- 1Department of Biology, Clarkson University, Potsdam, NY, United States
- 2National Institute of Biological Resources, Incheon, South Korea
- 3Department of Biotechnology, Sungshin Women’s University, Seoul, South Korea
The genus Hosta (Agavoideae and Asparagaceae) is one of the most popular landscaping and ornamental plants native to temperate East Asia. Their popularity has led to extensive hybridization to develop various cultivars. However, their long history of hybridization, cultivation, and selection has brought about taxonomic confusion in the Hosta species delimitation along with their indistinguishable morphology. Here, we conducted the first broad phylogenetic analyses of Hosta species based on the most comprehensive genomic data set to date. To do so, we captured 246 nuclear gene sequences and plastomes from 55 accessions of Korean Hosta species using the Hyb-Seq method. As a result, this study provides the following novel and significant findings: (1) phylogenetic analyses of the captured sequences retrieved six species of Hosta in South Korea compared to five to eleven species based on the previous studies, (2) their phylogenetic relationships suggested that the large genome size was ancestral and the diversification of Korean Hosta species was accompanied by decreases in genome sizes, (3) comparison between nuclear genes and plastome revealed several introgressive hybridization events between Hosta species, and (4) divergence times estimated here showed that Hosta diverged 35.59 million years ago, while Korean Hosta species rapidly diversified during the late Miocene. Last, we explored whether these genomic data could be used to infer the origin of cultivars. In summary, this study provides the most comprehensive genomic resources to be used in phylogenetic, population, and conservation studies of Hosta, as well as for unraveling the origin of many cultivars.
Introduction
The genus Hosta Tratt. is an economically important group in terms of ethnobotany and horticulture. Some Hosta species have been cultivated for medicinal purposes since they are known to produce phytochemicals having antioxidant and anti-inflammatory activities (Mimaki et al., 1997; Yang et al., 2017; Chu et al., 2019). Many Hosta species are also popular as landscaping and ornamental plants because of their astonishing foliage and shade tolerance. Thus, they have been extensively hybridized to develop various cultivars, resulting in about 6,000 cultivars in the world (The_American_Hosta_Society, 2020). However, their long history of hybridization, cultivation, and selection along with their indistinguishable morphologies has brought about taxonomic confusion in Hosta species delimitation. Therefore, as few as 23 species (Zonneveld and Van Iren, 2001) or as many as 40 species (Fujita, 1976; Schmid, 1991; Chen and Boufford, 2000; Tamura and Fujita, 2013) have been reported from temperate East Asia, i.e., China, Japan, South Korea, and Russia.
Taxonomically, Hosta belongs to the Agavoideae bimodal karyotype clade (ABK clade) of Asparagaceae and diverged at the crown node of the ABK clade during the Oligocene (∼20–28 million years ago, Ma) (McKain et al., 2012, 2016). However, since only one Hosta species was included in those studies, further analyses with more Hosta species are required to estimate more accurate divergence times for Hosta.
The most recent taxonomic revision of Hosta was done based on pollen viability and nuclear DNA content, by supplementing the previous work, and it proposed three subgenera: Hosta, Bryocles, and Giboshi (Zonneveld and Van Iren, 2001). Subgenus Hosta is composed of one species, H. plantaginea (Lam.) Asch., while Bryocles and Giboshi contain nine species from China and South Korea and 13 species from Japan and the Russian Far East, respectively. They further divided subgenera into several sections according to their geographical distribution and nuclear DNA contents. However, their treatment did not correspond to sections previously proposed (Maekawa, 1940; Schmid, 1991), which several lines of evidence supported, such as plastid DNA (Chung, 1991), isozymes (Chung et al., 1991a), pollen morphology (Chung and Jones, 1989), and morphological characters (Chung et al., 1991b). For example, Zonneveld and Van Iren (2001) placed H. clausa Nakai and H. ventricosa (Salisb.) Stearn in different sections, but these two species were considered to have descended from a common ancestor based on pollen morphology (Chung and Jones, 1989) and karyotypes (Kaneko, 1968). Thus, further investigation of the taxonomic treatment of Hosta is required.
In Korea, five to eleven species have been described based on morphological characteristics, such as rhizome, petiole color, inflorescence, and floral shape (Chung and Chung, 1988; Chung and Kim, 1991; Lee, 1996; Chung, 2007; Jo and Kim, 2017), and all of them belong to subgenus Bryocles (Zonneveld and Van Iren, 2001). However, the lack of distinct traits with taxonomic value makes it hard to reach a consensus regarding the entity of Hosta species. Moreover, continuous variation in morphological traits and frequent hybridization have been problematic with regards to species delimitation. For example, H. venusta F. Maek. has been recognized as a species endemic to Mt. Halla (Jeju Island, South Korea), with its entity confirmed by several studies (Chung, 1990; Chung et al., 1991a,b). Notably, it was inferred that H. venusta might have recently derived from H. minor (Baker) Nakai based on morphology and distribution patterns (Chung, 1990; Chung et al., 1991a,b). In addition, Sauve et al. (2005) differentiated these two species relying on ten random amplified polymorphic DNA (RAPD) markers. However, Jo and Kim (2017) treated H. venusta as a variety of H. minor based on its morphological characteristics, overlapping with those of H. minor. Thus, investigating on whether these two species are distinct requires more genomic data.
To date, Chung and colleagues have performed the most comprehensive work on Korean Hosta species (Chung, 1990; Chung et al., 1991b). Their studies on Hosta from 45 populations recognized six species based on morphometric multivariate analysis and distribution patterns. Based on gross morphology, H. yingeri S. B. Jones and H. capitata (Koidz.) Nakai were distinguished from all other taxa, while H. clausa was differentiated from the other three taxa by the shape of its rhizome. The remaining three species, H. minor, H. venusta, and H. jonesii M.G. Chung, were separated by the length of their floral lobes and leaf blades. Later, based on the variation of six isozymes, Chung et al. (1991a) confirmed four groups of species: (1) H. minor, H. venusta, and H. jonesii, (2) H. yingeri, (3) H. capitata, and (4) H. clausa. Their species delimitation was mainly based on the similarity of morphological traits and a small set of isozyme data; however, their evolutionary relationships were not inferred.
Recently, the phylogenetic relationships of the six Korean Hosta species were reconstructed based on complete plastomes (Lee et al., 2019). This study distinguished six species, relying on four genic and intergenic regions, but nucleotide diversity among these species was very low (an average of 0.07% from pairwise comparison). The phylogenetic analysis showed that three species, H. minor, H. venusta, and H. jonesii are closely related. Additionally, they found that H. yingeri formed a clade with H. jonesii, which conflicts with previous studies (Chung, 1990; Chung et al., 1991b). However, as their work included one plastome per species, population level sampling and sequence data from the nuclear genome are needed to evaluate phylogenetic relationships of Korean Hosta species and to investigate whether hybridization has occurred or not.
Recent progress in high-throughput sequencing (HTS) subsampling methods has enabled us to evaluate phylogenetic relationships of species much more efficiently, by increasing the number of nuclear genes that can easily and inexpensively be sequenced (McKain et al., 2018). Notably, Hyb-Seq, a modified target sequencing method, is an effective way to capture DNA sequences from many single-to-low-copy nuclear genes, as well as a way to obtain regions in high-copy number, i.e., organellar genomes (Weitemier et al., 2014). Since this organellar DNA can provide important information for reticulate evolution and introgression, having both nuclear and organellar DNA makes Hyb-Seq a very powerful approach.
Here, we used Hyb-Seq to capture 246 single-to-low-copy nuclear gene sequences and complete plastomes. We focused on Korean Hosta species to test whether these captured genomic resources possessed sufficient phylogenetic signal that could help unravel Hosta evolution. Therefore, we included 55 accessions representing six Korean Hosta species and two Chinese ones and aimed to answer the following questions: (1) What are the phylogenetic relationships for these Hosta species inferred from multiple accessions based on both nuclear and plastid sequences? (2) Are nuclear and plastid phylogenetic relationships congruent with each other? (3) When did Korean Hosta species diverge? Lastly, (4) Are these genomic data appropriate to infer the origin of extant cultivars? Considering many putative Hosta species are cultivars maintained through time via vegetative propagation, this study provides a cornerstone for the development of potential molecular markers to unravel the origin of said cultivars, as well as their entity, which can be of use in breeding programs.
Materials and Methods
RNA Extraction, Library Preparation, and Sequencing
To select single-to-low-copy nuclear genes for target capture probe design, we first profiled the leaf transcriptome of H. venusta. Total RNAs from leaf tissue were isolated, with the RNeasy Plant Mini Kit (Qiagen, Hilden, Germany), and treated with Ribo-Zero rRNA Removal kit (Plant leaf) (Illumina, San Diego, CA, United States). RNA was quantified with a Victor2 spectrophotometer (PerkinElmer, Shelton, CT, United States) using the Quant-iTTM RiboGreenTM RNA Assay Kit (Invitrogen, Carlsbad, CA, United States). A total of 4 ng RNA was used for library preparation with the TruSeq Stranded Total RNA Sample Preparation kit (Illumina, San Diego CA, United States) and an alternate fragmentation protocol (fragmentation at 94°C for 2 min) was followed to obtain insert sizes of 280 bp, as described in the manufacturer’s protocol. The obtained libraries were quantified using the Agilent DNA 1000 Kit (Agilent, Santa Clara, CA, United States) and the SsofastTM EvaGreen supermix (Bio-Rad Laboratories Inc., Hercules, CA, United States). A final library concentration of 3 nM was sequenced on an Illumina NextSeq 500 sequencer (Illumina, San Diego, CA, United States), with 150 paired-end sequencing (2 × 150 bp), at Macrogen (Seoul, South Korea). Reads generated from the transcriptome of H. venusta were assembled de novo using Trinity v2.2.0 (Grabherr et al., 2011) with default options.
Probe Design
To determine target genes and to design probes, we first assembled H. venusta transcripts using 29,222,496 filtered reads; fastq files with raw reads (R1 and R2) were deposited to the GenBank Sequence Read Archive (PRJNA673211). The assembled H. venusta transcriptome was compared to the proteomes of Arabidopsis thaliana, Populus trichocarpa, Oryza sativa, and Vitis vinifera, as references, using the MarkerMiner pipeline (ver. 1.0) (Chamala et al., 2015). As a result, 283 orthologous genes (494,539 bp of total exon length) were selected for probe design based on GC content (40∼60%), number of hits (≤3), and E-value (≤1E–28) from BLAST searches against the Asparagus genome (Harkess et al., 2017), which is the closest relative to Hosta among fully sequenced plant genomes. The comparison of our target genes with a commercially available universal gene capture enrichment panel, Angiosperm353, resulted in only 30 overlapping genes at both nucleotide and protein levels. The final 4,247 probes (each 120 bp long; an average of 15 probes per gene) were synthesized at Agilent Technologies (Santa Clara, CA, United States) with 3× tiling across target exons. These probes targeted 676 exons (171,365 bp) covering 283 genes (436 kb of coding regions) total.
