
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Physiol., 26 April 2022
Sec. Respiratory Physiology and Pathophysiology
Volume 13 - 2022 | https://doi.org/10.3389/fphys.2022.813478
This article is part of the Research TopicProtecting the Acutely Injured Lung: Physiologic, Mechanical, Inflammatory, and Translational PerspectivesView all 17 articles
High-frequency oscillatory ventilation (HFOV) is a type of invasive mechanical ventilation that employs supra-physiologic respiratory rates and low tidal volumes (VT) that approximate the anatomic deadspace. During HFOV, mean airway pressure is set and gas is then displaced towards and away from the patient through a piston. Carbon dioxide (CO2) is cleared based on the power (amplitude) setting and frequency, with lower frequencies resulting in higher VT and CO2 clearance. Airway pressure amplitude is significantly attenuated throughout the respiratory system and mechanical strain and stress on the alveoli are theoretically minimized. HFOV has been purported as a form of lung protective ventilation that minimizes volutrauma, atelectrauma, and biotrauma. Following two large randomized controlled trials showing no benefit and harm, respectively, HFOV has largely been abandoned in adults with ARDS. A multi-center clinical trial in children is ongoing. This article aims to review the physiologic rationale for the use of HFOV in patients with acute respiratory failure, summarize relevant bench and animal models, and discuss the potential use of HFOV as a primary and rescue mode in adults and children with severe respiratory failure.
Acute respiratory distress syndrome (ARDS) is a disease of acute onset characterized by significant hypoxemia and typical radiographic findings that affects both children and adults, and is an important cause of morbidity and mortality worldwide (Force et al., 2012; Pediatric Acute Lung Injury Consensus Conference, 2015; Thompson et al., 2017; Khemani et al., 2019). Whether pulmonary injury is the result of a direct (e.g., pneumonia, smoke inhalation, lung contusion) or indirect (e.g., sepsis, blood transfusion) insult, disease distribution in ARDS is heterogeneous, with more severe involvement of the dependent and relative sparing of the non-dependent lung regions (Ware and Matthay, 2000; Gattinoni et al., 2017b). This heterogeneous distribution of lung disease poses a challenge to the clinician instituting positive pressure ventilation, as different areas of the lung will have vastly different compliance and resistance. Non-dependent or uninjured alveoli (with better compliance) are at risk of overdistension, while dependent or injured alveoli (with worse compliance) are at risk of de-recruitment and repeated opening and closing with each respiratory cycle (Ware and Matthay, 2000; Gattinoni et al., 2017b).
When precisely employed, mechanical ventilation (MV) is a life-saving intervention, yet care must be taken to avoid ventilator-induced lung injury (VILI) (Tremblay and Slutsky, 2006; Beitler et al., 2016). Several factors have been identified as contributors to VILI. These include injury from the delivery of excessive pressure (barotrauma or stress) or tidal volume (VT) (volutrauma or strain), injury from the cyclic opening and closing of alveoli (atelectrauma), toxicity caused by high inspired fraction of inspired oxygen (FiO2), and injury resulting from cytokine release that can affect end-organ function (biotrauma) (Tremblay and Slutsky, 2006; Beitler et al., 2016). The landmark ARMA trial comparing mechanical ventilation with a tidal volume of 12 ml/kg to 6 ml/kg (both calculated using predicted body weight) found a significant mortality benefit with the application of a lower VT strategy and confirmed the role of high VT in VILI (Acute Respiratory Distress Syndrome et al., 2000). This study renewed interest in high-frequency oscillatory ventilation (HFOV) as an ultra-protective lung protective strategy capable of delivering very low VT. MV strategies aimed at avoiding VILI are termed “lung protective,” and generally operate in a theoretical “safe zone” on the deflating limb of the static pressure/volume curve (Figure 1).
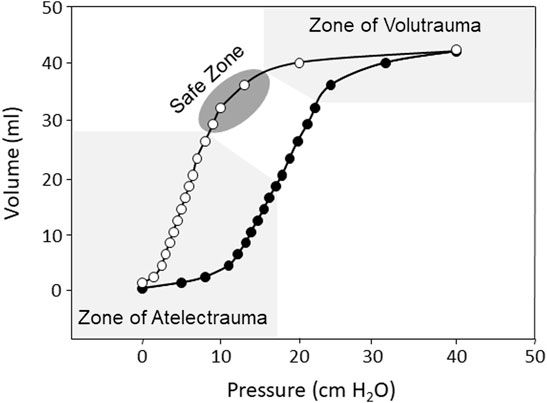
FIGURE 1. Representation of the inspiratory (black circles) and expiratory (white circles) static pressure-volume curves from a rabbit saline lavage model of ARDS showing hysteresis between the inspiratory and expiratory curves, the zone of volutrauma and atelectrauma (light gray), and the theoretical safe zone of ventilation (dark gray).
A study by Amato et al., published in 2015, elegantly illustrated the direct association between driving pressure (plateau pressure minus measured PEEP) and mortality in ARDS (Amato et al., 2015). Subsequent studies have also implicated driving pressure as a key variable that is associated with mortality in ARDS (Guerin et al., 2016; Goligher et al., 2021). Recent studies have highlighted the role of total energy delivered during each tidal breath to be an important factor in VILI (Gattinoni et al., 2017a). This concept, referred to as mechanical power, incorporates all mechanical ventilator settings, including respiratory rate, driving pressure, PEEP, and inspiratory flow (Gattinoni et al., 2017a). Mechanical power is an appealing concept because it accounts for the energy required to distend the lung, move gas, and maintain lung volume (Gattinoni et al., 2017a). The lung injury resultant from energy transmission from the various elements that determine mechanical power is termed ergotrauma. Indeed, mechanical power and the resultant ergotrauma have been directly associated with unfavorable outcomes in both adults and children with ARDS (Costa et al., 2021; Bhalla et al., 2022). A recent study found only respiratory rate and driving pressure to be independent predictors of mortality among variables included in the calculation for mechanical power (Costa et al., 2021). The simplified equation using these variables for estimation of mechanical power [(4 × ΔP) + RR] had a similar predictive value for mortality as mechanical power calculated using the more complex original method (Costa et al., 2021). Thus, mechanical power is an intriguing concept, but its utility as a modifiable parameter needs confirmation in a clinical trial.
Although lung protective ventilation can certainly be achieved through carefully conducted conventional mechanical ventilation (CMV), the lower VT, lower alveolar pressure swings, and higher mean airway pressure (mPaw) generally employed during various forms of high frequency ventilation make these modalities theoretically well suited for lung protection (Figure 2). There are four types of high frequency ventilation in clinical use: HFOV, high frequency jet ventilation, high-frequency percussive ventilation, and high frequency flow interruption (Keszler et al., 2015; Miller A. G. et al., 2021). This review will focus on the physiologic rationale for the use of HFOV in patients with acute respiratory failure, summarize data from relevant bench and animal models, and discuss the potential use of HFOV as a primary and rescue mode in adults and children with severe respiratory failure.
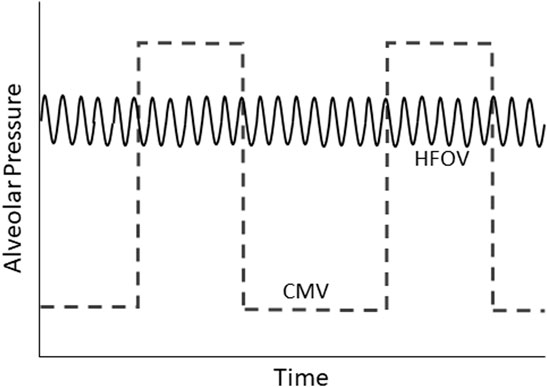
FIGURE 2. Schematic representation of alveolar pressure over time during conventional mechanical ventilation (CMV) and high-frequency oscillatory ventilation (HFOV).
HFOV is a form of MV that uses a constant distending pressure, usually reported as the mPaw, coupled with sinusoidal or square flow oscillations at supra-physiologic respiratory frequencies (Rettig et al., 2015; Miller A. G. et al., 2021). Respiratory frequencies used in clinical practice range from 5 to 15 Hz (i.e., 300 to 900 breaths per minute) with a small delivered VT, generally around 1–3 ml/kg, or lower than the anatomic dead space (Rettig et al., 2015; Miller A. G. et al., 2021). The constant distending pressure allows for alveolar recruitment while avoiding repetitive opening and closing of alveoli (atelectrauma), and has been shown to improve oxygenation (Rettig et al., 2015; Meyers et al., 2019). HFOV may also decrease the occurrence of volutrauma and barotrauma (Rettig et al., 2015).
HFOV differs from CMV and high frequency jet ventilation in that both inspiration and expiration are active (Miller A. G. et al., 2021; Miller A. G. et al., 2021). Oxygenation and ventilation are fairly independent during HFOV with oxygenation being controlled by FiO2 and mPaw while ventilation is controlled by VT (amplitude) and frequency (f) (Kneyber et al., 2012; Miller A. G. et al., 2021). Various mechanisms contribute to gas exchange during HFOV; these include gas flow turbulence in large airways, bulk convection, turbulent flow with radial mixing, pendelluft, asymmetric inspiratory and expiratory velocity profiles, Taylor dispersion, collateral ventilation, and cardiogenic mixing (Slutsky and Drazen, 2002; Pillow, 2005) (Figure 3).
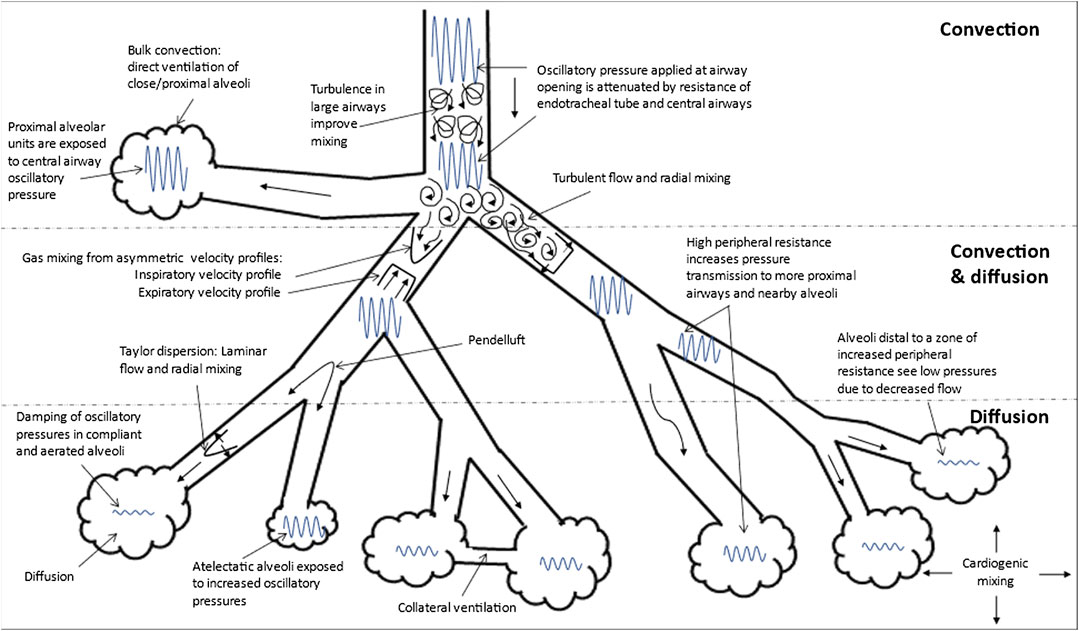
FIGURE 3. Gas Transport Mechanisms During High Frequency Oscillatory Ventilation (HFOV). Adapted from references: (Slutsky and Drazen, 2002; Pillow, 2005). The gas exchange mechanisms that function in each region (convection, convection and diffusion and diffusion alone) are shown. The various mechanisms that contribute to gas transport during HFOV are: 1) turbulence in large airways producing improved mixing; 2) bulk convection (direct ventilation of close alveoli); 3) turbulent flow with lateral convective mixing; 4) pendelluft (asynchronous flow among alveoli due to asymmetries in airflow impedance); 5) asymmetric inspiratory and expiratory velocity profiles (gas mixing due to velocity profiles that are axially asymmetric resulting in streaming of fresh gas toward alveoli along the inner wall of the airway and the streaming of alveolar gas away from the alveoli along the outer wall); 6) Taylor dispersion (laminar flow with lateral transport by diffusion); 7) collateral ventilation through non-airway connections between neighboring alveoli; and 8) cardiogenic mixing (rhythmic, pulsatile nature of the heart conferring a mixing of gases). The extent to which the oscillatory waveform is attenuated is also shown in this figure. Atelectatic alveoli will experience higher oscillatory pressure and lesser damping compared to normally aerated alveoli. Increase in peripheral resistance, other the other hand increase pressure transmission to more proximal airways and nearby alveoli such that alveoli distal to this zone of increased peripheral resistance experience lower pressures due to decreased flow.
