
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol. , 04 April 2025
Sec. Pharmacology of Infectious Diseases
Volume 16 - 2025 | https://doi.org/10.3389/fphar.2025.1546787
Objective: To explore the effectiveness and safety of azvudine and nirmatrelvir-ritonavir in a real-world setting.
Methods: This retrospective cohort study included adult patients with confirmed COVID-19 who received azvudine or nirmatrelvir-ritonavir treatment at Shanghai Changhai Hospital between 1 November 2022, and 30 March 2023. Data were collected from the hospital’s electronic medical record system using a standardized data extraction form. Propensity score matching (PSM) was used to control for potential confounding factors. The primary outcome was the incidence of composite disease progression, defined as the occurrence of death, ICU admission, invasive respiratory support, or high-flow oxygen therapy. Multivariable Cox regression analysis was performed to identify the factors independently associated with the composite progression outcomes.
Results: This study included 476 patients: 296 treated with azvudine and 180 treated with nirmatrelvir-ritonavir. After PSM, 139 patients were included in each group. There were no statistically significant differences between the two groups regarding the composite outcome (log-rank: P = 0.475; HR: 0.82, 95%CI: 0.46–1.43, P = 0.478), death (log-rank: P = 0.526; HR: 0.82, 95%CI: 0.44–1.52, P = 0.528), ICU admission (log-rank: P = 0.525; HR: 0.69, 95%CI: 0.22–2.18, P = 0.526), invasive ventilation (log-rank: P = 0.814; HR: 1.20, 95%CI: 0.27–5.39, P = 0.814), or oxygen use (log-rank: P = 0.370; HR: 1.44, 95%CI: 0.65–3.18, P = 0.372). The multivariable analysis showed that the antiviral drug (HR = 0.861, 95%CI: 0.486–1.524, P = 0.607) was not independently associated with the composite outcome. Only severe COVID-19 was independently associated with the composite outcome (HR = 3.322, 95%CI: 1.569–7.031, P = 0.002). The safety outcomes were similar between the two groups.
Conclusion: This real-world study demonstrates comparable efficacy and safety profiles between azvudine and nirmatrelvir-ritonavir in treating COVID-19 patients, regardless of disease severity or baseline characteristics. The findings support azvudine as a practical alternative for treatment selection, particularly in resource-constrained settings or for patients with contraindications to specific therapies. Clinical decisions should prioritize patient-specific needs, accessibility, and cost-effectiveness. Further large-scale prospective studies are needed to validate these observations and refine subgroup-specific treatment strategies.
COVID-19 is an infectious respiratory disease caused by SARS-CoV-2 and has imposed a huge burden on the socio-economic and medical systems around the globe (Koelle et al., 2022; Rothan and Byrareddy, 2020), with reported 775,251,779 cases and 7,043,660 deaths since early 2020 (World Health Organization, 2024). In February 2024, there were still 358 new severe cases and 22 deaths reported in China, indicating that COVID-19 continues to be a healthcare burden despite vaccination efforts (Lam et al., 2024; Schrimpf et al., 2023). Presently, the main strain prevalent in the world and in China is JN.1 (World Health Organization, 2024), which is highly infectious (Rubin, 2024). Currently, antiviral treatments such as nirmatrelvir/ritonavir (Paxlovid™) and azvudine (FNC) are the primary means of treating COVID-19 in China. Nirmatrelvir is a SARS-CoV-2 main protease (Mpro) inhibitor that prevents viral replication by blocking the cleavage of polyproteins, while ritonavir serves as a pharmacokinetic enhancer by inhibiting nirmatrelvir’s metabolism. Azvudine, a nucleoside analog, acts as an RNA-dependent RNA polymerase inhibitor that terminates viral RNA chain elongation, thereby inhibiting viral replication. Antiviral drugs can prevent disease progression or patient death by preventing virus replication, significantly reducing hospitalization and mortality rates among COVID-19 patients and shortening the time to turn negative (Zhang et al., 2021; Uraki et al., 2022; Li et al., 2022).
Indeed, a phase III study on nirmatrelvir-ritonavir showed that among COVID-19 patients over 65 years old, the rates of hospitalization and death due to COVID-19 were significantly lower among those receiving the antiviral treatment compared with those who did not (Arbel et al., 2022). A randomized controlled trial demonstrated that compared with traditional antiviral therapy, azvudine shortened the time for nucleic acid conversion to negative among patients with mild to moderate COVID-19 (Ren et al., 2020). The benefits are also seen in the real world, and azvudine significantly reduces the incidence of composite disease progression outcomes compared with supportive treatment among hospitalized COVID-19 patients (Sun et al., 2023). On the other hand, studies directly comparing nirmatrelvir-ritonavir vs. azvudine are rare, and additional research is necessary.
