
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
MINI REVIEW article
Front. Physiol., 09 February 2015
Sec. Cardiac Electrophysiology
Volume 6 - 2015 | https://doi.org/10.3389/fphys.2015.00034
This article is part of the Research TopicCa2+ Signaling and Heart RhythmView all 13 articles
The ability to dynamically regulate, traffic, retain, and recycle proteins within the cell membrane is fundamental to life and central to the normal function of the heart. In the cardiomyocyte, these pathways are essential for the regulation of Ca2+, both at the level of the plasma membrane, but also in local cellular domains. One intracellular pathway often overlooked in relation to cardiovascular Ca2+ regulation and signaling is the endosome-based trafficking pathway. Highlighting its importance, this system and its molecular components are evolutionarily conserved across all metazoans. However, remarkably little is known of how endosome-based protein trafficking and recycling functions within mammalian cells systems, especially in the heart. As the endosomal system acts to regulate the expression and localization of membrane proteins central for cardiac Ca2+ regulation, understanding the in vivo function of this system in the heart is critical. This review will focus on endosome-based protein trafficking in the heart in both health and disease with special emphasis for the role of endocytic regulatory proteins, C-terminal Eps15 homology domain-containing proteins (EHDs).
The capacity of a cell to regulate protein expression and localization within the plasma membrane is central to life. Endosome-based systems mediate a wide range of cellular processes including anterograde trafficking of newly formed proteins out of the Golgi apparatus to their proper locales, internalization of membrane proteins targeted for recycling or degradation, and nutrient endocytosis. Studying these systems in the intact mammalian cellular environment has proven difficult, mostly due to the lack of available tools and model systems. Only within the last 15 years have we begun teasing out the role of endosome-based protein trafficking and targeting recycling in vivo. We now know that endosome-based systems are critical for such cellular processes as cell motility (Traynor and Kay, 2007), cell division (Boucrot and Kirchhausen, 2007), cell-cell junction regulation (Palacios et al., 2002), epithelial polarity (Shivas et al., 2010), and neuronal plasticity (Wang et al., 2008). A growing body of evidence has implicated endosomal trafficking in the development and regulation of membrane excitability in neurons (Sun et al., 2014), pancreatic cells (Manna et al., 2010), and cardiac muscle cells (McEwen et al., 2007; Kruse et al., 2009; Ishii et al., 2012; Curran et al., 2014).
In cardiomyocytes, membrane excitability depends on the proper expression and organization of multiple ion channels, pumps, exchangers, and transporters within the plasma membrane to regulate intracellular ion signaling pathways. As the endosomal system acts to regulate the expression and localization of membrane proteins, the potential exists that this system may be able to modulate excitability in the heart. This regulatory capacity, therefore, makes it an attractive candidate for new therapeutic intervention in the treatment of arrhythmia and sudden cardiac death.
Only recently were efforts undertaken to determine the in vivo role of endosomal pathways in the cardiomyocyte. With the development of new tools, years of discovery in surrogate cell systems may now be translated into mammalian cardiovascular biology. Years of investigation have demonstrated that C-terminal Eps15 homology domain-containing (EHD) proteins have a functional role in each segment of endosome-mediated recycling, degradation, and trafficking. EHDs have therefore recently attracted significant attention as potential therapeutic targets to modulate endosomal function (Gudmundsson et al., 2010, 2012; Curran et al., 2014). As cardiac arrhythmia may arise from dysfunctional expression and organization of multiple membrane proteins leading to altered Na+ and Ca2+ homeostasis, therapeutically modulating EHD proteins may prove efficacious in the treatment of arrhythmia and sudden cardiac death.
EHDs are endocytic regulatory proteins. Discovered in the last decade, four EHD gene products (EHD1–4) are ubiquitously, albeit differentially-expressed across all tissue types (Pohl et al., 2000). These proteins are highly conserved throughout mammalian biology. Indeed, the human and mouse isoforms of EHD1 share 99.6% sequence similarity (Naslavsky and Caplan, 2005). EHD orthologs in non-mammalian species conserve this similarity as well. These data strongly indicate that this family of proteins plays a similar and central role in metazoan cell biology.
The protein family acquires its name from the presence of an epidermal growth factor receptor substrate 15 (Eps15) homology (EH) domain within the C-terminus (Figures 1A,B) (Lee et al., 2005). By itself, the EH domain is well known to mediate protein/protein interactions (Salcini et al., 1997; Paoluzi et al., 1998; Doria et al., 1999; Confalonieri and Di Fiore, 2002; Naslavsky and Caplan, 2005). This domain typically interacts with proteins that contain an NPF (asparagine-proline-phenylalanine) motif (Morgan et al., 2003; Henry et al., 2010; Kieken et al., 2010). Over 50 proteins containing at least one EH domain have been identified in the eukaryotic proteome (Polo et al., 2003; Miliaras and Wendland, 2004). In the C. elegans proteome there are more than 800 proteins that contain at least one NPF motif, with more expected in eukaryotes (Pant et al., 2009). Therefore, the potential for protein/protein interactions in this system is substantial. With particular importance to this review, proteins containing an EH domain are often associated with vesicular trafficking, transport, and sorting (Santolini et al., 1999; Confalonieri and Di Fiore, 2002).
