- 1Department of Internal Medicine, General Hospital Kraljevo, Kraljevo, Serbia
- 2Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 3Department of Internal Medicine, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 4Clinic for Rheumatology and Allergology, University Clinical Center Kragujevac, Kragujevac, Serbia
- 5Department of Biochemistry, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 6Department of Laboratory Diagnostics, University Clinical Center Kragujevac, Kragujevac, Serbia
- 7Department of Physical Medicine and Rehabilitation, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 8Center for Molecular Medicine and Stem Cell Research, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 9Department of Physiology, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 10Department of Surgery, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 11Department for Anesthesiology and Reanimation, University Clinical Center Kragujevac, Kragujevac, Serbia
- 12Clinic for Endocrinology, University Clinical Center Kragujevac, Kragujevac, Serbia
- 13Department of Psychiatry, Faculty of Medical Sciences, University of Kragujevac, Kragujevac, Serbia
- 14Psychiatry Clinic, University Clinical Center Kragujevac, Kragujevac, Serbia
Objective: Many studies so far have shown that antipsychotic therapy may have an effect on the development of metabolic syndrome in patients diagnosed with schizophrenia. Our goal was to determine whether our respondents are at risk for developing metabolic syndrome and who is more predisposed to it.
Methods: In a stable phase, 60 patients diagnosed with schizophrenia were equally divided into three groups according to the drug (risperidone, clozapine, and aripiprazole monotherapy). Control group had 20 healthy examinees. Patients were evaluated first using The Positive and Negative Syndrome Scale (PANSS). Prolactin, lipid status, glycemia, insulin, cytokine values (IL-33, TGF-β, and TNF-α) and C-reactive protein (CRP) were measured. Also, Body mass index (BMI), Homeostatic Model Assesment for Insulin Resistance (HOMA index), waist and hip circumference (WHR) and blood pressure (TA) measurement were performed in the study.
Results: Patients treated with risperidone compared to healthy control subjects and aripiprazol group of patients had statistically significant difference in prolactin levels. In clozapine group compared to healthy control group values of HDL cholesterol and glucose level were statistically significant different. In aripiprazole group compared to healthy control group value of BMI was statistically significant different. Statistically significant correlations were found in TNF-α with glucose and HOMA index in risperidone treated patients and with BMI in clozapine group of patients; IL-33 with glucose in risperidone and with BMI in clozapine group of patients and TGF-β with glucose in risperidone group, with insulin and HOMA index in clozapine group and statistically significant negative correlation with LDL cholesterol in aripiprazole group of patients.
Conclusion: Patients on risperidone and clozapine therapy may be at greater risk of developing metabolic syndrome than patients treated with aripiprazole. Statistically significant difference in concentration of TNF-α and TGF-β was in the group of patients treated with risperidone compared to healthy control group.
Introduction
Schizophrenia is a serious illness in psychiatry. The disease itself is accompanied by numerous changes in the functioning of these people in everyday life. Then, treatment with antipsychotics in patients with schizophrenia is often with side effects. It is also inevitable to monitor the immune response, activation of the inflammatory process, how it maintains and regulates itself in patients. Hence, it is very difficult to approach health improvement conditions of these patients, choose the appropriate antipsychotic and then, as far as possible, influence the adverse effects of the therapy itself and other associated factors that may contribute to it.
When we take into account previous research, we see that patients are at risk of developing diabetes, heart disease and changes in bone density, both due to therapy and its side effects and due to the lifestyle of these patients (1–4). Also, the role of cytokines, immune markers, is increasingly present in research in patients with schizophrenia. Determining the role of individual interleukins in different phases and during the disease itself is of great importance for further approach not only in the diagnosis, but also in the treatment of schizophrenia (5–9).
In this study, we have tried to point out certain differences in the treatment of antipsychotics, specifically those treated with risperidone, clozapine and aripiprazole, both in relation to healthy subjects and in their relationship with each other. That side effects in terms of metabolic disorders are less common in patients treated with aripiprazole. To determine the possible connection of cytokines with the examined variables and the clinical symptomatology according to the used antipsychotic. As well as the presence and relationship of cytokines (IL-33, TGF-β, and TNF-α) in the mentioned groups, taking into account that all respondents were in a stable phase of schizophrenia.
Materials and methods
Participants
The research study included patients diagnosed with schizophrenia at the Clinic of Psychiatry, University of Kragujevac, according to The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Structured Clinical Interview for DSM (SCID) was used to confirm the diagnosis of schizophrenia and in the control group to exclude the existence of any mental illness. Patients on risperidone, clozapine and aripiprazole therapy were selected. The total number of respondents was n = 60, equally divided by the mentioned groups of drugs. The condition to be in the group of subjects diagnosed with schizophrenia was that they had been on antipsychotic therapy for more than 6 months. The control group of n = 20 healthy subjects corresponded to the group of patients by age and gender. Subjects in the control group had not previously suffered from any psychiatric disorder, nor had such a family member. In all participants, the presence of cardiovascular, endocrinological, immunological and systemic diseases was excluded and thus the absence of therapy that may effect on bone metabolism or prolactin levels in the blood (antihypertensive, glucocorticoid, methotrexate and other immunosuppressant, antiepileptic therapy, etc.). Both the subjects diagnosed with schizophrenia and the control group respondents were persons over 18 years of age. Participants were introduced to the course of the study and signed written informed consent to participate after being given the explanation. The study was approved by the Ethics Committee.
Clinical assessments
All patients were assessed for clinical symptoms of schizophrenia using SCI-PANSS-The Positive and Negative Syndrome Scale (PANSS).
Laboratory measurements
Biochemical analyses were done for all subjects, laboratory analysis that included determination of: prolactin, cholesterol, triglycerides, HDL cholesterol, LDL cholesterol, glycemia, insulin, C- reactive protein (CRP), all at 8 a.m., as well as serum cytokine concentrations of IL-33, TGF-β, and TNF-α (IL, Interleukin; TNF, Tumor Necrosis Factor; TGF, Transforming growth factor). Cytokine concentrations (IL-33, TGF-β, and TNF-α) were measured by ELISA according to the established manufacturer’s protocol (R&D Systems, Minneapolis, MN, United States).
Measurements and indices made in the study
Body mass index (BMI), Homeostatic Model Assesment for Insulin Resistance (HOMA index) and waist and hip circumference (WHR) and blood pressure (TA), were performed in the study.
BMI is calculated by dividing the subject’s weight by the height in square meters. Based on the obtained value, we can assess the degree of obesity or malnutrition according to certain categories or consider as normal weight (Underweight is <18.5; Normal weight is in a range 18.5–24.9; Overweight is in a range 25–29.9; Obesity BMI is of 30 or greater).
The HOMA index is used to assess the existence of insulin resistance, based oncalculating the values of insulin and glucose concentration. Higher value of this index indicates the existence of insulin resistance (cut-off value is >2).
The waist-to-hip ratio (WHR) allows us to establish the distribution of fat and thus determine whether we are a certain health risk (WHR of 1.0 or higher increases the risk for all obesity- related diseases).
Blood pressure (TA) is measured with a certain device, the obtained values of systolic and diastolic blood pressure are expressed in millimeters of mercury (mm Hg). By measuring blood pressure, and in accordance with the obtained values, we can determine whether patients are in the risk of developing cardiovascular disease and stroke in the first place (normal TA is when systolic pressure is less than 120 mmHg and diastolic pressure is less than 80 mmHg).
