
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Pharmacol. , 21 February 2022
Sec. Pharmacology of Anti-Cancer Drugs
Volume 13 - 2022 | https://doi.org/10.3389/fphar.2022.801733
This article is part of the Research Topic Pharmacological and Biochemical Perspectives of Kinase Inhibitors in Cancer and COVID-19 Therapeutics, Volume I View all 8 articles
 Rajeev K. Singla1,2
Rajeev K. Singla1,2 Sahar Behzad3,4
Sahar Behzad3,4 Johra Khan5,6
Johra Khan5,6 Christos Tsagkaris7
Christos Tsagkaris7 Rupesh K. Gautam8
Rupesh K. Gautam8 Rajat Goyal8
Rajat Goyal8 Hitesh Chopra9
Hitesh Chopra9 Bairong Shen1*
Bairong Shen1*Endometrial cancer (EC) is the sixth most prevalent type of cancer among women. Kinases, enzymes mediating the transfer of adenosine triphosphate (ATP) in several signaling pathways, play a significant role in carcinogenesis and cancer cells’ survival and proliferation. Cyclin-dependent kinases (CDKs) are involved in EC pathogenesis; therefore, CDK inhibitors (CDKin) have a noteworthy therapeutic potential in this type of cancer, particularly in EC type 1. Natural compounds have been used for decades in the treatment of cancer serving as a source of anticancer bioactive molecules. Many phenolic and non-phenolic natural compounds covering flavonoids, stilbenoids, coumarins, biphenyl compounds, alkaloids, glycosides, terpenes, and terpenoids have shown moderate to high effectiveness against CDKin-mediated carcinogenic signaling pathways (PI3K, ERK1/2, Akt, ATM, mTOR, TP53). Pharmaceutical regimens based on two natural compounds, trabectedin and ixabepilone, have been investigated in humans showing short and midterm efficacy as second-line treatments in phase II clinical trials. The purpose of this review is twofold: the authors first provide an overview of the involvement of kinases and kinase inhibitors in the pathogenesis and treatment of EC and then discuss the existing evidence about natural products’ derived kinase inhibitors in the management of the disease and outline relevant future research.
Kinases play a significant role in tumorigenesis, cancer cells’ survival, and proliferation. Different studies on human kinases were able to report 555 types of protein kinases based on their sequence analysis and evolutionary history (Baier and Szyszka, 2020). Out of this, 497 were grouped into eukaryotic protein kinases and 58 as atypical kinases. Kinases constitute 2% of the total human genome and the first group of enzymes that play a significant role in oncogenic evolution and are a potential drug target in cancer therapy (Schwartz and Murray, 2011). Mutations and transformations in tyrosine kinases are found to occur in different oncogenic conditions. PI3KCA is a member of the PI3K family with dual specificity of lipid and protein kinases associated with ovarian, breast, endometrial, and colorectal cancer (Gocek et al., 2014). Protein kinases serve as regulators of many cellular pathways which are related to diseases, sometimes as triggers and sometimes as therapeutic mediation points. A growing body of research focuses on natural-source-derived kinase inhibitors as anticancer therapeutics.
Cyclin-dependent kinases (CDK) regulate essential complexes of cell cycle advancement and alteration. In cell cycle regulation, 11 molecules of the CDK family function as leads at three checkpoints: G1, G2, and M checkpoints. CDK4 and 6 D and CDK2-E cyclin regulate G1 and S phase transition, CDK2-cyclin A and CDK1-cyclin B regulate G2, and CDK2-cyclin A and CDK1-cyclin B are also responsible for the phosphorylation of FoxM1 (Forkhead box protein M1) that codes for the transition of a cell from G2 to M phases (Gocek et al., 2014; Finn et al., 2016; Jorda et al., 2018). CDK7 functions as a regulator of different CDKs during different phases of the cell cycle. As CDKs play an important role in ontogenesis in endometrial cancer (EC), CDK inhibitors (CDKin) have a noteworthy therapeutic potential in EC (Sanchez-Martinez et al., 2015). CDKin is classified into 4 different generations based on their development and mechanism of action. The majority of first- and second-generation CDKin have nonspecific action and can produce toxic effects. Dinaciclib, a second-generation CDKin, shows significant activity against the breast cancer cell line by inhibiting Myc. Palbociclib, ribociclib, and abemaciclib are some of the third- and fourth-generation CDKin which are found specific for both CDKs and tumor cell lines (Finn et al., 2016).
Recent studies screening natural compounds as kinase inhibitors indicated particular natural compounds, mostly polyphenols, with a major potential to mitigate kinase mutation. The bioactive compounds directly bind with tyrosine kinases receptors and can regulate many cell signaling pathways by altering their phosphorylation nature.
Unopposed estrogen exposure causes endometrioid tumor development, leading to gland hyperplasia and eventually dysplasia. Intrinsic factors that increase blood estrogen levels, such as obesity and type II diabetes mellitus, are highly associated with endometrial cancer (Onstad et al., 2016). All of them have been highly associated with EC of the corpus (or central) uteri, and in some cases with EC affecting the lower uterine area. Individual subtypes do not have distinctive gross appearances that identify them as such (cell type). The low-volume disease rarely finds residual disease by diagnostic curettage (Ambros and Kurman, 1992). Polypoid nodules and friable nodules develop with localized disease. Widespread involvement of the endometrium, but without a discernible exophytic component, suggests induration of the uterine surface. Necrosis and hemorrhage may be seen. They are characterized as being lighter in color because the areas of myometrial invasion are easily identifiable as light-gray or white zones that stand out clearly from the untreated myometrium. It is usually seen when an expansion of the original lesion is detected.
In addition to extrinsic factors, such as estrogen-only hormone replacement therapy and tamoxifen, intrinsic factors such as inheritance and early puberty can also increase the risk for endometrial adenocarcinoma (Whitney et al., 2004). Endometrial atrophy (reduction in the size of the endometrium) frequently accompanies serous endometrial cancer, and certain genetic anomalies have also been linked to the development of this disease. Estrogen-related cancers are often low-grade and have a favorable prognosis and are known as Type I tumors (Sherman et al., 1995). These endometrial hyperplasia lesions tend to have an immature, undifferentiated look due to the growth of hyperplasia around them. Lopsidedly, 80% of spontaneous tumors are due to them. PTEN and other targets are downregulated, resulting in constitutive activation of Akt and mTOR (Enomoto et al., 1991; Okuda et al., 2010).
Type II tumors are poorly differentiated and are typically seen in older postmenopausal women. When it comes to type II estrogen-independent breast cancers, endometrial atrophy is usually present. There are documented links between ErbB2, P16, and TP53 mutations and the development of type II cancers (Zagouri et al., 2010; Singh et al., 2011). In general, tumors with these features are frequently found to be at an advanced stage or have already metastasized, and this predicts a bad prognosis (Leslie et al., 2005). 6-month survival is quite low in this type of cancer, even with extensive treatment, including chemotherapy and radiation.
Type I and type II carcinomas differ both molecularly and clinically (Lax, 2004). Most type I carcinomas have a modest number of somatic copy number alterations as measured, whereas most type II carcinomas have a large number of them and aneuploidy. Other genes frequently mutated in the process of developing type I tumors include PTEN (about 50% of cases), KRAS (about 20%–30% of cases), ARID1A (a gene encoding a member of the low-grade endometrioid carcinoma subtype—about 40% of this tumor subtype), CTNNB1 (ß-catenin) (approximately 30% of low-grade endometrioid carcinomas), and PIK3R1 (about 20%–45% of cases), whereas mutations of TP53 (approximately 80%–90% of cases), FBXW7 (approximately 20%–30% of cases), and PPP2R1A (approximately 20%–30% of cases) are more frequently found in type II cancers (Tashiro et al., 1997; Kuhn et al., 2012; Matias-Guiu and Prat, 2013; Yeramian et al., 2013). Additionally, 25%–40% of type I carcinomas have a mutant phenotype leading to microsatellite instability (MSI), while this is quite less common in type II carcinomas. MSI typically involves repeated sequences (that is, MSI increases the mutation rate) (Catasus et al., 1998). Also, mutations in PIK3CA are present in about similar numbers in both types of cancer: of these mutations, 15%–20% are found in type I, while the remaining 80%–85% are found in type II (Oda et al., 2005; Bashir et al., 2014). Also, around 30% of grade 3 endometrioid carcinomas include mutations in the TP53 gene (Lax et al., 2000).
It is another example of the importance of risk factors being taken into consideration while doing molecular pathogenesis research. While the constant replacement of the surface epithelium of the ovary may result in p53 mutations piling up in the ovarian or fallopian tube epithelium, it is postulated that p53 mutations accumulate in the surface epithelium of the ovary or fallopian tube, causing genetic harm (Fathalla, 1971; Lee et al., 2007). Conversely, endometrial cancer seems to be without this similar mechanism. “Recurrent uterine lining growth and disruption” may occur, even if the term “incessant ovulation” just means “recurrent menstruation.” In the case of DNA replication errors during cell division, the probability of random genetic mutations developing increases with the number of cell divisions. There are thousands of mutations occurring in each cell cycle to increase the risk of multistep carcinogenesis, yet this may amount to tens of thousands of mutations per gram of proliferative tissue (Moolgavkar and Knudson, 1981; Cairns, 1998). Spontaneous mutations of another tumor-suppressor gene, PTEN, are often seen in the histologically normal endometria of regularly cycling premenopausal women.
The presence of somatically acquired inactivating mutations and/or deletions of the PTEN tumor-suppressor gene in 30%–50% of sporadic endometrial carcinomas favors PI3K/Akt/mTOR signaling in endometrial cancer (Tashiro et al., 1997; Ali, 2000). In the most recent studies, PTEN inactivation has been discovered in up to 83% of the endometrioid endometrial adenocarcinomas (Mutter, 2000). The PI3K/Akt signaling pathway is controlled by the enzyme protein tyrosine phosphatase-T, or PTEN, which inhibits it. PI3K is constitutively active whenever it is activated by mutations that activate PI3K kinase. Downstream pAkt is downstream of Akt, whereas downstream of PTEN is downstream of mTOR. mTOR is a member of the phosphatidylinositol kinase-related kinases. The catalytic activity of an enzyme is regulated by the mitogen-activated protein kinase (MAPK) and the phosphatidylinositol 3-kinase (PI3K)/Akt pathway. mTORC1 activity is regulated by p70S6K and 4E-binding protein 1 (4E-BP1) (Rosenwald et al., 1993).
MSI and mutations in K-ras, B-Raf, FGFR2, PI3K, and beta-catenin may also be found in endometrioid endometrial cancer (Horst et al., 2012; Katoh et al., 2012). This activation of the enzyme K-Ras (which results in an active MAPK) regulates the expression of the proteins ER and beta-catenin, thus controlling their activity. ER actively binds to promoters of pro-growth genes, resulting in higher rates of transcription and, subsequently, cell proliferation. In estrogen-only hormone replacement therapy, the risk of endometrial cancer is greatly increased (Furness et al., 2012).
Tumor cell growth in a cancer patient is influenced by the biological microenvironment inside and outside the cell (Zheng et al., 2021). Factors like mutated p53, PTEN, and KRAS play a crucial role in endometrial cancer progression, but metastasis occurs only after the support of the microenvironment (Richter et al., 2020). Cells like endothelial cells, macrophages, myofibroblasts, and inflammatory cells communicate via cytokines, ligands receptors, and growth factors with EC cells and create a suitable microenvironment for tumor cell invasion and metastasis. The factors of microenvironment-derived signals and stromal cell-derived proteins are potential targets as biomarkers for the detection of metastatic EC (Cheng et al., 2021).
