- 1State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, National Clinical Research Center for Infectious Diseases, Collaborative Innovation Center for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang, China
- 2Department of Hepatology, Qilu Hospital, Cheeloo College of Medicine, Shandong University, Jinan, Shandong, China
On 5 April 2022, the World Health Organization was notified of 10 cases of severe acute hepatitis of unknown etiology in children under 10 years of age in the United Kingdom. Although the exact cause of a proportion of pediatric acute hepatitis and acute liver failure cases was unclear, the above event has caused widespread concern worldwide. As of 14 September 2022, approximately 1,296 probable cases of acute hepatitis of unknown etiology have been reported from 37 countries/regions, of which approximately 55 required or received liver transplantation and 29 died. Although the etiology of acute hepatitis of unknown origin in children remains unclear, many hypotheses have been proposed about the disease. Instead of individual factors such as “adenovirus infection,” “SARS-CoV-2 related,” and “Adeno-associated virus 2 with helper virus coinfection,” it is more likely due to a combination of factors. Accordingly, there is an urgent need for more data and research to clarify the disease etiology. This review aims to provide a historical perspective of acute hepatitis of unknown etiology in children in the past decades and summarize the current hypothesis and evidence on this emerging disease.
1 Introduction
The purpose of this article is to give a framework and reference for understanding the etiology of acute hepatitis of unknown origin in children, taking into consideration newly published relevant literature and information data.
On 31 March 2022, severe acute hepatitis of unknown origin was reported for the first time among children in Scotland for causes other than common hepatitis A-E virus infection (Marsh et al., 2022). Since then, similar cases have been reported in many countries or regions worldwide, and the number of cases has rapidly increased, causing widespread concern. Acute hepatitis is caused by various pathogenic factors invading the liver, damaging hepatocytes and liver function, and resulting in severe clinical symptoms. At first, patients experience fatigue, fever and loss of appetite, and in more severe cases, nausea, vomiting or tea-colored urine occur, followed by jaundice. The disease course does not exceed half a year. These common pathogenic factors are viruses, bacteria, parasites, chemical poisons, drugs and poisons, alcohol, etc.
This article aims to provide a framework and reference for understanding the etiology of acute hepatitis of unknown origin in children, taking into consideration newly published relevant literature and information data.
2 Methods
We sought to conduct a review of the literature to better understand acute hepatitis of unknown origin in children. PubMed database was retrieved using the search terms “pediatrics” or “children” and “hepatitis” or “liver failure” to identify relevant studies published from inception until 14 September 2022. The full text of relevant titles and abstracts was then reviewed for inclusion. In addition, citations and references of included studies were searched to identify any additional studies. English-language peer-reviewed and non-peer-reviewed studies describing acute hepatitis in children, pediatric acute liver failure (PALF), adenovirus, SARS-CoV-2, adeno-associated virus 2, and autoimmune hepatitis were included.
3 Disease background
3.1 Determinate and indeterminate pediatric acute hepatitis and liver failure
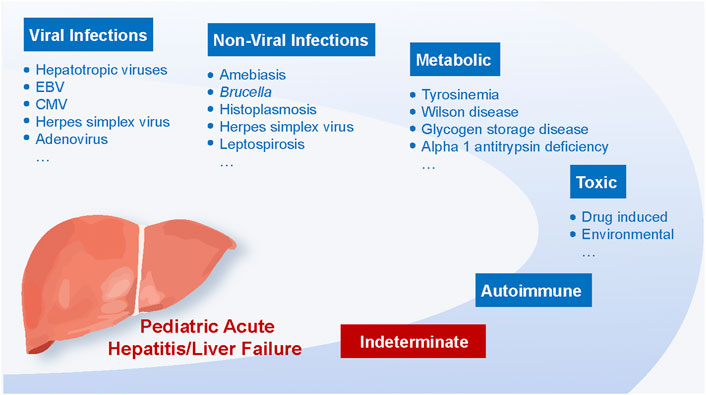
Over the past few decades, acute hepatitis and liver failure in children have become major public health issues worldwide (Daniels et al., 2009). Infectious, toxic, autoimmune, and metabolic mechanisms are the most common causes (Figure 1) (Devictor et al., 2011).
Most cases can be attributed to viral infections, with hepatotropic viruses being the primary culprit (Daniels et al., 2009; Zeng et al., 2021). Globally, acute hepatitis A virus (HAV) infection has long been considered the most prevalent cause of acute hepatitis in children (Rahman et al., 2022). However, its prevalence varies by geographical area and vaccination rates (Daniels et al., 2009; Zeng et al., 2021). Studies in the United Kingdom and India indicated that HAV infection accounts for 39% and 48.6% of acute hepatitis in hospitalized children, respectively (Braccio et al., 2017; Sood et al., 2019). Since the beginning of the century, routine childhood immunization has been promoted continuously in numerous nations, including the United States and Europe. In this regard, from 1995 to 2007, the incidence of acute hepatitis A in the United States population decreased by 92%, especially for children under 15 years old (Daniels et al., 2009). In addition to HAV, sporadic and epidemic infections of the Hepatitis E virus (HEV) represent a leading cause of acute viral hepatitis in children, primarily in developing nations (Verghese and Robinson, 2014; Fischler et al., 2016). HEV seroprevalence in children increases with age (Hodges et al., 1998) and is related to residence and socioeconomic status (Arankalle et al., 2001). Two early studies with small sample sizes from Egypt showed that acute sporadic hepatitis E accounted for 22.2% (under 11 years old) (el-Zimaity et al., 1993) and 12% (under 13 years old) (Hyams et al., 1992) of children admitted to hospital for acute hepatitis.
Besides hepatotropic pathogens, other viruses such as Epstein-Barr virus (EBV), cytomegalovirus (CMV), herpes simplex virus, adenovirus, and coxsackievirus can also cause acute liver failure in children, especially in those with immunodeficiency disorders (Gallegos-Orozco and Rakela-Brödner, 2010; Tsunoda et al., 2017). Real-time PCR and serological testing can assist in the detection of non-hepatitis virus-related acute hepatitis in children. A recent study investigated the incidence of CMV and EBV in children hospitalized with acute liver failure. Of the children excluded from hepatitis virus A, B, and C infection, 18% and 9% were positive for both DNA and IgM assay of CMV and EBV, respectively (Tsunoda et al., 2017). Another prospective study proposed that human herpesvirus-6 could cause acute liver failure in children (Chevret et al., 2008).
Other non-viral infections can potentially cause acute hepatitis in children, including amebiasis (Gupta et al., 2022), Brucella (Hassouneh et al., 2019), histoplasmosis, leptospirosis (Aygün et al., 2016), etc. A new study suggested that hepatitis was the most common laboratory abnormality in children with brucellosis (nearly three-quarters) (Hassouneh et al., 2019). In recent years, pediatric autoimmune hepatitis (AIH) has been increasingly reported (Mack et al., 2020). In the United States, the prevalence of AIH is 3 per 100,000 children, and the estimated annual incidence per 100,000 children is 0.4, comparable to Canada (0.23) (Deneau et al., 2013; Jiménez-Rivera et al., 2015).
In addition, drugs represent another potential cause of acute hepatitis. In developed countries, acetaminophen is the leading cause of drug-induced liver injury in children, followed by medications such as anti-tuberculosis and anti-epileptic agents (Squires et al., 2006). As for newborns and infants, acute liver failure is most commonly caused by inherited metabolic diseases (Durand et al., 2001), including Wilson disease, alpha-1 antitrypsin deficiency, tyrosinemia, glycogen storage disease, etc.
Although in-depth and comprehensive research has been conducted on the etiology of pediatric acute hepatitis and acute liver failure along with the continuous promotion of unprecedented metabolic and viral screening, a definite cause cannot be found in a substantial portion of children (Aydoğdu et al., 2003; Squires et al., 2006; Black, 2009; Narkewicz et al., 2009). When a specific diagnosis is not established in PALF, these children are categorized as indeterminate (iPALF) (Alonso et al., 2017). The Pediatric Acute Liver Failure study group, composed of centers from the United States, Canada, and the United Kingdom, released the initial data of 348 cases in 2006 (Squires et al., 2006). The outcomes of this prospective multicenter research revealed that for nearly half of children with pediatric acute liver failure, a definitive diagnosis could not be determined, consistent with the subsequent results published by this study group in 2008 and the conclusions of studies from Turkey and Germany (Aydoğdu et al., 2003; Squires et al., 2006; Narkewicz et al., 2009; Kathemann et al., 2015). Besides, children with ambiguous diagnoses were younger and more likely to receive liver transplantation after admission than children with a definitive diagnosis (Liu et al., 2006; Narkewicz et al., 2009). In subsequent studies, Narkewicz et al. evaluated the diagnostic mode of 329 children who were eventually diagnosed with indeterminate acute liver failure between 1999 and 2008. The results showed inadequate screening for metabolic and autoimmune acute liver failure causes. Furthermore, an efficient diagnostic evaluation pattern for pediatric acute liver failure based on age and clinical manifestations should be established to help pediatricians formulate timely and effective treatment strategies (Narkewicz et al., 2009). Mechanisms driving injury of iPALF have been postulated to be secondary to overwhelming inflammatory responses and immune dysregulation (Alonso et al., 2017). Recent efforts to further characterize iPALF have identified a subset of patients with unusual liver histology, including a dense CD103+CD8+ T-cell infiltrate, further suggesting liver injury is immune-mediated (Squires et al., 2022).
