
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Psychiatry , 25 February 2025
Sec. Public Mental Health
Volume 16 - 2025 | https://doi.org/10.3389/fpsyt.2025.1446897
Introduction: Previous studies have found that depressive patients tend to have low levels of creatinine; however, the extent to which creatinine levels are associated with depression has been poorly investigated. Therefore, this study aimed to explore the relationship between creatinine levels and depression.
Methods: The participants and follow-up data from the China Health and Retirement Longitudinal Study (CHARLS), as well as metabolomics data from the Metabolite Network of Depression Database (MENDA), were collected. The 10-item Center for Epidemiologic Studies Depression Scale (CESD-10) was used to assess the severity of depression. Spearman correlation analysis, spline regression, and binary logistic regression models were employed to explore the relationship between creatinine levels and depression.
Results: A total of 7,826 participants and 3,886 follow-up participants were included in the CHARLS 2011 and 2015 surveys. Of these, 37.9% (2,966/7,826) and 34.6% (13,44/3,886) of participants experienced depression in CHARLS 2011 and 2015, respectively. The creatinine level was negatively correlated with the total CESD-10 score and dimensions scores, showing an inverse dose–response relationship between creatinine levels and depression. Compared with participants with high creatinine levels, those with middle creatinine levels were associated with a higher risk of depression (OR = 1.22, 95% CI = 1.08–1.38), while participants with low creatinine levels had the highest risk of depression (OR = 1.30, 95% CI = 1.13–1.49) in the fully adjusted model. Similar results were observed in the follow-up data, and the MENDA metabolomics data validated the negative correlation between creatinine levels and the severity of depression.
Conclusion: Lower levels of creatinine were closely associated with a higher risk of depression, and it could serve as a potential marker for identifying individuals at high risk of depression.
Depression is a serious mental illness characterized by persistent sadness and loss of interest (1), which significantly impacts the quality of life. Previous studies have shown that the lifetime prevalence of depression is 6.8%–20.6% (2, 3), and it has become one of the leading causes of years lived with disability worldwide (4). As depression and depressive symptoms are dynamic and changeable throughout a person’s life, they are common in the population, especially among middle-aged and elderly adults (5). As China gradually transitions into an aging society, this challenge will become increasingly serious.
In recent years, some available and economical blood biomarkers have been proposed (6–8). Creatinine, a metabolite in the blood, comes from normal muscle wear and tear and can be filtered through the glomerulus and then excreted in urine. It is widely used in the evaluation of renal function, such as the glomerular filtration rate. Some studies have found that depression is related to a decline in renal function (9–11). In addition, creatinine levels are parallel to muscle mass, so they can also be used as an evaluation indicator of muscle mass (12, 13). Some studies have also found that muscle loss may be a risk factor for depression (14, 15). Nevertheless, in most of these studies, creatinine has been used as an indicator to evaluate renal function and sarcopenia, and the direct relationship between creatinine and depression has not been clarified.
Furthermore, increasing evidence shows that energy metabolism is disturbed in depressive patients (15–17). Creatine, an important intermediate of energy metabolism, is widely involved in the recycling process of adenosine phosphate (ATP) (18–20). Importantly, creatinine is the metabolite of creatine, so we speculate that creatinine may be closely related to depression. Some metabolomic evidence has emerged (21–23). For example, different teams from China both found that creatinine levels were downregulated in the urine of chronic unpredictable mild stress rats (21, 23). Interestingly, urine creatinine levels were increased in depression patients after being treated with antidepressants (22). More importantly, creatinine levels are a common indicator that can be directly accessed from the blood in clinical practice. Thus, this study aims to explore the relationship between creatinine and depression through the China Health and Retirement Longitudinal Study (CHARLS).
CHARLS is an ongoing national longitudinal survey designed to better understand the socioeconomic determinants and consequences of aging. The survey collects comprehensive data on demographics, physical and psychosocial health, and socioeconomic factors (24). In the CHARLS baseline survey, participants were selected using multi-stage probability sampling. Specifically, 150 counties were randomly selected from 28 provinces, and three villages or communities in each county were selected using the probability-proportional-to-size (PPS) sampling technique (24). The initial sample of CHARLS (wave 1: 2011–2012) includes 17,708 individuals, with follow-up occurring every 2 years (25). Furthermore, the study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline. More detailed information is available on the CHARLS project website (http://charls.pku.edu.cn/). We downloaded the harmonized data and blood data from the official website for further analysis.
To minimize bias, demographic covariates were also included for adjustment when evaluating the association between creatinine and depression. These covariates included age, gender, body mass index (BMI), education level, marital status, residence, smoking, drinking, hypertension, diabetes, heart problems, kidney problems, and liver problems, all of which were extracted from the questionnaire. Education level was categorized as “less than lower secondary”, “upper secondary and vocal training”, and “tertiary”. Marital status was categorized as “married” and “unmarried”. Residence was categorized as “urban” and “rural”. Health-related behaviors included whether the respondent reported ever smoking (“no”, “yes”) or drinking any alcohol before (“no”, “yes”). Disease histories included whether the respondent reported being diagnosed with hypertension, diabetes, heart, kidney, or liver problems (“no”, “yes”).
The well-trained staff of the China Center for Disease Control and Prevention (CDC) collected venous blood from each participant and sent it to the local laboratory at 4°C. The blood samples were separated into plasma and buffy coat, frozen at − 20°C, and sent to China CDC in Beijing within 2 weeks. Finally, they were stored at − 80°C until tested at the Youanmen Center for Clinical Laboratory of Capital Medical University. Serum creatinine was measured using a rate-blanked and compensated Jaffe creatinine method (26). Notably, CHARLS only collected blood samples in 2011 (wave 1) and 2015 (wave 3), so this study includes only the 2011 baseline data and 2015 follow-up data. The participants from wave 1 are shown in Figure 1.
