- 1School of Traditional Chinese Medicine, Yunnan University of Chinese Medicine, Kunming, China
- 2College of Pharmacy, Kunming Medical University, Kunming, China
Clerodendranthus spicatus (CS), is an herbaceous perennial belonging to the family Lamiaceae. The herb is extensively employed in traditional Chinese medicine for the mitigation of nephritis, cystitis, kidney stones, urological tract stones, gout, and other urinary conditions. Numerous research studies have been conducted in the past to explore the traditional medicinal value, phytochemical composition, pharmacological effects, and quality control measures associated with this plant. This has motivated us to systematically search various online databases such as Google Scholar, PubMed, Science Direct, Elsevier, CNKI, Scopus, Embase, and Web of Science using specific keywords to get the most recent research information findings related to this plant. Phytochemical investigations have identified that this plant predominantly contains flavonoids, terpenoids, phenylpropanoids, and volatile oil compounds. Certain constituents have been employed as markers in quality assessment research, and some were recognized as bioactive agents in the management of specific ailments. These components have demonstrated notable effectiveness in combating bacterial infections, reducing inflammation, providing antioxidant properties, managing hyperuricemia, and offering renal protection. Notably, clinical trials have confirmed its remarkable efficacy in treating urinary inflammation and stones. We acquired recent research findings concerning CS in the fields of phytochemistry, pharmacology, quality control, and clinical applications via online search. These findings have been summarized and analysed to offer a valuable reference for further comprehensive research, development, and utilization of CS.
1 Introduction
Ethnic medicine Clerodendranthus spicatus (Thunb.) C.Y.Wu (CS, its Chinese name is “Shencha” or “kidney tea,” syn. Orthosiphon aristatus var. aristatus, Orthosiphon spicatus (Thunb.) Backer, Bakh.f. and Steenis, Clerodendranthus stamineus (Benth.) Kudô and Orthosiphon stamineus Benth.), a perennial herb of the Lamiaceae family, has been listed in the latest revision of “The Plant List” (http://www.theplantlist.org/) and Medicinal Plant Names Services (http://mpns.kew.org), and commonly known as “cat’s whiskers grass” or “cat’s whiskers tea” (Guo, 2020), with referring as “Yanuomiao” in Dai medicine, is native to the southwest region of Yunnan province in China (Zhao et al., 2004). According to traditional medicinal practices, it is cooling in nature, a mild taste with slight bitterness, and is believed to exhibit properties such as heat-clearing, dehumidifying, stone-expelling, and diuretic effects. As a result, it has gained popularity as a treatment agent for ailments such as cystitis, nephritis, urinary calculus, arteriosclerosis, gout, jaundice, colds for more than two millennia (Hou et al., 2011).
Contemporary phytochemical analysis has isolated over 300 compounds in CS including flavonoids, terpenoids, phenols, volatile oils, and others (Zhang Y. et al., 2021). Pharmacological investigations confirmed that the plant possesses anti-inflammatory, antibacterial, antioxidant, anti-cancer, anti-gout, and nephroprotective (Hou et al., 2011). The purpose of this review is to provide up-to-date evidence-based information on the phytochemistry, pharmacology, quality control, and clinical application of CS. Therefore, this review summarizes the uniqueness and diversity of the structure of the compounds isolated from this plant, and the latest evidence on the effectiveness of pharmacological effects, and proposes the direction of future in-depth research on this plant as an update and supplement to the information from the previous review (Zhang et al., 2023; Zhang Y. et al., 2021). Therefore, this study provides the latest knowledge and conclusions for an in-depth understanding of this medicinal herb, which may stimulate more comprehensive research, development and utilization. Figure 1 is a representative image of the species CS with its vegetative body and blooms.

Figure 1. (A) Vegetative body (B) Blooms (Su et al., 2024).
2 Methodology
The relevant information and data on the phytochemistry, pharmacology, quality control, and clinical application of CS in this review were extracted and collected from published articles in related fields. The chemical structures of the compounds mentioned in the article were prepared using the ChemDraw program, and keywords such as CS, OS, shencha, cat’s whiskers grass, cat’s whiskers tea, research progress, phytochemistry, chemical composition, separation and identification, pharmacological activity, activity quality control, quality standards, and clinical application were used, and Google Scholar, PubMed, and Science Direct were used, Elsevier, Scopus, Embase, Web of Science and China National Knowledge Infrastructure (CNKI), Wanfang, the Chinese Scientific and Technological Periodical Database, the Chinese Biomedical Database (CBM) and other websites for an exhaustive online search. These terms can be used separately or in combination. Then, the retrieved literature was screened and classified according to four aspects: chemical composition, pharmacological effects, quality control and clinical application. The review includes research articles, master’s theses and doctoral dissertations published in English and Chinese, which provide complete information and data.
3 Phytochemistry
The research extensively reviewed the literature on the chemical constituents of CS spanning the last 2 decades. To date, over 300 distinct chemical compounds have been isolated, predominantly encompassing flavonoids, terpenoids, anthraquinones, phenylpropanoids, and volatile oils, with flavonoids, phenylpropanoids, and terpenoids possessing the highest content, also serving as the primary active principles within CS.
3.1 Flavonoids
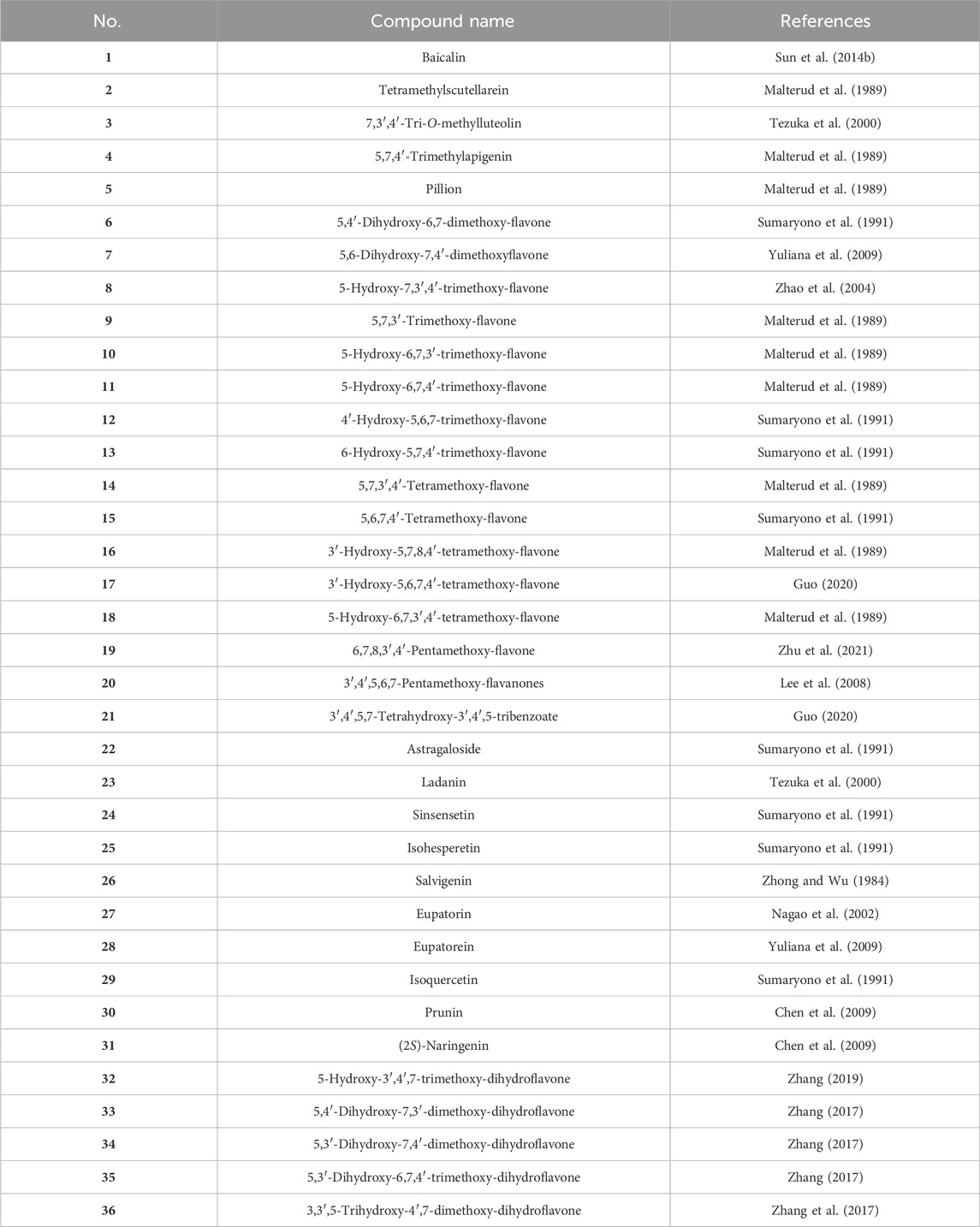
Upon extensive exploration of CS, 36 flavonoid derivatives (1–36, Table 1; Figure 2) have been isolated and identified from the overground part, including 29 flavonoids (1–29) and seven dihydroflavonoids (30–36). Notably, Baicalin (1) mitigated renal fibrosis via enhancing CPT1α-regulated fatty acid oxidation in diabetic nephropathy (Hu et al., 2024), and flavonoids (2, 5, 7, 17, 24, 27, and 28) displayed a diuretic effect by binding to adenosine A1 receptor (Yuliana et al., 2009).
3.2 Terpenoids
CS is rich in terpenoids, predominantly comprising diterpenoids (46–122) and triterpenoids (123–142). Recently, researchers have used more advanced extraction and separation techniques to isolate straight chain sesquiterpenoids (37–38) and norsesquiterpenoids (39–45).
3.2.1 Sesquiterpenoids
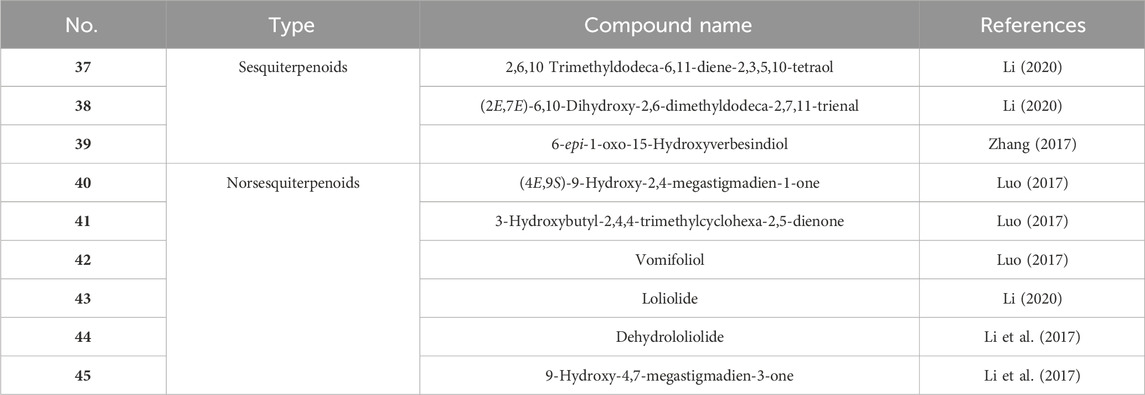
Currently, only nine sesquiterpenoids (Table 2; Figure 3, 37–45) have been isolated from CS. Among them, Loliolide (43) can be used as a potential anti-aging bioagent (Zhang et al., 2023), the compounds Loliolide (43) and Dehydrololiolide (44) have anticholinesterase activities (Xu et al., 2022). In addition, the compound Vomifoliol (42) may inhibit the nuclear factor of activated T-cells (NFAT) signaling pathway with calcineurin (CN) as the target enzyme, thereby inhibiting the immune response of Jurkat cells (Zhang X. et al., 2021).
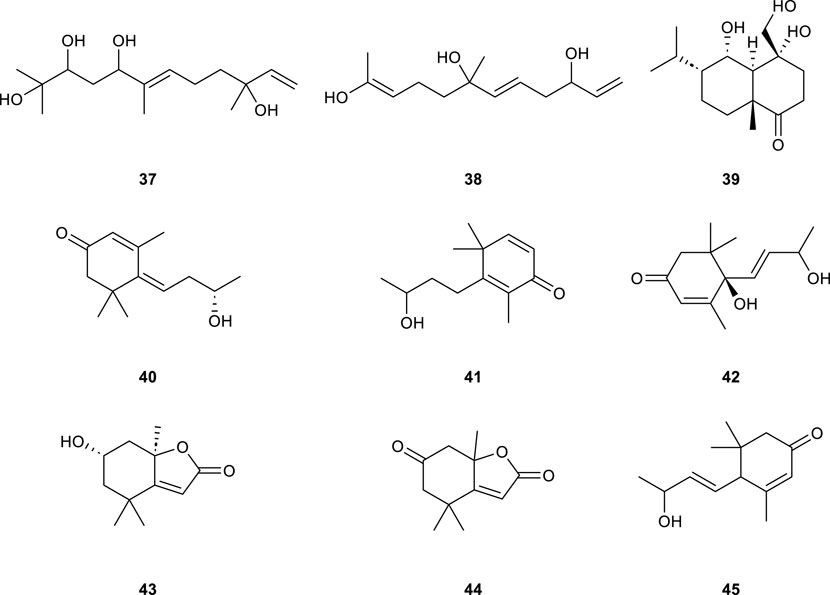
Figure 3. Chemical structures of sesquiterpenoids compounds isolated from Clerodendranthus spicatus.
