- 1Department of Biology, American University of Beirut, Beirut, Lebanon
- 2Department of Nutrition, University of Petra, Amman, Jordan
- 3CNRS, Centrale Marseille, iSm2, Aix-Marseille University, Marseille, France
Anchusa strigosa Banks and Sol. is a rough flowering plant of the Boraginaceae family native to Eastern Mediterranean region that is widely used in traditional herbal medicine, mainly for the treatment of wounds, abdominal pain, and arthritis, to name a few. This article aims to gather knowledge related to the medicinal properties of A. strigosa. Specifically, it summarizes its traditional uses and pharmacological activities in the treatment of various diseases. Moreover, its botanical, ecological, and phytochemical characteristics are also discussed. Research showed that this plant is rich in pyrrolizidine alkaloids, particularly in the leaves. Other bioactive metabolites identified in this plant include flavonoids, phenolic acids, triterpenes, organic acids, and volatile organic compounds. These phytochemicals are responsible for the reported pharmacological properties of A. strigosa, including antimicrobial, antioxidant, anticancer, anti-inflammatory, antiarthritic, gastric protective, antidiabetic, and pro-wound healing. This warrants further investigation into the molecular mechanism of action behind the observed effects to elucidate its therapeutic potential. Nevertheless, more research on this plant is needed to ensure its efficacy and safety.
1 Introduction
Historically, plants and natural products have been widely used in folk medicine as traditional remedies. Moreover, their use in primary healthcare has been expanding rapidly, with complementary and alternative medicine becoming mainstream in both developing and developed countries due to the wide acceptance of natural remedies and their perception as generally safe. Now, plant-based natural products play an important role in modern drug development, owing to the diversity and structural complexity of their metabolites and their unique properties. In fact, plants have contributed to the development of many drugs either directly or indirectly by using the core structure of natural bioactive metabolites as scaffolds. For example, morphine, which is found in Papaver somniferum, was the first natural product introduced and used as a therapeutic drug in 1826. And aspirin was developed as a semi-synthetic drug in 1899 to treat pain, fever, and inflammation, as a derivative of salicilin from Salix alba. Other examples of plant-derived drugs include Paclitaxel from Taxus brevifolia, which is chemotherapeutic agent used for the treatment of various cancers, and Artemisinin from Artemisia annua, used for the treatment of multidrug-resistant malaria, to name a few.
Anchusa is a major genus of rough flowering plants that belongs to the Boraginaceae family, with around 34 accepted species growing mainly in a temperate biome. It is native to Europe, the Middle East and North Africa region, Western Asia, and South Africa (Inflammation, 2023). Species of this genus have been used in traditional medicine for the treatment of various ailments (Al-Snafi, 2014), including arthritis (Lev and Amar, 2000; El Beyrouthy et al., 2008; Polat et al., 2013), abdominal pain (Al-Khalil, 1995; Polat et al., 2013; Mükemre et al., 2015), kidney stones (Mordi et al., 2021), and for wound healing (Honda et al., 1996; Lardos, 2006; Hudaib et al., 2008; Qasem, 2015).
A. officinalis L., A. strigosa Banks and Sol., and A. azurea Mill. are the most studied species of this genus, with insight given into their ethnobotanical uses and their phytochemical and pharmacological properties. Of particular interest to this review is A. strigosa, where we provide an up-to-date and comprehensive overview of its phytochemical properties of A. strigosa and pharmacological activities, with the aim to expose its potential as an attractive source of medicinal agents and call for further investigation into its therapeutic value.
2 Methods
Literature search was conducted using the keywords and MeSH terms ‘Anchusa strigosa Banks & Sol.’2023, “A. strigosa,” AND (“phytochemical content,” “pharmacological properties, or activities, or effects, or roles,” “anti-inflammatory,” “antioxidant,” “anticancer,” “ethnopharmacology,” “traditional uses,” “medicinal uses,” “antimicrobial,” “antibacterial,” or “antifungal”) in major scientific literature databases such as PubMed, Scopus, ScienceDirect, SciFinder, Medicinal and Aromatic Plants Abstracts, Dr. Duke’s Phytochemical and Ethnobotanical Databases, Chemical Abstracts, and Henriette′s Herbal Homepage. Google and Google Scholar were also used for general web searches. The search period covered articles published between 1984 and 2023. The search yielded 23 research articles on A. strigosa.
3 Ehtnopharmacological uses
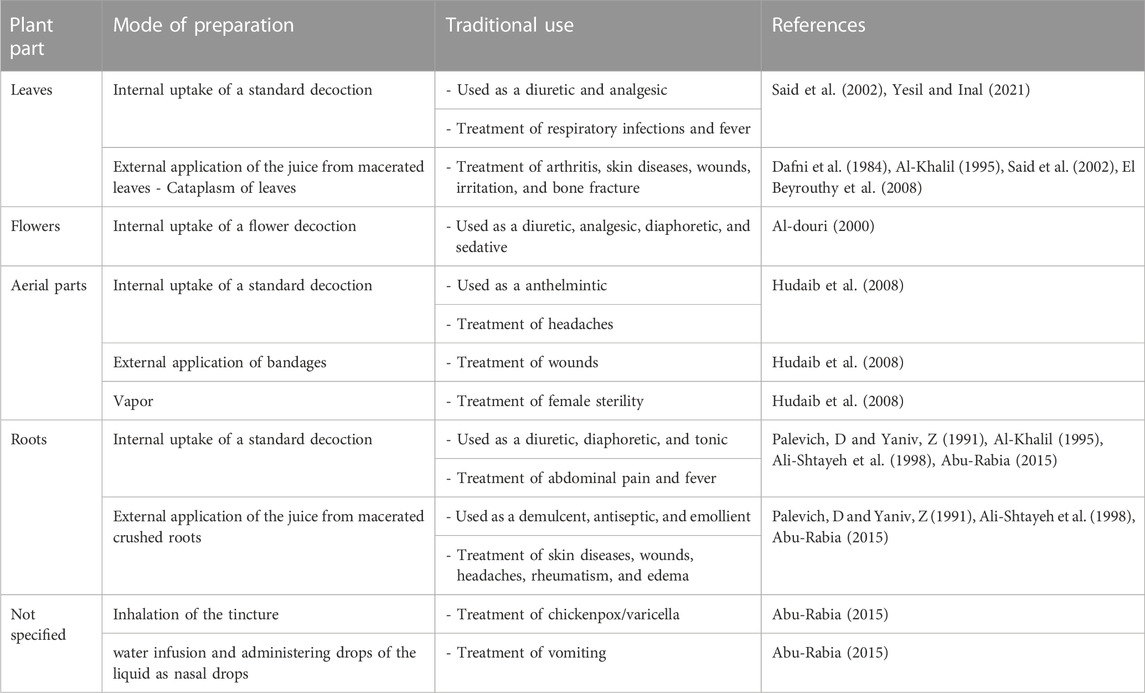
A. strigosa Banks & Sol. is a non-succulent and short-lived perennial plant widespread in the Eastern Mediterranean region, particularly found in Greece, Turkey, Israel, Lebanon, Syria, and Iran. It is commonly known as prickly alkanet or strigose bugloss, and as “lisan al-thawr,” “balghasoun,” or “himhim” in Arabic. It is a drought-hardy wild plant considered as a “famine food” and known for its traditional culinary applications, particularly in Palestinian cuisine (Qasem, 2015; Yeşil et al., 2019; Fullilove, 2022; Baydoun et al., 2023). It is additionally used in folk medicine practices (Yeşil et al., 2019; Baydoun et al., 2023) for the treatment of skin diseases (Said et al., 2002; Abu-Rabia, 2015), wounds (Dafni et al., 1984; Palevich, D and Yaniv, Z, 1991; Ali-Shtayeh et al., 1998; Said et al., 2002; Hudaib et al., 2008), arthritis (Ali-Shtayeh et al., 2000; Said et al., 2002; El Beyrouthy et al., 2008), and abdominal pain (Palevich, D and Yaniv, Z, 1991; Al-Khalil, 1995; Ali-Shtayeh et al., 1998; Abu-Rabia, 2015), among others. A summary of the traditional uses of A. strigosa is shown in Table 1.