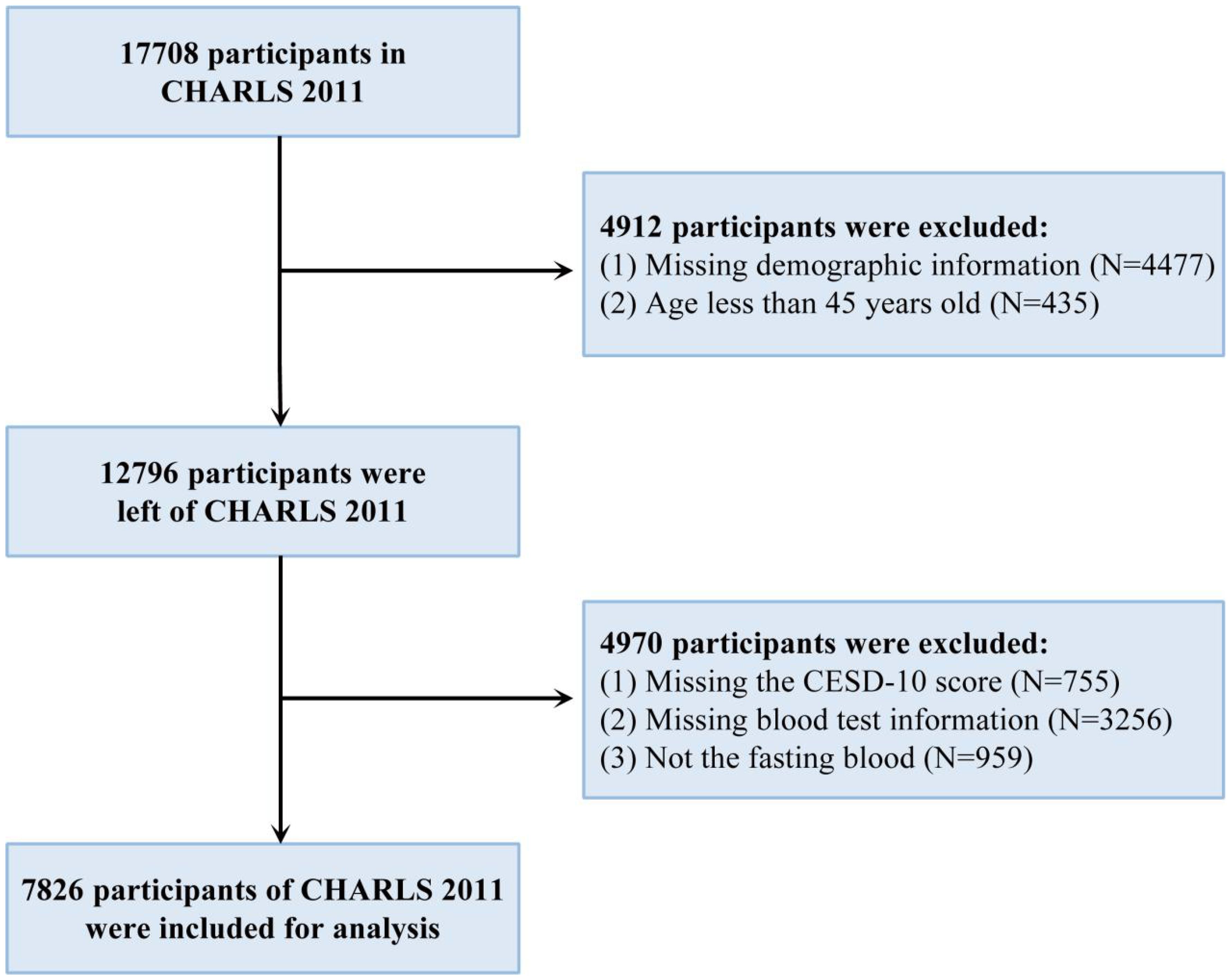
Figure 1. The flowchart of the included participants in wave 1. CHARLS, China Health and Retirement Longitudinal Study; CESD-10, 10-item center for epidemiologic studies depression scale.
Depression was evaluated using the 10-item Center for Epidemiological Studies Depression Scale (CESD-10). The scale consists of 10 items, each rated on a 3-point (0, 1, 2, 3) Likert scale. Responses varied from “rarely or none of the time (< 1 day)”, “some or a little of the time (1–2 days)”, “occasionally or a moderate amount of time (3–4 days)”, and “most or all of the time (5–7 days)”, respectively. Therefore, the total score on the CESD-10 scale ranged from 0 to 30. Depression was considered present when the cumulative score was ≥ 10 (11, 27, 28). The CESD-10 scale has been shown to have good reliability and validity in the Chinese population (29).
For this study, we used data from CHARLS in 2011 and 2015. The inclusion criteria were as follows: (1) age ≥ 45 years old; (2) availability of completed blood test information; and (3) availability of completed CESD-10 information. The exclusion criteria were as follows: (1) age < 45 years; (2) lack of basic demographic information; (3) lack of blood test information; (4) nonfasting blood test; and (5) missing CESD-10 information.
All statistical analyses were performed in R (version 4.1.0) and SPSS (version 25.0). Mean and standard deviation were used to describe quantitative data, while frequency and percentage were used to describe qualitative data. Spearman correlation coefficient was used to explore the correlation between creatinine levels and CESD-10 scores. The dose–response relationship between creatine levels and depression was preliminarily examined using spline regression models. Subsequently, creatinine levels were divided into three groups (< 0.689, 0.689–0.836, and ≥ 0.836 mg/dL) according to the tertiles. The t-test, one-way ANOVA, or Chi-square test was used to describe the difference between the groups.