DNA Extraction, Library Preparation, In-Solution Hybridization, and Sequencing
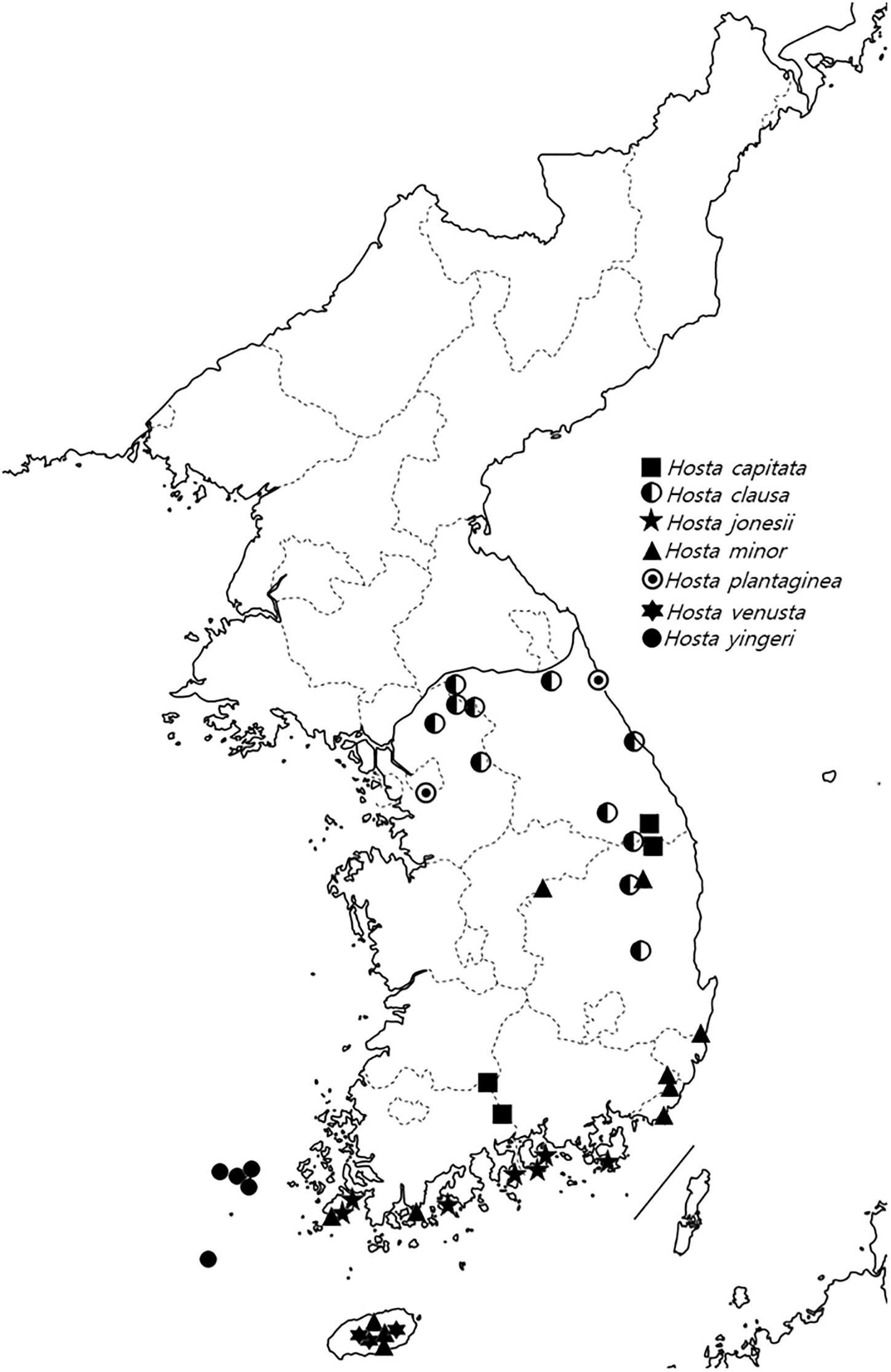
We sampled 64 accessions belonging to seven species of Hosta, including nine cultivars (Figure 1 and Supplementary Table 1). Voucher specimens for these samples were deposited at the herbarium of the National Institute of Biological Resources (NIBR), Incheon, South Korea. DNAs were extracted from either silica-gel-dried or herbarium material using a DNeasy Plant Mini Kit (Qiagen, Hilden, Germany) and were quantified using a Quant-iTTM PicoGreenTM dsDNA Assay Kit (Invitrogen, Carlsbad, CA, United States) with a SynergyTM LX Multi-Mode Microplate Reader (BioTek, Winooski, VT, United States). Libraries were prepared from a total of 50 ng of DNA using the TruSeq® Nano DNA Library Prep kit (Illumina, San Diego CA, United States) with 2 × 8 bp dual indexes to allow for multiplexed sequencing. After quantification, equimolar samples were pooled (16 libraires per reaction), hybridized with biotin-labeled baits to retrieve our targets using the probe kit described above. Captured DNA fragments were enriched in a 14–16 PCR cycle to generate the final libraries. A final concentration of 3 nM library was pooled and sequenced on an Illumina HiSeq 3000 (2 × 150 bp) (Illumina, San Diego, CA, United States) at RAPiD Genomics (Gainesville, FL, United States).
Contig Assembly and Multi-Sequence Alignment
Filtering of low-quality reads and adapter trimming were conducted by Skewer (v0.2.2) (Jiang et al., 2014) with options (mq = “30”, st = “3”, er = “0.1”, ml = “25”, a1 = “AGATCGGAAGAGCACACGTCTGAACTCCAGTCA”, a2 = “AGATCGGAAGAGCGTCGTGTAGGGAAAGAGTGT”). We employed HybPiper pipeline v.1.2 (Johnson et al., 2016) to process the trimmed data, using the BWA aligner (Li and Durbin, 2009) and the SPAdes v.3.10.1 assembler (Bankevich et al., 2012). Assembled contigs containing both on-target exons and off-target partial introns were combined into supercontigs. Potential paralogs flagged by the HybPiper paralog_investigator.py script were excluded from further analyses. Each supercontig was aligned using MAFFT v.7.402 (Katoh et al., 2002). Because the alignment included sample-specific indels, we removed them via Phyutility v.2.2.6 (Smith and Dunn, 2008) using -clean 0.7 option. Each cleaned supercontig file was concatenated, when necessary, according to sample ID. A total of 246 genes was used in downstream analyses.
Genome Skimming and Plastome Assembly
For plastome assembly, 64 genomic DNA libraries were sequenced on an Illumina HiSeq 3000 (2 X 150 bp), and the reads were trimmed as described above. Reconstruction of the plastome was performed using MITObim v.1.9.1 (Hahn et al., 2013). In short, the reads derived from a plastome were retrieved by mapping the trimmed reads onto the plastome sequence of H. minor (MK732316). Then, an initial reference for the species in question was built from the mapping result. The reads with overlap to this initial reference were fished, and these reads subsets were mapped to said initial reference. Thus, the reference extended iteratively by repeatedly fishing and mapping until all gaps were closed and the number of reads remained stationary. The assembled sequences were reevaluated by mapping the reads to previously published Hosta plastome sequences (Lee et al., 2019) using Geneious v.2019.2.31.
Sequence Diversity and Phylogenetic Inference
To estimate how much genetic diversity exists within and between Hosta species, we calculated nucleotide diversity (π) and genetic differentiation (Fst) among species using DnaSP v.6.12.03 (Rozas et al., 2017). Nuclear and plastid phylogenies of Hosta were inferred from the cleaned concatenated nuclear and plastome data matrices, respectively. Molecular evolution models were evaluated using ModelFinder (Kalyaanamoorthy et al., 2017), and maximum likelihood (ML) trees were inferred in IQ-TREE v.1.6.11 (Nguyen et al., 2015) with the standard nonparametric bootstrap method (-b 1000). Four plastome sequences of Yucca queretaroensis (NC_032713.1), Y. brevifolia (NC_032711.1), Agave attenuata (NC_032696.1), and Hesperoyucca whipplei (NC_032705.1) were used as outgroups in the plastome ML analysis, while the nuclear concatenated ML tree was rooted using two H. plantaginea species since they were inferred as sister to the remaining taxa in the plastome ML tree. We also inferred a species tree using the multispecies coalescent model, in which individual nuclear gene trees were constructed using RAxML v.8.2.12 under the GTRCAT substitution model and 200 bootstrap replicates by slow ML optimization (Stamatakis, 2014). Then ASTRAL-III v.5.6.2 (Zhang et al., 2018) was run to obtain a species tree, along with quartet support values that were visualized as pie charts relying on M. G. Johnson’s script2. We further evaluated gene conflict by measuring gene concordance factors (gCF) and site concordance factors (sCF) using IQ-TREE v.2.1.0 (Nguyen et al., 2015; Minh et al., 2020). It is thought that the plastome evolves as a single locus at a relatively constant rate, but we investigated whether this assumption holds for Hosta species by partitioning the plastome dataset into 130 genes and 117 intergenic areas to calculate gCF and sCF. Finally, the concatenated ML tree obtained from nuclear data was employed to reconstruct genome size ancestral states for Hosta species using the R package phytools v.0.7-70 under Brownian motion (Revell, 2012). The genome size data were obtained from Zonneveld and Van Iren (2001). Tree files, datasets and code used in R v.4.0.2 (R Core_Team, 2000) are available through the figshare data repository3.
