
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Physiol. , 10 November 2015
Sec. Craniofacial Biology and Dental Research
Volume 6 - 2015 | https://doi.org/10.3389/fphys.2015.00326
This article is part of the Research Topic Dental and periodontal tissues formation and regeneration: Current approaches and future challenges View all 26 articles
 Masaki Sato1
Masaki Sato1 Tadashi Furuya1,2
Tadashi Furuya1,2 Maki Kimura1
Maki Kimura1 Yuki Kojima1
Yuki Kojima1 Masakazu Tazaki1
Masakazu Tazaki1 Toru Sato2
Toru Sato2 Yoshiyuki Shibukawa1*
Yoshiyuki Shibukawa1*Extracellular ATP released via pannexin-1 channels, in response to the activation of mechanosensitive-TRP channels during odontoblast mechanical stimulation, mediates intercellular communication among odontoblasts in dental pulp slice preparation dissected from rat incisor. Recently, odontoblast cell lines, such as mouse odontoblast lineage cells, have been widely used to investigate physiological/pathological cellular functions. To clarify whether the odontoblast cell lines also communicate with each other by diffusible chemical substance(s), we investigated the chemical intercellular communication among cells from mouse odontoblast cell lines following mechanical stimulation. A single cell was stimulated using a glass pipette filled with standard extracellular solution. We measured intracellular free Ca2+ concentration ([Ca2+]i) by fura-2 in stimulated cells, as well as in cells located nearby. Direct mechanical stimulation to a single odontoblast increased [Ca2+]i, which showed sensitivity to capsazepine. In addition, we observed increases in [Ca2+]i not only in the mechanically stimulated odontoblast, but also in nearby odontoblasts. We could observe mechanical stimulation-induced increase in [Ca2+]i in a stimulated human embryo kidney (HEK) 293 cell, but not in nearby HEK293 cells. The increase in [Ca2+]i in nearby odontoblasts, but not in the stimulated odontoblast, was inhibited by adenosine triphosphate (ATP) release channel (pannexin-1) inhibitor in a concentration- and spatial-dependent manner. Moreover, in the presence of phospholipase C (PLC) inhibitor, the increase in [Ca2+]i in nearby odontoblasts, following mechanical stimulation of a single odontoblast, was abolished. We could record some inward currents evoked from odontoblasts near the stimulated odontoblast, but the currents were observed in only 4.8% of the recorded odontoblasts. The results of this study showed that ATP is released via pannexin-1, from a mechanically stimulated odontoblast, which transmits a signal to nearby odontoblasts by predominant activation of PLC-coupled nucleotide receptors.
Odontoblasts are dentin-forming cells that secrete dentin matrix proteins during physiological and pathological tooth formation. In the pathological setting, such as an enamel lesion, reactionary dentin is formed by various external stimuli applied to the dentin surface. The thermal, chemical, mechanical, and osmotic stimuli applied to the exposed dentin surface increase the hydrodynamic force and the velocity of dentinal fluid movement inside dentinal tubules (Andrew and Matthews, 2000; Charoenlarp et al., 2007). This increase in fluid movement induces deformation of plasma membrane of the odontoblast processes within the dentinal tubules (Magloire et al., 2010; Lin et al., 2011). Recent studies have indicated that odontoblasts express mechanosensitive ionic channels such as transient receptor potential (TRP) channel-vanilloid subfamily member-1, -2, and -4 (TRPV1, TRPV2, TRPV4) and TRP-ankyrin subfamily member-1 (TRPA1) (Magloire et al., 2010; Sato et al., 2013; Tsumura et al., 2013). Odontoblast cell membrane deformation activates various mechanosensitive-TRP channels as mechanosensors (Son et al., 2009; Tsumura et al., 2012; Shibukawa et al., 2015).
We have previously reported functional expression of G-protein- and phospholipase C (PLC)-coupling nucleotide receptor in odontoblasts (Shibukawa and Suzuki, 2003). These receptors are responsible for receipt of extracellular ATP released not only by dental pulp tissue damage (Cook et al., 1997; Liu et al., 2015), but also from other cells in the dental pulp, so as to establish intercellular communication. Most recently, we have reported that extracellular ATP released via pannexin-1 channels, in response to the activation of mechanosensitive-TRP channels during odontoblast mechanical stimulation, mediates intercellular chemical communication between odontoblasts and trigeminal ganglion (TG) neurons, to drive the sensory transduction mechanism for dental pain. We refer to this mechanism as “odontoblast hydrodynamic receptor theory” (Shibukawa et al., 2015). In addition, odontoblasts also established intercellular communication with each other via ATP/ADP and their nucleotide receptors, P2Y1 and P2Y12, expressed on these cells. This previous research was conducted by using primary cultured TG neurons and odontoblasts. The odontoblasts were obtained from dental pulp slice preparation from rat tissues (Shibukawa et al., 2015). Although the dental pulp slice preparations have been well established (Okumura et al., 2005; Son et al., 2009; Magloire et al., 2010; Tsumura et al., 2010, 2012, 2013; Shibukawa et al., 2015), odontoblast cell lines, such as mouse odontoblast lineage cells (OLCs; Arany et al., 2006; Fujisawa et al., 2012) or human dental pulp cells with odontoblastic differentiation (HDPs; Kitagawa et al., 2007), have also been widely used to investigate physiological/pathological odontoblast cellular functions (Ichikawa et al., 2012; Sato et al., 2013). Cooperative cellular function via intercellular signal communication plays an important role in the formation of hard tissues, including teeth (Iwamoto et al., 2010). Therefore, to clarify whether the odontoblast cell lines communicate with each other by diffusible chemical substance(s), we investigated the intercellular odontoblast signal communication when direct and focal mechanical stimulation was applied to single living odontoblasts.
