
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Plant Sci. , 21 March 2025
Sec. Plant Abiotic Stress
Volume 16 - 2025 | https://doi.org/10.3389/fpls.2025.1535395
Background: Stem tissue structures are the basis of stem function and are essential for maintaining the normal physiological metabolism of aquatic plants. Water and sediment conditions are important factors affecting the functional characteristics and physiological metabolism of Brassenia schreberi. Due to pollution and other water and sediment issues caused by human activities, the natural habitat and population size of B. schreberi have dramatically decreased. Understanding the responses of the functional characteristics of B. schreberi to water and sediment conditions is the key to its scientific conservation and management.
Objectives and methods: This study selected Beihai Wetland in Tengchong, China, which boasts the largest natural habitat of B. schreberi, as the research site. To detect the response strategies of B. schreberi to water and sediment conditions, the photosynthetic parameters and stem structural characteristics of this species at 17 locations, as well as the water and sediment nutrient parameters at these locations were measured. We examined the relationships between the trait characteristics of B. schreberi and the water and sediment parameters by using correlation analysis. The aim was to explore the effects of sediment nutrients and water quality on the photosynthetic and stem structural characteristics of B. schreberi.
Results and conclusions: B. schreberi with higher coverage exhibited higher stomatal conductance (Gs) and transpiration rate (Tr), but lower vascular bundle area and ventilation hole area (P<0.05), while the net photosynthetic rate (Pn) maintained content, indicating lower utilization efficiency of water and CO2. Water temperature (WT), sediment nitrogen content (ω(N)) and water dissolved oxygen (DO) were the main parameters affecting the characters of B. schreberi. The Pn, was significantly negatively correlated with ω(N), while it was positively correlated with DO and sediment phosphorus content (P<0.05). The findings indicate that B. schreberi requires good water quality to maintain a high photosynthetic rate and is prone to phosphorus limitation, but it has low requirements for sediment nitrogen content. The findings of this study provide a scientific basis for the habitat restoration and species-specific management of B. schreberi in degraded wetlands.
Brasenia schreberi, also known as watershield, is a perennial floating-leaved aquatic macrophyte in the Nymphaeaceae family (also listed in the Cabombaceae family) (Li et al., 2021, 2018). It has a wide yet and sporadic geographical distribution in temperate and tropical regions of Asia, Africa, Australia, North and South America and India (Drzymulska, 2018; Kim et al., 2012). Floating-leaved plants, such as B. schreberi, usually plays as a pioneer in the formation of plant communities in many freshwater ecosystems owing to its robust ability for vegetative propagation, and is also a dominant species in areas where the water level is too deep for emergent plants to grow well (Yang et al., 2020; Grasset et al., 2015; Bornette and Puijalon, 2011). It also plays an important role in water purification and other ecological functions. Additionally, B. schreberi is a traditional aquatic vegetable in Asia and is also a traditional medicinal plant in China, with high economic value for both edible and medicinal uses (Ran et al., 2020; Wang et al., 2017). However, the natural habitats suitable for the growth of B. schreberi are being seriously lost, the distribution range is narrowing, and the numbers of natural populations are decreasing sharply due to excessive harvesting, environmental pollution and climate change, thus many countries have listed it as an endangered and rare species for priority protection (Yang et al., 2020; Li et al., 2018; Xie et al., 2018). The natural B. schreberi is officially listed as a national key protected wild plant by the State Council of China in 1999, and is currently listed as a national level II protection of wild plants (Li et al., 2021). Natural populations of B. schreberi in China are mainly growing in unpolluted freshwater ponds, lakes, swamps, and even wild farmlands in the provinces of Yunnan, Hunan, Hubei, Jiangxi, and Taiwan etc. In the biodiversity-rich Yunnan region, natural populations of B. schreberi are only found in Tengchong Beihai wetland, where hosts the largest wild B. schreberi population area in China, covering 100 hectares, which is greater than the combined area of natural B. schreberi populations in other regions of China. B. schreberi in the Beihai wetland can form pure populations and serves as an important pioneer and dominant species, playing a significant role in the ecological functions of the wetland. Given the current conservation status of the species, the adaptive mechanisms of its natural populations are key scientific issues that need to be thoroughly investigated.
Plant functional traits can effectively regulate the functional response of plants to environmental changes since they are highly sensitive to these changes, and thus they are often used to explore the adaptation mechanisms of plants to the environment (Cheng et al., 2022; Liu et al., 2021). Nevertheless, previous studies have predominantly concentrated on the responses of leaf functional traits to environmental conditions and their role in plant adaptation to environmental changes (such as Islam et al., 2024; Chen et al., 2024; Wang et al., 2022; Slot et al., 2021; Wright et al., 2004), with relatively less attention devoted to the functional traits of other plant structures, such as stems. Stem is the middle part connecting plant leaves and root system. It plays a crucial role in water and material transport, mechanical support, defense, and lodging resistance throughout plant lifecycle (Łoboda et al., 2018; Bociag et al., 2009; Yiotis et al., 2009). The composition structures of stem are generally composed of epidermal structures, vascular bundle structures and stem tissue cells. Stems of wetland plants also possess aerenchyma, which is not found in most terrestrial plants. The stem of B. schreberi is composed of primary structures, including the epidermis and mucus, cortex, abscission layer, aerenchyma, and vascular bundles (Yang et al., 2020; Hu et al., 2018). Epidermal structure, covering the surface of all plant organs, serves as a natural protective barrier between plant and external environment (Han et al., 2021). It is composed of thick-walled cells that adhere firmly to each other and exhibit specific mechanical properties that confer the necessary strength for plant growth (Ristic and Jenks, 2002). The epidermis of B. schreberi serves as its barrier structure, which is highly sensitive to the growth environment and is a vital component that determines its normal survival (Drzymulska, 2018; Hu et al., 2018). Vascular system is the mechanical support system of higher plants that maintains plant morphology and supports upright growth. It is also the long-distance transport system for water, minerals, and photosynthetic products within plants, which dominates the redistribution and transfer of substances between different parts of plants (Zhang et al., 2022). Parenchyma cells have the function of maintaining plant tissue structure and limiting tissue growth rate (Serrano-Mislata and Sablowski, 2018; Lee, 2018; Brulé et al., 2019). Aerenchyma allows wetland plants to ventilate and store oxygen in hypoxic environments, facilitating the diffusion of oxygen from leaves to the roots and from the rhizosphere to the outside environment, therefor enhancing plant photosynthetic rate and photosynthetic rate (Yang et al., 2020; Seago et al., 2005). The larger the plant’s aerenchyma, the greater the amount of oxygen released from the root tips (Wang and Reid, 2020; Armstrong et al., 2006; Seago et al., 2005). These tissue structures are the basis of stem function and are essential for maintaining the normal physiological metabolism of plants. Stem biomass of B. schreberi constitutes the majority of its total biomass, and the physiological functions of stem structural characteristics are of significant importance for its adaptation to different water depths (Zhang et al., 2018). Investigating the environmental response strategies of B. schreberi from the perspective of stem functional traits is a crucial part of elucidating its adaptation mechanisms and will significantly contribute to the conservation of the species. While existing research on the stem functional traits of B. schreberi has largely focused on the structural composition and performance of its stems, few studies have quantitatively measured these traits and established quantitative relationships with the environment to explore its response strategies to environmental conditions.