The mechanism of gas exchange during HFOV varies according to the method of oscillation generation, attenuation of the pressure waveform, and efficiency of volume delivery (Pillow et al., 2001; John et al., 2014). HFOV produces biphasic pressure waveforms and diverts fresh bias flow to the patient at frequencies greater than 3 Hz (Keszler et al., 2015). The matching of positive and negative pressure deflections results in both inspiratory and expiration phases being active and can be achieved by a linear motor piston pump, an electromagnetically-driven vibrating diaphragm device, or an expiratory venturi jet (Keszler et al., 2015).
HFOV can be delivered via dedicated HFOV ventilator [e.g., Sensormedics 3100A and 3100B (Carefusion, Yorba Linda, California, United States)], or hybrid ventilators. Available HFOV ventilators and their mechanism of action are summarized in Table 1. Dedicated HFOV and hybrid ventilators differ in how they generate oscillations, ability to measure VT, availability of volume-targeted mode, range of settings for flow, pressure amplitude, frequency, and I:E ratio (Table 1) (Grazioli et al., 2015; Keszler et al., 2015; Tingay et al., 2015).
Oxygenation during HFOV is directly correlated with alveolar recruitment (i.e. alveolar surface area available for gas exchange), which is controlled largely by mPaw (Meyers et al., 2019). Optimizing mPaw strikes a balance between avoidance of de-recruitment and overdistension (Meyers et al., 2019).
Ventilation efficiency during HFOV (Q) can be expressed as (Slutsky et al., 1980; Boynton et al., 1989; Pillow, 2005):
VT is inversely proportional to frequency and directly proportional to amplitude (Sedeek et al., 2003; Miller A. G. et al., 2021). Therefore, the higher the frequency, the lower the VT; the higher the amplitude, the higher the VT (Sedeek et al., 2003; Miller A. G. et al., 2021). Similarly, a higher inspiratory time percentage results in increased ventilation due to higher VT (Miller A. G. et al., 2021). However, with the commonly used inspiratory time percentage of 33% [inspiratory (I) to expiratory (E) ratio of 1:2], the effect of decreasing amplitude has a greater impact on decreasing VT compared to increasing frequency (Sedeek et al., 2003). In contrast, with the inspiratory percentage set at 50% (I:E of 1:1), changes in frequency have a more pronounced effect on delivered VT compared to changes in amplitude (Sedeek et al., 2003). Ventilation is also affected by endotracheal tube (ETT) length and diameter, presence of a leak around the ETT, airway resistance, and respiratory system compliance (Van de Kieft et al., 2005). VT has been demonstrated in theoretical models, animals, and humans to have a greater effect on gas exchange than frequency during HFOV (Boynton et al., 1989; Pillow, 2005).
Diameter and length of the ETT affect VT delivery during HFOV. An increase in resistance is observed as ETT diameter deceases and as the length of the ETT increases, resulting in smaller delivered VT (Pillow et al., 2002; Van de Kieft et al., 2005; Custer et al., 2011). Creation of a cuff leak increases inhaled VT but reduces exhaled VT, and moves the source of fresh gas more distally towards the tip of the ETT (Van de Kieft et al., 2005; Van de Kieft et al., 2005; Bostick et al., 2012a). The Sensormedics 3100B has been shown to generate negative pressure during the exhalation and entrain CO2 into the inspiratory limb of the circuit, which can be reduced with the creation of cuff leak (Bostick et al., 2012a). Lastly, in a bench and clinical study, VT was found to be higher and CO2 elimination greater when the piston position for the Sensormedics 3100A was displaced towards the left compared to when the piston was in the center or displaced to the right for any given amplitude, frequency and inspiratory time (Hamel et al., 2005).
Airway resistance and compliance affect CO2 clearance during HFOV (Kneyber et al., 2012). The amplitude of the tracheal oscillatory pressure waveform decreases with increasing peripheral resistance, resulting in reduction of transmission of pressure over the airways to the alveoli (van Genderingen et al., 2001; Pillow, 2005; Kneyber et al., 2012). The opposite happens with reduced compliance, in which there is increase in pressure transmission to the alveoli and bronchi (van Genderingen et al., 2001; Pillow, 2005; Kneyber et al., 2012). Hence pressure transmission to the alveoli is the highest in patients with low compliance and low resistance.
Spontaneous breathing during HFOV may improve oxygenation and ventilation (van Heerde et al., 2009; van Heerde et al., 2010; Kneyber et al., 2012). However, due to limitations on maximal bias flow delivered during HFOV, spontaneous breathing may be challenging for older children who have higher inspiratory flow demands than the bias flow being delivered by the ventilator, resulting in an imposed increased work of breathing (van Heerde et al., 2006; Kneyber et al., 2012; Bordessoule et al., 2018). Hence, while neonates often tolerate spontaneous breathing during HFOV without the need for deep sedation or neuromuscular blockade, that is generally not the case for children and adults (Kneyber et al., 2012; Bordessoule et al., 2018).
While the physiology of gas exchange is similar between adults and children, some important differences exist between the two. Infants and children have shorter time constants, with resulting differing HFOV setting requirements. In general, children are managed with higher frequencies compared to adults, although significant variation in management exists (Arnold et al., 2000; de Jager et al., 2019). Thus, an infant on HFOV may be managed on a frequency of 10–12 Hz while an adult or larger child may require a frequency of 5–8 Hz. However, this will vary depending on the HFOV strategy used, severity of lung injury, and lung mechanics.
In clinical practice, oxygenation is managed by adjusting the mPaw or the fraction of inspired oxygen (FiO2). If oxygenation is below goal, the mPaw generally is increased in 1–2 cmH2O until oxygenation improves. Some centers may also employ a recruitment maneuver (e.g., rapid increase in mPaw by 10–15 cmH2O for 30–60 s) to improve oxygenation; although this strategy can result in hemodynamic compromise. Other strategies include incremental recruitment/decruitment maneuvers to find the optimal mPaw (de Jager et al., 2019). Paradoxically, excessive mPaw may result in worsening of oxygenation due to overdistension and in some cases a trial of decreased mPaw may be warranted. If oxygenation is above goal and the FiO2 is already within a non-toxic range (i.e., ≤0.50) mPaw generally is decreased in steps of 1–2 cmH2O as part of a weaning strategy. The frequency of adjustments is dependent upon patient characteristics and local practice.
Ventilation is controlled by the pressure amplitude and frequency (respiratory rate). To increase ventilation and decrease PaCO2, the amplitude can be increased or the frequency decreased. Different strategies are used depending on local practice, with some centers using a fixed frequency (higher frequencies being most protective) while adjusting amplitude to affect PaCO2, some maximize amplitude and use frequency as the main variable affecting ventilation, and others use a combination of the two strategies. The ideal method is unknown as direct comparisons have not been performed. Similar to oxygenation, overdistension may impair ventilation while lung recruitment can result in increased CO2 clearance without changes to frequency or amplitude.
Bench models of HFOV have demonstrated that adjustments to frequency have a larger effect on delivered VT than changes to amplitude (Van de Kieft et al., 2005; Wong et al., 2017). For instance, a 2 Hz increase in frequency results in a 21% decrease in VT, while a 10 cm H2O increase in pressure amplitude is necessary for an equivalent decrease in VT (Hager et al., 2007). When studied in patients, increasing frequency by 2 Hz decreased VT by 23%, while increasing amplitude by 10 cmH2O resulted in a 5.6% increase in VT (Hager et al., 2007). VT delivery also decreases as ETT size is reduced, due to the higher resistance across the smaller tube. Increasing bias flow from 20 to 30 L/min increases VT by 11% but this relationship is not linear; further increasing the bias flow from 30 to 40 L/min only results in a 3% increase in VT (Hager et al., 2007). Increasing bias flow has been shown to improve CO2 clearance up to 30 L/min (Nagano et al., 2018). CO2 clearance is most efficient with the R100 at 50% inspiratory time and least efficient with the 3100B at an inspiratory time 33% (Yumoto et al., 2019).
In the R100 ventilator with a 50% inspiratory time, VT was lower with smaller ETTs and higher frequencies (Hirao et al., 2009). When comparing VT delivery between the R100 and 3100B, VT was higher at similar settings with the R100 but comparable at 9 Hz and when inspiratory time was set at 50% on the 3100B (Iguchi et al., 2010). Other studies have found similar differences in VT delivery. s(Custer et al., 2011). Pressure delivery is attenuated when I:E is set at 1:2 using the 3100B (Hirayama et al., 2014).
Pendelluft has been observed between lung units, largely occurring during expiration (Lee et al., 2006). An adult bench model found negative pressure within the inspiratory limb of the circuit and CO2 rebreathing that became detectable when amplitude was >70 cmH2O and continued to increase as amplitude increased. CO2 rebreathing was eliminated by instituting an ETT cuff leak and increasing bias flow (Bostick et al., 2012b). Pressure amplitude significantly decreases throughout the respiratory system and this pressure attenuation is directly proportional to resistance increase and inversely proportional to compliance (Rozanek et al., 2012).
A computational model found that the resonant frequency of the non-injured lung is 8 Hz while the injured lung has a resonant frequency of 17 Hz (Herrmann et al., 2016). Due to the heterogeneous nature of disease distribution in most patients, individual lung sections may have different optimal frequencies, making the frequency section challenging in clinical practice (Herrmann et al., 2016).
Early animal models provided significant insights into the mechanisms of HFOV and were well summarized in a prior review (Kacmarek and Malhotra, 2005). These early models indicated that HFOV settings required to provide normocapnia were determined by (VT*f) n = 0.73*W*(VT/VL)−1.1 and that the f* VT during HFOV was higher than during CMV (Kacmarek and Malhotra, 2005). Early experiments also found that partial pressure of arterial CO2 (PaCO2) was held constant at different I:E ratio if mean lung volume and VT were held constant up to a frequency of 9 Hz. This is due to gas velocity profiles being unaffected by bulk flow rate. Additional models found that as VT increases, gas transport changes from dispersion (less efficient) to bulk gas flow (convection). Another study found that VT *f remained constant with frequencies of 3, 6, and 9 Hz. Regional gas distribution was most homogenous at 9 Hz compared to CMV and to lower frequencies. The ability to adequately exchange gas with HFOV was demonstrated by Bohn et al., in 1980 (Bohn et al., 1980). Another seminal study in HFOV found that VT *f was not the only determinant of CO2 clearance and that Taylor laminar and turbulent dispersion, pendelluft and asymmetrical velocity profiles were also factors. Early animal studies also showed that VT was directly related to amplitude and inversely proportional to frequency (Hz) (Kacmarek and Malhotra, 2005).
Studies using animal models have consistently found improved oxygenation with HFOV (Meyer et al., 2006; Ronchi et al., 2011; Li et al., 2015; Fioretto et al., 2019). However, HFOV did not offer an advantage over protective CMV for various markers of lung injury, both in a rabbit model of lung lavage (Rotta et al., 2001) or acid aspiration (Allardet-Servent et al., 2008). In an ex-vivo rabbit model of air leak, both stroke volume and mPaw influenced air leak flow, but mPaw appeared to be the main independent driver of air leak (Liu et al., 2007). Circuit disconnection was evaluated in a tween pig model of lung injury showing that disconnections resulted in worsening compliance and increase in FiO2 requirement, with these effects persisting over time (Kubiak et al., 2010). A study evaluating stepwise decreases in mPaw in a tween pig model of lung injury found that the titrated HFOV group had more atelectasis, fibrin, congestion, PMN invasion, and regional overdistension (Maggio et al., 2010).
In a rat model of saline lavage or lung injury from lipopolysaccharide administration, HFOV use resulted in decreased lung inflammation compared to CMV with low PEEP, but was similar to the decrease in inflammation observed in CMV with optimized PEEP (Krebs et al., 2010). A saline lavage model of sheep found that transpulmonary pressure was lowest at 9 Hz, which coincided with the lowest degree of lung inflammation (Liu et al., 2013). A porcine model of oleic acid lung injury found greater lung strain at lower frequency, with the lowest strain noted at a frequency of 20 Hz (Herrmann et al., 2020).
HFOV and protective MV have similar hemodynamic effects (Roosens et al., 2006). HFOV improves oxygenation without significant depression of cardiac function (Nakagawa et al., 2007). It also did not have deleterious effects on cerebral and systemic hemodynamics in a porcine model when mPaw was set 5 cmH2O above the CMV mPaw (Heuer et al., 2012). A porcine model of saline lavage found that mean arterial pressure and cardiac output increased during a decremental mPaw maneuver while central venous and wedge pressure decreased (Liu et al., 2020).
A study in pigs using saline lavage to cause severe lung injury found that normocapnia could not be achieved by HFOV or conventional CMV without extracorporeal CO2 removal (Brederlau et al., 2007). A similar study also found that high frequency improved lung recruitment (Muellenbach et al., 2008). HFOV with extracorporeal membrane oxygenation (ECMO) has been shown to attenuate lung inflammation in a saline lavage pig model compared to a pressure control strategy with a VT of 6 ml/kg (Muellenbach et al., 2010).
