- 1Hunan Key Laboratory of Oral Health Research, Department of Periodontics and Oral Medicine, Xiangya Stomatological Hospital, Xiangya School of Stomatology, Hunan Clinical Research Center of Oral Major Diseases and Oral Health, Academician Workstation for Oral-maxillofacial and Regenerative Medicine, Central South University, Changsha, China
- 2Department of Metabolism and Endocrinology, National Clinical Research Center for Metabolic Disease, The Second Xiangya Hospital, Central South University, Changsha, Hunan, China
Objectives: Animal models of oral submucous fibrosis (OSF) are essential for the studying on the pathogenesis of this disease. Current research on animal models of OSF requires further investigation. In this review, we aim to summarize the strengths and weaknesses of existing OSF animal models, as well as the recent progress in this field.
Subject and methods: OSF is an oral potentially malignant disorder (OPMD) characterized by fibrotic bands, burning sensations, and limited mouth opening. Numerous experimental animal models have been developed to replicate the pathological processes in patients with OSF. Therefore, we systematically evaluated existing animal models of OSF classifying them according to the elements of building an animal model.
Results: In this study, we propose that the elements of animal models for OSF include inducers, animal species, and methods of intervention. Additionally, we highlighted the advantages and limitations of these models and provided directions for future research.
Conclusion: Using human-like animals as experimental subjects, combining both physical and chemical stimulation, and adjusting the dosage and type of inducer may represent the direction of future studies in this field.
1 Introduction
Oral submucous fibrosis (OSF) is a disease characterized by submucous fibrosis, ulceration, a burning sensation, and limited mouth opening. It was first identified as a distinct disease by Indian researchers in 1953 (Pindborg et al., 1964). It has become increasingly prevalent in Asian regions, especially in Vietnam, India, and several Chinese provinces. As a result, it has emerged as a significant global healthcare concern (McGurk and Craig, 1984; Shah et al., 2001). In mainland China, the reported prevalence of OSF ranges from 0.9% to 4.7% (Liu et al., 2015), and from 2.5% to 3.0% in India (Kumbhalwar et al., 2022), with these rates continuing to rise. OSF is a potentially malignant disorder with a high malignant transformation rate (1.5%–15%) (Reichart and Phillipsen, 1998), posing a serious threats human life. Therefore, the prevention and treatment of OSF are critical issues.
1.1 The etiology of OSF
It is widely accepted that the pathogenic factors of OSF include areca nut chewing (Reichart and Phillipsen, 1998; Zain et al., 1999; Lee et al., 2003; Chung et al., 2005; Oakley et al., 2005; Reichart and Nguyen, 2008), nutritional disorders (Jani et al., 2017; Sachdev et al., 2018), genetic predisposition (Chiu et al., 2002; Chen et al., 2004; Xie et al., 2012), and immunologic factors (Pillai et al., 1987; Liu et al., 2022). A large body of epidemiological, in vivo, and in vitro studies has demonstrated that betel nut is a major causative factor in the development of OSF (Lee et al., 2003; Chung et al., 2005; Hazarey et al., 2007; Maher et al., 1994; Yang et al., 2001; Yang et al., 2005; Chen et al., 2006; Yen et al., 2007). Betel nut contains numerous bioactive components, including alkaloids, flavonoids, phenolic compounds, and essential oils. Among these, the most important and widely studied component is arecoline, which is considered the primary active ingredient responsible for inducing OSF.
Key components found in betel nut include:
Arecoline: A primary alkaloid known to promote fibroblast proliferation and collagen deposition, contributing to the fibrotic process in OSF.
Arecaidine: Another alkaloid that may have similar effects to arecoline in the development of OSF.
Tannins: Polyphenolic compounds that can contribute to tissue irritation and fibrosis.
Flavonoids: Antioxidant compounds that may have both protective and harmful effects, depending on their interaction with other components.
The high concentration of arecoline in betel nut is believed to be the most significant factor in the induction of OSF, acting through various molecular pathways to stimulate fibroblasts, inhibit collagen degradation, and promote extracellular matrix accumulation, ultimately leading to fibrosis and subsequent tissue scarring. Understanding the pathogenesis and pathological characteristics of OSF is crucial. However, its exact pathogenesis remains unclear.
1.2 The pathogenesis of OSF
Currently, the most widely accepted theories suggest that OSF pathogenesis involves abnormal expression of inflammatory cytokines and growth factors, such as TGF-β, TNF-α, IGF-1, b-FGF, and CTGF (Chiu et al., 2001; Tsai et al., 2005; Bishen et al., 2008; Khan et al., 2011; Moutasim et al., 2011; Khan et al., 2012; Chang et al., 2013; Kale et al., 2013; Yadahalli et al., 2022), abnormal activation of the NF-κB, c-Jun N-terminal kinase (JNK), and p38 MAPK pathways (Deng et al., 2009; Pitiyage et al., 2011; Liu et al., 2021), imbalance between matrix metalloproteinases (MMPs) and tissue inhibitors of matrix metalloproteinases (TIMPs) (Illeperuma et al., 2010), elevated levels of salivary copper and copper-dependent enzyme lysyl oxidase (Shieh et al., 2009), abnormal activation of the MEK, PI3K, and cyclooxygenase-2 (COX-2) signaling pathways, and the subsequent increase in heat shock protein (HSP) 47 (Utsunomiya et al., 2005; Yang et al., 2008), as well as the generation of reactive oxygen species (ROS) (Deng et al., 2009; Pitiyage et al., 2011). These changes contribute to decreased collagen degradation, increased collagen accumulation, excessive extracellular matrix (ECM) deposition and remodeling, ultimately resulting in the fibrosis characteristic of OSF. Furthermore, excessive apoptosis of endothelial cells in OSF leads to vascular endothelial damage, impairing vascular function and causing epithelial atrophy (Tseng et al., 2012). The histopathological features of OSF include epithelial atrophy, collagen fiber accumulation in the lamina propria and submucosa, reduced vascularity, and vessel occlusion (Figure 1), which collectively compromise the function and architecture of the oral mucosa.
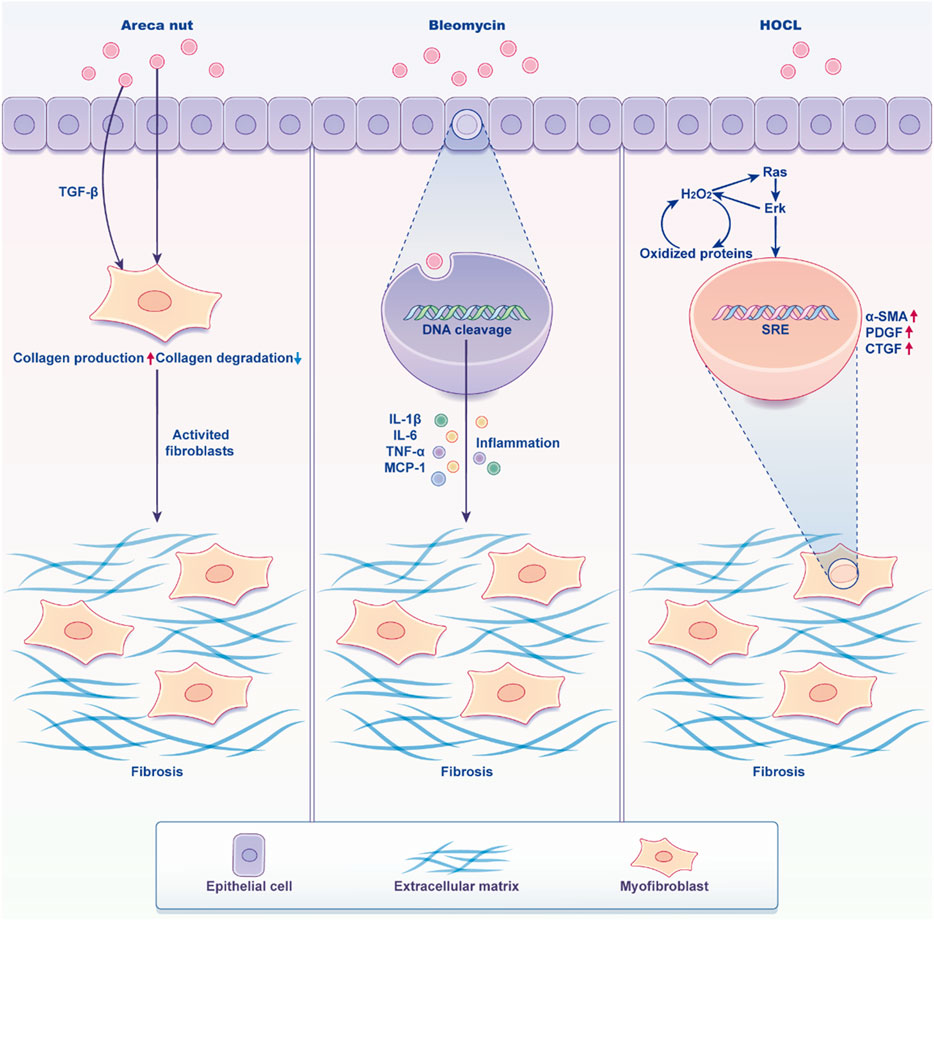
Figure 1. Characters of oral mucosa when stimulated by inducers. Arecoline prompts the epithelial cells of the oral mucosa to express significant levels of TGF-β1, which subsequently induces the activation of fibroblasts into myofibroblasts. This transformation results in an increased secretion of collagen fibers and reduced degradation, ultimately culminating in fibrosis. On the other hand, Bleomycin exerts a toxic effect on cells, leading to DNA fragmentation and the secretion of a large amount of cellular inflammatory factors. These factors activate fibroblasts, causing them to produce excessive collagen fibers, ultimately resulting in fibrosis. Additionally, HOCL stimulates tissues to generate a substantial amount of ROS, which enhances the phosphorylation of ERK1/2 and activates the Ras pathway and gives the diseased fibroblasts a high proliferation phenotype, leading to fibrosis. (Created by Figdraw).