Aquatic plants are the primary producers of wetlands and serve as the main transmitters of material and energy flow in lake ecosystems, and they also purify eutrophic water quality (Zhao et al., 2023; Dhote and Dixit, 2009). Additionally, macroaquatic plants play key roles in providing habitat, refugia, and food for biota in shallow lakes (O’Hare et al., 2018). The growth of aquatic plants is influenced by various environmental factors and is an important indicator of ecosystem health status (Dhote and Dixit, 2009). In the context of global environmental changes, more attention are paid to the responses and adaptations of aquatic plants and their ecosystems to climatic conditions at large spatial scales. However, aquatic plants exhibit strong cryptic characteristics that their sensitivity to climatic conditions is often lower than that of terrestrial plants, while they are more sensitive to the microenvironmental conditions. Water and sediment microenvironment conditions among many environmental factors, especially the sediment nutrient conditions, are important factors affecting the functional characteristics of aquatic plants (Pan et al., 2020; Ogdahl and Steinman, 2015). Water quality and sediment nitrogen and phosphorus contents are also the most important environmental factors affecting the growth, reproduction, and survival of B. schreberi. Related studies have shown that B. schreberi adapts to oligotrophic aquatic environment, and has higher requirements for water quality and sediment nutrients (Drzymulska, 2018; Xie et al., 2018). Habitat protection, especially water environment protection, is crucial for maintaining the population of B. schreberi (Chen et al., 2024; Drzymulska, 2018; Xie et al., 2018). A small amount of fertilizer added to the field water can lead to the decay of B. schreberi within a few days (Zhang et al., 2015). The permanganate index, total N content, electrical conductivity, and dissolved oxygen content of water, and organic carbon content and total nitrogen content of sediment collectively explained 82.2% of the changes in the mucilage accumulation of B. schreberi (Xie et al., 2018). Dissolved oxygen content, nitrogen and phosphorus content and water temperature are the main water factors affecting the leaf economical traits of B. schreberi; and the photosynthetic rate is significantly positively correlated with the dissolved oxygen content, ammonium nitrogen content and nitrate nitrogen content of water (Chen et al., 2024). The yield and mucilage thickness of B. schreberi are significantly correlated with soil organic matter, total nitrogen, and available nitrogen content (Wang et al., 2020). The aboveground biomass, belowground biomass and numbers of stems and stem nodes of B. schreberi are significantly higher in treatments with nitrogen, phosphorus, and organic fertilizers added to the sediment compare to the control group without fertilization (Zheng et al., 2018). Existing research suggests that the growth of B. schreberi depends on high-quality water and nutrient-rich sediment, but these claims remain to be thoroughly validated and still require additional supporting evidence. Moreover, the contributions of key sediment nutrients—carbon, nitrogen, and phosphorus—to the growth of B. schreberi are not yet well understood.
Based on the above background, how the stem structural characteristics of B. schreberi influence its photosynthetic physiological functions, and how these parameters vary along gradients of water environment and sediment nutrients such as nitrogen and phosphorus, are the scientific questions that this study aims to address. This study hypothesizes that the environmental variation within our set of wetlands (both water and sediment characteristics) led to significant variation among stem and photosynthetic traits of B. schreberi, the survival of B. schreberi require good water quality and nutrient-enriched sediments, and close correlations exist between plant traits and dissolved oxygen content and some pollution indicators of water, as well as the nutrient content of carbon, nitrogen and phosphorus in the sediment. To verify the hypotheses, a study was conducted at Tengchong Beihai wetland, and its natural B. schreberi was taken as the research object. The focus of the study was on the important stem nutritional structure of B. schreberi, by measuring photosynthetic carbon assimilation parameters as well as stem structural characteristics. This study explores the ecological response strategies of the stem functional characteristics of B. schreberi to water and sediment conditions, and verifies the characteristics of water environment and sediment conditions for the growth of B. schreberi. This study will provide a case for understanding the functional adaptation strategies of aquatic plants, and provide a theoretical basis for the scientific protection and rational utilization of B. schreberi.
The study site is located in the Beihai Wetland Provincial Nature Reserve (N 25°06′42″-25°08′49″, E 98°30′55″-98°35′02″) in Yunnan Province of China. The Beihai Wetland, with a mean altitude of 1,731 m, is surrounded by mountains, exhibiting the characteristics of “basin-lakeside-hillside”. The annual mean temperature is 14.7°C, which is lower than that in most areas with the same latitude and altitude. The annual mean rainfall in the area is 1,750 mm, and the climate is cool and humid. The annual evaporation is 1575 mm, and the annual mean humidity is 79%. According to the Research Report of the Water Quality in 2022, the water in Beihai Wetland is clear, with good water environment conditions. The overall water environment quality category is Class II, and some local spots are Class III. The eutrophication of the water is mild to moderate. The lake is rich in submerged plants such as Hydrilla verticillata, Utricularia aurea, and Myriophyllum spicatum, as well as floating-leaf plants like B. schreberi and Trapa incisa.
The current water surface area of Beihai Wetland is about 300 hm², mainly consisting of two parts: the northern and southern areas. The northern part is the original Beihai Wetland area, with an average water depth of 3 m and a maximum depth of 10 m. The area with a water depth of less than 3 m is sparsely distributed with B. schreberi, with a coverage of about 70%. The northeastern part has a large area of marshy floating mat meadows. The southern part was originally paddy fields reclaimed by farmers along the lake. It was restored to a wetland through a “farmland-to-wetland” project carried out by the local government from 2010 to 2015. The current water depth in the southern part ranges from 0.5 to 2 m. The southwestern part has a large area of overlapping B. schreberi plants, with a coverage reaching 100%. B. schreberi is the dominant species in the southern farmland-to-wetland area of Beihai Wetland and is also the main area for the distribution of this species in Beihai Wetland.
According to the field investigation, the growth of B. schreberi in Beihai Lake can be roughly divided into two cases. One is that B. schreberi is sporadically distributed in the eastern and northern of Beihai Lake, and the coverage of B. schreberi is about 70%. Another case is that in the western and the southern parts with the B. schreberi is distributed in a large area, and the coverage of the distribution points reaches 100%. According to the ecological environment conditions and the growth and distribution of B. schreberi, 17 research points were selected in the distribution range of B. schreberi in Beihai Lake, including 8 points with 70% coverage and 9 points with 100% coverage (Figure 1). In order to avoid the influence of asexual reproduction of B. schreberi, five healthy and similarly growing B. schreberi were selected as the study objects at each point, and each plant was separated by more than 5 m. To avoid differences in traits caused by variations in water depth, the water depth at all our sampling sites was maintained at around 1.5 meters, as this depth is the optimal growth depth for B. schreberi (Zhang et al., 2018).
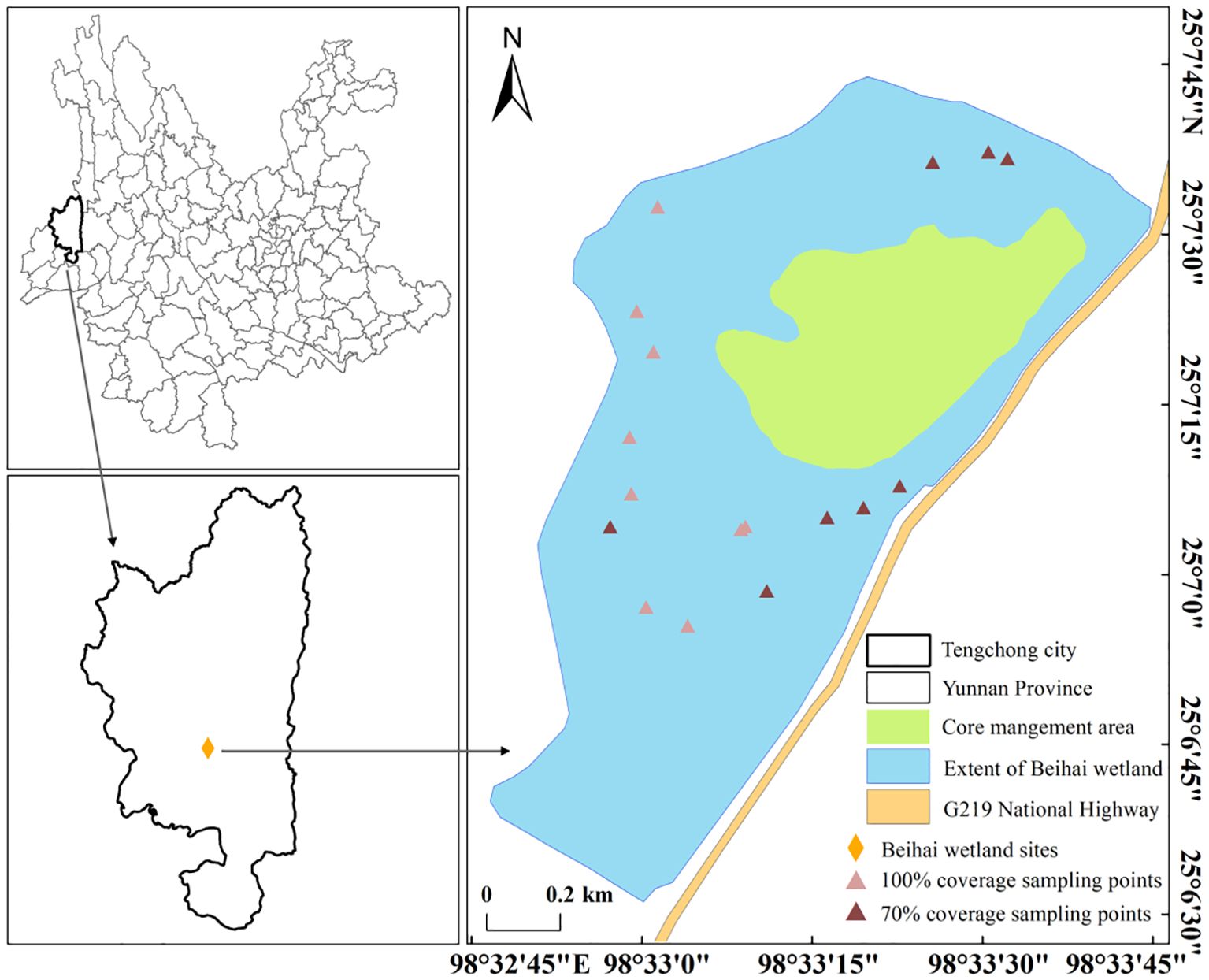
Figure 1. The sampling points were set up in the Beihai Wetland in this study. In the figure, the brownish-red points represent sampling points with a coverage of 70%, which are mainly located in the eastern and northern parts of the wetland, and are sparsely distributed. The pink points represent sampling points with a coverage of 100%, which are mainly located in the southern and southwestern parts of the wetland, and are densely clustered.
