
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol. , 18 March 2025
Sec. Obstetric and Pediatric Pharmacology
Volume 16 - 2025 | https://doi.org/10.3389/fphar.2025.1558966
This article is part of the Research Topic Exploring Adverse Drug Reactions, Medication Adherence, and Forensic Markers in Pediatrics and Obstetrics View all 8 articles
Introduction: The combination of remimazolam and esketamine effectively alleviates adverse hemodynamic effects, such as tachycardia and hypertension, during intubation. However, the dosage for achieving optimal intubation conditions when co-administered with remifentanil remains unestablished. Therefore, this study aimed to determine the effective doses of remimazolam and esketamine for endotracheal intubation without muscle relaxants in pediatric patients using Dixon’s up-and-down method.
Methods: This prospective, non-controlled, non-randomized clinical trial sequentially allocated 41 children aged 3–6 into two phases. All patients underwent tracheal intubation under general anesthesia. Patients received a fixed dose of remifentanil at 2.5 μg/kg via a pump over 90 s. In the first phase, the induction dose of remimazolam was set at 0.2 mg/kg. The first patient received esketamine at a dose of 0.5 mg/kg, administered with a dose gradient of 0.2 mg/kg based on Dixon’s up-and-down method. 50% effective dose (ED50) and 95% effective dose (ED95) for esketamine were then measured through probit regression analysis. Similarly, in the second phase, the ED95 of esketamine was fixed. The first patient received remimazolam at a dose of 0.2 mg/kg, administered at a dose gradient of 0.1 mg/kg. ED50 and ED95 for remimazolam were then measured. Intubation conditions were assessed via the Copenhagen scale. Heart rate (HR) and mean arterial pressure (MAP) were recorded at the following time points: Just before intubation (T1) and 1 min after intubation (T2). Adverse events were also recorded during anesthesia induction.
Results: At a fixed dose of remifentanil (2.5 μg/kg), the ED50 of esketamine was 0.74 mg/kg (95% confidence interval [CI]: 0.61–0.89 mg/kg), while the ED95 was 0.97 mg/kg (95% CI: 0.85–1.75 mg/kg). The ED50 of remimazolam was 0.39 mg/kg (95% CI: 0.29–0.53 mg/kg), while the ED95 was 0.56 mg/kg (95% CI: 0.46–1.47 mg/kg). Hemodynamic stability was maintained during anesthesia induction, with no significant adverse events observed.
Conclusion: The ED50 and ED95 values of remimazolam and esketamine in this study provide initial dosing references for pediatric endotracheal intubation without muscle relaxants. A fixed dose of 2.5 μg/kg remifentanil combined with these agents is safe and effective in children aged 3–6 years, though further multicenter studies are recommended for validation.
Clinical trial registration: www.chictr.org.cn, number: ChiCTR2200063847.
Endotracheal intubation is crucial in pediatric anesthesia, often requiring sedatives and analgesics to facilitate intubation and reduce patient discomfort. Muscle relaxants aid pediatric tracheal intubation but risk prolonged paralysis and neuromuscular recovery issues (Tamire et al., 2021). Intubation without muscle relaxants has recently been explored as an alternative approach, with many studies demonstrating that omitting muscle relaxants during pediatric anesthesia intubation can effectively alleviate muscular discomfort and expedite postoperative recovery (Karanth et al., 2018; Naziri et al., 2015; Rizvanović et al., 2017).
Remimazolam, a fast-acting benzodiazepine, offers quick onset and recovery, and can be swiftly reversed with flumazenil, indicating significant potential for pediatric anesthesia (Buchwald, 2020; Kilpatrick et al., 2007). However, esketamine, a stereoisomer of ketamine, provides potent analgesic effects with mild sedative properties and is known for its protective airway reflexes and hemodynamic stability (Ihmsen et al., 2001; Turner, 2019). We hypothesized that combining remimazolam and esketamine may offer an effective strategy for endotracheal intubation in pediatric patients, potentially eliminating the need for traditional muscle relaxants.
Determining the optimal dosing of these agents is crucial to enhancing their therapeutic efficacy while minimizing potential adverse effects. However, studies on their use in pediatric anesthesia are limited, particularly regarding their application in intubation without muscle relaxants. This study investigated the effective doses of remimazolam and esketamine for endotracheal without muscular relaxation in pediatric patients aged 3–6 years.
