
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Plant Sci. , 21 February 2025
Sec. Plant Symbiotic Interactions
Volume 16 - 2025 | https://doi.org/10.3389/fpls.2025.1542585
This article is part of the Research Topic Integrative Techniques to Alleviate Abiotic Stress in Plants Using Plant Growth Promoting Bacteria and Fungi: Mechanisms, Interactions, and Applications View all 4 articles
 Zhengmei Li1†
Zhengmei Li1† Yueyu Ye1†
Yueyu Ye1† Xia Wang1†
Xia Wang1† Suyu Peng1†
Suyu Peng1† Beibei Chen2
Beibei Chen2 Shiqing Li1
Shiqing Li1 Haimin Chen3
Haimin Chen3 Dongfeng Yang3
Dongfeng Yang3 Fusheng Jiang2
Fusheng Jiang2 Chunchun Zhang1*
Chunchun Zhang1* Meiya Li1,4*
Meiya Li1,4*Mycorrhizal fungi have been shown to promote seed germination and seedling growth in Orchidaceae plants. In the present study, a mycorrhizal fungus designated as BJ1 was isolated from the roots of Bletilla striata (Thunb.) Reiehb.f. Fluorescence staining and morphological analysis revealed that this fungus exhibited characteristics highly similar to those of Tulasnella. Subsequently, the strain was confirmed as a new strain of Tulasnella through sequencing and phylogenetic analysis of four loci: the internal transcribed spacer region ITS1-ITS4 (ITS), ATP synthase (C14436), glutamate synthase (C4102), and ATP deconjugase (C3304). Additionally, we investigated the in vitro biological activity of strain BJ1 and its effects on germination and growth of B. striata seeds. The results indicated that BJ1 is capable of producing plant cell-degrading enzymes, including pectinase and protease. Furthermore, it demonstrates an ability to solubilize inorganic phosphorus and synthesize indoleacetic acid (IAA). Nevertheless, it does not exhibit laccase activity or possess the capacity to produce siderophores, nor can it solubilize organic phosphorus. Microscopic observations revealed that strain BJ1 mainly colonizes the base of the B. striata protocorm, thereby enhancing seed germination, growth, and expansion. Notably, by the fourth week of germination, 74.23% of seeds in the symbiotic group had developed to stage 5, a significantly higher proportion compared to 50.43% in the non-symbiotic group. Additionally, the length, width, and fresh weight of seeds in the symbiotic group were 2.2 times, 1.8 times, and 3.7 times greater than those in the non-symbiotic group, respectively. Furthermore, by adding L-tryptophan as a substrate during co-cultivation with BJ1, there was a significant enhancement in IAA synthesis capability; this also led to a marked acceleration in the symbiotic germination process of B. striata seeds. These results suggest that strain BJ1 holds significant potential for application in the artificial propagation of B. striata seedlings. It can enhance propagation efficiency and improve seedling quality, thereby playing a crucial role in the conservation and sustainable development of germplasm resources of endangered orchids.
Bletilla striata (Thunb.) Reiehb.f. (B. striata) is a perennial herb belonging to the Orchidaceae family, mainly distributed in Japan, South Korea and coastal areas of China. As a traditional Chinese herbal medicine, the underground part (tuber) of B. striata has been utilized for thousands of years due to its properties such as astringency, hemostasis, detumescence, and promotion of granulation. This herbal remedy is predominantly employed in the treatment of traumatic hemorrhage, hematemesis, chapped skin, swelling, and ulcer (Kong et al., 2021; Zhao, 1963). B. striata not only possesses significant medicinal value but also serves as an essential raw material in industrial production (Pan et al., 2018).
Previously, B. striata available on the market was mainly sourced from wild collections. This situation can be attributed to the low rate of natural seedling formation due to inadequate seed development, and the underdeveloped artificial propagation techniques. However, with the expansion of market demand, overexploitation of wild B. striata populations has led to a significant depletion of its wild resources. In recent years, advancements in artificial breeding technology have led to the establishment of various seedling cultivation methods for B. striata, such as ramet propagation (Zhang et al., 2019; Ren et al., 2016), tissue culture techniques (Zou et al., 2013), seed direct seeding, and symbiotic germination, which have effectively mitigated resource shortages in the market (Pan et al., 2018). Ramet propagation is a crucial early-stage method characterized by simple operation procedures with high seedling rates and short cultivation periods (Ren et al., 2016). However, this approach suffers from low propagation efficiency; prolonged vegetative propagation through ramets can result in virus accumulation along with variety degradation that adversely affects yield quality (Zhang et al., 2019). Tissue culture offers higher propagation efficiency but incurs substantial costs related to equipment and labor, as well as involving multiple complex procedures (Zhang et al., 2019). Direct seeding technology is simple; however it incurs high management costs while yielding relatively low seedling rates. In comparison to these three aforementioned reproductive methods, symbiotic germination presents distinct advantages. This technique not only enhances the germination rate as well as both seedling success rates and transplant survival rates for B. striata seeds but also saves costs, thereby better aligning with market demands and simultaneously protecting wild B. striata resources (Pan et al., 2018; Zhang et al., 2005; Xu et al., 2019).
The earliest technology for symbiotic germination can be traced back to 1899, when Bernard discovered that fungal infection is essential for the germination and growth of orchid seeds. Fungi provide the necessary nutrients required for their germination, leading to the breakthrough establishment of symbiotic germination technique. This method plays a crucial role in both research and conservation efforts concerning Orchidaceae plants (Xu and Xu, 2007; Gao et al., 2019; Fan et al., 2003). Mycorrhizal fungi include ectomycorrhiza and endophytic mycorrhiza. Endophytic mycorrhizal fungi are those whose mycelia can invade the root cells of higher plants while coexisting harmoniously with them, thereby promoting plant growth (Zhao et al., 2021). The mycelium of mycorrhizal fungi penetrates plant root cells to form specialized structures such as arbuscular (the main site for nutrient transfer between fungi and plants) and vesicles (which form in cortical cells and act as storage organs for lipids and other resources), facilitating efficient nutrient exchange. Mycorrhizal fungi can engage in bidirectional nutrient exchange with plants through their intricate mycelial networks in the soil. Plants supply the fungi with fixed carbon, such as sugars, while the fungi provide essential minerals, thereby enhancing the efficiency of nutrient uptake by plants (Harley and Smith, 1983). Furthermore, Mycorrhizal fungi not only promote seed germination and growth but also enhance the utilization rates of phosphorus and potassium, secrete plant growth-regulating hormones, thereby promoting overall plant development and significantly improving host adaptability to adverse environmental conditions (Zhao et al., 2021; Yang et al., 2024; Miura et al., 2018). Furthermore, in certain medicinal plants, mycorrhizal fungi can also increase the content of their active components (Wang et al., 2023).
Orchidaceae seeds lack complete endosperm, making them challenging to germinate and grow under natural conditions. These seeds interact with microorganisms to form a unique symbiotic system for germination and growth (Yang et al., 2020). At present, a large number of symbiotic fungi have been isolated and identified from Orchidaceae plants. For example, Gao et al. (2019) identified Tulasnella sp.–a fungus capable of promoting seed germination in approximately forty different Orchidaceae species-while Ceratobasidium sp. has been shown to facilitate seed germination in over twenty species within this family. The symbiotic relationship between mycorrhizal fungi and Orchidaceae seeds plays an irreplaceable role in supporting their growth under natural conditions. As an effective strategy for conserving Orchidaceae resources, such fungi have been widely utilized in the reproduction and cultivation of these Orchidaceae plants (Batty et al., 2006).
B. striata exhibits seed germination challenges similar to those faced by other members of the Orchidaceae family. Therefore, finding mycorrhizal fungi that can facilitate the symbiotic germination and growth of B. striata seeds is essential for achieving efficient and rapid propagation of this species. Numerous studies have reported on mycorrhizal fungi that promote the germination and growth of B. striata seeds. For example, Guo and Xu (1992) found that four fungal species, including Myeena osmundieola Lange, could promote both seed germination and seedling development in B. striata seeds. Xu et al. (2019) investigated four different mycorrhizal fungi species regarding their effects on seed germination and seedling growth in B. striata; they found significant variations in symbiotic performance among different mycorrhizal fungi when interacting with B. striata.
In our current study, we isolated a mycorrhizal fungus designated as BJ1 from the roots of B. striata; it was identified as belonging to Tulasnella sp., exhibiting strong capabilities for promoting germination and growth. We systematically examined its biological characteristics-including enzyme activity related to parasitism and nutrient absorption-as well as its capacity for plant hormone production-and analyzed its impact on the symbiotic germination process of B. striata seeds over time. Our findings provide valuable technical support for the efficient cultivation of high-quality seedlings of B. striata in the future horticultural practices.
The strain BJ1 was isolated from the fibrous roots of B. striata (Figure 1; Supplementary Figure S1) and has been deposited at the China General Microbiological Culture Collection Center, with a preservation No. 40773. The BJ1 culture was transferred to potato dextrose agar (PDA) and incubated at 28°C for 1 week (Zhu et al., 2012; Daniel et al., 2022). The basic morphological characteristics of BJ1 colonies were observed and recorded, including color, morphology, growth rate, and cell characteristics. Then, the microstructure of hyphae was observed using both a light microscope and an electron microscope (field emission scanning electron microscopy, FESEM; SU8010, Japan), focusing on hyphal structure and cell morphology.

Figure 1. Biomorphology of B striata. B striata plant (A), fibrous root (B), tuber (C) and fruit clips (D).
The BJ1 strain was inoculated onto four different culture media: ½ FIM medium, E-medium, 3MN/Magali’s Modified Melin-Norkrans medium and ¼ PDA medium. A sterile slide was then diagonally inserted into the culture medium and incubated in a biochemical incubator (SPX-70BX, Tianjin Instrument Co., LTD, China) at 27°C in the dark for 5 days (Arifin et al., 2020). Once the slide was covered with growth from the BJ1 strain, it was stained with Hoechst 33342. The Hoechst 33342 solution was prepared according to the protocol reported by Arifin et al. (2020), utilizing phosphate buffer solution at pH 7.8 to dissolve Hoechst 33342, which facilitating clearer observation of both the cell nucleus and septum of the strain. After staining with Hoechst 33342 solution in darkness for 20 min and rinsing with sterile water, samples were scanned using a fluorescence scanning analyzer (VS120-S6-W, OLYMPUS, Japan) at magnifications of ×40 and ×100. Finally, morphological identification of the fungi was conducted following guidelines outlined in the Fungi Identification Handbook (Wei, 1979).
According to the report by Ruibal et al. (2013), PCR amplification was conducted on four gene loci: the ribosomal internal transcribed spacer (ITS1-ITS4), glutamic synthetase (C4102), ATP synthetase (C14436), and ATP helicase (C3304), using the specific primers and PCR protocols detailed in Table 1. The PCR products were subsequently separated and purified via agarose gel electrophoresis and submitted to Shanghai Sangon Biotechnology Co., Ltd. for sequencing. The obtained sequences were analyzed using MEGA v.7 software. The evolutionary history was inferred by using the Maximum Likelihood method based on the Kimura 2-parameter model. Initial tree(s) for the heuristic search were obtained by applying Neighbor-Join and BioNJ algorithms to a matrix of pairwise distances estimated using the Maximum Composite Likelihood (MCL) approach, and then selecting the topology with superior log likelihood value. The analysis involved 22 nucleotide sequences. Codon positions included were 1st+2nd+3rd+Noncoding. All positions containing gaps and missing data were eliminated. There were a total of 535 positions in the final dataset.