A binary logistic regression model was used to explore the relationship between creatinine levels and depression. Model 1 was an unadjusted model; model 2 adjusted for the basic demographic information, including age, gender, BMI, education level, marital status, and residence; model 3 adjusted for health-related behaviors, including drinking and smoking; model 4 adjusted for disease history, including hypertension, diabetes, heart problems, kidney problems, and liver problems; model 5 was the fully adjusted model, including age, gender, education level, marital status, residence, drinking, smoking, hypertension, diabetes, heart problems, kidney problems, and liver problems. A p-value < 0.05 (two-sided) was considered to be statistically significant.
As described in the Figure 1, a total of 7,826 participants were included in the main analysis. The mean age of the participants was 59.11 years ± 9.23 years old; 3,681 (47.0%) participants were men. Most of the participants had an education level of less than lower secondary, were married, lived in rural areas, and did not have a history of smoking, drinking, hypertension, diabetes, heart problems, kidney problems, or liver problems (Table 1). A total of 37.9% (2,966/7,826) of the participants experienced depression.
Correlation analysis results showed that the creatinine level was negatively correlated with the total score and each dimension score of the CESD-10 scale (p < 0.05, Supplementary Figure S1). We then used a spline regression model to preliminary explore the shape of the association between creatinine levels and depression. The restricted cubic spline function used the default three knots (at the 10th, 50th, and 90th percentiles). Both the unadjusted and fully adjusted models showed a dose–response relationship between the creatinine levels and depression (Figure 2). Therefore, we divided the participants into groups according to the tertiles of creatinine levels for further analysis.
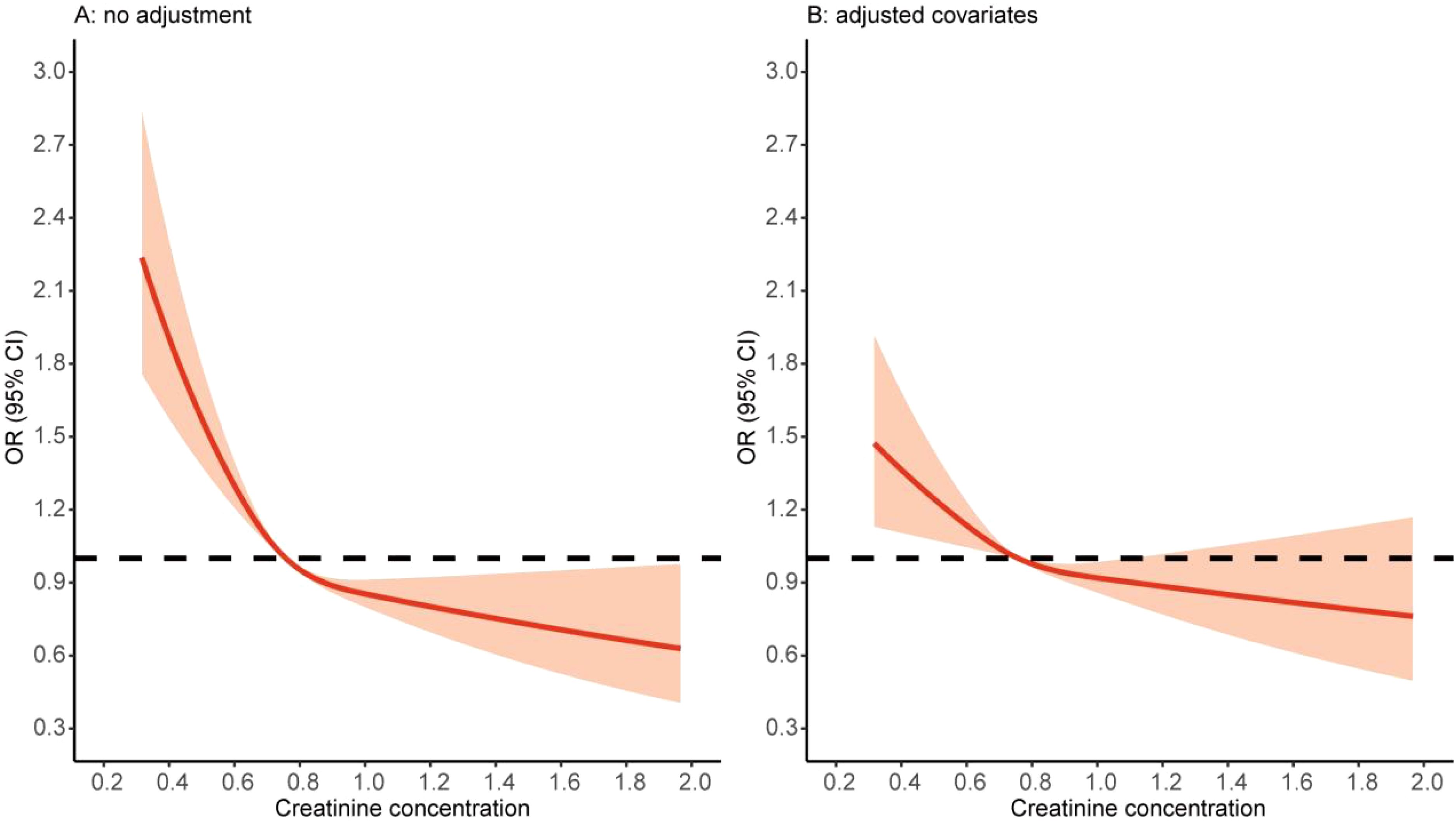
Figure 2. Restricted cubic spline of the creatinine and depression. (A) Without any adjustment of demographic covariates. (B) Adjustment for all demographic covariates, including age, sex, BMI, education level, marital status, residence, smoking, drinking, hypertension, diabetes, heart problems, kidney problems, and liver problems. OR, odds ratio; CI, confidence interval.