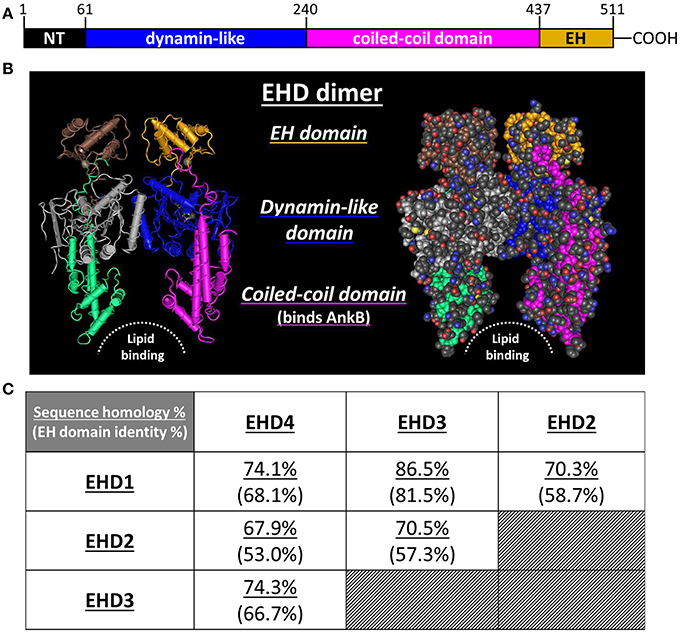
Figure 1. EHD protein structure and sequence homology. (A) Each EHD protein is contains an N-terminal region (NT), a dynamin-like domain, a coiled-coil domain, and a C-terminal EH domain. Typically, the EH domain mediates protein:protein interactions with EHDs. (B) Ribbon and space filling models of an EHD protein, based on the known crystal structure of EHD2 determined by Daumke et al. (2007). (C) The homology of total amino acid sequence and EH domain identity amongst the four EHD proteins expressed in mammals (Naslavsky and Caplan, 2011). Note that the overall sequence homology is greater than the homology shared amongst the EH domains.
Amongst the identified proteins containing one or more EH domains, the overwhelming majority of them contain this domain within the N-terminus. Only a small handful have an EH domain in the C-terminus (Confalonieri and Di Fiore, 2002). Notably however, EHD1–4 all express the EH domain within the C-terminus. The EHD family share high homology with the C. elegans endocytic regulator protein, receptor-mediated endocytosis 1 (RME-1), which also has a C-terminal EH domain. RME-1 is known to mediate endosomal trafficking. Therefore, the distinctive location of the EH domain in mammalian paralogs suggested an in vivo functional role of these proteins. When RME-1 function is disrupted in C. elegans, endosome-based protein recycling is significantly impaired (Grant et al., 2001). Critically, Lin et al. found that expressing the human RME-1 ortholog, EHD1, in C. elegans could fully rescue this phenotype (Lin et al., 2001). This is unambiguous evidence that not only are C-terminal EHD proteins directly involved in endosomal trafficking, but their function is highly conserved across metazoans.
One or more EHDs have been demonstrated to play key roles within every endosome-based protein trafficking compartment. EHDs promote anterograde trafficking from the TGN to the plasma membrane, along with internalization, recycling, and degradation pathways (see Naslavsky and Caplan for an expert review, Naslavsky and Caplan, 2011). While the specific molecular roles of EHD1–4 at the level of the vesicle remains poorly defined, they are hypothesized to serve two simultaneous functions. First, EHDs act as scaffolding proteins for key molecular players within the endosome. EHD1–4 have all been demonstrated to associate with Rab effector proteins (e.g., Rab11-Fip2, Rabenosyn-5, MICAL-L1) within membranous vesicular compartments in vivo (Naslavsky et al., 2004, 2006; George et al., 2007; Sharma et al., 2008, 2010). EHD proteins may recruit Rab effector proteins to the vesicle, although this function is not consistently observed (Naslavsky et al., 2004, 2006). Once present within the vesicle, the Rab effector can bind to individual Rab proteins that, in turn, recruit motor proteins such as myosin and dynein (Roland et al., 2007; Horgan and McCaffrey, 2011; Schafer et al., 2014). Indeed, recent reports have linked EHD1 with dynein motors (Rahajeng et al., 2010).
A second, more established role for EHDs is that of membrane scission. An elegant study by Daumke et al. demonstrated that when the G domain of an EHD is bound to ATP it will dimerize, forming either hetero- or homo-dimers (Figure 1B). This creates a membrane binding pocket within the protein complex (Daumke et al., 2007). These dimers then oligomerize to form ring-like structures around membranous. Upon ATP hydrolysis, the membrane binding pocket collapses, destabilizing the associated membrane, effectively pinching off the vesicle from the tubule to facilitate its transport. Indeed, based on this function, EHD4 is often termed “Pincher” in the literature (Shao et al., 2002; Smith et al., 2004). Combined with the scaffolding role and the association with Rab and Rab effectors, these findings nicely situate EHD proteins to be central players in endosome-based protein trafficking in vivo.