1.3 The purpose of this study
The purpose of this review is to provide a comprehensive overview of the various experimental models used to study oral submucous fibrosis (OSF). Specifically, this review aims to:
Summarize Different Animal Models: Categorize and describe the various animal models based on species, inducing agents, intervention techniques, and outcome observation indicators, providing a structured comparison.
Highlight the Mechanisms of OSF: Focus on the mechanisms induced by various agents and techniques in these models, contributing to a deeper understanding of the pathogenesis of OSF.
Guide Future Research: Offer insights into future research directions, recommend optimal models for studying different aspects of OSF, and suggest improvements or new experimental designs.
Support Translation to Human Disease: Assist researchers in selecting models that best reflect the pathophysiology of OSF in humans, thereby enhancing the translational potential of experimental findings.
2 Method
The study was based on data obtained from a systematic search conducted on Google Scholar, PubMed, and Scopus. The search employed Medical Subject Headings (MeSH) and Boolean operators: (“oral submucous fibrosis” OR “OSF”) AND (“animal models”). No restrictions were applied regarding the year of publication.
The inclusion criteria are as follows:
1. Original experimental articles;
2. Studies involving the establishment of an animal model of oral submucous fibrosis (OSF)
3. Articles providing a detailed description of the conditions and methods used to create the OSF animal model, including the species of experimental animals, inducing agents, induction methods, induction duration, and criteria for evaluating results.
The exclusion criteria are as follows:
1. Literature where the full text is unavailable
2. Literature discussing the impact of OSF animal models on diseases affecting systems other than the oral cavity.
3 The existing animal models of OSF
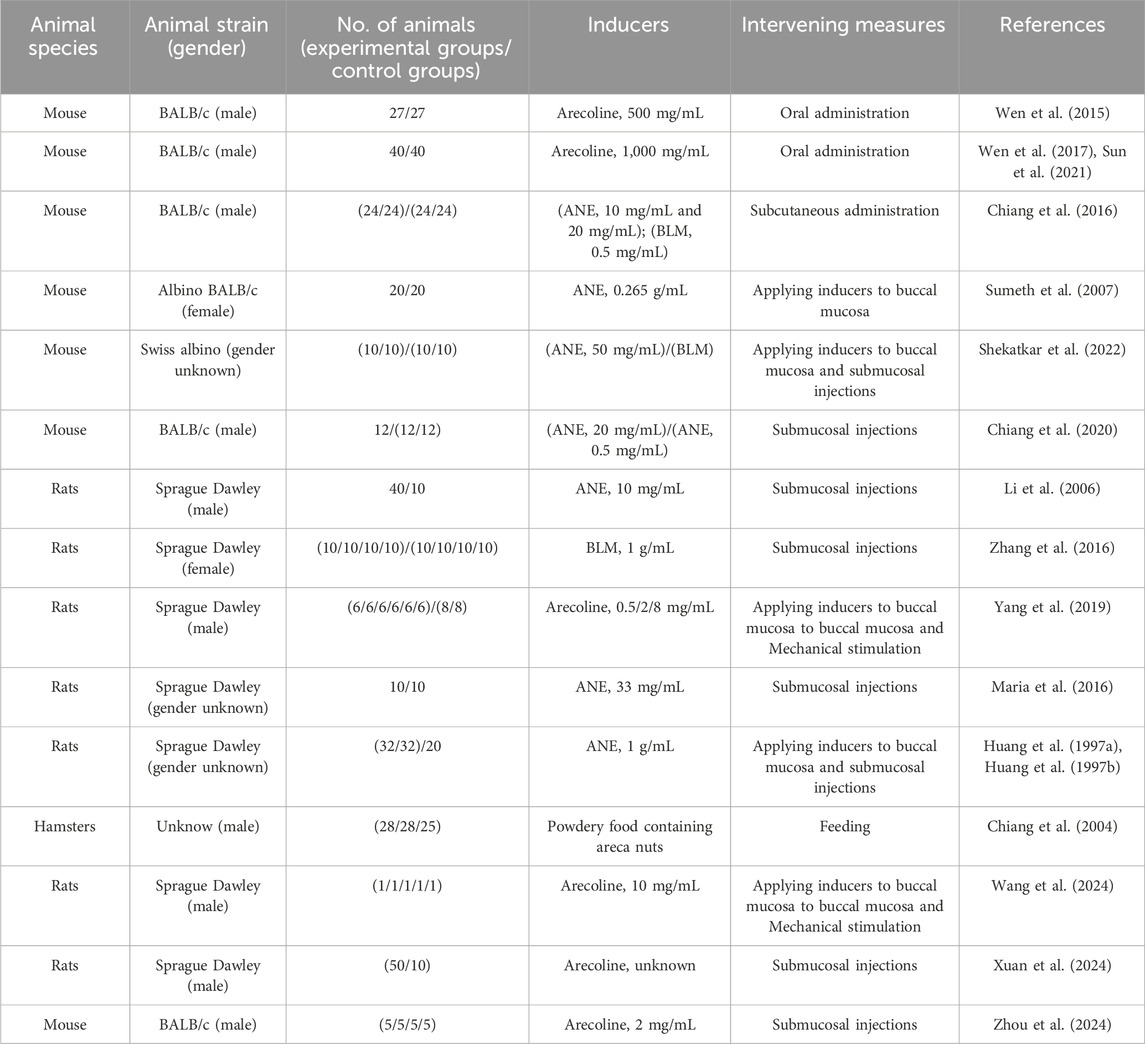
Various animal species have been used in OSF research, including rodents (rats, mice and hamsters), rabbits, and other less commonly utilized mammals. Each species offers distinct advantages depending on the specific research objectives. In previous studies, rodents have been the most commonly used species for experimental models. Therefore, this study will focus on the application of rodent models in OSF research. The existing animal models of OSF can be classified based on inducing agents, intervention techniques and pathological tissue outcomes as follows. This classification aids in identifying the most appropriate animal model for studying specific aspects of OSF pathology and evaluating potential therapeutic interventions (Table 1).
3.1 By inducing agents
3.1.1 Areca nut extract
According to the International Agency for Research on Cancer (Raghavan and Baruah, 1958; Shivashankar et al., 1969; Arjungi, 1976; Betel-quid and areca-nut, 2004), the primary chemical constituents of areca nuts include carbohydrates, fats, proteins, crude fiber, polyphenols (flavonols and tannins), alkaloids, and mineral matter. While alkaloids are the most significant components, other constituents also play crucial roles in the development of oral submucous fibrosis (OSF). Research has shown that certain polyphenols can exacerbate OSF by cross-linking collagen fibers (Sharan et al., 2012). Additionally, areca nuts contain high levels of copper, which is released into the oral environment during chewing and can activate lysyl oxidase, leading to collagen cross-linking and extracellular matrix (ECM) remodeling. One study suggested that these findings indicate copper’s potential local effect on the pathogenesis of OSF(Raja et al., 2007). Therefore, in developing animal models of OSF, it is important to consider all components of areca nut extract (ANE), including alkaloids. Additionally, some researchers used (Saikia and Vaidehi, 1983; Khrime et al., 1991; Chiang et al., 2004) areca nut powder as an inducer. While the results of these experiments were not uniformly ideal, the choice of using areca nut powder inspired future studies. Feeding experimental animals with areca nut powder or applying a paste made from areca nuts to their oral mucosa mimics the process of chewing areca nuts. Therefore, these approaches could offer new directions for selecting inducers for OSF (Table 2).