Univariate analysis showed a significant statistical significance in age, gender, education level, marital status, residence, drinking, smoking, hypertension, heart problems, and kidney problems. Compared with the lower creatinine level group, the higher creatinine level group tended to be older, have a higher proportion of men, have higher education, be married, live in urban areas, and have a history of smoking, drinking, hypertension, heart problems, and kidney problems (Table 1). There was no statistical significance for BMI, diabetes, and liver problems (p > 0.05).
Compared with the lower creatinine level group, the one-way ANOVA results showed that the higher creatinine level group had a lower depression score (Table 2), regardless of the total CESD-10 score or each dimension score (Figure 3). Almost all of the dimension items showed a downward trend (p < 0.05). Moreover, the proportion of participants with depression was significantly lower (Table 1).

Figure 3. CESD-10 score tendency among three groups according to the tertiles of creatinine level. (A) Total CESD-10 score and creatinine level. (B) Dimension score of CESD-10 and creatinine. CESD-10, 10-item center for epidemiological studies depression scale.
In addition, we also explored the source dimensions of depression. The mean value of the total score on the CESD-10 scale was 0.852, while the items Fearl-10, Goingl-7, and Flonel-5 were far lower than the threshold. In contrast, the items Fhopel-9, Sleeprl-3, and Whappyl-4 were far higher than the threshold, indicating that the depression in middle-aged and elderly adults in China mainly stemmed from the items Fhopel-9, Sleeprl-3, and Whappyl-4.
The logistic regression model results are shown in Table 3. In the unadjusted model, participants with lower creatinine levels were more likely to suffer from depression (middle level: OR = 1.41, 95% CI = 1.26–1.57; low level: OR = 1.67, 95% CI = 1.49–1.88). To exclude the influence of confounding covariates, models 2, 3, and 4 adjusted for demographic information, health-related behaviors, and disease histories, respectively, and the results were consistent with those of the crude model. In model 5 (fully adjusted model), compared with the higher creatinine level group, the OR of the middle-level group was 1.22 (95% CI = 1.08-1.38), and for the low-level group, it was 1.30 (95% CI = 1.13–1.49), respectively. The p-values for the trend in all the models were statistically significant. The above results indicate that a lower creatinine level is significantly associated with a higher risk of depression.
In the follow-up data of CHARLS 2015 (wave 3), a total of 3,886 participants with blood tests were included (Supplementary Table S1). Of these, 34.6% (1,344/3,886) of the participants experienced depression. Consistent with the above results, the total CESD-10 score decreased as the creatinine level increased (Supplementary Table S2). Depression was primarily associated with the dimension items Fhopel-5, Sleeprl-7, and Whappyl-8 (Supplementary Figure S2). Both the crude model and adjusted model found that lower creatinine level was significantly associated with a higher risk of depression (Supplementary Table S3).
Based on the depression measured in wave 1 and wave 3, we divided the participants into four groups: nondepression (NN), depression-relief (YN), new-onset depression (NY), and persistent depression (YY), and compared their CESD-10 score and creatinine levels (Supplementary Figure S3). The paired t-test showed that the creatinine levels of participants in all four groups in 2015 were generally significantly higher than those in 2011 (p < 0.05), which was consistent with the fact that the depression rate in 2015 was lower than that in 2011. The CESD-10 score in the YN and NY groups in 2015 was significantly different from 2011 (p < 0.01), while there was no statistical difference between the NN and YY groups (p > 0.05). Furthermore, we compared the CESD-10 score and creatinine level of each group in 2015. The results showed that the CESD-10 score of the YN group was significantly higher than that of the NN group (p < 0.01), and the creatinine level was significantly lower than that of the NN group (p = 0.03), indicating that the YN group had more severe depression, and the creatinine level had not returned to normal. Meanwhile, the CESD-10 score of the YY group was significantly higher than that of the NY group (p < 0.01), and the creatinine level was significantly lower in the YY group (p = 0.03), indicating the participants with a long-term depression course exhibited lower creatinine levels and more severe depression.
Furthermore, we explored the correlation of creatinine concentration between wave 1 and wave 3. The Spearman analysis results showed a correlation coefficient of 0.70 (p < 0.001, Supplementary Figure S4), which indicates that the baseline and follow-up creatinine levels were consistent and stable.
Furthermore, we validated the correlation between creatinine levels and the severity of depression at the metabolomics level by downloading and analyzing creatinine metabolic data and Hamilton Depression Scale (HAMD) scores of major depressive disorder (MDD) patients from the Metabolite Network of Depression Database (MENDA, http://menda.cqmu.edu.cn:8080/index.php) (30). Correlation analysis showed that the creatinine level was significantly negatively correlated with the HAMD score both in the plasma (r = − 0.40, p < 0.001) and urine (r = − 0.44, p < 0.001) of adult MDD patients. Additionally, we observed that the creatinine level was significantly negatively correlated with the HAMD score in the plasma (r = − 0.84, p < 0.001) of MDD patients in children and adolescents (Supplementary Figure S5).
Depression is a mental disease that seriously damages the social function of patients (31, 32) and is widespread among middle-aged and elderly adults (25, 28). However, depression is often overlooked and undertreated, as patients are frequently burdened with other chronic conditions, such as hypertension, diabetes, and heart disease (33–35). In addition, some studies have found that the effect of antidepressants on young people is significantly better than on the elderly (36, 37). As China becomes the world’s fastest aging country, there is an urgent need to closely address depression in middle-aged and elderly adults.