4 Epidemiology
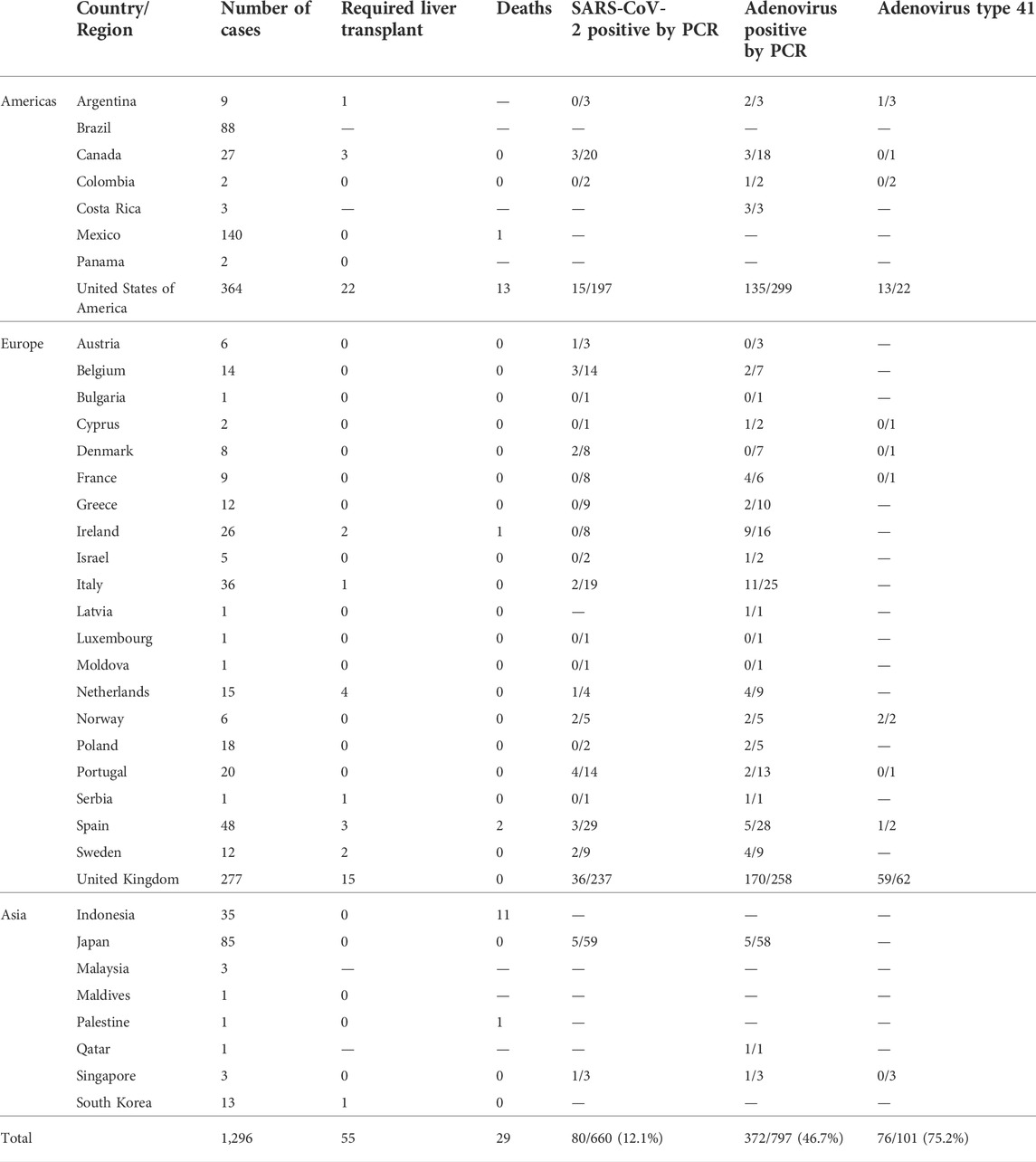
As of 14 September 2022, approximately 1,296 probable cases of acute hepatitis of unknown etiology have been reported from 37 countries/regions, of which approximately 55 required or received liver transplantation and 29 died. The highest incidence was in the United States (n = 364), followed by the United Kingdom (n = 277), Mexico (n = 140), Brazil (n = 88), and Japan (n = 85). Based on the available data, 12% (80/660) of the tested cases were positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) polymerase chain reaction (PCR), 46.7% (372/797) were positive for adenovirus PCR, and 75.2% (76/101) of the detected adenovirus cases were type 41 (World Health Organization, 2022b; European Centre for Disease Prevention and Control, 2022; Government of Canada, 2022). More details are shown in Table 1 and Figure 2.
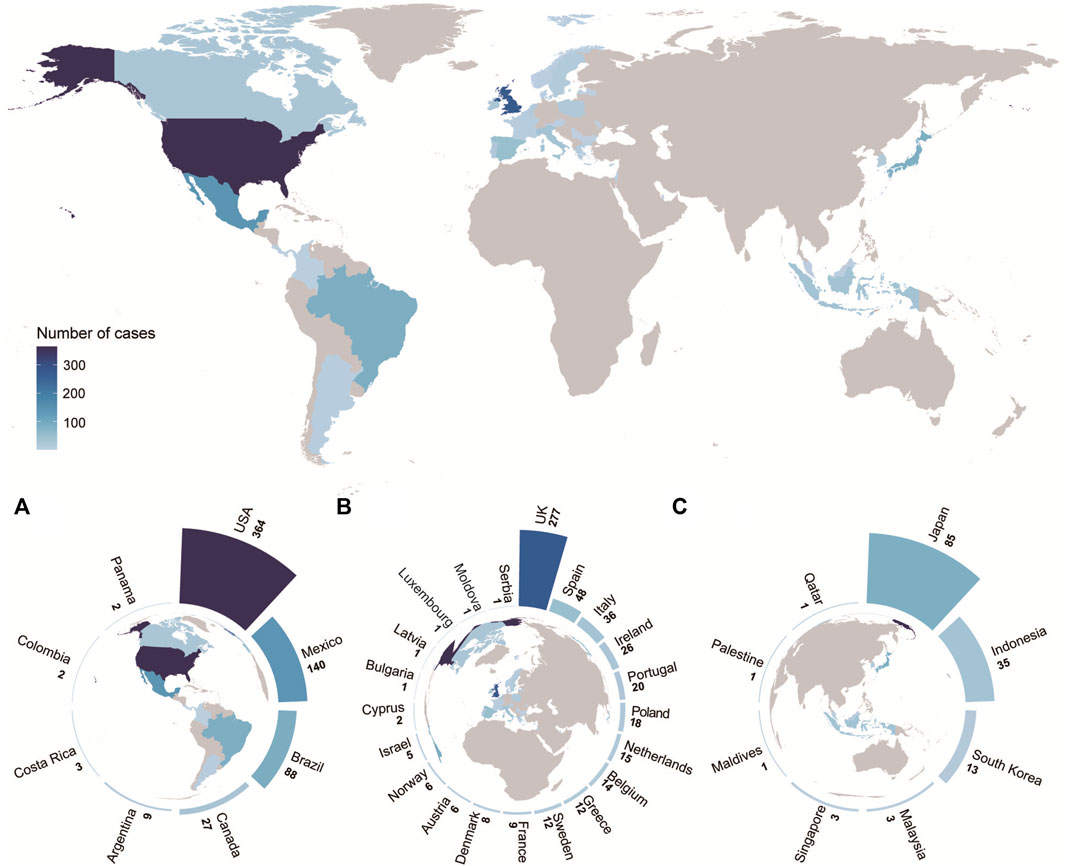
FIGURE 2. World distribution of possible cases, as of 14 September 2022 (n = 1,296) (A) Distribution of possible cases in the Americas. (B) Distribution of possible cases in Europe. (C) Distribution of possible cases in Asia. The above four figures share the same legend. Further details are provided in Table 1.