BJ1 was cultured on Potato Dextrose Agar (PDA) for 5 to 8 days. Subsequently, the culture was punched in the medium using the end of a sterile gun head (φ = 10 mm) and inoculated at the center of various screening media: PDA-ABTS (2, 2’-diazo-bis-3-ethylbenzothiazolin-6- sulfonic acid) screening medium (ABTS 0.03%, KNO3 3 g/L in PDA medium, sterilized at 121°C for 20 min), pectinase screening medium (NH4Cl 0.4g/L, K2HPO4 1 g/L, MgSO4·7H2O 2 g/L, FeSO4·7H2O 0.01 g/L, pectin 8 g/L, agar 20 g/L; pH 7, sterilized at 121°C for 20 min), and protease screening medium (skimmed milk powder 75 g/L, agar 15 g/L, sterilized at 110°C for 10 min) respectively (Zhu, 2018; Chi et al., 2022; Zhou et al., 2020). Each treatment group was set up with three biological replicates and incubated in a biochemical incubator at 25°C in the dark for six days. The enzymatic activity was evaluated based on the presence of a clear zone around these colonies. The enzymatic index (EI) was calculated using the formula EI = diameter (mm) of the discolored halo/diameter (mm) of the colonies (DeFátima et al., 2021).
BJ1 was inoculated onto Monkina phosphorus-free medium, which consisted of C6H12O6 10.00 g/L, (NH4)2SO4 0.50 g/L, NaCl 0.30 g/L, KCl 0.30 g/L, FeSO4·7H2O 0.03 g/L, MnSO4·4H2O 0.03 g/L, MgSO4·7H2O 0.30 g/L, Ca3(PO4) 2 5.00 g/L, agar 20.00 g/L, and water up to 1000 mL; the pH was maintained between 7.00 and 7.50, with sterilization conducted at 115°C for 30 min. Additionally, BJ1 was cultured on organic phosphorus solid medium prepared by cooling 1000 mL of beef extract peptone medium to below 50°C, followed by the immediate addition of 60 mL sterile saline and egg yolk liquid in a ratio of 1:1 before pouring into plates. Observation of a clear halo surrounding the colonies indicated that the strain exhibited phosphate-solubilizing activity (Li et al., 2021; Chen et al., 2009; Xue et al., 2019).
BJ1 was cultured at 25°C for a period of 7 days, after which it was transferred to CAS medium. The composition of the CAS medium included: 60.5 mg of CAS, 72.9 mg of cetyltrimethyl ammonium bromide (HDTMA), 10 mL of a 1 mmol/L FeCl3·6H2O (dissolved in 10 mmol/L HCl), along with 50 mL of a 0.1 mol/L phosphate buffer, 9.0 g of agar, and deionized water to make up a total volume of 1000 mL (Xu et al., 2022). The appearance of an orange siderophore chelate indicates that the strain exhibits siderophore-producing activity.
The BJ1 strain was inoculated into PDA liquid medium, either supplemented with L-tryptophan (100 mg/L) or without supplementation. Cultures were incubated at 25°C on a shaker (SKY-211B, Jingda Instrument manufacturing Co., LTD, China) set to 200 rpm for 5 days. Following incubation, the cultures were centrifuged at 12,000 rpm for 2 min. The supernatants were then mixed in a 1:1 ratio with Salkowski reagent (50 mL, 35% of perchloric acid, 1 mL 0.5 M FeCl3 solution) and transferred to a colorimetric cup for observation of any color changes. The presence of indole-3-acetic acid (IAA) or similar compounds in the supernatant will result in the development of a pink color upon reaction with Salkowski reagent (Wang et al., 2020; Zhang et al., 2016). A darker hue indicates a higher secretion capacity (Patel et al., 2018).
Mycelium blocks (9 mm in diameter) of the BJ1 strain, cultivated on PDA for 5 days were transferred into 200 mL of PDA liquid supplemented with 100 mg/L L-tryptophan and cultured at 25°C and agitated at 200 rpm. The fermentation supernatant and mycelium were subsequently collected, and the IAA secretion capability of BJ1 strain was evaluated using fermentation broth from the BJ1 strain without L-tryptophan as a control (Zhang et al., 1999; Cheng et al., 2022).
(Extraction of fermentation broth) A total of 500 mL each of BJ1 fermentation broth containing L-tryptophan and that not containing L-tryptophan was transferred to a round-bottom flask and concentrated to 35 mL under reduced pressure at 50°C. Then, 105 mL of absolute ethanol was added to make a final concentration of ethanol of approximately 75%. After filtration and concentration, the residues were dissolved in 75% ethanol to 5 mL and filtered using a 0.22 μm Millipore filter (Merck Millipore, Germany) for High Performance Liquid Chromatography (HPLC) analysis.
(Extraction of mycelial contents) The hyphae were dried, ground into powder, weighed (6.0 g), and refluxed with 100 mL of 75% ethanol for two hours. A portion measuring five milliliters from this mycelium extract was taken for Salkowski detection. The extracts were concentrated under reduced pressure and dissolved with 75% ethanol to 5 mL, and then filtered through a 0.22 μm Millipore filter for HPLC analysis.
Gibberellin (GA3), IAA, abscisic acid (ABA), 1-naphthylacetic acid (NAA), 6-benzylaminopurine (6-BA), and kinetin (KT) were accurately weighed and dissolved in 75% ethanol to prepare a standard solution, each with a concentration of 1 mg/mL. The IAA stock solution was diluted with 75% ethanol to prepare a series of IAA standard solutions, which were then injected into the HPLC system for the construction of the standard curve (Wang et al., 2018; Shah et al., 2021). Samples were injected into an UltiMate3000 HPLC system (Diane, California, USA) coupled with a GL SCIENCES Inertsil ODS-EP C18 column (4.6 × 250 mm, 5 μm), and gradient elution with acetonitrile and 0.1% formic acid aqueous solution at a flow rate of 1 mL/min as detailed in Table 2. Detection was carried out at a wavelength of 270 nm (Wu, 2019).
Fully mature B. striata fruit clips were selected for measurement. The seeds of B. striata were found to be approximately 1 to 2 millimeters in length and 0.2 millimeters in width, with a thousand-seed weight ranging from 1~2×10-3 g. The fruit clips underwent disinfection through a sequential treatment involving immersion in 75% ethanol for 5 minutes, followed by exposure to a solution of 4% sodium hypochlorite for an additional 7 minutes; they were then rinsed five times with sterile water. The fruit clip was then dissected, and the seeds were suspended in a 1% agar solution. Subsequently, approximately 100 seeds were evenly dispersed on agar medium for symbiotic cultivation, with non-symbiotic cultivation serving as the control. Seeds were cultured in an artificial climate chamber (LISK Instrument Equipment Co., LTD, NanJing, China) at a temperature of 25°C under conditions of 12 h light and 12 h darkness. The germination rate of B. striata seeds was monitored weekly over a four-week period according to criteria specified in previous publications (Stewart and Zettler, 2002; Tian et al., 2023). The stages of seed germination and seedling development in Orchidaceae plants are classified as follows: stage 0 indicates no germination; stage 1 involves rupture of the testa due to embryo expansion; stage 2 marks the formation of protocorms through spherical expansion of the embryo; stage 3 is characterized by the appearance of protomeristems; stage 4 denotes leaf emergence; and stage 5 signifies leaf elongation leading to seedling formation. Furthermore, from each treatment group, three sets of protocorms were randomly collected on a weekly basis, with each set consisting of 15 protocorms. Their length, width, and fresh weight were measured using the method described by Yamamoto (Yamamoto et al., 2017).
Symbiotic and non-symbiotic protocorms were randomly selected to observe fungal colonization. The protocorms were transferred to 1.5 mL Eppendorf tubes, treated with a 10% KOH solution by heating in a water bath at 90°C for 8 hours. Following this, the KOH solution was removed, and the protocorms were washed three times. They were then sequentially soaked in 10% hydrogen peroxide solution and 5% acetic acid for 1 h each. Finally, the protocorms were stained with a 0.04% Trypan blue solution for 10 min. Fungal colonization was observed with a microscope (Jiang et al., 2019).
B. striata seeds were evenly distributed across four distinct types of 1.5% agar medium, which included symbiotic agar media both with and without the addition of 100 mg/L L-tryptophan, as well as non-symbiotic agar media under the same conditions. The cultures were incubated in an artificial climate chamber maintained at 25°C with a photoperiod of 12 h light and 12 h darkness. Fifteen B. striata protocorms were collected every 7 days. The fresh weight, width, and length of B. striata seeds were determined. The synergistic effect of L-tryptophan in conjunction with BJ1 on the growth rate of B. striata seeds was evaluated using the growth rate model G = (a − b)/b × 100% (Yamamoto et al., 2017; Jahn et al., 2021).
GraphPad Prism 8.0 (GraphPad Software, La Jolla, CA, USA) was utilized for data processing, statistical analysis, and graphical representation. All data are expressed as mean ± standard deviation. One-way ANOVA was employed to compare differences between groups, followed by Tukey’s test for multiple comparisons. An independent-samples t-test was used to analyze the differences between the two groups. p < 0.05 was considered statistically significant.
After an incubation period of 8 days at 25°C, the BJ1 strain exhibited the development of white to cream-colored colonies on PDA, characterized by concentric rings and undulated, submerged edges. The aerial mycelium was not well-developed (Figure 2A). Microscopic observation showed that BJ1 could produce elliptical monilioidcells at the apex of the mycelia (Figure 2B), a finding further corroborated by scanning electron microscopy (Figures 2C, D). In addition, many mycelia frequently formed clusters and produced lateral branches at typical right angles or acute angles (Figures 2E, F) (Freitas et al., 2020; Linde et al., 2017). Scanning electron microscopy revealed that the monilioid cells of the BJ1 strain exhibited significant morphological variability, typically appearing spherical or ellipsoidal with diameters ranging from 5 to 10 µm. These cells formed chain-like structures, which could be branched, with each branch usually comprising 2 to 5 monilioid cells. The nuclear staining indicates binuclear cells (Figure 2F). There are structural similarities between the morphological observations and those described for Tulasnella sp.