The physiological functions of stems are mainly reflected in the transport of water, substances, and air, as well as providing structural support and protection for the internal tissues of plant. In this study, we selected vascular bundles, ventilation holes, and epidermal structural traits that correspond to these functions, as well as photosynthetic parameters that can directly reflect the physiological performance of these structures, for quantitative measurement. In June 2022, the field photosynthetic parameters were measured during the peak growth period of B. schreberi, and the daily measurement period was between 9:00-12:00 a.m. The net photosynthetic rate (Pn, μmol·m−2·s−1), stomatal conductance (Gs, mol·m−2·s−1) and transpiration rate (Tr, mmol·m−2·s−1) of healthy and mature leaves were measured and recorded in situ using LI-6800 photosynthetic fluorescence measuring instrument (LI-6800, LICOR, Nebraska, USA). Before the determination, a small CO2 cylinder was installed and the instrument was preheated for 30 minutes. The CO2 concentration in the leaf chamber was set at 420 μmol·mol-1. The leaf chamber temperature and the air relative humidity were maintaining natural conditions. The leaf chamber temperature is 25-27°C and the air relative humidity is 75-80%. In the determination, the leaves of B. schreberi were first induced by 1800 μmol·m−2·s−1 light for 2 min to maintain the maximum stomatal conductance, and then the light intensity was adjusted to 1500 μmol·m−2·s−1, and the photosynthetic parameters were determined after the leaf chamber CO2 concentration was matched and balanced.
After measuring the photosynthetic physiological parameters, cut off a section of the stems of the B. schreberi that is about 5 cm long, and about 50 cm away from the leaves. Mark the sections well and immerse them in FAA fixative solution (volume ratio of 70% alcohol, 100% glacial acetic acid, and 38% formaldehyde is 90:5:5) for at least 48 hours. After preservation in the preservation box, they were brought back to the laboratory to determine the anatomical structure of the stem sections. In the laboratory, the cross section of the stem of B. schreberi was sliced with double-sided stainless blade, stained with 1% toluidine blue for 1 min, and made into temporary water. The slices were observed and photographed under an optical microscope (Figure 2). Photographs of epidermal cells and cuticle were taken in the epidermis; then avoid the epidermis and take photos of the vascular bundle structure and the ventilation hole in the middle part. The vascular bundle area (BA, μm2), ventilation hole area (VA, μm2), cuticle thickness (CT, μm) and epidermal thickness (ET, μm) of the stem were measured and counted by Image J processing software (http://rsb.info.6nih.gov/ij/). The methods for measuring photosynthetic and stem structural parameters have been refined and perfected through our long-term use, and they are now well-established and capable of meeting the requirements for trait measurement.

Figure 2. Stem anatomical structures of B. schreberi. co, cortex; ep, epidermis; cu, cuticle; vb, vascular bundle; v, conduit; ae, aerenchyma; vh, ventilation hole; s, mucilage hair.
To minimize the impact of asexual reproduction in B. schreberi, a spacing of at least 5 meters should be maintained between each plant during sampling. At each sampling point, five plants are selected to measure functional traits. To ensure robust statistics, when measuring field photosynthetic parameters, an indefinite number greater than five plants (usually 8 to 10) are chosen for measurement. This approach stabilizes the values of most photosynthetic parameters. After discarding the abnormally high and low values, five stable photosynthetic parameters are selected. The plants corresponding to these five parameters are then recorded for photosynthetic traits and subsequently measured for stem structural parameters. Six values were counted for each trait of each plant to ensure that each trait of each study point had 30 statistical values.
Sediment nutrients are essential for the growth of floating-leaved aquatic plants and significantly impact their growth, ecological functions, and water purification capabilities. Carbon, nitrogen, and phosphorus—macronutrients—are particularly critical, as they determine the metabolic processes and overall growth of these plants and are central to research. Carbon forms the backbone of organic compounds in plants, providing the energy and material basis necessary for their growth and development. Nitrogen and phosphorus, on the other hand, are integral components of many vital organic compounds and directly engage in processes such as photosynthesis, respiration, and energy transfer. They also serve as key constituents of numerous intermediates in photosynthetic metabolism. In this study, a total of 1 kg of sediment samples were collected at each sampling point using a fixed-depth peat drill (Eikel Kampala 0423SA, Netherlands). The samples were then brought back to the laboratory and allowed to air dry naturally. After drying, the samples were finely ground using a soil crusher and sieved through a 100-mesh screen before being sealed and stored. These sediment samples were sent to a third-party testing agency at the Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, to determine the mass fractions (ω, g·kg-1) of carbon (C), nitrogen (N), and phosphorus (P) elements.
Besides the sediment nutrient conditions, water quality is another crucial microenvironmental factor influencing the growth of B. schreberi. Key indicators for assessing water quality include water temperature (WT, °C), pH value (pH, mol·L-1), dissolved oxygen content (DO, mg·L-1), nitrogen and phosphorus concentrations, biochemical oxygen demand (BOD, mg·L−1), five-day chemical oxygen demand (COD, mg·L−1) and potassium permanganate index (CODMn, mg·L−1). WT is one of the most basic parameters in water quality monitoring, directly affecting the living conditions of aquatic organisms, the amount of dissolved oxygen, and the reaction rates of chemical substances. The pH, which measures the strength of water’s acidity or alkalinity, also impacts the solubility and toxicity of chemical substances in water. DO is fundamental for the respiration of aquatic organisms and is directly related to their survival. The nitrogen and phosphorus content in water is an important indicator for evaluating the degree of eutrophication. Excess levels of these nutrients can lead to the rapid growth of planktonic plants, causing water turbidity and negatively affecting the growth of large aquatic plants. BOD, COD, and CODMn also reflect the amount of organic matter in water, which is associated with the degree of eutrophication. The DO, pH and WT were measured in situ at each sampling point using a multi-parameter water quality analyzer (YSI 650 MDS). Subsequently, 500 mL of water was collected from each sampling point and brought back to the laboratory. In the laboratory, 40 mL of water from each sampling point was filtered and then analyzed using a continuous flow analyzer (Germany SEAL Analytical AA3) to determine and calculate the total nitrogen volume fraction (Nwater, mg·L−1) and total phosphorus volume fraction (Pwater, mg·L−1). The remaining water samples were sent to a third-party professional testing institution to determine the ammonia nitrogen volume fraction (NH4+, mg·L−1), nitrate nitrogen volume fraction (NO3−, mg·L−1), BOD, COD and CODMn.
The data from this study were analyzed using the statistical analysis softwares of SPSS (v.25, https://spss.en.softonic.com) and Canoco (5.0 https://www.canoco5.com). The data were firstly tested for normality, and the results showed that the data followed a normal distribution. To compare the differences in the functional traits of B. schreberi at two different canopy coverages, the independent samples t-test was employed with a significance level of P<0.05, and the homogeneity of variances between the two groups of data were tested using Levene’s test. Pearson correlation analysis was used to detect the biovariable correlations between the functional traits of B. schreberi and environmental condition factors with a significance level of P<0.05. Principal component analysis (PCA) of the functional traits of B. schreberi revealed that the total variance was less than 3, and then Redundancy analyses (RDA) were conducted to further identify the key water and sediment factors that influence the functional traits and the relationships between the traits and environmental factors.
