
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Pharmacol. , 24 March 2025
Sec. Experimental Pharmacology and Drug Discovery
Volume 16 - 2025 | https://doi.org/10.3389/fphar.2025.1504245
 Yinchao Ma1†
Yinchao Ma1† Ge Wei2†
Ge Wei2† Zhichen Dong2†
Zhichen Dong2† Ziyuan Wang2
Ziyuan Wang2 Xinlong Zhai2
Xinlong Zhai2 Yuan Liu2
Yuan Liu2 Huan Chen1
Huan Chen1 Yaning Fu1
Yaning Fu1 Hongwei Hou1*
Hongwei Hou1* Qingyuan Hu1*
Qingyuan Hu1* Ming Chu1,2*
Ming Chu1,2*Solanesol, identified as Nonaprenol alcohol, predominates in the Solanaceae family. This compound exists as a white to pale yellow solid at room temperature, characterized by low polarity and water insolubility. Its unique chemical structure—featuring nine non-conjugated double bonds and low polarity—confers remarkable biological activities. Recent studies have demonstrated that solanesol exhibits polypharmacological properties, including antimicrobial, antioxidant, anti-inflammatory, and membrane-stabilizing effects. Mechanistically, solanesol suppresses ROS generation and inhibits pro-inflammatory cytokines (IL-1β, TNF-α). Preclinical studies highlight its therapeutic potential in inflammatory disorders (periodontitis, neuropathic pain) and neurodegenerative diseases (Alzheimer’s, Parkinson’s). However, current research still faces critical bottlenecks, such as a lack of in vivo pharmacokinetic data, unclear molecular targets, and insufficient toxicity assessments. Future studies urgently need to integrate experimental approaches, including target screening, nanotechnology-based delivery systems, and multi-omics analysis, to elucidate its mechanisms of action and promote clinical translation. As a compound that combines natural safety with multi-target effects, solanesol is not only a research focus for the development of novel drugs but also a bridge connecting natural products and precision medicine, poised to lead the innovation of next-generation biocompatible therapies.
Solanesol, also known as Nonaprenol alcohol (C45H74O), is a long-chain unsaturated fatty alcohol belonging to the triterpenoid class (Yan et al., 2019) (Figure 1). Solanesol is primarily found naturally in plants of the Solanaceae family, including tobacco, potatoes, tomatoes, eggplants, and peppers. Among these, tobacco contains the highest concentration of solanesol, reaching up to 4.13% of the tobacco’s dry weight (Xiang et al., 2017). The compound was first isolated from tobacco by Rowland and colleagues in 1956 (Rowland et al., 1956). In the early 1980s, Japan achieved the initial successful extraction of solanesol from tobacco leaves. Since the 1990s, China has been extracting solanesol from waste tobacco extracts using methods such as supercritical fluid extraction, ultrasonic-assisted extraction, microwave-assisted extraction, and solvent extraction. The extraction technologies of solanesol have garnered significant attention due to their critical applications in pharmaceutical and industrial fields. Current mainstream methods include: ammonia leaching pretreatment-assisted extraction, dynamic saponification extraction, ultrasonic-assisted extraction, molecular distillation, supercritical CO2 fluid extraction, and bio-enzymatic extraction (Yan et al., 2019). Additionally, molecularly imprinted polymers (MIPs) have emerged as a novel purification strategy, enabling selective separation through specific molecular recognition (Long et al., 2015; Ma et al., 2016; Duan et al., 2019). These methods exhibit distinct advantages in efficiency, environmental friendliness, and cost-effectiveness.
Solanesol appears as a white or faintly yellow solid at room temperature, has low polarity, is insoluble in water, and is soluble in hexane and ethanol (Liu et al., 2007). The structure of solanesol, composed of nine isoprene units including nine non-conjugated double bonds, gives it strong biological activity and makes it prone to reactions such as oxidation, addition, and dehydrogenation (Yan et al., 2015). Current research has found that solanesol, as an oil-in-water (O/W) emulsion, has good stability and exhibits comparable physicochemical stability to squalene under storage conditions of 5°C and 25°C. At storage conditions of 40°C, the physical stability of the solanesol emulsion is also good (Fisher et al., 2023). Currently, solanesol is primarily utilized in synthesizing compounds containing the isoprenoid structure, such as coenzyme Q10, vitamin K2, and vitamin E, establishing its indispensable role as an intermediate in ubiquinone-type medications. Recent studies have identified that solanesol can also act as an “active carrier” in the development of novel nano-drug delivery systems for synthesizing anticancer drugs (Gendron et al., 2023). Notably, solanesol inherently exhibits antibacterial, antioxidant, anti-inflammatory, and membrane-stabilizing properties (Table 1) (Zhang et al., 2024), and demonstrates promising therapeutic effects in animal models of inflammatory diseases and central nervous system (CNS) diseases. The exploration of bioactivities and pharmacological effects of natural products derived from plants has recently gained significant momentum. For example, the study by Ullah et al. revealed that nanocomposites based on natural products can exhibit robust antibacterial and anti-biofilm activities through synergistic molecular interactions (Ullah et al., 2024). Therefore, this review summarizes the research progress of solanesol’s polypharmacological actions, aiming to identify critical research gaps and provide a theoretical foundation for further exploration of its medicinal value.