According to ECDC and The European Surveillance System (TESSy), the majority (76.2%) of cases were 5 years old or younger, with no significant difference in gender (European Centre for Disease Prevention and Control, 2022).
5 Clinical features
The clinical manifestations are characterized by an acute onset of clinical symptoms, including weakness and poor appetite, nausea, vomiting, diarrhea, abdominal pain, and other gastrointestinal symptoms, followed by jaundice. Moreover, children may present with pale stools, liver enlargement, fever, respiratory symptoms, or splenomegaly. In rare cases, the disease may progress to acute liver failure within a short period, with progressive worsening of jaundice and hepatic encephalopathy. Rapid disease progression may require liver transplantation and even lead to death in severe cases.
According to the description in Technical Brief 4 published by The UK Health Security Agency (UKHSA), most patients present with jaundice (69.2%), followed by vomiting (57.9%), and pale stools (40.5%). Patients may also present with gastrointestinal symptoms, including diarrhea (41.5%), abdominal pain (38.5%), and nausea (26.2%). In addition, lethargy (47.7%), fever (23.1%) and some rare respiratory symptoms (17.9%) have been reported (UK Health Security Agency, 2022c).
6 Case definitions
Currently, different organizations/countries use different case definitions of confirmed, probable, and epi-linked. The WHO, United Kingdom, Scotland, the European Union, the United States, and Canada categorize cases as confirmed, probable, and epi-linked, using different case definitions based on case age, reporting window, and other etiologic specifications and exclusions. Table 2 summarizes different case definitions in different organizations (World Health Organization, 2022a; UK Health Security Agency, 2022b; Centre for Disease Prevention and Control, 2022; European Centre for Disease Prevention and Control, 2022; Government of Manitoba, 2022).
7 Diagnosis
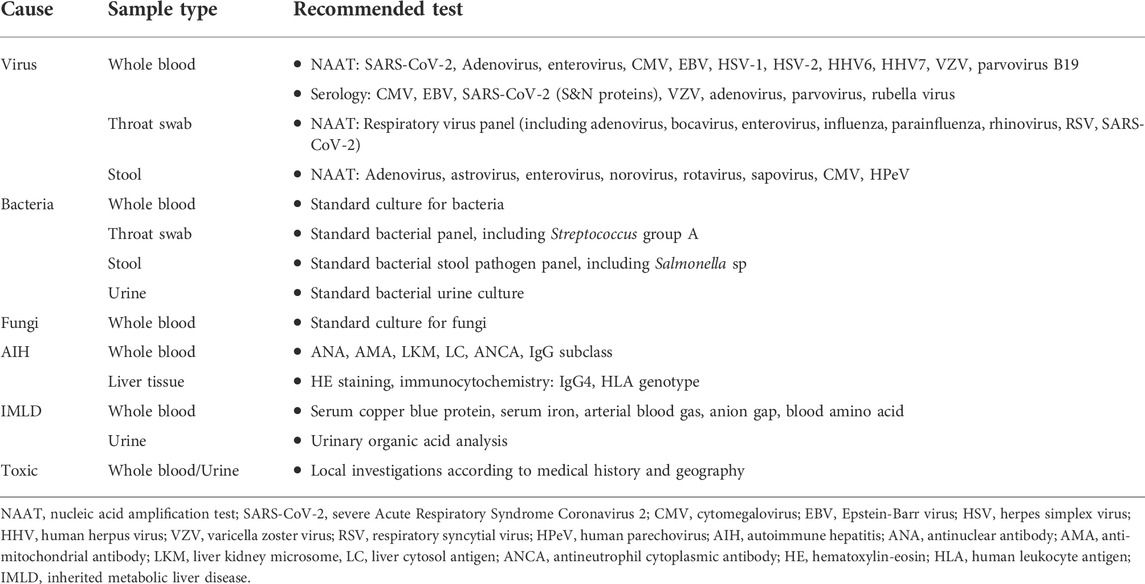
Patients who meet one of the definitions of probable cases and epi-linked cases should be comprehensively investigated, including the collection of epidemiological and family history. Routine laboratory tests should be performed, including blood cell analysis, biochemical tests, coagulation tests, plasma ammonia, inflammatory markers, and abdominal imaging. All instances that meet the case criteria should undergo testing of whole blood, serum, urine, feces, respiratory samples, and liver biopsy (if available). Laboratory tests for infectious and non-infectious causes are shown in Table 3.
8 Working hypotheses on possible etiology
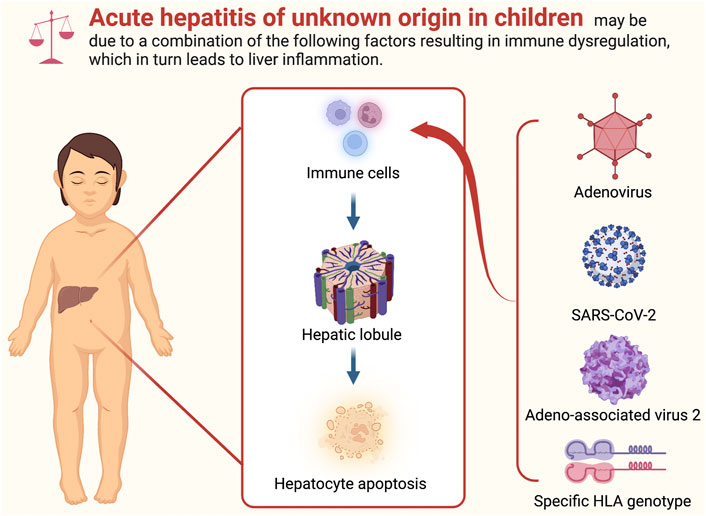
The primary hypotheses about the cause of acute hepatitis in children are as follows:
(1) Adenovirus infection (e.g., abnormal susceptibility or host response due to lack of exposure during the COVID-19 pandemic.; a large-scale outbreak of normal adenovirus infection; coinfection with or priming by another pathogen)
(2) SARS-CoV-2 related (e.g., sequelae from SARS-CoV-2 infection; a postinfectious SARS-CoV-2 syndrome (similar to the multisystem inflammatory syndrome in children); as superantigens; undescribed sign or symptom of a recently emerged variants of concern (VOC) (i.e., Omicron))
(3) Adeno-associated virus 2 with helper virus coinfection (e.g., adenovirus; human herpesvirus 6B)
(4) Immunodeficiency (e.g., a specific HLA genotype: DRB1*04:01)
(5) A novel pathogen not detectable with current diagnostics either acting alone or as a coinfection
(6) A non-infectious cause (e.g., toxin, drug, or environmental exposure)
8.1 Adenovirus infection
By far, adenovirus is most frequently identified in test samples. As of 28 July 2022, adenovirus has been found in 65.9% (170/258) of cases investigated in the United Kingdom (UK Health Security Agency, 2022c). Type 41 represented 95.2% (59/62) of these adenovirus-positive cases and was found in several cases reported in the United States (Baker et al., 2022; UK Health Security Agency, 2022c).
Recently, the UK Health Security Agency has documented an increase in adenovirus-positive stool samples from children aged 1–4 years compared to pre-pandemic levels, as well as a rise in the occurrence of hepatitis among children aged 1 to 4 (UK Health Security Agency, 2022b). However, on June 14, the CDC released a preliminary survey highlighting that current data do not indicate that pediatric hepatitis or adenovirus types 40/41 are above pre-pandemic baseline levels of COVID-19 (Kambhampati et al., 2022).
Although adenovirus infection is currently the most widely accepted hypothesis, it cannot fully explain the severity of clinical symptoms as the underlying cause. Biopsies of liver specimens from the United Kingdom, Israel, and Alabama, United States, did not reveal adenovirus inclusions, and the adenovirus load of affected children was generally low (Baker et al., 2022; UK Health Security Agency, 2022b; Marsh et al., 2022). Much controversy surrounds the ability of the “adenovirus hypothesis” to explain the pathogenesis of acute hepatitis of unknown origin in children.
It is well-established that adenovirus infections are prevalent in children. Indeed, children’s tonsils and adenoids are common host organs for adenoviruses, with a reported positivity rate of 76% for adenovirus PCR (Jaggi et al., 2013). About 30% of immunocompetent children develop persistent or resident infections after the acute phase of adenovirus infection. Such persistent infections are common in mucosal lymphocytes and are more common in young children, fading away with age (Garnett et al., 2002; Kosulin, 2019).
Then, during systemic immunocompromise, mucosal-site adenoviruses (as well as various other viruses) have the potential to enter the bloodstream and reactivate, eventually presenting as disseminated infection. Current evidence suggests that acute liver injury and liver failure, regardless of the cause, represent a state of immunodeficiency. However, whether liver damage or adenovirus viremia is the cause remains unclear.