Compared to B. schreberi with a coverage of 70%, the species with 100% coverage exhibited significantly higher stomatal conductance (Gs) and transpiration rate (Tr), but no significant difference were observed in net photosynthetic rate (Pn) between the two coverage (P>0.05) (Figure 3), indicating that stronger stomatal exchange of water and vapor, and higher leaf water transpiration exhibited in B. schreberi with 100% coverage, yet its net photosynthetic rate remains stable, compared to it with 70% coverage. B. schreberi with 100% coverage exhibited significantly lower vascular bundle area (BA) and ventilation hole area (VA) (P<0.05), while no significant differences were observed in cuticle thickness (CT) and epidermal thickness (ET) between the two coverage (P>0.05) (Figure 3), indicating that plant under 100% coverage showed lower water and air transportion, while its mechanical resistance of the epidermal structure remains stable.
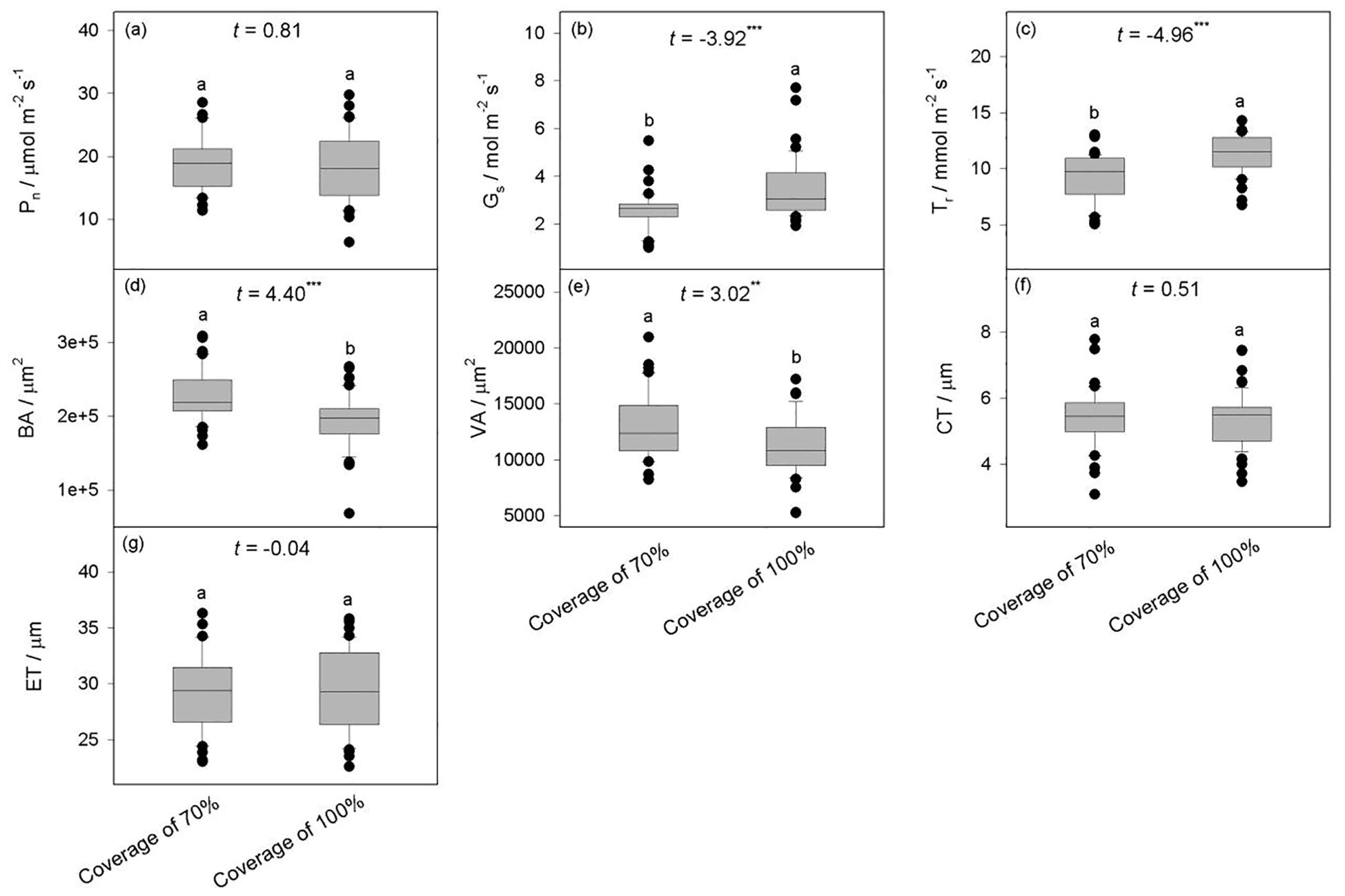
Figure 3. Differences in the characteristics of B. schreberi between two canopy coverage. (a–c) showed the differences in photosynthetic parameters that higher Gs and Tr were observed under the 100% coverage, while there was no significant difference in Pn between the two coverage. (d–g) showed the differences in stem structural characteristics that smaller BA and VA were detected under the 100% coverage, while no differences detected in CT and ET between the two coverage. Pn, net photosynthetic rate (a); Gs, stomatal conductance (b); Tr, transpiration rate (c); BA, vascular bundle area (d); VA, ventilation hole area (e); CT, cuticle thickness (f); ET, epidermal thickness (g). **, P<0.01; ***, P<0.001.
A redundancy analysis (RDA) was used to detect the impact of environmental factors on plant traits. The first two axes of RDA explained 45.3% and 24.2% of the total variance variation, respectively (Figure 4). WT, ω(N) and DO were the main parameters affecting the characters of B. schreberi (Figure 4, Table 1). WT and DO mainly negtively correlated with the axis 1, while the ω(N) mainly positively correlated with the axis 2 (Figure 4). Among the traits of B. schreberi, Gs and Tr were mainly positively correlated with the axis 1, while the BA and VA were mainly negatively correlated with axis 1; Pn and CT were mainly negatively correlated with axis 2 (Figure 4). Among the environmental parameters, the WT has the highest explanatory power, reaching 18.3%; followed by ω(N) and DO with explanatory powers of 13.1% and 11.8%, respectively (Table 1). The explans and contributions of these three parameters have reached significant levels (Table 1). The other parameters and their explanatory powers were ω(P) 8.4%, Nwater 7.8%, CODMn 5.2%, COD 4.9%, ω(C) 4.5%, Pwater 4.1%, NH4+ 4%, pH 3.7%, BOD 3.4%, and NO3- 2.1% (Table 1). Ranking of the contributions of the environmental parameters is consistent with their explans.