Spontaneous breathing during HFOV was evaluated using a custom demand flow valve in a saline lavage pig model of lung injury and found improved gas exchange with spontaneous breathing (van Heerde et al., 2009), possibly by shifting ventilation to more dependent lung zones (van Heerde et al., 2010). Transpulmonary pressure monitoring may help identify the lowest mPaw required to improve oxygenation and may result in fewer hemodynamic adverse effects of HFOV that occur when higher than necessary mPaw is employed (Karmrodt et al., 2006; Klapsing et al., 2018).
Early case series of HFOV in adults reported improvements in oxygenation with variable effects on hemodynamics (Fort et al., 1997; Claridge et al., 1999; Mehta et al., 2001). Subsequent studies confirmed these results, but most enrolled less than 50 subjects (Fort et al., 1997; Claridge et al., 1999; Mehta et al., 2001; Andersen et al., 2002; David et al., 2003; Mehta et al., 2004; Ferguson et al., 2005; Finkielman et al., 2006; Pachl et al., 2006; Fessler et al., 2008; Kao et al., 2011; Niwa et al., 2011; Camporota et al., 2013; Jog et al., 2013; Naorungroj et al., 2015; Thind et al., 2021). HFOV has also been described in three small case series of burn patients involving 6 to 30 subjects with mortality between 32 and 83% (Cartotto et al., 2001; Cartotto et al., 2004; Cartotto et al., 2009). Two of these studies showed an improvement in arterial partial pressure of oxygen (PaO2)/FiO2 (P/F) during HFOV (Cartotto et al., 2001; Cartotto et al., 2004), but oxygenation index (OI) only improved in subjects without inhalation injury (Cartotto et al., 2009). HFOV has also been used successfully in patients with elevated intracranial pressure (David et al., 2005), chronic obstructive pulmonary disease exacerbation failing CMV (Frerichs et al., 2012), and in conjunction with a extracorporeal CO2 removal (Lubnow et al., 2010).
The two largest case series included 156 (Mehta et al., 2004) and 102 (Camporota et al., 2013) subjects, with mortality rates of 63 and 48%, respectively. The first, published in 2004, found improvements in oxygenation during HFOV, and that mortality was associated with delayed HFOV initiation (Mehta et al., 2004). The second was published in 2013 and showed higher survival to be associated with younger age, greater initial improvement in P/F, and lower illness severity (Camporota et al., 2013).
While most studies have set the HFOV mPaw 3–5 cmH2O above the CMV mPaw, others have evaluated strategies to optimize lung volumes and set optimal mPaw (Ferguson et al., 2005). The TOOLs study evaluated the combination of HFOV and recruitment maneuvers in 25 adults with early ARDS. A recruitment maneuver (40 cmH2O for 40 s) was performed and mPaw was increased until FIO2 was <0.60, then targeted between 30 and 22 cmH2O before decreasing FIO2 (Ferguson et al., 2005). This resulted in significant improvements in P/F and OI, and ICU mortality was 44%. The recruitment maneuvers were well-tolerated, with 3.3% were stopped due to hemodynamic instability (Ferguson et al., 2005). Casserly et al. evaluated a method to determine the optimal mPaw in seven subjects and assessed changes in end-expiratory lung volume by measuring chest wall dimensions (Casserly et al., 2013). After a recruitment maneuver (40 cmH2O for 40 s), the mPaw was set at 35 cmH2O for 15 min, then reduced by 2.5 cmH2O every 15 min until the PaO2 was <60 mmHg or mPaw was 15 cmH2O. Lung volume was found to increase in a sigmoid shape, as did PaO2, although PaCO2 had a U-shaped curve as mPaw increased (Casserly et al., 2013).
In a study of 131 subjects with 60% mortality, HFOV was associated with significant increases in fentanyl, midazolam, and cisatracurium use, but no increase in propofol use over the first 4 days (Burry et al., 2013).
An early crossover study of HFOV in 16 adults with severe ARDS found that HFOV resulted in worsening right ventricular function and decreased cardiac index once mPaw was >5 cmH2O above the mPaw on CMV (Guervilly et al., 2012). Another study in 12 adults with ARDS found improvement in P/F with HFOV and tracheal insufflation with no difference in cardiac index and higher central venous saturation (Vrettou et al., 2014). The relationship between mPaw and esophageal pressure has been shown to be linear and highly correlated with set mPaw (Guervilly et al., 2016).
A crossover trial evaluated short-term prone positioning during HFOV compared to supine/prone CMV. Patients undergoing CMV had the PEEP set 2 cmH2O above the lower inflection point of the pressure-volume curve, while those undergoing HFOV had the mPaw was set 5 cmH2O above the CMV mPaw. Prone positioning improved P/F in both groups, but P/F did not improve during HFOV when patients were supine. Inflammatory markers were lower in the prone HFOV group (Papazian et al., 2005). A different crossover trial evaluating HFOV plus prone positioning in 43 subjects with ARDS found that HFOV maintained lung recruitment from prone positioning, and that P/F was higher in the HFOV prone and CMV prone groups (Papazian et al., 2005).
Due to the significant improvement in oxygenation observed in case series and observational studies, there was great enthusiasm for the use of HFOV as a strategy to improve mortality. Two early RCTs evaluated the efficacy of HFOV in adult ARDS. The MOAT trial conducted between 1997 and 2000 (Derdak et al., 2002) enrolled 148 subjects with ARDS, with 75 in the HFOV group and 73 in CMV group. Groups were similar at baseline, although neither group was receiving lung protective ventilation (PIP 39 vs. 38 cmH2O, VT 10.5 vs. 10.1 ml/kg) at enrollment. Mortality was 37% in the HFOV group and 52% in the CMV group but did not reach statistical significance. Survivors had lower OI after 24 h, regardless of group assignment (Derdak et al., 2002). Another RCT enrolled 61 subjects with ARDS from 1997–2001 (Bollen et al., 2005); it was stopped due to slow enrollment and found no difference in mortality between groups. Of note, the control group in this trial did not receive lung-protective ventilation (Bollen et al., 2005).
In 2013, the OSCAR and OSCILLATE trials were published (Ferguson et al., 2013; Young et al., 2013). The OSCAR trial randomized 397 subjects with ARDS (P/F < 200 on a minimum of 5 cmH2O PEEP, ventilated for <48 h) to HFOV and 398 to CMV in the United Kingdom. HFOV was set with a frequency of 10 Hz, mPaw 5 cmH2O above plateau pressure on CMV, bias flow 20 L/min, and an inspiratory time of 50%. Ventilation was managed to keep pH > 7.25 by maximizing the amplitude prior to adjusting the Hz, with a minimum of 5 Hz. The oxygenation target was a PaO2 between 60 and 75 mmHg. The CMV group was not controlled but centers were encouraged to target a VT 6–8 ml/kg and use a PEEP:FIO2 table. The groups were well-matched prior to randomization. This study showed no differences in mortality or ventilator-free days between the HFOV and CMV groups (Young et al., 2013).
The OSCILLATE trial randomized 548 subjects with ARDS and a P/F ≤ 200 with an FiO2 ≥ 0.50 to HFOV or CMV (Ferguson et al., 2013). HFOV was managed using a recruitment maneuver (40 cmH2O for 40 s) at initiation, then mPaw was set at 30 cmH2O and subsequently adjusted using a mPaw:FIO2 table, with the FIO2 needing to be ≤0.40 before the mPaw was decreased <30 cmH2O. The highest frequency possible was used to maintain pH > 7.25. Groups were similar at baseline, although vasopressor use was high in both groups at enrollment. Vasopressor and neuromuscular blockage use increased over time in the HFOV group. HFOV subjects received higher mPaw throughout and had a more positive fluid balance, while CMV subjects received a VT of 6.1 ml/kg and a PEEP of 18 cmH2O (Ferguson et al., 2013). This trial was stopped early due to higher mortality in subjects randomized to HFOV. A post-hoc analysis of 4 RCTs of HFOV found it likely to be harmful in mild to moderate ARDS but possibly beneficial in those with severe disease (P/F < 64) (Meade et al., 2017). The adult RCTs are summarized in Table 2.
Few studies have been published after the publication of OSCAR and OSCILLATE, and there has been a significant decrease in HFOV use in clinical practice (Tatham et al., 2021). A large cohort study of rescue strategies in adult patients with severe acute hypoxemic respiratory failure found that HFOV was used in 6% of all subjects and was the second most common rescue strategy after inhaled pulmonary vasodilators (Moreno Franco et al., 2016). A survey of critical care specialists found that only 8% would consider HFOV as rescue while 26% would likely never utilize it (Alhurani et al., 2016). Additional studies have shown HFOV use to be rare in the current era, with one reporting 4.3% of rescue cases (Duan et al., 2017), another reporting 1.7% of ventilator epochs (Jabaley et al., 2018), and only 10% of ICUs in the UK reporting any HFOV use (Jha et al., 2021). In 2021, the use of HFOV was reported in a single center study of 48 subjects with a OI 36 and a mortality rate of 92% (Thind et al., 2021).
Multiple meta-analyses following the publication of the OSCAR and OSCILLATE trials (Ferguson et al., 2013; Young et al., 2013) concluded that HFOV does not improve mortality compared to CMV; it also showed similar risk as CMV for barotrauma or hypotension but lower rate of treatment failure (Gu et al., 2014; Huang et al., 2014; Maitra et al., 2015; Sud et al., 2016; Goligher et al., 2017). Following those trials, multiple clinical practice guidelines (CPGs) recommended against the routine use of HFOV in the adult population (Claesson et al., 2015; Cho et al., 2016; Fan et al., 2017). A CPG from the American Thoracic Society, European Society of Intensive Care Medicine, and the Society of Critical Care Medicine suggested that future research on HFOV should employ lower mPaw strategies, have HFOV settings titrated to lung mechanics, and focus on its role as a rescue therapy (Fan et al., 2017).
Non-systematic reviews have suggested HFOV has not been shown to improve outcomes due to inadequately low frequency, excessive mPaw, and the need for neuromuscular blockade (Nguyen et al., 2016). Others have suggested the failure of HFOV was related to overdistension of non-injured alveoli, transmission of high mechanical power, excessive mPaw in subjects with poor recruitability, and adverse hemodynamics, particularly right ventricular dysfunction/failure (Dreyfuss et al., 2015; Sklar et al., 2017). The resonant frequency of the lung may also factor into patient outcomes, although identifying the optimal frequency in clinical practice is still a major challenge as it will vary among patients or even within the same patient at different stages of lung disease (Sklar et al., 2017). Despite limited data, HFOV may benefit patients with P/F < 65 and should be used cautiously in subjects with significant (pH < 7.23) respiratory acidosis (Sklar et al., 2017; Fielding-Singh et al., 2018).
Despite the strong physiologic rationale for its use and reproducible improvements in gas exchange, available data has not demonstrated a major outcome benefit for the routine use of HFOV in adults. Enthusiasm for HFOV has significantly decreased following the publication of the OSCAR and OSCILLATE trials, and its use has been largely abandoned in adult patients. This is despite subgroup analyses suggesting HFOV may have of benefit in profoundly hypoxemic patients (i.e., P/F is <64) (Sklar et al., 2017; Fielding-Singh et al., 2018). These RCTs enrolled subjects with a P/F < 200, used aggressive recruitment maneuvers, and the OSCILLATE trial enrolled a large number of subjects with hemodynamic instability requiring vasopressor, along with an aggressive mPaw protocol (Ferguson et al., 2013; Young et al., 2013). It is possible that these trials enrolled subjects who were not ill enough to benefit from HFOV, or that subjects with poorly recruitable lung were harmed by the high mPaw strategy employed (both from a barotrauma and preload-dependency standpoint). No clinical trials performed to date have evaluated lung recruitability prior to enrollment. In addition, the OSCAR trial did not control CMV in the control group, only had a single ventilator available for each site, used an HFOV ventilator with the inspiratory time fixed at 50%, and staff received limited education to use the ventilator (Young et al., 2013). Importantly, the management of HFOV is complex and can be affected by the ventilator used, specific HFOV strategy, staff education, and operator familiarity with the device or strategy. Together, available data illustrate the inherent challenges of studying complex interventions such as HFOV, and the fact that optimization of HFOV settings may not have been achieved in the adult RCTs. In addition, existing studies are limited by the inherent heterogeneity of diseases and concurrent treatment strategies employed in HFOV studies.
For adult patients, it is uncertain whether additional large clinical trials will be performed. If that were the case, future trials should focus on patients with a greater disease severity (P/F < 75 or pH < 7.20) and enroll patients in units where ECMO is not readily available. Trials should also attempt to include transpulmonary pressure monitoring and electric impedance tomography, along with evaluation of lung recruitability prior to enrollment. Caution should be exercised if incorporating recruitment maneuvers into a trial protocol, as these have not been shown to improve outcomes (Pensier et al., 2019; Ibarra-Estrada et al., 2021).