3.1.2 Arecoline
Arecoline is the principal alkaloid found in areca nuts. Studies have demonstrated that arecoline can stimulate fibroblasts in vitro (Harvey et al., 1986; Jeng et al., 1996). Regarding the mechanism of arecoline-induced OSF, recent research has shown that arecoline induces the expression of several molecules, leading to an increase in plasminogen activator inhibitor-1 (PAI-1), insulin-like growth factor-1 (IGF-1), nuclear factor kappa-light-chain enhancer of activated B cells (NF-κB), and vimentin (Tsai et al., 2005; Chang et al., 2002; Yang et al., 2003; Ni et al., 2007; Chang et al., 2014), promotion of TGF-β activity (Kong et al., 2018), and depletion of cellular glutathione (GSH) (Shieh et al., 2003). These processes ultimately result in the accumulation of extracellular matrix (ECM), the primary pathological characteristic of OSF.
Furthermore, many studies have shown that arecoline promotes the progression of OSF by stimulating reactive oxygen species (ROS) generation (Pitiyage et al., 2011). Excessive reactive oxygen species (ROS) can induce apoptosis (programmed cell death) in endothelial cells, thereby compromising the integrity of blood vessels, which contributes to the pathological alterations in the oral mucosa. These findings suggest that arecoline may be a primary pathogenic factor in the development of oral submucous fibrosis (OSF).
3.1.3 Bleomycin (BLM)
BLM is a glycopeptide antibiotic isolated from the fermentation broth of Streptomyces verticillus (Kong et al., 2018). It was initially used as an anti-cancer agent due to its cytotoxicity. Previous studies (Williamson et al., 2015; He et al., 2016) reported that BLM induces DNA oxidation, which leads to DNA strand scission, resulting in cell cycle arrest, apoptosis, and a dysfunctional repair response. Later, it was discovered that BLM could also cause severe lung and skin fibrosis (Williamson et al., 2015; Li et al., 2022). Following this discovery, researchers began using BLM to develop lung and skin fibrosis models (Luzina et al., 2013; Rangarajan et al., 2018). The animals in these studies showed pathological changes in their skin, such as the over-synthesis of ECM (Davies, 2016; Do and Eming, 2016), similar to those observed in OSF. This provided the theoretical foundation for using BLM as an inducer to create an animal model of OSF.
3.1.4 Hypochlorous acid (HOCl)
Hypochlorous acid (HOCl) is a solid oxidant catalyzed and produced by the heme enzyme myeloperoxidase (MPO) (Winterbourn, 2008), and it can kill pathogens under pathological conditions (Ulfig and Leichert, 2021). However, high concentrations of HOCl in vivo may lead to the development of several major inflammatory pathologies, including cardiovascular disease, neurodegenerative disorders, rheumatoid arthritis, chronic kidney disease, and certain cancers (Witko-Sarsat et al., 1996; Descamps-Latscha et al., 2005; Kisic et al., 2016; Aratani, 2018; Pravalika et al., 2018; Ndrepepa, 2019; Davies and Hawkins, 2020). The mechanism involves oxidative damage to proteins (Winter et al., 2008), DNA (Prütz, 1996), and lipids (Winterbourn et al., 1992). Consequently, some researchers have used HOCl to establish animal models of systemic sclerosis (SSc) (Fonteneau et al., 2017), particularly models of skin fibrosis (Ge et al., 2022; Mohammadi et al., 2022; Yamamoto et al., 2022).
Studies have shown that typical pathological fibrosis changes occur in the skin of experimental animals treated with HOCl, such as the infiltration of CD4+ T cells and macrophages (Gustafsson et al., 1990), and an increase in various cytokines, including transforming growth factor-β (TGF-β), interleukin-1 (IL-1), interleukin-6 (IL-6), tumor necrosis factor (TNF-α), matrix metalloproteinase-2 (MMP-2), and matrix metalloproteinase-6 (MMP-6). These changes may lead to fibroblast activation and extracellular matrix (ECM) synthesis (Ho et al., 2014). These alterations are observed in the early phase following HOCl administration. In the subsequent intermediate stage, a decrease in MMP/TIMP1 (Servettaz et al., 2009) and the loss of adipose tissue (Varga and Marangoni, 2017) could be observed, potentially resulting in (Maria et al., 2018) damage to ECM degradation and thinning of the epithelium.
Therefore, using HOCl provides valuable insights for establishing animal models of oral submucous fibrosis (OSF), as the characteristics of the intermediate and early phases are similar to the pathological changes seen in OSF. Therefore, adjusting the dosage and duration of HOCl exposure may offer a viable alternative for inducing OSF in animal models.
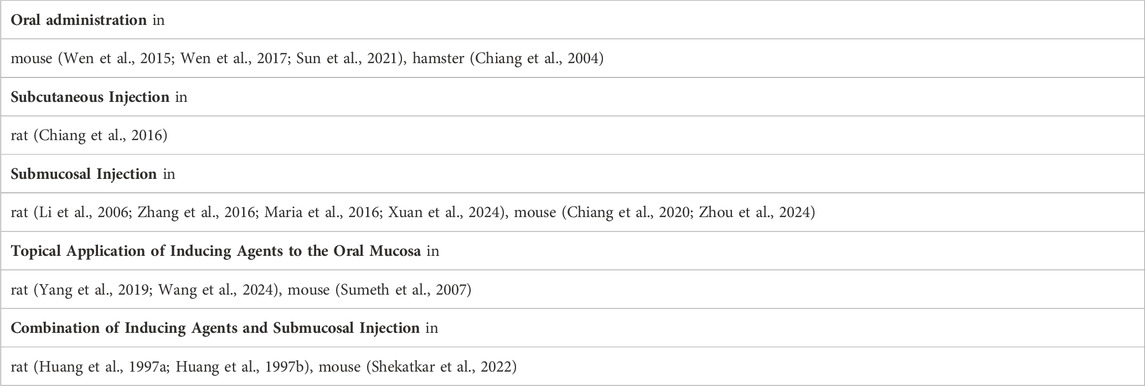
3.2 By intervention technique
Different intervention techniques include oral administration, subcutaneous injection, buccal submucosal injections, application of inducers to the buccal mucosa, or a combination of inducers and submucosal injections. Each of these methods has its own advantages and disadvantages, which are summarized below (Table 3).
3.2.1 Oral administration
3.2.1.1 Advantages
Non-invasive: Oral administration is simple and non-invasive, making it easier to handle animals. Systemic Exposure: Facilitates systemic exposure to inducing agents, which can replicate the chronic nature of OSF seen in humans, particularly with agents like areca nut extract or other fibrogenic substances. Mimics Human Behavior: Reflects real-world exposure, such as tobacco or areca nut chewing, which contributes to OSF in humans.
3.2.1.2 Disadvantages
Variable Absorption: Absorption and bioavailability of inducing agents may vary, leading to inconsistent results. Slow Onset: Oral administration may result in a slower onset of OSF symptoms, requiring longer experimental durations.
3.2.2 Subcutaneous injection
3.2.2.1 Advantages
Controlled Dosage: Subcutaneous injection allows for precise control over the dosage and delivery of inducing agents, ensuring consistent exposure. Systemic Effect: Promotes systemic exposure to agents, especially those that require gradual absorption or release.
3.2.2.2 Disadvantages
Invasive: Injection can cause discomfort or stress in animals, which may affect the validity of results. Local Reactions: May induce local tissue reactions, which might not accurately replicate the oral mucosa environment.
3.2.3 Submucosal injection
3.2.3.1 Advantages
Targeted Induction: Delivers the inducing agent directly to the site of interest, closely mimicking the localized nature of OSF in humans. Faster Onset: Induces fibrosis more rapidly, providing quicker experimental outcomes. Mimics Human Disease: Targets the oral mucosa directly, reflecting how OSF develops in humans due to areca nut chewing or other local irritants.
3.2.3.2 Disadvantages
Invasive: Injections are invasive and may cause local injury, which could affect tissue integrity or influence fibrosis development. Limited to Small Areas: This method is limited to localized fibrosis in the oral mucosa, making it difficult to model systemic effects.
3.2.4 Topical application of inducing agents to the oral mucosa
3.2.4.1 Advantages
Non-invasive: This method is non-invasive and relatively easy to apply to animal models. Mimics Human Exposure: Direct application to the oral mucosa closely mimics how environmental factors (e.g., areca nut or tobacco) contribute to OSF in humans.
3.2.4.2 Disadvantages
Limited Penetration: Topical application may not penetrate deeply enough into tissues, potentially reducing the efficacy of the model in replicating the full fibrotic process. Inconsistent Application: The area of application may not be uniform, leading to variations in the degree of fibrosis across the tissue.