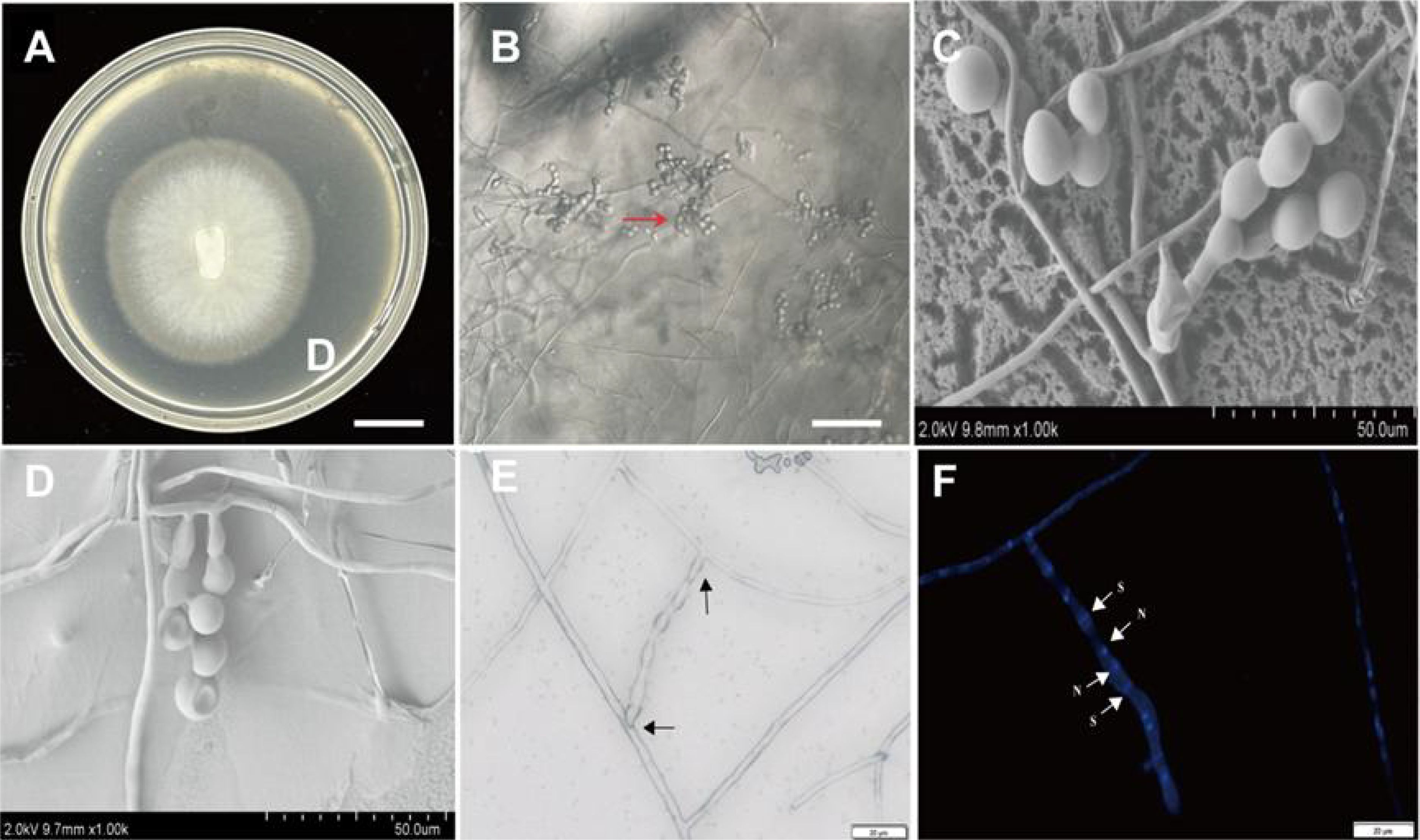
Figure 2. Morphological characteristics of BJ1. (A) Colony morphology of BJ1 cultured for 8 days (bar = 1 cm); (B) morphology of mycelium and monilioid cells under a microscope (bar =100 μm); (C) Morphology of hyphae and monilioid cells under a scanning electron microscope (bar = 50 μm); (D) mycelium interweaving and branching at right angles under a scanning electron microscope (bar = 50 μm); (E) The morphology of the hyphae under visible light, showing with branching at right angles, cylindrical hyphae, with cylindrical terminal elements (black arrow) bars = 50 µm; (F) The morphology of hyphae stained with Hoechst 33342, showing binucleate cells (white arrow, N = nuclei; S = septa);. Bars = 20 µm.
According to the culture medium types used in reported articles (½ FIM medium, E-medium, 3MN/Magali’s Modified Melin-Norkrans medium and ¼ PDA medium) and the same medium conditions, BJ1 was cultured in the same way (Arifin et al., 2020; Linde et al., 2017; Ruibal et al., 2013). The results showed that after the same culture for 4 weeks, BJ1 hyphae had completely covered the entire medium, and the hyphae growth rate was faster than that of the known Tulasnella mycorrhizal fungus. The aerial hyphae of the BJ1 strain are more developed; it is quite different from the reported new Tulasnella mycorrhizal fungi (Figure 3). Nevertheless, a more rigorous and systematic comparison of the related strains under standardized conditions is essential to elucidate their differences in growth characteristics.
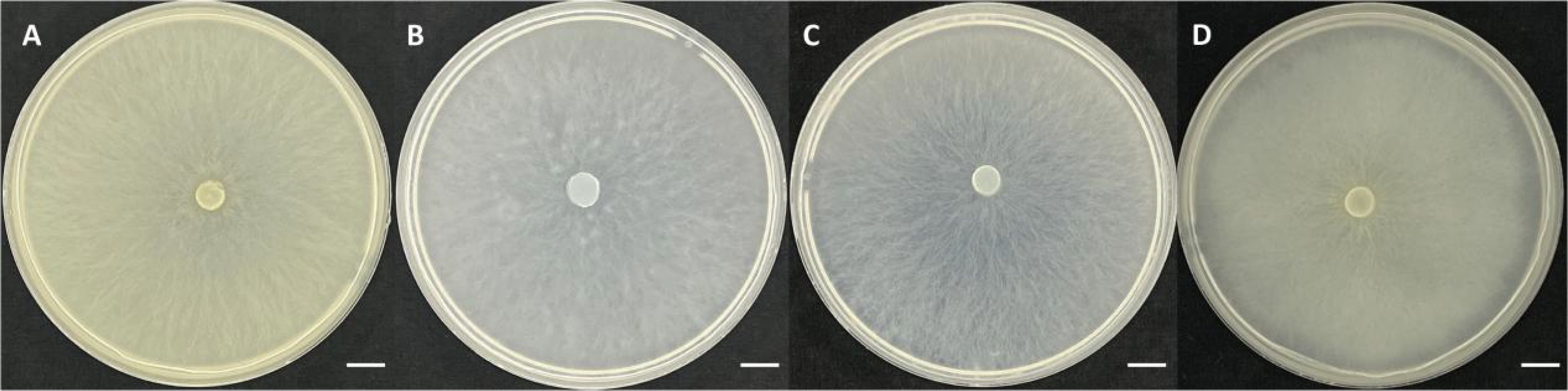
Figure 3. The morphology of strain BJ1 cultured on (A) ¼ PDA, (B) ½ FIM, (C) 3MN + vitamin, and (D) E-medium. Bars = 1cm.
Molecular identification indicated that BJ1 (OR186273.1) and Tulasnella sp. (OQ678396.1) clustered within the same phylogenetic branch. A comparison of their ITS sequences showed a query cover of 93%, with a similarity percentage of only 90.56% (Figure 4). The BJ1 strain was still branching independently, according to the results of the phylogenetic tree that was built using the other loci (C14436, C4102, and C3304). All three loci had high bootstrap (>90) and BPP values (Supplementary Figures S2-S4). Consequently, the strain BJ1 is likely representative of a novel species within the Tulasnella genus that has not been previously documented.
The results demonstrated that BJ1 could secrete extracellular enzymes, including pectinase and protease (Figures 5A, B), with respective activity indices of 0.30 ± 0.05 and 1.32 ± 0.08. However, the absence of a discernible transparent circle on the PDA-ABTS screening medium suggests that BJ1 may have a limited ability to secrete laccase.
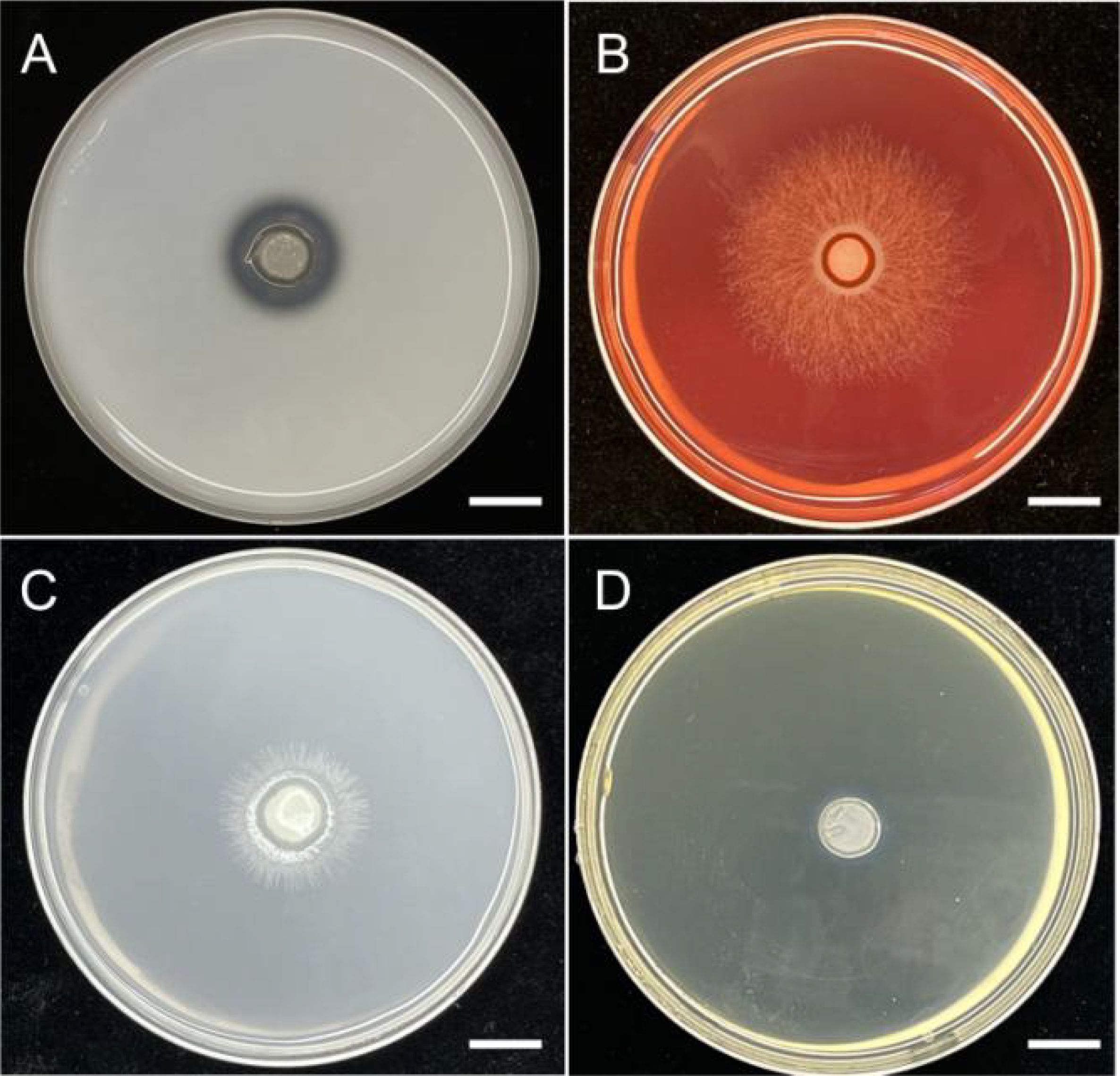
Figure 5. Enzyme activity analysis of BJ1 strain. Transparent circles appear on the culture medium in primary screening of (A) protease, (B) pectinase, (C) inorganic phosphate-solubilizing activity, and (D) siderophore-producing activity (bar = 1 cm).
The strain inoculated on Montina inorganic phosphorus medium was distinctly surrounded by a transparent circle (Figure 5C), indicating that the BJ1 strain possesses a certain capacity to dissolve inorganic phosphorus, with an enzyme index of 0.59 ± 0.03. However, no noticeable transparent circle was observed around the strain on the organic phosphorus solid medium, implying that the BJ1 strain may not be capable of dissolving organic phosphorus (Table 3). In addition, when the BJ1 strain was inoculated onto the CAS plate, it did not produce yellow siderophore, indicating that BJ1 lacks the ability to produce siderophore (Figure 5D).