Nearly all that is known regarding the EHD protein family has resulted from investigations using surrogate cell systems or model organisms. The study of endosomal systems in vivo has been hampered by the lack of appropriate tools. Only recently has a concerted effort been made to develop the molecular and biochemical tools and animal models necessary to study these systems in vivo. For this reason, remarkably little is known not just about how these systems function in the heart but even about the identity of the various molecular players involved.
In 2010, Gudmundsson et al. were the first to report that EHD1–4 were differentially expressed in the four chambers of the heart (Gudmundsson et al., 2010). EHD1–4 were each shown to localize within the perinuclear junction and also in discrete puncta spanning from the nucleus out to the plasma membrane, locales consistent with their role in endosomal trafficking (Sharma et al., 2009). Further, a direct protein/protein interaction was reported between EHD3 and the cytoskeletal membrane adapter protein, ankyrin-B (AnkB) (Gudmundsson et al., 2010). Interestingly, this protein/protein interaction was not mediated through the EH domain of EHD3 but rather through the coiled-coil domain. Early in the initial studies of EHD1–4, Naslavsky and Caplan astutely noted that the overall identity shared by EHD1–4 was higher than that shared between their individual EH domains (Figure 1C). They speculated that the conserved functions of EHD1–4 may not rely on the EH domain (Naslavsky and Caplan, 2005). The surprising finding that EHD3 interacts with AnkB through the coiled-coil domain supports this speculation and has potentially broader functional implications for EHD1–4.
Recall, the EH domain directly mediates protein/protein interactions. Moreover, in the case of EHD proteins, the EH domain has been demonstrated to mediate interactions with Rab effectors and Rab proteins. By interacting with cargo proteins, such as AnkB, through the coiled-coil region, this would free up the EH domain to still conduct its business with the motor protein complex (Rabs/Rab effectors). This places the EHD protein in a central position within the vesicular complex. It not only binds to, and recruits the motor complex, it may also simultaneously mediate cargo retention within the endosome. This observation may prove fundamental to our understanding of how EHD-dependent endosomal trafficking occurs in vivo.
In the heart, AnkB regulates cardiac calcium and contractility by controlling the proper targeting and retention of the Na/Ca exchanger (NCX), Na/K ATPase (NKA), inositol 1,4,5-trisphosphate receptor, and protein phosphatase 2A (Mohler et al., 2005; Degrande et al., 2013). Their appropriate subcellular localization is critical to maintaining proper cardiac function. The importance of the relationship between AnkB and these proteins is highlighted in patients harboring single point variants within ANK2, encoding for AnkB (Mohler et al., 2003, 2007). These variants result in a loss-of-function of AnkB and the mislocalization of the associated proteins. Consequently, cardiac function is severely compromised. These patients suffer from a complex arrhythmogenic phenotype ranging from ventricular and atrial fibrillation, sinus node disease, atrioventricular conduction block, and sudden cardiac death (Mohler et al., 2003).
Given the direct interaction between AnkB and EHD3, it was hypothesized that silencing EHD3 expressing in the cardiomyocyte would result a mislocalization of AnkB and its binding partners. Indeed, this was observed. Upon EHD3 silencing by siRNA, AnkB and NCX localization were disrupted. Both proteins were mislocalized within a perinuclear compartment. These data suggest that the cell is still synthesizing the protein, but it was not being trafficked to or retained at the proper subcellular location. In line with this, the NCX-mediated membrane current (INCX) was significantly downregulated (Gudmundsson et al., 2010). Interestingly, this same report also demonstrated that upon EHD3 overexpression in wild type myocytes, INCX increased. This suggests that endosomal pathways could potentially be targeted to fine tune membrane excitability. This was the first evidence that EHD3 (or any EHD) played a functional role in cardiomyocytes to regulate intracellular calcium.
Using newly established mouse models of EHD deficiency, Curran et al. provided the first data on the role of EHDs in the intact heart. EHD3 was found to play critical roles in maintaining membrane excitability and proper Ca2+ homeostasis in vivo (Curran et al., 2014). Hearts from EHD3−/− mice showed dysregulated AnkB and NCX trafficking and targeting. In isolated adult ventricular myocytes lacking EHD3, these proteins were mislocalized within a perinuclear compartment. Interestingly, in the EHD3−/− mouse, NCX protein expression was down by only 20%, while INCX was down by approximately 50%. A similar finding was reported for the L-type Ca channel (LTCC). The loss of EHD3 led to significant mislocalization of the protein. While overall LTCC protein expression was down less than 20%, the peak LTCC-mediated membrane current (ICa,L) was down by approximately 67%. Together, these data suggest that the myocyte is still synthesizing the NCX and LTCC protein, but the loss of EHD3 has limited its ability to properly traffic them to their correct subcellular localizations. Currently, there is no known interaction between the LTCC and AnkB, implying that the LTCC is being trafficked in an EHD3-dependent manner that does not require AnkB. This expands the purview of EHD3 function beyond that of just AnkB-mediated targeting to include direct or indirect interactions with other ion channels and transporters.
