
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Psychiatry , 21 March 2025
Sec. Mood Disorders
Volume 16 - 2025 | https://doi.org/10.3389/fpsyt.2025.1467876
 Ting-Hui Liu1
Ting-Hui Liu1 Jheng-Yan Wu2
Jheng-Yan Wu2 Po-Yu Huang3
Po-Yu Huang3 Wan-Hsuan Hsu3
Wan-Hsuan Hsu3 Min-Hsiang Chuang3
Min-Hsiang Chuang3 Ya-Wen Tsai4
Ya-Wen Tsai4 Kuang-Yang Hsieh1*
Kuang-Yang Hsieh1* Chih-Cheng Lai5,6*
Chih-Cheng Lai5,6*Background: Given that atrial fibrillation (AF) s associated with a high risk of psychiatric disorders, understanding the potential benefits of catheter ablation is clinically significant. This study was conducted to examine whether catheter ablation can prevent psychiatric disorders in patients with AF.
Methods: A retrospective cohort study was conducted over two years using data from the TriNetX electronic health record network. The study included adults diagnosed with AF and treated with either antiarrhythmic or rate-control medications. Participants were divided into two groups: those who underwent catheter ablation and a control group without ablation. The primary outcome measured was a composite of anxiety, depression, and insomnia occurrence within one to three years post-treatment. Secondary outcomes included individual psychiatric disorders, suicidal ideation or attempts, dementia, cerebral infarction, and atopic dermatitis (as a negative control).
Results: We included 21,019 patients in each matched group. The ablation group demonstrated a lower risk of the primary combined outcome (hazard ratio(HR):0.873, 95% confidence interval (CI) 0.784–0.973, p<0.01), and secondary outcomes including anxiety (HR:0.822, 95% CI:0.700–0.964; p=0.016), depression (HR:0.614, 95% CI:0.508–0.743; p<0.001), suicidal ideation or attempts (HR:0.392, 95% CI:0.165–0.934; p=0.028), dementia (HR:0.569, 95% CI:0.422–0.767; p<0.001), and cerebral infarction (HR:0.704, 95% CI:0.622–0.797; p<0.001) compared to the non-ablation group.
Conclusions: In patients with atrial fibrillation, catheter ablation was associated with a reduced risk of developing psychiatric disorders, including anxiety, depression, insomnia, suicidal ideation or attempt, and dementia, in comparison to those who did not undergo ablation. Clinicians should consider incorporating psychiatric risk factors into their comprehensive patient assessment when evaluating candidates for catheter ablation.
Atrial fibrillation (AF) is a prevalent form of symptomatic arrhythmia, impacting approximately 59.7 million individuals worldwide (1). This condition not only predisposes affected individuals to severe cardiovascular complications but also substantially diminishes their overall quality of life (2, 3). While the connection between AF and cardiovascular complications like stroke and heart failure is well-documented (4, 5), growing research reveals its significant impact on mental health (6–9). Patients with AF show higher rates of anxiety, depression, and other psychiatric disorders – a link that may be explained by several biological mechanisms. These include chronic inflammation, disrupted blood flow patterns, and the physical and emotional stress of managing a chronic heart condition (7, 10, 11). Understanding these connections is crucial, as mental health challenges can significantly affect how patients manage their AF, follow treatment plans, and perceive their illness, ultimately influencing their overall health outcomes.
Catheter ablation, a therapeutic modality that aims to restore sinus rhythm by isolating the pulmonary veins, has demonstrated promising results in reducing AF burden and associated complications (12). Randomized controlled trials (RCTs) have provided evidence that catheter ablation is a well-tolerated procedure for managing AF in patients who exhibit non-responsiveness to antiarrhythmic drugs (13, 14). Moreover, recent evidence from RCTs suggests that, in comparison to antiarrhythmic drugs, using catheter ablation as an initial treatment can help reduce the recurrence of AF and the need for hospitalizations (15, 16).
Beyond its antiarrhythmic benefits, there is a growing interest in exploring whether catheter ablation can potentially reduce the risk of psychiatric disorders in adults with AF. A recent multicenter RCT known as the Randomized Evaluation of the Impact of Catheter Ablation on Psychological Distress in Atrial Fibrillation (REMEDIAL) trial revealed that catheter ablation for AF led to a significant improvement in anxiety, depression, and psychological distress when compared to medication management (17). It is worth noting that this trial had a relatively small cohort size, including only 100 individuals. At present, there is a noticeable absence of large-scale real-world data that investigate the impact of catheter ablation on psychiatric disorders in patients with AF.
To address this research gap, we conducted a retrospective cohort study using TriNetX, a global health research network that provides comprehensive, updated, and longitudinal patient data from healthcare organizations. Our study aimed to assess the association between catheter ablation and psychiatric disorder risk in adults with AF.
This retrospective cohort study used data from the TriNetX research network, providing access to the de-identified records of 250 million unique patients sourced from over 120 healthcare organizations (HCOs) spanning 19 countries. TriNetX represents a global coalition of health research entities that gather anonymized patient data from electronic health records (EHRs). The dataset includes an extensive array of patient information such as demographics, clinical diagnoses, performed procedures, prescription details, laboratory findings, genetic information, and documentation of healthcare organization visits. In this analysis, we used Research Network that includes data on more than 30 million patients from 80 healthcare organizations. TriNetX facilitates patient-level data analysis through integrated tools and provides researchers with synthesized results (18, 19). Access to comprehensive information in the database is available online. The requirement for written informed consent was exempted due to the anonymized nature of the TriNetX data. The study protocol received approval from the Institutional Review Board of Chi Mei Medical Center (approval no. 11206-E02).
The TriNetX Research database comprising 80 HCOs as of October 15, 2023, was used in the patient selection process. We began with individuals who visited HCOs more than twice. The inclusion criteria for our cohort were an age of at least 18 years, a diagnosis of AF, and a documented history of receiving antiarrhythmic or rate-control medications. AF was defined according to the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) code I48.0 (Paroxysmal atrial fibrillation), I48.1 (Persistent atrial fibrillation), and I48.2 (Chronic atrial fibrillation) (20). We employed RxNorm, ATC, and VA codes to ascertain the usage of antiarrhythmic or rate-control medications. Next, the cohort was categorized into two groups based on the treatment type: those who underwent catheter ablation and those who did not. Catheter ablation was identified using the procedure codes listed in Supplementary Tables S1, S2. Patients with psychiatric diagnoses one year before and one year after the index date were excluded. The index date, as defined for each cohort (further details are available in Supplementary Tables S3, S4), signifies the date when patients first fulfilled the specific cohort criteria. For the ablation group, this date corresponds to the procedure date, while for the non-ablation group, it corresponds to the date of diagnosis. Further details regarding the inclusion and exclusion criteria, and additional patient information, are provided in Supplementary Tables S1 and S2.