Mouse odontoblast lineage cells (OLC) (Arany et al., 2006; Fujisawa et al., 2012) were cultured in an alpha-minimum essential medium containing 10% fetal bovine serum, 1% penicillin-streptomycin, and 1% fungizone (Life technologies, Carlsbad, CA, USA) at 37°C with 5% CO2. The cells were positive for various odontoblast representative transcripts of dentin sialophosphoprotein, dentin matrix protein-1, and nestin, generously provided by Dr. Masayuki Tokuda, Kagoshima University, Kagoshima, Japan. HEK293 cells were cultured in Dulbecco's modified Eagle's medium containing 10% fetal bovine serum, 1% penicillin-streptomycin, and 1% fungizone at 37°C with 5% CO2.
Standard extracellular solution (standard ECS) was composed of 135 mM NaCl, 5 mM KCl, 2.5 mM CaCl2, 0.5 mM MgCl2, 10 mM NaHCO3, 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), and 10 mM glucose and pH was adjusted to 7.4 by tris (hydroxymethyl) aminomethane (Tris) (328.5 mOsm/L). For Ca2+-free ECS, we removed extracellular Ca2+ (0 mM) from the standard ECS. Mefloquine and U73122 were obtained from TOCRIS Cookson (Bristol, UK). Capsazepine was obtained from Wako pure chemical (Osaka, Japan). All stock solutions were prepared in dimethyl sulfoxide. These stock solutions were diluted with standard ECS to the appropriate concentration before use. Except where indicated, all reagents were obtained from Sigma Chemical Co. (St. Louis, MO, USA).
Direct mechanical stimulation was applied using a fire-polished glass micropipette with a tip diameter of 2–3 μm. The stimulation micropipettes were pulled from glass pipette tubes (Harvard apparatus, UK) by using a DMZ Universal Puller (Zeitz instruments, Martinsried, Germany) and the micropipettes were filled with standard ECS. The micropipette was operated using a micromanipulator (NHW-3, Narishige, Tokyo, Japan). The micropipette was placed at a site just above the cell attachment position and was gently moved by 4.3, 8.5, or 12.8 μm in the vertically downward direction at 2.2 μm/s velocity to depress the cell membrane, to generate a focused mechanical stimulation.
Detached odontoblasts resuspended in standard ECS were placed into culture dishes at 37°C and 5% CO2 for 1 h. Images of odontoblasts in standard ECS were acquired using an intensified charge-coupled device camera (Hamamatsu Photonics, Shizuoka, Japan) mounted on a microscope (Olympus, Tokyo, Japan). The odontoblast radius was determined from the images obtained (AQUACOSMOS, HCImage, Hamamatsu Photonic, Shizuoka, Japan). Changes in peripheral cell length by mechanical stimulation were normalized to that found without any stimulation in the standard ECS.
Odontoblasts were incubated for 60 min (37°C) in standard ECS containing 10 μM fura-2 acetoxymethyl ester (Dojindo Laboratories, Kumamoto, Japan) and 0.1% (w/v) F-127 pluronic acid (Life technologies), followed by rinsing with fresh standard ECS. [Ca2+]i was measured using an AQUACOSMOS and HCImage system with an excitation wavelength selector and an intensified charge-coupled device camera incorporated onto a microscope. Fura-2 fluorescence emission was measured at 510 nm in response to alternating excitation wavelengths of 340 nm (F340) and 380 nm (F380). [Ca2+]i was measured as the fluorescence ratio (RF340∕F380) at two excitation wavelengths of 380 nm and 340 nm, and then expressed as F/F0 units; the RF340∕F380 value (F) was normalized to the resting value (F0).
Patch-clamp recordings of the whole-cell configuration were performed under voltage-clamp conditions. Patch pipettes (2–5 MΩ) were pulled from capillary tubes by using a DMZ Universal Puller (Zeitz Instruments, Martinsried, Germany), and the pipettes were filled with an intracellular solution. The intracellular solution contained 140 mM KCl, 10 mM NaCl, and 10 mM HEPES (pH adjusted to 7.2 by Tris). Whole-cell currents were measured using a patch-clamp amplifier (L/M-EPC-7+; Heka Elektronik, Lambrecht, Germany), and were monitored and stored using pCLAMP software (Molecular Devices, Sunnyvale, CA, USA), after digitizing the analog signals at 1 kHz (DigiData 1440A, Molecular Devices) and filtering the signals digitally at 100 Hz using pCLAMP. The data were analyzed offline by using pCLAMP and the technical graphics/analysis program ORIGIN (MicroCal Software, Northampton, MA, USA). All experiments were conducted at room temperature (30 ± 1.0°C).
Data are expressed as the mean ± standard deviation (SD) or standard error (SE) of the mean of N observations, where N represents the number of tested cells or separate experiments, respectively. Statistical differences were evaluated using analysis of variance (ANOVA) and the Steel-Dwass post-hoc test, and P < 0.05 was considered to be significant.
Cell peripheral length was calculated to measure changes in cell size with or without exposure by penetration of a glass micropipette at three different depths (4.3, 8.5, and 12.8 μm). The normalized peripheral length of cells significantly increased as the glass pipette depth was measured from 0 to 8.5 μm (Figure 1A, P < 0.05), indicating that direct mechanical stimulation using a glass pipette induced stretching of the odontoblast plasma membrane. The peripheral length of cells without mechanical stimulation was found to be 46.2 ± 1.4 μm (N=11).