Divergence Time Estimation
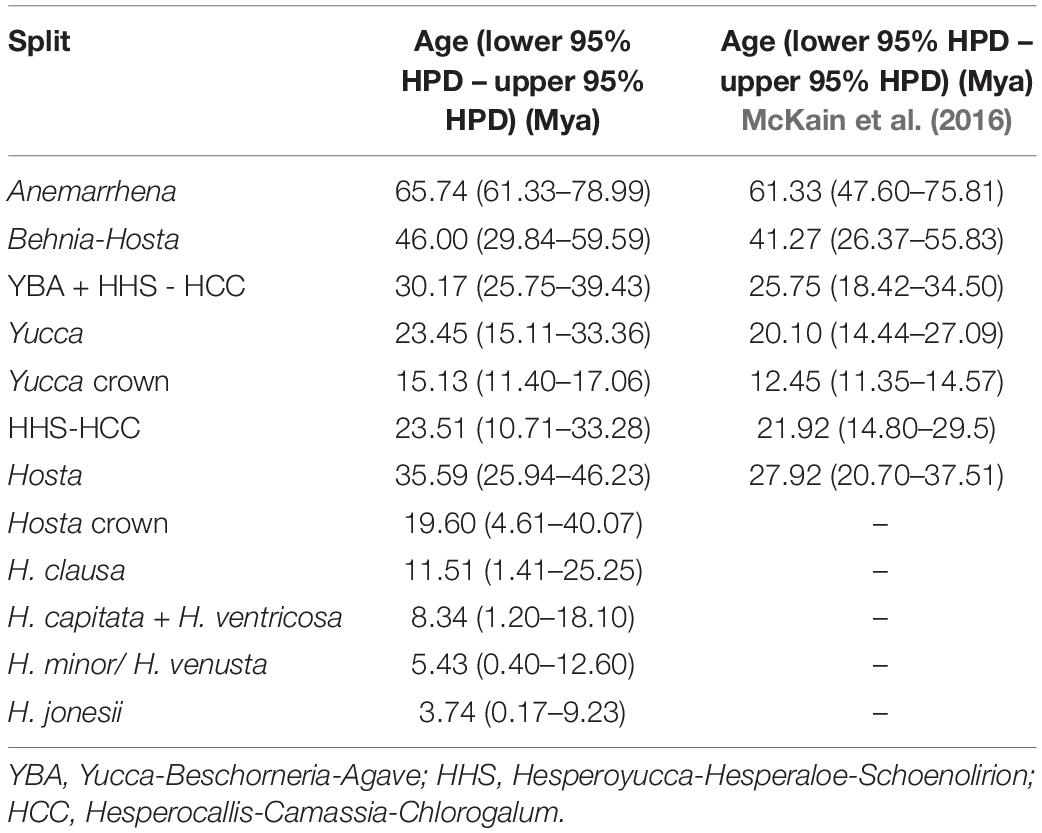
To estimate divergence times for Korean Hosta species, we only considered the plastome. Although nucleotide diversity was higher among nuclear genes, we were concerned rate heterogeneity among-sites and among-lineages, and recombination within and between loci, could severely impact divergence time estimates. Additionally, the 246 nuclear genes obtained here exhibited high among-gene incongruence (normalized quartet score = 0.61). Ideally, a wide range of outgroups with well-known fossils would be needed to calibrate our data matrices; however, those outgroups are missing from our nuclear dataset. Therefore, we employed the plastome which, in theory, acts as a single locus with no recombination and relatively homogenous evolutionary rate. A total of 79 protein-coding plastome genes (accD, atpA, atpB, atpE, atpF, atpH, atpI, ccsA, cemA, clpP, infA, matK, ndhA, ndhB, ndhC, ndhD, ndhE, ndhF, ndhG, ndhH, ndhI, ndhJ, ndhK, pbf1, petA, petB, petD, petG, petL, petN, psaA, psaB, psaC, psaI, psaJ, psbA, psbB, psbC, psbD, psbE, psbF, psbH, psbI, psbJ, psbK, psbL, psbM, psbT, psbZ, rbcL, rpl2, rpl14,rpl16,rpl20, rpl22, rpl23, rpl32, rpl33, rpl36, rpoA, rpoB, rpoC1, rpoC2, rps11, rps12, rps14, rps15, rps16, rps18, rps19, rps2, rps3, rps4, rps7, rps8, ycf1, ycf2, ycf3, and ycf4), commonly conserved in all taxa surveyed, were employed from 40 taxa of Asparagales, including 16 Hosta accessions (Supplementary Table 2). The aligned sequences contained 68,766 bp, and a ML phylogeny was inferred as described above. Since the monophyly of each subfamily and the genus Hosta was confirmed (Supplementary Figure 1), we constrained these clades for the BEAST 2 run. Divergence times were estimated using BEAST v.2.6.2 (Bouckaert et al., 2019) after generating BEAST input XML files using BEAUti v2.6.2. We followed the model parameters and the method used in McKain et al. (2016). In short, a GTRGAMMA evolutionary model was used with 10 gamma rate categories. A log-normal relaxed clock (Drummond et al., 2006) was the clock model, using the ML tree estimated above as the starting tree, and with speciation modeled by the Yule process. Age calibrations were implemented as follows: a log-normal distribution with a mean of 1.0, a standard deviation of 1.0, and an offset of 11.28 for the crown group of Yucca (Tidwell and Parker, 1990) and a log-normal distribution with a mean of 1.0, a standard deviation of 1.0, and an offset of 61.33 for Anemarrhena (McKain et al., 2016). We ran five separate runs for a total of 50 million generations, sampling every 1000. Convergence was assessed with Tracer v.1.7.1, and runs were continued to reach effective sample size (ESS) values of at least 200. Runs were combined using LogCombiner v.2.6.2 with a 10% burnin, and a maximum clade credibility (MCC) tree with mean node heights was created using a sample of 22,424 trees in TreeAnnotator v.2.6.2. The chronogram was drawn using FigTree v.1.4.34.
Results
Target Nuclear Dataset
De novo assembly of H. venusta transcripts resulted in 68,043 clustered unigenes, with a total of 1,013 transcripts as putative single-copy orthologs to four species. Among these, 283 transcripts, identified as putative orthologs, for at least three reference species, were selected as target genes. After target enrichment and sequencing, nine out of 64 accessions were excluded due to their low coverage (<5%). As for 55 accessions, an average of 4,982,380 reads were obtained per sample, and 74.5% of them were mapped to target genes (Supplementary Table 1). Average reads mapping on target equaled 680.67 (minimum = 325.19, maximum = 2688.39), and the general error rate (sequence divergence compared to the reference) was 2.84% on average (Supplementary Table 1). In agreement with using transcripts from H. venusta as the reference transcriptome, H. venusta (Hosven_15311) showed the least sequence divergence (2.40%), while H. yingeri (Hosyin_0616_1) exhibited the highest sequence divergence (3.32%) (Supplementary Table 1). These sequence divergences reflect their respective phylogenetic distances. For example, H. minor and H. venusta exhibited similar sequence divergences of 2.65% on average, while H. yingeri showed the highest degree of sequence divergence among seven species, supporting its isolation from the other species. We analyzed potential paralogs to evaluate their effect on phylogenetic inference, and we observed that relationships inferred were not congruent. Since high discordance was observed among ortholog genes, probably due to a rapid radiation in Hosta species (see below), we excluded paralogs from further analyses. After the data clean-up and the exclusion of paralogs, a total of 246 nuclear genes (489,796 bp) were included in the final alignment. The length of assembled sequences ranges from 359,467 bp in H. minor (Hosmin_15305_3) to 480,309 bp in H. clausa (Hoscla13), with an average of 3.67% gaps in the final alignment (Supplementary Table 1). When recovered sequences were parsed into exons and flanking regions, an average of 223 kb corresponded to exon regions, equal to 130% of target exons (171 kb, meaning their length was greater than expected) and 51% of target genes (436 kb of coding regions). Also, our designed probe set captured an additional average of 211 kb of flanking non-coding regions, including introns and 5′ or 3′-untranslated regions (UTRs). The final alignment was deposited to the figshare data repository (see text footnote 3).
Plastome Assembly
The genome skimming resulted in an average of 2,521,082 filtered reads per sample. An average of 2.25% filtered reads was mapped to their own assembled references, ranging from 0.20% in H. yingeri (Hosyin_16285) to 7.19% in H. clausa (Hoscla_16) (Supplementary Table 1). The size of assembled plastomes ranges from 154,648 bp in H. yingeri (Hosyin_16285; 98.7% of the complete plastome) to 156,756 bp in H. yingeri (Hoysin_s_n; 100% of the complete plastome). Previously reported sequence variation in the intergenic region between trnK-UUU and trnQ-UUG (Lee et al., 2019) was observed; that is, a 278 bp-deletion was found in all H. capitata species investigated here, except for a single cultivar (Hoscap72). Compared to four outgroup taxa, plastome sequences of Hosta species (156,455 bp on average) were shorter (157,776 bp on average), due to the loss of exon 2 of the rps16 gene, as previously known from H. ventricosa (Steele et al., 2012; McKain et al., 2016). The final alignment (159,590 bp) was also stored in the figshare data repository (see text footnote 3).
Sequence Divergence and Genetic Differentiation
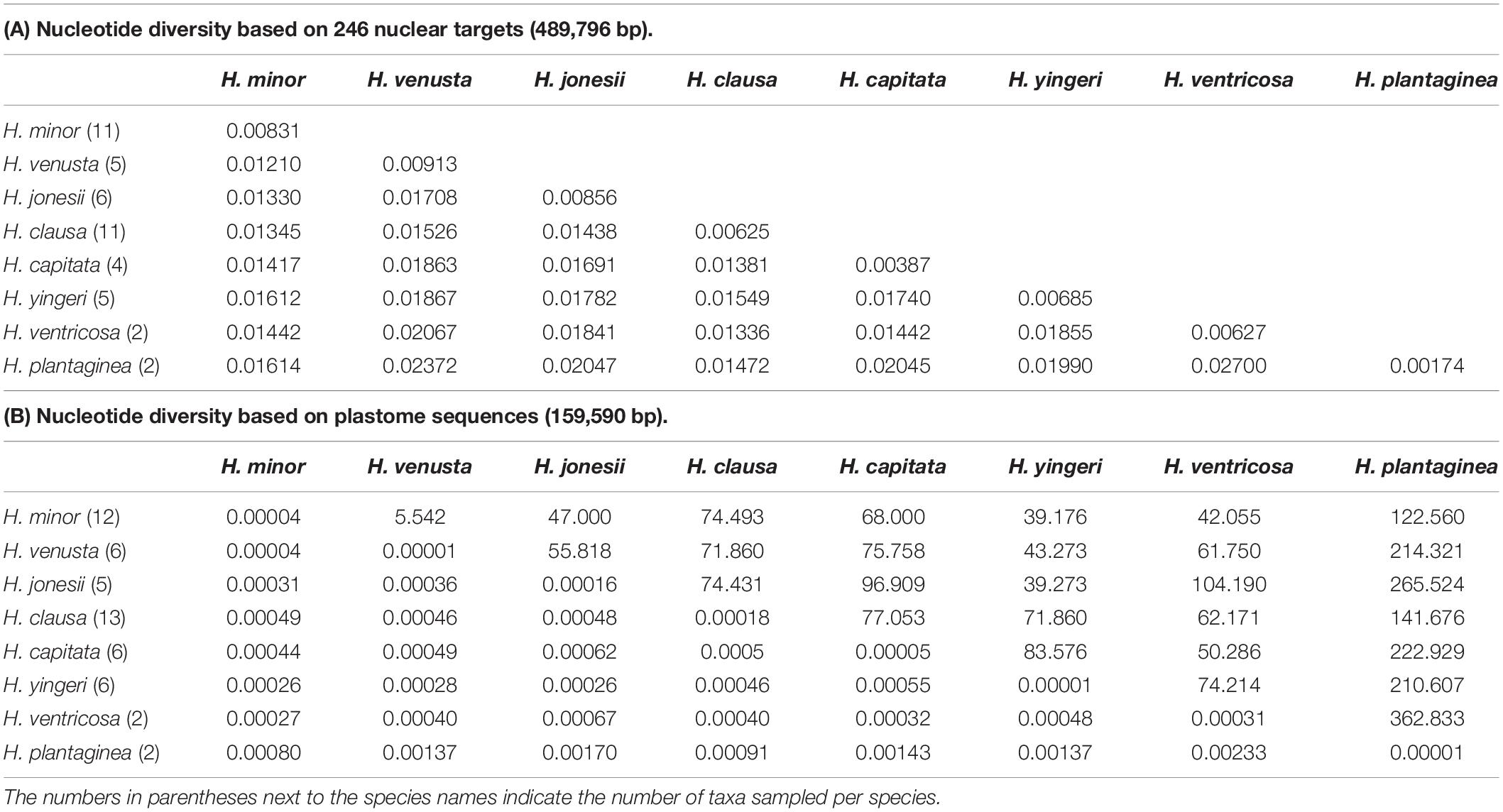
We examined sequence divergence by calculating nucleotide diversity (π) within and among species groups. In general, the nucleotide diversities of nuclear transcripts were much higher than those of plastome sequences (Table 1). The nucleotide diversity for each species ranges from 0.17% (H. clausa) to 0.91% (H. venusta) across the 246 nuclear target genes, while it ranges from 0% (H. plantaginea) to 0.02% (H. clausa) for the plastome sequences (Table 1). Among species, the nucleotide diversity between H. plantaginea and H. ventricosa showed the highest value in both nuclear DNA (2.7%) and plastid data (0.23%) (Table 1). Meanwhile, the nucleotide diversity between H. minor and H. venusta was 1.2% and 0.004% for the nuclear and plastid data, respectively, but H. venusta accessions were embedded within the H. minor clade in both ML trees (see below).