Unfortunately, the available studies comparing azvudine vs. nirmatrelvir-ritonavir report conflicting results. Indeed, some studies report favorable outcomes with (Gao et al., 2023; Zhao et al., 2023), while others favor azvudine (Han et al., 2024; Deng et al., 2023; Wei et al., 2023) or report no differences between the two drugs (Chen et al., 2024; Wang S. et al., 2024). Therefore, there is still no conclusive evidence regarding the real-world head-to-head comparison of the treatment effects of the two drugs, and further research is still needed to explore the efficacy of azvudine and nirmatrelvir-ritonavir in the treatment of COVID-19.
Therefore, the present study aimed to explore the therapeutic effects and safety of azvudine and nirmatrelvir-ritonavir in a real-world setting. The results could help define the value of the two drugs in managing COVID-19.
This retrospective cohort study was conducted at Shanghai Changhai Hospital, a tertiary care hospital in China. The target population included all adult COVID-19 patients who received antiviral therapy during the study period. A consecutive sampling method was employed to enroll eligible patients treated with azvudine or nirmatrelvir-ritonavir between 1 November 2022, and 30 March 2023. Propensity score matching (PSM) was applied to mitigate selection bias and potential confounding. Follow-up period was defined as the interval between hospital admission and discharge. All patients included in the final analysis had complete follow-up data throughout their entire hospitalization. The study was approved by the ethics review committee of Shanghai Changhai Hospital (#CHEC-2023-100). The requirement for individual informed consent was waived by the committee because of the retrospective nature of the study.
The inclusion criteria were 1) age ≥18 years, 2) diagnosed with COVID-19 infection according to the “Diagnosis and Treatment Protocol for COVID-19 Pneumonia” (i.e., RT-PCR Ct value < 35), and 3) received azvudine or nirmatrelvir-ritonavir. The exclusion criteria were 1) the antiviral drug was used for <2 days, 2) received other antiviral treatments, 3) concurrent use of azvudine and nirmatrelvir-ritonavir, 4) patients diagnosed with COVID-19 after hospitalization, or 5) missing key clinical information (Figure 1).
The antiviral treatment plan was selected by the patients and physicians after a comprehensive discussion based on the clinical features, physician’s experience, and according to the “Diagnosis and Treatment Protocol for COVID-19 Pneumonia”. The oral administration plan for nirmatrelvir-ritonavir was 300 mg nirmatrelvir +100 mg ritonavir (twice a day) for 5 days. The oral administration plan for azvudine was 5 mg once daily for 7 days or longer (not exceeding 14 days). The dosage of azvudine could be adjusted based on renal function if necessary. Other supportive treatments such as assisted ventilation, glucocorticoid therapy, non-steroidal anti-inflammatory therapy, nutritional support, anticoagulant therapy, etc., were applied routinely and according to the “Diagnosis and Treatment Protocol for COVID-19 Pneumonia”.
Patient demographic information and clinical data were collected from the medical record system of Shanghai Changhai Hospital, including age, sex, body mass index (BMI), vaccination status, smoking and drinking history, comorbid high-risk factors (including cardiovascular and cerebrovascular diseases [CVD, including hypertension], chronic lung diseases, type 2 diabetes mellitus [T2DM], chronic liver and kidney diseases, cancer, and immune deficiency), time from diagnosis to medication use, severity of COVID-19, concomitant medication use, history of comorbidities (including CVD, T2DM, liver disease, kidney disease, immune system diseases, schizophrenia, and cancer), and laboratory test results.
The primary outcome was the incidence of composite disease progression, defined as the occurrence of death, intensive care unit (ICU) admission, invasive respiratory support, or high-flow oxygen therapy, whichever occurred first. The secondary outcome measures included the incidence of individual composite outcome components, length of hospital stay, time to negative nucleic acid conversion (from medication start to negativity), changes in Ct values, and cumulative occurrence of abnormal laboratory indicators.
To mitigate potential confounding effects from clinical baseline variables (age, sex, vaccination status, smoking status, presence of high-risk factors, COVID-19 severity, and Diagnosis drug level) that may influence therapeutic outcomes, propensity score matching was employed. The caliper value was set at 0.1. The logistic regression was used to calculate the closest neighbor distance for 1:1 matching. The standardized mean difference (SMD) was used to assess the balance of the baseline variables between the two groups before and after propensity score matching, with an SMD of less than 0.1 indicating a good balance between the two groups.
In addition to propensity score matching, we employed inverse probability of treatment weighting (IPTW) as a sensitivity analysis to utilize the full sample and validate the robustness of our findings. The same set of covariates used in the propensity score model was included in the IPTW model. Balance after IPTW was evaluated using SMD, with values less than 0.1 indicating adequate balance.