Statistical analysis
Statistical data processing was performed in IBM SPSS Statistics v.20. Data are presented descriptively using mean and standard deviation or median for continuous variables and using absolute and relative frequencies for categorical variables. The normality of the data distribution was analyzed using the Shapiro-Wilk normality test. For the analysis of continuous variables (age, PANSS, metabolic variables, cytokines, and cytokine ratio) in relation to groups of subjects according to the drug they use, one-factor ANOVA was used for different groups if data followed normal distribution or Kruskal–Wallis test if data did not follow normal distribution. For the analysis of continuous variables with each other, the method of linear correlation was applied, Pearson’s correlation coefficient was interpreted if the data followed the normal distribution or Spearman’s correlation coefficient if the data did not follow the normal distribution. To analyze the gender of the respondents in relation to the groups of respondents, according to the drug they use, the chi-square test for independence was applied.
The results were considered statistically significant if the p-value was less than or equal to 0.05. In the case of subsequent tests, since four groups were compared with each other, that we had six subsequent tests, we considered the results statistically significant if the p-value was less than 0.0083.
Strength of study
Using G-Power software (G*Power 3.1.2), and based on a certain sample size of 20 respondents per group, the strength of the study is 82.44%.
Results
Demographic characteristics and clinical scale for the assessment and clinical monitoring of the course of schizophrenia and pharmacotherapeutic response, scales of positive and negative symptoms of schizophrenia (Positive and Negative Syndrom Scale)
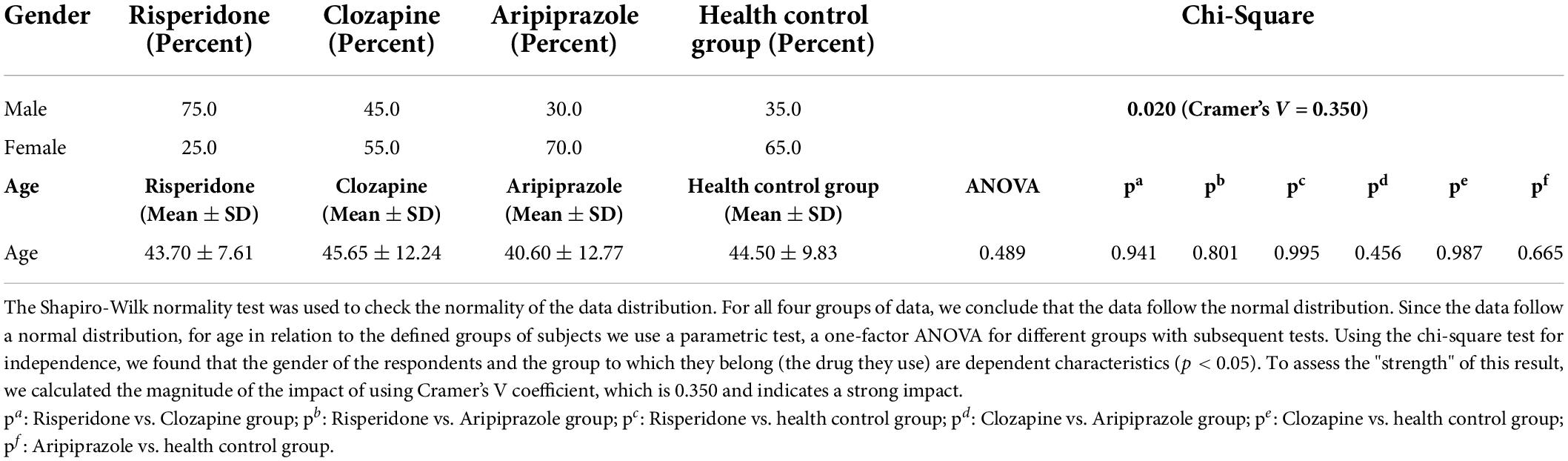
n the group of patients treated with risperidone participated n = 15 male and n = 5 female respondents (75.0% male/25.0% female respondents). In the group with clozapine in treatment there were n = 9 male and n = 11 female respondents (45.0% male/55.0% female respondents). In the third group of patients treated with aripiprazole there were n = 6 male and female n = 14 respondents (30.0% male/70% female respondents). When it comes to the control group, it consisted of n = 8 male and n = 12 female subjects (35.0% male/65% female subjects). We calculated the magnitude of the impact using Cramer’s V coefficient, which was 0.350 and indicated a strong impact. It was observed that only in the group using Risperidone were more men while in the group using Aripiprazole and in the control group more women.
In the group of patients treated with risperidone mean age (SD) was 43.70 ± 7.61, in clozapine group mean age (SD) was 45.65 ± 12.24, in aripiprazol treated patients mean age (SD) was 40.60 ± 12.77 and in health control group mean age (SD) was 44.50 ± 9.83. There was no statisticaly significant difference between any mentioned groups in mean age (Table 1).
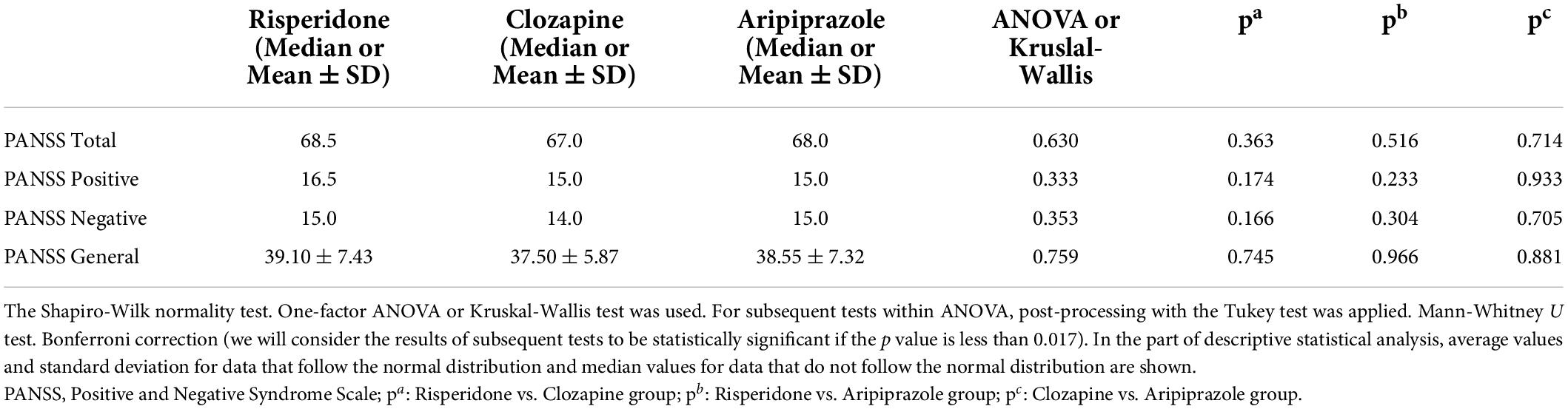
A clinical scale for the assessment and clinical monitoring of the course of schizophrenia and pharmacotherapeutic response, Scales of positive and negative symptoms of schizophrenia (Positive and Negative Syndrome Scale – PANSS), was used in all subjects with schizophrenia, and compared to one another gave us the results that are shown in next table (Table 2). As we can see in Table 2, results in each group of used antipsychotics and PANSS scale were not statistically significant.