3.2.2 Diterpenoids
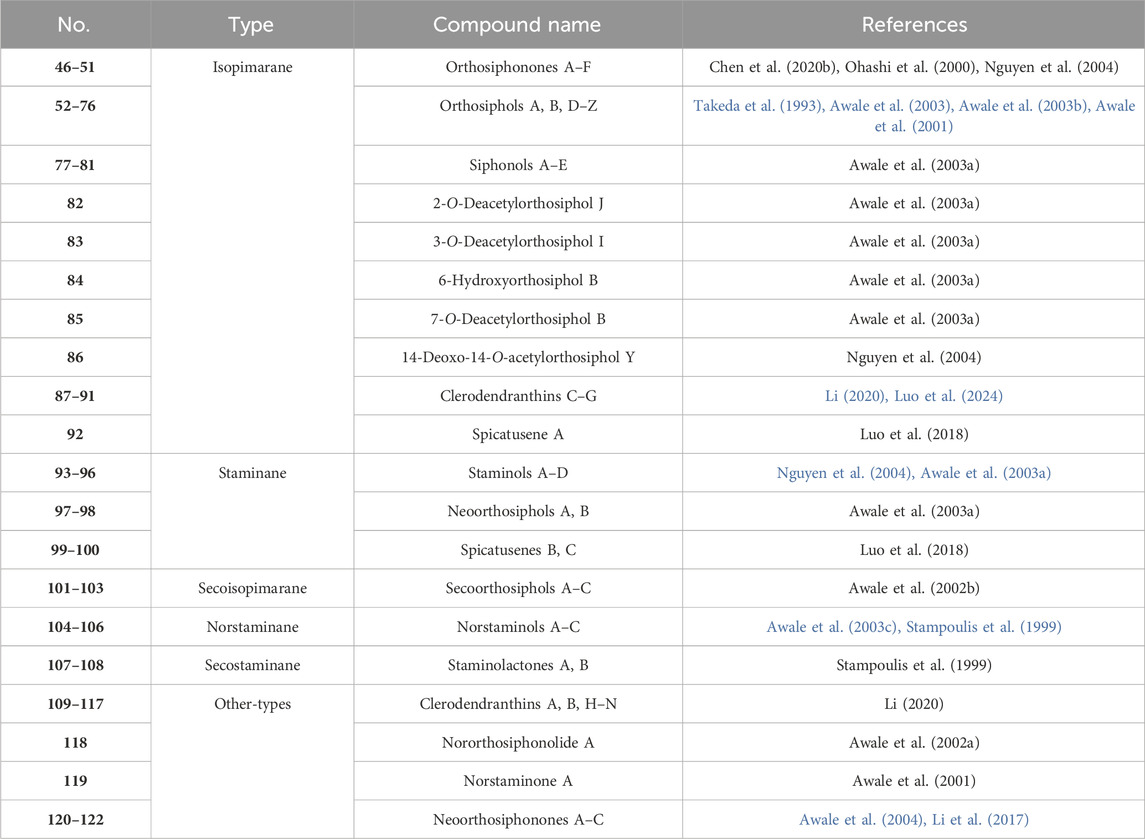
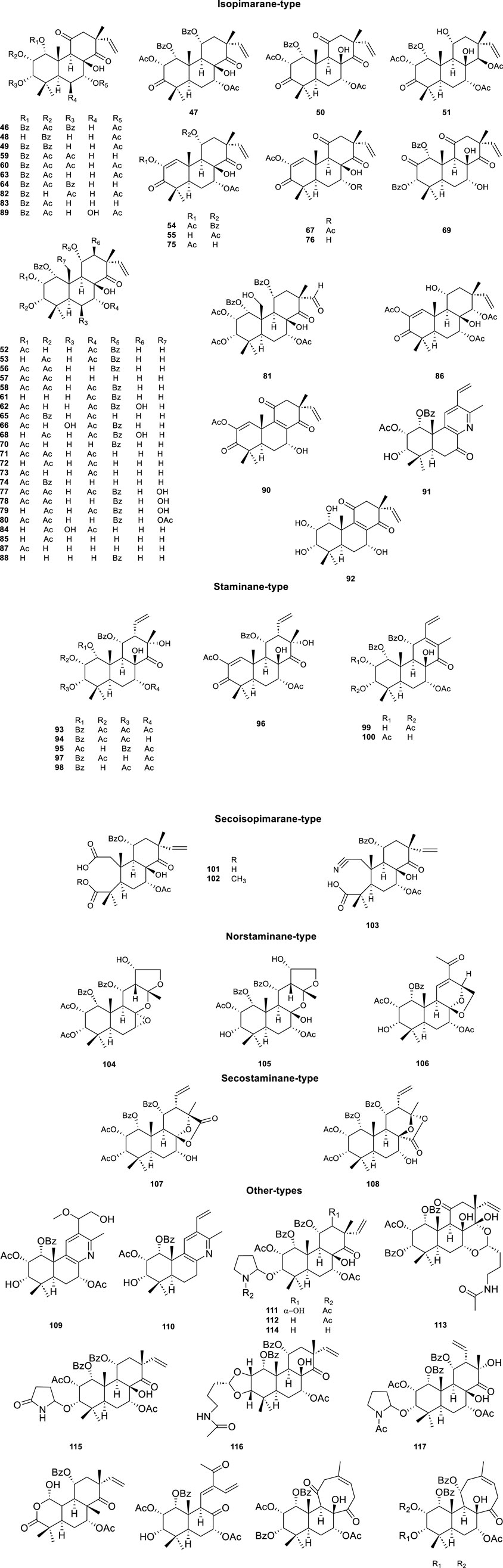
As the characteristic and effective ingredients of CS, a total of 73 diterpenoids (Table 3; Figure 4, 46–123) have been isolated, which were divided into five main categories according to the skeleton type: isopimarane, staminane, secoisopimarane, norstaminane, and secostaminane.
The highly oxygenated diterpenoids in CS exhibited various pharmacological properties including anti-inflammation, anti-tumor activity, renal fibrosis prevention, gout management, and diabetes control (Zhang et al., 2023). Highly oxygenated diterpenoids exhibited notable anti-inflammatory activity, specifically the Orthosiphols B, D, and M (53, 54 and 63), as well as Orthosiphonone A and Neoorthosiphol A, surpassing aspirin (Chen et al., 2020a). Orthosiphols A, D (52, 54) and Orthosiphonone A (46) modulated gout pathogenesis via the anti-inflammatory and analgesic cascade (Chen et al., 2020b). The compounds Siphonols A–C (77-79) and Siphonol E (81) showed more potent inhibitory effects on the nitric oxide (NO) production in lipopolysaccharide (LPS)-activated macrophage-like J774.1 cells than a positive control NG-monomethyl-L-arginine (L-NMMA), and Siphonols A–E (77-81) represented the first examples of isopimarane-type diterpenes oxygenated at C-20 (Awale et al., 2003). The compounds Spicatusenes B and C (99 and 100) exhibited anti-inflammatory properties in vitro by inhibiting productions of inflammatory mediators (IL-8, IL-1β, and TNF-α) (Chen et al., 2020a). Additionally, the Spicatusene C (100), Orthosiphols K, L, N, R, and W (61, 62, 64, 68, 73), along with Neoorthosiphol A (97) and Norstaminol B (106) demonstrated antifibrotic effects in TGF-β1-induced rat kidneys (Luo et al., 2018). Seven derivatives of orthosiphol K (61) were modified by different reagents and conditions (Figure 5). Subsequently, the anti-rheumatoid arthritis activity of these diterpenoid derivatives was evaluated on a TNF-α-induced human rheumatoid fibroblast-like synovial cell model. Of these compounds, compound 2 showed the strongest activity. Based on their inhibitory effect on IL-1b release levels, their structure-activity relationship was preliminarily derived: vinyl-migrated sea pine type diterpenoids showed no activity compared to isopimarane-type diterpenoids. The C-11 OBz group and the C-7 OAc group are important for maintaining activity. For substituents on the ring A, the electron-withdrawing group on the 2-hydroxyl group decreased the activity while the donor group improved the activity. The oxidation of 3-OH to a carbonyl group decreases activity, while Ac substitution on it may increase activity. Thus, the substituents on rings C and A significantly affect the activity of isopimarane-type diterpenoids. The above studies not only enriched the diversity of the structure of diterpenoids, but also showed that isopimarane-type diterpenoids may be a good precursor for the development of anti-rheumatoid arthritis drugs (Luo et al., 2024).
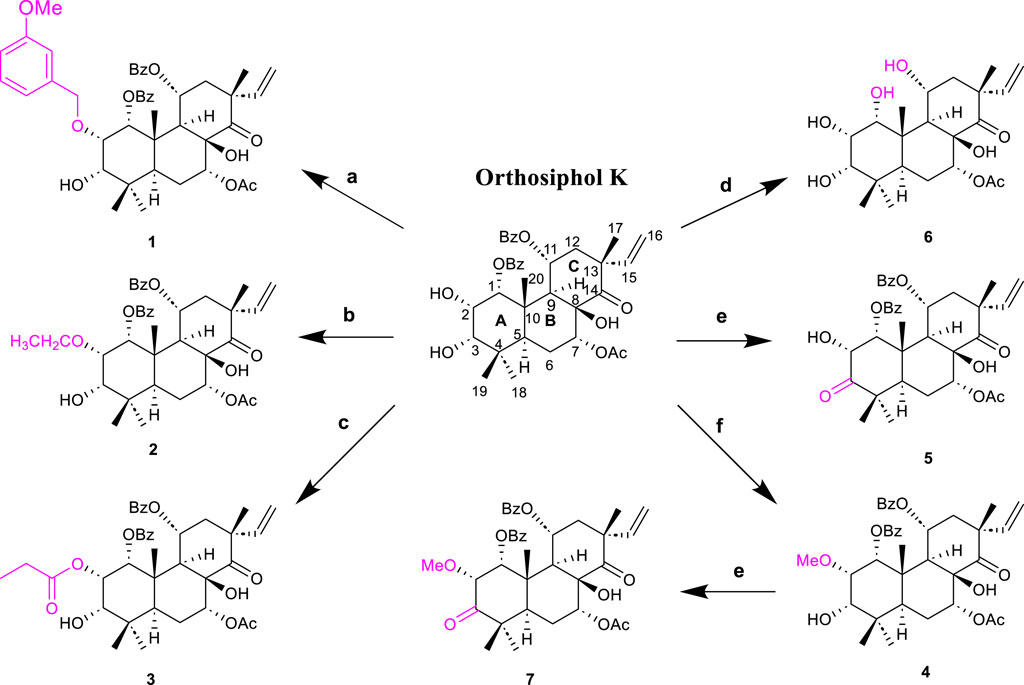
Figure 5. Structures and reactions in modifications of orthosiphol K (61). Reagents and conditions: (A) Cs2CO3, DMF, 3-OMePhCH2Br, 80°C, reflux; (B) Cs2CO3, DMF, CH3CH2Br, 80°C, reflux; (C) Propanoic acid, DMAP, EDCI, CH2Cl2, 0°C–25°C, 6 h; (D) Cs2CO3, DMF, MeI, r.t., 24 h; (E) Jones reagent, acetone, 0°C–25°C, 1.5–2 h; (F) NH4OH/MeOH, 110°C, reflux, 0.5–1 h (Luo et al., 2024).