The statistical analyses were performed using R 4.3.2 (The R Project for Statistical Computing, www.r-project.org). The continuous data were presented as means ± standard deviations or medians (ranges) and tested between groups using Student’s t-test. The categorical data were presented as n (%) and analyzed using the chi-squared test or Fisher’s exact test. Kaplan-Meier (KM) cumulative incidence curves were plotted for the incidence of the composite disease progression outcome or individual endpoints, and the log-rank test was used to compare the differences between groups. Hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) were calculated using univariate Cox proportional hazards regression models. The association between age and survival was examined using restricted cubic spline (RCS) regression. The optimal number of knots was determined based on the Akaike Information Criterion (AIC) minimization principle. To identify factors independently associated with composite progression outcomes, multivariate Cox regression analysis was conducted on the matched dataset. Additionally, subgroup analyses were performed to evaluate the impact of baseline variables on therapeutic efficacy, with results visualized through forest plots. Two-sided P-values <0.05 were considered statistically significant.
This retrospective cohort study enrolled 476 hospitalized COVID-19 patients, comprising 296 azvudine recipients and 180 nirmatrelvir-ritonavir recipients. Following 1:1 propensity score matching (PSM), 139 matched pairs were established with all standardized mean differences below 0.1, as detailed in Table 1. Subsequent inverse probability treatment weighting (IPTW) analysis demonstrated adequate balance across all baseline variables (standardized differences <0.1), although minor residual imbalances persisted in age (p = 0.036) and smoking status (p = 0.094). Comprehensive baseline characteristics for both analytic approaches are presented in Table 1.
As shown in Figure 2 and Table 2, there were no statistically significant differences between the azvudine and nirmatrelvir-ritonavir groups regarding the composite outcome (log-rank: P = 0.650; HR: 0.87, 95%CI: 0.47–1.60, P = 0.652), death (log-rank: P = 0.779; HR: 0.91, 95%CI: 0.47–1.77, P = 0.780), ICU admission (log-rank: P = 0.903; HR: 1.08, 95%CI: 0.31–3.75, P = 0.904), invasive ventilation (log-rank: P = 0.523; HR: 0.58, 95%CI: 0.11–3.20, P = 0.534), or oxygen use (log-rank: P = 0.265; HR: 0.63, 95%CI: 0.28–1.44, P = 0.272).
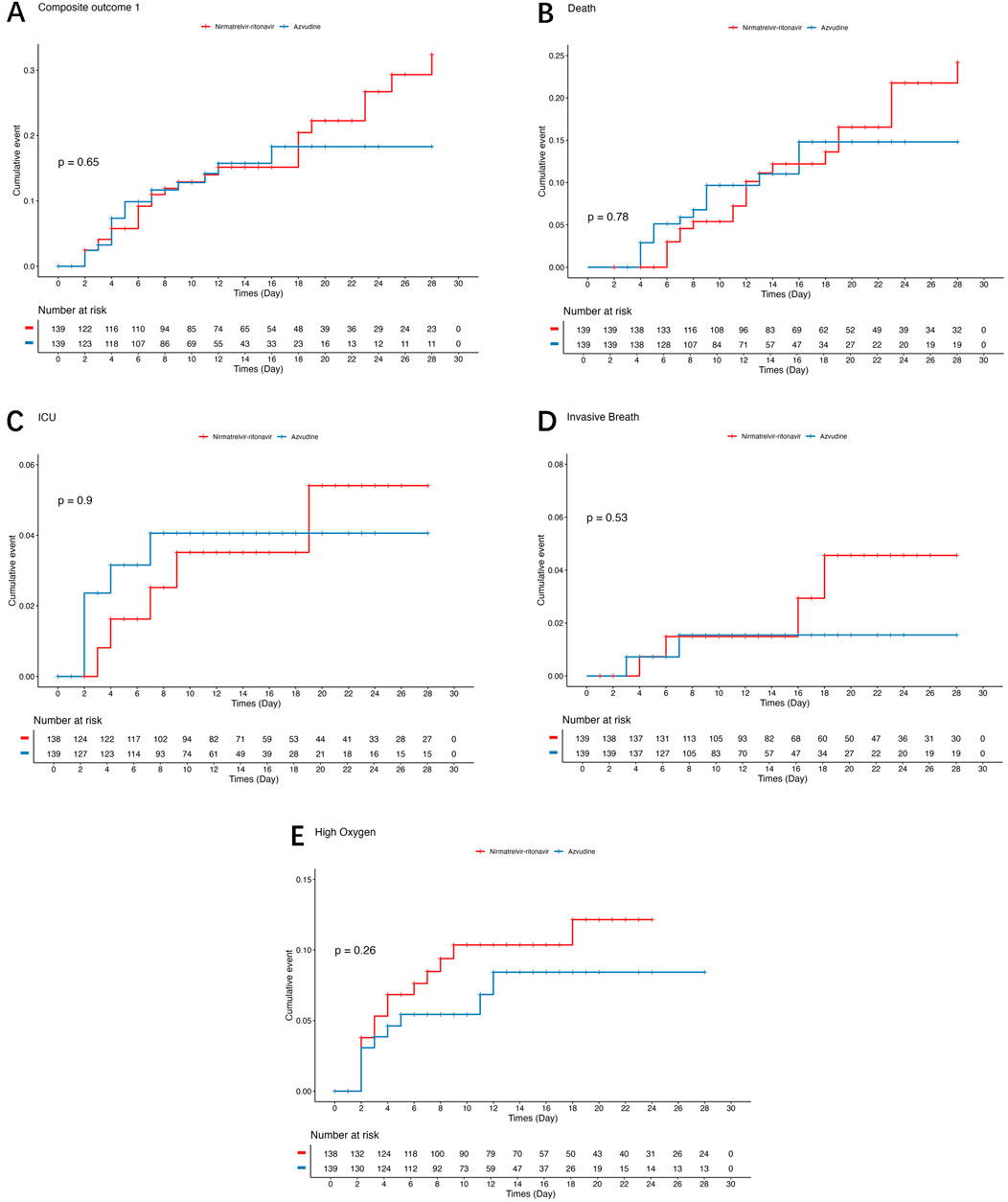
Figure 2. Kaplan-Meier curves for composite disease progression and individual outcomes. (A) Composite outcome of disease progression. (B) Death. (C) Invasive ventilation. (D) Intensive care unit admission. (E) Oxygen use.