Finally, the patient selection involved the use of propensity score matching on a 1:1 basis, matching for age at the index, race, sex, and comorbid medical conditions. This resulted in two comparable groups for the study: the ablation and non-ablation groups.
To mitigate baseline characteristic disparities between the ablation and non-ablation cohorts, we accounted for 23 variables (21, 22). The covariates employed for 1:1 propensity score matching encompassed age at index event, gender (female or male), race (Caucasian, African American, Asian, or indeterminate), along with a range of comorbid conditions. These conditions comprised hypertension, diabetes mellitus, hyperlipidemia, chronic kidney disease, ischemic heart disease, heart failure, cerebral infarction, atherosclerosis, peripheral vascular diseases, neoplasms, chronic obstructive pulmonary disease (COPD), asthma, chronic liver diseases, substance use disorders, and alcohol-related disorders. This comprehensive approach aimed to ensure a rigorous comparison and enhance the reliability of the study outcomes.
The primary outcome of our study was the composite incidence of specific psychiatric disorders, including anxiety, depression, and insomnia within a one- to three-year period following the index date. The index date, defined for each cohort (details provided in Supplementary Tables S3, S4), marked the day when patients initially met the specified cohort criteria (23). These particular psychiatric disorders, including anxiety, depression, and insomnia, were identified according on ICD-10-CM as previously described (Supplementary Table S5) (24).
For secondary outcomes, we examined each of these psychiatric disorders individually, along with outcomes related to suicidal ideation or attempts, and dementia. Additionally, we included two outcome categories to provide a comprehensive assessment. The first, serving as a positive control, was cerebral infarction, while the second, serving as a negative control, was atopic dermatitis (25). This approach allowed us to thoroughly evaluate the impact of interventions across a diverse range of health outcomes.
The TriNetX platform’s integrated propensity score-matching function was employed to establish a 1:1 correspondence between subjects in both the ablation and non-ablation groups. The matching was executed using a nearest-neighbor greedy algorithm with a caliper limit set at 0.1 of the pooled standard deviation, optimizing the pairing of participants. Subsequently, standardized differences were calculated to evaluate the equivalence of the matched groups, with absolute values below 0.1 denoting a robust match (26).
Subsequently, we performed a Kaplan-Meier analysis, followed by log-rank tests with calculations of hazard ratios (HR) at 95% confidence intervals (CI) to compare the two groups (27). Statistical significance was set at p< 0.05. HR was used to describe the relative risk of outcomes by comparing time-to-event rates and was calculated using a proportional hazard model, which is a built-in function in TriNetX (28).
For the subgroup analysis, we compared the primary and secondary outcomes between the two groups, stratified by age (18–64 and ≥65 years), sex (male and female), race (white and non-white), and presence of comorbidities (paroxysmal AF, non-paroxysmal AF, heart failure, cancer, COPD, diabetes mellitus, dyslipidemia, and chronic liver disease) (20, 29).
The final cohort comprised 21,019 individuals who underwent catheter ablation and 21,017 individuals who did not undergo ablation, serving as the comparator cohort (Figure 1). The baseline characteristics differed significantly between the ablation and non-ablation groups. The mean age at index for the ablation group was 63.9 ± 10.4 years, whereas the non-ablation group exhibited an older demographic with a mean age of 71.5 ± 12.0 years. The sex distribution in both groups before matching showed a higher proportion of males (68.6% and 59.5% in the ablation and non-ablation groups, respectively). In terms of racial demographics, the majority were Caucasian, constituting 86.64% of the ablation group and 77.69% of the non-ablation group. Comorbidity analysis revealed that hypertensive diseases were the most prevalent, affecting 50.35% and 43.35% of the patients in the ablation and non-ablation groups, respectively. Dyslipidemia was also present, with rates of 37.47 and 33.13% in the ablation and non-ablation groups, respectively. Regarding comorbidities, conditions such as peripheral vascular diseases, neoplasms, and COPD were more common in the non-ablation group. The matching process substantially minimized the demographic and comorbidity disparities between the two groups, achieving balance through propensity score matching. (Table 1).