The LTCC and NCX are intimately involved in EC coupling and are central players in the maintenance of Ca2+ homeostasis in the heart (Bers, 2002). The ICa,L triggers further Ca2+ release from the sarcoplasmic reticulum (SR) through the SR Ca2+ release channel, ryanodine receptor (RyR), a process called Ca2+-induced Ca2+ release (Bers, 2001). This Ca2+ release induces muscle contraction. The amount of Ca2+ released ultimately dictates the strength of contraction. Ca2+ release is primarily dictated by two mechanisms: the size of the Ca2+ trigger and the size of the SR Ca2+ load. If one of these is downregulated and the other remains the same, the result will be diminished Ca2+ release and strength of contraction, or vice versa.
Therefore, one would expect that dysregulation of the LTCC and NCX observed in the EHD3-deficient mouse would have ramifications on EC coupling. Indeed, in ventricular myocytes isolated from EHD3−/− mice, the average SR Ca2+ load was increased approximately 40%. This is likely a compensatory response to the downregulation of the ICa,L and INCX. The loss of these two membrane currents would favor Ca2+ retention in the cell, thereby increasing the SR Ca2+ concentration (Bers et al., 1996; McCall et al., 1998). This increase in SR Ca2+ concentration would have the effect of sensitizing the RyR to Ca2+, thereby promoting increased Ca2+ release in the face of downregulated Ca2+ trigger (Bassani et al., 1995). In this fashion, contractility was maintained in the EHD3-deficient heart, similar to observations in mice deficient in NCX (Pott et al., 2005).
EHD3-dependent trafficking mechanisms will likely include other ion channels and transporters. The action potential duration (APD) of EHD3−/− myocytes was approximately 60% shorter compared to WT (Curran et al., 2014). While the NCX and the LTCC both play roles in mediating the APD, even if combined the amount of membrane current lost due to dysregulation of these proteins in the EHD3−/− heart cannot account for such a drastic shortening of the APD. This strongly suggests that EHD3 mediates the trafficking of other ion channels or regulatory proteins pertinent to developing the action potential. Future investigations should aim at uncovering the relationship between EHD3 and these other proteins, particularly the family of potassium channels.
Beyond their roles in Ca2+ homeostasis and contraction in the heart, the NCX and LTCC also mediate automaticity and action potential conduction (Lyashkov et al., 2007; Monfredi et al., 2013a,b). In particular, they play vital roles within the “Ca2+ clock” machinery of the sinoatrial node (SAN) where they facilitate spontaneous membrane depolarization and the origin of the cardiac action potential (Maltsev and Lakatta, 2009; Lakatta et al., 2010). Further, LTCC function is required for the proper conduction of the action potential from the atria to the ventricle through the atrioventricular node (Zhang et al., 2011). Disruption of NCX and LTCC function would be expected to have considerable consequences on cardiac rhythm and action potential conduction.
EHD3 is expressed in the SAN (Curran et al., 2014). Given the functional role of EHD3 in trafficking and targeting the NCX and LTCC, dysregulation of cardiac automaticity in these mice would be predicted. Indeed, significant increases in heart rate variability and SAN pause were observed in the EHD3-deficient mouse. Furthermore, incidences of antrioventricular (AV) node conduction block were routinely detected. These observations strongly suggest that EHD3-dependent endosomal trafficking is required for proper cardiac automaticity and conduction. While no data yet exists investigating EHD3 function directly in SAN or AV node cells, it is intriguing to envision that NCX and LTCC trafficking and function are dysregulated in a similar manner as is observed in the ventricle. It is likely that the rhythm and conduction disturbances observed in EHD3-deficient mice are directly related to dysregulated NCX and LTCC function.
In nearly all forms of heart failure, a common observation is the increased expression and function of NCX within the membrane. While initially a compensatory mechanism, this increased NCX expression eventually becomes maladaptive and supplies an arrhythmogenic substrate (Pogwizd et al., 1992, 1999). This membrane remodeling is not limited to human, as increased NCX expression is commonly observed in animal models of heart failure (Baartscheer et al., 2003; Wei et al., 2007; Wang et al., 2009; Kohlhaas and Maack, 2010). This suggests that this response is evolutionarily conserved. Development of a specific pharmacological inhibitor of the NCX aimed at attenuating arrhythmogenesis is an area of active research. However, success in this endeavor has been elusive.
Recent data has indicated that EHD3 is also increased in human heart failure and in all animal models of heart failure yet examined (Gudmundsson et al., 2012). This observation coupled with what is now known about EHD3-dependent NCX trafficking in the heart provides a plausible molecular mechanism by which the heart mediates NCX expression in response to heart failure. These data imply that EHD3 may be involved in the electrical remodeling of the plasma membrane associated with heart failure. If this is the case, EHD-dependent endosomal trafficking may provide a new approach to developing novel therapeutics against arrhythmia. A significant amount of work must be undertaken to more fully describe the function and molecular machinery of these endosomal trafficking pathways in the heart.
A deeper understanding of endosomal trafficking may offer new avenues for therapies against arrhythmia and heart failure. The vast majority of arrhythmias are associated with ion channel or transporter dysfunction. For this reason, the field has pushed for ion channel-based therapeutic strategies. While the logic behind this approach is sound, it has unfortunately been met with limited success. In fact, as the CAST-II trial revealed, when anti-arrhythmic drugs were administered to patients, the rate of arrhythmogenesis in these patients increased (Greene et al., 1992). A secondary approach is therefore needed. Targeting the endosomal trafficking of specific ion channels and transporters may provide that approach.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
This work was funded by NIH Grants F32 HL114252 (to Jerry Curran), HL084583, HL083422, HL114383 (to Peter J. Mohler), and the American Heart Association.