Early case series in children, published prior to 2000, found improved oxygenation, with variable effects on hemodynamics and mortality rates between 0 and 48% (Arnold et al., 1993; Goodman and Pollack, 1998; Duval et al., 1999; Fedora et al., 2000; Winters et al., 2000). These studies were small, single center studies that included patients receiving HFOV for a variety of indications and disease severity. These were followed by a large, multicenter study of 290 subjects that found a mortality rate of 32% in subjects with a P/F 75–90 and an OI 27–33 (Arnold et al., 2000). Oxygenation improved during HFOV and mortality was associated immunocompromised state, OI after 24 h of HFOV, sepsis, and chronic lung disease (Arnold et al., 2000). A subsequent study of 112 subjects in the era of lung-protective ventilation was unable to identify any risk factors for mortality but also found improved oxygenation during HFOV (Babbitt et al., 2012). A study of 34 subjects identified improvement in oxygenation and organ dysfunction score as independent predictors of mortality (Chattopadhyay et al., 2020).
Children often develop acute hypoxemic respiratory failure after hematopoietic stem cell transplant and 91% of centers performing stem cell transplants used HFOV as rescue (McArthur et al., 2011). A multi-center retrospective study on the use of HFOV in 85 children following hematopoietic stem cell transplant found that those treated with HFOV were 3 times more likely to die than those who did not receive HFOV (Rowan et al., 2016). They suggested that, when considering the use of HFOV in this population, it should be initiated within 5 days of respiratory failure. Another study of children with severe PARDS following hematopoietic stem cell transplantation found that HFOV use was associated with an odds ratio of 6.28 (95% confidence interval 1.16–34.12) for death (van Gestel et al., 2008).
A secondary analysis of the PARDIE study dataset evaluating rescue strategies for severe hypoxemia found that nearly all centers had HFOV available and used it in 9.3% of subjects (Khemani et al., 2019; Rowan et al., 2020). This rate was similar to the use of prone positioning (10%), slightly lower than inhaled nitric oxide (13%) but higher than ECMO (3%) (Rowan et al., 2020). HFOV was used more frequently in middle income countries, in patients with higher illness severity, and immunocompromised patients.
The feasibility of a physiologic, open-lung recruitment strategy of HFOV was described in 115 subjects treated in a single center in the Netherlands (de Jager et al., 2019). The HFOV strategy consisted of a starting frequency of 12 Hz for all subjects and used incremental-decremental staircase adjustments to select the “optimal” mPaw on the deflation limb of pressure-volume curve (de Jager et al., 2019). Ventilation was controlled by adjusting the frequency, but only after maximizing the power (amplitude) setting (de Jager et al., 2019). The reported mortality for different PARDS severity was similar to the mortality reported in the PARDIE study (Khemani et al., 2019). A different report from the same group found minimal hemodynamic effects, with 88% sensitivity and 54% specificity for changes in lung volumes (de Jager et al., 2020). This is the HFOV strategy currently being investigated as part of the PROSPECT trial (https://prospect-network.org/).
The first pediatric RCT of HFOV, published in 1994, enrolled 70 children ≤35 kg with an OI > 13, acute diffuse lung injury, or barotrauma (Arnold et al., 1994); it showed significant improvements in oxygenation compared to CMV, but no difference in survival (59% for CMV vs. 66% for HFOV). The OI was the strongest predictor of mortality with an OR of 20.8 (95% confidence interval 3.4 to 128.4, p < 0.001)and HFOV subjects were less likely to require oxygen at discharge (Arnold et al., 1994).
More recently, three propensity score matched analyses of HFOV compared to CMV have been published. The first used the Virtual PICU Systems database and compared early vs. late HFOV to CMV (Gupta et al., 2014). Subjects were matched for age, weight, CPR, severity of illness, ECMO, dialysis, arterial catheter, central access, hemodynamics, diagnoses. This study found that HFOV was associated with an increased length of MV, ICU length of stay, and mortality (Gupta et al., 2014). Importantly, this database did not collect crucial variables such as ventilator settings, gas exchange, and measures of oxygenation. As such, the adequacy of propensity matching (and, thus, the study findings) should be taken with caution. A reanalysis of the RESTORE trial dataset used propensity score matching to compare early HFOV with CMV or late HFOV (Bateman et al., 2016) and found no significant association with mortality; however, secondary analyses revealed early HFOV was associated with a higher mortality after accounting for risk category, with the 2 highest risk groups having increased mortality. Subjects in the HFOV group spend more time on the ventilator (Bateman et al., 2016). HFOV use increased as OI increased, with subjects more likely to be placed on HFOV once OI was >8, and those with an OI ≥ 40 were 17 times more likely to be placed on HFOV (Bateman et al., 2016). The Pediatric Acute & Critical Care Medicine Asian Network (PACCMAN) group performed a propensity matched study using a large multicenter database of pediatric ARDS and found that, compared to CMV, HFOV was associated with increased mortality and fewer ICU free days, but no difference for ventilator free days (Wong et al., 2020).
These studies are significantly limited as most did not record granular respiratory details such as plateau pressure, VT, driving pressure, and rationale for starting HFOV. Despite efforts to control for illness severity, it is likely that patients receiving HFOV were in fact sicker or failing CMV prior to placement on HFOV, thus resulting in mismatched acuity between the groups. Importantly, the presence of shock, vasopressor use, and renal failure were not included in the models.
Three additional RCTs have been published in recent years. A small trial, published in 2016, randomized 18 children with severe ARDS to HFOV (n = 9) or CMV (n = 9) with lung recruitment maneuvers. HFOV improved oxygenation and was well-tolerated hemodynamically. The overall survival was 89%, with one death in each group. Of note, 3 (33%) subjects randomized to the CMV group crossed over to HFOV (Samransamruajkit et al., 2016). The second trial, published in 2017, compared HFOV to protective CMV in 200 subjects with pediatric ARDS (El-Nawawy et al., 2017). HFOV resulted in improved oxygenation and more rapid increase in P/F but no differences in mortality (43% for CMV vs. 45% in HFOV), days of MV, OI difference after 24 h, and PICU length of stay (El-Nawawy et al., 2017).
A RCT of 61 infants with ARDS after high-risk atrial septal defect or ventricular septal defect repair compared HFOV to CMV; both groups also received surfactant replacement therapy (Zheng et al., 2021). The primary outcome was improvement in arterial blood gases. CMV strategy called for an inverse I:E ratio, with PIP 18–25 cmH2O and PEEP 4–-6 cmH2O but actual values were not reported. HFOV resulted in relatively small differences in PaO2, P/F, PaCO2, and OI. The HFOV group had shorter time on mechanical ventilation, ICU length of stay, and total hospital length of stay (Zheng et al., 2021). Pediatric RCTs are summarized in Table 3.
A recent systematic review of pediatric RCTs, propensity score matched studies, and observational studies failed to show an advantage of HFOV over CMV, with no demonstrable reduction in mortality, time on MV, or barotrauma (Junqueira et al., 2021). Of note, the GRADE certainty was low or very low for all studied outcomes (Junqueira et al., 2021). Recent non-systemic reviews suggest that, while HFOV strategies still require refinement, HFOV remains a viable rescue therapy for severe pediatric ARDS (Moerer et al., 2017; Nardi et al., 2017; Ng and Ferguson, 2017). Kneyber et al. advanced that HFOV was not optimized in prior RCTs (Kneyber et al., 2012) and suggested starting HFOV if SpO2 <88%, PaO2 < 50 mmHg with an FIO2 > 0.60 on sufficient support or in the presence of refractory respiratory acidosis. The suggested strategy involves increasing mPaw using incremental mPaw steps while following an expected rise in SpO2 until overdistension is observed. This is followed by a stepwise reduction in mPaw until the point of derecruitment, then the mPaw is set 2–4 cmH2O above this decruitment point (Kneyber et al., 2012). This strategy also advocates the use of the highest tolerable frequency (Hz) to minimize VT, and is currently being investigated in the PROSPECT trial (https://prospect-network.org/).
Similar to adults, available data do not support the use of HFOV in children with severe ARDS, despite consistently observed improvement in oxygenation. Unlike adults, however, these data are significantly limited by low quality RCTs and HFOV is still widely utilized in pediatric ICUs. Hopefully, the ongoing PROSPECT trial will provide more definitive answers on the role of HFOV in pediatric ARDS. Beyond the PROSPECT trial, the use of HFOV as a rescue strategy should be investigated, perhaps through a large registry or database that includes granular variables to allow for improved patient-level matching of relevant characteristics. Physiologic studies should include electric impedance tomography to evaluate the continuous relationship between lung volume and gas exchange. We await the results of the PROSPECT trial, an international multicenter, two-by-two factorial, response-adaptive RCT evaluating CMV and HFOV, along with prone and supine positions in children with severe hypoxemic respiratory failure.
Positive pressure ventilation can have negative effects on right ventricular (RV) function and overall hemodynamics, with some investigators suggesting this as a possible reason why RCTs of HFOV have failed to show an outcome benefit (Dreyfuss et al., 2015; Sklar et al., 2017). When lung volume is excessively increased, there is a potential for an increase in West zone 1 (ventilation with no perfusion) lung units, which results in higher RV afterload from an increase in pulmonary vascular resistance (PVR). Conversely, the underinflated lung (i.e., below functional residual capacity), also lead to increased PVR and increased RV afterload (Simmons et al., 1961). Thus, the use of high mPaw during HFOV may be detrimental to RV function due to increased PVR. Available data, however, do not show significant hemodynamic effects during stepwise recruitment and de-recruitment maneuvers (de Jager et al., 2020). Additionally, the elevated mPaw used during HFOV may adversely affect preload and result in the need for intravascular fluid expansion that can lead to volume overload.
An observational study found that, when HFOV was initiated with a mPaw 5 cmH2O above CMV mPaw, no differences in mean arterial pressure or heart rate were noted, but right atrial pressure increased, cardiac index slightly decreased, and left ventricular end-diastolic pressure decreased in a study of nine subjects with ARDS (David et al., 2004). Another study found HFOV did not appear to have a large effect on left and right ventricular function, but the cardiac index decreased by 13% when HFOV mPaw was set 5 cmH2O above the CMV mPaw (Ursulet et al., 2015). Additional studies found no association between body mass index and mortality, and that the higher mortality observed in the OSCILLATE was not related to hemodynamic changes 2 h after HFOV initiation (Tlayjeh et al., 2019; Angriman et al., 2020). One study found that an acute cor pulmonale score ≥2 and a P/F ≥ 100 were directly associated with mortality during HFOV (Angriman et al., 2020).
Management of HFOV is complex and requires advanced skills, device specific training/competency, physiologic understanding, and critical thinking as the learning curve for HFOV is steep. HFOV management is challenging even for teams experienced in its use. Data evaluating HFOV education are sparse. Deficits in basic MV management and assessment for asynchrony by critical care physicians has been reported (Colombo et al., 2011). A narrative review concluded that there is a paucity of information describing MV education in graduate medical education (Keller et al., 2019). Likewise, there is a dearth of guidance to facilitate staff education and verify competency of the end user. Simulation based training has shown promise in improving the outcome of learners as an addition to traditional didactic teaching (Cook et al., 2011). High-fidelity simulation has been shown to improve knowledge and skills related to MV in anesthesiology residents (Spadaro et al., 2017). There remains no standard approach to teach HFOV management nor to assess staff competency.
Staff education may be an underappreciated factor in prior clinical trials, particularly the OSCAR trial in which a new HFOV ventilator was used and some centers may have had limited experience with HFOV (Young et al., 2013). Education is even more critical when complex maneuvers, such as dynamic sustained inflation for lung recruitment or staircase titration of mPaw, are used to determine optimal mPaw (de Jager et al., 2019). Training and education in the use of HFOV settings, such as amplitude, require the user to assess subjective parameters such as “chest wiggle” as a surrogate for appropriate ventilation (Meyers et al., 2019). Inexperienced team members may have difficulty properly assessing the degree of chest wiggle or how to react appropriately, as frequency adjustments are counter-intuitive compared to CMV. Better feedback and assessment tools are needed to guide learning objectives and determine end-user competency. Future exploration into teaching and training methods utilizing HFOV are warranted; in the meantime, yearly education and competency assessment for centers that do not routinely utilize HFOV is suggested (Thind et al., 2021).
HFOV has largely been abandoned in adults following two large clinical trials despite a strong physiologic rationale and promising animal data. Available data do not support its routine use in adult or pediatric ARDS but it may have utility in more severe disease and pediatric data are of low quality. The mode is complex and patient outcomes may be affected by the ventilator used, HFOV strategy, and staff education.