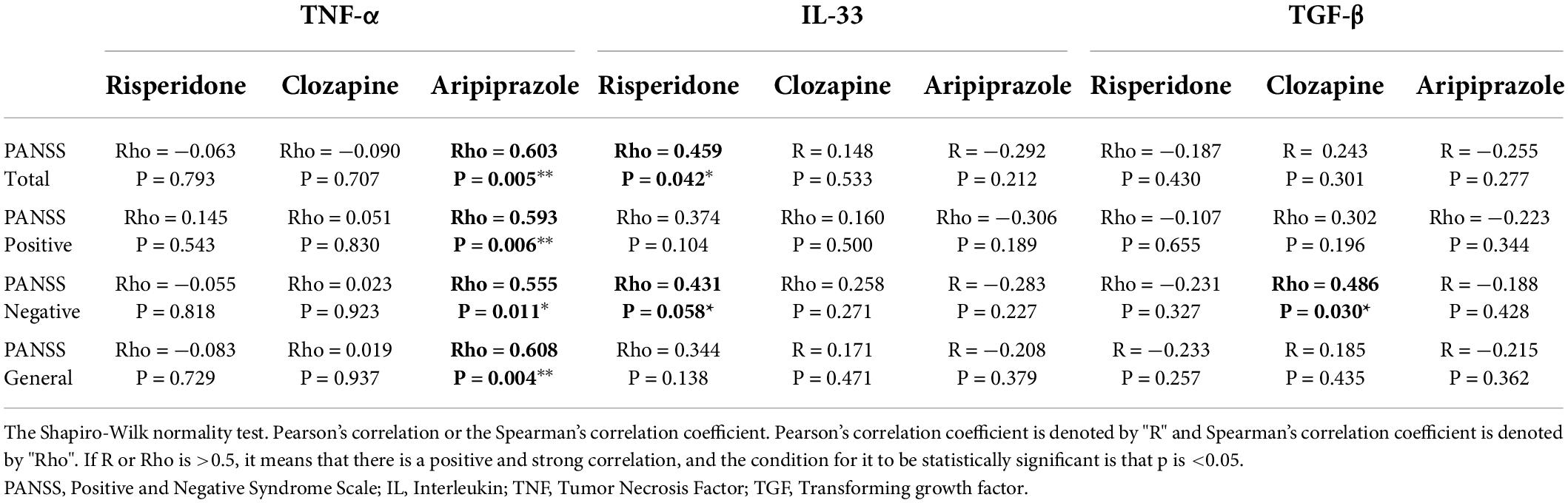
However, the results we obtained in the correlation between the cytokines and PANSS scale according to each group of used antipsychotics are as follows (Table 3).
In the group of patients treated with risperidone, there is a statistically significant correlation of IL-33 with PANSS total score (p = 0.042) and PANSS negative (p = 0.058) subscale. No other statistically significant correlation was found in other cytokines and PANSS scale in risperidone group of patients.
In the group of patients treated with clozapine, there is a statistically significant correlation between TGF-β and PANSS negative subscale (p = 0.030). No other statistically significant correlation was found in other cytokines and PANSS scale in clozapine group of patients.
When it comes to aripiprazole treated patients there is a statistically significant correlation of TNF-α with PANSS total score (p = 0.005), PANSS positive (p = 0.006), PANSS negative (p = 0.011) and PANSS general (p = 0.004) subscales. No statistically significant correlation between other cytokines and PANSS scale was found in aripiprazole group of patients.
Comparison of observed metabolic variables (prolactin level, lipid status measurements, glucose, insulin, body mass index, homeostatic model assesment for insulin resistance index and waist and hip circumference) between examined groups
Comparing all groups we have at our disposal and measured values that can be taken as predictors of metabolic syndrome, we came to the following results shown in Table 4.
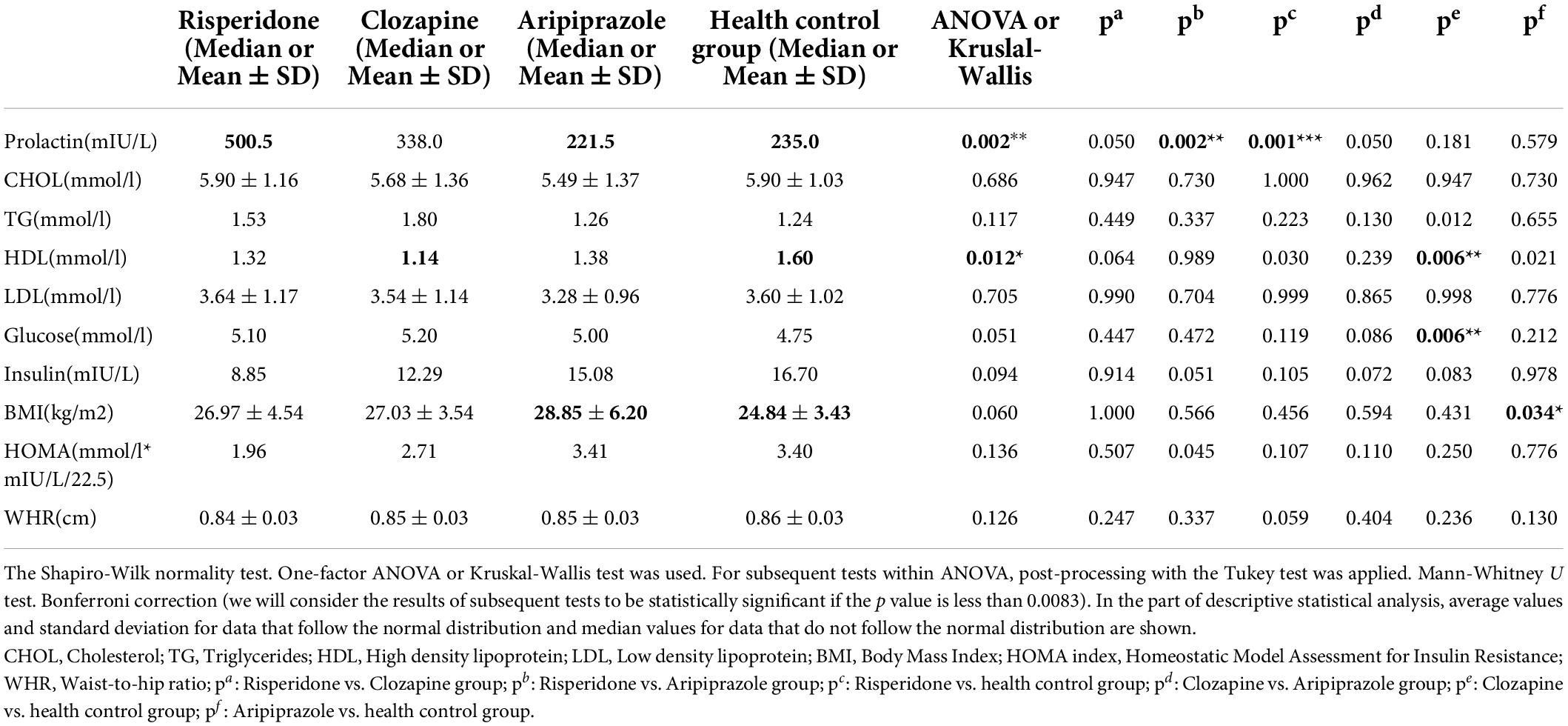
Table 4. Comparison of observed metabolic variables between the groups of patients treated with risperidone, clozapine, and aripiprazole toward the health control group and within them.
The median value of prolactin level was 500.5 in the group of patients treated with risperidone and 235.0 in the control group, thus this difference was statistically significant (p = 0.001).