We also investigated genetic differentiation among species. In general, estimated Fst values were higher for plastome sequences than for nuclear transcripts (Table 2). Most species had high Fst values (> 0.25), indicating significant genetic differentiation between species. However, H. minor and H. venusta accessions showed little genetic differentiation (plastome Fst = 0.2123, nuclear Fst = 0.0686) (Table 2).
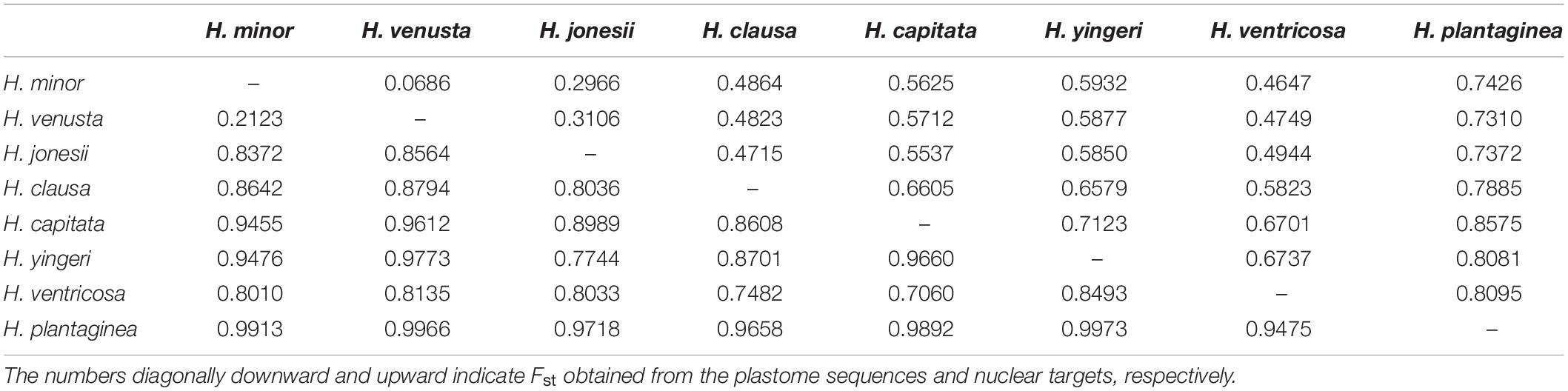
Table 2. Genetic differentiation (Fst) between Hosta species based on 246 nuclear targets and plastome sequences.
Phylogenetic Inference
We investigated phylogenetic relationships among seven Korean Hosta species using 246 single-copy nuclear orthologs and the whole plastome. The concatenated nuclear alignment included 54,754 parsimony informative sites (PIS, out of 489,796 bp, i.e., 11.2%), while the plastome sequence alignment contained 1,851 PIS (out of 159,590 bp, i.e., 1.2%).
Maximum likelihood (ML) and ASTRAL trees from both nuclear and plastome sequences showed congruence in terms of retrieving seven putative species: (1) H. minor/H. venusta, (2) H. jonesii, (3) H. clausa, (4) H. capitata, (5) H. ventricosa, (6) H. yingeri, and (7) H. plantaginea; all of them received maximum bootstrap support (BS) (Figure 2 and Supplementary Figures 2, 3). Although BS is 100% and local posterior probability (LPP) is 1 for these clades, underlying gene tree conflict seems to be extensive, for example, for the H. jonesii clade gCF is 8.2%, with 49.2% sCF, in the nuclear tree (Figure 2A). These values show that only 8.2% of 246 gene trees and 49.2% of phyloinformative alignment sites support this clade. In the ASTRAL tree, only 21 of 246 gene trees showed concordance for the same clade (Supplementary Figure 3), while gCF obtained from the plastome sequences was 9.3%, while sCF was 96.8% (Figure 2B).
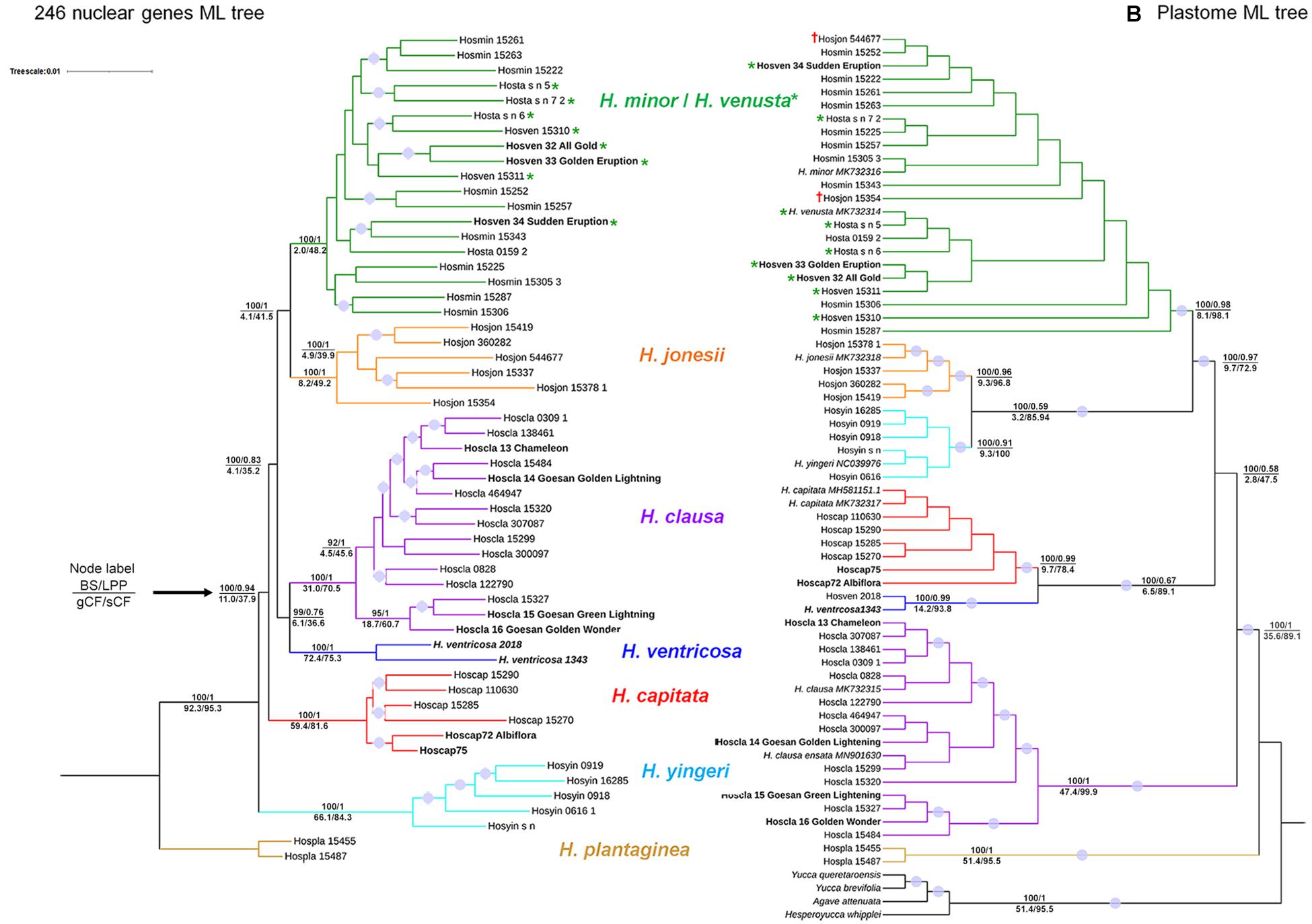
Figure 2. Comparison of maximum likelihood trees based on (A) nuclear DNA data matrix (246 loci, 489,796 bp) with a model of GTR + F + R3 and (B) plastome sequence matrix (159,590 bp) with a model of K3Pu + F + R2. The plastome tree has very short branch lengths for most clades (Supplementary Figure 2), so a cladogram was presented here. In both trees, the upper numbers on the node represent bootstrap support (BS) from 1,000 replicates for the concatenated dataset with IQ-TREE and local posterior probability (LPP) from the ASTRAL species tree, while the lower numbers indicate gene concordance factor (gCF) and site concordance factor (sCF) values. The values were not shown for branches in conflict between IQ-TREE and ASTRAL-III trees. In both trees, purple dots indicate ≥ 95 bootstrap support (BS). Bold taxa show cultivated species, while green asterisks indicate H. venusta. † shows H. jonesii accession which has the H. minor-like plastome.
In the plastome ML tree, H. plantaginea was inferred to be sister to the remaining species, as supported by 51% gCF and 95.5% sCF, along with 100% BS and 1 LPP (Figure 2B and Supplementary Figure 2). Branches subtending both H. plantaginea and the clade composed of the remaining Hosta species are notably long (Supplementary Figure 2). The sister relationship between H. plantaginea and the others was also observed in the nuclear ML tree and was overall highly supported (Figure 2).
Two species, H. minor and H. venusta, were not reciprocally monophyletic. In the nuclear ML tree, seven of eight accessions of H. venusta formed two clades nested within a H. minor clade, while the remaining accession (Hosven_34) was sister to a different H. minor accession (Hosmin_15343). In the plastome ML tree, H. venusta accessions were interspersed along a H. minor clade (Figure 2). This clade was defined by a high degree of gene tree discordance, for example, only 6 out of 246 gene trees support their grouping (Supplementary Figure 3).