3.2.3 Triterpenoids
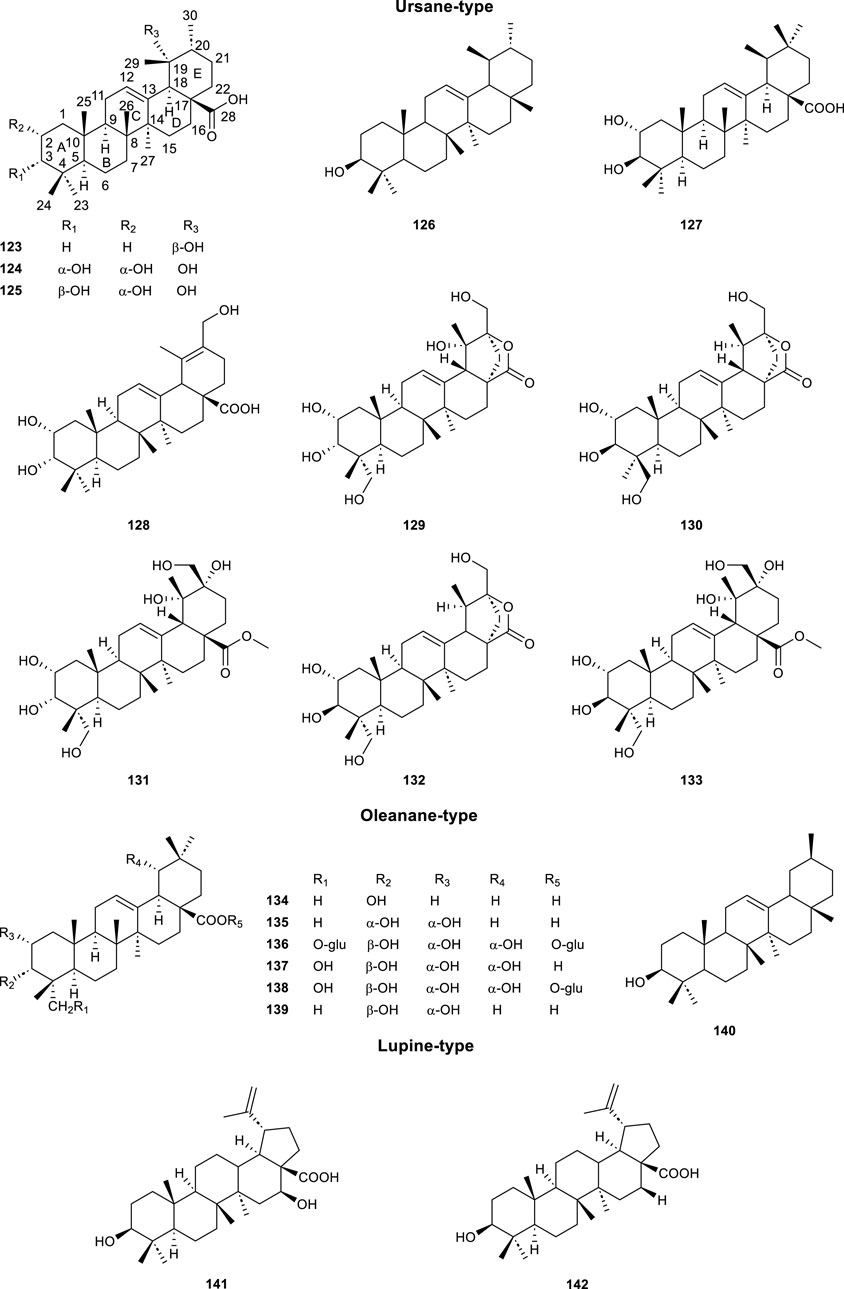
A total of 20 triterpenoids (Table 4; Figure 6, 123–142) was reported from CS. One of the representatives of pentacyclic triterpenoids was Ursolic acid (UA, 123), which possessed important biological effects, including anti-inflammatory, anticancer, antidiabetic, antioxidant and antibacterial effects (Mlala et al., 2019). It has been reported that the C-3 (hydroxyl), C12-C13 (double bond) and C-28 (carboxylic acid) positions of UA have been modified (such as acetylation, methylation, and esterification) to obtain several ursolic acid derivatives, which have stronger potency, bioavailability and water solubility than UA (Mlala et al., 2019). The α-Amyrin (126) demonstrated superior efficacy in stimulating uric acid excretion compared to benzbromarone (Chen et al., 2020b). Maslinic acid (139) was one of the representatives of oleanane-type triterpenoids, which has a variety of biological activities, such as anti-tumor, hypoglycemic, anti-inflammatory, and anti-parasitic (Yan et al., 2024). β-Amyrin (140) was displayed important anti-Toxoplasma activity (Cardona-Trujillo et al., 2024). Something special, Betulinic acid (141) was a triterpene natural product which has shown antiparasitic activity against Leishmania, Trypanosoma cruzi, and Plasmodium (Rocha et al., 2022).
3.3 Phenylpropanoids
Phenylpropanoids, which were characteristic constituents in CS, and exhibited numerous therapeutic properties, such as anticancer, antioxidant, anti-inflammatory, and neuroprotective effects, with 75 phenylpropanoids (Table 5; Figure 7, 143–217) was isolated from CS. Analysis via HPLC-ESI-MS showed the presence of compounds such as Caffeic acid (152), Ferulic acid (156), p-Hydroxybenzoic acid (147), Rosmarinic acid (158), Danshensu (168), and Protocatechuic acid (144) in aqueous extracts of CS, which delayed exogenous senescence by defending antioxidant activities and suppressing inflammation (Wang et al., 2016). Rosmarinic acid (158) was found as a major component responsible for the antioxidant activity in CS extract (Nuengchamnong et al., 2011), and Orthosiphoic acid B (172) and C (173) displayed modest anti-HIV-1 protease activity (Sun et al., 2014). Clerodens A–J (178–187), which with a distinctive bicycle [2.2.2] octane moiety, and Cleroden D (181) notably inhibited LPS-induced NO production in RAW 264.7 (Ma et al., 2015), while the Cleroden E (182) showed notable antibacterial potency to drug-resistant bacterial properties in vitro. Additionally, Helisterculin C (190), featuring a rare bicyclol [2.2.2] octane moiety previously isolated phenolic acid derivatives, Helisterculins C (190) and D (191), exhibited moderate neuroprotective activity (Zhou et al., 2017).
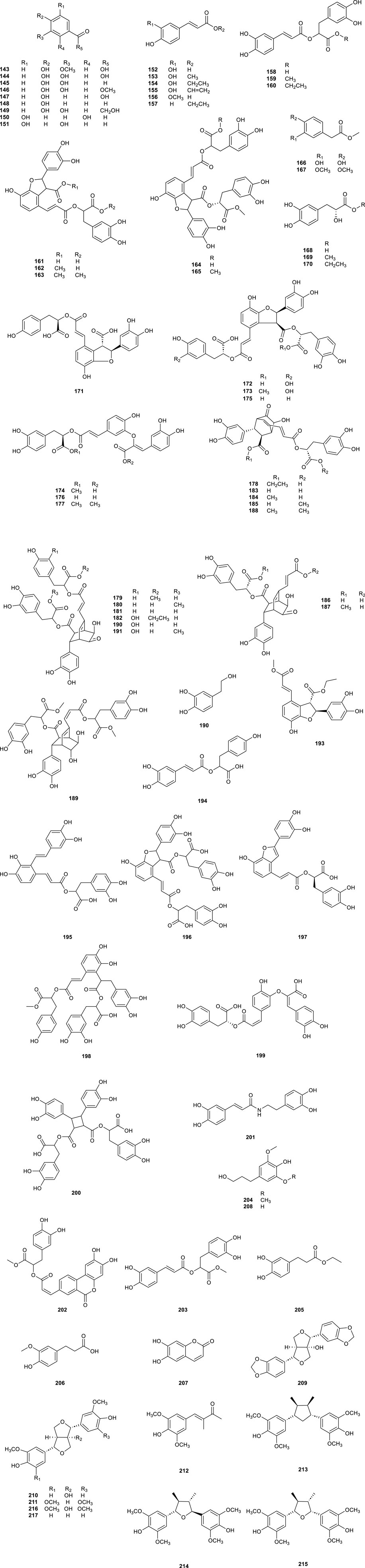
Figure 7. Chemical structures of phenylpropanoids compounds isolated from Clerodendranthus spicatus.
3.4 Volatile oil
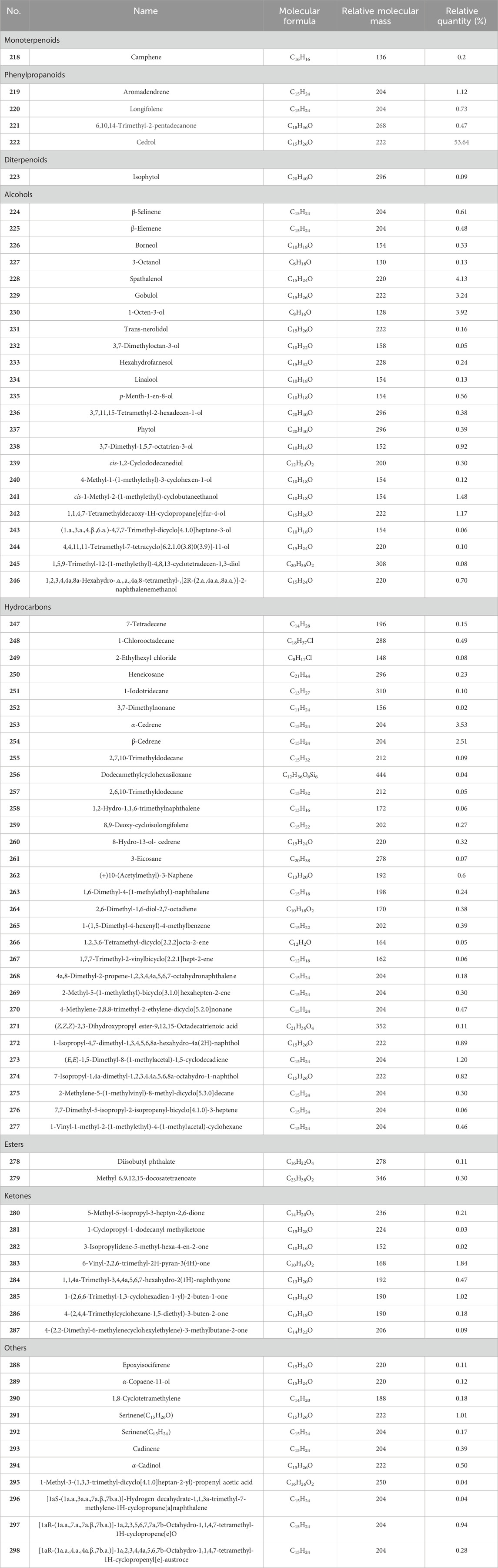
CS is abundant in volatile oil constituents, yet there are few studies on their pharmacological activity and medical application, predominantly highlighting chemical composition identification and content determination. 81 chemical constituents were identified (Table 6; Figure 8, 218–298) in volatile oils extracted via conventional steam distillation and solvent extraction coupled with gas chromatography–mass spectrometry (GC-MS), primarily comprising alcohols, alkenes, ketones, naphthalene, esters, and alkanes. Cedrol (222) as the most abundant sesquiterpene (Li et al., 2022), comprising 53.64%, which exhibiting antibacterial activity (Ma, 2013). According to the literature, Cedrol (222) has antibacterial activity (Chang et al., 2023).
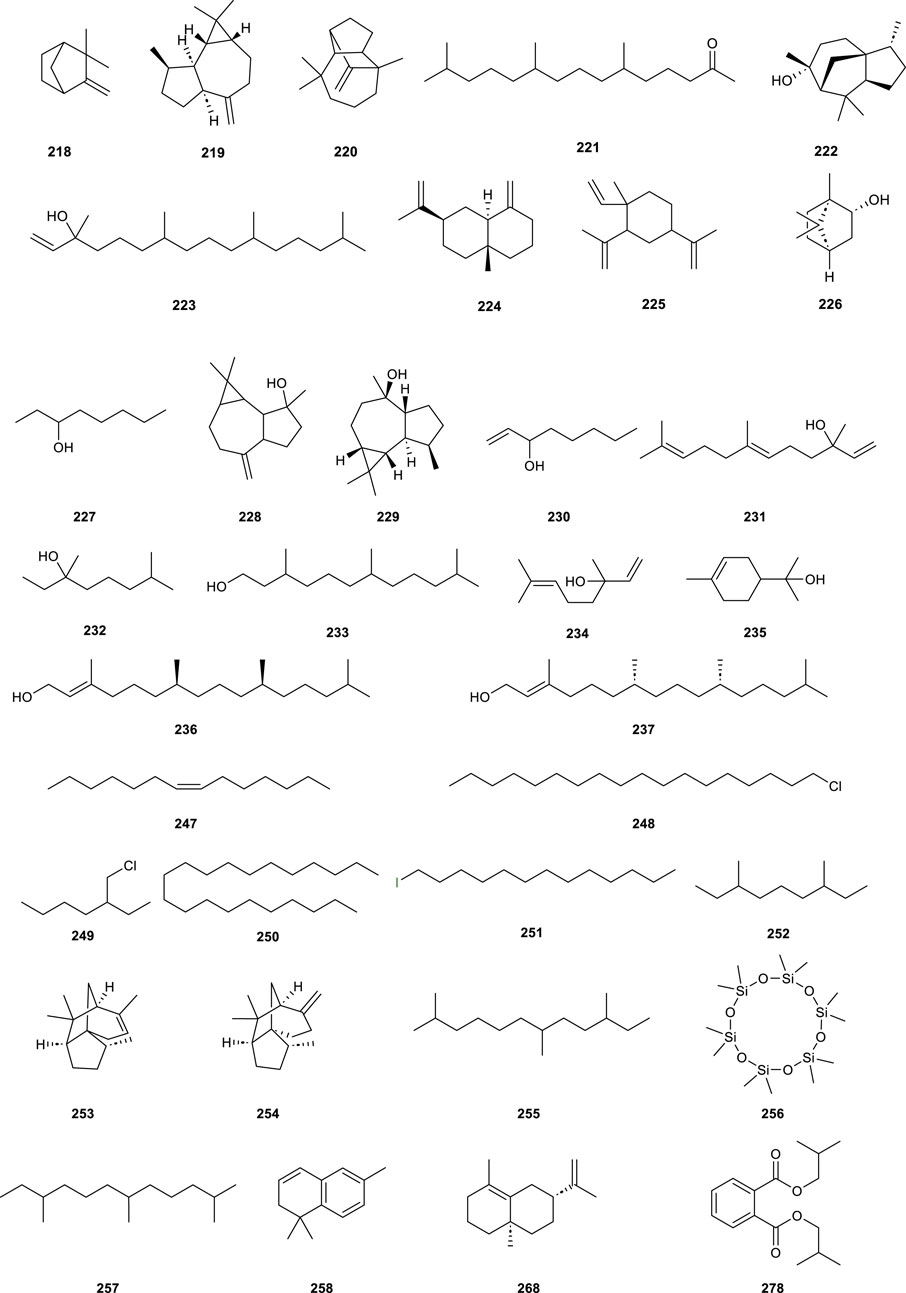
Figure 8. Chemical structures of partially volatile oil compounds isolated from Clerodendranthus spicatus.