This prospective, non-controlled, non-randomized clinical trial employed Dixon’s up-and-down sequential allocation method. Approved by the Ethics Committee of the Affiliated Shunde Hospital of Jinan University (JDSY-LL-2022042, 02/04/2022), the trial was registered with the Chinese Clinical Trial Registry (www.chictr.org.cn, number: ChiCTR2200063847, 19/09/2022). Participants were recruited from the Hernia Surgery Department or the Otolaryngology Department of our hospital. All procedures strictly adhere to the relevant guidelines and regulations established by the Affiliated Shunde Hospital of Jinan University and comply with the Declaration of Helsinki and CIOMS guidelines, ensuring compliance with international ethical standards. Parents or guardians were provided with detailed information about the study’s risks and benefits, and written informed consent was obtained before enrollment.
Inclusion criteria: Participants were children aged 3 to 6, undergoing adenotonsillectomy or pediatric hydrocele surgery under general anesthesia with endotracheal intubation. They were classified as American Society of Anesthesiologists (ASA) physical status I or II, with a body mass index (BMI) of 13–22 kg/m2 and weighing 10–40 kg. There were no gender restrictions, and participants were normally developed.
Exclusion criteria: Allergy to remimazolam or esketamine, upper respiratory tract infection, severe heart and lung disease, airway hyperresponsiveness, and difficult airways.
The pediatric patients fasted for over 6 h and refrained from drinking clear liquids for at least 2 h before operation. Electrocardiogram, peripheral capillary oxygen saturation (SpO2), heart rate (HR), and mean arterial pressure (MAP) were monitored using a multifunction device upon entering the operating room. Patients received oxygen supplementation via a face mask at a flow rate of 3 L/min. The baseline of SpO2, HR, and MAP were measured 3 min after resting.
All pediatric patients were administered remifentanil (Jiangsu Enhua Pharmaceutical Co., China, diluted to 20 μg/mL with normal saline, lot number: TRF23I02) at a dose of 2.5 μg/kg using standard solution (pump, administered over 90 s) (Bai et al., 2024; Hirano et al., 2023). In the first phase, 0.2 mg/kg of remimazolam (Jiangsu Hengrui Medicine Co., China, diluted to 1 mg/mL with normal saline; lot number 220326 A K) was injected intravenously (no less than 10 s) in a fixed dose during anesthesia induction. Different dosages of esketamine (Jiangsu Hengrui Medicine Co., China, diluted to 2.5 mg/mL with normal saline; lot number 230908 B L) were administered using Dixon’s up-and-down sequential method. After intubation, analgosedation was maintained with a continuous infusion of remifentanil at 0.1–0.2 μg/kg/min and intermittent boluses of remimazolam (0.05–0.1 mg/kg) as needed to maintain adequate anesthesia depth. Esketamine was not administered post-intubation to avoid potential cumulative effects. In the first phase, the first patient received esketamine at 0.5 mg/kg, and subsequent patients were administered at a 0.2 mg/kg dose gradient. Once the eyelash reaction disappeared, indicating adequate anesthesia depth, an experienced anesthesiologist performed intubation using a visual laryngoscope, and the intubation response was evaluated. If the response to intubation was negative (effective), the dose gradient during induction was reduced for the subsequent case. Conversely, if the response was positive (ineffective), the dose gradient was increased. This process continued until the test was stopped after the occurrence of six positive and negative crosspoints (Pace and Stylianou, 2007). In the second phase, different dosages of remimazolam were administered intravenously, followed by a fixed dose of esketamine, which was the ED95 determined in the first phase. The first patient received 0.2 mg/kg of remimazolam, with subsequent doses administered in 0.1 mg/kg increments or decrements. The same assessment method used in the first phase was employed to evaluate the tracheal intubation and the response to intubation. Children were sequentially enrolled and assigned to receive either esketamine or remimazolam at varying doses based on the response of the previous patient, following Dixon’s up-and-down sequential allocation method.
Primary outcome measures: The endotracheal intubation response was assessed based on the Copenhagen scale (Viby-Mogensen et al., 1996). The Copenhagen scale is a validated tool for assessing intubation conditions, evaluating factors such as jaw relaxation, vocal cord visibility, and limb movement during intubation.
