- 1The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou, China
- 2Key Unit of Methodology in Clinical Research, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou, China
- 3Foshan Hospital of Traditional Chinese Medicine, Foshan, China
- 4Guangzhou Nansha Hospital of TCM, Guangzhou, China
- 5State Key Laboratory of Dampness Syndrome of Chinese Medicine, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou, China
- 6Guangdong Provincial Key Laboratory of Clinical Research on Traditional Chinese Medicine Syndrome, Guangzhou, China
- 7Department of Global Public Health, Health Systems and Policy, Karolinska Institute, Stockholm, Sweden
Background: Chinese medicine (CM) is widely used for treating hyperlipidemias, especially in China. However, the heterogeneity of outcomes measured and reported across trials exacerbates the obstacles of evidence synthesis and effectiveness comparison. In this study, we develop a core outcome set (COS) for CM clinical trials for hyperlipidemia (COS-CM-Hyperlipidemia) to tackle the outcome issues.
Methods: We generated candidate outcomes through a systematic review of interventional and observational studies of Chinese medicine for hyperlipidemias. The comprehensive search strategy was employed. Study selection and data collection were independently done by two researchers. We searched clinical trial registry platform to supplement the outcomes list extracted by systematic review. Then, we conducted a three-round Delphi survey. The stakeholders were hyperlipidemia patients, clinicians or researchers, in either CM/integrated Chinese or Western medicine, clinical pharmacy, clinical epidemiology or statisticians, or editors of important relevant journals and an ethicist. They used a 9-point Likert scale to determine how important they felt each outcome was in determining treatment success. A consensus meeting was held to confirm the final COS, based on the Delphi survey results.
Results: We identified a total of 433 outcomes from 3,547 articles, and 28 outcomes from 367 registered trials. After standardization, we selected 71 outcomes to develop a preliminary outcome list for further consensus. After three Delphi survey rounds and one consensus meeting, the most important outcomes were determined for COS-CM-Hyperlipidemia. It included cardiovascular events, low-density lipoprotein cholesterol, risk of cardiovascular disease, total cholesterol, carotid intima-media thickness, high-density lipoprotein cholesterol, triglycerides, cerebrovascular events, adverse drug reactions and patient-reported symptoms.
Conclusion: COS-CM-Hyperlipidemia may improve outcome reporting consistency in clinical trials. Further work is needed to explore the optimal methods for measuring these outcomes.
Registration: The Core Outcome Measures in Effectiveness Trials Initiative (COMET): http://www.cometinitiative.org/studies/details/983. Registered on 25 April 2017.
1 Introduction
Cardiovascular diseases (CVDs) are the leading cause of death globally, taking an estimated 17.9 million lives each year.1 People with hyperlipidemia are at roughly twice the risk of developing CVDs as those without (Karr, 2017). High cholesterol (one type of hyperlipidemia) is one of the primary causal risk factors for CVDs and is 1 of 7 critical metrics the American Heart Association has used to define cardiovascular health in adults and children (Virani et al., 2020). According to the 2017 Global Burden of Disease Study (GBD 2017 Risk Factor Collaborators, 2018), of the leading risk factors for global mortality, high low-density lipoprotein cholesterol (LDL-C) remained the fifth-leading risk factor for mortality in both 1990 and 2017, accounting for 4.3 million deaths in 2017 (Virani et al., 2020).
Statins are the cornerstone of hyperlipidemia therapy, in addition to healthy lifestyle interventions (Grundy et al., 2019). Chinese medicine (CM) is widely used for treating hyperlipidemias, especially in China (Liu et al., 2011; Liao et al., 2014; Yao et al., 2016; Wang and Qiu, 2019). Thus, there has been an increasing focus on trials of CM on hyperlipidemias, yet there remains a lack of studies with high methodological quality. Researchers have suggested that this may be attributable to heterogeneity in the outcomes measured and reported across trials (Zhang et al., 2013; Xing et al., 2014). The lack of standardization of hyperlipidemia outcomes exacerbates the obstacles to evidence synthesis and CM effectiveness comparison (Williamson et al., 2017). Developing a core outcome set (COS) is one approach to addressing this lack of quality and standardization.
A COS represents the minimum outcomes that should be measured and reported in all clinical trials for a specific condition to facilitate the comparison and combination of trials while researchers continue to explore other outcomes (Williamson et al., 2017).2 CM hyperlipidemia treatment involves CM patterns (syndromes, or zheng in Chinese) which need to be considered by syndrome differentiation according to a patient’s clinical manifestations, including their pulse and tongue. Thus, outcome assessment may deviate from that used in Western medicine (Zhang et al., 2013; Xing et al., 2014; Qiu et al., 2021). Therefore, the aim of this study was to develop a COS for clinical trials of Chinese Medicine hyperlipidemia treatments (COS-CM-Hyperlipidemia).