The median value of HDL cholesterol was 1.14 in the group of patients treated with clozapine, and 1.60 in the control group, so this difference was statistically significant (p = 0.006). The median glucose level was 5.20 in the group of patients treated with clozapine and 4.75 in the control group, and this difference was statistically significant (p = 0.006).
Mean ± SD value of BMI was 28.85 ± 6.20 in the aripiprazole group of patients and 24.84 ± 3.43 in the control group, thus this difference was statistically significant (p = 0.034).
No other statistically significance differences were found in the values of all others variables between the patients in each drug group and subjects in the control group.
Comparison within groups of patients on the mentioned therapy in observed metabolic variables showed the difference only in measured levels of prolactin.
In risperidone group median value of prolactin level was 500.5 and in aripiprazole group of patients 221.5, so the difference was statistically significant (p = 0.002).
Correlation of observed metabolic variables and cytokines
When it comes to the measured values of cytokines in these three groups based on used therapy and according to the already mentioned observed variables in the study, we came to the following results in correlation within them (Table 5).

Table 5. Correlation of observed metabolic variables and measured cytokines in each group of patients.
In the risperidone group of patients, there was a statistically significant correlation in TNF-α with glucose (p = 0.020) and HOMA index (p = 0.025), between IL-33 and glucose (p = 0.001) and TGF- β with glucose (p = 0.028).
In the clozapine group of patients, there was a statistically significant correlation between TNF-α and BMI (p = 0.001), IL-33 and BMI (p = 0.003) and TGF-β with insulin (p = 0.004) and HOMA index (p = 0.002).
In the aripiprazole group, there was one statistically significant negative correlation between TGF-β and LDL cholesterol (p = 0.018).
No other significant correlation was found with any other metabolic variables measured and cytokines in these three groups.
Comparison of cytokine concentration values of IL-3, TGF-β, and TNF-α
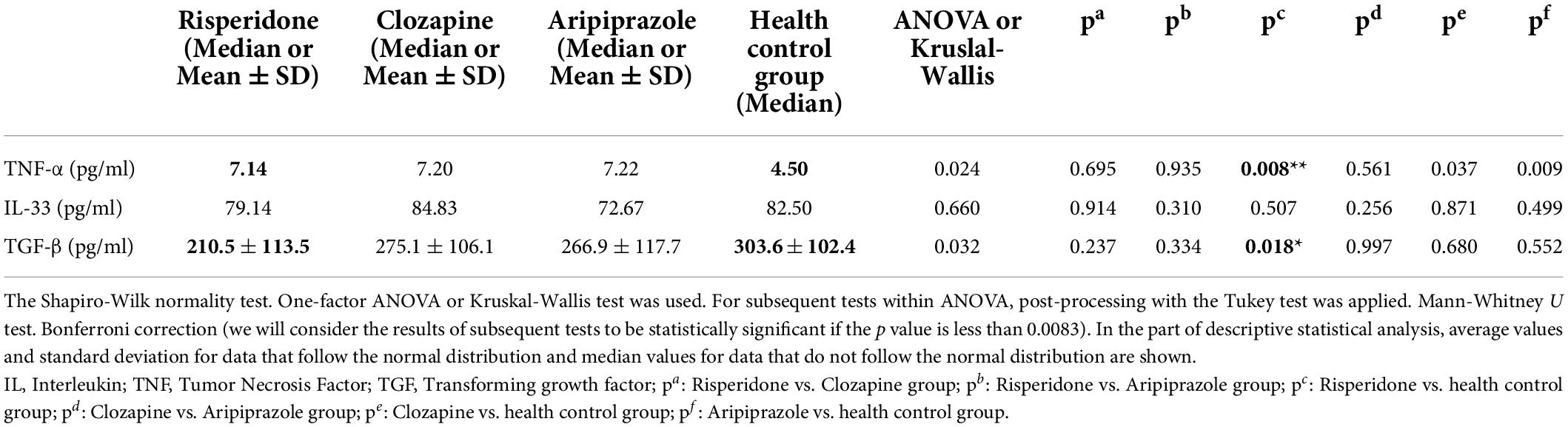
Our study was done in the stable phase of schizophrenia. Results of the cytokine concentration values measured in this study are compared within each of examined groups (Table 6).
In risperidone group of patients compared to the subjects in control group there was a statistically significant difference found in the values of the concentration in two cytokines, TNF-α (p = 0.008) and TGF-β (p = 0.018).
There are no other statistically significant differences found in any other comparisons according to the given groups and cytokines.
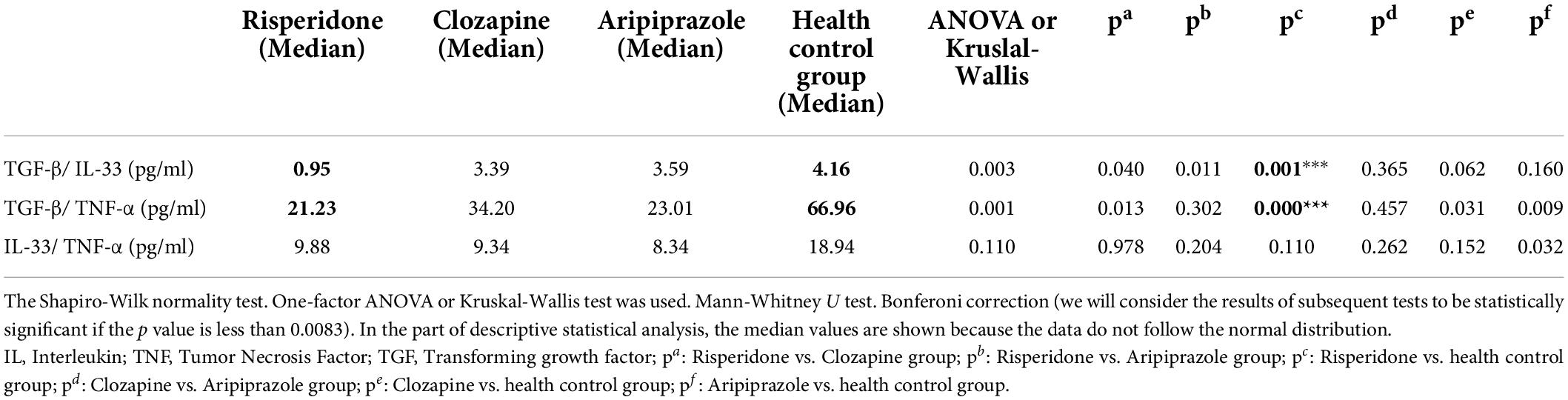
Cytokine ratio of TGF-β/IL-33, TGF-β/TNF-α, and IL-33/TNF-α in all mentioned groups
We also determined the ratio of these cytokines in all examined groups. The ratio of anti- inflammatory cytokines to pro-inflammatory cytokines (TGF-β/IL-33 and TGF-β/TNF-α) and the interrelationship of proinflammatory cytokines (IL-33/TNF-α) gave the following results (Table 7).
Risperidone group of patients compared to healthy control group difference was statistically significant in TGF-β/IL-33 ratio (p = 0.011) and in TGF-β/TNF-α ratio (p = 0.000).
There are no statistically significant differences in all other comparisons according to the given groups and the ratio of these cytokines.
Values of CRP in each patient of mentioned groups and of course in health control group were within the reference value. Blood pressure values measured in all groups were within normal blood pressure values.