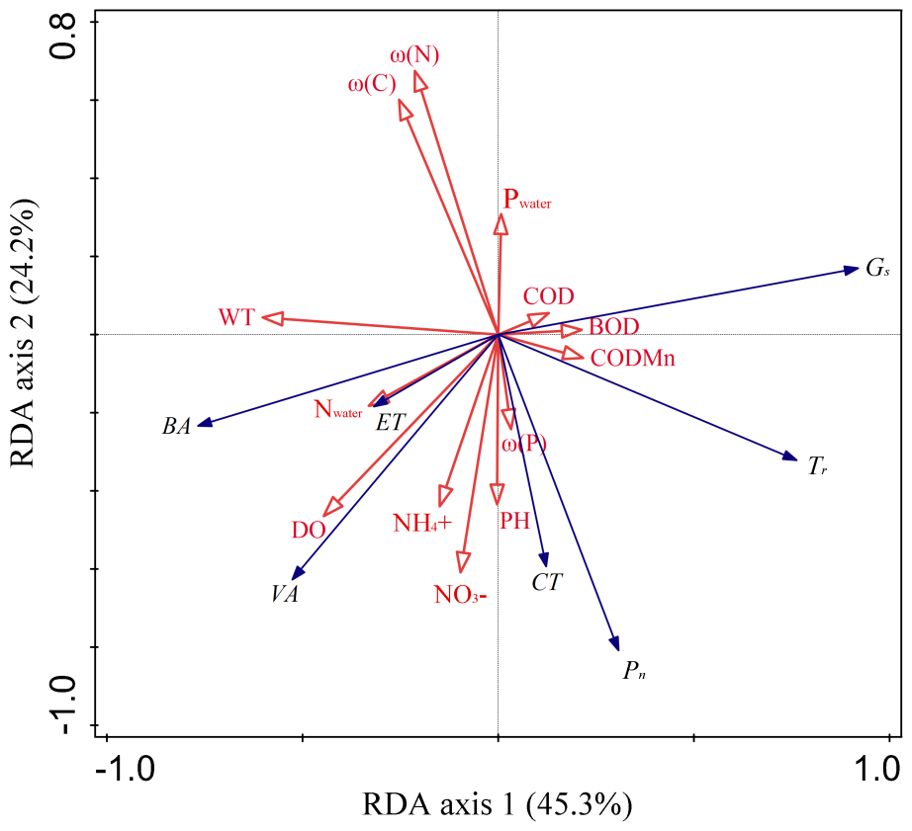
Figure 4. The first two axes of RDA between plant functional traits and environmental factors. The first two axes of RDA explained 69.5% of the total variance variation. The six functional traits are all located far from the origin, indicating high sensitivity to water and sediment parameters. Among them, Gs, Tr, BA, and VA were mainly distributed along the first axis, while Pn and CT were primarily distributed along the second axis. Among the environmental parameters, WT, and DO were mainly distributed along the first axis, while ω(N) and ω(C) were primarily distributed along the second axis. Pn, net photosynthetic rate; Gs, stomatal conductance; Tr, transpiration rate; BA, vascular bundle area; VA, ventilation hole area; CT, cuticle thickness; ET, epidermal thickness. ω(C), sediment mass fraction of carbon; ω(N), sediment mass fraction of nitrogen; ω(P), sediment mass fraction of phosphorus; DO, dissolved oxygen content; pH, water pH value; WT, water temperature; Nwater, water total nitrogen volume fraction; Pwater, water total phosphorus volume fraction; NH4+, ammonia nitrogen volume fraction; NO3−, nitrate nitrogen volume fraction; BOD, biochemical oxygen demand; COD, five-day chemical oxygen demand; CODMn, potassium permanganate index.
With the biovariate correlations between plant traits and sediment elements, the Pn, Tr, CT and ET were all significantly negatively correlated with ω(C) and ω(N), while the Pn was positively correlated with ω(P) (P<0.05; Table 2). This result indicated that excessive carbon and nitrogen in the sediment showed reduce the net photosynthetic rate of B. schreberi and thin its epidermal barrier structure, thereby weakening the protective function of the barrier.
With the biovariate correlations of plant traits and water parameters, The Pn, VA and CT were all significantly positively correlated with NH4+ and NO3- (P<0.05; Table 2). Pn, BA and VA were all significantly positively correlated with DO, the latter two traits were also both significantly positively correlated with WT, while Tr was significantly negatively correlated with DO and WT (P<0.05; Table 2). Besides, Tr and ET were positively correlated with CODMn; VA and CT were both positively correlated with pH (P<0.05; Table 2). Nwater, Pwater, COD and BOD contribute little to the plant characteristics, with most of the correlations were insignificant but the correlation between Nwater and ET (Table 2). Overall, DO, WT, pH, NH4+, and NO3− were mainly positively correlated with Pn, BA, VA, and CT, indicated that higher levels of these parameters could promote the net photosynthetic rate of B. schreberi, enhance the transport volumes of water and air, and stabilize the cuticle barrier structure.
Some relations existed in the plant characteristics of B. schreberi, reflecting their functional associations. Gs was positively correlated with Tr (Table 3), indicates that the process of water and vapor exchange through stomata is coupled with the process of water transpiration loss, and they together regulate net photosynthetic production to maintain stability. Larger BA and VA can transport more water and air at one time, but they correspond to lower transport efficiency. The BA, VA and ET were significantly positively correlated with each other, and the three traits were all negatively correlated with Gs; BA also significantly negatively correlated with Tr (Table 3). These results indicated that B. schreberi, under higher coverage, exhibited higher stomatal water and vapor exchange and transpirational water loss, corresponding to higher water and air transport efficiency. However, the mechanical stability of the epidermal barrier decreased.
Stomatal conductance and transpiration rate are generally positively correlated with photosynthetic rate since they both reflect the ability of water and CO2 exchange during the photosynthetic carbon assimilation, although under some extreme conditions (such as drought, high temperature, or salt stress), plants may respond to stress by closing their stomata, which can lead to a decrease in stomatal conductance and transpiration rate. The B. schreberi with 100% coverage had higher stomatal conductance and transpiration rate compared to this species with 70% coverage (Figure 3), indicating that B. schreberi with higher coverage had higher photosynthetic water vapor and CO2 exchange capacity, and higher photosynthetic rate in theory. However, there was no significant difference in the net photosynthetic rate between the two groups, indicating that the higher coverage of B. schreberi reduced the utilization efficiency of water and CO2, the same photosynthetic capacity needed to consume more water and CO2 at higher coverage (Peixoto et al., 2016; Oliveira-Junior et al., 2018; Liu et al., 2024). Even if the increase of water vapor and CO2 exchange increased the actual photosynthetic rate of B. schreberi, the intense plant respiration under hypoxic conditions would lead to a decrease or no change in photosynthetic rate (Dusenge et al., 2019). The smaller stem vascular bundles and ventilation holes of B. schreberi with higher coverage (Figure 3), may be a response to higher water vapor and air exchange capacity. Higher water vapor exchange capacity requires faster water and air transport, so higher water and air transport efficiency is required (Pan et al., 2021; Zhao et al., 2022). Small vascular bundles and ventilation holes generally correspond to a greater numbers of the structures, which decrease the risk of cavitation with the increase of velocity in the process of transporting water and air, thus increasing the relative area of transportation and improving the efficiency of water and air transportation (Caraco et al., 2006; Qaderi et al., 2019). Therefore, B. schreberi with high coverage will reduce their utilization efficiency of water and CO2, while increase their efficiency of water and air transportation, indicating that high coverage exacerbates the habitat stress on B. schreberi. This result supports the hypothesis of this study that the growth of B. schreberi requires good water quality. Under high cover conditions in Tengchong Beihai wetland, the water where B. schreberi grows has significantly lower dissolved oxygen content (DO=1.9 mg/L) and significantly lower water temperature (WT=21.8 ℃) reflecting deteriorated water quality. Previous studies also pointed the high coverage of floating leaves and floating plants will lead to a serious decrease in the amount of oxygen available in the lower layer of the plant (Oliveira-Junior et al., 2018; Henriot et al., 2019). The respiration of the leaves and roots in the lower layer of the plant is strong, and the plant must transport more air to maintain the survival of the plant (Dai et al., 2012; Tang et al., 2017; Oliveira-Junior et al., 2018).
Water and sediment serve as vital sources of nutrients and energy required for the growth and development of aquatic plants, and their environmental conditions significantly influence the expression of functional traits in these plants (Pan et al., 2020; Henriot et al., 2019; Xie et al., 2018). Water temperature and dissolved oxygen content are the most significant water factors affecting the photosynthetic physiology and stem structural traits of B. schreberi (Figure 4; Tables 2, 3). Floating-leaved plants reduce water temperature and dissolved oxygen content through reducing water transmittance and gas exchange between water and air, which in turn limits water reoxygenation and photosynthetic enzyme activity, respectively, increases photorespiration and plant respiration, and reduces net photosynthetic rate of B. schreberi (Jiang et al., 2022; Said et al., 2021). Stable net photosynthetic rate is the foundation to ensure plant normal growth, reproduction, and dispersion under stress conditions (Lamers et al., 2020; Li et al., 2021). Higher transpiration rate and stomatal conductance, along with smaller vascular bundle area and aerenchyma area, are the typical phenotypic traits that contribute to higher photosynthetic rate. Therefore, the net photosynthetic rate of B. schreberi may be enhanced by increased water-vapor exchange capacity and improved water and air transport efficiency, thus the photosynthetic limitation caused by low temperatures can be alleviated and stable net photosynthetic rate can be maintained. The significant correlations of stomatal conductance and transpiration rate to vascular bundle area and aerenchyma area, indicating that these four traits play similar roles in maintaining the stability of plant photosynthetic function and close functional relationships exist in the traits. The significant positive correlation between net photosynthetic rate and dissolved oxygen also suggests that a higher oxygen supply is the foundation for ensuring higher photosynthetic rate in plants.