Currently, there is also literature that proves solanesol has antimicrobial effects. In 2007, Chen and colleagues utilized agar diffusion and twofold dilution methods to assess the antimicrobial activity of solanesol against a variety of standard bacterial strains (including Staphylococcus aureus, Bacillus cereus, Escherichia coli, and Pseudomonas aeruginosa) as well as clinically isolated strains (such as Staphylococcus aureus, Bacillus subtilis, Mycobacterium phlei, Escherichia coli, and Pseudomonas aeruginosa). The findings revealed that solanesol exhibited significant inhibitory effects against both Gram-negative bacteria (E. coli and P. aeruginosa) and Gram-positive bacteria (S. aureus and M. phlei), albeit showing weaker activity against B. subtilis and B. cereus (Chen et al., 2007). In 2008, Guo et al. utilized three extraction methods—supercritical extraction, Soxhlet extraction, and ultrasonic extraction—to isolate solanesol from tobacco leaves. The study found that solanesol, irrespective of the extraction method used, demonstrated the strongest antibacterial effect against S. aureus, followed by B. subtilis, with the least effectiveness observed against E. coli (Guo et al., 2008).
Solanesol, containing nine non-conjugated double bonds, has the capability to absorb free radicals (Qi et al., 2005). In 2008, Huang and colleagues utilized supercritical carbon dioxide with ethanol as a co-solvent to extract solanesol from tobacco, discovering that the antioxidant activity of the extracts was closely correlated with the yield of solanesol, with a determination coefficient of 0.946 (Huang et al., 2008). Further research by Ma and others revealed that solanesol possesses strong in vitro antioxidant activity, with its capacity to scavenge superoxide anions and hydroxyl radicals being comparable to that of the water-soluble vitamin E (Trolox), and it can inhibit microsomal lipid peroxidation (Ma et al., 2011). In 2014, Bai and team demonstrated that solanesol could effectively absorb ultraviolet radiation, scavenge lipid radicals, and inhibit tyrosinase activity, suggesting its potential in preventing skin aging and the formation of age spots (Bai et al., 2014). In 2015, Yao and colleagues found in an alcohol-induced L02 hepatocyte oxidative damage model that solanesol could disrupt the heat shock protein (Hsp) 90-heat shock factor 1 (HSF1) complex, promote the binding of Hsp90 to Keap1 (Kelch-like ECH-associated protein 1), thereby releasing nuclear factor-erythroid 2 related factor 2 (Nrf2). Nrf2 then enters the nucleus and binds to the antioxidant response element (ARE), initiating the transcription of downstream genes such as H O -1, GCLC and GCLM (Yao et al., 2015) (Figure 2). HO-1, a rate-limiting enzyme in heme catabolism, breaks down heme into biliverdin, iron, and carbon monoxide, exhibiting antioxidant effects (Mansouri et al., 2022). The enzyme complex formed by GCLC and GCLM, known as glutamate cysteine ligase (GCL), is the rate-limiting enzyme in the biosynthesis of glutathione (GSH), affecting intracellular GSH levels (Liu et al., 2022). GSH protects cells from oxidative stress induced by high levels of reactive oxygen species (ROS), maintaining the cellular redox state. Additionally, Solanesol disrupts the Hsp90-HSF1 complex, releasing HSF1 which then enters the nucleus and binds to the heat shock element (HSE). This initiates the transcription of downstream HSP70 genes, further activating the Keap1/Nrf2/ARE pathway and enhancing antioxidant effects (Yao et al., 2015). In 2023, Liu and team studied a high-glucose-induced L02 hepatocyte oxidative damage model and found that solanesol could decrease the expression of Keap1 and promote the translocation of Nrf2 to the nucleus, activating the Keap1/Nrf2/ARE pathway. This activation increases the expression of downstream HO1 and NQO1, inhibits the expression and secretion of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and lactate dehydrogenase (LDH), and improves the activities of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px). Thus, it balances intracellular ROS levels, inhibits lipid peroxidation of biomembranes, restores mitochondrial membrane potential, and protects L-02 cells under high-glucose conditions (Liu et al., 2023) (Figure 2).
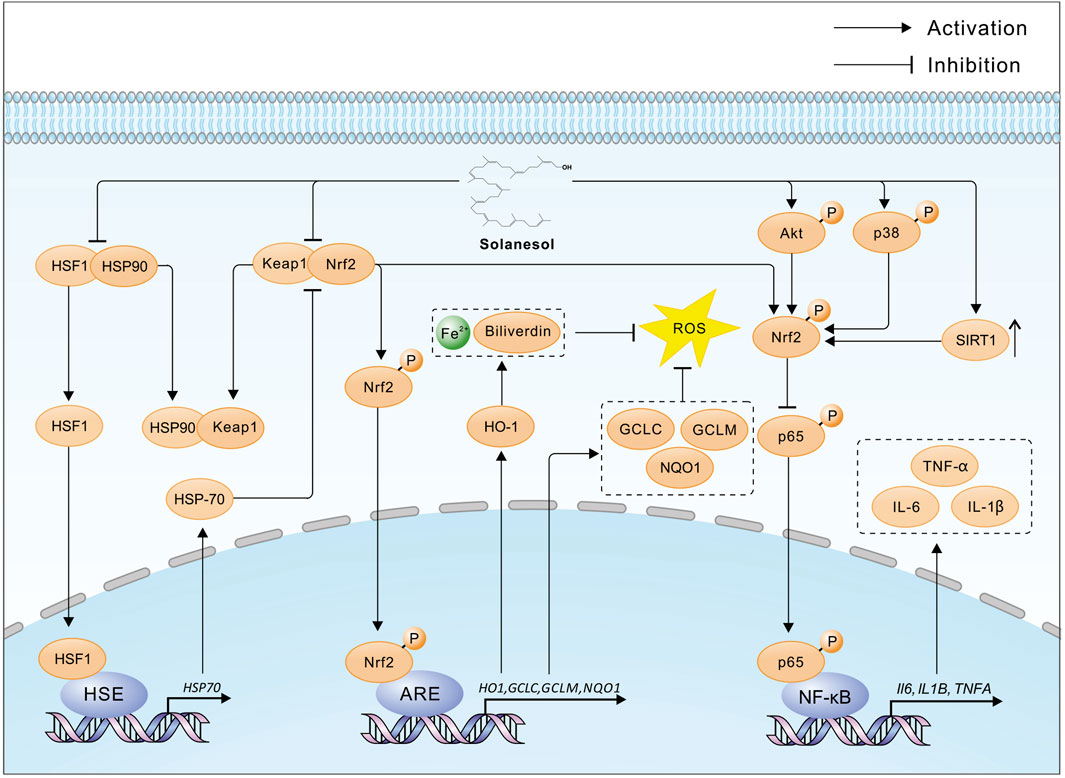