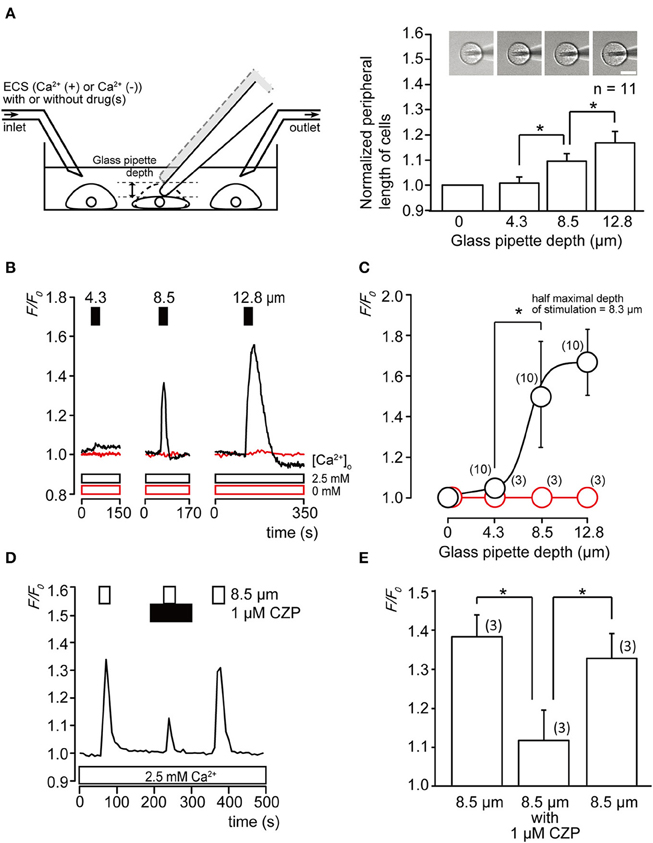
Figure 1. Cell deformation by direct stimulation induces increase in [Ca2+]i (A) Single-cell direct mechanical stimulation with glass micropipette in the recording bath. The relationship between changes in the peripheral length of cells and intensity of mechanical stimulation in terms of depth reached by the pipette. Normalized cell size increased with increasing mechanical stimulation, each normalized cell size was 1.02 ± 0.03-fold by 4.3 μm, 1.10 ± 0.03-fold by 8.5 μm, and 1.16 ± 0.05-fold by 12.8 μm of the vertical downward displacement of the pipette. Bars represent mean ± SD of 11 cells. Statistically significant differences between values are indicated by asterisks. *P < 0.05 (A right). Top panel shows images of cells during each stimulation. Bar indicates 10 μm. (B) Traces of transient increase in [Ca2+]i during a series of mechanical stimulations induced by vertical micropipette displacements at depths of 4.3, 8.5, and 12.8 μm (upper filled boxes) in standard ECS with extracellular 2.5 mM Ca2+ (black lines and black line boxes) or without extracellular Ca2+ (0 mM) (red lines and red line boxes). (C) Linear plot of F/F0 value against direct mechanical stimulation intensity with extracellular 2.5 mM Ca2+ (black) or without extracellular Ca2+ (red). The numbers in parentheses indicate the number of tested cells. (D) Mechanical stimulation-induced Ca2+ influx was inhibited by TRPV1 antagonist (upper black box). [Ca2+]i increase was elicited by 8.5 μm direct mechanical stimulation (upper white boxes). (E) Summary bar graphs of the increase in [Ca2+]i induced by mechanical stimulation (to 8.5 μm) without (left column) or with (middle column) application of TRP channel antagonist. Recovery effect is shown in the right column. The resting value, F/F0 = 1.0. Each bar indicates the mean ± SD for three cells. Statistically significant differences between columns (shown by solid lines) are indicated by asterisks. *P < 0.05.
A series of direct mechanical stimulations (4.3–12.8 μm) elicited transient increases in intracellular free Ca2+ concentration ([Ca2+]i) (Figure 1B, black lines) in the presence of extracellular Ca2+, while in the absence of extracellular Ca2+ (Ca2+-free extracellular solution; Ca2+-free ECS), mechanical stimulation-induced [Ca2+]i increases were completely abolished (Figure 1B, red line), indicating that mechanical stimulation of the odontoblasts elicited Ca2+ influx. The linear plot in Figure 1C shows F/F0 values as a function of applied direct mechanical stimulation. Dependence of [Ca2+]i on mechanical stimulation by the pipette was confirmed by fitting the data to the following function:
where K is the half-maximal depth of a mechanical stimulation applied on the odontoblast (8.3 μm), Amax is maximal and Amin is minimal F/F0 response. The x indicates applied depth of the glass pipette.
To further investigate the Ca2+ influx pathway activated by direct mechanical stimulation, the effects of nonselective TRPV1 and TRP Melastatin subfamily member 8 (TRPM8) channel antagonist, capsazepine (CZP), on direct mechanical stimulation-induced Ca2+ influx were examined. In the presence of extracellular Ca2+ (2.5 mM), [Ca2+]i in odontoblasts showed a rapid and transient increase during direct mechanical stimulation (8.5 μm), which was significantly and reversibly inhibited by 1 μM CZP to 32.4 ± 12.1% (N=3) (Figures 1D,E).
We measured the increase in [Ca2+]i by direct mechanical stimulation not only in the single odontoblast, but also in the neighboring odontoblasts. Direct mechanical stimulation of the single odontoblast increased [Ca2+]i transiently. We also observed an increase in nearby odontoblasts (Figure 2A). The increase in [Ca2+]i in nearby odontoblasts was reduced by increasing their distance from the stimulated odontoblast (Figure 2A). The plot in Figure 2B shows F/F0 values as a function of distance from stimulated odontoblast to nearby odontoblasts. The spatial constant (τ), was obtained by fitting the data to the exponential decay function. The spatial constant was 30.1 μm. Direct mechanical stimulation of a human embryonic kidney (HEK) 293 cell increased [Ca2+]i transiently, whereas [Ca2+]i increase in HEK293 cells near the stimulated cell was not observed (Figure 2C). The plot in Figure 2D shows F/F0 values as a function of distance from stimulated HEK293 cells to nearby HEK293 cells. The spatial constant could not be measured for HEK293 cells.