2 Methods
This study has been registered on the Core Outcome Measures in Effectiveness Trials (COMET) website (No. 983)3 and the protocol has been published (Li et al., 2019). The conduct of this COS development adhered to the COMET handbook as much as possible (Williamson et al., 2017), and its results have been reported following the Core Outcome Set–STAndards for Reporting (the COS-STAR Statement) (Kirkham et al., 2016).
We established a work group and a study advisor group (SAG), and used group discussion, a Delphi survey and consensus meeting methods sequentially to develop COS-CM-Hyperlipidemia.
Ethical approval was obtained from the ethics committee of Guangdong Provincial Hospital of Chinese Medicine (GPHCM). Informed consent was obtained from all participants. All personal information about potential and enrolled participants will remain confidential.
2.1 Participants
2.1.1 Study Advisory Group
We established a SAG with participants from various stakeholder groups to guarantee quality and efficiency in COS-CM-Hyperlipidemia development. The SAG was composed of nine members—two endocrinologists and one cardiovascular expert from GPHCM, two GPHCM outpatients, two methodologists from GPHCM, one cardiovascular clinical trial researcher from GPHCM, one ethicist from the GPHCM ethics committee, and one statistician from GPHCM. The SAG was responsible for confirming the candidate outcome set for the Delphi survey, participating in the consensus meeting, process coordination, as well as data analysis and interpretation.
2.1.2 Delphi Survey Panel Assembly
According to the principle of representativeness and authority, we recruited experts in CM/integrated Chinese and Western medicine, clinical pharmacology, clinical epidemiology and statistics, medical journal editors and patients to participate in the Delphi survey. We expected to select 50 participants using a snowball sampling method. We identified a preliminary list of experts by reviewing the authors of high-impact papers and selected preliminary patients from a pool of GPHCM outpatients. Then, the preliminary stakeholders recommended whomever else they thought should be included as relevant stakeholders.
2.1.3 Consensus Meeting Participants
We adopted the purposeful sampling method to select participants who had completed all three rounds of the Delphi survey and invited at least two representatives from patients, endocrinologists, cardiologists, hyperlipidemia specialist nurses, and hyperlipidemia researchers to the consensus meeting. Additionally, all members of the SAG took part in the consensus meeting.
2.2 Information Sources
We conducted a systematic review of literature on studies of CM for hyperlipidemia (Li et al., 2021). Then, we searched three English databases (PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), and Embase) and three Chinese databases (China National Knowledge Infrastructure (CNKI), Chinese BioMedical Database (CBM), and Wanfang Database) in October 2017, with no time restriction. In order to collect comprehensive outcomes, we also searched two clinical trial registries (http://www.chictr.org.cn/index.aspx, https://clinicaltrials.gov/) to retrieve any outcomes used in clinical trials between 1 January 2016, and 1 January 2019.
We included randomized controlled trials, non-randomized controlled trials, case series, case-control, cohort studies, and systematic reviews evaluating CM for hyperlipidemia. Studies were excluded if 1) they lacked either clear diagnosis and effectiveness assessment standards or hyperlipidemia outcome reporting; 2) patients had only been treated by Western medicine.
Then, two reviewers (GL and RH) independently extracted the data, and entered it into a database using EpiData 3.1 (EpiData Association, Denmark). Information included the characteristics of each study (e.g., title, publishing journal, author(s), year of publication, country, authors’ affiliation(s), funding, diagnosis criteria, patient source, type of hyperlipidemia complications, follow-up duration, number of patients who withdrew, intervention details and CM syndrome pattern), study design, treatment, blood lipids, names of outcomes and whether they had been specified as primary or secondary outcomes, definitions of outcomes, time-point and method of outcome measurement and adverse events. Disagreements were resolved by discussion or consulting a third researcher (ZW).
After data extraction, we assigned the outcomes to one of eight domains: 1) mortality-related outcomes, 2) pathological or pathophysiological outcomes, 3) response rate-related outcomes, 4) cardiovascular events, 5) symptoms or function-related outcomes, 6) adverse events or safety-related outcomes, 7) patient-reported outcomes, and 8) resource utilization-related outcomes. In order to generate a candidate outcome set for consensus, the SAG members scored all of the outcomes in each domain to determine whether or not they would be included.
2.3 Consensus Process
We conducted a three-round Delphi survey to assess experts’ opinions on the importance of the candidate outcomes. Then we held a consensus meeting, attended by key stakeholders, to finalize COS-CM-Hyperlipidemia.