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
AM receives a monthly stipend as a Section Editor for RESPIRATORY CARE. AR has received honoraria from Vapotherm and Breas US for consulting and lecturing.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Alhurani R. E., Oeckler R. A., Franco P. M., Jenkins S. M., Gajic O., Pannu S. R. (2016). Refractory Hypoxemia and Use of Rescue Strategies. A U.S. National Survey of Adult Intensivists. Ann. ATS 13 (7), 1105–1114. doi:10.1513/AnnalsATS.201508-560OC
Allardet-Servent J., Bregeon F., Delpierre S., Steinberg J.-G., Payan M.-J., Ravailhe S., et al. (2008). High-frequency Percussive Ventilation Attenuates Lung Injury in a Rabbit Model of Gastric Juice Aspiration. Intensive Care Med. 34 (1), 91–100. doi:10.1007/s00134-007-0848-z
Amato M. B. P., Meade M. O., Slutsky A. S., Brochard L., Costa E. L. V., Schoenfeld D. A., et al. (2015). Driving Pressure and Survival in the Acute Respiratory Distress Syndrome. N. Engl. J. Med. 372 (8), 747–755. doi:10.1056/NEJMsa1410639
Andersen F. A., Guttormsen A. B., Flaatten H. K. (2002). High Frequency Oscillatory Ventilation in Adult Patients with Acute Respiratory Distress Syndrome - a Retrospective Study. Acta Anaesthesiol Scand. 46 (9), 1082–1088. doi:10.1034/j.1399-6576.2002.460905.x
Angriman F., Ferreyro B. L., Donaldson L., Cuthbertson B. H., Ferguson N. D., Bollen C. W., et al. (2020). The Harm of High-Frequency Oscillatory Ventilation (HFOV) in ARDS Is Not Related to a High Baseline Risk of Acute Cor Pulmonale or Short-Term Changes in Hemodynamics. Intensive Care Med. 46 (1), 132–134. doi:10.1007/s00134-019-05806-8
Arnold J. H., Anas N. G., Luckett P., Cheifetz I. M., Reyes G., Newth C. J. L., et al. (2000). High-frequency Oscillatory Ventilation in Pediatric Respiratory Failure: a Multicenter Experience. Crit. Care Med. 28 (12), 3913–3919. doi:10.1097/00003246-200012000-00031
Arnold J. H., Hanson J. H., Toro-Figuero L. O., Gutiérrez J., Berens R. J., Anglin D. L. (1994). Prospective, Randomized Comparison of High-Frequency Oscillatory Ventilation and Conventional Mechanical Ventilation in Pediatric Respiratory Failure. Crit. Care Med. 22 (10), 1530–1539. doi:10.1097/00003246-199422100-00006
Arnold J. H., Truog R. D., Thompson J. E., Fackler J. C. (1993). High-frequency Oscillatory Ventilation in Pediatric Respiratory Failure. Crit. Care Med. 21 (2), 272–278. doi:10.1097/00003246-199302000-00021
Babbitt C. J., Cooper M. C., Nussbaum E., Liao E., Levine G. K., Randhawa I. S. (2012). High-frequency Oscillatory Ventilation in Pediatric Acute Hypoxemic Respiratory Failure: Disease-specific Morbidity Survival Analysis. Lung 190 (6), 685–690. doi:10.1007/s00408-012-9417-5
Bateman S. T., Borasino S., Asaro L. A., Cheifetz I. M., Diane S., Wypij D., et al. (2016). Early High-Frequency Oscillatory Ventilation in Pediatric Acute Respiratory Failure. A Propensity Score Analysis. Am. J. Respir. Crit. Care Med. 193 (5), 495–503. doi:10.1164/rccm.201507-1381OC
Beitler J. R., Malhotra A., Thompson B. T. (2016). Ventilator-induced Lung Injury. Clin. Chest Med. 37 (4), 633–646. doi:10.1016/j.ccm.2016.07.004
Bhalla A. K., Klein M. J., Modesto I Alapont V., Emeriaud G., Kneyber M. C. J., Medina A., et al. (2022). Mechanical Power in Pediatric Acute Respiratory Distress Syndrome: a PARDIE Study. Crit. Care 26 (1), 2. doi:10.1186/s13054-021-03853-6
Bohn D. J., Miyasaka K., Marchak B. E., Thompson W. K., Froese A. B., Bryan A. C. (1980). Ventilation by High-Frequency Oscillation. J. Appl. Physiol. 48 (4), 710–716. doi:10.1152/jappl.1980.48.4.710
Bollen C. W., van Well G. T. J., Sherry T., Beale R. J., Shah S., Findlay G., et al. (2005). High Frequency Oscillatory Ventilation Compared with Conventional Mechanical Ventilation in Adult Respiratory Distress Syndrome: a Randomized Controlled Trial [ISRCTN24242669]. Crit. Care 9 (4), R430–R439. doi:10.1186/cc3737
Bordessoule A., Piquilloud L., Lyazidi A., Moreira A., Rimensberger P. C. (2018). Imposed Work of Breathing during High-Frequency Oscillatory Ventilation in Spontaneously Breathing Neonatal and Pediatric Models. Respir. Care 63 (9), 1085–1093. doi:10.4187/respcare.05703
Bostick A. W., Naworol G. A., Britton T. J., Ori T. R., French S. K., Derdak S. (2012a). Inspiratory Limb Carbon Dioxide Entrainment during High-Frequency Oscillatory Ventilation: Characterization in a Mechanical Test Lung and Swine Model. Respir. Care 57 (11), 1865–1872. doi:10.4187/respcare.01563
Bostick A. W., Naworol G. A., Britton T. J., Ori T. R., French S. K., Derdak S. (2012b). Inspiratory Limb Carbon Dioxide Entrainment during High-Frequency Oscillatory Ventilation: Characterization in a Mechanical Test Lung and Swine Model. Respir. Care 57 (11), 1865–1872. doi:10.4187/respcare.01563
Boynton B. R., Hammond M. D., Fredberg J. J., Buckley B. G., Villanueva D., Frantz I. D. (1989). Gas Exchange in Healthy Rabbits during High-Frequency Oscillatory Ventilation. J. Appl. Physiol. 66 (3), 1343–1351. doi:10.1152/jappl.1989.66.3.1343
Brederlau J., Muellenbach R., Kredel M., Kuestermann J., Anetseder M., Greim C., et al. (2007). Combination of Arteriovenous Extracorporeal Lung Assist and High-Frequency Oscillatory Ventilation in a Porcine Model of Lavage-Induced Acute Lung Injury: a Randomized Controlled Trial. J. Trauma 62 (2), 336–346. doi:10.1097/01.ta.0000221667.32598.71
Acute Respiratory Distress Syndrome Brower R. G., Matthay M. A., Morris A., Schoenfeld D., Thompson B. T., Wheeler A. (2000). Ventilation with Lower Tidal Volumes as Compared with Traditional Tidal Volumes for Acute Lung Injury and the Acute Respiratory Distress Syndrome. N. Engl. J. Med. 342 (18), 1301–1308. doi:10.1056/NEJM200005043421801
Burry L. D., Seto K., Rose L., Lapinsky S. C., Mehta S. (2013). Use of Sedation and Neuromuscular Blockers in Critically Ill Adults Receiving High-Frequency Oscillatory Ventilation. Ann. Pharmacother. 47 (9), 1122–1129. doi:10.1177/1060028013503121
Camporota L., Sherry T., Smith J., Lei K., McLuckie A., Richard B. (2013). Physiological Predictors of Survival during High-Frequency Oscillatory Ventilation in Adults with Acute Respiratory Distress Syndrome. Crit. Care 17 (2), R40. doi:10.1186/cc12550
Cartotto R., Cooper A. B., Esmond J. R., Gomez M., Fish J. S., Smith T. (2001). Early Clinical Experience with High-Frequency Oscillatory Ventilation for ARDS in Adult Burn Patients. J. Burn Care Rehabil. 22 (5), 325–333. doi:10.1097/00004630-200109000-00006
Cartotto R., Ellis S., Gomez M., Cooper A., Smith T. (2004). High Frequency Oscillatory Ventilation in Burn Patients with the Acute Respiratory Distress Syndrome. Burns 30 (5), 453–463. doi:10.1016/j.burns.2004.01.015
Cartotto R., Walia G., Ellis S., Fowler R. (2009). Oscillation after Inhalation: High Frequency Oscillatory Ventilation in Burn Patients with the Acute Respiratory Distress Syndrome and Co-existing Smoke Inhalation Injury. J. Burn Care Res. 30 (1), 119–127. doi:10.1097/BCR.0b013e3181920fe6
Casserly B., Dennis McCool F., Sethi J. M., Kawar E., Read R., Levy M. M. (2013). A Method for Determining Optimal Mean Airway Pressure in High-Frequency Oscillatory Ventilation. Lung 191 (1), 69–76. doi:10.1007/s00408-012-9434-4
Chattopadhyay A., Gupta S., Sankar J., Kabra S. K., Lodha R. (2020). Outcomes of Severe PARDS on High-Frequency Oscillatory Ventilation - A Single Centre Experience. Indian J. Pediatr. 87 (3), 185–191. doi:10.1007/s12098-019-03134-9
Cho Y.-J., Moon J. Y., Shin E.-S., Kim J. H., Jung H., Park S. Y., et al. (2016). Clinical Practice Guideline of Acute Respiratory Distress Syndrome. Tuberc. Respir. Dis. 79 (4), 214–233. doi:10.4046/trd.2016.79.4.214
Claesson J., Freundlich M., Gunnarsson I., Laake J. H., Vandvik P. O., Varpula T., et al. (2015). S Candinavian Clinical Practice Guideline on Mechanical Ventilation in Adults with the Acute Respiratory Distress Syndrome. Acta Anaesthesiol Scand. 59 (3), 286–297. doi:10.1111/aas.12449
Claridge J. A., Hostetter R. G., Lowson S. M., Young J. S. (1999). High-frequency Oscillatory Ventilation Can Be Effective as rescue Therapy for Refractory Acute Lung Dysfunction. Am. Surg. 65 (11), 1092–1096.
Colombo D., Cammarota G., Alemani M., Carenzo L., Barra F. L., Vaschetto R., et al. (2011). Efficacy of Ventilator Waveforms Observation in Detecting Patient-Ventilator Asynchrony*. Crit. Care Med. 39 (11), 2452–2457. doi:10.1097/CCM.0b013e318225753c
Cook D. A., Hatala R., Brydges R., Zendejas B., Szostek J. H., Wang A. T., et al. (2011). Technology-Enhanced Simulation for Health Professions Education. JAMA 306 (9), 978–988. doi:10.1001/jama.2011.1234
Costa E. L. V., Slutsky A. S., Brochard L. J., Brower R., Serpa-Neto A., Cavalcanti A. B., et al. (2021). Ventilatory Variables and Mechanical Power in Patients with Acute Respiratory Distress Syndrome. Am. J. Respir. Crit. Care Med. 204 (3), 303–311. doi:10.1164/rccm.202009-3467OC
Custer J. W., Ahmed A., Kaczka D. W., Mulreany D. G., Hager D. N., Simon B. A., et al. (2011). In Vitro performance Comparison of the Sensormedics 3100A and B High-Frequency Oscillatory Ventilators. Pediatr. Crit. Care Med. 12 (4), e176–e180. doi:10.1097/PCC.0b013e3181fe3028
David M., Karmrodt J., Weiler N., Scholz A., Markstaller K., Eberle B. (2005). High-frequency Oscillatory Ventilation in Adults with Traumatic Brain Injury and Acute Respiratory Distress Syndrome. Acta Anaesthesiol Scand. 49 (2), 209–214. doi:10.1111/j.1399-6576.2004.00570.x
David M., von Bardeleben R. S., Weiler N., Markstaller K., Scholz A., Karmrodt J., et al. (2004). Cardiac Function and Haemodynamics during Transition to High-Frequency Oscillatory Ventilation. Eur. J. Anaesthesiology 21 (12), 944–952. doi:10.1017/s026502150400032810.1097/00003643-200412000-00004
David M., Weiler N., Heinrichs W., Neumann M., Joost T., Markstaller K., et al. (2003). High-frequency Oscillatory Ventilation in Adult Acute Respiratory Distress Syndrome. Intensive Care Med. 29 (10), 1656–1665. doi:10.1007/s00134-003-1897-6
de Jager P., Burgerhof J. G. M., Koopman A. A., Markhorst D. G., Kneyber M. C. J. (2020). Physiologic Responses to a Staircase Lung Volume Optimization Maneuver in Pediatric High-Frequency Oscillatory Ventilation. Ann. Intensive Care 10 (1), 153. doi:10.1186/s13613-020-00771-8
de Jager P., Kamp T., Dijkstra S. K., Burgerhof J. G. M., Markhorst D. G., Curley M. A. Q., et al. (2019). Feasibility of an Alternative, Physiologic, Individualized Open-Lung Approach to High-Frequency Oscillatory Ventilation in Children. Ann. Intensive Care 9 (1), 9. doi:10.1186/s13613-019-0492-0
Derdak S., Mehta S., Stewart T. E., Smith T., Rogers M., Buchman T. G., et al. (2002). High-Frequency Oscillatory Ventilation for Acute Respiratory Distress Syndrome in Adults. Am. J. Respir. Crit. Care Med. 166 (6), 801–808. doi:10.1164/rccm.2108052
Dreyfuss D., Ricard J.-D., Gaudry S. (2015). Did Studies on HFOV Fail to Improve ARDS Survival Because They Did Not Decrease VILI? on the Potential Validity of a Physiological Concept Enounced Several Decades Ago. Intensive Care Med. 41 (12), 2076–2086. doi:10.1007/s00134-015-4062-0
Duan E. H., Adhikari N. K. J., D’Aragon F., Cook D. J., Mehta S., Alhazzani W., et al. (2017). Management of Acute Respiratory Distress Syndrome and Refractory Hypoxemia. A Multicenter Observational Study. Ann. ATS 14 (12), 1818–1826. doi:10.1513/AnnalsATS.201612-1042OC
Duval E. L. I. M., Leroy P. L. J. M., Gemke R. J. B. J., van Vught A. J. (1999). High-frequency Oscillatory Ventilation in RSV Bronchiolitis Patients. Respir. Med. 93 (6), 435–440. doi:10.1053/rmed.1999.0578
El-Nawawy A., Moustafa A., Heshmat H., Abouahmed A. (2017). High Frequency Oscillatory Ventilation versus Conventional Mechanical Ventilation in Pediatric Acute Respiratory Distress Syndrome: A Randomized Controlled Study. TurkJPediatr 59 (2), 130–143. doi:10.24953/turkjped.2017.02.004
Fan E., Del Sorbo L., Goligher E. C., Hodgson C. L., Munshi L., Walkey A. J., et al. (2017). An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: Mechanical Ventilation in Adult Patients with Acute Respiratory Distress Syndrome. Am. J. Respir. Crit. Care Med. 195 (9), 1253–1263. doi:10.1164/rccm.201703-0548ST
Fedora M., Klimovic M., Seda M., Dominik P., Nekvasil R. (2000). Effect of Early Intervention of High-Frequency Oscillatory Ventilation on the Outcome in Pediatric Acute Respiratory Distress Syndrome. Bratisl Lek Listy 101 (1), 8–13.