These two ML trees showed other disparities. For example, H. yingeri was sister to the remaining species, other than H. plantaginea, in the nuclear ML tree, while this same species was sister to a H. jonesii clade in the plastome ML tree (Figure 2). Also, two H. jonesii accessions (Hosjon_544677, Hosjon_15354) were nested in the H. minor/H. venusta clade in the plastome ML tree (Figure 2). These accessions formed a clade with H. minor/H. venusta in some trees obtained from the partitioned analysis of nuclear data (Supplementary Figure 4). In addition, three species clades, H. clausa, H. capitata, and H. ventricosa, exhibited topological differences between the nuclear and plastome trees. In the nuclear tree, H. capitata was sister to all other species, except for H. yingeri and H. plantaginea, while H. clausa was sister to H. ventricosa, and both were sister to the clade including H. minor, H. venusta, and H. jonesii. In the plastome tree, however, a clade of H. clausa species was sister to all other species except for H. plantaginea, while H. capitata was sister to H. ventricosa (Figure 2). These overall disparities can be explained by incongruence, as shown by the low values of gCF (Figure 2) and quartet support values for the clades (Supplementary Figure 3), as well as by the low normalized quartet score (NQS) values for the nuclear genes (0.61) and the plastome sequences (0.43). However, the NQS value for the plastome sequences was much lower than the one estimated by Gonçalves et al. (2019) for 89 rosid species (0.8), probably due to the lack of phylogenetic signal resulting from the low genetic diversity (Table 1).
Using the tree inferred from nuclear DNA data, we reconstructed the genome size ancestral state in Hosta. The result points to large genome size (19.9 ± 1.1 pg) as the ancestral state for the lineage leading to Korean Hosta species that have smaller genome sizes, while the H. plantaginea clade seems to have experienced an increase in its genome size sometime during its evolutionary history (Figure 3).
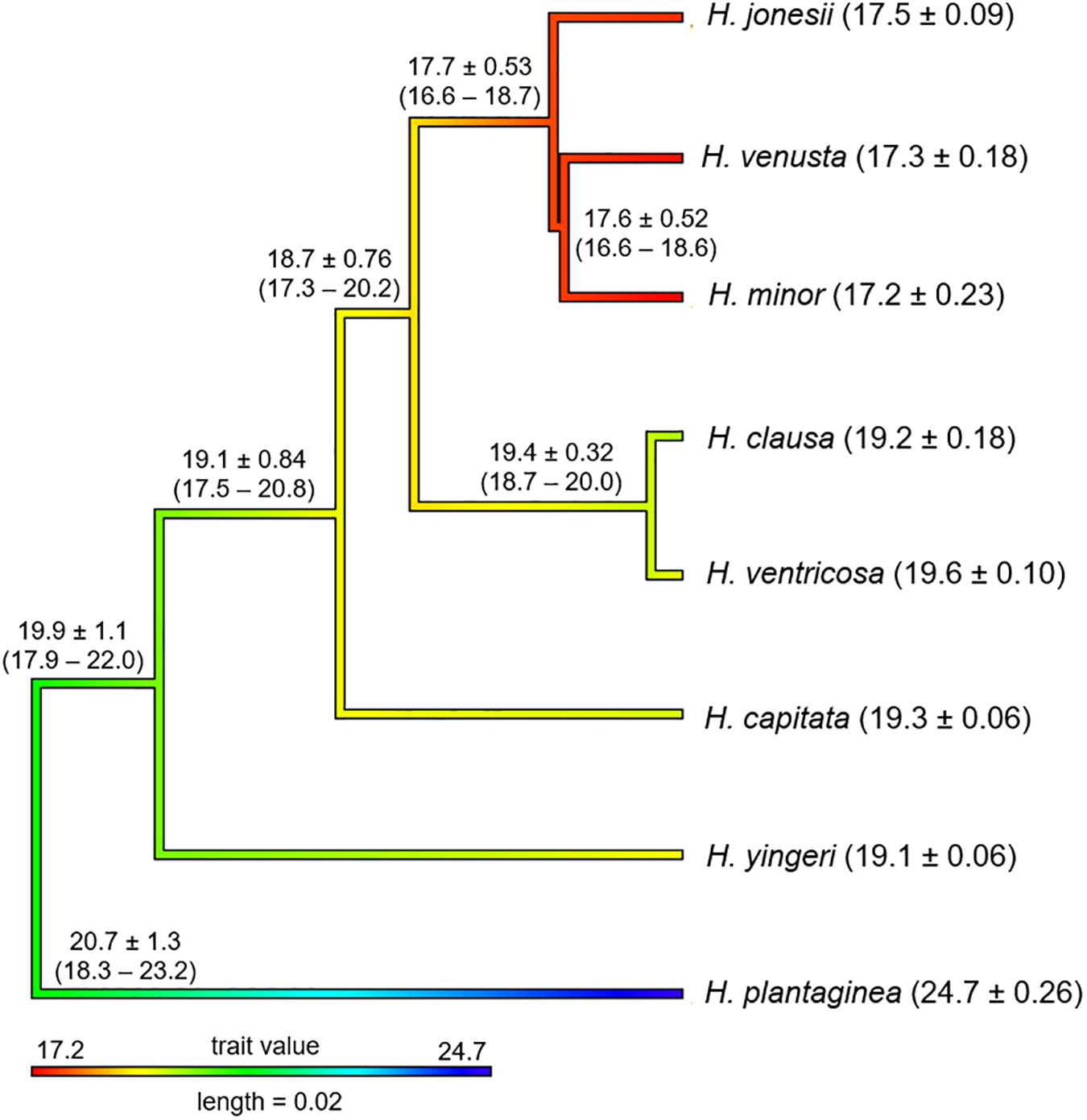
Figure 3. Ancestral character reconstruction of genome size evolution along the branches and nodes of the tree obtained from 246 nuclear target genes. The numbers in parentheses next to the species names show nuclear DNA content (2C) (average ± standard deviation) from Zonneveld and Van Iren (2001). The 2C value was reduced to half in H. ventricosa (2n = 4x = 120). The numbers near the branches and nodes represent the ancestral character estimates ± standard deviation with lower and upper 95% of confidence intervals.
To evaluate the application of the genomic resources generated here for inferring the origin of cultivars, we included several cultivar accessions. Since our sampling is limited to Hosta species in South Korea, we utilized nine cultivars for which their parentage is known. All these cultivars were clustered with their parental species, for example, cultivar “Chameleon” (Hoscla_13) was nested in the H. clausa clade, while cultivar “Albiflora” (Hoscap72) was nested in the H. capitata clade (Figure 2), their known respective parentals.
Divergence Time Estimates
To estimate divergence times for Korean Hosta species, we used 79 plastome protein-coding genes from 40 Asparagales taxa relying on two calibration points, and the purported ages of Yucca and Anemarrhena. Estimated divergence times for lineage splits were different from those of McKain et al. (2016). We inferred the stem age of Hosta at 35.59 Ma (late Eocene) (95% highest posterior density (HPD) interval: 25.94–46.23 Ma) (Figure 4 and Table 3), while McKain et al. (2016) estimated this same stem age at 27.92 Ma (middle Oligocene) (95% HPD interval: 37.51–20.70 Ma). Here, we first report a divergence time estimate for the Hosta crown group and the Korean Hosta species. The Hosta crown group diverged 19.60 Ma (early Miocene), while the Korean species diverged during the late Miocene (11.51 Ma for H. clausa, 3.74 Ma for H. jonesii) (Figure 4 and Table 3).
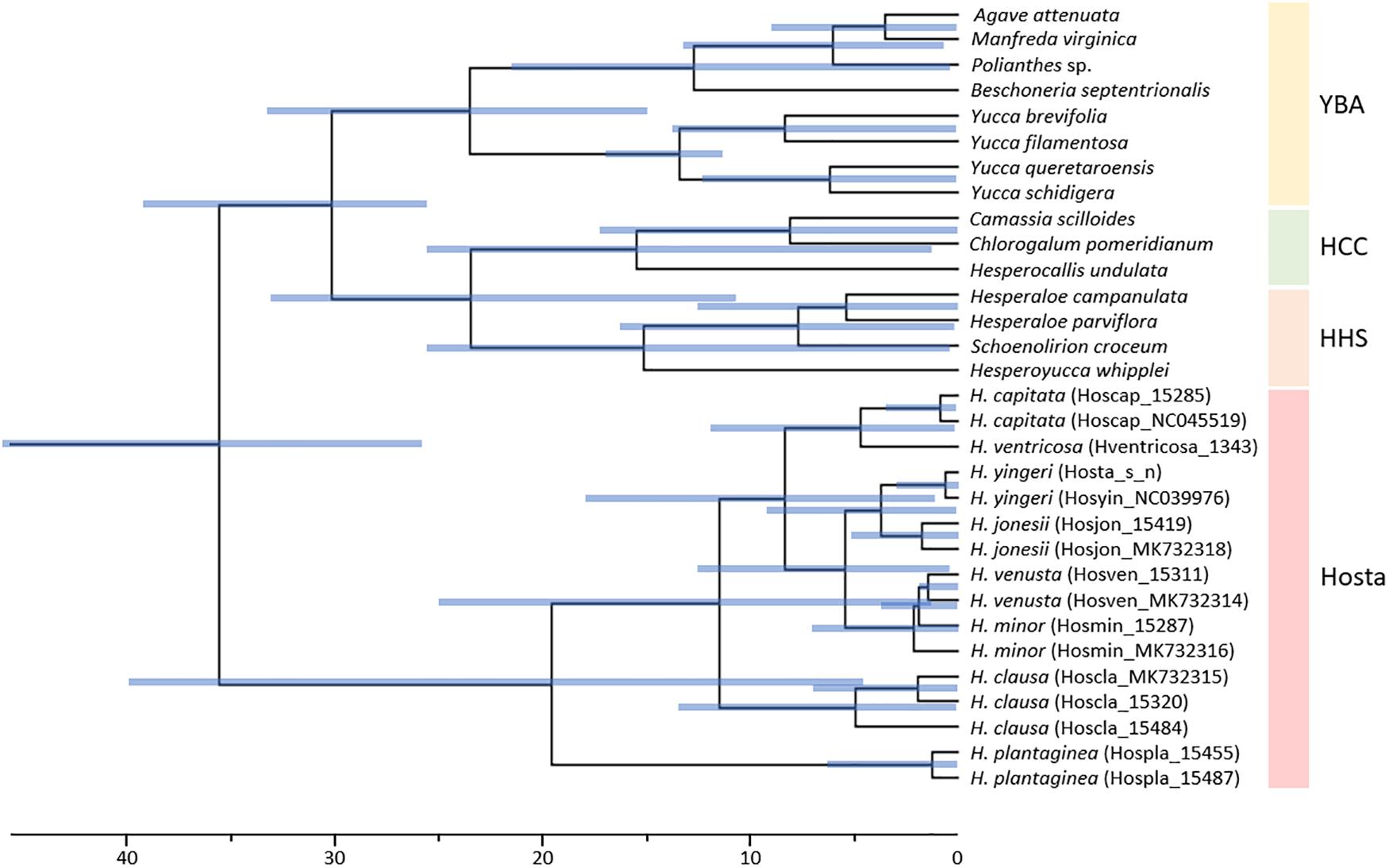
Figure 4. Divergence time estimate for Korean Hosta species inferred from 79 protein-coding plastome genes. Blue bars represent 95% highest posterior density (HPD) intervals. YBA, Yucca-Beschorneria-Agave; HHS, Hesperoyucca-Hesperaloe-Schoenolirion; HCC, Hesperocallis-Camassia-Chlorogalum.