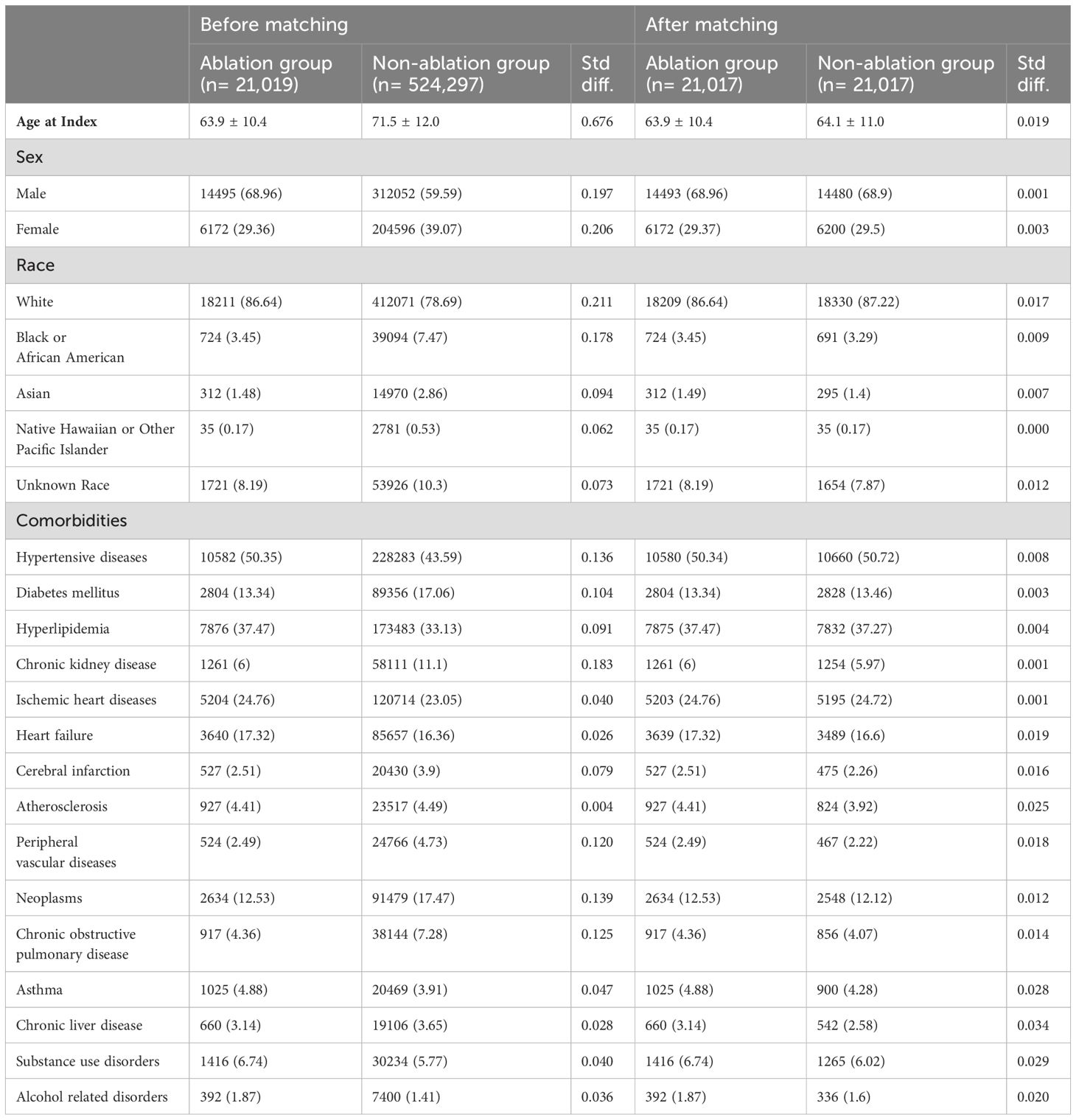
Table 1. Comparison of characteristics of ablation group and non-ablation group before and after matching.
In our propensity score-matched cohorts, the ablation group exhibited a reduced incidence of psychiatric disorders compared to the non-ablation group, with an HR of 0.873 (95% CI, 0.784–0.973) (Table 2). Specifically, during the follow-up period, 598 individuals (2.8%) with AF who underwent catheter ablation developed various psychiatric disorders, including anxiety, depression, and insomnia. In contrast, 728 individuals representing 3.5% of the group with AF who did not undergo catheter ablation had these disorders. These differences persisted consistently throughout the follow-up period, indicating that the ablation group had a lower risk of developing psychiatric disorders than the non-ablation group (log-rank p < 0.01; Figure 2).
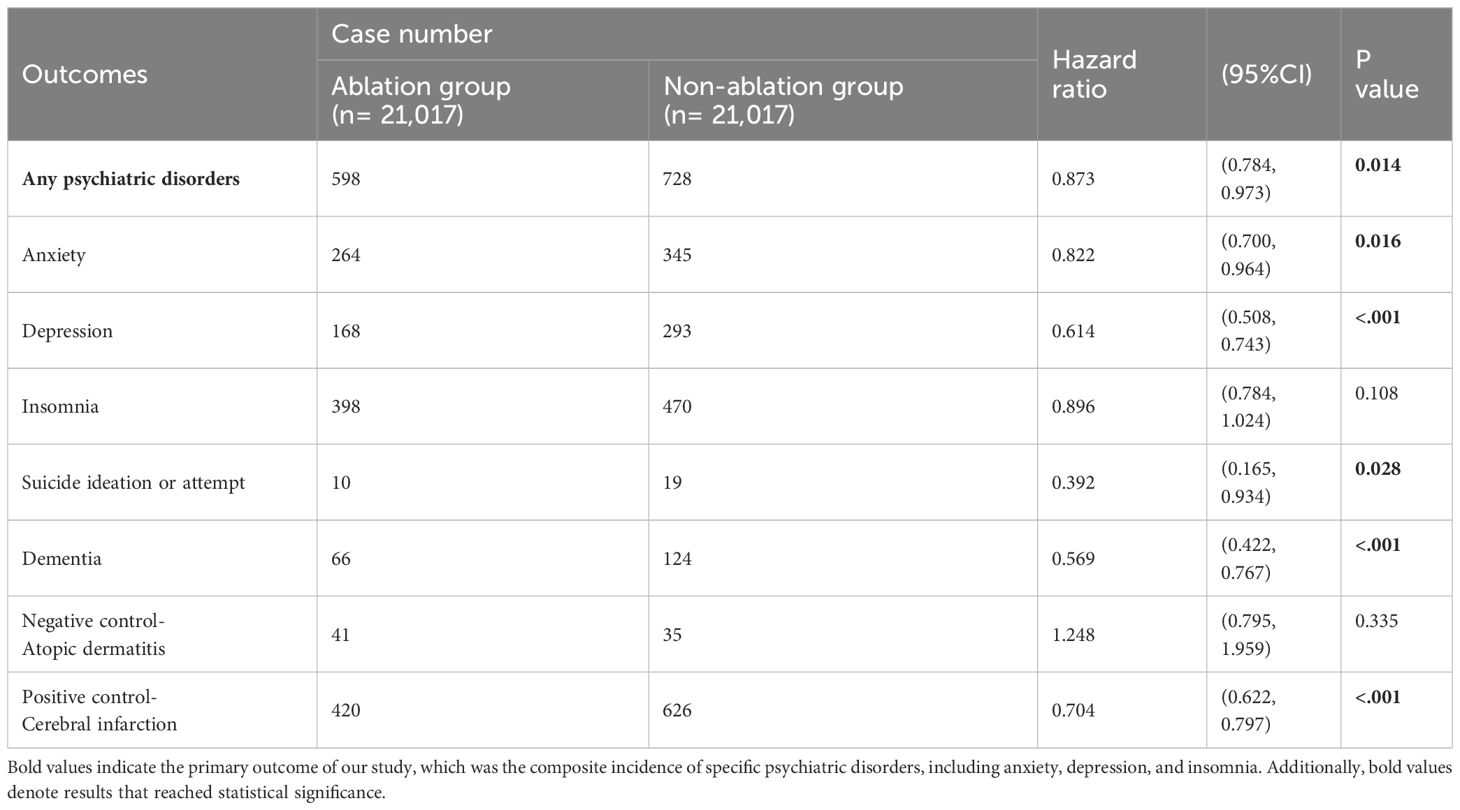
Table 2. The hazard ratio and cases number for comparing matched ablation group and non-ablation group for the primary composite outcome and secondary outcomes.