4 General characteristics
4.1 Botanical characteristics
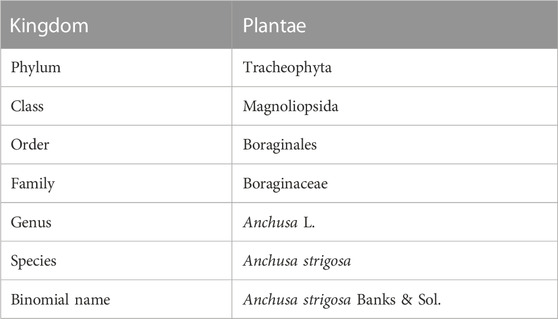
The taxonomic classification of A. strigosa is illustrated in Table 2. It is a hardy perennial weed with a bristly inflorescence stem that can grow up to a meter in height. It blooms in the late spring season from March to August, with trumpet-shaped flowers that are small (10–15 mm), tubular and distributed in an irregular pattern, but mostly dense clusters at the tips of the stems (Figure 1) (Tohmé and Tohmé, 2007). They can have a pale blue, violet, or white color, depending on the habitat. The age of the flower can also affect the color of the corolla, as it was observed that young flowers were violet, while mature ones were blue and produced a larger amount of nectar (Kadmon et al., 1991). The base of A. strigosa consists of a rosette of leaves that resemble the tongue of a ruminate by having a rough and a prickly texture, hence its Arabic designation “lisan al-thawr.” Moreover, it is characterized by oblong petiolate basal leaves and linear upper leaves, both covered in short and stiff hairs, as well as verrucose, exhibiting wart-like outgrowths. Finally, the roots of A. strigosa contain anchusin, a red-coloring dye that is used as food coloring and in cosmetics (Majumdar, D. N. and Chakravarty, G. C., 1940).

FIGURE 1. (A) Anchusa strigosa plant, (B) and (C) Anchusa strigosa flowers, and (D) Anchusa strigosa leaves. Images were obtained from https://www.gbif.org/occurrence/gallery?taxon_key=8192155 (accessed on 14 September 2023).
4.2 Ecological characteristics
The Mediterranean region harbors one of the richest sources of medicinal plants and hence has a long history of herbal remedies as an important source of treatment for several diseases (Saad et al., 2005). Of particular interest, A. strigosa, which is found in the Eastern Mediterranean region, naturally occurs in forests, rocky slopes, steppe vegetation, deserts, and along roadsides (Tohmé and Tohmé, 2007). It is native to Lebanon (Tohmé and Tohmé, 2007; El Beyrouthy et al., 2008; Baydoun et al., 2023; Chebaro et al., 2023), Syria (Alallan et al., 2018; Sulaiman et al., 2022), Palestine (Israel) (Dafni et al., 1984; Ali-Shtayeh et al., 1998, 2000; Lev and Amar, 2000; Said et al., 2002; Abutbul et al., 2005; Fullilove, 2022; Yarmolinsky et al., 2022), Jordan (Al-Khalil, 1995; Alali et al., 2007; Hudaib et al., 2008; Abbas et al., 2009; Dibas et al., 2017; Qasem, 2020; Al-Khatib et al., 2021), Iraq (Al-douri, 2000; Ghalib and Kadhim, 2021; Khit et al., 2023), Iran (Keshavarzi et al., 2017; Mordi et al., 2021), Turkey (Honda et al., 1996; Polat et al., 2013; Mükemre et al., 2015; Yeşil et al., 2019; Yesil and Inal, 2021), Greece (Selvi and Bigazzi, 2003), and Cyprus (Inflammation, 2023).
A. strigosa is a tenacious wild plant, with the ability to withstand periods of drought, and to adapt to a wide range of habitats, which is a characteristic of most plants of the Boraginaceae family. Due to its prominence in Palestinian cuisine and its ability to grow without human help and adapt to a wide range of habitats and conditions, such as disturbed soils and waste grounds, A. strigosa has been classified as a “famine food” (Fullilove, 2022). In fact, it is commonly used as food ingredient in conflict areas (Sulaiman et al., 2022), suggesting its important economic value and relevance in society.
4.3 Phytochemical characteristics
4.3.1 Phytochemical composition
A. strigosa is a rich source of secondary metabolites, particularly pyrrolizidine alkaloids (PAs), which are a class of plant toxin found in wide variety of plant families, and notably among the Boraginaceae (Ibanez, 2005; Tamariz et al., 2018; Günthardt et al., 2021). They are essentially stored in plants as protoxins and play in important role in the plant defense response against insect herbivores and pathogens. In fact, in a study investigating the concentration of PAs in A. strigosa, it was shown that the leaves were the richest in PAs (23.63 mg/g of dried part), followed by the flowers (19.77 mg/g), and the roots (1.80 mg/g), further supporting the protective effects of PAs against pests (Siciliano et al., 2005). A summary of the PAs identified in A. strigosa is summarized in Table 3. PAs exhibit great structural variety, and their toxicity is mostly associated with the presence of a double bond in the necine base. These are referred to 1,2-unsaturated PAs and are metabolized to PA radicals in the intestine and liver, causing liver injury, mainly resulting in hepatic sinusoidal obstruction syndrome, as well as liver fibrosis (Neuman et al., 2015; Yang et al., 2019b, Yang et al., 2019a). They have also been linked to genotoxicity, neurological damage and potential tumorigenic effects in humans and animals (Chen et al., 2010). However, the beneficial use of PAs to treat diseases has been gaining interest in recent decades due to their numerous biological and pharmacological activities (Wei et al., 2021). For instance, PAs isolated from plants have been shown to possess anti-microbial, anti-inflammatory, anti-diabetic, anti-ulcer and anti-cancer properties, among others (Schramm et al., 2019). For example, indicine N-oxide has been used in the treatment of leukemia and PAs isolated from various plants have been shown to induce autophagy and apoptosis in several cancer cell lines including lung cancer cells and human colorectal cancer cells (Wei et al., 2021). Overall, PAs have indeed beneficial pharmacological properties with promising therapeutic applications. However, the use of PA-containing medicine is associated with many risks and should be used with caution. Their recommended dosage is still under debate and often lacks clinical evidence. Nevertheless, their potential use in cancer treatment has stimulated the interest of research groups to develop methods for the targeted delivery of PAs to cancer cells, therefore limiting their toxicity to the liver (drugtargetreview, 2022).
A comparison between the phytochemical composition of the different parts of the A. strigosa plant shows that the leaves are particularly rich in phenolic acids, flavonoids, and PAs, while the roots were particularly rich in phenolic glycosides, PAs, and triterpenes (Table 3). Moreover, the flowers are rich in fixed and volatile oils, constituting 4.4% of the dry weight, with fixed oils and volatile oils comprising 52.8% and 26.1% of the total lipid content, respectively (Al-Salihi F. et al., 2009). Moreover, the phytochemical composition of plant materials varies with the extraction process and the type of the solvent used, often leading to the identification of different bioactive metabolites. For instance, in a study investigating the composition of A. strigosa leaves, the authors showed that 38 metabolites were identified in the aqueous extract obtained by maceration, whereas 39 metabolites were detected from the ultrasound-assisted technique (Chebaro et al., 2023). More importantly, the amount of the major phytochemical metabolites with potent well-documented anticancer activities such as rosmarinic acid, syringic acid, and kaempferol, were present in higher amounts in the maceration extract, and consequently conferred more potent biological activities (Chebaro et al., 2023). Some of the major phytochemicals found in A. strigosa are listed in Table 3.
4.3.2 Antioxidant capacity
Oxidative stress is identified as the major cause for the development and progression of aging and several pathophysiological conditions such as cancer, inflammation, and neurodegenerative disorders. It is caused by an imbalance between the production and elimination of reactive oxygen species (ROS) and free radicals. The accumulation of these very reactive molecules causes cellular damage because of their unpaired electrons. The levels of ROS and free radicals are normally regulated by endogenous antioxidants or boosted by exogenous sources such as natural plant products. In fact, plants have the ability to synthesize a wide range of phytochemicals that are known to possess potent antioxidant effects that counteract the toxic effects of free radicals. Moreover, natural antioxidant metabolites are regarded as safer alternatives than synthetic ones such as butyl hydroxy anisole (BHA) and butylated hydroxytoluene (BHT), which are widely used as preservatives in food, cosmetic formulations, and other consumer products, and unfortunately associated with potentially toxic and carcinogenic effects (Malkinson, 1983; Lourenço et al., 2019; Felter et al., 2021).
The antioxidant potential of A. strigosa has been investigated by using different solvents for the extraction of plant material and assessed through various assays, as tabulated in Table 4. Both aqueous and methanolic extracts showed significant scavenging activities of the 2,2'-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical, which was attributed to the total phenolic content of the extracts (Alali et al., 2007). Similar results were obtained with a methanolic extract from A. strigosa flowers using the DPPH radical scavenging and β-carotene bleaching (BCB) assays (Al-Khateeb et al., 2019), as well as the aqueous and hydro-ethanolic extracts of A. strigosa aerial parts using the DPPH and ferric reducing power (FRAP) assays (Dibas et al., 2017). More specifically, the hydro-ethanolic extract exhibited stronger antioxidant activity that was correlated with higher phenolic and flavonoid contents. The antioxidant capacity of the leaves was also assessed (Chebaro et al., 2023). Results showed that the aqueous extracts exhibited strong free-radical scavenging activity, which was associated with the presence of secondary metabolites with antioxidant properties. Indeed, the extract showed high concentrations of phenolics, flavonoids, and other metabolites that may be responsible for the observed effect. It is important to mention that the antioxidant capacity of A. strigosa has been assessed through in vitro assays, which are often prone to errors due to the chemical diversity of phytochemicals. Result should be cautiously interpreted and supported by additional evidence of their beneficial antioxidant use in vivo.