3.5 Others
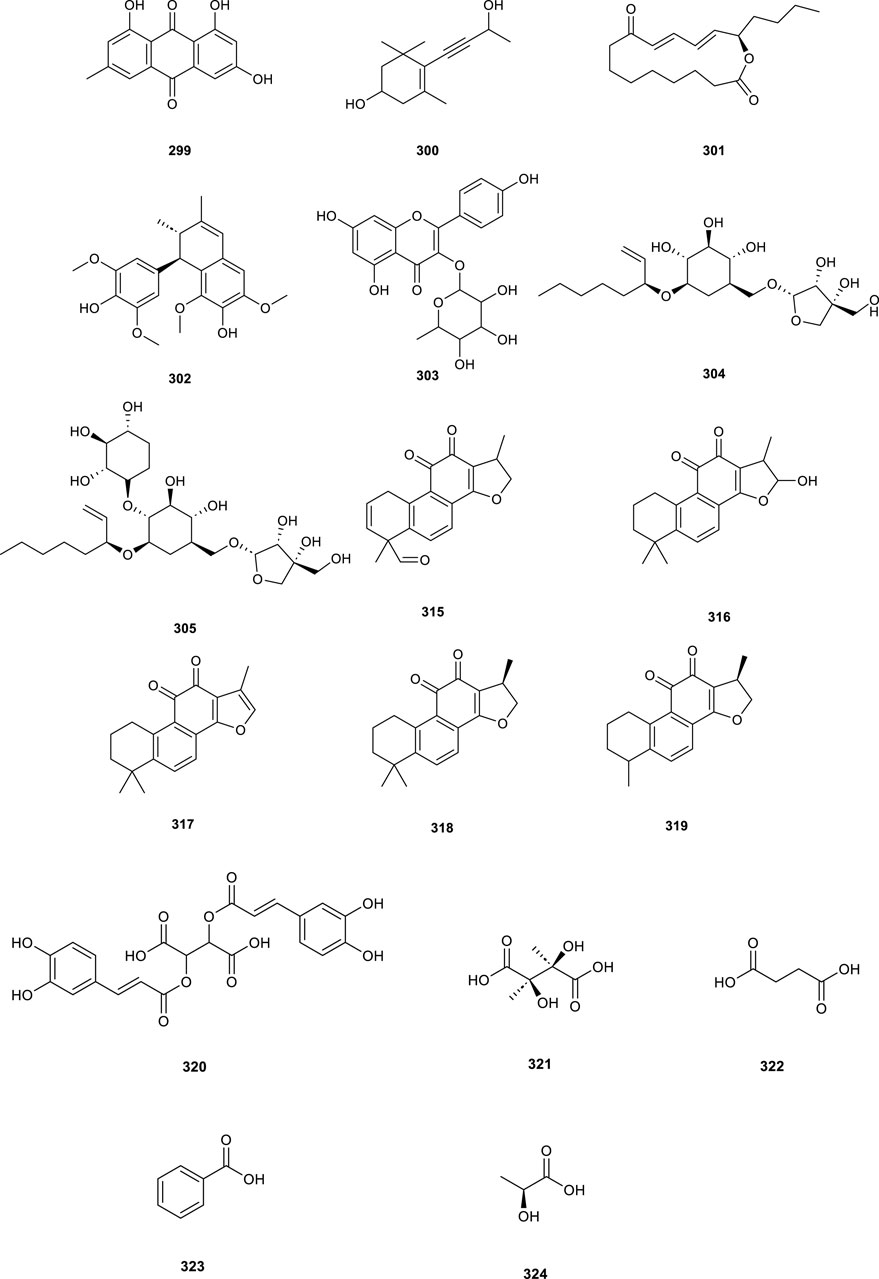
Additional compounds isolated from CS include anthraquinones, cyanosides, alkaloids, organic acids, and diverse types of components (Table 7; Figure 9, 299–324). Notably, amides compounds (311–313) demonstrated anti-inflammatory properties, confirming their role as major anti-inflammatory constituents (Li et al., 2022).
4 Pharmacology
Previously documented studies indicate CS possesses diverse pharmacological activities such as anti-inflammatory, antibacterial, antioxidant, anti-neoplastic, anti-gout, and renal protection properties. Notably, it is frequently utilized for treating nephritis, gout, urinary tract infections, kidney stones, and related conditions. An in-depth analysis on these pharmacological actions was thus discussed subsequently.
4.1 Anti-inflammatory
Thousands of years of ethnic medicine practice has proved that CS has superior efficacy in the treatment of urinary system inflammation, and modern phytochemical and biological activity research has unearthed the material basis for CS to exert anti-inflammatory effects. The hyperoxy diterpenoids derived from ethanolic extract of CS exhibited anti-inflammatory potency superior to aspirin in vitro by inhibiting the pro-inflammatory factors TNF-α, IL-1β, and IL-8 stimulated via LPS in renal epithelial cells (HK-2 cells) (Chen et al., 2020a). In addition, phenolic acid, the Clerodens A–D (178–181), with a dicyclic [2.2.2] octene framework, was isolated from the extract CS demonstrating the anti-inflammatory activity by suppressing NO produced by RAW 264.7 cells induced by LPS (Ma et al., 2015) The total flavonoids of CS proved effective against bacterial prostatitis in rats, potentially related to inhibiting TNF-α and IL-8 inflammation and reducing oxidative stress (Chen and Yang, 2016). Three identified amide compounds, N-cis-Feruloyltyramine (312), N-trans-Feruloyltyramine (313), and trans-N-Cinnam-oyltyramine (314), isolated from the middle and low polarity parts of CS, which exhibited potent anti-inflammatory activity in the LPS-stimulated RAW 264.7 macrophage inflammation model. The results of this study support the idea that amide compounds were one of the main components of the anti-inflammatory effect of CS (Li et al., 2022).
4.2 Antibacterial
Nowadays, bacterial infections have increased all over the world and antibiotics resistance has emerged as a challenging healthcare issue, therefore screening of medicinal plants to explore new antibacterial agents has become a top priority (Alshawsh et al., 2012). The aqueous extract of CS suppresses the expansion of Gram-negative bacilli and Staphylococci such as Escherichia coli and Klebsiella pneumonia (Yi et al., 2013). Cleroden E (182) with a bicyclic [2.2.2] octene functional showed moderate antibacterial activity against various clinically isolated drug-resistant strains such as Staphylococcus aureus ATCC 33591 (MRSA), Staphylococcus epidermidis 09-3 (MRSE), Enterococcus faecalis ATCC 51299 VRE, E. coli 09-1 ESBLs, and Klebsiella pneumoniae ATCC BAA-2146 NDM-1, exhibiting minimum inhibitory concentrations of 3.2, 11.8, 3.2, 6.4, and 3.2 μg/mL in vitro (Li et al., 2016). In conclusion, this plant exhibited promising antibacterial properties, particularly against Gram-positive bacteria. Both past medicinal practices and modern scientific research have highlighted the potential of CS as an antimicrobial agent.
4.3 Antioxidant
The physiological and pathological processes of many diseases are accompanied by free radical-mediated lipid peroxidation, such as arteriosclerosis, heart diseases, aging process and cancer. The petroleum ether (PE), chloroform (CE), ethyl acetate (EAE), and water (WE) extracts of CS demonstrated DPPH, O2−, ⋅OH, Fe2+, and ROS scavenging activities in vitro. Significantly, EAE and WE extract exhibited potency to decrease MDA levels and enhance GSH-Px levels in renal homogenates, concurrently suppressing mitochondrial swelling under concentration-dependent conditions (Chen et al., 2014). Besides, research noted that the methanol extract of CS exhibits strong scavenging capabilities for superoxide ions and DPPH free radicals in vitro, attributing its efficacy to Sinsensetin (24) and 3′-Hydroxy-5,6,7,4′-tetramethoxyflavone (17) (Yam et al., 2007). Other studies showed that there were significant correlations between phenolic compounds and antioxidant properties of medicinal plants. The ethyl acetate fraction (EAF) of aqueous-methanolic extract of CS, which had the highest total phenol content and high total flavonoid content, and the EAF fraction showed excellent antioxidant activity in a number of subsequent antioxidant tests such as β-carotene bleaching (BCB) and DPPH free radical determination (Abdelwahab et al., 2011). Even more surprisingly, a number of phenolic compounds that exert antioxidant effects were identified in another study, such as Helisterculin C (190), Salvianolic acid B (196), Orthosiphoic acid E (175) and Ethyl caffeate (154) might be the main antioxidant constituents (Wang et al., 2023). These findings supported the idea that the antioxidant activity of phenolic compounds was found to be mainly due to their scavenging and redox properties, through neutralizing and quenching free radicals (Alshawsh et al., 2012). It was envisaged that phenolic compounds in CS will be used as an antioxidant to support the treatment of many major diseases in the future.
The randomized trial in a mouse model of UV-induced skin damage demonstrated that the WE extract of CS successfully reduced the levels of inflammatory cytokines such as IL-1β, IL-6, TNF-α, COX-2, and PGE2, rejuvenated collagen density, and restricted on production of matrix metalloproteinases (Wang et al., 2016), suggesting that WE with ability to postpone exogenously induced aging via antioxidant and anti-inflammatory properties.
4.4 Anti-tumor
Tumor/cancer is a prominent cause of mortality in humans, and numerous studies have proposed that naturally occurring compounds derived from plants could serve as potential agents for combating tumors (Xu et al., 2015). Recent research on the anti-tumour effects of CS has mainly focused on non-small cell lung, colon and liver cancers Studies in vivo and in vitro have proved that CS has an anti-tumor effect. Investigations into the in-vitro anti-tumor effects of CS demonstrated that EAE extract potent inhibitory activity against human non-small cell lung cancer cells A549, human ileocecal cancer cells HCT-8, and human hepatocellular carcinoma cells BEL-7402, exhibiting IC50 values of 194.61, 283.05, and 238.61 μg/mL (Zheng et al., 2019). Furthermore, extract (C5EOSEW5050ESA) (400 mg/kg) of CS potently suppresses the Notch signaling cascade by diminishing Notch1 ICD, HES-1, and HEY-2 signalings, thereby impeding Notch signaling function, minimizing expression of multidrug resistance genes, and epithelial-mesenchymal transition markers in gemcitabine-resistant cells, potentially auguring as a complementary treatment agent for drug-resistant pancreatic cancer (Yehya et al., 2022).
In-vivo experiments, the 50% ethanol extract (EE) of CS at a dose of 200 mg/kg resulted in an 83.39% ± 4.1% reduction in colorectal tumor proliferation in athymic mice. In-depth mechanistic studies have convinced that CS exerts the antiangiogenic effect by blocking VEGF signaling pathway, and eventually the antiangiogenic property could be the principle factor for the potent antitumor property of CS, and the antiangiogenic effect may be due to the collective contribution of phytochemicals particularly, Rosmarinic acid (158), Eupatorine (27), Sinensetin (24), Betulinic acid (141), and 3′-Hydroxy-5,6,7,4′-tetramethoxyflavone (17) in CS (Ahamed et al., 2012).
4.5 Anti-hyperuricemia and anti-gout
Many studies have shown that hyperuricemia (HUA) and gout are independent risk factors for chronic kidney disease (CKD), obesity, hypertension, type 2 diabetes, cardiovascular and cerebrovascular diseases, and are independent predictors of premature mortality (Bardin and Richette, 2017). CS has been extensively utilized the treatment of HUA and gout in folk and clinical practice, demonstrating notable effectiveness. Ethyl acetate extract (OSE) of CS that was shown to have curative effects in a mouse model of potassium oxazinate (PO) induced HUA after treatment at a dose of 500–2,000 mg/kg for 7 days. It is hypothesized that its impact may be achieved by decreasing xanthine oxidase (XOD) and adenosine deaminase (ADA) activity in the liver, downregulating the mRNA and protein expression levels of uric acid transporters URAT 1 and GLUT 9 and upregulating OAT 1 and OAT 3 in the renal. Subsequently, eight components were identified from the OSE extracts by UPLC/ESI-MS, including Protocatechualdehyde (145), Caffeic acid (152), Rosmarinic acid (158), Caffeic acid tetramer, Lithospermic acid (161), Salvianolic acid B (196), isorhamnetin-3-O-hexoside and caffeic acid derivative (Xu et al., 2020). Furthermore, the anti-HUA activity of Orthosiphols A, B, N, and α-Amyrin (52, 53, 64 and 126) isolated from the EAE extract of CS was superior to that of benbromarone, and most diterpenes with high oxygen content have significant anti-gout arthritis and analgesic activity, the results exhibited that CS modulated pathological state of gout mainly via the anti-inflammatory and analgesic cascade. (Chen et al., 2020b). Similarly, WE extracts of different doses of CS have exhibited significant anti-hyperuricemia and anti-gout activities through diverse pharmacodynamic mechanisms, such as: 1. CS improved the pathological state of HUA by regulating the structure of intestinal flora (increased the abundance of Roseburia and Enterorhabdus, and decreased the abundance of Ileibacterium and UBA1819) and remodeling metabolic disorders (returned the levels of differential metabolite to normal) (Chen et al., 2023); 2. CS exerted anti-HUA activity via changing the composition of intestinal microbiota (reduced the abundance of unidentified-Ruminococcaceae and Lachnospiraceae-NK4A136-group), metabolites (increased 17 metabolites such as lactose, 4-oxovaleric acid, butyric acid, etc., decreased 55 metabolites such as flavin adenine dinucleotide and xanthine, and metabolic pathway enrichment analysis found that CS was mainly involved in purine metabolism) and significantly up-regulating the expression level of the intestinal uric acid excretion transporter ATP-binding cassette subfamily G member 2 (ABCG 2) (Zhu et al., 2023); 3. CS demonstrated the ability to impede the progression of epithelial–mesenchymal transition (EMT) in renal tubular cells of rats with hyperuricemia nephropathy and HUA-HK 2 cells by inhibiting the NF-κB/Snail signaling pathway, thereby alleviating renal injury and showcasing significant anti-hyperuricemia and nephroprotective properties of CS (Wu et al., 2023).