In this study, we found that serum creatinine concentration was negatively correlated with depression, and lower creatinine levels were associated with a higher risk of depression, based on data from a large, nationally representative cohort of middle-aged and elderly adults in China. Even after adjusting for many possible confounding variables, our study still provides convincing results. To our knowledge, this is the first study to explore the relationship between creatinine and depression in this population.
Creatine is a component of vertebrate muscle, synthesized in the liver and transported throughout the body, where it is then phosphorylated to store energy for muscle contraction. It is widely involved in the recycling process of adenosine phosphate (18–20) and is closely related to depression (19). For example, animal models of depression have found that creatine is downregulated in various tissues, such as the hippocampus, prefrontal lobe, plasma, serum, and urine (38–42). In addition, Zhou et al. observed that creatine was downregulated in the plasma of children and adolescents with depression through metabolomics (43). While exogenous supplementation of creatine or ATP can alleviate depression (44, 45), the possible explanation is that creatine can accelerate the clinical response to selective serotonin reuptake inhibitors, promote the differentiation of neurons, and thus improve the overall efficacy (17, 18, 46). Therefore, creatine’s effect on energy metabolism may be part of the basis for its treatment of depression.
Creatinine is the crystal end product of creatine metabolism, which exists in urine, muscle, and blood. Serum creatinine is the product of human muscle metabolism. Previous studies have found that creatinine is disturbed in depression. For example, Peng et al. found that the creatinine level in young depressive patients was decreased (47). Paradoxically, Lee et al. found that the creatinine level in Korean female depressive patients was increased, with no significant change in men (48). This could be due to the differences in eating habits and geography. Nevertheless, the vast majority of studies favored a lower creatinine level in depressive patients. For example, Pu et al. found a significant decrease in creatinine in the peripheral blood of depressive patients through a meta-analysis (7). Zhou et al. found a significant decrease in creatinine in the plasma of children and adolescents with depression through metabolomics (43). Using a data-driven approach to integrate metabolomics data from existing animal models of depression, Pu et al. found that creatinine was decreased in various tissues (19). After the antidepressant intervention, the creatinine or creatine levels in both blood and urine were significantly upregulated (49–51). Moreover, Niklasson et al. found that the creatinine level in cerebrospinal fluid was negatively correlated with suicidal ideation in depressive patients (52). Consistent with the above findings, we found that creatinine level was negatively correlated with CESD-10 score and HAMD score and that there was a dose–response relationship between creatinine level and depression. This association persisted even after we adjusted for demographic information, health-related behaviors, and disease histories. These results suggest that the creatinine level is disturbed in depression, regardless of tissue type, age, and species, and that creatinine may play a key role in the development of depression.
Moreover, we also explored the sources of depression and found that positive factors and sleep restlessness were the main sources of depression. According to previous studies, poor sleep is common among middle-aged and elderly adults (53, 54), and it has exceeded 20% in China (55, 56). It is well-known that sleep deprivation can lead to decreased alertness and difficulty in performing mental tasks (18), whereas subjects treated with creatine showed a lower decay of psychomotor activity attenuation and improved mood in tests (57, 58). These studies illustrate the effectiveness of creatine in improving sleep deprivation and mental fatigue, providing some evidence for guiding the alleviation of depression among middle-aged and elderly adults in China.
Taken together, our study provides evidence that a lower level of creatinine is independently associated with a higher risk of depression in Chinese middle-aged and elderly adults. These findings may contribute to the further development of depression biomarkers.
There are also some limitations in this study. Firstly, we could not determine the causal relationship between creatinine and depression. Secondly, depression was assessed using the CESD-10, a subjective self-assessment scale, instead of evaluation by a professional psychiatry doctor. Thirdly, although we adjusted for some confounding variables, there may be other factors, such as the family histories of depression and renal function, leading to potential residual bias. Fourthly, body composition and physical exercise may affect creatinine levels; however, the cohort did not provide this information. Therefore, the impact of these factors on depression warrants further research. Fifthly, muscles not only produce creatinine but also brain-derived neurotrophic factor (BDNF), which is significantly associated with depression. Thus, BDNF should also be considered in subsequent studies. Sixthly, a longer follow-up period is needed to confirm the relationship between creatinine and depression. Lastly, further studies are required to explore the molecular mechanisms linking creatinine and depression.
The data in this study were publicly available datasets, which can be found at http://charls.pku.edu.cn/.