Ferguson N. D., Chiche J.-D., Kacmarek R. M., Hallett D. C., Mehta S., Findlay G. P., et al. (2005). Combining High-Frequency Oscillatory Ventilation and Recruitment Maneuvers in Adults with Early Acute Respiratory Distress Syndrome: The Treatment with Oscillation and an Open Lung Strategy (TOOLS) Trial Pilot Study*. Crit. Care Med. 33 (3), 479–486. doi:10.1097/01.ccm.0000155785.23200.9e
Ferguson N. D., Cook D. J., Guyatt G. H., Mehta S., Hand L., Austin P., et al. (2013). High-frequency Oscillation in Early Acute Respiratory Distress Syndrome. N. Engl. J. Med. 368 (9), 795–805. doi:10.1056/NEJMoa1215554
Fessler H. E., Hager D. N., Brower R. G. (2008). Feasibility of Very High-Frequency Ventilation in Adults with Acute Respiratory Distress Syndrome*. Crit. Care Med. 36 (4), 1043–1048. doi:10.1097/01.CCM.0b013e318168fcab
Fielding-Singh V., Matthay M. A., Calfee C. S. (2018). Beyond Low Tidal Volume Ventilation. Crit. Care Med. 46 (11), 1820–1831. doi:10.1097/CCM.0000000000003406
Finkielman J. D., Gajic O., Farmer J. C., Afessa B., Hubmayr R. D. (2006). The Initial Mayo Clinic Experience Using High-Frequency Oscillatory Ventilation for Adult Patients: a Retrospective Study. BMC Emerg. Med. 6, 2. doi:10.1186/1471-227X-6-2
Fioretto J. R., Pires R. B., Klefens S. O., Kurokawa C. S., Carpi M. F., Bonatto R. C., et al. (2019). Inflammatory Lung Injury in Rabbits: Effects of High-Frequency Oscillatory Ventilation in the Prone Position. J. Bras. Pneumol. 45 (5), e20180067. doi:10.1590/1806-3713/e20180067
Force A. D. T., Ranieri V. M., Rubenfeld G. D., Thompson B. T., Ferguson N. D., Caldwell E., et al. (2012). Acute Respiratory Distress Syndrome. JAMA 307 (23), 2526–2533. doi:10.1001/jama.2012.5669
Fort P., Farmer C., Westerman J., Johannigman J., Beninati W., Dolan S., et al. (1997). High-frequency Oscillatory Ventilation for Adult Respiratory Distress Syndrome-A Pilot Study. Crit. Care Med. 25 (6), 937–947. doi:10.1097/00003246-199706000-00008
Frerichs I., Achtzehn U., Pechmann A., Pulletz S., Schmidt E. W., Quintel M., et al. (2012). High-frequency Oscillatory Ventilation in Patients with Acute Exacerbation of Chronic Obstructive Pulmonary Disease. J. Crit. Care 27 (2), 172–181. doi:10.1016/j.jcrc.2011.04.008
Gattinoni L., Marini J. J., Collino F., Maiolo G., Rapetti F., Tonetti T., et al. (2017a). The Future of Mechanical Ventilation: Lessons from the Present and the Past. Crit. Care 21 (1), 183. doi:10.1186/s13054-017-1750-x
Gattinoni L., Tonetti T., Quintel M. (2017b). Regional Physiology of ARDS. Crit. Care 21 (Suppl. 3), 312. doi:10.1186/s13054-017-1905-9
Goligher E. C., Costa E. L. V., Yarnell C. J., Brochard L. J., Stewart T. E., Tomlinson G., et al. (2021). Effect of Lowering Vt on Mortality in Acute Respiratory Distress Syndrome Varies with Respiratory System Elastance. Am. J. Respir. Crit. Care Med. 203 (11), 1378–1385. doi:10.1164/rccm.202009-3536OC
Goligher E. C., Munshi L., Adhikari N. K. J., Meade M. O., Hodgson C. L., Wunsch H., et al. (2017). High-Frequency Oscillation for Adult Patients with Acute Respiratory Distress Syndrome. A Systematic Review and Meta-Analysis. Ann. ATS 14 (Suppl. ment_4), S289–S296. doi:10.1513/AnnalsATS.201704-341OT
Goodman A. M., Pollack M. M. (1998). Hemodynamic Effects of High-Frequency Oscillatory Ventilation in Children. Pediatr. Pulmonol. 25 (6), 371–374. doi:10.1002/(sici)1099-0496(199806)25:6<371::aid-ppul3>3.0.co;2-k
Grazioli S., Karam O., Rimensberger P. C. (2015). New Generation Neonatal High Frequency Ventilators: Effect of Oscillatory Frequency and Working Principles on Performance. Respir. Care 60 (3), 363–370. doi:10.4187/respcare.03048
Gu X.-l., Wu G.-n., Yao Y.-w., Shi D.-h., Song Y. (2014). Is High-Frequency Oscillatory Ventilation More Effective and Safer Than Conventional Protective Ventilation in Adult Acute Respiratory Distress Syndrome Patients? A Meta-Analysis of Randomized Controlled Trials. Crit. Care 18 (3), R111. doi:10.1186/cc13900
Guérin C., Papazian L., Papazian L., Reignier J., Ayzac L., Loundou A., et al. (2016). Effect of Driving Pressure on Mortality in ARDS Patients during Lung Protective Mechanical Ventilation in Two Randomized Controlled Trials. Crit. Care 20 (1), 384. doi:10.1186/s13054-016-1556-2
Guervilly C., Forel J.-M., Hraiech S., Demory D., Allardet-Servent J., Adda M., et al. (2012). Right Ventricular Function during High-Frequency Oscillatory Ventilation in Adults with Acute Respiratory Distress Syndrome. Crit. Care Med. 40 (5), 1539–1545. doi:10.1097/CCM.0b013e3182451b4a
Guervilly C., Forel J.-M., Hraiech S., Roch A., Talmor D., Papazian L. (2016). Effect of High-Frequency Oscillatory Ventilation on Esophageal and Transpulmonary Pressures in Moderate-To-Severe Acute Respiratory Distress Syndrome. Ann. Intensive Care 6 (1), 84. doi:10.1186/s13613-016-0181-1
Gupta P., Green J. W., Tang X., Gall C. M., Gossett J. M., Rice T. B., et al. (2014). Comparison of High-Frequency Oscillatory Ventilation and Conventional Mechanical Ventilation in Pediatric Respiratory Failure. JAMA Pediatr. 168 (3), 243–249. doi:10.1001/jamapediatrics.2013.4463
Hager D. N., Fessler H. E., Kaczka D. W., Shanholtz C. B., Fuld M. K., Simon B. A., et al. (2007). Tidal Volume Delivery during High-Frequency Oscillatory Ventilation in Adults with Acute Respiratory Distress Syndrome*. Crit. Care Med. 35 (6), 1522–1529. doi:10.1097/01.CCM.0000266586.04676.55
Hamel D. S., Katz A. L., Craig D. M., Davies J. D., Cheifetz I. M. (2005). Carbon Dioxide Elimination and Gas Displacement Vary with Piston Position during High-Frequency Oscillatory Ventilation. Respir. Care 50 (3), 361–366.
Herrmann J., Gerard S. E., Shao W., Hawley M. L., Reinhardt J. M., Christensen G. E., et al. (2020). Quantifying Regional Lung Deformation Using Four-Dimensional Computed Tomography: A Comparison of Conventional and Oscillatory Ventilation. Front. Physiol. 11, 14. doi:10.3389/fphys.2020.00014
Herrmann J., Tawhai M. H., Kaczka D. W. (2016). Regional Gas Transport in the Heterogeneous Lung during Oscillatory Ventilation. J. Appl. Physiol. 121 (6), 1306–1318. doi:10.1152/japplphysiol.00097.2016
Heuer J. F., Sauter P., Barwing J., Herrmann P., Crozier T. A., Bleckmann A., et al. (2012). Effects of High-Frequency Oscillatory Ventilation on Systemic and Cerebral Hemodynamics and Tissue Oxygenation: an Experimental Study in Pigs. Neurocrit. Care 17 (2), 281–292. doi:10.1007/s12028-011-9566-z
Hirao O., Iguchi N., Uchiyama A., Mashimo T., Nishimura M., Fujino Y. (2009). Influence of Endotracheal Tube Bore on Tidal Volume during High Frequency Oscillatory Ventilation: a Model Lung Study. Med. Sci. Monit. 15 (1), MT1–4.
Hirayama T., Nagano O., Shiba N., Yumoto T., Sato K., Terado M., et al. (2014). Mean Lung Pressure during Adult High-Frequency Oscillatory Ventilation: an Experimental Study Using a Lung Model. Acta Med. Okayama 68 (6), 323–329. doi:10.18926/AMO/53021
Huang C.-T., Lin H.-H., Ruan S.-Y., Lee M.-S., Tsai Y.-J., Yu C.-J. (2014). Efficacy and Adverse Events of High-Frequency Oscillatory Ventilation in Adult Patients with Acute Respiratory Distress Syndrome: a Meta-Analysis. Crit. Care 18 (3), R102. doi:10.1186/cc13880
Ibarra-Estrada M. Á., García-Salas Y., Mireles-Cabodevila E., López-Pulgarín J. A., Chávez-Peña Q., García-Salcido R., et al. (2021). Use of Airway Pressure Release Ventilation in Patients with Acute Respiratory Failure Due to Coronavirus Disease 2019. Crit. Care Med. Publish Ahead of Print. doi:10.1097/CCM.0000000000005312
Iguchi N., Hirao O., Uchiyama A., Mashimo T., Nishimura M., Fujino Y. (2010). Evaluation of Performance of Two High-Frequency Oscillatory Ventilators Using a Model Lung with a Position Sensor. J. Anesth. 24 (6), 888–892. doi:10.1007/s00540-010-1032-0
Jabaley C. S., Groff R. F., Sharifpour M., Raikhelkar J. K., Blum J. M. (2018). Modes of Mechanical Ventilation Vary between Hospitals and Intensive Care Units within a university Healthcare System: a Retrospective Observational Study. BMC Res. Notes 11 (1), 425. doi:10.1186/s13104-018-3534-z
Jha A., Vasques F., Sanderson B., Daly K., Glover G., Ioannou N., et al. (2021). A Survey on the Practices and Capabilities in the Management of Respiratory Failure in South East England. J. Intensive Care Soc. 22 (2), 175–181. doi:10.1177/1751143720928895
Jog S., Patel D., Dravid T., Rajhans P., Akole P., Pawar B., et al. (2013). Early Application of High Frequency Oscillatory Ventilation in 'H1N1 Influenza' Related ARDS Is Associated with Better Outcome: a Retrospective Study. Intensive Care Med. 39 (6), 1146–1147. doi:10.1007/s00134-013-2878-z
John J., Harcourt E. R., Davis P. G., Tingay D. G. (2014). Dräger VN500's Oscillatory Performance Has a Frequency-dependent Threshold. J. Paediatr. Child. Health 50 (1), 27–31. doi:10.1111/jpc.12398
Junqueira F. M. D., Nadal J. A. H., Brandão M. B., Nogueira R. J. N., Souza T. H. (2021). High‐frequency Oscillatory Ventilation in Children: A Systematic Review and Meta‐analysis. Pediatr. Pulmonology 56 (7), 1872–1888. doi:10.1002/ppul.25428
Kacmarek R. M., Malhotra A. (2005). High-frequency Oscillatory Ventilation: What Large-Animal Studies Have Taught Us! Crit. Care Med. 33 (3 Suppl. l), S148–S154. doi:10.1097/01.ccm.0000156786.43935.a0
Kao K.-C., Yang C.-T., Hu H.-C., Ting H.-C., Huang C.-T., Chou L.-T., et al. (2011). Resolution of Organ Functional Scores to Predict the Outcome in Adult Acute Respiratory Distress Syndrome Patients Receiving High-Frequency Oscillatory Ventilation. J. Trauma 71 (6), 1536–1542. doi:10.1097/TA.0b013e3182332102
Karmrodt J., David M., Yuan S., Markstaller K. (2006). Alternative Protocol to Initiate High-Frequency Oscillatory Ventilation: an Experimental Study. Crit. Care 10 (5), R138. doi:10.1186/cc5052
Keller J. M., Claar D., Ferreira J. C., Chu D. C., Hossain T., Carlos W. G., et al. (2019). Mechanical Ventilation Training during Graduate Medical Education: Perspectives and Review of the Literature. J. Grad Med. Educ. 11 (4), 389–401. doi:10.4300/JGME-D-18-00828.1
Keszler M., Pillow J. J., Courtney S. E. (2015). Pediatric and Neonatal Mechanical Ventilation. New York: Springer.