Moreover, almost all previously reported cases of adenoviral hepatitis involved immunodeficiency. In this respect, adenoviral inclusion bodies were detected in all liver biopsies of adenoviral hepatitis. Few cases of acute severe hepatitis or liver failure due to adenovirus infection have been reported in immunocompetent children. However, the serotype human adenovirus (HAdV) 41 has not been linked to hepatitis or liver failure (Rocholl et al., 2004; Ozbay Hoşnut et al., 2008; Lynch and Kajon, 2016; Kang, 2017).
Although adenovirus is infrequently related to fulminant liver failure in immunocompetent children, other factors may enhance susceptibility; hence, adenovirus remains a potential contributing factor in addition to other factors such as abnormal susceptibility or host response.
Interestingly, it has been reported that the outbreak control during the 2019 coronavirus epidemic may have reduced young children’s exposure to common pathogens, leading to the development of immune systems different from their peers before the 2019 coronavirus epidemic. When young children are subsequently infected with common pathogens such as adenovirus, the immune response or disease manifestations differ from their peers. According to the UKHSA, adenovirus, enterovirus, human metapneumovirus, rhinovirus, and norovirus have been significantly overrepresented among children under 10 years old since the end of 2021. Respiratory syncytial viruses have been reported since late summer 2021, possibly due to behavioral alterations and population vulnerability following the pandemic’s low incidence (UK Health Security Agency, 2022b).
8.2 SARS-CoV-2 related
Up to now, more than 10% of positive SARS-CoV-2 cases have been detected by PCR, while the positive rate of SARS-CoV-2 serological tests is about 70%. Serological tests are underway to explore previous infections further. (World Health Organization, 2022a; European Centre for Disease Prevention and Control, 2022). Most patients were only tested for SARS-CoV-2 PCR but not for SARS-CoV-2 serology. Notably, SARS-CoV-2 PCR can only determine if the child was infected with SARS-CoV-2 then, but serological testing is indicated to assess whether the child has been infected with SARS-CoV-2 in the past.
SARS-CoV-2 can result in liver function impairment in COVID-19 patients. Recent research by Ellen K. Kendall et al. revealed that children infected with COVID-19 had a two-fold higher risk of liver injury than children infected with other respiratory infections. This finding implies that COVID-19 causes acute and chronic hepatic complications in pediatric patients (Kendall et al., 2022).
Recently, Shiri Cooper et al. analyzed the clinical presentation and laboratory test results of five hospitalized children who recovered from COVID-19 and subsequently exhibited liver injury. It has been suggested that SARS-CoV-2 infection is more likely to cause acute hepatitis of unknown etiology in children. The two patterns of long-term hepatic manifestations of COVID-19 in children include acute liver failure and acute hepatitis with cholestasis (Cooper et al., 2022).
In 2020, Yijin Wang et al. studied 156 COVID-19 patients from two designated centers in China and compared the clinical characteristics of patients with and without increased aminotransferases. The findings indicate that SARS-CoV-2 infection of the liver is a major contributor to the impairment of liver function in COVID-19 patients. However, the median levels of ALT were 50 U/L and 19 U/L, and AST levels were 45.5 U/L and 19 U/L in the abnormal and normal transaminase groups, respectively (Wang et al., 2020). Even the transaminase values in the abnormal transaminase group were much lower than those in the case definition.
However, two independent research groups in Glasgow and London, UK, have recently found no direct association between acute hepatitis of unknown origin in children and SARS-CoV-2 infection. The prevalence of SARS-CoV-2 antibodies did not differ between cases and age-matched controls. Nevertheless, they could not completely rule out an immune-mediated post-COVID-19 phenomenon in susceptible children (Ho et al., 2022; Morfopoulou et al., 2022).
8.2.1 Vaccine
It has been suggested that vaccination with the SARS-CoV-2 mRNA vaccine can trigger T-cell-dominant autoimmune hepatitis mediated by vaccination-induced antigen-specific tissue-resident immunity necessitating systemic immunosuppression (Boettler et al., 2022). Cases of SARS-CoV-2 vaccine-induced autoimmune hepatitis have also been reported prior to October 2021 (Bril et al., 2021).
However, there is no proof to substantiate that acute hepatitis of unknown origin in children is associated with the side effects of SARS-CoV-2 vaccines. According to the WHO, the hypothesis that these cases result from the side effects of SARS-CoV-2 vaccines is not supported, given that most afflicted children were not vaccinated (World Health Organization, 2022a).
8.2.2 MIS-C and SARS-CoV-2 as superantigens
Multisystem inflammatory syndrome in children (MIS-C) often occurs 6–8 weeks after SARS-CoV-2 infection. MIS-C may manifest clinically as persistent fever, gastrointestinal symptoms (abdominal discomfort, vomiting, diarrhea), rash, and conjunctivitis. Patients often present with three to 5 days of fever, followed by the onset of shock and/or multisystem involvement.
Acute hepatitis has been associated with multisystem inflammatory syndrome in children. Amanda Cantor et al. concluded from a small retrospective cohort study that acute hepatitis is an outstanding manifestation of MIS-C (Cantor et al., 2020).
Recently, Petter Brodin and Moshe Arditi have proposed a novel potential mechanism for acute hepatitis of unknown origin in children - superantigens-mediated, adenovirus-induced immune activation by SARS-CoV-2 infection, representing an alternative manifestation of MIS-C (Brodin and Arditi, 2022).
SARS-CoV-2 infection in children represents a viral reservoir in the body (Brodin, 2022). Specifically, the persistence of SARS-CoV-2 in the gastrointestinal tract of children can result in the sustained release of viral proteins from intestinal epithelial cells, thus causing immune activation. The superantigen motif may mediate this recurrent immune activation in the spike protein of SARS-CoV-2, which, similar to staphylococcal enterotoxin B, can trigger widespread and nonspecific t-cell activation (Cheng et al., 2020). This superantigen-mediated activation of immune cells has been recognized as a mechanism of MIS-C.
Accordingly, Petter Brodin and Moshe Arditi speculated that recent cases of acute hepatitis in children may have resulted from the emergence of a viral reservoir in the gut following infection with SARS-CoV-2, followed by adenovirus infection and the dysregulation of the body’s immune response in response to a combination of triggers from the adenovirus and SARS-CoV-2 S protein, leading to the development of acute hepatitis. Importantly, immunomodulatory therapy should be considered for acute hepatitis of unknown origin in children if evidence of superantigen-mediated immune activation is available (Brodin and Arditi, 2022).
A cohort study by Rita Robin involving 113 persons found that a small proportion of COVID-19 patients still had viral RNA in their feces after testing negative for respiratory samples. Interestingly, the condition reportedly persists for about 4% of patients for 7 months or longer, and these patients are more likely to experience persistent gastrointestinal symptoms (Rubin, 2022). These findings add to the growing body of evidence that SARS-CoV-2 invades the gut and provide circumstantial evidence for the speculations of Petter Brodin and Moshe Ardit.
Nonetheless, this theory does not account for the immunopathological liver damage in acute hepatitis of unknown origin in children, whereas MIS-C leads to inflammatory damage in multiple organ systems. Moreover, only a tiny proportion of children with unexplained hepatitis in children present with fever, whereas all MIS-C have fever >3 days.
During the epidemic of the delta variant in India (April 2021 to July 2021), Covid-19-associated hepatitis was reported in 37 children 2–6 weeks after infection with SARS-CoV-2, which was named COVID-19 Associated Hepatitis in Children (CAH-C), and these cases were similar to recent cases of acute hepatitis of unknown origin in children, with a median AST 942.45 (301.67, 2002.05) U/L and a median ALT of 1,326.25 (492.12, 2124.92) U/L, and no jaundice. Radha Kanta Ratho et al. found that CAH-C cases differed from MIS-C, which lacked the significantly elevated inflammatory markers or systemic disturbances seen with MIS-C during all testing periods. The more severe form of MIS-C is characterized by polyclonal T-cell activation, whereas the milder form of CAH-C involves polyclonal B-cell activation (Ratho et al., 2022).
Recently, Keith Sacco et al. described different immunological features in pediatric COVID-19 (pCOVID-19) and MIS-C. Pediatric COVID-19 is characterized by a substantial type I interferon (IFN) response, whereas MIS-C is related to type II IFN-dependent and NF-κB-dependent characteristics, activation of the extracellular matrix, and higher amounts of circulating SARS-CoV-2 stinger protein. However, the only strains included in the study were the ancestral Wuhan strain, the B1.177 (European lineage) variant and the 1.1.7 (Alpha) variant. Thus, the effect of Delta and Omicron variants on the innate and adaptive immune responses in children with pCOVID-19 and MIS-C requires further study (Sacco et al., 2022).