Figure 2. The multiple pharmacological mechanisms of solanesol. HSF1, Heat shock factor protein 1; HSP90, Heat shock protein 90; Keap1, Kelch-like ECH-associated protein 1; Nrf2, Nuclear factor erythroid 2-related factor 2; HSE, Heat shock element; ARE, Antioxidant response element; HO-1, Heme oxygenase 1; GCLC, Glutamate--cysteine ligase catalytic subunit; GCLM, Glutamate--cysteine ligase regulatory subunit; NQO1, NAD(P)H dehydrogenase [quinone] 1; ROS, Reactive oxygen species; HSP70, Heat shock protein 70; Akt, RAC-alpha serine/threonine-protein kinase; p38, Mitogen-activated protein kinase 14; SIRT1, Sirtuin 1; p65, RELA proto-oncogene, NF-kB subunit; IL-1β, Interleukin 1 beta; IL-6, Interleukin 6; TNF-α, tumor necrosis factor alpha; Solanesol disrupts the HSF1-HSP90 and Keap1-Nrf2 complexes, facilitating the liberation and nuclear translocation of Nrf2, which in turn initiates the transcription of ARE-mediated genes such as HO1, GCLC, GCLM, and NQO1. The enzymatic action of HO-1 results in an increased cytosolic concentration of Fe2+ and biliverdin, which suppresses the generation of ROS. Additionally, HSF1 promotes the transcription of HSP70, further enhancing the release of Nrf2 and bolstering the antioxidative defense. Furthermore, solanesol activates Nrf2 through the phosphorylation of Akt and p38 proteins, along with upregulating the expression of SIRT1, thereby inhibiting the phosphorylation of the p65 protein and subsequent transcription of pro-inflammatory cytokines such as IL6, IL1B, and TNFA, exerting an anti-inflammatory effect.
The anti-inflammatory effects of solanesol were further explored by Yao et al., in 2017 within an LPS-stimulated macrophage model (RAW264.7 cells). The study illustrated that solanesol activates Nrf2 through phosphorylation of p38 and Akt, promoting Nrf2’s nuclear translocation and the initiation of HO1 transcription. This process subsequently inhibited the secretion levels of IL-1β, IL-6, and TNF-α in LPS-stimulated RAW264.7 cells (Yao et al., 2017; Campbell et al., 2021) (Figure 2). Additionally, solanesol was shown to induce the expression of microtubule-associated protein 1 light chain 3B-II (LC3B-II) in a dose- and time-dependent manner, exerting its anti-inflammatory effect through the induction of autophagy. In the same year, Sridevi and colleagues demonstrated the significant in vivo anti-inflammatory effects of solanesol by preparing a solanesol gel and testing it in a rat paw edema experiment (Sridevi et al., 2017).
In 2018, research by Eriksson et al. revealed that solanesol could mimic components of the inner mitochondrial membrane (IMM), integrating into liposomes to decrease the permeability of IMM to small hydrophilic solutes without affecting lipid stacking within the IMM (Eriksson et al., 2018). Another study further indicated that solanesol enhances the membrane’s resistance to deformation, thereby improving E. coli’s osmotic pressure resistance (Eriksson et al., 2019).
In recent years, there has been a growing amount of research on solanesol in animal models of inflammatory diseases, such as periodontitis, chronic inflammatory pain, and anxiety induced by inflammation (Figure 3). In 2018, Zhang and colleagues conducted a study on the therapeutic effects of solanesol at three different concentrations—15 mg/kg, 30 mg/kg, and 60 mg/kg—on a rat model of experimental periodontitis (Zhang et al., 2018; Shang et al., 2023; Sczepanik et al., 2020). The research found that all three doses of solanesol significantly reduced the levels of IL-1β, TNF-α, and prostaglandin E2 (PGE2) in rat plasma. Additionally, solanesol increased the activity of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px), decreased malondialdehyde (MDA) content, improved systemic oxidative stress, alleviated alveolar bone loss, and mitigated periodontal inflammation. Intraplantar injection of complete Freund’s adjuvant (CFA) is a classic mouse model of anxiety-like behavior induced by neuroinflammation (Lazarević et al., 2024). Previously, a study found that a 50 mg/kg dose of solanesol could reverse mechanical allodynia and thermal hypersensitivity induced by CFA injection, alleviate anxiety-like syndromes associated with chronic inflammatory pain, and significantly reduce the levels of the inflammatory factors TNF-α and IL-1β in the mouse spinal cord. Immunohistochemical staining results further showed that the activation of microglia and astrocytes induced by CFA injection was significantly inhibited (Wang et al., 2024). After treating mice with 50 mg/kg solanesol for 1 week in a CFA-induced neuroinflammation mouse model, Ding et al. found a significant improvement in mouse anxiety behavior, reduced activation of microglia and astrocytes in the anterior cingulate cortex, downregulated TIA1, thereby lowering the levels of pro-inflammatory cytokines IL-1β and TNF-α. These studies reveal the potential of solanesol in treating inflammatory diseases, warranting further exploration of the mechanisms by which solanesol acts in inflammatory conditions, and providing direction for future pharmacological research on solanesol (Ding et al., 2024).