Discussion
Species Entity and Phylogenomic Relationships of Korean Hosta Species
Previously, many researchers have proposed evolutionary relationships for Korean Hosta species, but those were based on limited information. Recently, Lee et al. (2019) employed whole plastome sequences, but only included an individual per species in their analysis. Therefore, this study provides the first instance of phylogenetic inference based on comprehensive data for most genomic compartments: 246 single-to-low-copy nuclear genes and whole plastome sequences from 55 accessions of Korean Hosta species.
In both nuclear and plastome ML trees, H. plantaginea was sister to or distinctly separated from the remaining Hosta species, respectively (Figure 2 and Supplementary Figure 2). This separation agrees with its geographic origin (China) and its unique floral characteristics. Hosta plantaginea is the only species which blooms at night, has a fragrance, and possesses the largest flower among Hosta species with white perianths and yellow anthers. The genomic distinctness of H. plantaginea was shown earlier based on RAPD analysis (Sauve et al., 2005) and by means of having the largest genome size among this seven-species group (Zonneveld and Van Iren, 2001). Therefore, the placement of H. plantaginea in its own subgenus (i.e., Hosta) seems to be reasonable (Maekawa, 1940; Fujita, 1976).
The remaining species belong to subgenus Bryocles, among which either H. yingeri or H. clausa may be sister to all other taxa (Figure 2). Both ML trees retrieved six species in South Korea, but they were not all monophyletic due to the inclusion of H. ventricosa. Unlike other Hosta species, H. ventricosa is a tetraploid species, native to southern China, but it has been widely cultivated in China and western countries as a medicinal plant and as an ornamental, respectively (Mimaki et al., 1997; Yang et al., 2017; Chu et al., 2019). Previous studies suggested that H. ventricosa might have shared a common ancestor with H. clausa during their evolutionary history based on pollen morphology (Chung and Jones, 1989), DNA content (Zonneveld and Van Iren, 2001), and chromosome shape (Kaneko, 1968). This inference was supported in the nuclear ML tree, but not in the plastome ML tree which showed that H. ventricosa and H. capitata might have shared their maternal parent (Figure 2B). Since only two cultivated H. ventricosa accessions were included in this study, more samples of H. ventricosa, both natural and cultivated, should be included in future phylogenetic analysis to disentangle relationships among these three species.
In the nuclear ML tree, H. yingeri was sister to all other Korean taxa although its position was not fully supported. In contrast to the high BS and LPP support values, gCF and sCF values showed high incongruence among gene trees for its placing (Figure 2A). Hosta yingeri is endemic to Heuksan Island in South Korea, which is 97 km from the mainland, and is easily distinguishable from other Hosta species based on its floral traits, for example, a funnel-shaped flower with distinct stamens (3 + 3) and persistent bract at flowering (Jo and Kim, 2017). However, in the plastome ML tree, all five accessions of H. yingeri were sister to H. jonesii, and the genetic diversity between these two species was very low (0.026%; Table 1). This result indicates these two species might have shared their maternal parent, or that H. yingeri might have captured the plastome of H. jonesii through introgressive hybridization. Based on the divergence times estimated, H. jonesii and H. yingeri might share a most recent common ancestor (MRCA) 6.59 Ma, which corresponds to the late Miocene (Table 3). Also, considering their distribution (Figure 1), they might previously have had close contact. Therefore, the isolation of H. yingeri at Heuksan Island for a long period might have facilitated its morphological and nuclear differentiation. The hybridization experiments between H. yingeri and other Korean Hosta species resulted in less than 60% of pollen viability (Zonneveld and Van Iren, 2001), supporting its reproductive isolation along with its unique floral morphological features. Reproductive isolation is commonly found in island species (reviewed in Crawford and Archibald, 2017), which often exhibit incongruence between nuclear and plastome markers (French et al., 2016; Hughes et al., 2018; Waselkov et al., 2018; Thureborn et al., 2019). In addition, its genome size is much more similar to that of H. capitata and H. clausa, when compared to H. jonesii (Zonneveld and Van Iren, 2001), in agreement with their evolutionary relationships, as shown in the nuclear tree (Figure 4). Therefore, along with its unique floral characteristics, H. yingeri’s geographical isolation and its genome size support its position sister to all other Korean Hosta, rather than a sister relationship to H. jonesii.
The position of the H. clausa and H. capitata clades showed disparity between our two trees (Figure 2), as stated above. In the nuclear tree, H. capitata was sister to the remaining four species, while H. clausa was sister to these four species in the plastome tree (Figure 2). These two species are morphologically distinct. For instance, H. capitata and H. clausa can be distinguished because the former has a head-like inflorescence and short creeping rhizome, while the latter has a spike-like inflorescence and rhizome with underground stolon. Observed gene tree conflicts in both nuclear and plastome trees suggests either a lack of phylogenetic signal or incomplete lineage sorting (ILS) in Korean Hosta species may be at work. Their ambiguous phylogenetic relationships could thus be explained in terms of their rapid diversification during a relatively short period, as implied by the short branches in both trees and the overlapping divergence times estimated (Figures 2, 4).
The last three species, H. jonesii, H. minor, and H. venusta, formed a clade and their close relationship has been suggested in previous studies (Chung, 1991; Chung et al., 1991a,b). These three species have similar genome size, of about 17.5 pg (Zonneveld and Van Iren, 2001), and their interspecific nucleotide diversity was inferred to be very low (Table 1). Indeed, their low genetic diversity implies close relatedness and shared evolutionary histories, which are further supported by their morphological traits, such as ovate-to-lanceolate leaf blade, bract state at flowering, fruit color, and intermediate size of pistil and anthers (Jo and Kim, 2017). Especially, H. venusta accessions failed to form a clade, but were instead nested in the H. minor clade (Figure 2). The monophyletic group containing both H. minor and H. venusta was supported by only six gene trees out of 246 (Supplementary Figure 3), suggesting a high degree of underlying gene tree conflict or a lack of phylogenetic signal for the nuclear dataset. The latter could be the case, considering the genetic diversity between these two purported species (0.94% in the nuclear data, 0.004% in the plastome data) (Table 1). The genetic differentiation between these two purported species was also extremely low, 0.0868 and 0.2123 in nuclear and plastome datasets, respectively (Table 2). These values are much lower than those for other species, for example, the genetic differentiation between H. jonesii and H. minor was three to four times higher, compared to that between H. venusta and H. minor (0.2934 in nuclear data and 0.8372 in plastome data; Table 2). Thus, H. minor and H. venusta may be the same species, with a Fst < 0.25, and given the threshold previously estimated for species and populations from 97 studies of pairs of closely related species and populations (Hey and Pinho, 2012). Consequently, H. venusta, endemic to Jeju Island, should be recognized as a variety of H. minor, since they also present overlapping morphological characteristics (Jo and Kim, 2017), along with their low genetic diversity as shown here. However, there seems to be clinal variation in H. venusta along altitudes in Mt. Halla, Jeju Island (C. E. Lim, pers. comm.). Therefore, further analysis with more accessions of H. minor and H. venusta species from Jeju Island should be carried out to determine its taxonomic rank. Regardless of the species entity of H. venusta, the dispersal of H. minor from mainland Korea to Jeju Island might trace back to as early as 2 Ma when Jeju island was formed through volcanism (Woo et al., 2013).
Hosta jonesii occurs in Korea’s southern islands and its close relationship to H. minor was proposed in previous studies (Chung, 1991; Chung et al., 1991a,b). This study also supports their close phylogenetic relationship, but two accessions of H. jonesii were found to contain the plastome of H. minor (Hosjon_544677, Hosjon_15354) (Figure 2). Since all accession of H. jonesii belong to one clade in the nuclear ML tree, this cytonuclear discordance between the nuclear and plastome ML trees might be the result of introgressive hybridization between H. minor and H. jonesii. We observed these two accessions formed a clade with H. minor/H. venusta in some trees, indeed supporting a possible introgression (Supplementary Figure 4). These two species share their geographical distribution (Figure 1), thus, their close contact might have enabled their hybridization.
Hosta is well known for extensive hybridization due to its cultivar development, but no previous study has managed to detect hybridization events in nature. Earlier studies on the Korean Hosta species also failed to detect hybridization, since they examined either nuclear or plastid markers, focusing only on a small portion of the genome. Considering the low genetic diversities among Hosta species, the lack of information might be the main barrier to uncover hybridization events. In this study, we were able to reveal several hybridization events based on 246 nuclear coding genes and plastome sequences. Thus, this study suggests that a more genome-wide search is required for species groups with low genetic diversity to detect any hybridization or introgression events.
Taxonomic Implications and Genome Size Evolution in Korean Hosta Species
A well-resolved phylogeny provides a backbone for investigating various biological aspects, such as taxonomic treatment and the evolution of morphological traits and genome sizes for a given lineage of interest. As for the taxonomic implications, this work showed that all six Korean species and H. ventricosa belong to subgenus Bryocles, since they form a monophyletic group. However, their placement into sections shows some disparities compared to previous treatments. Hosta ventricosa was assigned to section Bryocles due to its tetraploid status (Maekawa, 1937), while the rest of species are diploid. However, in our trees H. ventricosa forms a clade with H. clausa, which is a member of section Stolonifera, along with H. yingeri and H. capitata. Additionally, section Stolonifera was not supported by this study, as its three species were found to be paraphyletic (Figure 2). Based on the phylogeny obtained here, we can recognize three sections that consist of H. yingeri only, H. capitata only, and both H. clausa and H. ventricosa, respectively. The remaining three species of H. minor/H. venusta and H. jonesii were previously included in section Lamellatae (Zonneveld and Van Iren, 2001), in agreement with our reconstruction. However, the other two Japanese species, H. tibae and H. tsushimensis, should be examined to support the legitimacy of section Lamellatae. In addition, the legitimacy of subgenus Bryocles should also be evaluated by including Japanese Hosta species, most of which are thought to belong to subgenus Giboshi (Zonneveld and Van Iren, 2001).