Stromal myofibroblasts (SMs) are the most dominant contributors in the microenvironment of EC and many other types of the cancer cell. SMs produce an environment of cytokines to support the growth of EC, their mobility, and metastasis. Some studies show that the interaction between SMs and EC is with the help of hepatocyte growth factor (HGF) pathways (Cancer Genome Atlas Research Network, 2017). The pathway begins with the production of HGFs by endometrial myofibroblasts followed by the interaction of HGF with the receptor on EC cells inducing the EC cell’s invasion and metastasis. Cancer-associated fibroblasts (CAFs) are the plastic host mesenchyme fibroblasts present in the microenvironment surrounding the EC tumor cells. These CAFs are activated by tumor cells, which appear like normal tissue-resident fibroblasts with modified functions like cardinal elongated fibroblastic function, and a mesenchymal marker. CAFs support EC growth, tumor progression, and drug resistance development, but they still do not play a direct role in tumor management (Soslow et al., 2019). CAFs take part in a matrix organization, and stromal structure formation is also known as stromagenesis. This stroma functions as a path to control metastasis of tumor cells and their adhesion to normal cells, migration, and invasion. Recent research based on ex vivo and in vivo studies reported that myofibroblasts stimulate EC progression using stromal-derived factor-1 (SDF-1) for chemoattracting the tumor niche. SDF-1 binds with chemokine receptor 4 (CXCR4) to control normal and malignant cell trafficking. Studies based on qPCR and immunohistochemistry found high-level CXCR4 mRNA in EC tissue samples indicating its involvement in triggering endometrial tumor cell invasion (Soslow et al., 2019).
Macrophages, due to their dual function, can either promote the invasion of a cancer cell or inhibit its progression. They are the major component of the tumor cell microenvironment as they release many cytokines, growth factors, and chemokines and also help in EC tumor invasion. The two basic macrophage phenotypes recorded are M1 and M2 (Akbani and Levine, 2016; Cancer Genome Atlas Research Network, 2017). M1 is considered an antitumor with its cytotoxic potential, and M2 is considered a pro-tumor with its tissue and wound healing properties. High-grade EC or type II endometrioid carcinomas carry more stromal M2 tumor-associated macrophages in comparison to type 1 adenocarcinoma. In vivo studies have demonstrated that the colony-stimulating factor-1 (CSF-1) when expressed on EC facilitates macrophage recruitment through blood vessels (Yamada et al., 2016). These macrophages remain in the perivascular area of the uterus and induce endometrial carcinogenesis by producing TNF -α (tumor necrosis factor-α), IL-6, (interleukin-6), and IL-1β (interleukin-1 β). These macrophage-derived cytokines help in tumor cell growth and migration to the secondary site (Hu et al., 2016) (Figure 1).
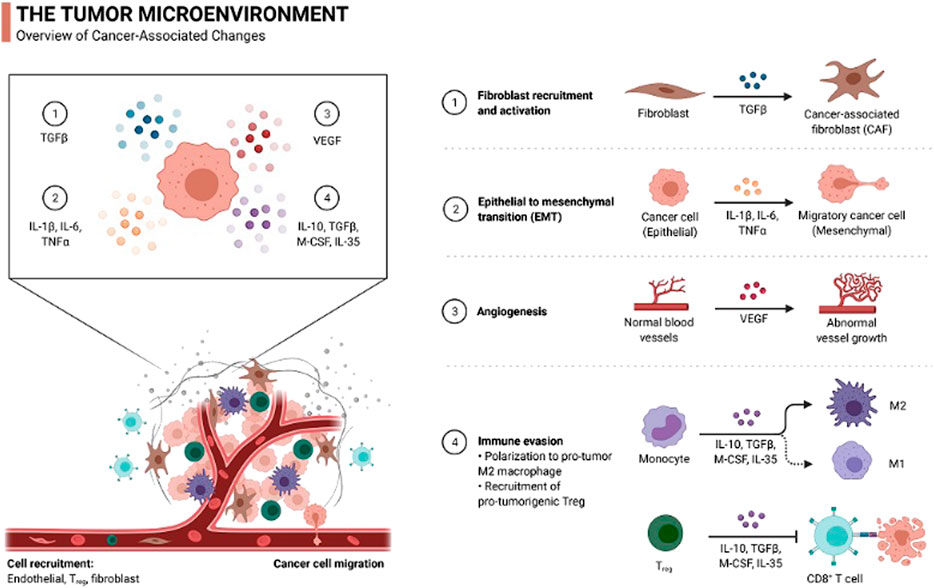
FIGURE 1. Different types of cells and their function in the EC microenvironment. The microenvironment consists of different components such as myofibroblasts, epithelial cells, and immune cells. (1) TGF-β produced by influx monocytes helps in the conversion of fibroblasts to cancer-associated fibroblasts (CAF). (2) Macrophages induce endometrial carcinogenesis by producing TNF-α, IL-6, and IL-1β that facilitate the conversion of cancer cells to migratory mesenchyme cancer cells. (3) VEGF produced by tumor cells promotes angiogenesis. (4) High-grade EC carries more M2 macrophages, which facilitates more macrophage recruitment via blood vessels.
Endometrial or uterine cancer and its metastasis are under the control of many regulatory signaling pathways. Different signaling pathways in the development of EC identified based on many current studies include the APC/β-catenin pathway based on WNT/β-catenin signaling transduction cascade (Katoh and Katoh, 2007), ErbB signaling pathway (member of the receptor tyrosine kinase family), mTOR/APC signaling pathway, VEGF ligand-receptor signaling pathway, and p53/p21 signaling pathway. These pathways help in escaping apoptosis, inducing cell proliferation, stopping or suspending cell differentiation, and enhancing angiogenesis (Lustig and Behrens, 2003). Here we are discussing some of these signaling pathways in more detail.
Wnt signaling factors are responsible for cell growth, movement, and differentiation in embryonic growth. Wnt is found to be responsible for the varied signaling cascade activation in tumor formation. Wnt signaling pathways are classified into two types based on the involvement of canonical: canonical and Wnt/β-catenin-dependent signaling pathways and non-canonical Wnt/calcium pathway. β-Catenin-dependent signaling pathways are found associated with human EC/uterine cancer. During the menstruation cycle, the estradiol works as an enhancer for Wnt/β-catenin-dependent signaling pathways, whereas progesterone is found to inhibit Wnt signaling by FOXO1 and DKK1 expression (Jenwitheesuk et al., 2017). The activation of the Wnt signaling pathway causes endometrial hyperplasia resulting in EC.
Tp53 and p16 are two genes connected with apoptotic disorders significant in many human cancers (Markowska et al., 2014). p53 is related to repairing the damages in DNA, and any mutation in Tp53 results in abnormal p53 protein production which leads to uncontrolled cancer cell growth as found in EC patients. p16 gene mutations change epigenetic modifications and enhance EC carcinogenesis (Kobayashi et al., 2013).
Since ancient times, several medicinal plant extracts and their active components have been demonstrated to have potential uses as anticancer agents (Shen and Singla, 2020; Madaan et al., 2021; Marzocco et al., 2021; Singla, 2021; Singla et al., 2021c; Singla et al., 2021d). Over the last three decades, drug discoveries based on natural products have received considerable interest (Singla et al., 2020a; Singla et al., 2020b; Bansal et al., 2021; Singla et al., 2021b; Sultana et al., 2021). An investigation indicates that a minimum of one-third of the marketed drugs either originated or were derived from different natural resources. Numerous structures of natural product-derived drugs have comprehensively been reported in the literature (Lagana et al., 2019; Singla and Dubey, 2019; Singla et al., 2019; Singla et al., 2021a). Paclitaxel (from the Taxus species), camptothecin (from Camptotheca acuminata Decne.), and vincristine and vinblastine (from Catharanthus roseus (L.) G. Don) are some of the most common plant-derived anticancer products. Besides, many other phytochemicals exhibited remarkable effects against various types of cancer. They show such effectiveness by alternating the cancer initiation, development, and progression as well as interrupting several mechanisms like differentiation, cellular proliferation, angiogenesis, apoptosis, and metastasis (Dutta et al., 2019; Gouveia et al., 2019).
A vast number of phytochemicals and natural products demonstrate efficacy via numerous signaling pathways to exert tumor inhibitory and antiproliferative effects. Curcumin, caffeic acid, viscolin, pulchranin, retinoids, resveratrol, and guanidine alkaloids exhibit anticancer potential through the regulation of activator protein-1, along with other mechanisms. Many oncogenic transcription factors, such as NF-κB, FoxM1, HIF1, Wnt/β-catenin, and Hh/GLI, are targeted by several potent natural structures. Some of these natural compounds include celastrol, oleanolic acid, ursolic acid, boldine, emodin, resveratrol, genistein, dentatin, garcinol, curcumin, and epigallocatechin gallate (Tewari et al., 2019).
Kinase proteins play a vital role in metabolism, cell signaling, protein regulation, cell trafficking, secretion processes, and many other cellular pathways. Their prominent physiological role is reflected in pharmacological research, where kinase-associated preparations can be found in up to one-quarter of contemporary drug discovery. Among them, the key players are PIK3CA, BRAF, and epidermal growth factor receptor (EGFR), which activate various tumor cell signaling pathways in endometrial cancer cells (Bhullar et al., 2018). Cyclin-dependent kinase Cdc2 (cell division cycle 2), also known as Cdk, is an important regulator at G2/M transition as one of the major checkpoints in the cell cycle. Wee1 kinase and Cdc25C phosphatase are responsible for the regulation of the activity of Cdc2 by phosphorylation and dephosphorylation. Cdc2 binds to cyclin B1 to form a complex that is activated at the onset of mitosis by dephosphorylation of the inhibitory sites on Cdc2 by functional Cdc25C. p21WAF1/CIP1, a member of the CDK inhibitor (CDKI) family, can be bound to cyclin B1–Cdc2 complexes and inactivate it.
Therefore, natural kinase inhibitors (Figure 2 represents some of the key examples) for the management of endometrial/uterine cancer are being discussed below.
Quercetin is a wildly distributed flavonoid present in a variety of fruits, vegetables, leaves, seeds, and grains. Prior studies have shown its cytotoxic activity against several cancerous cell lines including skin, lung, colon, and breast (Tang et al., 2020). The growth of endometrial cancer cells (Ishikawa cell line) was treated by quercetin (1, 10, and 100 μM) and inhibited by 3%, 51%, and 87%, respectively. It could also significantly reduce the gene and protein expression of EGF and cyclin D1 at 100 μM. In another study, it was reported that phosphatidylinositol 4-kinase (PI kinase) and phosphatidylinositol 4-phosphate 5-kinase (PIP kinase) were inhibited by quercetin (Kaneuchi et al., 2003). Kinetic analysis has shown that quercetin is a remarkable ATP competitive CK2 inhibitor. Some other kinases such as MEK-1, GSK-3, Hck, and IKKα/β are also inhibited by quercetin at very low concentrations (Hou and Kumamoto, 2010; Baier et al., 2017).
Eupatilin (5,7-dihydroxy-3′,4′,6-trimethoxyflavone), an O-methylated flavone mainly found in Artemisia plants, can enhance the activation and phosphorylation of Chk2/Cdc2 checkpoint proteins causing cell cycle arrest in endometrial cancer cells. In addition, eupatilin regulated the phosphorylation of protein kinases ERK1/2, Akt, ATM, and Chk2. It can inhibit the cell growth of Hec1A and KLE (human endometrial cancer cells) with IC50 values 82.2 and 85.5 μM, respectively, more potent than that of cisplatin (IC50 = 225.6 and 186.3 μM) (Cho et al., 2011; Nageen et al., 2020). The IC50 of jaceosidin, another abundant flavone in Artemisia species, in Hec1A and KLE cells was 70.54 and 147.14 μM, respectively, which was also markedly lower than that of cisplatin. It has been demonstrated that jaceosidin induces growth inhibition in human endometrial cancer cells by increasing the population of cells in the G2/M phase of the cell cycle as well as eupatilin. It can increase p21 expression in Hec1A cells and induce cell cycle arrest by ATM phosphorylation. Moreover, the cell cycle arrest in the G2/M phase is a result of phosphorylation of the Chk1 and Chk2 proteins, and subsequently phosphorylation and inactivation of Cdc25C.
Resveratrol has stilbenoid chemistry which is produced by more than 70 species of plants in response to stress, injury, infection, or ultraviolet radiations (Sirotkin, 2021). Resveratrol was found to have an apoptotic induction in uterine cancer cells. It has been shown that at a low dose of 10 μM, it can increase apoptosis in HeLa, EN-1078D, HEC-1A, and RL95-2 cells. However, it was revealed in Ishikawa cells at a much higher concentration. Resveratrol showed variable effects on protein kinase C in vitro (Sexton et al., 2006). It has a strong inhibitory activity on PKCα isomers (IC50 = 2.0 μM), whereas other isomers are not affected by resveratrol significantly. It is also described as a moderate inhibitor of several tyrosine kinases and serine/threonine protein kinases, such as Src, ERK1/2, JNK1/2, p38, PKC, PI3K, PKB, and IKK (Slater et al., 2003).
