- 1Institute of Innovative Traditional Chinese Medications, Hunan Academy of Chinese Medicine, Changsha, China
- 2TCM and Ethnomedicine Innovation and Development International Laboratory, Innovative Material Medical Research Institute, School of Pharmacy, Hunan University of Chinese Medicine, Changsha, China
- 3Department of Chemistry, School of Sciences, IFTM University, Moradabad, Uttar Pradesh, India
- 4Forestry Bureau of Hengnan County, Hengyang, China
Background: The Ehretia genus, comprising 66 species in the Boraginaceae family, has a history of ethnomedicinal use for various ailments. This review focuses on the botany, traditional uses, phytochemistry, pharmacology, toxicology, clinical studies, cultivation, and commercial potential of the Ehretia genus, with the goal of enhancing current research and applications.
Methods: Literatures related to Ehretia species were compiled using keywords such as “Ehretia,” “traditional use,” “chemical constituents,” and “bioactivity” from scientific databases, including “China Knowledge Resource Integrated Databases (CNKI),” “Flora of China,” “Google Scholar,” “Hunan Library,” “Plants of the World Online,” and “Web of Science” and so on.
Results: From 1980 to August 2024, only 101 compounds have been identified within this genus, primarily consisting of flavonoids, phenylpropanoids, phenolics, benzoquinones, triterpenoids, and fatty acids, with phenylpropanoids being the main components. Extracts and compounds from Ehretia species exhibited various bioactivities, including antioxidant, hepatoprotective, analgesic, anti-inflammatory, antibacterial, and anticancer effects, etc.
Conclusion: Research on the Ehretia genus is limited, with many species remaining underexamined in terms of phytochemistry and pharmacology. Few active compounds have been isolated and assessed for biological activities, and there is a lack of investigation into their mechanisms of action. Despite its documented uses, Ehretia species remains less explored scientifically than other Boraginaceae genera, presenting significant research opportunities. Further comprehensive studies are necessary to deepen our understanding of this diverse genus and validate its therapeutic potential.
1 Introduction
The Ehretia genus, comprising 66 accepted species, belongs to the Boraginaceae family and is primarily distributed across tropical Asia, Africa, Australia, North and South America (http://www.plantsoftheworldonline.org). These plants exhibit wide adaptability and strong stress resistance, thriving in warm, moist environments with deep, fertile, well-drained soil (pH 5.5–6.5) and an annual average temperature above 10°C. Ehretia species grow in various habitats, from tropical forests to dry regions, showcasing resilience compared to less adaptive genera like Myosotis (Mandal and Joshi, 2014; Retief and Van Wyk, 2001; Shukla and Kaur, 2018). The genus includes only tree or shrub species (http://www.iplant.cn/). Various parts of the Ehretia plants-roots, leaves, flowers, barks, fruits, and heartwoods-are used in herbal medicine and consumed as food (Li et al., 2010a; Lim et al., 2023; Hadjichambis et al., 2008; Zara et al., 2012). In folklore, Ehretia species were employed as ethnomedicine to treat a variety of cough, inflammation, swellings and syphilis, et al. (Iqbal et al., 2005). Notably, some species such as Ehretia corylifolia, E. longiflora, E. macrophylla, E. thyrsiflora, and E. tsangii are recognized for their antipyretic and detoxifying properties in traditional Chinese medicine (Zeng and Zeng, 1994). The bitter tea known as “Kudingcha” in Chinese has historically been made locally using the leaves of E. thyrsiflora (He and Liu, 1992). “Tsaang gubat” also known as E. microphylla, is presently included in the Philippine National Formulary and is recognised by the Department of Health as one of the top therapeutic plants recommended in the nation (Legaspi and Bagaoisan, 2020).
According to literatures, more than 100 compounds have been identified from Ehretia genus. These compounds including flavonoids, phenylpropanoids, simple phenolics, benzoquinones, triterpenoids, fatty acids and so on (Chien et al., 2012; Le et al., 2021; Li et al., 2009; Yamamura et al., 1995; Yoshikawa et al., 1995). The extracts and compounds of Ehretia species exhibited a range of bioactivities, such as antioxidant, anti-diabetic, hepatoprotective, analgesic, anti-inflammatory, antibacterial, anticancer, antihemolytic, anti-arthritic, wound healing effects, etc (Harne et al., 2021; Jan et al., 2023; Kaur et al., 2019; Kaur et al., 2022; Memon et al., 2022; Panja et al., 2020; Rangnathrao and Shanmugasundaram, 2019; Waheed et al., 2019).
To date, only two review articles focus on the genus Ehretia, with the most recent one occurring 6 years ago (Li et al., 2010a; Shukla and Kaur, 2018). This review aims to provide a comprehensive overview of the botany, ethnomedicinal values, phytochemistry, pharmacology, toxicology, clinical studies, cultivation, and commercial value of the genus Ehretia (Mitsi and Echeverría, 2024). Due to the genus’s diversity, only species with proven medicinal characteristics will be highlighted. Additionally, this review addresses the limitations of current research and suggests directions for future studies, offering insights to guide upcoming research on Ehretia species.
2 Ethnomedicinal uses of genus Ehretia
The Flora Reipubae Popularis Sinicae categorizes all plants in the genus Ehretia as either trees or shrubs (Table 1) (Editorial Committee of the Flora of China, 1985). This genus is predominantly found in southern Asia, China, Africa, North and South America (Figure 1). In China, twelve species and one variant are identified, primarily in provinces south of the Yangtze River, with additional occurrences in southern Gansu, Henan, Shaanxi, and Qinghai. It is only known that 12 species of the genus are utilized in traditional medicine (Table 2). Rich folklore medical applications for Ehretia plants include treatments for syphilis, eczema, stomach disorders, cough, diarrhea, and chest pains (Arenas et al., 2013; Mncwangi et al., 2012; Torane et al., 2011; Waheed et al., 2019). The Ehretia genus also contributes significantly to forest ecosystems as a source of food for birds and insects due to its fleshy drupes and nectar-rich flowers. Unlike invasive or fast-spreading members of Boraginaceae (e.g., Heliotropium indicum), Ehretia species are generally regarded as ecologically harmonious, adding to their conservation importance.
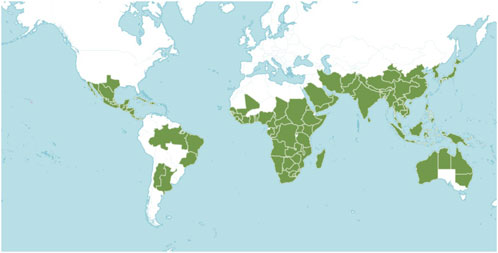
Figure 1. Geographical distribution of Ehretia species plants in the world (Cite from http://www.plantsoftheworldonline.org).
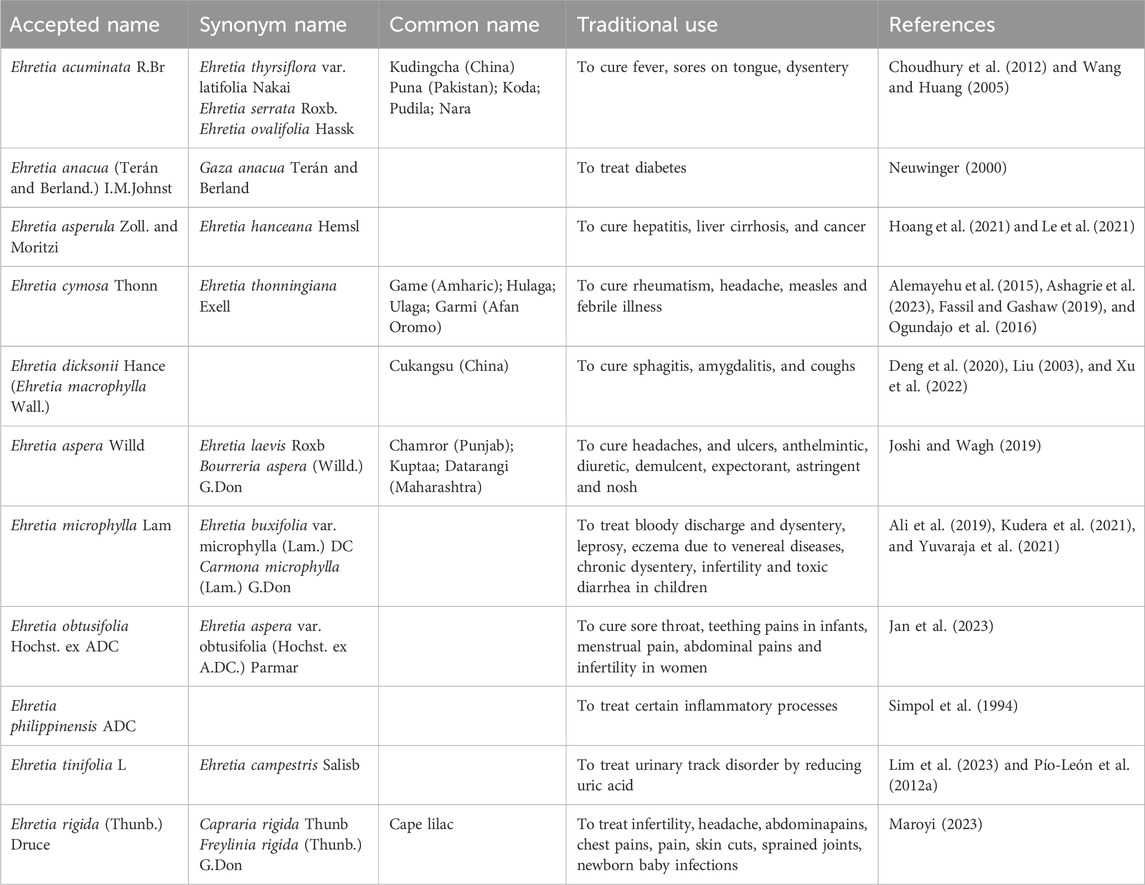
Table 2. Record of the traditional uses of some Ehretia species (http://www.plantsoftheworldonline.org).