If the intubation response is negative, the drug dosage is effective, and the intubation conditions are satisfactory (Viby-Mogensen, et al., 1996). Specifically, this situation is reflected in the relaxation of the jaw during laryngoscopic intubation, allowing for smooth laryngoscopy, clear visibility of the vocal cords, and no coughing or limb movement responses. Additionally, the absence of responses to intubation stimuli means that the conditions for tracheal intubation have reached an excellent standard, with changes in MAP and HR before and after intubation being less than 20%. Conversely, if the intubation response is positive, it indicates that the drug dosage used is ineffective. If the mandible was stiff and the intubation condition was unsatisfactory, 0.2 mg/kg of mivacurium was added. If the MAP or HR increased by over 20% within 2 min after tracheal intubation, remimazolam was added to deepen anesthesia.
The secondary outcomes included HR and MAP measured at the following time points: Just before intubation (T1) and 1 min after intubation (T2). Hypotension, hypertension, bradycardia, tachycardia, SpO2 < 90%, bucking, mandibular stiffness, body movement, and glottal closure were recorded.
Adverse events were managed as follows: hypotension (MAP decreased by 20% or more from baseline) was treated with intravenous (IV) ephedrine 3–6 mg; hypertension (systolic pressure ≥140 mmHg or diastolic pressure ≥90 mmHg) was treated with IV remimazolam 0.1 mg/kg and sufentanil 5 μg; SpO2 < 90% was managed by applying positive pressure oxygen via a mask and increasing the depth of anesthesia; bradycardia (HR < 60 beats/min) was treated with IV atropine 0.2–0.3 mg; and tachycardia (HR exceeded 20% of baseline) was addressed with IV remimazolam 0.1 mg/kg or sufentanil 5 µg.
The sample size determination was performed using Dixon’s up-and-down method, requiring seven crosspoints (effective to ineffective) for statistical analysis (Pace and Stylianou, 2007). Statistical analyses were conducted using the Statistical Package for the Social Sciences (SPSS) software, version 26.0 (IBM Corp., Armonk, NY, United States). Data are presented as means ± standard deviation (SD) or as frequencies and percentages (n [%]). Comparisons between phases were conducted using the t-test, while categorical data were analyzed using the chi-square test or Fisher’s exact test. ED50 and ED95 of remimazolam and esketamine were determined through probit regression analysis, with a p-value of less than 0.05 considered indicative of statistical significance.
This study enrolled 44 pediatric patients across two phases and excluded 3 between January 2023 and February 2024. In the first phase, 21 pediatric patients were included for analysis, while 20 pediatric patients were included in the second phase, as displayed in the study flowchart (Figure 1). In the first phase, 10 cases were classified as “effective,” while 11 cases were identified as “ineffective” (Figure 2). In the second phase, 9 cases were classified as “effective” while 11 cases were identified as “ineffective” (Figure 3). There were no statistically significant differences in ASA, age, height, weight, and BMI among all patients (p > 0.05) as presented in Tables 1, 2.
Probit regression analysis, based on a fixed dose of 2.5 μg/kg remifentanil, revealed that the ED50 of esketamine was 0.74 mg/kg (95% confidence interval [CI]: 0.61–0.89 mg/kg) and the ED95 was 0.97 mg/kg (95% CI: 0.85–1.75 mg/kg) in the first phase. Based on the ED95 of esketamine in the first phase, the ED50 of remimazolam was 0.39 mg/kg (95% CI: 0.29–0.53 mg/kg), and the ED95 was 0.56 mg/kg (95% CI: 0.46–1.47 mg/kg) (Figure 4).
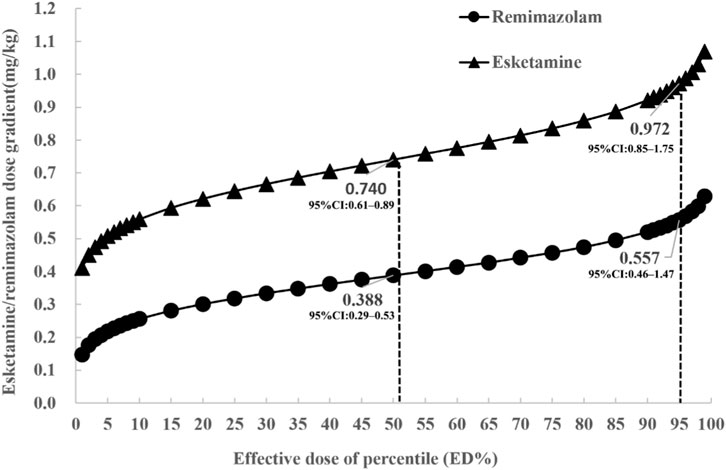
Figure 4. Dose⁃effect analysis of the induction doses of esketamine and remimazolam on inhibition of tracheal intubation response in both phases.