Throughout the follow-up period, the ablation group had 264 cases of anxiety, whereas the non-ablation group had 345 cases, resulting in an HR of 0.822 (95% CI:0.700–0.964; p=0.016). In the context of depression, there were 168 cases in the ablation group and 293 cases in the non-ablation group, with an HR of 0.614 (95% CI:0.508–0.743; p<0.001). Regarding suicidal ideation or attempts, there were 10 cases in the ablation group versus 19 cases in the non-ablation group, and the HR was 0.392 (95% CI:0.165–0.934; p=0.028). Regarding dementia, there were 66 cases in the ablation group and 124 cases in the non-ablation group, yielding an HR of 0.569 (95% CI:0.422–0.767; p<0.001). The HR for atopic dermatitis, the negative control, was 1.248 (95% CI:0.795–1.959; p=0.335). Eventually, for the positive control (cerebral infarction), the HR was 0.704 (95% CI:0.622–0.797; p<0.001).
In most subgroup analyses, the ablation group showed a consistently and significantly lower risk of developing psychiatric disorders (Figure 3). However, there were certain exceptions, including those aged > 65 years, non-Caucasian populations, and those with certain comorbidities. In these cases, only a non-significant lower risk of psychiatric disorders was observed.
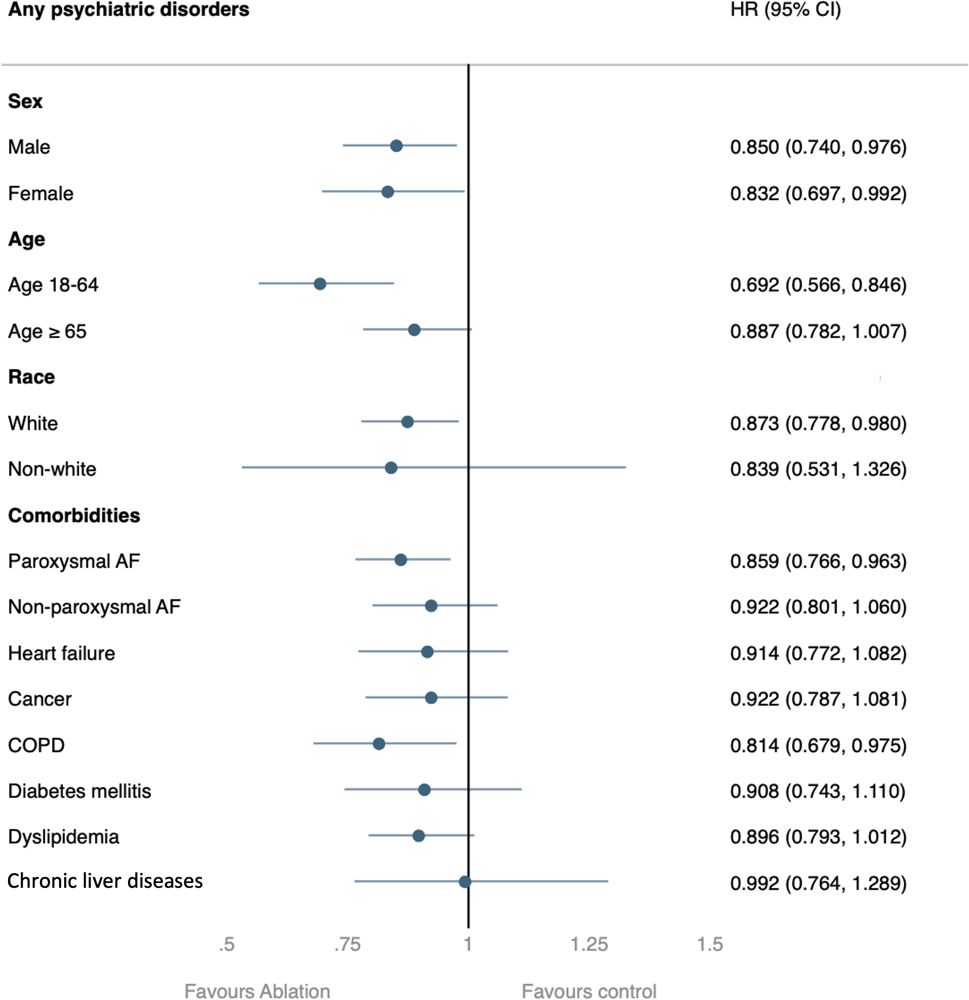
Figure 3. Subgroup analysis of the risk of the primary outcome between the ablation group and non-ablation group. AF: atrial fibrillation; COPD: chronic obstruction pulmonary disease.
In terms of individual outcomes, the analysis revealed that the ablation group exhibited a significantly lower risk of anxiety than the non-ablation group, particularly in the 18–64 years age subgroup and among patients with paroxysmal AF (Supplementary Figure S1). Concerning depression, most subgroups demonstrated a significantly lower risk in the ablation group, except for non-Caucasian individuals and those with liver diseases (Supplementary Figure S2). In the case of insomnia, only the 18–64 years age subgroup, patients with paroxysmal atrial fibrillation, and those with COPD demonstrated a reduced risk in the ablation group (Supplementary Figure S3). However, no significant differences were observed in suicide risk across the subgroups (Supplementary Figure S4). In the case of dementia, the majority of subgroups exhibited a significantly lower risk in the ablation group, except for the 18–64 age group, non-Caucasian individuals, patients with diabetes mellitus, and those with chronic liver diseases (Supplementary Figure S5). In the context of the negative control, atopic dermatitis, no significant differences were found between groups in any of the subgroup analyses (Supplementary Figure S6). Finally, for the positive control outcome of cerebral infarction, all subgroups showed a significantly lower risk in the ablation group than in the non-ablation group, except for patients with chronic liver diseases (Supplementary Figure S7).