Among the earliest species in the Ehretia genus is E. acuminata, which has been traditionally used to treat fever, tongue sores, diarrhea, and other ailments in China and India (Choudhury et al., 2012; Shukla et al., 2019b; Wang and Huang, 2005). The leaves of E. thyrsiflora (syn. E. acuminata) serve as one of the sources for Kudingcha, a bitter tea popular in southern China (Li et al., 2009). In Nigeria, folk medicine utilizes the leaves of E. anacua to cure diabetes (Abimbola et al., 2021). E. asperula, endemic to northern Vietnam, has a history of use in treating cancer, liver cirrhosis, and hepatitis (Hoang et al., 2021; Le et al., 2021). The small tree E. cymose is predominantly found in the secondary jungles and savannas of Africa. Its fruits are black, ovoid to spherical, measuring 2–6 mm in length, and the leaves are oval in shape (Sarkodie et al., 2015). In several parts of Ethiopia, the leaves of E. cymose are traditionally employed to treat feverish illnesses, rheumatism, headaches, and measles (Figure 2) (Alemayehu et al., 2015; Ashagrie et al., 2023; Fassil and Gashaw, 2019; Ogundajo et al., 2016).
Folk medicine for alleviating swelling often utilizes the bark of E. dicksonii (Xu et al., 2022). For respiratory conditions such as sphagitis, amygdalitis, and coughs, the fruit of E. macrophylla-identified as the same species as E. dicksonii-along with herbal tea, serves as traditional remedies (Deng et al., 2020; Liu, 2003). E. laevis, a deciduous shrub native to tropical regions of Asia and Australia, is recognized for its significant therapeutic value in traditional medicine (Velappan and Thangaraj, 2014). In Uttarakhand and other sub-Himalayan regions of India, the herbal medicine is used to treat jaundice (Rangnathrao and Shanmugasundaram, 2019). Its utilization can be traced back to ancient medical systems, including Ayurveda and Siddha (Sharma et al., 2021). The plant uses its inner bark for sustenance and nutrition. This plant’s leaves can be used to cure headaches, skin conditions, and ulcers. This plant’s fruit is frequently used to treat lung and spleen ailments, urinary tract issues, astringent, deworming, diuretic, analgesic, and expectorant conditions (Joshi and Wagh, 2019).
Carmona retusa (syn. E. microphylla) is a striking shrub reaching heights of 1.5–4.0 m, characterized by its glossy, coarse, dark green leaves. In addition to medicinal uses, certain species like E. microphylla are also cultivated for ornamental purposes and bonsai (Aarthi et al., 2014; Mageswari and Karpagam, 2015; Mageswari et al., 2012). E. microphylla is particularly significant in the Philippines and India, especially within the Ayurvedic and Siddha medical traditions (Chopra et al., 1956; Selvanayagam et al., 1996). A leaf infusion serves as a tea alternative and cough suppressant for conditions involving bloody discharge and diarrhea (Ali et al., 2019; Yuvaraja et al., 2021). In Siddha Materia Medica, it is used to treat leprosy, sexually transmitted infection-related dermatitis, chronic dysentery, infertility, and toxic diarrhea in children (Kudera et al., 2021). Additionally, the root acts as an antidote for vegetable poisoning and treats syphilis and cachexia (Aarthi et al., 2014).
In Zimbabwe, various compounds from E. obtusefolia are utilized to cure infertility in women, menstrual cramps, stomach spasms, sore throats, and children’s teething pain (Jan et al., 2023). E. philippinensis, endemic to low- and medium-altitude forests in the Philippines, is known for its stem bark and leaves that aid in treating certain inflammatory processes (Simpol et al., 1994). The small deciduous tree or shrub E. rigida, also known as the “puzzle bush,” has its stems used medicinally by the Zulu people, and its fruit is edible (Steyn, 1998).
E. tinifolia, an evergreen tree native to Mexico and the United States, can grow up to 25 m. Its small, round, fragrant yellow fruit is commonly consumed, while the bark aids wound recovery, and the flowers and leaves can alleviate symptoms of bloody vomiting (Hadjichambis et al., 2008; Pío-León et al., 2012b; Lim et al., 2023; Pío-León et al., 2012a). In conclusion, various Ehretia species are primarily employed for diverse ailments in Asian countries, indicating a substantial potential for bioactive compounds. Consequently, these species warrant further biological and chemical investigation.
3 Phytochemistry
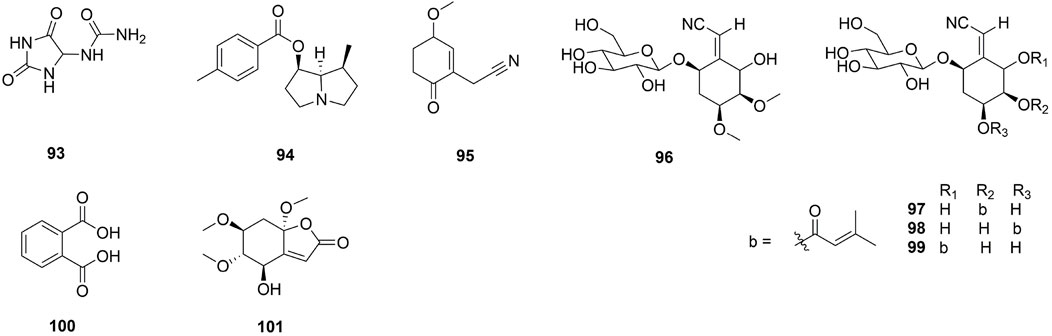
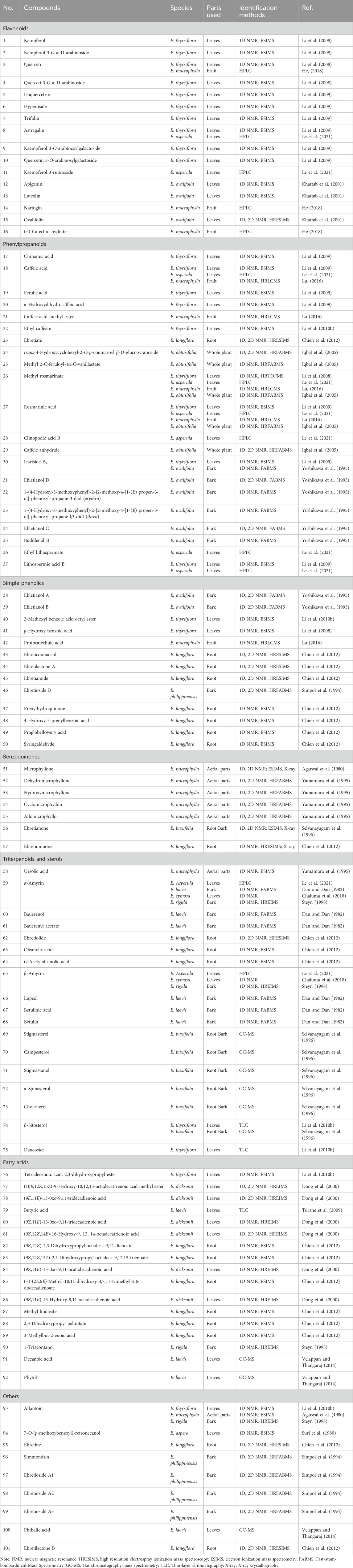
One hundred and one compounds were identified from Ehretia genus, including flavonoids, phenypropanoids, simple phenolics, benzoquinones, triterpenoids, sterols, fatty acids and other compounds (Figures 3–9). These compounds have been identified in a variety of Ehretia species, including the fruit, roots, leaves, and bark (Table 3). Certain chemicals exhibited numerous bioactivities both in vitro and in vivo.
3.1 Flavonoids
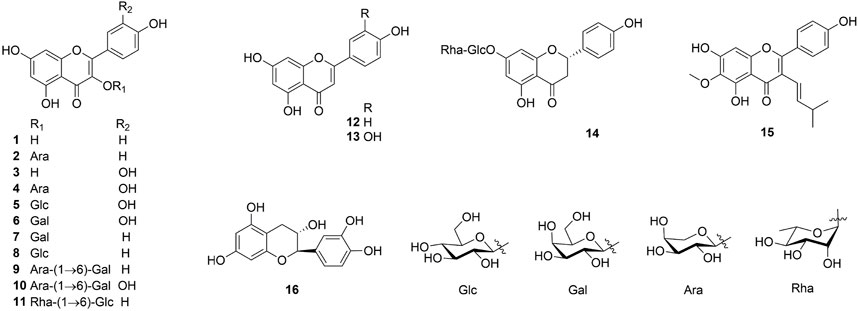
Flavonoids and their derivatives comprise the primary compounds of the Ehretia species. More than 16 compounds, including flavonoids and its glucoside (1–13, 15), a flavanonol (14) and a flavanol (16) have been identified from leaves of E. thyrsiflora, E. Asperula, E. ovalifolia and fruits of E. macrophylla. Flavonoid glycosides comprise a diverse range of sugars, including glucose (Glc), galactose (Gla), arabinose (Ara) and rhamnose (Rha). Compounds 9–11 amd 14 have two different sugars. Ovalifolin (15) is a new isopentenyl flavonoid got from E. ovalifolia leaves (Khattab et al., 2001). (+)-Catechin hydrate (16) is a flavanol identified from the fruit of E. macrophylla (He, 2018). Each of them contains only hydroxyl groups and lacks methoxyl groups on their aromatic rings.