2.3.1 Delphi Survey
For each topic domain, we sorted candidate items alphabetically. For patient panelists, we presented lay equivalents of each outcome instead of scientific terms (Allin et al., 2016). For some complicated or difficult to comprehend outcomes, we enclosed a definition and explanation in the Delphi survey. In Round 1, participants could suggest outcomes not included in the questionnaire which they felt were important. In Delphi Rounds 2 and 3, outcome scores from the questionnaire assigned in the previous round were presented, and the distribution of each outcome from each stakeholder group was attached.
2.3.2 Scoring Importance of Outcome
To evaluate the outcomes’ importance, we used a 9-point Likert scale, where 1, 2 and 3 meant “unimportant” 4, 5 and 6 meant “important, but not essential”; and 7, 8 and 9 meant “essential” (Guyatt et al., 2011). At the end of each round, we performed data analysis on individual stakeholder groups, and as a whole. After Delphi Round 1, some of the outcomes recommended by the experts would enter the second round after discussion by the SAG. All scored outcomes were included in Delphi Round 2. Outcomes that were scored 7–9 by a majority of the panelists in Round two were retained for Delphi Round 3. After conducting the Delphi Round three analysis, we assigned the outcomes to one of three categories: ‘consensus out’ (≥70% scoring 1 to 3 AND <15% scoring 7 to 9 in each stakeholder group), ‘consensus in’ (<15% scoring 1 to 3 AND ≥ 70% scoring 7 to 9 in each stakeholder group) or ‘without consensus’ (other conditions) (Harman et al., 2013; Hirsch et al., 2016; Williamson et al., 2017).
2.3.3 Consensus Meeting
We reported the results of each Delphi round and the classification of candidate outcomes from the third Delphi round. Outcomes of “consensus in” were considered either “yes” for selected or “no” for not selected. Those voted for by at least 70% of the participants were included in the final COS-CM-Hyperlipidemia. Outcomes scored “consensus out” were excluded, and outcomes of “without consensus” were discussed and re-scored using the same 9-point Likert scale until a final consensus was reached (Allin et al., 2016; Iyengar et al., 2016). The definitions of outcomes were also provided to the expert panel. The same consensus criteria in round three were used at the meeting.
2.4 Data Management and Quality Control
This study process was implemented in accordance with the pre-developed protocol following the COMET handbook. We double-checked the data collected during three rounds of the survey process and confirmed the accurate entry and summary. All data from the consensus meeting have been checked through the meeting minutes.