8.2.3 The recent emergence of VOCs
Notably, given that most of the above emergence studies were conducted before the emergence of the Omicron variant (and even the Delta variant), there are big gaps in our knowledge of new symptoms or sequelae associated with the recent emergence of VOCs.
On April 26, a seroepidemiological study published by the United States CDC showed that as of February 2022, about 75% of children and adolescents had serological evidence of past SARS-CoV-2 infection. Since December 2021, about one-third of children and adolescents have new seropositive (Clarke et al., 2022).
Hiroshi Nishiura et al. analyzed the cumulative incidence of COVID-19 from 1 December 2021, to 27 April 2022, in 38 countries of the Organization for Economic Cooperation and Development (OECD) and Romania. Using Welch’s analysis of variance (ANOVA), it was found that countries with hepatitis cases were more likely to experience more Omicron cases, suggesting that prior exposure to the Omicron variant (B.1.1.529) might enhance children’s risk of severe hepatitis, highlighting the importance of cofactor research (Nishiura et al., 2022).
However, data from the whole population may not be representative of the child population. Recently Yi H et al. collected children-specific population data from the ECDC and analyzed the link between Omicron infection in children and the emergence of acute hepatitis of unknown etiology. A positive correlation between cumulative Omicron cases in children and hepatitis cases was demonstrated by Spearman correlation analysis in hepatitis-detected countries. Nonetheless, the absolute number of cumulative Omicron cases is not a positive indicator compared to the cumulative incidence. The cumulative incidence of Omicron cases in children from countries with and without hepatitis detection was not significantly different. Therefore, it is hypothesized that acute hepatitis may be triggered by adenovirus, with SARS-CoV-2 acting as a cofactor that facilitates the progression of adenovirus infection to acute hepatitis (Yi et al., 2022).
There may be a link between the emergence of Omicron and acute hepatitis of unknown origin in children. Nonetheless, further studies are needed to validate this hypothesis.
8.3 Adeno-associated virus 2 with helper virus coinfection
In July, two non-peer-reviewed studies from the United Kingdom provided new insights into the possible etiology of the disease: children may be infected with both viruses and harbor genes that lead to an overactive immune response. In addition to the many studies that have reported the detection of adenovirus 41 in children with acute hepatitis of unknown origin, these two studies detected another virus, adeno-associated virus 2 (AAV2), in most children and carried a specific HLA genotype (DRB1*04:01).
AAV2 is a human parvovirus that depends on coinfection with helper viruses such as adenovirus or herpesvirus for efficient reproduction. Many people are infected with AAV2 by age 10, although the virus can remain latent in cells until a helper virus activates it.
A study conducted in Glasgow, Scotland, reported nine early cases and 58 controls. NGS and PCR detected AAV2 in plasma samples from nine patients and liver samples from four patients but not in matched control children. In addition, they found a higher prevalence of the class II HLA-DRB1*04:01 allele in eight of nine children (89%) compared to this HLA gene among Scottish blood donors (15.6%). HAdV (species C and F) and human herpesvirus 6B (HHV6B) were detected in 6/9 and 3/9 affected cases, including 3/4 and 2/4 liver biopsies, respectively (Ho et al., 2022).
Another study by a team in London investigated 28 cases of acute hepatitis in children of unknown origin, including liver samples from five children who required transplantation and blood samples from the remaining children who did not. 16 of the 17 samples tested for AAV2 were positive during RNA sequencing of liver samples (Morfopoulou et al., 2022).
These two studies propose a new etiological hypothesis that acute hepatitis in children of unknown origin may result from the interplay between AAV2, helper virus (e.g., adenovirus), and genetic susceptibility (a specific HLA genotype).
Although genetic material from AAV2 was discovered in the liver cells of patients, neither viral proteins nor actual copies of the virus were detected, emphasizing that AAV2 may trigger an immunological response that is harmful to the organ rather than directly harming liver cells.
There were a few limitations associated with these two studies. First, the number of study subjects was small. Second, current evidence only suggests a correlation with AAV2 with no proof of a causal relationship. It is highly conceivable that AAV2 is only a biomarker for coinfection with ADV or even HHV6B. Many scientists are still cautious about the conclusions of these two studies, and further mechanistic studies and large-sample research studies are warranted to confirm them.
8.4 Immunodeficiency
Gao, SH. hypothesized that children with severe acute hepatitis may have an underlying immunodeficiency, given that most cases were in children aged five or younger. In addition to immune insufficiency in children of these ages and immunodeficiency due to lack of pathogen exposure in children as a result of COVID-19 prevention and control measures, primary immunodeficiency should also be investigated (Gao et al., 2022).
In July, the Glasgow Research Group revealed that most children (8/9) carried a specific HLA genotype (DRB1*04:01) (Ho et al., 2022), associated with some autoimmune diseases, such as autoimmune hepatitis (AIH) and rheumatoid arthritis. It is well known that AIH can be classified into two subtypes; type 1 and type 2. Approximately two-thirds of pediatric AIH cases are AIH-1, which usually appears in adolescence, whereas AIH-2 affects younger children, including infants. Up to 67% of patients in children present with an acute onset. Fulminant manifestation is more frequent in AIH-2. AIH-1 susceptibility is conferred by MHC class II HLA DRB1*03 at all ages, whereas DRB1*04 is susceptible to late-onset disease; AIH-2 is associated with possession of DRB1*07 and DRB1*03 (Mieli-Vergani et al., 2018; Di Giorgio et al., 2020; Terziroli Beretta-Piccoli et al., 2022). The class II HLA DRB1*0401 allele may represent a disease susceptibility gene, but the local population structure may overestimate the significance (Ho et al., 2022). Indeed, it has been established that HLA serotypes vary across ethnic groups and regions. AIH-1 is closely related to DRB1*0301 and DRB1*0401 alleles in Caucasian adults; DRB1*0405 in Japan and Argentina (Terziroli Beretta-Piccoli et al., 2022). Moreover, DRB1*04 does not increase children’s susceptibility to AIH and even exerts a protective effect compared to adult patients (Gregorio et al., 1997).
Therefore, genetic testing, major histocompatibility complex molecular phenotyping, and immune function testing, including testing for humoral, cellular, and innate immune responses, should be undertaken on these children.
8.5 A novel adenovirus/a new variant of SARS-CoV-2/a new variant of another known pathogen/a new pathogen
In addition, acute hepatitis in children of unknown origin is likely caused by the emergence of a novel adenovirus/a new variant of SARS-CoV-2/a new variant of another known pathogen (cytomegalovirus, EBV, respiratory syncytial virus, enterovirus, parainfluenza virus, etc.)/a new pathogen with altered characteristics, with or without the cofactors mentioned above.
Suppose it is a new variant of adenovirus. It is of interest whether it is related to the recombination and mutation of adenovirus under selection pressure that may occur after mass vaccination with adenovirus vector vaccines.
There is a lack of data to support this hypothesis, especially when whole-genome sequencing results from multiple cases have not been established. However, the phenomenon of a novel adenovirus/a novel variant of SARS-CoV-2/a novel variant of other known pathogens/a new pathogen arising from naturally occurring genome recombination is highly possible.
8.6 A non-infectious cause
Toxicological investigations have revealed no major findings, with no indication of paracetamol, fluconazole, or aflatoxin B1-induced toxicity (UK Health Security Agency, 2022b).
Environmental exposure that may influence adenovirus infection has not been elucidated. According to the UKHSA report, approximately 70% of children affected whose families were interviewed reported recent contact with pet dogs. In the extended investigations, however, there was no indication that dogs played a role in hepatic syndrome, and no public health issues were associated with dog exposure and hepatic syndrome (UK Health Security Agency, 2022a).
With limited studies based on liver biopsy samples, there is little evidence to support direct viral infection of HAdV-F41, SARS-CoV-2, or AAV2 in explanted livers, and neither immunohistochemistry nor proteomic analysis has been used to detect viral proteins. Instead, studies have found evidence of high HLA class II allele expression in transplant cases, which increases the genetic risk of unknown hepatitis in these children (Ho et al., 2022; Morfopoulou et al., 2022). Nevertheless, we cannot completely rule out an immune-mediated phenomenon in susceptible children with prior viral infections. Therefore, the first four hypotheses mentioned above remain the most likely hypotheses.
9 Treatment
Treatment of hepatitis depends on the underlying etiology. Due to the lack of clarity surrounding disease etiology, no specific treatment is available. It is vital to administer comprehensive treatment, monitor conditional changes, dynamically assess laboratory indicators, be alert to liver failure, and prevent complications.