5 Biological activities of Anchusa strigosa
In recent years, several studies demonstrated the vast range of biological and pharmacological properties of A. strigosa including antimicrobial, pro-wound healing, antioxidant, anti-inflammatory, anticancer, antiarthritic, gastric protective, and antidiabetic effects. These are summarized in Figure 2.
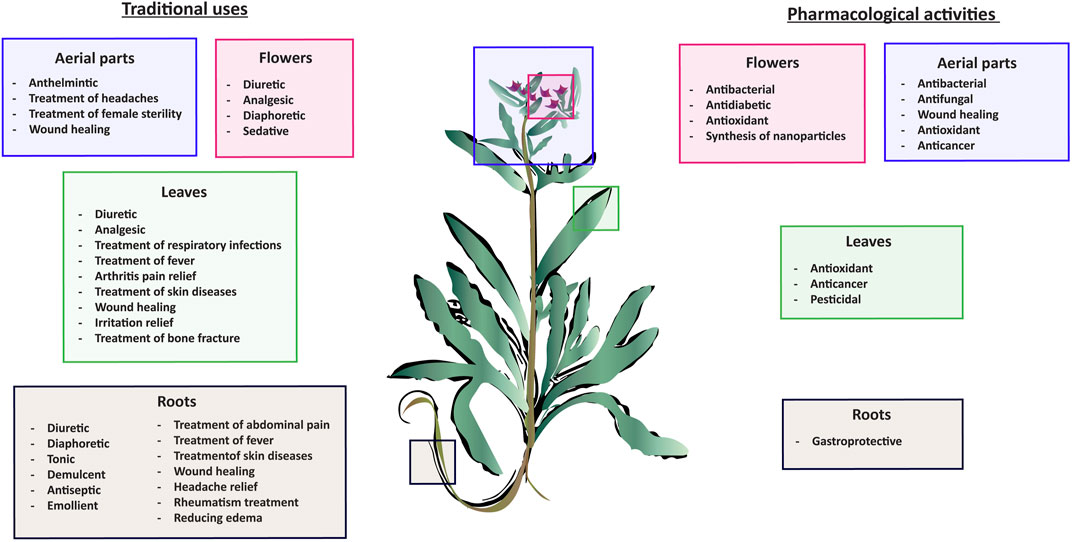
FIGURE 2. Summary of the main traditional uses of Anchusa strigosa and tested pharmacological activities.
5.1 Antimicrobial activities
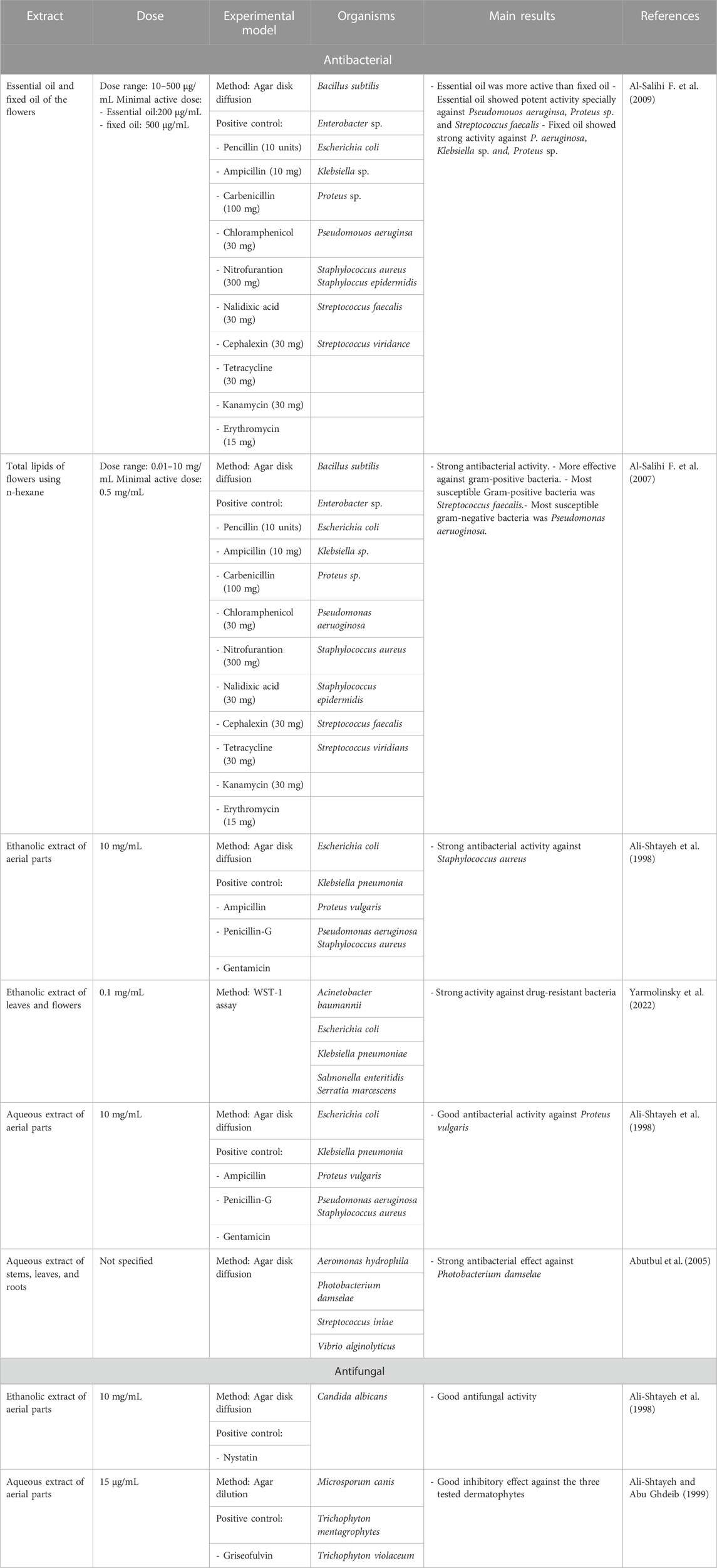
Antimicrobial resistance is a major global challenge and a serious threat to humans, which is caused by the extensive and inappropriate use of antibiotics. As traditional drug therapies have been losing their effectiveness, there has been an urgent need to develop new antimicrobial agents using novel therapies based on natural metabolites due to the chemical diversity and efficacy. In fact, extracts from medicinal plants have been widely reported to exhibit antimicrobial activities. They act by inhibiting the growth of a variety of pathogens, including bacteria, fungi, and viruses, in addition to enhancing the activity of antibiotics, and may help overcome antimicrobial resistance (Vaou et al., 2021). The effectiveness of A. strigosa extracts against a broad range of pathogenic microorganisms, including gram-positive and gram-negative bacteria, fungi, yeast, and pests has been extensively analyzed and listed in Table 5.
Results showed that lipid extracts from A. strigosa flowers exhibit potent antimicrobial activity, particularly against gram-positive bacteria such as Streptococcus faecalis and Staphylococcus aureus and against the gram-negative bacteria Pseudomouos aeruginsa, (Al-Salihi F. et al., 2007, 2009). Further analysis of the total lipids extract showed the presence of two phospholipids (phosphatidyl serine and phosphatidylethanolamine) and a triglyceride (tripalmitin) (Al-Salihi F. et al., 2007). Moreover, the authors showed that the essential oil extract from A. strigosa flowers showed stronger activity than the fixed oil extract (Al-Salihi F. et al., 2009). Several studies demonstrated the antimicrobial effects of A. strigosa ethanolic extracts. For example, Ali-Shtayeh et al. (1998) demonstrated its effect against the gram-positive bacteria Staphylococcus aureus, as well the yeast Candida albicans. And Yarmolinsky et al. (2022) showed that the crude extract as well as the isolated metabolite kaempferol and its glycoside derivatives significantly inhibited the growth of gram-negative bacteria that show drug-resistance such as Escherichia coli, Klebsiella pneumoniae, Acinetobacter baumannii, Serratia marcescens, and Salmonella enteritidis. Aqueous extracts also showed potent antimicrobial activity against the gram-negative bacteria Proteus vulgaris (Ali-Shtayeh et al., 1998) and bacterial pathogens that infect fish such as P. damselae (Abutbul et al., 2005), as well as antifungal activity against Microsporum canis, Trichophyton mentagrophytes, and Trichophyton violaceum (Ali-Shtayeh and Abu Ghdeib, 1999).