The secretion of growth hormone represents one molecular mechanism through which mycorrhizal fungi facilitate the growth and germination of orchid seeds (Patel et al., 2018; Wu et al., 2002). The results indicated that the unfermented control PDA liquid medium, regardless of whether L-tryptophan was present, did not exhibit a significant color reaction; In contrast, both the fermentation supernatant and mycelial extract from BJ1 cultured in PDA liquid medium supplemented with L-tryptophan displayed a distinct pale pink coloration. Surprisingly, neither the BJ1 fermentation broth nor the mycelia extract of PDA liquid culture without L-tryptophan showed any notable color change (Figure 6A). These findings indicated that strain BJ1 is capable of synthesizing IAA when L-tryptophan serves as a substrate; however, no positive reaction was detected in the absence of L-tryptophan, which may be attributed to the low content of IAA. Furthermore, HPLC analysis was performed to analyze if BJ1 might produce other types of plant hormones aside from IAA. As depicted in Supplementary Figure S5, no additional hormones were detected in the concentrated extract of BJ1 fermentation broth apart from IAA. Thus, we further analyzed IAA production under varying fermentation conditions. Remarkable IAA peaks were detected in both the culture supernatant and mycelium extracts derived from BJ1 fermentation with L-tryptophan (Figure 6B, 1 and 2). In addition, traces of IAA were also found in both the fermentation supernatant and mycelium extract without L-tryptophan (Figure 6B, 3 and 4), but the content was significantly lower than those obtained with L-tryptophan supplementation. The content of IAA under different culture conditions was also quantified based on a standard curve: Y = 14.90X − 0.076, R2 = 0.999. The concentrations measured for IAA in fermentation broths with and without L-tryptophan were determined to be 0.0217 mg/mL and 0.0076 mg/mL, respectively; similarly, for dried mycelia they were recorded at concentrations of 0.0188 mg/g and 0.0061 mg/g, respectively.
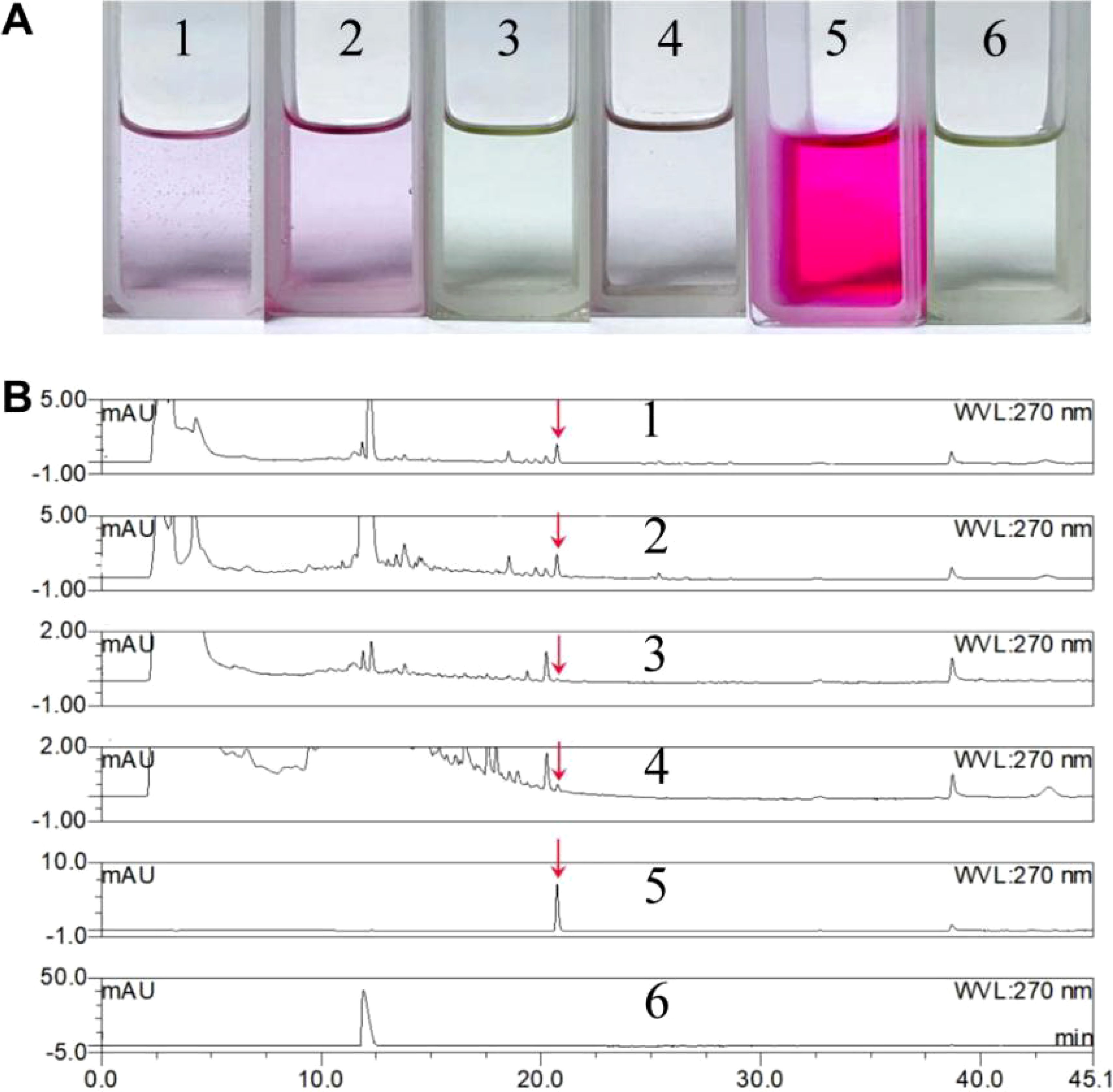
Figure 6. Qualitative and quantitative analysis of IAA. (A) Salkowski colorimetry; (B) HPLC analysis. 1 BJ1 fermentation (with L-tryptophan) broth extract, 2 BJ1 fermentation (with L-tryptophan) mycelia extract, 3 BJ1 fermentation (without L-tryptophan) broth extract, 4 BJ1 fermentation (without L-tryptophan) mycelia extract, 5 the standard of IAA, and 6 the standard of L-tryptophan. The red arrow indicates the peak of IAA.
According to the criteria established by Stewart and Zettler (2002) for the germination and growth of Orchidaceae plants, we calculated the symbiotic germination rates of mycorrhizal fungi BJ1 and B. striata seeds at different time intervals (Figure 7). The results showed that mycorrhizal fungus BJ1 significantly promoted the germination of B. striata seeds. The germination rate in the symbiotic group was notably higher than that in the control group (Table 4). In the first week, both groups reached stage 2 with percentages of 71.32 ± 8.12% for the symbiotic group and 71.17 ± 8.15% for the control group, showing no significant difference between them. However, only the symbiotic group reached stage 3 at a rate of 12.42 ± 4.11%, while no seeds from the non-symbiotic group reach this stage. Similarly, during weeks 2 and 3 of cultivation, growth in the symbiotic group outpaced that in the control group, with percentages reaching stages 4 and 5 at rates of 16.16 ± 4.25% and 15.22 ± 3.49%, respectively. By week 4, a significantly higher percentage of seeds in the symbiotic group had attained stage 5 (74.23 ± 11.21%) compared to those in the control group (53.23 ± 11.35%). Significant differences were observed between these two groups.
Furthermore, we conducted weekly comparisons regarding growth morphology, size, and weight among representative seeds from each treatment group (Figure 8A). After 1 week of culture, B. striata seeds began absorbing water and expanding into rounder shapes; however, there was no significant difference noted between the symbiotic group and non-symbiotic group. After 2–4 weeks of culture, the disparity in length and width between both groups progressively increased. The symbiotic group showed incremental growth with a higher growth rate, whereas the non-symbiotic group showed steady growth with a lower growth rate. This may be attributed to the symbiotic relationship between strain BJ1 and B. striata seeds, which accelerates its nutrient supply and plant hormone secretion, thereby promoting the rapid growth of the B. striata protocorm (Figure 8B). After 2–4 weeks of culture, significant statistical differences in protocorm weight were also noted between the symbiotic and non-symbiotic groups (Figure 8C).
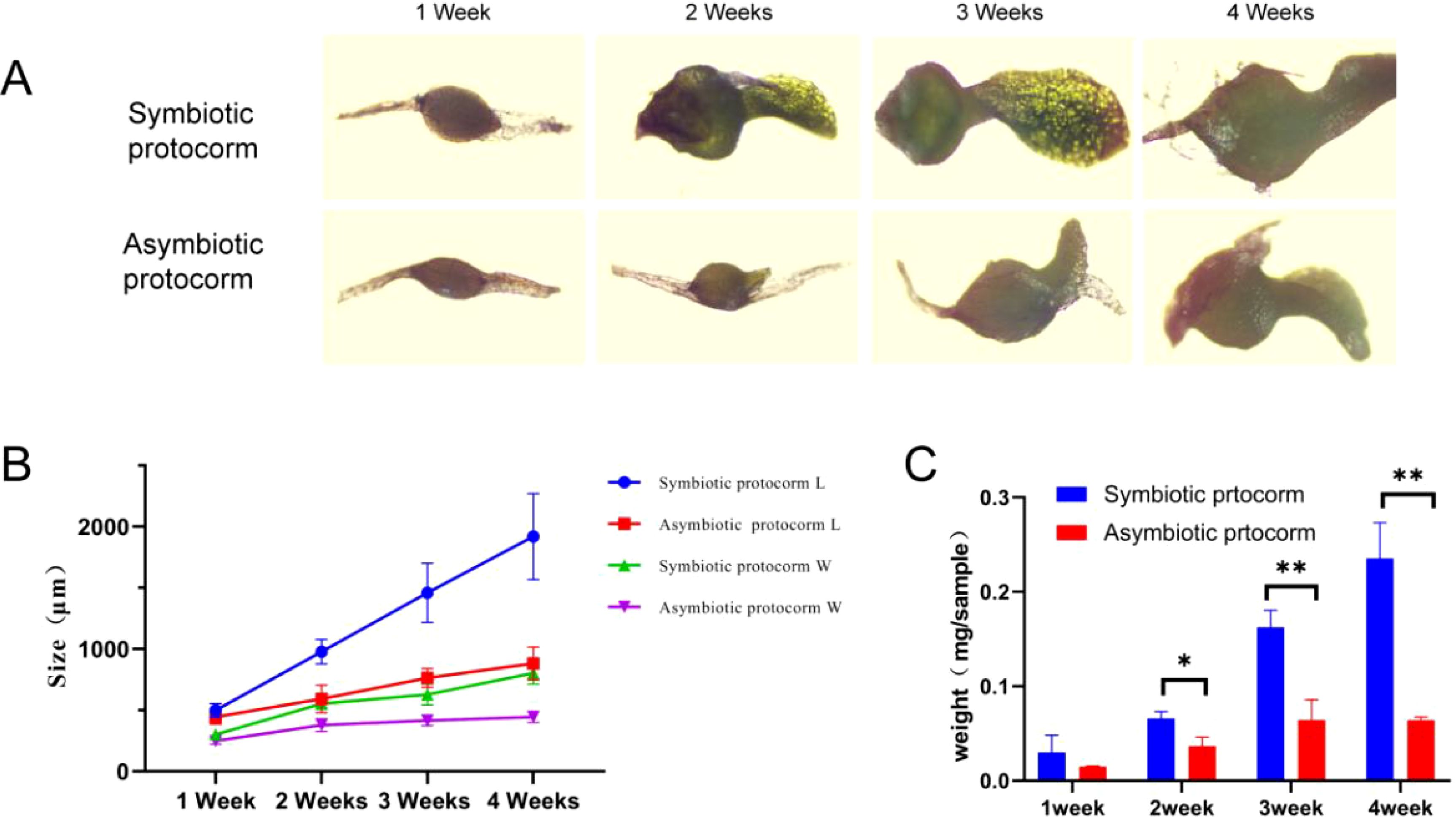
Figure 8. Effect of BJ1 symbiosis on the growth of B striata protocorm. (A) Effects of symbiosis and non-symbiosis culture on the growth of B striata protocorms. Scale = 500 μm; (B) length and width changes of B striata protocorms under symbiotic and non-symbiotic conditions. Solid circles and triangles represent the length (L) and width (W) of the symbiotic group, respectively. Rectangles and inverted triangles represent the length (L) and width (W) of non-symbiotic groups, respectively; (C) weight of symbiotic and non-symbiotic B striata protocorms. The error line represents the standard error of the average value of 15 protocorms. Independent-samples t-tests were used to determine the statistically significant differences in seed weight: *p < 0.05, **p < 0.01.