Khemani R. G., Smith L., Lopez-Fernandez Y. M., Kwok J., Morzov R., Klein M. J., et al. (2019). Paediatric Acute Respiratory Distress Syndrome Incidence and Epidemiology (PARDIE): an International, Observational Study. Lancet Respir. Med. 7 (2), 115–128. doi:10.1016/S2213-2600(18)30344-8
Klapsing P., Moerer O., Wende C., Herrmann P., Quintel M., Bleckmann A., et al. (2018). High-frequency Oscillatory Ventilation Guided by Transpulmonary Pressure in Acute Respiratory Syndrome: an Experimental Study in Pigs. Crit. Care 22 (1), 121. doi:10.1186/s13054-018-2028-7
Kneyber M. C., van Heerde M., Markhorst D. G. (2012). Reflections on Pediatric High-Frequency Oscillatory Ventilation from a Physiologic Perspective. Respir. Care 57 (9), 1496–1504. doi:10.4187/respcare.01571
Krebs J., Pelosi P., Tsagogiorgas C., Zoeller L., Rocco P. R., Yard B., et al. (2010). Open Lung Approach Associated with High-Frequency Oscillatory or Low Tidal Volume Mechanical Ventilation Improves Respiratory Function and Minimizes Lung Injury in Healthy and Injured Rats. Crit. Care 14 (5), R183. doi:10.1186/cc9291
Kubiak B. D., Albert S. P., Gatto L. A., Trikha G., El-Zammar O., Nieman G. F. (2010). Loss of Airway Pressure during HFOV Results in an Extended Loss of Oxygenation: a Retrospective Animal Study. J. Surg. Res. 162 (2), 250–257. doi:10.1016/j.jss.2009.04.026
Lee W. J., Kawahashi M., Hirahara H. (2006). Experimental Analysis of Pendelluft Flow Generated by HFOV in a Human Airway Model. Physiol. Meas. 27 (8), 661–674. doi:10.1088/0967-3334/27/8/001
Li Q.-j., Yuan Y., Li Y.-m., Sun L.-y., Yuan S.-y. (2015). Effect of High Frequency Oscillatory Ventilation on EVLW and Lung Capillary Permeability of Piglets with Acute Respiratory Distress Syndrome Caused by Pulmonary and Extrapulmonary Insults. J. Huazhong Univ. Sci. Technol. [Med. Sci. 35 (1), 93–98. doi:10.1007/s11596-015-1395-4
Liu S., Yi Y., Wang M., Chen Q., Huang Y., Liu L., et al. (2013). Higher Frequency Ventilation Attenuates Lung Injury during High-Frequency Oscillatory Ventilation in Sheep Models of Acute Respiratory Distress Syndrome. Anesthesiology 119 (2), 398–411. doi:10.1097/ALN.0b013e31829419a6
Liu S., Zhao Z., Tan L., Wang L., Möller K., Frerichs I., et al. (2020). Optimal Mean Airway Pressure during High-Frequency Oscillatory Ventilation in an Experimental Model of Acute Respiratory Distress Syndrome: EIT-Based Method. Ann. Intensive Care 10 (1), 31. doi:10.1186/s13613-020-0647-z
Liu T.-Y., Lien S.-H., Hua Y.-M., Soong W.-J., Lu P., Yuh Y.-S. (2007). Determinants of Airleak Flow during Humming V High-Frequency Oscillatory Ventilation in an Open-Compartment Ex-Vivo Model of Airleak. Pediatr. Pulmonol. 42 (12), 1104–1109. doi:10.1002/ppul.20669
Lubnow M., Luchner A., Philipp A., Buchner S., Jeron A., Karagiannidis C., et al. (2010). Combination of High Frequency Oscillatory Ventilation and Interventional Lung Assist in Severe Acute Respiratory Distress Syndrome. J. Crit. Care 25 (3), 436–444. doi:10.1016/j.jcrc.2009.11.004
Maggio D., Sadowitz B., Vieau C., Albert S. P., Kubiak B. D., Roy S., et al. (2010). Titration of Mean Airway Pressure and FiO2 during High Frequency Oscillatory Ventilation in a Porcine Model of Acute Lung Injury. J. Surg. Res. 164 (1), e147–e153. doi:10.1016/j.jss.2010.07.002
Maitra S., Bhattacharjee S., Khanna P., Baidya D. K. (2015). High-frequency Ventilation Does Not Provide Mortality Benefit in Comparison with Conventional Lung-Protective Ventilation in Acute Respiratory Distress Syndrome. Anesthesiology 122 (4), 841–851. doi:10.1097/ALN.0000000000000306
McArthur J., Pettersen G., Jouvet P., Christensen M., Tamburro R. (2011). The Care of Critically Ill Children after Hematopoietic SCT: a North American Survey. Bone Marrow Transpl. 46 (2), 227–231. doi:10.1038/bmt.2010.89
Meade M. O., Young D., Hanna S., Zhou Q., Bachman T. E., Bollen C., et al. (2017). Severity of Hypoxemia and Effect of High-Frequency Oscillatory Ventilation in Acute Respiratory Distress Syndrome. Am. J. Respir. Crit. Care Med. 196 (6), 727–733. doi:10.1164/rccm.201609-1938OC
Mehta S., Granton J., MacDonald R. J., Bowman D., Matte-Martyn A., Bachman T., et al. (2004). High-Frequency Oscillatory Ventilation in Adults. Chest 126 (2), 518–527. doi:10.1378/chest.126.2.518
Mehta S., Lapinsky S. E., Hallett D. C., Merker D., Groll R. J., Cooper A. B., et al. (2001). Prospective Trial of High-Frequency Oscillation in Adults with Acute Respiratory Distress Syndrome. Crit. Care Med. 29 (7), 1360–1369. doi:10.1097/00003246-200107000-00011
Meyer J., Cox P. N., McKerlie C., Bienzle D. (2006). Protective Strategies of High-Frequency Oscillatory Ventilation in a Rabbit Model. Pediatr. Res. 60 (4), 401–406. doi:10.1203/01.pdr.0000238248.42925.f4
Miller A. G., Bartle R. M., Feldman A., Mallory P., Reyes E., Scott B., et al. (2021a). A Narrative Review of Advanced Ventilator Modes in the Pediatric Intensive Care Unit. Translational Pediatr. 10, 2700–2719. doi:10.21037/tp-20-332
Miller A. G., Bartle R. M., Rehder K. J. (2021b). High-frequency Jet Ventilation in Neonatal and Pediatric Subjects: A Narrative Review. Respir. Care 66 (6), 845–856. doi:10.4187/respcare.08691
Moerer O., Tonetti T., Quintel M. (2017). Rescue Therapies for Acute Respiratory Distress Syndrome. Curr. Opin. Crit. Care 23 (1), 52–59. doi:10.1097/MCC.0000000000000374
Moreno Franco P., Enders F., Wilson G., Gajic O., Pannu S. R. (2016). A Comparative Effectiveness Study of Rescue Strategies in 1,000 Subjects with Severe Hypoxemic Respiratory Failure. Respir. Care 61 (2), 127–133. doi:10.4187/respcare.04162
Muellenbach R. M., Kredel M., Wilhelm J., Küstermann J., Fink L., Siebenliest G., et al. (2010). High-frequency Oscillation Combined with Arteriovenous Extracorporeal Lung Assist Reduces Lung Injury. Exp. Lung Res. 36 (3), 148–158. doi:10.3109/01902140903214683
Muellenbach R. M., Kuestermann J., Kredel M., Johannes A., Wolfsteiner U., Schuster F., et al. (2008). Arteriovenous Extracorporeal Lung Assist Allows for Maximization of Oscillatory Frequencies: A Large-Animal Model of Respiratory Distress. BMC Anesthesiol 8, 7. doi:10.1186/1471-2253-8-7
Myers M., Rodrigues N., Ari A. (2019). High-frequency Oscillatory Ventilation: A Narrative Review. Cjrt 55, 40–46. doi:10.29390/cjrt-2019-004
Nagano O., Yumoto T., Nishimatsu A., Kanazawa S., Fujita T., Asaba S., et al. (2018). Bias Flow Rate and Ventilation Efficiency during Adult High-Frequency Oscillatory Ventilation: a Lung Model Study. ICMx 6 (1), 11. doi:10.1186/s40635-018-0176-3
Nakagawa R., Koizumi T., Ono K., Tsushima K., Yoshikawa S., Kubo K., et al. (2007). Cardiovascular Responses to High-Frequency Oscillatory Ventilation during Acute Lung Injury in Sheep. J. Anesth. 21 (3), 340–347. doi:10.1007/s00540-007-0508-z
Naorungroj T., Vilaichone W., Tongyoo S., Thamrongpairoj P., Permpikul C. (2015). High-frequency Oscillatory Ventilation for Patients during Exudative Phase of Severe ARDS. J. Med. Assoc. Thai 98 (4), 343–351.