5.2 Antifeedant effect
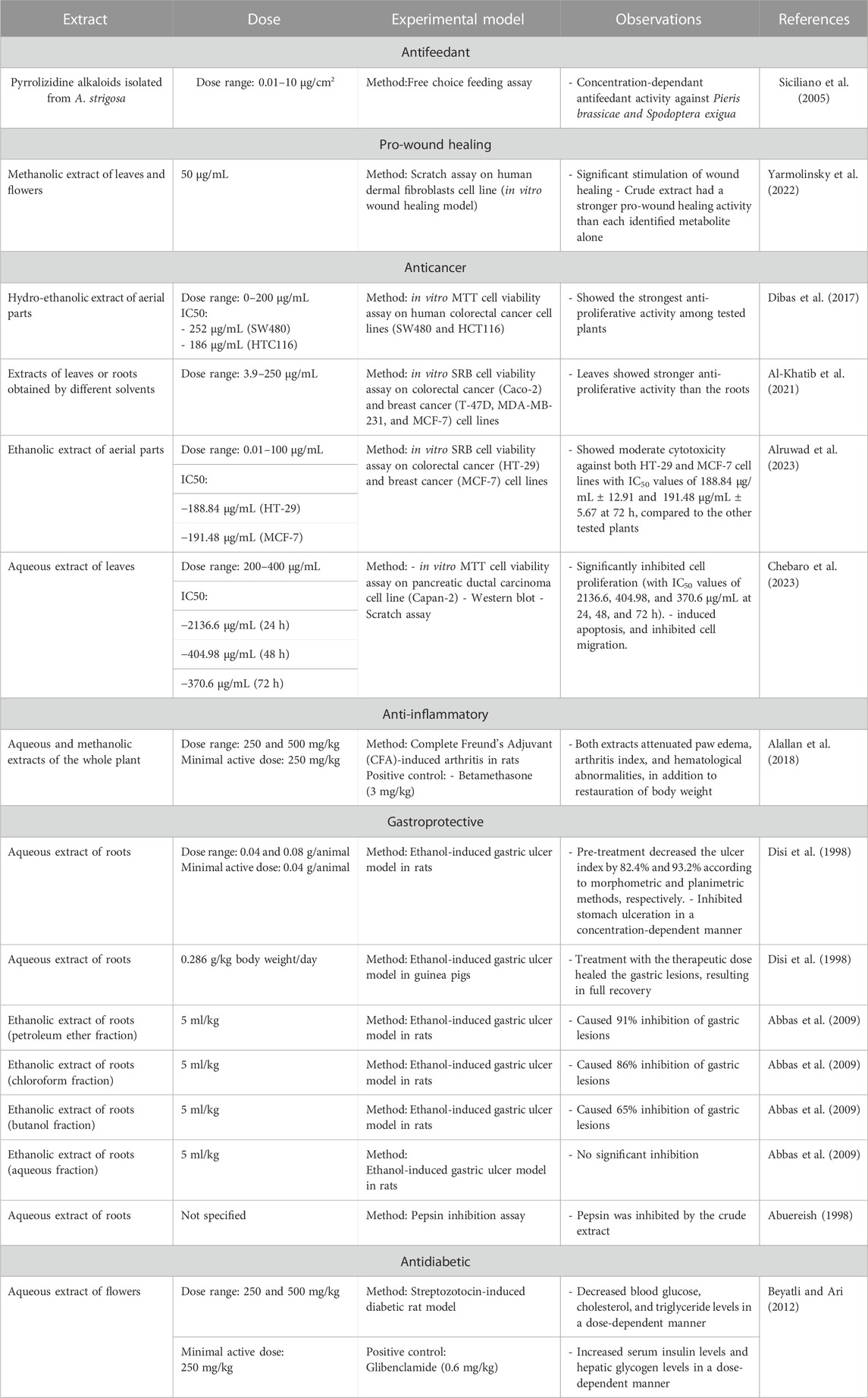
Plant extracts have long been shown to exhibit potent antifeeding activities against insects. PAs isolated from the methanolic extract of A. strigosa leaves showed potent antifeedant activity against the generalist beet armyworm Spodoptera exigua and the Pieris brassicae specialist larvae (Siciliano et al., 2005). Interestingly, only 1,2-unsaturated PAs showed antifeedant activity against the tested pests, further supporting their high toxic potential.
5.3 Wound healing properties
Wound healing is dynamic process that is characterized by four major stages: homeostasis, inflammation, proliferation, and remodeling. Each of these stages is driven by biological and chemical processes that protect the area from infection and lead to the regeneration of damaged tissue. A. strigosa has been widely used in traditional medicine for the treatment of topical wounds. As such, the mechanism of action behind the pro-wound healing activity of A. strigosa leaves and flowers was investigated in a study using a human dermal fibroblasts cell line (Yarmolinsky et al., 2022). Results showed that the crude methanolic extract significantly stimulated wound healing by increasing the rate of gap closure in the cultured cells. Moreover, it is the synergistic action between the isolated metabolites that confers the plant extract its pro-wound healing property. The authors identified quercetin 3-O-rutinoside, ellagic acid, kaempferol, and kaempferol 3-O-β-rhamnopyranosyl(1→6)-β-glucopyranoside as the pro-wound healing metabolites of the extract, calling for further investigation behind the mechanism of action of the tested metabolites. This is presented in Figure 3 and Table 6. While the in vitro human dermal fibroblast model provides a quick and inexpensive method to screen for the pro-wound healing property of an extract, it mainly focuses on the proliferation and migration of fibroblasts and does not assess other factors in the wound healing process. More importantly, the safety profile of the extract should be further investigated using in vivo studies by assessing its effect through systemic (oral or parenteral) and topical administrations.
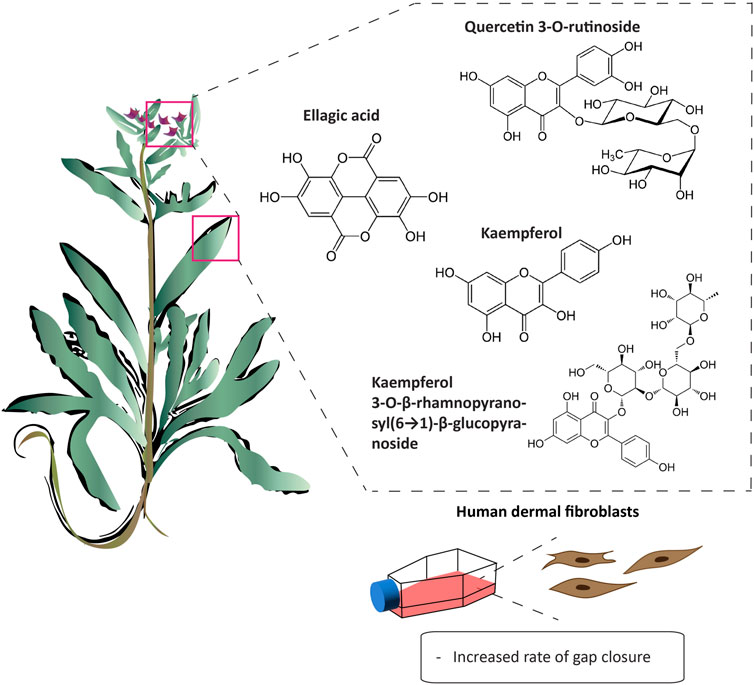
FIGURE 3. Anchusa strigosa exhibits wound healing properties. A. strigosa stimulates wound healing by increasing the rate of gap closure in cultured human dermal fibroblast cells. This is due to the synergistic action between metabolites isolated from the extract, with quercetin 3-O-rutinoside, ellagic acid, kaempferol, and kaempferol 3-O-β-rhamnopyranosyl(1→6)-β- as the main wound healing components.
5.4 Anticancer activities
Cancer is a global health burden and one of the leading causes of mortality worldwide. Despite recent advances in cancer therapy, adverse side effects and multidrug resistance continues to be a major challenge to conventional treatment regimens, which has fueled recent interest in the search for new bioactive metabolites from natural plant sources (Gezici and Şekeroğlu, 2019; Khan et al., 2020). In a study screening different plants in Jordan for their cytotoxicity against human colorectal cancer, the authors showed that the hydro-ethanolic extract of aerial parts of A. strigosa showed the strongest anti-proliferative activity among the tested plants with IC50 values of 186 and 252 μg/mL against the SW480 and HCT116 cell lines, respectively (Dibas et al., 2017). The anticancer properties of A. strigosa extracts from roots and leaves was also tested against several cancer cell lines including the colorectal carcinoma Caco-2, the human breast ductal carcinoma T-47D, the human breast carcinoma MDA-MB-231, and the breast adenocarcinoma MCF-7. Results showed that the leaves exhibited stronger anti-proliferative activity than the roots, probably due to the higher concentration of tannins and PAs (Al-Khatib et al., 2021). The cytotoxicity of the areal parts of A. strigosa was further confirmed in a study using an ethanolic extract against the HT-29 colorectal and MCF-7 breast cancer cell lines (Alruwad et al., 2023). Moreover, the aqueous extract of A. strigosa leaves showed strong anticancer activity against the aggressive pancreatic ductal carcinoma capan-2 cells (Chebaro et al., 2023). Notably, the extract exhibited potent inhibitory effects on capan-2 cells’ proliferation and migration, along with an induction of cell-cell aggregation and apoptosis. The anticancer properties of A. strigosa are shown in Figure 4 and summarized in Table 6. Overall, A. strigosa leaves prove to be an effective source of bioactive metabolites with anticancer potential that still warrants further investigation.