Trypan blue staining demonstrated that the strain BJ1 primarily colonized at the base of the protocorm (Figure 9A); conversely, no mycelia clusters were observed in the control group (Figure 9B). This observation aligns with findings reported by Jiang (Jiang et al., 2019).
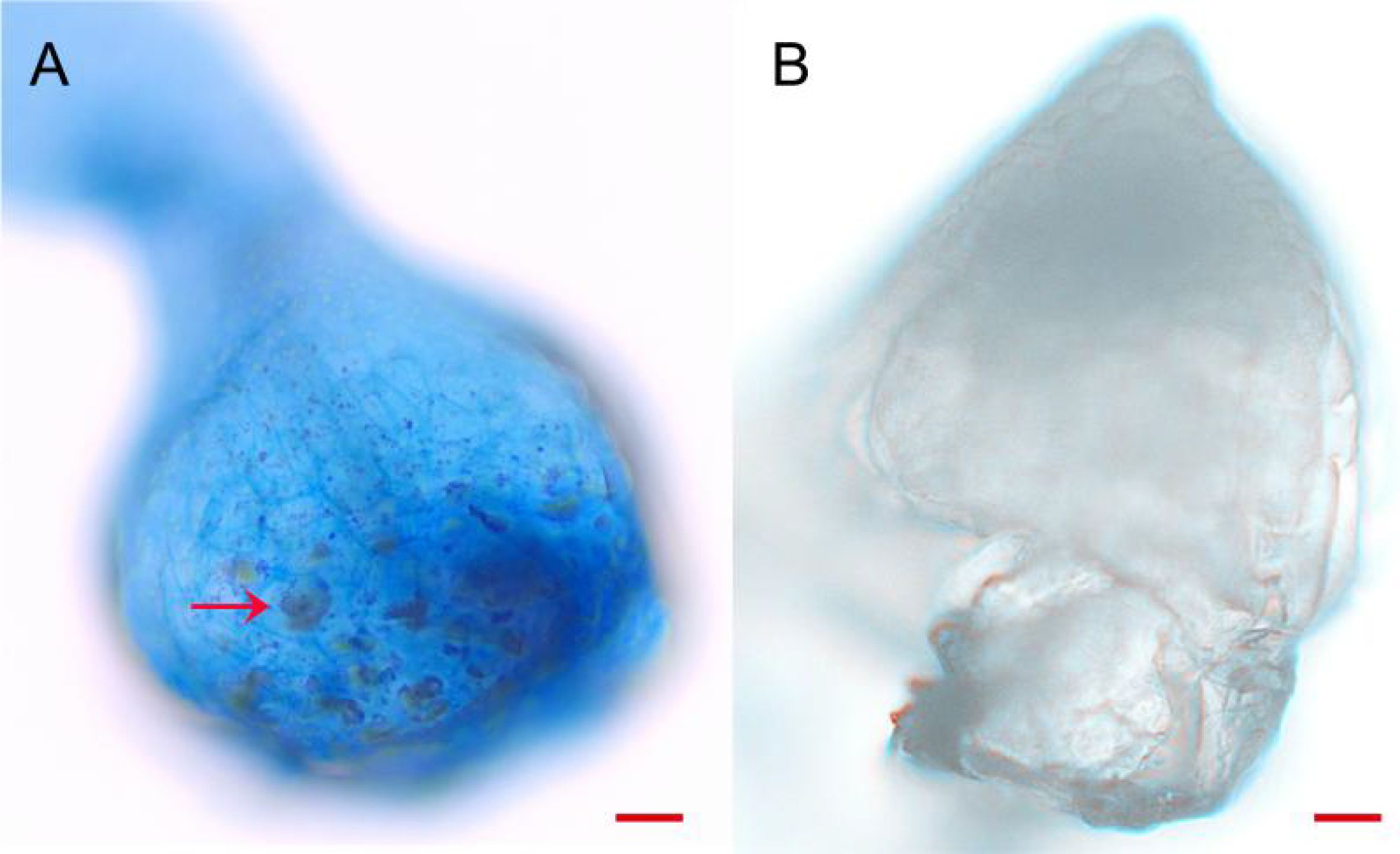
Figure 9. Trypan blue staining of endophytic colonization. (A) Endophytic colonization in protocorm of B striata (arrow) (bar = 50 μm); (B) protocorm of B. striata without BJ1 symbiosis (bar = 50 μm).
BJ1 can utilize L-tryptophan as a substrate to promote IAA synthesis; thus, we investigated whether the combination of L-tryptophan and BJ1 could synergistically enhance the growth of B. striata seeds. As presented in Figure 10, the length, width, and weight of the B. striata protocorm in the control group (without BJ1 and L-tryptophan treatment) gradually increased in the second and third weeks, but no significant growth was observed in the fourth week. Both L-tryptophan and BJ1 effectively promoted the growth of the B. striata protocorms after co-culturing with either L-tryptophan or BJ1 for 4 weeks, with BJ1 demonstrating superior efficacy compared to L-tryptophan. When seeds were co-cultured with both L-tryptophan and BJ1 for 4 weeks, their effects on both length and weight of the B. striata protocorms were significantly greater than those observed when seeds were co-cultured solely with either L-tryptophan or BJ1 individually. In particular, the weight of fresh protocorms co-cultured with L-tryptophan and BJ1 was 2.6 times that of the control, even heavier than the total weight of protocorms co-cultured with L-tryptophan or BJ1 alone, with the growth rate for weight reaching 155.32%. Collectively, these findings support the notion that L-tryptophan and BJ1 can work synergistically to promote both growth and development in B. striata seeds.
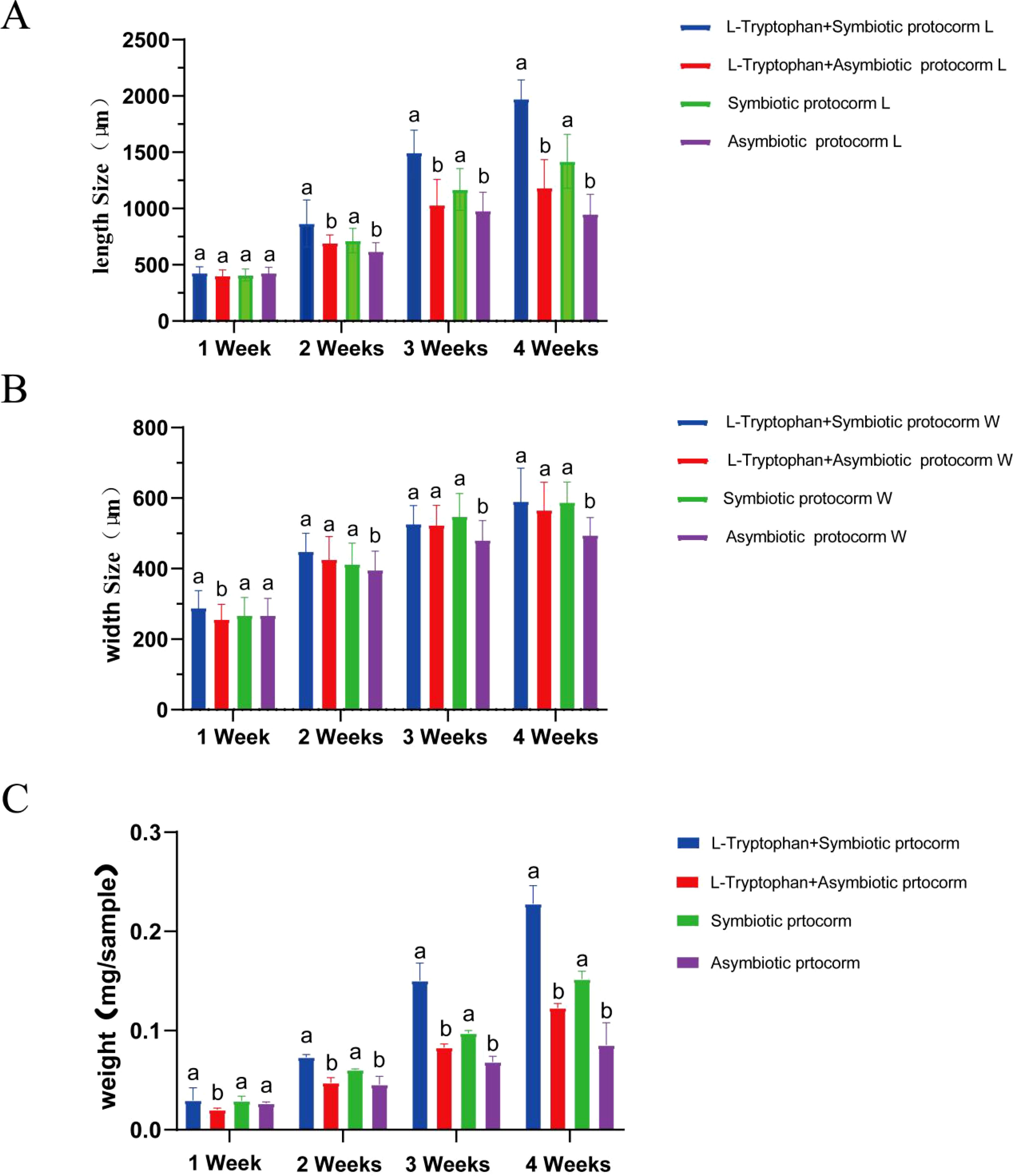
Figure 10. BJ1 and L-tryptophan synergistically promote the growth of B striata seeds. Changes in protocorm (A) length, (B) width, and (C) weight of B striata under different culture conditions. Standard error of the average value of 15 protocorms. One-way ANOVA was used for comparison between groups followed by Tukey’s test for multiple comparisons. Different letters indicate significant differences between groups in the same week, p < 0.05.
It is widely recognized that mycorrhizal fungi are essential for the germination and growth of orchid seed in their natural environments (McCormick et al., 2018). Hundreds of fungal species have been isolated and identified from various orchid species, including those belonging to Basidiomycota and Deuteromycota. The most prevalent orchid mycorrhizal fungus is Rhizoctonia sensu lato, which encompasses three asexual genera: Ceratorhiza, Epulorhiza, and Moniliopsis. Additionally, these strains represent the predominant groups that significantly facilitate orchid seed germination and growth (Watkinson and Winkel, 2024). Further investigation into the biological characteristics of these mycorrhizal fungi and their symbiotic mechanisms with orchid plants can provide a robust theoretical foundation for optimizing seedling cultivation techniques and achieving sustainable green planting practices. Additionally, such research will contribute significantly to the conservation of orchid germplasm resources. In this study, we investigated the biological properties of Tulasnella sp. mycorrhizal fungus BJ1, which was isolated from the fibrous roots of B. striata, along with its role in promoting germination and growth of B. striata seeds.