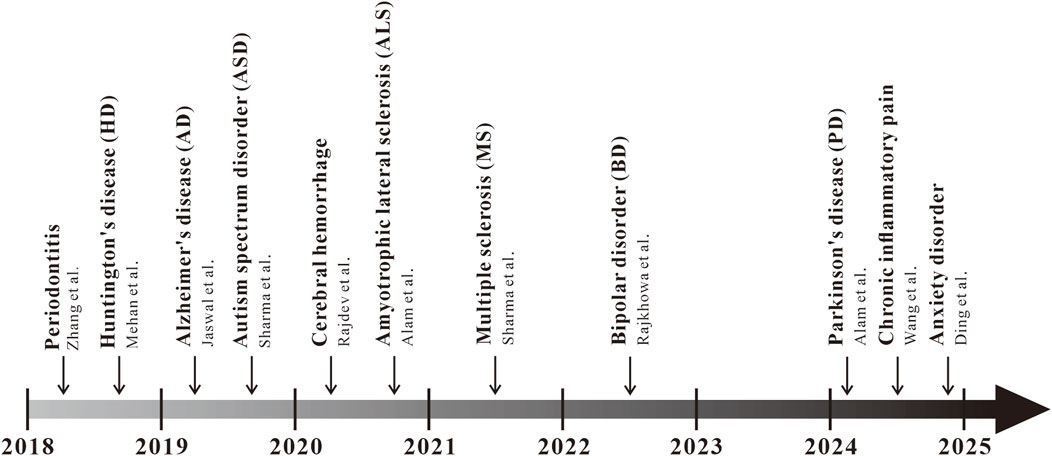
Figure 3. Progress of the pharmacological action of solanesol in diseases Starting from 2018, a multitude of studies have gradually focused on the pharmacological action of solanesol in diseases, such as periodontitis, Huntington’s disease (HD), Alzheimer’s disease (AD), autism spectrum disorder (ASD), cerebral hemorrhage, amyotrophic lateral sclerosis (ALS), multiple sclerosis (MS), bipolar disorder (BD), chronic inflammatory pain, and anxiety disorders.
Over the past decade, a substantial body of research has focused on the neuroprotective effects of solanesol, demonstrating promising results in various animal disease models (Figure 3). In 2018, Mehan and colleagues explored solanesol’s neuroprotective effects against Huntington’s disease (HD), suggesting that solanesol may slow disease progression by improving mitochondrial dysfunction and reducing oxidative stress-induced neuronal damage (Jiang et al., 2023; Mehan et al., 2018a). Further research by Mehan and colleagues demonstrated that solanesol at doses of 5, 10, and 15 mg/kg could mitigate HD-like symptoms in rats, including memory impairments, reduced grip strength, postural abnormalities, and cognitive deficits induced by 3-nitropropionic acid, by activating coenzyme Q10 to repair mitochondrial damage. Significant improvements were observed in the histological alterations of the hippocampus, basal ganglia, and cerebral cortex in rats, along with alleviation of inflammation and oxidative damage in rat brain tissues (Mehan et al., 2018b). In 2019, Jaswal et al. created an Alzheimer’s disease (AD)-like rat model using intracerebroventricular injections of streptozotocin (Jaswal et al., 2019). They discovered that a 21-day combined treatment with solanesol (10 mg/kg) and epigallocatechin gallate (20 mg/kg) effectively reduced Ca2+ overload, lower levels of lipid peroxidation, superoxide dismutase (SOD), glutathione (GSH), and cytochrome C by closing the mitochondrial permeability transition pore, significantly improving the rats’ memory and learning capabilities. In the same year, Sharma from Mehan’s team utilized an intracerebroventricular injection of propionic acid to create an autism rat model, investigating solanesol’s neuroprotective effects (Hirota and King, 2023; Sharma et al., 2019). The study found that solanesol alone at doses of 40 and 60 mg/kg or in combination with drugs like aripiprazole, risperidone, clozapine, and donepezil could restore mitochondrial respiratory chain complex enzyme activities (complexes I, II, and V) and coenzyme Q10 activity, increase levels of neurotransmitters dopamine, acetylcholine, and glutamate, reduce levels of inflammation markers TNF-α and IL-1β, decrease oxidative stress markers acetylcholinesterase (AchE), GSH, and LDH, and improve cognitive deficits and biochemical markers in rats. Long-term treatment with solanesol and these drugs significantly improved long-term memory, depressive behavior, and muscle coordination in autistic rats, demonstrating its neuroprotective effects against autism.