Honokiol (3′,5-di-(2-propenyl)-1,1′-biphenyl-2,4′-diol) is a bioactive natural lignan derivative isolated from the leaves and barks of Magnolia spp. It belongs to the class of neolignans. Honokiol showed anti-inflammatory, anti-angiogenic, antioxidative, and anticancer activities (Arora et al., 2012). Further research has demonstrated that honokiol is involved in the inhibition of the PI3K/Akt/mTOR pathway and regulation of the EGFR signaling pathway through suppression of EGFR expression and phosphorylation. Moreover, honokiol inhibits the activation of the mTOR via inhibition of the ERK1/2 pathway (Banik et al., 2019).
Osthole (7-methoxy-8-(3-methyl-2-butenyl)-2H-1-benzopyran-2-one) is a natural coumarin first derived from Cnidium plant. Angelica, Archangelica, Citrus, and Clausena are other sources of osthole. Due to its multiple bioactive properties, it has been considered a promising multitarget drug (Zhang et al., 2015). Experiments involving osthole treatment showed that osthole exposure significantly suppressed the growth of human endometrial cancer cell lines (JEC, KLE, and Ishikawa) in a dose- and time-dependent manner, and osthole displayed the most toxicity on JEC cells. Additionally, osthole could increase the expression of PTEN and inhibit PI3K/AKT activity, a key factor in the progression of endometrial cancer, and consequently lead to apoptosis of JEC cells (Liang et al., 2021).
Esculetin is a coumarin derivative that suppresses endometrial cancer proliferation and induces apoptosis by downregulating the BCLXL and XIAP via hnRNPA1. Its anticancer actions have been analyzed in vivo in nude mouse xenograft models of human endometrial cancer (Jiang et al., 2021).
Psammaplin A is a natural histone deacetylase inhibitor, extracted from the sponge’s species, i.e., Poecillastra sp. and Jaspis sp., which causes the cell cycle arrest and precipitates cell apoptosis in uterine tumor cells, accompanied by the activation of p21WAF1 and downregulation of pRb and cyclins/CDKs via the p53-independent pathway (Ahn et al., 2008).
The action of psammaplysene A on cell proliferation and viability has been investigated by using the BrdU incorporation and cell viability assay, respectively, which showed that it induces cell apoptosis in uterine tumor cells via FOXO1 (Berry et al., 2009).
The in vitro studies revealed that curcumin prevents the endometrial carcinoma (EC) cells from invading and migrating. It has anti-metastatic properties due to a decrease in the functioning of matrix metalloproteinases (i.e., MMP-2 and MMP-9) which causes the incursion of myometrial carcinoma to the lymph nodes in type-II of EC (Pourhanifeh et al., 2020).
Plumbagin is a naphthoquinone derivative found primarily in medicinal plants and belongs to the secondary metabolites of plants that persuade the arrest of the Ishikawa cell cycle and cell apoptosis by the Akt/PI3K signaling pathway in uterine tumor cells (Zhang X. et al., 2021).
Asparanin A is a spirosteroid saponin (25S-5β-spirostan-3β-ol-3-O-β-Dglucopyranosyl-(1→2)-β-D-glucopyranoside), which is also known as timosaponin A-III, isolated from Asparagus officinalis L. root. It has been also isolated from other medicinal herbs Anemarrhena asphodeloides Bunge and Asparagus cochinchinensis (Lour.) Merr. Asparanin A showed various cytotoxic effects in several cancer cell lines such as liver, colon, and pancreatic cancer. It highly inhibited Ishikawa cell (human EC) proliferation in a time- and dose-dependent manner (IC50 = 9.34 μM). It was found to be a very potent reducer of the level of PI3K, AKT, and mTOR pathway in Ishikawa cells, and the expression of p-AKT was markedly reduced. Simultaneously, a significant apoptosis induction via the mitochondrial pathway has been reported by Asparanin A (Zhang et al., 2019). In a multi-omics study, Zhang et al. revealed that apoptosis and autophagy marker proteins including PARP, Bcl-2, AMPK, mTOR, and p62 were notably downregulated, while the cleaved-PARP (c-PARP), ULK1, and LC3II/LC3I were upregulated in Ishikawa cells. It is also found that Asparanin A could induce DNA damage in Ishikawa cells and activate the p53 signaling pathway. The genes ATM, ATR, and CHEK2 were highly expressed and downregulated significantly (p < 0.01), which revealed that DNA damage occurred in Ishikawa cells (Zhang F. et al., 2021).
Steroidal saponin and dioscin (diosgenyl 2,4-di-O-α-L-rhamnopyranosyl-β-D-glucopyranoside) have been isolated from several natural sources, most of which are Dioscoreaceae plants. It is one of the steroidal saponin ingredients from a famous Chinese medicinal plant, Polygonatum odoratum (Mill.) Druce (Asparagaceae). Polygonatum is recently considered as a promising new anticancer agent. Numerous steroidal compounds have been reported from Polygonatum with strong immunomodulatory and antitumor activity on different types of cell lines (Yang et al., 2019). Dioscin exhibited the lowest cytotoxicity effects, among them on Ishikawa cells (IC50 = 2.37 μM). Previous studies revealed that dioscin can inhibit cancer cell proliferation at different stages through various pathways. It led to the downregulation of Cyclin D, CDK 6, Cyclin A2, Cyclin E, and CDK2 in the G0/G1 phase of the cell cycle through the p21/Cyclin E/CDK2 pathway. Some other signaling pathways such as PI3K/Akt/mTOR and p38/MAPK/JNK are also reported for the anticancer activities of dioscin (Li X. L. et al., 2021). Molecular docking suggests that dioscin can bind to AKT and mTOR to inhibit their phosphorylation (Mao et al., 2020).
Caudatin, a natural glycosidic compound extracted from the Chinese herb “baishouwu,” is a root tuber of Vincetoxicum auriculatum (Royle ex Wight) Kuntze that promotes cellular morphological changes and inhibits the proliferation of the cell, formation of colonies, and migration and persuades to apoptosis. The anticancer potential of caudatin has been investigated in vivo in HEC-1A and HeLa cell lines by using the MTT assay method on BALB/c mice. Caudatin inhibits the tumor progression by targeting the cytochrome c/caspase and TNFAIP1/NF-κB signaling pathways, as well as reducing the clonogenicity and viability of uterine cancerous cells (Tan et al., 2016).
Chen and his team had evaluated the potential of wogonoside, a potent flavonoid from Scutellaria baicalensis Georgi, for the treatment and management of endometrial cancer. They had observed that Wogonoside could upregulate the Mammalian Ste20-like kinase 1, followed by activation of the Hippo signaling pathway. Wogonoside was also found to potentiate the level of ER stress (Chen et al., 2019).
Berberine (2,3-methylenedioxy-9,10-dimenthoxyprotoberberine chloride) is currently a well-known dietary supplement with numerous pharmacological activities such as antibacterial, anti-inflammatory, antioxidant, and antitumor activities. It is an isoquinoline alkaloid that is found in the roots, rhizomes, and stem bark of Berberis spp. (Mortazavi et al., 2020). Berberine showed remarkable inhibitory effects on cell proliferation in different cancer types through various pathways. It exhibited significant cytotoxic effects on human EC cell lines (AN3 CA, HEC-1-A, and KLE cells) in a time- and dose-dependent manner. With the various concentrations of berberine, the levels of p-Akt and Akt were markedly declined in all three cells. These results suggested that berberine certainly can also halt cell cycle progression and trigger apoptosis via modulation of the PI3K/Akt signaling pathway in endometrial cancers (Kuo et al., 2015). Experiments have exhibited that berberine can also reduce the HER-2 expression or reduce the phosphorylation on various tumor cells (Pierpaoli et al., 2015).
Sweet wormwood (Artemisia annua L.) is a popular medicinal herb, and it has been used as a remedy for chill and fever in East Asia. Several sesquiterpene lactones such as artemisinin have been isolated from sweet wormwood. Recent studies have shown that artemisinin and its derivatives display strong antitumor effects and apoptosis induction in various human cancer cell model systems such as colon, melanoma, breast, ovarian, prostate, central nervous system, leukemic, renal, and endometrial cancer cells (Wong et al., 2017).
It was reported that artemisin dose-dependently had a remarkable cytotoxic activity against Ishikawa human endometrial cancer cell lines. It can induce a G1 cell cycle arrest of Ishikawa cells by downregulating the transcript and protein levels of CDK2 and CDK4 (Tran et al., 2014; Li D. et al., 2021).
Methyl jasmonate is a methyl ester compound that acts as a plant signal-regulating plant morphogenesis and responses to abiotic and biotic stresses. It was isolated as a fragrant compound present in Jasminum essential oil and other plant species for the first time (González-Aguilar et al., 2006). It exhibited a potent pro-apoptotic effect and significant cytotoxicity in human endometrioid Ishikawa and ECC-1 cell lines (Type I) and uterine serous papillary (USPC-1 and USPC-2; Type II). Methyl jasmonate seems to be involved through the PI3K/AKT pathway in sarcoma cell toxicity (Elia and Flescher, 2008). It also enhanced ERK1/2 activity in breast cancer cells. Several studies have revealed that an overexpression of receptor tyrosine kinase (IGF1R) which mediates actions of insulin-like growth factor 1 (IGF1) is related to endometrial cancer. Since AKT and ERK1/2 play important roles in IGF1R signaling, the apoptotic and anti-proliferative action of methyl jasmonate in endometrial cancer cells can be enhanced by co-targeting the insulin-like growth factor-1 receptor (IGF1R) signaling pathway. In addition, it showed remarkable cytotoxic activity in combination with NVP-AEW541, as a selective IGF1R tyrosine kinase inhibitor (Bruchim et al., 2014).
Curcusone C, a rhamnofolane diterpene, present in Jatropha curcashas, was found to significantly produce dose-dependent antiproliferative and apoptotic effects in endometrial cancer Ishikawa and HEC-1A cells. Along with other mechanisms, Curcusone C was observed as a MAPK activator and modulating the MAPK/ERK pathway as ERK inhibitor U0126 could attenuate the effect of Curcusone C (An et al., 2020).
Nerolidol is a well-known sesquiterpene alcohol reported from various aromatic essential oils. Dong and the team had studied its significance in the treatment and management of uterine fibroids (UF) by testing the cis- and trans-nerolidol in ELT3 cells, a rat leiomyoma cell line. A dose-dependent antiproliferative activity of nerolidol was reported along with induction of cell cycle arrest in the G1-phase, through the modulation of the ATM/Akt pathway, downregulation of cyclin-dependent kinase 4 (CDK4), and CDK6 protein expression (Dong et al., 2021).
Hinokitiol showed significant anti-proliferative activity against human endometrial cancer cell lines (Ishikawa, HEC-1A, and KLE). The IC50 value was 13.33 μM in the Ishikawa cells, 49.51 μM in the HEC-1A cells, and 4.69 μM in the KLE cells (Chen et al., 2021). Zhang and the team had studied the antiproliferative effects of garcinol against endometrial cancer cells (Ishikawa (ISH) and HEC-1B cell lines). They had reported that garcinol was exhibiting dose-dependent activity with cell cycle arrest at the G1 phase and G2/M phase, by downregulating CDK2, CDK4, cyclin D1, and cyclin B1 as well as upregulating phosphorylated c-JUN N-terminal kinase (JNK) and p-c-JUN (Zhang et al., 2020).
Pheophorbide A is a natural compound that is extracted from Scutellaria barbata D. Don, a traditional Chinese medicine. It has been found to exhibit a dose-dependent growth inhibitory impact on human endocrine sarcoma cell lines (MESSA) (Mansoori et al., 2019).
EC, which arises from the uterine lining, is one of the most common gynecological cancers. Various techniques to treat endometrial cancer have been explored throughout the years, among which natural herbal treatments are now proving to be a beneficial strategy (El Khoury et al., 2019). Scientific evidence revealed that phytochemicals have been found to exhibit substantial anticancer potential. Preclinical screening models have been resulting in potential lead compounds for the development of anticancer drugs through pervasive information about preliminary efficiency, pharmacokinetics studies, toxicity profile, and safety measures, which aid to determine whether a compound ought to be taken further for clinical trial purposes in bench-to-bedside processes of drug development (Choudhari et al., 2019).