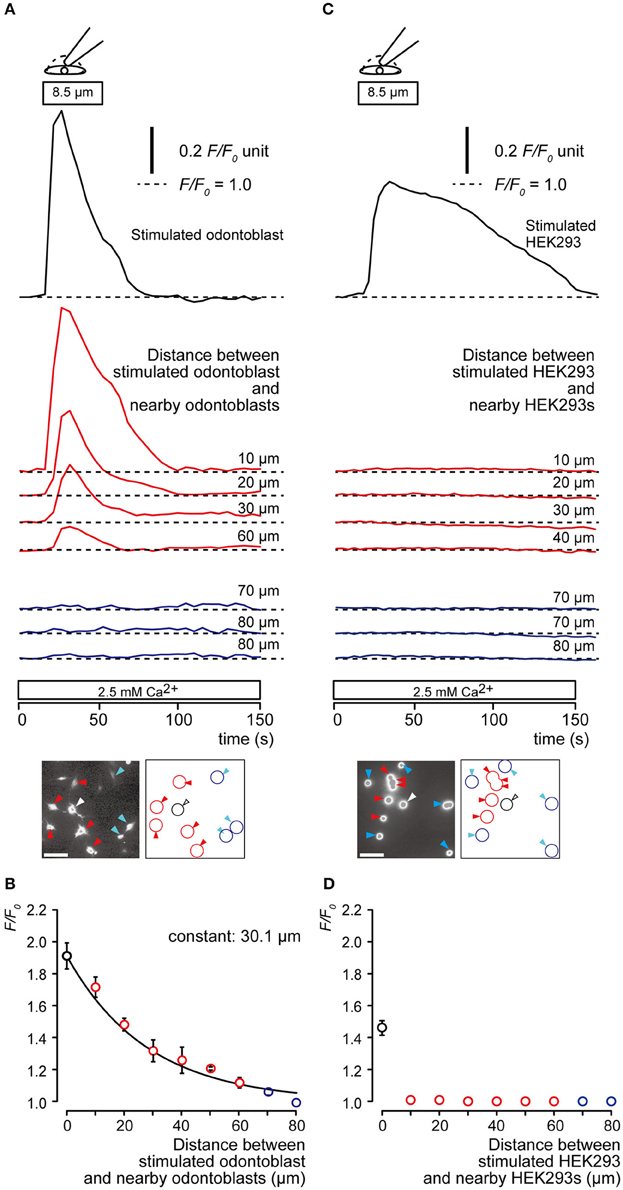
Figure 2. Communication between a mechanical stimulated odontoblast and nearby odontoblasts. (A,C) Representative traces showing transient increase in [Ca2+]i from a mechanically stimulated cell (upper black traces), nearby (middle red traces) and distant cells (lower blue cells) in odontoblast (A) and HEK293 cell (C) during focal, direct mechanical stimulation of a single cell. Cell deformation was induced by displacement of a micropipette to a depth of 8.5 μm (upper open boxes and drawings) in a single cell in standard ECS (2.5 mM Ca2+). Bottom in (A,C) show images of relative fluorescence ratio (F0) before recordings (white bars show 50 μm), and their schematic representations. Stimulated cells are shown by white arrowhead and black lines, nearby cells are indicated by red arrowheads and red lines, and distant cells are represented by blue arrowheads and blue lines. Each horizontal dotted line shows base line (as F/F0 = 1.0) for each response. Responses from nearby (four red traces) or distant cells (three blue traces) were recorded 10–80 μm away from stimulated cells. (B,D) F/F0 values (data points) as a function of the distance from the stimulated cell (0 μm) to nearby (red symbols) or distant cells (blue symbols) for odontoblasts (A) and HEK293 cells (D). The superimposed lines denote the best fit of a single exponential function. This analysis yielded spatial constants (τ) for the distance-dependent decrease in [Ca2+]i. The increase of [Ca2+]i was observed in neither nearby nor distant HEK293 cells. Data points represent the mean ± SE from 3 to 32 cells in 3 separate experiments in (B) and 3 to 25 cells in 3 separate experiments in (D).
Using 10 μM mefloquine, which is an inhibitor of the pannexin-1 channel, mechanical-stimulation-induced [Ca2+]i increase was observed in stimulated odontoblast, whereas in nearby odontoblasts no significant increase in [Ca2+]i was observed (Figure 3A). In addition, mefloquine (0.1, 1.0, and 10 μM) inhibited [Ca2+]i response in nearby odontoblasts in a concentration- and spatial-dependent manner (Figures 3B,C). The administration of 1.0 μM U73122, an inhibitor of phospholipase C, significantly suppressed mechanical stimulation-induced increase in [Ca2+]i in nearby odontoblasts, but not the stimulated cell (Figure 3D). U73122 (0.01, 0.1, and 1.0 μM) inhibited [Ca2+]i responses in nearby odontoblasts in a concentration- and spatial-dependent manner (Figures 3E,F). The decrease in [Ca2+]i in nearby odontoblasts with increasing distance from stimulated odontoblasts occurred rapidly (Figures 3B,E) in a dose-dependent manner on mefloquine (Figure 3C) and U73122 treatment (Figure 3F), showing significant decrease in spatial constants.