3.2 Phenypropanoids
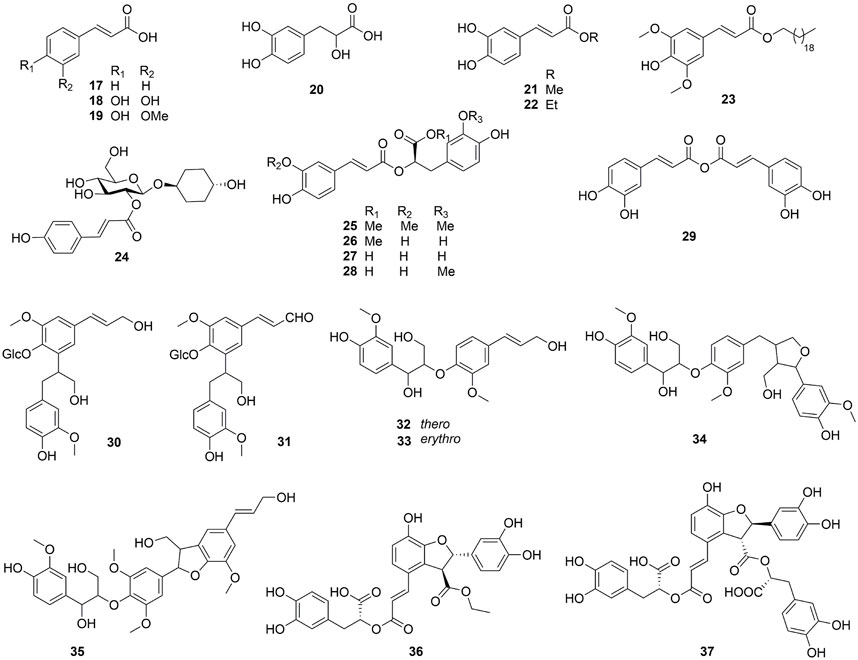
This genus is rich in phenypropanoids (17–37), which usually show C6-C3 carbon skeleton. The compounds can be categorized into phenylpropionic acids and lignans and got from the fruit, leaves and bark of Ehretia species. Compounds 17–22 are the simple phenylpropionic acids. Ehretiate (23) is a new trans-icosanyl sinapate that has been isolated from the root of E. longiflora (Chien et al., 2012). Rosmarinic acid (27) is an ester of caffeic acid and 3,4-dihydroxyphenyllactic acid that possesses a diverse range of activities (Ijaz et al., 2023; Petersen, 2013; Petersen and Simmonds, 2003). The Labiatae and Boraginaceae family contain the greatest concentration of rosmarinic acid (Dresler et al., 2017). Three novel compounds were obtained from the dried whole plant of E. obtusifolia: trans-4-hydroxycyclohexyl-2-O-p-coumaroyl β-D-glucopyranoside (24), methyl 2-O-feruloyl-1α-O-vanillactate (25) and caffeic anhydride (29); Ehletianol C (34), buddlenol B (35) and ethyl lithospermate (36) are lignans containing 3 phenylpropanes (Le et al., 2021; Yoshikawa et al., 1995). Four phenylpropanes connected by C-C and C-O-C bonds combine to generate the lignan known as lithospermic acid B (37) (Li et al., 2009).
3.3 Simple phenolics
The novel phenolic compounds, called ehletianols A and B (38 and 39), were separated from the bark of E. ovalifolia and contain two sugars (Yoshikawa et al., 1995). Simple phenolics with carboxyl or ester groups are compounds 40–44. The root of E. longiflora yielded ehretiamide (45), a novel phenolic containing acetyl group (Chien et al., 2012). Ehretioside B (46) is a new phenolic which connected with a cyanomethylene acid group (Simpol et al., 1994). Simple phenolics containing an isopentene or aldehyde group include prenylhydroquinone (47), 4-hydroxy-3-prenylbenzoic (48), proglobeflowery acid (49) (Chien et al., 2012).
3.4 Benzoquinones
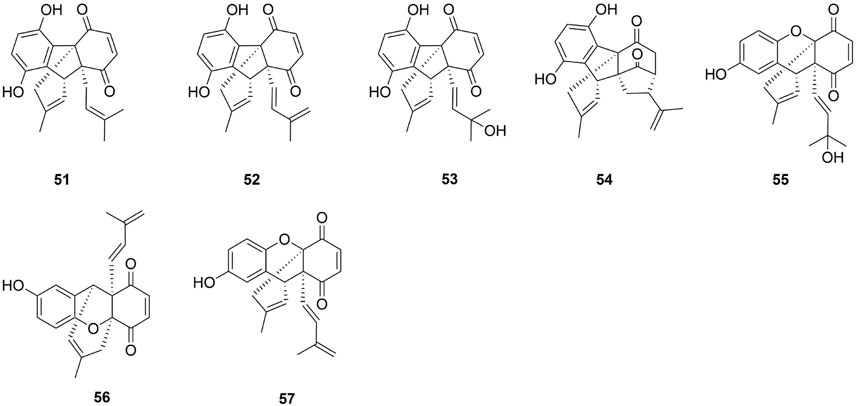
Seven benzoquinones dimeric prenylbenzoquinone moieties were identified in the aerial portions of E. microphylla, the root bark of E. buxifolia, and the root of E. longiflora. The absolute configuration of three novel compounds-microphyllone (51), ehretianone (56) and ehretiquinone (57)-was determined using X-ray crystallographic analysis (Agarwal et al., 1980; Chien et al., 2012; Selvanayagam et al., 1996). Compounds 52–55 are the congeners of microphyllone (51) (Yamamura et al., 1995).
3.5 Triterpenoids and sterols
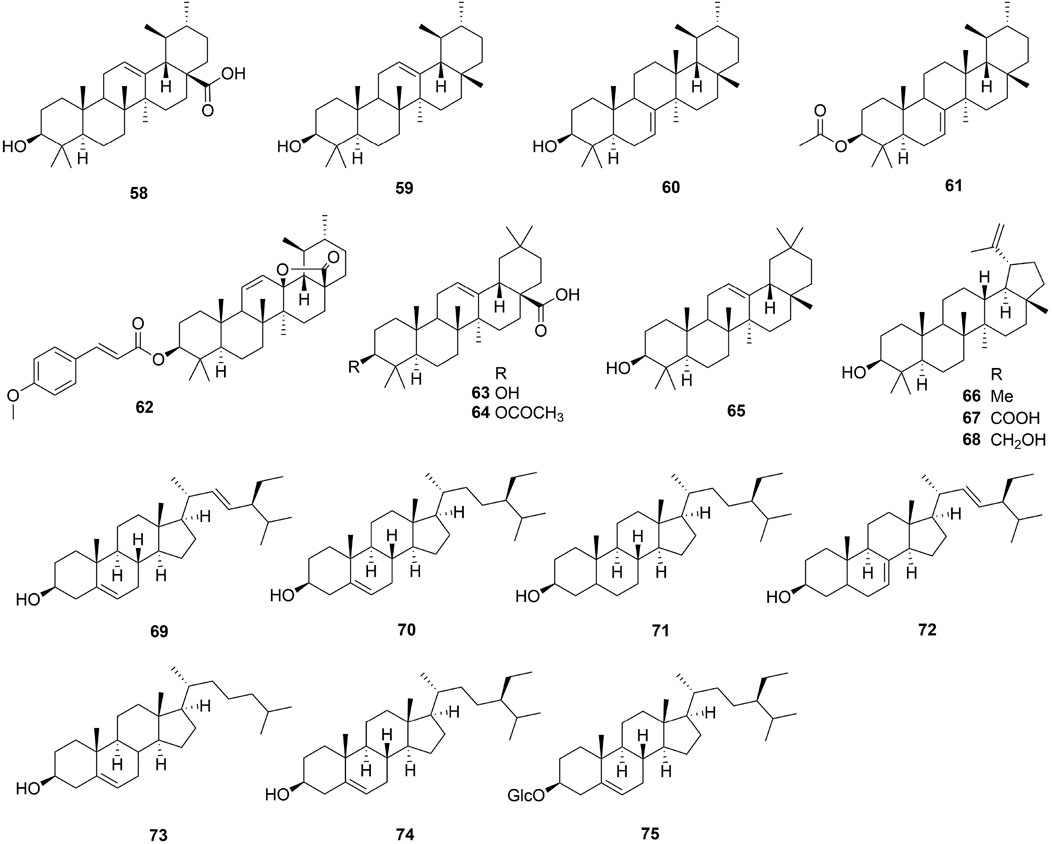
To now, only 3 types of triterpenoids (ursulane type, oleanane type and lupinane type) were reported from the Ehretia genus. These are all common triterpenoids, except for ehretiolide (62) (Chien et al., 2012). Ehretiolide (62) is a new ursulane type triterpenoid containing the O-(E)-4′-methoxylcinnamoyl and lactone groups. Additionally, the GC-MS detected every sterol with the exception of the daucoster (75), which was determined by TLC (Li et al., 2010b).
3.6 Fatty acids
This genus has been observed to contain 17 fatty acids. Fatty acid esters make up the majority of these molecules, with fatty alcohols making up the minority.
3.7 Others
A derivative of hydantolurea, allantoin (93) was isolated from E. thyrsiflora leaves, E. microphylla aerial parts, and E. rigida bark (Agarwal et al., 1980; Li et al., 2010b; Steyn, 1998). A novel pyrrolizidine, 7-O-(p-methoxybenzoyl)-retronecanol (94) has been isolated from E. aspera leaves (Suri et al., 1980). Ehretine (95) is a new alkaloid which having a cyclohex-2-en-1-one moiety (Chien et al., 2012). Simmondsin (96), ehretioside A1 (97), ehretioside A2 (98), ehretioside A3 (99) are four cyanoglucosides which were isolated from E. philippinensis bark (Simpol et al., 1994).
4 Biological activity
4.1 Antioxidant activity
The ethyl acetate (EA) extracts from the bark of E. acuminata exhibited potent antioxidant activity, with IC50 values of 22 μg/mL, 26 μg/mL, and 140 μg/mL for ABTS, DPPH, and NO radical scavenging assays, respectively (Table 4). In comparison, the standard ascorbic acid (AA) had IC50 values of 28 μg/mL, 30 μg/mL, and 175 μg/mL, respectively. This suggests that the bark of this plant possesses significant health benefits due to its antioxidant properties, as supported by previous research (Kaur et al., 2019). The EA extract from the fruit of E. acuminata also demonstrated strong free radical scavenging capacity, as reported by Shukla group, with IC50 values of 40 μg/mL, 50 μg/mL, and 380 μg/mL for ABTS, DPPH, and NO assays, respectively. For the same assays, the standard AA had IC50 values of 23 μg/mL, 27 μg/mL, and 230 μg/mL, respectively (Shukla et al., 2019b). Notably, the EA extract from the leaves of E. acuminata exhibited the highest antioxidant activity, with IC50 values of 90 μg/mL, 99 μg/mL, and 250 μg/mL for ABTS, DPPH, and NO assays, respectively (Shukla et al., 2019a). In vitro antioxidant and radical scavenging assays conducted on E. serrata fruits and leaves revealed that the EA fraction of the leaf extract exhibited the highest activity. This is attributed to its high phenolic content. Additionally, the EA fraction of the fruit extract showed the highest lipid peroxidation value, followed by the leaf fraction. Significant free radical scavenging potential was also observed in the chloroform (CH) and 1-butanolic fractions of the leaf extract (Zara et al., 2012). R28 cells were used to evaluate the protective effects of phytochemicals and ethanolic extracts from E. asperula leaves against oxidative stress-induced retinal cell loss and excitotoxicity. The results indicated that both 70% and 95% ethanolic leaf extracts enhanced cell viability under these conditions. Furthermore, methyl rosmarinic acid (26) and rosmarinic acid (27) were particularly effective in preventing retinal cell death and increasing ROS levels in cells exposed to glutamate/BSO-induced excitotoxicity/oxidative stress. These findings suggest that E. asperula leaves may have therapeutic potential for treating retinal degeneration (Le et al., 2021).