The relationship between age and survival outcomes was assessed using RCS regression. Based on the AIC minimization principle, models with 4 knots demonstrated optimal fit. Both unadjusted and covariate-adjusted RCS analyses revealed a predominantly linear association between age and survival (P for non-linearity = 0.319 and 0.083, respectively), as illustrated in Supplementary Figures S2. All covariates were modeled as follows: age as a continuous variable (in years), sex as a categorical variable (male/female), vaccination status as a categorical variable (vaccinated/non-vaccinated/unknown), smoking status as a categorical variable (yes/no/unknown), high-risk factors as a binary variable (yes/no), COVID-19 severity as a categorical variable (mild/moderate/severe), and diagnosis drug level as a categorical variable (<5, >5, unknown).
The multivariable Cox analysis showed that the antiviral drug (HR = 0.79, 95%CI: 0.42–1.49, P = 0.465) was not independently associated with the composite outcome (Table 3).
The subgroup analysis showed no statistically significant differences across all subgroups based on sex, smoking, vaccination, drug use at diagnosis, COVID-19 type (Figure 3).
As shown in Figure 4, the patterns of changes in the Ct values were similar between the two groups, with a little dip in the Ct value of the O gene after 2–3 days of treatment.
The cumulative incidence of laboratory abnormalities within 60 days was similar between the two groups (azvudine vs. nirmatrelvir-ritonavir) for alkaline phosphatase (ALP) (21.6% vs. 25.9%) (Figure 5A), alanine aminotransferase (ALT) (36.0% vs. 43.9%) (Figure 5B), aspartate aminotransferase (AST) (33.8% vs. 35.3%) (Figure 5C), blood urea nitrogen (BUN) (48.9% vs. 52.5%) (Figure 5D), creatinine (CRE) (32.4% vs. 33.1%) (Figure 5E), lactate dehydrogenase (LDH) (56.8% vs. 55.4%) (Figure 5F), and total bilirubin (TBIL) (12.9% vs. 13.7%) (Figure 5G). No other adverse events were reported in either group.
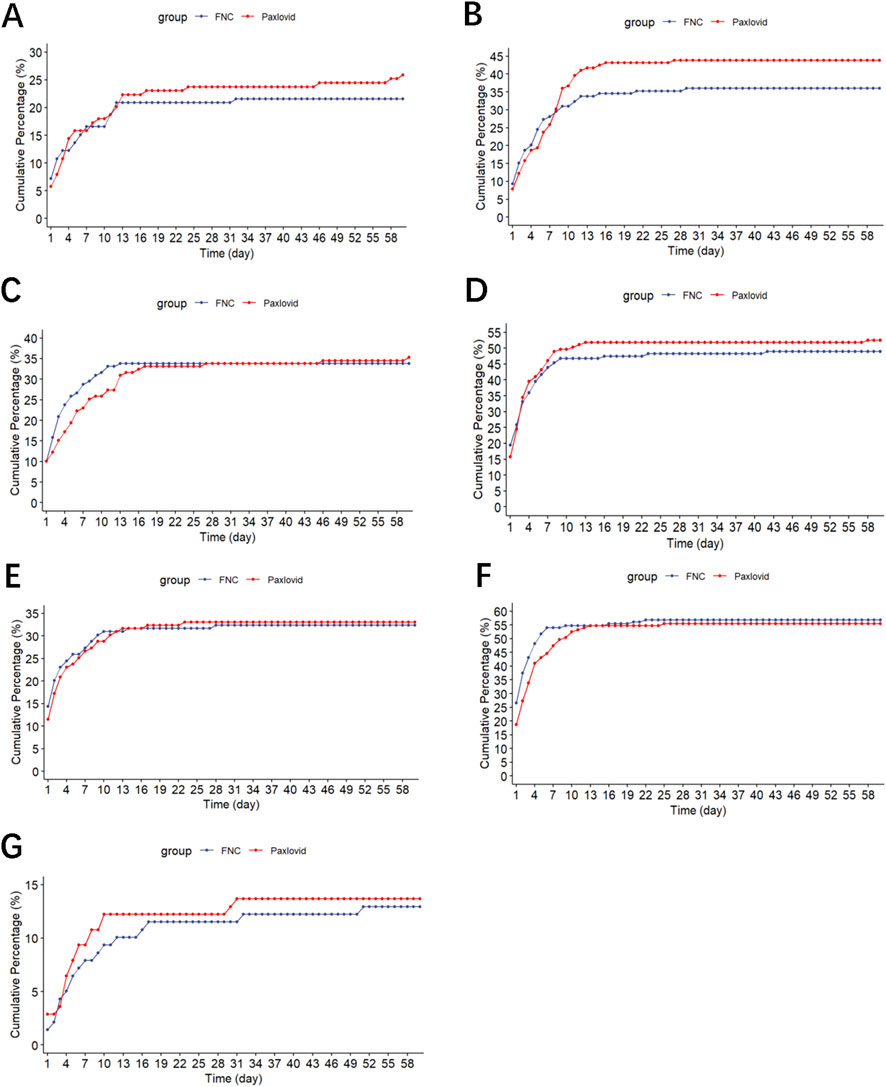
Figure 5. Cumulative Incidence of abnormal laboratory test values. (A) Alkaline phosphatase (ALP). (B) Alanine transaminase. (C) Aspartate transaminase (D) Blood urea nitrogen. (E) Creatinine. (F) Lactate dehydrogenase. (G) Total bilirubin.
This study explored the therapeutic effects and safety of azvudine and nirmatrelvir-ritonavir in a real-world setting. The results suggest that azvudine exhibits similar real-world therapeutic effects and safety compared with nirmatrelvir-ritonavir. There were no differences between the two drugs in terms of the composite outcome that included death, ICU admission, invasive ventilation, and oxygen use or the individual outcomes.