Ecteinascidin-743 (Figure 3) (ET-743), also known as Trabectedin, is a new marine-derived tetrahydroisoquinoline natural compound, isolated from Ecteinascidia turbinata Herdman. It binds to the minor groove of DNA in a sequence-specific manner, affecting the transcription-regulated genes selectively. After demonstrating a higher therapeutic index and potency in preclinical research, ET-743 rapidly proceeded to the phase-I clinical trials. In a clinical phase II investigation, ET-743 was shown to be effective as a second-line treatment therapy for individuals with recurrent or persistent EC (McMeekin et al., 2004).
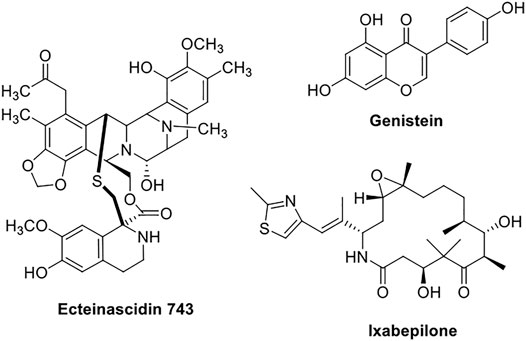
FIGURE 3. Chemical structures of phytochemicals (Ecteinascidin-743, genistein, and Ixabepilone) used in endometrial cancer in clinical studies.
Genistein (Figure 3) is a naturally occurring isoflavone derivative that binds to the endoplasmic reticulum (ER) as an agonist or antagonist, acting as a selective ER modulator having full agonistic activity on the α-receptor and partial agonistic activity on the β-receptor, demonstrating a modulatory action of genistein on cell apoptosis and growth-related pathways in the hyperplastic endometrium. In the presence of a hyperestrogenic environment in premenopausal women with endometrial hyperplasia, genistein exhibited a substantial reduction in the endometrial hyperplasia and associated symptoms (Bitto et al., 2015).
Ixabepilone (Figure 3) is an analog of epothilone B, a natural product. The response rate of ixabepilone in patients with persistent or recurrent endometrial cancer who have progressed despite standard therapy was examined in a clinical phase II investigation. A total of 52 patients were enrolled in this research, with 50 of them being eligible. Ixabepilone exhibited modest activity with short-term duration as a second-line treatment in endometrial adenocarcinoma (Dizon et al., 2009).
No matter how many in vitro preclinical studies have shown the potent anticancer capabilities of various natural substances, the findings have been unable to be translated into human trials because of several roadblocks (Leonarduzzi et al., 2010; Jeetah et al., 2014). In the human body, low aqueous solubility, low stability, and poor oral bioavailability are all major problems for natural polyphenolic medicines (Khan et al., 2013; Gohulkumar et al., 2014; Singla et al., 2021c). Because these compounds undergo substantial digestion in the small intestine and liver, only certain conjugates reach the circulation and reach their intended organs. It has been shown that flavonoids can be converted into their various methylated, sulfated, and glucuronidated forms by the phase II enzymes catechol-O-methyltransferases (COMT), SULT 1A1, and uridine diphosphate-glucuronosyltransferases (UDPGTs). However, the potential bioactive properties of these derivatives are still largely unknown. As a result, the quantities of parent chemicals (aglycones) in human plasma are substantially lower than their pharmacologically active levels utilized in preclinical research, staying only in the nanomolar range (Muqbil et al., 2011). Future therapeutic applications of natural polyphenols need the development of innovative drug delivery systems. Nanotechnology has lately emerged as a helpful tool for overcoming the difficulties associated with the in vivo use of polyphenols. Nano-sized carriers have been employed to convey parent chemicals directly to the regions where they are required, such as malignant tissues, to demonstrate their powerful anticancer properties to their full therapeutic advantage. To maximize effectiveness and reduce probable systemic damage, tailored treatment may be necessary (Rahman et al., 2014; Singla et al., 2018; Madaan et al., 2021).
The size, shape, and other features of nanoparticles may be tailored to precisely target certain cells. They may be either passive or aggressive in their targeting of the tumor cells. Anticancer medications including nanostructures are manufactured in such a manner that they directly interact with tumor cells via ligand–receptor or antigen–antibody recognition. They may interact with cancer cells passively as well. Some researchers have suggested a possible explanation for the preferential accumulation of nanoparticles in solid tumors: There may be abnormal angiogenesis in these tumors that enhances the nanoparticle’s penetration and retention. Targeted administration improves bioavailability and lowers drug concentrations elsewhere in the body, therefore reducing toxicity to a negligible extent.
Additionally, nanoformulations encounter several challenges, including manufacture, distribution, interaction with biological systems, and toxicity (Ahmad et al., 2010; Akhter et al., 2011). In nanoformulation, particle size and size distribution are the most essential properties that affect biodistribution, pharmacokinetics, and safety. While tiny nanoparticles (less than 30 nm in diameter) are quickly eliminated by the excretory system, bigger nanoparticles (more than 200 nm) are taken up by mononuclear phagocytic cells (Desai, 2012). Nanoparticle stability and opsonization are further influenced by the features of the nanoparticle’s surface, including its charge, hydrophobicity, and functional groups (Gessner et al., 2000; Moghimi et al., 2001). As far as nanoformulation is concerned, the main issue is that nanoparticles themselves may be antigenic agents (Trynda-Lemiesz, 2004; Desai, 2012). As a consequence of plasma proteins opsonizing nanoparticles and recognizing them as foreign particles, the complement system may be activated, resulting in life-threatening allergy and hypersensitive reactions, anaphylactic shock, and humoral and cellular immunological responses. It has been shown that nanoformulations may cause thrombogenicity and hemolysis (Dobrovolskaia and McNeil, 2007).
Due to their limited solubility, tyrosine kinase inhibitors (TKIs) have a highly varied bioavailability, which is shown by several characteristics influencing absorption from the digestive tract. In terms of absorption, TKIs are largely reliant on the pH of the solution in which they are dissolved. It is determined whether or not TKIs take on the ionized or nonionized form in the circulation based on luminal pH and pKa, due to their weakly basic nature. Because of the stomach’s acidic environment, the ionized form of the drug dissolves more easily than the nonionized version. Because antacids and PPIs raise the stomach pH, they may reduce the bioavailability of TKIs when used in combination with these medications. Treatment failure and subtherapeutic exposure will occur from a lack of solubility of both forms of the medicine (Augustijns et al., 2014; van Leeuwen et al., 2014; Puszkiel et al., 2017). The passive diffusion of lipophilic and non-polarized TKIs through the lipid bilayer membrane is possible in a normal pH environment. Transmembrane channel-creating proteins carry polarized TKIs, while nonpolarized TKIs do not (Managuli et al., 2018). TKIs have several drawbacks that make them difficult to utilize in clinical practice, and nanomedicine has the potential to overcome these issues. Aside from preserving TKIs from the gastrointestinal tract’s severe environment, nanomedicines increase the intestinal absorption of TKIs and offer TKIs with a regulated release and sustained ionization rate. Nanomedicines are being developed to protect TKIs from the severe environment of the intestines. Even though the majority of TKIs are meant to be taken orally, nearly all of the FDA-approved nanomedicines are designed for intravenous use. Oral nanomedicine delivery presents many unique obstacles; hence, the development of new and better nanovesicles is needed.
Coupling nanotechnology has made it possible to alter the pharmacokinetics and pharmacodynamics of drug molecules. As reported by Han et al., quercitin which is the most abundantly available flavonoid found its use in anti-fibrosis treatment. However, bio-pharmaceutically, quercetin belongs to the BCS Class IV category, thus limiting its absorption. Han et al. prepared quercitin-based nanoparticles using the pulsed laser ablation method with increased solubility and drug release activity (Han et al., 2018). Pharmacologically, nanoparticles showed multifunctional effects via inhibition of Aβ aggregation, destabilizing Aβ fibrils, decreasing Aβ-induced oxidative stress and Aβ-mediated cytotoxicity, and opening new channels for the treatment of amyloid-related diseases. Another group of researchers increased the bioavailability of quercitin via preparing nanosuspensions consisting of stabilizers such as d-alpha-tocopherol acid polyethylene glycol succinate (TPGS)/soybean lecithin and metabolism inhibitors, i.e., piperine and sodium oleate (Li H. et al., 2021). The presence of stabilizers and metabolism inhibitors increased the oral bioavailability as evident by in vivo studies in animal models in comparison to traditional nanosuspensions.
Resveratrol’s poor aqueous solubility and light sensitivity are a major hurdle in its therapeutic efficacy. On exposure to light, the trans-isomer gets isomerized to cis-form which is less pharmaceutically active. Also, it has poor bioavailability due to pre-systemic metabolism. To tackle this, Peñalva et al. reported a formulation containing casein nanoparticles entrapping resveratrol for increasing its oral bioavailability with a payload of 30 μg/mg nanoparticle (Peñalva et al., 2018). The in vivo results showed zero-order release from nanoparticles and release was independent of pH conditions of gastro environment and release the resveratrol in the intestinal region thus decreasing the rate of pre-systemic metabolism.
Honokiol has poor aqueous solubility acting as a rate-limiting step in its biological efficacy. Researchers developed nanoparticles based on honokiol, using the liquid antisolvent precipitation technique which reduces the particle size of molecules and increases their aqueous solubility (Wu et al., 2018). The nanoparticles were prepared using the vacuum freeze-drying method and using HP-β-CD as a cryoprotectant. The solubility of nanoparticles increased remarkably (46.52 mg/ml in comparison to 0.08 mg/ml of free honokiol). Also, the in vitro study showed an increase in drug release from nanoparticles due to their amorphous nature and reduction in size, also on the same context the bioavailability also increased in animal models. Another group of researchers also reported nanosuspension of honokiol and an increase in bioavailability and its application in cardio-cerebrovascular diseases (Han et al., 2014).
Similarly, another group of researchers developed gold nanoparticles for photothermal and chemotherapy based on encapsulation of Eupatilin isolated from Artemisia argyi H.Lév. & Vaniot (Sun et al., 2021). The nanoparticles were able to permeate through the cytomembrane and locate themselves near the nucleus. Due to the synergism between NIR laser radiation and gold nanoparticles, the chemo photothermal activity of the molecule was enhanced. Radiation with laser light releases the eupatilin and increases the tumor cell killing ability.
Osthol, a coumarin derivative derived from Cnidium monnieri (L.) Cusson, found its use in gynecological disorders. Chemically it is lipophilic and has poor aqueous solubility and bioavailability. To enhance its bioavailability, researchers developed enteric-coated nanoparticles based on Eudragit S100 (Li et al., 2018). The nanoparticles showed an increase in plasma concentration and bioavailability in comparison to that per se. The presence of fluconazole inhibited the CYP3A4 pathway regulators; as a result, the metabolism of osthol is decreased and bioavailability increased. Curcumin has a similar kind of physical properties such as low aqueous solubility and bioavailability, thus limiting its use. Gupta et al. developed curcumin-encapsulated lipid nano-constructs (CLEN) (Gupta et al., 2020). The CLEN complexes showed an increase in solubility by a factor of 1.4 × 106 times in comparison to 11 ng/ml (normal curcumin). Other authors have also documented literature citing the benefits of nano-curcumin (Rahimi et al., 2016; Chopra et al., 2021).
Plumbagin is another natural compound possessing low aqueous solubility found in the Plumbaginaceae family. It mainly found its role in anticancer, antidiabetic, anti-inflammatory, etc., activities. Chrastina et al. developed nanoemulsions based on Plumbagin for enhancing its anticancer effect on the prostate tumor (Chrastina et al., 2018). Nanoemulsions consisted of oleic acid, polysorbate 80, and plumbagin. The nanoemulsions showed one phase exponential release profile of plumbagin with a half-life of 6.1 h in gastric fluid and 7.0 h in intestinal fluid. Also, plumbagin nanoemulsions showed increased cytotoxicity toward PTEN-P2 cells in comparison to free drugs. Also, another group of researchers developed plumbagin-coupled silver nanoparticles for their anticancer activity on HeLa cells (Appadurai and Rathinasamy, 2015). Plumbagin showed mitotic blockage and inhibition of the clonogenic survival rate along with silver nanoparticles, due to an increase in internalization of plumbagin.