In 2020, Alam from Mehan’s team investigated the therapeutic effects of solanesol alone at doses of 15 and 30 mg/kg and in combination with riluzole in a methylmercury-induced amyotrophic lateral sclerosis (ALS) rat model (Alam et al., 2020). The study found that long-term treatment elevated adenylate cyclase and mitochondrial coenzyme Q10 levels, inhibited inflammation and oxidative stress, reversed alterations in neurochemical substances, and improved grip strength and cognitive function in rats. Furthermore, Rajdev from Mehan’s team induced cerebral hemorrhage in rats by injecting autologous blood into the brain and found that long-term treatment with solanesol alone at doses of 40 and 60 mg/kg or in combination with drugs like donepezil, memantine, celecoxib, and pregabalin restored mitochondrial respiratory chain complex enzyme activities (complexes I, II, and V) and coenzyme Q10 levels in brain mitochondria (Sheth, 2022; Rajdev et al., 2020). This treatment improved levels of dopamine, glutamate, gamma-aminobutyric acid (GABA), and acetylcholine (Ach), reduced acetylcholinesterase (AchE) levels, suppressed the expression of neuroinflammatory cytokines (TNF-α, IL-6, IL-1β), and improved brain oxidative stress markers such as nitrate concentration, GSH, and SOD. It significantly ameliorated neuronal mitochondrial damage-associated post-hemorrhagic behavioral and neurochemical dysfunctions. In 2021, the team led by Mehan, with Sharma et al., investigated the effects of long-term treatment with solanesol at doses of 40 mg/kg and 80 mg/kg in a rat model of multiple sclerosis (MS) induced by ethidium bromide (Charabati et al., 2023; Sharma et al., 2021). Long-term treatment with both doses of solanesol was shown to upregulate the levels of SIRT-1 and myelin basic protein in rat brain tissues, restore the activities of mitochondrial respiratory chain complexes (complexes I, II, and V), improve neurotransmitter levels, inhibit the production of inflammatory cytokines TNF-α and IL-1β, significantly reduce oxidative stress levels, regulate apoptosis-related factors such as caspase-3, Bax, and Bcl-2, decrease the volume of brain demyelination, repair the pathological state of the rat brain, and improve motor functions, grip strength, memory, and cognitive abilities in rats, demonstrating significant neuroprotective effects. In 2022, Rajkhowa and colleagues conducted research on rats induced with bipolar disorder (BD)-like symptoms using ouabain (OUA) (Nierenberg et al., 2023; Rajkhowa et al., 2022). They discovered that solanesol, at doses of 40 mg/kg and 80 mg/kg, could increase the levels of SIRT-1 in cerebrospinal fluid, brain homogenate, and plasma samples. This increase led to improvements in the levels of apoptosis markers—caspase-3, Bax, and Bcl-2—restoration of mitochondrial respiratory chain complex enzymes (complexes I, II, IV, and V), reduction in the expression of inflammatory cytokines TNF-α and IL-1β, elevation of serotonin and acetylcholine levels, decrease in dopamine and glutamate levels, reduction of oxidative stress, and restoration of altered Na/K ATPase levels. Additionally, the combination of solanesol at a dose of 80 mg/kg with lithium significantly reduced brain neuroglial cell proliferation, restored neuronal cell structures, decreased cell apoptosis, and alleviated manic-like and depressive-like behaviors in BD model rats.
In 2024, research has found that in a zebrafish Parkinson’s disease (PD) model induced by Tramadol, motor dysfunction and motor coordination in the zebrafish PD model were reversed after treatment with solanesol alone at doses of 25, 50, and 100 mg/kg, as well as in combination with standard drugs (Morris et al., 2024; Alam et al., 2024). Following treatment with solanesol, the levels of dopamine and norepinephrine in the brains of zebrafish were restored, serotonin levels were increased, GABA levels were improved, the neurotransmitter system was repaired, levels of TNF-α and IL-1β were reduced, and levels of IL-10 were increased. Additionally, solanesol significantly reduced the levels of oxidative stress induced by Tramadol, improved lipid peroxidation, decreased the concentration of malondialdehyde (MDA), and restored the levels of reduced glutathione (GSH) and superoxide dismutase (SOD). Histopathological results showed that after treatment with solanesol at various concentrations, neuronal cell damage and neuronal cell density were both improved in a dose-dependent manner.
Solanesol, a long-chain unsaturated fatty alcohol composed of nine isoprene units with nine non-conjugated double bonds, exhibits unique chemical properties that underpin its diverse bioactivities. Its low polarity and insolubility in water, coupled with solubility in organic solvents like hexane and ethanol, facilitate its integration into lipid membranes and nanocarrier systems. The extended hydrophobic chain and conjugated double-bond system contribute to its strong antioxidant capacity by scavenging free radicals and stabilizing lipid peroxidation. Furthermore, the structural flexibility of solanesol enables interactions with key molecular targets. Despite significant progress in elucidating solanesol’s bioactivities, current research gaps remain. The precise molecular targets and downstream signaling pathways of solanesol in various disease models remain incompletely characterized, necessitating advanced techniques such as artificial intelligence (AI) and machine learning-based target prediction, activity-based protein profiling, or molecular docking to identify binding partners. Additionally, the structure-activity relationship (SAR) of solanesol, particularly how specific structural motifs (e.g., double-bond configuration, chain length) influence its pharmacological efficacy, requires systematic exploration.
Future research should prioritize translating preclinical findings into clinical applications. While animal studies demonstrate solanesol’s therapeutic potential in neurodegenerative and inflammatory diseases, rigorous pharmacokinetic studies and human trials are essential to validate its safety and efficacy. Emerging applications in nanotechnology also hold promise. Solanesol’s amphiphilic nature and compatibility with lipid-based systems position it as an ideal candidate for advanced drug delivery platforms, including liposomes or polymeric nanoparticles, to improve drug solubility and targeted delivery. Collaborative efforts integrating computational biology, structural biology, and synthetic chemistry will be pivotal in unlocking solanesol’s full medicinal potential. By addressing these challenges and opportunities, solanesol could emerge as a cornerstone in the development of next-generation therapeutics for complex diseases, bridging the gap between natural product discovery and clinical innovation.
So far, solanesol has been shown to disrupt the HSF1-HSP90 and Keap1-Nrf2 complexes, phosphorylate Akt and p38, and thereby facilitate the release and nuclear translocation of Nrf2. This activation initiates the transcription of downstream genes such as HO1, increasing the cytoplasmic levels of Fe2+ and biliverdin, and inhibiting the production of ROS. Additionally, solanesol enhances the activity of mitochondrial respiratory chain complexes and improves neurotransmitter levels, inhibiting the secretion of inflammatory cytokines. Solanesol, apart from exhibiting antibacterial, antioxidant, anti-inflammatory, and membrane-stabilizing effects, has also shown promising therapeutic potential in animal models of inflammatory diseases and central nervous system disorders. Therefore, as a natural product, solanesol is worth further exploration. Although significant progress has been made in the pharmacological research of solanesol, there are still shortcomings. Current studies are limited to the pharmacological effects of solanesol and some superficial mechanistic explorations, without clarifying the specific targets of solanesol’s action. The exploration of signaling pathways is also not comprehensive or in-depth. Moreover, existing animal experiments have only explored the role of solanesol in central nervous system diseases, with only partial validation in cell experiments for other diseases. Therefore, the purpose of this review is to summarize the latest research progress of solanesol, providing ideas for subsequent research on the pharmacological effects of solanesol.