The high phenolic content of E. macrophylla peel extracts is a key factor contributing to their remarkable antioxidant activity, as evidenced by PSC and ORAC values of 859.85 ± 70.32 μmol vit. C equiv./g and 2877.01 ± 163.80 μmol trolox equiv./g, respectively (Lu, 2016). According to Huma, there was a notable reduction in DPPH after 60 min (94.1%) in the stem bark extract of E. serrata and 30 min (85.01%) in the fruit extract of E. obtusifolia at the dosage level of 1,000 μg/mL (Huma, 2019). Pío-León research group demonstrated that the antioxidant activity of E. tinifolia fruits was comparable to or higher than that of various tropical fruits, including guava, orange, and prickly pears. The 70% ethanolic extract of E. laevis flowers exhibited robust antioxidant effects, as shown by DPPH and NO reducing power assays (Rangnathrao and Shanmugasundaram, 2019). Compared to rodents subjected to paracetamol-induced toxicity, the CH and EA extracts of E. microphylla significantly increased antioxidant parameters (CAT, SOD, GSH, and GPx) in rats (Yuvaraja et al., 2021). The methanol fraction of E. cymosa leaves exhibited significantly stronger (P < 0.05) scavenging activities for DPPH (0.47 mg/mL), ABTS (0.49 mg/mL), and -OH radical (0.55 mg/mL) (Ogundajo and Ashafa, 2017). Two polyphenols found in E. tinifolia Linnaeus, TEAC (4,134 ± 9.7 μM TE/g dry extract) and DPPH (EC50 = 0.32 ± 0.03 mg/mL), demonstrated significant free radical scavenging capacity (Deng et al., 2020). After extracting active compounds from the dry fruit of E. macrophylla using 70% ethanol, four distinct solvents (EA, n-butanol, CH, and PE) were used to remove the remaining contents. Methyl rosmarinate (26) showed strong anti-oxidation action, with ORAC, PSC, and CAA values of 28,976.58 ± 3,589.36 μmol TE/g DW, 32,139.75 ± 3,979.76 μmol EVc/g DW, and 317.77 ± 36.69 μmol QE/100μmol, respectively. The EA fraction demonstrated strong antioxidant activity, with higher ORAC and PSC values compared to methyl caffeate (21) (He, 2018). Ehretiquinone (57), isolated from the root of E. longiflora, inhibited superoxide formation generated by N-formyl methionyl leucyl phenylalanine (fMLP) with an IC50 value of 0.36 ± 0.03 μM (Chien et al., 2012).
4.2 Anti-diabetic activity
The antidiabetic potential of various bark extracts of E. acuminata was evaluated in vitro using α-amylase and α-glycosidase inhibition assays. Among the tested extracts, only the CH extract demonstrated significant inhibitory activity against both enzymes, with IC50 values of 43.35 μg/mL for α-amylase and 42.90 μg/mL for α-glycosidase. These findings suggest that E. acuminata bark contains bioactive compounds with promising antidiabetic property (Kaur et al., 2022). In a separate study, researchers investigated the effects of an aqueous extract of E. anacua leaves on alloxan-induced diabetic rats. Histological examination of pancreatic tissues revealed that blood glucose levels were significantly higher in diabetic rats compared to the control group. Treatment with the aqueous extract at doses of 50, 100, and 200 mg/kg reduced blood glucose levels and preserved pancreatic histoarchitecture. Microscopic examination of pancreatic tissue showed significant architectural damage in the alloxan-treated group, while the plant extract-treated group exhibited normal architecture. This indicates that the extract mitigates alloxan-induced toxicity. These results support the use of the extract as a dietary supplement with hypoglycemic and antidiabetic activities for functional foods (Abimbola et al., 2021). The bioactive compounds in E. cymosa leaves were also studied for their antidiabetic properties. Methanol and EA fractions exhibited statistically significant inhibition (P < 0.05) against α-amylase (IC50: 2.11 mg/mL and 2.75 mg/mL, respectively) and α-glucosidase (IC50: 0.66 mg/mL and 0.60 mg/mL, respectively). Additionally, the methanol fraction inhibited α-amylase through a competitive mechanism and α-glucosidase through a noncompetitive mechanism. These findings suggest that E. cymosa leaves contain bioactive compounds with therapeutic potential for diabetes treatment (Ogundajo and Ashafa, 2017). Antihyperglycemic efficacy of a 70% ethanolic fraction of the whole E. cymosa plant was investigated in vivo using Sprague Dawley rats. Additional ex vivo experiments were performed to determine the modulatory effects on the absorption of glucose in the intestines. Irrespective of the dosage, the extract substantially decreased the fasting blood glucose level and equally reduced the amount of glucose absorbed by the rat intestinal sacs. The findings validate the conventional application of E. cymosa extract as a pharmaceutical treatment for diabetes in susceptible mice (Sarkodie et al., 2015). Phenolic compounds from E. macrophylla fruit exhibited effective hypoglycemic activity by inhibiting α-glucosidase and α-amylase, increasing glucose consumption and glycogen accumulation, and reducing G6Pase and PEPCK activity. Thus, E. macrophylla fruits have the potential to enhance human health as metabolites in functional foods, offering further health and economic benefits (Deng et al., 2020). E. tinifolia extracts showed no inhibitory effect on lipase but selectively inhibited α-glucosidase and α-amylase (Monroy-García et al., 2021). Sequential Soxhlet extraction of E. acuminata leaves yielded both nonpolar and polar extracts, which were evaluated for antidiabetic potential using spectrophotometric analysis. The CH extract had the highest antidiabetic activity, with an IC50 ranging from 260 to 265 μg/mL (Shukla et al., 2021).
4.3 Analgesic and anti-inflammatory activity
The volume of the paw edoema was substantially reduced in the carrageenan- and formalin-induced rat paw edoema methods when the paw edoema was generated by the herbal hydrogel containing the methanol extract of E. laevis leaves (HEL3). HEL3 exhibited 78.75% and 79.51% inhibition, respectively, compared to the standard indomethacin, which showed 80.24% and 81.12% inhibition at 360 min. HEL3 demonstrated potent anti-inflammatory and wound healing properties (Memon et al., 2022). The mechanisms underlying the anti-inflammatory properties of the methanol extract of E. tinifolia (ETME) were elucidated. ETME significantly increased total GSH levels and decreased pro-inflammatory cytokine and NO production. Additionally, ETME reduced the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 in response to LPS. The study’s results suggest that ETME may be capable of protecting Kupffer cells from NF-κB and MAPKs, as well as LPS-induced oxidative stress and heightened inflammatory responses can be mitigated through the activation of the antioxidant pathway involving Nrf2 and HO-1 (Lim et al., 2023). The spectrophotometric technique was employed to evaluate the anti-inflammatory properties of the leaf extracts (PE, CH, EA, EOL, and aqua) of E. acuminata. Shukla group reported that the EA extract exhibited the most potent anti-inflammatory activity (Shukla et al., 2021). When tested in vitro with hen egg albumin, the EA and ethanol extracts of E. acuminata bark shown potent anti-inflammatory properties (IC50 values of 170 μg/mL and 172 μg/mL, respectively). According to the findings, the bark of E. acuminata contains chemicals that show promise as probable anti-inflammatory agents (Kaur et al., 2022). Compound 77, extracted from E. dicksonii, demonstrated a 43% inhibitory effect on TPA-exposed inflammation in the ears of mouse when administered at a dose of 500 μg. Compounds 78, 80, 81, 86 and 84 exhibited potent activity with, IE500 μg of 32%, 19%, 39%, 63%, and 79%, respectively (Dong et al., 2000). Acetic acid-induced writhing and hot plate experiments, carrageenan-induced paw edoema, and cotton-pellet-exposed granuloma models were employed to evaluate the analgesic and anti-inflammatory properties of E. cymosa leaves. The findings of this investigation substantiate the conventional application of E. cymosa in the management of a diverse array of inflammatory and painful conditions. Specifically, the 80% methanol, aqueous, EA, and CH extracts of the plant demonstrated substantial analgesic and anti-inflammatory effects (Ashagrie et al., 2023). The four distinct solvents (EA, n-butanol, CH, and PE) were used to extract the compounds that were shown to be effective from the dried fruit of E. macrophylla after 70% ethanol was used. Strong anti-inflammatory activity was demonstrated by the EA and CH fractions, with IC50 values for preventing NO release of 19.10 ± 0.31 μg/mL and 19.48 ± 0.25 μg/mL, respectively.
Methyl caffeate and methyl rosmarinate, two compounds that were extracted from E. macrophylla, showed better anti-inflammatory properties than the other four fractions; these compounds notably reduced the levels of TNF-α, IL-1β, and iNOS at 2 μg/mL and 1.25 μg/mL, respectively (He, 2018). Jyothirmai group demonstrated that the CH, methanolic, and aqueous extracts of E. laevis exhibited significant anti-inflammatory efficacy by reducing paw volume at varying concentrations (Jyothirmai et al., 2016). With the exception of stem bark, E. serrata demonstrated a highly significant analgesic effect at all dose levels (100, 200, and 300 mg/kg) over a 60-min period. The stem bark of E. obtusifolia exhibited highly significant (P < 0.01) results at all concentrations after 60 min. The anti-inflammatory effects of E. serrata and E. obtusifolia were found to be highly significant (P < 0.01) at all concentrations, including 100 mg/kg, 200 mg/kg, and 300 mg/kg, after two and 3 h (Huma, 2019).
4.4 Hepatoprotective activity
The hepatoprotective effect of the hydroalcoholic (70% ethanol) and EA fractions of E. laevis flowers were evaluated using Wistar rats. Both extracts demonstrated significant dose-dependent protection against alterations in serum aspartate ASAT, ALAT, ALP, and TP at oral doses of 100 and 200 mg/kg. Additionally, they provided dose-dependent protection against liver tissue modifications, such as necrosis, fatty degeneration, and lymphatic infiltration, induced by paracetamol. The EA fraction exhibited superior activity compared to the hydroalcoholic extract. These findings suggest that E. laevis extracts have promising potential as preventive treatments for liver damage (Rangnathrao and Shanmugasundaram, 2019). Similarly, the CH and EA extracts of E. microphylla significantly protected rats from paracetamol-induced liver toxicities. Elevated levels of SGOT, SGPT, ALP, triglycerides, and total cholesterol in paracetamol-induced liver injury were effectively reduced by pretreatment with E. microphylla extracts (CH and EA, 200 mg/kg), comparable to silymarin (100 mg/kg). In summary, E. microphylla extracts demonstrated substantial hepatoprotective benefits in the context of paracetamol-induced liver injury in rats. The hepatoprotective properties of E. microphylla may be attributed to the presence of flavonoids and phenolic compounds (Yuvaraja et al., 2021).