Randomized controlled trials demonstrated the efficacy and safety of azvudine (de Souza et al., 2023; Wang Y. et al., 2024) or nirmatrelvir-ritonavir (Liu et al., 2023; Amani and Amani, 2023) for managing COVID-19. Still, clinical trials are performed in highly selected patients, limiting the generalizability of the conclusions. Real-world studies are complementary to clinical trials (Kim et al., 2018). Furthermore, no clinical trials directly compared azvudine vs. nirmatrelvir-ritonavir. Real-world studies can provide some evidence pending a clinical trial.
Gao et al. (2023) reported a retrospective study that showed that compared with patients receiving azvudine, those receiving nirmatrelvir-ritonavir showed faster virus suppression and earlier nucleic acid negative conversion during the initial hospitalization stage (Gao et al., 2023). It is similar to another recent retrospective study that showed that in terms of conversion time, azvudine and nirmatrelvir-ritonavir had similar efficacy, while nirmatrelvir-ritonavir was able to suppress the virus faster in patients with mild COVID-19 (Zhao et al., 2023). Nirmatrelvir-ritonavir might have an advantage in reducing early mortality compared with azvudine (Han et al., 2024). On the other hand, other retrospective studies found that compared with nirmatrelvir-ritonavir, azvudine improved the composite disease progression outcomes among hospitalized COVID-19 patients (Deng et al., 2023; Wei et al., 2023). Of note, Deng et al. (Deng et al., 2023) observed a significant difference for the composite outcome (P = 0.026) but not for the individual outcomes (all P > 0.05). Chen et al. (Chen et al., 2024) reported that azvudine was comparable to nirmatrelvir/ritonavir and molnupiravir in adult patients with mild-to-moderate COVID-19 in terms of nucleic acid negative conversion, hospital stay, and adverse events. Similar results were reported in older adults with severe COVID-19 (Wang S. et al., 2024). The study found no differences between the two drugs in the composite outcome of disease progression or the individual outcomes, which contradicts previous studies but agrees with others (Chen et al., 2024; Wang S. et al., 2024). Therefore, further exploration is needed to compare the head-to-head efficacy of these two drugs. Based on the available evidence, one or the other can be used to manage patients with COVID-19. The choice should be made based on drug availability and the financial capacities of the patients.