Future research ought to emphasize the from-the-bench-to-the-clinic orientation. In silico, in vitro, and in vivo studies need to expand the reported knowledge about the effect of phytochemicals in EC. It is important to investigate how genetic variance, host–microbiome, and tumor microenvironment can affect the response to treatment. Intra-tumoral and inter-tumoral heterogeneity, the ability of tumors to contain and or differentiate toward different subtypes of cancer cells, is an important factor that can spearhead resistance to treatment and downplay the effect of CDKin in EC. Regular tissue and liquid biopsies have made the continuous monitoring of the tumors’ genomic profile possible and can provide explanations in case of decreased efficacy or failure of potent compounds in the treatment of EC. Organoids offer a promising platform for investigating and modulating the tumors’ microenvironment before researching animal models and humans. In the context of clinical trials, future research should investigate the etiology of treatment failure, when it occurs in a significant portion of the participants, to improve the design of future clinical trials and promote patient and tumor-specific treatment. It is also important to investigate when host parameters (microbiome, inflammation, levels of circulating tumor genetic material) affect the bioavailability, distribution, and metabolism of CDKin in the course of treatment.
Beyond precision medicine and translational research in oncology, it is important to associate findings of EC epidemiology studies with the presence of particular natural compounds in the nutrition or the living environment of patients (Chen et al., 2017; Chen et al., 2020). Potential correlations between the presence of particular compounds in areas where the frequency of EC is lower or the prognosis of the disease is better can indicate phytochemicals with major potential for further investigation. Surveying cultural perceptions that can affect the adherence of patients to adjunct treatments based on phytochemicals can also help design real-world clinical studies and interventions.
EC is a prevalent type of cancer accounting for significant disease burden, morbidity, and mortality. It is divided into two types. Type 1 is more prevalent, has a better prognosis, and is associated with several mutations and satellite instability mechanisms involving CDKs (PTEN, KRAS, ARID1A, CTNNB1, and PIK3RA1). Therefore, CDKin has major therapeutic potential in the management of the disease. Phenolic and non-phenolic natural compounds (Figure 4) serve as sources of CDKin, which can be applied in clinical practice.
In in vitro and in vivo studies, flavonoids such as quercetin and eupatilin compete with MEK-1, GSK-3, Hck, IKKα/β, and ERK1/2, Akt, ATM, Chk2, respectively. Stilbenoids such as resveratrol act as moderate inhibitors of tyrosine and serine/threonine protein kinases (Src, ERK1/2, JNK1/2, p38, PKC, PI3K, PKB, and IKK) and exert variable effects on PKC. Biphenyl compounds such as honokiol are involved in the inhibition of the PI3K/Akt/mTOR pathway. Coumarins such as osthole can increase the expression of PTEN and inhibit PI3K/AKT. Glycosides such as asparanin A have an inhibitory effect on the PI3K, AKT, and mTOR pathways, while alkaloids have such an effect on PI3K and AKT. Terpenes and terpenoids can downregulate the transcript and protein levels of CDK2 and CDK4. Phase two clinical trials investigating the effects of trabectedin, a marine-derived tetrahydroisoquinoline natural compound, and ixabepilone, an analog of epothilone B, reported short- and mid-term efficacy of these regimens as second-line treatments. On these grounds, it is a dire need to pursue translational research in the field, mapping the potential therapeutic effect of natural compounds’ derived CDKin and accelerating their proper investigation in clinical trials.
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
This work was supported by the National Natural Science Foundation of China (32070671), the COVID-19 research projects of West China Hospital Sichuan University (Grant no. HX-2019-nCoV-057), and the regional innovation cooperation between Sichuan and Guangxi Provinces (2020YFQ0019).
Author RS is honorary-based associated with the iGlobal Research and Publishing Foundation (iGRPF), India.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors, and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Ahmad, M. Z., Akhter, S., Jain, G. K., Rahman, M., Pathan, S. A., Ahmad, F. J., et al. (2010). Metallic Nanoparticles: Technology Overview & Drug Delivery Applications in Oncology. Expert Opin. Drug Deliv. 7 (8), 927–942. doi:10.1517/17425247.2010.498473
Ahn, M. Y., Jung, J. H., Na, Y. J., and Kim, H. S. (2008). A Natural Histone Deacetylase Inhibitor, Psammaplin A, Induces Cell Cycle Arrest and Apoptosis in Human Endometrial Cancer Cells. Gynecol. Oncol. 108 (1), 27–33. doi:10.1016/j.ygyno.2007.08.098
Akbani, R., and Levine, D. A. (2016). “Abstract 133: Integrated Molecular Characterization of Uterine Carcinosarcoma in the Cancer Genome Atlas (TCGA) Project,” in AACR 107th Annual Meeting 2016, New Orleans, LA, April 16-20, 2016 (New Orleans: Molecular and Cellular Biology, Genetics). doi:10.1158/1538-7445.am2016-133
Akhter, S., Ahmad, Z., Singh, A., Ahmad, I., Rahman, M., Anwar, M., et al. (2011). Cancer Targeted Metallic Nanoparticle: Targeting Overview, Recent Advancement and Toxicity Concern. Curr. Pharm. Des. 17 (18), 1834–1850. doi:10.2174/138161211796391001
Ali, I. U. (2000). Gatekeeper for Endometrium: the PTEN Tumor Suppressor Gene. J. Natl. Cancer Inst. 92 (11), 861–863. doi:10.1093/jnci/92.11.861
Ambros, R. A., and Kurman, R. J. (1992). Combined Assessment of Vascular and Myometrial Invasion as a Model to Predict Prognosis in Stage I Endometrioid Adenocarcinoma of the Uterine Corpus. Cancer 69 (6), 1424–1431. doi:10.1002/1097-0142(19920315)69:6<1424::aid-cncr2820690620>3.0.co;2-5
An, J., Li, L., and Zhang, X. (2020). Curcusone C Induces Apoptosis in Endometrial Cancer Cells via Mitochondria-dependent Apoptotic and ERK Pathway. Biotechnol. Lett. 43 (1), 329–338. doi:10.1007/s10529-020-03027-4
Appadurai, P., and Rathinasamy, K. (2015). Plumbagin-silver Nanoparticle Formulations Enhance the Cellular Uptake of Plumbagin and its Antiproliferative Activities. IET Nanobiotechnol 9 (5), 264–272. doi:10.1049/iet-nbt.2015.0008
Arora, S., Singh, S., Piazza, G. A., Contreras, C. M., Panyam, J., and Singh, A. P. (2012). Honokiol: a Novel Natural Agent for Cancer Prevention and Therapy. Curr. Mol. Med. 12 (10), 1244–1252. doi:10.2174/156652412803833508
Augustijns, P., Wuyts, B., Hens, B., Annaert, P., Butler, J., and Brouwers, J. (2014). A Review of Drug Solubility in Human Intestinal Fluids: Implications for the Prediction of Oral Absorption. Eur. J. Pharm. Sci. 57, 322–332. doi:10.1016/j.ejps.2013.08.027
Baier, A., Galicka, A., Nazaruk, J., and Szyszka, R. (2017). Selected Flavonoid Compounds as Promising Inhibitors of Protein Kinase CK2α and CK2α', the Catalytic Subunits of CK2. Phytochemistry 136, 39–45. doi:10.1016/j.phytochem.2016.12.018
Baier, A., and Szyszka, R. (2020). Compounds from Natural Sources as Protein Kinase Inhibitors. Biomolecules 10 (11). doi:10.3390/biom10111546
Banik, K., Ranaware, A. M., Deshpande, V., Nalawade, S. P., Padmavathi, G., Bordoloi, D., et al. (2019). Honokiol for Cancer Therapeutics: A Traditional Medicine that Can Modulate Multiple Oncogenic Targets. Pharmacol. Res. 144, 192–209. doi:10.1016/j.phrs.2019.04.004
Bansal, H., Singla, R. K., Behzad, S., Chopra, H., Grewal, A. S., and Shen, B. (2021). Unleashing the Potential of Microbial Natural Products in Drug Discovery: Focusing on Streptomyces as Antimicrobials Goldmine. Ctmc 21, 2374–2396. doi:10.2174/1568026621666210916170110
Bashir, S., Jiang, G., Joshi, A., Miller, C., Matrai, C., Yemelyanova, A., et al. (2014). Molecular Alterations of PIK3CA in Uterine Carcinosarcoma, clear Cell, and Serous Tumors. Int. J. Gynecol. Cancer 24 (7), 1262–1267. doi:10.1097/IGC.0000000000000183
Berry, E., Hardt, J. L., Clardy, J., Lurain, J. R., and Kim, J. J. (2009). Induction of Apoptosis in Endometrial Cancer Cells by Psammaplysene A Involves FOXO1. Gynecol. Oncol. 112 (2), 331–336. doi:10.1016/j.ygyno.2008.10.017
Bhullar, K. S., Lagarón, N. O., McGowan, E. M., Parmar, I., Jha, A., Hubbard, B. P., et al. (2018). Kinase-targeted Cancer Therapies: Progress, Challenges and Future Directions. Mol. Cancer 17 (1), 48. doi:10.1186/s12943-018-0804-2
Bitto, A., Granese, R., Polito, F., Triolo, O., Giordano, D., Squadrito, F., et al. (2015). Genistein Reduces Angiogenesis and Apoptosis in Women with Endometrial Hyperplasia. Btat 2015 (5), 27–32. doi:10.2147/btat.S67368
Bruchim, I., Sarfstein, R., Reiss, A., Flescher, E., and Werner, H. (2014). IGF1R Tyrosine Kinase Inhibitor Enhances the Cytotoxic Effect of Methyl Jasmonate in Endometrial Cancer. Cancer Lett. 352 (2), 214–219. doi:10.1016/j.canlet.2014.06.013
Byron, S. A., Gartside, M., Powell, M. A., Wellens, C. L., Gao, F., Mutch, D. G., et al. (2012). FGFR2 Point Mutations in 466 Endometrioid Endometrial Tumors: Relationship with MSI, KRAS, PIK3CA, CTNNB1 Mutations and Clinicopathological Features. PLoS ONE 7 (2), e30801. doi:10.1371/journal.pone.0030801
Cairns, J. (1998). Mutation and Cancer: the Antecedents to Our Studies of Adaptive Mutation. Genetics 148 (4), 1433–1440. doi:10.1093/genetics/148.4.1433
Cancer Genome Atlas Research Network (2017). Electronic address, a.a.d.h.e., and Cancer Genome Atlas Research, NIntegrated Genomic Characterization of Pancreatic Ductal Adenocarcinoma. Cancer Cell 32 (2), 185–203. e113. doi:10.1016/j.ccell.2017.07.007
Catasús, L., Matias-Guiu, X., Machin, P., Muñoz, J., and Prat, J. (1998). BAX Somatic Frameshift Mutations in Endometrioid Adenocarcinomas of the Endometrium: Evidence for a Tumor Progression Role in Endometrial Carcinomas with Microsatellite Instability. Lab. Invest. 78 (11), 1439–1444.