The rhizomes of floating-leaved plants are rooted in the sediment, and obtained nutrients from the sediment, making sediment nutrient conditions an important factor that affects their growth strategies (Henriot et al., 2019; Titus and Gary Sullivan, 2001). Sediment nitrogen content exhibits significant negative correlations with net photosynthetic rate, transpiration rate, epidermal thickness, and cuticular thickness of B. schreberi (Table 2), indicating that excessively high nitrogen levels in the sediment are not conducive to the photosynthetic production of B. schreberi, and also limit the epidermic water retention and mechanical support capabilities of its stem epidermis structure. Nitrogen is a key component of plant chlorophyll, proteins, and some other components (Larson and Funk, 2016). In general, the availability of nitrogen in the environment is a crucial factor determining plant growth, and high levels of environmental nutrients can promote rapid plant reproduction and expansion (Wright et al., 2004). Some former studies also pointed the growth of B. schreberi requires good water quality and nutrient-enriched sediments based on mucilage content (Li et al., 2021; Xie et al., 2018), and its artificial propagation often requires applying sufficient base fertilizer before planting (Zhang et al., 2018), while our results are not entirely consistent with this statement based on net photosynthetic rate. The distribution sites in this study are mainly located in areas where farmland has been restored to wetlands and water has been stored, resulting in relatively high levels of nutrient elements, providing sufficient nutrients for the growth of B. schreberi. Therefore, this study has, to a certain extent, verified that the growth of B. schreberi requires fertile sediment. However, high sediment nitrogen inhibited the photosynthetic physiology of B. schreberi, thus a limit should be placed on the nitrogen demand of B. schreberi, indicating that excessive sediment nitrogen has an inhibitory effect on photosynthetic physiology, while moderate nitrogen content is the optimal condition.
Under hypoxic wetland conditions, nitrogen in the sediment is converted into higher concentrations of ammonium nitrogen through the process of denitrification. Elevated ammonium concentrations typically facilitate the production of various phytotoxic compounds in the rhizosphere, which inhibit the photosynthetic physiological processes of plants and restrict the increase in plant growth characteristics (Ponnamperuma, 1972; Pezeshki, 2001). For instance, high ammonium concentrations and temperatures in wetlands are usually significantly and negatively correlated with the growth and biomass production of rhizomes (Henriot et al., 2019; Klok and van der Velde, 2017). Size parameters are considerded as growth traits, while substances like the mucilage of B. schreberi, are primarily secondary metabolites (Henriot et al., 2019). A higher amount of secondary metabolites often incurs greater construction costs, thereby weakening growth traits, and therefore, under high-nitrogen substrate conditions, the accumulation of more mucilage (such as Li et al., 2021; Xie et al., 2018) is a stratage for plants to alleviate stress, but it may not conducive to their good growth and reproduction that the latter are frequently related to plant competitive ability (Grime, 2006). In the conservation and management of rare aquatic plants such as B. schreberi in wetland protected areas, environmental conditions should be carefully controlled (for example, by increasing dissolved oxygen and reducing sediment nitrogen content) to ensure that more photosynthetic products are allocated to plant growth rather than to the production of secondary metabolites. Conversely, for artificial cultivation aimed at obtaining higher yields of secondary metabolites, while ensuring the basic growth conditions of the plants, efforts can be made to direct more photosynthetic products towards the synthesis of these secondary metabolites.
Phosphorus is a key constituent of nucleic acids, energy carrier adenosine triphosphate (ATP), and numerous enzymes. It plays a vital role in photosynthetic processes like photophosphorylation and the Calvin cycle, and is essential for cell division and nutrient uptake in plants. Therefore, the supply of phosphorus is crucial for the healthy growth and high yield of plants. The significant positive correlation between sediment phosphorus content and net photosynthetic rate (Table 2) indicates that compared to sediment nitrogen, sediment phosphorus content has a stronger limiting effect on the photosynthetic production of B. schreberi. Similarly, some growth traits, such as leaf size and stem size also have previously been demonstrated to increase with nutrient content and particularly with phosphates (Henriot et al., 2019; Klok and van der Velde, 2017). Compared to the effects of nitrogen, phosphorus has a greater impact on the growth and reproduction traits of Nuphar lutea (a close relative of B. schreberi), with its rhizome size and number of flowers are significantly positively correlated with the phosphorus content in the sediment, while they show negative correlations with the nitrogen content in the sediment (Henriot et al., 2019). In salt marshes, the addition of phosphorus or a combination of nitrogen and phosphorus induces a rapid shift in community dominance from microalgae to higher plants, particularly Eleocharis spp. and Typha domingensis, and additionally, phosphorus addition results in a four- to five-fold increase in tissue phosphorus content in Eleocharis compared to control plants (Rejmánková et al., 2008). In phosphorus-limited wetlands, aquatic plants often show high sensitivity to phosphorus. Even minor additions of phosphorus can spur rapid plant growth and boost photosynthetic rate, thus enhancing their competitive ability. The significant correlation between the photosynthetic rate of B. schreberi and phosphorus in Tengchong Beihai wetland indicates that this wetland is phosphorus-limited.
Epidermal structure is an important barrier in plants and maintaining a healthy epidermal structure is essential for plants to grow and thrive (Yang et al., 2020). Epidermal structure provides protection against external factors such as pathogens, insects, and harsh environmental conditions, while also regulating water loss and gas exchange through its pores, stomata, and waxy coatings (Thompson and Gilbert, 2014). In addition, the epidermal structure also contributes to the mechanical support of the plant body. Thin and discontinuous cuticle of epidermis may contribute to aquatic plants sensitivity to water pollution, like Genlisea and B. schreberi (Yang et al., 2020; Płachno et al., 2005). The significant negative correlations of sediment nitrogen content to epidermal thickness and cuticular thickness suggests that high sediment nitrogen levels can thin out the cuticle and epidermis of B. schreberi, thereby weakening its protective barrier structure. This indicates that while nitrogen is a necessary nutrient for plant growth, excessive nitrogen in the sediment can have negative impacts on the epidermal integrity and function of this aquatic plant (Zhang et al., 2015; Xie et al., 2018).
This study aims to investigate the relationships between the stem structure and photosynthetic traits of B. schreberi and environmental variables within a single wetland over a one-year study period. As B. schreberi is a long-lived plant species, the relationship between its environment and species traits may occur over a longer timescale than the duration of this study. Similarly, its large rhizomes may integrate responses to environmental signals over a very large scale, potentially leading to an attenuation of the measured responses at the rhizome fragment level, despite previous studies highlighting the reactivity of plant ramets to habitat variability. Although the environmental characteristics of the Tengchong Beihai wetland show some variation, we may still have underestimated the impact of environmental features on plant traits at broader and larger scales. Additionally, the timescale of the study does not allow for the assessment of environmental changes that occur over longer periods. For example, the continuous input of nutrient-rich agricultural water from surrounding areas and increasing biotic competition and invasions within the wetland may have adverse effects on B. schreberi over timescales exceeding one year, gradually degrading its health over time. Our study primarily used correlation analysis to detect the close relationships between traits and the environment; however, these relationships still need to be further validated through causal analysis and controlled experimental approaches, which will be the focus of our future work.
Dissolved oxygen, water temperature, nitrogen and phosphorus contents in the sediment are the primary factors influencing the stem structure and photosynthetic traits of B. schreberi. High plant coverage of B. schreberi results in decreased water temperature and dissolved oxygen levels. In response, the plant exhibits higher stomatal conductance and transpiration rates, while the size of stem vascular bundles and aerenchyma tissues decreases, the net photosynthetic rate remains constant, indicating reduced efficiency in the utilization of water and CO2 and B. schreberi requires good water quality for growth. Its high photosynthetic rate does not depend on high sediment nitrogen content but is significantly positively correlated with sediment phosphorus content, suggesting that it is prone to phosphorus limitation. Based on these findings, in the conservation and management of rare aquatic plants like B. schreberi in wetland protected areas, it is crucial to control environmental conditions to enhance plant photosynthesis, morphological size, and other growth characteristics, thereby boosting plant competition. Additionally, efforts should be made to reduce the production of secondary metabolites, such as mucilage. For example, increasing dissolved oxygen levels, moderately raising water temperature, and managing sediment nitrogen while increasing sediment phosphorus content are recommended strategies. Expanding the research scope, conducting continuous dynamic studies, and developing models to validate the causal relationships between traits and environmental factors will be key priorities for future work.