4.5 Muscle relaxant and antispasmodic activity
The bark extracts of E. acuminata exhibited concentration-dependent increases in intestinal motility in experimental animals. These extracts also demonstrated antispasmodic, analgesic, and muscle relaxant activities at 300 mg/kg, with no observed acute toxic effects in tested mice. Specifically, the antispasmodic activity was measured at 11 ± 1, 9 ± 1, and 11 ± 1; analgesic activity at 10 ± 1, 16 ± 1, and 11 ± 1; and muscle relaxant activity at 6 ± 1, 5 ± 1, and 5 ± 1. This study provides significant evidence supporting the pharmacological use of E. acuminata as an analgesic, antispasmodic, and muscle relaxant (Jan et al., 2023). Additionally, the methanolic extracts of the fruit, stem bark, and leaves of E. serrata and E. obtusifolia were evaluated for antispasmodic activity in Swiss albino rodents. The methanolic extract of the stem bark showed highly significant results (P < 0.01) at concentrations of 200 and 300 mg/kg. E. obtusifolia exhibited enhanced smooth muscle relaxation at low doses. The highly significant (P < 0.01) activities of the leaf (200 and 300 mg/kg), fruit (300 mg/kg), and stem bark extracts (all concentrations) scientifically validated their ethnopharmacological use as antispasmodic agents (Huma, 2019).
4.6 Antibacterial activity
The antibacterial potential of phytoconstituents was examined by assessing their ability to disrupt bacterial cell permeability and inhibit bacterial growth. The methanolic extract of E. serrata leaves demonstrated effective antibacterial activity against five tested bacteria, with ZOI ranging from 10.3 to 29.0 mm. The MIC values ranged from 0.8 to 5.1 mg/mL (Waheed et al., 2019). The CH, methanolic, and aqueous extracts of E. laevis exhibited exceptional antibacterial efficacy against both gram-positive and gram-negative bacteria, with the methanolic fraction showing the most potent activity (Jyothirmai et al., 2016). Two compounds, prenylhydroquinone (47) and ehretiquinone (57), were isolated from the methanolic fraction of E. longiflora root. These compounds exhibited antitubercular effect inhibit Mycobacterium tuberculosis strain H37Rv, with MIC values of 26.2 and 25.0 μg/mL, respectively (Chien et al., 2012). The ethanol extract of E. acuminata leaves exhibited the most extensive ZOI (12–18 mm) against various food poisoning bacteria (Shukla et al., 2021). The agar diffusion assay revealed that the 70% ethanolic fraction of the entire plant of E. cymosa inhibited P. aeruginosa, E. coli, B. subtilis, and S. aureus (Sarkodie et al., 2015). The extract exhibited inhibitory effect against P. aeruginosa, E. coli, B. subtilis, and S. aureus (Sarkodie et al., 2015). The methanolic extract of E. serrata leaves exhibited a substantial antibacterial zone (7 mm) at a dosage level of 1,000 μg/mL, in contrast to the reference drug’s (31.5 mm). The leaf extract of E. obtusifolia exhibited a more potent antibacterial zone (7 mm) than the reference medication (30.2 mm) at a dosage level of 1,000 μg/mL (Huma, 2019).
4.7 Anticancer activity
The leaves of E. laevis were utilized to synthesize spherical silver nanoparticles with diameters ranging from 25 to 35 nm. These nanoparticles exhibited potent anticancer activity, demonstrating high stability in solution and significant cytotoxicity. Specifically, the median lethal concentrations (LC50) for HeLa and MCF-7 cells were 12.7 μg/mL and 14.5 μg/mL, respectively (Panja et al., 2020). The effective compounds were extracted from E. macrophylla fruit by 70% ethanol, and then extracted with four different solvents. The EA and CH fractions exhibited significant anti-proliferation activity in tested dosage. Methyl caffeate (21) demonstrated stronger anti-proliferation effect on Caco-2 cells (EC50 = 132.73 ± 18.19 μM), while EC50 value of methyl caffeate on HepG2 cells (EC50 = 48.35 ± 0.61 μM) (He, 2018). E. microphylla (extract of leaves with flower buds) showed a significant and highly selective antiproliferative effect on cancer cells (Kudera et al., 2021). At a concentration of 50 μg/mL, the CH extract of E. microphylla leaves inhibited MCF-7 and A-549 cancer cell lines by 85.55% and 77.93%, respectively. The DAPI staining method was employed to investigate the mechanism of cell death, which revealed alterations in nuclear morphology in MCF-7 cell lines in the form of distinct changes that were observed during the staining process (Sharma et al., 2022). The extract of the fruit of E. macrophylla showed dose-dependent antiproliferative activity, possibly influenced by the synergistic and additive effects of individual phenolics (Deng et al., 2020). Both E. tinifolia and Sideroxylon lanuginosum Michaux (Sapotaceae) exhibited antiproliferative activities against HeLa, HT-29 and MCF-7 cells (Monroy-García et al., 2021).
4.8 Antihemolytic activity
The antihemolytic properties of the leaf extracts (PE, CH, EA, ethanol, and aqua) of E. acuminata were assessed using spectrophotometric technique. The EA extract demonstrated the most potential for antihemolytic activity, with an IC50 of 90 μg/mL (Shukla et al., 2021). It was demonstrated that E. tinifolia polyphenols had the ability to reduce hemolysis (IC50 = 58.55 ± 2.4 μg/mL) (Monroy-García et al., 2021).
4.9 Anti-arthritic activity
The methanolic extract of E. laevis leaves (500 mg/kg) in mice prevented paw edoema from increasing by 60% and by 56%, as per Velappan and Thangaraj (Velappan and Thangaraj, 2014).
4.10 Wound healing activity
With healing times ranging from 7 days to a maximum of 66 days, E. laevis leaves show considerable healing properties in both infected and non-infected, chronic and fresh wounds. These properties become more effective as one ages. According to the study, the patient’s immune status remained unaffected because no antibiotics were administered (Thakre et al., 2016). The study also showed that the CH fraction of the EtOH extract of E. Laevis has the same wound-healing properties as Povidon iodine ointment in wistar rats (Harne et al., 2021). In 5% and 10% concentrations, the alcoholic extract ointment of C. retusa root, stem, and leaves would be able to stimulate wound healing activity. Swiss albino mice were employed to test the effectiveness of wound healing. The ointment prepared from different fraction showed significant effectiveness in wound contraction and faster wound closure compared to the standard Nitrofurazone (0.2%) ointment. As a result, the wound healing study demonstrated that C. retusa is available in facilitating wound closure (Mageswari et al., 2012).
4.11 Anti-amoebic activity
The leaves of Ampalaya (Momordica charantia L.) and Tsaang Gubat (E. microphylla) were processed to create a lyophilized aqueous extract. Against Entamoeba histolytica, tsaang gubat and ampalaya leaves did not show any anti-amoebic activity. In reality, they promoted the development of amoebae at all dose levels. The IC50 of the extracts of tsaang gubat and ampalaya leaves was greater than 500 μg/mL at 24, 48, and 72 h. These findings contradict the conventional application of these herbal medicines to alleviate diarrhoea (Maramba-Lazarte et al., 2020). The leaves of E. laevis were used to create the synthetic silver nanoparticles, which demonstrated larvicidal activity and efficient methylene blue dye degradation. The nanoparticles were also extremely stable in solution and active. After being treated for 72 h, it killed 70% ± 10.24% of Culex quinquefasciatus larvae at a dosage of 25 μg/mL. Within 8 h, the nanoparticles reduced Congo red by approximately 85% at a concentration of 200 μg/mL. Furthermore, when exposed to sunlight, the produced nanoparticles may function as a water purifying agent (Panja et al., 2020).
4.12 Lipoxygenase inhibitory activity
Compounds 77, 81, 84 and 86 isolated from the fresh leaves and twigs of E. dicksonii exhibited inhibitory effect against soybean lipoxygenase at a concentration of 10 μg/mL (Dong et al., 2000). Compounds 24–27 and 29 demonstrated concentration-dependent inhibition of lipoxygenase, with Ki values ranging from 0.85 to 57.6 μM. Compounds 26 and 27 were the most potent, having Ki values of 0.85 μM and 1.0 μM, respectively. Compounds 25–27 and 29 exhibited noncompetitive inhibition, whereas compound 24 was classified as an uncompetitive inhibitor of lipoxygenase (Iqbal et al., 2005).
4.13 Modulation of gut microbiota
A new polysaccharide called EWMFP was effectively isolated and identified from E. macrophylla fruit by Xu group. The impacts on the gastrointestinal microbiota of humans were investigated using an in vitro fermentation model that reproduces the micro-ecosystem of the human colon. With a molecular weight of 12.45 KDa, EWMFP is made up of four monosaccharides and is better than inulin at preserving microbial diversity. By the end of fermentation, EWMFP produces a higher concentration of butyrate than inulin and can modify the composition of gut microbes in a different way. Differing routes may lead to differing compositions of short chain fatty acids (SCFA) during fermentation between EWMFP and inulin. According to their research, EWMFP may have a new role as a prebiotic in controlling colonic health (Deng et al., 2020).
4.14 Other activities
Erhetianone (56), which was isolated from a methanolic preparation of the root bark of E. buxifolia, has been shown to have antisnake venom activity against the venom of Echis carinatus in rodents. The antisnake venom effect of compound 56 was determined by calculating the LD50 of E. carinatus venom in mice that was administered subcutaneously (Selvanayagam et al., 1996). It was discovered that the butanolic and EA extracts of E. philippinensis bark exhibited anti-histamine releasing activity (Simpol et al., 1994). The objective of this work was to investigate the impact of E. microphylla on the quantity of ovarian surface follicles, relative weight of the ovaries and uterus, and folliculogenesis in female Wistar albino rats. Hematology required the removal of the uterus and ovaries. The findings show that 1,000 mg/kg of powdered E. microphylla increased the levels of FSH, LH, and estradiol significantly. Additionally, increased folliculogenesis was found along with increased ovarian and uterine weight. As a result, the results point to a noteworthy stimulating effect on female reproductive function that might improve adult female rats’ fertility (Aarthi et al., 2012).