Chen, H. Y., Cheng, W. P., Chiang, Y. F., Hong, Y. H., Ali, M., Huang, T. C., et al. (2021). Hinokitiol Exhibits Antitumor Properties through Induction of ROS-Mediated Apoptosis and P53-Driven Cell-Cycle Arrest in Endometrial Cancer Cell Lines (Ishikawa, HEC-1A, KLE). Int. J. Mol. Sci. 22 (15), 8268. doi:10.3390/ijms22158268
Chen, S., Wu, Z., Ke, Y., Shu, P., Chen, C., Lin, R., et al. (2019). Wogonoside Inhibits Tumor Growth and Metastasis in Endometrial Cancer via ER Stress-Hippo Signaling axis. Acta Biochim. Biophys. Sinica. doi:10.1093/abbs/gmz109
Chen, Y., Yang, L., Hu, H., Chen, J., and Shen, B. (2017). How to Become a Smart Patient in the Era of Precision Medicine? Adv. Exp. Med. Biol. 1028, 1–16. doi:10.1007/978-981-10-6041-0_1
Chen, Y., Liu, X., Yu, Y., Yu, C., Yang, L., Lin, Y., et al. (2020). PCaLiStDB: a Lifestyle Database for Precision Prevention of Prostate Cancer. Database J. Biol. Databases Curation 2020. doi:10.1093/database/baz154
Cheng, P., Ma, J., Zheng, X., Zhou, C., and Chen, X. (2021). Bioinformatic Profiling Identifies Prognosis-Related Genes in the Immune Microenvironment of Endometrial Carcinoma. Sci. Rep. 11 (1), 12608. doi:10.1038/s41598-021-92091-5
Cho, J. H., Lee, J. G., Yang, Y. I., Kim, J. H., Ahn, J. H., Baek, N. I., et al. (2011). Eupatilin, a Dietary Flavonoid, Induces G2/M Cell Cycle Arrest in Human Endometrial Cancer Cells. Food Chem. Toxicol. 49 (8), 1737–1744. doi:10.1016/j.fct.2011.04.019
Chopra, H., Dey, P. S., Das, D., Bhattacharya, T., Shah, M., Mubin, S., et al. (2021). Curcumin Nanoparticles as Promising Therapeutic Agents for Drug Targets. Molecules 26 (16). doi:10.3390/molecules26164998
Choudhari, A. S., Mandave, P. C., Deshpande, M., Ranjekar, P., and Prakash, O. (2019). Phytochemicals in Cancer Treatment: From Preclinical Studies to Clinical Practice. Front. Pharmacol. 10, 1614. doi:10.3389/fphar.2019.01614
Chrastina, A., Baron, V. T., Abedinpour, P., Rondeau, G., Welsh, J., and Borgström, P. (2018). Plumbagin-Loaded Nanoemulsion Drug Delivery Formulation and Evaluation of Antiproliferative Effect on Prostate Cancer Cells. Biomed. Res. Int. 2018, 9035452–9035457. doi:10.1155/2018/9035452
Desai, N. (2012). Challenges in Development of Nanoparticle-Based Therapeutics. AAPS J. 14 (2), 282–295. doi:10.1208/s12248-012-9339-4
Dizon, D. S., Blessing, J. A., McMeekin, D. S., Sharma, S. K., DiSilvestro, P., and Alvarez, R. D. (2009). Phase II Trial of Ixabepilone as Second-Line Treatment in Advanced Endometrial Cancer: Gynecologic Oncology Group Trial 129-P. J. Clin. Oncol. 27 (19), 3104–3108. doi:10.1200/jco.2008.20.6995
Dobrovolskaia, M. A., and McNeil, S. E. (2007). Immunological Properties of Engineered Nanomaterials. Nat. Nanotechnol 2 (8), 469–478. doi:10.1038/nnano.2007.223
Dong, J. R., Chang, W. W., and Chen, S. M. (2021). Nerolidol Inhibits Proliferation of Leiomyoma Cells via Reactive Oxygen Species-Induced DNA Damage and Downregulation of the ATM/Akt Pathway. Phytochemistry 191, 112901. doi:10.1016/j.phytochem.2021.112901
Dutta, S., Mahalanobish, S., Saha, S., Ghosh, S., and Sil, P. C. (2019). Natural Products: An Upcoming Therapeutic Approach to Cancer. Food Chem. Toxicol. 128, 240–255. doi:10.1016/j.fct.2019.04.012
El Khoury, D., Matar, R., and Touma, T. (2019). Curcumin and Endometrial Carcinoma: an Old Spice as a Novel Agent. Int. J. Womens Health 11, 249–256. doi:10.2147/ijwh.S194262<
Elia, U., and Flescher, E. (2008). PI3K/Akt Pathway Activation Attenuates the Cytotoxic Effect of Methyl Jasmonate toward Sarcoma Cells. Neoplasia 10 (11), 1303–1313. doi:10.1593/neo.08636
Enomoto, T., Inoue, M., Perantoni, A. O., Buzard, G. S., Miki, H., Tanizawa, O., et al. (1991). K-ras Activation in Premalignant and Malignant Epithelial Lesions of the Human Uterus. Cancer Res. 51 (19), 5308–5314.
Fathalla, M. F. (1971). Incessant Ovulation-Aa Factor in Ovarian Neoplasia? Lancet 2 (7716), 163. doi:10.1016/s0140-6736(71)92335-x
Finn, R. S., Aleshin, A., and Slamon, D. J. (2016). Targeting the Cyclin-dependent Kinases (CDK) 4/6 in Estrogen Receptor-Positive Breast Cancers. Breast Cancer Res. 18 (1), 17. doi:10.1186/s13058-015-0661-5
Furness, S., Roberts, H., Marjoribanks, J., and Lethaby, A. (2012). Hormone Therapy in Postmenopausal Women and Risk of Endometrial Hyperplasia. Cochrane Database Syst. Rev. doi:10.1002/14651858.CD000402.pub4
Gessner, A., Waicz, R., Lieske, A., Paulke, B., Mäder, K., and Müller, R. H. (2000). Nanoparticles with Decreasing Surface Hydrophobicities: Influence on Plasma Protein Adsorption. Int. J. Pharm. 196 (2), 245–249. doi:10.1016/s0378-5173(99)00432-9
Gocek, E., Moulas, A. N., and Studzinski, G. P. (2014). Non-receptor Protein Tyrosine Kinases Signaling Pathways in normal and Cancer Cells. Crit. Rev. Clin. Lab. Sci. 51 (3), 125–137. doi:10.3109/10408363.2013.874403
Gohulkumar, M., Gurushankar, K., Rajendra Prasad, N., and Krishnakumar, N. (2014). Enhanced Cytotoxicity and Apoptosis-Induced Anticancer Effect of Silibinin-Loaded Nanoparticles in Oral Carcinoma (KB) Cells. Mater. Sci. Eng. C Mater. Biol. Appl. 41, 274–282. doi:10.1016/j.msec.2014.04.056
González-Aguilar, G., Tiznado-Hernandez, M., and Wang, C. (2006). Physiological and Biochemical Responses of Horticultural Products to Methyl Jasmonate. Stewart postharvest Rev. 2 (1), 1–9. doi:10.2212/spr.2006.1.1
Gouveia, D. N., Guimarães, A. G., Santos, W. B. D. R., and Quintans-Júnior, L. J. (2019). Natural Products as a Perspective for Cancer Pain Management: a Systematic Review. Phytomedicine 58, 152766. doi:10.1016/j.phymed.2018.11.026
Gupta, T., Singh, J., Kaur, S., Sandhu, S., Singh, G., and Kaur, I. P. (2020). Enhancing Bioavailability and Stability of Curcumin Using Solid Lipid Nanoparticles (CLEN): A Covenant for its Effectiveness. Front. Bioeng. Biotechnol. 8, 879. doi:10.3389/fbioe.2020.00879
Han, M., Yu, X., Guo, Y., Wang, Y., Kuang, H., and Wang, X. (2014). Honokiol Nanosuspensions: Preparation, Increased Oral Bioavailability and Dramatically Enhanced Biodistribution in the Cardio-Cerebro-Vascular System. Colloids Surf. B Biointerfaces 116, 114–120. doi:10.1016/j.colsurfb.2013.12.056
Han, Q., Wang, X., Cai, S., Liu, X., Zhang, Y., Yang, L., et al. (2018). Quercetin Nanoparticles with Enhanced Bioavailability as Multifunctional Agents toward Amyloid Induced Neurotoxicity. J. Mater. Chem. B 6 (9), 1387–1393. doi:10.1039/c7tb03053c
Horst, D., Chen, J., Morikawa, T., Ogino, S., Kirchner, T., and Shivdasani, R. A. (2012). Differential WNT Activity in Colorectal Cancer Confers Limited Tumorigenic Potential and Is Regulated by MAPK Signaling. Cancer Res. 72 (6), 1547–1556. doi:10.1158/0008-5472.Can-11-3222
Hou, D. X., and Kumamoto, T. (2010). Flavonoids as Protein Kinase Inhibitors for Cancer Chemoprevention: Direct Binding and Molecular Modeling. Antioxid. Redox Signal. 13 (5), 691–719. doi:10.1089/ars.2009.2816
Hu, H., Hang, J. J., Han, T., Zhuo, M., Jiao, F., and Wang, L. W. (2016). The M2 Phenotype of Tumor-Associated Macrophages in the Stroma Confers a Poor Prognosis in Pancreatic Cancer. Tumour Biol. 37 (7), 8657–8664. doi:10.1007/s13277-015-4741-z
Jeetah, R., Bhaw-Luximon, A., and Jhurry, D. (2014). Nanopharmaceutics: Phytochemical-Based Controlled or Sustained Drug-Delivery Systems for Cancer Treatment. J. Biomed. Nanotechnol 10 (9), 1810–1840. doi:10.1166/jbn.2014.1884
Jenwitheesuk, A., Boontem, P., Wongchitrat, P., Tocharus, J., Mukda, S., and Govitrapong, P. (2017). Melatonin Regulates the Aging Mouse Hippocampal Homeostasis via the Sirtuin1-FOXO1 Pathway. EXCLI J. 16, 340–353. doi:10.17179/excli2016-852
Jiang, R., Su, G., Chen, X., Chen, S., Li, Q., Xie, B., et al. (2021). Esculetin Inhibits Endometrial Cancer Proliferation and Promotes Apoptosis via hnRNPA1 to Downregulate BCLXL and XIAP. Cancer Lett. 521, 308–321. doi:10.1016/j.canlet.2021.08.039
Jorda, R., Hendrychová, D., Voller, J., Řezníčková, E., Gucký, T., and Kryštof, V. (2018). How Selective Are Pharmacological Inhibitors of Cell-Cycle-Regulating Cyclin-dependent Kinases? J. Med. Chem. 61 (20), 9105–9120. doi:10.1021/acs.jmedchem.8b00049
Kaneuchi, M., Sasaki, M., Tanaka, Y., Sakuragi, N., Fujimoto, S., and Dahiya, R. (2003). Quercetin Regulates Growth of Ishikawa Cells through the Suppression of EGF and Cyclin D1. Int. J. Oncol. 22 (1), 159–164. doi:10.3892/ijo.22.1.159
Katoh, M., and Katoh, M. (2007). WNT Signaling Pathway and Stem Cell Signaling Network. Clin. Cancer Res. 13 (14), 4042–4045. doi:10.1158/1078-0432.CCR-06-2316
Khan, N., Bharali, D. J., Adhami, V. M., Siddiqui, I. A., Cui, H., Shabana, S. M., et al. (2013). Oral Administration of Naturally Occurring Chitosan-Based Nanoformulated green tea Polyphenol EGCG Effectively Inhibits Prostate Cancer Cell Growth in a Xenograft Model. Carcinogenesis 35 (2), 415–423. doi:10.1093/carcin/bgt321
Kobayashi, H., Uekuri, C., Akasaka, J., Ito, F., Shigemitsu, A., Koike, N., et al. (2013). The Biology of Uterine Sarcomas: A Review and Update. Mol. Clin. Oncol. 1 (4), 599–609. doi:10.3892/mco.2013.124
Kuhn, E., Wu, R. C., Guan, B., Wu, G., Zhang, J., Wang, Y., et al. (2012). Identification of Molecular Pathway Aberrations in Uterine Serous Carcinoma by Genome-wide Analyses. J. Natl. Cancer Inst. 104 (19), 1503–1513. doi:10.1093/jnci/djs345
Kuo, H.-P., Lee, Y.-J., Hsu, C.-Y., Lee, S.-L., Hsu, S.-C., Chuang, T.-C., et al. (2015). Growth-suppressive Effect of Berberine on Endometrial Carcinoma Cells: Role of Mitochondrial and PI3K/Akt Pathway. J. Funct. Foods 17, 600–609. doi:10.1016/j.jff.2015.06.006
Laganà, P., Anastasi, G., Marano, F., Piccione, S., Singla, R. K., Dubey, A. K., et al. (2019). Phenolic Substances in Foods: Health Effects as Anti-inflammatory and Antimicrobial Agents. J. AOAC Int. 102 (5), 1378–1387. doi:10.5740/jaoacint.19-0131
Lax, S. F., Kendall, B., Tashiro, H., Slebos, R. J., and Hedrick, L. (2000). The Frequency of P53, K-Ras Mutations, and Microsatellite Instability Differs in Uterine Endometrioid and Serous Carcinoma: Evidence of Distinct Molecular Genetic Pathways. Cancer 88 (4), 814–824. doi:10.1002/(sici)1097-0142(20000215)88:4<814::Aid-cncr12>3.0.Co;2-u
Lax, S. F. (2004). Molecular Genetic Pathways in Various Types of Endometrial Carcinoma: from a Phenotypical to a Molecular-Based Classification. Virchows Arch. 444 (3), 213–223. doi:10.1007/s00428-003-0947-3
Lee, Y., Miron, A., Drapkin, R., Nucci, M. R., Medeiros, F., Saleemuddin, A., et al. (2007). A Candidate Precursor to Serous Carcinoma that Originates in the Distal Fallopian Tube. J. Pathol. 211 (1), 26–35. doi:10.1002/path.2091