The HR increased significantly at T2 in both phases (p < 0.05), this may be related to the sympathetic excitatory effect of esketamine and the stimulation of intubation. In the second phase, the MAP at T2 significantly increased compared to T1 (p = 0.006), whereas no significant differences in MAP were observed between T1 and T2 in the first phase (p = 0.269), as illustrated in Table 3. This difference may be attributed to the higher dose of esketamine used in the second phase, which could have led to greater sympathetic stimulation and subsequent elevation in blood pressure.
No statistically significant differences in HR and MAP between the phases at T1 and T2 time point (p > 0.05).
No significant difference was observed in the incidence of adverse events during anesthesia induction (p > 0.05) (Table 4). However, the incidence of tachycardia (85.7% in Phase 1 and 85% in Phase 2) highlights the need for careful monitoring of HR during anesthesia induction.
Based on a fixed dose of 2.5 μg/kg remifentanil, ED50/ED95 of esketamine was 0.74 mg/kg (95% CI:0.61–0.89) and 0.97 mg/kg (95% CI: 0.85–1.75), respectively. Similarly, ED50/ED95 of remimazolam was 0.39 mg/kg (95% CI: 0.29–0.53) and 0.56 mg/kg (95% CI: 0.46–1.47), respectively. These findings can provide initial dosing references for children aged 3–6 undergoing tracheal intubation without muscle relaxation.
The results indicated that during the second phase, MAP at T2 was significantly higher than at T1 (p < 0.05), whereas no significant differences in MAP were observed between T1 and T2 in the first phase (p > 0.05). The possible reason is that the second phase involved administering the ED95 of esketamine, which was marginally higher than the dose used in the first phase. Moreover, esketamine has the property of stabilizing hemodynamic parameters (Del Sant et al., 2020; Zhao et al., 2024), especially in pediatric cardiovascular anesthesia (Shimizu et al., 2024). In this study, the effect of esketamine on HR increased with dosage; however, its impact on blood pressure was not evident, possibly due to its effects on the sympathetic nervous system and catecholamine levels (Mihaljević et al., 2020). Although the incidence of tachycardia exceeded 80% in both phases and remained manageable without special treatment, these findings highlight the importance of continuous hemodynamic monitoring when using this drug combination in pediatric patients. Remifentanil primarily suppresses HR, while esketamine increases HR and bronchial relaxation (Zanos et al., 2018). Using a low dose of remifentanil reduces pharyngeal reflexes and sympathetic nerve stimulation during intubation (Kang et al., 2022), which helps avoid side effects from increased dosages of esketamine and remimazolam. This drug regimen, comprising fast-acting, short-duration medications, promotes quick recovery in children, prevents the onset of hyperalgesia (increased sensitivity to pain), and enhances patient comfort (Colvin et al., 2019). However, it is crucial to recognize that the safety results in this study are secondary and should be viewed cautiously because of the relatively small sample size. While no patient required treatment for bradycardia or hypotension during the trial, further studies with larger cohorts are needed to confirm the safety profile of this drug combination.
This study found that six children (two in the first phase and four in the second phase) experienced glottal closure during intubation. The symptoms resolved after deepening the anesthesia and administering muscle relaxants, indicating that using anesthesia without muscle relaxants in children can be risky. A research revealed that hypoxia and laryngospasm constitute approximately 30% of respiratory events, with intubation difficulties accounting for 13% and bronchospasm for 7% during pediatric anesthesia (Von Ungern-Sternberg and Habre, 2007). Consequently, implementing appropriate preventive and therapeutic measures in pediatric anesthesia is imperative.
Although the ED95 of esketamine in this study is larger than that reported in previous studies (Su et al., 2023; Wang et al., 2022; Zheng et al., 2023; Zheng et al., 2022; Zhong et al., 2023), it still falls within the recommended safe range of 0.5–1 mg/kg for induction and maintenance anesthesia (Kohtala, 2021; Trimmel et al., 2018). Possible reasons include: (1) Compared to propofol, remimazolam has a milder cardiovascular depressant effect, and its overall efficacy is lower (Fang et al., 2023); (2) the dosage requirements of esketamine can differ among children in various age groups (Leroux et al., 2021); (3) the effective dose of esketamine was determined by administering a relatively small dose of remimazolam at 0.2 mg/kg. Consequently, despite being slightly larger than expected, the determined ED95 for esketamine at 0 0.97 mg/kg (95% CI: 0.85–1.75 mg/kg) remains credible and suitable for clinical application.