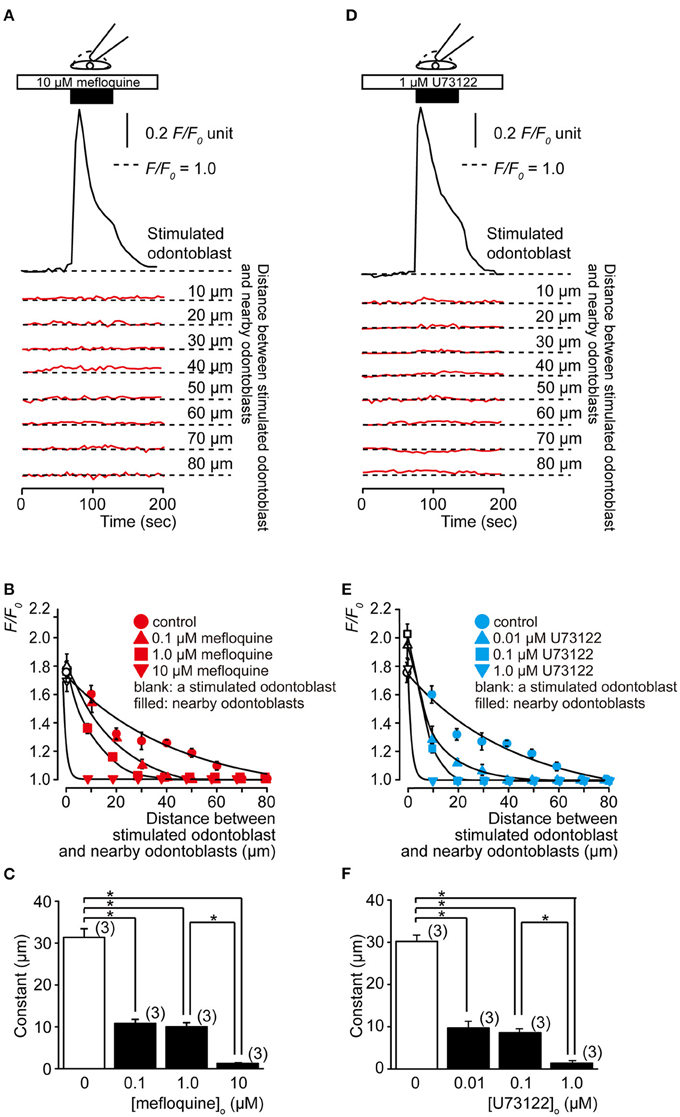
Figure 3. Intercellular odontoblast communication via ATP. Effects of pannexin-1 inhibitor, mefloquine, and phospholipase C (PLC) inhibitor, U73122, on the increase in [Ca2+]i following focal and direct mechanical stimulation of a single odontoblast. (A,D) Transient increase in [Ca2+]i from a mechanically stimulated odontoblast (upper black traces), nearby and distant odontoblasts (lower red traces), during single odontoblast stimulation in the presence of mefloquine (A) or U73122 (D). Mechanical stimulation was applied by displacement of a micropipette to a depth of 8.5 μm (upper filled boxes and drawings) in standard ECS (2.5 mM Ca2+). Horizontal dotted lines show the baseline (F/F0 = 1.0) for each response. Responses from nearby and distant odontoblast were recorded from cells located 10 to 80 μm away from the stimulated odontoblast. This distance is indicated on the right side of each trace. (B,E) F/F0 values (data points) as a function of the distance from a stimulated odontoblast (0 μm) to nearby odontoblast, with or without pannexin-1 inhibitor (0.1–10 μM mefloquine; red symbols in B) and phospholipase C inhibitor (0.01–1 μM U73122; blue symbols in E). Each mechanical stimulation induced [Ca2+]i increase in the stimulated odontoblast is shown by open symbols with each concentration of mefloquine or U73122. This response was not affected by treatment with mefloquine or U73122. The superimposed lines denote the best fit of a single exponential function. This analysis yielded spatial constants (τ) of the distance-dependent decreases in [Ca2+]i (solid lines in B,E) with or without (filled circles) mefloquine (B) and U73122 (E). Data points represent the mean ± SD from 3 to 41 cells in (B,E) from each three separate experiment. (C,F) Summary bar graphs of the mean τ values of the distance-dependent decrease in [Ca2+]i, measured from stimulated to nearby odontoblast, with pannexin-1 inhibitor (C) or phospholipase C inhibitor (F). Each bar indicates mean ± SD. The numbers in parentheses in (C,F) indicate the number of tested cells. Statistically significant differences between spatial constant values are indicated by asterisks. *P < 0.05.
The whole-cell patch-clamp method was used to record membrane currents that are activated by intercellular transmitter released from mechanically stimulated odontoblasts (Figure 4). We recorded an evoked inward current in only 2 out of 42 tested odontoblasts located 5 μm from the mechanically stimulated cell (in 42 experiments). These currents had delay times of 7–10 s in their activation.
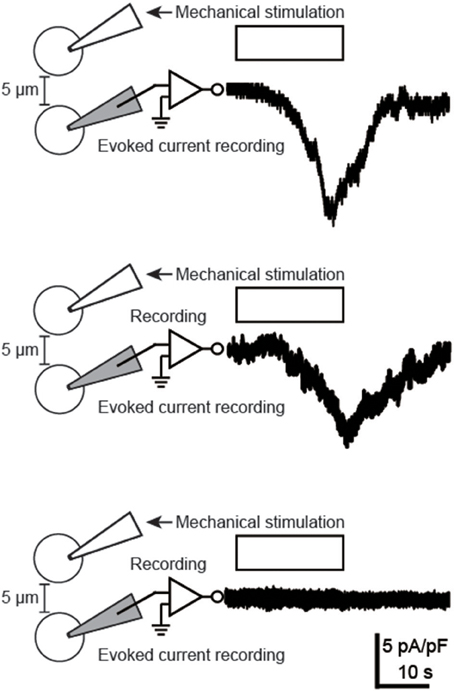
Figure 4. Evoked inward currents in neighboring odontoblasts following mechanical stimulation of a single odontoblast. Traces of inward currents (upper two traces) were recorded by using whole-cell patch-clamp recordings of the cells located 5 μm away from a stimulated odontoblast, following mechanical stimulation (the white boxes and the drawings on the left). Pictures of the whole-cell patch-clamp recording are also shown. Note that we could record evoked inward currents in only 2 out of 42 tested odontoblasts. Lower trace shows current from cell which did not elicit inward current, following mechanical stimulation. The recording was obtained from three separated cells.