Previously, it was proposed that the ancestral Hosta nuclear DNA content might be close to either 17 or 19 pg, and the speciation of Hosta might have been accompanied by increases in DNA content in dry habitats and by decreases in wet environments (Zonneveld and Van Iren, 2001). However, this idea contradicts recent works showing increased genome sizes could be associated with early flowering and a tendency to grow in wet environments (Knight et al., 2005; Vesely et al., 2012). Also, under nutrient-limited conditions species with large genomes would be less competitive than species with small genomes, as they would need more resources to maintain their large genomes (Smarda et al., 2013; Guignard et al., 2016). The ancestral state estimation for the genome size, given our Hosta species tree, shows that the MRCA of H. plantaginea and the remaining species might have had a large genome size (19.9 ± 1.1 pg), supporting Korean Hosta species may have diversified by reducing their DNA content (Figure 3). For example, H. yingeri, which is sister to all the remaining Korean Hosta species and H. ventricosa, has 19 pg of 2C value, while H. minor, which is furthest from the root, contains 17 pg of 2C value (Zonneveld and Van Iren, 2001). Although there is a trend of DNA content decrease in Korean Hosta species, there were substantial overlaps in confidence intervals among species. This might be explained by their rapid diversification in similar environmental conditions in South Korea, but the relationship between genome size and their ecological and evolutionary constraints should be further tested. Therefore, the inclusion of Japanese Hosta species that occur in diverse conditions could shed light on the speculation of the effect of genome size on Hosta speciation. The better resolved tree with Japanese taxa should provide a backbone for more accurate inference on the genome size evolution in Hosta.
Divergence Time Estimates for the Hosta Crown Group and Korean Hosta Species
Previous analysis of 69 plastome protein-coding genes estimated that Hosta diverged at the crown node of the ABK clade during the Oligocene (20–27 Ma) (McKain et al., 2012, 2016). However, our analysis with more Hosta species estimated its divergence during the late Eocene (38.38 Ma). Since our estimates for other lineages are not much different from the previous ones (Table 3), analysis with more Hosta taxa and with more genes (i.e., 79 plastome protein-coding genes) might result in different estimates for Hosta. Here, we estimated divergence times for the Hosta crown group and for Korean Hosta species for the first time. Particularly, 95% HPD intervals were very wide for Hosta clades (Figure 4 and Table 3), which might be due to the low sequence divergence among Hosta species, as shown by their short branch lengths (Supplementary Figures 1, 2) and low nucleotide diversities (Table 1). Although our estimates were not narrowed down to specific time points, the estimated times for Hosta species suggest rapid diversification of Korean Hosta species over a short period (∼ 10 Ma) during the Miocene.
During the early Miocene, the Japanese Islands started their separation from the continent, and this process was completed during the middle Miocene and resulted in the formation of the East Sea (Yabe et al., 2019). The divergence time we estimated for crown Korean Hosta dates back to the late Miocene, when the Korean Peninsula and the Japanese Islands were completely separated by the East Sea. Thus, we hypothesize that limited gene flow between Korean and Japanese Hosta species might have accelerated the speciation of Hosta species in South Korea, without necessarily increasing their genetic diversity. However, the phylogeny of Hosta without Japanese species does not allow to assess whether Korean Hosta evolved from Chinese vs. Japanese taxa. The latter case would involve the migration of Hosta species from China to either southeastern Russia-to-Japan or Japan and then onto South Korea. Therefore, it is critical to include Japanese Hosta species, which have the highest species diversity of Hosta (ca. 20 species), as well as Russian accessions, to figure out the origin of the Korean species.
Application of Hyb-Seq Results for Identifying the Origin of Cultivar Species
Hosta species have very showy foliage and shade tolerance, so they have considerable economic value as landscaping and ornamental plants. Their popularity, in turn, has brought about a long history of cultivation and extensive hybridization, resulting in many extant cultivars. However, their origins were not clearly recorded in many cases. Here, we tried to evaluate whether 246 single-copy nuclear genes and complete plastome sequences were useful for inferring the origin of cultivars. In this study, we examined nine cultivars, two H. capitata, three H. venusta, and four H. clausa cultivars, for which their origins are known. All nine cultivars were clustered with their corresponding parent species, but our results suggest that, for a given species, cultivars could have been developed from different individuals from different geographic regions. For example, two H. clausa cultivars (Hoscla_15 and Hoscla_16) were close to Hoscla_15327 collected from southern South Korea, while the other two cultivars were clustered with H. clausa species occurring in northern South Korea (Figure 1). Also, the results for the H. venusta cultivars show the importance of a well-resolved phylogeny in inferring the origin of cultivars. Although two H. venusta cultivars (Hosven_32, Hosven_33) formed a clade sister to H. venusta (Hosven_15311), Hosven_34 was sister to H. minor (Figure 2), not H. venusta. Considering these two species may instead be one and the same (see above), this placement is not surprising. Therefore, a well-resolved and supported phylogeny is critical to infer the origin of cultivars as well as the provision of genomic resources. Although our work shows the usefulness of these 246 single-copy nuclear genes and complete plastome sequences for exploring the origins of cultivars, these genomic resources should be further tested with more cultivars of known and unknown origins. Then, an extensive investigation can lead to the development of molecular markers useful for inferring their origin, which could be employed for evaluating whether Hosta species currently growing in a garden originated from South Korea, Japan, China, or even Russia. Also, those molecular markers could be used for cataloging cultivars, which is critical for the conservation of natural species. Finally, Hosta breeding programs will benefit from their known parentage in developing new cultivars.
Conclusion
The Hyb-Seq method allowed us to capture several hundreds of low-to-single-copy nuclear genes as well as to obtain full plastomes. Due to low genetic diversities, commonly used nuclear and plastome markers have shown inconsistent results with low resolution in the phylogeny of Hosta. Here, the genomic data obtained with Hyb-Seq successfully helped identify Korean Hosta species and permitted inferring a well-resolved and supported phylogeny. In addition, we detected several putative introgressive hybridization events, never before found. However, ILS could not be ruled out due to the high incongruence observed among nuclear genes. This gene tree discordance might result from a lack of phylogenetic signal for Korean Hosta species following their rapid diversification during a short time interval. Last, we demonstrated the applicability of genomic data from Hyb-Seq for tracing the origin of cultivated Hosta species. In summary, this study provides the most comprehensive genomic resources to date for phylogenetic inference, the development of potential molecular markers for population and conservation studies of Hosta, as well as for unraveling the origin of many cultivars.
Data Availability Statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Author Contributions
CL designed the project. CL and B-YL conceived ideas and prepared funding and samples. SK oversaw the procedure of data generation. M-JY analyzed the data and wrote the manuscript. All authors have read and approved the manuscript.
Funding
This research was supported by National Institute of Biological Resources (NIBR), Ministry of Environment, South Korea (NIBR201922101 and NIBR202022101). NIBR provided the fund required for the project and evaluated the process of running fund and overall performance of the project.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We appreciate parataxonomists of the Society for Korean Peninsula Plants (SKPP) who helped to collect Hosta samples in South Korea. We thank Jung Hwa Kang and Wan Hee Lee at Hantaek Botanical Garden for their help with Hosta cultivar samples, as well as Suhyeon Park at Sungshin Women’s University for her support in data generation. We would like to express our gratitude to three reviewers for their helpful comments that have improved the quality of this manuscript.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2021.645735/full#supplementary-material
Footnotes
- ^ http://www.geneious.com
- ^ https://github.com/mossmatters/MJPythonNotebooks
- ^ https://doi.org/10.6084/m9.figshare.14043965
- ^ http://tree.bio.ed.ac.uk/software/figtree/
References
Bankevich, A., Nurk, S., Antipov, D., Gurevich, A. A., Dvorkin, M., Kulikov, A. S., et al. (2012). Spades: a new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 19, 455–477. doi: 10.1089/cmb.2012.0021
Bouckaert, R., Vaughan, T. G., Barido-Sottani, J., Duchene, S., Fourment, M., Gavryushkina, A., et al. (2019). BEAST 2.5: An advanced software platform for Bayesian evolutionary analysis. PLoS Comput. Biol. 15:e1006650. doi: 10.1371/journal.pcbi.1006650
Chamala, S., Garcia, N., Godden, G. T., Krishnakumar, V., Jordon-Thaden, I. E., De Smet, R., et al. (2015). MarkerMiner 1.0: A new application for phylogenetic marker development using angiosperm transcriptomes. Appl. Plant Sci. 3:1400115. doi: 10.3732/apps.1400115
Chen, X., and Boufford, D. E. (2000). “Hosta,” in Flora of China, eds Z. Y. Wu and P. H. Raven (Beijing: Science Press), 204–205.
Chu, H. B., Li, N. N., Zhang, Z. P., Hu, X. Y., Yu, C. Y., and Hua, L. (2019). Steroidal glycosides from the underground parts of Hosta ventricosa and their anti-inflammatory activities in mice. Nat. Prod. Res. 35, 1766–1774. doi: 10.1080/14786419.2019.1638379
Chung, M. G. (1990). A biosystematic study on the genus Hosta (Liliaceae/Funkiaceae) in Korea and Tsushima Island of Japan. PhD dissertation. Athens, GA: University of Georgia.
Chung, M. G. (1991). Notes on the phylogenetic relationships among species of korean hosta (Liliaceae) based on chloroplast DNA restriction site data. Plant Species Biol. 6, 71–74. doi: 10.1111/j.1442-1984.1991.tb00211.x
Chung, M. G., Hamrick, J. L., Jones, S. B., and Derda, G. S. (1991a). Isozyme variation within and among populations of hosta (Liliaceae) in Korea. Syst. Bot. 16, 667–684. doi: 10.2307/2418869
Chung, M. G., Jones, S. B., Hamrick, J. L., and Chung, G. G. (1991b). Morphometric and isozyme analysis of the genus Hosta (Liliaceae) in Korea. Plant Species Biol. 6, 55–69. doi: 10.1111/j.1442-1984.1991.tb00210.x
Chung, M. G., and Jones, S. B. (1989). Pollen morphology of hosta tratt. (Funkiaceae) and related genera. Bull. Torrey Bot. Club. 116, 31–44.
Chung, Y. C. (2007). “Hosta,” in The Genera of Vascular Plants of Korea, ed. C. W. Park (Seoul, Korea: Academy Publishing Co.), 1304– 1306.