This is the first large-scale retrospective cohort study involving 42,034 individuals to assess the effects of catheter ablation on psychiatric disorders in patients with AF. Our primary finding was that patients who underwent catheter ablation after AF diagnosis demonstrated a lower risk of psychiatric disorders. These patients were less likely to experience depression, anxiety, or insomnia than those who did not undergo catheter ablation. Importantly, this protective effect was consistently observed across different demographic subgroups, including sex, race (Caucasian or non-Caucasian), age group between 18–59 years, as well as patients with paroxysmal AF or COPD. These findings suggest that catheter ablation may serve as a protective measure to reduce the risk of psychiatric disorders in patients with AF.
Notably, our results align with those of the REMEDIAL trial conducted by Al Kaisey et al. In their study involving participants with symptomatic AF, improvements in psychological symptoms such as anxiety and depression were observed following catheter ablation, but not with medical therapy (17). Although our study shares a common conclusion with the REMEDIAL trial, it is important to acknowledge the methodological differences between the two. Notably, our study had a larger cohort size and employed diagnostic codes as opposed to rating scales to measure the outcomes. Moreover, our present work, portrays the real-world scenario, offering insights that effectively reflect the everyday clinical practice, capturing a broader range of patient populations with uncontrolled variables, thus complementing the controlled environment of randomized trials. Despite their methodological disparities, both studies conducted using varying approaches have consistently demonstrated the potential benefits of catheter ablation in preventing psychiatric disorders in patients with AF, thereby reinforcing the robustness of this conclusion.
Our secondary findings indicate that catheter ablation significantly reduces the risk of various psychiatric conditions, such as anxiety, depression, insomnia, suicidal ideation or attempt, and dementia, in patients with AF compared to those who did not undergo ablation. For a comprehensive understanding of the potential mechanisms underlying this improvement, a study conducted by Hasebe et al. discovered that augmentation of parasympathetic reactivity to stress was correlated with reduced anxiety (30). This implies that modifications in cardiac autonomic function may play a pivotal role in the psychological improvement experienced by patients after undergoing catheter ablation for AF. Additionally, it is possible that by restoring regular heart rhythm, catheter ablation leads to improved cerebral blood flow- thereby potentially alleviating symptoms of depression, anxiety, and cognitive disorders (31, 32). Furthermore, the consequent decrease in systemic inflammation, commonly associated with AF, might further play a role in reducing psychiatric symptoms (32). Studies have demonstrated that systemic inflammation might be a shared mechanism contributing to psychiatric manifestations (33). Together, these findings suggest that catheter ablation’s efficacy in mitigating psychiatric conditions might stem from its ability to modulate cardiac autonomic function, enhance cerebral blood flow through rhythm regularization, and reduce systemic inflammation (34).
The observed variations in psychiatric risk reduction following catheter ablation across different subgroups suggest that its benefits on insomnia and dementia risk are influenced by neurophysiological, autonomic, and inflammatory mechanisms, as well as patient-specific disease burden (35–38). Our findings indicate that younger adults (18–64 years), those with paroxysmal AF, and patients with COPD experienced significant reductions in insomnia risk, likely due to greater neuroplasticity, enhanced autonomic regulation, and improved cerebrovascular reserve, which facilitate recovery from AF-related hypoperfusion, oxidative stress, and systemic inflammation (37, 39–42). In COPD, the elimination of AF-related sympathetic overactivation and intermittent hypoxia may stabilize sleep and cerebral oxygenation, reducing neuropsychiatric complications (43, 44). However, dementia risk reduction was more evident in older adults (≥65 years), white patients, and those with cardiovascular comorbidities. This may reflect the greater cumulative impact of AF-related hypoperfusion and microembolic events in these populations, where sinus rhythm restoration prevents further neurovascular injury (45, 46).
In contrast, younger patients, those with DM, and individuals with liver disease did not show significant dementia risk reduction, suggesting that the protective effects of ablation may be counteracted by pre-existing metabolic and systemic conditions. Younger individuals may have a lower baseline dementia risk and greater neuroplasticity, making short-term cognitive benefits less detectable (46, 47). However, in DM and liver disease, persistent systemic inflammation, insulin resistance, endothelial dysfunction, and neurotoxic metabolic effects may continue to drive cognitive decline regardless of AF status (48–51). Given that diabetes is a major risk factor for both vascular and Alzheimer’s-type dementia, the metabolic and microvascular changes associated with DM may override the cognitive benefits of sinus rhythm restoration (52, 53). Similarly, liver disease is associated with hepatic encephalopathy, altered ammonia metabolism, and systemic inflammation, all of which can contribute to dementia independent of AF (51, 54). These findings highlight the complex interplay between cardiovascular function, metabolic disease, and brain health, highlighting the need for prospective studies to investigate the mechanisms linking catheter ablation to psychiatric outcomes across diverse patient populations.
Our study revealed that patients who underwent catheter ablation exhibited a lower risk of experiencing suicidal ideation or attempting suicide than those who did not undergo catheter ablation. This finding aligns with that of a previous cohort study conducted by Walters et al., which involved 78 patients and revealed a high prevalence of severe psychological distress (35%) and suicidal ideation (20%) among individuals with AF (9). Notably, they found that effective catheter ablation of AF was associated with a substantial improvement in both psychological distress and reported suicidal ideation. However, a key distinction between our study and that of Walters et al. lies in our cohort selection. We excluded individuals with a prior history of psychiatric disorders and focused on new-onset psychiatric conditions following a diagnosis of AF. This design choice ensured that our findings were pertinent to the development of psychiatric disorders after an AF diagnosis, offering valuable insights into this specific patient condition.