In 1888, Schroter made the first discovery of the genus Tulasnella, which he named Tulasnella lilacina J. Schrot (Freitas et al., 2020). These fungi are found all over the world and mostly feed on decaying wood and liverwort or orchid mycobionts (Brundrett, 2002; Warcup, 1973). Through comprehensive research, it has been determined that Tulasnella sp. exhibits a lack of distinctive macromorphological characteristics, as well as a notable degree of morphological variation and overlapping dimensions among its microscopic components (Warcup, 1981). Therefore, distinguishing between different groups of Tulasnella sp. from a morphological perspective is challenging. In this study, the morphological characteristics of BJ1 strain were investigated. The results showed that there was no significant difference in morphological characteristics between BJ1 strain and reported Tulasnella sp fungi. However, when compared under the same medium conditions, the hyphae of BJ1 grew faster and the aerial hyphae were more developed (Figure 3). This indicated that BJ1 strains were different from other Tulasnella sp fungi. Since the reference strains mentioned in the literature were not available for direct comparison, further studies are required to confirm the observed differences in mycelial growth rate and colony morphology. The taxonomy of Tulasnella species presents considerable challenges owing to the ambiguous morphological and molecular characteristics they display. Cruz et al. (2014) propose an integrated approach that synthesizes molecular and morphological data for the identification of Tulasnella sp., thereby establishing a robust theoretical foundation for precise classification. Linde et al. (2017) used sequence data from 8 sequence loci (ITS, mtLSU, C4102, C14436, C3304, C12424, C4722, C10499) and coalescent-based species delimitation methods have revealed several species-level lineages of Tulasnella associated with the orchid in Australia. Arifin et al. (2020) identified new species of Tulasnella associated with Australian terrestrial orchids using 4 sequence loci (ITS, C14436, C4102, mtLSU). Due to the superior efficacy of the four sequence sites—the ribosomal internal transcribed spacer (ITS1-ITS4), glutamate synthase (C4102), ATP synthase (C14436), and ATP helicase (C3304)—in molecular identification of Tulasnella sp., this study selected these sequence sites for a more precise molecular identification of the BJ1 strain. The phylogenetic analysis revealed that the BJ1 strain maintained distinct independent branches (Figure 4; Supplementary Figures S2–S4). The low similarity to other Tulasnella species significantly enhances the probability that the BJ1 strain represents a novel strain.
According to the literature, mycelia invade the embryo cells from the base of the embryo before forming mycelia clusters within exodermal cells. These mycelia continue their invasion into plant cells for colonization; finally, dead mycelia will be digested and absorbed by the plant as nutrients to promote its growth. This process forms a symbiotic relationship with orchids. Trypan blue staining confirmed that the strain BJ1 primarily colonized at the base of the protocorm (Figure 9A), consistent with findings reported by Jiang et al. (2019). Mycorrhizal fungi often secrete catabolic enzymes such as protease and pectinase to degrade cell wall polymers (e.g., cellulose, xylan, and pectin), thereby facilitating invasion and parasitism (Kubicek et al., 2014). In addition, mycorrhizal fungi can promote the synthesis of plant secondary metabolites by secreting cell-degrading enzymes. They enhance plant stress resistance, facilitate nutrient absorption in plants, and contribute to ecological diversity and community stability (Fawad et al., 2024). In this study, we evaluated the enzyme activity of BJ1 and found that it exhibited comparable secretion capabilities of protease and pectinase to other Tulasnella sp. strains. However, while some of these fungi have strong secretion abilities, they may not necessarily promote seed germination (Tang, 2021). Some studies have reported that the fungus Tulasnella sp. may not have the ability to produce laccase (Tang, 2021), while others have shown that the fungus Tulasnella sp. can produce laccase but exhibits relatively low enzymatic activity (Zhao et al., 2021). However, strain BJ1 exhibited no detectable laccase activity (Table 3). Therefore, further investigation into the laccase production potential of Tulasnella sp. (BJ1) is warranted. Obviously, the symbiotic germination process is influenced by various factors beyond just the capacity to secrete cell-degrading enzymes. It is possible that multiple hydrolases function synergistically rather than contributing individually to this process or that the secretory capacity under pure culture conditions differs from that during a symbiotic relationship with seeds (Chen et al., 2022). Therefore, further investigation into the genetic basis of cell wall-degrading enzymes is warranted.
Plant growth requires the absorption of phosphorus and iron elements from the soil. Microorganisms that produce siderophore can fulfill plants’iron requirements, hasten the dissolution of iron ions in minerals, and facilitate the release of related elements from insoluble iron oxides (Ge et al., 2023). Phosphorus-solubilizing microorganisms enhance soil phosphorus availability, regulate soil conditions, and boost plant uptake and utilization of soil phosphorus. They also convert or activate otherwise inaccessible forms of phosphorus into available ones (Wang et al., 2022; Han et al., 2021). According to the analysis of phosphorus-dissolving and siderophore production carrier activity, it was found that BJ1 lacks both iron-producing carrier and organic phosphorus-dissolving capacity, but possesses the ability to dissolve inorganic phosphorus. This suggests that BJ1 strain is capable of converting inorganic phosphorus into soluble forms to support B. striata seeds absorption and utilization of phosphorus, thereby promoting their germination and growth (Long et al., 2016).
Mycorrhizal fungi are known to release symbiotic signaling factors, including plant hormones and related compounds, which activate genes associated with plant symbiotic pathways. This process ultimately facilitates the colonization of cortical cells in orchid tissue by mycorrhizal fungi, thereby promoting seed germination and growth (Favre-Godal et al., 2020). IAA is a crucial phytohormone responsible for various aspects of plant growth and development, including cell growth, root initiation, tropism, fruit ripening, and senescence. Numerous studies have shown that mycorrhizal fungi possess the capability to produce IAA and other plant growth hormones that enhance seed germination and overall plant development (Wang et al., 2021). In addition to IAA production, mycorrhizal fungi can produce gibberellins (GAs) and cytokinin. For instance, Fusarium sp., has been shown to generate GAs (Wu et al., 2002). To assess whether BJ1 has the ability to produce IAA, a Salkowski test was performed. The results indicated a clear positive reaction in the fermentation supernatant containing L-tryptophan; however, there was almost no positive reaction observed in the fermentation supernatant without L-tryptophan supplementation. This suggests that the BJ1 strain possesses genes related to IAA synthesis and can utilize L-tryptophan as a substrate for its production. HPLC analysis further confirmed BJ1’s capability to produce IAA. However, when L-tryptophan was not provided as a substrate, the IAA production by BJ1 was significantly reduced both inside and outside the mycelia compared to the culture condition where L-tryptophan was present (Figure 6). However, the ability of BJ1 strain to secrete IAA (0.0217 mg/mL) was significantly higher than that of other endophytic bacteria (0.0069 mg/mL) (Wu et al., 2022). It is known that L-tryptophan acts as a precursor for IAA biosynthesis through two distinct pathways: one dependent on L-tryptophan and another independent pathway (Wu et al., 2002; Idris et al., 2007). Evidently, BJ1 can promote IAA synthesis when utilizing L-tryptophan as a substrate, in line with existing literature (Pham et al., 2019). However, it remains uncertain whether BJ1 can independently produce IAA without relying on the L-tryptophan pathway due to potential residual presence of L-tryptophan in PDA medium. Furthermore, based on HPLC analysis and reference to standard plant growth hormone compounds, no other auxins were detected. This suggests at least partially, BJ1 promotes the germination and growth of B. striata seeds by secreting IAA.
We also evaluated the growth-promoting effects of BJ1. Notably, BJ1 exhibited a remarkable enhancement in growth, especially during the fourth week (Figures 7, 8), which aligns with findings related to mycorrhizal fungi within the same genus (Gao et al., 2019; Li et al., 2017). Given that L-tryptophan serves as a precursor for IAA synthesis in both plants and fungi, we supplemented the medium with L-tryptophan and monitored the growth of B. striata seeds. As expected, the addition of L-tryptophan resulted in enhanced seed growth compared to the control group (Figure 10); although this effect was less pronounced than that observed with BJ1. This could be attributed to BJ1’s capacity not only to synthesize IAA but also provide nutrients conducive to seed growth; it may also stem from seeds being less efficient than BJ1 in converting L-tryptophan into IAA. Moreover, co-culturing BJ1 with L-tryptophan led to as significant increase in both the growth and development of B. striata seeds. Particularly noteworthy was the remarkable synergistic effect on seed length and weight, indicating potential benefits for enhancing B. striata seedling cultivation. These mechanisms, especially how mycorrhizal fungi synthesize IAA, are important to understand the communication between these fungi and their host (Mehmood et al., 2019, 2020). However, the current study did not explore the regulation of genes associated with IAA biosynthesis in BJ1 and B. striata seeds, which offers new directions for future research. Furthermore, no significant difference in seedling growth was observed between the non-symbiotic group during the third and fourth weeks. In contrast, the symbiotic group with BJ1 continued to demonstrate pronounced growth effects (Figure 8). This phenomenon may be attributed to the promotion of plant hormone synthesis, such as IAA, by the BJ1 symbiosis or to the nutritional benefits provided by BJ1. Additional well-designed experiments are necessary to elucidate the underlying mechanisms. In addition, the germination of Orchidaceae seeds in association with Tulasnella sp. fungi was also influenced by environmental factors, including pH and temperature. Noushka and Lawrie (2018) identified two distinct Tulasnella fungal lineages that are specific to different habitats. One of these lineages was found to be more favorable for seed germination at lower temperatures (12°C-16°C) during the fall and winter seasons. They also found that adult orchid growth was inhibited when co-planted with certain common symbiotic orchid species. In this study, controlled laboratory conditions—specifically temperature, humidity, and pH—were employed to investigate the effects of the BJ1 strain on seed germination. The temperature suitable (25°C-27°C) for germination and growth of B. striata seeds was mainly considered. Unfortunately, the impact of the BJ1 strain on seed germination was not evaluated under natural conditions. Future field tests will be conducted to validate these findings. Numerous studies have demonstrated that mycorrhizal fungi exert both direct and indirect influences on the growth, development, and accumulation of secondary metabolites in plants through a variety of biological activities. Direct mechanisms involve the production of auxins (such as IAA, cytokinins, gibberellins) as well as carriers for phosphorus solubilization and iron production by mycorrhizal fungi. Indirect mechanisms encompass inhibiting factors that interfere with plant growth and exhibiting antibacterial activity. These biological activities enable mycorrhizal fungi to establish beneficial relationships with plants (Long et al., 2016; Li et al., 2017; Yan et al., 2019; Wu et al., 2022). Investigations into the diversity of mycorrhizal fungi within Orchidaceae have revealed that Tulasnella species are dominant in promoting plant growth (Gao et al., 2019; Li et al., 2017). Some reports suggest that this genus may influence plant secondary metabolism (Ghirardo et al., 2020) and also plays a role in nitrogen transfer and transport during plant development (Fochi et al., 2017). In addition, it has been reported that Tulasnella sp. fungi play a crucial role in enhancing nutrient uptake and stress tolerance in plants. This is particularly evident through their symbiotic relationships, which improve root function, facilitate nutrient absorption, and bolster resilience to various environmental challenges (Jian et al., 2022). Whether BJ1 exhibits these effects warrants further exploration. Additionally, the enhanced ability of strain BJ1 to produce IAA through L-tryptophan supplementation may contribute significantly to future advancements in biological fertilizer development.