On the premise of sufficient evidence to support viral infection, it is recommended to use broad-spectrum antiviral drugs. Cidofovir (CDV) is the mainstay antiviral drug used to treat adenovirus infection. Besides, cidofovir’s lipid ester analog, brincidofovir, may be a promising salvage therapy for treatment in cases of renal toxicity induced by cidofovir or unsatisfactory clinical response (Gu et al., 2021). The nucleoside analog filociclovir (FCV) has recently been reported as a potent inhibitor of HAdV-F41 in cell culture (Tollefson et al., 2022).
Recent investigations revealed that iPALF is characterized by excessive cellular and humoral systemic inflammatory responses (Alonso et al., 2017). Immunosuppressants have been used to treat PALF associated with activated CD8+ T cell hepatitis (Squires et al., 2022), including corticosteroids for nonspecific immunosuppression, and anti-thymocyte globulin, cyclosporine, or etoposide for more specific suppression of T cell responses. Anti-cytokine therapy may also play a potential role (Alonso et al., 2017). However, the risks and benefits of immunosuppressive therapy should be weighed.
Interestingly, it has been shown that plasmapheresis can improve short-term survival and combined with other extracorporeal therapies, it can serve as a bridge to transplantation (Squires et al., 2022). For children with poor response to active comprehensive medical treatment, liver transplantation should be considered if there is no contraindication.
10 Prevention and control
Until the etiology is further understood, the WHO recommends implementing general infection prevention and control (IPC) measures, including regular hand hygiene, wearing a well-fitted mask, social distancing, covering coughs and sneezes and avoiding touching eyes, nose or mouth, ensuring adequate ventilation when indoors, and attention to food and water hygiene (World Health Organization, 2022b). Health facilities should adhere to standard precautions for suspected or probable cases and undertake contact and droplet precautions (World Health Organization, 2022b).
11 Conclusion
In summary, acute hepatitis of unknown origin in children cannot be attributed to a single cause and is most likely caused by a combination of factors. Combined with the previous hypothesis, we speculate that for a subset of children with the class II HLA allele HLA-DRB1*04:01, previous infection with SARS-CoV-2 leads to superantigen-mediated immune activation. In addition, secondary ADV infection and coinfection or reactivated hepatotropic AAV2 infection lead to abnormal host response or susceptibility, resulting in severe immune dysregulation and liver inflammation. Nevertheless, further data and research are required to comprehend the specific mechanism fully.
In addition, the heterogeneous case definitions used by different organizations/countries increase the challenge of data collection systems and comparisons between data. Indeed, it is better to establish a uniform case definition to facilitate data statistics and analysis to ensure that better and more accurate conclusions can be drawn. The WHO has established a clinical Case Report Form and urges countries to participate in the Global Clinical Platform for all cases meeting the WHO case definition. A standardized global clinical data analysis can aid in comprehending the disease’s etiology, clinical manifestations, natural course, and severity and can better guide the public health response and establish clinical management guidelines, including investigation methodologies and interventions for infection prevention and control (World Health Organization, 2022b). Most importantly, clinicians should collaborate with researchers such as immunologists and epidemiologists to study the epidemiology and pathogenesis of this disease. For children with acute hepatitis of unknown etiology but serum transaminases (ALT, AST) below 500 U/L, close attention should also be paid to their disease progression. If a mechanism associated with a postinfectious inflammatory process is identified, early administration of corticosteroids or immunomodulators may prevent progression to acute liver failure (Alexander and Deep, 2022).
Author contributions
Study design: KG, YF, and YS. Data collection and Literature Search: KG, XX, YL, XY, JY, HT, and SY. Data analyses and results interpretations: KG and XX. Manuscript writing: KG, XX, and YS. Supervision: YF and YS. All authors have read and agreed to the final manuscript.
Funding
This study was supported by the National Key R&D Program of China (2021YFC2301800); Chinese National Natural Science Foundation (No. 81870425); The Fundamental Research Funds for the Central Universities (2021FZZX001-41); Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (No. 2022490480).
Acknowledgments
The authors acknowledge the efforts of the many scientists and clinicians worldwide who have worked on elucidating the aetiology and mechanisms of this disease. The graphical abstract was created with BioRender.com.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Alexander, E. C., and Deep, A. (2022). Characterization of a hepatitis outbreak in children, 2021 to 2022. JAMA Netw. Open 5 (10), e2237091. doi:10.1001/jamanetworkopen.2022.37091
Alonso, E. M., Horslen, S. P., Behrens, E. M., and Doo, E. (2017). Pediatric acute liver failure of undetermined cause: A research workshop. Hepatology 65 (3), 1026–1037. doi:10.1002/hep.28944
Arankalle, V. A., Chadha, M. S., Chitambar, S. D., Walimbe, A. M., Chobe, L. P., and Gandhe, S. S. (2001). Changing epidemiology of hepatitis A and hepatitis E in urban and rural India (1982-98). J. Viral Hepat. 8 (4), 293–303. doi:10.1046/j.1365-2893.2001.00279.x
Aydoğdu, S., Ozgenç, F., Yurtsever, S., Akman, S. A., Tokat, Y., and Yağci, R. V. (2003). Our experience with fulminant hepatic failure in Turkish children: Etiology and outcome. J. Trop. Pediatr. 49 (6), 367–370. doi:10.1093/tropej/49.6.367
Aygün, F. D., Avar-Aydın, P. Ö., Çokuğraş, H., and Camcıoğlu, Y. (2016). Different clinical spectrum of leptospirosis. Turk. J. Pediatr. 58 (2), 212–215. doi:10.24953/turkjped.2016.02.015
Baker, J. M., Buchfellner, M., Britt, W., Sanchez, V., Potter, J. L., Ingram, L. A., et al. (2022). Acute hepatitis and adenovirus infection among children - Alabama. MMWR. Morb. Mortal. Wkly. Rep. 71 (18), 638–640. doi:10.15585/mmwr.mm7118e1
Black, D. D. (2009). The continuing challenge of "indeterminate" acute liver failure in children. J. Pediatr. 155 (6), 769–770. doi:10.1016/j.jpeds.2009.07.060
Boettler, T., Csernalabics, B., Salié, H., Luxenburger, H., Wischer, L., Salimi Alizei, E., et al. (2022). SARS-CoV-2 vaccination can elicit a CD8 T-cell dominant hepatitis. J. Hepatol. 77, 653–659. doi:10.1016/j.jhep.2022.03.040
Braccio, S., Irwin, A., Riordan, A., Shingadia, D., Kelly, D. A., Bansal, S., et al. (2017). Acute infectious hepatitis in hospitalised children: A British paediatric surveillance unit study. Arch. Dis. Child. 102 (7), 624–628. doi:10.1136/archdischild-2016-311916
Bril, F., Al Diffalha, S., Dean, M., and Fettig, D. M. (2021). Autoimmune hepatitis developing after coronavirus disease 2019 (COVID-19) vaccine: Causality or casualty? J. Hepatol. 75 (1), 222–224. doi:10.1016/j.jhep.2021.04.003
Brodin, P., and Arditi, M. (2022). Severe acute hepatitis in children: Investigate SARS-CoV-2 superantigens. Lancet. Gastroenterol. Hepatol. 7 (7), 594–595. doi:10.1016/S2468-1253(22)00166-2
Brodin, P. (2022). SARS-CoV-2 infections in children: Understanding diverse outcomes. Immunity 55 (2), 201–209. doi:10.1016/j.immuni.2022.01.014
Cantor, A., Miller, J., Zachariah, P., DaSilva, B., Margolis, K., and Martinez, M. (2020). Acute hepatitis is a prominent presentation of the multisystem inflammatory syndrome in children: A single-center report. Hepatology 72 (5), 1522–1527. doi:10.1002/hep.31526
Centers for Disease Control and Prevention (2022). Children with acute hepatitis of unknown etiology investigation updates. [Online]. Available: https://www.cdc.gov/ncird/investigation/hepatitis-unknown-cause/updates.html (Accessed Sept 14, 2022).
Cheng, M. H., Zhang, S., Porritt, R. A., Noval Rivas, M., Paschold, L., Willscher, E., et al. (2020). Superantigenic character of an insert unique to SARS-CoV-2 spike supported by skewed TCR repertoire in patients with hyperinflammation. Proc. Natl. Acad. Sci. U. S. A. 117 (41), 25254–25262. doi:10.1073/pnas.2010722117
Chevret, L., Boutolleau, D., Halimi-Idri, N., Branchereau, S., Baujard, C., Fabre, M., et al. (2008). Human herpesvirus-6 infection: A prospective study evaluating HHV-6 DNA levels in liver from children with acute liver failure. J. Med. Virol. 80 (6), 1051–1057. doi:10.1002/jmv.21143
Clarke, K. E. N., Jones, J. M., Deng, Y., Nycz, E., Lee, A., Iachan, R., et al. (2022). Seroprevalence of infection-induced SARS-CoV-2 antibodies - United States, september 2021-february 2022. MMWR. Morb. Mortal. Wkly. Rep. 71 (17), 606–608. doi:10.15585/mmwr.mm7117e3
Cooper, S., Tobar, A., Konen, O., Orenshtein, N., Kropach, N., Landau, Y., et al. (2022). Long COVID-19 liver manifestation in children. J. Pediatr. Gastroenterol. Nutr. 75, 244–251. doi:10.1097/MPG.0000000000003521
Daniels, D., Grytdal, S., and Wasley, A. (2009). Surveillance for acute viral hepatitis - United States, 2007. Surveillance Summaries (Washington, D.C.): Morbidity and Mortality Weekly Report.