The data analyzed in this study is subject to the following licenses/restrictions: The associated data of this paper are not suitable for sharing but can be obtained from the author upon reasonable request. Please contact the author at c20wNTEwMjE1QDE2My5jb20=.
TW: Data curation, Writing – original draft. HY: Data curation, Writing – original draft. HC: Data curation, Writing – review & editing. WZ: Data curation, Writing – review & editing. ZL: Data curation, Writing – review & editing. QL: Data curation, Writing – review & editing. MS: Investigation, Project administration, Writing – original draft, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work is financially supported by the Agricultural Joint Special Project of General Program under the Science and Technology Department of Yunnan Province (202101BD070001-099), the Open Fund of the Key Laboratory for Plateau Wetland Protection, Restoration, and Ecological Services (Education) in Yunnan Province (202105AG070002), and the Funded Project of Yunnan Biodiversity Protection Foundation.
We would like to thank Du Kang for collecting materials. We also need to thank the boatmen of Beihai Wetland Provincial Nature Reserve in Tengchong for helping us rowing.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Armstrong, A. F., Logan, D. C., Atkin, O. K. (2006). On the developmental dependence of leaf respiration: responses to short-and long-term changes in growth temperature. Am. J. Bot. 93, 1633–1639. doi: 10.3732/ajb.93.11.1633
Bociag, K., Galka, A., Lazarewicz, T., Szmeja, J. (2009). Mechanical strength of stems in aquatic macrophytes. Acta Societatis Botanicorum Poloniae 78, 181–187. doi: 10.5586/asbp.2009.022
Bornette, G., Puijalon, S. (2011). Response of aquatic plants to abiotic factors: a review. Aquat. Sci. 73, 1–14. doi: 10.1007/s00027-010-0162-7
Brulé, V., Rafsanjani, A., Asgari, M. (2019). Three-dimensional functional gradients direct stem curling in the resurrection plant Selaginella lepidophylla. J. R. Soc. Interface 16, 20190454. doi: 10.1098/rsif.2019.0454
Caraco, N., Cole, J., Findlay, S. (2006). Vascular plants as engineers of oxygen in aquatic systems. BioScience 56, 219–225. doi: 10.1641/0006-3568(2006)056[0219:vpaeoo]2.0.co;2
Chen, H., Sun, M., Liu, Z., Yang, H. (2024). Response of leaf economic traits of natural Brasenia schreberi to water environment in high altitude area. Chin. J. Ecol. 43, 1763. doi: 10.13292/j.1000-4890.202406.042
Cheng, J. J., Zheng, X. N., Zhang, Z. Y., Chen, J. (2022). Analysis on differences in leaf functional traits of desert plants under interspecific association pattern in different water-salt environments of Ebinur Lake. J. Plant Resour. Environ. 31, 18–25. doi: 10.3969/j.issn.1674-7895.2022.0303
Dai, Y., Jia, C., Liang, W. (2012). Effects of the submerged macrophyte Ceratophyllum demersum L. @ on restoration of a eutrophic water body and its optimal coverage. Ecol. Eng. 40, 113–116. doi: 10.1016/j.ecoleng.2011.12.023
Dhote, S., Dixit, S. (2009). Water quality improvement through macrophytes - a review. Environ. Monit. Assess. 152, 149–153. doi: 10.1007/s10661-008-0303-9
Drzymulska, D. (2018). On the history of brasenia schreb. in the european pleistocene. Vegetation History Archaeobotany 27, 527–534. doi: 10.1007/s00334-017-0652-9
Dusenge, M. E., Duarte, A. G., Way, D. A. (2019). Plant carbon metabolism and climate change: elevated CO2 and temperature impacts on photosynthesis, photorespiration and respiration. New Phytol. 221, 32–49. doi: 10.1111/nph.15283
Grasset, C., Delolme, C., Arthaud, F. (2015). Carbon allocation in aquatic plants with contrasting strategies: the role of habitat nutrient content. J. Vegetation Sci. 26, 946–955. doi: 10.1111/jvs.12298
Grime, J. P. (2006). Trait convergence and trait divergence in herbaceous plant communities: mechanisms and consequences. J. vegetation Sci. 17, 255–260. doi: 10.1111/j.1654-1103.2006.tb02444.x
Han, G., Li, Y., Qiao, Z. (2021). Advances in the regulation of epidermal cell development by C2H2 zinc finger proteins in plants. Front. Plant Sci. 12. doi: 10.3389/fpls.2021.754512
Henriot, C. P., Cuenot, Q., Levrey, L. H., Loup, C., Chiarello, L., Masclaux, H., et al. (2019). Relationships between key functional traits of the waterlily Nuphar lutea and wetland nutrient content. PeerJ 7, e7861. doi: 10.7717/peerj.7861
Hu, J., Chen, Z., Yang, M., Zhang, R., Cui, Y. (2018). A multiscale fusion convolutional neural network for plant leaf recognition. IEEE Signal Process. Lett. 25, 853–857. doi: 10.1109/LSP.2018.2809688
Islam, T., Hamid, M., Nawchoo, I. A., Khuroo, A. A. (2024). Leaf functional traits vary among growth forms and vegetation zones in the Himalaya. Sci. Total Environ. 906, 167274. doi: 10.1016/j.scitotenv.2023.167274
Jiang, W. Y., Zhang, Y. H., Liu, Y. C. (2022). The effect of abrupt increase in water temperature on the survival and growth of eelgrass Zostera marina. Aquat. Bot. 183, 103572. doi: 10.1016/j.aquabot.2022.103572
Kim, H., Na, S. H., Lee, S. Y., Jeong, Y. M., Hwang, H. J., Hur, J. Y., et al. (2012). Structure–function studies of a plant tyrosyl-DNA phosphodiesterase provide novel insights into DNA repair mechanisms of Arabidopsis thaliana. Biochem. J. 443, 49–56. doi: 10.1042/BJ2011130
Klok, P. F., van der Velde, G. (2017). Plant traits and environment: floating leaf blade production and turnover of waterlilies. PeerJ 5, e3212. doi: 10.7717/peerj.3212
Lamers, J., van der Meer, T., Testerink, C. (2020). How plants sense and respond to stressful environments. Plant Physiol. 182, 1624–1635. doi: 10.1104/pp.19.01464
Larson, J. E., Funk, J. L. (2016). Seedling root responses to soil moisture and the identification of a belowground trait spectrum across three growth forms. New Phytol. 210, 827–838. doi: 10.1111/nph.13829
Lee, H. (2018). Stem cell maintenance and abiotic stress response in shoot apical meristem for developmental plasticity. J. Plant Biol. 61, 358–365. doi: 10.1007/s12374-018-0301-6
Li, L., Dou, N., Zhang, H. (2021). The versatile GABA in plants. Plant Signaling Behav. 16, 1862565. doi: 10.1080/15592324.2020.1862565
Li, Z. Z., Gichira, A. W., Wang, Q. F. (2018). Genetic diversity and population structure of the endangered basal angiosperm Brasenia schreberi (Cabombaceae) in China. PeerJ Life Environ. 6, e5296. doi: 10.7717/peerj.5296
Liu, H., Liu, G., Xing, W. (2021). Functional traits of submerged macrophytes in eutrophic shallow lakes affect their ecological functions. Sci. Total Environ. 760, 143332. doi: 10.1016/j.scitotenv.2020.143332
Liu, Z., Zhao, Y., Yu, H., Zhao, Y., Guo, H., Sun, M. (2024). Response of the functional traits of Schoenoplectus tabernaemontani to simulated warming in the Napahai wetland of northwestern Yunnan, China. Front. Ecol. Evol. 12. doi: 10.3389/fevo.2024.1399584
Łoboda, A. M., Przyborowski, Ł., Karpiński, M. (2018). Biomechanical properties of aquatic plants: The effect of test conditions. Limnology Oceanography: Methods 16, 222–236. doi: 10.1002/lom3.10239
O’Hare, M. T., Aguiar, F. C., Asaeda, T. (2018). Plants in aquatic ecosystems: current trends and future directions. Hydrobiologia 812, 1–11. doi: 10.1007/s10750-017-3190-7