5 Toxicology
The alcoholic extract from E. microphylla leaves did not exhibit any chromosome-fragmentation generating activity in the mutagenicity or genotoxicity assays (Balboa and Sylianco, 1993; Legaspi and Bagaoisan, 2020). Only one report on acute oral toxicity for E. laevis adheres to the OECD recommendation of 423. All of the methanolic extracts from the fruits, stems, and foliage in this investigation were determined to be safe at a dosage of 2,000 mg/kg (Sharma et al., 2021; Velappan and Thangaraj, 2014). The safety and effectiveness of E. laevis and E. microphylla extracts and fractions for a range of illnesses are not adequately supported by the few and inadequate toxicity reports that are currently available. Moreover, there is a dearth of information in the literature regarding this genus’s toxicity.
6 Clinical studies
The tsaang gubat tablets contains a 10% leaf aqueous extract that has been shown to be both safe and efficacious in the treatment of gastrointestinal and biliary colic pain. In a Phase II clinical trial conducted in Pila and Victoria, Laguna, the formulation was administered to five male patients who were diagnosed with acute colic due to lax bowel movements. The following effects were observed after the treatment: (1) all patients reported experiencing relief from intestinal spasms or colic, and (2) the onset of colic relief typically occurred 20–30 min after the dosage. Efficacy, tolerability, and acceptability of the tsaang gubat tablet at a dose of 150 mg/kg were comparable to those of dicycloverine at a dose of 0.5 mg/kg in Phase II clinical trials conducted at the Tarlac Provincial Hospital. The patients were 110 outpatients with acute mild, moderate, or severe biliary colic. The laboratory tests conducted subsequent to the administration of the tsaang gubat tablet did not reveal any abnormalities, and the patients reported no adverse effects (Legaspi and Bagaoisan, 2020). It has been registered with the Philippine Food and Drug Administration, is currently listed in the Philippine National Formulary and has been licensed to a limited number of local pharmaceutical enterprises.
7 Cultivation and commercial value
In recent years, there are more and more reports about the cultivation of this genus with the development and utilization of some plants of Ehretia genus. It is mainly divided into three cultivation methods. The first one is seed cultivation. The plant fruit begins in ripe and can be collected centrally when the fruit changes from green to orange-red or yellow. The fruit was peeled and stratified after harvest. The seeds are sprouted and sown when the soil thaws in the following spring. The other two methods are root and branch cottage (Huma, 2019; Zhou, et al., 2012). As a kind of Kudingcha, the leaves of E. thyrsiflora have great commercial value as a health tea in China (Wang and Huang, 2005). Furthermore, Tupipa (the fruit of E. macrophylla) has been developed as a new food in Hengnan County, China, to promote local economic development and help rural revitalization. Up to now, the county planting area of more than 30,000 mus and the sales of Tupipa products reached 100 million yuan in 2023 (http://lyj.hunan.gov.cn/). E. tinifolia produces small, globe-shaped yellow drupes measuring up to 8 mm in diameter. These fruits have a sweet taste and have been utilized as both food and medicinal plants in various regions of Mexico and the United States (Monroy-García et al., 2021). E. microphylla is considered the most promising species for large-scale production and is used extensively in traditional Philippine medicine. Based on the findings of early experiments, recommendations were made on the cultivation, harvesting, and storage (where applicable) of vegetative propagation (Mageswari and Karpagam, 2015). Some plants of Ehretia genus are often used as herb medicine, food, street tree and yard planting, so the cultivation and commercial value of this genus have certain reference value.
8 Conclusion and perspectives
The biology, ethnopharmacology, phytochemistry, pharmacology, toxicity, clinical studies, cultivation and commercial value of the Ehretia genus were summarized in this paper. This genus is widely distributed across various regions, with some species being particularly diverse. It has been extensively utilized in traditional medicine and food practices. Although there are commonalities in medicinal applications, these vary significantly depending on the species and geographical location. In fact, Ehretia species are used to treat skin conditions, pain and inflammation, and digestive issues in many different countries. Numerous pharmacological activities, including the antioxidant, anti-diabetic, and anti-inflammatory characteristics, have been studied in vivo and in vitro in several species. While most studies have concentrated on crude extracts, certain active compounds have also been evaluated. There is evidence that flavonoids and phenylpropanoids have antioxidant properties.
In Asian countries, ethnic populations employ E. leavis as masticatories. Undiscovered wound-healing properties have been shown by E. leavis. The finest aspect is that it produces a lot of material without requiring the plant to be uprooted since its leaves are effective. Indian scientists have conducted several studies to enhance the germination of multifunctional trees such as E. leavis. To learn more about the characteristics of the genus Ehretia, molecular studies have also been conducted on a few species. E. tinifolia has recently been the subject of study that indicates beneficial responses against cardiovascular illnesses, atherosclerosis, and diabetes problems (Panja et al., 2020).
However, many questions remain regarding our comprehensive understanding of the Ehretia genus, necessitating further research. First, given their historical applications, certain species warrant more in-depth investigation. Notably, E. microphylla is extensively used in China, India, and the Philippines to treat gastrointestinal and biliary colic, diarrhea, spasms, and inflammation. Studies on this species’ antispasmodic activity have revealed its antioxidant, hepatoprotective, antibacterial, anti-inflammatory, antidiabetic, anticancer, and wound-healing properties. These findings may lead to the discovery of novel bioactive compounds. Therefore, additional research should focus on traditionally used plants that have been overlooked but offer clear benefits. Second, despite limited documentation of traditional usage for most species within this genus, abundant resources allow for continued scientific exploration into these species.
Thirdly, despite the fact that extracts from a number of Ehretia species have shown strong activity, comparatively few of the chemicals causing these effects have been found in relation to the number of pharmacological investigations that have been carried out. For instance, E. anacua is said to exhibit strong anti-diabetic effects in vivo; yet, this plant’s secondary compounds have not been found. Therefore, bio-guided isolation should be used to identify the bioactive compounds from these species. Moreover, there is a great chance of finding novel active chemicals in the genus Ehretia because of its high degree of endemism. Lastly, although systematic study is still lacking, research on this species focuses mostly on the investigation of its chemicals and biological activity. Establishing quality standards and conducting more complete investigations are vital for future studies to guarantee the integrity and efficacy of the findings pertaining to this genus. It is recommended to add techniques or approaches that can address these gaps in the future research (e.g., metabolomics, high-throughput screening, HPLC-MS). Furthermore, some of the literatures are not high-level papers, and the experimental data can only be used for reference.
In conclusion, further research is required because the phytochemistry and pharmacology of the Ehretia genus have not been thoroughly examined. To date, 101 compounds have been identified from this genus, with phenylpropanoids being the most active class. Rosmarinic acid showed different strong activity in this genus. Several species’ medicinal potential has been highlighted by numerous biological investigations, providing a strong basis for further investigation. It is imperative that further research be carried out in vivo with suitable dosage levels and controls. To guarantee safety and effectiveness, the side effects connected to the effective doses should also be thoroughly assessed. Given the rich resource base and significant medical value of this genus, continued research is warranted. This paper contributes scientific value to the ongoing development of the genus.
Author contributions
SJ: Writing–original draft, Writing–review and editing. MW: Writing–original draft. AK: Writing–review and editing. LJ: Investigation, Validation, Writing–review and editing. YC: Formal Analysis, Investigation, Writing–review and editing. JL: Data curation, Writing–review and editing. ML: Investigation, Writing–review and editing. HW: Investigation, Writing–review and editing. DW: Conceptualization, Writing–review and editing. YP: Conceptualization, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This work was financially supported by the key Research and Development Plan Project of Hunan province (2021SK2007), Natural Science Foundation of Hunan province (2024JJ8186).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Aarthi, V., Anbu, J., Nazeer Ahamed, K., Anjana, A., and Velpandian, V. (2012). Effect of Ehretia microphylla lamk on stimulation of reproductive function and ovarian folliculogenesis in rats. Int. J. Pharm. Bio. Sci. 3, 273–280.
Aarthi, V., Shakila, R., Sasikala, E., and Pitchiahkumar, M. (2014). Pharmacognostical studies on Ehretia microphylla lamk. Asian J. Trad. Med. 9, 118–129.
Abimbola, O. W., Ademola, O. J., and Olawande, F. T. (2021). Effect of administration of Ehretia anacua aqueous extract on blood glucose level in alloxan-induced diabetic rat. World J. Adv. Res. Rev. 11, 001–009. doi:10.30574/wjarr.2021.11.2.0068
Agarwal, S. K., Rastogi, R. P., Van Koningsveld, H., Goubitz, K., and Olthof, G. J. (1980). The molecular structure of 4a, 5, 8, 8a-tetrahydro-11, 14-dimethoxy-7-methyl-4a-(3-methyl-2-butenyl)-5, 8a-o-benzeno-1, 4-naphthoquinone. Tetrahedron 36, 1435–1438. doi:10.1016/0040-4020(80)85058-7
Alemayehu, G., Asfaw, Z., and Kelbessa, E. (2015). Ethnobotanical study of medicinal plants used by local communities of minjar-shenkora district, North shewa zone of amhara region, Ethiopia. J. Med. Plants Stud. 3, 1–11.
Ali, S. A., Sharief, N. H., and Mohamed, Y. S. (2019). Hepatoprotective activity of some medicinal plants in Sudan. Evid-Based Compl. Alt. Med. 2019, 2196315–2196316. doi:10.1155/2019/2196315
Arenas, P. M., Molares, S., Aguilar Contreras, A., Doumecq, B., and Gabrielli, F. (2013). Ethnobotanical, micrographic and pharmacological features of plant-based weight-loss products sold in naturist stores in Mexico City: the need for better quality control. Acta Bot. Bras. 27, 560–579. doi:10.1590/s0102-33062013000300014
Ashagrie, G., Abebe, A., and Umer, S. (2023). Analgesic and anti-inflammatory activities of 80% methanol extract and solvent fractions of Ehretia cymosa Thonn (boraginaceae) leaves in rodents. J. Exp. Pharm. 15, 63–79. doi:10.2147/JEP.S396769
Balboa, J., and Sylianco, C. (1993). Antigenotoxic effects of drug preparations from lagundi, tsaang gubat and ulasimang bato. Philipp. J. Philipp. J. Sci. 122, 1–14.
Chaluma, S., Ruth, S., Gemechu, G., Hailemichael, T., Aman, D., Teshome, A., et al. (2018). Antibacterial triterpenoid from the leaves extract of Ehretia cymosa. Ethiop. J. Sci. Sust. Dev. 5, 42–53.