In general, the isolated mycorrhizal fungus BJ1 from B. striata, is classified as a new species within the genus Tulasnella sp., significantly increased the germination rate of B. striata seeds from 93.04% to 96.07% by the fourth week of symbiosis. Furthermore, it enhanced the proportion of protocorms that progressed to stage 5 from 50.43% to 74.23%. Additionally, the length, width, and fresh weight of protocorms in the symbiotic group were 2.2 times, 1.8 times, and 3.7 times greater than those in the non-symbiotic group, respectively. However, there exists a limited systematic understanding of the symbiotic germination mechanisms employed by such mycorrhizal fungi. The current work demonstrates that BJ1 promotes the growth of B. striata seeds at least partially through the dissolution of inorganic phosphorus and the production of plant auxin IAA. In addition, when combined with L-tryptophan, BJ1 exhibits a synergistic effect on promoting both growth and development in B. striata seeds, which holds significant implications for the efficient cultivation of B. striata seedlings. Nevertheless, further elucidation is required to clarify the underlying mechanisms governing symbiotic germination between BJ1 and B. striata seeds. Additionally, it remains necessary to explore whether BJ1 can effectively promote seed germination and growth, enhance resistance against pathogens in B. striata and its potential impact on the quality of traditional Chinese medicine derived from B. striata in soil-based trials.
The research and collection of materials for the seed plant of B. striata mentioned in this article comply with relevant institutional guidelines and legislation in China. All methods are carried out in accordance with the relevant guidelines in the methods section. Professor Shuili Zhang from Zhejiang University of Traditional Chinese Medicine officially identified the plant materials used in the study. Our team has obtained permission to collect plant materials. The voucher number (20221021-ND) and seeds are stored in the laboratory of the Institute of Traditional Chinese Medicine Resources, Zhejiang University of Traditional Chinese Medicine. The storage temperature of seeds is 4°C.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
ZL: Writing – original draft. YY: Writing – original draft, Data curation. XW: Data curation, Formal analysis, Writing – original draft. SP: Data curation, Writing – original draft. BC: Data curation, Formal analysis, Writing – original draft. SL: Funding acquisition, Writing – original draft. HC: Methodology, Writing – original draft. DY: Methodology, Writing – original draft. FJ: Methodology, Writing – review & editing. CZ: Writing – review & editing. ML: Writing – review & editing.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. The present research was carried out with the financial support of the Science and Technology Plan of Traditional Chinese Medicine of Zhejiang Province (grant number 2021ZX008).
We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2025.1542585/full#supplementary-material
Arifin, A. R., May, T. W., Linde, C. C. (2020). New species of Tulasnella associated with Australian terrestrial orchids in the Cryptostylidinae and Drakaeinae. Mycologia 1, 113. doi: 10.1080/00275514.2020.1813473
Batty, A. L., Brundrett, M. C., Dixon, K. W. (2006). New methods to import symbiotic propagation of temperature terrestrial orchid seedlings from axenic culture to soil. Aust. J. Bot. 54, 367–374. doi: 10.1071/BT04023
Brundrett, M. C. (2002). Coevolution of roots and mycorrhizas of land plants. New Phytol. 154, 275–304. doi: 10.1046/j.1469-8137.2002.00397.x
Chen, J., Tang, Y., Kohler, A., Lebreton, A., Xing, Y., Zhou, D., et al. (2022). Comparative transcriptomics analysis of the symbiotic germination of D. officinale (Orchidaceae) with emphasis on plant cell wall modification and cell wall-degrading enzymes. Front. Plant Sci. 13, 880600. doi: 10.3389/fpls.2022.880600
Chen, Q. Y., Hu, Z. Q., Liu, L. S. (2009). Isolation and identification of six organophosphorus- solubilizing bacteria. J. Anhui Agric. Univ. 36, 417–421. doi: 10.13610/j.cnki.1672-352x.2009.03.006
Cheng, S., Jiang, J. W., Tan, L. T., Deng, J. X., Liang, P. Y., Su, H., et al. (2022). Plant growth-promoting ability of mycorrhizal fusarium strain KB-3 enhanced by its IAA producing endohyphal bacterium, klebsiella aerogenes. Front. Microbiol. 13, 855399. doi: 10.3389/fmicb.2022.855399
Chi, B. B., Ni, Y., Chen, H. Y., Liu, H. Y. (2022). Identification of a moderately halophilic bacterium (Aspergillus aculeatus GLUT-01) producing pectinase and optimization of enzyme-producing conditions. Jiangsu Agric. Sci. 50, 239–246. doi: 10.15889/j.issn.1002-1302.2022.02.039
Cruz, D., Suárez, J. P., Kottke, I., Piepenbring, M. (2014). Cryptic species revealed by molecular phylogenetic analysis of sequences obtained from basidiomata of Tulasnella. Mycologia 106, 708–722. doi: 10.3852/12-386
Daniel, H., Huo, W. W., Zhang, F. L., Liu, L., Cao, F. X., Yu, X. Y., et al. (2022). Isolation and identification of endophytic fungi and screening of biocontrol bacteria from the roots of Orchid chinensis. Biol. Resour. 44, 189–197. doi: 10.14188/j.ajsh.2022.02.008
DeFátima, D. D. G., Cota, L. V., Figueiredo, J. E. F., Aguiar, F. M., Da, S. D. D., De, P. L. U. G., et al. (2021). Antifungal activity of bacterial strains from maize silks against Fusarium verticillioides. Arch. Microbiol. 28, 204(1):89. doi: 10.1007/s00203-021-02726-4
Fan, C. M., Li, Z. L., He, Y. Q. (2003). Advances in tissue culture and molecular biology of orchid. Acta Horticult. Sin. 30, 487–491. doi: 10.16420/j.issn.0513-353x.2003.04.035
Favre-Godal, Q., Gourguillon, L., Lordel-Madeleine, S. (2020). Orchids and their mycorrhizal fungi: an insufficiently explored relationship. Mycorrhiza 30, 5–22. doi: 10.1007/s00572-020-00934-2
Fawad, Z., Muhammad, H., Wajid, A. K. (2024). The pivotal role of arbuscular mycorrhizal fungi in enhancing plant biomass and nutrient availability under drought stress conditions: A global meta-analysis. Sci. Total Environ. 955, 176960–176960. doi: 10.1016/j.scitotenv.2024.176960
Fochi, V., Chitarra, W., Kohler, A., Voyron, S., Singan, V. R., Lindquist, E. A., et al. (2017). Fungal and plant gene expression in the Tulasnella calospora-Serapias vomeracea symbiosis providers clubs about nitrogen pathways in orchid mycorrhizas. N. Phytol. 213, 365–379. doi: 10.1111/nph.14279
Freitas, E. F. S., Da, S. M., Cruz, E. D. S., Mangaravite, E., Bocayuva, M. F., Veloso, T. G. R., et al. (2020). Diversity of mycorrhizal Tulasnella associated with epiphytic and rupicolous orchids from the Brazilian Atlantic Forest, including four new species. Sci. Rep. 10, 7069. doi: 10.1038/s41598-020-63885-w
Gao, Y., Guo, S. X., Xing, X. K. (2019). Research progress on fungal diversity and symbiotic germination mechanism of Orchidaceae seeds. Acta Fungi Sin. 38, 18. doi: 10.13346/j.mycosystema.190163
Ge, M. M., Bo, Y. L., Liu, C., Liu, F. C., Xie, X. X., Dong, Y. L., et al. (2023). Screening of soil iron producing carrier bacteria and their activation and utilization of iron oxides. Microbiol. Bull. 50 (03), 1062–1072. doi: 10.13344/j.microbiol.china.220643
Ghirardo, A., Fochi, V., Lange, B., Witting, M., Schnitzler, J. P., Perotto, S., et al. (2020). Metabolomic adjustments in the orchid mycorrhizal fungus Tulasnella calospora during symbiosis with Serapias vomeracea. New Phytol. 228, 1939–1952. doi: 10.1111/nph.v228.6
Guo, S. X., Xu, J. T. (1992). Relationship between seed germination and seedling growth of Bletilla striata and four fungi such as Osmunda japonica. J. Chin. Acad. Med. Sci. 14, 6.
Han, Y., Liu, S., Chen, F., Deng, X., Miao, Z., Wu, Z., et al. (2021). Characteristics of plant growth-promoting rhizobacteria SCPG-7 and its effect on the growth of Capsicum annuum L. Environ. Sci. pollut. Res. Int. 28, 11323–11332. doi: 10.1007/s11356-020-11388-6
Idris, E. E., Iglesias, D. J., Talon, M., Borriss, R. (2007). Tryptophan-dependent production of indole-3-acetic acid (IAA) affects level of plant growth promotion by Bacillus amyloliquefaciens. Mol. Plant Microbe Interact;FZB 42, 619–626. doi: 10.1094/MPMI-20-6-0619
Jahn, L., Hofmann, U., Ludwig-Müller, J. (2021). Indole-3-Acetic acid is synthesized by the endophyte Cyanodermella asteris via a Tryptophan-dependent and -independent way and mediates the interaction with a non-host plant. Int. J. Mol. Sci. 22 (5), 2651. doi: 10.3390/ijms22052651
Jian, Z., Zhenjian, L., Siyu, W. (2022). Correlations between the phylogenetic relationship of 14 tulasnella strains and their promotion effect on dendrobium crepidatum protocorm. Horticulturae 8, 1213–1213. doi: 10.3390/horticulturae8121213
Jiang, J., Zhang, K., Cheng, S., Nie, Q., Zhou, S. X., Chen, Q., et al. (2019). Fusarium Oxysporum KB-3 from Bletilla striata: an orchid mycorrhizal fungus. Mycorrhiza 29, 531–540. doi: 10.1007/s00572-019-00904-3
Kong, W. H., Xu, J. B., Cui, Q. (2021). Research progress on chemical constituents, pharmacological effects and extraction technology of Bletilla striata polysaccharide. Chin. Med. Inf. 38, 69–78. doi: 10.19656/j.cnki.1002-2406.20210914
Kubicek, C. P., Starr, T. L., Glass, N. L. (2014). Plant cell wall-degrading enzymes and their secretion in plant-pathogenic fungi. Annu. Rev. Phytopathol. 52, 427–451. doi: 10.1146/annurev-phyto-102313-045831
Li, X. Z., Song, M. L., Yao, X. (2017). The effect of seed-borne fungi and epichlo endophyte on seed germination and biomass of elymus sibiricus. Front. Microbiol. 8. doi: 10.3389/fmicb.2017.02488
Li, Y., Hua, C. Y., Zong, K. (2021). Identification and phylogenetic analysis of a fungus isolated from rice seeds. Acta Agronom. Sin. 11, 36–40.