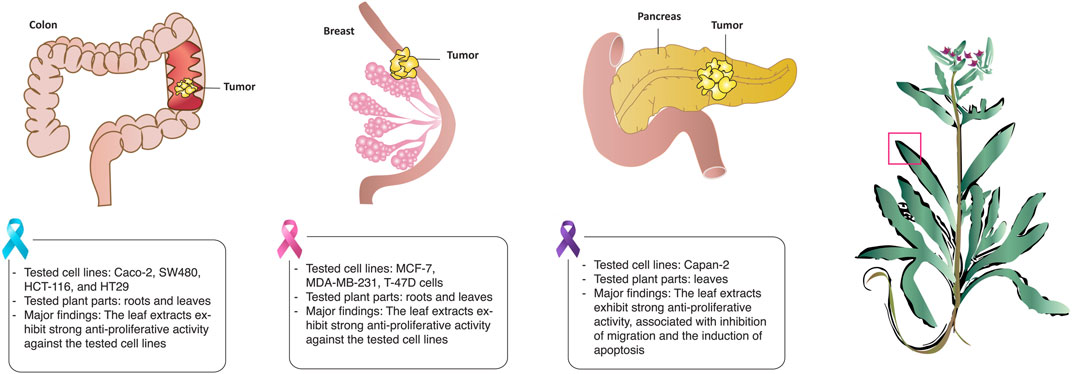
FIGURE 4. Anchusa strigosa shows strong antitumorigenic properties against several cancers. A. strigosa leaf extracts reduces the proliferation and growth rate of colorectal, breast, and pancreatic cancer cells in vitro.
5.5 Anti-inflammatory and anti-arthritic activities
Inflammation is a natural response by the body to fight an infection or repair an injury (National Institute of Environmental Health Sciences, Inflammation, 2023). However, when inflammation becomes chronic, it may lead to several diseases, including cardiovascular diseases, cancer, diabetes, neurodegenerative diseases, and arthritis. The anti-inflammatory property of A. strigosa was investigated first using Complete Freund’s Adjuvant (CFA)-induced paw edema in rats (Alallan et al., 2018). Treatment with the aqueous and methanolic extracts of the whole plant both showed significant reduction in swelling, similar to the betamethasone control, suggesting that A. strigosa could be a source of bioactive metabolites involved in the acute inflammatory response. The extracts were further investigated for the anti-arthritic effect, using CFA-induced arthritis in rats, as model for chronic immune-mediated joint inflammation (Alallan et al., 2018). Results showed that the arthritis index was significantly lowered with both extracts. This was accompanied by a restauration of body weight, which was significantly lowered due to CFA treatment, in addition to an attenuation of hematological abnormalities, showing increased levels of hemoglobin and a reduction of elevated white blood cell levels following treatment. These findings are summarized in Table 6. These indicate that A. strigosa have potential therapeutic activities in the treatment of rheumatoid arthritis and inflammatory diseases.
5.6 Gastroprotective properties
A. strigosa has been traditionally used in the treatment of gut and digestive disorders such as abdominal pain, diarrhea, and vomiting. And over the last few decades, several studies have demonstrated its gastroprotective properties, mostly in the management and treatment of gastric ulcers, using particularly the roots (Figure 5; Table 6). For example, in a study using ethanol-induced ulcer in laboratory animals, treatment with an aqueous extract from A. strigosa roots before ulcer induction showed a protective antigastritis effect and protected the stomach from ulcer formation, as observed by a lower lesion index (Disi et al., 1998). It was also shown to be effective in the treatment of ulcers, showing even complete healing and recovery within 25 days after ulcer formation (Disi et al., 1998). Fractionation studies were carried out on the A. strigosa root extract in an effort to identify the metabolites responsible for its gastroprotective effect. As such, four triterpenoids were isolated from the petroleum ether fraction, which showed strongest activity, and were identified as: oleanolic acid, β-amyrin, crataegolic acid, and β-sitosteryl glucoside (Abbas et al., 2009). These have been previously isolated from various medicinal plants and were shown to have strong anti-ulcerogenic effects (Xiao et al., 1992; Navarrete et al., 2002; Arrieta et al., 2003; Rodríguez, 2003). Furthermore, a pepsin inhibitor was isolated from the aqueous extracts of A. strigosa roots, confirming the gastroprotective effect of A. strigosa (Abuereish, 1998). In fact, excessive secretion of acid and pepsin is a major cause of hemorrhagic damage of the gastric mucosa and at the root of the genesis and chronicity of ulceration. As such, inhibition of pepsin activity has been a major line in the treatment of ulcers. These results suggest that A. strigosa extracts and isolated metabolites should be further exploited as a source of potential therapeutic agents in the treatment of gastric ulcers.
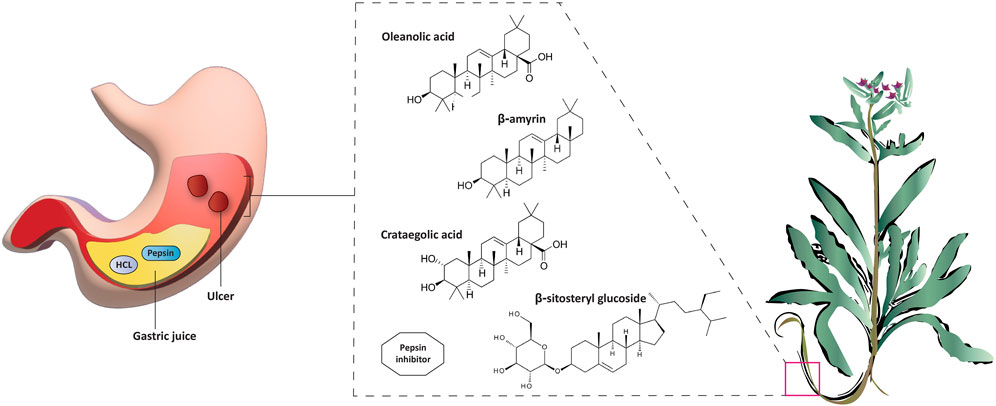
FIGURE 5. Anchusa strigosa has gastroprotective properties. A. strigosa root extracts show a protective antigastritis effect by preventing the formation of ulcers in laboratory animals administered with ethanol to cause gastric necrotic damage. The extracts are also effective in the treatment of ulcers. These contain several metabolites that are known to have anti-ulcerogenic effects, including a pepsin inhibitor.
5.7 Antidiabetic effect
Diabetes is a serious metabolic disorder that is caused by a deregulation of blood sugars due to the inefficient production of insulin by the pancreas or when the body is unable to respond and use effectively that insulin. If left uncontrolled, the elevated blood glucose concentration, or hyperglycemia, can lead to serious damage to blood vessels and nerves that can lead to permanent vision loss, kidney failure, and cardiovascular complications. Medicinal plants have long played an important role in the management and treatment of diabetes before the discovery of drugs. Today, there has been a resurgence of public interest in therapies from natural sources due to their cost-effectiveness and associated limited side effects. The oral administration of an aqueous extract from A. strigosa flowers significantly decreased blood sugar levels, as well as cholesterol and triglyceride levels in a streptozotocin-induced diabetic rat model (Figure 6; Table 6) (Beyatli and Ari, 2012). Moreover, the body weight of rats improved, and the levels of hepatic glycogen increased, possibly due to the reactivation of glycogen synthase and observed increase in serum insulin levels. These results show that A. strigosa flowers could be a potential source of anti-hyperglycaemic and hypolipidemic agents in the treatment of diabetes that warrants further investigation.