This is the first large-scale retrospective cohort study to evaluate the preventive effect of catheter ablation on psychiatric disorders in patients with AF. Despite potential biases, such as misdiagnosis or documentation errors associated with registry databases, we leveraged a comprehensive and dynamically updated population-based database. This enabled us to examine diverse global populations within the current timeframe. While propensity score matching helps balance observed covariates, we acknowledge that unobserved confounders, such as socioeconomic status, medication adherence, may still exist. To minimize this residual confounding, we conducted several sensitivity analyses and included multiple relevant covariates in our matching process. Additionally, the possibility of selective reporting of results must be considered; however, we minimized this risk by reporting all measured outcomes, including both significant and non-significant findings, to ensure transparency. Our methodological approach was further strengthened by the use of both positive and negative controls, with the latter helping validate our analytical methods. For instance, the non-significant finding for atopic dermatitis (p=0.335) serves as an appropriate negative control, as catheter ablation would not be expected to affect dermatological conditions. We specifically excluded patients with prior psychiatric disorders to ensure clean ascertainment of new-onset conditions, which strengthens our ability to establish temporal relationships, though we acknowledge this may limit generalizability to the broader AF population. While our use of diagnostic codes follows established methodology in large-scale epidemiological studies, we recognize this limits the precision of psychiatric assessment. All HR for individual psychiatric outcomes were statistically significant and less than 1, with the exception of insomnia. For insomnia, although the hazard ratio was less than one (suggesting a protective effect of catheter ablation), the result was not statistically significant (p=0.108), possibly due to limited statistical power. Similarly, despite finding a significantly reduced risk of insomnia in the subgroups of young adults, patients with COPD, and paroxysmal AF, non-significant findings were observed in subgroups with other comorbidities and older adults. This may be attributed to the smaller sample sizes in these subgroups. Further large-scale studies are warranted to assess the association with various psychiatric diseases among specific subgroups with a small sample size in this study.
This retrospective cohort study demonstrated that catheter ablation in patients with AF might be associated with a significantly lower risk of developing psychiatric disorders, including anxiety, depression, insomnia, suicidal ideation or attempt, and dementia, compared to those who did not undergo ablation. While these findings suggest potential psychiatric benefits beyond the established cardiovascular advantages of ablation, further prospective studies are needed to confirm whether this association should influence the timing of ablation decisions. When evaluating candidates for catheter ablation, clinicians may consider incorporating psychiatric risk factors as part of their comprehensive patient assessment, alongside established clinical indicators.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
The requirement for written informed consent was exempted due to the anonymized nature of the TriNetX data. The study protocol received approval from the Institutional Review Board of Chi Mei Medical Center (approval no. 11206-E02).
T-HL: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft. J-YW: Data curation, Formal analysis, Investigation, Writing – original draft. P-YH: Data curation, Formal analysis, Investigation, Writing – original draft. W-HH: Data curation, Formal analysis, Investigation, Writing – original draft. M-HC: Data curation, Formal analysis, Investigation, Writing – original draft. Y-WT: Data curation, Formal analysis, Investigation, Writing – original draft. K-YH: Conceptualization, Investigation, Supervision, Writing – review & editing. C-CL: Conceptualization, Supervision, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2025.1467876/full#supplementary-material
1. Kornej J, Benjamin EJ, Magnani JW. Atrial fibrillation: global burdens and global opportunities. Heart. (2021) 107(7):516–8. doi: 10.1136/heartjnl-2020-318480
2. Pierre-Louis IC, Saczynski JS, Lopez-Pintado S, Waring ME, Abu HO, Goldberg RJ, et al. Characteristics associated with poor atrial fibrillation-related quality of life in adults with atrial fibrillation. J Cardiovasc Med. (2023) 24:422–9. doi: 10.2459/JCM.0000000000001479
3. Piersma F, Breel J, Krul S, Eberl S, Wensing A, Deutekom F, et al. Atrial fibrillation: a retrospective chart review of complications, morbidity and mortality at 30 days. Eur Heart J. (2022) 43(Supplement_2)ehac544. 2750. doi: 10.1093/eurheartj/ehac544.2750
4. Ling L-H, Kistler PM, Kalman JM, Schilling RJ, Hunter RJ. Comorbidity of atrial fibrillation and heart failure. Nat Rev Cardiol. (2016) 13:131–47. doi: 10.1038/nrcardio.2015.191
5. Essa H, Hill AM, Lip GY. Atrial fibrillation and stroke. Card Electrophysiol Clin. (2021) 13:243–55. doi: 10.1016/j.ccep.2020.11.003
6. Yossef M, Amer R, Elsokkary H, Shama G. Psychiatric symptoms in patients with non-valvular atrial fibrillation. Middle East Curr Psychiatry. (2022) 29:105. doi: 10.1186/s43045-022-00268-z
7. Patel D, Mc Conkey ND, Sohaney R, Mc Neil A, Jedrzejczyk A, Armaganijan L. A systematic review of depression and anxiety in patients with atrial fibrillation: the mind-heart link. Cardiovasc Psychiatry Neurol. (2013) 2013(1):159850. doi: 10.1155/2013/159850