In this study, we demonstrated that intercellular signal transduction between mechanically stimulated and neighboring odontoblasts occurs via activation of mechanosensitive TRP channels, release of ATP from the pannexin-1 channel, and activation of PLC-coupled receptors. Since we previously reported that TRPM8 channels are not expressed in mouse odontoblasts, CPZ-sensitive mechanical stimulation-induced [Ca2+]i response in the present study is mediated by TRPV1 channels. Activation of TRPV1 channels, acting as sensor proteins (Magloire et al., 2010; Sato et al., 2013; Tsumura et al., 2013), induces the release of ATP via pannexin-1 channels from mechanically stimulated odontoblasts. ATP released from mechanically stimulated odontoblasts, as an intercellular transmitter in the extracellular medium, then activates PLC-coupled ATP/ADP receptors in neighboring odontoblasts. These results are in line with our previous results showing that mechanically stimulated odontoblasts release ATP via pannexin-1 channels to the neighboring odontoblasts, which activates the P2Y1 and P2Y12 receptors (Shibukawa et al., 2015).
The changes in the peripheral length and the increase in [Ca2+]i depended on the intensity of the direct mechanical stimulation applied to odontoblasts. In the previous and present study, repeated mechanical stimulation to the odontoblasts induced increase in [Ca2+]i repeatedly (Shibukawa et al., 2015). Although these results suggested that the stimuli did not induce any unfavorable effects to the cells, application of mechanical stimulation was limited to three times in the present study to avoid cell damage.
Treatment of the culture medium with an inhibitor of ATP-releasing pannexin-1 channels abolishes the increase in [Ca2+]i in neighboring odontoblasts, but not in mechanically stimulated odontoblasts, indicating that ATP released via pannexin-1 acts as an intercellular transmitter for odontoblast–odontoblast chemical communication. In previous immunohistochemical studies, pannexin-1 was found to be localized throughout the cell body of odontoblasts (Shibukawa et al., 2015). In addition, TRPV4 and TRPA1 channel activation also elicits ATP release in odontoblasts (Egbuniwe et al., 2014). When direct mechanical stimulation was applied to HEK293 cells, internal Ca2+ was increased in the mechanically stimulated cells, but not in the neighboring cells. This is consistent with previous results showing that pannexin-1 is not expressed in HEK293 cells (Langlois et al., 2014), and suggests that intercellular communication via diffusible substances (i.e., intercellular transmitters) is established specifically in inter-odontoblast cellular networks in the dental pulp.
Odontoblasts express connexin (Cx) hemi-channels, forming a gap junction and establishing cell–cell communication (Goldberg et al., 1981; Sasaki et al., 1982; Callé, 1985; Goldberg and Sasaki, 1985; About et al., 2001; Muramatsu et al., 2004; Ikeda and Suda, 2013). Connexin 43, a gap junction protein, is expressed in odontoblasts (Ikeda and Suda, 2013; Muramatsu et al., 2013), and seems to play a role in electrical communication between odontoblasts. It has been reported that connexin 43 also releases ATP extracellularly in the pathological setting, such as low (almost 0 mM) extracellular Ca2+ circumstance or high (c.a. +60 mV) membrane potential. Since we did not expose the odontoblast to such extracellular conditions, contribution of connexin 43 to intercellular odontoblasts chemical communication is unlikely (Shibukawa et al., 2015).
Use of U73122, an inhibitor of phospholipase C, did not affect calcium response in the mechanically stimulated odontoblast, but abolish it in the neighboring odontoblasts. P2Y receptors are coupled with PLC and activated principally by endogenous nucleotides, ATP, and ADP (Burnstock, 2007). A specific enzyme, nucleoside triphosphate diphosphohydrolase-2 (NTPDase2) (Liu et al., 2012), is expressed in the Schwann cells of the dental pulp and/or the odontoblast membrane, and has been reported to hydrolyze extracellular ATP to ADP. Thus, both, released ATP and ADP from degradation of the ATP by NTPDase2, activate P2Y receptors in nearby odontoblasts. In addition, an ATP derivative, 2-methylthio-ATP, mobilizes [Ca2+]i in the odontoblasts (Shibukawa and Suzuki, 2003), showing expression of the P2Y1 and P2Y12 receptors. We previously reported that P2Y1 and P2Y12 receptor activation in odontoblasts established inter-odontoblast communication (Shibukawa et al., 2015).
On the other hand, as shown in Figure 4, we could successfully record evoked inward currents in the odontoblast located 5 μm away from the stimulated odontoblast. The evoked inward currents via intercellular transmitters were likely mediated by the activation of ionotropic receptors and/or ionic channels. Mechanical stimulation of the odontoblasts induces a release of intercellular transmitters, not only of ATP (as in the present study and in Shibukawa et al., 2015), but also of glutamate (our personal communication). However, the present results indicate that the ionotropic receptors (such as ionotropic ATP (P2Xs) and/or glutamate receptors) and ionic channel activation are hardly involved in the inter-odontoblast communication, since the evoked currents elicited by mechanical stimulation could be observed in only 4.8% of the odontoblasts recorded (2/42 cells) in the present study. We previously reported that the P2X3 receptor is not expressed in odontoblasts, while the P2X3 receptor in the TG neurons plays an important role in the sensory transduction sequence by receiving ATP from mechanically stimulated odontoblasts (Shibukawa et al., 2015). Although, we observed several P2X subtype expressions, except for the P2X3 receptor, in odontoblasts (personal communication), these P2X receptor subtypes need a relative high concentration of extracellular ATP to be activated. Therefore, ATP released in response to TRPV1 channel activation during mechanical stimulation predominantly activates P2Y receptors on neighboring odontoblasts to mediate intercellular odontoblasts communication.