In our research patients were taking antipsychotics in the maintenance doses. There were no significant differences in dose range between groups compared on equivalents based on Chlorpromazine. It is provided in Supplementary Material. This was main reason that there was not statistically significant difference in influence on variables (symptomatology, metabolic parameters, and cytokines), between antipsychotic doses, compared on equivalents based on Chlorpromazine. Also, we compared influence of different doses in the same antipsychotic group and there was not statistically significant difference, too.
Discussion
The therapeutic approach to the treatment of schizophrenia is long-term and involves the use of antipsychotics, which, although necessary in the treatment in addition to the desired effect on the disease itself, may result in an increased risk of metabolic syndrome and cardiovascular events as side effects (10).
Of all the drugs, antipsychotics most often lead to hyperprolactinaemia (10–12). Lacto trophic cells located in the anterior pituitary gland are responsible for the secretion of the hormone prolactin and its synthesis and release are controlled by peptides, neurotransmitters and steroids. Dopamine plays a main inhibitory role on the membrane of lactotroph cells, binding to dopamine D2 receptors. By blocking D2 receptors, antipsychotics eliminate the inhibitory effect of dopamine on prolactin secretion and thus can all cause hyperprolactinaemia. The faster the antipsychotic separates from the D2 receptor, the lower the plasma prolactin increase appears to be. Due to all the listed properties of antipsychotics, we refer to them as prolactin-raising (conventional neuroleptics, amisulpride, risperidone) and prolactin-sparing (clozapine, aripiprazole, olanzapine) antipsychotics (13).
The aspect of the patient in which we establish obesity, high blood pressure values, hyperglycemia, lipid status disorder, and also insulin resistance refer us to the possible metabolic syndrome which should be recognized in order to prevent further cardiovascular adverse events (14, 15). It is known that due to the interaction of antipsychotics with serotonergic, histaminergic and dopaminergic neurotransmitter systems, there is a change in appetite and food intake, which becomes increased (16–18). Antipsychotic therapy is long-lasting and it can lead to weight gain (19). In patients treated with clozapine and olanzapine (except in relation to clozapine) antipsychotics, the risk of metabolic syndrome was significantly higher, and in patients treated with aripiprazole (except in relation to amisulpride) significantly lower than with other antipsychotics (20). It is suggested that hyperprolactinaemia has a role in the pathogenesis of obesity and glucose intolerance and thus can lead to changes in metabolic profile and finally metabolic syndrome (21). Therefore, patients with diagnosed schizophrenia are at higher risk of developing hypertension, cardiovascular disease, diabetes, and osteoporosis (22).
In the present study, statistically significant difference in prolactin level was found in risperidone treated patients compared to the control group and compared to aripiprazole treated patients. One study says that in patients with schizophrenia and hyperprolactinemia adding aripiprazole could be an option to lower its concentration (23). In relation to the gender of the patients, the type of antipsychotics and the duration of the disease, the level of prolactin will also depend (24). Also, one study showed that adjunctive aripiprazole indicated better values of hyperprolactinemia and improvement of clinical symptoms of schizophrenia (25).
It is known that when we say good cholesterol, we think of high-density lipoprotein (HDL). If the values are lower, than considering the anamnesis, clinical history and additional examinations of the patients can help us discover the risk of developing the metabolic syndrome (26). In our study, statistically significant differences in values of HDL cholesterol and glucose level were in clozapine treated patients compared to the control group.
In aripiprazole group of patients compared to healthy control group we found statistically significant difference in BMI value.
Monitoring metabolic parameters in one study in patients on clozapine treatment and long-acting injectable antipsychotics suggested that the first group of patients was at higher risk of developing metabolic syndrome (27). Antipsychotic therapy affects lipid and glycoregulatory disorders, as well as mood swings in patients with schizophrenia (28). Analyzing metabolic disorders as a consequence of the use of antipsychotics, one study indicated that patients treated with olanzapine and clozapine had the worst profile, in contrast to patients treated with aripiprazole, brexpiprazole, cariprazine, lurasidone and ziprasidone (29). The level of HDL cholesterol in overweight patients with schizophrenia differed depending from the antipsychotic they were treated (30).
In patients treated with risperidon a statistically significant positive correlation in TNF-α with glucose levels and HOMA index, IL-33, and TGF-β also with glucose levels was found.
In clozapine group of patients statistically significant correlation of TNF-α and IL-33 with BMI was established and TGF-β with insulin level and HOMA index. It has previously been shown that there is a positive association between IL-1β, IL-6, and TNF-α and prolactin levels in patients with schizophrenia (31). Other study has shown clozapine effect on weight gain and that there was a positive correlation between TNF α and BMI (32). Another study showed expression of certain cytokines (IL-2, IL-6, IL-17, and TNF-α) after chronic clozapine therapy (33).
In aripiprazol treated patients statistically significant negative correlation was between TGF and LDL cholesterol. Without other correlations found in the group of patients treated with aripiprazole, it indicates a lower risk of developing metabolic disorders in this group of patients. It is in accordance with another study which indicates that the use of aripiprazole in patients with milder clinical symptoms or clozapine therapy in refractory patients who have already used antipsychotics should be considered (34).
In obese individuals, we know that occurs a change in the balance between pro-inflammatory (T helper 1 and T helper 17 lymphocytes) and anti-inflammatory (T helper 2 and regulatory T lymphocytes) CD4 + cells. With obesity, and especially in those patients who already have the metabolic syndrome beside, the number of anti-inflammatory regulatory T lymphocytes in adipose tissue decreases (35–37). Based on immunological changes, a recent study indicated that long-term inflammation may be an independent factor in the development of diabetes mellitus type 2 (38). Adiponectin, leptin, resistin, MCP-1, TNF-α, IL-6, IL-1β, IL-10, and TGF-β are the ones that have been most researched so far (39, 40).
Earlier studies have shown that the course of the disease and the effectiveness of therapy are associated with certain immune markers. Thus, an association with acute symptoms in affective disorders and schizophrenia and elevated values of IL-33 has been shown (41). Also, significant association between cognition in patients with schizophrenia and IL-33 levels and the soluble form of IL 33 receptor (sST2) was found (42). In the next study, which examined the values of Gal-3, IL-33, and sST2 in schizophrenia patients, but in different stages of disease, the obtained result indicated that in the phase of exacerbation compared to the control population and patients in remission were higher values of IL -33 and sST2 (43).
In our study in all three groups of respondents risperidone, clozapine and aripiprazole patients, a certain correlation was found among the cytokines (IL-33, TGF-β, and TNF-α) and the SCI- PANSS scale. In the group of patients treated with risperidone, IL-33 significantly correlated with PANSS total score and PANSS negative subscale. In clozapine patients group, TGF-β significantly correlated with PANSS negative subscale. In the group of patients treated with aripiprazole, TNF-α significantly correlated with all subsets of PANSS scale.
One study compared the IFN-γ/TGF-β ratio and the IL-17/TGF-β ratio to the PANSS scale. In the first case, a negative correlation was shown between the mentioned cytokines and the total PANSS score, and in the second case between the mentioned cytokines and the subscale of PANSS negative and PANSS general psychopathology (44). In another study, IL-17 and TGF-β values had a positive correlation with the PANSS total scale (45). In the study we mentioned earlier (43) alongside of observing cytokine values in certain stages of the disease it was examined whether there is a correlation of IL-33 with the PANSS scale. They came to the conclusion that it correlates with the PANSS positive and the PANSS general subscale (43). Other studies have shown a negative correlation of IL-10 with the PANSS scale (positive, negative and total subscale) in chronic schizophrenia patients treated with aripiprazole (34).