8. Serpytis R, Navickaite A, Serpytiene E, Barysiene J, Marinskis G, Jatuzis D, et al. Impact of atrial fibrillation on cognitive function, psychological distress, quality of life, and impulsiveness. Am J Med. (2018) 131(6):703. e1–e5. doi: 10.1016/j.amjmed.2017.12.044
9. Walters TE, Wick K, Tan G, Mearns M, Joseph SA, Morton JB, et al. Psychological distress and suicidal ideation in patients with atrial fibrillation: prevalence and response to management strategy. J Am Heart Assoc. (2018) 7(18):e005502. doi: 10.1161/JAHA.117.005502
10. Lakkireddy D, Atkins D, Pillarisetti J, Ryschon K, Bommana S, Drisko J, et al. Effect of yoga on arrhythmia burden, anxiety, depression, and quality of life in paroxysmal atrial fibrillation: the YOGA My Heart Study. J Am Coll Cardiol. (2013) 61(11):1177–82. doi: 10.1016/j.jacc.2012.11.060
11. Ladwig K-H, Goette A, Atasoy S, Johar H. Psychological aspects of atrial fibrillation: A systematic narrative review. Curr Cardiol Rep. (2020) 22:137. doi: 10.1007/s11886-020-01396-w
12. Rottner L, Bellmann B, Lin T, Reissmann B, Tönnis T, Schleberger R, et al. Catheter ablation of atrial fibrillation: state of the art and future perspectives. Cardiol Ther. (2020) 9(1):45–58. doi: 10.1007/s40119-019-00158-2
13. Jaïs P, Cauchemez B, Macle L, Daoud E, Khairy P, Subbiah R, et al. Catheter ablation versus antiarrhythmic drugs for atrial fibrillation: the A4 study. Circulation. (2008) 118(24):2498–505. doi: 10.1161/CIRCULATIONAHA.108.772582
14. Lin C, Sun M, Liu Y, Su Y, Liang X, Ma S, et al. Catheter ablation vs. drug therapy in the treatment of atrial fibrillation patients with heart failure: An update meta-analysis for randomized controlled trials. Front Cardiovasc Med. (2023) 10:1103567. doi: 10.3389/fcvm.2023.1103567
15. Andrade JG, Wells GA, Deyell MW, Bennett M, Essebag V, Champagne J, et al. Cryoablation or drug therapy for initial treatment of atrial fibrillation. N Engl J Med. (2021) 384(4):305–15. doi: 10.1056/NEJMoa2029980
16. Wazni OM, Dandamudi G, Sood N, Hoyt R, Tyler J, Durrani S, et al. Cryoballoon ablation as initial therapy for atrial fibrillation. N Engl J Med. (2021) 384(4):316–24. doi: 10.1056/NEJMoa2029554
17. Al-Kaisey AM, Parameswaran R, Bryant C, Anderson RD, Hawson J, Chieng D, et al. Atrial fibrillation catheter ablation vs medical therapy and psychological distress: A randomized clinical trial. JAMA. (2023) 330(10):925–33. doi: 10.1001/jama.2023.14685
18. Liu TH, Liao HY, Chang CC, Lai CC. Effectiveness of molnupiravir for treating COVID-19 in patients with psychiatric disorders. Front Pharmacol. (2024) 15:1384264. doi: 10.3389/fphar.2024.1384264
19. Liu TH, Wu JY, Huang PY, Hsu WH, Chuang MH, Tsai YW, et al. Clinical effectiveness of nirmatrelvir plus ritonavir for patients with COVID-19 and preexisting psychiatric disorders. Expert Rev anti-infective Ther. (2024) 22(10):885–93. doi: 10.1080/14787210.2024.2351555
20. Harrison SL, Buckley BJ, Austin P, Lane DA, Lip GY. Catheter ablation and lower risk of incident dementia and mortality in older adults with atrial fibrillation. J Am Geriatr Soc. (2023) 71(11):3357–66. doi: 10.1111/jgs.v71.11
21. Liu TH, Wu JY, Huang PY, Tsai YW, Lai CC. The effect of nirmatrelvir plus ritonavir on the long-term risk of epilepsy and seizure following COVID-19: A retrospective cohort study including 91,528 patients. J Infect Mar. (2023) 86:256–308. doi: 10.1016/j.jinf.2023.01.014
22. Liu T-H, Huang P-Y, Wu J-Y, Chuang M-H, Hsu W-H, Tsai Y-W, et al. Post-COVID-19 condition risk in patients with intellectual and developmental disabilities: a retrospective cohort study involving 36,308 patients. BMC Med. (2023) 21:505. doi: 10.1186/s12916-023-03216-8
23. Liu TH, Chuang MH, Wu JY, Huang PY, Tsai YW, Hsu WH, et al. Effectiveness of oral antiviral agents on long-term cardiovascular risk in nonhospitalized patients with COVID-19: A multicenter matched cohort study. J Med Virol. (2023) 95(8):e28992. doi: 10.1002/jmv.28992
24. Liu TH, Wu JY, Huang PY, Tsai YW, Lai CC. The effect of nirmatrelvir-ritonavir on the long-term risk of neuropsychiatric sequelae following COVID-19. J Med Virol. (2023) 95:e28951. doi: 10.1002/jmv.28951
25. Taquet M, Dercon Q, Luciano S, Geddes JR, Husain M, Harrison PJ. Incidence, co-occurrence, and evolution of long-COVID features: A 6-month retrospective cohort study of 273,618 survivors of COVID-19. PloS Med. (2021) 18:e1003773. doi: 10.1371/journal.pmed.1003773
26. Haukoos JS, Lewis RJ. The propensity score. JAMA. (2015) 314:1637–8. doi: 10.1001/jama.2015.13480
27. Liu T-H, Wu J-Y, Huang P-Y, Tsai Y-W, Lai C-C. Effect of COVID-19 on the long-term cardiovascular outcomes among patients with alcohol use disorder: A retrospective cohort study including 45,842 patients. J Addict Med. (2023) 17(6):e382–7. doi: 10.1097/adm.0000000000001232
28. Liu T-H, Ho C-H, Chen DT-L, Wu J-Y, Huang P-Y, Lai C-C, et al. Omega-3 polyunsaturated fatty acids and the psychiatric post-acute sequelae of COVID-19: A one-year retrospective cohort analysis of 33,908 patients. Brain Behav Immun. (2023) 114:453–61. doi: 10.1016/j.bbi.2023.09.008
29. Liu T-H, Hsu W-H, Tsai Y-W, Wu J-Y, Huang P-Y, Chuang M-H, et al. Clinical effectiveness of nirmatrelvir plus ritonavir in the treatment of COVID-19 for patients with dementia. J Am Med Dir Assoc. (2023) 24(8):1159–62. doi: 10.1016/j.jamda.2023.05.034
30. Hasebe H, Shinba T. Decreased anxiety after catheter ablation for paroxysmal atrial fibrillation is associated with augmented parasympathetic reactivity to stress. Heart Rhythm O2. (2020) 1:189–99. doi: 10.1016/j.hroo.2020.05.008