The studies involving humans were approved by Biomedical Ethics Review Committee of Peking University (IRB00001052-11015). The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
FL: Conceptualization, Data curation, Formal analysis, Writing – original draft, Writing – review & editing. XZ: Data curation, Formal analysis, Methodology, Writing – original draft. CW: Conceptualization, Data curation, Formal analysis, Supervision, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
We would like to thank the participants involved in this study. We also extend our thanks to the National Development Institute of Peking University and the Chinese Center for Social Sciences Survey of Peking University for providing the CHARLS data.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2025.1446897/full#supplementary-material
1. Mann JJ. The medical management of depression. N Engl J Med. (2005) 353:1819–34. doi: 10.1056/NEJMra050730
2. Hasin DS, Sarvet AL, Meyers JL, Saha TD, Ruan WJ, Stohl M, et al. Epidemiology of adult DSM-5 major depressive disorder and its specifiers in the United States. JAMA Psychiatry. (2018) 75:336–46. doi: 10.1001/jamapsychiatry.2017.4602
3. Huang Y, Wang Y, Wang H, Liu Z, Yu X, Yan J, et al. Lu J et al: Prevalence of mental disorders in China: a cross-sectional epidemiological study. Lancet Psychiatry. (2019) 6:211–24. doi: 10.1016/S2215-0366(18)30511-X
4. Disease GBD, Injury I, Prevalence C. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. (2018) 392:1789–858. doi: 10.1016/S0140-6736(18)32279-7
5. Sousa RM, Ferri CP, Acosta D, Albanese E, Guerra M, Huang Y, et al. Pichardo GR et al: Contribution of chronic diseases to disability in elderly people in countries with low and middle incomes: a 10/66 Dementia Research Group population-based survey. Lancet. (2009) 374:1821–30. doi: 10.1016/S0140-6736(09)61829-8
6. Pan JX, Xia JJ, Deng FL, Liang WW, Wu J, Yin BM, et al. Wang HY et al: Diagnosis of major depressive disorder based on changes in multiple plasma neurotransmitters: a targeted metabolomics study. Transl Psychiatry. (2018) 8:130. doi: 10.1038/s41398-018-0183-x
7. Pu J, Liu Y, Zhang H, Tian L, Gui S, Yu Y, et al. Ran Y et al: An integrated meta-analysis of peripheral blood metabolites and biological functions in major depressive disorder. Mol Psychiatry. (2021) 26:4265–76. doi: 10.1038/s41380-020-0645-4
8. Zheng P, Fang Z, Xu XJ, Liu ML, Du X, Zhang X, et al. Metabolite signature for diagnosing major depressive disorder in peripheral blood mononuclear cells. J Affect Disord. (2016) 195:75–81. doi: 10.1016/j.jad.2016.02.008
9. Jia F, Li X, Liu F, Shi X, Liu H, Cao F. Association of renal function and depressive symptoms: Evidence from the China health and retirement longitudinal study. J Psychosom Res. (2020) 137:110224. doi: 10.1016/j.jpsychores.2020.110224
10. Li H, Wang A, Qi G, Guo J, Li X, Wang W, et al. Cystatin C and risk of new-onset depressive symptoms among individuals with a normal creatinine-based estimated glomerular filtration rate: A prospective cohort study. Psychiatry Res. (2019) 273:75–81. doi: 10.1016/j.psychres.2019.01.009
11. Zhang Z, He P, Liu M, Zhou C, Liu C, Li H, et al. Wu Q et al: Association of Depressive Symptoms with Rapid Kidney Function Decline in Adults with Normal Kidney Function. Clin J Am Soc Nephrol. (2021) 16:889–97. doi: 10.2215/CJN.18441120
12. Kakita D, Matsuzawa R, Yamamoto S, Suzuki Y, Harada M, Imamura K, et al. Shimokado K et al: Simplified discriminant parameters for sarcopenia among patients undergoing haemodialysis. J Cachexia Sarcopenia Muscle. (2022) 13:2898–907. doi: 10.1002/jcsm.13078
13. Tang T, Xie L, Hu S, Tan L, Lei X, Luo X, et al. Serum creatinine and cystatin C-based diagnostic indices for sarcopenia in advanced non-small cell lung cancer. J Cachexia Sarcopenia Muscle. (2022) 13:1800–10. doi: 10.1002/jcsm.12977
14. Kim NH, Kim HS, Eun CR, Seo JA, Cho HJ, Kim SG, et al. Park MH et al: Depression is associated with sarcopenia, not central obesity, in elderly korean men. J Am Geriatr Soc. (2011) 59:2062–8. doi: 10.1111/j.1532-5415.2011.03664.x
15. Lin S, Huang L, Luo ZC, Li X, Jin SY, Du ZJ, et al. Luo ZY et al: The ATP Level in the Medial Prefrontal Cortex Regulates Depressive-like Behavior via the Medial Prefrontal Cortex-Lateral Habenula Pathway. Biol Psychiatry. (2022) 92:179–92. doi: 10.1016/j.biopsych.2022.02.014
16. Teng T, Clarke G, Maes M, Jiang Y, Wang J, Li X, et al. Liu X et al: Biogeography of the large intestinal mucosal and luminal microbiome in cynomolgus macaques with depressive-like behavior. Mol Psychiatry. (2022) 27:1059–67. doi: 10.1038/s41380-021-01366-w
17. Yoon S, Kim JE, Hwang J, Kim TS, Kang HJ, Namgung E, et al. Renshaw PF et al: Effects of Creatine Monohydrate Augmentation on Brain Metabolic and Network Outcome Measures in Women With Major Depressive Disorder. Biol Psychiatry. (2016) 80:439–47. doi: 10.1016/j.biopsych.2015.11.027
18. Balestrino M, Adriano E. Beyond sports: Efficacy and safety of creatine supplementation in pathological or paraphysiological conditions of brain and muscle. Med Res Rev. (2019) 39:2427–59. doi: 10.1002/med.21590