3.2.5 Combination of inducing agents and submucosal injection
3.2.5.1 Advantages
Comprehensive Induction: This combination ensures both localized and systemic effects, leading to a more robust model of OSF. Enhanced Model Precision: Allows researchers to control both local and systemic exposure to inducing agents, making the model more accurate in mimicking human disease.
3.2.5.2 Disadvantages
Invasive and Complex: This method is more invasive than single interventions and introduces additional variables that may affect outcomes. Increased Animal Stress: Multiple interventions can increase animal stress and discomfort, potentially influencing the results.
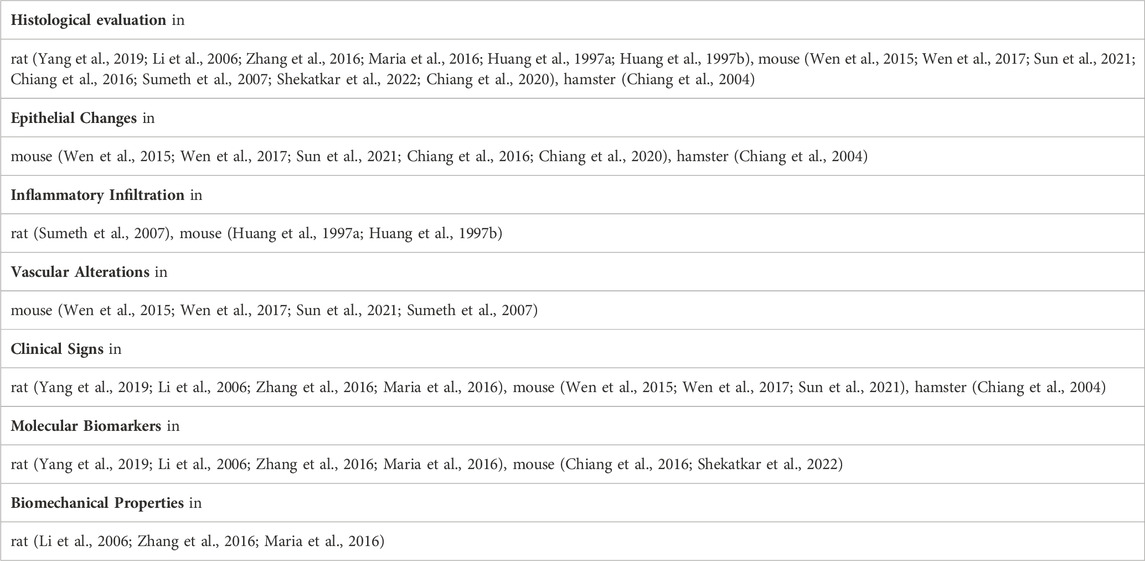
3.3 By outcome observation indicators
When measuring certain indicators, experimental animals may need to be euthanized to obtain tissue samples or perform invasive procedures. However, other indicators can be assessed using non-invasive detection methods, which enable researchers to monitor disease progression or physiological changes without causing harm or distress to the animals (Table 4).
3.3.1 Histological evaluation
Assessment of tissue changes through histological staining techniques, such as Masson’s trichrome, Hematoxylin and Eosin (H&E) staining, or immunohistochemistry, to analyze fibrosis, collagen deposition, and inflammation. When measuring this indicator, experimental animals typically need to be euthanized to obtain tissue samples.
3.3.2 Epithelial changes
Observation of epithelial hyperplasia, thinning, or atrophy as part of the disease progression. When measuring this indicator, experimental animals typically need to be euthanized to obtain tissue samples.
3.3.3 Inflammatory infiltration
Presence of inflammatory cells (e.g., lymphocytes, neutrophils) in the submucosal area, particularly during the early stages of OSF. When measuring this indicator, experimental animals typically need to be euthanized to obtain tissue samples.
3.3.4 Vascular alterations
Changes in blood vessels, such as thickening of the vessel walls or reduced vascularity. When measuring this indicator, experimental animals typically need to be euthanized to obtain tissue samples.
3.3.5 Clinical signs
These may include restriction of mouth opening, changes in mucosal appearance (e.g., whitening, stiffness), and alterations in tissue pliability or flexibility, often assessed through clinical examination. This detection method is non-invasive and does not cause harm to the animals.
3.3.6 Molecular biomarkers
Measurement of specific proteins or genes related to fibrosis, such as collagen types I and III, TGF-β, and other markers associated with fibrogenesis. The methods used for detecting these biomarkers include qPCR, Western blotting, immunohistochemistry, and other related techniques. The biological materials analyzed are typically derived from the buccal mucosa or skin tissues of the experimental animals. When measuring this indicator, experimental animals typically need to be euthanized to obtain tissue samples.
3.3.7 Biomechanical properties
Quantification of the mechanical properties of the oral mucosa, such as tissue elasticity and stiffness, which are affected by the fibrotic process. This detection method is non-invasive and does not cause harm to the animals.
4 Discussion
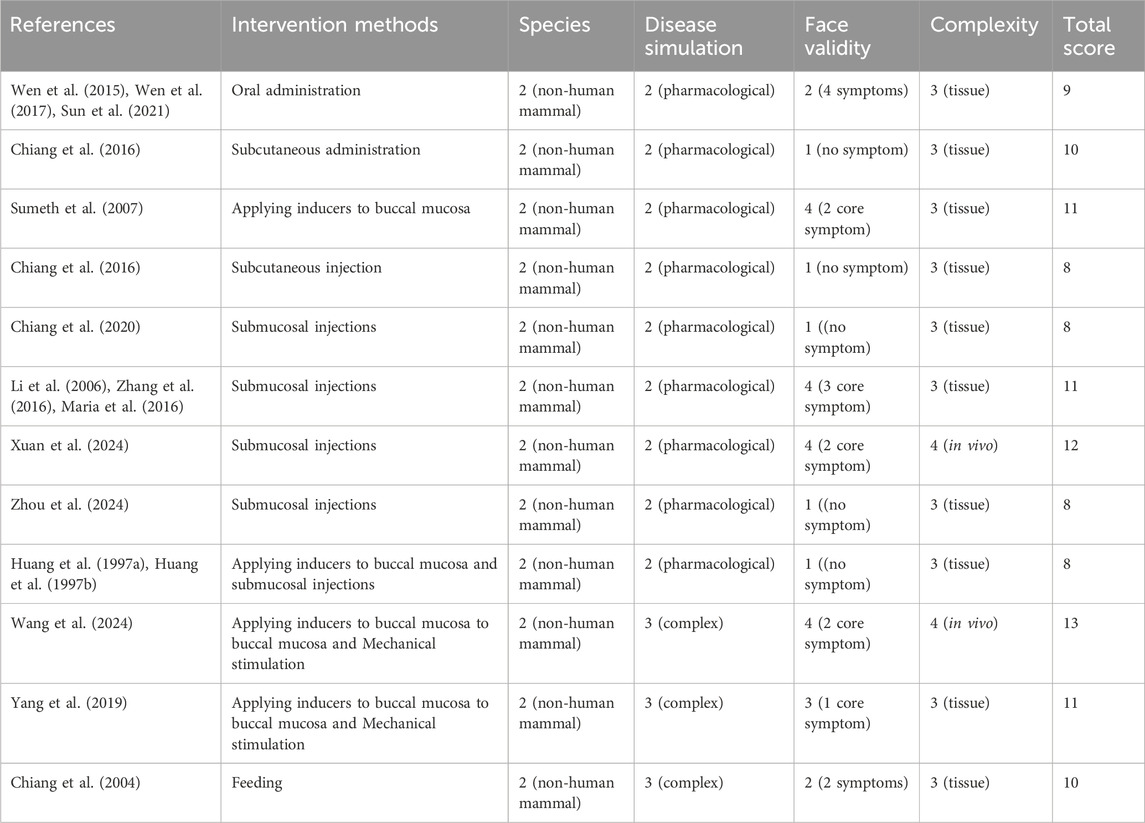
After years of research, significant advances have been made in the study of animal models of oral submucous fibrosis (OSF). However, challenges persist in this field. We evaluate all current research on OSF animal models using a standardized framework (Denayer et al., 2014) to identify the most suitable cases for each intervention method and the animal models that most closely replicate human OSF (Tables 5, 6). This standardized scoring system includes the following criteria: animal species, disease induction method, face validity, and the complexity of outcome assessment indicators (Denayer et al., 2014).