31. Wutzler A, Nee J, Boldt LH, Kühnle Y, Gräser S, Schröder T, et al. Improvement of cerebral oxygen saturation after successful electrical cardioversion of atrial fibrillation. Europace Feb. (2014) 16(2):189–94. doi: 10.1093/europace/eut246
32. Chiappelli J, Adhikari BM, Kvarta MD, Bruce HA, Goldwaser EL, Ma Y, et al. Depression, stress and regional cerebral blood flow. J Cereb Blood Flow Metab. (2023) 43(5):791–800. doi: 10.1177/0271678X221148979
33. Réus GZ, Fries GR, Stertz L, Badawy M, Passos I, Barichello T, et al. The role of inflammation and microglial activation in the pathophysiology of psychiatric disorders. Neuroscience. (2015) 300:141–54. doi: 10.1016/j.neuroscience.2015.05.018
34. Su K-p. Inflammation in psychopathology of depression: clinical, biological, and therapeutic implications. Biomedicine. (2012) 2:68–74. doi: 10.1016/j.biomed.2012.03.002
35. Bunch TJ, Bair TL, Crandall BG, Cutler MJ, Day JD, Graves KG, et al. Stroke and dementia risk in patients with and without atrial fibrillation and carotid arterial disease. Heart rhythm Jan. (2020) 17:20–6. doi: 10.1016/j.hrthm.2019.07.007
36. Mittal S, Musat DL, Hoskins MH, Prillinger JB, Roberts GJ, Nabutovsky Y, et al. Clinical outcomes after ablation of the AV junction in patients with atrial fibrillation: impact of cardiac resynchronization therapy. J Am Heart Assoc. (2017) 6(12):e007270. doi: 10.1161/jaha.117.007270
37. Al-Bashaireh AM, Alkouri O, Alharbi A, Khader Y, Hammoudeh A, Aljawarneh Y, et al. Factors associated with quality of life among people with atrial fibrillation: Jordan atrial fibrillation registry study. Medicina (Kaunas Lithuania). (2024) 60(8):1262. doi: 10.3390/medicina60081262
38. Alonso A, Arenas de Larriva AP. Atrial fibrillation, cognitive decline and dementia. Eur Cardiol Summer. (2016) 11:49–53. doi: 10.15420/ecr.2016:13:2
39. Wang YH, Lai CC, Wang CY, Wang HC, Yu CJ, Chen L. Risks of pneumonia in COPD patients with new-onset atrial fibrillation. J Clin Med. (2018) 7(9):229. doi: 10.3390/jcm7090229
40. Simons SO, Elliott A, Sastry M, Hendriks JM, Arzt M, Rienstra M, et al. Chronic obstructive pulmonary disease and atrial fibrillation: an interdisciplinary perspective. Eur Heart J. (2021) 42(5):532–40. doi: 10.1093/eurheartj/ehaa822
41. Harada M, Van Wagoner DR, Nattel S. Role of inflammation in atrial fibrillation pathophysiology and management. Circ J. (2015) 79:495–502. doi: 10.1253/circj.CJ-15-0138
42. Ren X, Wang X, Yuan M, Tian C, Li H, Yang X, et al. Mechanisms and treatments of oxidative stress in atrial fibrillation. Curr Pharm Design. (2018) 24(26):3062–71. doi: 10.2174/1381612824666180903144042
43. Linz D, Elliott AD, Hohl M, Malik V, Schotten U, Dobrev D, et al. Role of autonomic nervous system in atrial fibrillation. Int J Cardiol. (2019) 287:181–8. doi: 10.1016/j.ijcard.2018.11.091
44. Lu Z, Nie L, He B, Yu L, Salim M, Huang B, et al. Increase in vulnerability of atrial fibrillation in an acute intermittent hypoxia model: importance of autonomic imbalance. Autonomic Neurosci. (2013) 177(2):148–53. doi: 10.1016/j.autneu.2013.03.014
45. Kochhäuser S, Lohmann H, Ritter M, Leitz P, Güner F, Zellerhoff S, et al. Neuropsychological impact of cerebral microemboli in ablation of atrial fibrillation. Clin Res Cardiol. (2015) 104:234–40. doi: 10.1007/s00392-014-0777-0
46. Anselmino M, Scarsoglio S, Saglietto A, Gaita F, Ridolfi L. Transient cerebral hypoperfusion and hypertensive events during atrial fibrillation: a plausible mechanism for cognitive impairment. Sci Rep. (2016) 6:28635. doi: 10.1038/srep28635
47. Chen J-H, Lin K-P, Chen Y-C. Risk factors for dementia. J Formosan Med Assoc. (2009) 108:754–64. doi: 10.1016/S0929-6646(09)60402-2
48. Li Y, Liu Y, Liu S, Gao M, Wang W, Chen K, et al. Diabetic vascular diseases: molecular mechanisms and therapeutic strategies. Signal Transduct Target Ther. (2023) 8:152. doi: 10.1038/s41392-023-01400-z
49. Lontchi-Yimagou E, Sobngwi E, Matsha TE, Kengne AP. Diabetes mellitus and inflammation. Curr Diabetes Rep. (2013) 13:435–44. doi: 10.1007/s11892-013-0375-y
50. Laleman W, Claria J, van der Merwe S, Moreau R, Trebicka J. Systemic inflammation and acute-on-chronic liver failure: too much, not enough. Can J Gastroenterol Hepatol. (2018) 2018:1027152. doi: 10.1155/2018/1027152
51. Ochoa-Sanchez R, Tamnanloo F, Rose CF. Hepatic encephalopathy: from metabolic to neurodegenerative. Neurochem Res. (2021) 46:2612–25. doi: 10.1007/s11064-021-03372-4
52. Athanasaki A, Melanis K, Tsantzali I, Stefanou MI, Ntymenou S, Paraskevas SG, et al. Type 2 diabetes mellitus as a risk factor for Alzheimer’s disease: Review and meta-analysis. Biomedicines. (2022) 10(4):778. doi: 10.3390/biomedicines10040778
53. Ahtiluoto S, Polvikoski T, Peltonen M, Solomon A, Tuomilehto J, Winblad B, et al. Diabetes, Alzheimer disease, and vascular dementia: a population-based neuropathologic study. Neurology. (2010) 75:1195–202. doi: 10.1212/WNL.0b013e3181f4d7f8
Keywords: atrial fibrillation, catheter ablation, anxiety, depression, insomnia, suicidal ideation or attempt, dementia
Citation: Liu T-H, Wu J-Y, Huang P-Y, Hsu W-H, Chuang M-H, Tsai Y-W, Hsieh K-Y and Lai C-C (2025) Association between catheter ablation and psychiatric disorder risk in adults with atrial fibrillation: a multi-institutional retrospective cohort study. Front. Psychiatry 16:1467876. doi: 10.3389/fpsyt.2025.1467876
Received: 21 July 2024; Accepted: 03 March 2025;
Published: 21 March 2025.
Edited by:
Vikash Jaiswal, JCCR Cardiology Research, IndiaReviewed by:
Danisha Kumar, Dow University of Health Sciences, PakistanCopyright © 2025 Liu, Wu, Huang, Hsu, Chuang, Tsai, Hsieh and Lai. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kuang-Yang Hsieh, a3loc2llaC50d0BnbWFpbC5jb20=; Chih-Cheng Lai, ZHRtZWQxNDFAZ21haWwuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.