This review systematically compiled studies on solanesol through searches in major databases, including NCBI (National Center for Biotechnology Information, https://pubmed.ncbi.nlm.nih.gov), CNKI (China National Knowledge Infrastructure, https://www.cnki.net/), and Elsevier (https://www.elsevier.cn/), among others. The search terms included “solanesol,” “nonaprenol alcohol,” “antimicrobial,” “antioxidant,” “anti-inflammatory,” “neuroprotective,” and “pharmacological mechanisms.” Inclusion criteria encompassed peer-reviewed articles in English or Chinese focusing on solanesol’s bioactivity, pharmacological effects, or mechanisms. Exclusion criteria included non-original research (e.g., reviews, conference abstracts) and studies without experimental validation.
YM: Investigation, Writing–original draft, Writing–review and editing. GW: Investigation, Writing–original draft, Writing–review and editing. ZD: Investigation, Writing–review and editing. ZW: Investigation, Writing–review and editing. XZ: Writing–review and editing. YL: Writing–review and editing. HC: Writing–review and editing. YF: Writing–review and editing. HH: Project administration, Writing–review and editing. QH: Project administration, Writing–review and editing. MC: Project administration, Writing–original draft, Writing–review and editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by Beijing Life Science Academy (BLSA), No: 2024500CB0030, 2023000CA0040]; the National Natural Science Foundation of China [grant number 81603119]; the Natural Science Foundation of Beijing Municipality [grant number 7174316]; the Peking University Medicine Seed Fund for Interdisciplinary Research supported by “the Fundamental Research Funds for the Central Universities” [grant number No. BMU2022MX017, No. BMU2022MX003].
We would like to thank for the Significant Science and Technology Project of Beijing Life Science Academy, National Natural Science Foundation of China, Natural Science Foundation of Beijing Municipality and Peking University Medicine Seed Fund for Interdisciplinary Research supported by “the Fundamental Research Funds for the Central Universities financial support.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Alam, M., Minz, E., Yadav, R., and Mehan, S. (2020). Neuroprotective potential of adenyl cyclase/cAMP/CREB and mitochondrial CoQ10 activator in amyotrophic lateral sclerosis rats. Curr. Bioact. Compd. 17 (5), e010621184055. doi:10.2174/1573407216999200723113054
Alam, M. R., Dobhal, V., and Singh, S. (2024). Neuroprotective potential of solanesol against tramadol induced zebrafish model of Parkinson's disease: insights from neurobehavioral, molecular, and neurochemical evidence. Drug Chem. Toxicol. 28, 1241–1256. doi:10.1080/01480545.2024.2355542
Bai, Q., Yu, J., Su, M., Bai, R., Katumata, G., Katumata, M., et al. (2014). Antioxidant function of solanesol and its inhibitory effect on tyrosinase. J. Biomed. Eng. 31 (4), 833–836.
Campbell, N. K., Fitzgerald, H. K., and Dunne, A. (2021). Regulation of inflammation by the antioxidant haem oxygenase 1. Nat. Rev. Immunol. 21 (7), 411–425. doi:10.1038/s41577-020-00491-x
Charabati, M., Wheeler, M. A., Weiner, H. L., and Quintana, F. J. (2023). Multiple sclerosis: neuroimmune crosstalk and therapeutic targeting. Cell 186 (7), 1309–1327. doi:10.1016/j.cell.2023.03.008
Chen, H. B., Zhang, J., Yu, H. X., and Hu, Q. L. (2007). In vitro study on the antibacterial of a medicinal intermediate, solanesol. Qilu Pharm. Aff. (09), 558–559.
Ding, S., Li, Y., Chen, Z., Hu, J., Li, J., Li, J., et al. (2024). Solanesol ameliorates anxiety-like behaviors via the downregulation of cingulate T cell-restricted intracellular antigen-1 in a complete Freund's adjuvant-induced mouse model. Int. J. Mol. Sci. 25 (18), 10165. doi:10.3390/ijms251810165
Duan, C., Chen, Z., Liu, X., Li, K., Wang, X., Jia, W., et al. (2019). Noble surface molecularly imprinted polymer modified titanium dioxide toward solanesol adsorption selectivity study. J. Mater. Res. 34, 3271–3287. doi:10.1557/jmr.2019.148
Eriksson, E. K., Agmo Hernández, V., and Edwards, K. (2018). Effect of ubiquinone-10 on the stability of biomimetic membranes of relevance for the inner mitochondrial membrane. Biochim. Biophys. Acta Biomembr. 1860 (5), 1205–1215. doi:10.1016/j.bbamem.2018.02.015
Eriksson, E. K., Edwards, K., Grad, P., Gedda, L., and Agmo Hernández, V. (2019). Osmoprotective effect of ubiquinone in lipid vesicles modelling the E. coli plasma membrane. Biochim. Biophys. Acta Biomembr. 1861 (7), 1388–1396. doi:10.1016/j.bbamem.2019.04.008
Fisher, K. J., Kinsey, R., Mohamath, R., Phan, T., Liang, H., Orr, M. T., et al. (2023). Semi-synthetic terpenoids with differential adjuvant properties as sustainable replacements for shark squalene in vaccine emulsions. NPJ Vaccines 8 (1), 14. doi:10.1038/s41541-023-00608-y
Gendron, A., Domenichini, S., Zanna, S., Gobeaux, F., Piesse, C., Desmaële, D., et al. (2023). Development and characterization of innovative multidrug nanoformulation for cardiac therapy. Mater. (Basel) 16 (5), 1812. doi:10.3390/ma16051812
Guo, Y., Ni, J. W., and Huang, W. (2008). Comparison on bioactivities of solanesol extracted from tobacco leaves by different methods. J. Anhui Agri. Sci. 36 (15), 6356–6359.