Based on the preceding explanation, it is evident that the hyperoxic diterpenoids and phenolic acids in CS as effective components have shown promising potential for the treatment of hyperuricemia and gout via multi-target and multi-pharmacodynamic mechanisms.
4.6 Nephroprotective
The EE extract of CS demonstrated potent nephroprotective effect via lowering melamine in mice renal, impeding stone formation, and mitigating renal injury (Zhang J. et al., 2021). A variety of flavonoids in CS that exhibited robust renal protection effects, with trials demonstrating substantial reductions in proteinuria, serum creatinine, and urea nitrogen in rats with acute renal failure model, as well as decreased oxidative stress. The therapeutic effect of CS on acute renal failure may be related to the nephroprotective effect mediated by activation of the ERK/CT-1 pathway (Guo et al., 2019a). Additionally, these flavonoids stimulate renal tubular epithelial cell proliferation in acute renal failure, curtail apoptosis, modify oxidative stress levels, suppress pro-apoptotic protein expression, and enhance renal tubular epithelial cell repair in acute renal failure (Guo et al., 2019b).
4.7 Other effects
Two novel phenolic acid Helisterculins C and D (190 and 191), derived from CS in vitro, demonstrated neuroprotective potential against 6-OHDA-mediated SH-SY5Y cell death, with IC50 values of 17.4 and 21.3 μM, respectively (Zhou et al., 2017). In addition, research indicated that 2-caffeoyl-L-tartaric acid and rosmarinic acid outperformed acarbose in reducing α-glucosidase activity in vitro (Zhang, 2017). In clinical practice, CS potent therapeutics towards urinary tract stones, with EAE extract preventing kidney stones via mitigating oxidative stress and inflammation amplified by glycerophospholipid metabolism (Zhu et al., 2021).
The research mentioned above highlights how pharmacological studies on CS not only validate its traditional uses but also uncover its potential applications in treating other diseases. The key components in CS, such as diterpenoids, flavonoids, and phenolic acids, play a crucial role in its pharmacological effects. These compounds enable CS to exhibit multiple therapeutic targets and mechanisms in combating inflammation, oxidative stress, tumors, hyperuricemia, gout, and nephroprotective. However, while CS has shown promising anti-tumor effects in vitro, further research is needed to explore its efficacy in vivo and understand the underlying pharmacological mechanisms.
5 Quality control
The current and previous editions of the Chinese Pharmacopoeia exclude CS from their permissible content, and the quality control measures for CS have traditionally relied on outdated provincial or local criteria, leading to inconsistencies in quality control research due to the absence of standardized guidelines. In order to obtain stable and reliable quality control methods to promote the development and utilization of CS, researchers were committed to enhancing and refining essential technologies in quality control research, including qualitative identification, quantitative analysis, and fingerprint analysis.
5.1 Qualitative identification
Qualitative identification is a significant channel to ensure the correctness, excellent quality, safety and effectiveness of medicinal materials, and Morphological identification, microscopic identification and thin layer chromatography (TLC) identification are momentous means for qualitative identification of medicinal materials (Wang et al., 2022). Jiao et al. used traditional methods to identify the traits of CS, and observed the cross-section of leaves and stems and the microscopic characteristics of powder of CS by paraffin sectioning method, which provided more detailed and comprehensive characteristics for the identification of CS (Jiao and Feng, 2013). In the study, researchers employed danshensu, rosmarinic acid, and ursolic acid as marker ingredients for TLC identification, replacing the conventional reference materials typically used in this analytical technique. Through systematic investigation of single factors and adaptability of the system, a straightforward, practical, reproducible, and specific TLC identification method was developed for quality control of CS (Gu, 2023).
5.2 Quantitative analysis
Ursolic acid is one of the main chemical substances that play pharmacological roles in CS, and some preparations have recognized it as a reference material for quality testing and control. Huang and colleagues (Huang et al., 2019) determined the ursolic acid content in the alcohol extract of CS using HPLC under the following optimal conditions: mobile phase: acetonitrile-5% tetrahydrofuran with 0.1% phosphoric acid in a ratio of 68:32; wavelength: 203 nm; column temperature: 33°C; flow rate: 1 mL/min; injection volume: 20 μL. The average recovery was 101.11% (RSD = 2.04%). This technique demonstrated high resolution and reduced analysis time, thereby offering a scientific foundation for the quality assessment of ursolic acid in CS alcohol extracts. In a separate investigation, the levels of danshensu and rosmarinic acid in CS were analyzed using HPLC. The analysis employed a Venusil C18 column with a mobile phase gradient elution consisting of acetonitrile and a 0.1% phosphoric acid aqueous solution, flowing at a rate of 1 mL/min. Detection was performed at a wavelength of 210 nm, with the column maintained at 30°C and an injection volume of 5.0 μL. The quantification of danshensu and rosmarinic acid ranged from 1.75 to 56 μg and 15–480 μg, respectively, demonstrating a strong linear correlation. This straightforward, consistent, and dependable method offered valuable insights for the quality control assessment of CS medicinal materials (Gu, 2023).
Chinese materials display varying qualities depending on their origin, harvesting season, climate, and other variables, influencing drug efficacy, with establishing an appropriate quality standard is vital for Chinese herbal quality standards. A noteworthy finding revealed that the extracts of CS from the same region, which summer harvests contained higher levels of total polyphenols, total flavonoids, and antioxidant capacity, compared to autumn, while rosmarinic acid content varied depending upon region and season (Xue et al., 2016). Moreover, in order to evaluate the difference in quality between CS with purple corollas and white corollas, the researchers employed a colorimetric approach to ascertain the levels of total phenolic acids and total flavonoids. Furthermore, the quantities of caffeic acid, rosmarinic acid, and eupatorin were determined using HPLC. The results revealed that the levels of total phenolic acids, total flavonoids, and eupatorin were higher in CS with purple corollas compared to those CS with white corollas, while the quantities of caffeic acid and rosmarinic acid were found to be similar (Li et al., 2023).
Dao-di medicinal materials are high-quality Chinese herbal products, considered the first choice in Traditional Chinese Medicine (TCM) due to their historical significance and superior efficacy. CS primarily grows in Yunnan, Guangxi, Hainan, other southern regions of China, especially the CS which from Yunnan has always had a well reputation in traditional applications and modern clinical research. Through origin analysis, it has been determined that CS from Yunnan exhibits superior quality, most notably the CS which was from Xishuangbanna with highest rosmarinic acid content compared with other origins (Lan et al., 2017).
5.3 Fingerprint analysis
The fingerprint analysis technique in traditional Chinese medicine enables a comprehensive assessment of the chemical composition present in traditional Chinese medicine and its formulations, facilitating a detailed description and evaluation of the overall drug quality. In a research study, through statistical analysis of the fingerprints of 13 batches of CS medicinal materials, 12 common peaks were identified, and two common components (danshensu and rosmarinic acid) were identified by comparison with control products. The results of similarity analysis indicated that the similarity among the 13 batches of CS medicinal materials ranged from 0.698 to 0.997. Notably, the similarity among the fingerprints of 12 batches exceeded 0.970, with the exception of the S4 sample, which exhibited a similarity lower than 0.970 when compared to the control fingerprint (Gu, 2023). In recent times, there has been notable progress in ultra-performance liquid chromatography (UPLC) technology, enhancing the efficiency of liquid chromatography separations by elevating resolution, sensitivity, and analysis speed beyond what is achievable with conventional HPLC techniques. In accordance with this, Guo et al. (2019) conducted an examination of the resemblance among the predominant peaks present in 8 distinct batches of CS medicinal materials using the UPLC technique in conjunction with the “Chromatographic Fingerprint Similarity Evaluation System of Traditional Chinese Medicine 2004A Edition.” They established the fingerprints for these 8 batches of CS medicinal materials, identified 15 distinctive common peaks, and ascertained the constituents of 5 chromatographic peaks (caffeic acid, rosmarinic acid, salvianolic acid B, sinsensetin and eupatorine) through comparison with a reference substance. For the fingerprint analysis of volatile components in CS, Liu et al. (2016) utilized GC-MS in conjunction with programmed temperature retention index technology to examine 12 batches of CS samples. They established a volatile components fingerprint using GC-MS and identified 13 common chromatographic peak components, predominantly comprising alcohols, ketones, naphthalene, alkanes, acids, aldehydes, esters, and other compounds. Furthermore, the “Chromatographic Fingerprint Similarity Evaluation System Software for Traditional Chinese Medicine” (2004A Edition) was employed to conduct a more in-depth analysis of the fingerprint, revealing a similarity coefficient exceeding 0.999 among the 12 batches of CS samples. This approach offers a comprehensive depiction of the volatile components present in CS, characterized by its specificity, thus serving as a valuable tool for ensuring the quality control of volatile components in this medicinal material.
CS has demonstrated positive outcomes in the treatment of urinary system inflammation, gout, and various other medical conditions. Numerous clinical cases have supported its efficacy in treatment. Therefore, the implementation of standardized quality control measures is imperative in ensuring its safe and effective application. The current body of research provides some valuable insights for the quality control of CS to a limited extent. However, the overall quality control standards for CS are typically inadequate, primarily focusing on qualitative identification and content determination of a limited number of components. These compounds are not exclusive to CS, leading to a lack of credibility in contemporary quality control studies. Future research should prioritize qualitative identification and content determination of unique constituents within CS, while also considering the establishment of a quality evaluation and control system that integrates fingerprinting and multi-component content determination.
6 Clinical applications
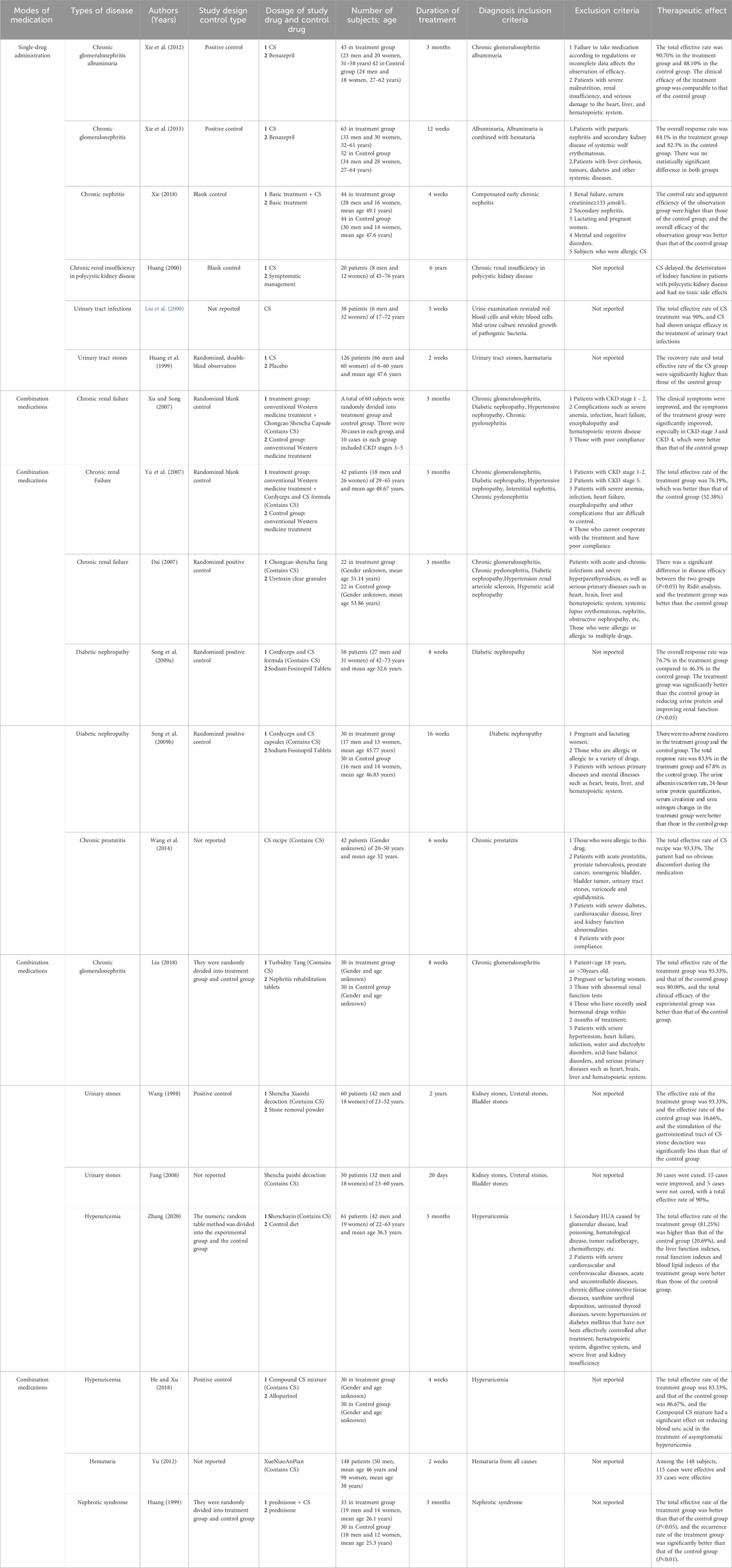
CS was prevalent in managing urinary tract stones, infections, nephrotic syndrome, renal failure, hyperuricemia, etc. Individually or in conjunction, it exhibited commendable efficacy, with minimal suspicious adverse reactions. The detailed data is delineated in Table 8.