At different stages of schizophrenia, the challenge for researchers is to determine which cytokines are more or less present, the ratio of pro-inflammatory to anti-inflammatory cytokines. One study showed that at the very beginning of the disease there was an increased level of both pro-inflammatory and anti-inflammatory cytokines, and later only a prolonged pro-inflammatory response. Of course, more research studies are needed on this topic (46, 47). In one study, they compared the levels of different cytokines in the first episode before drug administration and later after in stable phase treatment. The results indicated that the level of type 2 cytokines that was increased at the beginning, after the therapy with antipsychotics, was reduced (48). It is also explained that in the beginning of the disease due to the pronounced pro-inflammatory response and regulation of chronic inflammation, the presence of anti-inflammatory activity is increased, as it will counteract or limit the first mentioned response (48).
In our study statistically significant difference in TGF-β concentration was found between the risperidone treated patients and the control group. We also founded a statistically significant difference in TNF-α concentration between risperidone and the control group.
TGF-β/IL-33 and TGF-β/TNF-α ratio were statistically significant in risperidone treated patients compared to the control group. There was no statistically significant difference found among any group of subjects in IL-33/TNF-α ratio. Other studies suggest similar results (48, 49). A clear change in the ratio of Th1 and Th2 cells was observed in schizophrenia (50). We have already mentioned that the immune response plays a big role in the disease of schizophrenia and of course it is liable to change after the inclusion of antipsychotics. A higher Th1/Th2 ratio was observed in those patients, but it was weakened after the effect of the therapy. It is also noticed that TGF-β plays a role in attenuating Th1 receptor activity (51).
One study provided an explanation and indicated the role of cytokines in schizophrenia, and thus they could influence the prevention, regulation and treatment of schizophrenia as key molecular targets (52). Cytokines that have changed depending on the clinical status of schizophrenia, so for example the TGF-β marker seemed to be state-related marker and it was increased during acute exacerbation, but after antipsychotic treatment normalized. On the other hand, TNF-α was shown as trait marker, since the levels remained elevated in both of these stages (53). Also, the researchers’ interest is more focused both on examining metabolic and inflammatory changes in patients with chronic schizophrenia (54).
It is necessary to supplement research on the influence of all risk factors, influence and connection among antipsychotics and cytokines. Encourage them to be active, have a hygienic dietary regime and a healthier lifestyle (55–58). Limitations of the study were: limited examined group of patients to the University Clinical Center of Kragujevac (we did not take into account other centers nearby, which would for certain give us a larger number of respondents); cytokine concentration were not measured before antipsychotic treatment; potential introduction of Cariprazine and Paliperidone were not observed, because the number of patients on these drugs was small in our environment.
Conclusion
Patients treated with risperidone and clozapine may be at greater risk than patients treated with aripiprazole for the development of metabolic syndrome and later on cardiovascular events. This is probably due to the influence of cytokines and their relationship.
In the group of patients treated with risperidone, glucose correlates with all three cytokines, TNF-α, IL-33, TGF-β, while HOMA index correlates with TNF-α. When it comes to clozapine group of patients BMI correlates with TNF-α and IL-33, until HOMA index and insulin correlate with TGF-β.
There is also a difference in the concentration of cytokines TNF-α and TGF-β and cytokine ratio of TGF-β/IL-33 and TGF-β/TNF-α, between patients treated with risperidone and a healthy control group.
Statistically significant difference in prolactin levels between risperidone and aripiprazole treated patients is found.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving human participants were reviewed and approved by the Ethics Committee, University Clinical Center Kragujevac. The patients/participants provided their written informed consent to participate in this study.
Author contributions
All authors were included in the designing of the manuscript, drafting the work, critical revision, and final approval for all aspects of the work, and the final version to be published.
Funding
This research was funded by the Faculty of Medical Sciences, University of Kragujevac (project JP 09/19).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2022.925757/full#supplementary-material
References
1. Juan DM, Sonia A, Gabriel R, Francisco LM. Metabolomic connections between schizophrenia, antipsychotic drugs and metabolic syndrome: a variety of players. Curr Pharm Des. (2021) 27:4049–61. doi: 10.2174/1381612827666210804110139
2. Sandra H, Ekaterina Y, Ahmad K, Stuart DR. Cardiovascular effects of psychotic illnesses and antipsychotic therapy. Heart. (2019) 105:1852–9. doi: 10.1136/heartjnl-2017-312107
3. Marc DH, Johan D, Brendon S. Relationship between antipsychotic medication, serum prolactin levels and osteoporosis/osteoporotic fractures in patients with schizophrenia: a critical literature review. Expert Opin Drug Saf. (2016) 15:809–23. doi: 10.1517/14740338.2016.1167873
4. Bruce JK, Hong LS, Virginia LS, Jayanthi J. Bone loss associated with hyperprolactinemia in patients with schizophrenia. Clin Schizophr Relat Psychoses. (2013) 7:115–23. doi: 10.3371/CSRP.KISE.020113
5. Elżbieta K, Ewa BB, Justyna A, Adam W, Paulina ŻJ. Alarmins (IL-33, sST2, HMGB1, and S100B) as potential biomarkers for schizophrenia. Psychiatr Res. (2021) 138:380–7. doi: 10.1016/j.jpsychires.2021
6. Dorota F, Błażej M, Edyta PA, Lidia K, Anna T, Paweł S, et al. Sex differences in TGFB-β signaling with respect to age of onset and cognitive functioning in schizophrenia. Neuropsychiatr Dis Treat. (2015) 11:575–84. doi: 10.2147/NDT.S74672
7. Alireza M, Ehsan R, Vahid GA. Brain, blood, cerebrospinal fluid, and serum biomarkers in schizophrenia. Psychiatry Res. (2018) 265:25–38. doi: 10.1016/j.psychres.2018.04.036
8. Yayan L, Hongbo H, Jie Z, Yufen O, Ni F. Changes in serum TNF-α, IL-18, and IL-6 concentrations in patients with chronic schizophrenia at admission and at discharge. Compr Psychiatry. (2019) 90:82–7. doi: 10.1016/j.comppsych.2019.01.003