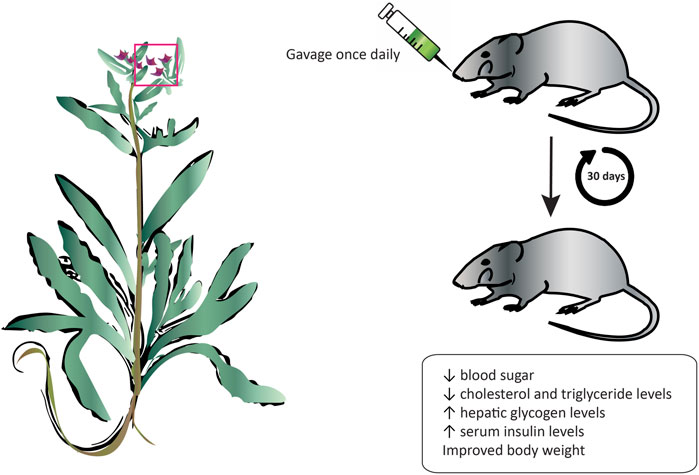
FIGURE 6. Anchusa strigosa exhibits an antidiabetic effect. The oral administration of A. strigosa flower extract significantly improved the glycemic and lipidemic profiles of streptozotocin-induced diabetic rats.
5.8 Other activities
Over the last decade, nanotechnology has been gaining tremendous impetus and holds great promise in various domains, including technology industries, agriculture, food, cosmetics, and medicine. It involves the manipulation of materials into their nanosize, and making use of their unique chemical, physical, mechanical, and optical properties. However, the synthesis and application of nanotechnology is not without its challenges and available techniques are expensive and require a lot of material and energy. Recently, there has been growing interest in the green synthesis of nanoparticles through the use of microorganisms and plants. These provide active biomolecules that facilitate the bioreduction of metal ions to their elemental form, producing nanoparticles in the 1- to 100-nm range (Zafar, 2023). It is a clean, safe, cost-effective, and environmentally friendly approach that does not require the use of toxic chemicals and high energy processes. Moreover, it results in the production of more stable nanoparticles compared to other traditional synthesis methods that can also support mass production (Huston et al., 2021). Contextually, an A. strigosa flower extract was used in the green synthesis of copper oxide nanoparticles. The obtained nanoparticles were very stable and effective in the removal of basic safranine dye from aqueous solutions (Khit et al., 2023).
6 Safety of Anchusa strigosa
Toxicological studies are crucial when developing a botanical drug to ensure its efficacy and safety profile prior to administration to humans, as these can have adverse effects on the body or interfere with other drugs (Thakkar et al., 2020; Hossain et al., 2022). The incorporation of A. strigosa in the Levantine cuisine for many years without any reported toxicity provides some assurance for the safety of this plant (Qasem, 2015; Yeşil et al., 2019; Fullilove, 2022; Baydoun et al., 2023). Moreover, acute toxicity studies of the aqueous A. strigosa flower extract showed no mortality or toxic reactions to rats administered orally with the extract at 1, 2, and 4 g/kg of body weight during the 72 h treatment period (Beyatli and Ari, 2012). However, the aqueous extract of A. strigosa roots used for the gastroprotective studies showed an intraperitoneal lethal dose 50 (LD50) of 0.08 g extract/kg body weight in mice, which is considered high compared to other plants (Disi et al., 1998). The authors argued that the extract dose required for ulcer treatment is much lower than the lethal dose. In fact, subchronic toxicity studies of this extract on rats showed no histological changes when water intake was replaced with 75 ml of the extract at concentrations of 2.865, 3.57, and 4.284 g/l per animal per day for 90 days, except for observed depressive effects on the central nervous system and general weakness at doses higher than 3.57 g/L. Moreover, the ingestion of a therapeutic dose of 0.286 g/day/kg body weight administered to guinea pigs with ethanol-induced ulcer had no toxic effect on the animals during the 24-day study (Disi et al., 1998). While the acute toxicity tests described here provide a preliminary and positive effects with no obvious toxicity, subacute and chronic studies have demonstrated some side effects. Whereas these toxicology studies provide some reassurance regarding the use of A. strigosa as a botanical drug, they fail to highlight the fact that this plant has high levels of PAs, which are a concerning risk and safety issue, particularly related to chronic toxicity. The intake of PAs is associated with liver damage, whereas the prolonged exposure to PAs has been linked with genotoxic and carcinogenic effects. Overall, these observations indicate that further toxicology screenings and additional confirmation testing need to be carried out to validate the safe usage of this plant.
7 Conclusion and future perspectives
Plant materials have long been used in the treatment and prevention of human diseases. Now more than ever, plants are seen as potential lead metabolites for drug development. As such, research on plant products has been gaining more and more interest in recent years. However, in plant-derived drug discovery, plant metabolites should be optimized for their efficacy and follow a thorough assessment for their toxicity. Phytochemical and pharmacological studies on A. strigosa have shown that it is a rich source of bioactive metabolites with a wide range of applications, further supporting its ethnopharmacological uses. However, A. strigosa is also rich in PAs, which are toxins produced by the plant as a defense mechanism against insects and herbivores. Toxicokinetics of PAs show that they are readily absorbed via the gastrointestinal tract and distributed to the liver, where they are metabolized to highly reactive pyrroles that cause damage to the liver, kidneys, and lungs. Therefore, the use of plants rich in PAs has been controversial over their associated risks and safety, impeding their clinical application. In parallel, PAs have been drawing greater attention as promising drug leads due their vast pharmacological properties including anti-microbial, anti-inflammatory, and anti-cancer activities, among others. Therefore, assessing the toxicity profile of A. strigosa extracts is of vital importance. And strategies to reduce the toxicity of PAs, while maintaining their bioactivity and improving their therapeutic effect is key to move forward to clinical trials and drug development.
Author contributions
ZC: Conceptualization, Methodology, Writing-original draft. AB: Funding acquisition, Supervision, Writing–review and editing. MM: Funding acquisition, Methodology, Supervision, Writing–review and editing. EB: Conceptualization, Resources, Supervision, Writing–eview and editing. JM: Conceptualization, Methodology, Supervision, Writing–original draft, Writing–review and editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by a URB grant from the American University of Beirut to EB and a University of Petra grant to AB.
Acknowledgments
All authors thank the URB of the American University of Beirut and the University of Petra for their funding.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Abbas, M., Disi, A., and Al-Khalil, S. (2009). Isolation and identification of anti-ulcer components from Anchusa strigosa root. Jordan J. Pharm. Sci. 2, 10.
Abuereish, G. M. (1998). Pepsin inhibitor from roots of Anchusa strigosa. Phytochemistry 48, 217–221. doi:10.1016/S0031-9422(97)01131-X
Abu-Rabia, A. (2015). Indigenous medicine among the bedouin in the Middle East. China, Berghahn Books.
Abutbul, S., Golan Goldhirsh, A., Barazani, O., Ofir, R., and Zilberg, D. (2005). Screening of desert plants for use against bacterial pathogens in fish. Israeli J. Aquac. - Bamidgeh 57, 71–80. doi:10.46989/001c.20405
Alali, F. Q., Tawaha, K., El-Elimat, T., Syouf, M., El-Fayad, M., Abulaila, K., et al. (2007). Antioxidant activity and total phenolic content of aqueous and methanolic extracts of Jordanian plants: an ICBG project. Nat. Prod. Res. 21, 1121–1131. doi:10.1080/14786410701590285
Alallan, L., Agha, M. I. H., Omerein, A. N., and Balkhi, M. H. A. (2018). Anti-arthritic effects of Anchusa strigosa extracts on complete Freund’s adjuvant-induced arthritis in rats. J. Pharmacogn. Phytochem. 7 (6), 679–685.
Al-douri, N. A. (2000). A survey of medicinal plants and their traditional uses in Iraq. Pharm. Biol. 38, 74–79. doi:10.1076/1388-0209(200001)3811-BFT074
Ali-Shtayeh, M. S., and Abu Ghdeib, S. I. (1999). Antifungal activity of plant extracts against dermatophytes. Mycoses 42, 665–672. doi:10.1046/j.1439-0507.1999.00499.x
Ali-Shtayeh, M. S., Yaghmour, R. M.-R., Faidi, Y. R., Salem, K., and Al-Nuri, M. A. (1998). Antimicrobial activity of 20 plants used in folkloric medicine in the Palestinian area. J. Ethnopharmacol. 60, 265–271. doi:10.1016/S0378-8741(97)00153-0
Ali-Shtayeh, M. S., Yaniv, Z., and Mahajna, J. (2000). Ethnobotanical survey in the Palestinian area: a classification of the healing potential of medicinal plants. J. Ethnopharmacol. 73, 221–232. doi:10.1016/S0378-8741(00)00316-0
Al-Khalil, S. (1995). A survey of plants used in Jordanian traditional medicine. Int. J. Pharmacogn. 33, 317–323. doi:10.3109/13880209509065385
Al-Khateeb, E. H., Al-Assi, G. A., Shakya, A. K., Al-Rawi, N., and Shalan, N. (2019). Antioxidant potential of pistacia vera L. Fruit hull, Anchusa strigosa flowers and Ilex paraguariensis A. St.-Hil. Leaves extract. Orient. J. Chem. 35, 982–986. doi:10.13005/ojc/350309
Al-Khatib, A. O., Khateeb, I., Dahab, R., and Al-Rawi, N. (2021). The cytotoxic effect of the extract of Anchusa strigosa (him him) grown in Jordan against different cancer cell lines. Baghdad Sci. J. 18, 0070. doi:10.21123/bsj.2021.18.1.0070
Alruwad, M. I., Sabry, M. M., Gendy, A. M., El-Dine, R. S., and El Hefnawy, H. M. (2023). In vitro cytotoxic potential of selected Jordanian flora and their associated phytochemical analysis. Plants 12, 1626. doi:10.3390/plants12081626
Al-Salihi, F., Al-Ameri, A., and Al-Juobory, T. (2007). Antimicrobial activity of total lipids extracted from Anchusa strigosa Lab. J. Surra Man Raa 3 (6), 11–20.