Nardi N., Mortamet G., Ducharme-Crevier L., Emeriaud G., Jouvet P. (2017). Recent Advances in Pediatric Ventilatory Assistance. F1000Res 6, 290. doi:10.12688/f1000research.10408.1
Ng J., Ferguson N. D. (2017). High-frequency Oscillatory Ventilation. Curr. Opin. Crit. Care 23 (2), 175–179. doi:10.1097/MCC.0000000000000387
Nguyen A. P., Schmidt U. H., MacIntyre N. R. (2016). Should High-Frequency Ventilation in the Adult Be Abandoned? Respir. Care 61 (6), 791–800. doi:10.4187/respcare.04584
Niwa T., Hasegawa R., Ryuge M., Kawase M., Kondoh Y., Taniguchi H. (2011). Benefits and Risks Associated with the R100 High Frequency Oscillatory Ventilator for Patients with Severe Hypoxaemic Respiratory Failure. Anaesth. Intensive Care 39 (6), 1111–1119. doi:10.1177/0310057X1103900539
Pachl J., Roubík K., Waldauf P., Frič M., Zábrodský V. (2006). Normocapnic High-Frequency Oscillatory Ventilation Affects Differently Extrapulmonary and Pulmonary Forms of Acute Respiratory Distress Syndrome in Adults. Physiol. Res. 55 (1), 15–24. doi:10.33549/physiolres.930775
Papazian L., Gainnier M., Marin V., Donati S., Arnal J.-M., Demory D., et al. (2005). Comparison of Prone Positioning and High-Frequency Oscillatory Ventilation in Patients with Acute Respiratory Distress Syndrome*. Crit. Care Med. 33 (10), 2162–2171. doi:10.1097/01.ccm.0000181298.05474.2b
Pediatric Acute Lung Injury Consensus Conference (2015). Pediatric Acute Respiratory Distress Syndrome: Consensus Recommendations from the Pediatric Acute Lung Injury Consensus Conference. Pediatr. Crit. Care Med. 16 (5), 428–439. doi:10.1097/PCC.0000000000000350
Pensier J., de Jong A., Hajjej Z., Molinari N., Carr J., Belafia F., et al. (2019). Effect of Lung Recruitment Maneuver on Oxygenation, Physiological Parameters and Mortality in Acute Respiratory Distress Syndrome Patients: a Systematic Review and Meta-Analysis. Intensive Care Med. 45 (12), 1691–1702. doi:10.1007/s00134-019-05821-9
Pillow J. J. (2005). High-frequency Oscillatory Ventilation: Mechanisms of Gas Exchange and Lung Mechanics. Crit. Care Med. 33 (Mar), S135–S141. doi:10.1097/01.ccm.0000155789.52984.b7
Pillow J. J., Sly P. D., Hantos Z., Bates J. H. T. (2002). Dependence of Intrapulmonary Pressure Amplitudes on Respiratory Mechanics during High-Frequency Oscillatory Ventilation in Preterm Lambs. Pediatr. Res. 52 (4), 538–544. doi:10.1203/00006450-200210000-00013
Pillow J. J., Wilkinson M. H., Neil H. L., Ramsden C. A. (2001). In VitroPerformance Characteristics of High-Frequency Oscillatory Ventilators. Am. J. Respir. Crit. Care Med. 164 (6), 1019–1024. doi:10.1164/ajrccm.164.6.2005008
Rettig J. S., Smallwood C. D., Walsh B. K., Rimensberger P. C., Bachman T. E., Bollen C. W., et al. (2015). High-Frequency Oscillatory Ventilation in Pediatric Acute Lung Injury. Crit. Care Med. 43 (12), 2660–2667. doi:10.1097/CCM.0000000000001278
Ronchi C. F., dos Anjos Ferreira A. L., Campos F. J., Kurokawa C. S., Carpi M. F., de Moraes M. A., et al. (2011). High-frequency Oscillatory Ventilation Attenuates Oxidative Lung Injury in a Rabbit Model of Acute Lung Injury. Exp. Biol. Med. (Maywood) 236 (10), 1188–1196. doi:10.1258/ebm.2011.011085
Roosens C. D., Ama R., Leather H. A., Segers P., Sorbara C., Wouters P. F., et al. (2006). Hemodynamic Effects of Different Lung-Protective Ventilation Strategies in Closed-Chest Pigs with normal Lungs. Crit. Care Med. 34 (12), 2990–2996. doi:10.1097/01.CCM.0000242758.37427.16
Rotta A. T., Gunnarsson B., Fuhrman B. P., Hernan L. J., Steinhorn D. M. (2001). Comparison of Lung Protective Ventilation Strategies in a Rabbit Model of Acute Lung Injury. Crit. Care Med. 29 (11), 2176–2184. doi:10.1097/00003246-200111000-00021
Rowan C. M., Gertz S. J., McArthur J., Fitzgerald J. C., Nitu M. E., Loomis A., et al. (2016). Invasive Mechanical Ventilation and Mortality in Pediatric Hematopoietic Stem Cell Transplantation. Pediatr. Crit. Care Med. 17 (4), 294–302. doi:10.1097/PCC.0000000000000673
Rowan C. M., Klein M. J., Hsing D. D., Dahmer M. K., Spinella P. C., Emeriaud G., et al. (2020). Early Use of Adjunctive Therapies for Pediatric Acute Respiratory Distress Syndrome: A PARDIE Study. Am. J. Respir. Crit. Care Med. 201 (11), 1389–1397. doi:10.1164/rccm.201909-1807OC
Rožánek M., Horáková Z., Čadek O., Kučera M., Roubík K. (2012). Damping of the Dynamic Pressure Amplitude in the Ventilatory Circuit during High-Frequency Oscillatory Ventilation. Biomed. Tech. (Berl) 57. doi:10.1515/bmt-2012-4481
Samransamruajkit R., Prapphal N., Deerojanawong J., Sritippayawan S., Rassameehirun C., Pongsanon K., et al. (2016). A Comparison of Clinical Efficacy between High Frequency Oscillatory Ventilation and Conventional Ventilation with Lung Volume Recruitment in Pediatric Acute Respiratory Distress Syndrome: A Randomized Controlled Trial. Indian J. Crit. Care Med. 20 (2), 72–77. doi:10.4103/0972-5229.175940
Sedeek K. A., Takeuchi M., Suchodolski K., Kacmarek R. M. (2003). Determinants of Tidal Volume during High-Frequency Oscillation*. Crit. Care Med. 31 (1), 227–231. doi:10.1097/00003246-200301000-00035
Simmons D. H., Linde L. M., Miller J. H., O'Reilly R. J. (1961). Relation between Lung Volume and Pulmonary Vascular Resistance. Circ. Res. 9, 465–471. doi:10.1161/01.res.9.2.465
Sklar M. C., Fan E., Goligher E. C. (2017). High-Frequency Oscillatory Ventilation in Adults with ARDS. Chest 152 (6), 1306–1317. doi:10.1016/j.chest.2017.06.025
Slutsky A. S., Drazen J. M., Ingram R. H., Kamm R. D., Shapiro A. H., Fredberg J. J., et al. (1980). Effective Pulmonary Ventilation with Small-Volume Oscillations at High Frequency. Science 209 (4456), 609–611. doi:10.1126/science.6771872
Slutsky A. S., Drazen J. M. (2002). Ventilation with Small Tidal Volumes. N. Engl. J. Med. 347 (9), 630–631. doi:10.1056/NEJMp020082
Spadaro S., Karbing D. S., Fogagnolo A., Ragazzi R., Mojoli F., Astolfi L., et al. (2017). Simulation Training for Residents Focused on Mechanical Ventilation. Simul Healthc. Publish Ahead of Print (6), 349–355. doi:10.1097/SIH.0000000000000249
Sud S., Sud M., Friedrich J. O., Wunsch H., Meade M. O., Ferguson N. D., et al. (2016). High-frequency Oscillatory Ventilation versus Conventional Ventilation for Acute Respiratory Distress Syndrome. Cochrane Database Syst. Rev. 2018, CD004085. doi:10.1002/14651858.CD004085.pub4
Tatham K. C., Ferguson N. D., Zhou Q., Hand L., Austin P., Taneja R., et al. (2021). Evolution of Practice Patterns in the Management of Acute Respiratory Distress Syndrome: A Secondary Analysis of Two Successive Randomized Controlled Trials. J. Crit. Care 65, 274–281. doi:10.1016/j.jcrc.2021.06.017
Thind G. S., Hatipoğlu U., Chatburn R. L., Krishnan S., Duggal A., Mireles-Cabodevila E. (2021). Evaluation of High-Frequency Oscillatory Ventilation as a Rescue Strategy in Respiratory Failure. Respir. Care 66, 1746–1751. doi:10.4187/respcare.08936
Thompson B. T., Chambers R. C., Liu K. D. (2017). Acute Respiratory Distress Syndrome. N. Engl. J. Med. 377 (6), 562–572. doi:10.1056/NEJMra1608077
Tingay D. G., John J., Harcourt E. R., Black D., Dargaville P. A., Mills J. F., et al. (2015). Are All Oscillators Created Equal? In Vitro Performance Characteristics of Eight High-Frequency Oscillatory Ventilators. Neonatology 108 (3), 220–228. doi:10.1159/000431216
Tlayjeh H., Arabi Y. M., Ferguson N. D., Zhou Q., Lamontagne F., Arroliga A., et al. (2019). Body Mass Index and Mortality in Subjects with ARDS: Post-hoc Analysis of the OSCILLATE Trial. Respir. Care 64 (9), 1042–1048. doi:10.4187/respcare.06675
Tremblay L. N., Slutsky A. S. (2006). Ventilator-induced Lung Injury: from the Bench to the Bedside. Intensive Care Med. 32 (1), 24–33. doi:10.1007/s00134-005-2817-8
Ursulet L., Roussiaux A., Belcour D., Ferdynus C., Gauzere B.-A., Vandroux D., et al. (2015). Right over Left Ventricular End-Diastolic Area Relevance to Predict Hemodynamic Intolerance of High-Frequency Oscillatory Ventilation in Patients with Severe ARDS. Ann. Intensive Care 5 (1), 25. doi:10.1186/s13613-015-0068-6
Van de Kieft M., Dorsey D., Morison D., Bravo L., Venticinque S., Derdak S. (2005). High-frequency Oscillatory Ventilation: Lessons Learned from Mechanical Test Lung Models. Crit. Care Med. 33 (3 Suppl. l), S142–S147. doi:10.1097/01.ccm.0000155924.74942.7f
van Genderingen H. R., Versprille A., Leenhoven T., Markhorst D. G., van Vught A. J., Heethaar R. M. (2001). Reduction of Oscillatory Pressure along the Endotracheal Tube Is Indicative for Maximal Respiratory Compliance during High‐frequency Oscillatory Ventilation: A Mathematical Model Study. Pediatr. Pulmonol. 31 (6), 458–463. doi:10.1002/ppul.1075
van Gestel J. P. J., Bollen C. W., Bierings M. B., Boelens J. J., Wulffraat N. M., van Vught A. J. (2008). Survival in a Recent Cohort of Mechanically Ventilated Pediatric Allogeneic Hematopoietic Stem Cell Transplantation Recipients. Biol. Blood Marrow Transplant. 14 (12), 1385–1393. doi:10.1016/j.bbmt.2008.09.020
van Heerde M., Roubik K., Kopelent V., Kneyber M. C. J., Markhorst D. G. (2010). Spontaneous Breathing during High-Frequency Oscillatory Ventilation Improves Regional Lung Characteristics in Experimental Lung Injury. Acta Anaesthesiol Scand. 54 (10), 1248–1256. doi:10.1111/j.1399-6576.2010.02323.x
van Heerde M., Roubik K., Kopelent V., Plötz F. B., Markhorst D. G. (2009). Demand Flow Facilitates Spontaneous Breathing during High-Frequency Oscillatory Ventilation in a Pig Model. Crit. Care Med. 37 (3), 1068–1073. doi:10.1097/CCM.0b013e318196153b
van Heerde M., van Genderingen H., Leenhoven T., Roubik K., Plötz F., Markhorst D. (2006). Imposed Work of Breathing during High-Frequency Oscillatory Ventilation: a Bench Study. Crit. Care 10 (1), R23. doi:10.1186/cc3988
Vrettou C. S., Zakynthinos S. G., Malachias S., Mentzelopoulos S. D. (2014). The Effect of High-Frequency Oscillatory Ventilation Combined with Tracheal Gas Insufflation on Extravascular Lung Water in Patients with Acute Respiratory Distress Syndrome: a Randomized, Crossover, Physiologic Study. J. Crit. Care 29 (4), 568–573. doi:10.1016/j.jcrc.2014.03.020
Ware L. B., Matthay M. A. (2000). The Acute Respiratory Distress Syndrome. N. Engl. J. Med. 342 (18), 1334–1349. doi:10.1056/NEJM200005043421806
Winters J. W., Willing M. A., Sanfilippo D. (2000). Heliox Improves Ventilation during High-Frequency Oscillatory Ventilation in Pediatric Patients. Pediatr. Crit. Care Med. 1 (1), 33–37. doi:10.1097/00130478-200007000-00006
Wong J. J.-M., Liu S., Liu S., Dang H., Anantasit N., Phan P. H., et al. (2020). The Impact of High Frequency Oscillatory Ventilation on Mortality in Paediatric Acute Respiratory Distress Syndrome. Crit. Care 24 (1), 31. doi:10.1186/s13054-020-2741-x
Wong R., Deakers T., Hotz J., Khemani R. G., Ross P. A., Newth C. J. (2017). Volume and Pressure Delivery during Pediatric High-Frequency Oscillatory Ventilation. Pediatr. Crit. Care Med. 18 (4), e189–e194. doi:10.1097/PCC.0000000000001089
Young D., Lamb S. E., Shah S., MacKenzie I., Tunnicliffe W., Lall R., et al. (2013). High-frequency Oscillation for Acute Respiratory Distress Syndrome. N. Engl. J. Med. 368 (9), 806–813. doi:10.1056/NEJMoa1215716
Yumoto T., Fujita T., Asaba S., Kanazawa S., Nishimatsu A., Yamanouchi H., et al. (2019). Comparison of the Ventilation Characteristics in Two Adult Oscillators: a Lung Model Study. ICMx 7 (1), 15. doi:10.1186/s40635-019-0229-2
Zheng Y.-R., Lei Y.-Q., Liu J.-F., Wu H.-L., Xu N., Huang S.-T., et al. (2021). Effect of High-Frequency Oscillatory Ventilation Combined with Pulmonary Surfactant in the Treatment of Acute Respiratory Distress Syndrome after Cardiac Surgery: A Prospective Randomised Controlled Trial. Front. Cardiovasc. Med. 8, 675213. doi:10.3389/fcvm.2021.675213
Keywords: mechanical ventilation (lung protection) strategy, high-frequency ventilation with oscillations, high-frequency ventilation, children, ARDS, review (article), lung injury
Citation: Miller AG, Tan HL, Smith BJ, Rotta AT and Lee JH (2022) The Physiological Basis of High-Frequency Oscillatory Ventilation and Current Evidence in Adults and Children: A Narrative Review. Front. Physiol. 13:813478. doi: 10.3389/fphys.2022.813478
Received: 11 November 2021; Accepted: 08 April 2022;
Published: 26 April 2022.
Edited by:
Maurizio Cereda, University of Pennsylvania, United StatesReviewed by:
Vasilios E. Papaioannou, Democritus University of Thrace, GreeceCopyright © 2022 Miller, Tan, Smith, Rotta and Lee. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Andrew G. Miller, YW5kcmV3LmcubWlsbGVyQGR1a2UuZWR1; Jan Hau Lee, Z21zbGpoQG51cy5lZHUuc2c=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.