Chung, Y. C., and Chung, Y. H. (1988). A taxonomic study of the genus Hosta in Korea. Korean J. Pl. Taxon. 18, 161–172. doi: 10.11110/kjpt.1988.18.2.161
Crawford, D. J., and Archibald, J. K. (2017). Island floras as model systems for studies of plant speciation: Prospects and challenges. J. Syst. Evol. 55, 1–15. doi: 10.1111/jse.12234
Drummond, A. J., Ho, S. Y., Phillips, M. J., and Rambaut, A. (2006). Relaxed phylogenetics and dating with confidence. PLoS Biol. 4:e88. doi: 10.1371/journal.pbio.0040088
French, P. A., Brown, G. K., and Bayly, M. J. (2016). Incongruent patterns of nuclear and chloroplast variation in Correa (Rutaceae): Introgression and biogeography in south-eastern Australia. Plant Syst. Evol. 302, 447–468. doi: 10.1007/s00606-016-1277-7
Gonçalves, D. J. P., Simpson, B. B., Ortiz, E. M., Shimizu, G. H., and Jansen, R. K. (2019). Incongruence between gene trees and species trees and phylogenetic signal variation in plastid genes. Mol Phylogen. Evol. 138, 219–232. doi: 10.1016/j.ympev.2019.05.022
Grabherr, M. G., Haas, B. J., Yassour, M., Levin, J. Z., Thompson, D. A., Amit, I., et al. (2011). Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 29, 644–652. doi: 10.1038/nbt.1883
Guignard, M. S., Nichols, R. A., Knell, R. J., Macdonald, A., Romila, C. A., Trimmer, M., et al. (2016). Genome size and ploidy influence angiosperm species’ biomass under nitrogen and phosphorus limitation. New Phytol. 210, 1195–1206. doi: 10.1111/nph.13881
Hahn, C., Bachmann, L., and Chevreux, B. (2013). Reconstructing mitochondrial genomes directly from genomic next-generation sequencing reads-a baiting and iterative mapping approach. Nucleic Acids Res. 41:e129. doi: 10.1093/nar/gkt371
Harkess, A., Zhou, J. S., Xu, C. Y., Bowers, J. E., Van Der Hulst, R., Ayyampalayam, S., et al. (2017). The asparagus genome sheds light on the origin and evolution of a young Y chromosome. Nat. Commun. 8:1279. doi: 10.1038/s41467-017-01064-8
Hey, J., and Pinho, C. (2012). Population genetics and objectivity in species diagnosis. Evolution 66, 1413–1429. doi: 10.1111/j.1558-5646.2011.01542.x
Hughes, M., Peng, C. I., Lin, C. W., Rubite, R. R., Blanc, P., and Chung, K. F. (2018). Chloroplast and nuclear DNA exchanges among Begonia sect. Baryandra species (Begoniaceae) from Palawan island, Philippines, and descriptions of five new species. PLoS One 13:e0194877. doi: 10.1371/journal.pone.0194877
Jiang, H., Lei, R., Ding, S. W., and Zhu, S. (2014). Skewer: A fast and accurate adapter trimmer for next-generation sequencing paired-end reads. BMC Bioinform. 15:182. doi: 10.1186/1471-2105-15-182
Jo, H., and Kim, M. (2017). A taxonomic study of the genus Hosta in Korea. Korean J. Pl. Taxon. 47, 27–45. doi: 10.11110/kjpt.2017.47.1.27
Johnson, M. G., Gardner, E. M., Liu, Y., Medina, R., Goffinet, B., Shaw, A. J., et al. (2016). HybPiper: Extracting coding sequence and introns for phylogenetics from high-throughput sequencing reads using target enrichment. Appl. Plant Sci. 4:1600016. doi: 10.3732/apps.1600016
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., Von Haeseler, A., and Jermiin, L. S. (2017). ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods 14, 587–589. doi: 10.1038/nmeth.4285
Kaneko, K. (1968). Cytological studies on some species of Hosta. III. Karyotypes of H. clausa, H. clausa var. normalis and H. ventricosa. Bot. Mag. (Tokyo) 81, 396–403.
Katoh, K., Misawa, K., Kuma, K., and Miyata, T. (2002). MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 30, 3059–3066. doi: 10.1093/nar/gkf436
Knight, C. A., Molinari, N. A., and Petrov, D. A. (2005). The large genome constraint hypothesis: evolution, ecology and phenotype. Ann. Bot. 95, 177–190. doi: 10.1093/aob/mci011
Lee, S. R., Kim, K., Lee, B. Y., and Lim, C. E. (2019). Complete chloroplast genomes of all six Hosta species occurring in Korea: molecular structures, comparative, and phylogenetic analyses. BMC Genom. 20:833. doi: 10.1186/s12864-019-6215-y
Li, H., and Durbin, R. (2009). Fast and accurate short read alignment with burrows-wheeler transform. Bioinformatics 25, 1754–1760. doi: 10.1093/bioinformatics/btp324
McKain, M. R., Johnson, M. G., Uribe-Convers, S., Eaton, D., and Yang, Y. (2018). Practical considerations for plant phylogenomics. Appl. Plant Sci. 6:e1038. doi: 10.1002/aps3.1038
McKain, M. R., Mcneal, J. R., Kellar, P. R., Eguiarte, L. E., Pires, J. C., and Leebens-Mack, J. (2016). Timing of rapid diversification and convergent origins of active pollination within Agavoideae (Asparagaceae). Am. J. Bot. 103, 1717–1729. doi: 10.3732/ajb.1600198
McKain, M. R., Wickett, N., Zhang, Y., Ayyampalayam, S., Mccombie, W. R., Chase, M. W., et al. (2012). Phylogenomic analysis of transcriptome data elucidates co-occurrence of a paleopolyploid event and the origin of bimodal karyotypes in Agavoideae (Asparagaceae). Am. J. Bot. 99, 397–406. doi: 10.3732/ajb.1100537
Mimaki, Y., Kameyama, A., Kuroda, M., Sashida, Y., Hirano, T., Oka, K., et al. (1997). Steroidal glycosides from the underground parts of Hosta plantaginea var. japonica and their cytostatic activity on leukaemia HL-60 cells. Phytochemistry 44, 305–310. doi: 10.1016/s0031-9422(96)00548-1
Minh, B. Q., Hahn, M. W., and Lanfear, R. (2020). New methods to calculate concordance factors for phylogenomic datasets. Mol. Biol. Evol. 37, 2727–2733. doi: 10.1093/molbev/msaa106
Nguyen, L. T., Schmidt, H. A., Von Haeseler, A., and Minh, B. Q. (2015). IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274. doi: 10.1093/molbev/msu300
R Core_Team (2000). R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing.
Revell, L. J. (2012). phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223. doi: 10.1111/j.2041-210X.2011.00169.x
Rozas, J., Ferrer-Mata, A., Sanchez-Delbarrio, J. C., Guirao-Rico, S., Librado, P., Ramos-Onsins, S. E., et al. (2017). DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol. Biol. Evol. 34, 3299–3302. doi: 10.1093/molbev/msx248
Sauve, R. J., Zhou, S. P., Yu, Y. C., and Schmid, W. G. (2005). Randomly amplified polymorphic DNA analysis in the genus Hosta. HortScience 40, 1243–1245. doi: 10.21273/Hortsci.40.5.1243
Schmid, W. G. (1991). The genus Hosta – Giboshi Zoku (ギボウシ属). London and Portland: Batsford/Timber Press.
Smarda, P., Hejcman, M., Brezinova, A., Horova, L., Steigerova, H., Zedek, F., et al. (2013). Effect of phosphorus availability on the selection of species with different ploidy levels and genome sizes in a long-term grassland fertilization experiment. New Phytol. 200, 911–921. doi: 10.1111/nph.12399
Smith, S. A., and Dunn, C. W. (2008). Phyutility: A phyloinformatics tool for trees, alignments and molecular data. Bioinformatics 24, 715–716. doi: 10.1093/bioinformatics/btm619
Stamatakis, A. (2014). RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313. doi: 10.1093/bioinformatics/btu033
Steele, P. R., Hertweck, K. L., Mayfield, D., Mckain, M. R., Leebens-Mack, J., and Pires, J. C. (2012). Quality and quantity of data recovered from massively parallel sequencing: Examples in Asparagales and Poaceae. Am. J. Bot. 99, 330–348. doi: 10.3732/ajb.1100491
Tamura, M. N., and Fujita, N. (2013). “Hosta,” in Flora of Japan IVb, eds K. Iwatsuki, T. Yamazaki, D. E. Boufford, and H. Ohba (Tokyo, Japan: Heibonsha).
The_American_Hosta_Society (2020). Genus Hosta Registration Publications [Online]. Available Online at: https://www.hostaregistrar.org/hosta_registration_lists.html: The American Hosta Society. [Accessed June 9 2020]
Thureborn, O., Razafimandimbison, S. G., Wikstrom, N., Khodabandeh, A., and Rydin, C. (2019). Phylogeny of Anthospermeae of the coffee family inferred using clock and nonclock models. Int. J. Plant Sci. 180, 386–402. doi: 10.1086/703353
Tidwell, W., and Parker, L. (1990). Protoyucca shadishii gen. et sp. nov., an arbo-rescent monocotyledon with secondary growth from the middle miocene of northwestern Nevada, U.S.A. Rev. Palaeobot. Palynol. 62, 79–95. doi: 10.1016/0034-6667(90)90018-E
Vesely, P., Bures, P., Smarda, P., and Pavlicek, T. (2012). Genome size and DNA base composition of geophytes: the mirror of phenology and ecology? Ann. Bot. 109, 65–75. doi: 10.1093/aob/mcr267
Waselkov, K. E., Boleda, A. S., and Olsen, K. M. (2018). A phylogeny of the genus Amaranthus (Amaranthaceae) based on several low-copy nuclear loci and chloroplast regions. Syst. Bot. 43, 439–458. doi: 10.1600/036364418x697193
Weitemier, K., Straub, S. C., Cronn, R. C., Fishbein, M., Schmickl, R., Mcdonnell, A., et al. (2014). Hyb-Seq: Combining target enrichment and genome skimming for plant phylogenomics. Appl. Plant Sci. 2:42. doi: 10.3732/apps.1400042
Woo, K., Sohn, Y., Ahn, U., Yoon, S., and Spate, A. (2013). ”Geology of Jeju Island,” in Jeju Island Geopark - A Volcanic Wonder of Korea. Geoparks of the World (closed) (Development and Management). Berlin, Heidelberg: Springer.
Yabe, A., Jeong, E., Kim, K., and Uemura, K. (2019). Oligocene–Neogene fossil history of Asian endemic conifer genera in Japan and Korea. J. Syst. Evol. 57, 114–128. doi: 10.1111/jse.12445
Yang, L., Jiang, S. T., Zhou, Q. G., Zhong, G. Y., and He, J. W. (2017). Chemical constituents from the flower of Hosta plantaginea with cyclooxygenases inhibition and antioxidant activities and their chemotaxonomic significance. Molecules 22:1825. doi: 10.3390/molecules22111825
Zhang, C., Rabiee, M., Sayyari, E., and Mirarab, S. (2018). ASTRAL-III: polynomial time species tree reconstruction from partially resolved gene trees. BMC Bioinform. 19:153. doi: 10.1186/s12859-018-2129-y
Keywords: Hosta, Korean Hosta, Hyb-seq, target enrichment, phylogenomics, divergence time estimates
Citation: Yoo M-J, Lee B-Y, Kim S and Lim CE (2021) Phylogenomics With Hyb-Seq Unravels Korean Hosta Evolution. Front. Plant Sci. 12:645735. doi: 10.3389/fpls.2021.645735
Received: 23 December 2020; Accepted: 16 June 2021;
Published: 08 July 2021.
Edited by:
Lisa Pokorny, Center for Plant Biotechnology and Genomics, National Institute of Agricultural and Food Research and Technology, SpainReviewed by:
Lauren Eserman, Atlanta Botanical Garden, United StatesYannick Woudstra, Royal Botanic Gardens, United Kingdom
Nathanael Walker-Hale, University of Cambridge, United Kingdom
Copyright © 2021 Yoo, Lee, Kim and Lim. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chae Eun Lim, Y2hhZWxpbUBrb3JlYS5rcg==
 Mi-Jeong Yoo
Mi-Jeong Yoo Byoung-Yoon Lee2
Byoung-Yoon Lee2