Baartscheer, A., Schumacher, C. A., Belterman, C. N., Coronel, R., and Fiolet, J. W. (2003). [Na+]i and the driving force of the Na+/Ca2+-exchanger in heart failure. Cardiovasc. Res. 57, 986–995. doi: 10.1016/S0008-6363(02)00848-9
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Bassani, J. W., Yuan, W., and Bers, D. M. (1995). Fractional SR Ca release is regulated by trigger Ca and SR Ca content in cardiac myocytes. Am. J. Physiol. 268, C1313–C1319.
Bers, D. M. (2001). Excitation-Contraction Coupling and Cardiac Contractile Force. (Dordrecht: Kluwer Academic Publishers). doi: 10.1007/978-94-010-0658-3
Bers, D. M. (2002). Cardiac excitation-contraction coupling. Nature 415, 198–205. doi: 10.1038/415198a
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Bers, D. M., Bassani, J. W., and Bassani, R. A. (1996). Na-Ca exchange and Ca fluxes during contraction and relaxation in mammalian ventricular muscle. Ann. N.Y. Acad. Sci. 779, 430–442. doi: 10.1111/j.1749-6632.1996.tb44818.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Boucrot, E., and Kirchhausen, T. (2007). Endosomal recycling controls plasma membrane area during mitosis. Proc. Natl. Acad. Sci. U.S.A. 104, 7939–7944. doi: 10.1073/pnas.0702511104
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Confalonieri, S., and Di Fiore, P. P. (2002). The Eps15 homology (EH) domain. FEBS Lett. 513, 24–29. doi: 10.1016/S0014-5793(01)03241-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Curran, J., Makara, M. A., Little, S. C., Musa, H., Liu, B., Wu, X., et al. (2014). EHD3-dependent endosome pathway regulates cardiac membrane excitability and physiology. Circ. Res. 115, 68–78. doi: 10.1161/CIRCRESAHA.115.304149
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Daumke, O., Lundmark, R., Vallis, Y., Martens, S., Butler, P. J., and McMahon, H. T. (2007). Architectural and mechanistic insights into an EHD ATPase involved in membrane remodelling. Nature 449, 923–927. doi: 10.1038/nature06173
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Degrande, S. T., Little, S. C., Nixon, D. J., Wright, P., Snyder, J., Dun, W., et al. (2013). Molecular mechanisms underlying cardiac protein phosphatase 2A regulation in heart. J. Biol. Chem. 288, 1032–1046. doi: 10.1074/jbc.M112.426957
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Doria, M., Salcini, A. E., Colombo, E., Parslow, T. G., Pelicci, P. G., and Di Fiore, P. P. (1999). The eps15 homology (EH) domain-based interaction between eps15 and hrb connects the molecular machinery of endocytosis to that of nucleocytosolic transport. J. Cell Biol. 147, 1379–1384. doi: 10.1083/jcb.147.7.1379
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
George, M., Ying, G., Rainey, M. A., Solomon, A., Parikh, P. T., Gao, Q., et al. (2007). Shared as well as distinct roles of EHD proteins revealed by biochemical and functional comparisons in mammalian cells and C. elegans. BMC Cell Biol. 8:3. doi: 10.1186/1471-2121-8-3
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Grant, B., Zhang, Y., Paupard, M. C., Lin, S. X., Hall, D. H., and Hirsh, D. (2001). Evidence that RME-1, a conserved C. elegans EH-domain protein, functions in endocytic recycling. Nat. Cell. Biol. 3, 573–579. doi: 10.1038/35078549
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Greene, H. L., Roden, D. M., Katz, R. J., Woosley, R. L., Salerno, D. M., and Henthorn, R. W. (1992). The Cardiac Arrhythmia Suppression Trial: first CAST… then CAST-II. J. Am. Coll. Cardiol. 19, 894–898. doi: 10.1016/0735-1097(92)90267-Q
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Gudmundsson, H., Curran, J., Kashef, F., Snyder, J. S., Smith, S. A., Vargas-Pinto, P., et al. (2012). Differential regulation of EHD3 in human and mammalian heart failure. J. Mol. Cell. Cardiol. 52, 1183–1190. doi: 10.1016/j.yjmcc.2012.02.008