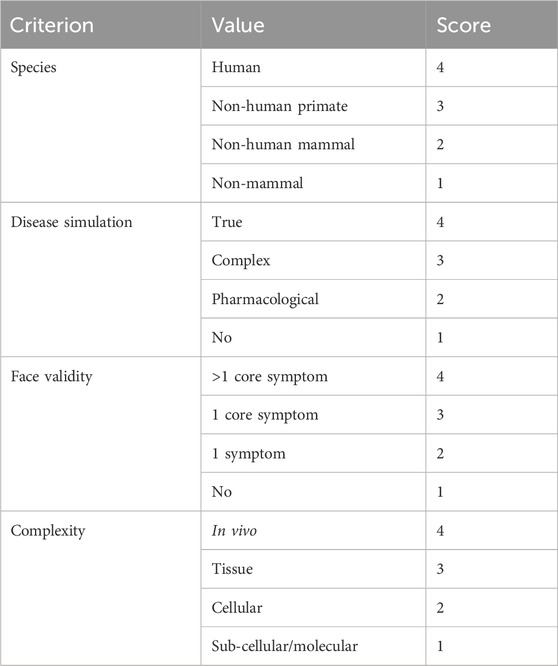
Table 5. Proposed validity scoring system. Adapted from Denayer et al., 2014.
Disease simulation refers to how the disease is simulated in the study, including the realistic simulation of disease state (true): the animal model accurately replicates the pathological characteristics of the disease, including its clinical features, underlying mechanisms, and progression; the use of multiple methods to induce disease, the use of pharmacological agents, or the failure to induce the disease.
Face validity is a measure based on subjective evaluation, assessing the appropriateness or relevance of the model. In this study, face validity refers to the disease symptoms observed in the experimental animals, including core symptoms directly related to OSF, such as reduced mouth opening and the appearance of white patches on the buccal mucosa. Other general symptoms, such as weight loss and fur discoloration, are less closely related to OSF but may still be observed.
Complexity refers to the biological levels at which the outcome indicators are assessed, including the in vivo, tissue, cellular, subcellular, or molecular levels.
Using this scoring system, we evaluated existing rodent OSF models. The study by Wang, S.Y., et al., conducted in 2024, received the highest score. Future OSF animal model construction can reference this study and potentially improve upon it, such as by incorporating non-human primates or other model species.
Mice and rats are commonly used as experimental animals in OSF research. Mice offer advantages such as high fertility, docile temperament, genetic purity, and increased sensitivity to carcinogens. SD rats, being omnivorous like humans, also have a gentle nature and social advantages. Furthermore, these animals are small, inexpensive, and easy to care for, which contributes to their widespread use as OSF models. Hamsters, with their two cheek pouches, have a structure similar to that of humans and could be considered for future studies. Some research has also used other animals, such as New Zealand white rabbits (Song, 2009), although these studies lacked detailed experimental data, which limits their reliability.
Given the ongoing challenges in previous research, such as long experimental periods and low success rates, the proper dosage of inducers still requires further investigation and refinement. Continued efforts are necessary for advancing animal models of OSF, and we hope that our study will contribute to future advancements in this field. Although this article provides a comprehensive overview of current disease models for OSF, it does not offer a definitive conclusion on which model is optimal. However, researchers can select one or more appropriate experimental models based on their specific objectives and conditions to enhance the credibility of their research findings.
5 Summary and future directions
Establishing animal models is essential for studying oral submucous fibrosis (OSF); however, a universally accepted standard model does not yet exist. This may be due to a lack of consensus on the most effective inducers, animal species, and intervention methods. To address this challenge, it is crucial to explore these factors in greater depth and develop more refined strategies for future research. Given that the exact mechanisms of OSF remain unclear, further investigation and better-designed animal models are critical.
The pathogenic factors of OSF include both physical and chemical stimuli. Purely physical stimuli are insufficient to induce the development of OSF(89); however, chemical stimulation with ANE can induce typical fibrotic changes in vivo (Khrime et al., 1991). Endoscopic examination reveals whitening of the esophagus in some OSF patients (Misra et al., 1998), and pathological analysis shows esophageal fibrosis in approximately two-thirds of patients (Shilpa et al., 2011). These findings suggest that chemical stimuli may serve as the initiating factor, while physical stimuli play a promoting role. Therefore, we believe that only by combining both physical and chemical stimuli can an animal model that closely mimics OSF be induced.
Author contributions
LZ: Conceptualization, Writing–original draft, Writing–review and editing. JC: Data curation, Investigation, Writing–review and editing. FL: Investigation, Writing–review and editing. BL: Conceptualization, Funding acquisition, Project administration, Resources, Writing–review and editing. JL: Conceptualization, Formal Analysis, Investigation, Supervision, Validation, Visualization, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This research was funded by the Hunan Province Natural Science Foundation (2022JJ30871), Changsha Natural Science Foundation (kq2202489) and Research Project Funded by the Hunan Provincial Health Care Special Fund (A2024-01).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Aratani Y. (2018). Myeloperoxidase: its role for host defense, inflammation, and neutrophil function. Arch. Biochem. Biophys. 640, 47–52. doi:10.1016/j.abb.2018.01.004
Betel-quid and areca-nut (2004). Betel-quid and areca-nut chewing and some areca-nut derived nitrosamines. IARC Monogr. Eval. Carcinog. Risks Hum. 85, 1–334.
Bishen K. A., Radhakrishnan R., Satyamoorthy K. (2008). The role of basic fibroblast growth factor in oral submucous fibrosis pathogenesis. J. Oral Pathol. Med. 37 (7), 402–411. doi:10.1111/j.1600-0714.2008.00649.x
Chang J. Z., Yang W. H., Deng Y. T., Chen H. M., Kuo M. Y. (2013). EGCG blocks TGFβ1-induced CCN2 by suppressing JNK and p38 in buccal fibroblasts. Clin. Oral Investig. 17 (2), 455–461. doi:10.1007/s00784-012-0713-5
Chang Y. C., Tsai C. H., Lai Y. L., Yu C. C., Chi W. Y., Li J. J., et al. (2014). Arecoline-induced myofibroblast transdifferentiation from human buccal mucosal fibroblasts is mediated by ZEB1. J. Cell Mol. Med. 18 (4), 698–708. doi:10.1111/jcmm.12219
Chang Y.-C., Tsai C.-H., Tai K.-W., Yang S.-H., Chou M.-Y., Lii C.-K. (2002). Elevated vimentin expression in buccal mucosal fibroblasts by arecoline in vitro as a possible pathogenesis for oral submucous fibrosis. Oral Oncol. 38 (5), 425–430. doi:10.1016/s1368-8375(01)00083-5
Chen H. M., Hsieh R. P., Yang H., Kuo Y. S., Kuo M. Y., Chiang C. P. (2004). HLA typing in Taiwanese patients with oral submucous fibrosis. J. Oral Pathol. Med. 33 (4), 191–199. doi:10.1111/j.0904-2512.2004.00195.x
Chen P. C., Pan C. C., Kuo C., Lin C. P. (2006). Risk of oral nonmalignant lesions associated with human papillomavirus infection, betel quid chewing, and cigarette smoking in Taiwan: an integrated molecular and epidemiologic study. Arch. Pathol. Lab. Med. 130 (1), 57–61. doi:10.1043/1543-2165(2006)130[57:ROONLA]2.0.CO;2
Chiang C. P., Chang M. C., Lee J. J., Chang J. Y., Lee P. H., Hahn L. J., et al. (2004). Hamsters chewing betel quid or areca nut directly show a decrease in body weight and survival rates with concomitant epithelial hyperplasia of cheek pouch. Oral Oncol. 40 (7), 720–727. doi:10.1016/j.oraloncology.2003.12.015
Chiang M. H., Chen P. H., Chen Y. K., Chen C. H., Ho M. L., Wang Y. H. (2016). Characterization of a novel dermal fibrosis model induced by areca nut extract that mimics oral submucous fibrosis. PLoS One 11 (11), e0166454. doi:10.1371/journal.pone.0166454
Chiang M. H., Lee K. T., Chen C. H., Chen K. K., Wang Y. H. (2020). Photobiomodulation therapy inhibits oral submucous fibrosis in mice. Oral Dis. 26 (7), 1474–1482. doi:10.1111/odi.13409
Chiu C. J., Chang M. L., Chiang C. P., Hahn L. J., Hsieh L. L., Chen C. J. (2002). Interaction of collagen-related genes and susceptibility to betel quid-induced oral submucous fibrosis. Cancer Epidem Biomar 11 (7), 646–653.
Chiu C. J., Chiang C. P., Chang M. L., Chen H. M., Hahn L. J., Hsieh L. L., et al. (2001). Association between genetic polymorphism of tumor necrosis factor-alpha and risk of oral submucous fibrosis, a pre-cancerous condition of oral cancer. J. Dent. Res. 80 (12), 2055–2059. doi:10.1177/00220345010800120601
Chung C. H., Yang Y. H., Wang T. Y., Shieh T. Y., Warnakulasuriya S. (2005). Oral precancerous disorders associated with areca quid chewing, smoking, and alcohol drinking in southern Taiwan. J. Oral Pathol. Med. 34 (8), 460–466. doi:10.1111/j.1600-0714.2005.00332.x
Davies M. J. (2016). Protein oxidation and peroxidation. Biochem. J. 473 (7), 805–825. doi:10.1042/BJ20151227
Davies M. J., Hawkins C. L. (2020). The role of myeloperoxidase in biomolecule modification, chronic inflammation, and disease. Antioxid. Redox Signal 32 (13), 957–981. doi:10.1089/ars.2020.8030
Denayer T., St?Hr T., Van Roy M. (2014). Animal models in translational medicine: Validation and prediction. New Horizons Transl. Med. 2 (1), 5–11.