Al-Salihi, F., Yasseen, A., and Al-Salihi, S. (2009). Antimicrobial activity of volatile oil and fixed oil extracted from Anchusa strigosa Lab. Tikrit J. Pure Sci. 14 (2), 21–24.
Al-Snafi, A. E. (2014). The pharmacology of Anchusa italica and Anchusa strigosa–A review. Int. J. Pharm. Pharm. Sci. 6, 7–10.
Anchusa, L. (2023). plants of the world online. Available at: http://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:4451-1 [Accessed August 3, 2023].
Anchusa strigosa Banks & Sol (2023). Plants of the world online. Available at: http://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:113367-1 (Accessed August 3, 2023).
Arrieta, J., Benitez, J., Flores, E., Castillo, C., and Navarrete, A. (2003). Purification of gastroprotective triterpenoids from the stem bark of amphipterygium adstringens; role of prostaglandins, sulfhydryls, nitric oxide and capsaicin-sensitive neurons. Planta Med. 69, 905–909. doi:10.1055/s-2003-45098
Baydoun, S., Hani, N., Nasser, H., Ulian, T., and Arnold-Apostolides, N. (2023). Wild leafy vegetables: a potential source for a traditional Mediterranean food from Lebanon. Front. Sustain. Food Syst. 6, 991979. doi:10.3389/fsufs.2022.991979
Beyatli, A., and Ari, N. (2012). Antidiabetic activity ofThe aqueous extract of Anchusa strigosa lab in streptozotocin diabetic rats. Int. J. Pharm. 2, 445–449.
Braca, A., Bader, A., Siciliano, T., Morelli, I., and De Tommasi, N. (2003). New pyrrolizidine alkaloids and glycosides from Anchusa strigosa. Planta Med. 69, 835–841. doi:10.1055/s-2003-43202
Chebaro, Z., Abdallah, R., Badran, A., Hamade, K., Hijazi, A., Maresca, M., et al. (2023). Study of the antioxidant and anti-pancreatic cancer activities of Anchusa strigosa aqueous extracts obtained by maceration and ultrasonic extraction techniques. Frontiers in Pharmacology 14.doi:10.3389/fphar.2023.1201969
Chen, T., Mei, N., and Fu, P. P. (2010). Genotoxicity of pyrrolizidine alkaloids. J. Appl. Toxicol. 30, 183–196. doi:10.1002/jat.1504
Dafni, A., Yaniv, Z., and Palevitch, D. (1984). Ethnobotanical survey of medicinal plants in northern Israel. J. Ethnopharmacol. 10, 295–310. doi:10.1016/0378-8741(84)90017-5
Dibas, J. I., Yaghi, B. M., Mansi, I. A., Mhaidat, N. M., and Al-Abrouni, K. F. (2017). Screening for cytotoxic and antioxidant activity of selected wild plants at shafa badran, amman, Jordan. Res. J. Pharm. Biol. Chem. Sci. 9.
Disi, A. M., Tamimi, S. O., and Abuereish, G. M. (1998). Effects of Anchusa strigosa root aqueous extract on gastric ethanol-induced ulcer in laboratory animals. J. Ethnopharmacol. 60, 189–198. doi:10.1016/S0378-8741(97)00134-7
drugtargetreview (2022). Drug target review. Available at: https://www.drugtargetreview.com/news/105481/scientists-inhibit-cancer-cell-growth-using-pyrrolizidine-alkaloid/ (Accessed October 29, 2023).
El Beyrouthy, M., Arnold, N., Delelis-Dusollier, A., Dupont, F., Annick, D. D., et al. (2008). Plants used as remedies antirheumatic and antineuralgic in the traditional medicine of Lebanon. J. Ethnopharmacol. 120, 315–334. doi:10.1016/j.jep.2008.08.024
Felter, S. P., Zhang, X., and Thompson, C. (2021). Butylated hydroxyanisole: carcinogenic food additive to be avoided or harmless antioxidant important to protect food supply? Regul. Toxicol. Pharmacol. 121, 104887. doi:10.1016/j.yrtph.2021.104887
Fullilove, C. (2022). “Famine foods” and the values of biodiversity preservation in Israel-Palestine. Isis 113, 625–636. doi:10.1086/721288
Gezici, S., and Şekeroğlu, N. (2019). Current perspectives in the application of medicinal plants against cancer: novel therapeutic agents. Anticancer Agents Med. Chem. 19, 101–111. doi:10.2174/1871520619666181224121004
Ghalib, S. A., and Kadhim, E. (2021). The investigation of some phytochemical compounds found in Anchusa strigosa L. Grown naturally in Iraq. IJPS 30, 179–188. doi:10.31351/vol30iss1pp179-188
Günthardt, B. F., Wettstein, F. E., Hollender, J., Singer, H., Härri, J., Scheringer, M., et al. (2021). Retrospective HRMS screening and dedicated target analysis reveal a wide exposure to pyrrolizidine alkaloids in small streams. Environ. Sci. Technol. 55, 1036–1044. doi:10.1021/acs.est.0c06411
Honda, G., Yeşilada, E., Tabata, M., Sezik, E., Fujita, T., Takeda, Y., et al. (1996). Traditional medicine in Turkey. VI. Folk medicine in west anatolia: afyon, kütahya, denizli, muğla, aydin provinces. J. Ethnopharmacol. 53, 75–87. doi:10.1016/S0378-8741(96)01426-2
Hossain, C. M., Gera, M., and Ali, K. A. (2022). Current status and challenges of herbal drug development and regulatory aspect: a global perspective. Asian J. Pharm. Clin. Res., 31–41. doi:10.22159/ajpcr.2022.v15i12.46134
Hudaib, M., Mohammad, M., Bustanji, Y., Tayyem, R., Yousef, M., Abuirjeie, M., et al. (2008). Ethnopharmacological survey of medicinal plants in Jordan, Mujib Nature Reserve and surrounding area. J. Ethnopharmacol. 120, 63–71. doi:10.1016/j.jep.2008.07.031
Huston, M., DeBella, M., DiBella, M., and Gupta, A. (2021). Green synthesis of nanomaterials. Nanomater. (Basel) 11, 2130. doi:10.3390/nano11082130
Ibanez, G. (2005). “Pyrrolizidine alkaloids,” in Encyclopedia of toxicology. Editor P. Wexler Second Edition (New York: Elsevier), 585–587. doi:10.1016/B0-12-369400-0/00822-X
Inflammation (2023). Inflammation. China: National Institute of Environmental Health Sciences. Available at: https://www.niehs.nih.gov/health/topics/conditions/inflammation/index.cfm (Accessed April 11, 2023).