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Gudmundsson, H., Hund, T. J., Wright, P. J., Kline, C. F., Snyder, J. S., Qian, L., et al. (2010). EH domain proteins regulate cardiac membrane protein targeting. Circ. Res. 107, 84–95. doi: 10.1161/CIRCRESAHA.110.216713
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Henry, G. D., Corrigan, D. J., Dineen, J. V., and Baleja, J. D. (2010). Charge effects in the selection of NPF motifs by the EH domain of EHD1. Biochemistry 49, 3381–3392. doi: 10.1021/bi100065r
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Horgan, C. P., and McCaffrey, M. W. (2011). Rab GTPases and microtubule motors. Biochem. Soc. Trans. 39, 1202–1206. doi: 10.1042/BST0391202
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Ishii, K., Norota, I., and Obara, Y. (2012). Endocytic regulation of voltage-dependent potassium channels in the heart. J. Pharmacol. Sci. 120, 264–269. doi: 10.1254/jphs.12R12CP
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kieken, F., Sharma, M., Jovic, M., Giridharan, S. S., Naslavsky, N., Caplan, S., et al. (2010). Mechanism for the selective interaction of C-terminal Eps15 homology domain proteins with specific Asn-Pro-Phe-containing partners. J. Biol. Chem. 285, 8687–8694. doi: 10.1074/jbc.M109.045666
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kohlhaas, M., and Maack, C. (2010). Adverse bioenergetic consequences of Na+-Ca2+ exchanger-mediated Ca2+ influx in cardiac myocytes. Circulation 122, 2273–2280. doi: 10.1161/CIRCULATIONAHA.110.968057
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kruse, M., Schulze-Bahr, E., Corfield, V., Beckmann, A., Stallmeyer, B., Kurtbay, G., et al. (2009). Impaired endocytosis of the ion channel TRPM4 is associated with human progressive familial heart block type I. J. Clin. Invest. 119, 2737–2744. doi: 10.1172/JCI38292
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lakatta, E. G., Maltsev, V. A., and Vinogradova, T. M. (2010). A coupled SYSTEM of intracellular Ca2+ clocks and surface membrane voltage clocks controls the timekeeping mechanism of the heart's pacemaker. Circ. Res. 106, 659–673. doi: 10.1161/CIRCRESAHA.109.206078
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lee, D. W., Zhao, X., Scarselletta, S., Schweinsberg, P. J., Eisenberg, E., Grant, B. D., et al. (2005). ATP binding regulates oligomerization and endosome association of RME-1 family proteins. J. Biol. Chem. 280, 17213–17220. doi: 10.1074/jbc.M412751200
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lin, S. X., Grant, B., Hirsh, D., and Maxfield, F. R. (2001). Rme-1 regulates the distribution and function of the endocytic recycling compartment in mammalian cells. Nat. Cell Biol. 3, 567–572. doi: 10.1038/35078543
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lyashkov, A. E., Juhaszova, M., Dobrzynski, H., Vinogradova, T. M., Maltsev, V. A., Juhasz, O., et al. (2007). Calcium cycling protein density and functional importance to automaticity of isolated sinoatrial nodal cells are independent of cell size. Circ. Res. 100, 1723–1731. doi: 10.1161/CIRCRESAHA.107.153676
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Maltsev, V. A., and Lakatta, E. G. (2009). Synergism of coupled subsarcolemmal Ca2+ clocks and sarcolemmal voltage clocks confers robust and flexible pacemaker function in a novel pacemaker cell model. Am. J. Physiol. Heart Circ. Physiol. 296, H594–H615. doi: 10.1152/ajpheart.01118.2008
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Manna, P. T., Smith, A. J., Taneja, T. K., Howell, G. J., Lippiat, J. D., and Sivaprasadarao, A. (2010). Constitutive endocytic recycling and protein kinase C-mediated lysosomal degradation control K(ATP) channel surface density. J. Biol. Chem. 285, 5963–5973. doi: 10.1074/jbc.M109.066902
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
McCall, E., Ginsburg, K. S., Bassani, R. A., Shannon, T. R., Qi, M., Samarel, A. M., et al. (1998). Ca flux, contractility, and excitation-contraction coupling in hypertrophic rat ventricular myocytes. Am. J. Physiol. 274, H1348–H1360.