7 Conclusions and future perspectives
This review summarized the most recent research on CS, focusing on its chemical constituents, pharmacological activity, quality control and clinical applications. The insights presented in this review serve as a valuable resource for guiding future investigations and advancements in this field. CS is a widely recognized herbal remedy in China and various Southeast Asian nations. It is extensively documented in ancient Chinese materia medica books, boasting a rich history of use and renowned efficacy in treating urological ailments. This reputation has piqued the curiosity of researchers worldwide, prompting comprehensive investigations into its properties and applications. Presently, over 300 compounds have been isolated from CS, expanding its pharmaceutical applications from the urinary system to various physiological systems and treatments for ailments such as the circulatory and nervous systems, with exhibiting notable pharmacological activities in anti-inflammatory, antibacterial, antioxidant, antitumor, anti-gout, renal protection, neuroprotection, and hypoglycemia, notably the active components such as total flavonoids, phenolic acids, and diterpenoids with high oxygen content, are hypothesized to be pivotal in anti-inflammatory, renal protection, neuroprotection, and urate-lowering activities. Preparations of traditional Chinese medicine that include CS, as well as CS on its own, have demonstrated positive outcomes in clinical interventions for conditions such as urinary tract infection, urinary tract stones, nephritis, renal insufficiency, chronic renal failure, diabetic nephropathy, hyperuricemia, and other related diseases. The favorable pharmacological activities and clinical efficacy can be attributed to the diverse therapeutic effects of compounds or constituents found in CS, which act on multiple targets and pathways.
Despite the considerable body of scientific research conducted on CS, there remain certain constraints that require immediate disposition. First of all, research on the phytochemical and pharmacological properties of CS has primarily concentrated on phenylpropanoids, flavonoids, and terpenoids, with limited emphasis on other compounds such as polysaccharides and saponins. Furthermore, existing pharmacological investigations have predominantly provided superficial assessments of drug efficacy, lacking in-depth exploration of the mechanisms underlying the anti-inflammatory, antibacterial, neuroprotective, and other pharmacological effects associated with CS. Hence, it is imperative to conduct further research on the pharmacodynamic material basis and mechanisms of action of CS in the treatment of significant medical conditions. This will establish a robust groundwork for CS as a therapeutic agent for a broader range of diseases. Secondly, despite the existence of numerous quality control studies focusing on CS, these studies often fail to select the signature components of CS as the control objects, thus rendering the current research on quality standards inconclusive. Given the intricate and diverse nature of the components found in CS, as well as their pharmacological activity being influenced by the synergistic effects of multiple components, a mere examination of individual components is insufficient for evaluating the quality of an active ingredient. Therefore, it is feasible to establish a quality evaluation and control system that integrates fingerprinting and multi-component content determination. Enhancing the quality control system for CS necessitates not only the thorough investigation by scholars in the field, but also the policy backing from relevant governmental bodies. This entails the development of unified and scientifically sound quality control standards through comprehensive and systematic research, as well as the timely inclusion of CS in the Chinese Pharmacopoeia. Such efforts are crucial for the advancement and effective utilization of CS. Moreover, clinicians predominantly rely on ancient Chinese materia medica books and personal experience when utilizing CS in patient treatment. While CS has demonstrated effectiveness, there remains a deficiency in comprehensive preclinical assessments of its safety and efficacy. Addressing this gap necessitates collaborative efforts across various disciplines and departments to establish a more robust scientific foundation supporting the secure, efficient, and consistent application of CS in clinical practice.
In anticipation of the future, it is recommended that pertinent governmental agencies enhance the promotion and policy supporting of CS to broaden its impact. Scholars are advised to draw inspiration from traditional Chinese medicine and ethnic medicinal practices, while integrating cutting-edge scientific technologies and research methodologies to explore more compounds with significant activity in CS and improve their pharmacodynamic mechanism research. Furthermore, it is imperative to conduct spectrum-effect relationship studies on CS, develop robust quality control criteria focusing on the active pharmaceutical ingredients and characteristic components present in CS. By integrating the quality of CS with its therapeutic efficacy to offer a more comprehensive theoretical and practical foundation for utilizing CS in disease prevention and treatment.
Author contributions
XJ: Investigation, Software, Writing–original draft. QJ: Project administration, Validation, Writing–review and editing. PZ: Conceptualization, Project administration, Resources, Supervision, Writing–review and editing. ZT: Conceptualization, Supervision, Validation, Writing–review and editing. LiL: Funding acquisition, Writing–review and editing. LuL: Funding acquisition, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This work was funded by the Major Science and Technology Special Project from Yunnan Provincial Science and Technology Department (202202AA100009, 202302AA310006), Key Laboratory of Yunnan Provincial Department of Education on Substance Benchmark Research of Ethnic Medicines (2022YGZ02), the Dai Medicine Key Discipline of Pharmacy State Administration of Traditional Chinese Medicine, and Open and Shared Public Science and Technology Service Platform of Traditional Chinese Medicine Science and Technology Resources in Yunnan for the financial support.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Abdelwahab, S. I., Mohan, S., Mohamed Elhassan, M., Al-Mekhlafi, N., Mariod, A. A., Abdul, A. B., et al. (2011). Antiapoptotic and antioxidant properties of Orthosiphon stamineus Benth (Cat’s Whiskers): intervention in the Bcl-2-mediated apoptotic pathway. Evid. Based Compl. Altern. Med. 2011, 156765. doi:10.1155/2011/156765
Ahamed, M. B., Aisha, A. F., Nassar, Z. D., Siddiqui, J. M., Ismail, Z., Omari, S. M., et al. (2012). Cat’s whiskers tea (Orthosiphon stamineus) extract inhibits growth of colon tumor in nude mice and angiogenesis in endothelial cells via suppressing VEGFR phosphorylation. Nutr. Cancer 64 (1), 89–99. doi:10.1080/01635581.2012.630160
Alshawsh, M. A., Abdulla, M. A., Ismail, S., Amin, Z. A., Qader, S. W., Hadi, H. A., et al. (2012). Free radical scavenging, antimicrobial and immunomodulatory activities of Orthosiphon stamineus. Molecules 17 (5), 5385–5395. doi:10.3390/molecules17055385
Awale, S., Tezuka, Y., Banskota, A. H., Adnyana, I. K., and Kadota, S. (2003a). Highly-oxygenated isopimarane-type diterpenes from Orthosiphon stamineus of Indonesia and their nitric oxide inhibitory activity. Chem. Pharm. Bull. 51 (3), 268–275. doi:10.1248/cpb.51.268
Awale, S., Tezuka, Y., Banskota, A. H., Adnyana, I. K., and Kadota, S. (2003b). Nitric oxide inhibitory isopimarane-type diterpenes from Orthosiphon stamineus of Indonesia. J. Nat. Prod. 66 (2), 255–258. doi:10.1021/np020455x
Awale, S., Tezuka, Y., Banskota, A. H., and Kadota, S. (2003). Siphonols A-E: novel nitric oxide inhibitors from Orthosiphon stamineus of Indonesia. Bioorg. Med. Chem. Lett. 13 (1), 31–35. doi:10.1016/s0960-894x(02)00854-5
Awale, S., Tezuka, Y., Banskota, A. H., and Kadota, S. (2003c). Inhibition of NO production by highly-oxygenated diterpenes of Orthosiphon stamineus and their structure-activity relationship. Biol. Pharm. Bull. 26 (4), 468–473. doi:10.1248/bpb.26.468
Awale, S., Tezuka, Y., Banskota, A. H., Kouda, K., Tun, K. M., and Kadota, S. (2001). Five novel highly oxygenated diterpenes of Orthosiphon stamineus from Myanmar. J. Nat. Prod. 64 (5), 592–596. doi:10.1021/np000607t
Awale, S., Tezuka, Y., Banskota, A. H., Kouda, K., Tun, K. M., and Kadota, S. (2002a). Four highly oxygenated isopimarane-type diterpenes of Orthosiphon stamineus. Planta Medica. 68 (3), 286–288. doi:10.1055/s-2002-23137
Awale, S., Tezuka, Y., Kobayashi, M., Ueda, J., and Kadota, S. (2004). Neoorthosiphonone A: a nitric oxide (NO) inhibitory diterpene with new carbon skeleton from Orthosiphon stamineus. ChemInform 35, 1359–1362. doi:10.1002/chin.200421169
Awale, S., Tezuka, Y., Shimoji, S., Taira, K., and Kadota, S. (2002b). Secoorthosiphols A-C: three highly oxygenated secoisopimarane-type diterpenoids from Orthosiphon stamineus. Tetrahedron Lett. 43 (8), 1473–1475. doi:10.1016/s0040-4039(02)00037-0
Bardin, T., and Richette, P. (2017). Impact of comorbidities on gout and hyperuricaemia: an update on prevalence and treatment options. BMC Med. 15 (1), 123. doi:10.1186/s12916-017-0890-9
Cardona-Trujillo, M. C., Jiménez-González, F. J., Veloza, L. A., and Sepúlveda-Arias, J. C. (2024). In vitro anti-toxoplasma activity of extracts obtained from tabebuia rosea and tabebuia chrysantha: the role of β-amyrin. Molecules 29 (5), 920. doi:10.3390/molecules29050920
Chang, K. F., Liu, C. Y., Huang, Y. C., Hsiao, C. Y., and Tsai, N. M. (2023). Downregulation of VEGFR2 signaling by cedrol abrogates VEGF-driven angiogenesis and proliferation of glioblastoma cells through AKT/P70S6K and MAPK/ERK1/2 pathways. Oncol. Lett. 26 (2), 342. doi:10.3892/ol.2023.13928
Chen, D. L., Long, H. M., Zhang, H. M., Li, N., and Liu, S. H. (2014). In vitro antioxidant and mitochondria protective activities of Clerodendranthus spicatus extracts. Nat. Prod. Res. Dev. 26 (03), 392–397. doi:10.16333/j.1001-6880.2014.03.021
Chen, T., and Yang, Q. W. (2016). Study on the protective effect of total flavonoids of Clerodendranthus Spicatus against chronic bacterial prostatitis in rats. J. Hubei Univ. Nationalities·Medical Ed. 33 (04), 1–3. doi:10.13501/j.cnki.42-1590/r.2016.04.002
Chen, W. D., Zhao, Y. L., Dai, Z., Zhou, Z. S., Zhu, P. F., Liu, Y. P., et al. (2020a). Bioassay-guided isolation of anti-inflammatory diterpenoids with highly oxygenated substituents from kidney tea (Clerodendranthus spicatus). J. food Biochem. 44 (12), e13511. doi:10.1111/jfbc.13511
Chen, W. D., Zhao, Y. L., Sun, W. J., He, Y. J., Liu, Y. P., Jin, Q., et al. (2020b). “Kidney tea” and its bioactive secondary metabolites for treatment of gout. J. Agric. food Chem. 68 (34), 9131–9138. doi:10.1021/acs.jafc.0c03848
Chen, X. F., Ma, G. X., Huang, Z., Wu, T. Y., Xu, X. D., and Zhong, X. M. (2017). Identification of water-soluble phenolic acids from Clerodendranthus spicatus. Chin. Traditional Herb. Drugs 48 (13), 2614–2618.
Chen, Y., Pei, C., Chen, Y., Xiao, X., Zhang, X., Cai, K., et al. (2023). Kidney tea ameliorates hyperuricemia in mice via altering gut microbiota and restoring metabolic profile. Chemico-biological Interact. 376, 110449. doi:10.1016/j.cbi.2023.110449
Chen, Y. L., Tan, J. J., Lu, L. L., Tan, C. H., Jiang, S. H., and Zhu, D. Y. (2009). Study on Water-soluble constituents of Clerodendranthus spicatus. Chin. Traditional Herb. Drugs 40 (05), 689–693. doi:10.3321/j.issn:0253-2670.2009.05.006
Cho, D. H., Yun, J. H., Heo, J., Lee, I. K., Lee, Y. J., Bae, S., et al. (2023). Identification of loliolide with anti-aging properties from Scenedesmus deserticola JD052. J. Microbiol. Biotechnol. 33 (9), 1250–1256. doi:10.4014/jmb.2304.04044
Dai, L. J. (2007). Influence of microinflamtion and clinical study on treating non-dialysis chronic renal failure patients with ChongcaoShenchaFang. Dissertation thesis. Harbing: Heilongjiang University of Chinese Medicine.
Fan, F. F., Li, X. B., Qiu, M. F., Zhang, L. L., Zhao, Y. H., Wang, M. Y., et al. (2013). Chemical studies on the N-butanol extract of Dai medicine “Yanuomiao,” the whole plant of Clerodendranthus Spicatus. Adv. Mod. Biomed. Sci. 13 (32), 6227–6230+6254. doi:10.13241/j.cnki.pmb.2013.32.006
Fang, S. Y. (2008). 50 cases of urinary calculi were treated with self-simulated Shencha paishi decoction. Inn. Mong. J. Tradit. Chin. Med. (08), 24.