Ogdahl, M. E., Steinman, A. D. (2015). Factors influencing macrophyte growth and recovery following shoreline restoration activity. Aquat. Bot. 120, 363–370. doi: 10.1016/j.aquabot.2014.10.006
Oliveira-Junior, E. S., Tang, Y., Berg, S. J. P. (2018). The impact of water hyacinth (Eichhornia crassipes) on greenhouse gas emission and nutrient mobilization depends on rooting and plant coverage. Aquat. Bot. 145, 1–9. doi: 10.1016/j.aquabot.2017.11.005
Pan, Y., Cieraad, E., Armstrong, J. (2020). Global patterns of the leaf economics spectrum in wetlands. Nat. Commun. 11, 4519. doi: 10.1038/s41467-020-18354-3
Pan, Z. L., Guo, W., Zhang, Y. J. (2021). Leaf trichomes of Dendrobium species (epiphytic orchids) in relation to foliar water uptake, leaf surface wettability, and water balance. Environ. Exp. Bot. 190, 104568. doi: 10.1016/j.envexpbot.2021.104568
Peixoto, R. B., Marotta, H., Bastviken, D. (2016). Floating aquatic macrophytes can substantially offset open water CO2 emissions from tropical floodplain lake ecosystems. Ecosystems 19, 724–736. doi: 10.1007/s10021-016-9964-3
Pezeshki, S. R. (2001). Wetland plant responses to soil flooding. Environ. Exp. Bot. 46, 299–312. doi: 10.1016/S0098-8472(01)00107-1
Płachno, B. J., Adamus, K., Faber, J., Kozłowski, J. (2005). Feeding behaviour of carnivorous Genlisea plants in the laboratory. Acta Botanica Gallica 152, 159–164. doi: 10.1080/12538078.2005.10515466
Ponnamperuma, F. N. (1972). The chemistry of submerged soils. Adv. Agron. 24, 29–96. doi: 10.1016/S0065-2113(08)60633-1
Qaderi, M. M., Martel, A. B., Dixon, S. L. (2019). Environmental factors influence plant vascular system and water regulation. Plants 8, 65. doi: 10.3390/plants8030065
Ran, X., Zhao, F., Wang, Y., Liu, J., Zhuang, Y., Ye, L., et al. (2020). Plant Regulomics: a data-driven interface for retrieving upstream regulators from plant multi-omics data. Plant J. 101, 237–248. doi: 10.1111/tpj.14526
Rejmánková, E., Macek, P., Epps, K. (2008). Wetland ecosystem changes after three years of phosphorus addition. Wetlands. 28, 914–927. doi: 10.1672/07-150.1
Ristic, Z., Jenks, M. A. (2002). Leaf cuticle and water loss in maize lines differing in dehydration avoidance. J. Plant Physiol. 159, 645–651. doi: 10.1078/0176-1617-0743
Said, N. E., McMahon, K., Lavery, P. S. (2021). Accounting for the influence of temperature and location when predicting seagrass (Halophila ovalis) photosynthetic performance. Estuarine Coast. Shelf Sci. 257, 107414. doi: 10.1016/j.ecss.2021.107414
Seago, J. L., Jr., Marsh, L. C., Stevens, K. J. (2005). A re-examination of the root cortex in wetland flowering plants with respect to aerenchyma. Ann. Bot. 96, 565–579. doi: 10.1093/aob/mci211
Serrano-Mislata, A., Sablowski, R. (2018). The pillars of land plants: new insights into stem development. Curr. Opin. Plant Biol. 45, 11–17. doi: 10.1016/j.pbi.2018.04.016
Slot, M., Cala, D., Aranda, J., Virgo, A., Michaletz, S. T., Winter, K. (2021). Leaf heat tolerance of 147 tropical forest species varies with elevation and leaf functional traits, but not with phylogeny. Plant Cell Environ. 44, 2414–2427. doi: 10.1111/pce.14060
Tang, Y., Harpenslager, S. F., van Kempen, M. M. L. (2017). Aquatic macrophytes can be used for wastewater polishing but not for purification in constructed wetlands. Biogeosciences 14, 755–766. doi: 10.5194/bg-14-755-2017
Thompson, K., Gilbert, F. (2014). Phenological synchrony between a plant and a specialised herbivore. Basic Appl. Ecol. 15, 353–361. doi: 10.1016/j.baae.2014.05.003
Titus, J. E., Gary Sullivan, P. (2001). Heterophylly in the yellow waterlily, Nuphar variegata (Nymphaeaceae): effects of [CO2], natural sediment type, and water depth. Am. J. Bot. 88, 1469–1478. doi: 10.2307/3558455
Wang, J., Li, M., Yu, C., Fu, G. (2022). The change in environmental variables linked to climate change has a stronger effect on aboveground net primary productivity than does phenological change in alpine grasslands. Front. Plant Sci. 12. doi: 10.3389/fpls.2021.798633
Wang, S., Reid, M. C. (2020). Kinetics of nitrous oxide mass transfer from porewater into root aerenchyma of wetland plants. J. Environ. Qual. 4, 1–13. doi: 10.1002/jeq2.20162
Wang, Q., Wang, F. Y., Xiong, L. H. (2017). The role of wetland in water quality purification and maintenance of coastal reservoirs: a case of Qingcaosha Reservoir. J. Sustain. Urbanization Plann. Prog. 2, 16–18. doi: 10.26798/JSUPP.2017.02.00
Wright, I. J., Reich, P. B., Westoby, M., Ackerly, D. D., Baruch, Z., Bongers, F., et al. (2004). The worldwide leaf economics spectrum. Nature 428, 821–827. doi: 10.1038/nature02403
Xie, C., Li, J., Pan, F. (2018). Environmental factors influencing mucilage accumulation of the endangered Brasenia schreberi in China. Sci. Rep. 8, 17955. doi: 10.1038/s41598-018-36448-3
Yang, C., Zhang, X., Seago, J. L., Jr., Wang, Q. (2020). Anatomical and histochemical features of Brasenia schreberi (Cabombaceae) shoots. Flora 263, 151524. doi: 10.1016/j.flora.2019.151524
Yiotis, C., Petropoulou, Y., Manetas, Y. (2009). Evidence for light-independent and steeply decreasing PSII efficiency along twig depth in four tree species. Photosynthetica 47, 223–231. doi: 10.1007/s11099-009-0036-1
Zhang, G., Cui, K., Li, G. (2022). Stem small vascular bundles have greater accumulation and translocation of non-structural carbohydrates than large vascular bundles in rice. Physiologia Plantarum 174, e13695. doi: 10.1111/ppl.13695
Zhang, Y., Wang, L., Hu, Y. (2015). Water organic pollution and eutrophication influence soil microbial processes, increasing soil respiration of estuarine wetlands: site study in Jiuduansha wetland. PloS One 10, e0126951. doi: 10.1371/journal.pone.0126951
Zhang, L. H., Zeng, C. S., Tong, C. (2018). Effects of nitrogen addition and submergence on the growth, nutrients, and carbon sequestration characteristics of Cyperus malaccensis. Acta Ecologica Sin. 38, 4932–4941. doi: 10.5846/stxb201705030813
Zhao, Y., Sun, M., Guo, H. (2022). Responses of leaf hydraulic traits of Schoenoplectus tabernaemontani to increasing temperature and CO2 concentrations. Botanical Stud. 63, 2. doi: 10.1186/s40529-022-00331-2
Zhao, J., Yang, P., Lin, Y. (2023). The effect of underwater supplemental light on the growth of V. spinulosa Yan and the restoration process of water. Process Saf. Environ. Prot. 169, 328–336. doi: 10.1016/j.psep.2022.11.043
Keywords: aquatic plants, functional traits, sediment nutrients, water quality, ecological response
Citation: Wang T, Yang H, Chen H, Zhang W, Liu Z, Li Q and Sun M (2025) Growth of Brasenia schreberi requries good water quality and appropriate sediment nitrogen content. Front. Plant Sci. 16:1535395. doi: 10.3389/fpls.2025.1535395
Received: 10 January 2025; Accepted: 04 March 2025;
Published: 21 March 2025.
Edited by:
Majid Sharifi-Rad, Zabol University, IranReviewed by:
Kaleem Mehmood, University of Swat, PakistanCopyright © 2025 Wang, Yang, Chen, Zhang, Liu, Li and Sun. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mei Sun, c20wNTEwMjE1QDE2My5jb20=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.