We previously showed that accumulated [Ca2+]i following TRPV1 channel activation is extruded by Na+-Ca2+ exchangers (NCXs) in odontoblasts (Tsumura et al., 2012). Our results suggest that communication between odontoblasts mediated by ATP plays an important role in reactionary dentin formation following dentin stimulation by increased intracellular Ca2+ signaling by TRPV-mediated Ca2+ influx and P2Y receptor-mediated Ca2+ mobilization as well as subsequent Ca2+ extrusion by NCXs.
The results from this study show that ATP is released from a mechanically stimulated odontoblast via pannexin-1 by TRP channel activation, and it transmits a signal to nearby odontoblasts by activating P2Y receptors. The results also strongly suggest that odontoblasts communicate with each other to drive their cellular functions such as enhancement of dentin formation. Our results also indicate that odontoblasts are sensory receptor cells (Shibukawa et al., 2015) that detect hydrodynamic force within dentinal tubules (Liu et al., 2015) via mechanosensitive TRP channels. Additionally, ATP released from odontoblasts via pannexin-1 pathway acts as the intercellular transmitter between odontoblasts by activating the P2Y receptors. Taken together with our previous results (Tsumura et al., 2010, 2012, 2013; Sato et al., 2013; Shibukawa et al., 2015), the present study also strongly suggests that the intercellular signaling via released ATP in response to activation of odontoblasts as mechano-sensitive sensory receptor cells are capable of activating two specific and separate pathways underlying odontoblast function: (1) the selective activation of P2Y receptors in odontoblasts establishes inter-odontoblast network that may drive dentin formation, and (2) specific activation of P2X3 receptors in TG neurons mediates the sensory transduction sequence for dentin (Shibukawa et al., 2015). In addition, the results also prove that odontoblast cell lines are useful in studying the cellular functions that are mediated by intercellular signaling.
MS, TF, MK, and YK carried out the measurement intracellular Ca2+ signaling. YS, TS, and MT participated in the design of the study. MS and TF performed the statistical analysis. YS conceived of the study, and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
This research was supported by a Grant-in-Aid (No. 26861559/15K11129/15K11056) for Scientific Research from the MEXT of Japan.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
About, I., Proust, J. P., Raffo, S., Mitsiadis, T. A., and Franquin, J. C. (2001). In vivo and in vitro expression of connexin 43 in human teeth. Connect. Tissue Res. 43, 232–237. doi: 10.1080/03008200290000952
Andrew, D., and Matthews, B. (2000). Displacement of the contents of dentinal tubules and sensory transduction in intradental nerves of the cat. J. Physiol. 529(Pt 3), 791–802. doi: 10.1111/j.1469-7793.2000.00791.x
Arany, S., Nakata, A., Kameda, T., Koyota, S., Ueno, Y., and Sugiyama, T. (2006). Phenotype properties of a novel spontaneously immortalized odontoblast-lineage cell line. Biochem. Biophys. Res. Commun. 342, 718–724. doi: 10.1016/j.bbrc.2006.02.020
Burnstock, G. (2007). Physiology and pathophysiology of purinergic neurotransmission. Physiol. Rev. 87, 659–797. doi: 10.1152/physrev.00043.2006
Callé, A. (1985). Intercellular junctions between human odontoblasts. a freeze-fracture study after demineralization. Acta Anat. 122, 138–144. doi: 10.1159/000145995
Charoenlarp, P., Wanachantararak, S., Vongsavan, N., and Matthews, B. (2007). Pain and the rate of dentinal fluid flow produced by hydrostatic pressure stimulation of exposed dentine in man. Arch. Oral Biol. 52, 625–631. doi: 10.1016/j.archoralbio.2006.12.014
Cook, S. P., Vulchanova, L., Hargreaves, K. M., Elde, R., and McCleskey, E. W. (1997). Distinct ATP receptors on pain-sensing and stretch-sensing neurons. Nature 387, 505–508. doi: 10.1038/387505a0
Egbuniwe, O., Grover, S., Duggal, A. K., Mavroudis, A., Yazdi, M., Renton, T., et al. (2014). TRPA1 and TRPV4 activation in human odontoblasts stimulates ATP release. J. Dent. Res. 93, 911–917. doi: 10.1177/0022034514544507
Fujisawa, M., Tokuda, M., Morimoto-Yamashita, Y., Tatsuyama, S., Arany, S., Sugiyama, T., et al. (2012). Hyperosmotic stress induces cell death in an odontoblast-lineage cell line. J. Endod. 38, 931–935. doi: 10.1016/j.joen.2012.03.023
Goldberg, M., Escaig, F., and Septier, D. (1981). Freeze fracturing and thin-section study of intercellular junctions between odontoblasts and the rat incisor. changes induced by vinblastine. J. Biol. Buccale 9, 141–153.
Goldberg, M., and Sasaki, T. (1985). Intramembrane particle distribution on the plasma membrane of ruffle-ended and smooth ended maturation ameloblasts of the rat incisors. J. Biol. Buccale 13, 251–260.