Gu, Y. Z. (2023). Preliminary study on chemical constituents and quality standard of Clerodendranthus spicatus. Dissertation thesis. Nanchang: Jiangxi University of Chinese Medicine. doi:10.27180/d.cnki.gjxzc.2023.000355
Guo, Y. X., Ge, P. Y., and Hu, M. R. (2019a). Effects of Clerodendranthus spicatus flavonoids on oxidative stress in rats with acute renal failure through erk/ct-1 pathway. World Chin. Med. 15 (18), 2700–2703. doi:10.3969/j.issn.1673-7202.2020.18.006
Guo, Y. X., Hu, M. R., and Ge, P. Y. (2019b). Study on the protective effect of Clerodendranthus spicatus flavone on renal tubular epithelial cells in acute renal failure. Mod. Traditional Chin. Med. Materia Materia-World Sci. Technol. 22 (06), 1773–1779. doi:10.11842/wst.20191125003
Guo, Z. L. (2020). Multicomponent analysis of Orthosiphon stamineus Benth. By LC-MS technologies and pharmacokinetics of phenols. Master’s thesis. Hangzhou: Zhejiang University of Technology. doi:10.27463/d.cnki.gzgyu.2020.000021
Guo, Z. L., Xia, J. J., Han, N., Gu, J. P., Su, F., Liang, X. R., et al. (2019). Establishment of Clerodendranthus spicatus UPLC fingerprint and evaluation of its antioxidant activity. Bull. Ferment. Sci. Technol. 49 (04), 187–191. doi:10.16774/j.cnki.issn.1674-2214.2020.04.001
He, B. Y., and Xu, J. J. (2018). Study on the intervention of compound Clerodendranthus spicatus mixture on asymptomatic hyperuricemia. Abstract compilation of papers of the 2018 Academic Annual Meeting of the Kidney Disease Professional Committee of the Chinese Association of Integrative Medicine 1.
Hossain, M. A., and Ismail, Z. (2005). A new lupene-type triterpene from the leaves of Orthosiphon stamineus. Indian J. Chem. 44B, 436–437. doi:10.1002/chin.200525181
Hou, Z. Y., Wang, L. Q., and Liang, Z. S. (2011). Recent research progress on pharmacological effects of extracts of Clerodendranthus spicatus. China Med. Pharm. 1 (02), 26–27+30.
Hu, H., Li, W., Hao, Y., Peng, Z., Zou, Z., and Liang, W. (2024). Baicalin ameliorates renal fibrosis by upregulating CPT1α-mediated fatty acid oxidation in diabetic kidney disease. Phytomedicine 122, 155162. doi:10.1016/j.phymed.2023.155162
Huang, B. (1999). Clinical observations on treatment with Clerodendranthus spicatus combined with prednison for 33 cases with nephrotic syndrome. Chinese Journal of Integrated Traditional and Western Medicine in Intensive and Critical Care (12), 550–551. doi:10.3321/j.issn:1008-9691.1999.12.008
Huang, K. M. (2000). Clinical application of Clerodendranthus spicatus in the treatment of chronic renal insufficiency in polycystic kidney disease. Chin. J. Dis. Control Prev. (01), 19. doi:10.3969/j.issn.1674-3679.2000.01.037
Huang, R. G., Shen, W. T., Zheng, Z. X., and Xu, Y. R. (1999). Therapeutic effect of Clerodendranthus spicatus on urinary tract stones. Journal of Fujian Medical University, (01), 402–405. doi:10.3969/j.issn.1672-4194.1999.04.013
Huang, T. Z., Ding, W. N., Zhang, L. J., Zheng, H. R., and He, X. L. (2019). Establishment of HPLC analytical method for ursolic acid in alcohol extract of Clerodendranthus spicatus and its methodology. Shandong Chem. Ind. 49 (19), 93–95+97. doi:10.19319/j.cnki.issn.1008-021x.2020.19.038
Jiao, A. J., and Feng, J. (2013). Pharmacognosy identification of Clerodendranthus spicatus. J. Guangxi Med. Univ. 30 (02), 190–191. doi:10.16190/j.cnki.45-1211/r.2013.02.005
Lan, L. L., Fan, Q. H., Cao, C., Liu, M. C., Zou, X. H., Zeng, Y. E., et al. (2017). Determining the contents of rosemary acid and caffeic acid in Dai medicine Clerodendranthus spicatus and developing its fingerprint. China J. Traditional Chin. Med. Pharm. 32 (06), 2740–2745.
Lee, S. H., Moon, B. H., Park, Y. H., et al. (2008). Methyl substitution effects on 1H and 13C NMR data of methoxyflavones. Bulletin-Korean Chem. Soc. 29 (9), 1793–1796. doi:10.5012/bkcs.2008.29.9.1793
Li, Q. (2020). Study on the chemical constituents and their biological activity of the medicinal plant Clerodendranthus spicatus. Dissertation thesis. Wuhan: South-Central University for Nationalities. doi:10.27710/d.cnki.gznmc.2020.000490
Li, Q., He, Y. N., Shi, X. W., Kang, L. Y., Niu, L. Y., Wang, X. G., et al. (2016). Clerodens E-J, antibacterial caffeic acid derivatives from the aerial part of Clerodendranthus spicatus. Fitoterapia 114, 110–114. doi:10.1016/j.fitote.2016.08.021
Li, X. H., Li, H. T., Nie, Q., Li, X. L., Lin, Y. F., Zheng, M. Y., et al. (2023). Morphological characteristics and quality comparison of Clerodendranthus spicatus with purple corollas and white corollas. J. Chin. Med. Mater. 46 (06), 1381–1386. doi:10.13863/j.issn1001-4454.2023.06.010
Li, X. Z., Yan, Y. M., and Cheng, Y. X. (2017). Compounds from Clerodendranthus spicatus. Nat. Prod. Res. Dev. 29 (02), 183–189. doi:10.16333/j.1001-6880.2017.2.001
Li, Y. M., Xiang, B., Li, X. Z., Yan, Y. M., and Cheng, Y. X. (2017). New Diterpenoids from Clerodendranthus spicatus. Nat. Prod. Bioprospect. 7 (3), 263–267. doi:10.1007/s13659-017-0128-8
Li, Y. P., Pan, Z. H., Fu, Y. X., Ning, D. S., Li, L. C., and Hai, H. (2022). Anti-inflammatory constituents of Clerodendranthus spicatus. Guihaia 42 (09), 1480–1486. doi:10.11931/guihaia.gxzw202108015
Li, Y. T., Huang, R. G., and Zheng, X. Z. (2002). Research progress of Clerodendranthus spicatus. Chin. J. Integr. Tradit. West. Med. 22 (06), 470–472. doi:10.7661/CJIM.2002.6.470
Liu, B., Liu, G. L., Li, Y. W., Mou, L. L., Fang, H., Zhang, N., et al. (2016). GC-MS Fingerprint of Clerodendranthus spicatus. Chin. J. Exp. Traditional Formulae 22 (13), 59–62. doi:10.13422/j.cnki.syfjx.2016130059
Liu, M. J. (2018). The clinical observation of descending turbidity tang treatment on the type of spleen-kidney qi deficiency and dampness-heat of chronic glomerulonephritis. Dissertation thesis. Kun Ming: Yunnan University of Chinese Medicine.
Liu, Y., and Qi, L. H. (1999). Clerodendranthus spicatus treated urinary tract infections in 38 cases. Fujian J. Tradit. Chin. Med. (05), 46–47.
Long, H. M., Luo, Y., Cheng, H. Y., Xu, M. J., Wang, X. C., and Yan, J. (2017). Study on anti-cancer activity of flavonoids of Clerodendranthus Spicatus. J. Gannan Med. Univ. 37 (02), 179–184. doi:10.3969/j.issn.1001-5779.2017.02.002
Luo, Y. (2017). Chemical compositions from Clerodendranthus spicatus. Dissertation thesis. Luzhou: Southwest Medical University. doi:10.3969/j.issn.1007-6883.2013.03.011
Luo, Y., Cheng, L. Z., Luo, Q., Yan, Y. M., Wang, S. M., Sun, Q., et al. (2017). New ursane-type triterpenoids from Clerodendranthus spicatus. Fitoterapia 119, 69–74. doi:10.1016/j.fitote.2017.04.001
Luo, Y., Li, X. Z., Xiang, B., Luo, Q., Liu, J. W., Yan, Y. M., et al. (2018). Cytotoxic and renoprotective diterpenoids from Clerodendranthus spicatus. Fitoterapia 125, 135–140. doi:10.1016/j.fitote.2018.01.003
Luo, Y. X., Gong, X., Su, Z. C., Mo, J. F., Li, D. L., Wu, R. H., et al. (2024). Isolation, structure modification, and anti-rheumatoid arthritis activity of isopimarane-type diterpenoids from Orthosiphon aristatus. J. enzyme inhibition Med. Chem. 39 (1), 2296355. doi:10.1080/14756366.2023.2296355
Ma, G. X., Zhang, X. P., Li, P. F., Sun, Z. H., Zhu, N. L., Zhu, Y. D., et al. (2015). Four new phenolic acid with unusual bicycle [2.2.2] octane moiety from Clerodendranthus spicatus and their anti-inflammatory activity. Fitoterapia 105, 61–65. doi:10.1016/j.fitote.2015.06.010
Ma, Z. Y. (2013). Chemical Constituents of Volatile oil from Clerodendranthus spicatus. J. Hanshan Normal Univ. 34 (03), 50–54+59.
Malterud, K. E., Hanche-Olsen, I. M., and Smith-Kielland, I. (1989). Flavonoids from Orthosiphon spicatus. Planta Medica. 55 (6), 569–570. doi:10.1055/s-2006-962099
Mlala, S., Oyedeji, A. O., Gondwe, M., and Oyedeji, O. O. (2019). Ursolic acid and its derivatives as bioactive agents. Molecules 24 (15), 2751. doi:10.3390/molecules24152751
Nagao, T., Abe, F., Kinjo, J., and Okabe, H. (2002). Antiproliferative constituents in plants 10. Flavones from the leaves of Lantana montevidensis Briq. and consideration of structure-activity relationship. Biol. Pharm. Bull. 25 (7), 875–879. doi:10.1248/bpb.25.875
Nguyen, M. T., Awale, S., Tezuka, Y., Chien-Hsiung, C., and Kadota, S. (2004). Staminane- and isopimarane-type diterpenes from Orthosiphon stamineus of Taiwan and their nitric oxide inhibitory activity. J. Nat. Prod. 67 (4), 654–658. doi:10.1021/np030471+
Nuengchamnong, N., Krittasilp, K., and Ingkaninan, K. (2011). Characterisation of phenolic antioxidants in aqueous extract of Orthosiphon grandiflorus tea by LC-ESI-MS/MS coupled to DPPH assay. Food Chem. 127 (3), 1287–1293. doi:10.1016/j.foodchem.2011.01.085
Ohashi, K., Bohgaki, T., Matsubara, T., and Shibuya, H. (2000). Indonesian medicinal plants. XXIII. Chemical structures of two new migrated pimarane-type diterpenes, neoorthosiphols A and B, and suppressive effects on rat thoracic aorta of chemical constituents isolated from the leaves of Orthosiphon aristatus (Lamiaceae). Chem. Pharm. Bull. 48 (3), 433–435. doi:10.1248/cpb.48.433
Rocha, V., Quadros, H., Meira, C., Silva, L., Carvalho, D., Hodel, K., et al. (2022). Potential of triterpenic natural compound betulinic acid for neglected tropical diseases new treatments. Biomedicines 10 (4), 831. doi:10.3390/biomedicines10040831
Song, L. Q., Jin, L. X., and Song, Y. X. (2009b). The clinical study of the effect of Diabetic nephropathy treated by Chongcao Shencha Capsul. Chin. Archives Traditional Chin. Med. 27 (04), 679–681.
Song, L. Q., Pei, C. P., and Song, Y. X. (2009a). The clinical study of ChongcaoShenchaFang in the treatment of diabetic nephropathy. Inf. Traditional Chin. Med. 26 (04), 38–39.
Stampoulis, P., Tezuka, Y., Banskota, A. H., Tran, K. Q., Saiki, I., and Kadota, S. (1999). Staminolactones A and B and norstaminol A: three highly oxygenated staminane-type diterpenes from Orthosiphon stamineus. Org. Lett. 1 (9), 1367–1370. doi:10.1021/ol990216+
Su, Y., Li, B., Liang, W., Wen, H., and Wei, W. (2024). The complete chloroplast genome of Orthosiphon aristatus (Blume) Miq. (Lamiaceae). Mitochondrial DNA. Part B, Resour. 9 (1), 79–82. doi:10.1080/23802359.2023.2301012
Sumaryono, W., Proksch, P., Wray, V., Witte, L., and Hartmann, T. (1991). Qualitative and quantitative analysis of the phenolic constituents from Orthosiphon aristatus. Planta Medica. 57 (2), 176–180. doi:10.1055/s-2006-960060
Sun, Z. C., Zheng, Q. X., Ma, G. X., Zhang, X. P., Yuan, J. Q., Wu, H. F., et al. (2014a). Four new phenolic acids from Clerodendranthus spicatus. Phytochem. Lett. 8, 16–21. doi:10.11669/cpj.2014.01.006
Sun, Z. C., Zheng, Q. X., Wu, H. F., Ma, G. X., Xu, X. D., and Yang, J. S. (2014b). Water-soluble Constituents of Clerodendranthus spicatus. Chin. Pharm. J. 49 (01), 22–25.
Takeda, Y., Matsumoto, T., Terao, H., Terao, H., Futatsuishi, Y., Nohara, T., et al. (1993). Orthosiphol D and E, monior diterpenes from Orthosiphon stamineus. Phytochemistry 33 (2), 411–415. doi:10.1016/0031-9422(93)85530-5
Tan, J. J., Tan, C. H., Chen, Y. L., Jiang, S. H., and Zhu, D. Y. (2009). Chemical constituents of Clerodendranthus spicatus. Nat. Prod. Res. Dev. 21 (04), 608–611+592. doi:10.16333/j.1001-6880.2009.04.033
Tezuka, Y., Stampoulis, P., Banskota, A. H., Awale, S., Tran, K. Q., Saiki, I., et al. (2000). Constituents of the Vietnamese medicinal plant Orthosiphon stamineus. Chem. Pharm. Bull. 48 (11), 1711–1719. doi:10.1248/cpb.48.1711
Wang, C., Wang, J., Gao, M., Gao, P., Gao, D., Zhang, H., et al. (2022). Radix Ranunculi ternati: review of its chemical constituents, pharmacology, quality control and clinical applications. J. Pharm. Pharmacol. 74 (7), 930–952. doi:10.1093/jpp/rgac018
Wang, C., Xia, L. Y., Zhang, J. J., Wang, J. X., Li, W., and Gao, X. M. (2014). A clinical report on the treatment of chronic prostatitis with Clerodendranthus spicatus formula. Pharm. Clin. Chin. Materia Medica. 5 (05), 43–45.