19. Pu J, Liu Y, Gui S, Tian L, Yu Y, Song X, et al. Zheng P et al: Metabolomic changes in animal models of depression: a systematic analysis. Mol Psychiatry. (2021) 26:7328–36. doi: 10.1038/s41380-021-01269-w
20. Wallimann T, Tokarska-Schlattner M, Schlattner U. The creatine kinase system and pleiotropic effects of creatine. Amino Acids. (2011) 40:1271–96. doi: 10.1007/s00726-011-0877-3
21. Tian JS, Shi BY, Xiang H, Gao S, Qin XM, Du GH. 1H-NMR-based metabonomic studies on the anti-depressant effect of genipin in the chronic unpredictable mild stress rat model. PloS One. (2013) 8:e75721. doi: 10.1371/journal.pone.0075721
22. Tian JS, Peng GJ, Gao XX, Zhou YZ, Xing J, Qin XM, et al. Dynamic analysis of the endogenous metabolites in depressed patients treated with TCM formula Xiaoyaosan using urinary (1)H NMR-based metabolomics. J Ethnopharmacol. (2014) 158 Pt A:1–10. doi: 10.1016/j.jep.2014.10.005
23. Zheng S, Yu M, Lu X, Huo T, Ge L, Yang J, et al. Urinary metabonomic study on biochemical changes in chronic unpredictable mild stress model of depression. Clin Chim Acta. (2010) 411:204–9. doi: 10.1016/j.cca.2009.11.003
24. Zhao Y, Hu Y, Smith JP, Strauss J, Yang G. Cohort profile: the China health and retirement longitudinal study (CHARLS). Int J Epidemiol. (2014) 43:61–8. doi: 10.1093/ije/dys203
25. Liang J, Li X, Huang X, Xie W, Zheng F. Progression of depressive symptoms after early exposure to famine: The China Health and Retirement Longitudinal Study. J Affect Disord. (2023) 322:46–51. doi: 10.1016/j.jad.2022.11.025
26. Chen X, Crimmins E, Hu PP, Kim JK, Meng Q, Strauss J, et al. Venous blood-based biomarkers in the China health and retirement longitudinal study: rationale, design, and results from the 2015 wave. Am J Epidemiol. (2019) 188:1871–7. doi: 10.1093/aje/kwz170
27. Boey KW. Cross-validation of a short form of the CES-D in Chinese elderly. Int J Geriatr Psychiatry. (1999) 14:608–17. doi: 10.1002/(SICI)1099-1166(199908)14:8<608::AID-GPS991>3.0.CO;2-Z
28. Xie Y, Ma M, Wu W, Zhang Y, Zhang Y, Tan X. Factors associated with depressive symptoms among the elderly in China: structural equation model. Int Psychogeriatr. (2021) 33:157–67. doi: 10.1017/S1041610220001398
29. Chen H, Mui AC. Factorial validity of the Center for Epidemiologic Studies Depression Scale short form in older population in China. Int Psychogeriatr. (2014) 26:49–57. doi: 10.1017/S1041610213001701
30. Pu J, Yu Y, Liu Y, Tian L, Gui S, Zhong X, et al. Liu L et al: MENDA: a comprehensive curated resource of metabolic characterization in depression. Brief Bioinform. (2020) 21:1455–64. doi: 10.1093/bib/bbz055
31. Wang Y, Wang G, Zhang N, Huang J, Wu W, Jia F, et al. Hong W et al: Association between residual symptoms and social functioning in patients with depression. Compr Psychiatry. (2020) 98:152164. doi: 10.1016/j.comppsych.2020.152164
32. Zhou E, Ma S, Kang L, Zhang N, Wang P, Wang W, et al. Sun S et al: Psychosocial factors associated with anxious depression. J Affect Disord. (2023) 322:39–45. doi: 10.1016/j.jad.2022.11.028
33. Boima V, Tetteh J, Yorke E, Archampong T, Mensah G, Biritwum R, et al. Older adults with hypertension have increased risk of depression compared to their younger counterparts: Evidence from the World Health Organization study of Global Ageing and Adult Health Wave 2 in Ghana. J Affect Disord. (2020) 277:329–36. doi: 10.1016/j.jad.2020.08.033
34. de Jonge P, Roy JF, Saz P, Marcos G, Lobo A, Investigators Z. Prevalent and incident depression in community-dwelling elderly persons with diabetes mellitus: results from the ZARADEMP project. Diabetologia. (2006) 49:2627–33. doi: 10.1007/s00125-006-0442-x
35. Stubbs B, Vancampfort D, Veronese N, Kahl KG, Mitchell AJ, Lin PY, et al. Carvalho AF et al: Depression and physical health multimorbidity: primary data and country-wide meta-analysis of population data from 190 593 people across 43 low- and middle-income countries. Psychol Med. (2017) 47:2107–17. doi: 10.1017/S0033291717000551
36. Knochel C, Alves G, Friedrichs B, Schneider B, Schmidt-Rechau A, Wenzler S, et al. Treatment-resistant late-life depression: challenges and perspectives. Curr Neuropharmacol. (2015) 13:577–91. doi: 10.2174/1570159X1305151013200032
37. Zhu Y, Guo X, Zhang X, Shi X, Yang Y, Zhang Q. Sex differences in the relationship of serum creatinine to cystatin C ratio and depressive symptoms among middle-aged and older adults in China. J Affect Disord. (2022) 319:57–61. doi: 10.1016/j.jad.2022.09.030
38. Li Y, Wu L, Chen C, Wang L, Guo C, Zhao X, et al. Serum metabolic profiling reveals the antidepressive effects of the total iridoids of valeriana jatamansi jones on chronic unpredictable mild stress mice. Front Pharmacol. (2020) 11:338. doi: 10.3389/fphar.2020.00338
39. Liu L, Zhou X, Zhang Y, Pu J, Yang L, Yuan S, et al. Hippocampal metabolic differences implicate distinctions between physical and psychological stress in four rat models of depression. Trans Psychiatry. (2018) 8:4. doi: 10.1038/s41398-017-0018-1
40. Liu T, Zhou N, Xu R, Cao Y, Zhang Y, Liu Z, et al. A metabolomic study on the anti-depressive effects of two active components from Chrysanthemum morifolium. Artif Cells Nanomed Biotechnol. (2020) 48:718–27. doi: 10.1080/21691401.2020.1774597