Chien, Y. C., Lin, C. H., Chiang, M. Y., Chang, H. S., Liao, C. H., Chen, I. S., et al. (2012). Secondary compounds from the root of Ehretia longiflora and their biological activities. Phytochemistry 80, 50–57. doi:10.1016/j.phytochem.2012.04.013
Chopra, R. N., Nayar, S. L., Chopra, I. C., Asolkar, L., and Kakkar, K. (1956). Glossary of Indian medicinal plants, New Delhi. Counc. Sci. Ind. Res. 55, 1–329.
Choudhury, S., Sharma, P., Choudhury, M. D., and Sharma, G. D. (2012). Ethnomedicinal plants used by Chorei tribes of Southern Assam, North eastern India. Asian pac. J. Trop. Dis. 2, 141–147. doi:10.1016/s2222-1808(12)60140-6
Deng, N., Zheng, B. S., Li, T., Hu, X. D., and Liu, R. H. (2020). Phenolic profiles, antioxidant, antiproliferative, and hypoglycemic activities of Ehretia macrophyla Wall. (EMW) fruit. J. Food Sci. 85, 2177–2185. doi:10.1111/1750-3841.15185
Dong, M., Oda, Y., and Hirota, M. (2000). (10E,12Z,15Z)-9-hydroxy-10,12,15-octadecatrienoic acid methyl ester as an anti-inflammatory compound from Ehretia dicksonii. Biosci. Biotech. Bioch. 64, 882–886. doi:10.1271/bbb.64.882
Dresler, S., Szymczak, G., and Wójcik, M. (2017). Comparison of some secondary metabolite content in the seventeen species of the Boraginaceae family. Pharm. Biol. 55, 691–695. doi:10.1080/13880209.2016.1265986
Fassil, A., and Gashaw, G. (2019). An ethnobotanical study of medicinal plants in chiro district, West Harare, Ethiopia. Afr. J. Plant Sci. 13, 309–323. doi:10.5897/ajps2019.1911
Hadjichambis, A. C., Paraskeva-Hadjichambi, D., Della, A., Elena Giusti, M., De Pasquale, C., Lenzarini, C., et al. (2008). Wild and semi-domesticated food plant consumption in seven circum-Mediterranean areas. Int. J. Food Sci. Nutr. 59, 383–414. doi:10.1080/09637480701566495
Harne, K., Tekade, P., and Thakre, R. (2021). Wound healing activity of various fractions from an extract of Ehretia laevis Roxb. (Khandu Chakka) leaves in animal model. J. Adv. Sci. Res. 2021, 100–104.
He, L. L. (2018). The separation and purification of effective constituent in fruit of Ehretia macrophylla Wall, and the bioactivity study. South China Univ. Technol.
He, Z. D., and Liu, Y. Q. (1992). Glycosides from Ligustrum purpurascens. Acta Bot. Yunnanica 14, 328–336.
Hoang, T. L. H., Jang, D. C., Nguyen, Q. T., Na, W. H., Kim, I. S., and Vu, N. T. (2021). Biochar-improved growth and physiology of Ehretia asperula under water-deficit condition. Appl. Sci. 11, 10685. doi:10.3390/app112210685
Huma, Z.-e. (2019). Pharmacognostic evaluation of Ehretia serrata roxb. And Ehretia obtusifolia hocht. A. DC. Family Boraginaceae. Univ. Peshawar.
Ijaz, S., Iqbal, J., Abbasi, B. A., Ullah, Z., Yaseen, T., Kanwal, S., et al. (2023). Rosmarinic acid and its derivatives: current insights on anticancer potential and other biomedical applications. Biomed. and Pharmacother. 162, 114687. doi:10.1016/j.biopha.2023.114687
Iqbal, K., Nawaz, S. A., Malik, A., Riaz, N., Mukhtar, N., Mohammad, P., et al. (2005). Isolation and lipoxygenase-inhibition studies of phenolic constituents from Ehretia obtusifolia. Biomed. Pharmacother. 2, 104–111. doi:10.1002/cbdv.200490161
Jan, H. U., Saeed, A., Parveen, G., Mukhtar, N., Siraj, M., Sami, A., et al. (2023). Pharmacognostic study of Ehretia acuminata R. Br. Proceed. Pakistan Acad.Sci.: B. Life environm. Sci. 60, 267–272.
Joshi, U. P., and Wagh, R. D. (2019). Phytochemical screening and HPTLC fingerprinting profile of bark extracts of Ehretia laevis. Int. J. Pharm. Life Sci. 10, 6075–6080. doi:10.53560/PPASB(60-2)743
Jyothirmai, N., Nagaraju, B., and Kumar, J. S. (2016). Evaluation of anti-inflammatory and anti-bacterial activities of different solvent extracts of Ehretia laevis Roxb. J. Pharm. Sci. Res. 8, 715–720.
Kaur, A., Shukla, A., and Shukla, R. K. (2019). Comparative evaluationof ABTS, DPPH, FRAP, nitric oxide assays for antioxidant potential, phenolic and flavonoid content of Ehretia acuminata R. Br. bark. Int. Res. J. Pharm. 9, 100–104. doi:10.7897/2230-8407.0912301
Kaur, A., Shukla, A., and Shukla, R. K. (2022). In vitro antidiabetic and anti-inflammatory activities of the bark of Ehretia acuminata R. Br. Indian J. Nat. Prod. Res. 12, 538–543.
Khattab, A. M., Grace, M. H., and El-Khrisy, E. A. (2001). A new flavone derivative from Ehretia ovalifolia leaves. Die Pharm. 56, 661–662. doi:10.56042/ijnpr.v12i4.29108
Kudera, T., Fiserova, B., Korytakova, M., Doskocil, I., Salmonova, H., Tulin, E. E., et al. (2021). In vitro selective antibacterial and antiproliferative effects of ethanolic extracts from Cambodian and Philippine plants used in folk medicine for diarrhea treatment. Front. Pharmacol. 12, 746808. doi:10.3389/fphar.2021.746808
Le, T. T., Kang, T. K., Do, H. T., Nghiem, T. D., Lee, W. B., and Jung, S. H. (2021). Protection against oxidative stress-induced retinal cell death by compounds isolated from Ehretia asperula. Nat. Prod. Commun. 16, 1934578X211067986. doi:10.1177/1934578x211067986
Legaspi, C. L. B., and Bagaoisan, D.-M. A. (2020). Ehretia microphylla tablet formulation for biliary and gastrointestinal colic: a review of its phytochemical constituents, pharmacologic activities and clinical researches. Acta Med. Philipp. 54, 80–85. doi:10.47895/amp.v54i1.1108
Li, L., Peng, Y., Xu, L. J., Li, M. H., and Xiao, P. G. (2009). Flavonoid glycosides and phenolic acids from Ehretia thyrsiflora. Biochem. Syst. Ecol. 36, 915–918. doi:10.1016/j.bse.2008.11.008
Li, L., Peng, Y., Yao, X., Xu, L. J., Wulan, T. N., Liu, Y., et al. (2010a). Chemical constituents and biological activities of plants from the genus Ehretia Linn. Chin. Herb. Med. 2, 106–111.
Li, L., Shi, R. B., Xu, L. J., Peng, Y., and Xiao, P. G. (2010b). Chemical contituents in leaves of Ehretia thyrsiflora. China J. Chin. Mat. Med. 35, 331–332. doi:10.3969/j.issn.1674-6384.2010.02.002
Li, L., Xu, L. J., He, Z. D., Yang, Q. Q., Pen, Y., and Xiao, P. G. (2008). Chemical study on ethylacetate portion of Ehretia thyrsiflora, Boraginaceae species Kudingcha. China J. Chin. Mat. Med. 33, 2121–2123.
Lim, J. S., Lee, S. H., Yun, H., Lee, D. Y., Cho, N., Yoo, G., et al. (2023). Inhibitory effects of Ehretia tinifolia extract on the excessive oxidative and inflammatory responses in lipopolysaccharide-stimulated mouse kupffer cells. Antioxidants 12, 1792–1809. doi:10.3390/antiox12101792
Liu, R. H. (2003). Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 78, 517S-520S–520S. doi:10.1093/ajcn/78.3.517S
Lu, J. C. (2016). “Research on processes of Ehretia macrophylla wall,” in Fruit and structural identifications of separated phenolic compounds. South China Univ. Technol.
Mageswari, S., and Karpagam, S. (2015). Evaluation of antimicrobial studies on root of Carmona retusa (Vahl.) Masam. Int. J. Curr. Pharm. Res. 7, 60–63.
Mageswari, S., Karpagam, S., and Reddy, G. A. (2012). Evaluation of wound healing activity of the plant Carmona retusa (Vahl.) Masam., in mice. Int. J. Intell. Inf. Technol. 4, 1–4.
Mandal, G., and Joshi, S. P. (2014). Analysis of vegetation dynamics and phytodiversity from three dry deciduous forests of Doon Valley, Western Himalaya, India. J. Asia-Pacific Biodivers. 7, 292–304. doi:10.1016/j.japb.2014.07.006
Maramba-Lazarte, C. C., Rivera, P. T., and Villacorte, E. A. (2020). Philippine Tsaang Gubat (Ehretia microphylla Lam) and ampalaya (Momordica charantia L.) leaf extracts lack amoebicidal activity in vitro. Acta Med. Philipp. 54, 5–10. doi:10.47895/amp.v54i1.1081
Maroyi, A. (2023). A systematic review on biological and medicinal properties of Ehretia rigida (Thunb.) Druce (Ehretiaceae) in Southern Africa. Plant Sci. Today 10, 74–82.
Memon, F. S., Silawat, N., and Jain, N. K. (2022). Phytochemical and pharmacological screening of Ehretia Laevis extract for anti-inflammatory activity. J. Pharm. Neg. Res. 3, 4854–4859.