Deng Y. T., Chen H. M., Cheng S. J., Chiang C. P., Kuo M. Y. (2009). Arecoline-stimulated connective tissue growth factor production in human buccal mucosal fibroblasts: modulation by curcumin. Oral Oncol. 45 (9), e99–e105. doi:10.1016/j.oraloncology.2009.04.004
Descamps-Latscha B., Witko-Sarsat V., Nguyen-Khoa T., Nguyen A. T., Gausson V., Mothu N., et al. (2005). Advanced oxidation protein products as risk factors for atherosclerotic cardiovascular events in nondiabetic predialysis patients. Am. J. Kidney Dis. 45 (1), 39–47. doi:10.1053/j.ajkd.2004.09.011
Do N. N., Eming S. A. (2016). Skin fibrosis: models and mechanisms. Curr. Res. Transl. Med. 64 (4), 185–193. doi:10.1016/j.retram.2016.06.003
Fonteneau G., Bony C., Goulabchand R., Maria A. T. J., Le Quellec A., Rivière S., et al. (2017). Serum-Mediated oxidative stress from systemic sclerosis patients affects mesenchymal stem cell function. Front. Immunol. 8, 988. doi:10.3389/fimmu.2017.00988
Ge Y., Luo J., Li D., Li C., Huang J., Yu H., et al. (2022). Deficiency of vitamin D receptor in keratinocytes augments dermal fibrosis and inflammation in a mouse model of HOCl-induced scleroderma. Biochem. Biophys. Res. Commun. 591, 1–6. doi:10.1016/j.bbrc.2021.12.085
Gustafsson R., Tötterman T. H., Klareskog L., Hällgren R. (1990). Increase in activated T cells and reduction in suppressor inducer T cells in systemic sclerosis. Ann. Rheum. Dis. 49 (1), 40–45. doi:10.1136/ard.49.1.40
Harvey W., Scutt A., Meghji S., Canniff J. P. (1986). Stimulation of human buccal mucosa fibroblasts in vitro by betel-nut alkaloids. Arch. Oral Biol. 31 (1), 45–49. doi:10.1016/0003-9969(86)90112-3
Hazarey V. K., Erlewad D. M., Mundhe K. A., Ughade S. N. (2007). Oral submucous fibrosis: study of 1000 cases from central India. J. Oral Pathol. Med. 36 (1), 12–17. doi:10.1111/j.1600-0714.2006.00485.x
He Y., Lan Y., Liu Y., Yu H., Han Z., Li X., et al. (2016). Pingyangmycin and bleomycin share the same cytotoxicity pathway. Molecules 21 (7), 862. doi:10.3390/molecules21070862
Ho Y. Y., Lagares D., Tager A. M., Kapoor M. (2014). Fibrosis--a lethal component of systemic sclerosis. Nat. Rev. Rheumatol. 10 (7), 390–402. doi:10.1038/nrrheum.2014.53
Huang S., Ling T., Wu H. (1997a). Experimental study on aqueous areca nut extracts inducing oral submucous fibrosis in rats. I. Observation of histomorphology. Hua Xi Kou Qiang Yi Xue Za Zhi 15 (2), 91–93.
Huang S., Ling T., Wu H. (1997b). Experimental study on aqueous areca nut extracts inducing oral submucous fibrosis in rats. II. Effection of mast cells on collagen metabolism. Hua Xi Kou Qiang Yi Xue Za Zhi 15 (2), 94–96.
Illeperuma R. P., Ryu M. H., Kim K. Y., Tilakaratne W. M., Kim J. (2010). Relationship of fibrosis and the expression of TGF-β1, MMP-1, and TIMP-1 with epithelial dysplasia in oral submucous fibrosis. J. Oral Pathol. Med. 15 (1), 21–28. doi:10.3353/omp.15.21
Jani Y. V., Chaudhary A. R., Dudhia B. B., Bhatia P. V., Soni N. C., Patel P. S. (2017). Evaluation of role of trace elements in oral submucous fibrosis patients: a study on Gujarati population. J. Oral Maxillofac. Pathol. 21 (3), 455. doi:10.4103/jomfp.JOMFP_106_14
Jeng J. H., Lan W. H., Hahn L. J., Hsieh C. C., Kuo M. Y. (1996). Inhibition of the migration, attachment, spreading, growth and collagen synthesis of human gingival fibroblasts by arecoline, a major areca alkaloid, in vitro. J. Oral Pathol. Med. 25 (7), 371–375. doi:10.1111/j.1600-0714.1996.tb00281.x
Kale A. D., Mane D. R., Shukla D. (2013). Expression of transforming growth factor β and its correlation with lipodystrophy in oral submucous fibrosis: an immunohistochemical study. Med. Oral Patol. Oral Cir. Bucal 18 (1), e12–e18. doi:10.4317/medoral.18226
Khan I., Agarwal P., Thangjam G. S., Radhesh R., Rao S. G., Kondaiah P. (2011). Role of TGF-β and BMP7 in the pathogenesis of oral submucous fibrosis. Growth factors. 29 (4), 119–127. doi:10.3109/08977194.2011.582839
Khan I., Kumar N., Pant I., Narra S., Kondaiah P. (2012). Activation of TGF-β pathway by areca nut constituents: a possible cause of oral submucous fibrosis. PLoS One 7 (12). doi:10.1371/journal.pone.0051806
Khrime R. D., Mehra Y. N., Mann S. B., Mehta S. K., Chakraborti R. N. (1991). Effect of instant preparation of betel nut (pan masala) on the oral mucosa of albino rats. Indian J. Med. Res. 94, 119–124.
Kisic B., Miric D., Dragojevic I., Rasic J., Popovic L. (2016). Role of myeloperoxidase in patients with chronic kidney disease. Oxid. Med. Cell Longev. 2016, 1069743. doi:10.1155/2016/1069743
Kong J., Yi L., Xiong Y., Huang Y., Yang D., Yan X., et al. (2018). The discovery and development of microbial bleomycin analogues. Appl. Microbiol. Biotechnol. 102 (16), 6791–6798. doi:10.1007/s00253-018-9129-8
Kumbhalwar A., Shetiya S. H., Kakodkar P., Mehta V., Mathur A., Porwal P. (2022). Prevalence of precancerous lesions and conditions in India: a systematic review and meta-analysis. World J. Methodol. 12 (4), 293–304. doi:10.5662/wjm.v12.i4.293
Lee C. H., Ko Y. C., Huang H. L., Chao Y. Y., Tsai C. C., Shieh T. Y., et al. (2003). The precancer risk of betel quid chewing, tobacco use and alcohol consumption in oral leukoplakia and oral submucous fibrosis in southern Taiwan. Br. J. Cancer 88 (3), 366–372. doi:10.1038/sj.bjc.6600727
Li S., Shi J., Tang H. (2022). Animal models of drug-induced pulmonary fibrosis: an overview of molecular mechanisms and characteristics. Cell Biol. Toxicol. 38 (5), 699–723. doi:10.1007/s10565-021-09676-z
Li Y. C., Tan J., Chen A., Peng C. X., Chen M., Liu Y. P., et al. (2006). Effect of Danxuankukang on TGF in rats with oral submucosal fibrosis β-1 Effect of expression. New Chin. Med. (11), 89–91.