In the present study, the choice of antiviral was not independently associated with the composite outcome. Only COVID-19 severity was independently associated with the composite outcome, which is unsurprising considering that it is the most important prognostic indicator in patients with COVID-19 (Wiersinga et al., 2020; Tan et al., 2021). Furthermore, the performance of the two drugs was similar across the various subgroups. Azvudine appears to be safe in patients on hemodialysis, suggesting that it can be used safely in patients with chronic kidney disease (Shang et al., 2023). On the other hand, nirmatrelvir-ritonavir is contraindicated in patients with severe or end-stage kidney disease, although the literature suggests it might be safe (Cai et al., 2023). Additional studies are necessary to determine the categories of patients that are the best fit for a specific treatment.
In this study, abnormalities in ALP (21.6% vs. 25.9%), ALT (36.0% vs. 43.9%), AST (33.8% vs. 35.3%), BUN (48.9% vs. 52.5%), CRE (32.4% vs. 33.1%), LDH (56.8% vs. 55.4%), and TBIL (12.9% vs. 13.7%) were observed. Of note, it was impossible to determine whether a given abnormality in a given patient was due to the antiviral drug, COVID-19, or comorbidities. Nevertheless, those abnormalities were observed in previous studies of azvudine and nirmatrelvir-ritonavir (Zhang et al., 2021; Ren et al., 2020; Sun et al., 2023; Gao et al., 2023; Zhao et al., 2023; Han et al., 2024; Deng et al., 2023; Wei et al., 2023; Chen et al., 2024; Wang et al., 2024a; de Souza et al., 2023; Wang Y. et al., 2024; Liu et al., 2023; Amani and Amani, 2023; Shang et al., 2023; Cai et al., 2023), and no new safety signals were observed.
A rebound phenomenon has been described for the Ct value after starting nirmatrelvir-ritonavir or molnupiravir (Parums, 2022; Anderson et al., 2022; Charness et al., 2022; Rubin, 2022), while it was suggested that such a rebound was not observed with azvudine (da Silva et al., 2023). Although the present study showed a modest dip in the O-gene Ct values at the beginning of treatment with the two drugs, no evident rebound was observed.
A strength of this study was the inclusion of all patients with COVID-19 who were hospitalized at the study hospital during the study period. In China, hospitalization was mandatory during the COVID-19 pandemic whenever an RT-PCR test was positive, resulting in the inclusion of all patients. Nevertheless, this study had limitations. The patients were from a single hospital, resulting in a small sample size. Subgroup analyses were conducted post-matching without subgroup-specific adjustments, and the results should be interpreted cautiously as exploratory due to limited sample sizes within certain subgroups, which may reduce statistical power. We did conduct an IPTW analysis, and the conclusions were consistent with our previous findings. Our empirical assessments of non-linearity confirmed that modeling age as a continuous linear term in our Cox proportional hazards models and propensity score calculations was appropriate for our dataset. This approach allowed us to maximize statistical efficiency while accurately representing the relationship between age and the outcomes of interest. In addition, although the “Diagnosis and Treatment Protocol for COVID-19 Pneumonia” was applied to all patients, there is a risk of bias due to local practice, limiting generalizability. The study was retrospective, limiting the data to those available in the patient charts. Larger prospective studies and randomized controlled trials are needed in the future.
In conclusion, azvudine exhibits similar real-world therapeutic effects and safety compared with nirmatrelvir-ritonavir in patients with COVID-19, irrespective of patient characteristics or COVID-19 severity. These findings have important clinical implications for treatment selection, particularly in resource-limited settings or for patients with contraindications to either medication. Our results suggest that clinicians can consider both drugs as viable treatment options, with the choice guided by patient-specific factors, drug availability, and cost considerations. Future prospective studies with larger sample sizes are warranted to confirm these findings and potentially identify specific patient subgroups that might benefit more from one treatment over the other.
The datasets presented in this article are not readily available because It’s patient privacy. We can’t provide any information. Requests to access the datasets should be directed to Y2hlbmppbmd4aWFfMjAxMkAxNjMuY29t.
The studies involving humans were approved by Ethics Committee of the First Hospital Affiliated to Naval Medical University. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and institutional requirements.