3 Results
This study was completed according to the study protocol (Li et al., 2019). One deviation occurred in the data analysis of Delphi Round 2. Since the protocol defined criteria as ‘any outcomes whose median is greater than or equal to 4 (by any stakeholder group) will continue to Delphi Round 3’ being unable to exclude any outcomes, we changed the criteria to ‘outcomes that were scored 7–9 by more than 50% panelists in Round two were retained for Delphi round 3’. The other deviation was that: Due to the COVID-19 outbreak, the face-to-face consensus meeting had to be held as a combination web/in-person meeting.
3.1 Outcomes Extracted in the Systematic Review
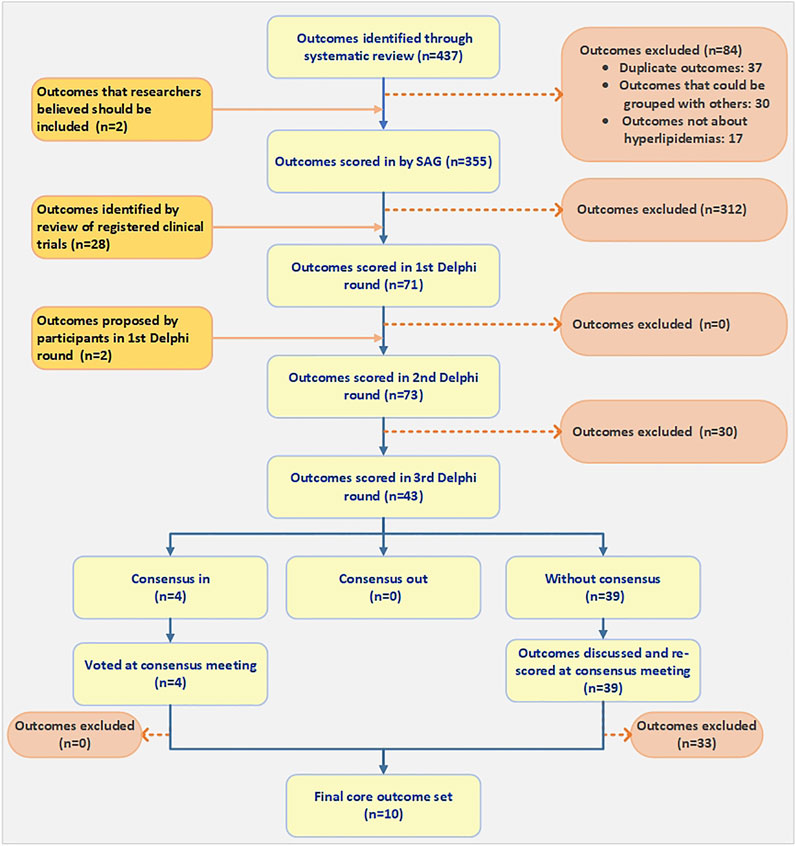
The systematic review identified 51,905 articles; 3,547 (3,461 in Chinese and 86 in English) were eligible for inclusion following abstract, title, and full-text screening. Supplementary File 1 is the PRISMA flowchart. A total of 433 unique outcomes were identified and reported, which were grouped into eight outcome domains. Researchers reviewed these 433 outcomes, identified 37 duplicates, grouped 25 closely related outcomes, and deleted 18 outcomes that were definitively not about hyperlipidemia. Because this list of outcomes was too long to be used for a Delphi survey, and in order to optimize the Delphi survey, SAG members evaluated the remaining 353 outcomes and two other outcomes that researchers believed should be considered and excluded 312 of them from the candidate outcome set. Additionally, we obtained 367 registered trials on Chinese medicine for hyperlipidemias, and from them, we derived 28 new outcomes. Therefore, a final inventory of 71 outcomes was entered into the first Delphi round (Figure 1) (Supplementary File 2). We also conducted an updated search from November 2017 to November 2020 to identify more new outcomes. Ultimately, we found 19 new outcomes, of which 1 outcome could be grouped with other outcomes, and we deleted the other 18 outcomes after discussion by SAG members (Li et al., 2021). Supplementary File 3 is a list of all 452 original outcomes.
3.2 Delphi Survey
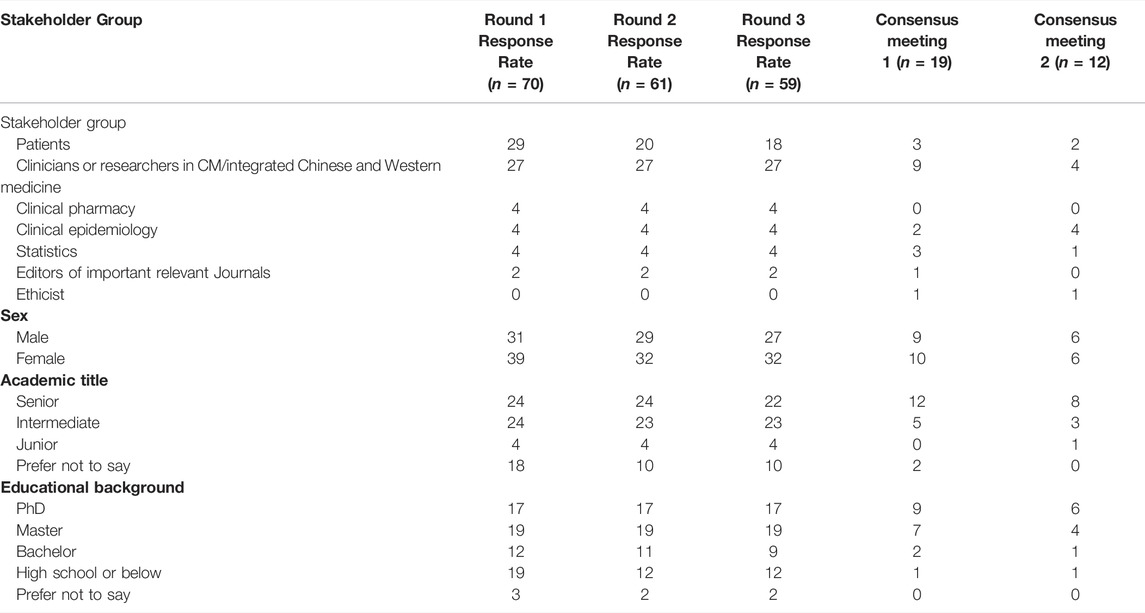
Participants’ characteristics for each round are presented in Table 1. A total of ninety experts and patients were invited to participate in the Delphi Round one survey. 70 (75.27%) of them completed the survey and six additional outcomes were suggested by respondents (see Supplementary File 2). All outcomes moved on to the next round. After being assessed by SAG members, two of the six additional outcomes, fatty liver and cerebrovascular events, were included in the second round (n = 73) (see Figure 1 and Supplementary File 2).