Mitsi, C., and Echeverría, J. (2024). The genus Haplopappus: botany, phytochemistry, traditional uses, and pharmacological properties. Front. Pharmacol. 15, 1490243. doi:10.47750/pnr.2022.13.S10.588
Mncwangi, N., Chen, W., Vermaak, I., Viljoen, A. M., and Gericke, N. (2012). Devil's Claw-A review of the ethnobotany, phytochemistry and biological activity of Harpagophytum procumbens. J. Ethnopharmacol. 143, 755–771. doi:10.1016/j.jep.2012.08.013
Monroy-García, I. N., Carranza-Torres, I. E., Carranza-Rosales, P., Oyón-Ardoiz, M., García-Estévez, I., Ayala-Zavala, J. F., et al. (2021). Phenolic profiles and biological activities of extracts from edible wild fruits Ehretia tinifolia and Sideroxylon lanuginosum. Foods 10, 2710–2725. doi:10.3390/foods10112710
Ogundajo, A., and Ashafa, A. T. (2017). Phytochemical compositions and in vitro assessments of antioxidant and antidiabetic potentials of fractions from Ehretia cymosa Thonn. Pharm. Mag. 13, S470-S480–S480. doi:10.4103/pm.pm_118_17
Ogundajo, A. L., Nnaemeka, C. O., Olawunmi, R. O., and Ogunwande, I. A. (2016). Chemical constituents of essential oil of Ehretia cymosa Thonn. Br. J. Appl. Sci. Technol. 14, 1–6. doi:10.9734/bjast/2016/24240
Panja, S., Choudhuri, I., Khanra, K., Pati, B., and Bhattacharyya, N. (2020). Biological and photocatalytic activity of silver nanoparticle synthesized from Ehretia laevis Roxb. leaves extract. Nano Biom. Eng. 12, 104–113. doi:10.5101/nbe.v12i1.p104-113
Petersen, M. (2013). Rosmarinic acid: new aspects. Phytochem. Rev. 12, 207–227. doi:10.1007/s11101-013-9282-8
Petersen, M., and Simmonds, M. S. (2003). Rosmarinic acid. Phytochemistry 62, 121–125. doi:10.1016/s0031-9422(02)00513-7
Pío-León, J. F., Díaz-Camacho, S. P., López, M. G., Montes-Avila, J., López-Angulo, G., and Delgado-Vargas, F. (2012a). Características fisicoquímicas, nutricias y antioxidantes del fruto de Ehretia tinifolia. Rev. Mex. Biodivers. 83, 273–280. doi:10.22201/ib.20078706e.2012.1.1148
Pío-León, J. F., Díaz-Camacho, S. P., López, M. G., Montes-Avila, J., López-Angulo, G., and Delgado-Vargas, F. (2012b). Physicochemical, nutritional, and antioxidant characteristics of the fruit of Ehretia tinifolia. Rev. Mex. Biodivers. 83, 273–280. doi:10.22201/ib.20078706e.2012.1.1148
Rangnathrao, T. S., and Shanmugasundaram, P. (2019). Antioxidant and hepatoprotective activity of Ehretia laevis Roxb against paracetamol induced acute hepatotoxicityin wistar rats. Res. J. Pharm. Technol. 12, 6143–6148. doi:10.5958/0974-360x.2019.01067.9
Retief, E., and Van Wyk, A. E. (2001). The genus Ehretia (boraginaceae: ehretioideae) in southern Africa. Bothalia-african biodiv. Conserv. 31, 9–23. doi:10.4102/abc.v31i1.494
Sarkodie, J., Squire, S., Kretchy, I., Domozoro, C., Ahiagbe, K., Twumasi, M., et al. (2015). The antihyperglycemic, antioxidant and antimicrobial activities of Ehretia cymosa. J. Pharm. Phytochem. 4, 105–111.
Selvanayagam, Z. E., Gnanavendhan, S. G., Balakrishna, K., Rao, R. B., Sivaraman, J., Subramanian, K., et al. (1996). Ehretianone, a novel quinonoid xanthene from Ehretia buxifolia with antisnake venom activity. J. Nat. Prod. 59, 664–667. doi:10.1021/np960355p
Sharma, P., Shri, R., and Kumar, S. (2022). Phytochemical and in vitro cytotoxic screening of chloroform extract of Ehretia microphylla lamk. Stresses 2, 384–394. doi:10.3390/stresses2040027
Sharma, P., Shri, R., Ntie-Kang, F., and Kumar, S. (2021). Phytochemical and ethnopharmacological perspectives of Ehretia laevis. Molecules 26, 3489–3517. doi:10.3390/molecules26123489
Shukla, A., and Kaur, A. (2018). A systematic review of traditional uses bioactive phytoconstituents of genus Ehretia. Asian J. Pharm. Clin. Res. 11, 88–100. doi:10.22159/ajpcr.2018.v11i6.25178
Shukla, A., Kaur, A., Shukla, R., and Anchal, (2019a). A comparative study of in vitro antioxidant potential, photoprotective screening of Ehretia acuminata R. Br. leaves. Indian Drugs 56, 30–36. doi:10.53879/id.56.09.11593
Shukla, A., Kaur, A., and Shukla, R. K. (2021). Evaluation of different biological activities of leaves of Ehretia acuminata R. Br. Indian Drugs 58, 42–49. doi:10.53879/id.58.04.12201
Shukla, A., Kaur, A., Shukla, R. K., and Anchal, (2019b). Comparative evaluation of antioxidant capacity, total flavonoid and phenolic content of Ehretia acuminata R. Br. fruit. Res. J. Pharm. Technol. 12, 1811–1816. doi:10.5958/0974-360x.2019.00302.0
Simpol, L. R., Otsuka, H., Ohtani, K., Kasai, R., and Yamasaki, K. (1994). Nitrile glucosides and rosmarinic acid, the histamine inhibitor from Ehretia philippinensis. Phytochemistry 36, 91–95. doi:10.1016/s0031-9422(00)97019-5
Steyn, T. (1998). The chemical constituents of Ehretia rigida, Apodytes dimidiata and Ocotea kenyensis. Citeseer.
Suri, O. P., Jamwal, R. S., Suri, K. A., and Atal, C. K. (1980). Ehretinine, a novel pyrrolizidine alkaloid from Ehretia aspera. Phytochemistry 19, 1273–1274. doi:10.1016/0031-9422(80)83115-3
Thakre, R., Bhutada, S., Chouragade, B., Khobragade, P., and Ketaki, H. (2016). Unexplored wound healing property of Ehtretia laevis Roxb. (Khandu Chakka) plant. Int. J. Res. Ayurveda Pharm. 7, 54–57. doi:10.7897/2277-4343.075219
Torane, R. C., Kamble, G. S., Kale, A. A., Gadkari, T. V., and Deshpande, N. R. (2011). Quantification of dioctyl phthalate from Ehretia laevis Roxb by HPTLC. J. Chem. Pharm. Res. 3, 48–51.
Torane, R. C., Ruikar, A. D., Chandrachood, P. S., and Deshpande, N. R. (2009). Study of amino acids and carbohydrates from the leaves of Ehretia laevis. Asian J. Chem. 21, 1636–1638.
Velappan, S., and Thangaraj, P. (2014). Phytochemical constituents and antiarthritic activity of Ehretia laevis Roxb. J. Food Biochem. 38, 433–443. doi:10.1111/jfbc.12071
Waheed, A., Chohan, M. M., Ahmed, D., and Ullah, N. (2019). The first report on the in vitro antimicrobial activities of extracts of leaves of Ehretia serrata. Saudi J. Biol. Sci. 26, 1253–1261. doi:10.1016/j.sjbs.2018.05.025
Wang, Y. C., and Huang, T. L. (2005). Screening of anti-Helicobacter pylori herbs deriving from Taiwanese folk medicinal plants. FEMS Immunol. Med. Microbiol. 43, 295–300. doi:10.1016/j.femsim.2004.09.008
Xu, X. G., Cheng, Y., Tong, L. L., Tian, L., and Xia, C. L. (2022). The complete chloroplast genome sequence of Ehretia dicksonii Hance (Ehretiaceae). Mitochondrial DNA. B 7, 661–662. doi:10.1080/23802359.2022.2061873
Yamamura, S., Simpol, L. R., Ozawa, K., Ohtani, K., Otsuka, H., Kasai, R., et al. (1995). Antiallergic dimeric prenylbenzoquinones from Ehretia microphylla. Phytochemistry 39, 105–110. doi:10.1016/0031-9422(94)00845-k
Yende, S. R., Shah, S. K., Arora, S. K., Moharir, K. S., and Lohiya, G. K. (2021). In silico prediction of phytoconstituents from Ehretia laevis targeting TNF-α in arthritis. Dig. Chin. Med. 4, 180–190. doi:10.1016/j.dcmed.2021.09.003
Yoshikawa, K., Kageyama, H., and Arihara, S. (1995). Phenolic glucosides and lignans from Ehretia ovalifolia. Phytochemistry 39, 659–664. doi:10.1016/0031-9422(94)00931-i
Yuvaraja, K. R., Santhiagu, A., Jasemine, S., and Gopalasatheeskumar, K. (2021). Hepatoprotective activity of Ehretia microphylla on paracetamol induced liver toxic rats. J. Res. Pharm. 25, 1–98. doi:10.35333/jrp.2021.286
Zara, S., Ahmed, D., Baig, H., and Ikram, M. (2012). Evaluation of antioxidant activities of various solvent extracts of fruits and leaves of Ehretia serrata. Asian J. Chem. 24, 4345–4351.
Zeng, M. Y., and Zeng, J. F. (1994). China traditional Chinese medicine resources Zhi Yao. Beijing: Science Press, 1034–1035.
Keywords: Ehretia genus, traditional uses, phytochemistry, pharmacology, clinical studies
Citation: Jiang S, Wang M, Kaur A, Jiang L, Cai Y, Luo J, Li M, Wang H, Wan D and Peng Y (2025) Ehretia genus: a comprehensive review of its botany, ethnomedicinal values, phytochemistry, pharmacology, toxicology and clinical studies. Front. Pharmacol. 16:1526359. doi: 10.3389/fphar.2025.1526359
Received: 11 November 2024; Accepted: 03 February 2025;
Published: 21 February 2025.
Edited by:
Rajeev K. Singla, West China Hospital, Sichuan University, ChinaReviewed by:
Smith B. Babiaka, University of Tuebingen, GermanyU.G. Chandrika, University of Sri Jayewardenepura, Sri Lanka
Jitendra Pandey, University of Hawaii at Manoa, United States
Copyright © 2025 Jiang, Wang, Kaur, Jiang, Cai, Luo, Li, Wang, Wan and Peng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dan Wan, MzUwMjMyMjUyQHFxLmNvbQ==; Yanmei Peng, MjcxODUzMTQ1QHFxLmNvbQ==
†These authors have contributed equally to this work
 Sai Jiang
Sai Jiang Mengyun Wang2†
Mengyun Wang2† Amanpreet Kaur
Amanpreet Kaur Lin Jiang
Lin Jiang Yuan Cai
Yuan Cai