JC: Writing – original draft. ZL: Writing – original draft. RL: Methodology, Writing – review and editing. CS: Data curation, Writing – review and editing. YY: Formal Analysis, Writing – review and editing. ZW: Conceptualization, Project administration, Validation, Writing – review and editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors extend their gratitude to the custodians of the data sets utilized in this study for their invaluable assistance and support in the collation of the data.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2025.1546787/full#supplementary-material
Amani, B., and Amani, B. (2023). Efficacy and safety of nirmatrelvir/ritonavir (Paxlovid) for COVID-19: a rapid review and meta-analysis. J. Med. Virol. 95 (2), e28441. doi:10.1002/jmv.28441
Anderson, A. S., Caubel, P., Rusnak, J. M., and Investigators, E.-H. T. (2022). Nirmatrelvir-ritonavir and viral load rebound in covid-19. N. Engl. J. Med. 387 (11), 1047–1049. doi:10.1056/NEJMc2205944
Arbel, R., Wolff Sagy, Y., Hoshen, M., Battat, E., Lavie, G., Sergienko, R., et al. (2022). Nirmatrelvir use and severe covid-19 outcomes during the omicron surge. N. Engl. J. Med. 387 (9), 790–798. doi:10.1056/NEJMoa2204919
Cai, H., Yan, J., Liu, S., Li, P., Ding, L., Zhan, Y., et al. (2023). Paxlovid for hospitalized COVID-19 patients with chronic kidney disease. Antivir. Res. 216, 105659. doi:10.1016/j.antiviral.2023.105659
Charness, M. E., Gupta, K., Stack, G., Strymish, J., Adams, E., Lindy, D. C., et al. (2022). Rebound of SARS-CoV-2 infection after nirmatrelvir-ritonavir treatment. N. Engl. J. Med. 387 (11), 1045–1047. doi:10.1056/NEJMc2206449
Chen, M. P., Jiang, D. X., Rang, J. X., Zhuo, H. B., and Zhou, Z. G. (2024). Comparison of azvudine, molnupiravir, and nirmatrelvir/ritonavir in adult patients with mild-to-moderate COVID-19: a retrospective cohort study. Sci. Rep. 14 (1), 3318. doi:10.1038/s41598-024-53862-y
da Silva, R. M., Gebe Abreu Cabral, P., de Souza, S. B., Arruda, R. F., Cabral, S. P. F., de Assis, A., et al. (2023). Serial viral load analysis by DDPCR to evaluate FNC efficacy and safety in the treatment of mild cases of COVID-19. Front. Med. (Lausanne) 10, 1143485. doi:10.3389/fmed.2023.1143485
Deng, G., Li, D., Sun, Y., Jin, L., Zhou, Q., Xiao, C., et al. (2023). Real-world effectiveness of Azvudine versus nirmatrelvir-ritonavir in hospitalized patients with COVID-19: a retrospective cohort study. J. Med. Virol. 95 (4), e28756. doi:10.1002/jmv.28756
de Souza, S. B., Cabral, P. G. A., da Silva, R. M., Arruda, R. F., Cabral, S. P. F., de Assis, A., et al. (2023). Phase III, randomized, double-blind, placebo-controlled clinical study: a study on the safety and clinical efficacy of AZVUDINE in moderate COVID-19 patients. Front. Med. (Lausanne) 10, 1215916. doi:10.3389/fmed.2023.1215916
Gao, Y., Luo, Z., Ren, S., Duan, Z., Han, Y., Liu, H., et al. (2023). Antiviral effect of azvudine and nirmatrelvir-ritonavir among hospitalized patients with COVID-19. J. Infect. 86 (6), e158–e160. doi:10.1016/j.jinf.2023.03.023
Han, X., Gao, D., Li, C., Yuan, X., Cui, J., Zhao, W., et al. (2024). Real-world effectiveness of nirmatrelvir-ritonavir versus azvudine in hospitalized patients with COVID-19 during the omicron wave in Beijing: a multicenter retrospective cohort study. BMC Infect. Dis. 24 (1), 57. doi:10.1186/s12879-023-08965-8
Kim, H. S., Lee, S., and Kim, J. H. (2018). Real-world evidence versus randomized controlled trial: clinical research based on electronic medical records. J. Korean Med. Sci. 33 (34), e213. doi:10.3346/jkms.2018.33.e213
Koelle, K., Martin, M. A., Antia, R., Lopman, B., and Dean, N. E. (2022). The changing epidemiology of SARS-CoV-2. Science 375 (6585), 1116–1121. doi:10.1126/science.abm4915
Lam, I. C. H., Zhang, R., Man, K. K. C., Wong, C. K. H., Chui, C. S. L., Lai, F. T. T., et al. (2024). Persistence in risk and effect of COVID-19 vaccination on long-term health consequences after SARS-CoV-2 infection. Nat. Commun. 15 (1), 1716. doi:10.1038/s41467-024-45953-1
Li, P., Wang, Y., Lavrijsen, M., Lamers, M. M., de Vries, A. C., Rottier, R. J., et al. (2022). SARS-CoV-2 Omicron variant is highly sensitive to molnupiravir, nirmatrelvir, and the combination. Cell Res. 32 (3), 322–324. doi:10.1038/s41422-022-00618-w
Liu, J., Pan, X., Zhang, S., Li, M., Ma, K., Fan, C., et al. (2023). Efficacy and safety of Paxlovid in severe adult patients with SARS-Cov-2 infection: a multicenter randomized controlled study. Lancet Reg. Health West Pac 33, 100694. doi:10.1016/j.lanwpc.2023.100694