Hirota, T., and King, B. H. (2023). Autism spectrum disorder: a review. JAMA 329 (2), 157–168. doi:10.1001/jama.2022.23661
Huang, W., Li, Z., Niu, H., Wang, J., and Qin, Y. (2008). Bioactivity of solanesol extracted from tobacco leaves with carbon dioxide-ethanol fluids. Biochem. Eng. J. 42, 92–96. doi:10.1016/j.bej.2008.06.002
Jaswal, P., Riya, R., and Singh, G. (2019). Neuroprotective potential of Solanesol and Epigallocatechin gallate on ICV-STZ induced cognitive dysfunction in rats. J. Neurol. Sci. 405S, 19. doi:10.1016/j.jns.2019.10.245
Jiang, A., Handley, R. R., Lehnert, K., and Snell, R. G. (2023). From pathogenesis to therapeutics: a review of 150 Years of Huntington's disease research. Int. J. Mol. Sci. 24 (16), 13021. doi:10.3390/ijms241613021
Lazarević, M., Stanisavljević, S., Nikolovski, N., Dimitrijević, M., and Miljković, Đ. (2024). Complete Freund's adjuvant as a confounding factor in multiple sclerosis research. Front. Immunol. 15, 1353865. doi:10.3389/fimmu.2024.1353865
Liu, J. C., Li, D. Q., Zhou, R. Q., and Hao, F. (2007). Solubilities of solanesol in acetonitrile, ethanol and n-hexane from 285 to 310K. Korean J. Chem. Eng. 24, 113–115. doi:10.1007/s11814-007-5019-y
Liu, T., Sun, L., Zhang, Y., Wang, Y., and Zheng, J. (2022). Imbalanced GSH/ROS and sequential cell death. J. Biochem. Mol. Toxicol. 36 (1), e22942. doi:10.1002/jbt.22942
Liu, Y., Lin, M., Mu, X., Qin, L., Deng, J., Liu, Y., et al. (2023). Protective effect of solanesol in glucose-induced hepatocyte injury: mechanistic insights on oxidative stress and mitochondrial preservation. Chem. Biol. Interact. 383, 110676. doi:10.1016/j.cbi.2023.110676
Long, J. P., Chen, Z. B., Liu, X. J., and Du, X. Y. (2015). Preparation and adsorption property of solanesol molecular imprinted polymers. Des. Monomers Polym. 18, 641–649. doi:10.1080/15685551.2015.1070495
Ma, X., Meng, Z., Qiu, L., Chen, J., Guo, Y., Yi, D., et al. (2016). Solanesol extraction from tobacco leaves by Flash chromatography based on molecularly imprinted polymers. J. Chromatogr. B 1020, 1–5. doi:10.1016/j.jchromb.2016.03.007
Ma, Y., Hou, T., Zhang, J., Zhou, H., Xu, R., Liu, Y., et al. (2011). Solanesol antioxidation. Food Res. 32 (02), 8–12.
Mansouri, A., Reiner, Ž., Ruscica, M., Tedeschi-Reiner, E., Radbakhsh, S., Bagheri, E. M., et al. (2022). Antioxidant effects of statins by modulating Nrf2 and Nrf2/HO-1 signaling in different diseases. J. Clin. Med. 11 (5), 1313. doi:10.3390/jcm11051313
Mehan, S., Monga, V., Rani, M., Dudi, R., and Ghimire, K. (2018b). Neuroprotective effect of solanesol against 3-nitropropionic acid-induced Huntington's disease-like behavioral, biochemical, and cellular alterations: restoration of coenzyme-Q10-mediated mitochondrial dysfunction. Indian J. Pharmacol. 50 (6), 309–319. doi:10.4103/ijp.IJP_11_18
Mehan, S., Rajput, M., Dudi, R., and Ghimire, K. (2018a). Neuroprotective strategies of solanesol in mitochondrial impairment in experimentally induced Huntington disease. J. Pharm. Toxicol. 1 (1), 3–7.