Deneau, M., Jensen, M. K., Holmen, J., Williams, M. S., Book, L. S., and Guthery, S. L. (2013). Primary sclerosing cholangitis, autoimmune hepatitis, and overlap in Utah children: Epidemiology and natural history. Hepatology 58 (4), 1392–1400. doi:10.1002/hep.26454
Devictor, D., Tissieres, P., Afanetti, M., and Debray, D. (2011). Acute liver failure in children. Clin. Res. Hepatol. Gastroenterol. 35 (6-7), 430–437. doi:10.1016/j.clinre.2011.03.005
Di Giorgio, A., Hadzic, N., Dhawan, A., Deheragoda, M., Heneghan, M. A., Vergani, D., et al. (2020). Seamless management of juvenile autoimmune liver disease: Long-term medical and social outcome. J. Pediatr. 218, 121–129. doi:10.1016/j.jpeds.2019.11.028
Durand, P., Debray, D., Mandel, R., Baujard, C., Branchereau, S., Gauthier, F., et al. (2001). Acute liver failure in infancy: A 14-year experience of a pediatric liver transplantation center. Am. J. Trop. Med. Hyg. 139 (6), 871–876. doi:10.1067/mpd.2001.119989
el-Zimaity, D. M., Hyams, K. C., Imam, I. Z., Watts, D. M., Bassily, S., Naffea, E. K., et al. (1993). Acute sporadic hepatitis E in an Egyptian pediatric population. Am. J. Trop. Med. Hyg. 48 (3), 372–376. doi:10.4269/ajtmh.1993.48.372
European Centre for Disease Prevention and Control (2022). Joint ECDC-WHO Regional Office for Europe hepatitis of unknown origin in children surveillance bulletin. [Online]. Available at:https://cdn.ecdc.europa.eu/novhep-surveillance/(Accessed Sept 14, 2022).
Fischler, B., Baumann, U., Dezsofi, A., Hadzic, N., Hierro, L., Jahnel, J., et al. (2016). Hepatitis E in children: A position paper by the espghan hepatology committee. J. Pediatr. Gastroenterol. Nutr. 63 (2), 288–294. doi:10.1097/MPG.0000000000001231
Gallegos-Orozco, J. F., and Rakela-Brödner, J. (2010). Hepatitis viruses: Not always what it seems to be. Rev. Med. Chil. 138 (10), 1302–1311./S0034-98872010001100016.
Gao, S.-H., Gong, M.-C., and Song, H.-M. (2022). Acute severe hepatitis of unknown origin in children: Considerations from the perspective of immunology. World J. Pediatr. 18 (8), 529–532. doi:10.1007/s12519-022-00580-y
Garnett, C. T., Erdman, D., Xu, W., and Gooding, L. R. (2002). Prevalence and quantitation of species C adenovirus DNA in human mucosal lymphocytes. J. Virol. 76 (21), 10608–10616. doi:10.1128/jvi.76.21.10608-10616.2002
Government of Canada (2022). Acute severe hepatitis in children. [Online]. Available at:https://www.canada.ca/en/public-health/services/diseases/acute-hepatitis-children.html (Accessed Sept 16, 2022).
Government of Manitoba (2022). Acute, severe hepatitis of unknown origin in children. [Online]. Available at:https://www.gov.mb.ca/health/publichealth/diseases/acute_hepatitis.html (Accessed Jun 17, 2022).
Gregorio, G. V., Portmann, B., Reid, F., Donaldson, P. T., Doherty, D. G., McCartney, M., et al. (1997). Autoimmune hepatitis in childhood: A 20-year experience. Hepatology 25 (3), 541–547. doi:10.1002/hep.510250308
Gu, J., Su, Q.-Q., Zuo, T.-T., and Chen, Y.-B. (2021). Adenovirus diseases: A systematic review and meta-analysis of 228 case reports. Infection 49 (1), 1–13. doi:10.1007/s15010-020-01484-7
Gupta, S., Smith, L., and Diakiw, A. (2022). Amebiasis and amebic liver abscess in children. Pediatr. Clin. North Am. 69 (1), 79–97. doi:10.1016/j.pcl.2021.08.003
Hassouneh, L., Quadri, S., Pichilingue-Reto, P., Chaisavaneeyakorn, S., Cutrell, J. B., Wetzel, D. M., et al. (2019). An outbreak of brucellosis: An adult and pediatric case series. Open Forum Infect. Dis. 6 (10), ofz384. doi:10.1093/ofid/ofz384
Ho, A., Orton, R., Tayler, R., Asamaphan, P., Tong, L., Smollett, K., et al. (2022). Adeno-associated virus 2 infection in children with non-A-E hepatitis.medRxiv, 2022.2007.2019.22277425. doi: doi:10.1101/2022.07.19.22277425
Hodges, M., Sanders, E., and Aitken, C. (1998). Seroprevalence of hepatitis markers; HAV, HBV, HCV and HEV amongst primary school children in Freetown, Sierra Leone. West Afr. J. Med. 17 (1), 36–37.
Hyams, K. C., McCarthy, M. C., Kaur, M., Purdy, M. A., Bradley, D. W., Mansour, M. M., et al. (1992). Acute sporadic hepatitis E in children living in Cairo, Egypt. J. Med. Virol. 37 (4), 274–277. doi:10.1002/jmv.1890370407
Jaggi, P., Kajon, A. E., Mejias, A., Ramilo, O., and Leber, A. (2013). Human adenovirus infection in kawasaki disease: A confounding bystander? Clin. Infect. Dis. 56 (1), 58–64. doi:10.1093/cid/cis807
Jiménez-Rivera, C., Ling, S. C., Ahmed, N., Yap, J., Aglipay, M., Barrowman, N., et al. (2015). Incidence and characteristics of autoimmune hepatitis. Pediatrics 136 (5), e1237–e1248. doi:10.1542/peds.2015-0578
Kambhampati, A. K., Burke, R. M., Dietz, S., Sheppard, M., Almendares, O., Baker, J. M., et al. (2022). Trends in acute hepatitis of unspecified etiology and adenovirus stool testing results in children - United States, 2017-2022. MMWR. Morb. Mortal. Wkly. Rep. 71 (24), 797–802. doi:10.15585/mmwr.mm7124e1
Kang, G. (2017). “Viral diarrhea,” in International encyclopedia of public health. Editor S. R. Quah (Elsevier), 260–267.