Parums, D. V. (2022). Editorial: rebound COVID-19 and cessation of antiviral treatment for SARS-CoV-2 with paxlovid and molnupiravir. Med. Sci. Monit. 28, e938532. doi:10.12659/MSM.938532
Ren, Z., Luo, H., Yu, Z., Song, J., Liang, L., Wang, L., et al. (2020). A randomized, open-label, controlled clinical trial of azvudine tablets in the treatment of mild and common COVID-19, a pilot study. Adv. Sci. (Weinh) 7 (19), e2001435. doi:10.1002/advs.202001435
Rothan, H. A., and Byrareddy, S. N. (2020). The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 109, 102433. doi:10.1016/j.jaut.2020.102433
Rubin, R. (2022). From positive to negative to positive again-the mystery of why COVID-19 rebounds in some patients who take paxlovid. JAMA 327 (24), 2380–2382. doi:10.1001/jama.2022.9925
Rubin, R. (2024). As COVID-19 cases surge, here’s what to know about JN.1, the latest SARS-CoV-2 variant of interest. JAMA 331 (5), 382–383. doi:10.1001/jama.2023.27841
Schrimpf, A., Bleckwenn, M., and Braesigk, A. (2023). COVID-19 continues to burden general practitioners: impact on workload, provision of care, and intention to leave. Healthc. (Basel) 11 (3), 320. doi:10.3390/healthcare11030320
Shang, S., Fu, B., Geng, Y., Zhang, J., Zhang, D., Xiao, F., et al. (2023). Azvudine therapy of common COVID-19 in hemodialysis patients. J. Med. Virol. 95 (8), e29007. doi:10.1002/jmv.29007
Sun, Y., Jin, L., Dian, Y., Shen, M., Zeng, F., Chen, X., et al. (2023). Oral Azvudine for hospitalised patients with COVID-19 and pre-existing conditions: a retrospective cohort study. EClinicalMedicine 59, 101981. doi:10.1016/j.eclinm.2023.101981
Tan, E., Song, J., Deane, A. M., and Plummer, M. P. (2021). Global impact of coronavirus disease 2019 infection requiring admission to the ICU: a systematic review and meta-analysis. Chest 159 (2), 524–536. doi:10.1016/j.chest.2020.10.014
Uraki, R., Kiso, M., Iida, S., Imai, M., Takashita, E., Kuroda, M., et al. (2022). Characterization and antiviral susceptibility of SARS-CoV-2 Omicron BA.2. Nature 607 (7917), 119–127. doi:10.1038/s41586-022-04856-1
Wang, S., Sun, J., Zhang, X., Li, M., Qin, B., Liu, M., et al. (2024a). Antiviral effectiveness and survival correlation of azvudine and nirmatrelvir/ritonavir in elderly severe patients with COVID-19: a retrospective real-world study. EClinicalMedicine 69, 102468. doi:10.1016/j.eclinm.2024.102468
Wang, Y., Xie, H., Wang, L., Fan, J., Zhang, Y., Pan, S., et al. (2024b). Effectiveness of azvudine in reducing mortality of COVID-19 patients: a systematic review and meta-analysis. Virol. J. 21 (1), 46. doi:10.1186/s12985-024-02316-y
Wei, A. H., Zeng, L., Wang, L., Gui, L., Zhang, W. T., Gong, X. P., et al. (2023). Head-to-head comparison of azvudine and nirmatrelvir/ritonavir for the hospitalized patients with COVID-19: a real-world retrospective cohort study with propensity score matching. Front. Pharmacol. 14, 1274294. doi:10.3389/fphar.2023.1274294
Wiersinga, W. J., Rhodes, A., Cheng, A. C., Peacock, S. J., and Prescott, H. C. (2020). Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA 324 (8), 782–793. doi:10.1001/jama.2020.12839
World Health Organization (2024). COVID-19 epidemiological update. Available online at: https://www.who.int/publications/m/item/covid-19-epidemiological-update-edition-1662024. (Accessed December 12, 2024).
Zhang, J. L., Li, Y. H., Wang, L. L., Liu, H. Q., Lu, S. Y., Liu, Y., et al. (2021). Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients. Signal Transduct. Target Ther. 6 (1), 414. doi:10.1038/s41392-021-00835-6
Keywords: COVID-19, azvudine, nirmatrelvir-ritonavir, real-world study, composite outcome
Citation: Chen J, Liu Z, Liu R, Su C, Yang Y and Wang Z (2025) Composite outcome of oral azvudine vs. nirmatrelvir-ritonavir in COVID-19 patients: a retrospective cohort study. Front. Pharmacol. 16:1546787. doi: 10.3389/fphar.2025.1546787
Received: 22 January 2025; Accepted: 25 March 2025;
Published: 04 April 2025.
Edited by:
Eleonore Fröhlich, Medical University of Graz, AustriaReviewed by:
Himmatul Ulya, Muria Kudus University, IndonesiaCopyright © 2025 Chen, Liu, Liu, Su, Yang and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhuo Wang, d3p0Z3l4MjIzQDE2My5jb20=; Yunyun Yang, MTMyNjIyNDY5NjVAMTYzLmNvbQ==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.