McEwen, D. P., Schumacher, S. M., Li, Q., Benson, M. D., Iniguez-Lluhi, J. A., Van Genderen, K. M., et al. (2007). Rab-GTPase-dependent endocytic recycling of Kv1.5 in atrial myocytes. J. Biol. Chem. 282, 29612–29620. doi: 10.1074/jbc.M704402200
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Miliaras, N. B., and Wendland, B. (2004). EH proteins: multivalent regulators of endocytosis (and other pathways). Cell Biochem. Biophys. 41, 295–318. doi: 10.1385/CBB:41:2:295
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Mohler, P. J., Davis, J. Q., and Bennett, V. (2005). Ankyrin-B coordinates the Na/K ATPase, Na/Ca exchanger, and InsP3 receptor in a cardiac T-tubule/SR microdomain. PLoS Biol. 3:e423. doi: 10.1371/journal.pbio.0030423
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Mohler, P. J., Healy, J. A., Xue, H., Puca, A. A., Kline, C. F., Allingham, R. R., et al. (2007). Ankyrin-B syndrome: enhanced cardiac function balanced by risk of cardiac death and premature senescence. PLoS ONE 2:e1051. doi: 10.1371/journal.pone.0001051
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Mohler, P. J., Schott, J. J., Gramolini, A. O., Dilly, K. W., Guatimosim, S., Dubell, W. H., et al. (2003). Ankyrin-B mutation causes type 4 long-QT cardiac arrhythmia and sudden cardiac death. Nature 421, 634–639. doi: 10.1038/nature01335
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Monfredi, O., Maltseva, L. A., Spurgeon, H. A., Boyett, M. R., Lakatta, E. G., and Maltsev, V. A. (2013b). Beat-to-beat variation in periodicity of local calcium releases contributes to intrinsic variations of spontaneous cycle length in isolated single sinoatrial node cells. PLoS ONE 8:e67247. doi: 10.1371/journal.pone.0067247
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Monfredi, O., Maltsev, V. A., and Lakatta, E. G. (2013a). Modern concepts concerning the origin of the heartbeat. Physiology (Bethesda) 28, 74–92. doi: 10.1152/physiol.00054.2012
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Morgan, J. R., Prasad, K., Jin, S., Augustine, G. J., and Lafer, E. M. (2003). Eps15 homology domain-NPF motif interactions regulate clathrin coat assembly during synaptic vesicle recycling. J. Biol. Chem. 278, 33583–33592. doi: 10.1074/jbc.M304346200
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Naslavsky, N., Boehm, M., Backlund, P. S. Jr., and Caplan, S. (2004). Rabenosyn-5 and EHD1 interact and sequentially regulate protein recycling to the plasma membrane. Mol. Biol. Cell 15, 2410–2422. doi: 10.1091/mbc.E03-10-0733
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Naslavsky, N., and Caplan, S. (2005). C-terminal EH-domain-containing proteins: consensus for a role in endocytic trafficking, EH? J. Cell Sci. 118, 4093–4101. doi: 10.1242/jcs.02595
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Naslavsky, N., and Caplan, S. (2011). EHD proteins: key conductors of endocytic transport. Trends Cell Biol. 21, 122–131. doi: 10.1016/j.tcb.2010.10.003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Naslavsky, N., Rahajeng, J., Sharma, M., Jovic, M., and Caplan, S. (2006). Interactions between EHD proteins and Rab11-FIP2: a role for EHD3 in early endosomal transport. Mol. Biol. Cell 17, 163–177. doi: 10.1091/mbc.E05-05-0466
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Palacios, F., Schweitzer, J. K., Boshans, R. L., and D'Souza-Schorey, C. (2002). ARF6-GTP recruits Nm23-H1 to facilitate dynamin-mediated endocytosis during adherens junctions disassembly. Nat. Cell Biol. 4, 929–936. doi: 10.1038/ncb881
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pant, S., Sharma, M., Patel, K., Caplan, S., Carr, C. M., and Grant, B. D. (2009). AMPH-1/Amphiphysin/Bin1 functions with RME-1/Ehd1 in endocytic recycling. Nat. Cell Biol. 11, 1399–1410. doi: 10.1038/ncb1986
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Paoluzi, S., Castagnoli, L., Lauro, I., Salcini, A. E., Coda, L., Fre, S., et al. (1998). Recognition specificity of individual EH domains of mammals and yeast. EMBO J. 17, 6541–6550. doi: 10.1093/emboj/17.22.6541
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pogwizd, S. M., Hoyt, R. H., Saffitz, J. E., Corr, P. B., Cox, J. L., and Cain, M. E. (1992). Reentrant and focal mechanisms underlying ventricular tachycardia in the human heart. Circulation 86, 1872–1887. doi: 10.1161/01.CIR.86.6.1872
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pogwizd, S. M., Qi, M., Yuan, W., Samarel, A. M., and Bers, D. M. (1999). Upregulation of Na(+)/Ca(2+) exchanger expression and function in an arrhythmogenic rabbit model of heart failure. Circ. Res. 85, 1009–1019. doi: 10.1161/01.RES.85.11.1009
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pohl, U., Smith, J. S., Tachibana, I., Ueki, K., Lee, H. K., Ramaswamy, S., et al. (2000). EHD2, EHD3, and EHD4 encode novel members of a highly conserved family of EH domain-containing proteins. Genomics 63, 255–262. doi: 10.1006/geno.1999.6087
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Polo, S., Confalonieri, S., Salcini, A. E., and Di Fiore, P. P. (2003). EH and UIM: endocytosis and more. Sci STKE 2003:re17. doi: 10.1126/stke.2132003re17.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pott, C., Philipson, K. D., and Goldhaber, J. I. (2005). Excitation-contraction coupling in Na+-Ca2+ exchanger knockout mice: reduced transsarcolemmal Ca2+ flux. Circ. Res. 97, 1288–1295. doi: 10.1161/01.RES.0000196563.84231.21