41. Veeraiah P, Noronha JM, Maitra S, Bagga P, Khandelwal N, Chakravarty S, et al. Dysfunctional glutamatergic and gamma-aminobutyric acidergic activities in prefrontal cortex of mice in social defeat model of depression. Biol Psychiatry. (2014) 76:231–8. doi: 10.1016/j.biopsych.2013.09.024
42. Zhao L, Xiong Z, Lu X, Zheng S, Wang F, Ge L, et al. Metabonomic evaluation of chronic unpredictable mild stress-induced changes in rats by intervention of fluoxetine by HILIC-UHPLC/MS. PloS One. (2015) 10:e0129146. doi: 10.1371/journal.pone.0129146
43. Zhou X, Liu L, Lan X, Cohen D, Zhang Y, Ravindran AV, et al. Yang L et al: Polyunsaturated fatty acids metabolism, purine metabolism and inosine as potential independent diagnostic biomarkers for major depressive disorder in children and adolescents. Mol Psychiatry. (2019) 24:1478–88. doi: 10.1038/s41380-018-0047-z
44. Cao X, Li LP, Wang Q, Wu Q, Hu HH, Zhang M, et al. Xiong WC et al: Astrocyte-derived ATP modulates depressive-like behaviors. Nat Med. (2013) 19:773–7. doi: 10.1038/nm.3162
45. Cunha MP, Pazini FL, Lieberknecht V, Rodrigues ALS. Subchronic administration of creatine produces antidepressant-like effect by modulating hippocampal signaling pathway mediated by FNDC5/BDNF/Akt in mice. J Psychiatr Res. (2018) 104:78–87. doi: 10.1016/j.jpsychires.2018.07.001
46. Lyoo IK, Yoon S, Kim TS, Hwang J, Kim JE, Won W, et al. A randomized, double-blind placebo-controlled trial of oral creatine monohydrate augmentation for enhanced response to a selective serotonin reuptake inhibitor in women with major depressive disorder. Am J Psychiatry. (2012) 169:937–45. doi: 10.1176/appi.ajp.2012.12010009
47. Peng YF, Xiang Y, Wei YS. The significance of routine biochemical markers in patients with major depressive disorder. Sci Rep. (2016) 6:34402. doi: 10.1038/srep34402
48. Lee BJ. Association of depressive disorder with biochemical and anthropometric indices in adult men and women. Sci Rep. (2021) 11:13596. doi: 10.1038/s41598-021-93103-0
49. Liu X, Zheng X, Du G, Li Z, Qin X. Brain metabonomics study of the antidepressant-like effect of Xiaoyaosan on the CUMS-depression rats by (1)H NMR analysis. J Ethnopharmacol. (2019) 235:141–54. doi: 10.1016/j.jep.2019.01.018
50. Pu J, Liu Y, Gui S, Tian L, Yu Y, Wang D, et al. Chen Y et al: Effects of pharmacological treatment on metabolomic alterations in animal models of depression. Transl Psychiatry. (2022) 12:175. doi: 10.1038/s41398-022-01947-5
51. Zhou Y, Lu L, Li Z, Gao X, Tian J, Zhang L, et al. Antidepressant-like effects of the fractions of Xiaoyaosan on rat model of chronic unpredictable mild stress. J Ethnopharmacol. (2011) 137:236–44. doi: 10.1016/j.jep.2011.05.016
52. Niklasson F, Agren H. Brain energy metabolism and blood-brain barrier permeability in depressive patients: analyses of creatine, creatinine, urate, and albumin in CSF and blood. Biol Psychiatry. (1984) 19:1183–206.
53. Gindin J, Shochat T, Chetrit A, Epstein S, Ben Israel Y, Levi S, et al. van Hout H et al: Insomnia in long-term care facilities: a comparison of seven European countries and Israel: the Services and Health for Elderly in Long TERm care study. J Am Geriatr Soc. (2014) 62:2033–9. doi: 10.1111/jgs.2014.62.issue-11
54. Kim KW, Kang SH, Yoon IY, Lee SD, Ju G, Han JW, et al. Prevalence and clinical characteristics of insomnia and its subtypes in the Korean elderly. Arch Gerontol Geriatr. (2017) 68:68–75. doi: 10.1016/j.archger.2016.09.005
55. Yue Z, Zhang Y, Cheng X, Zhang J. Sleep quality among the elderly in 21st century shandong province, China: A ten-year comparative study. Int J Environ Res Public Health. (2022) 19. doi: 10.3390/ijerph192114296
56. Zhong BL, Li HJ, Xu YM, Jiang XF. Clinical insomnia among elderly primary care attenders in Wuhan, China: A multicenter cross-sectional epidemiological study. Front Public Health. (2022) 10:1026034. doi: 10.3389/fpubh.2022.1026034
57. McMorris T, Harris RC, Howard AN, Langridge G, Hall B, Corbett J, et al. Creatine supplementation, sleep deprivation, cortisol, melatonin and behavior. Physiol Behav. (2007) 90:21–8. doi: 10.1016/j.physbeh.2006.08.024
58. McMorris T, Harris RC, Swain J, Corbett J, Collard K, Dyson RJ, et al. Effect of creatine supplementation and sleep deprivation, with mild exercise, on cognitive and psychomotor performance, mood state, and plasma concentrations of catecholamines and cortisol. Psychopharmacol (Berl). (2006) 185:93–103. doi: 10.1007/s00213-005-0269-z
Keywords: depression, creatinine, association, Chinese, CHARLS, MENDA
Citation: Liu F, Zhong X and Wang C (2025) Lower creatinine levels are associated with an increased risk of depression: evidence from the China Health and Retirement Longitudinal Study. Front. Psychiatry 16:1446897. doi: 10.3389/fpsyt.2025.1446897
Received: 10 June 2024; Accepted: 04 February 2025;
Published: 25 February 2025.
Edited by:
Shen Liu, Anhui Agricultural University, ChinaReviewed by:
Adriano Martimbianco De Assis, Catholic University of Pelotas, BrazilCopyright © 2025 Liu, Zhong and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Cuiting Wang, d2FuZ2N1aXRpbmc4ODc3QDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.