The standard sample size for the modified sequential method is 20–40 cases (Wei et al., 2015). This test included over 20 cases in each stage, fully complying with the testing methodology’s requirements. The primary objective of this study was to assess the changes in intubation conditions and hemodynamics both before and after intubation, regardless of the surgical. The study results revealed that no significant circulatory inhibition was observed with increased dosages of esketamine. Moreover, a higher dose of remimazolam increased effectiveness in inhibiting reactions to endotracheal intubation. However, lower doses of remimazolam resulted in elevated reactions, such as body movement and coughing, and substantial fluctuations in MAP and HR before and after intubation.
This study has several limitations. First, our study focused on children aged 3–6 years, and the dosing requirements may differ for younger or older pediatric patients. Younger children, for example, may require lower doses due to differences in drug metabolism, while older children may need higher doses to achieve the same effect. Future studies should explore the dosing requirements across a broader age range to provide more clinical practice recommendations. Second, lack of bispectral index monitoring for anesthesia depth is a notable limitation, as it could have provided more precise data on the depth of sedation during intubation. Finally, while our findings provide valuable insights, the single-center design may limit the generalizability of our findings to other settings with different patient populations or clinical practices. Future multicenter studies with larger cohorts are needed to validate these findings and make them applicable to a larger group of children.
In conclusion, the ED50 and ED95 values of remimazolam and esketamine determined in this study provide initial dosing references for clinical practice. Administering a fixed dose of 2.5 μg/kg remifentanil combined with these agents can be safely and effectively used for endotracheal intubation without muscle relaxants in pediatric patients aged 3–6 years. However, further validation through larger, multicenter studies is recommended.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by the Ethics Committee of the Affiliated Shunde Hospital of Jinan University. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants’ legal guardians/next of kin. Written informed consent was obtained from the individual(s), and minor(s)’ legal guardian/next of kin, for the publication of any potentially identifiable images or data included in this article.
JC: Methodology, Project administration, Writing–original draft, Writing–review and editing. YM: Formal Analysis, Writing–original draft, Writing–review and editing. XC: Data curation, Writing–original draft, Writing–review and editing. HS: Data curation, Writing–original draft, Writing–review and editing. ZC: Investigation, Software, Writing–original draft, Writing–review and editing. ZZ: Funding acquisition, Resources, Writing–original draft, Writing–review and editing.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. Medical Scientific Research Project of Foshan Municipal Health Bureau, Number:20230245; Foshan Self-funded Science and Technology Innovation Project, Number: 2220001003819; Key medical talents training project of Shunde District; Medical Science and Technology Research Foundation of Guangdong Province, Number: A2022515. Medical. Association Foundation of Jinan University (YXJC2022003).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Bai, C., Xu, M., Guo, Y., Jin, Y., and Zhao, X. (2024). Clinical application and research progress of remimazolam for pediatric patients. Drug Des. Devel Ther. 18, 1221–1229. doi:10.2147/dddt.S453440
Buchwald, P. (2020). Soft drugs: design principles, success stories, and future perspectives. Expert Opin. Drug Metab. Toxicol. 16, 645–650. doi:10.1080/17425255.2020.1777280