Liu B., Cai J., Li W., Liu G., Chen J. (2022). Oral submucous fibrosis induced by graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Oral Oncol. 130, 105919. doi:10.1016/j.oraloncology.2022.105919
Liu B., Shen M., Xiong J., Yuan Y., Wu X., Gao X., et al. (2015). Synergistic effects of betel quid chewing, tobacco use (in the form of cigarette smoking), and alcohol consumption on the risk of malignant transformation of oral submucous fibrosis (OSF): a case-control study in Hunan Province, China. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 120 (3), 337–345. doi:10.1016/j.oooo.2015.04.013
Liu J., Li F., Liu B., Yao Z., Li L., Liu G., et al. (2021). Adipose-derived mesenchymal stem cell exosomes inhibit transforming growth factor-β1-induced collagen synthesis in oral mucosal fibroblasts. Exp. Ther. Med. 22 (6), 1419. doi:10.3892/etm.2021.10854
Luzina I. G., Kopach P., Lockatell V., Kang P. H., Nagarsekar A., Burke A. P., et al. (2013). Interleukin-33 potentiates bleomycin-induced lung injury. Am. J. Respir. Cell Mol. Biol. 49 (6), 999–1008. doi:10.1165/rcmb.2013-0093OC
Maher R., Lee A. J., Warnakulasuriya K. A., Lewis J. A., Johnson N. W. (1994). Role of areca nut in the causation of oral submucous fibrosis: a case-control study in Pakistan. J. Oral Pathol. Med. 23 (2), 65–69. doi:10.1111/j.1600-0714.1994.tb00258.x
Maria A. T. J., Toupet K., Maumus M., Rozier P., Vozenin M. C., Le Quellec A., et al. (2018). Fibrosis development in HOCl-induced systemic sclerosis: a multistage process hampered by mesenchymal stem cells. Front. Immunol. 9, 2571. doi:10.3389/fimmu.2018.02571
Maria S., Kamath V. V., Satelur K., Rajkumar K. (2016). Evaluation of transforming growth factor beta1 gene in oral submucous fibrosis induced in Sprague-Dawley rats by injections of areca nut and pan masala (commercial areca nut product) extracts. J. Cancer Res. Ther. 12 (1), 379–385. doi:10.4103/0973-1482.148729
McGurk M., Craig G. T. (1984). Oral submucous fibrosis: two cases of malignant transformation in Asian immigrants to the United Kingdom. Br. J. Oral Maxillofac. Surg. 22 (1), 56–64. doi:10.1016/0266-4356(84)90010-x
Misra S. P., Misra V., Dwivedi M., Gupta S. C. (1998). Oesophageal subepithelial fibrosis: an extension of oral submucosal fibrosis. Postgrad. Med. J. 74 (878), 733–736. doi:10.1136/pgmj.74.878.733
Mohammadi M., Kohan L., Saeidi M., Saghaeian Jazi M., Mohammadi S. (2022). The antifibrotic effects of naringin in a hypochlorous acid (HOCl)-induced mouse model of skin fibrosis. Immunopharm immunot 44 (5), 704–711. doi:10.1080/08923973.2022.2077217
Moutasim K. A., Jenei V., Sapienza K., Marsh D., Weinreb P. H., Violette S. M., et al. (2011). Betel-derived alkaloid up-regulates keratinocyte alphavbeta6 integrin expression and promotes oral submucous fibrosis. J. Pathol. 223 (3), 366–377. doi:10.1002/path.2786
Ndrepepa G. (2019). Myeloperoxidase - a bridge linking inflammation and oxidative stress with cardiovascular disease. Clin. Chim. Acta 493, 36–51. doi:10.1016/j.cca.2019.02.022
Ni W. F., Tsai C. H., Yang S. F., Chang Y. C. (2007). Elevated expression of NF-kappaB in oral submucous fibrosis--evidence for NF-kappaB induction by safrole in human buccal mucosal fibroblasts. Oral Oncol. 43 (6), 557–562. doi:10.1016/j.oraloncology.2006.06.007
Oakley E., Demaine L., Warnakulasuriya S. (2005). Areca (betel) nut chewing habit among high-school children in the Commonwealth of the Northern Mariana Islands (Micronesia). B World Health Organ 83 (9), 656–660.
Pillai M. R., Balaram P., Abraham T., Nair M. K. (1987). Lymphocyte populations in premalignant lesions and cancer of the oral cavity. Neoplasma 34 (4), 469–479.
Pindborg J. J., Chawla T. N., Srivastava A. N., Gupta D., Mehrotra M. L. (1964). Clinical aspect of oral submucous fibrosis. Acta Odontol. Scand. 22, 679–691. doi:10.3109/00016356409058581
Pitiyage G. N., Slijepcevic P., Gabrani A., Chianea Y. G., Lim K. P., Prime S. S., et al. (2011). Senescent mesenchymal cells accumulate in human fibrosis by a telomere-independent mechanism and ameliorate fibrosis through matrix metalloproteinases. J. Pathol. 223 (5), 604–617. doi:10.1002/path.2839
Pravalika K., Sarmah D., Kaur H., Wanve M., Saraf J., Kalia K., et al. (2018). Myeloperoxidase and neurological disorder: a crosstalk. ACS Chem. Neurosci. 9 (3), 421–430. doi:10.1021/acschemneuro.7b00462
Prütz W. A. (1996). Hypochlorous acid interactions with thiols, nucleotides, DNA, and other biological substrates. Arch. Biochem. Biophys. 332 (1), 110–120. doi:10.1006/abbi.1996.0322
Raghavan V., Baruah H. (1958). Arecanut: India’s popular masticatory—history, chemistry and utilization. Econ. Bot. 12 (4), 315–345. doi:10.1007/bf02860022
Raja K. B., Hazarey V. K., Peters T. J., Warnakulasuriya S. (2007). Effect of areca nut on salivary copper concentration in chronic chewers. Biometal 20 (1), 43–47. doi:10.1007/s10534-006-9013-3
Rangarajan S., Bone N. B., Zmijewska A. A., Jiang S., Park D. W., Bernard K., et al. (2018). Metformin reverses established lung fibrosis in a bleomycin model. Nat. Med. 24 (8), 1121–1127. doi:10.1038/s41591-018-0087-6
Reichart P. A., Nguyen X. H. (2008). Betel quid chewing, oral cancer and other oral mucosal diseases in Vietnam: a review. J. Oral Pathol. Med. 37 (9), 511–514. doi:10.1111/j.1600-0714.2008.00669.x
Reichart P. A., Phillipsen H. P. (1998). Betel chewer's mucosa--a review. J. Oral Pathol. Med. 27 (6), 239–242. doi:10.1111/j.1600-0714.1998.tb01949.x
Sachdev P. K., Freeland-Graves J., Beretvas S. N., Sanjeevi N. (2018). Zinc, copper, and iron in oral submucous fibrosis: a meta-analysis. Int. J. Dent. 2018, 3472087. doi:10.1155/2018/3472087
Saikia M., Vaidehi M. P. (1983). Studies on the pathological effects of feeding betel-nut meal in albino rats. Br. J. Exp. Pathol. 64 (5), 515–517.
Servettaz A., Goulvestre C., Kavian N., Nicco C., Guilpain P., Chéreau C., et al. (2009). Selective oxidation of DNA topoisomerase 1 induces systemic sclerosis in the mouse. J. Immunol. 182 (9), 5855–5864. doi:10.4049/jimmunol.0803705
Shah B., Lewis M. A., Bedi R. (2001). Oral submucous fibrosis in a 11-year-old Bangladeshi girl living in the United Kingdom. Br. Dent. J. 191 (3), 130–132. doi:10.1038/sj.bdj.4801117
Sharan R. N., Mehrotra R., Choudhury Y., Asotra K. (2012). Association of betel nut with carcinogenesis: revisit with a clinical perspective. PLoS One 7 (8), e42759. doi:10.1371/journal.pone.0042759
Shekatkar M., Kheur S., Sanap A., Undale V., Kharat A., Bhalchim V., et al. (2022). A novel approach to develop an animal model for oral submucous fibrosis. Med. Oncol. 39 (11), 162. doi:10.1007/s12032-022-01760-6
Shieh D. H., Chiang L. C., Shieh T. Y. (2003). Augmented mRNA expression of tissue inhibitor of metalloproteinase-1 in buccal mucosal fibroblasts by arecoline and safrole as a possible pathogenesis for oral submucous fibrosis. Oral Oncol. 39 (7), 728–735. doi:10.1016/s1368-8375(03)00101-5
Shieh T. M., Tu H. F., Ku T. H., Chang S. S., Chang K. W., Liu C. J. (2009). Association between lysyl oxidase polymorphisms and oral submucous fibrosis in older male areca chewers. J. Oral Pathol. Med. 38 (1), 109–113. doi:10.1111/j.1600-0714.2008.00695.x
Shilpa B. J., Ashok L., Veerendra Swamy S. M. (2011). Esophageal changes in oral submucous fibrosis using fiber-optic endoscopy. J. Investig. Clin. Dent. 2 (1), 10–15. doi:10.1111/j.2041-1626.2010.00037.x
Shivashankar S., Dhanaraj S., Mathew A., Srinivasa Murthy S., Vyasamurthy M., Govindarajan V. (1969). Physical and chemical characteristics of processed arecanuts. J. Food Sci. Technol. (Mysore). 6 (2), 113–116.
Song Z. F. (2009). Effect of collagenase treatment on oral submucosal fibrosis on patients' mouth opening and masticatory function. China Pract. Med. 4 (11), 122–123.