9. Cigdem S, Noushin Z, Ayse K, Yazgul D, Muazzez G, Ismet K, et al. Reduced regulatory T cells with increased proinflammatory response in patients with schizophrenia. Psychopharmacology (Berl). (2020) 237:1861–71. doi: 10.1007/s00213-020-05504-0
10. Walter M, Chiara C, Anna C. Hyperprolactinemia induced by antipsychotics: from diagnosis to treatment approach. Endocr Metab Immune Disord Drug Targets. (2017) 17:38–55. doi: 10.2174/1871530317666170424102332
11. Spyridon S, Dimitrios T, Myrto S, Georgios P. Antipsychotic drugs: from receptor-binding profiles to metabolic side effects. Curr Neuropharmacol. (2018) 16:1210–23. doi: 10.2174/1570159X15666170630163616
12. Jasmin G, Roisin W, Caroline T, Caroline G, Natalie T, Jayashri K. Antipsychotic-induced hyperprolactinemia: synthesis of world-wide guidelines and integrated recommendations for assessment, management and future research. Psychopharmacology (Berl). (2017) 234:3279–97. doi: 10.1007/s00213-017-4730-6
13. Besnard I, Auclair V, Callery G, Gabriel-Bordenave C, Roberge C. Antipsychotic-drug- induced hyperprolactinemia: physiopathology, clinical features and guidance. Encephale. (2014) 40:86–94. doi: 10.1016/j.encep.2012.03.002
14. Robert HE, Scott MG, Paul ZZ. The metabolic syndrome. Lancet. (2005) 365:1415–28. doi: 10.1016/S0140-6736(05)66378-7
15. Tajamul H, Mushtaq AM, Sheikh S, Mahak S, Rajesh KC. Prevalence of metabolic syndrome among psychiatric inpatients: a hospital based study from Kashmir. J Clin Diagn Res. (2017) 11:VC05–08. doi: 10.7860/JCDR/2017/25801.10011
16. Starrenburg FCJ, Bogers JPAM. How can antipsychotics cause diabetes mellitus? Insights based on receptor-binding profiles, humoral factors and transporter proteins. Eur Psychiatry. (2009) 24:164–70. doi: 10.1016/j.eurpsy.2009.01.001
17. Dennis HK, Matthew JM, Stephen MS. Building a better antipsychotic: receptor targets for the treatment of multiple symptom dimensions of schizophrenia. Neurotherapeutics. (2009) 6:78–85. doi: 10.1016/j.nurt.2008.10.020
18. Fabio P, Vincenzo DL, Andrea DB. Weight gain, schizophrenia and antipsychotics: new findings from animal model and pharmacogenomics studies. Schizophr Res Treat. (2011) 2011:459284. doi: 10.1155/2011/459284
19. Maarten B, Annemarie F, Jouke J, Jim van O, Marjan Dr. Almost all antipsychotics result in weight gain: a meta-analysis. PLoS One. (2014) 9:e94112. doi: 10.1371/journal.pone.0094112
20. Davy V, Brendon S, Alex JM, Marc DH, Martien W, Philip BW, et al. Risk of metabolic syndrome and its components in people with schizophrenia and related psychotic disorders, bipolar disorder and major depressive disorder: a systematic review and meta-analysis. World Psychiatry. (2015) 14:339–47. doi: 10.1002/wps.20252
21. Renata SA, Dario DA, Rosa P, Rosario P, Annamaria C. The effects of hyperprolactinemia and its control on metabolic diseases. Expert Rev Endocrinol Metab. (2018) 13:99–106. doi: 10.1080/17446651.2018.1434412
22. Haishan W, Lu D, Lipin Z, Jingping Z, Lehua L, Jindong C. Osteoporosis associated with antipsychotic treatment in schizophrenia. Int J Endocrinol. (2013) 2013:167138. doi: 10.1155/2013/167138
23. Javier L, Itziar M, Alexandre GR, Clemente GR, Benedicto CF, José Antonio M, et al. Pharmacological treatment strategies for lowering prolactin in people with a psychotic disorder and hyperprolactinaemia: a systematic review and meta-analysis. Schizophr Res. (2020) 222:88–96. doi: 10.1016/j.schres.2020.04.031
24. Liana D, Ana-Maria R, Ion P, Cristina Ana B, Victor D, Sorin U, et al. Prolactin response to antipsychotics: an inpatient study. PLoS One. (2020) 15:e0228648. doi: 10.1371/journal.pone.0228648
25. Wei Z, Dong-Bin C, Xin-Hu Y, Gabor SU, Chee HN, Zhan-Ming S, et al. Adjunctive aripiprazole for antipsychotic-related hyperprolactinaemia in patients with first-episode schizophrenia: a meta-analysis. Gen Psychiatr. (2019) 32:e100091. doi: 10.1136/gpsych-2019-100091
26. Mayo Clinic Staff. HDL Cholesterol: How to Boost Your ‘Good’ Cholesterol? Your Cholesterol Levels are an Important Measure of Heart Health. For HDL Cholesterol, or “Good” Cholesterol, Higher Levels are Better. Available online at: https://www.mayoclinic.org/diseases-conditions/high-blood-cholesterol/in-depth/hdl-cholesterol/art-20046388 (accessed November 10, 2020).
27. Lydon A, Vallely J, Tummon A, Maher S, Sabri S, McLoughlin J, et al. Routine screening and rates of metabolic syndrome in patients treated with clozapine and long-acting injectable antipsychotic medications: a cross-sectional study. Ir J Psychol Med. (2021) 38:40–8. doi: 10.1017/ipm.2020.12
28. Adam W, Dominik S, Iwona K. Levels of triglycerides, cholesterol, LDL, HDL and glucose in patients with schizophrenia, unipolar depression and bipolar disorder. Diabetes Metab Syndr. (2015) 9:168–76. doi: 10.1016/j.dsx.2015.04.004
29. Pillinger T, McCutcheon RA, Vano L, Mizuno Y, Arumuham A, Hindley G, et al. Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia, predictors of metabolic dysregulation, and association with psychopathology: a systematic review and network meta-analysis. Lancet Psychiatry. (2020) 7:64–77. doi: 10.1016/S2215-0366(19)30416-X
30. Shin O, Takuro S, Yutaro S, Manabu Y, Kazutaka S, Takao M, et al. High-density lipoprotein- cholesterol and antipsychotic medication in overweight inpatients with schizophrenia: post-hoc analysis of a Japanese nationwide survey. BMC Psychiatry. (2018) 18:180. doi: 10.1186/s12888-018-1764-1
31. Xueqin S, Xiaoduo F, Jianjiang Z, Hui Z, Xue L, Lijuan P, et al. Prolactin serum levels correlate with inflammatory status in drug-naïve first-episode schizophrenia. World J Biol Psychiatry. (2014) 15:546–52. doi: 10.3109/15622975.2014.922699
32. Michael K, Andreas S, Alexander S, Hubertus H, Mira AD, Peter MW, et al. Effects of clozapine and olanzapine on cytokine systems are closely linked to weight gain and drug- induced fever. Psychoneuroendocrinology. (2009) 34:118–28. doi: 10.1016/j.psyneuen.2008.08.016
33. Xiaoping Y, Song W, Yudong S, Yating Y, Yulong Z, Lei X, et al. Pro-inflammatory cytokine levels are elevated in female patients with schizophrenia treated with clozapine. Psychopharmacology (Berl). (2022) 239:765–71. doi: 10.1007/s00213-022-06067-y
34. Jarosław S, Monika RC, Elżbieta Ś, Piotr G. Therapeutic effect of aripiprazole in chronic schizophrenia is accompanied by anti-inflammatory activity. Pharmacol Rep. (2015) 67:353–9. doi: 10.1016/j.pharep.2014.09.007
35. Jeffrey D, Zubair S, Nilay S, Bradley N, Dean M, Vimal N, et al. Visceral adipose inflammation in obesity is associated with critical alterations in tregulatory cell numbers. PLoS One. (2011) 6:e16376. doi: 10.1371/journal.pone.0016376