Colvin, L. A., Bull, F., and Hales, T. G. (2019). Perioperative opioid analgesia-when is enough too much? A review of opioid-induced tolerance and hyperalgesia. Lancet 393, 1558–1568. doi:10.1016/s0140-6736(19)30430-1
Del Sant, L. C., Sarin, L. M., Magalhães, E. J. M., Lucchese, A. C., Tuena, M. A., Nakahira, C., et al. (2020). Effects of subcutaneous esketamine on blood pressure and heart rate in treatment-resistant depression. J. Psychopharmacol. 34, 1155–1162. doi:10.1177/0269881120922955
Fang, Y. B., Wang, C. Y., Gao, Y. Q., Cai, Y. H., Chen, J., Zhang, X. L., et al. (2023). The safety and efficacy of remimazolam tosylate for induction and maintenance of general anesthesia in pediatric patients undergoing elective surgery: study protocol for a multicenter, randomized, single-blind, positive-controlled clinical trial. Front. Pharmacol. 14, 1090608. doi:10.3389/fphar.2023.1090608
Hirano, T., Kimoto, Y., Kuratani, N., Cavanaugh, D., and Mason, K. P. (2023). Remimazolam for pediatric procedural sedation: results of an institutional pilot program. J. Clin. Med. 12, 5937. doi:10.3390/jcm12185937
Ihmsen, H., Geisslinger, G., and Schüttler, J. (2001). Stereoselective pharmacokinetics of ketamine: R(-)-ketamine inhibits the elimination of S(+)-ketamine. Clin. Pharmacol. Ther. 70, 431–438. doi:10.1067/mcp.2001.119722
Kang, S., Chae, Y. J., Bae, S. Y., Song, J. Y., and Joe, H. B. (2022). Remifentanil requirement for acceptable intubation conditions with two different doses of ketamine without a neuromuscular blocking agent in pediatric patients. Eur. Rev. Med. Pharmacol. Sci. 26, 1632–1639. doi:10.26355/eurrev_202203_28231
Karanth, H., Raveendra, U. S., Shetty, R. B., Shetty, P., and Thalanjeri, P. (2018). Comparative evaluation between sevoflurane and propofol for endotracheal intubation without muscle relaxants in pediatric cleft surgeries. Anesth. Essays Res. 12, 434–439. doi:10.4103/aer.AER_38_18
Kilpatrick, G. J., McIntyre, M. S., Cox, R. F., Stafford, J. A., Pacofsky, G. J., Lovell, G. G., et al. (2007). CNS 7056: a novel ultra-short-acting Benzodiazepine. Anesthesiology 107, 60–66. doi:10.1097/01.anes.0000267503.85085.c0
Kohtala, S. (2021). Ketamine-50 years in use: from anesthesia to rapid antidepressant effects and neurobiological mechanisms. Pharmacol. Rep. 73, 323–345. doi:10.1007/s43440-021-00232-4
Leroux, S., Mechinaud-Heloury, F., and Jacqz-Aigrain, E. (2021). Contribution of population pharmacokinetics of glycopeptides and antifungals to dosage adaptation in paediatric onco-hematological malignancies: a review. Front. Pharmacol. 12, 635345. doi:10.3389/fphar.2021.635345
Mihaljević, S., Pavlović, M., Reiner, K., and Ćaćić, M. (2020). Therapeutic mechanisms of ketamine. Psychiatr. Danub 32, 325–333. doi:10.24869/psyd.2020.325
Naziri, F., Amiri, H. A., Rabiee, M., Banihashem, N., Nejad, F. M., Shirkhani, Z., et al. (2015). Endotracheal intubation without muscle relaxants in children using remifentanil and propofol: comparative study. Saudi J. Anaesth. 9, 409–412. doi:10.4103/1658-354x.159465
Pace, N. L., and Stylianou, M. P. (2007). Advances in and limitations of up-and-down methodology: a précis of clinical use, study design, and dose estimation in anesthesia research. Anesthesiology 107, 144–152. doi:10.1097/01.anes.0000267514.42592.2a
Rizvanović, N., Čaušević, S., and Šabanović, A. (2017). Conditions of endotracheal intubation with and without muscle relaxant in children. Med. Glas. (Zenica) 14, 41–48. doi:10.17392/865-16
Shimizu, T., Kanazawa, T., Matsuoka, Y., Yoshida, T., Sakura, T., Shimizu, K., et al. (2024). General anesthesia with remimazolam during minimally invasive cardiac surgery for atrial septal defect: a pediatric case report. A A Pract. 18, e01735. doi:10.1213/xaa.0000000000001735
Su, M., Zhu, Y., Liu, S., Song, L., Qu, J., Zhang, Y., et al. (2023). Median effective dose (ED(50)) of esketamine combined with propofol for children to inhibit response of gastroscope insertion. BMC Anesthesiol. 23, 240. doi:10.1186/s12871-023-02204-y