We invited seventy participants who had completed the Round one survey to re-rate the second round of the Delphi survey, and received 61 (87.14%) responses. The questionnaire listed the outcome scores voted for in the first round, as well as the distribution of each outcome from each stakeholder group. The 43 outcomes which were scored 7–9 by a majority of the panelists moved on to Round 3, while the others were excluded.
We invited the sixty-one participants who had completed Round two to re-rate the third round of the Delphi survey, and received 59 (96.72%) responses. Also, the questionnaire at this stage presented the voting results of the second round, and the distribution of each outcome from each stakeholder group was attached. After analysis, we categorized four outcomes (cardiovascular events, LDL-C, risk of cardiovascular disease and total cholesterol (TC)) as ‘consensus in’, while others were ‘without consensus’ (see Table 2). All outcomes were then discussed at the consensus meeting.
3.3 Consensus Meeting
Due to the COVID-19 outbreak and the large number of outcomes that needed a consensus, the consensus meeting was divided into two meetings. The first meeting was held on 21 August 2020, and involved 19 participants representing six stakeholders (see Table 1). The second meeting was held on 16 September 2020, and involved 12 participants representing five stakeholders (see Table 1). At the beginning of each consensus meeting, we showed the results of the three Delphi survey rounds to the participants. Outcomes of “consensus in” in the third round were voted on as either “yes” or “no” anonymously. After discussion and voting, all four “consensus in” outcomes were included in the final COS-CM-Hyperlipidemia. Outcomes of “without consensus” were discussed and re-scored using the same 9-point Likert scale.
At the end, 10 outcomes were included in the final COS-CM-Hyperlipidemia: cardiovascular events, LDL-C, risk of cardiovascular disease, TC, CIMT, HDL-C, TG, cerebrovascular events, adverse drug reaction, and patient-reported symptoms (see Table 3).
4 Discussion
In this study, we identified 10 core outcomes that should be reported in all future trials involving hyperlipidemia patients receiving CM. The final COS-CM-Hyperlipidemia includes cardiovascular events, LDL-C, risk of cardiovascular disease, TC, CIMT, HDL-C, TG, cerebrovascular events, adverse drug reactions, and patient-reported symptoms. This is the first study to report the development of a COS on clinical trials of CM for hyperlipidemias.
Adoption of this COS will standardize outcome selection and reporting; this will improve the relevance and interpretability of future systematic review activities, thereby promoting evaluation of the effects of CM for hyperlipidemia. From a clinical data perspective, adoption of the COS-CM-Hyperlipidemia will contribute to the standardization of the outcome data acquisition, hence, facilitating the harmonization and standardization of source data. Thereby, it can promote clinical data sharing and merging.
When this COS is applied, three points should be noted. First, COS is a minimum outcome set, so besides outcome in COS, studies with different purposes can add other outcomes if necessary. Second, outcomes in COS do not distinguish between primary outcomes and secondary outcomes, and studies can utilize one or more outcomes in this COS as the primary outcome(s) according to their main purposes. Third, although outcome measure points are not restricted, they should be defined based on the trial objective, in terms of scientificity, rationality and feasibility (Jin et al., 2020).
There are several limitations in this study. Firstly, there is a limited geographical representation of stakeholders. Most of the experts and patients were from China, so their perspectives may not reflect those of other regions overseas. Secondly, the consensus process was divided into two parts, and some of the experts participated in the meeting via conference calls instead of face-to-face. This may have led to insufficient discussion, and thus affected the consensus results. Thirdly, we changed the outcome criteria retained for Round 3. The original criterion was that any outcomes whose median was ≥ 4 (by any stakeholder group) would continue to Delphi Round 3 (Li et al., 2019). However, when we conducted the analysis using this criterion, we found that our pre-defined criteria were too loose to eliminate outcomes. After reading the COS handbook (Williamson et al., 2017) and consulting with the SAG, we changed the criteria such that outcomes that were scored 7–9 by a majority of the panelists in Round 2 were retained for Delphi Round 3. This change may have affected the Delphi results. Although we extended Delphi Round 1’s response time and also sent personalized reminder emails or made direct telephone calls, the attrition rate in Delphi Round 1 was 30%, which exceeded the acceptable range in most situations (Williamson et al., 2017). Attrition bias may have occurred due to this lower response rate. However, in Delphi Rounds 2 and 3, the response rates were higher and acceptable. Fifthly, we did not recommend any outcome measurement instruments for this COS. This is because we knew that incomparable scores from different instruments, as well as variability in the measures’ quality (i.e., reliability and validity), would skew reporting, making it difficult to compare and combine the findings in systematic reviews and meta-analyses (Prinsen et al., 2016; Williamson et al., 2017). Therefore, we will search, evaluate, and provide generic recommendations on the selection of outcome measurement instruments for outcomes included in this COS in the future.