Morris, H. R., Spillantini, M. G., Sue, C. M., and Williams-Gray, C. H. (2024). The pathogenesis of Parkinson's disease. Lancet 403 (10423), 293–304. doi:10.1016/S0140-6736(23)01478-2
Nierenberg, A. A., Agustini, B., Köhler-Forsberg, O., Cusin, C., Katz, D., Sylvia, L. G., et al. (2023). Diagnosis and treatment of bipolar disorder: a review. JAMA 330 (14), 1370–1380. doi:10.1001/jama.2023.18588
Qi, H., Zhang, Q., Zhao, T., Chen, R., Zhang, H., Niu, X., et al. (2005). Antioxidant activity of different sulfate content derivatives of polysaccharide extracted from Ulva pertusa (Chlorophyta) in vitro. Int. J. Biol. Macromol. 37 (4), 195–199. doi:10.1016/j.ijbiomac.2005.10.008
Rajdev, K., Siddiqui, E. M., Jadaun, K. S., and Mehan, S. (2020). Neuroprotective potential of solanesol in a combined model of intracerebral and intraventricular hemorrhage in rats. IBRO Rep. 8, 101–114. doi:10.1016/j.ibror.2020.03.001
Rajkhowa, B., Mehan, S., Sethi, P., Prajapati, A., Suri, M., Kumar, S., et al. (2022). Activating SIRT-1 signalling with the mitochondrial-CoQ10 activator solanesol improves neurobehavioral and neurochemical defects in ouabain-induced experimental model of bipolar disorder. Pharm. (Basel) 15 (8), 959. doi:10.3390/ph15080959
Rowland, R., Latimer, P., and Giles, J. (1956). Flue-cured tobacco. I. Isolation of solanesol, an unsaturated alcohol. J. Amer. Chem. Soc. 78, 4680–4683. doi:10.1021/ja01599a041
Sczepanik, F. S. C., Grossi, M. L., Casati, M., Goldberg, M., Glogauer, M., Fine, N., et al. (2020). Periodontitis is an inflammatory disease of oxidative stress: we should treat it that way. Periodontol 84 (1), 45–68. doi:10.1111/prd.12342
Shang, J., Liu, H., Zheng, Y., and Zhang, Z. (2023). Role of oxidative stress in the relationship between periodontitis and systemic diseases. Front. Physiol. 14, 1210449. doi:10.3389/fphys.2023.1210449
Sharma, N., Upadhayay, S., Shandilya, A., Sahu, R., Singh, A., Rajkhowa, B., et al. (2021). Neuroprotection by solanesol against ethidium bromide-induced multiple sclerosis-like neurobehavioral, molecular, and neurochemical alterations in experimental rats. Phytomedicine Plus 1 (4), 100051. doi:10.1016/j.phyplu.2021.100051
Sharma, R., Rahi, S., and Mehan, S. (2019). Neuroprotective potential of solanesol in intracerebroventricular propionic acid induced experimental model of autism: insights from behavioral and biochemical evidence. Toxicol. Rep. 6, 1164–1175. doi:10.1016/j.toxrep.2019.10.019
Sheth, K. N. (2022). Spontaneous intracerebral hemorrhage. N. Engl. J. Med. 387 (17), 1589–1596. doi:10.1056/NEJMra2201449
Sridevi, P., Vijayanand, P., and Raju, M. B. (2017). Formulation and evaluation of anti-inflammatory herbal gel containing isolated solanesol. Ann. Phytomed. 6, 127–131. doi:10.21276/ap.2017.6.1.19
Ullah, N., Hasnain, S. Z. U., Baloch, R., Amin, A., Nasibova, A., Selakovic, D., et al. (2024). Exploring essential oil-based bio-composites: molecular docking and in vitro analysis for oral bacterial biofilm inhibition. Front. Chem. 12, 1383620. doi:10.3389/fchem.2024.1383620
Wang, Y. Y., Li, Y. F., and Zhou, Z. F. (2024). Solanesol alleviates CFA-induced chronic inflammatory pain via inhibition of proinflammatory cytokines in spinal glial cells. Heliyon 10 (15), e34870. doi:10.1016/j.heliyon.2024.e34870
Xiang, D., Yao, Z., Liu, Y., Gai, X., Du, Y., Zhang, Z., et al. (2017). Analysis on solanesol content and genetic diversity of Chinese flue-cured tobacco (nicotiana tabacum L.). Crop Sci. 57 (2), 847–855. doi:10.2135/cropsci2016.07.0568
Yan, N., Liu, Y., Gong, D., Du, Y., Zhang, H., and Zhang, Z. (2015). Solanesol: a review of its resources, derivatives, bioactivities, medicinal applications, and biosynthesis. Phytochem. Rev. 14, 403–417. doi:10.1007/s11101-015-9393-5
Yan, N., Liu, Y., Liu, L., Du, Y., Liu, X., Zhang, H., et al. (2019). Bioactivities and medicinal value of solanesol and its accumulation, extraction Technology, and determination methods. Biomolecules 9 (8), 334. doi:10.3390/biom9080334
Yao, X., Bai, Q., Yan, D., Li, G., Lü, C., and Xu, H. (2015). Solanesol protects human hepatic L02 cells from ethanol-induced oxidative injury via upregulation of HO-1 and Hsp70. Toxicol Vitro 29 (3), 600–608. doi:10.1016/j.tiv.2015.01.009
Yao, X., Lu, B., Lü, C., Bai, Q., Yan, D., Wu, Y., et al. (2017). Solanesol induces the expression of heme oxygenase-1 via p38 and Akt and suppresses the production of proinflammatory cytokines in RAW264.7 cells. Food Funct. 8 (1), 132–141. doi:10.1039/c6fo01073c
Zhang, R., Wang, X., Ren, X., Li, Q., Tang, J., Tang, L., et al. (2018). The effect of solanesol on experimental models of periodontitis in rats. Chin. J. Ethnomed. Ethnopharm. 27 (06), 31–40.
Keywords: solanesol, antimicrobial, antioxidant, anti-inflammatory, membrane stabilization
Citation: Ma Y, Wei G, Dong Z, Wang Z, Zhai X, Liu Y, Chen H, Fu Y, Hou H, Hu Q and Chu M (2025) Solanesol: a promising natural product. Front. Pharmacol. 16:1504245. doi: 10.3389/fphar.2025.1504245
Received: 30 September 2024; Accepted: 04 March 2025;
Published: 24 March 2025.
Edited by:
Muhammad Furqan Akhtar, Riphah International University (Lahore), PakistanReviewed by:
Ahmed Esmat Abdel Moneim, Helwan University, EgyptCopyright © 2025 Ma, Wei, Dong, Wang, Zhai, Liu, Chen, Fu, Hou, Hu and Chu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hongwei Hou, cXNmY3RjQDE2My5jb20=; Qingyuan Hu, aHVxeTE5NjVAMTYzLmNvbQ==; Ming Chu, ZmFtb3VzQGJqbXUuZWR1LmNu
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.