Sumeth P. M. W., Gunasinghe D., Perera P. A., Ranasinghe A., Amaratunga P., Warnakulasuriya S., et al. (2007). Development of an in vivo mouse model to study oral submucous fibrosis. J. Oral Pathol. Med. 36 (5), 273–280. doi:10.1111/j.1600-0714.2007.00523.x
Sun Y., Wang T., Wen Q. T., Yu D. H., Chen J. X. (2021). VEGF gene transfection restores the angiogenesis of oral submucous fibrosis in mice. Ann. Transl. Med. 9 (11), 930. doi:10.21037/atm-21-2213
Tsai C. H., Yang S. F., Chen Y. J., Chou M. Y., Chang Y. C. (2005). The upregulation of insulin-like growth factor-1 in oral submucous fibrosis. Oral Oncol. 41 (9), 940–946. doi:10.1016/j.oraloncology.2005.05.006
Tseng S. K., Chang M. C., Su C. Y., Chi L. Y., Chang J. Z., Tseng W. Y., et al. (2012). Arecoline induced cell cycle arrest, apoptosis, and cytotoxicity to human endothelial cells. Clin. Oral Investig. 16 (4), 1267–1273. doi:10.1007/s00784-011-0604-1
Ulfig A., Leichert L. I. (2021). The effects of neutrophil-generated hypochlorous acid and other hypohalous acids on host and pathogens. Cell Mol. Life Sci. 78 (2), 385–414. doi:10.1007/s00018-020-03591-y
Utsunomiya H., Tilakaratne W. M., Oshiro K., Maruyama S., Suzuki M., Ida-Yonemochi H., et al. (2005). Extracellular matrix remodeling in oral submucous fibrosis: its stage-specific modes revealed by immunohistochemistry and in situ hybridization. J. Oral Pathol. Med. 34 (8), 498–507. doi:10.1111/j.1600-0714.2005.00339.x
Varga J., Marangoni R. G. (2017). Systemic sclerosis in 2016: dermal white adipose tissue implicated in SSc pathogenesis. Nat. Rev. Rheumatol. 13 (2), 71–72. doi:10.1038/nrrheum.2016.223
Wang S. Y., Zhang S. J., Meng H. F., Xu H. Q., Guo Z. X., Yan J. F., et al. (2024). DPSCs regulate epithelial-T cell interactions in oral submucous fibrosis. Stem Cell Res. Ther. 15 (1), 113. doi:10.1186/s13287-024-03720-5
Wen Q. T., Wang T., Yu D. H., Wang Z. R., Qing H. Y., Sun Y. (2015). Establishment of arecoline chemistry-induced Balb/c mouse oral submucous fibrosis model. J. Guangxi Med. Univ. 32 (06), 876–878.
Wen Q. T., Wang T., Yu D. H., Wang Z. R., Sun Y., Liang C. W. (2017). Development of a mouse model of arecoline-induced oral mucosal fibrosis. Asian Pac J. Trop. Med. 10 (12), 1177–1184. doi:10.1016/j.apjtm.2017.10.026
Williamson J. D., Sadofsky L. R., Hart S. P. (2015). The pathogenesis of bleomycin-induced lung injury in animals and its applicability to human idiopathic pulmonary fibrosis. Exp. Lung Res. 41 (2), 57–73. doi:10.3109/01902148.2014.979516
Winter J., Ilbert M., Graf P. C., Ozcelik D., Jakob U. (2008). Bleach activates a redox-regulated chaperone by oxidative protein unfolding. Cell 135 (4), 691–701. doi:10.1016/j.cell.2008.09.024
Winterbourn C. C. (2008). Reconciling the chemistry and biology of reactive oxygen species. Nat. Chem. Biol. 4 (5), 278–286. doi:10.1038/nchembio.85
Winterbourn C. C., van den Berg J. J., Roitman E., Kuypers F. A. (1992). Chlorohydrin formation from unsaturated fatty acids reacted with hypochlorous acid. Arch. Biochem. Biophys. 296 (2), 547–555. doi:10.1016/0003-9861(92)90609-z
Witko-Sarsat V., Friedlander M., Capeillère-Blandin C., Nguyen-Khoa T., Nguyen A. T., Zingraff J., et al. (1996). Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 49 (5), 1304–1313. doi:10.1038/ki.1996.186
Xie H., Liu J., Ling T. Y. (2012). Expression of cytochrome P450 related genes in oral submucous fibrosis tissue. Zhonghua Kou Qiang Yi Xue Za Zhi 47 (12), 743–747. doi:10.3760/cma.j.issn.1002-0098.2012.12.012
Xuan Z., An L., Keke Z., Mingyu R. (2024). Effect of Jiedu Huayu decoction on oral mucosal Axin and β-catenin expression in oral submucosal fibrosis model rats. J. Tradit. Chin. Med. 44 (4), 688–693. doi:10.19852/j.cnki.jtcm.20240617.001
Yadahalli R., Sarode G. S., Sarode S. C., Khan Z. A., Vyas N., Kharat A. H., et al. (2022). CC group of chemokines and associated gene expression of transcription factors: deciphering immuno-pathogenetic aspect of oral submucous fibrosis. Dm-Dis Mon. 69, 101351. doi:10.1016/j.disamonth.2022.101351
Yamamoto A., Saito T., Hosoya T., Kawahata K., Asano Y., Sato S., et al. (2022). Therapeutic effect of cyclin-dependent kinase 4/6 inhibitor on dermal fibrosis in murine models of systemic sclerosis. Arthritis Rheumatol. 74 (5), 860–870. doi:10.1002/art.42042
Yang B., Fu M. F., Tang Z. G. (2019). Rat model with oral submucous fibrosis induced by arecoline and mechanical stimulation. Hua Xi Kou Qiang Yi Xue Za Zhi 37 (3), 260–264. doi:10.7518/hxkq.2019.03.006
Yang S.-F., Hsieh Y.-S., Tsai C.-H., Chou M.-Y., Chang Y.-C. (2003). The upregulation of type I plasminogen activator inhibitor in oral submucous fibrosis. Oral Oncol. 39 (4), 367–372. doi:10.1016/s1368-8375(02)00123-9
Yang S. F., Tsai C. H., Chang Y. C. (2008). The upregulation of heat shock protein 47 expression in human buccal fibroblasts stimulated with arecoline. J. Oral Pathol. Med. 37 (4), 206–210. doi:10.1111/j.1600-0714.2007.00633.x
Yang Y. H., Lee H. Y., Tung S., Shieh T. Y. (2001). Epidemiological survey of oral submucous fibrosis and leukoplakia in aborigines of Taiwan. J. Oral Pathol. Med. 30 (4), 213–219. doi:10.1034/j.1600-0714.2001.300404.x
Yang Y. H., Lien Y. C., Ho P. S., Chen C. H., Chang J. S., Cheng T. C., et al. (2005). The effects of chewing areca/betel quid with and without cigarette smoking on oral submucous fibrosis and oral mucosal lesions. Oral Dis. 11 (2), 88–94. doi:10.1111/j.1601-0825.2004.01061.x
Yen A. M., Chen S. C., Chen T. H. (2007). Dose-response relationships of oral habits associated with the risk of oral pre-malignant lesions among men who chew betel quid. Oral Oncol. 43 (7), 634–638. doi:10.1016/j.oraloncology.2006.05.001
Zain R. B., Ikeda N., Gupta P. C., Warnakulasuriya S., van Wyk C. W., Shrestha P., et al. (1999). Oral mucosal lesions associated with betel quid, areca nut and tobacco chewing habits: consensus from a workshop held in Kuala Lumpur, Malaysia, November 25-27, 1996. J. Oral Pathol. Med. 28 (1), 1–4. doi:10.1111/j.1600-0714.1999.tb01985.x
Zhang S. S., Gong Z. J., Xiong W., Wang X., Min Q., Luo C. D., et al. (2016). A rat model of oral submucous fibrosis induced by bleomycin. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 122 (2), 216–223. doi:10.1016/j.oooo.2015.07.042
Keywords: oral submucous fibrosis, animal model, areca nut, HOCl, bleomycin
Citation: Zhang L, Chen J, Li F, Liu B and Liu J (2025) Choosing the proper animal model for oral submucous fibrosis research: considerations and challenges. Front. Physiol. 16:1501158. doi: 10.3389/fphys.2025.1501158
Received: 24 September 2024; Accepted: 28 January 2025;
Published: 06 March 2025.
Edited by:
Christine Hong, University of California, San Francisco, United StatesReviewed by:
Rucha Arun Bapat, University of Southern California, United StatesZhang Xiaoqi, Sichuan University, China
Copyright © 2025 Zhang, Chen, Li, Liu and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Binjie Liu, bGl1YmluamllQDE4OC5jb20=Junjie Liu, MTY2ODEyMDA4QGNzdS5lZHUuY24=
 Linlin Zhang
Linlin Zhang Jun Chen
Jun Chen Fuxingzi Li2
Fuxingzi Li2 Binjie Liu
Binjie Liu