Ichikawa, H., Kim, H.-J., Shuprisha, A., Shikano, T., Tsumura, M., Shibukawa, Y., et al. (2012). Voltage-dependent sodium channels and calcium-activated potassium channels in human odontoblasts in vitro. J. Endod. 38, 1355–1362. doi: 10.1016/j.joen.2012.06.015
Ikeda, H., and Suda, H. (2013). Odontoblastic syncytium through electrical coupling in the human dental pulp. J. Dent. Res. 92, 371–375. doi: 10.1177/0022034513478430
Iwamoto, T., Nakamura, T., Doyle, A., Ishikawa, M., de Vega, S., Fukumoto, S., et al. (2010). Pannexin 3 regulates intracellular ATP/cAMP levels and promotes chondrocyte differentiation. J. Biol. Chem. 285, 18948–18958. doi: 10.1074/jbc.M110.127027
Kitagawa, M., Ueda, H., Iizuka, S., Sakamoto, K., Oka, H., Kudo, Y., et al. (2007). Immortalization and characterization of human dental pulp cells with odontoblastic differentiation. Arch. Oral Biol. 52, 727–731. doi: 10.1016/j.archoralbio.2007.02.006
Langlois, S., Xiang, X., Young, K., Cowan, B. J., Penuela, S., and Cowan, K. N. (2014). Pannexin 1 and pannexin 3 channels regulate skeletal muscle myoblast proliferation and differentiation. J. Biol. Chem. 289, 30717–30731. doi: 10.1074/jbc.M114.572131
Lin, M., Luo, Z. Y., Bai, B. F., Xu, F., and Lu, T. J. (2011). Fluid mechanics in dentinal microtubules provides mechanistic insights into the difference between hot and cold dental pain. PLoS ONE 6:e18068. doi: 10.1371/journal.pone.0018068
Liu, X., Wang, C., Fujita, T., Malmstrom, H. S., Nedergaard, M., Ren, Y. F., et al. (2015). External dentin stimulation induces ATP release in human teeth. J. Dent. Res. 94, 1259–1266. doi: 10.1177/0022034515592858
Liu, X., Yu, L., Wang, Q., Pelletier, J., Fausther, M., Sévigny, J., et al. (2012). Expression of ecto-ATPase NTPDase2 in human dental pulp. J. Dent. Res. 91, 261–267. doi: 10.1177/0022034511431582
Magloire, H., Maurin, J. C., Couble, M. L., Shibukawa, Y., Tsumura, M., Thivichon-Prince, B., et al. (2010). Topical review. dental pain and odontoblasts: facts and hypotheses. J. Orofac. Pain 24, 335–349.
Muramatsu, T., Hamano, H., Ogami, K., Ohta, K., Inoue, T., and Shimono, M. (2004). Reduction of connexin 43 expression in aged human dental pulp. Int. Endod. J. 37, 814–818. doi: 10.1111/j.1365-2591.2004.00880.x
Muramatsu, T., Hashimoto, S., Shibukawa, Y., Yuasa, K., Furusawa, M., and Shimono, M. (2013). Immunoelectron microscopic observation of connexin43 in rat odontoblasts. Microsc. Res. Tech. 76, 988–991. doi: 10.1002/jemt.22271
Okumura, R., Shima, K., Muramatsu, T., Nakagawa, K.-I., Shimono, M., Suzuki, T., et al. (2005). The odontoblast as a sensory receptor cell? the expression of TRPV1 (VR-1) channels. Arch. Histol. Cytol. 68, 251–257. doi: 10.1679/aohc.68.251
Sasaki, T., Nakagawa, K., and Higashi, S. (1982). Ultrastructure of odontoblasts in kitten tooth germs as revealed by freeze-fracture. Arch. Oral Biol. 27, 897–904. doi: 10.1016/0003-9969(82)90048-6
Sato, M., Sobhan, U., Tsumura, M., Kuroda, H., Soya, M., Masamura, A., et al. (2013). Hypotonic-induced stretching of plasma membrane activates transient receptor potential vanilloid channels and sodium-calcium exchangers in mouse odontoblasts. J. Endod. 39, 779–787. doi: 10.1016/j.joen.2013.01.012
Shibukawa, Y., Sato, M., Kimura, M., Sobhan, U., Shimada, M., Nishiyama, A., et al. (2015). Odontoblasts as sensory receptors: transient receptor potential channels, pannexin-1, and ionotropic ATP receptors mediate intercellular odontoblast-neuron signal transduction. Pflügers Arch. 467, 843–863. doi: 10.1007/s00424-014-1551-x
Shibukawa, Y., and Suzuki, T. (2003). Ca2+ signaling mediated by IP3-dependent Ca2+ releasing and store-operated Ca2+ channels in rat odontoblasts. J. Bone Miner. Res. 18, 30–38. doi: 10.1359/jbmr.2003.18.1.30
Son, A. R., Yang, Y. M., Hong, J. H., Lee, S. I., Shibukawa, Y., and Shin, D. M. (2009). Odontoblast TRP channels and thermo/mechanical transmission. J. Dent. Res. 88, 1014–1019. doi: 10.1177/0022034509343413
Tsumura, M., Okumura, R., Tatsuyama, S., Ichikawa, H., Muramatsu, T., Matsuda, T., et al. (2010). Ca2+ extrusion via Na+-Ca2+ exchangers in rat odontoblasts. J. Endod. 36, 668–674. doi: 10.1016/j.joen.2010.01.006
Tsumura, M., Sobhan, U., Muramatsu, T., Sato, M., Ichikawa, H., Sahara, Y., et al. (2012). TRPV1-mediated calcium signal couples with cannabinoid receptors and sodium-calcium exchangers in rat odontoblasts. Cell Calcium 52, 124–136. doi: 10.1016/j.ceca.2012.05.002
Keywords: odontoblast, transient receptor potential channel, pannexin-1, adenosine triphosphate, P2Y, paracrine
Citation: Sato M, Furuya T, Kimura M, Kojima Y, Tazaki M, Sato T and Shibukawa Y (2015) Intercellular Odontoblast Communication via ATP Mediated by Pannexin-1 Channel and Phospholipase C-coupled Receptor Activation. Front. Physiol. 6:326. doi: 10.3389/fphys.2015.00326
Received: 08 July 2015; Accepted: 27 October 2015;
Published: 10 November 2015.
Edited by:
Thimios Mitsiadis, University of Zurich, SwitzerlandReviewed by:
Imad About, Aix-Marseille Université, FranceCopyright © 2015 Sato, Furuya, Kimura, Kojima, Tazaki, Sato and Shibukawa. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yoshiyuki Shibukawa, eXNoaWJ1a2FAdGRjLmFjLmpw
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.