Next, we will collaborate with systematic review groups, clinicians, journal editors, and other stakeholders to promote the broad application of COS in hyperlipidemia. We hope this COS will be recommended by relevant industry associations. With the development of medical research, the knowledge of disease, diagnosis, treatment, and evaluation will be constantly updated, so the COS needs constant evaluation and upgrades following the latest achievements of basic and clinical research. With the constant evaluation and update, the COS keeps its value and advantage in clinical research on CM in hyperlipidemia by adding new outcomes to ensure practicability and advancement (Li et al., 2019).
This COS involved the outcomes of interventional clinical trials, not only from the perspective of disease, but also combined with the purpose of CM intervention so that the effects of CM interventions can be evaluated. Our study is similar to Qiu et al.‘s study in which they developed a COS to support CM COVID-19 treatment (Qiu et al., 2020), and Sun et al.‘s study in which they developed a COS for CM for chronic hepatitis B (Sun et al., 2020). Both of these two studies are also aimed to develop a COS for CM. what is more, the methodology and procedure of our study are similar to these two studies, too. Besides, we think this COS is appropriate for western medicine for hyperlipidemia as well, because the outcomes included in this COS are generally used. For the efficacy evaluation of CM, CM patterns have been thought as important as other general outcomes that are appropriate for both CM and western medicine (Zhang et al., 2013; Xing et al., 2014; Qiu et al., 2021). A syndrome score is the commonly used outcome for assessing the effect of CM patterns. For example, Peng et al.‘s study found that compared with western medicine, CM can significantly improve the curative effect of CM syndromes in hyperlipidemia patients with turbid phlegm syndrome (Peng et al., 2017). However, there is a lack of agreeable and universal standards for CM syndrome score, many works need to do to promote its applicability and acceptability (Luo et al., 2015). Developing a scientific, standard syndrome scale or a new more specific outcome for evaluating the effect of patterns may be one of the solutions.
In conclusion, this study has identified a COS for clinical trials of CM for hyperlipidemias. Adoption of this COS will standardize outcome selection and reporting. We also hope the use of this COS will improve the quality of (and reduce waste in) human, physical and financial resources in clinical trials of CM for hyperlipidemias. Further work is needed to explore the optimal methods for measuring these outcomes.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding authors.
Ethics Statement
Ethical approval to conduct this study has been granted by the Ethics Committee at Guangdong Provincial Hospital of Chinese Medicine. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
XC, GL, and ZW devised the study. RH and ML conducted the Delphi surveys and collected the responses. GL and RH performed data extraction and analysis. GL drafted the manuscript. XC supervised the work. ZW resolved conflicting opinions between researchers and reviewed the results. All authors read and approved the final version of the manuscript prior to submission.
Funding
This work was supported by the Specific Research Fund for TCM Science and Technology of Guangdong Provincial Hospital of Chinese Medicine (No. YN2015QN11). The funders had no role in the design of the study; in the collection, analysis or interpretation of data; in the writing of the article, or in the decision to publish the results.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
The study team would like to thank anyone who contributed to the consensus meeting, and to those who took part in the Delphi survey.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2022.847101/full#supplementary-material
Abbreviations
CVDs, cardiovascular diseases; COS, core outcome set; CM, Chinese medicine; COS-CM-Hyperlipidemia, COS for clinical trials of Chinese Medicine for hyperlipidemias; COMET, Core Outcome Measures in Effectiveness Trials; LDL-C, low density lipoprotein cholesterol; TC, total cholesterol; CIMT, carotid intima-media thickness; HDL-C, high-density lipoprotein cholesterol; TG, triglycerides; VLDL-C, very low-density lipoprotein cholesterol; AI, arteriosclerosis index; BMI, body mass index; AST, aspartate aminotransferase; ALT, alanine aminotransferase; Cr, creatinine; BUN, blood urea nitrogen; AEs, adverse events.