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Rahajeng, J., Giridharan, S. S., Naslavsky, N., and Caplan, S. (2010). Collapsin response mediator protein-2 (Crmp2) regulates trafficking by linking endocytic regulatory proteins to dynein motors. J. Biol. Chem. 285, 31918–31922. doi: 10.1074/jbc.C110.166066
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Roland, J. T., Kenworthy, A. K., Peranen, J., Caplan, S., and Goldenring, J. R. (2007). Myosin Vb interacts with Rab8a on a tubular network containing EHD1 and EHD3. Mol. Biol. Cell. 18, 2828–2837. doi: 10.1091/mbc.E07-02-0169
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Salcini, A. E., Confalonieri, S., Doria, M., Santolini, E., Tassi, E., Minenkova, O., et al. (1997). Binding specificity and in vivo targets of the EH domain, a novel protein-protein interaction module. Genes Dev. 11, 2239–2249. doi: 10.1101/gad.11.17.2239
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Santolini, E., Salcini, A. E., Kay, B. K., Yamabhai, M., and Di Fiore, P. P. (1999). The EH network. Exp. Cell Res. 253, 186–209. doi: 10.1006/excr.1999.4694
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Schafer, J. C., Baetz, N. W., Lapierre, L. A., McRae, R. E., Roland, J. T., and Goldenring, J. R. (2014). Rab11-FIP2 interaction with MYO5B regulates movement of Rab11a-containing recycling vesicles. Traffic 15, 292–308. doi: 10.1111/tra.12146
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Shao, Y., Akmentin, W., Toledo-Aral, J. J., Rosenbaum, J., Valdez, G., Cabot, J. B., et al. (2002). Pincher, a pinocytic chaperone for nerve growth factor/TrkA signaling endosomes. J. Cell Biol. 157, 679–691. doi: 10.1083/jcb.200201063
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Sharma, M., Giridharan, S. S., Rahajeng, J., Caplan, S., and Naslavsky, N. (2010). MICAL-L1: an unusual Rab effector that links EHD1 to tubular recycling endosomes. Commun. Integr. Biol. 3, 181–183. doi: 10.4161/cib.3.2.10845
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Sharma, M., Giridharan, S. S., Rahajeng, J., Naslavsky, N., and Caplan, S. (2009). MICAL-L1 links EHD1 to tubular recycling endosomes and regulates receptor recycling. Mol. Biol. Cell 20, 5181–5194. doi: 10.1091/mbc.E09-06-0535
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Sharma, M., Naslavsky, N., and Caplan, S. (2008). A role for EHD4 in the regulation of early endosomal transport. Traffic 9, 995–1018. doi: 10.1111/j.1600-0854.2008.00732.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Shivas, J. M., Morrison, H. A., Bilder, D., and Skop, A. R. (2010). Polarity and endocytosis: reciprocal regulation. Trends Cell Biol. 20, 445–452. doi: 10.1016/j.tcb.2010.04.003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Smith, C. A., Dho, S. E., Donaldson, J., Tepass, U., and McGlade, C. J. (2004). The cell fate determinant numb interacts with EHD/Rme-1 family proteins and has a role in endocytic recycling. Mol. Biol. Cell 15, 3698–3708. doi: 10.1091/mbc.E04-01-0026
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Sun, H., Lu, L., Zuo, Y., Wang, Y., Jiao, Y., Zeng, W. Z., et al. (2014). Kainate receptor activation induces glycine receptor endocytosis through PKC deSUMOylation. Nat. Commun. 5, 4980. doi: 10.1038/ncomms5980.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Traynor, D., and Kay, R. R. (2007). Possible roles of the endocytic cycle in cell motility. J. Cell Sci. 120, 2318–2327. doi: 10.1242/jcs.007732
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Wang, S., Ziman, B., Bodi, I., Rubio, M., Zhou, Y. Y., D'Souza, K., et al. (2009). Dilated cardiomyopathy with increased SR Ca2+ loading preceded by a hypercontractile state and diastolic failure in the alpha(1C)TG mouse. PLoS ONE 4:e4133. doi: 10.1371/journal.pone.0004133
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Wang, Z., Edwards, J. G., Riley, N., Provance, D. W. Jr. Karcher, R., Li, X. D., et al. (2008). Myosin Vb mobilizes recycling endosomes and AMPA receptors for postsynaptic plasticity. Cell 135, 535–548. doi: 10.1016/j.cell.2008.09.057
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Wei, S. K., McCurley, J. M., Hanlon, S. U., and Haigney, M. C. (2007). Gender differences in Na/Ca exchanger current and beta-adrenergic responsiveness in heart failure in pig myocytes. Ann. N.Y. Acad. Sci. 1099, 183–189. doi: 10.1196/annals.1387.026
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Zhang, Q., Timofeyev, V., Qiu, H., Lu, L., Li, N., Singapuri, A., et al. (2011). Expression and roles of Cav1.3 (alpha1D) L-type Ca(2)+ channel in atrioventricular node automaticity. J. Mol. Cell. Cardiol. 50, 194–202. doi: 10.1016/j.yjmcc.2010.10.002
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Keywords: endosome, protein trafficking, cardiac membrane excitability, Ca2+ homeostasis, heart failure
Citation: Curran J, Makara MA and Mohler PJ (2015) Endosome-based protein trafficking and Ca2+ homeostasis in the heart. Front. Physiol. 6:34. doi: 10.3389/fphys.2015.00034
Received: 29 October 2014; Accepted: 21 January 2015;
Published online: 09 February 2015.
Edited by:
Christopher Huang, University of Cambridge, UKReviewed by:
Ravi C. Balijepalli, University of Wisconsin, USACopyright © 2015 Curran, Makara and Mohler. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jerry Curran, The Davis Heart and Lung Research Institute, The Ohio State University, 473 W. 12th Ave., Columbus, OH 43210, USA e-mail:amVycnkuY3VycmFuQG9zdW1jLmVkdQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.