Leonarduzzi, G., Testa, G., Sottero, B., Gamba, P., and Poli, G. (2010). Design and Development of Nanovehicle-Based Delivery Systems for Preventive or Therapeutic Supplementation with Flavonoids. Curr. Med. Chem. 17 (1), 74–95. doi:10.2174/092986710789957760
Leslie, K. K., Stein, M. P., Kumar, N. S., Dai, D., Stephens, J., Wandinger-Ness, A., et al. (2005). Progesterone Receptor Isoform Identification and Subcellular Localization in Endometrial Cancer. Gynecol. Oncol. 96 (1), 32–41. doi:10.1016/j.ygyno.2004.09.057
Li, D., Zhang, J., and Zhao, X. (2021). Mechanisms and Molecular Targets of Artemisinin in Cancer Treatment. Cancer Invest. 39 (8), 675–684. doi:10.1080/07357907.2021.1954190
Li, H., Li, M., Fu, J., Ao, H., Wang, W., and Wang, X. (2021). Enhancement of Oral Bioavailability of Quercetin by Metabolic Inhibitory Nanosuspensions Compared to Conventional Nanosuspensions. Drug Deliv. 28 (1), 1226–1236. doi:10.1080/10717544.2021.1927244
Li, L. P., Wang, X. J., Zhang, J. Y., Zhang, L. L., Cao, Y. B., Gu, L. Q., et al. (2018). Antifungal Activity of Osthol in vitro and Enhancement in vivo through Eudragit S100 Nanocarriers. Virulence 9 (1), 555–562. doi:10.1080/21505594.2017.1356503
Li, X. L., Ma, R. H., Ni, Z. J., Thakur, K., Cespedes-Acuña, C. L., Wang, S., et al. (2021). Dioscin Inhibits Human Endometrial Carcinoma Proliferation via G0/G1 Cell Cycle Arrest and Mitochondrial-dependent Signaling Pathway. Food Chem. Toxicol. 148, 111941. doi:10.1016/j.fct.2020.111941
Liang, L., Yang, B., Wu, Y., and Sun, L. (2021). Osthole Suppresses the Proliferation and Induces Apoptosis via Inhibiting the PI3K/AKT Signaling Pathway of Endometrial Cancer JEC Cells. Exp. Ther. Med. 22 (4), 1171. doi:10.3892/etm.2021.10605
Lustig, B., and Behrens, J. (2003). The Wnt Signaling Pathway and its Role in Tumor Development. J. Cancer Res. Clin. Oncol. 129 (4), 199–221. doi:10.1007/s00432-003-0431-0
Madaan, R., Singla, R. K., Kumar, S., Dubey, A. K., Kumar, D., Sharma, P., et al. (2021). Bergenin - a Biologically Active Scaffold: Nanotechnological Perspectives. Ctmc 21. doi:10.2174/1568026621666211015092654
Managuli, R. S., Raut, S. Y., Reddy, M. S., and Mutalik, S. (2018). Targeting the Intestinal Lymphatic System: a Versatile Path for Enhanced Oral Bioavailability of Drugs. Expert Opin. Drug Deliv. 15 (8), 787–804. doi:10.1080/17425247.2018.1503249
Mansoori, B., Mohammadi, A., Amin Doustvandi, M., Mohammadnejad, F., Kamari, F., Gjerstorff, M. F., et al. (2019). Photodynamic Therapy for Cancer: Role of Natural Products. Photodiagnosis Photodyn Ther. 26, 395–404. doi:10.1016/j.pdpdt.2019.04.033
Mao, W., Yin, H., Chen, W., Zhao, T., Wu, S., Jin, H., et al. (2020). Network Pharmacology and Experimental Evidence Reveal Dioscin Suppresses Proliferation, Invasion, and EMT via AKT/GSK3b/mTOR Signaling in Lung Adenocarcinoma. Drug Des. Devel Ther. 14, 2135–2147. doi:10.2147/DDDT.S249651
Markowska, A., Pawałowska, M., Lubin, J., and Markowska, J. (2014). Signalling Pathways in Endometrial Cancer. Contemp. Oncol. (Pozn) 18 (3), 143–148. doi:10.5114/wo.2014.43154
Marzocco, S., Singla, R. K., and Capasso, A. (2021). Multifaceted Effects of Lycopene: A Boulevard to the Multitarget-Based Treatment for Cancer. Molecules 26 (17). doi:10.3390/molecules26175333
Matias-Guiu, X., and Prat, J. (2013). Molecular Pathology of Endometrial Carcinoma. Histopathology 62 (1), 111–123. doi:10.1111/his.12053
McMeekin, D. S., Manikas, G., Crispens, M., Orza, A. M., Braly, P., Doering, D., et al. (2004). A Phase II Study of Trabectedin (ET-743) as a Second Line Therapy in Patients with Persistent or Recurrent Endometrial Carcinoma. Jco 22 (14_Suppl. l), 5086. doi:10.1200/jco.2004.22.90140.5086
Moghimi, S. M., Hunter, A. C., and Murray, J. C. (2001). Long-circulating and Target-specific Nanoparticles: Theory to Practice. Pharmacol. Rev. 53 (2), 283–318.
Moolgavkar, S. H., and Knudson, A. G. (1981). Mutation and Cancer: a Model for Human Carcinogenesis. J. Natl. Cancer Inst. 66 (6), 1037–1052. doi:10.1093/jnci/66.6.1037
Mortazavi, H., Nikfar, B., Esmaeili, S. A., Rafieenia, F., Saburi, E., Chaichian, S., et al. (2020). Potential Cytotoxic and Anti-metastatic Effects of Berberine on Gynaecological Cancers with Drug-Associated Resistance. Eur. J. Med. Chem. 187, 111951. doi:10.1016/j.ejmech.2019.111951
Muqbil, I., Masood, A., Sarkar, F. H., Mohammad, R. M., and Azmi, A. S. (2011). Progress in Nanotechnology Based Approaches to Enhance the Potential of Chemopreventive Agents. Cancers (Basel) 3 (1), 428–445. doi:10.3390/cancers3010428
Mutter, G. L., Lin, M. C., Fitzgerald, J. T., Kum, J. B., Baak, J. P., Lees, J. A., et al. (2000). Altered PTEN Expression as a Diagnostic Marker for the Earliest Endometrial Precancers. J. Natl. Cancer Inst. 92 (11), 924–930. doi:10.1093/jnci/92.11.924
Nageen, B., Sarfraz, I., Rasul, A., Hussain, G., Rukhsar, F., Irshad, S., et al. (2020). Eupatilin: a Natural Pharmacologically Active Flavone Compound with its Wide Range Applications. J. Asian Nat. Prod. Res. 22 (1), 1–16. doi:10.1080/10286020.2018.1492565
Oda, K., Stokoe, D., Taketani, Y., and McCormick, F. (2005). High Frequency of Coexistent Mutations of PIK3CA and PTEN Genes in Endometrial Carcinoma. Cancer Res. 65 (23), 10669–10673. doi:10.1158/0008-5472.Can-05-2620
Okuda, T., Sekizawa, A., Purwosunu, Y., Nagatsuka, M., Morioka, M., Hayashi, M., et al. (2010). Genetics of Endometrial Cancers. Obstet. Gynecol. Int. 2010, 984013–984018. doi:10.1155/2010/984013
Onstad, M. A., Schmandt, R. E., and Lu, K. H. (2016). Addressing the Role of Obesity in Endometrial Cancer Risk, Prevention, and Treatment. J. Clin. Oncol. 34 (35), 4225–4230. doi:10.1200/JCO.2016.69.4638
Peñalva, R., Morales, J., González-Navarro, C., Larrañeta, E., Quincoces, G., Peñuelas, I., et al. (2018). Increased Oral Bioavailability of Resveratrol by its Encapsulation in Casein Nanoparticles. Ijms 19 (9), 2816. doi:10.3390/ijms19092816
Pierpaoli, E., Damiani, E., Orlando, F., Lucarini, G., Bartozzi, B., Lombardi, P., et al. (2015). Antiangiogenic and Antitumor Activities of Berberine Derivative NAX014 Compound in a Transgenic Murine Model of HER2/neu-Positive Mammary Carcinoma. Carcinogenesis 36 (10), 1169–1179. doi:10.1093/carcin/bgv103
Pourhanifeh, M. H., Darvish, M., Tabatabaeian, J., Fard, M. R., Mottaghi, R., Azadchehr, M. J., et al. (2020). Therapeutic Role of Curcumin and its Novel Formulations in Gynecological Cancers. J. Ovarian Res. 13 (1), 130. doi:10.1186/s13048-020-00731-7
Puszkiel, A., Noé, G., Thomas-Schoemann, A., Bellesoeur, A., Boudou-Rouquette, P., Vidal, M., et al. (2017). Pharmacokinetics and Pharmacodynamics of Tyrosine Kinase Inhibitors in the Treatment of Metastatic Renal Cell Carcinoma. Int. J. Pharmacokinet. 2 (4), 257–283. doi:10.4155/ipk-2017-0008
Rahimi, H. R., Nedaeinia, R., Sepehri Shamloo, A., Nikdoust, S., and Kazemi Oskuee, R. (2016). Novel Delivery System for Natural Products: Nano-Curcumin Formulations. Avicenna J. Phytomed 6 (4), 383–398.
Rahman, M., Beg, S., Ahmed, A., and Swain, S. (2014). Emergence of Functionalized Nanomedicines in Cancer Chemotherapy: Recent Advancements, Current Challenges and Toxicity Considerations. Pnm 3 (2), 128–139. doi:10.2174/18779123113036660002
Richter, C., Mayhew, D., Rennhack, J. P., So, J., Stover, E. H., Hwang, J. H., et al. (2020). Genomic Amplification and Functional Dependency of the Gamma Actin Gene ACTG1 in Uterine Cancer. Int. J. Mol. Sci. 21 (22). doi:10.3390/ijms21228690
Rosenwald, I. B., Rhoads, D. B., Callanan, L. D., Isselbacher, K. J., and Schmidt, E. V. (1993). Increased Expression of Eukaryotic Translation Initiation Factors eIF-4E and eIF-2 Alpha in Response to Growth Induction by C-Myc. Proc. Natl. Acad. Sci. U S A. 90 (13), 6175–6178. doi:10.1073/pnas.90.13.6175
Sánchez-Martínez, C., Gelbert, L. M., Lallena, M. J., and de Dios, A. (2015). Cyclin Dependent Kinase (CDK) Inhibitors as Anticancer Drugs. Bioorg. Med. Chem. Lett. 25 (17), 3420–3435. doi:10.1016/j.bmcl.2015.05.100
Schwartz, P. A., and Murray, B. W. (2011). Protein Kinase Biochemistry and Drug Discovery. Bioorg. Chem. 39 (5-6), 192–210. doi:10.1016/j.bioorg.2011.07.004
Sexton, E., Van Themsche, C., Leblanc, K., Parent, S., Lemoine, P., and Asselin, E. (2006). Resveratrol Interferes with AKT Activity and Triggers Apoptosis in Human Uterine Cancer Cells. Mol. Cancer 5 (1), 45. doi:10.1186/1476-4598-5-45
Sherman, M. E., Sturgeon, S., Brinton, L., and Kurman, R. J. (1995). Endometrial Cancer Chemoprevention: Implications of Diverse Pathways of Carcinogenesis. J. Cell Biochem Suppl 23, 160–164. doi:10.1002/jcb.240590921
Singh, M., Darcy, K. M., Brady, W. E., Clubwala, R., Weber, Z., Rittenbach, J. V., et al. (2011). Cadherins, Catenins and Cell Cycle Regulators: Impact on Survival in a Gynecologic Oncology Group Phase II Endometrial Cancer Trial. Gynecol. Oncol. 123 (2), 320–328. doi:10.1016/j.ygyno.2011.07.005
Singla, R. K., Agarwal, T., He, X., and Shen, B. (2021a). Herbal Resources to Combat a Progressive & Degenerative Nervous System Disorder- Parkinson's Disease. Curr. Drug Targets 22 (6), 609–630. doi:10.2174/1389450121999201013155202
Singla, R. K., and Dubey, A. K. (2019). Molecules and Metabolites from Natural Products as Inhibitors of Biofilm in Candida Spp. Pathogens. Curr. Top. Med. Chem. 19 (28), 2567–2578. doi:10.2174/1568026619666191025154834
Singla, R. K., Guimarães, A. G., and Zengin, G. (2020a). Editorial: Application of Plant Secondary Metabolites to Pain Neuromodulation. Front. Pharmacol. 11, 623399. doi:10.3389/fphar.2020.623399
Singla, R. K., Kumar, R., Khan, S., MohitKumari, K., Kumari, K., and Garg, A. (2019). Natural Products: Potential Source of DPP-IV Inhibitors. Curr. Protein Pept. Sci. 20 (12), 1218–1225. doi:10.2174/1389203720666190502154129
Singla, R. K. (2021). Secondary Metabolites as Treatment of Choice for Metabolic Disorders and Infectious Diseases and Their Metabolic Profiling - Part 3. Curr. Drug Metab. 22 (6), 412–414. doi:10.2174/138920022206210708103019
Singla, R. K., and Singla, R. K. (2020). Secondary Metabolites as Treatment of Choice for Metabolic Disorders and Infectious Diseases and Their Metabolic Profiling: Part 1. Curr. Drug Metab. 21 (14), 480–481. doi:10.2174/138920022107200925101631
Singla, R. K., Sultana, A., Alam, M. S., and Shen, B. (2020b). Regulation of Pain Genes-Capsaicin vs Resiniferatoxin: Reassessment of Transcriptomic Data. Front. Pharmacol. 11, 551786. doi:10.3389/fphar.2020.551786
Singla, R. K., De, B., and Nagal, A. (2018). “Multimodal Perspectives of Nanotechnology and Nanoparticles in Drug Delivery” in Proceedings of MOL2NET 2018, International Conference on Multidisciplinary Sciences, 4th edition, Basel, June 4, 2018 (Basel, Switzerland: MDPI Sciforum).