Kathemann, S., Bechmann, L. P., Sowa, J.-P., Manka, P., Dechêne, A., Gerner, P., et al. (2015). Etiology, outcome and prognostic factors of childhood acute liver failure in a German Single Center. Ann. Hepatol. 14 (5), 722–728. doi:10.1016/s1665-2681(19)30767-7
Kendall, E. K., Olaker, V. R., Kaelber, D. C., Xu, R., and Davis, P. B. (2022). Elevated liver enzymes and bilirubin following SARS-CoV-2 infection in children under 10. medRxiv, . doi: doi:10.1101/2022.05.10.22274866
Kosulin, K. (2019). Intestinal HAdV infection: Tissue specificity, persistence, and implications for antiviral therapy. Viruses 11 (9), E804. doi:10.3390/v11090804
Liu, E., MacKenzie, T., Dobyns, E. L., Parikh, C. R., Karrer, F. M., Narkewicz, M. R., et al. (2006). Characterization of acute liver failure and development of a continuous risk of death staging system in children. J. Hepatol. 44 (1), 134–141. doi:10.1016/j.jhep.2005.06.021
Lynch, J. P., and Kajon, A. E. (2016). Adenovirus: Epidemiology, global spread of novel serotypes, and advances in treatment and prevention. Semin. Respir. Crit. Care Med. 37 (4), 586–602. doi:10.1055/s-0036-1584923
Mack, C. L., Adams, D., Assis, D. N., Kerkar, N., Manns, M. P., Mayo, M. J., et al. (2020). Diagnosis and management of autoimmune hepatitis in adults and children: 2019 practice guidance and guidelines from the American association for the study of liver diseases. Hepatology 72 (2), 671–722. doi:10.1002/hep.31065
Marsh, K., Tayler, R., Pollock, L., Roy, K., Lakha, F., Ho, A., et al. (2022). Investigation into cases of hepatitis of unknown aetiology among young children, Scotland, 1 January 2022 to 12 April 2022. Euro Surveill. Bull. Eur. Sur Les. Mal. Transm. = Eur. Commun. Dis. Bull. 27 (15). doi:10.2807/1560-7917.ES.2022.27.15.2200318
Mieli-Vergani, G., Vergani, D., Baumann, U., Czubkowski, P., Debray, D., Dezsofi, A., et al. (2018). Diagnosis and management of pediatric autoimmune liver disease: ESPGHAN hepatology committee position statement. J. Pediatr. Gastroenterol. Nutr. 66 (2), 345–360. doi:10.1097/MPG.0000000000001801
Morfopoulou, S., Buddle, S., Torres Montaguth, O. E., Atkinson, L., Guerra-Assunção, J. A., Storey, N., et al. (2022). Genomic investigations of acute hepatitis of unknown aetiology in children. medRxiv. doi: doi:10.1101/2022.07.28.22277963
Narkewicz, M. R., Dell Olio, D., Karpen, S. J., Murray, K. F., Schwarz, K., Yazigi, N., et al. (2009). Pattern of diagnostic evaluation for the causes of pediatric acute liver failure: An opportunity for quality improvement. J. Pediatr. 155 (6), 801–806. doi:10.1016/j.jpeds.2009.06.005
Nishiura, H., Jung, S.-M., and Hayashi, K. (2022). High population burden of Omicron variant (B.1.1.529) is associated with the emergence of severe hepatitis of unknown etiology in children. Int. J. Infect. Dis. 122, 30–32. doi:10.1016/j.ijid.2022.05.028
Ozbay Hoşnut, F., Canan, O., Ozçay, F., and Bilezikçi, B. (2008). Adenovirus infection as possible cause of acute liver failure in a healthy child: A case report. Turk. J. Gastroenterol. 19 (4), 281–283.
Rahman, M. A., Chakma, U., Kumer, A., Rahman, M. R., and Matin, M. M. (2022). Uridine-derived 4-aminophenyl 1-thioglucosides: DFT optimized FMO, ADME, and antiviral activities study. doi: doi:10.33263/BRIAC131.052
Ratho, R. K., Asati, A. A., Mishra, N., Jain, A., and Rawat, S. K. (2022). COVID-19 Associated Hepatitis in Children (CAH-C) during the second wave of SARS-CoV-2 infections in Central India: Is it a complication or transient phenomenon. medRxiv. doi: doi:10.1101/2021.07.23.21260716
Rocholl, C., Gerber, K., Daly, J., Pavia, A. T., and Byington, C. L. (2004). Adenoviral infections in children: The impact of rapid diagnosis. Pediatrics 113 (1), e51–e56. doi:10.1542/peds.113.1.e51
Rubin, R. (2022). SARS-CoV-2 RNA can persist in stool months after respiratory tract clears virus. JAMA 327 (22), 2175–2176. doi:10.1001/jama.2022.7892
Sacco, K., Castagnoli, R., Vakkilainen, S., Liu, C., Delmonte, O. M., Oguz, C., et al. (2022). Immunopathological signatures in multisystem inflammatory syndrome in children and pediatric COVID-19. Nat. Med. 28 (5), 1050–1062. doi:10.1038/s41591-022-01724-3
Sood, V., Lal, B. B., Gupta, E., Khanna, R., Siloliya, M. K., and Alam, S. (2019). Hepatitis A virus-related pediatric liver disease burden and its significance in the Indian subcontinent. Indian Pediatr. 56 (9), 741–744. doi:10.1007/s13312-019-1640-1
Squires, J. E., Alonso, E. M., Ibrahim, S. H., Kasper, V., Kehar, M., Martinez, M., et al. (2022). North American society for pediatric gastroenterology, hepatology, and nutrition position paper on the diagnosis and management of pediatric acute liver failure. J. Pediatr. Gastroenterol. Nutr. 74 (1), 138–158. doi:10.1097/MPG.0000000000003268
Squires, R. H., Shneider, B. L., Bucuvalas, J., Alonso, E., Sokol, R. J., Narkewicz, M. R., et al. (2006). Acute liver failure in children: The first 348 patients in the pediatric acute liver failure study group. J. Pediatr. 148 (5), 652–658. doi:10.1016/j.jpeds.2005.12.051
Terziroli Beretta-Piccoli, B., Mieli-Vergani, G., and Vergani, D. (2022). Autoimmmune hepatitis. Cell. Mol. Immunol. 19 (2), 158–176. doi:10.1038/s41423-021-00768-8
Tollefson, A. E., Hussein, I. T. M., Toth, K., and Bowlin, T. L. (2022). Filociclovir is a potent inhibitor of human adenovirus F41. Antivir. Res. 208, 105431. doi:10.1016/j.antiviral.2022.105431
Tsunoda, T., Inui, A., Iwasawa, K., Oikawa, M., Sogo, T., Komatsu, H., et al. (2017). Acute liver dysfunction not resulting from hepatitis virus in immunocompetent children. Pediatr. Int. 59 (5), 551–556. doi:10.1111/ped.13249
UK Health Security Agency (2022a). Investigation into acute hepatitis of unknown aetiology in children in england: Technical briefing 1. [Online]. Available at:https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1071198/acute-hepatitis-technical-briefing-1_4_.pdf (Accessed Jun 17, 2022).
UK Health Security Agency (2022b). Investigation into acute hepatitis of unknown aetiology in children in england: Technical briefing 3. [Online]. Available at:https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1077027/acute-hepatitis-technical-briefing_3.pdf (Accessed Jun 17, 2022).
UK Health Security Agency (2022c). Investigation into acute hepatitis of unknown aetiology in children in england: Technical briefing 4. [Online]. Available at:https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1094573/acute-hepatitis-technical-briefing-4.pdf (Accessed Sept 14, 2022).
Verghese, V. P., and Robinson, J. L. (2014). A systematic review of hepatitis E virus infection in children. Clin. Infect. Dis. 59 (5), 689–697. doi:10.1093/cid/ciu371
Wang, Y., Liu, S., Liu, H., Li, W., Lin, F., Jiang, L., et al. (2020). SARS-CoV-2 infection of the liver directly contributes to hepatic impairment in patients with COVID-19. J. Hepatol. 73 (4), 807–816. doi:10.1016/j.jhep.2020.05.002
World Health Organization (2022a). Disease outbreak news; acute hepatitis of unknown aetiology in children - multi-country. [Online]. Available at:https://www.who.int/emergencies/disease-outbreak-news/item/DON-389 (Accessed Jun 17, 2022).
World Health Organization (2022b). Severe acute hepatitis of unknown aetiology in children - multi-country. [Online]. Available at:https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON400 (Accessed Sept 14, 2022).
Yi, H., Lin, Y., Lu, B., and Mao, Y. (2022). The origin of severe hepatitis of unknown aetiology in children: SARS-CoV-2 or adenovirus? J. Hepatology. doi:10.1016/j.jhep.2022.08.032
Keywords: acute hepatitis, adenovirus, SARS-CoV-2, multisystem inflammatory syndrome in children, adeno-associated virus 2, immunodeficiency
Citation: Gong K, Xu X, Yao J, Ye S, Yu X, Tu H, Lan Y, Fan Y-c and Shi Y (2022) Acute hepatitis of unknown origin in children: A combination of factors. Front. Pharmacol. 13:1056385. doi: 10.3389/fphar.2022.1056385
Received: 28 September 2022; Accepted: 31 October 2022;
Published: 11 November 2022.
Edited by:
Talha Bin Emran, Begum Gulchemonara Trust University, BangladeshReviewed by:
Mohsina Patwekar, Luqman college of pharmacy, GulbargaMohammed Mahbubul Matin, University of Chittagong, Bangladesh
Chih-Cheng Lai, Chi Mei Medical Center, Taiwan
Ayane Alves Sarmento, Federal University of Rio Grande do Norte, Brazil
Copyright © 2022 Gong, Xu, Yao, Ye, Yu, Tu, Lan, Fan and Shi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yu Shi, emp1c2hpeXVAemp1LmVkdS5jbg==; Yu-chen Fan, ZmFueXVjaGVuQHNkdS5lZHUuY24=
†These authors have contributed equally to this work and share first authorship
 Kai Gong
Kai Gong Xianbin Xu
Xianbin Xu Junjie Yao1
Junjie Yao1 Yan Lan
Yan Lan Yu-chen Fan
Yu-chen Fan Yu Shi
Yu Shi