Wang, L., Zhang, X., Li, Y. X., Xu, L. Q., Li, C. L., Zhang, Z. B., et al. (2016). Aqueous extract of Clerodendranthus spicatus exerts protective effect on uv-induced photoaged mice skin. Evidence-Based Compl. Altern. Med. 2016, 9623957. doi:10.1155/2016/9623957
Wang, M., Liang, J. Y., and Chen, X. Y. (2007). Water-soluble components of Clerodendranthus Spicatus. Chin. J. Nat. Med. (01), 27–30.
Wang, Q., Li, N., Wang, Y., Li, R., Jia, Y., Zhou, J., et al. (2023). Studies on the key constituents and the related mechanisms of Clerodendranthus spicatus in the treatment of diabetes based on network pharmacology. J. Ethnopharmacol. 303, 115949. doi:10.1016/j.jep.2022.115949
Wang, Q. Q. (1998). Clinical report on the treatment of urinary stones by Shencha Xiaoshi decoction. Zhejiang J. Integr. Traditional Chin. West. Med. (04), 206.
Wu, S., Yan, M., Liu, J., Li, Y., Tian, R., Li, C., et al. (2023). Clerodendranthus spicatus inhibits epithelial-mesenchymal transition of renal tubular cells through the NF-κB/Snail signalling pathway in hyperuricaemia nephropathy. Pharm. Biol. 61 (1), 1274–1285. doi:10.1080/13880209.2023.2243086
Xie, L. P., Lan, F., Xiang, C. C., Shi, W., Xie, Y. X., and Tao, Z. H. (2013). Clinical observation of 63 cases of Clerodendranthus spicatus in the treatment of chronic glomerulonephritis. Guangxi J. Traditional Chin. Med. 6 (05), 29–31. doi:10.3969/j.issn.1003-0719.2013.05.012
Xie, L. P., Xiang, C. C., Shi, W., Xie, Y. X., and Tang, Y. (2012). A clinical study of 43 cases of Clerodendranthus spicatus in the treatment of proteinuria in chronic glomerulonephritis. Jiangsu J. Traditional Chin. Med. 44 (02), 21–22. doi:10.3969/j.issn.1672-397X.2012.02.014
Xie, Q. (2018). Effects of Clerodendranthus spicatus on intraglomerular C4d deposition and serum hepatocyte growth factor expression in patients with chronic nephritis. Chin. J. Integr. Traditional West. Nephrol. 19 (07), 604–606.
Xu, H., Zhao, X., Liu, X., Xu, P., Zhang, K., and Lin, X. (2015). Antitumor effects of traditional Chinese medicine targeting the cellular apoptotic pathway. Drug Des. Dev. Ther. 9, 2735–2744. doi:10.2147/DDDT.S80902
Xu, W., Wang, J., Ju, B., Lan, X., Ying, X., and Stien, D. (2022). Seven compounds from Portulaca oleracea L. and their anticholinesterase activities. Nat. Prod. Res. 36 (10), 2547–2553. doi:10.1080/14786419.2021.1916928
Xu, W. H., Wang, H. T., Sun, Y., Xue, Z. C., Liang, M. L., and Su, W. K. (2020). Antihyperuricemic and nephroprotective effects of extracts from Orthosiphon stamineus in hyperuricemic mice. J. Pharm. Pharmacol. 72 (4), 551–560. doi:10.1111/jphp.13222
Xu, Y. Q., and Song, L. Q. (2007). Observation of the clinical efficacy of ChongcaoShenchaFang in the treatment of different stages of chronic kidney disease. Chin. J. Integr. Traditional West. Nephrol. (07), 415–416. doi:10.3969/j.issn.1009-587X.2007.07.018
Xue, H. Q., Cai, X., Xiong, H. H., Hang, Y. Q., Xiao, C. F., and Lu, L. H. (2016). Nain nutrients and antioxidant capacity of Orthosiphon stamineus in different parts. Acta Agric. Shanghai 32 (03), 30–35. doi:10.15955/j.issn1000-3924.2016.03.06
Yam, M. F., Basir, R., Asmawi, M. Z., and Ismail, Z. (2007). Antioxidant and hepatoprotective effects of Orthosiphon stamineus Benth. standardized extract. Am. J. Chin. Med. 35 (1), 115–126. doi:10.1142/S0192415X07004679
Yan, R., Liu, L., Huang, X., Quan, Z. S., Shen, Q. K., and Guo, H. Y. (2024). Bioactivities and structure-activity relationships of maslinic acid derivatives: a review. Chem. Biodivers. 21 (2), e202301327. doi:10.1002/cbdv.202301327
Yehya, A. H. S., Subramaniam, A. V., Asif, M., Kaur, G., Abdul Majid, A. M. S., and Oon, C. E. (2022). Anti-tumour activity and toxicological studies of combination treatment of Orthosiphon stamineus and gemcitabine on pancreatic xenograft model. World J. Gastroenterol. 28 (32), 4620–4634. doi:10.3748/wjg.v28.i32.4620
Yi, F., He, Y. J., Liang, K., and Wang, Z. Q. (2013). Bacteriostatic experiment for aqueous extract of Clerodendranthus spicatus in vitro. Med. J. Natl. Defending Forces Southwest China 23 (10), 1058–1059. doi:10.3969/j.issn.1004-0188.2013.10.005
Yu, S. M., Guo, D. D., and Song, L. Q. (2007). Clinical observation of ChongcaoShenchaFang in the treatment of chronic renal failure in 42 cases. Acta Chin. Med. Pharmacol. (06), 44–46. doi:10.19664/j.cnki.1002-2392.2007.06.020
Yu, X. J. (2012). XueNiaoAnPian treatment of hematuria was clinically observed in 148 cases. China Health Care Nutr. 22 (20), 4633. doi:10.3969/j.issn.1004-7484(x).2012.11.493
Yuliana, N. D., Khatib, A., Link-Struensee, A. M., Ijzerman, A. P., Rungkat-Zakaria, F., Choi, Y. H., et al. (2009). Adenosine A1 receptor binding activity of methoxy flavonoids from Orthosiphon stamineus. Planta Medica. 75 (2), 132–136. doi:10.1055/s-0028-1088379
Zhang, H. L. (2019). Study on chemical constituents and biological activities of ethyl acetate extract of Clerodendranthus spicatus cultivated in Hainan. Dissertation thesis. Haikou: Hainan University.
Zhang, J. Q., Huang, R. G., Zheng, X. Z., and Lin, Z. M. (2021). Effects of Clerodendranthus spicatus Extracts on Melamine-induced Kidney Stones Model in Mice. Straits Pharm. 33 (10), 4–7. doi:10.3969/j.issn.1006-3765.2021.10.002
Zhang, L. (2020). The clinical observation of shenchayin in the treatment of hyperuricemia and the investigation of patients′ dietary habits. Dissertation thesis. Kun Ming: Yunnan University of Chinese Medicine.
Zhang, R. R. (2017). Study on Chemical constituents and biological activities of cultivated Clerodendranthus spicatus in Hainan. Dissertation thesis. Haikou: Hainan University.
Zhang, R. R., Mei, W. L., Huang, S. Z., Li, W., Zhou, L. M., Zhao, Y. X., et al. (2017). Chemical constituents from the cultivated Clerodendranthus spicatus (Thunb.) C. Y. Wu in Hainan. J. Trop. Subtropical Bot. 25 (02), 182–188.
Zhang, X., Li, G., Deng, Q., Xu, Z., Cen, J., and Xu, J. (2021). Vomifoliol isolated from mangrove plant Ceriops tagal inhibits the NFAT signaling pathway with CN as the target enzyme in vitro. Bioorg. Med. Chem. Lett. 48, 128235. doi:10.1016/j.bmcl.2021.128235
Zhang, Y. Q., Fu, Y., Ruan, J., Gao, Q., Yang, D., Wang, D., et al. (2023). Highly oxygenated diterpenoids in Clerodendranthus spicatus and their bioactivity, A review. Biochem. Syst. Ecol. 106, 104580. doi:10.1016/j.bse.2022.104580
Zhang, Y. Y., Wu, J. C., Li, S. P., Li, N. L., Huang, Y. F., and Lin, Q. H. (2021). Research progress on chemical constituents and pharmacological actions of Clerodendranthus spicatus. Acta Chin. Med. Pharmacol. 49 (01), 112–120. doi:10.19664/j.cnki.1002-2392.210025
Zhao, A. H., Zhao, Q. S., Li, R. T., and Sun, H. D. (2004). Chemical constituents from Clerodendranthus spicatus. Yunnan Botanical Res. (05), 563–568.
Zheng, Q., Sun, Z., Zhang, X., Yuan, J., Wu, H., Yang, J., et al. (2012). Clerodendranoic acid, a new phenolic acid from Clerodendranthus spicatus. Molecules 17 (11), 13656–13661. doi:10.3390/molecules171113656
Zheng, Y. H., Pan, X. M., and Li, L. T. (2019). Study on anti-tumor active parts in vitro of the extracts from Clerodendranthus spicatus. J. Anhui Agric. Sci. 48 (12), 177–179.
Zhong, J. Y., and Wu, Z. S. (1984). Chemical constituents from Clerodendranthus spicatus. Yunnan Botanical Res. (03), 344–345.
Zhou, H. C., Yang, L., Guo, R. Z., and Li, J. (2017). Phenolic acid derivatives with neuroprotective effect from the aqueous extract of Clerodendranthus spicatus. J. Asian Nat. Prod. Res. 19 (10), 974–980. doi:10.1080/10286020.2016.1277707
Zhu, C., Niu, H., Bian, M., Zhang, X., Zhang, X., and Zhou, Z. (2023). Study on the mechanism of Orthosiphon aristatus (Blume) Miq. in the treatment of hyperuricemia by microbiome combined with metabonomics. J. Ethnopharmacol. 317, 116805. doi:10.1016/j.jep.2023.116805
Zhu, C., Niu, H., Nie, A., and Bian, M. (2021). Bioactivity-guided separation of potential α-glycosidase inhibitor from clerodendranthus spicatus based on HSCCC coupled with molecular docking. Sci. Rep. 11 (1), 6914. doi:10.1038/s41598-021-86379-9
Zou, J., Zhu, Y. D., and Zhao, W. M. (2008). Two new alkyl glycosides from Clerodendranthus spicatus. J. Asian Nat. Prod. Res. 10 (7-8), 603–607. doi:10.1080/10286020802133076
Glossary
ABCG 2 ATP-Binding Cassette subfamily G member 2
ADA Adenosine Deaminase
BCB β-carotene Bleaching
CE Chloroform Extract
CS Clerodendranthus spicatus
CBM Chinese Biomedical Database
CN Calcineurin
CNKI China National Knowledge Infrastructure
CKD Chronic Kidney Disease
DPPH 2,2-diphenyl-1-picrylhydrazyl
EAF Ethyl Acetate Fraction
EAE Ethyl Acetate Extract
EE Ethanol Extract
EMT Epithelial–Mesenchymal Transition
GC-MS Gas Chromatography–Mass Spectrometry
GLUT 9 Glucose Transporter 9
HUA Hyperuricemia
HPLC High Performance Liquid Chromatography
L-NMMA NG-Monomethyl-L-Arginine
MDA Malondialdehyde
NFAT Nuclear Factor of Activated T-cells
NO Nitric Oxide
OAT 1 Organic Anion Transporter 1
OAT 3 Organic Anion Transporter 3
OSE Orthosiphon stamineus Ethyl acetate extract
PE Petroleum Ether extract
PO Potassium Oxazinate
TCM Traditional Chinese Medicine
TLC Thin Layer Chromatography
UPLC Ultraperformance Liquid Chromatography
UA Ursolic Acid
URAT 1 Urate Transporter 1
WE Water Extract
XOD Xanthine Oxidase
Keywords: Clerodendranthus spicatus, chemical constituents, pharmacology, review, quality control, clinical application
Citation: Jiao X, Jin Q, Zhu P, Tan Z, Li L and Liu L (2025) Clerodendranthus spicatus: a comprehensive review of the chemical constituents, pharmacology, quality control and clinical applications. Front. Pharmacol. 16:1452797. doi: 10.3389/fphar.2025.1452797
Received: 21 June 2024; Accepted: 21 February 2025;
Published: 13 March 2025.
Edited by:
Tsong-Long Hwang, Chang Gung University of Science and Technology, TaiwanReviewed by:
Ritesh Raju, Western Sydney University, AustraliaMohamed Ezzat Abouelela, Al-Azhar University, Egypt
Copyright © 2025 Jiao, Jin, Zhu, Tan, Li and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lu Liu, dG9kYXlsaXVsdUAxNjMuY29t; Li Li, Njc1Njk3NTA5QHFxLmNvbQ==
 Xingmeng Jiao1
Xingmeng Jiao1 Zhuomin Tan
Zhuomin Tan Lu Liu
Lu Liu