Kadmon, R., Shmida, A., and Selten, R. (1991). Within-plant foraging behaviour of bees and its relationship to nectar distribution in Anchusa strigosa. null 40, 283–294. doi:10.1080/0021213X.1991.10677206
Keshavarzi, M., Nasrollahi, F., and Sheidai, M. (2017). Cytogenetic study of the genus Anchusa L. (Boraginaceae) in Iran. Caryologia 70, 357–365. doi:10.1080/00087114.2017.1367172
Khan, T., Ali, M., Khan, A., Nisar, P., Jan, S. A., Afridi, S., et al. (2020). Anticancer plants: a review of the active phytochemicals, applications in animal models, and regulatory aspects. Biomolecules 10, 47. doi:10.3390/biom10010047
Khit, S. A., Shaheed, I. M., and Kareem, E. T. (2023). in A green synthesis of copper oxide nanoparticles using Anchusa strigosa L. flowers extract and study their impact for water pollutant removal. Editor I. Baghdad doi:10.1063/5.0156952
Lardos, A. (2006). The botanical materia medica of the Iatrosophikon—a collection of prescriptions from a monastery in Cyprus. J. Ethnopharmacol. 104, 387–406. doi:10.1016/j.jep.2005.12.035
Lev, E., and Amar, Z. (2000). Ethnopharmacological survey of traditional drugs sold in Israel at the end of the 20th century. J. Ethnopharmacol. 72, 191–205. doi:10.1016/S0378-8741(00)00230-0
Lourenço, S. C., Moldão-Martins, M., and Alves, V. D. (2019). Antioxidants of natural plant origins: from sources to food industry applications. Molecules 24, 4132. doi:10.3390/molecules24224132
Majumdar, D. N., and Chakravarty, G. C. (1940). The constituents of alkanet root (Anchusa tinctoria, LAM) Part II. Anchusin and its derivcatives. J. Indian Chem. Soc. 17. doi:10.5281/zenodo.6612807
Malkinson, A. M. (1983). Review: putative mutagens and carcinogens in foods III. Butylated hydroxytoluene (BHT). Environ. Mutagen. 5, 353–362. doi:10.1002/em.2860050313
Mordi, A., Teimorian, M., Shakiba, B., and Moudi, E. (2021). Traditional botanical flora of medicinal plants in the treatment of kidney stones in Iran. J. Biol. Res. - Bollettino della Soc. Italiana di Biol. Sperimentale 94. doi:10.4081/jbr.2021.9869
Mukemre, M., Behçet, L., and Çakılcıoğlu, U. (2015). Ethnobotanical study on medicinal plants in villages of Çatak (Van-Turkey). J. Ethnopharmacol. 166, 361–374. doi:10.1016/j.jep.2015.03.040
Navarrete, A., Trejo-Miranda, J. L., and Reyes-Trejo, L. (2002). Principles of root bark of Hippocratea excelsa (Hippocrataceae) with gastroprotective activity. J. Ethnopharmacol. 79, 383–388. doi:10.1016/S0378-8741(01)00414-7
Neuman, M. G., Cohen, L., Opris, M., Nanau, R. M., and Jeong, H. (2015). Hepatotoxicity of pyrrolizidine alkaloids. J. Pharm. Pharm. Sci. 18, 825–843. doi:10.18433/J3BG7J
Polat, R., Cakilcioglu, U., and Satıl, F. (2013). Traditional uses of medicinal plants in Solhan (Bingöl—Turkey). J. Ethnopharmacol. 148, 951–963. doi:10.1016/j.jep.2013.05.050
Qasem, J. R. (2015). Prospects of wild medicinal and industrial plants of saline habitats in the Jordan Valley. Pak. J. Bot. 47, 551–570.
Qasem, J. R. S. (2020). The coloured atlas of medicinal and aromatic plants of Jordan and their uses, Volume Two. Cambridge: Cambridge Scholars Publishing.
Rodríguez, J., Astudillo, L., and Schmeda-Hirschmann, G. (2003). Oleanolic acid promotes healing of acetic acid-induced chronic gastric lesions in rats. Pharmacol. Res. 48, 291–294. doi:10.1016/S1043-6618(03)00155-5
Saad, B., Azaizeh, H., and Said, O. (2005). Tradition and perspectives of arab herbal medicine: a review. Evid. Based Complement. Altern. Med. 2, 475–479. doi:10.1093/ecam/neh133
Said, O., Khalil, K., Fulder, S., and Azaizeh, H. (2002). Ethnopharmacological survey of medicinal herbs in Israel, the golan heights and the west bank region. J. Ethnopharmacol. 83, 251–265. doi:10.1016/S0378-8741(02)00253-2
Schramm, S., Köhler, N., and Rozhon, W. (2019). Pyrrolizidine alkaloids: biosynthesis, biological activities and occurrence in crop plants. Molecules 24, 498. doi:10.3390/molecules24030498
Selvi, F., and Bigazzi, M. (2003). Revision of genus Anchusa (Boraginaceae-Boragineae) in Greece. Botanical J. Linn. Soc. 142, 431–454. doi:10.1046/j.1095-8339.2003.00206.x
Siciliano, T., Leo, M. D., Bader, A., Tommasi, N. D., Vrieling, K., Braca, A., et al. (2005). Pyrrolizidine alkaloids from Anchusa strigosa and their antifeedant activity. Phytochemistry 66, 1593–1600. doi:10.1016/j.phytochem.2005.05.002
Sulaiman, N., Pieroni, A., Sõukand, R., and Polesny, Z. (2022). Food behavior in emergency time: wild plant use for human nutrition during the conflict in Syria. Foods 11, 177. doi:10.3390/foods11020177
Tamariz, J., Burgueño-Tapia, E., Vázquez, M. A., and Delgado, F. (2018). “Chapter one - pyrrolizidine alkaloids,” in The alkaloids: chemistry and biology. Editor H.-J. Knölker (Academic Press), 1–314. doi:10.1016/bs.alkal.2018.03.001
Thakkar, S., Anklam, E., Xu, A., Ulberth, F., Li, J., Li, B., et al. (2020). Regulatory landscape of dietary supplements and herbal medicines from a global perspective. Regul. Toxicol. Pharmacol. 114, 104647. doi:10.1016/j.yrtph.2020.104647
Tohmé, G., and Tohmé, H. S. (2007). Illustrated flora of Lebanon. Beirut, Lebanon: National Council for Scientific Research.
Vaou, N., Stavropoulou, E., Voidarou, C., Tsigalou, C., and Bezirtzoglou, E. (2021). Towards advances in medicinal plant antimicrobial activity: a review study on challenges and future perspectives. Microorganisms 9, 2041. doi:10.3390/microorganisms9102041
Wei, X., Ruan, W., and Vrieling, K. (2021). Current knowledge and perspectives of pyrrolizidine alkaloids in pharmacological applications: a mini-review. Molecules 26, 1970. doi:10.3390/molecules26071970
Xiao, M., Yang, Z., Jiu, M., You, J., and Xiao, R. (1992). The antigastroulcerative activity of beta-sitosterol-beta-D-glucoside and its aglycone in rats. Hua Xi Yi Ke Da Xue Xue Bao 23, 98–101.
Yang, M., Ma, J., Ruan, J., Ye, Y., Fu, P. P.-C., and Lin, G. (2019a). Intestinal and hepatic biotransformation of pyrrolizidine alkaloid N-oxides to toxic pyrrolizidine alkaloids. Archives Toxicol. 93, 2197–2209. doi:10.1007/s00204-019-02499-2
Yang, X.-Q., Ye, J., Li, X., Li, Q., and Song, Y.-H. (2019b). Pyrrolizidine alkaloids-induced hepatic sinusoidal obstruction syndrome: pathogenesis, clinical manifestations, diagnosis, treatment, and outcomes. World J. Gastroenterology 25, 3753–3763. doi:10.3748/wjg.v25.i28.3753
Yarmolinsky, L., Budovsky, A., Khalfin, B., Yarmolinsky, L., and Ben-Shabat, S. (2022). Medicinal properties of Anchusa strigosa and its active compounds. Molecules 27, 8239. doi:10.3390/molecules27238239
Yeşil, Y., Çelik, M., and Yılmaz, B. (2019). Wild edible plants in Yeşilli (Mardin-Turkey), a multicultural area. J. Ethnobiol. Ethnomed 15, 52. doi:10.1186/s13002-019-0327-y
Yesil, Y., and Inal, I. (2021). Ethnomedicinal plants of hasankeyf (batman-Turkey). Front. Pharmacol. 11, 624710. doi:10.3389/fphar.2020.624710
Zafar, S. (2023). “Synthesis, properties, and characterization of biological nanomaterial,” in Nanomaterials: the building blocks of modern technology: synthesis, properties and applications smart nanomaterials technology. Editors T. Khan, M. Jawaid, K. A. Ahmad, and B. Singh (Singapore: Springer Nature), 91–114. doi:10.1007/978-981-99-4149-0_5
Keywords: Anchusa strigosa, traditional uses, herbal medicine, phytochemistry, pharmacology
Citation: Chebaro Z, Badran A, Maresca M, Baydoun E and Mesmar JE (2023) Ethnopharmacology and therapeutic potential of Anchusa strigosa: a comprehensive review. Front. Pharmacol. 14:1301154. doi: 10.3389/fphar.2023.1301154
Received: 24 September 2023; Accepted: 09 November 2023;
Published: 24 November 2023.
Edited by:
Nunziatina De Tommasi, University of Salerno, ItalyReviewed by:
Mahdi Moridi Farimani, Shahid Beheshti University, IranDaniela Russo, University of Basilicata, Italy
Marinella De Leo, University of Pisa, Italy
Copyright © 2023 Chebaro, Badran, Maresca, Baydoun and Mesmar. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Joelle Edward Mesmar, am0xMDRAYXViLmVkdS5sYg==; Elias Baydoun, ZWxpYXNiYXlAYXViLmVkdS5sYg==; Marc Maresca, bS5tYXJlc2NhQHVuaXYtYW11LmZy
 Ziad Chebaro
Ziad Chebaro Adnan Badran
Adnan Badran Marc Maresca
Marc Maresca Elias Baydoun
Elias Baydoun Joelle Edward Mesmar
Joelle Edward Mesmar