36. Jagannathan-Bogdan M, McDonnell ME, Shin H, Rehman Q, Hasturk H, Apovian CM, et al. Elevated proinflammatory cytokine production by a skewed T cell compartment requires monocytes and promotes inflammation in type 2 diabetes. J Immunol. (2011) 186:1162–72. doi: 10.4049/jimmunol.1002615
37. Annemarie K, Sari S, Dawn EA, Lauren JK, Eleanor MS, Alka MK, et al. Body fat distribution and inflammation among obese older adults with and without metabolic syndrome. Obesity (Silver Spring). (2010) 18:2354–61. doi: 10.1038/oby.2010.86
38. Nathalie E, Sylvie LP, Jacques P, André JS, Nicolas P. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract. (2014) 105:141–50. doi: 10.1016/j.diabres.2014.04.006
39. Tahereh F, Silvia L, Ali Mohammad PS, Milad A, Marjan T, Mehdi S, et al. An overview of the role of adipokines in cardiometabolic diseases. Molecules. (2020) 25:5218. doi: 10.3390/molecules25215218
40. Mathias F, Matthias B. Adipokines in health and disease. Trends Pharmacol Sci. (2015) 36:461–70. doi: 10.1016/j.tips.2015.04.014
41. Gianluca P, Giovanni G, Marco C, Maria R, Anna M, Antonio B, et al. IL-33 in mental disorders. Medicina (Kaunas). (2021) 57:315. doi: 10.3390/medicina57040315
42. Salvina Maria de CC, Aline Silva M, Ingrid C, Silva D, Amanda de O, Breno FC, et al. Serum levels of interleukin-33 and its soluble form receptor (sST2) are associated with cognitive performance in patients with schizophrenia. Compr Psychiatry. (2017) 74:96–101. doi: 10.1016/j.comppsych.2017.01.008
43. Milica MB, Slavica MJ, Ivan PJ, Nevena G, Nebojsa NA, Miodrag LL. IL-33/ST2 pathway and galectin-3 as a new analytes in pathogenesis and cardiometabolic risk evaluation in psychosis. Front Psychiatry. (2018) 9:271. doi: 10.3389/fpsyt.2018.00271
44. Milica B, Ivan J, Gordana R, Slavica DJD, Vesna S, Nebojsa A, et al. Antipsychotics can modulate the cytokine profile in schizophrenia: attenuation of the type-2 inflammatory response. Schizophr Res. (2013) 147:103–9. doi: 10.1016/j.schres.2013.03.027
45. Haozhe L, Qinting Z, Ningning L, Fan W, Hui X, Zongfeng Z, et al. Plasma levels of Th17- related cytokines and complement C3 correlated with aggressive behavior in patients with schizophrenia. Psychiatry Res. (2016) 246:700–6. doi: 10.1016/j.psychres.2016.10.061
46. Jayant M, Aparna S, Ravi Philip R, Muthuramalingam A, Vikas M, Negi VS, et al. An exploratory study of immune markers in acute and transient psychosis. Asian J Psychiatr. (2017) 25:219–23. doi: 10.1016/j.ajp.2016.11.010
47. Sara M, Ameneh ZS, Nima R. Cytokine alterations in schizophrenia: an updated review. Front Psychiatry. (2019) 10:892. doi: 10.3389/fpsyt.2019.00892
48. Milica B, Ivan J, Gordana R, Slavica DJD, Dragic B, Nebojsa A, et al. Elevated serum level of type-2 cytokine and low IL-17 in first episode psychosis and schizophrenia in relapse. J Psychiatr Res. (2012) 46:1421–6. doi: 10.1016/j.jpsychires.2012.08.016
49. Toby P, Emanuele FO, Stefan B, Valeria M, Robert AMCC, Oliver DH. A meta-analysis of immune parameters, variability, and assessment of modal distribution in psychosis and test of the immune subgroup hypothesis. Schizophr Bull. (2019) 45:1120–33. doi: 10.1093/schbul/sby160
50. Sonnig Sue-Whei C, Michael R, Markus S, Norbert M. Is T-helper type 2 shift schizophrenia-specific? Primary results from a comparison of related psychiatric disorders and healthy controls. Psychiatry Clin Neurosci. (2013) 67:228–36. doi: 10.1111/pcn.12040
51. Yong-Ku K, Aye-Mu M, Bun-Hee L, Chang-Su H, Heon-Jeong L, Dae-Jin K, et al. Th1, Th2 and Th3 cytokine alteration in schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. (2004) 28:1129–34. doi: 10.1016/j.pnpbp.2004.05.047
52. Marcella R, Erica C, Nigel HG. Cytokine imbalance in schizophrenia. from research to clinic: potential implications for treatment. Front Psychiatry. (2021) 12:536257. doi: 10.3389/fpsyt.2021.536257
53. Brian JM, Peter B, Wesley S, Andrew M, Brian K. Meta-analysis of cytokine alterations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. (2011) 70:663–71. doi: 10.1016/j.biopsych.2011.04.013
54. Balõtšev R, Koido K, Vasar V, Janno S, Kriisa K, Mahlapuu R, et al. Inflammatory, cardio- metabolic and diabetic profiling of chronic schizophrenia. Eur Psychiatry. (2017) 39:1–10. doi: 10.1016/j.eurpsy.2016.05.010
55. Jothimani G, Sailaxmi G, Dinakaran D, Venkatasubramanian G, Marimuthu P. Exercise, diet and educational interventions for metabolic syndrome in persons with schizophrenia: a systematic review. Asian J Psychiatr. (2018) 36:73–85. doi: 10.1016/j.ajp.2018.06.018
56. Clara MFP, Vicente Molina R, Juan I, Franch V. Metabolic syndrome and atypical antipsychotics: possibility of prediction and control. Rev Psiquiatr Salud Ment. (2017) 10:38–44. doi: 10.1016/j.rpsm.2016.09.003
57. Stefan L, Andrea C, Loukia S, Dimitris M, Deniz O, Franziska R, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. (2013) 382:951–62. doi: 10.1016/S0140-6736(13)60733-3
Keywords: schizophrenia, cytokine, metabolic syndrome, risperidone, clozapine, aripiprazole
Citation: Koricanac A, Tomic Lucic A, Veselinovic M, Bazic Sretenovic D, Bucic G, Azanjac A, Radmanovic O, Matovic M, Stanojevic M, Jurisic Skevin A, Simovic Markovic B, Pantic J, Arsenijevic N, Radosavljevic GD, Nikolic M, Zornic N, Nesic J, Muric N and Radmanovic B (2022) Influence of antipsychotics on metabolic syndrome risk in patients with schizophrenia. Front. Psychiatry 13:925757. doi: 10.3389/fpsyt.2022.925757
Received: 21 April 2022; Accepted: 30 June 2022;
Published: 25 July 2022.
Edited by:
Izabela Guimaraes Barbosa, Federal University of Minas Gerais, BrazilReviewed by:
Breno Fiuza Cruz, Federal University of Minas Gerais, BrazilMariusz Stanisław Wiglusz, Medical University of Gdańsk, Poland
Copyright © 2022 Koricanac, Tomic Lucic, Veselinovic, Bazic Sretenovic, Bucic, Azanjac, Radmanovic, Matovic, Stanojevic, Jurisic Skevin, Simovic Markovic, Pantic, Arsenijevic, Radosavljevic, Nikolic, Zornic, Nesic, Muric and Radmanovic. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Aleksandra Tomic Lucic, YXRvbWljbHVjaWNAZ21haWwuY29t
 Aleksandra Koricanac
Aleksandra Koricanac Aleksandra Tomic Lucic3,4*
Aleksandra Tomic Lucic3,4* Mirjana Veselinovic
Mirjana Veselinovic Danijela Bazic Sretenovic
Danijela Bazic Sretenovic Aleksandra Jurisic Skevin
Aleksandra Jurisic Skevin Bojana Simovic Markovic
Bojana Simovic Markovic Nebojša Arsenijevic
Nebojša Arsenijevic Gordana D. Radosavljevic
Gordana D. Radosavljevic Maja Nikolic
Maja Nikolic Nemanja Muric
Nemanja Muric Branimir Radmanovic
Branimir Radmanovic