Footnotes
1WHO. Cardiovascular Diseases (2021). https://www.who.int/health-topics/cardiovascular-diseases#tab=tab_1 [accessed 12 March 2021]
2Core Outcome Measures in Effectiveness Trials (2021). https://www.comet-initiative.org/[Accessed 12 March 2021]
3Zehuai Wen, Xiankun Chen, Geng Li, et al. (2021). A core outcome set for clinical trials of Chinese Medicine in hyperlipidemia. http://www.comet-initiative.org/studies/details/983 [Accessed 12 March 2021]
References
Allin, B., Bradnock, T., Kenny, S., Walker, G., and Knight, M. (2016). NETS1HD: Study Protocol for Development of a Core Outcome Set for Use in Determining the Overall success of Hirschsprung's Disease Treatment. Trials 17, 577. doi:10.1186/s13063-016-1693-6
GBD 2017 Risk Factor Collaborators (2018). Global, Regional, and National Comparative Risk Assessment of 84 Behavioural, Environmental and Occupational, and Metabolic Risks or Clusters of Risks for 195 Countries and Territories, 1990-2017: a Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 392, 1923–1994. doi:10.1016/S0140-6736(18)32225-6
Grundy, S. M., Stone, N. J., Bailey, A. L., Beam, C., Birtcher, K. K., Blumenthal, R. S., et al. (2019). 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 73, e285–e1143. doi:10.1016/j.jacc.2018.11.003
Guyatt, G. H., Oxman, A. D., Kunz, R., Atkins, D., Brozek, J., Vist, G., et al. (2011). GRADE Guidelines: 2. Framing the Question and Deciding on Important Outcomes. J. Clin. Epidemiol. 64, 395–400. doi:10.1016/j.jclinepi.2010.09.012
Harman, N. L., Bruce, I. A., Callery, P., Tierney, S., Sharif, M. O., O'brien, K., et al. (2013). MOMENT Management of Otitis Media with Effusion in Cleft Palate: Protocol for a Systematic Review of the Literature and Identification of a Core Outcome Set Using a Delphi Survey. Trials 14, 70. doi:10.1186/1745-6215-14-70
Hippisley-Cox, J., Coupland, C., Robson, J., and Brindle, P. (2010). Derivation, Validation, and Evaluation of a New QRISK Model to Estimate Lifetime Risk of Cardiovascular Disease: Cohort Study Using QResearch Database. BMJ 341, c6624. doi:10.1136/bmj.c6624
Hirsch, M., Duffy, J. M., Barker, C., Hummelshoj, L., Johnson, N. P., Mol, B., et al. (2016). Protocol for Developing, Disseminating and Implementing a Core Outcome Set for Endometriosis. BMJ open 6, e013998. doi:10.1136/bmjopen-2016-013998
Iyengar, S., Williamson, P. R., Schmitt, J., Johannsen, L., Maher, I. A., Sobanko, J. F., et al. (2016). Development of a Core Outcome Set for Clinical Trials in Rosacea: Study Protocol for a Systematic Review of the Literature and Identification of a Core Outcome Set Using a Delphi Survey. Trials 17, 429. doi:10.1186/s13063-016-1554-3
Jin, X., Pang, B., Zhang, J., Liu, Q., Yang, Z., Feng, J., et al. (2020). Core Outcome Set for Clinical Trials on Coronavirus Disease 2019 (COS-COVID). Engineering (Beijing) 6, 1147–1152. doi:10.1016/j.eng.2020.03.002
Kirkham, J. J., Gorst, S., Altman, D. G., Blazeby, J. M., Clarke, M., Devane, D., et al. (2016). Core Outcome Set-STAndards for Reporting: The COS-STAR Statement. Plos Med. 13, e1002148. doi:10.1371/journal.pmed.1002148
Li, G., Han, R., Cao, W., Wen, Z., and Chen, X. (2021). Outcome Reporting Variability in Trials of Chinese Medicine for Hyperlipidemia: A Systematic Review for Developing a Core Outcome Set. Evid. Based Complement. Alternat Med. 2021, 8822215. doi:10.1155/2021/8822215
Li, G., Zhou, L., Ouyang, W., Xuan, M., Lu, L., Li, X., et al. (2019). A Core Outcome Set for Clinical Trials of Chinese Medicine for Hyperlipidemia: a Study Protocol for a Systematic Review and a Delphi Survey. Trials 20, 14. doi:10.1186/s13063-018-3082-9
Liao, J., Tian, J., Li, T., Song, W., Zhao, W., and Du, J. (2014). Xuefuzhuyu Decoction for Hyperlipidemia: a Systematic Review and Meta-Analysis of Randomized Clinical Trails. J. Tradit Chin. Med. 34, 411–418. doi:10.1016/s0254-6272(15)30040-6
