:122–4.
23. Ferrari L, Turrini G, Rostello C, Guidi A, Casartelli A, Piaia A, et al. Evaluation of two combinations of Domitor, Zoletil 100, and Euthatal to obtain long-term nonrecovery anesthesia in Sprague-Dawley rats. Comp Med. (2005) 55:256–64.
24. Redmond AH, Herman MI. Ingestion of Telazol®, a veterinary anaesthetic, by a 16 year-old male. Clin Intensive Care. (2002) 13:139–42. doi: 10.3109/tcic.13.2-3.139.142
25. Lee YK, Kang IG, Park CS, Heo SJ, Chai YS, Park SS, et al. A case of mental change in a patient who received a zoletil injection. J Korean Soc Clin Toxicology. (2013) 11:23–7.
26. Pavarin RM, Marani S, Turino E. Ketamine abusers referring to emergency departments in northern Italy: a cross- sectional study. Ann Ist Super Sanita. (2019) 55:338–44. doi: 10.4415/ANN_19_04_06
27. Hörtnagl H, Tasan RO, Wieselthaler A, Kirchmair E, Sieghart W, Sperk G. Patterns of mRNA and protein expression for 12 GABAA receptor subunits in the mouse brain. Neuroscience. (2013) 236:345–72. doi: 10.1016/j.neuroscience.2013.01.008
28. Hanchar HJ, Dodson PD, Olsen RW, Otis TS, Wallner M. Alcohol-induced motor impairment caused by increased extrasynaptic GABA(A) receptor activity. Nat Neurosci. (2005) 8:339–45. doi: 10.1038/nn1398
29. Boecker H, Wills AJ, Ceballos-Baumann A, Samuel M, Thompson PD, Findley LJ, et al. The effect of ethanol on alcohol-responsive essential tremor: a positron emission tomography study. Ann Neurol. (1996) 39:650–8. doi: 10.1002/ana.410390515
30. Xu K, Lipsky RH. Repeated ketamine administration alters N-methyl-D-aspartic acid receptor subunit gene expression: implication of genetic vulnerability for ketamine abuse and ketamine psychosis in humans. Exp Biol Med (Maywood). (2015) 240:145–55. doi: 10.1177/1535370214549531
31. de la Peña JB, Yoon SY, de la Peña IC, Lee HL, I de la Peña IJ, Cheong JH. Pre-exposure to related substances induced place preference and self-administration of the NMDA receptor antagonist–benzodiazepine combination, zoletil. Behav Pharmacol. (2013) 24:20–8. doi: 10.1097/FBP.0b013e32835cf442
32. de la Peña JBI, dela Peña IJI, Lee HL, dela Peña I, Shin CY, Sohn AR, et al. Pre-exposure to ethanol, but not to caffeine and nicotine, induced place preference and self-administration of the NMDA receptor antagonist-benzodiazepine combination, Zoletil®. Pharmacol Biochem Behavior. (2013) 110:231–7. doi: 10.1016/j.pbb.2013.07.016
33. de la Pena JB, Lee HC, de la Pena IC, Woo TS, Yoon SY, Lee HL, et al. Rewarding and reinforcing effects of the NMDA receptor antagonist-benzodiazepine combination, Zoletil(R): difference between acute and repeated exposure. Behav Brain Res. (2012) 233:434–42. doi: 10.1016/j.bbr.2012.05.038
34. Zuo D, Liu Y, Liu Z, Cui J, Zhou X, Liu Y, et al. Alcohol aggravates ketamine-induced behavioral, morphological and neurochemical alterations in adolescent rats: The involvement of CREB-related pathways. Behav Brain Res. (2018) 349:80–90. doi: 10.1016/j.bbr.2018.05.003
35. Li Q, Wu HR, Fan SJ, Liu DX, Jiang H, Zhang Q, et al. The effects of sub-anesthetic ketamine plus ethanol on behaviors and apoptosis in the prefrontal cortex and hippocampus of adolescent rats. Pharmacol Biochem Behav. (2019) 184:172742. doi: 10.1016/j.pbb.2019.172742
36. Lin HC, Thurmon JC, Benson GJ, Tranquilli WJ. Telazol–a review of its pharmacology and use in veterinary medicine. J Vet Pharmacol Ther. (1993) 16:383–418. doi: 10.1111/j.1365-2885.1993.tb00206.x
37. Roberts E, Copeland C, Robson D, McNeill A. Drug-related deaths associated with vaping product use in the United Kingdom. Addiction. (2021) 116:2908–11. doi: 10.1111/add.v116.10
Keywords: tiletamine, alcohol, primidone, phencyclidine use disorder, polydrug use, withdrawal, e-cigarettes, case report
Citation: Zhou B, Yang S, Zhou X, Chen Q, Tu E, Zhang B, Shi L and Zhou X (2025) Severe tremors induced by tiletamine e-cigarette and alcohol use: a case report. Front. Psychiatry 16:1537822. doi: 10.3389/fpsyt.2025.1537822
Received: 01 December 2024; Accepted: 26 February 2025;
Published: 03 April 2025.
Edited by:
Matej Stuhec, University of Maribor, SloveniaReviewed by:
Juan Sebastian Izquierdo-Condoy, University of the Americas, EcuadorCopyright © 2025 Zhou, Yang, Zhou, Chen, Tu, Zhang, Shi and Zhou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xuhui Zhou, MzMwMTE1QGhudWNtLmVkdS5jbg==
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.