Singla, R. K., Gupta, R., Joon, S., Gupta, A. K., and Shen, B. (2021b). Isolation, Docking and In Silico ADME-T Studies of Acacianol: Novel Antibacterial Isoflavone Analogue Isolated from Acacia Leucophloea Bark. Cdm 22, 893–904. doi:10.2174/1389200222666211005091417
Singla, R. K., Sai, C. S., Chopra, H., Behzad, S., Bansal, H., Goyal, R., et al. (2021c). Natural Products for the Management of Castration-Resistant Prostate Cancer: Special Focus on Nanoparticles Based Studies. Front. Cell Dev. Biol. 9. doi:10.3389/fcell.2021.745177
Singla, R. K., Sharma, P., Dubey, A. K., Gundamaraju, R., Kumar, D., Kumar, S., et al. (2021d). Natural Product-Based Studies for the Management of Castration-Resistant Prostate Cancer: Computational to Clinical Studies. Front. Pharmacol. 12. doi:10.3389/fphar.2021.732266
Sirotkin, A. V. (2021). Effects of Resveratrol on Female Reproduction: A ReviewPhytother Res. 35 (10), 5502–5513. doi:10.1002/ptr.7185
Slater, S. J., Seiz, J. L., Cook, A. C., Stagliano, B. A., and Buzas, C. J. (2003). Inhibition of Protein Kinase C by Resveratrol. Biochim. Biophys. Acta 1637 (1), 59–69. doi:10.1016/S0925-4439(02)00214-4
Soslow, R. A., Tornos, C., Park, K. J., Malpica, A., Matias-Guiu, X., Oliva, E., et al. (2019). Endometrial Carcinoma Diagnosis: Use of FIGO Grading and Genomic Subcategories in Clinical Practice: Recommendations of the International Society of Gynecological Pathologists. Int. J. Gynecol. Pathol. 38 (Suppl. 1), S64–S74. doi:10.1097/PGP.0000000000000518
Sultana, A., Singla, R. K., He, X., Sun, Y., Alam, M. S., and Shen, B. (2021). Topical Capsaicin for the Treatment of Neuropathic Pain. Curr. Drug Metab. 22 (3), 198–207. doi:10.2174/1389200221999201116143701
Sun, Y.-w., Liang, H., Zong, K.-q., Che, X., and Meng, D.-l. (2021). Green and Facile Preparation and Dual-Enhancement Cytotoxicity of Eupatilin Loaded on Hollow Gold Nanoparticles under Near-Infrared Light. New J. Chem. 45 (34), 15676–15681. doi:10.1039/d1nj02276h
Tan, Z. W., Xie, S., Hu, S. Y., Liao, T., Liu, P., Peng, K. H., et al. (2016). Caudatin Targets TNFAIP1/NF-κB and Cytochrome C/caspase Signaling to Suppress Tumor Progression in Human Uterine Cancer. Int. J. Oncol. 49 (4), 1638–1650. doi:10.3892/ijo.2016.3662
Tang, S. M., Deng, X. T., Zhou, J., Li, Q. P., Ge, X. X., and Miao, L. (2020). Pharmacological Basis and New Insights of Quercetin Action in Respect to its Anti-cancer Effects. Biomed. Pharmacother. 121, 109604. doi:10.1016/j.biopha.2019.109604
Tashiro, H., Blazes, M. S., Wu, R., Cho, K. R., Bose, S., Wang, S. I., et al. (1997). Mutations in PTEN Are Frequent in Endometrial Carcinoma but Rare in Other Common Gynecological Malignancies. Cancer Res. 57 (18), 3935–3940.
Tewari, D., Patni, P., Bishayee, A., Sah, A. N., and Bishayee, A. (2019). Natural Products Targeting the PI3K-Akt-mTOR Signaling Pathway in Cancer: A Novel Therapeutic Strategy. Semin. Cancer Biol. doi:10.1016/j.semcancer.2019.12.008
Tran, K. Q., Tin, A. S., and Firestone, G. L. (2014). Artemisinin Triggers a G1 Cell Cycle Arrest of Human Ishikawa Endometrial Cancer Cells and Inhibits Cyclin-dependent Kinase-4 Promoter Activity and Expression by Disrupting Nuclear Factor-κB Transcriptional Signaling. Anticancer Drugs 25 (3), 270–281. doi:10.1097/CAD.0000000000000054
Trynda-Lemiesz, L. (2004). Paclitaxel-HSA Interaction. Binding Sites on HSA Molecule. Bioorg. Med. Chem. 12 (12), 3269–3275. doi:10.1016/j.bmc.2004.03.073
van Leeuwen, R. W., van Gelder, T., Mathijssen, R. H., and Jansman, F. G. (2014). Drug-drug Interactions with Tyrosine-Kinase Inhibitors: a Clinical Perspective. Lancet Oncol. 15 (8), e315–26. doi:10.1016/s1470-2045(13)70579-5
Whitney, C. W., Brunetto, V. L., Zaino, R. J., Lentz, S. S., Sorosky, J., Armstrong, D. K., et al. (2004). Phase II Study of Medroxyprogesterone Acetate Plus Tamoxifen in Advanced Endometrial Carcinoma: a Gynecologic Oncology Group Study. Gynecol. Oncol. 92 (1), 4–9. doi:10.1016/j.ygyno.2003.09.018
Wong, Y. K., Xu, C., Kalesh, K. A., He, Y., Lin, Q., Wong, W. S. F., et al. (2017). Artemisinin as an Anticancer Drug: Recent Advances in Target Profiling and Mechanisms of Action. Med. Res. Rev. 37 (6), 1492–1517. doi:10.1002/med.21446
Wu, W., Wang, L., Wang, L., Zu, Y., Wang, S., Liu, P., et al. (2018). Preparation of Honokiol Nanoparticles by Liquid Antisolvent Precipitation Technique, Characterization, Pharmacokinetics, and Evaluation of Inhibitory Effect on HepG2 Cells. Int. J. Nanomedicine 13, 5469–5483. doi:10.2147/ijn.S178416
Yamada, K., Uchiyama, A., Uehara, A., Perera, B., Ogino, S., Yokoyama, Y., et al. (2016). MFG-E8 Drives Melanoma Growth by Stimulating Mesenchymal Stromal Cell-Induced Angiogenesis and M2 Polarization of Tumor-Associated Macrophages. Cancer Res. 76 (14), 4283–4292. doi:10.1158/0008-5472.CAN-15-2812
Yang, L., Ren, S., Xu, F., Ma, Z., Liu, X., and Wang, L. (2019). Recent Advances in the Pharmacological Activities of Dioscin. Biomed. Res. Int. 2019, 5763602. doi:10.1155/2019/5763602
Yeramian, A., Moreno-Bueno, G., Dolcet, X., Catasus, L., Abal, M., Colas, E., et al. (2013). Endometrial Carcinoma: Molecular Alterations Involved in Tumor Development and Progression. Oncogene 32 (4), 403–413. doi:10.1038/onc.2012.76
Zagouri, F., Bozas, G., Kafantari, E., Tsiatas, M., Nikitas, N., Dimopoulos, M. A., et al. (2010). Endometrial Cancer: What Is New in Adjuvant and Molecularly Targeted Therapy? Obstet. Gynecol. Int. 2010, 749579–749611. doi:10.1155/2010/749579
Zhang, F., Zhang, Y. Y., Sun, Y. S., Ma, R. H., Thakur, K., Zhang, J. G., et al. (2019). Asparanin A from Asparagus Officinalis L. Induces G0/G1 Cell Cycle Arrest and Apoptosis in Human Endometrial Carcinoma Ishikawa Cells via Mitochondrial and PI3K/AKT Signaling Pathways. J. Agric. Food Chem. 68 (1), 213–224. doi:10.1021/acs.jafc.9b07103
Zhang, F., Zhang, Y.-Y., Ma, R.-H., Thakur, K., Han, J., Hu, F., et al. (2021). Multi-omics Reveals the Anticancer Mechanism of asparagus Saponin-Asparanin A on Endometrial Cancer Ishikawa Cells. Food Funct. 12 (2), 614–632. doi:10.1039/d0fo02265a
Zhang, M., Lu, Q., Hou, H., Sun, D., Chen, M., Ning, F., et al. (2021). Garcinol Inhibits the Proliferation of Endometrial Cancer Cells by Inducing Cell Cycle Arrest. Oncol. Rep. 45 (2), 630–640. doi:10.3892/or.2020.7900
Zhang X., X., Kan, H., Liu, Y., and Ding, W. (2021). Plumbagin Induces Ishikawa Cell Cycle Arrest, Autophagy, and Apoptosis via the PI3K/Akt Signaling Pathway in Endometrial Cancer. Food Chem. Toxicol. 148, 111957. doi:10.1016/j.fct.2020.111957
Zhang, Z. R., Leung, W. N., Cheung, H. Y., and Chan, C. W. (2015). Osthole: A Review on its Bioactivities, Pharmacological Properties, and Potential as Alternative Medicine. Evid. Based Complement. Alternat Med. 2015, 919616. doi:10.1155/2015/919616
Keywords: hormone-sensitive cancer, metastasis, natural products, medicinal plants, endometrial cancer
Citation: Singla RK, Behzad S, Khan J, Tsagkaris C, Gautam RK, Goyal R, Chopra H and Shen B (2022) Natural Kinase Inhibitors for the Treatment and Management of Endometrial/Uterine Cancer: Preclinical to Clinical Studies. Front. Pharmacol. 13:801733. doi: 10.3389/fphar.2022.801733
Received: 25 October 2021; Accepted: 17 January 2022;
Published: 21 February 2022.
Edited by:
Balakumar Chandrasekaran, ITM University, IndiaReviewed by:
Biswa Mohan Sahoo, Roland Institute of Pharmaceutical Sciences, IndiaCopyright © 2022 Singla, Behzad, Khan, Tsagkaris, Gautam, Goyal, Chopra and Shen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bairong Shen, YmFpcm9uZy5zaGVuQHNjdS5lZHUuY24=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.