Liu, Z. L., Liu, J. P., Zhang, A. L., Wu, Q., Ruan, Y., Lewith, G., et al. (2011). Chinese Herbal Medicines for Hypercholesterolemia. Cochrane Database Syst. Rev., CD008305. doi:10.1002/14651858.CD008305.pub2
Luo, H., Liao, X., and Wang, Q. (2015). Application of Chinese Medical Syndrome Scores in Effectiveness Evaluation: a Critical Appraisal of 240 Randomized Controlled Trials. Zhongguo Zhong Xi Yi Jie He Za Zhi 35, 1261–1266. doi:10.7661/CJIM.2015.10.1261
Peng, D., Wang, Y., Liu, X., Xie, R., and Li, X. (2017). Meta-analysis of Curative Effect of Invigorating Spleen to Resolving Phlegm on Turbid Phlegm Syndrome of Hyperlipidemia. Tianjin J. Traditional Chin. Med. 34, 733–737. doi:10.11656/j.issn.1672-1519.2017.11.05
Prinsen, C. A., Vohra, S., Rose, M. R., Boers, M., Tugwell, P., Clarke, M., et al. (2016). How to Select Outcome Measurement Instruments for Outcomes Included in a "Core Outcome Set" - a Practical Guideline. Trials 17, 449. doi:10.1186/s13063-016-1555-2
Qiu, R., Zhao, C., Liang, T., Hao, X., Huang, Y., Zhang, X., et al. (2020). Core Outcome Set for Clinical Trials of COVID-19 Based on Traditional Chinese and Western Medicine. Front. Pharmacol. 11, 781. doi:10.3389/fphar.2020.00781
Qiu, R., Sun, R., Zhong, C., Li, M., Hu, J., Zhao, C., et al. (2021). Methods of Developing Core Traditional Chinese Medicine Syndromes Set. Chin. J. Evid Based. Med. 21, 1353–1357. doi:10.7507/1672-2531.202106083
Sun, Y., He, L., Luo, L., and Yu, C. (2020). Core Outcome Sets of Traditional Chinese Medicine on the Treatments of Chronic Hepatitis B Clinical Trials. China J. Traditional Chin. Med. Pharm. 35, 4684–4688.
Virani, S. S., Alonso, A., Benjamin, E. J., Bittencourt, M. S., Callaway, C. W., Carson, A. P., et al. (2020). Heart Disease and Stroke Statistics-2020 Update: A Report from the American Heart Association. Circulation 141, e139–e596. doi:10.1161/CIR.0000000000000757
Wang, S., and Qiu, X. J. (2019). The Efficacy of Xue Fu Zhu Yu Prescription for Hyperlipidemia: A Meta-Analysis of Randomized Controlled Trials. Complement. Ther. Med. 43, 218–226. doi:10.1016/j.ctim.2019.02.008
Williamson, P. R., Altman, D. G., Bagley, H., Barnes, K. L., Blazeby, J. M., Brookes, S. T., et al. (2017). The COMET Handbook: Version 1.0. Trials 18, 280. doi:10.1186/s13063-017-1978-4
Xing, D., Zhang, J., and Zhang, B. (2014). Approach to Developing Core Outcome Sets for Clinical Trials of Traditional Chinese Medicine. Chin. J. Tradit Chin. Med. Pharm. 29, 1352–1355.
Yao, H., Zhang, Z., Wang, J., Zuo, J., Chen, Y., Zhu, L., et al. (2016). Efficacy and Safety of Yinchenwuling Powder for Hyperlipidemia: a Systematic Review and Meta-Analysis. J. Tradit Chin. Med. 36, 135–143. doi:10.1016/s0254-6272(16)30019-x
Keywords: hyperlipidemia, core outcome set, Chinese medicine, systematic review, delphi survey
Citation: Li G, Han R, Lin M, Wen Z and Chen X (2022) Developing a Core Outcome Set for Clinical Trials of Chinese Medicine for Hyperlipidemia. Front. Pharmacol. 13:847101. doi: 10.3389/fphar.2022.847101
Received: 01 January 2022; Accepted: 04 April 2022;
Published: 02 May 2022.
Edited by:
Juei-Tang Cheng, Chang Jung Christian University, TaiwanReviewed by:
Eric De Groot, University of Amsterdam, NetherlandsDaniela Oliveira De Melo, Federal University of São Paulo, Brazil
Copyright © 2022 Li, Han, Lin, Wen and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiankun Chen, eGlhbmt1bi5jaGVuQGd6dWNtLmVkdS5jbg==; Zehuai Wen, d2VuemhAZ3p1Y20uZWR1LmNu
†These authors have contributed equally to this work
 Geng Li
Geng Li Ruxue Han
Ruxue Han Mingjun Lin
Mingjun Lin Zehuai Wen
Zehuai Wen Xiankun Chen
Xiankun Chen