Linde, C. C., May, T. W., Phillips, R. D., Ruibal, M., Smith, L. M., Peakall, R. (2017). New species of Tulasnella associated with terrestrial orchids in Australia. IMA Fungus 8, 27–47. doi: 10.5598/imafungus.2017.08.01.03
Long, X., Yan, X., Hong, J. L., Liang, Z. S., Chen, H. M. (2016). Research progress of endophytes in plant seeds. Zhejiang Agric. Sci. 57, 1319–1324. doi: 10.16178/j.issn.0528-9017.20160851
McCormick, M. K., Whigham, D. F., Canchani-Viruet, A. (2018). Mycorrhizal fungi affect orchid distribution and population dynamics. New Phytol. 219, 1207–1215. doi: 10.1111/nph.2018.219.issue-4
Mehmood, A., Hussain, A., Irshad, M., Hamayun, M., Iqbal, A., Khan, N. (2019). In Vitro Production of IAA by Endophytic Fungus Aspergillus awamori and Its Growth Promoting Activities in Zea mays. Symbiosis 77, 225–235. doi: 10.1007/s13199-018-0583-y
Mehmood, A., Hussain, A., Irshad, M., Hamayun, M., Iqbal, A., Tawab, A., et al. (2020). Yucasin and cinnamic acid inhibit IAA and flavonoids biosynthesis minimizing interaction between maize and endophyte aspergillus nomius. Symbiosis 81, 149–160. doi: 10.1007/s13199-020-00690-z
Miura, C., Yamaguchi, K., Miyahara, R., Yamamoto, T., Fuji, M., Yagame, T., et al. (2018). The mycoheterotrophic symbiosis between orchids and mycorrhizal fungi possesses major components shared with mutualistic plant-mycorrhizal symbioses. Mol. Plant Microbe Interact. 31, 1032–1047. doi: 10.1094/MPMI-01-18-0029-R
Noushka, R. A. C., Lawrie, C. C. (2018). Linde. Matching symbiotic associations of an endangered orchid to habitat to improve conservation outcomes. Ann. Bot. 122, 947–959. doi: 10.1093/aob/mcy094
Pan, Y. C., Li, L., Xiao, S. J., Zhang, S. B., Shangguan, Y. N., Xu, D. L. (2018). Research progress on breeding technology of Bletilla striata. Chin. Patent Med. 40, 1142–1149. doi: 10.3969/j.issn.1001-1528.2018.05.029
Patel, D., Patel, A., Vora, D., Menon, S., Vadakan, S., Acharya, D., et al. (2018). A resourceful methodology to profile indolic auxins produced by rhizo-fungi using spectrophotometry and HPTLC. 3 Biotech. 8, 413. doi: 10.1007/s13205-018-1428-3
Pham, M. T., Huang, C. M., Kirschner, R. (2019). The plant growth-promoting potential of the mesophilic wood-rot mushroom Pleurotus pulmonarius. J. Appl. Microbiol. 127, 1157–1171. doi: 10.1111/jam.v127.4
Ren, F. M., Yan, L., Ying, L. I., Yang, T. J., Feng, H., Song, J. Y. (2016). Resource distribution and breeding of medicinal plants in genus Bletilla Reichb. f. Chin. Tradit. Herb Drugs 47, 4478–4487.
Ruibal, M. P., Peakall, R., Smith, L. M., Linde, C. C. (2013). Phylogenetic and microsatellite markers for Tulasnella (Tulasnellaceae) mycorrhizal fungi associated with Australian orchids. Appl. Plant Sci. 1, 1200394. doi: 10.3732/apps.1200394
Shah, S., Chand, K., Rekadwad, B. (2021). A Prospectus of plant growth promoting endophytic bacterium from orchid (Vanda cristata). BMC Biotechnol. 21, 1–9. doi: 10.1186/s12896-021-00676-9
Stewart, S. L., Zettler, L. W. (2002). Symbiotic germination of three semi-aquatic rein orchids (Habenaria repens, H. quinquiseta, H. macroceratitis) from Florida. Aquat. Bot. 72, 25–35. doi: 10.1016/S0304-3770(01)00214-5
Tang, Y. J. (2021). Study on the specificity of symbiotic germination of medicinal Dendrobium seeds and mycorrhizal fungi and its mechanism of action. (Beijing: Peking Union Medical College (Tsinghua University Medical Department) Chinese Academy of Medical Sciences), 32–39.
Tian, F., Wang, J. C., Bai, X. X., Yang, Y. B., Huang, L., Liao, X. F. (2023). Symbiotic seed germination and seedling growth of mycorrhizal fungi in Paphiopedilum hirsutissimun (Lindl.Ex Hook.) Stein from China. Plant Signal Behav. 18, 2293405. doi: 10.1080/15592324.2023.2293405
Wang, J., Fan, Y. H., Shang, S., Li, X. P., Zhang, Y. M., Wu, T., et al. (2022). Screening and identification of a rhizosphere phosphate solubilizing bacterium and its role in dissolving phosphorus and promoting growth. China Soil Fertil. 6, 195–203. doi: 10.11838/sfsc.1673-6257.21131
Wang, S., Ji, B., Su, X. (2020). Isolation of endophytic bacteria from Rehmannia glutinosa Libosch and their potential to promote plant growth. J. Gen. Appl. Microbiol. 66 (5), 279–288. doi: 10.2323/jgam.2019.12.001
Wang, T., Song, Z., Wang, X. (2018). Functional insights into the roles of hormones in the dendrobium officinale-tulasnella sp. germinated seed symbiotic association. Int. J. Mol. Entities 19 (11), 3484–3484. doi: 10.3390/ijms19113484
Wang, X., Gao, J., Li, N. (2023). Serendipita officinale sp. nov. (Serendipitaceae): a new species of orchid mycorrhizal fungus. Phytotaxa 630, 229–240. doi: 10.11646/phytotaxa.630.3.4
Wang, Y., Zhang, W., Liu, W., Ahammed, G. J., Wen, W., Guo, S., et al. (2021). Auxin is involved in arbuscular mycorrhizal fungi-promoted tomato growth and NADP-malic enzymes expression in continuous cropping substrates. BMC Plant Biol. 21, 48. doi: 10.1186/s12870-020-02817-2
Warcup, J. H. (1973). Symbiotic germination of some Australian terrestrial orchids. New Phytol. 72, 387–392. doi: 10.1111/j.1469-8137.1973.tb02046.x
Warcup, J. H. (1981). The mycorrhizal relationships of Australian orchids. New Phytol. 87, 371–381. doi: 10.1111/j.1469-8137.1981.tb03208.x
Watkinson, J. I., Winkel, B. S. J. (2024). Diversity of unique, nonmycorrhizal endophytic fungi in cultivated Phalaenopsis orchids: A pilot study. Plant Environ. Interact. 5, e10146. doi: 10.1002/pei3.10146
Wei, J. C. (1979). Handbook of Fungal Identification (Beijing: Shanghai Science and Technology Press).
Wu, J., Qian, J., Zheng, S. (2002). A preliminary study on ingredient of secretion from fungi of orchid mycorrhiza. Ying Yong Sheng Tai Xue Bao 13, 845–848.
Wu, J. H. (2019). Gentiana straminea endophytic fungi diversity research. (Sichuan: Sichuan Normal University). doi: 10.27347/d.cnki.gssdu.2019.001323
Wu, Y., Xiao, S., Qi, J., Gong, Y., Li, K. (2022). Pseudomonas fluorescens BsEB-1: an endophytic bacterium isolated from the root of Bletilla striata that can promote its growth. Plant Signal Behav. 17, 2100626. doi: 10.1080/15592324.2022.2100626
Xu, H., Xu, L. (2007). Research progress of tissue culture of Cymbidium chinensis. J. Consump. 12, 249.
Xu, J. L., Zhang, P., Li, M. F. (2022). Isolation, optimization of culture conditions and preliminary application of iron-producing carrier strains. Bull. Microbiol. 49, 1004–1016. doi: 10.13344/j.microbiol.china.210729
Xu, L. L., Zhang, Y., Zhao, M., Li, X., Nan, M., Meng, F. (2019). Effects of mycorrhizal fungi on seed germination and seedling rooting of Bletilla striata. Acta Fungi Sin. 38, 1440–1449. doi: 10.13346/j.mycosystema.190175
Xue, Y. Y., Ye, W., Yang, S. (2019). Isolation and identification of a phosphorus-solubilizing strain and its phosphorus-solubilizing and growth-promoting effect. Agric. Res. Arid Areas 37, 253–262. doi: 10.7606/j.issn.1000-7601.2019.04.34
Yamamoto, T., Miura, C., Fuji, M. (2017). Quantitative evaluation of protocol growth and functional colonization in Bletilla striata (Orchidaceae) revals less-productive symbiosis with a non-native symbiotic function. BMC Plant Biol. 17 (1), 50. doi: 10.1186/s12870-017-1002-x
Yan, L., Zhu, J., Zhao, X., Shi, J., Jiang, C., Shao, D. (2019). Beneficial effects of endophytic fungi colonization on plants. Appl. Microbiol. Biotechnol. 103, 3327–3340. doi: 10.1007/s00253-019-09713-2
Yang, J. W., Chen, X. M., Meng, Z. X. (2020). Effects of S7 (Tulasnella sp.) and its extract on seed germination of Dendrobium candidum. Sci. China: Life Sci. 50, 559–570. doi: 10.1360/SSV-2019-0305
Yang, J., Li, N. Q., Gao, J. Y. (2024). Roles of mycorrhizal fungi on seed germination of two Chinese medicinal orchids: need or do not need a fungus? Front. Plant Sci. 15, 1415401. doi: 10.3389/fpls.2024.1415401
Zhang, D. Y., Liu, Y., Wu, Y. (2016). Screening, identification and effect of IAA-producing bacteria in peanut rhizosphere. Chin. J. Oil Crops 38, 104–110. doi: 10.7505/j.issn.1007-9084.2016.01.016
Zhang, J. H., Wang, C. L., Guo, S. X. (1999). Plant hormones produced by five endophytic fungi of Orchidaceae medicinal plants. J. Chin. Acad. Med. Sci. 21, 460–465. doi: CNKI:SUN:ZYKX.0.1999-06-010
Zhang, J. X., Fu, Z. H., Li, H. L. (2005). Relationship between embryo development and seed germination of Bletilla striata. J. Subtrop. Plant Sci. 34, 4. doi: 10.3969/j.issn.1009-7791.2005.04.009
Zhang, M., Shao, Q., Xu, E., Wang, Z., Wang, Z., Yin, L. (2019). Bletilla striata: a review of seedling propagation and cultivation modes. Physiol. Mol. Biol. Plants 25, 601–609. doi: 10.1007/s12298-019-00644-w
Zhao, X. M. (1963). Gleanings of Compendium of Materia Medica. (Beijing: People's Medical Publishing House).
Zhao, D. K., Selosse, M. A., Wu, L., Luo, Y., Shao, S. C., Ruan, Y. L. (2021). Orchid reintroduction based on seed germination-promoting mycorrhizal fungi derived from protocorms or seedlings. Front. Plant Sci. 12, 701152. doi: 10.3389/fpls.2021.701152
Zhao, Z., Shao, S., Liu, N., Liu, Q., Jacquemyn, H., Xing, X. (2021). Extracellular enzyme activities and carbon/nitrogen utilization in mycorrhizal fungi isolated from epiphytic and terrestrial orchids. Front. Microbiol. 2, 787820. doi: 10.3389/fmicb.2021.787820
Zhou, X. Y., Mimapuchi, Ma, X. Y., Ma, Z. G., Ding, G. T. (2020). Isolation and identification of a hemoglobin-decomposing strain and determination of its protease activity. J. Microbiol. 40, 66–71. doi: 10.3969/j.issn.1005-7021.2020.02.008
Zhu, X. H. (2018). Isolation and identification of laccase-producing bacteria and determination of laccase activity. J. Jiamusi Vocational Coll. 11, 388–389. doi: CNKI:SUN:JMSJ.0.2018-11-253
Zhu, X. M., Hu, H., Li., S. Y. (2012). Interaction between endophytic fungi and two Paphiopedilum species in co-culture. J. Plant Classific. Resour. 34, 171–178. doi: 10.3724/SP.J.1143.2012.11144
Keywords: symbiotic germination, mycorrhizal fungus, Bletilla striata, Tulasnella sp., indoleacetic acid
Citation: Li Z, Ye Y, Wang X, Peng S, Chen B, Li S, Chen H, Yang D, Jiang F, Zhang C and Li M (2025) Mycorrhizal fungus BJ1, a new species of Tulasnella sp.: its biological characteristics and promoting effect on seed germination of Bletilla striata. Front. Plant Sci. 16:1542585. doi: 10.3389/fpls.2025.1542585
Received: 10 December 2024; Accepted: 29 January 2025;
Published: 21 February 2025.
Edited by:
Muhammad Qadir, Hunan University, ChinaCopyright © 2025 Li, Ye, Wang, Peng, Chen, Li, Chen, Yang, Jiang, Zhang and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Meiya Li, bG1laXlhQDEyNi5jb20=; Chunchun Zhang, MjAwODEwMjZAemNtdS5lZHUuY24=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.