Tamire, T., Garbessa, B., Gebeyehu, G., and Getachew, L. (2021). Tracheal Intubation-related adverse events in pediatrics anesthesia in Ethiopia. Paediatr. Anaesth. 31, 515–521. doi:10.1111/pan.14143
Trimmel, H., Helbok, R., Staudinger, T., Jaksch, W., Messerer, B., Schöchl, H., et al. (2018). S(+)-ketamine: current trends in emergency and intensive care medicine. Wien Klin. Wochenschr 130, 356–366. doi:10.1007/s00508-017-1299-3
Turner, E. H. (2019). Esketamine for treatment-resistant depression: seven concerns about efficacy and FDA approval. Lancet Psychiatry 6, 977–979. doi:10.1016/s2215-0366(19)30394-3
Viby-Mogensen, J., Engbaek, J., Eriksson, L. I., Gramstad, L., Jensen, E., Jensen, F. S., et al. (1996). Good clinical research practice (GCRP) in pharmacodynamic studies of neuromuscular blocking agents. Acta Anaesthesiol. Scand. 40, 59–74. doi:10.1111/j.1399-6576.1996.tb04389.x
Von Ungern-Sternberg, B. S., and Habre, W. (2007). Pediatric anesthesia - potential risks and their assessment: part I. Paediatr. Anaesth. 17, 206–215. doi:10.1111/j.1460-9592.2006.02097.x
Wang, J., Hu, W., Zhao, X., Ren, W., Huang, X., and Zhang, B. (2022). Sedative effect and safety of different doses of S-ketamine in combination with propofol during gastro-duodenoscopy in school-aged children: a prospective, randomized study. BMC Anesthesiol. 22, 346. doi:10.1186/s12871-022-01885-1
Wei, L., Deng, X., Sui, J., Wang, L., and Liu, J. (2015). Dexmedetomidine improves intubating conditions without muscle relaxants in children after induction with propofol and remifentanil. Anesth. Analg. 121, 785–790. doi:10.1213/ane.0000000000000812
Zanos, P., Moaddel, R., Morris, P. J., Riggs, L. M., Highland, J. N., Georgiou, P., et al. (2018). Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacol. Rev. 70, 621–660. doi:10.1124/pr.117.015198
Zhao, X., Zhou, Z., Li, Z., Hu, Z., and Yu, Y. (2024). Use of esketamine for tracheoscopic drug injection: a randomized controlled trial. Front. Med. (Lausanne) 11, 1479741. doi:10.3389/fmed.2024.1479741
Zheng, X., Huang, J., Wei, S., Tao, Y., Shen, Y., Wang, Y., et al. (2023). Efficacy and safety comparison of esketamine-propofol with nalbuphine-propofol for upper gastrointestinal endoscopy in children: a multi-center randomized controlled trial. Front. Pediatr. 11, 1126522. doi:10.3389/fped.2023.1126522
Zheng, X. S., Shen, Y., Yang, Y. Y., He, P., Wang, Y. T., Tao, Y. Y., et al. (2022). ED(50) and ED(95) of propofol combined with different doses of esketamine for children undergoing upper gastrointestinal endoscopy: a prospective dose-finding study using up-and-down sequential allocation method. J. Clin. Pharm. Ther. 47, 1002–1009. doi:10.1111/jcpt.13635
Zhong, Y., Jiang, M., Wang, Y., Su, T., Lv, Y., Fan, Z., et al. (2023). Evaluating efficacy and safety of sub-anesthetic dose esketamine as an adjuvant to propofol/remifentanil analgosedation and spontaneous respiration for children flexible fibreoptic bronchoscopy: a prospective, double-blinded, randomized, and placebo-controlled clinical trial. Front. Pharmacol. 14, 1184663. doi:10.3389/fphar.2023.1184663
Keywords: remimazolam, esketamine, effective dose, without muscle relaxants, dixon’s up-and-down method
Citation: Chen J, Mai Y, Cheng X, Sun H, Chen Z and Zhang Z (2025) Effective doses of remimazolam and esketamine combined with remifentanil for endotracheal intubation without muscle relaxants in pediatric patients. Front. Pharmacol. 16:1558966. doi: 10.3389/fphar.2025.1558966
Received: 11 January 2025; Accepted: 28 February 2025;
Published: 18 March 2025.
Edited by:
Angelo Montana, Marche Polytechnic University, ItalyReviewed by:
Catherine M. T. Sherwin, University of Western Australia, AustraliaCopyright © 2025 Chen, Mai, Cheng, Sun, Chen and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhongqi Zhang, anh6enExMUAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.